The Importance of Diversity in Pancreatic Cancer Clinical Trials
Pancreatic cancer is defined by the lethality of the disease. With an estimated 5-year survival rate of 9% and increasing incidence, it is projected to soon become the second leading cause of cancer deaths in the United States (US).1 Pancreatic cancer does not affect all populations equally, as significant health disparities exist between race, ethnicity and gender. Over the last two decades, Black individuals have persistently had a 19% higher incidence of pancreatic cancer when compared to other racial groups.2 They present at younger ages with more advanced disease. Moreover, Black patients with pancreatic cancer experience a higher age-adjusted mortality rate compared to all other racial groups (13.3 versus 11.0 per 100,000 population of all other racial/ethnic groups combined). Native American patients also have higher rates of death from pancreatic cancer with an estimated 5-year survival rate of 6.7%.2 In contrast, Hispanic patients diagnosed with pancreatic cancer have the longest median and overall survival when compared to other ethnic groups.3 These disparities are not entirely accounted for by comorbidities, socioeconomic status and access to high-quality healthcare alone, suggesting that biologic processes may be partially responsible.4
Though it is critical to address the systemic injustices, institutional racism and disadvantages faced by minority populations, ancestral differences in tumor biology cannot be overlooked. Recently, increased focus has been applied to exploring racial and ethnic differences in patient and tumor biology through a multi-omics approach. Oncologic disparities have been identified based on differences in germline and somatic mutations, molecular subtypes, tumor immune response, epigenetics and microbiota.5 Black patients with pancreatic cancer are more likely to have KRAS somatic mutations and HER2 protein overexpression when compared to White patients.6 Germline loss-of-function mutations in BRCA 1/2 genes and in CDKN2A tumor suppressor genes are also more common in patients with African ancestry. Patients with germline BRCA and CDKN2A mutations are at an increased risk for pancreatic cancer and may exhibit different treatment responses to targeted therapies.7,8 Given these and other racial and ethnic differences in pancreatic cancer tumor biology, it is unlikely that all patients will exhibit similar outcomes to trialed therapeutics.
In order to address these inequities, Congress issued the Final Rule for Clinical Trials Registration and Results Information Submission (42 CFR Part 11) requiring race and ethnicity reporting in ClinicalTrials.gov for clinical trial participants. Given the implementation of these requirements, we sought to assess the current state of diversity and inclusion and by investigating trends in demographic data reporting and assessing its impact on the diversity of pancreatic cancer clinical trials. We hypothesized that racial and ethnic minority patients with pancreatic cancer continue to be underrepresented, despite the federal requirement for more transparent trial demographic reporting. As a consequence, participation underrepresentation in clinical trials likely perpetuates the health disparities found in pancreatic cancer treatment and outcomes.
Assessing Pancreatic Cancer Clinical Trials
To evaluate trends in diversity and inclusion, a comprehensive review of pancreatic cancer clinical trials was performed using the US National Library of Medicine’s ClinicalTrials.gov database. Search criteria included “pancreatic cancer” and “pancreatic carcinoma.” Subtopics included “pancreatic neoplasm,” “pancreas cancer,” “cancer of the pancreas,” “cancer of pancreas,” “pancreatic tumors,” “carcinoma of the pancreas,” and “neoplasm of pancreas.” The search was filtered to include adult study participants (> 18 years of age) and interventional (clinical trial) studies. Trials were limited to “with results,” which includes those with study results information included in the study record under the Study Results tab. All study phases were included. The study phase labelled not applicable (N/A) describes device or behavioral interventions that are not defined by the Food and Drug Administration (FDA). The database was queried on December 21, 2020 and this initial search yielded 337 clinical trial results. One hundred and thirty clinical trials were excluded because they were conducted outside of the US (n=44), included other cancer types (n=83) or other pancreatic pathologies (n=3). These exclusion criteria were selected to cleanly analyze pancreatic cancer clinical trials within the US. Descriptive statistics were recorded from provided study results. Results were cross-referenced with formally linked publications on ClinicalTrials.gov as well as PubMed.gov using trial registry numbers in the secondary source identification (SI) field. Results were reported on ClinicalTrials.gov by gender (female, male), ethnicity (Hispanic or Latino, Not Hispanic or Latino, Unknown or Not Reported) and race (American Indian or Alaska Native, Asian, Native Hawaiian or Other Pacific Islander, Black or African American, White, More than one race, and Unknown or Not reported). Due to the lack of racial and ethnic demographic reporting from 2005–2007, these data were excluded from longitudinal analysis (n=1). Clinical trial sponsors were categorized by primary sponsor type into university/cancer center, National Cancer Institute (NCI), pharmaceuticals/biotech, and private hospitals.
The enrollment fraction (EF) was calculated for each trial and subgroup population. The term EF was initially described by Murthy et al. and is defined as the number of clinical trial participants divided by the US incidence of cancer in each race, ethnicity and gender subgroup.10 Wilcoxon rank sum tests were used to determine whether the center of the distribution of EF ratios among all trials differed from 1. Incident cases of pancreatic cancer were collected through the North American Association of Central Cancer Registries (NAACCR) from 2013–2017 and age-adjusted to the 2000 US Standard Population.11 Pancreatic cancer incidence was unavailable for “multiracial” and “unknown” racial and ethnic subgroups so EF analysis could not be calculated. All analyses were performed using the R statistical software package (V4.0.2, The R Foundation for Statistical Computing).
Are trials reporting data participant race, ethnicity and gender?
Two-hundred seven pancreatic cancer clinical trials comprised of 8,429 pooled participants were included with results reported between 2005 and 2020. In this timespan, race and ethnicity were reported in only 49.3% and 34.7% of studies respectively, whereas 99% of studies reported participants’ gender (supplemental table 1). Reporting of participant race and ethnicity data however has improved substantially over time (p<0.0001) (Figure 1A). In 2019, 100% of trials reported data on race and 73.3% of trials reported data on ethnicity. In 2020, 100% of race and ethnicity data was reported, though only two trials meeting inclusion criteria had publicly reported results at the time of data analysis. These data represent an encouraging trend toward improved sponsor reporting of race and ethnicity data over the last decade.
Figure 1.
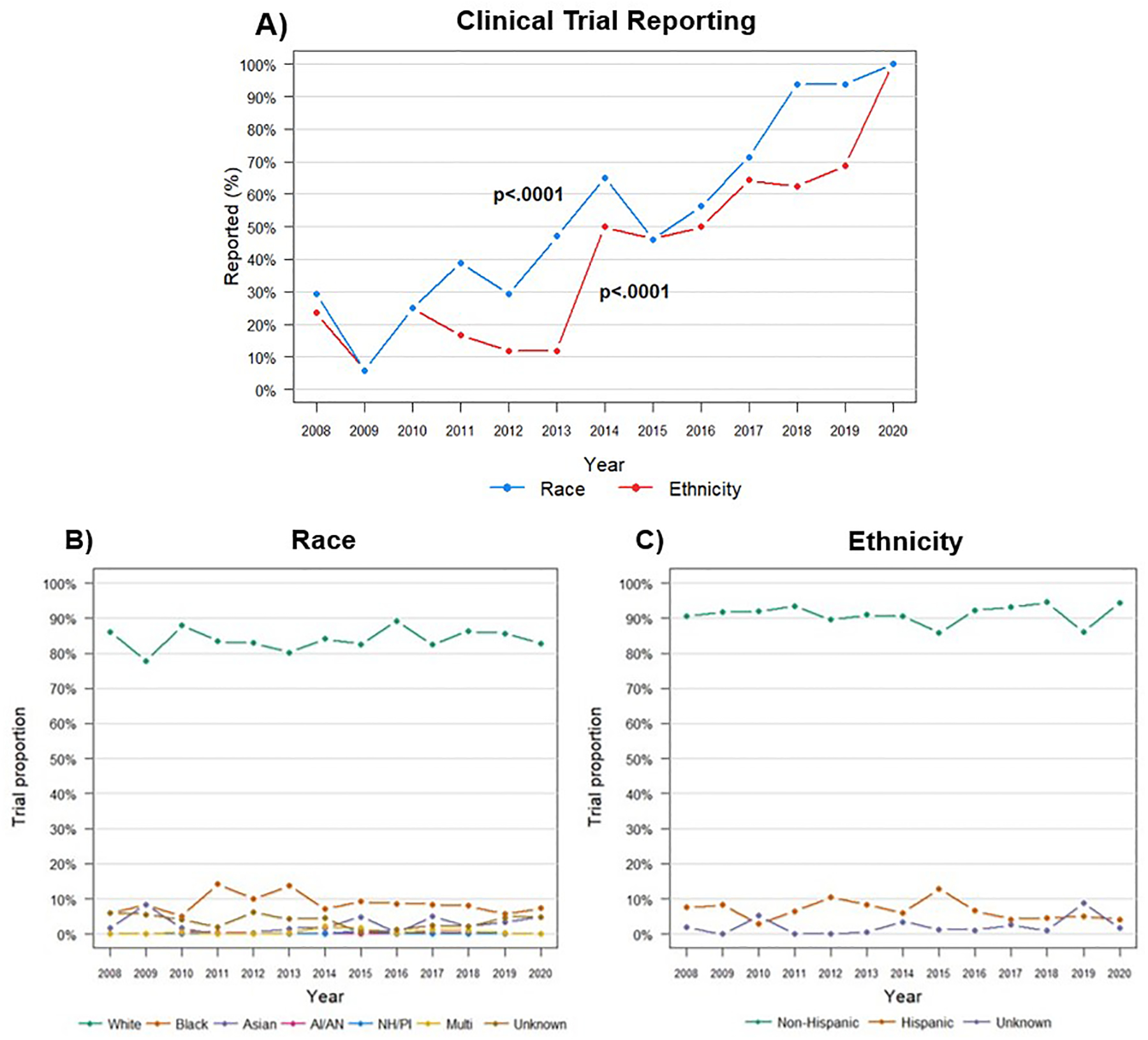
Longitudinal analysis of race and ethnicity reporting and trial diversity
Abbreviations: AI/AN, American Indian/Alaskan Native, NH/PI, Native Hawaiian/Pacific Islander, Multi, Multiracial.
Reporting of race and ethnicity has continued to improve over time (A), whereas racial and ethnic diversity of pancreatic cancer clinical trial participants has remained unchanged (B and C).
Is the diversity of pancreatic cancer clinical trials representative of the pancreatic cancer incidence?
Despite a positive trajectory in the reporting of race and ethnicity data, non-White and Hispanic patients remain markedly underrepresented in pancreatic cancer clinical trials. White patients made up the majority (84.7%, EF=1.05, p=0.002) of pancreatic cancer clinical trial participants whereas Black (8.2%, EF=0.43, p=0.002), Asian/Pacific Islander (2.4%, EF=0, p=<0.0001), American Indian or Alaskan Native (0.3%, EF=0, p=<0.0001) and Hispanic (6%, EF=0.47, p=<0.0001) patients were disproportionately missing (Table 1). Trial participants were also more likely to be male (54.8%, EF=1.07, p=.0006) than female (45.2%, EF=0.93, p=.001). Indicated by a significantly low median EF, underrepresentation of non-White and Hispanic patients was not the results of a few trials, but rather systemic underrepresentation across pancreatic cancer clinical trials.
Table 1.
Diversity of pancreatic cancer clinical trial participants
| Participant demographic | N (%) | Proportion of Incident Cases (%)a | Median EFb | P valuec |
|---|---|---|---|---|
| Race (n=102 trials with race data, n=4374 participants) | ||||
| White | 3704 (84.7) | 82.3 | 1.05 | .002 |
| Black | 360 (8.2) | 12.4 | 0.43 | <.0001 |
| Asian/Pacific Islander | 105 (2.4) | 3.3 | 0 | <.0001 |
| American Indian/ Alaska native | 11 (0.3) | 0.4 | 0 | <.0001 |
| Multiraciald | 32 (0.7) | NA | NA | NA |
| Unknownd | 162 (3.7) | NA | NA | NA |
| Ethnicity (n=73 trials with ethnicity data, n=2981 participants) | ||||
| Non-Hispanic | 2711 (90.9) | 91.5 | 1.02 | .271 |
| Hispanic | 180 (6.0) | 8.5 | 0.47 | <.0001 |
| Unknownd | 90 (3.0) | NA | NA | NA |
| Gender (n=204 trials with gender data, n=8278 participants) | ||||
| Female | 3744 (45.2) | 48.4 | 0.93 | .001 |
| Male | 4534 (54.8) | 51.4 | 1.07 | .0006 |
Abbreviations: EF, enrollment fraction
Incident cases 2013–2017, age-adjusted to the 2000 US Standard Population.
Median EF of each individual trial with demographic data reported.
Wilcoxon rank sum tests determined distribution of EF ratios were different from 1.
Incident cases were not reported in NAACCR for multiracial and unknown subgroups.
Additionally, non-White, Hispanic and female patients with pancreatic cancer were underrepresented across nearly all phases of clinical trials (supplemental table 2). In fact, when studies were limited to pancreatic cancer clinical trials in the US, no Black or American Indian/Alaskan Native patients were documented in phase 3 clinical trials and no Asian/Pacific Islander and American Indian/Alaskan Native patients were reported in phase 4 clinical trials. Based on enrollment fraction, non-White and Hispanic patients with pancreatic cancer were underrepresented in clinical trials sponsored by universities and cancer centers, pharmaceutical and biotech companies and the NCI (supplemental table 3). Female patients were also underrepresented by all sponsor types except the NCI.
Although progress was made in demographic reporting (Figure 1A), trends in participant racial and ethnic diversity remained relatively unchanged from 2008 to 2020 (Figure 1B, 1C). These data indicate that non-White, Hispanic and female patients with pancreatic cancer continue to be disproportionately represented across pancreatic cancer clinical trials relative to the disease burden. This underrepresentation spans clinical phases and sponsors, and there has been no meaningful improvement in participant diversification in the last decade.
Discussion
The lack of diversity found in pancreatic cancer clinical trials is a salient example of broader disparities in diversity and inclusion found across clinical trials. Enrollment disparities in cancer clinical trials was first noted by Murthy et al. as they evaluated racial and ethnic representation in breast, colorectal, lung and prostate cancer clinical trials from 2000–2002 within the NCI clinical trial cooperative group.10 They noted that racial and ethnic minorities, women and the elderly were less likely to enroll in cancer clinical trials.10 Subsequently, Duma et al. explored racial, ethnic and gender disparities in common cancer clinical trials.12 They found a decrease in recruitment of minority patients from 2003–2016 as well as low recruitment of Black, Hispanic and female patients in cancer clinical trials.12 Further supporting these findings in a study focused on prostate cancer clinical trials, Rencsok et al. found that the majority of participants were White men.13 Moreover, African and Caribbean countries with predominantly Black populations represented only 3% of prostate cancer clinical trials from 1987 to 2016.13 Underrepresentation of racial and ethnic minority populations has also been found in surgical oncology trials as well as clinical trials leading to US FDA approval of oncology drugs14,15.
Throughout the last three decades, federal initiatives have aimed to increase the accrual of racial/ethnic minority patients in clinical trials. In 1990, the Minority-Based Community Clinical Oncology Program was founded with the goal to increase the NCI’s clinical trial network to minority populations and decrease racial/ethnic disparities.16 Coupled with the NIH Revitalization Act of 1993 and FDA Modernization Act of 1997, initial progress was made to improve the heterogeneity of clinical trial participants. Unfortunately, despite this early progress, national accrual of minority patients in clinical trials has remained poor over the last two decades.17 Spurred by Section 907 of the FDA Safety and Innovation Act of 2012, the FDA has recently instituted multiple initiatives to improve the representation of racial and ethnic minorities in clinical trials. This includes the FDA’s Action Plan to enhance the collection and availability of demographic subgroup data.18 Though the implementation of these initiatives has improved data collection and availability, the evidence provided in this study indicates that we are not only falling short of achieving representation, but that participant diversity has not improved.
The challenges associated with improving clinical trial diversity are multifaceted and complex though they may be simplified by categorization based on patients, providers and systemic challenges. Historically, distrust has been quoted as a key challenge in the recruitment of minority patients in clinical trials. This distrust has been attributed to the historic atrocities and exploitation of minority populations; particularly the Tuskegee Syphilis Study in which Black men were unknowingly given syphilis and treatment was withheld.19 Despite this rightful distrust of the medical system, Black and Hispanic patients are in actuality equally as willing as White patients to participate in clinical trials.20 Factors that disproportionally impact minority populations such as poor health literacy, low socioeconomic status and access to care also largely affect willingness or ability to enroll in a clinical trial.21
Though much emphasis has been placed on overcoming patient factors, literature suggests that physician bias, false perceptions and prejudices may influence participation in clinical trials. In a study by van Ryn et al., physicians were more likely to believe that Black patients are less intelligent and less likely to adhere to medical treatment.22 As a result, physicians do not recommend clinical trials to minority patients because of concerns for compliance to trial protocols. Notably, racial/ethnic disparities are negated when patients are given equal opportunity or explicitly offered participation in a clinical trial.23 Moreover, only 2.3% of practicing oncologists are Black or African American and only 5.8% of practicing oncologists are Hispanic, likely largely impacting trial referral practices and patient trust.24
Systemic barriers in clinical trial design and infrastructure also may limit minority patient participation. These include limited number of trials and resources, poor community engagement, and strict trial eligibility criteria. Patients of racial and ethnic minority backgrounds are also more likely to receive care at institutions with insufficient resources.21 Additionally, strict eligibility criteria often excludes many minority patients interested in participating in clinical trials. In a study by Adams-Campbell et al., the majority of Black patients were rendered ineligible for clinical trials secondary to medical morbidities.25
In the face of these challenges, it is imperative to equitably include racial and ethnic minorities in clinical trials. Emphasis on improved communication, trust and education between both patients and providers is critical. If clinical trials do not reflect the patient population they serve, disparities in treatment and outcomes will be perpetuated. The inclusion of racial and ethnic minority populations in pancreatic cancer clinical trials will take concerted and intentional efforts toward diversification.
Limitations:
This study has several limitations that warrant further discussion. The exclusion of trials including other cancer types and pancreatic pathologies may underestimate the number of patients with pancreatic cancer who participated in disease-agnostic phase 1 and 2 clinical trials. Additionally, by including only studies within the US, this may limit the number of phase 3 and phase 4 clinical trials captured as well as global generalizability. These latter limitations allow for us to offer a more focused assessment relative to US minority patient participation in US-based pancreatic cancer clinical trials.
Within the NAACCR database, race is characterized as White, Black, Asian/Pacific Islander and American Indian/Alaska Native. The assumption that participants are siloed neatly into each demographic category may be an overgeneralization of the true diversity of the US population. Despite these limitations, these data suggest that though demographic reporting has improved over time, the diversity of clinical trial participants is disproportionate to the disease burden of pancreatic cancer.
Conclusions:
These findings emphasize the persistent trend of underrepresentation of racial and ethnic minority patients in pancreatic cancer clinical trials. Representation of minority patients in pancreatic cancer clinical trials is a critical link between oncologic innovation and patient outcomes. Though prior initiatives have experienced varying levels of success, we propose that future interventions will need to be implemented on multiple levels in order to effectively include minority populations in pancreatic cancer clinical trials. In order to improve the diversity of pancreatic cancer clinical trials, we propose the following potential next steps:
Promotion of inclusivity through the reevaluation of strict eligibility criteria, particularly those that disproportionally exclude patients from racial and ethnic minority backgrounds.
Clinical trial accountability from sponsor organizations to support the inclusion of a diverse patient population.
Improved diversity of both providers and coordinators conducting pancreatic cancer clinical trials.
Creation of pancreatic cancer clinical trial environments that are free of both explicit and implicit bias through dedicated workforce training.
Conduction of pancreatic cancer clinical trials in locations that are accessible to racially and ethnically diverse patient populations.
Continued efforts toward patient advocacy and public policy in order to promote future change in the diversity and inclusion of pancreatic cancer clinical trials.
Through the implementation of these recommendations, we may continue to work toward improving the diversity and inclusion of participants in pancreatic cancer clinical trials. Enrollment of a diverse patient population will allow for the translation of new discoveries into tangible benefits for all patients suffering from pancreatic cancer.
Supplementary Material
Grant support:
This work is supported by the National Human Genome Research Institute (T32 HG0008958 to KMH and ANR) and the National Cancer Institute (R01CA242003 to JGT) of the National Institutes of Health and the Joseph and Ann Matella Fund for Pancreatic Cancer Research (JGT).
Abbreviations:
- BRCA 1/2
Breast cancer gene
- CDKN2A
Cyclin Dependent Kinase Inhibitor 2A
- EF
Enrollment Fraction
- FDA
Food and Drug Administration
- HER2
Human epidermal growth factor receptor 2
- KRAS
Kirsten rat sarcoma
- N/A
Not applicable
- NAACCR
North American Association of Central Cancer Registries
- NCI
National Cancer Institute
- NIH
National Institutes of Health
- US
United States
Footnotes
Disclosures: Authors have no conflicts of interest.
Data Transparency Statement: Data, analytic methods, and study materials will be made available to other researchers upon request.
References:
- 1.Rahib L, Smith BD, Aizenberg R, et al. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014;74(11):2913–21. [DOI] [PubMed] [Google Scholar]
- 2.SEER*Explorer: An interactive website for SEER cancer statistics [Internet]. Surveillance Research Program, National Cancer Institute. [Cited 2021 Feb 1]. Available from https://seer.cancer.gov/explorer/. [Google Scholar]
- 3.Riner AN, Underwood PW, Yang K, et al. Disparities in Pancreatic Ductal Adenocarcinoma-The Significance of Hispanic Ethnicity, Subgroup Analysis, and Treatment Facility on Clinical Outcomes. Cancer Med. 2020;9(12):4069–4082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Arnold LD, Patel AV, Yan Y, et al. Are racial disparities in pancreatic cancer explained by smoking and overweight/obesity? Cancer Epidemiol Biomarkers Prev. 2009;18(9):2397–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vick AD, Hery DN, Markowiak SF, Brunicardi FC. Closing the Disparity in Pancreatic Cancer Outcomes: A Closer Look at Nonmodifiable Factors and Their Potential Use in Treatment. Pancreas. 2019;48(2):242–249. [DOI] [PubMed] [Google Scholar]
- 6.Pernick NL, Sarkar FH, Philip PA, et al. Clinicopathologic analysis of pancreatic adenocarcinoma in African Americans and Caucasians. Pancreas. 2003;26(1):28–32. [DOI] [PubMed] [Google Scholar]
- 7.Golan T, Kindler HL, Park JO, et al. Geographic and Ethnic Heterogeneity of Germline. J Clin Oncol. 2020;38(13):1442–1454. [DOI] [PubMed] [Google Scholar]
- 8.McWilliams RR, Wieben ED, Chaffee KG, et al. Germline Rare Coding Variants and Risk of Pancreatic Cancer in Minority Populations. Cancer Epidemiol Biomarkers Prev. 2018;27(11):1364–1370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.ClinicalTrials.gov Background - ClinicalTrials.gov. https://clinicaltrials.gov/ct2/about-site/background. Accessed March 10, 2021.
- 10.Murthy VH, Krumholz HM, Gross CP. Participation in cancer clinical trials: race-, sex-, and age-based disparities. JAMA. 2004;291(22):2720–6. [DOI] [PubMed] [Google Scholar]
- 11.Wilson JP, Goldberg DW, Swift JN (2009). Central Cancer Registry Geocoding Needs. USC GIS Research Laboratory Techincal Report No. 13.
- 12.Duma N, Vera Aguilera J, Paludo J, et al. Representation of Minorities and Women in Oncology Clinical Trials: Review of the Past 14 Years. J Oncol Pract. 2018;14(1):e1–e10. [DOI] [PubMed] [Google Scholar]
- 13.Rencsok EM, Bazzi LA, McKay RR, et al. Diversity of Enrollment in Prostate Cancer Clinical Trials: Current Status and Future Directions. Cancer Epidemiol Biomarkers Prev. 2020;29(7):1374–1380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Stewart JH, Bertoni AG, Staten JL, et al. Participation in surgical oncology clinical trials: gender, race/ethnicity, and age-based disparities. Ann Surg Oncol. 2007;14(12):3328–34. [DOI] [PubMed] [Google Scholar]
- 15.Loree JM, Anand S, Dasari A, et al. Disparity of Race Reporting and Representation in Clinical Trials Leading to Cancer Drug Approvals From 2008 to 2018. JAMA Oncol. 2019:e191870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.McCaskill-Stevens W, McKinney MM, Whitman CG, et al. Increasing minority participation in cancer clinical trials: the minority-based community clinical oncology program experience. J Clin Oncol. 2005;23(22):5247–54. [DOI] [PubMed] [Google Scholar]
- 17.Chen MS, Lara PN, Dang JH, et al. Twenty years post-NIH Revitalization Act: enhancing minority participation in clinical trials (EMPaCT): laying the groundwork for improving minority clinical trial accrual: renewing the case for enhancing minority participation in cancer clinical trials. Cancer. 2014;120 Suppl 7:1091–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Enhancing the Diversity of Clinical Trial Populations- Eligibility Criteria, Enrollment Practices, and Trial Designs for Industry. Guidance for Industry (fda.gov) Accessed March 15, 2021.
- 19.Hamel LM, Penner LA, Albrecht TL, et al. Barriers to Clinical Trial Enrollment in Racial and Ethnic Minority Patients With Cancer. Cancer Control. 2016;23(4):327–337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wendler D, Kington R, Madans J, et al. Are racial and ethnic minorities less willing to participate in health research? PLoS Med. 2006;3(2):e19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Clark LT, Watkins L, Piña IL, et al. Increasing Diversity in Clinical Trials: Overcoming Critical Barriers. Curr Probl Cardiol. 2019;44(5):148–172. [DOI] [PubMed] [Google Scholar]
- 22.van Ryn M, Burke J. The effect of patient race and socio-economic status on physicians’ perceptions of patients. Soc Sci Med. 2000;50(6):813–28. [DOI] [PubMed] [Google Scholar]
- 23.Kehl KL, Arora NK, Schrag D, et al. Discussions about clinical trials among patients with newly diagnosed lung and colorectal cancer. J Natl Cancer Inst. 2014;106(10). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Facts & Figures: Diversity in Oncology. https://www.asco.org/practice-policy/cancer-care-initiatives/diversity-oncology-initiative/facts-figures-diversity. Accessed March 21, 2021.
- 25.Adams-Campbell LL, Ahaghotu C, Gaskins M, et al. Enrollment of African Americans onto clinical treatment trials: study design barriers. J Clin Oncol. 2004;22(4):730–4. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


