Abstract
We report the development of a multiplex PCR protocol for the diagnosis of staphylococcal infection. The protocol was designed to (i) detect any staphylococcal species to the exclusion of other bacterial pathogens (based on primers corresponding to Staphylococcus-specific regions of the 16S rRNA genes), (ii) distinguish between S. aureus and the coagulase-negative staphylococci (CNS) (based on amplification of the S. aureus-specific clfA gene), and (iii) provide an indication of the likelihood that the staphylococci present in the specimen are resistant to oxacillin (based on amplification of the mecA gene). The expected fragments were amplified from each of 60 staphylococcal isolates (13 oxacillin-resistant S. aureus isolates, 23 oxacillin-sensitive S. aureus isolates, 17 oxacillin-resistant CNS, and 7 oxacillin-sensitive CNS). No amplification products were observed with template DNA from nonstaphylococcal species, and the efficiency of amplification of staphylococcal targets was not adversely affected by the presence of DNA from other bacterial species in the same sample. The utility of the protocol for the analysis of clinical samples was verified by analysis of aliquots taken directly from BacT/Alert blood culture bottles. Of 77 blood cultures tested, only 7 yielded results inconsistent with those of conventional methods of diagnosis and susceptibility testing. Of those, one was identified as a CNS species by PCR and S. aureus by conventional methods. We also identified two isolates that were mecA positive but were oxacillin sensitive according to conventional methods. The other four samples failed to yield any amplification product even with a control set of primers corresponding to a conserved region of the eubacterial rRNA genes.
The staphylococci are among the most prominent of all nosocomial pathogens. Although Staphylococcus aureus is clearly the primary pathogen, the coagulase-negative staphylococci (CNS) are also capable of causing disease (18). That is particularly true of Staphylococcus epidermidis, which is a frequent cause of infections associated with indwelling medical devices (1, 6) However, the prevalence of S. epidermidis as a commensal bacterium has the adverse diagnostic consequence of false-positive culture results owing to contamination of the specimen during collection (35). For that reason, it is important to distinguish between S. aureus and CNS in clinical samples and to confirm the presence of CNS before making a diagnostic decision (35).
The major concern with regard to the treatment of staphylococcal infections is the continued emergence of antibiotic-resistant strains. Indeed, over 90% of all nosocomial isolates are resistant to penicillin, and an increasing number are resistant to the semisynthetic, β-lactamase-resistant derivatives represented by oxacillin (2, 14). Moreover, oxacillin-resistant strains are often resistant to other antimicrobial agents commonly used to treat staphylococcal infection (33). Therapeutic options in such cases are often limited to the glycopeptide antibiotics (e.g., vancomycin) or the newly approved drugs linezolid (23) and quinupristin-dalfopristin (32). Recent reports describing S. aureus isolates with reduced susceptibility to vancomycin emphasize the tenuous nature of our reliance on such a limited group of drugs (12, 30). To delay the emergence of resistant strains and prolong the utility of currently available antibiotics, it is imperative that the use of these drugs be restricted to those cases in which they are absolutely necessary, the primary example being a serious infection caused by an oxacillin-resistant strain.
Based on the preceding discussion, the most important considerations with respect to the diagnosis of staphylococcal infections are (i) identification of staphylococci in clinical specimens, (ii) differentiation of S. aureus from the less-pathogenic CNS, and (iii) determination of whether isolates of either group are resistant to oxacillin. In most laboratories, the accurate assessment of these issues is dependent on the phenotypic characterization of cultured bacteria. However, there are numerous reports describing the use of PCR for the identification and characterization of staphylococcal isolates (3, 4, 7, 10, 11, 16, 18, 19, 24, 25, 31, 34). To maximize sensitivity, most protocols focused on amplification of conserved regions of eubacterial rRNA genes and required additional steps (e.g., hybridization with species-specific probes) to establish a diagnosis (7, 10, 11, 18, 25). Other protocols were directed toward the specific detection of S. aureus and focused on amplification of genes found only in that species. Specific examples include the genes encoding nuclease (nuc) and coagulase (coa) and an undefined 442-bp DNA fragment amplified from the S. aureus chromosome (3, 4, 19, 25). Given the importance of detecting oxacillin resistance, some protocols focused directly on amplification of the mecA gene either alone or in a multiplex format capable of simultaneously amplifying additional markers (7, 16, 24, 25, 34).
One of the most comprehensive studies employing a multiplex format examined 786 bacterial isolates (including 686 staphylococcal isolates) using primer pairs corresponding to the eubacterial 16S rRNA genes, a Staphylococcus-specific region of the 16S rRNA genes, the coa gene, and the mecA gene (25). This protocol was both rapid (∼4 h) and specific; however, it was evaluated using isolated bacterial colonies and therefore required culture prior to analysis. Additionally, reports describing polymorphisms within coa (9, 13, 26) suggest that protocols that focus on coa as a distinguishing characteristic might be subject to errors of amplification and/or interpretation. A more recent report described a procedure that was capable of detecting multiple target genes and could be used for the direct analysis of positive blood cultures (15). However, the protocol utilized independent amplification reactions for each target gene and could not distinguish between S. aureus and other staphylococcal species. It also had a sensitivity limit of approximately 109 CFU, which may exceed the density of bacteria present in at least some positive blood cultures.
We describe a multiplex PCR protocol that can be applied directly to the analysis of positive blood cultures. The protocol uses primer pairs corresponding to (i) regions of the 16S rRNA genes that are unique to staphylococci, (ii) the S. aureus-specific clfA gene, encoding a surface-associated fibrinogen-binding protein (20), and (iii) the mecA gene, which is the primary determinant of oxacillin-resistance in both S. aureus and the CNS species (7). The specificity and reproducibility of the protocol were verified using 60 confirmed staphylococcal isolates, many of which were previously shown to represent distinct clonal variants (27, 29, 30). The applicability of the protocol to the direct analysis of clinical samples was tested using template DNA obtained directly from positive blood culture bottles. Of 77 samples tested, only 7 yielded results inconsistent with those obtained using conventional diagnostic and susceptibility testing protocols.
MATERIALS AND METHODS
Bacterial strains and susceptibility testing.
Staphylococcal isolates used to develop and evaluate our protocol were obtained from the clinical laboratory at the University of Arkansas for Medical Sciences (UAMS) or were obtained from Fred Tenover at the Centers for Disease Control and Prevention (CDC). The UAMS S. aureus isolates (n = 18) were previously shown to be distinct clonal variants based on genomic fingerprinting with probes corresponding to the collagen adhesin (cna), fibronectin-binding protein (fnbA and fnbB), and β-toxin (hlb) genes (29). The CDC isolates (n = 18) were chosen from a group of strains that were previously used to evaluate epidemiological typing protocols (30). The choice of CDC strains was based on subsequent fingerprinting experiments (27), isolation of strains from different geographic locations, and differences in oxacillin susceptibility (30). With the exception of UAMS-88, which is a confirmed S. epidermidis isolate, the CNS isolates (n = 24) were not distinguished from each other at the species level. Bacteria were maintained on Trypticase soy agar (TSA) without antibiotic selection. Oxacillin resistance was determined by the broth dilution method. Resistance was defined as a MIC of ≥4 μg/ml (30).
Isolates of Escherichia coli, beta-hemolytic Streptococcus spp., Bacillus spp., and Corynebacterium spp. were obtained from the clinical laboratory at UAMS and were not differentiated beyond the level indicated. Isolates of Pseudomonas aeruginosa were kindly provided by Shouguang Jin (University of Florida, Gainesville). Genomic DNA from Neisseria gonorrhoeae was kindly provided by David Dyer (University of Oklahoma Health Sciences Center, Oklahoma City).
To test the utility of our protocol in the presence of blood products, we inoculated BacT/Alert blood culture bottles (Organon Teknika Corporation, Durham, N.C.) with an isolate of oxacillin-resistant S. aureus (ORSA), oxacillin-sensitive S. aureus (OSSA), oxacillin-resistant CNS (ORCNS), or oxacillin-sensitive CNS (OSCNS). After growth for 15 h at 37°C, template DNA was isolated and processed for PCR as described below. We also used the ORSA isolate to examine the sensitivity of our protocol. To minimize the number of nonviable bacteria, UAMS-601 was inoculated into a BacT/Alert blood culture bottle at a starting density of <100 cell per ml and a sample was harvested within 15 h of incubation at 37°C. Serial 10-fold dilutions were prepared using sterile BacT/Alert medium containing 10 ml of venous blood as a diluent. Viable counts were determined by plating appropriately diluted aliquots on TSA. At the same time, a 1-ml sample from each dilution was processed for template DNA.
To determine whether our protocol could be used for the direct analysis of clinical samples, we carried out a study in which aliquots from positive blood cultures were obtained from the UAMS clinical laboratory and processed for PCR as described below. These studies were done in a blinded fashion, such that the investigators carrying out the PCR analysis were unaware of the results obtained by the clinical laboratory and vice versa.
Primer design.
We designed three primer pairs that would collectively allow us to accomplish all three of our diagnostic objectives (Table 1). The first corresponds to regions of the 16S rRNA genes that are conserved among staphylococci and are unique by comparison to other eubacterial species. The second corresponds to the S. aureus clfA gene, which encodes a surface-exposed fibrinogen-binding protein (20). The choice of clfA was based on previous work from our laboratory suggesting that clfA is present in the chromosome of all S. aureus strains (28) and reports demonstrating the existence of multiple polymorphisms within the S. aureus coa gene (9, 13, 26). The third pair corresponds to the mecA gene, which encodes the unique penicillin-binding protein (PBP2a or PBP2′) that is most directly associated with oxacillin resistance in both S. aureus and the CNS species (2, 7).
TABLE 1.
Nucleotide sequences of PCR primers
| Target gene | Primera
|
Length (bp) | |
|---|---|---|---|
| 5′ | 3′ | ||
| Staphylococcal 16S rRNA | CCTATAAGACTGGGATAACTTCGGG | CTTTGAGTTTCAACCTTGCGGTCG | 791 |
| clfA | GCAAAATCCAGCACAACAGGAAACGA | CTTGATCTCCAGCCATAATTGGTGG | 638 |
| mecA | TCCAGGAATGCAGAAAGACCAAAGC | GACACGATAGCCATCTTCATGTTGG | 499 |
| Eubacterial 16S rRNAb | AACTGGAGGAAGGTGGGGAT | AGGAGGTGATCCAACCGCA | 371 |
All primers are written 5′ to 3′ as synthesized. Design parameters for the clfA, mecA, and staphylococcal rRNA gene primers are discussed in the text.
Primers for the eubacterial rRNA genes are from Schmitz et al. (25). The eubacterial rRNA primers were used to confirm the presence of genomic DNA and were not designed for use in our multiplex protocol.
Primers were designed to yield amplification products that ranged between 400 and 800 bp and differed by at least 100 bp (Table 1). Accession numbers of the specific sequences used to design each primer pair were X52593 (mecA), Z18852 (clfA), and X68417 and Z22809 (rRNA genes for S. aureus and S. epidermidis, respectively). All primers used in the multiplex protocol were 24 to 25 bp long with a G+C content of 48 to 50%. We also synthesized a fourth primer pair that corresponds to a region of the rRNA genes that is conserved in all eubacteria (25). These primers, which amplify a 371-bp fragment, were used in independent amplifications (i.e., not as part of the multiplex protocol) to ensure that the lack of an amplification product from species other than staphylococci reflected the specificity of our protocol rather than the lack of suitable template DNA.
Preparation of template DNA.
Template DNA was obtained from pure cultures of bacteria and from uncharacterized positive blood cultures. Pure cultures were used to assess the specificity and reproducibility of our amplification protocol. Specifically, each of 60 verified staphylococcal isolates (13 ORSA, 23 OSSA, 17 ORCNS, and 7 OSCNS isolates) were grown overnight in tryptic soy broth (TSB). A 100-μl aliquot of the overnight culture (approximately 5 × 108 CFU) was processed for template DNA as described below. To test whether the presence of DNA from other species interfered with the amplification of staphylococcal targets, two different experiments were done. In the first, all nonstaphylococcal species were mixed together in a single TSB culture with and without the ORSA strain UAMS-601. The mixed culture was grown overnight and processed for template DNA as described below. In the second, each nonstaphylococcal species was grown in TSB and processed for template DNA. Equal volumes of each DNA preparation were then mixed together with and without template DNA from UAMS-601.
The utility of our protocol for the direct analysis of clinical samples was tested using samples taken from positive BacT/Alert blood culture bottles. A 1.0-ml sample was removed under aseptic conditions and centrifuged in a microcentrifuge at 15,000 rpm (21,000 × g) for 1 min. The supernatant was discarded, and the pellet was resuspended in 560 μl of TE buffer (10 mM Tris [pH 7.5] and 1 mM EDTA). Then, 5 μl of RNase (10 mg/ml) and 5 μl of lysostaphin (10 mg/ml) were added and mixed vigorously. After incubation at 37°C for 1 h, 30 μl of 10% sodium dodecyl sulfate, 5 μl of RNase, and 10 μl of proteinase K (10 mg/ml) were added and the incubation was continued for an additional hour. NaCl (100 μl, 5 M) was added, followed by 80 μl of prewarmed (65°C) CTAB-NaCl (10% hexadecyltrimethyl ammonium bromide in 0.7 M NaCl). Following a 10 min incubation at 65°C, an equal volume of chloroform was added, and the suspension was mixed by vortexing. After centrifugation for 5 min at 15,000 rpm, the viscous upper phase was removed and transferred to a new 1.5-ml microcentrifuge tube. The suspension was extracted twice with an equal volume of phenol-chloroform-isoamyl alcohol (25:24:1) and once with chloroform-isoamyl alcohol. The upper aqueous phase was transferred to a new 1.5-ml microcentrifuge tube, and the DNA was precipitated by adding 500 μl of isopropanol. After centrifugation at 15,000 rpm for 10 min., the isopropanol was removed and the pellet was washed with 1.0 ml of 70% ethanol. The DNA pellet was then dried and resuspended in 30 μl of sterile water.
PCR protocol.
PCR was done using a master mix containing 69 μl of sterile water, 10 μl of 10× amplification buffer, 10 μl of 2 mM deoxynucleoside triphosphates, 3 μl of 3.75 mM MgCl2, 1 μl of a 10 pM stock of each primer, and 0.5 μl (2.5 U) of Taq polymerase. Amplification buffer (10×), deoxynucleoside triphosphates, MgCl2, and Taq polymerase were all from the Taq PCR core kit (Qiagen, Inc., Valencia, Calif.). Template DNA (1 μl) was added to a 0.5-ml thin-walled PCR tube, followed by the addition of 99 μl of PCR master mix. After mixing, the sample was pulse centrifuged for 5 s and then overlaid with 50 μl of mineral oil. Cycling parameters were (i) 94°C for 3.0 min, (ii) 94°C for 1.5 min, (iii) 55°C for 1 min, (iv) 72°C for 1 min, (v) 36 cycles of steps 2 through 4 inclusive, and (vi) 72°C for 10 min. Aliquots (5 μl) of the amplification products were analyzed by agarose gel electrophoresis using 1.0% LE agarose (FMC Bioproducts, Rockland, Maine) containing 0.5 μg of ethidium bromide per ml. Gels were visualized and photographed using a GDS7500 gel documentation system (UVP Inc., Upland, Calif.).
To verify the identity of each amplification product, representative DNA fragments amplified from UAMS-601 were gel purified and cloned using the pCRII-TOPO vector (Invitrogen Corp., Carlsbad, Calif.). Cloned fragments were sequenced using the M13 forward and reverse primers and an ABI 377 automated DNA sequencer (Applied Biosystems, Foster City, Calif.).
Conventional diagnostic methods.
Samples from positive blood cultures were examined by Gram staining and plated on TSA containing 5% sheep blood to obtain isolated colonies. Staphylococci were differentiated from other gram-positive cocci based on hemolysis pattern and the production of coagulase and/or catalase. S. aureus was distinguished from CNS using the Staphaurex latex agglutination test (Murex Biotech Ltd., Dartford, Kent, United Kingdom). The oxacillin MIC was determined using the Vitek susceptibility testing system (bioMérieux Inc., St. Louis, Mo.).
RESULTS
Analysis of cultured bacteria.
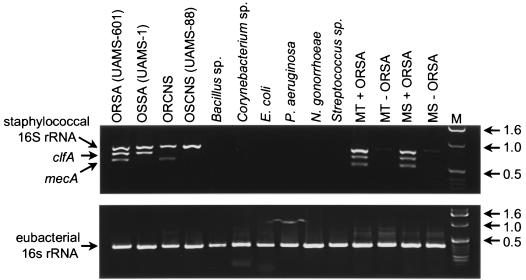
Using genomic DNA from an ORSA strain (UAMS-601), we successfully amplified DNA fragments of approximately 800, 650, and 500 bp (Fig. 1). Based on sequences in the GenBank database, the sizes of these amplification products were consistent with the predicted sizes for the staphylococcal rRNA, clfA, and mecA targets, respectively. The identity of all three fragments was subsequently confirmed by DNA sequencing (data not shown). Sequencing data also confirmed that the actual sizes of the staphylococcal rRNA, clfA, and mecA amplification products matched the predicted sizes of 791, 638, and 499 bp, respectively.
FIG. 1.
Specificity of multiplex PCR. Template DNA was isolated from TSB cultures of the indicated bacteria and subjected to PCR as described in the text. The approximate number of bacteria in the starting sample was 5 × 108 CFU. MT refers to cases in which template DNA was derived from mixed cultures of bacteria containing all of the nonstaphylococcal species with or without the ORSA strain UAMS-601. MS refers to those cases in which template DNA was derived from pure cultures of each nonstaphylococcal species and then mixed prior to analysis with or without template DNA from UAMS-601. (Top) Multiplex PCR utilizing primers for staphylococcal rRNA, clfA, and mecA; (bottom) PCR using primers for conserved regions of eubacterial rRNA genes. Lane M, molecular size markers. Molecular sizes (in kilobases) are on the right.
When genomic DNA from UAMS-1 was used as a template, only the 638- and 791-bp fragments were amplified (Fig. 1). The successful amplification of these two fragments, together with the failure to amplify the 499-bp mecA fragment, was consistent with the observation that UAMS-1 is an OSSA isolate (8). As expected, the ORCNS isolate used in these experiments yielded amplification products of 499 and 791 bp while the OSCNS isolate (UAMS-88) yielded only the 791-bp fragment (Fig. 1). No amplification products were observed using template DNA from any of the nonstaphylococcal species. The fact that a 371-bp fragment corresponding to a conserved region of the eubacterial rRNA genes was amplified from all species (Fig. 1) confirms that the failure of the multiplex protocol to amplify DNA fragments from the nonstaphylococcal species was not due to the absence of template DNA.
Importantly, the ORSA fragment pattern observed when UAMS-601 was examined alone was also observed when UAMS-601 was examined as part of a mixed culture (Fig. 1). That was true whether DNA was derived from a mixed culture or was derived from pure cultures and then mixed together. However, in some cases, we did observe a faint band of approximately 800 bp when mixed cultures were examined in the absence of staphylococcal DNA (Fig. 1). Amplification of this fragment could reflect cross-contamination of the micropipettes but, given our use of pipette tips containing filter barriers, more likely reflects similarities in rRNA genes among eubacterial species. The presence of this fragment would be irrelevant in all cases other than OSCNS isolates, and it would probably not cause a serious diagnostic problem because it is both inconsistent and inefficient. It should also be noted that this fragment was observed only when genomic DNA was prepared from pure cultures containing a large number of bacteria (≥108 CFU); it was not observed in any of the blood cultures tested, including the positive cultures that did not contain staphylococci.
To assess the reproducibility of our protocol, we extended our analysis to include 34 additional S. aureus isolates and 22 additional CNS isolates. Based on oxacillin-resistance profiles as determined by broth MIC, the expected fragment pattern was observed with all 56 isolates (data not shown). Specifically, the three-band (499, 638, and 791 bp) profile observed with the ORSA strain UAMS-601 was also observed with each of 12 additional ORSA strains. The 499-bp mecA fragment was absent in each of 22 additional OSSA isolates. The 638-bp clfA fragment was absent in all 22 of the additional CNS isolates, while the 499-bp fragment was absent only in the 7 CNS isolates that were sensitive to oxacillin (data not shown).
Analysis of blood cultures.
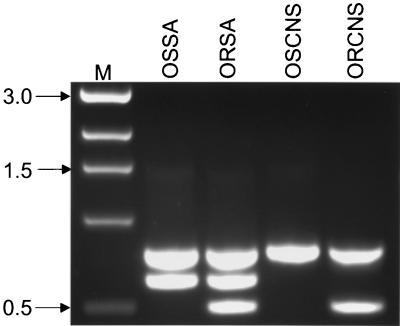
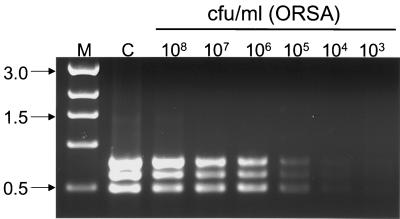
The analysis of blood cultures prepared with ORSA, OSSA, ORCNS, and OSCNS isolates demonstrated that the specificity of our protocol was not altered by the presence of blood products (Fig. 2). We also demonstrated that unambiguous results were obtained with blood cultures containing at least 105 CFU of viable bacteria (Fig. 3). Although that is a relatively high concentration of bacteria, it was well below the level of viable bacteria contained in the positive blood cultures we tested. Specifically, we did viable counts on randomly chosen cultures and found that the minimum concentration of bacteria present before a positive culture was detected was approximately 108 CFU per ml (data not shown).
FIG. 2.
Specificity of blood culture PCR. Blood culture bottles were inoculated with approximately 10 OSSA, ORSA, OSCNS, or ORCNS isolates. Aliquots were processed for template DNA after the culture was identified as positive as described in the text. Lane M, molecular size markers. Approximate sizes (in kilobases) of the amplification products are on the left.
FIG. 3.
Sensitivity of blood culture PCR. A blood culture bottle containing 10 ml of venous blood was inoculated with the ORSA strain UAMS-601 and incubated for 15 h at 37°C. Serial dilutions were prepared using medium from a sterile blood culture as a diluent. DNA isolated from a 1-ml aliquot of cultures containing the indicated number of viable bacteria was subjected to PCR. Lane C, positive control with template DNA derived from a TSB culture of UAMS-601; lane M, molecular size markers. Molecular sizes (in kilobases) are on the left.
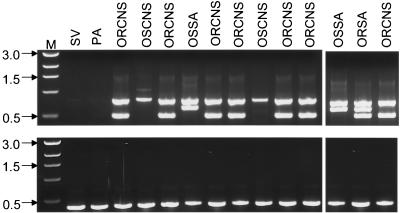
We subsequently tested the utility of our protocol in a blind comparison of 77 positive blood cultures (Fig. 4). These cultures were chosen based only on the detection of a positive culture; the bacteria present in the culture were not characterized prior to PCR analysis. The cultures were simultaneously characterized in the clinical laboratory using conventional methods to determine identity (S. aureus versus CNS species) and antibiotic susceptibility. The results were compared only at the completion of the study. As shown in Table 2, our protocol correctly identified the staphylococcal pathogen and assessed oxacillin resistance in 70 of 77 of the samples tested. Of the seven cultures that did not yield consistent results, one was identified as CNS by our protocol and S. aureus by the clinical lab. We also identified two mecA-positive clinical isolates that were sensitive to oxacillin as determined by the clinical laboratory. The remaining four samples failed to yield any amplification product even when analyzed using the 16S rRNA eubacterial primers (Table 2).
FIG. 4.
Analysis of blood cultures by PCR. Template DNA was isolated from positive blood cultures obtained from the clinical laboratory prior to phenotypic characterization of the bacteria present in the sample. The results shown were chosen because they include all four classes of staphylococci (ORSA, OSSA, ORCNS, and OSCNS) and representative nonstaphylococcal species (SV, viridans group streptococci; PA, P. aeruginosa). (Top) Results obtained with the multiplex protocol; (bottom) results obtained with the eubacterial primers. Lane M, molecular size markers. Molecular sizes (in kilobases) are on the left.
TABLE 2.
Summary of blood culture PCR results
| Category | No. of isolates
|
Total | |
|---|---|---|---|
| Confirmeda | Inconsistentb | ||
| ORSA | 5 | 0 | 5 |
| OSSA | 5 | 1 | 6 |
| ORCNS | 28 | 2 | 30 |
| OSCNS | 4 | 3 | 7 |
| Other | 28 | 1 | 29 |
| Total | 70 | 7 | 77 |
All three parameters evaluated by PCR were confirmed by phenotypic analysis.
At least one of the PCR results did not agree with the phenotypic analysis. The specific nature of these inconsistencies is discussed in the text.
DISCUSSION
The diagnosis of staphylococcal infections is a time-consuming process that is generally dependent on the phenotypic characterization of cultured bacteria (17, 33). However, an increasing number of investigators have employed the tools of molecular biology to facilitate the diagnostic process. Based on its speed and sensitivity, the preferred approach has been the use of PCR to amplify specific target genes (3, 4, 7, 10, 11, 16, 18, 19, 24, 25, 31, 34). In one of the first reports, Greisen et al. (10) used universal primers to amplify a region of the 16S rRNA genes that is conserved among diverse bacterial genera. The PCR was coupled to hybridization analysis employing a series of oligonucleotide probes, some of which were designed to detect S. aureus and the CNS species. A third probe was designed to detect species commonly found as contaminants in clinical samples. This approach was very sensitive; however, the reliance on Southern blots for a definitive diagnosis increased the time, cost, and technical expertise required to carry out the protocol.
Geha et al. (7) also employed universal primers corresponding to highly conserved regions of the eubacterial 16S rRNA genes; however, to increase the focus on the staphylococci, they included a second set of primers corresponding to mecA. Amplification of mecA was found to be a very reliable, although not absolute, indicator of oxacillin resistance. However, based on the use of universal primers for amplification of the rRNA genes, this protocol did not provide any specificity with regard to the identity of the bacterial species present in the sample. For that reason, it had no diagnostic value with respect to oxacillin-sensitive staphylococci. Salisbury et al. (24) attempted to solve this problem by modifying the rRNA gene primers to make them correspond precisely to the 16S rRNA gene sequence found in S. aureus. Not surprisingly, these primers also amplified an rRNA gene fragment from the limited number of CNS species tested. However, because the experiments did not include any nonstaphylococcal species (24), it remained unclear whether their protocol would eliminate false-positive reactions with bacterial species other than the staphylococci. Schmitz et al. (25) solved the problem of differentiating between the staphylococci and other bacterial species by including four primer pairs, one of which corresponded to conserved regions of eubacterial rRNA genes while another was designed to specifically amplify the staphylococcal rRNA genes. The other two primer pairs targeted the coagulase gene (coa) and mecA. The inclusion of four primer pairs meant that the protocol could detect the presence of staphylococci to the exclusion of other eubacterial pathogens, differentiate between S. aureus and other staphylococcal species, and provide an indication of whether staphylococcal isolates were oxacillin resistant (25). Examination of 686 staphylococcal isolates and 100 eubacterial isolates revealed a 100% correlation with the eubacterial rRNA primers, the staphylococcal rRNA primers, and the coa primers. Indeed, the only exceptions to the correlation between PCR and phenotype were five strains that carried mecA but were oxacillin sensitive and five oxacillin-resistant strains that did not yield a mecA amplification product (25). Using our protocol, we identified two strains that fell into the former category but none that fell into the latter. The detection of oxacillin-sensitive strains that carry mecA is not particularly surprising given the multifactorial nature of oxacillin resistance (2). While the detection of such strains by PCR would delay treatment with a preferred class of drugs, the delay would not extend beyond the time frame associated with conventional diagnostic methods, since treatment could be modified as soon as phenotypic susceptibility tests were completed. Also, a primary objective of methods aimed at the direct detection of mecA is to limit the use of alternative drugs (e.g., vancomycin and linezolid) as much as possible, and that objective is not compromised by the use of alternative drugs in the limited number of cases involving mecA-positive, oxacillin-sensitive strains. The existence of oxacillin-resistant strains that are not detected by PCR is more troublesome because these cases are more likely to result in a treatment failure. Although we did not detect any mecA-negative, oxacillin-resistant strains, the relative sample sizes employed in our study (n = 137) and that of Schmitz et al. (n = 686) preclude us from drawing any conclusions about the relative efficiency of our mecA amplification protocol. It does seem clear based on their results that current PCR-based protocols are most appropriately applied as screens that can be used to augment, but not supplant, conventional methods of susceptibility testing.
Most PCR protocols were developed and evaluated using isolated bacterial colonies (25). With respect to blood samples, that requires cultural amplification first by broth culture and subsequently by agar plating. The need for isolated colonies therefore eliminates much of the time savings associated with PCR. Other investigators have reported the direct application of PCR to clinical samples. For example, Mariani et al. (18) developed a PCR protocol capable of detecting bacteria in synovial fluid. While this protocol could detect as few as 100 cells per ml, that level of sensitivity was dependent on Southern blotting of the amplification products (18). Moreover, the protocol employed universal primers and a hybridization probe corresponding to the E. coli 16S rRNA gene. The use of such generic methods eliminates the possibility of a species-specific diagnosis and greatly increases the possibility of false-positive reactions reflecting contamination of the sample during processing. This problem was overcome by Canvin et al. (4), who used a protocol directed toward amplification of the S. aureus nuclease gene (nuc) to track the presence of S. aureus in the synovial fluid of a patient suffering from septic arthritis. Similarly, Carroll et al. (5) reported a mecA PCR protocol that could be used with samples taken directly from BacT/Alert blood culture bottles. Although relatively accurate with respect to providing an indication of oxacillin resistance, the protocol did not provide any diagnostic information with respect to the staphylococcal species present in the sample. More recently, Jaffe et al. (15) reported a blood culture protocol that could distinguish between S. aureus and coagulase-negative species and provide an indication of oxacillin resistance. Although it addressed all of the most relevant diagnostic issues, the protocol was based on independent amplifications of each target gene and had a relatively high sensitivity limit of 109 CFU. In fact, our analysis of positive blood cultures indicated that the cell density consistently exceeded 108 CFU per ml but sometimes failed to reach 109 CFU per ml, particularly if the samples were taken as soon as the culture was identified as positive (data not shown). Moreover, 11 of 77 samples contained more than one bacterial species (data not shown). In such cases, it is certainly possible that the culture will be identified as positive before the density of staphylococci in the culture reaches 109 CFU per ml. The presence of multiple bacterial species also makes it imperative that the sensitivity of the protocol not be limited by the presence of DNA from nonstaphylococcal species.
We believe that our protocol addresses all of these issues in that it can detect any staphylococcal species even in the presence of other bacteria and can distinguish between clinically relevant groups at a level of detection that eliminates the need to isolate bacteria from positive blood cultures. Of the 77 blood cultures examined, only 7 yielded results that were inconsistent with those obtained by the clinical laboratory. Two of these were the mecA-positive, oxacillin-sensitive strains discussed above. A third was a strain that we identified as a CNS species while the clinical lab identified it as S. aureus. There are several possible explanations for this discrepancy. For instance, the Staphaurex assay used by the clinical lab to identify S. aureus is based on the production of protein A and/or a fibrinogen-binding protein. Although the primary fibrinogen-binding protein is ClfA, S. aureus does produce other fibrinogen-binding proteins (e.g., ClfB) (21). At least some S. epidermidis strains are also capable of binding fibrinogen (22). These observations suggest that the discrepancy was probably due to a positive Staphaurex assay rather than a PCR failure; however, it remains possible that this isolate had a clfA polymorphism that prevented amplification but did not limit the ability to bind fibrinogen. It should be noted that we also examined 33 additional blood cultures, all of which were correctly characterized with respect to the presence of S. aureus versus CNS species (data not shown). Because they were isolated from only one of several blood samples, these isolates were considered contaminants obtained during the collection procedure and were not submitted for susceptibility testing. Based on that, we could not evaluate the results of our mecA amplification and did not include the results obtained with these samples in our study. However, it remains noteworthy that our protocol correctly assessed all of the parameters for which comparative data was available. The other four samples that did not yield consistent results in our assay and in the clinical lab all failed to yield any amplification product. At present, it is not possible to determine whether these results reflect an inherent limitation of the protocol or a technical error.
Although we evaluated a number of protocols for the isolation of template DNA, the CTAB protocol was the only one that yielded consistent results. Based largely on this, it takes up to 8 h to obtain results using our PCR analysis. However, that does not extend the analysis time beyond a same-day (24-h) diagnosis. Importantly, 11 of the positive blood cultures we examined did not yield any amplification product and were subsequently found to contain pure cultures of other gram-positive cocci, including Streptococcus spp. (n = 6), Enterococcus spp. (n = 4), and a Micrococcus sp. (n = 1). This further emphasizes the specificity of our assay and its ability to discriminate between closely related species. These results also indicate that our protocol could be used for the rapid and accurate analysis of any positive blood culture as soon as that culture is found to contain gram-positive cocci. Finally, efforts to optimize our DNA isolation protocol, together with continuing advances in PCR technology, strongly suggest that the time frame can be reduced even further without compromising the specificity or sensitivity of the protocol.
ACKNOWLEDGMENTS
This work was supported by grant R29-AI37729 from the National Institute of Allergy and Infectious disease.
The technical assistance of Marcella Gardner, Toni Darville, Scott Allmendinger, and William Lee Mason is greatly appreciated.
REFERENCES
- 1.Archer G L. Staphylococcus epidermidis and other coagulase-negative staphylococci. In: Mandell G L, Douglas R G Jr, Bennett J E, editors. Principles and practice of infectious diseases. New York, N.Y: Churchill Livingstone; 1990. pp. 1511–1518. [Google Scholar]
- 2.Berger-Bachi B. Resistance not mediated by beta-lactamase (methicillin resistance) In: Crossley K B, Archer G L, editors. The staphylococci in human disease. New York, N.Y: Churchill Livingstone; 1997. pp. 158–174. [Google Scholar]
- 3.Brakstad O G, Aasbakk K, Maeland J A. Detection of Staphylococcus aureus by PCR amplification of the nuc gene. J Clin Microbiol. 1992;30:1654–1660. doi: 10.1128/jcm.30.7.1654-1660.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Canvin J M G, Goutcher S C, Hagig M, Gemmel C G, Sturrock R D. Persistence of Staphylococcus aureus as detected by PCR in the synovial fluid of a patient with septic arthritis. Br J Rheumatol. 1997;36:203–206. doi: 10.1093/rheumatology/36.2.203. [DOI] [PubMed] [Google Scholar]
- 5.Carroll K C, Leonard R B, Newcomb-Gayman P L, Hillyard D R. Rapid detection of the staphylococcal mecA gene from BACTEC blood culture bottles by the polymerase chain reaction. Am J Clin Pathol. 1996;106:600–605. doi: 10.1093/ajcp/106.5.600. [DOI] [PubMed] [Google Scholar]
- 6.Foster T J, McDevitt D. Molecular basis of adherence of staphylococci to biomaterials. In: Bisno A L, Waldvogel F A, editors. Infections associated with indwelling medical devices. Washington, D.C.: American Society for Microbiology; 1994. pp. 31–44. [Google Scholar]
- 7.Geha D J, Uhl J R, Gustaferro C A, Persing D H. Multiplex PCR for identification of methicillin-resistant staphylococci in the clinical laboratory. J Clin Microbiol. 1994;32:1768–1772. doi: 10.1128/jcm.32.7.1768-1772.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gillaspy A F, Hickmon S G, Skinner R A, Thomas J R, Nelson C L, Smeltzer M S. Role of the accessory gene regulator (agr) in the pathogenesis of staphylococcal osteomyelitis. Infect Immun. 1995;63:3373–3380. doi: 10.1128/iai.63.9.3373-3380.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Goh S H, Byrne S K, Zhang J L, Chow A W. Molecular typing of Staphylococcus aureus on the basis of coagulase gene polymorphisms. J Clin Microbiol. 1992;30:1642–1645. doi: 10.1128/jcm.30.7.1642-1645.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Greisen K, Loeffelholz M, Purohit A, Leong D. PCR primers and probes for the 16S rRNA gene of most species of pathogenic bacteria, including bacteria found in cerebrospinal fluid. J Clin Microbiol. 1994;32:335–351. doi: 10.1128/jcm.32.2.335-351.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gribaldo S, Cookson B, Saunders N, Marples R, Stanley J. Rapid identification by specific PCR of coagulase-negative staphylococcal species important in hospital infection. J Med Microbiol. 1997;46:45–53. doi: 10.1099/00222615-46-1-45. [DOI] [PubMed] [Google Scholar]
- 12.Hiramatsu K, Hanaki H, Ino T, Yabuta K, Oguri T, Tenover F C. Methicillin-resistant Staphylococcus aureus clinical strain with reduced vancomycin susceptibility. J Antimicrob Chemother. 1997;40:135–136. doi: 10.1093/jac/40.1.135. [DOI] [PubMed] [Google Scholar]
- 13.Hookey J V, Richardson J F, Cookson B D. Molecular typing of Staphylococcus aureus based on PCR restriction fragment length polymorphism and DNA sequence analysis of the coagulase gene. J Clin Microbiol. 1998;36:1083–1089. doi: 10.1128/jcm.36.4.1083-1089.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hussain Z, Stoakes L, Lannigan R, Longo S, Nancekivell B. Evaluation of screening and commercial methods for detection of methicillin resistance in coagulase-negative staphylococci. J Clin Microbiol. 1998;36:273–274. doi: 10.1128/jcm.36.1.273-274.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jaffe R I, Lane J D, Albury S V, Niemeyer D M. Rapid extraction from and direct identification in clinical samples of methicillin-resistant staphylococci using the PCR. J Clin Microbiol. 2000;38:3407–3412. doi: 10.1128/jcm.38.9.3407-3412.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kamph G, Weist K, Swidsinski S, Kegel M, Ruden H. Comparison of screening methods to identify methicillin-resistant Staphylococcus aureus. Eur J Clin Microbiol Infect Dis. 1997;16:301–307. doi: 10.1007/BF01695635. [DOI] [PubMed] [Google Scholar]
- 17.Kloos W E, Bannerman T L. Staphylococcus and Micrococcus. In: Murray P R, Baron E J, Pfaller M A, Tenover F C, Yolken R H, editors. Manual of clinical microbiology. Washington, D.C.: ASM Press; 1995. pp. 282–298. [Google Scholar]
- 18.Mariani B D, Martin D S, Levine M J, Booth R E, Tuan R S. Polymerase chain reaction detection of bacterial infection in total knee arthroplasty. Clin Orthop Relat Res. 1996;331:11–22. doi: 10.1097/00003086-199610000-00003. [DOI] [PubMed] [Google Scholar]
- 19.Martineau F, Picard F J, Roy P H, Ouellette M, Bergeron M G. Species-specific and ubiquitous-DNA-based assays for rapid identification of Staphylococcus aureus. J Clin Microbiol. 1998;36:618–623. doi: 10.1128/jcm.36.3.618-623.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.McDevitt D, Francois P, Vaudaux P, Foster T J. Molecular characterization of the clumping factor (fibrinogen receptor) of Staphylococcus aureus. Mol Microbiol. 1994;11:237–248. doi: 10.1111/j.1365-2958.1994.tb00304.x. [DOI] [PubMed] [Google Scholar]
- 21.Ni Eldhin D, Perkins S, Francois P, Vaudaux P, Hook M, Foster T J. Clumping factor B (ClfB), a new surface-located fibrinogen-binding adhesin of Staphylococcus aureus. Mol Microbiol. 1998;30:245–257. doi: 10.1046/j.1365-2958.1998.01050.x. [DOI] [PubMed] [Google Scholar]
- 22.Nilsson M, Frykberg L, Flock J I, Pei L, Lindberg M, Guss B. A fibrinogen-binding protein of Staphylococcus epidermidis. Infect Immun. 1998;66:2666–2673. doi: 10.1128/iai.66.6.2666-2673.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Noskin G A, Siddiqui F, Storsor V, Hacek D, Peterson L R. In vitro activities of linezolid against important gram-positive bacterial pathogens including vancomycin-resistant enterococci. Antimicrob Agents Chemother. 1999;43:2059–2062. doi: 10.1128/aac.43.8.2059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Salisbury S M, Sabatini L M, Spiegal C A. Identification of methicillin-resistant staphylococci by multiplex PCR assay. Am J Clin Pathol. 1997;107:368–373. doi: 10.1093/ajcp/107.3.368. [DOI] [PubMed] [Google Scholar]
- 25.Schmitz F J, MacKenzie C R, Hofmann B, Verhoef J, Finken-Eigen M, Heinz H P, Kohrer K. Specific information concerning taxonomy, pathogenicity and methicillin resistance of staphylococci obtained by multiplex PCR. J Med Microbiol. 1997;46:773–778. doi: 10.1099/00222615-46-9-773. [DOI] [PubMed] [Google Scholar]
- 26.Schwarzkopf A, Karch H. Genetic variation in Staphylococcus aureus coagulase genes: potential and limits for use as epidemiological marker. J Clin Microbiol. 1994;32:2407–2412. doi: 10.1128/jcm.32.10.2407-2412.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Smeltzer M S, Gillaspy A F, Pratt F L, Thames M D. Comparative evaluation of use of cna, fnbA, fnbB, and hlb for genomic fingerprinting in the epidemiological typing of Staphylococcus aureus. J Clin Microbiol. 1997;35:2444–2449. doi: 10.1128/jcm.35.10.2444-2449.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Smeltzer M S, Gillaspy A F, Pratt F L, Jr, Thames M D, Iandolo J J. Prevalence and chromosomal map location of Staphylococcus aureus adhesin genes. Gene. 1997;196:249–259. doi: 10.1016/s0378-1119(97)00237-0. [DOI] [PubMed] [Google Scholar]
- 29.Smeltzer M S, Pratt F L, Jr, Gillaspy A F, Young L A. Genomic fingerprinting for epidemiological differentiation of Staphylococcus aureus clinical isolates. J Clin Microbiol. 1996;34:1364–1372. doi: 10.1128/jcm.34.6.1364-1372.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tenover F C, Arbeit R, Archer G, Biddle J, Byrne S, Goering R, Hancock G, Hebert G A, Hill B, Hollis R, Jarvis W R, Kreiswirth B, Eisner W, Maslow J, McDougal L K, Miller J M, Mulligan M, Pfaller M A. Comparison of traditional and molecular methods of typing isolates of Staphylococcus aureus. J Clin Microbiol. 1994;32:407–415. doi: 10.1128/jcm.32.2.407-415.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vannuffel P, Gigi A, Ezzidine H, Vandercam B, Delmee M, Wauters G, Gala J L. Specific detection of methicillin-resistant Staphylococcus species by multiplex PCR. J Clin Microbiol. 1994;33:2864–2867. doi: 10.1128/jcm.33.11.2864-2867.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Von Eiff C, Reinert R R, Kresken M, Brauers J, Hafner D, Peters G. Nationwide German multicenter study on prevalence of antibiotic resistance in staphylococcal bloodstream isolates and comparative in vitro activities of quinupristin-dalfopristin. J Clin Microbiol. 2000;38:2819–2823. doi: 10.1128/jcm.38.8.2819-2823.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Waldvogel F A. Staphylococcus aureus (including toxic shock syndrome) In: Mandell G L, Douglas R G Jr, Bennett J E, editors. Principles and practice of infectious diseases. New York, N.Y: Churchill Livingstone; 1990. pp. 1489–1510. [Google Scholar]
- 34.Wallet F, Roussel-Delvallez M, Courcol R J. Choice of a routine method for detecting methicillin-resistance in staphylococci. J Antimicrob Chemother. 1996;37:901–909. doi: 10.1093/jac/37.5.901. [DOI] [PubMed] [Google Scholar]
- 35.Weinstein M P, Mirritt S, van Pelt L, McKinnon M, Zimmer M, Kloos B L, Reller L B. Clinical importance of identifying coagulase-negative staphylococci isolated from blood cultures: evaluation of MicroScan rapid and dried overnight gram-positive panels versus a conventional reference method. J Clin Microbiol. 1998;36:2089–2092. doi: 10.1128/jcm.36.7.2089-2092.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]