Abstract
Pheochromocytomas and paragangliomas are the most heritable endocrine tumors. In addition to the inherited mutation other driver mutations have also been identified in tumor tissues. All these genetic alterations are clustered in distinct groups which determine the pathomechanisms. Most of these tumors are benign and their surgical removal will resolve patient management. However, 5–15% of them are malignant and therapeutical possibilities for them are limited. This review provides a brief insight about the tumorigenesis associated with pheochromocytomas/paragangliomas in order to present them as potential therapeutical targets.
Keywords: pheochromocytoma, paraganglioma, hereditary tumor, susceptibility genes, germline, somatic, metastatic, treatment, personalized medicine
1. Introduction
Pheochromocytomas (PCC) and paragangliomas (PGL, together PPGL) are rare endocrine tumors originating from the chromaffin cells of the embryonic crest. PCC originate from the adrenal medulla whereas PGLs are extra-adrenally located in the abdomen, thorax, pelvis and neck. These tumors secrete catecholamines, except for the head and neck PGLs (HNPGL), which arise from the parasympathetic ganglia. The catecholamine excess prompts the classic, frequently paroxysmal symptoms such as headaches, palpitation, diaphoresis, syncope, abdominal pain, panic attacks, tremor, pallor and diarrhea. These symptoms are generally accompanied by hypertension and massive release of catecholamines can lead to cardiovascular complications as well [1,2]. Overall, approx. 40% of these tumors are developed due to inherited mutations of the various, growing number of PPGL genes which make PPGL the tumors with the highest susceptibility for inherited cancer syndrome [3,4]. Genetic testing is a cornerstone in patient management, affecting both treatment selection and clinical follow-up [5,6]. Although the tissue origin of these tumors is identical, the differences in tumorigenesis are remarkably distinct. Different molecular clusters are distinguished based on the pathogenesis [7]. Cluster 1 (which is further divided into two sub-clusters) represents the pseudohypoxia pathway with either germline or somatic mutation of EGLN1, EGLN2, DLST, FH, IDH3B, MDH2, SDHA, SDHAF2, SDHB, SDHC, SDHD or VHL and the exclusively postzygotic mutations of EPAS1, IDH1 and IDH2 genes. Cluster 2 stands for the kinase-signaling pathway group with germline or somatic mutations in NF1, MAX, MERTK, MET, MYCN, RET or TMEM127 and mutations only reported in sporadic PPGL in BRAF, HRAS and FGFR1 genes. Cluster 3 is a somatic cluster which represents the Wnt signaling pathway with MAML3 fusion genes and the mutations in CSDE1 gene. These clusters are eventually expanded by new candidate genes and novel potential clusters are emerging as well. Categorizing this heterogeneous, complex tumor group yields an opportunity for personalized patient management which results in a better clinical outcome. The diagnosis of malignant disease is established (radiologically or histologically) upon the presence of metastases, which is defined by the presence of chromaffin tissue where chromaffin cells are not typically found. The most common metastatic sites are lymph nodes, bone, liver and lungs [1]. Metastatic disease is rare, but almost half of the cases are associated with SDHB mutation-related PPGL, as patients with germline SDHB mutation have an overall 25% to 50% risk of developing metastatic disease [8,9]. The five-year overall survival ranges from 50% to 70% [10,11,12,13] The risk factors for metastatic disease additionally to germline SDHB-mutation comprise of noradrenergic or dopaminergic phenotype, size (>5 cm), and extra-adrenal location [14,15,16].
This brief review intends to give an insight into the specific molecular pathomechanism of the PPGL clusters and highlights the differences between them regarding clinical outcome and patient management. The clinical relevance of genetic background of PPGL tumors is also summarized to draw the attention to the existing genotype–phenotype correlations and their implication in clinical use.
2. Hereditary Factors
2.1. Pseudohypoxic Cluster
Pseudohypoxic cluster can be divided into two further subgroups: tricarboxylic acid (TCA) cycle-related and VHL/EGLN1/EPAS1-related mutations (Table 1).
Table 1.
Cluster differentiation of the currently known PPGL susceptibility genes.
| Germline and Somatic | Exclusively Somatic | ||
|---|---|---|---|
| Cluster 1: Pseudohypoxic cluster | Cluster 1A | SDHA/B/C/D/AF2, FH, MDH2, IDH3B, GOT2, DLST, SLC25A11 | IDH1/2 |
| Cluster 1B | EGLN1, VHL | EPAS1 | |
| Cluster 2: Kinase signalling cluster | RET, MERTK, MET, NF1, MAX, TMEM127 | FGFR1, HRAS, BRAF | |
| Cluster 3: Wnt signaling cluster | - | CSDE1, UBTF-MAML3 | |
Mutations in the genes encoding the several members of the tricarboxylic acid (TCA) cycle enzymes predispose to PPGL. The four succinate dehydrogenase subunits (SDHA, SDHB, SDHC, and SDHD), the SDH assembly factor (SDHAF2), fumarate hydratase (FH) and malate dehydrogenase 2 (MDH2) have been identified in about 12% to 16% of PPGL patients. Mutation in SDHx or FH genes, represent a high, 20–50% risk for metastatic disease [1,4,8,9,17,18]. Mutations in the genes encoding SDHx and FH result in increased levels of succinate and fumarate, respectively. Upon accumulation, these metabolites act as oncometabolites, altering cellular processes and promoting the survival and proliferation of tumor cells [19]. The elevated level of succinate or fumarate activates the pseudohypoxic pathway by inhibiting Hypoxia-Inducible Factor- (HIF) prolyl hydroxylases (and therefore stabilizing HIF under normoxic conditions) and promotes the hypermethylation of histone and DNA as competitive inhibitors of alpha-ketoglutarate-dependent dioxygenases. These include Jumonji domain-containing histone demethylases and the ten-eleven translocase (TET) family of DNA demethylases. Certain methylated genes, like phenyl-ethanolamine N-methyltransferase (PNMT), linked to neuroendocrine differentiation, may play a role in SDHx-mutated PGL development [20]. Moreover, hypermethylated genomic regions often lead to the silencing of tumor suppressor genes promoting cancer development.
Succinate dehydrogenase (SDH) converts succinate to fumarate as part of the TCA cycle and catalyzes the reduction of ubiquinone to ubiquinol as part of the electron transport chain. The biochemical phenotype of SDHx tumors is characterized by the hypersecretion of dopamine (and its metabolite methoxytyramine) alone or dopamine and norepinephrine. SDHx mutations are accompanied by intra-adrenal tumors less commonly. SDHC and SDHD mutation-associated PGLs are predominantly located in the head and neck region compared to SDHB PGLs that are commonly found in the abdomen [21,22]. The prevalence of malignancy is higher in SDHB-related PPGLs (~30%) compared to SDHA, SDHC and SDHD (incidence of metastasis ~0–4%). The high metastatic risk of SHDB-related PPGLs still remains largely unclear despite extensive research [23].
SUCLG2, a GTP-specific succinyl-CoA synthetase, is a subunit of SUCL succinyl-CoA ligase (SUCL), an enzyme that provides substrate for succinate dehydrogenase (SDH; mitochondrial complex II [CII]). Recently, SUCLG2 has been identified as a novel candidate gene in hereditary PPGL. In the work of Vanova et al. 2021, SUCLG2-mutated PPGL tumors and SUCLG2-deficient chromaffin cells revealed the decrease in the level of the SDHB subunit of SDH, and faulty assembly of the complex II, resulting in aberrant respiration and elevated succinate accumulation [24].
Fumarate hydratase (or fumarase) catalyzes the hydration of fumarate to L-malate in the TCA cycle. Heterozygous germline mutations in FH gene are associated with hereditary leiomyomatosis and renal cell cancer (HLRCC) syndrome and less commonly hereditary PPGL [25].
Germline mutations in malate dehydrogenase 2 (MDH2) have been linked to PPGL tumorigenesis and malignant PPGL development. MDH2 encodes the mitochondrial malate dehydrogenase, which converts malate to oxaloacetate. Accumulation of malate inhibits the α-ketoglutarate dependent enzymes, including HIF-prolyl hydroxylases, and DNA and histone demethylases, leading to a pseudohypoxic and hypermethylator phenotype, oncogenic pathway activation and PPGL formation [26].
Recently, a truncating germline isocitrate dehydrogenase 3B (IDH3B) mutation was found in a patient with PGL. Isocitrate dehydrogenases IDH1 and IDH2 are NADPH-dependent enzymes catalyzing the reversible oxidative decarboxylation of isocitrate to α-ketoglutarate. Unlike IDH1 and IDH2, IDH3 is NAD-dependent and catalyzes the irreversible conversion of isocitrate to α-ketoglutarate. IDH3B mutated tumor samples showed an elevated α-ketoglutarate/isocitrate ratio, which resulted in a hypermethylator phenotype [27].
In another recent work it was shown that SLC25A11 encoding the mitochondrial alpha-ketoglutarate/malate carrier is a novel paraganglioma susceptibility gene. The loss-of-function germline mutations in the SLC25A11 gene decrease alpha-ketoglutarate levels and alter the alpha-ketoglutarate/succinate ratio, which is associated with hypermethylator phenotype and metabolic reprogramming. Moreover, SLC25A11 mutations are strongly associated with the development of metastatic PPGL [28].
Glutamic-oxaloacetic transaminase 2 (GOT2) is a mitochondrial enzyme, which catalyzes irreversible transamination of aspartate and alpha-ketoglutarate to form oxaloacetate and glutamate and is also involved in stimulating the malate/aspartate shuttle. Gain-of-function mutation in GOT2 gene was recently reported in a patient with metastatic PGL. This mutation showed an elevated succinate/fumarate ratio similar to that observed for SDHx PPGLs [27]. This mutation increases intracellular aspartate level as well, which promotes cancer cell proliferation [29].
Mutations in the dihydrolipoamide S-Succinyltransferase encoding DLST gene lead to the accumulation of 2-hydroxyglutarate which similarly to succinate and malate is considered to be an oncometabolite by inhibiting the α-ketoglutarate dependent enzymes (the HIF-prolyl hydroxylases, DNA and histone demethylases). PPGL associated with DLST mutations expressed similar expression and methylation profile to EPAS-1 related PPGL [30].
Under normoxic conditions, the oxygen-dependent EGLN1 (also known as PHD2) enzyme containing the prolyl hydroxylase domain (PHD) binds a hydroxyl group to the HIF-1α and HIF-2α (also known as EPAS1) subunit. This signal is recognized by VHL (Von Hippel–Lindau) protein, which targets the HIF-1α and HIF-2α subunit for proteasomal degradation. Under hypoxic conditions, HIF-1α and HIF-2α accumulate and form a heterodimer with HIFβ, which translocates to the nucleus and functions as a transcription factor [17]. HIF activates the transcription of genes that affect multiple cellular processes and play a role in the process of tumor formation, invasion and survival [31,32]. A common feature of gene mutations associated with the pseudohypoxic pathway is that they induce a hypoxic response under normoxic conditions during constitutive activation of the HIF pathway.
Mutations in the VHL/EGLN1/EPAS1 pathway are 25% hereditary with moderate metastatic risk [4,17]. Germline mutations in the VHL gene cause Von Hippel–Lindau disease, which associates with hemangioblastomas, clear cell renal cell carcinomas, and pheochromocytomas [33]. Of all apparently sporadic PPGL patients, 1% to 13% have germline VHL mutations, [4]. The pheochromocytomas in VHL patients are most commonly intra-adrenal [22,34]. Germline or somatic loss-of-function mutation of EGLN1 or VHL leads to stabilization of HIF-2α, which causes increased transcription of HIF target genes affecting angiogenesis, proliferation and migration.
2.2. Kinase Signaling Cluster
PPGL belonging to this cluster exhibit mostly benign, well differentiated, mostly epinephrine secreting tumors that predominantly occur in the adrenal gland. Germline mutations are present in 20% of cases. Often multiple tumors are present especially in association with the germline mutations of RET gene, but MAX and TMEM127 mutations are associated with multiplex neoplasms as well. Approximately 50–60% of PPGL belong to this cluster. [4]. Unlike cluster 1, these tumors have an intact phenyl-ethanolamine N-methyltransferase (PMNT) function, therefore the epinephrine (Epi)/norepinephrine (NE) levels are elevated. In this cluster syndromic PPGLS are developed due to the activating germline mutations of proto-oncogene RET, MERTK, MET or the deactivating mutations of tumor suppressor NF1, MAX, MYCN and TMEM127.
Mutations of a tyrosine kinase receptor encoding RET gene lead to the continuous activation of the ERK/MAPK pathway. RET mutations associate with the multiplex endocrine neoplasia type 2 (MEN2) syndrome, which is characterized by the development of medullary thyroid cancer, parathyroid tumors and PCC [35,36]. Three different clinical presentations can be developed based on the mutated codons of the RET gene. MEN2A syndrome associated RET mutations results in the ligand-free homodimerization of the RET receptor which activates the PI3K-AKT-mTOR, RAS-RAF-MAPK and JUN kinase pathways. MEN2B-associated mutations affecting the tyrosine kinase domain cause ligand-independent activation of the RET receptor even in monomeric form [36,37,38,39]. The third manifestation is characterized by the sole presence of familial medullary thyroid carcinoma and in terms of pathogenesis it is a variant of MEN2A syndrome. However, the classical categorization of a kindred as FMTC is difficult. In order to not miss PPGL in small kindreds, the diagnosis of FMTC requires more than 10 carriers in the family, multiple carriers or affected members over the age of 50 years, and an adequate medical history, particularly in older members [40]. Others proposed that FMTC can be established in families with four or more cases of MTC in the absence of PCC or parathyroid adenoma/hyperplasia [41,42,43].
Germline activating mutation of MERTK (also known as c-Mer) has been described in a single patient with MEN2-like phenotype (medullary thyroid carcinoma and PCC) and recurrent and metastatic PGL without the presence of germline mutation in the coding and splicing regions of RET or any other PPGL driver gene [44]. MERTK encodes a tyrosine kinase receptor, which similarly to RET, activates the Raf-MEK-Erk and PI3K-Akt-mTOR pathways among others, and has been associated with various cancers [45,46].
The third tyrosine kinase receptor which is associated with inherited PPGL is encoded by the MET gene. Mutations of MET also lead to the continuous activation of the encoded tyrosine kinase receptor and therefore the activation of RAS/MAPK pathway. Germline MET mutations in association with PPGL are rare, but somatic mutations of MET commonly contribute to disease progression [47].
The NF1 gene encodes the neurofibromin tumorsuppressor which inactivates the GAP and RAS proteins. The inactivating mutations of NF1 lead to the well-characterized neurofibromatosis type 1 syndrome (café-au-lait spots, axillary/inguinal freckling, neurofibromas, Lisch nodules, optical gliomas, skeletal abnormalities and increased risk for certain tumors) where the penetrance of PPGL is relatively low [48,49,50].
The MAX gene encodes a basic helix-loop-helix (bHLH) zipper protein which forms a complex with the MYC transcription factor. Most mutations affect the bHLH region, inhibiting the binding of the MAX to other proteins-including the MYC. The inactivating mutations result in the overexpression of MYC associated genes. The MYC–MAX complex regulates more than a thousand genes that regulate the growth, life span and morphology of the cells and the cell cycle [51]. Familial PCCs in association with germline MAX mutations are often bilateral (in approx. 50%) but somatic mutations are documented, as well [52,53,54]. Interestingly, MAX mutation-related PPGL exhibit low PNMT expression which results in a noradrenergic phenotype with normal or slightly elevated metanephrine levels [55].
Germline mutations of TMEM127 are a rare cause of familial PPGL syndrome. The endomembrane protein TMEM127 acts as a negative regulator of mTOR pathway. Inactivating mutations of TMEM127 lead to the decreased phosphorylation and the consecutive activation of mTOR signaling [56,57,58]. PCC is present in one-third of the germline TMEM127 mutation carriers who develop bilateral tumors in 33% of the cases [54,58,59].
KIF1B mutations in PPGL have been described both somatic and germline [60]. However, as these variants are rare, further studies are required to clarify their role in PPGL development, as recent reports have questioned its involvement in PPGL tumorigenesis [61].
3. Somatic Alterations
3.1. Pseudohypoxia Cluster
The solely somatic gain-of-function mutations of HIF-2α/EPAS1 stabilize the HIF-2α protein under normoxic conditions, which similarly to VHL and EPAS1 mutations leads to neovascularization, proliferation and cell migration. Somatic mutations of EPAS1 have been linked to PPGL formation [62,63]. Germline EPAS1 mutations were reported in a family associated with recurrent PPGL but loss of heterozygosity or additional somatic mutations were not confirmed at the somatic level. Therefore, it is suspected that germline EPAS1 mutations are more predisposing than causative factors in PPGL tumorigenesis [64].
Isocitrate dehydrogenases IDH1 (cytoplasmatic enzyme) and IDH2 (mitochondrial, TCA cycle-related enzyme) are reported to be exclusively somatically mutated in PPGL. IDH mutations lead to a neomorphic function, catalyzing the reduction of α-ketoglutarate to 2-hydroxyglutarate, leading to the pseudohypoxic and hypermethylator phenotype described in the previous section [65].
Mosaicism in cluster 1-related PPGL was reported in EPAS1, VHL and SDHB genes [66].
3.2. Kinase Signaling Cluster
In addition to the previously listed both germline and somatic mutated PPGL genes, there are some exclusively somatically mutated ones.
Continuous activating mutations of tyrosine kinase receptor FGFR1 and mutations of the tyrosine kinase HRAS and BRAF activate the RAS/Raf/MAPK/ERK pathway. Pathogenic variants of these genes in association with PPGL were documented exclusively as somatic mutations [44,47,67,68,69,70,71]. Mutations of HRAS in sporadic PPGL are common, whereas FGFR1 mutations are uncommon and BRAF mutations are rarely present [72].
3.3. Wnt Signaling Cluster
To date, only two genes, the CSDE1 (cold shock domain E1) and MAML3 (mastermind like transcriptional coactivator 3), have been identified and included in this purely sporadic group. Somatic mutations of CSDE1 and somatic gene fusions of UBTF (Upstream Binding Transcription Factor) and MAML3 have been reported in a few cases and might be responsible for approx. 5–10% of sporadic PPGL. The PNMT function is intact, therefore elevated Epi/NE ratios can be observed [7].
Somatic gene fusions of UBTF-MAML3 lead to protein gain of function, which leads to DNA hypomethylation and the activation of Wnt and Hedgehog signaling pathways. The fusion genes were reported in a subset of patients with aggressive phenotype and without germline mutation in PPGL driver genes [7,73].
CSDE1 encodes a pluripotent tumor suppressor which participates in cell-type specific apoptosis and differentiation and plays an important role in mRNA stability [74]. As CSDE1 is located at 1p13.2 chromosomic region it could serve as a potential target for 1p deletion observed in PPGL in addition to SDHB loss [7].
4. Potential Role of Novel Genes and Clusters
With the emergence and fine-tuning of next-generation sequencing techniques, novel genes and clusters are regularly proposed and described in association with PPGL tumorigenesis.
The Cancer Genome Atlas (TCGA) describes a small fraction of PPGL presenting a fourth cluster that bears a cortical admixture signature [7]. This proposition is currently debated as it is questioned whether tumor samples could have been contaminated with non-tumoral tissue. As MAX-mutated PPGL were also assigned to this cluster, it raises further questions to be clarified in the future [4]. Additional studies are necessary in order to validate whether it is a unique cluster or just an artifact related to tissue processing.
Recently, the RNA exonuclease 2 protein encoding REXO2 has been suggested as a potential PPGL susceptibility gene [75]. However, as the study described a single family and tumor tissue was not available for assessing allelic loss or loss of protein expression, careful consideration is recommended.
5. Disease-Modifying Factors and Somatic Mosaicism
As the current classification for malignant PPGL is based only on the presentation of distant metastases, eager attempts are made to identify potential disease modifying genetic alterations that could predict disease progression and may serve as cornerstones for the currently limited therapy [13].
Actin-dependent motor Myosin Vb gene B (MYO5B) encodes a motor protein which interacts with Rab-GTPases. Somatic mutations of MYO5B were identified in SDH-deficient tumors and are reported to accelerate tumor progression [76,77]. Similarly to MYO5B, the somatic mutations of MYCN gene have been reported in malignant PPGL harboring germline SDHB mutation [76]. MYCN is a member of the MYC family of transcription factors and encodes the N-myc proto-oncogene protein which regulates various pathways affecting cell survival, tumorigenesis, tumor progression and metastasis development [78].
Genes involved in the process of chromatin remodeling are described to affect PPGL tumorigenesis: Somatic inactivating mutations of ATRX have been reported to drive tumor progression in clinically aggressive PPGL [47,79]. The histone 3.3 encoding H3F3A somatic mutations [44] and the mostly somatic, occasionally germline mutations of the histone methyltransferase encoding KMTD2 [80], have been associated with PPGL tumorigenesis.
Genes related to cancer development have also been linked to PPGL. Both the mutations of cell cycle regulator CDK2NA and the TP53 were reported in PPGL tumors on the somatic level [71].
Somatic mosaicism refers to the presence of a genetically distinct cell population within an organism, hence the potential presence of a pathogenic variant in PPGL genes occurring during the early postzygotic development. Somatic cases have been identified more often since the introduction of NGS technology into genetic diagnostics. Due to its nature, mosaicism can lead to attenuated/partial phenotype of a disease. Indeed, in PPGL, in addition to germline and somatic mutations, mosaicism has been reported in several genes including EPAS1, H3F3A, VHL and SDHB [44,64,66,81,82]. In mosaic cases the well-described genotype–phenotype correlations are not absolutely reliable, similarly to the prediction of other manifestation of the disease. Mosaicism represents a significant difficulty regarding genetic diagnostics and clinical management as the interpretation of the relevance of such variants are challenging.
6. Clinical Relevance of Genetic Background of PPGL
The basis of the clinical relevance of genetic background of PPGL is the well-known genotype–phenotype association described in detail elsewhere [1,18].
From a clinical point of view alterations of the PPGL genes can be divided into two groups: 10 genes (RET, VHL, NF1, SDHD, SDHAF2, SDHC, SDHB, SDHA, TMEM127, MAX) that currently have well-defined genotype–phenotype correlations that can be used in the clinical approaches and the group of other, continuously emerging genes without soundly established genotype–phenotype associations [1,18].
After the PPGL diagnosis is verified, germline genetic testing is recommended due to the high hereditary background [6]. The genetic diagnosis (identification of the alterations of the 10 clinically relevant genes) further guides the tailoring of imaging studies and subsequent medical management based on the well-known genotype–phenotype associations [1]. Thus, PPGL diagnosed as part of complex genetic tumor syndrome, gene-specific further medical investigations and surveillance protocols are recommended (see details in [1]). Examples include medical test and surveillance for additional manifestations of the syndromes in case of pathogenic variants of RET or VHL genes; MR imaging of the abdomen in case of MAX and TMEM127 pathogenic variants, MR imaging of the skull base and neck in case of SDHC and SDHAF2 pathogenic variants, while MRI of the skull base and neck, thorax, retroperitoneum, and pelvis are all recommended when a pathogenic alteration in SDHA, SDHB or SDHD genes is found.
However, when pathogenic gene variants in the other group of PPGL genes are detected, hereditary predisposition is established, but in the lack of well-described genotype-phenotype correlations, only general medical testing, surveillance and family screening can be proposed [1].
The genetic background influences the initial and follow-up biochemical testing, imaging and therapeutic options [1]. The diagnostic and therapeutic relations of PPGL genetics are continuously developing similarly to molecular pathology with several important discoveries summarized below.
7. Using the Genetic Background for Individualized Patient Management
As novel genes and pathomechanisms are described yearly, it becomes evident that uniform diagnostic and therapeutic approaches are not sufficient for each patient affected by PPGL. Germline mutations affect the location and number of tumors, the biochemical characteristics, the risk for malignancy and, therefore, the overall prognosis [83]. Cluster differentiation yields the possibility to personalize the diagnostic and therapeutic approaches. As this has been extensively reviewed in literature [84], this paragraph briefly summarizes the most important hallmarks of the different clusters and how they can be exploited in the clinical setting.
7.1. Implications of Genetic Background for Individualized Diagnostic Approach in Metastatic PPGL
The diagnosis of malignant PPGL is only established by the detection of distant metastasis [6]. Various types of predictive systems have been suggested in order to predict metastatic disease [85]. In terms of genetic background, germline SDHB mutations predispose to higher risk of malignancy [6]. In limited cohorts, mutations of FH [25], MAX [86], SDHD [87], SLC25A11 [28], TERT [88,89] and gene fusions of MAML3 [7] have been reported to be more prevalent among malignant PPGL compared to benign tumors. Somatic mutations of ATRX are also associated with aggressive phenotype and this represents an independent risk factor for the clinical progression of PPGL [79,90]. Regardless of genotype, for primary and metastatic tumor localization, computed tomography (CT) and magnetic resonance imaging (MRI) are used as an initial step.
The awareness of the pathological background of different PPGLs affects the diagnostic imaging [91]. For the initial screening of SDHx mutation carriers, for recurrent cases, tumor staging, metastatic tumors and for the initial localization of occult or ectopic tumors, radionuclide-based functional imaging studies are recommended [5,92]. These can be 123I-metaiodobenzylguanidine (MIBG), 6-18F-fluoro-L-dopa (18F-FDOPA), 18F-fluorodeoxyglucose (18F-FDG), and somatostatin receptor (SSTR) scintigraphy with either using PET technology [gallium-68 DOTATATE (68Ga-DOTATATE)] or traditional γ-camera [technetium-99m-tektrotyd (99mTc-tektrotyd) and indium-111-octreoscan (111In-octreoscan)].
The choice of the nuclear imaging method and of the PET tracer is influenced by the genetic background, the localization and the behavior of PPGL [93,94,95]. Cluster 1A SDHx mutation related PPGL exhibit strong somatostatin receptor 2 (SSTR2) expression therefore functional imaging with SSTR analogue 68Ga-DOTATATE positron emission tomography-computed tomography (PET/CT) offers the most sensitive functional imaging method for the diagnosis and screening of these PPGLs [96,97,98,99]. 18F-FDG-PET imaging for SDHx-related PPGL offers the second most sensitive method especially for rapidly progressive or metastatic disease [13,96,100,101,102]. 18F-FDOPA uptake is shown uniquely in parasympathetic HNPGL, therefore, 18F-FDOPA-PET may be used for SDHD-related HNPGLs [96,103,104,105]. For FH mutation related PPGL 18F-FDOPA-PET is likely to be the most sensitive method but as germline FH mutations are associated with malignant phenotype, 18F-FDG-PET should be considered as a second option [96,106].
Cluster 1B VHL, EPAS1, PHD1/2-related PPGLs show a stronger L-type amino-acid transporter compared to SSTR2 expression, thus, 18F-FDOPA imaging is more sensitive in these tumors [96,107,108].
Among cluster 2 PPGL, MEN2 and NF1 syndrome-associated cases should undergo 18F-FDOPA-PET or 123I-MIBG imaging [100].
123I-MIBG scan is mandatory for candidate patients for 131I-MIBG therapy [13]. It has to be noted, however, that in contrast to 68Ga-DOTATATE-PET and 18F-FDG-PET, MIBG scintigraphy yields lower sensitivity for the diagnosis of VHL and SDHx mutation-related PPGL and metastatic lesions [13,109,110,111] and prediction of MIBG uptake based on the genetic background of PPGL alone is difficult [112].
7.2. Universal and Individualized Therapeutic Approach in Metastatic PPGL
The first line of therapy for PPGL remains the surgical resection with preoperative alpha blockade in order to reduce the catecholamine-related symptoms [6,113]. Alpha blockade is recommended before systematic therapies for disseminated disease as well [13] and should be continued for at least 3 days after both ablative or systemic therapies [13]. Cortical sparing resection is recommended in all hereditary cases due to the high risk or bilateral tumor development and low risk for malignancy which is typically descriptive for cluster 2 tumors [6].
The indication for non-surgical therapies includes irresectable tumors, high tumor burden or metastatic disease. The options consist of radiotherapy, radiofrequency or cryo ablation, tumor embolization or systematic radionuclide therapy and cyclophosphamide–vincristine–dacarbazine (CVD) or temozolomide chemotherapy [13]. In the largest retrospective study, CVD treatment for metastatic PPGL yielded 37% radiographic partial response and 40% biochemical response [114]. In retrospective studies, temozolomide treatment also showed radiographic and biochemical response to therapy [115,116].
MIBG exploits the expression of the norepinephrine transporter for recycling and uptake of amines via a sodium-dependent cell membrane noradrenaline transporter (NET) into the cell. MIBG serves as a substrate for this enzyme and the accumulation of this radiopharmaceutical is taken advantage of both in imaging (123I-MIBG) and therapy (131I-MIBG), as well. In a prospective registrational trial (NCT00874614) for hypertensive metastatic MIBG-avid PPGL treated with High-Specific-Activity 131I-MIBG, 25% of the patients showed at least 50% reduction in all antihypertensive medication for at least 6 months and tumor size reduction was achieved in 23%, whereas stable disease was the case in 69% of the patients [117]. Additionally, despite the expected worse prognosis for patients with lung or liver metastasis, similar survival was observed when compared to those patients who did not have such metastasis [117].
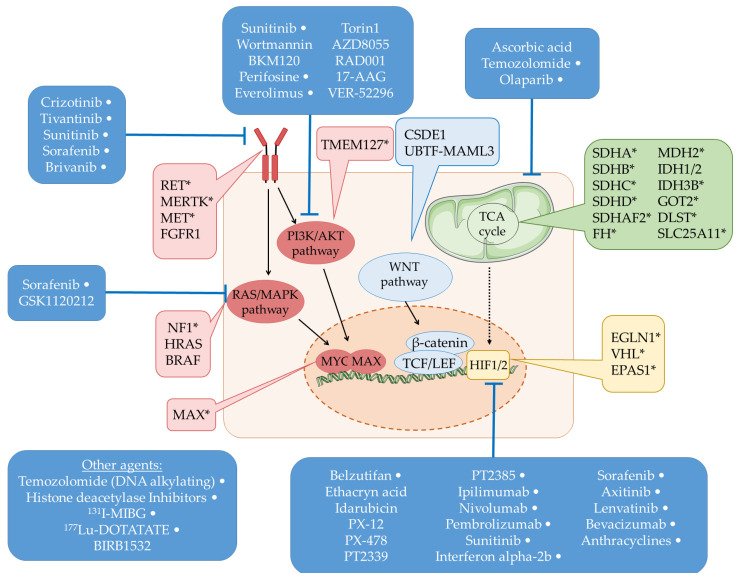
Recently, novel, often cluster and molecular signature-specific therapeutic agents have emerged as promising options for the treatment of metastatic PPGL (Figure 1).
Figure 1.
Schematic representation of the main pathways associated with PPGL tumorigenesis. The genes marked with ‘*’ are germline susceptible PPGL genes as well. The therapeutic agents targeting the associated genes or pathways are also displayed. The therapeutic agents marked with ‘White dot’ represent the specific agents which are involved in clinical trials or approved in the clinical setting, whereas the unmarked agents are being studied in in vitro or in vivo models currently.
7.3. Targeted Therapy for Patients with Cluster 1 Genetic Alterations
As the hallmark of Cluster 1 PPGL is the stabilization of HIF under normoxic conditions, an evident treatment option would be HIF inhibition for these PPGLs. A HIF-2 antagonist, belzutifan, was recently approved as a treatment option as it showed promising results in case of VHL mutated and clear cell renal carcinoma, and is currently being evaluated regarding PPGL treatment [118].
It has been shown that SDHB-mutated PPGLs exhibit dysregulation in oxygen metabolic pathways (pseudohypoxia and increased reactive oxygen species); it has been suggested that targeting the redox balance pathway could be a potential therapeutic approach [119]. Indeed, a recent study reported that targeting SDHB was a promising therapeutic strategy for SDHB-mutated PPGL [119]. Namely, the use of pharmacologic ascorbic acid successfully suppressed SDHB-low metastatic lesions and prolonged overall survival preclinical animal model with PPGL allografts [119].
PPGLs of different clusters exhibit different metabolite profiles. In contrast to sporadic and RET mutation associated PPGL, Cluster 1A related PPGL with impaired TCA cycle exhibit lower levels of lactic acid [120], and increased glutamine/glutamate metabolism to support cellular anabolism [121]. A mitochondrial enzyme, glutaminase-1 (GLS-1), was a target of SDH-related studies: SDHB knockout cells are more sensitive to GLS-1 inhibitors [122,123]. Increased GLS-1 expression in PPGL was associated with more aggressive phenotype and shorter survival [122,124]. The glutaminase inhibitor telaglenastat (CB-839) is currently being studied within the confines of SDH-associated gastrointestinal stromal tumors and non-gastrointestinal stromal tumors as well (NCT02071862). In vitro studies of telaglenastat have demonstrated that the loss of both the respiratory complex I and II is required for effective suppression of proliferation and cell survival [125]. Complex I and II are parts of the mitochondrial electron transport chain which participates in the oxidative phosphorylation. Deciphering the molecular aspects of these tumors will yield novel targets for therapy in the future.
Pseudohypoxia deregulates the cellular energetics which prevent immune system recognition. Namely pseudohypoxia leads to T cell dysfunction, impairs tumor infiltration by T cells and induces resistance to cytotoxic T cells. Ipilimumab, nivolumab and pembrolizumab and currently evaluated as potential therapeutic options to address this phenomenon [126].
As previously mentioned, alongside HNPGLs irrespective of genetic background, SHDx mutation related PGLs show increased SSTR-2 and -3 expression supporting the role of SSTR-targeted therapies for these tumors [127,128]. 177Lu-DOTATATE treatment was recently approved by the US Government Food and Drug Administration following a phase 2 trial with promising results [117]. 177Lu-DOTATATE therapy should be considered for MIBG negative but SSTR-PET-positive patients with malignant PPGL. Compared to MIBG therapy, 177Lu-DOTATATE offers milder side effects and the reports on biochemical and radiographic response are encouraging [129,130,131,132].
7.4. Targeted Therapy for Patients with Alterations of Tyrosine Kinase Signaling Cluster
Tyrosine kinase inhibitors with antiangiogenic potential (axitinib, cabozantinib, dovitinib, lenvantinib, pazopanib and sunitinib) are reported with limited but promising preliminary data for treatment of disseminated PPGL [133]. The potential roles of MET, FGFR1 and TERT inhibitors in PPGL have also been raised and have been tested in clinical trials [84,134].
Pathogenic variants of TMEM127 result in an increase in the mTOR signal that may promote the occurrence of PCC, and the degree of its activation may be related to tumor invasion and metastasis. This gives the rationale of the application of another, commonly used targeted therapeutic approach, mTOR inhibition (everolimus) in PPGL [84,134].
Targeted therapy has many advantages (e.g., fewer side effects, longer survival time, and, consequently, better quality of life) in many tumor types. As neuroendocrine tumors, including PPGL, are generally considered chemo- and radioresistant tumors, targeted therapies in this field hold great promise. It is important to highlight that despite many new discoveries related to PPGL-targeted treatments, the resulting data are still limited and, in many cases, experimental. However, in the near future, they will provide a reference for clinical targeted therapy for PPGL.
8. Conclusions
In the last two decades, as the extraordinary diverse genetic background of PPGL has unfolded, these tumors have become the center of attention for studies in genetics, oncology and biochemistry. These studies are the flagships of translational medicine: the emergence of the high-throughput next-generation sequencing methods has led to the identification of numerous PPGL genes and redefined the clinical genetic screening of these patients and their families; these were closely followed by the studies deciphering the tumorigenesis which yielded novel potential prognostic factors, diagnostic and therapeutic options. Although these tumors are rare, their diversity highlights that even an infrequent tumor should not be treated uniformly. As our knowledge increases, the next decades certainly will be spent under the aegis of personalized and precision healthcare for the patients affected by these tumors.
Author Contributions
Conceptualization, B.S. and A.P.; writing—original draft preparation, B.S. and E.S.; writing—review and editing, H.B. and A.P.; visualization, B.S. and E.S.; supervision, A.P.; project administration, A.P.; funding acquisition, A.P. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Laboratories Excellence program (under the National Tumorbiology Laboratory project (NLP-17).
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Neumann H.P.H., Young W.F., Jr., Eng C. Pheochromocytoma and Paraganglioma. N. Engl. J. Med. 2019;381:552–565. doi: 10.1056/NEJMra1806651. [DOI] [PubMed] [Google Scholar]
- 2.Jain A., Baracco R., Kapur G. Pheochromocytoma and paraganglioma—an update on diagnosis, evaluation, and management. Pediatric Nephrol. 2020;35:581–594. doi: 10.1007/s00467-018-4181-2. [DOI] [PubMed] [Google Scholar]
- 3.Dahia P.L. Pheochromocytoma and paraganglioma pathogenesis: Learning from genetic heterogeneity. Nat. Rev. Cancer. 2014;14:108–119. doi: 10.1038/nrc3648. [DOI] [PubMed] [Google Scholar]
- 4.Crona J., Taïeb D., Pacak K. New Perspectives on Pheochromocytoma and Paraganglioma: Toward a Molecular Classification. Endocr. Rev. 2017;38:489–515. doi: 10.1210/er.2017-00062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lenders J.W.M., Kerstens M.N., Amar L., Prejbisz A., Robledo M., Taieb D., Pacak K., Crona J., Zelinka T., Mannelli M., et al. Genetics, diagnosis, management and future directions of research of phaeochromocytoma and paraganglioma: A position statement and consensus of the Working Group on Endocrine Hypertension of the European Society of Hypertension. J. Hypertens. 2020;38:1443–1456. doi: 10.1097/HJH.0000000000002438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lenders J.W., Duh Q.Y., Eisenhofer G., Gimenez-Roqueplo A.P., Grebe S.K., Murad M.H., Naruse M., Pacak K., Young W.F., Jr. Pheochromocytoma and paraganglioma: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2014;99:1915–1942. doi: 10.1210/jc.2014-1498. [DOI] [PubMed] [Google Scholar]
- 7.Fishbein L., Leshchiner I., Walter V., Danilova L., Robertson A.G., Johnson A.R., Lichtenberg T.M., Murray B.A., Ghayee H.K., Else T., et al. Comprehensive Molecular Characterization of Pheochromocytoma and Paraganglioma. Cancer Cell. 2017;31:181–193. doi: 10.1016/j.ccell.2017.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Andrews K.A., Ascher D.B., Pires D.E.V., Barnes D.R., Vialard L., Casey R.T., Bradshaw N., Adlard J., Aylwin S., Brennan P., et al. Tumour risks and genotype-phenotype correlations associated with germline variants in succinate dehydrogenase subunit genes SDHB, SDHC and SDHD. J. Med. Genet. 2018;55:384–394. doi: 10.1136/jmedgenet-2017-105127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fishbein L., Merrill S., Fraker D.L., Cohen D.L., Nathanson K.L. Inherited mutations in pheochromocytoma and paraganglioma: Why all patients should be offered genetic testing. Ann. Surg. Oncol. 2013;20:1444–1450. doi: 10.1245/s10434-013-2942-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ayala-Ramirez M., Feng L., Johnson M.M., Ejaz S., Habra M.A., Rich T., Busaidy N., Cote G.J., Perrier N., Phan A., et al. Clinical risk factors for malignancy and overall survival in patients with pheochromocytomas and sympathetic paragangliomas: Primary tumor size and primary tumor location as prognostic indicators. J. Clin. Endocrinol. Metab. 2011;96:717–725. doi: 10.1210/jc.2010-1946. [DOI] [PubMed] [Google Scholar]
- 11.Fishbein L., Ben-Maimon S., Keefe S., Cengel K., Pryma D.A., Loaiza-Bonilla A., Fraker D.L., Nathanson K.L., Cohen D.L. SDHB mutation carriers with malignant pheochromocytoma respond better to CVD. Endocr. Relat. Cancer. 2017;24:L51-l55. doi: 10.1530/ERC-17-0086. [DOI] [PubMed] [Google Scholar]
- 12.Hamidi O., Young W.F., Jr., Gruber L., Smestad J., Yan Q., Ponce O.J., Prokop L., Murad M.H., Bancos I. Outcomes of patients with metastatic phaeochromocytoma and paraganglioma: A systematic review and meta-analysis. Clin. Endocrinol. 2017;87:440–450. doi: 10.1111/cen.13434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fishbein L., Del Rivero J., Else T., Howe J.R., Asa S.L., Cohen D.L., Dahia P.L.M., Fraker D.L., Goodman K.A., Hope T.A., et al. The North American Neuroendocrine Tumor Society Consensus Guidelines for Surveillance and Management of Metastatic and/or Unresectable Pheochromocytoma and Paraganglioma. Pancreas. 2021;50:469–493. doi: 10.1097/mpa.0000000000001792. [DOI] [PubMed] [Google Scholar]
- 14.Crona J., Lamarca A., Ghosal S., Welin S., Skogseid B., Pacak K. Genotype-phenotype correlations in pheochromocytoma and paraganglioma: A systematic review and individual patient meta-analysis. Endocr. Relat. Cancer. 2019;26:539–550. doi: 10.1530/ERC-19-0024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hescot S., Curras-Freixes M., Deutschbein T., van Berkel A., Vezzosi D., Amar L., de la Fouchardière C., Valdes N., Riccardi F., Do Cao C., et al. Prognosis of Malignant Pheochromocytoma and Paraganglioma (MAPP-Prono Study): A European Network for the Study of Adrenal Tumors Retrospective Study. J. Clin. Endocrinol. Metab. 2019;104:2367–2374. doi: 10.1210/jc.2018-01968. [DOI] [PubMed] [Google Scholar]
- 16.Eisenhofer G., Lenders J.W., Siegert G., Bornstein S.R., Friberg P., Milosevic D., Mannelli M., Linehan W.M., Adams K., Timmers H.J., et al. Plasma methoxytyramine: A novel biomarker of metastatic pheochromocytoma and paraganglioma in relation to established risk factors of tumour size, location and SDHB mutation status. Eur. J. Cancer. 2012;48:1739–1749. doi: 10.1016/j.ejca.2011.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kantorovich V., Pacak K. New insights on the pathogenesis of paraganglioma and pheochromocytoma. F1000Res. 2018;7:F1000 Faculty Rev-1500. doi: 10.12688/f1000research.14568.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Neumann H.P., Young W.F., Krauss T., Bayley J.-P., Schiavi F., Opocher G., Boedeker C.C., Tirosh A., Castinetti F., Ruf J., et al. 65 YEARS OF THE DOUBLE HELIX: Genetics informs precision practice in the diagnosis and management of pheochromocytoma. Endocr. Relat. Cancer. 2018;25:T201–T219. doi: 10.1530/ERC-18-0085. [DOI] [PubMed] [Google Scholar]
- 19.Ward P.S., Thompson C.B. Metabolic reprogramming: A cancer hallmark even warburg did not anticipate. Cancer Cell. 2012;21:297–308. doi: 10.1016/j.ccr.2012.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Baysal B.E., Maher E.R. 15 YEARS OF PARAGANGLIOMA: Genetics and mechanism of pheochromocytoma-paraganglioma syndromes characterized by germline SDHB and SDHD mutations. Endocr. Relat. Cancer. 2015;22:T71–T82. doi: 10.1530/ERC-15-0226. [DOI] [PubMed] [Google Scholar]
- 21.Kantorovich V., King K.S., Pacak K. SDH-related pheochromocytoma and paraganglioma. Best Pr. Res. Clin. Endocrinol. Metab. 2010;24:415–424. doi: 10.1016/j.beem.2010.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kavinga Gunawardane P.T., Grossman A. The clinical genetics of phaeochromocytoma and paraganglioma. Arch. Endocrinol. Metab. 2017;61:490–500. doi: 10.1590/2359-3997000000299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Benn D.E., Robinson B.G., Clifton-Bligh R.J. 15 YEARS OF PARAGANGLIOMA: Clinical manifestations of paraganglioma syndromes types 1–5. Endocr. Relat. Cancer. 2015;22:T91–T103. doi: 10.1530/ERC-15-0268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hadrava Vanova K., Pang Y., Krobova L., Kraus M., Nahacka Z., Boukalova S., Pack S.D., Zobalova R., Zhu J., Huynh T.T., et al. Germline SUCLG2 Variants in Patients with Pheochromocytoma and Paraganglioma. J. Natl. Cancer Inst. 2021;114:130–138. doi: 10.1093/jnci/djab158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Castro-Vega L.J., Buffet A., De Cubas A.A., Cascon A., Menara M., Khalifa E., Amar L., Azriel S., Bourdeau I., Chabre O., et al. Germline mutations in FH confer predisposition to malignant pheochromocytomas and paragangliomas. Hum. Mol. Genet. 2014;23:2440–2446. doi: 10.1093/hmg/ddt639. [DOI] [PubMed] [Google Scholar]
- 26.Cascon A., Comino-Mendez I., Curras-Freixes M., de Cubas A.A., Contreras L., Richter S., Peitzsch M., Mancikova V., Inglada-Perez L., Perez-Barrios A., et al. Whole-exome sequencing identifies MDH2 as a new familial paraganglioma gene. J. Natl. Cancer Inst. 2015;107:1915–1942. doi: 10.1093/jnci/djv053. [DOI] [PubMed] [Google Scholar]
- 27.Remacha L., Comino-Mendez I., Richter S., Contreras L., Curras-Freixes M., Pita G., Leton R., Galarreta A., Torres-Perez R., Honrado E., et al. Targeted Exome Sequencing of Krebs Cycle Genes Reveals Candidate Cancer-Predisposing Mutations in Pheochromocytomas and Paragangliomas. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2017;23:6315–6324. doi: 10.1158/1078-0432.CCR-16-2250. [DOI] [PubMed] [Google Scholar]
- 28.Buffet A., Morin A., Castro-Vega L.J., Habarou F., Lussey-Lepoutre C., Letouzé E., Lefebvre H., Guilhem I., Haissaguerre M., Raingeard I., et al. Germline Mutations in the Mitochondrial 2-Oxoglutarate/Malate Carrier SLC25A11 Gene Confer a Predisposition to Metastatic Paragangliomas. Cancer Res. 2018;78:1914–1922. doi: 10.1158/0008-5472.CAN-17-2463. [DOI] [PubMed] [Google Scholar]
- 29.Meléndez-Rodríguez F., Urrutia A.A., Lorendeau D., Rinaldi G., Roche O., Böğürcü-Seidel N., Ortega Muelas M., Mesa-Ciller C., Turiel G., Bouthelier A., et al. HIF1α Suppresses Tumor Cell Proliferation through Inhibition of Aspartate Biosynthesis. Cell Rep. 2019;26:2257–2265.e2254. doi: 10.1016/j.celrep.2019.01.106. [DOI] [PubMed] [Google Scholar]
- 30.Remacha L., Pirman D., Mahoney C.E., Coloma J., Calsina B., Currás-Freixes M., Letón R., Torres-Pérez R., Richter S., Pita G., et al. Recurrent Germline DLST Mutations in Individuals with Multiple Pheochromocytomas and Paragangliomas. Am. J. Hum. Genet. 2019;104:651–664. doi: 10.1016/j.ajhg.2019.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pollard P.J., Brière J.J., Alam N.A., Barwell J., Barclay E., Wortham N.C., Hunt T., Mitchell M., Olpin S., Moat S.J., et al. Accumulation of Krebs cycle intermediates and over-expression of HIF1alpha in tumours which result from germline FH and SDH mutations. Hum. Mol. Genet. 2005;14:2231–2239. doi: 10.1093/hmg/ddi227. [DOI] [PubMed] [Google Scholar]
- 32.Selak M.A., Armour S.M., MacKenzie E.D., Boulahbel H., Watson D.G., Mansfield K.D., Pan Y., Simon M.C., Thompson C.B., Gottlieb E. Succinate links TCA cycle dysfunction to oncogenesis by inhibiting HIF-alpha prolyl hydroxylase. Cancer Cell. 2005;7:77–85. doi: 10.1016/j.ccr.2004.11.022. [DOI] [PubMed] [Google Scholar]
- 33.Gaal J., van Nederveen F.H., Erlic Z., Korpershoek E., Oldenburg R., Boedeker C.C., Kontny U., Neumann H.P., Dinjens W.N., de Krijger R.R. Parasympathetic paragangliomas are part of the Von Hippel-Lindau syndrome. J. Clin. Endocrinol. Metab. 2009;94:4367–4371. doi: 10.1210/jc.2009-1479. [DOI] [PubMed] [Google Scholar]
- 34.Karasek D., Shah U., Frysak Z., Stratakis C., Pacak K. An update on the genetics of pheochromocytoma. J. Hum. Hypertens. 2013;27:141–147. doi: 10.1038/jhh.2012.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mulligan L.M., Kwok J.B., Healey C.S., Elsdon M.J., Eng C., Gardner E., Love D.R., Mole S.E., Moore J.K., Papi L., et al. Germ-line mutations of the RET proto-oncogene in multiple endocrine neoplasia type 2A. Nature. 1993;363:458–460. doi: 10.1038/363458a0. [DOI] [PubMed] [Google Scholar]
- 36.Santoro M., Carlomagno F., Romano A., Bottaro D.P., Dathan N.A., Grieco M., Fusco A., Vecchio G., Matoskova B., Kraus M.H., et al. Activation of RET as a dominant transforming gene by germline mutations of MEN2A and MEN2B. Science. 1995;267:381–383. doi: 10.1126/science.7824936. [DOI] [PubMed] [Google Scholar]
- 37.Asai N., Iwashita T., Matsuyama M., Takahashi M. Mechanism of activation of the ret proto-oncogene by multiple endocrine neoplasia 2A mutations. Mol. Cell. Biol. 1995;15:1613–1619. doi: 10.1128/MCB.15.3.1613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Califano D., Rizzo C., D’Alessio A., Colucci-D’Amato G.L., Cali G., Bartoli P.C., Santelli G., Vecchio G., de Franciscis V. Signaling through Ras is essential for ret oncogene-induced cell differentiation in PC12 cells. J. Biol. Chem. 2000;275:19297–19305. doi: 10.1074/jbc.M905866199. [DOI] [PubMed] [Google Scholar]
- 39.Segouffin-Cariou C., Billaud M. Transforming ability of MEN2A-RET requires activation of the phosphatidylinositol 3-kinase/AKT signaling pathway. J. Biol. Chem. 2000;275:3568–3576. doi: 10.1074/jbc.275.5.3568. [DOI] [PubMed] [Google Scholar]
- 40.Brandi M.L., Gagel R.F., Angeli A., Bilezikian J.P., Beck-Peccoz P., Bordi C., Conte-Devolx B., Falchetti A., Gheri R.G., Libroia A., et al. CONSENSUS: Guidelines for Diagnosis and Therapy of MEN Type 1 and Type 2. J. Clin. Endocrinol. Metab. 2001;86:5658–5671. doi: 10.1210/jcem.86.12.8070. [DOI] [PubMed] [Google Scholar]
- 41.Kloos R.T., Eng C., Evans D.B., Francis G.L., Gagel R.F., Gharib H., Moley J.F., Pacini F., Ringel M.D., Schlumberger M., et al. Medullary thyroid cancer: Management guidelines of the American Thyroid Association. Thyroid. 2009;19:565–612. doi: 10.1089/thy.2008.0403. [DOI] [PubMed] [Google Scholar]
- 42.Moline J., Eng C. Multiple endocrine neoplasia type 2: An overview. Genet. Med. Off. J. Am. Coll. Med. Genet. 2011;13:755–764. doi: 10.1097/GIM.0b013e318216cc6d. [DOI] [PubMed] [Google Scholar]
- 43.Eng C. Multiple Endocrine Neoplasia Type 2. In: Adam M.P., Ardinger H.H., Pagon R.A., Wallace S.E., Bean L.J.H., Mirzaa G., Amemiya A., editors. GeneReviews(®) University of Washington; Seattle, WA, USA: 1993. [PubMed] [Google Scholar]
- 44.Toledo R.A., Qin Y., Cheng Z.M., Gao Q., Iwata S., Silva G.M., Prasad M.L., Ocal I.T., Rao S., Aronin N., et al. Recurrent Mutations of Chromatin-Remodeling Genes and Kinase Receptors in Pheochromocytomas and Paragangliomas. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2016;22:2301–2310. doi: 10.1158/1078-0432.CCR-15-1841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cummings C.T., Deryckere D., Earp H.S., Graham D.K. Molecular pathways: MERTK signaling in cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2013;19:5275–5280. doi: 10.1158/1078-0432.CCR-12-1451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Graham D.K., Dawson T.L., Mullaney D.L., Snodgrass H.R., Earp H.S. Cloning and mRNA expression analysis of a novel human protooncogene, c-mer. Cell Growth Differ. 1994;5:647–657. [PubMed] [Google Scholar]
- 47.Castro-Vega L.J., Lepoutre-Lussey C., Gimenez-Roqueplo A.P., Favier J. Rethinking pheochromocytomas and paragangliomas from a genomic perspective. Oncogene. 2016;35:1080–1089. doi: 10.1038/onc.2015.172. [DOI] [PubMed] [Google Scholar]
- 48.Viskochil D., Buchberg A.M., Xu G., Cawthon R.M., Stevens J., Wolff R.K., Culver M., Carey J.C., Copeland N.G., Jenkins N.A., et al. Deletions and a translocation interrupt a cloned gene at the neurofibromatosis type 1 locus. Cell. 1990;62:187–192. doi: 10.1016/0092-8674(90)90252-A. [DOI] [PubMed] [Google Scholar]
- 49.Ballester R., Marchuk D., Boguski M., Saulino A., Letcher R., Wigler M., Collins F. The NF1 locus encodes a protein functionally related to mammalian GAP and yeast IRA proteins. Cell. 1990;63:851–859. doi: 10.1016/0092-8674(90)90151-4. [DOI] [PubMed] [Google Scholar]
- 50.Wallace M.R., Marchuk D.A., Andersen L.B., Letcher R., Odeh H.M., Saulino A.M., Fountain J.W., Brereton A., Nicholson J., Mitchell A.L., et al. Type 1 neurofibromatosis gene: Identification of a large transcript disrupted in three NF1 patients. Science. 1990;249:181–186. doi: 10.1126/science.2134734. [DOI] [PubMed] [Google Scholar]
- 51.Cascón A., Robledo M. MAX and MYC: A heritable breakup. Cancer Res. 2012;72:3119–3124. doi: 10.1158/0008-5472.CAN-11-3891. [DOI] [PubMed] [Google Scholar]
- 52.Blackwood E.M., Lüscher B., Eisenman R.N. Myc and Max associate in vivo. Genes Dev. 1992;6:71–80. doi: 10.1101/gad.6.1.71. [DOI] [PubMed] [Google Scholar]
- 53.Burnichon N., Cascón A., Schiavi F., Morales N.P., Comino-Méndez I., Abermil N., Inglada-Pérez L., de Cubas A.A., Amar L., Barontini M., et al. MAX mutations cause hereditary and sporadic pheochromocytoma and paraganglioma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2012;18:2828–2837. doi: 10.1158/1078-0432.CCR-12-0160. [DOI] [PubMed] [Google Scholar]
- 54.Bausch B., Schiavi F., Ni Y., Welander J., Patocs A., Ngeow J., Wellner U., Malinoc A., Taschin E., Barbon G., et al. Clinical Characterization of the Pheochromocytoma and Paraganglioma Susceptibility Genes SDHA, TMEM127, MAX, and SDHAF2 for Gene-Informed Prevention. JAMA Oncol. 2017;3:1204–1212. doi: 10.1001/jamaoncol.2017.0223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Eisenhofer G., Huynh T.T., Pacak K., Brouwers F.M., Walther M.M., Linehan W.M., Munson P.J., Mannelli M., Goldstein D.S., Elkahloun A.G. Distinct gene expression profiles in norepinephrine- and epinephrine-producing hereditary and sporadic pheochromocytomas: Activation of hypoxia-driven angiogenic pathways in von Hippel-Lindau syndrome. Endocr. Relat. Cancer. 2004;11:897–911. doi: 10.1677/erc.1.00838. [DOI] [PubMed] [Google Scholar]
- 56.Qin Y., Yao L., King E.E., Buddavarapu K., Lenci R.E., Chocron E.S., Lechleiter J.D., Sass M., Aronin N., Schiavi F., et al. Germline mutations in TMEM127 confer susceptibility to pheochromocytoma. Nat. Genet. 2010;42:229–233. doi: 10.1038/ng.533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Deng Y., Qin Y., Srikantan S., Luo A., Cheng Z.-M., Flores S.K., Vogel K.S., Wang E., Dahia P.L.M. The TMEM127 human tumor suppressor is a component of the mTORC1 lysosomal nutrient-sensing complex. Hum. Mol. Genet. 2018;27:1794–1808. doi: 10.1093/hmg/ddy095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yao L., Schiavi F., Cascon A., Qin Y., Inglada-Pérez L., King E.E., Toledo R.A., Ercolino T., Rapizzi E., Ricketts C.J., et al. Spectrum and Prevalence of FP/TMEM127 Gene Mutations in Pheochromocytomas and Paragangliomas. JAMA. 2010;304:2611–2619. doi: 10.1001/jama.2010.1830. [DOI] [PubMed] [Google Scholar]
- 59.Toledo S.P., Lourenço D.M., Jr., Sekiya T., Lucon A.M., Baena M.E., Castro C.C., Bortolotto L.A., Zerbini M.C., Siqueira S.A., Toledo R.A., et al. Penetrance and clinical features of pheochromocytoma in a six-generation family carrying a germline TMEM127 mutation. J. Clin. Endocrinol. Metab. 2015;100:E308–E318. doi: 10.1210/jc.2014-2473. [DOI] [PubMed] [Google Scholar]
- 60.Welander J., Andreasson A., Juhlin C.C., Wiseman R.W., Bäckdahl M., Höög A., Larsson C., Gimm O., Söderkvist P. Rare germline mutations identified by targeted next-generation sequencing of susceptibility genes in pheochromocytoma and paraganglioma. J. Clin. Endocrinol. Metab. 2014;99:E1352–E1360. doi: 10.1210/jc.2013-4375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cardot Bauters C., Leteurtre E., Carnaille B., Do Cao C., Espiard S., Penven M., Destailleur E., Szuster I., Lovecchio T., Leclerc J., et al. Genetic predisposition to neural crest-derived tumors: Revisiting the role of KIF1B. Endocr. Connect. 2020;9:1042–1050. doi: 10.1530/EC-20-0460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Comino-Méndez I., de Cubas A.A., Bernal C., Álvarez-Escolá C., Sánchez-Malo C., Ramírez-Tortosa C.L., Pedrinaci S., Rapizzi E., Ercolino T., Bernini G., et al. Tumoral EPAS1 (HIF2A) mutations explain sporadic pheochromocytoma and paraganglioma in the absence of erythrocytosis. Hum. Mol. Genet. 2013;22:2169–2176. doi: 10.1093/hmg/ddt069. [DOI] [PubMed] [Google Scholar]
- 63.Toledo R.A., Qin Y., Srikantan S., Morales N.P., Li Q., Deng Y., Kim S.-W., Pereira M.A.A., Toledo S.P.A., Su X., et al. In vivo and in vitro oncogenic effects of HIF2A mutations in pheochromocytomas and paragangliomas. Endocr. Relat. Cancer. 2013;20:349–359. doi: 10.1530/ERC-13-0101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lorenzo F.R., Yang C., Ng Tang Fui M., Vankayalapati H., Zhuang Z., Huynh T., Grossmann M., Pacak K., Prchal J.T. A novel EPAS1/HIF2A germline mutation in a congenital polycythemia with paraganglioma. J. Mol. Med. 2013;91:507–512. doi: 10.1007/s00109-012-0967-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Gaal J., Burnichon N., Korpershoek E., Roncelin I., Bertherat J., Plouin P.F., de Krijger R.R., Gimenez-Roqueplo A.P., Dinjens W.N. Isocitrate dehydrogenase mutations are rare in pheochromocytomas and paragangliomas. J. Clin. Endocrinol. Metab. 2010;95:1274–1278. doi: 10.1210/jc.2009-2170. [DOI] [PubMed] [Google Scholar]
- 66.Ben Aim L., Pigny P., Castro-Vega L.J., Buffet A., Amar L., Bertherat J., Drui D., Guilhem I., Baudin E., Lussey-Lepoutre C., et al. Targeted next-generation sequencing detects rare genetic events in pheochromocytoma and paraganglioma. J. Med. Genet. 2019;56:513–520. doi: 10.1136/jmedgenet-2018-105714. [DOI] [PubMed] [Google Scholar]
- 67.Currás-Freixes M., Inglada-Pérez L., Mancikova V., Montero-Conde C., Letón R., Comino-Méndez I., Apellániz-Ruiz M., Sánchez-Barroso L., Aguirre Sánchez-Covisa M., Alcázar V., et al. Recommendations for somatic and germline genetic testing of single pheochromocytoma and paraganglioma based on findings from a series of 329 patients. J. Med. Genet. 2015;52:647–656. doi: 10.1136/jmedgenet-2015-103218. [DOI] [PubMed] [Google Scholar]
- 68.Oudijk L., de Krijger R.R., Rapa I., Beuschlein F., de Cubas A.A., Dei Tos A.P., Dinjens W.N.M., Korpershoek E., Mancikova V., Mannelli M., et al. H-RAS Mutations Are Restricted to Sporadic Pheochromocytomas Lacking Specific Clinical or Pathological Features: Data From a Multi-Institutional Series. J. Clin. Endocrinol. Metab. 2014;99:E1376–E1380. doi: 10.1210/jc.2013-3879. [DOI] [PubMed] [Google Scholar]
- 69.Stenman A., Welander J., Gustavsson I., Brunaud L., Bäckdahl M., Söderkvist P., Gimm O., Juhlin C.C., Larsson C. HRAS mutation prevalence and associated expression patterns in pheochromocytoma. Genes Chromosomes Cancer. 2016;55:452–459. doi: 10.1002/gcc.22347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Luchetti A., Walsh D., Rodger F., Clark G., Martin T., Irving R., Sanna M., Yao M., Robledo M., Neumann H.P., et al. Profiling of somatic mutations in phaeochromocytoma and paraganglioma by targeted next generation sequencing analysis. Int. J. Endocrinol. 2015;2015:138573. doi: 10.1155/2015/138573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Castro-Vega L.J., Letouzé E., Burnichon N., Buffet A., Disderot P.H., Khalifa E., Loriot C., Elarouci N., Morin A., Menara M., et al. Multi-omics analysis defines core genomic alterations in pheochromocytomas and paragangliomas. Nat. Commun. 2015;6:6044. doi: 10.1038/ncomms7044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Toledo R. Genetics of Pheochromocytomas and Paragangliomas: An Overview on the Recently Implicated Genes MERTK, MET, Fibroblast Growth Factor Receptor 1, and H3F3A. Endocrinol. Metab. Clin. N. Am. 2017;46:459–489. doi: 10.1016/j.ecl.2017.01.009. [DOI] [PubMed] [Google Scholar]
- 73.Alzofon N., Koc K., Panwell K., Pozdeyev N., Marshall C.B., Albuja-Cruz M., Raeburn C.D., Nathanson K.L., Cohen D.L., Wierman M.E., et al. Mastermind Like Transcriptional Coactivator 3 (MAML3) Drives Neuroendocrine Tumor Progression. Mol. Cancer Res. MCR. 2021;19:1476–1485. doi: 10.1158/1541-7786.MCR-20-0992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Guo A.X., Cui J.J., Wang L.Y., Yin J.Y. The role of CSDE1 in translational reprogramming and human diseases. Cell Commun. Signal. 2020;18:14. doi: 10.1186/s12964-019-0496-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Laitman Y., Tzur S., Attai R., Tirosh A., Friedman E. Germline variant in REXO2 is a novel candidate gene in familial pheochromocytoma. Genet. Res. 2020;102:e3. doi: 10.1017/S0016672320000038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wilzén A., Rehammar A., Muth A., Nilsson O., Tešan Tomić T., Wängberg B., Kristiansson E., Abel F. Malignant pheochromocytomas/paragangliomas harbor mutations in transport and cell adhesion genes. Int. J. Cancer. 2016;138:2201–2211. doi: 10.1002/ijc.29957. [DOI] [PubMed] [Google Scholar]
- 77.Tomić T.T., Olausson J., Rehammar A., Deland L., Muth A., Ejeskär K., Nilsson S., Kristiansson E., Wassén O.N., Abel F. MYO5B mutations in pheochromocytoma/paraganglioma promote cancer progression. PLoS Genet. 2020;16:e1008803. doi: 10.1371/journal.pgen.1008803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Meškytė E.M., Keskas S., Ciribilli Y. MYC as a Multifaceted Regulator of Tumor Microenvironment Leading to Metastasis. Int. J. Mol. Sci. 2020;21:7710. doi: 10.3390/ijms21207710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Fishbein L., Khare S., Wubbenhorst B., DeSloover D., D’Andrea K., Merrill S., Cho N.W., Greenberg R.A., Else T., Montone K., et al. Whole-exome sequencing identifies somatic ATRX mutations in pheochromocytomas and paragangliomas. Nat. Commun. 2015;6:6140. doi: 10.1038/ncomms7140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Juhlin C.C., Stenman A., Haglund F., Clark V.E., Brown T.C., Baranoski J., Bilguvar K., Goh G., Welander J., Svahn F., et al. Whole-exome sequencing defines the mutational landscape of pheochromocytoma and identifies KMT2D as a recurrently mutated gene. Genes Chromosomes Cancer. 2015;54:542–554. doi: 10.1002/gcc.22267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Garcia-Carbonero R., Matute Teresa F., Mercader-Cidoncha E., Mitjavila-Casanovas M., Robledo M., Tena I., Alvarez-Escola C., Arístegui M., Bella-Cueto M.R., Ferrer-Albiach C., et al. Multidisciplinary practice guidelines for the diagnosis, genetic counseling and treatment of pheochromocytomas and paragangliomas. Clin. Transl. Oncol. 2021;23:1995–2019. doi: 10.1007/s12094-021-02622-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Buffet A., Smati S., Mansuy L., Ménara M., Lebras M., Heymann M.F., Simian C., Favier J., Murat A., Cariou B., et al. Mosaicism in HIF2A-related polycythemia-paraganglioma syndrome. J. Clin. Endocrinol. Metab. 2014;99:E369–E373. doi: 10.1210/jc.2013-2600. [DOI] [PubMed] [Google Scholar]
- 83.Alrezk R., Suarez A., Tena I., Pacak K. Update of Pheochromocytoma Syndromes: Genetics, Biochemical Evaluation, and Imaging. Front. Endocrinol. 2018;9:515. doi: 10.3389/fendo.2018.00515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Liu Y., Liu L., Zhu F. Therapies targeting the signal pathways of pheochromocytoma and paraganglioma. Onco Targets. 2019;12:7227–7241. doi: 10.2147/OTT.S219056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wang Y., Li M., Deng H., Pang Y., Liu L., Guan X. The systems of metastatic potential prediction in pheochromocytoma and paraganglioma. Am. J. Cancer Res. 2020;10:769–780. [PMC free article] [PubMed] [Google Scholar]
- 86.Comino-Méndez I., Gracia-Aznárez F., Schiavi F., Landa I., Leandro-García L., Letón R., Honrado E., Ramos-Medina R., Caronia D., Pita G., et al. Exome sequencing identifies MAX mutations as a cause of hereditary pheochromocytoma. Nat. Genet. 2011;43:663–667. doi: 10.1038/ng.861. [DOI] [PubMed] [Google Scholar]
- 87.Kimura N., Takekoshi K., Naruse M. Risk Stratification on Pheochromocytoma and Paraganglioma from Laboratory and Clinical Medicine. J. Clin. Med. 2018;7:242. doi: 10.3390/jcm7090242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Liu T., Brown T., Juhlin C., Stenman A., Wang N., Bäckdahl M., Healy J., Prasad M., Korah R., Carling T., et al. The activating TERT promoter mutation C228T is recurrent in subsets of adrenal tumors. Endocr. Relat. Cancer. 2014;21:427–434. doi: 10.1530/ERC-14-0016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Dwight T., Flynn A., Amarasinghe K., Benn D.E., Lupat R., Li J., Cameron D.L., Hogg A., Balachander S., Candiloro I.L.M., et al. TERT structural rearrangements in metastatic pheochromocytomas. Endocr. Relat. Cancer. 2018;25:1–9. doi: 10.1530/ERC-17-0306. [DOI] [PubMed] [Google Scholar]
- 90.Job S., Draskovic I., Burnichon N., Buffet A., Cros J., Lépine C., Venisse A., Robidel E., Verkarre V., Meatchi T., et al. Telomerase Activation and ATRX Mutations Are Independent Risk Factors for Metastatic Pheochromocytoma and Paraganglioma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2019;25:760–770. doi: 10.1158/1078-0432.CCR-18-0139. [DOI] [PubMed] [Google Scholar]
- 91.Flores S.K., Estrada-Zuniga C.M., Thallapureddy K., Armaiz-Peña G., Dahia P.L.M. Insights into Mechanisms of Pheochromocytomas and Paragangliomas Driven by Known or New Genetic Drivers. Cancers. 2021;13:4602. doi: 10.3390/cancers13184602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Amar L., Pacak K., Steichen O., Akker S.A., Aylwin S.J.B., Baudin E., Buffet A., Burnichon N., Clifton-Bligh R.J., Dahia P.L.M., et al. International consensus on initial screening and follow-up of asymptomatic SDHx mutation carriers. Nat. Rev. Endocrinol. 2021;17:435–444. doi: 10.1038/s41574-021-00492-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Favier J., Amar L., Gimenez-Roqueplo A.P. Paraganglioma and phaeochromocytoma: From genetics to personalized medicine. Nat. Rev. Endocrinol. 2015;11:101–111. doi: 10.1038/nrendo.2014.188. [DOI] [PubMed] [Google Scholar]
- 94.Ryder S.J., Love A.J., Duncan E.L., Pattison D.A. PET detectives: Molecular imaging for phaeochromocytomas and paragangliomas in the genomics era. Clin. Endocrinol. 2021;95:13–28. doi: 10.1111/cen.14375. [DOI] [PubMed] [Google Scholar]
- 95.Nölting S., Bechmann N., Taieb D., Beuschlein F., Fassnacht M., Kroiss M., Eisenhofer G., Grossman A., Pacak K. Personalized Management of Pheochromocytoma and Paraganglioma. Endocr. Rev. 2021;20:1–41. doi: 10.1210/endrev/bnab019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Taïeb D., Hicks R.J., Hindié E., Guillet B.A., Avram A., Ghedini P., Timmers H.J., Scott A.T., Elojeimy S., Rubello D., et al. European Association of Nuclear Medicine Practice Guideline/Society of Nuclear Medicine and Molecular Imaging Procedure Standard 2019 for radionuclide imaging of phaeochromocytoma and paraganglioma. Eur. J. Nucl. Med. Mol. Imaging. 2019;46:2112–2137. doi: 10.1007/s00259-019-04398-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Ziegler C.G., Brown J.W., Schally A.V., Erler A., Gebauer L., Treszl A., Young L., Fishman L.M., Engel J.B., Willenberg H.S., et al. Expression of neuropeptide hormone receptors in human adrenal tumors and cell lines: Antiproliferative effects of peptide analogues. Proc. Natl. Acad. Sci. USA. 2009;106:15879–15884. doi: 10.1073/pnas.0907843106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Van Essen M., Krenning E.P., De Jong M., Valkema R., Kwekkeboom D.J. Peptide Receptor Radionuclide Therapy with radiolabelled somatostatin analogues in patients with somatostatin receptor positive tumours. Acta. Oncol. 2007;46:723–734. doi: 10.1080/02841860701441848. [DOI] [PubMed] [Google Scholar]
- 99.Taïeb D., Jha A., Treglia G., Pacak K. Molecular imaging and radionuclide therapy of pheochromocytoma and paraganglioma in the era of genomic characterization of disease subgroups. Endocr. Relat. Cancer. 2019;26:R627–R652. doi: 10.1530/ERC-19-0165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Taïeb D., Timmers H.J., Hindié E., Guillet B.A., Neumann H.P., Walz M.K., Opocher G., de Herder W.W., Boedeker C.C., de Krijger R.R., et al. EANM 2012 guidelines for radionuclide imaging of phaeochromocytoma and paraganglioma. Eur. J. Nucl. Med. Mol. Imaging. 2012;39:1977–1995. doi: 10.1007/s00259-012-2215-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Jha A., de Luna K., Balili C.A., Millo C., Paraiso C.A., Ling A., Gonzales M.K., Viana B., Alrezk R., Adams K.T., et al. Clinical, Diagnostic, and Treatment Characteristics of SDHA-Related Metastatic Pheochromocytoma and Paraganglioma. Front. Oncol. 2019;9:53. doi: 10.3389/fonc.2019.00053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Janssen I., Blanchet E.M., Adams K., Chen C.C., Millo C.M., Herscovitch P., Taieb D., Kebebew E., Lehnert H., Fojo A.T., et al. Superiority of [68Ga]-DOTATATE PET/CT to Other Functional Imaging Modalities in the Localization of SDHB-Associated Metastatic Pheochromocytoma and Paraganglioma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2015;21:3888–3895. doi: 10.1158/1078-0432.CCR-14-2751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Jha A., Ling A., Millo C., Chen C., Gupta G., Viana B., Gonzales M., Adams K., Herscovitch P., Lin F., et al. Superiority of 68Ga-DOTATATE PET/CT to other functional and anatomic imaging modalities in the detection of SDHD-related pheochromocytoma and paraganglioma—A comparative prospective study. J. Nucl. Med. 2018;59:46. [Google Scholar]
- 104.Janssen I., Chen C.C., Taieb D., Patronas N.J., Millo C.M., Adams K.T., Nambuba J., Herscovitch P., Sadowski S.M., Fojo A.T., et al. 68Ga-DOTATATE PET/CT in the Localization of Head and Neck Paragangliomas Compared with Other Functional Imaging Modalities and CT/MRI. J. Nucl. Med. 2016;57:186–191. doi: 10.2967/jnumed.115.161018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Taïeb D., Pacak K. New Insights into the Nuclear Imaging Phenotypes of Cluster 1 Pheochromocytoma and Paraganglioma. Trends Endocrinol. Metab. 2017;28:807–817. doi: 10.1016/j.tem.2017.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Joshua A.M., Ezzat S., Asa S.L., Evans A., Broom R., Freeman M., Knox J.J. Rationale and Evidence for Sunitinib in the Treatment of Malignant Paraganglioma/Pheochromocytoma. J. Clin. Endocrinol. Metab. 2009;94:5–9. doi: 10.1210/jc.2008-1836. [DOI] [PubMed] [Google Scholar]
- 107.Därr R., Nambuba J., Del Rivero J., Janssen I., Merino M., Todorovic M., Balint B., Jochmanova I., Prchal J.T., Lechan R.M., et al. Novel insights into the polycythemia-paraganglioma-somatostatinoma syndrome. Endocr. Relat. Cancer. 2016;23:899–908. doi: 10.1530/ERC-16-0231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Janssen I., Chen C.C., Zhuang Z., Millo C.M., Wolf K.I., Ling A., Lin F.I., Adams K.T., Herscovitch P., Feelders R.A., et al. Functional Imaging Signature of Patients Presenting with Polycythemia/Paraganglioma Syndromes. J. Nucl. Med. 2017;58:1236–1242. doi: 10.2967/jnumed.116.187690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Gimenez-Roqueplo A.P., Caumont-Prim A., Houzard C., Hignette C., Hernigou A., Halimi P., Niccoli P., Leboulleux S., Amar L., Borson-Chazot F., et al. Imaging work-up for screening of paraganglioma and pheochromocytoma in SDHx mutation carriers: A multicenter prospective study from the PGL.EVA Investigators. J. Clin. Endocrinol. Metab. 2013;98:E162–E173. doi: 10.1210/jc.2012-2975. [DOI] [PubMed] [Google Scholar]
- 110.Kaji P., Carrasquillo J.A., Linehan W.M., Chen C.C., Eisenhofer G., Pinto P.A., Lai E.W., Pacak K. The role of 6-[18F] fluorodopamine positron emission tomography in the localization of adrenal pheochromocytoma associated with von Hippel-Lindau syndrome. Eur. J. Endocrinol. 2007;156:483–487. doi: 10.1530/EJE-06-0712. [DOI] [PubMed] [Google Scholar]
- 111.Kroiss A.S., Uprimny C., Shulkin B.L., Frech A., Tilg H., Gasser R.W., Sprinzl G.M., Gruber L., Thomé C., Plangger C., et al. Compared to (123)I-MIBG SPECT/CT, (18)F-DOPA PET/CT provides accurate tumor extent in patients with extra-adrenal paraganglioma. Ann. Nucl. Med. 2017;31:357–365. doi: 10.1007/s12149-017-1162-7. [DOI] [PubMed] [Google Scholar]
- 112.van Berkel A., Rao J.U., Lenders J.W., Pellegata N.S., Kusters B., Piscaer I., Hermus A.R., Plantinga T.S., Langenhuijsen J.F., Vriens D., et al. Semiquantitative 123I-Metaiodobenzylguanidine Scintigraphy to Distinguish Pheochromocytoma and Paraganglioma from Physiologic Adrenal Uptake and Its Correlation with Genotype-Dependent Expression of Catecholamine Transporters. J. Nucl. Med. 2015;56:839–846. doi: 10.2967/jnumed.115.154815. [DOI] [PubMed] [Google Scholar]
- 113.Fassnacht M., Assie G., Baudin E., Eisenhofer G., de la Fouchardiere C., Haak H.R., de Krijger R., Porpiglia F., Terzolo M., Berruti A. Adrenocortical carcinomas and malignant phaeochromocytomas: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2020;31:1476–1490. doi: 10.1016/j.annonc.2020.08.2099. [DOI] [PubMed] [Google Scholar]
- 114.Niemeijer N.D., Alblas G., van Hulsteijn L.T., Dekkers O.M., Corssmit E.P. Chemotherapy with cyclophosphamide, vincristine and dacarbazine for malignant paraganglioma and pheochromocytoma: Systematic review and meta-analysis. Clin. Endocrinol. 2014;81:642–651. doi: 10.1111/cen.12542. [DOI] [PubMed] [Google Scholar]
- 115.Kulke M.H., Stuart K., Enzinger P.C., Ryan D.P., Clark J.W., Muzikansky A., Vincitore M., Michelini A., Fuchs C.S. Phase II study of temozolomide and thalidomide in patients with metastatic neuroendocrine tumors. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2006;24:401–406. doi: 10.1200/JCO.2005.03.6046. [DOI] [PubMed] [Google Scholar]
- 116.Hadoux J., Favier J., Scoazec J.Y., Leboulleux S., Al Ghuzlan A., Caramella C., Déandreis D., Borget I., Loriot C., Chougnet C., et al. SDHB mutations are associated with response to temozolomide in patients with metastatic pheochromocytoma or paraganglioma. Int. J. Cancer. 2014;135:2711–2720. doi: 10.1002/ijc.28913. [DOI] [PubMed] [Google Scholar]
- 117.Pryma D.A., Chin B.B., Noto R.B., Dillon J.S., Perkins S., Solnes L., Kostakoglu L., Serafini A.N., Pampaloni M.H., Jensen J., et al. Efficacy and Safety of High-Specific-Activity (131)I-MIBG Therapy in Patients with Advanced Pheochromocytoma or Paraganglioma. J. Nucl. Med. 2019;60:623–630. doi: 10.2967/jnumed.118.217463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Deeks E.D. Belzutifan: First Approval. Drugs. 2021;81:1921–1927. doi: 10.1007/s40265-021-01606-x. [DOI] [PubMed] [Google Scholar]
- 119.Liu Y., Pang Y., Zhu B., Uher O., Caisova V., Huynh T.T., Taieb D., Hadrava Vanova K., Ghayee H.K., Neuzil J., et al. Therapeutic Targeting of SDHB-Mutated Pheochromocytoma/Paraganglioma with Pharmacologic Ascorbic Acid. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2020;26:3868–3880. doi: 10.1158/1078-0432.CCR-19-2335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Rao J.U., Engelke U.F.H., Sweep F.C.G.J., Pacak K., Kusters B., Goudswaard A.G., Hermus A.R.M.M., Mensenkamp A.R., Eisenhofer G., Qin N., et al. Genotype-Specific Differences in the Tumor Metabolite Profile of Pheochromocytoma and Paraganglioma Using Untargeted and Targeted Metabolomics. J. Clin. Endocrinol. Metab. 2015;100:E214–E222. doi: 10.1210/jc.2014-2138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Lussey-Lepoutre C., Hollinshead K.E.R., Ludwig C., Menara M., Morin A., Castro-Vega L.-J., Parker S.J., Janin M., Martinelli C., Ottolenghi C., et al. Loss of succinate dehydrogenase activity results in dependency on pyruvate carboxylation for cellular anabolism. Nat. Commun. 2015;6:8784. doi: 10.1038/ncomms9784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Sarkadi B., Meszaros K., Krencz I., Canu L., Krokker L., Zakarias S., Barna G., Sebestyen A., Papay J., Hujber Z., et al. Glutaminases as a Novel Target for SDHB-Associated Pheochromocytomas/Paragangliomas. Cancers. 2020;12:599. doi: 10.3390/cancers12030599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Kitazawa S., Ebara S., Ando A., Baba Y., Satomi Y., Soga T., Hara T. Succinate dehydrogenase B-deficient cancer cells are highly sensitive to bromodomain and extra-terminal inhibitors. Oncotarget. 2017;8:28922–28938. doi: 10.18632/oncotarget.15959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Kim H.M., Koo J.S. Expression of Glutamine Metabolism-Related and Amino Acid Transporter Proteins in Adrenal Cortical Neoplasms and Pheochromocytomas. Dis. Markers. 2021;2021:8850990. doi: 10.1155/2021/8850990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Lorendeau D., Rinaldi G., Boon R., Spincemaille P., Metzger K., Jager C., Christen S., Dong X., Kuenen S., Voordeckers K., et al. Dual loss of succinate dehydrogenase (SDH) and complex I activity is necessary to recapitulate the metabolic phenotype of SDH mutant tumors. Metab. Eng. 2017;43:187–197. doi: 10.1016/j.ymben.2016.11.005. [DOI] [PubMed] [Google Scholar]
- 126.Jimenez C., Subbiah V., Stephen B., Ma J., Milton D., Xu M., Zarifa A., Akhmedzhanov F.O., Tsimberidou A., Habra M.A., et al. Phase II Clinical Trial of Pembrolizumab in Patients with Progressive Metastatic Pheochromocytomas and Paragangliomas. Cancers. 2020;12:2307. doi: 10.3390/cancers12082307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Elston M.S., Meyer-Rochow G.Y., Conaglen H.M., Clarkson A., Clifton-Bligh R.J., Conaglen J.V., Gill A.J. Increased SSTR2A and SSTR3 expression in succinate dehydrogenase-deficient pheochromocytomas and paragangliomas. Hum. Pathol. 2015;46:390–396. doi: 10.1016/j.humpath.2014.11.012. [DOI] [PubMed] [Google Scholar]
- 128.Han S., Suh C.H., Woo S., Kim Y.J., Lee J.J. Performance of 68Ga-DOTA–Conjugated Somatostatin Receptor–Targeting Peptide PET in Detection of Pheochromocytoma and Paraganglioma: A Systematic Review and Metaanalysis. J. Nucl. Med. 2019;60:369–376. doi: 10.2967/jnumed.118.211706. [DOI] [PubMed] [Google Scholar]
- 129.Kong G., Grozinsky-Glasberg S., Hofman M.S., Callahan J., Meirovitz A., Maimon O., Pattison D.A., Gross D.J., Hicks R.J. Efficacy of Peptide Receptor Radionuclide Therapy for Functional Metastatic Paraganglioma and Pheochromocytoma. J. Clin. Endocrinol. Metab. 2017;102:3278–3287. doi: 10.1210/jc.2017-00816. [DOI] [PubMed] [Google Scholar]
- 130.Forrer F., Riedweg I., Maecke H.R., Mueller-Brand J. Radiolabeled DOTATOC in patients with advanced paraganglioma and pheochromocytoma. Q. J. Nucl. Med. Mol. Imaging. 2008;52:334–340. [PubMed] [Google Scholar]
- 131.Vyakaranam A.R., Crona J., Norlén O., Granberg D., Garske-Román U., Sandström M., Fröss-Baron K., Thiis-Evensen E., Hellman P., Sundin A. Favorable Outcome in Patients with Pheochromocytoma and Paraganglioma Treated with (177)Lu-DOTATATE. Cancers. 2019;11:909. doi: 10.3390/cancers11070909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Zandee W.T., Feelders R.A., Smit Duijzentkunst D.A., Hofland J., Metselaar R.M., Oldenburg R.A., van Linge A., Kam B.L.R., Teunissen J.J.M., Korpershoek E., et al. Treatment of inoperable or metastatic paragangliomas and pheochromocytomas with peptide receptor radionuclide therapy using 177Lu-DOTATATE. Eur. J. Endocrinol. 2019;181:45–53. doi: 10.1530/EJE-18-0901. [DOI] [PubMed] [Google Scholar]
- 133.Jimenez C., Fazeli S., Román-Gonzalez A. Antiangiogenic therapies for pheochromocytoma and paraganglioma. Endocr. Relat. Cancer. 2020;27:R239–R254. doi: 10.1530/ERC-20-0043. [DOI] [PubMed] [Google Scholar]
- 134.Pang Y., Liu Y., Pacak K., Yang C. Pheochromocytomas and Paragangliomas: From Genetic Diversity to Targeted Therapies. Cancers. 2019;11:436. doi: 10.3390/cancers11040436. [DOI] [PMC free article] [PubMed] [Google Scholar]