Abstract
The impact of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection during pregnancy on the developing fetal brain is poorly understood. Other antenatal infections such as influenza have been associated with adverse neurodevelopmental outcomes in offspring. Although vertical transmission has been rarely observed in SARS-CoV-2 to date, given the potential for profound maternal immune activation (MIA), impact on the developing fetal brain is likely. Here we review evidence that SARS-CoV-2 and other viral infections during pregnancy can result in maternal, placental, and fetal immune activation, and ultimately in offspring neurodevelopmental morbidity. Finally, we highlight the need for cellular models of fetal brain development to better understand potential short- and long-term impacts of maternal SARS-CoV-2 infection on the next generation.
Keywords: COVID-19, SARS-CoV-2, pregnancy, fetal brain, placenta, neurodevelopment
Potential transgenerational impact of viral infections in pregnancy
Some viral infections acquired antenatally can have lasting, potentially devastating, impacts on the developing fetal brain. Those viruses that demonstrate neuroinvasion and neurotropism – that is, the ability to invade the blood–brain barrier and infect neurologic tissues, such as Zika virus or cytomegalovirus – can directly damage the developing fetal brain if transplacental transmission occurs [1,2]. Other infections that do not cross the placental barrier, including viral infections such as influenza, have also been consistently associated with adverse neurodevelopmental outcomes in offspring, primarily via mechanisms related to maternal, placental, and subsequent fetal brain immune activation [3., 4., 5., 6., 7., 8.]. Adverse neurodevelopmental outcomes observed following prenatal infection range from autism spectrum disorder (ASD), attention deficit hyperactivity disorder, and cognitive dysfunction, to anxiety, depression, and schizophrenia [3,4,7., 8., 9., 10., 11., 12., 13.].
Past pandemics have provided a window into potential neurodevelopmental consequences in subsequent generations. For example, individuals who were fetuses during the 1957 influenza pandemic had an increased risk for being hospitalized for schizophrenia as adults [12]. The rubella pandemic of 1964 was associated with a 10–15-fold increase in ASD and schizophrenia in offspring [14]. In a cohort of more than 115 000 pregnancies, fevers in pregnancy were associated with an increased odds ratio of up to 3.1 for ASD [11].
The potential for neurodevelopmental morbidity in offspring exposed prenatally to SARS-CoV-2 is therefore of great concern. Given the large number of exposed individuals, even a modest increase in risk for adverse offspring neurodevelopment would still have a massive public health impact [15., 16., 17., 18., 19., 20.]. More than 59 million people in the United States and 300 million worldwide have been diagnosed with coronavirus disease 2019 (COVID-19)i, including over 155 500 pregnant women in the United States [21]. With approximately 140 million live births occurring annually worldwideii, and the prevalence of SARS-CoV-2 positivity in pregnant women as high as 15% in urban centers [22], the numbers of children exposed to maternal COVID-19 infection in utero worldwide could reach up to 20 million per year, particularly in the setting of low COVID-19 vaccine uptake in pregnant populations [23,24]. In fact, a 12-month follow-up of more than 7000 deliveries in a large hospital system, including more than 200 COVID-19-exposed pregnancies, suggested that prenatal SARS-CoV-2 infection is associated with an increased risk for offspring neurodevelopmental diagnoses [25].
There are multiple potential pathways by which a maternal infection with SARS-CoV-2 could impact the developing fetal brain [13,26., 27., 28.]: (i) via MIA (see Glossary) during key neurodevelopmental windows in pregnancy; (ii) via direct fetal infection of neurologic tissues via transplacental transmission of virus; or (iii) via compromised placental function resulting in adverse pregnancy outcomes associated with an increased risk of neurologic injury (e.g., fetal growth restriction, preterm birth, abruption). In this review, we present emerging evidence regarding the impact of SARS-CoV-2 infection during pregnancy on offspring neurodevelopmental outcomes, explore potential mechanisms by which prenatal SARS-CoV-2 exposure might impact the developing fetal brain, and discuss virus and host factors that might influence risk. Lastly, we discuss the need for cellular models to best study the impact of SARS-CoV-2 on the developing brain, and to identify individuals most at risk for adverse outcomes who may benefit from early intervention or therapeutics.
Early evidence of adverse neurodevelopmental outcomes in children with prenatal exposure to SARS-CoV-2
Mounting evidence suggests that SARS-CoV-2 infection can cause both acute and chronic neurologic and psychiatric sequelae in adult and pediatric populations [29., 30., 31., 32., 33., 34.]. A recent study from the UK suggests that 3.8% of children hospitalized with COVID-19 experience a neurologic complication that also has long-term consequences, ranging from behavioral change, hallucinations, and encephalopathy, to status epilepticus, encephalitis, Guillain–Barré/acute demyelinating syndromes, chorea, and psychosis [35]. A persistent problem in interpreting these data is the paucity of studies with well-matched controls – for example, to understand whether these sequelae reflect direct effects of SARS-CoV-2, or simply the consequences of any severe illness requiring hospitalization. One early report using neurocognitive assessments suggested that the pattern of deficits in adults was nonspecific, consistent with other complicated hospitalizations [36]. Still, in aggregate, these results suggest at least the possibility that SARS-CoV-2 – whether via inflammation or immune-mediated mechanisms or via direct infection of the central nervous system – can have a lasting impact on the developed (and therefore likely also on the developing) brain.
Early signals of adverse neurodevelopmental outcomes at 3–6 months, 1 year, and 1.5 years in infants and children exposed to SARS-CoV-2 in utero, and/or born during the COVID-19 pandemic are emerging [37., 38., 39., 40., 41.]. A longitudinal cohort study of 57 infants with prenatal exposure to SARS-CoV-2 in China identified deficits in the social–emotional domain of neurodevelopmental testing at 3 months of age [40]. A preliminary report of 298 infants born to women with SARS-CoV-2 infection during pregnancy found evidence of developmental delay in 10% of infants at 12 months of age, although neither study included a noninfected comparator group [41]. Another preliminary report of over 7000 infants born during the COVID-19 pandemic to both SARS-CoV-2 infected and noninfected mothers identified an association between maternal SARS-CoV-2 exposure and a neurodevelopmental diagnosis at 12 months [25]. Importantly, that study found that while the association between maternal SARS-CoV-2 exposure and offspring neurodevelopmental morbidity was enhanced by preterm delivery, the presence of a neurodevelopmental diagnosis was not entirely explained by prematurity, suggesting a more specific mechanism of effect than simply SARS-CoV-2 contributing to pregnancy complications. Whether a definitive connection exists between prenatal SARS-CoV-2 exposure and neurodevelopmental disorders in offspring is not yet known, in part because most children born to women infected in the first wave of the pandemic are still too young for reliable diagnosis of many neurodevelopmental conditions. Despite the significant limitations of epidemiologic and clinical data sets in defining causality or mechanism, these preliminary data demonstrate the potential for prenatal SARS-CoV-2 exposure to impact early neurodevelopmental outcomes.
MIA and placental immune activation in response to SARS-CoV-2 infection in pregnancy
MIA is a primary link between maternal viral infection and offspring neurodevelopmental disorders
Convergent data from animal models have demonstrated that MIA, rather than a specific virus or bacterial infection, is likely the final common pathway by which most maternal infections confer offspring neurodevelopmental morbidity [42,43]. Rodent and non-human primate models have utilized multiple techniques to activate the maternal immune system, including the viral mimic polyinosinic:polycytidylic (poly[I:C]), a synthetic double-stranded RNA, demonstrating alterations in offspring brain immune function, and behavioral and neuroimaging correlates of ASD and schizophrenia [42,44., 45., 46., 47., 48., 49., 50., 51., 52., 53.]. Rodent models of maternal influenza infection resulted in offspring behavioral phenotypes consistent with ASD and psychosis [54], but many of the same behavioral abnormalities were present when uninfected rodent mothers were injected with poly[I:C] [54], suggesting that MIA rather than any specific pathogen mediates the neurodevelopmental morbidity in offspring [53,55]. Immune cell populations, effector cytokines, and inflammatory markers critical in mediating abnormal offspring behavioral phenotypes in models of MIA include T helper 17 cells/interleukin (IL)-17α, IL-6, IL-1Rα, tumor necrosis factor-alpha (TNF-α), IL-10, C-reactive protein, and the complement system, among others [10,56., 57., 58., 59., 60., 61., 62., 63., 64., 65., 66., 67., 68.]. Many of these same cytokines and inflammatory markers are also dysregulated in COVID-19 infection in the non-pregnant population [69., 70., 71., 72., 73., 74., 75., 76.].
Immune activation at the maternal–fetal interface and in fetal cord blood has been observed in prenatal SARS-CoV-2 infection
Although the immune and inflammatory response to SARS-CoV-2 in pregnancy is only beginning to be characterized, early data show a proinflammatory cytokine phenotype in pregnant women with active SARS-CoV-2, with interferon-gamma (IFN-γ), IL-1β, and IL-6 most implicated, particularly in cases of severe COVID-19 disease [77,78]. Several studies have demonstrated the potential for maternal SARS-CoV-2 infection to stimulate an intense placental immune and inflammatory response [79., 80., 81.], both in the presence (rare) [81] and absence (more common) [80,82,83] of direct SARS-CoV-2 infection of the placenta. Substantial infiltration of maternal immune cells into the placenta has been observed in cases of severe maternal COVID-19 disease, adverse neonatal outcomes, and heavy placental SARS-CoV-2 viral burden [81]. In addition, maternal SARS-CoV-2 infection is associated with hyperplasia and/or increased density of fetal placental macrophages or Hofbauer cells, typically in the absence of Hofbauer cell infection [80,84]. In placental samples without evidence of direct SARS-CoV-2 infection, transcriptomic analysis has demonstrated upregulation of inflammatory pathways of maternal decidual natural killer and T cells and upregulation of interferon-stimulated genes (ISGs) in placental villous tissue [79,80]. Notable sex differences have been observed in the placental immune response to maternal SARS-CoV-2 [80], which suggests that sex plays an important role in fetal and offspring vulnerability to maternal SARS-CoV-2 infection.
Increased cord blood cytokine levels and altered cord blood immune cell profiles including increased proportion of natural killer cells, Vδ2+ γδ T cells, and regulatory T cells have been observed in maternal SARS-CoV-2 infection in the absence of fetal infection [85,86], pointing to the potential for MIA itself to generate a proinflammatory fetal response. Single-cell RNA sequencing of cord blood mononuclear cells demonstrates a significant impact of maternal SARS-CoV-2 on cord blood monocyte programs, with ISGs upregulated in CD14+ and CD16+ monocytes [87]. Importantly, observed fetal immune imprinting to date has been nonspecific to SARS-CoV-2, occurring in the absence of vertical transmission [86,87].
How can maternal and placental immune activation and inflammation impact the developing fetal brain?
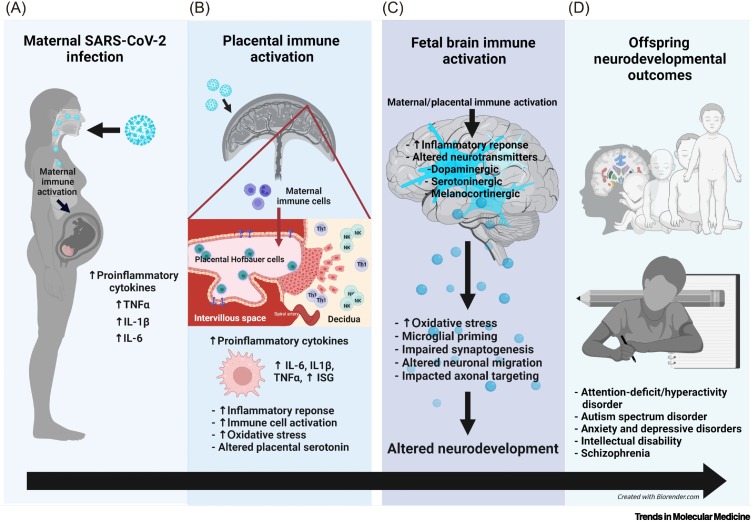
Several candidate mechanisms have been proposed in models of MIA which may apply to maternal SARS-CoV-2 infection as well (Figure 1 ).
-
(i)
MIA is associated with rapid induction of immune (e.g., type I–III IFN pathways) and proinflammatory responses (e.g., TNF-α, IL-6, IL-1β) in the placenta and the developing fetal brain, particularly in the setting of activation via Toll-like receptor (TLR) 7/8 signaling pathways, and to a lesser extent for immune activation via TLR3/TLR4 signaling pathways [42,88., 89., 90., 91.].
-
(ii)
Dysregulated placental serotonin signaling, as the placenta is the primary source of serotonin for the developing fetal brain [92,93]. Both maternal and placental immune activation and inflammation alter placental serotonin signaling, which in turn influences fetal brain development via impaired synaptogenesis, neuronal migration, and axonal targeting [93., 94., 95., 96., 97.].
-
(iii)
Maternal and placental immune activation are also associated with other alterations in fetal brain neurotransmitter signaling, including the dopaminergic, cholinergic, GABAergic, glutamatergic, and melanocortinergic systems (the latter mediated primarily by dysregulated leptin signaling) that influence fetal brain development and future risk for conditions such as schizophrenia, addiction, and disordered eating [49,98., 99., 100., 101., 102., 103.].
-
(iv)
Maternal and placental immune activation are associated with placental and fetal brain mitochondrial dysfunction, oxidative stress, and disrupted protein homeostasis [88,91,104,105]. The maternal and offspring gut microbiome may also be important modifiers of the impact of MIA on the developing brain and offspring outcomes [56,105,106].
Figure 1.
Coronavirus disease 2019 (COVID-19) in pregnancy and implications for offspring neurodevelopment.
(A) Maternal severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection results in maternal immune activation (MIA) and increased proinflammatory cytokines in the maternal periphery. (B) Maternal SARS-CoV-2 infection can impact the placenta via two mechanisms: (i) placental immune activation and inflammation resulting from MIA (likely most common) or (ii) direct placental infection with SARS-CoV-2 (rare per the current literature). Placental immune activation and inflammation are associated with placental Hofbauer cell activation or priming, increased natural killer (NK) and T-helper 1 (Th1) cells at the maternal–fetal interface, increased proinflammatory cytokine production, upregulation of interferon-stimulated genes (ISGs), placental serotonin dysregulation, and increased oxidative stress. When direct placental infection with SARS-CoV-2 occurs (which is rare with the ancestral and other pre-Delta strains), the syncytiotrophoblast and cytotrophoblast layers are most commonly infected, and once the virus gains access to the intervillous space, it can theoretically gain access to the fetal circulation. (C) Both MIA and placental immune activation can lead to fetal brain immune activation, inflammation, and altered neurotransmitter signaling, including the serotonergic, dopaminergic, melanocortinergic, GABAergic, and glutamatergic systems. Fetal brain immune responses are associated with microglial priming, altered neural progenitor cell proliferation, impaired neuronal migration, synaptogenesis, and axonal targeting, all of which can result in altered offspring neurodevelopment. (D) Offspring affected by MIA are at increased risk for attention hyperactivity deficit disorder, autism spectrum disorder, anxiety, depression, impaired cognition, learning disabilities, and schizophrenia.
MIA models have pointed to aberrant programming of fetal microglia and dysregulation of cytokine networks as key mechanisms underlying abnormal fetal brain development, with microglia primed toward a proinflammatory phenotype and altered synaptic pruning implicated in offspring morbidity [59,107., 108., 109., 110.]. Given the extent of synapse formation and pruning that occurs in fetal and neonatal life [111., 112., 113., 114.], developmental microglial function represents a critical target for investigation to better understand the impact of SARS-CoV-2-driven immune activation on the developing fetal brain. MIA has also been associated with abnormalities of offspring neural progenitor cell proliferation; impaired neuronal migration; alterations in neuronal density and perineuronal nets; altered dendrite structure, synaptogenesis, and synaptic function and plasticity; and changes in interhemispheric and corticolimbic connectivity [49,66,105,115]. Thus, in utero exposure to SARS-CoV-2-related MIA may be associated with altered development of multiple brain cell types and functions, with the timing of the insult, presence or absence of other exposures, and intrinsic fetal characteristics such as fetal sex or genetic susceptibility dictating fetal resilience or vulnerability.
Transplacental transmission of SARS-CoV-2 and direct fetal infection: uncommon in observations to date
Transplacental transmission of virus capable of infecting neural tissue can have lasting and devastating consequences on the developing fetal brain. A key factor in understanding potential fetal infection risk is whether maternally acquired SARS-CoV-2 can transmit across the placenta, the primary physiological and immunological barrier preventing viral transmission from the maternal to fetal circulation [116., 117., 118.]. The preponderance of evidence to date suggests that adverse neurodevelopmental effects of SARS-CoV-2 infection, at least with the ancestral strain and strains preceding the B.1.617.2 (Delta) variant, are more likely to occur via maternal and placental immune activation and downstream impact on the developing fetal brain, rather than via direct fetal infection with SARS-CoV-2 in utero [28,119]. Population-level data suggest that rates of SARS-CoV-2 positivity among newborns in SARS-CoV-2-exposed pregnancies range from 1% to 3% [83,120., 121., 122., 123., 124.], with placental infection being a relatively rare event. One meta-analysis of case reports and case series estimated the rate of placental infection as 7% [125], although prospective studies have identified even lower rates of placental infection [83,84,126,127]. Protective mechanisms against placental infection include low rates of maternal SARS-CoV-2 viremia, preserved immune defenses at the syncytiotrophoblast border, and the lack of coordinated expression of molecules required for SARS-CoV-2 attachment and entry (ACE2 and TMPRSS2) into the syncytiotrophoblast [80,83,128]. Evidence supporting fetal infection – that is, documented viral particles in the sterile amniotic space or fetal tissue – has been limited to case reports [129].
Consistent with the observed low rate of placental infection and vertical transmission, data are now available from completed pregnancies exposed to SARS-CoV-2 during all developmental windows (first through third trimesters). To date, no characteristic congenital syndrome has emerged after prenatal SARS-CoV-2 exposure that would suggest direct fetal infection [122,123,130,131]. Prenatal neuroimaging studies in pregnant cohorts have similarly failed to identify evidence of grossly visible fetal intracranial pathology associated with maternal SARS-CoV-2 infection, though the small number of women included with severe or critical illness versus mild disease may limit generalizability of these findings [132,133]. All these data point to maternal and placental immune activation and consequent fetal neuroimmune activation as primary drivers of neurodevelopmental morbidity in SARS-CoV-2-exposed offspring, rather than direct placental and fetal brain infection as has been observed with Zika virus or maternal cytomegalovirus infection [1,2].
Potential vulnerability of the fetal brain to SARS-CoV-2 infection
Despite the aforementioned observational data against direct fetal infection as a major mechanism of risk to the developing fetal brain, animal and cellular models have demonstrated the potential for SARS-CoV-2 to directly infect fetal brain tissue, should the placental barrier be broached. A comprehensive analysis of fetal brain sequencing data from publicly available data sets identified low expression of canonical spike (S) protein interactors ACE2 and TMPRSS2, but high expression of novel S protein interactors throughout gestation, with highest expression occurring in the second and third trimesters, suggestive of increased vulnerability during this window of gestation [134]. Limited evidence from adult human and animal models suggest that the SARS-CoV-2 virus can bypass or disrupt the blood–brain barrier and gain access to neurological tissue [135., 136., 137.]. Models using cerebral organoids with proteomic signatures similar to fetal brain tissue [138] have provided evidence of SARS-CoV-2 capability of neuroinvasion and neurotropism for the choroid plexus, cortical neurons, and glial cells [137,139., 140., 141.]. Taken together, these data raise the possibility that neurodevelopment at the cellular level could be directly impacted by prenatal infection with SARS-CoV-2, if viral particles gain access to the fetal circulation.
SARS-CoV-2 variants of concern and potential fetal risk
A key caveat in interpreting the available data is that observations primarily reflect the effects of ancestral viral strains, while less is known about placental and fetal effects of recent variants of concern, in particular the Delta and Omicron variants. It is plausible that mutations associated with enhanced immune escape or transmissibility could impact the risk for transplacental infection. Unfortunately, detecting anything other than a large or abrupt increase in vertical transmission will require the accumulation of data over time.
Prenatal SARS-CoV-2 variant exposure and placental impact
Recent evidence showing a near twofold increased risk of stillbirth associated with maternal SARS-CoV-2 infection, with a greater magnitude of association during the Delta-predominant period of the COVID-19 pandemic [142], has raised concerns that the SARS-CoV-2 virus itself might be directly responsible for adverse pregnancy outcomes [129]. Of critical importance is whether Delta-variant SARS-CoV-2 infection may differentially affect either (i) placental function or (ii) the integrity of the placental barrier to infection and thus the susceptibility for vertical transmission and/or more direct effects on the developing fetal brain.
As previously noted, the emergence of new strains complicates generalizations about SARS-CoV-2 mechanisms of risk. Delta variant infections have been linked to higher viral loads [143] and an increased risk of hospitalizations [144] in non-pregnant individuals compared with prior variants, as well as increased severity of maternal disease in pregnancy [145., 146., 147.]. Delta spike P681R mutation may also affect virulence and tissue tropism by enhanced S protein cleavability by furin [148,149], a transmembrane serine protease that is widely expressed by both the placental syncytiotrophoblast [150,151] and fetal brain tissue [134]. Although a characteristic histopathological signature associated with maternal SARS-CoV-2 infection was not clearly identified with the ancestral strain [83,84,152,153], SARS-CoV-2 placentitis – the triad of histiocytic intervillositis, perivillous fibrin, and villous trophoblastic necrosis in the setting of SARS-CoV-2 infection – has emerged as a histopathologic entity observed in association with both Alpha- and Delta-variant maternal SARS-CoV-2 infections, and has been linked to poor pregnancy outcomes including stillbirth in case reports [154,168]. Although definitive evidence linking Delta-variant SARS-CoV-2 placentitis to increased risk for fetal infection is lacking to date, these observations suggest the biological possibility that prenatal Delta-variant SARS-CoV-2 could lead to global placental dysfunction, and breach of the placental immune barrier.
SARS-CoV-2-associated preterm birth and neurodevelopmental risk
Prenatal SARS-CoV-2 exposure may also impact offspring neurodevelopment by contributing directly to adverse pregnancy outcomes. Indeed, SARS-CoV-2 infection acquired during pregnancy is associated with an increased risk of preeclampsia and preterm birth [25,122,155., 156., 157., 158.], outcomes which have independently been associated with offspring neurodevelopmental risk [159,160]. The association between SARS-CoV-2 and preterm birth complicates studies of neurodevelopment, because it impacts the ability to understand specificity of virus-associated risk. If the mechanism of action of SARS-CoV-2 is simply to cause maternal illness that, in more severe cases, contributes to preterm delivery, then full-term offspring should not experience elevated rates of neurodevelopmental sequelae, and efforts to investigate specific viral effects are unlikely to be necessary. By contrast, if this is not the case, it is critically important to understand the additive, or multiplicative, effect of the virus itself when preterm delivery occurs in the setting of or following maternal SARS-CoV-2 infection.
To date, only one study has addressed this question [25] and found that maternal infection was still associated with a greater risk for 12-month neurodevelopmental adverse outcomes after adjustment for preterm delivery. When analysis was limited to full-term deliveries, the magnitude of risk was diminished, and the 95% confidence interval included no effect, but risk in numeric terms remained substantial. While preliminary, this highlights the importance of well-controlled follow-up studies that evaluate the impact of gestational age at delivery.
The role of cellular models in understanding mechanisms of risk to the fetal brain
To date, efforts to understand MIA have largely focused on either large-scale human epidemiologic studies or human biological materials, which may indicate risk but are vulnerable to confounding, and animal models of disease, which allow more complete experimental control but may not recapitulate key elements of brain development. Cellular models can bridge these two investigative forms, particularly models that make use of primary human cells that may retain epigenomic effects of environment and infection. For example, in a prior work, patient-derived microglia-like cells differentiated from peripheral blood monocyte precursors demonstrated abnormal phagocytosis of synaptic materials, providing a potential mechanism of disease for schizophrenia or related disorders [161]. More recently, these protocols have been adapted to generate microglia-like cells from umbilical cord blood [162], providing a more direct means of characterizing the impact of in utero exposures for a specific individual. Another emerging strategy utilizes Hofbauer cells, fetal placental macrophages which may be isolated from the placenta after delivery and cultured [116]. An advantage of these cells is that, like microglia, they are yolk sac-derived, and thus may closely mimic the exposures and developmental processes of brain microglia [163].
Multiple published protocols also enable the generation of microglia-like cells from iPSCs. A limitation of this approach, compared with the prior two, is that generation of iPSCs entails disruption of the epigenome, prohibiting epigenomic investigations [164]. Alternatively, an advantage is that iPSCs can be expanded, banked, and used for large-scale experiments or screens. Regardless of source, such models provide an opportunity to conduct mechanistic studies in human cells to understand SARS-CoV-2-mediated effects.
Concluding remarks
The data presented here demonstrate the potential for maternal SARS-CoV-2 infection to drive maternal, placental, and fetal immune activation, and thus the potential for adverse fetal neurodevelopmental programming. Future studies will need to evaluate whether the fetoplacental immune responses observed in maternal SARS-CoV-2 infection are associated with longer-term neurodevelopmental and neuropsychiatric morbidity in offspring. While the majority of available data are for third trimester infections or active infection at the time of delivery, the implications of infection earlier in gestation on offspring outcomes will be critical to a holistic understanding of risk. An important observation is that immune activation need not impact only fully differentiated cells – indeed, by impacting yolk sac-derived precursor cells, an early (e.g., first trimester) insult may still impact brain development via the microglial progenitor pool [165., 166., 167.]. Thus, it is critical to understand not only direct/immediate effects on cells, but also persistent effects, as a putative mediator of neurodevelopmental consequences. How the timing of infection, variant strain, fetal sex, other prenatal exposures (e.g., maternal cardiometabolic conditions, substance use, stress, environmental or medication exposures, other infections in pregnancy), and perinatal/postnatal exposures (e.g., subsequent infant or child infection with SARS-CoV-2, breastfeeding status) intersect to impact offspring neurodevelopment will be important to a comprehensive understanding of the potential lasting impact of the COVID-19 pandemic on the next generation (Clinician’s corner and see Outstanding questions).
Clinician’s corner.
Although placental infection and vertical transmission after maternal SARS-CoV-2 infection are rare entities to date, early studies suggest that prenatal exposure to SARS-CoV-2 infection is associated with adverse neurodevelopmental outcomes in children from 3 months to 1.5 years of age.
Evaluating neurodevelopmental outcomes in children exposed to maternal SARS-CoV-2 infection prenatally at 2 years of age and beyond will be critical to gaining a full understanding of offspring neurodevelopmental risk.
The impact of additive or synergistic maternal risk factors such as maternal obesity, diabetes or hypertension, maternal substance use disorder, or other maternal bacterial or viral infections in pregnancy on offspring neurodevelopmental outcomes will be an important area for future study.
The impacts of virus and host factors such as variant strain, maternal disease severity, and fetal sex on offspring neurodevelopmental risk will be important to parse in future studies.
Alt-text: Clinician’s corner
Outstanding questions.
What will neurodevelopmental outcomes be in children exposed to prenatal SARS-CoV-2 infection at age 2 years and beyond?
Does trimester of maternal infection impact offspring neurodevelopmental outcomes?
Does prenatal SARS-CoV-2 infection that results in placental infection and/or vertical transmission have a differential (e.g., more severe) impact on offspring neurodevelopmental outcomes? Large, population-level studies will be needed to answer this question given the relative rarity of placental infection and vertical transmission.
How do variant strain and severity of maternal illness impact offspring neurodevelopmental risk?
What is the impact of fetal sex on offspring neurodevelopmental risk?
How do other pre- and perinatal exposures, and the postnatal environment, interact with prenatal SARS-CoV-2 infection to impact offspring neurodevelopmental risk?
How can cellular models be leveraged to understand neurodevelopmental risk at the individual level and guide precision interventions and/or therapeutics?
Alt-text: Outstanding questions
Acknowledgments
Acknowledgments
Support for this work was provided by K12HD103096 (to L.L.S.), R01HD100022-S2 (to A.G.E.), Simons Foundation SFARI Maternal COVID-19 Award (to A.G.E.), R01MH117177 (to E.L.S), R01MH124824 (to E.L.S.), March of Dimes and Silver Family Foundation grants (to J.O.L.), R01MH120227 (to R.H.P.), and Blyth Family Fund (to R.H.P.).
Declaration of interests
The authors report no conflicts of interest.
Glossary
- Histiocytic intervillositis
a placental lesion characterized by infiltration of maternal macrophages into the intervillous space; can be associated with adverse pregnancy outcomes.
- Hofbauer cells
specialized population of fetal macrophages that reside in the chorionic villous and are involved in immune regulation and transfer of ions, proteins, and nutrients across the maternal–fetal barrier.
- Interferon-stimulated genes
genes that can be expressed in response to stimulation by IFN, a primary mediator of the innate immune response to viral pathogens.
- iPSC
somatic cells that have been reprogrammed in culture into an embryonic-like, pluripotent state.
- Maternal immune activation (MIA)
elevated inflammatory markers above normal range during pregnancy, in the presence or absence of a pathogen.
- Microglia
specialized population of resident macrophages in the central nervous system that can direct inflammatory responses and support synaptic organization, neuronal development, brain protection, and repair.
- Peripheral blood monocytes
bone marrow-derived leukocytes with a single round nucleus isolated from peripheral blood, characterized by the ability to phagocytose, produce cytokines, and present antigen.
- Perivillous fibrin
a placental lesion in which fibrin is noted to be surrounding the chorionic villous.
- Polyinosinic:polycytidylic acid (poly[I:C])
an immunostimulant used to simulate viral infections; a synthetic analog of double-stranded RNA that resembles the molecular pattern of certain viruses.
- SARS-CoV-2 placentitis
the triad of histiocytic intervillositis, perivillous fibrin deposition, and villous trophoblastic necrosis in the setting of maternal SARS-CoV-2 infection.
- Spike (S) proteina
large, transmembrane glycoprotein located on the surface of SARS-CoV-2 that binds to host cell surface receptors to facilitate viral entry.
- Syncytiotrophoblast
specialized epithelial cells that line the placental chorionic villous, forming a physical barrier between the maternal and fetal circulations.
- Villous trophoblastic necrosis
necrotic trophoblasts identified in the chorionic villous on placental histopathology.
Resources
ihttps://coronavirus.jhu.edu/map.htmliihttps://ourworldindata.org/grapher/births-and-deaths-projected-to-2100References
- 1.Mlakar J., et al. Zika virus associated with microcephaly. N. Engl. J. Med. 2016;374:951–958. doi: 10.1056/NEJMoa1600651. [DOI] [PubMed] [Google Scholar]
- 2.Cheeran M.C.J., et al. Neuropathogenesis of congenital cytomegalovirus infection: disease mechanisms and prospects for intervention. Clin. Microbiol. Rev. 2009;22:99–126. doi: 10.1128/CMR.00023-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Al-Haddad B.J.S., et al. Long-term risk of neuropsychiatric disease after exposure to infection in utero. JAMA Psychiatry. 2019;76:594–602. doi: 10.1001/jamapsychiatry.2019.0029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cordeiro C.N., et al. Infections and brain development. Obstet. Gynecol. Surv. 2015;70:644–655. doi: 10.1097/OGX.0000000000000236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yockey L.J., et al. Contributions of maternal and fetal antiviral immunity in congenital disease. Science. 2020;368:608–612. doi: 10.1126/science.aaz1960. [DOI] [PubMed] [Google Scholar]
- 6.Zerbo O., et al. Maternal infection during pregnancy and autism spectrum disorders. J. Autism Dev. Disord. 2015;45:4015–4025. doi: 10.1007/s10803-013-2016-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Adams Waldorf K.M., McAdams R.M. Influence of infection during pregnancy on fetal development. Reproduction. 2013;146:R151–R162. doi: 10.1530/REP-13-0232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Al-Haddad B.J.S., et al. The fetal origins of mental illness. Am. J. Obstet. Gynecol. 2019;221:549–562. doi: 10.1016/j.ajog.2019.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kepinska A.P., et al. Schizophrenia and influenza at the centenary of the 1918–1919 Spanish influenza pandemic: mechanisms of psychosis risk. Front. Psychiatry. 2020;11:72. doi: 10.3389/fpsyt.2020.00072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jiang H.-Y., et al. Maternal infection during pregnancy and risk of autism spectrum disorders: a systematic review and meta-analysis. Brain Behav. Immun. 2016;58:165–172. doi: 10.1016/j.bbi.2016.06.005. [DOI] [PubMed] [Google Scholar]
- 11.Lee B.K., et al. Maternal hospitalization with infection during pregnancy and risk of autism spectrum disorders. Brain Behav. Immun. 2015;44:100–105. doi: 10.1016/j.bbi.2014.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mednick S.A. Adult schizophrenia following prenatal exposure to an influenza epidemic. Arch. Gen. Psychiatry. 1988;45:189. doi: 10.1001/archpsyc.1988.01800260109013. [DOI] [PubMed] [Google Scholar]
- 13.Brown A.S., Meyer U. Maternal immune activation and neuropsychiatric illness: a translational research perspective. Am. J. Psychiatry. 2018;175:1073–1083. doi: 10.1176/appi.ajp.2018.17121311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Patterson P.H. Immune involvement in schizophrenia and autism: etiology, pathology and animal models. Behav. Brain Res. 2009;204:313–321. doi: 10.1016/j.bbr.2008.12.016. [DOI] [PubMed] [Google Scholar]
- 15.Volkow N.D., et al. The healthy brain and child development study-shedding light on opioid exposure, COVID-19, and health disparities. JAMA Psychiatry. 2021;78:471–472. doi: 10.1001/jamapsychiatry.2020.3803. [DOI] [PubMed] [Google Scholar]
- 16.Lins B. Maternal immune activation as a risk factor for psychiatric illness in the context of the SARS-CoV-2 pandemic. Brain Behav. Immun. Health. 2021;16 doi: 10.1016/j.bbih.2021.100297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lopez-Diaz A., et al. COVID-19 infection during pregnancy and risk of neurodevelopmental disorders in offspring: time for collaborative research. Biol. Psychiatry. 2021;89:e29–e30. doi: 10.1016/j.biopsych.2020.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sakurada, et al. Neurodevelopmental disorders induced by maternal immune activation: toward a prevention strategy in the era of the COVID-19 pandemic. Psychiatry Int. 2020;1:24–26. [Google Scholar]
- 19.Figueiredo C.P., et al. SARS-CoV-2-associated cytokine storm during pregnancy as a possible risk factor for neuropsychiatric disorder development in post-pandemic infants. Neuropharmacology. 2021;201 doi: 10.1016/j.neuropharm.2021.108841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Okechukwu C. Inflammatory cytokines induced by severe acute respiratory syndrome coronavirus 2 infection during pregnancy may alter fetal brain development predisposing the offspring to neurodevelopmental disorders. Nigerian J. Exp. Clin. Biosci. 2021;9:58. [Google Scholar]
- 21.Centers for Disease Control and Prevention . Centers for Disease Control and Prevention; 2021. Data on COVID-19 During Pregnancy: Severity Of Maternal Illness. [Google Scholar]
- 22.Sutton D., et al. Universal screening for SARS-CoV-2 in women admitted for delivery. N. Engl. J. Med. 2020;382:2163–2164. doi: 10.1056/NEJMc2009316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shook L., et al. Countering COVID-19 vaccine hesitancy in pregnancy: the “4 Cs”. Am. J. Perinatol. 2021 doi: 10.1055/a-1673-5546. Published online October 19, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shook L., et al. COVID-19 vaccination in pregnancy and lactation: current research and gaps in understanding. Front. Cell. Infect. Microbiol. 2021;11:899. doi: 10.3389/fcimb.2021.735394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Edlow A.G., et al. Neurodevelopmental outcomes at one year in offspring of mothers who test positive for SARS-CoV-2 during pregnancy. medRxiv. 2021 doi: 10.1101/2021.12.15.21267849. Published online December 16, 2021. [DOI] [Google Scholar]
- 26.Racicot K., Mor G. Risks associated with viral infections during pregnancy. J. Clin. Invest. 2017;127:1591–1599. doi: 10.1172/JCI87490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Silasi M., et al. Viral infections during pregnancy. Am. J. Reprod. Immunol. 2015;73:199–213. doi: 10.1111/aji.12355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Granja M.G., et al. SARS-CoV-2 infection in pregnant women: neuroimmune-endocrine changes at the maternal-fetal interface. Neuroimmunomodulation. 2021;28:1–21. doi: 10.1159/000515556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mao L., et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:683–690. doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Han Y., et al. Neuropsychiatric manifestations of COVID-19, potential neurotropic mechanisms, and therapeutic interventions. Transl. Psychiatry. 2021;11:499. doi: 10.1038/s41398-021-01629-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rogers J.P., et al. Neurology and neuropsychiatry of COVID-19: a systematic review and meta-analysis of the early literature reveals frequent CNS manifestations and key emerging narratives. J. Neurol. Neurosurg. Psychiatry. 2021;92:932–941. doi: 10.1136/jnnp-2021-326405. [DOI] [PubMed] [Google Scholar]
- 32.Castro V.M., et al. Persistence of neuropsychiatric symptoms associated with SARS-CoV-2 positivity among a cohort of children and adolescents. medRxiv. 2021 doi: 10.1101/2021.09.28.21264259. Published online September 29, 2021. [DOI] [Google Scholar]
- 33.Lindan C.E., et al. Neuroimaging manifestations in children with SARS-CoV-2 infection: a multinational, multicentre collaborative study. Lancet Child Adolesc. Health. 2021;5:167–177. doi: 10.1016/S2352-4642(20)30362-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Singer T.G., et al. Coronavirus infections in the nervous system of children: a scoping review making the case for long-term neurodevelopmental surveillance. Pediatr. Neurol. 2021;117:47–63. doi: 10.1016/j.pediatrneurol.2021.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ray S.T.J., et al. Neurological manifestations of SARS-CoV-2 infection in hospitalised children and adolescents in the UK: a prospective national cohort study. Lancet Child Adolesc. Health. 2021;5:631–641. doi: 10.1016/S2352-4642(21)00193-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jaywant A., et al. Frequency and profile of objective cognitive deficits in hospitalized patients recovering from COVID-19. Neuropsychopharmacology. 2021;46:2235–2240. doi: 10.1038/s41386-021-00978-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Huang P., et al. Association between the COVID-19 pandemic and infant neurodevelopment: a comparison before and during COVID-19. Front. Pediatr. 2021;9 doi: 10.3389/fped.2021.662165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Deoni S.C., et al. Impact of the COVID-19 pandemic on early child cognitive development: initial findings in a longitudinal observational study of child health. medRxiv. 2021 doi: 10.1101/2021.08.10.21261846. Published online August 11, 2021. [DOI] [Google Scholar]
- 39.Shuffrey L.C., et al. Association of birth during the COVID-19 pandemic with neurodevelopmental status at 6 months in infants with and without in utero exposure to maternal SARS-CoV-2 infection. JAMA Pediatr. 2022 doi: 10.1001/jamapediatrics.2021.5563. Published online Jan 04, 2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang Y., et al. Impact of Covid-19 in pregnancy on mother's psychological status and infant's neurobehavioral development: a longitudinal cohort study in China. BMC Med. 2020;18:347. doi: 10.1186/s12916-020-01825-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ayed M., et al. Neurodevelopmental outcomes of infants secondary to in utero exposure to maternal SARS-CoV-2 infection: a national prospective study in Kuwait. medRxiv. 2021 doi: 10.1101/2021.11.12.21266291. Published online November 14, 2021. [DOI] [Google Scholar]
- 42.Careaga M., et al. Maternal immune activation and autism spectrum disorder: from rodents to nonhuman and human primates. Biol. Psychiatry. 2017;81:391–401. doi: 10.1016/j.biopsych.2016.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Santana-Coelho D., et al. Advancing autism research from mice to marmosets: behavioral development of offspring following prenatal maternal immune activation. Front Psychiatry. 2021;12 doi: 10.3389/fpsyt.2021.705554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bauman M.D., et al. Activation of the maternal immune system during pregnancy alters behavioral development of rhesus monkey offspring. Biol. Psychiatry. 2014;75:332–341. doi: 10.1016/j.biopsych.2013.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hsiao E.Y., et al. Modeling an autism risk factor in mice leads to permanent immune dysregulation. Proc. Natl. Acad. Sci. U. S. A. 2012;109:12776–12781. doi: 10.1073/pnas.1202556109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ito H.T., et al. Maternal immune activation alters nonspatial information processing in the hippocampus of the adult offspring. Brain Behav. Immun. 2010;24:930–941. doi: 10.1016/j.bbi.2010.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Malkova N.V., et al. Manganese-enhanced magnetic resonance imaging reveals increased DOI-induced brain activity in a mouse model of schizophrenia. Proc. Natl. Acad. Sci. U. S. A. 2014;111:E2492–E2500. doi: 10.1073/pnas.1323287111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Malkova N.V., et al. Maternal immune activation yields offspring displaying mouse versions of the three core symptoms of autism. Brain Behav. Immun. 2012;26:607–616. doi: 10.1016/j.bbi.2012.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bergdolt L., Dunaevsky A. Brain changes in a maternal immune activation model of neurodevelopmental brain disorders. Prog. Neurobiol. 2019;175:1–19. doi: 10.1016/j.pneurobio.2018.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Haddad F.L., et al. Maternal immune activation by poly I:C as a preclinical model for neurodevelopmental disorders: a focus on autism and schizophrenia. Neurosci. Biobehav. Rev. 2020;113:546–567. doi: 10.1016/j.neubiorev.2020.04.012. [DOI] [PubMed] [Google Scholar]
- 51.Bilbo S.D., et al. Beyond infection - maternal immune activation by environmental factors, microglial development, and relevance for autism spectrum disorders. Exp. Neurol. 2018;299:241–251. doi: 10.1016/j.expneurol.2017.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Meyer U. Prenatal poly(I:C) exposure and other developmental immune activation models in rodent systems. Biol. Psychiatry. 2014;75:307–315. doi: 10.1016/j.biopsych.2013.07.011. [DOI] [PubMed] [Google Scholar]
- 53.Boulanger-Bertolus J., et al. Increasing role of maternal immune activation in neurodevelopmental disorders. Front. Behav. Neurosci. 2018;12:230. doi: 10.3389/fnbeh.2018.00230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Shi L., et al. Maternal influenza infection causes marked behavioral and pharmacological changes in the offspring. J. Neurosci. 2003;23:297–302. doi: 10.1523/JNEUROSCI.23-01-00297.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Missault S., et al. The risk for behavioural deficits is determined by the maternal immune response to prenatal immune challenge in a neurodevelopmental model. Brain Behav. Immun. 2014;42:138–146. doi: 10.1016/j.bbi.2014.06.013. [DOI] [PubMed] [Google Scholar]
- 56.Choi G.B., et al. The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science. 2016;351:933–939. doi: 10.1126/science.aad0314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Wong H., Hoeffer C. Maternal IL-17A in autism. Exp. Neurol. 2018;299:228–240. doi: 10.1016/j.expneurol.2017.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Smith S.E., et al. Maternal immune activation alters fetal brain development through interleukin-6. J. Neurosci. 2007;27:10695–10702. doi: 10.1523/JNEUROSCI.2178-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Nunez J.L., et al. A novel model for prenatal brain damage. II. Long-term deficits in hippocampal cell number and hippocampal-dependent behavior following neonatal GABAA receptor activation. Exp. Neurol. 2003;181:270–280. doi: 10.3201/eid0906.020377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Boksa P. Effects of prenatal infection on brain development and behavior: a review of findings from animal models. Brain Behav. Immun. 2010;24:881–897. doi: 10.1016/j.bbi.2010.03.005. [DOI] [PubMed] [Google Scholar]
- 61.Coulthard L.G., et al. Complement: The emerging architect of the developing brain. Trends Neurosci. 2018;41:373–384. doi: 10.1016/j.tins.2018.03.009. [DOI] [PubMed] [Google Scholar]
- 62.Canetta S., et al. Elevated maternal C-reactive protein and increased risk of schizophrenia in a national birth cohort. Am. J. Psychiatry. 2014;171:960–968. doi: 10.1176/appi.ajp.2014.13121579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Girard S., et al. IL-1 receptor antagonist protects against placental and neurodevelopmental defects induced by maternal inflammation. J. Immunol. 2010;184:3997–4005. doi: 10.4049/jimmunol.0903349. [DOI] [PubMed] [Google Scholar]
- 64.Meyer U., et al. Adult behavioral and pharmacological dysfunctions following disruption of the fetal brain balance between pro-inflammatory and IL-10-mediated anti-inflammatory signaling. Mol. Psychiatry. 2008;13:208–221. doi: 10.1038/sj.mp.4002042. [DOI] [PubMed] [Google Scholar]
- 65.Rudolph M.D., et al. Maternal IL-6 during pregnancy can be estimated from newborn brain connectivity and predicts future working memory in offspring. Nat. Neurosci. 2018;21:765–772. doi: 10.1038/s41593-018-0128-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Spann M.N., et al. Maternal immune activation during the third trimester is associated with neonatal functional connectivity of the salience network and fetal to toddler behavior. J. Neurosci. 2018;38:2877–2886. doi: 10.1523/JNEUROSCI.2272-17.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Graham A.M., et al. Maternal systemic interleukin-6 during pregnancy is associated with newborn amygdala phenotypes and subsequent behavior at 2 years of age. Biol. Psychiatry. 2018;83:109–119. doi: 10.1016/j.biopsych.2017.05.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Rasmussen J.M., et al. Maternal Interleukin-6 concentration during pregnancy is associated with variation in frontolimbic white matter and cognitive development in early life. Neuroimage. 2019;185:825–835. doi: 10.1016/j.neuroimage.2018.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Qin C., et al. Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan, China. Clin. Infect. Dis. 2020;71:762–768. doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Liu J., et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55 doi: 10.1016/j.ebiom.2020.102763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Cao X. COVID-19: immunopathology and its implications for therapy. Nat. Rev. Immunol. 2020;20:269–270. doi: 10.1038/s41577-020-0308-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Cavalcante M.B., et al. Maternal immune responses and obstetrical outcomes of pregnant women with COVID-19 and possible health risks of offspring. J. Reprod. Immunol. 2021;143 doi: 10.1016/j.jri.2020.103250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Moore J.B., June C.H. Cytokine release syndrome in severe COVID-19. Science. 2020;368:473–474. doi: 10.1126/science.abb8925. [DOI] [PubMed] [Google Scholar]
- 74.Mehta P., et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Pacha O., et al. COVID-19: a case for inhibiting IL-17? Nat. Rev. Immunol. 2020;20:345–346. doi: 10.1038/s41577-020-0328-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wu D., Yang X.O. Th17 responses in cytokine storm of COVID-19: an emerging target of JAK2 inhibitor fedratinib. J. Microbiol. Immunol. Infect. 2020;53:368–370. doi: 10.1016/j.jmii.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Tanacan A., et al. The impact of COVID-19 infection on the cytokine profile of pregnant women: a prospective case-control study. Cytokine. 2021;140 doi: 10.1016/j.cyto.2021.155431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Sherer M.L., et al. Pregnancy alters interleukin-1 beta expression and antiviral antibody responses during severe acute respiratory syndrome coronavirus 2 infection. Am. J. Obstet. Gynecol. 2021;225 doi: 10.1016/j.ajog.2021.03.028. 301.e301.e1-301.e14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lu-Culligan A., et al. Maternal respiratory SARS-CoV-2 infection in pregnancy is associated with a robust inflammatory response at the maternal-fetal interface. Med (N Y) 2021;2:591–610.e510. doi: 10.1016/j.medj.2021.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Bordt E.A., et al. Maternal SARS-CoV-2 infection elicits sexually dimorphic placental immune responses. Sci. Transl. Med. 2021;13:eabi7428. doi: 10.1126/scitranslmed.abi7428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Argueta L.B., et al. SARS-CoV-2 infects syncytiotrophoblast and activates inflammatory responses in the placenta. bioRxiv. 2021 doi: 10.1101/2021.06.01.446676. Published online June 17, 2021. [DOI] [Google Scholar]
- 82.Lu-Culligan A., Iwasaki A. 2021. The New York Times 26 November. [Google Scholar]
- 83.Edlow A.G., et al. Assessment of maternal and neonatal SARS-CoV-2 viral load, transplacental antibody transfer, and placental pathology in pregnancies during the COVID-19 pandemic. JAMA Netw. Open. 2020;3 doi: 10.1001/jamanetworkopen.2020.30455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Hecht J.L., et al. SARS-CoV-2 can infect the placenta and is not associated with specific placental histopathology: a series of 19 placentas from COVID-19-positive mothers. Mod. Pathol. 2020;33:2092–2103. doi: 10.1038/s41379-020-0639-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Garcia-Flores V., et al. Maternal-fetal immune responses in pregnant women infected with SARS-CoV-2. Nat. Commun. 2022;13:320. doi: 10.1038/s41467-021-27745-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Gee S., et al. The legacy of maternal SARS-CoV-2 infection on the immunology of the neonate. Nat. Immunol. 2021;22:1490–1502. doi: 10.1038/s41590-021-01049-2. [DOI] [PubMed] [Google Scholar]
- 87.Matute J.D., et al. Single-cell immunophenotyping of the fetal immune response to maternal SARS-CoV-2 infection in late gestation. Pediatr Res. 2021 doi: 10.1038/s41390-021-01793-z. Published online November 8, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Baines K.J., et al. Maternal immune activation alters fetal brain development and enhances proliferation of neural precursor cells in rats. Front. Immunol. 2020;11:1145. doi: 10.3389/fimmu.2020.01145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kwon J., et al. Distinct trans-placental effects of maternal immune activation by TLR3 and TLR7 agonists: implications for schizophrenia risk. Sci. Rep. 2021;11:23841. doi: 10.1038/s41598-021-03216-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Mueller F.S., et al. Influence of poly(I:C) variability on thermoregulation, immune responses and pregnancy outcomes in mouse models of maternal immune activation. Brain Behav. Immun. 2019;80:406–418. doi: 10.1016/j.bbi.2019.04.019. [DOI] [PubMed] [Google Scholar]
- 91.Oskvig D.B., et al. Maternal immune activation by LPS selectively alters specific gene expression profiles of interneuron migration and oxidative stress in the fetus without triggering a fetal immune response. Brain Behav. Immun. 2012;26:623–634. doi: 10.1016/j.bbi.2012.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Bonnin A., et al. A transient placental source of serotonin for the fetal forebrain. Nature. 2011;472:347–350. doi: 10.1038/nature09972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Bonnin A., Levitt P. Fetal, maternal, and placental sources of serotonin and new implications for developmental programming of the brain. Neuroscience. 2011;197:1–7. doi: 10.1016/j.neuroscience.2011.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Bonnin A., et al. Serotonin modulates the response of embryonic thalamocortical axons to netrin-1. Nat. Neurosci. 2007;10:588–597. doi: 10.1038/nn1896. [DOI] [PubMed] [Google Scholar]
- 95.Mitchell A.J., et al. The influence of maternal metabolic state and nutrition on offspring neurobehavioral development: a focus on preclinical models. Biol. Psychiatry Cogn. Neurosci. Neuroimaging. 2021 doi: 10.1016/j.bpsc.2021.11.014. Published online December 13, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Rosenfeld C.S. Placental serotonin signaling, pregnancy outcomes, and regulation of fetal brain development. Biol. Reprod. 2020;102:532–538. doi: 10.1093/biolre/ioz204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Velasquez J.C., et al. Placental serotonin: implications for the developmental effects of SSRIs and maternal depression. Front. Cell. Neurosci. 2013;7:47. doi: 10.3389/fncel.2013.00047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Valleau J.C., Sullivan E.L. The impact of leptin on perinatal development and psychopathology. J. Chem. Neuroanat. 2014;61–62:221–232. doi: 10.1016/j.jchemneu.2014.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Meyer U., et al. Adult brain and behavioral pathological markers of prenatal immune challenge during early/middle and late fetal development in mice. Brain Behav. Immun. 2008;22:469–486. doi: 10.1016/j.bbi.2007.09.012. [DOI] [PubMed] [Google Scholar]
- 100.Meyer U., et al. Preliminary evidence for a modulation of fetal dopaminergic development by maternal immune activation during pregnancy. Neuroscience. 2008;154:701–709. doi: 10.1016/j.neuroscience.2008.04.031. [DOI] [PubMed] [Google Scholar]
- 101.Luchicchi A., et al. Maternal immune activation disrupts dopamine system in the offspring. Int. J. Neuropsychopharmacol. 2016;19:pyw007. doi: 10.1093/ijnp/pyw007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Grayson B.E., et al. Changes in melanocortin expression and inflammatory pathways in fetal offspring of nonhuman primates fed a high-fat diet. Endocrinology. 2010;151:1622–1632. doi: 10.1210/en.2009-1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Bitanihirwe B.K., et al. Late prenatal immune activation in mice leads to behavioral and neurochemical abnormalities relevant to the negative symptoms of schizophrenia. Neuropsychopharmacology. 2010;35:2462–2478. doi: 10.1038/npp.2010.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Simoes L.R., et al. Maternal immune activation induced by lipopolysaccharide triggers immune response in pregnant mother and fetus, and induces behavioral impairment in adult rats. J. Psychiatr. Res. 2018;100:71–83. doi: 10.1016/j.jpsychires.2018.02.007. [DOI] [PubMed] [Google Scholar]
- 105.Kalish B.T., et al. Maternal immune activation in mice disrupts proteostasis in the fetal brain. Nat. Neurosci. 2021;24:204–213. doi: 10.1038/s41593-020-00762-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Chu C., et al. The microbiota regulate neuronal function and fear extinction learning. Nature. 2019;574:543–548. doi: 10.1038/s41586-019-1644-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Nakai Y., et al. Apoptosis and microglial activation in influenza encephalopathy. Acta Neuropathol. 2003;105:233–239. doi: 10.1007/s00401-002-0605-x. [DOI] [PubMed] [Google Scholar]
- 108.Smolders S., et al. Controversies and prospects about microglia in maternal immune activation models for neurodevelopmental disorders. Brain Behav. Immun. 2018;73:51–65. doi: 10.1016/j.bbi.2018.06.001. [DOI] [PubMed] [Google Scholar]
- 109.Fernandez de Cossio L., et al. Prenatal infection leads to ASD-like behavior and altered synaptic pruning in the mouse offspring. Brain Behav. Immun. 2017;63:88–98. doi: 10.1016/j.bbi.2016.09.028. [DOI] [PubMed] [Google Scholar]
- 110.Zhao Q., et al. Maternal immune activation-induced PPARgamma-dependent dysfunction of microglia associated with neurogenic impairment and aberrant postnatal behaviors in offspring. Neurobiol. Dis. 2019;125:1–13. doi: 10.1016/j.nbd.2019.01.005. [DOI] [PubMed] [Google Scholar]
- 111.Lenz K.M., Nelson L.H. Microglia and beyond: innate immune cells as regulators of brain development and behavioral function. Front. Immunol. 2018;9:698. doi: 10.3389/fimmu.2018.00698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Paolicelli R.C., et al. Synaptic pruning by microglia is necessary for normal brain development. Science. 2011;333:1456–1458. doi: 10.1126/science.1202529. [DOI] [PubMed] [Google Scholar]
- 113.Paolicelli R.C., Ferretti M.T. Function and dysfunction of microglia during brain development: consequences for synapses and neural circuits. Front. Synaptic Neurosci. 2017;9:9. doi: 10.3389/fnsyn.2017.00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Stephan A.H., et al. The complement system: an unexpected role in synaptic pruning during development and disease. Annu. Rev. Neurosci. 2012;35:369–389. doi: 10.1146/annurev-neuro-061010-113810. [DOI] [PubMed] [Google Scholar]
- 115.Kreitz S., et al. Maternal immune activation during pregnancy impacts on brain structure and function in the adult offspring. Brain Behav. Immun. 2020;83:56–67. doi: 10.1016/j.bbi.2019.09.011. [DOI] [PubMed] [Google Scholar]
- 116.Megli C., Coyne C.B. Gatekeepers of the fetus: characterization of placental macrophages. J. Exp. Med. 2021;218 doi: 10.1084/jem.20202071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Narang K., et al. Placental immune responses to viruses: molecular and histo-pathologic perspectives. Int. J. Mol. Sci. 2021;22:2921. doi: 10.3390/ijms22062921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Raj R.S., et al. Influenza, immune system, and pregnancy. Reprod. Sci. 2014;21:1434–1451. doi: 10.1177/1933719114537720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Shah P.S., et al. Classification system and case definition for SARS-CoV-2 infection in pregnant women, fetuses, and neonates. Acta Obstet. Gynecol. Scand. 2020;99:565–568. doi: 10.1111/aogs.13870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Adhikari E.H., et al. Pregnancy outcomes among women with and without severe acute respiratory syndrome coronavirus 2 infection. JAMA Netw. Open. 2020;3 doi: 10.1001/jamanetworkopen.2020.29256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Dumitriu D., et al. Outcomes of neonates born to mothers with severe acute respiratory syndrome coronavirus 2 infection at a large medical center in New York City. JAMA Pediatr. 2020;175:157–167. doi: 10.1001/jamapediatrics.2020.4298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Woodworth K.R., et al. Birth and infant outcomes following laboratory-confirmed SARS-CoV-2 infection in pregnancy - SET-NET, 16 Jurisdictions, March 29-October 14, 2020. MMWR Morb. Mortal. Wkly Rep. 2020;69:1635–1640. doi: 10.15585/mmwr.mm6944e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Flaherman, V.J. et al. Infant outcomes following maternal infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): first report from the Pregnancy Coronavirus Outcomes Registry (PRIORITY) Study. Clin. Infect. Dis. 73, e2810–e2813. [DOI] [PMC free article] [PubMed]
- 124.Shook L.L., et al. Vertical transmission of SARS-CoV-2: consider the denominator. Am. J. Obstet. Gynecol. MFM. 2021;3 doi: 10.1016/j.ajogmf.2021.100386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Kotlyar A.M., et al. Vertical transmission of coronavirus disease 2019: a systematic review and meta-analysis. Am. J. Obstet. Gynecol. 2021;224:35–53.e33. doi: 10.1016/j.ajog.2020.07.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Tallarek A.C., et al. Inefficient placental virus replication and absence of neonatal cell-specific immunity upon SARS-CoV-2 infection during pregnancy. Front. Immunol. 2021;12 doi: 10.3389/fimmu.2021.698578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Egloff C., et al. Evidence and possible mechanisms of rare maternal-fetal transmission of SARS-CoV-2. J. Clin. Virol. 2020;128 doi: 10.1016/j.jcv.2020.104447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Fenizia C., et al. Unlikely SARS-CoV-2 transmission during vaginal delivery. Reprod. Sci. 2021;28:2939–2941. doi: 10.1007/s43032-021-00681-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Schwartz D.A., Levitan D. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infecting pregnant women and the fetus, intrauterine transmission, and placental pathology during the coronavirus disease 2019 (COVID-19) pandemic: it’s complicated. Arch. Pathol. Lab. Med. 2021;145:925–928. doi: 10.5858/arpa.2021-0164-ED. [DOI] [PubMed] [Google Scholar]
- 130.Dube R., Kar S.S. COVID-19 in pregnancy: the foetal perspective - a systematic review. BMJ Paediatr. Open. 2020;4 doi: 10.1136/bmjpo-2020-000859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Leyser M., et al. Potential risk of brain damage and poor developmental outcomes in children prenatally exposed to SARS-CoV-2: a systematic review. Rev. Paul Pediatr. 2021;40 doi: 10.1590/1984-0462/2022/40/2020415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Koliogiannis V., et al. Proceedings of Radiological Society of North America Annual Meeting, Chicago, IL, USA. 2021. SARS-CoV-2 infection during pregnancy: does fetal MRI show signs of impaired fetal brain development? [Google Scholar]
- 133.Soto-Torres E., et al. Ultrasound and Doppler findings in pregnant women with SARS-CoV-2 infection. Ultrasound Obstet. Gynecol. 2021;58:111–120. doi: 10.1002/uog.23642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Varma P., et al. Novel targets of SARS-CoV-2 spike protein in human fetal brain development suggest early pregnancy vulnerability. Front. Neurosci. 2020;14 doi: 10.3389/fnins.2020.614680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Reza-Zaldívar E.E., et al. Infection mechanism of SARS-COV-2 and its implication on the nervous system. Front. Immunol. 2020;11 doi: 10.3389/fimmu.2020.621735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Zhang L., et al. SARS-CoV-2 crosses the blood-brain barrier accompanied with basement membrane disruption without tight junctions alteration. Signal. Transduct. Target Ther. 2021;6:337. doi: 10.1038/s41392-021-00719-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Song E., et al. Neuroinvasion of SARS-CoV-2 in human and mouse brain. J. Exp. Med. 2021;218 doi: 10.1084/jem.20202135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Nascimento J.M., et al. Human cerebral organoids and fetal brain tissue share proteomic similarities. Front. Cell Dev. Biol. 2019;7:303. doi: 10.3389/fcell.2019.00303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Jacob F., et al. Human pluripotent stem cell-derived neural cells and brain organoids reveal SARS-CoV-2 neurotropism predominates in choroid plexus epithelium. Cell Stem Cell. 2020;27:937–950.e939. doi: 10.1016/j.stem.2020.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Pellegrini L., et al. SARS-CoV-2 infects the brain choroid plexus and disrupts the blood-CSF barrier in human brain organoids. Cell Stem Cell. 2020;27:951–961.e955. doi: 10.1016/j.stem.2020.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.McMahon C.L., et al. SARS-CoV-2 targets glial cells in human cortical organoids. Stem Cell Reports. 2021;16:1156–1164. doi: 10.1016/j.stemcr.2021.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.DeSisto C.L., et al. Risk for stillbirth among women with and without COVID-19 at delivery hospitalization - United States, March 2020-September 2021. MMWR Morb. Mortal. Wkly Rep. 2021;70:1640–1645. doi: 10.15585/mmwr.mm7047e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Wang Y., et al. Transmission, viral kinetics and clinical characteristics of the emergent SARS-CoV-2 Delta VOC in Guangzhou, China. EClinicalMedicine. 2021;40 doi: 10.1016/j.eclinm.2021.101129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Twohig K.A., et al. Hospital admission and emergency care attendance risk for SARS-CoV-2 Delta (B.1.617.2) compared with alpha (B.1.1.7) variants of concern: a cohort study. Lancet Infect. Dis. 2021 doi: 10.1016/S1473-3099(21)00475-8. Published online August 27, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Adhikari E.H., et al. Increasing severity of COVID-19 in pregnancy with Delta (B.1.617.2) variant surge. Am. J. Obstet. Gynecol. 2022;226:149–151. doi: 10.1016/j.ajog.2021.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Wang A.M., et al. Association of the Delta (B.1.617.2) variant of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) with pregnancy outcomes. Obstet. Gynecol. 2021;138:838–841. doi: 10.1097/AOG.0000000000004595. [DOI] [PubMed] [Google Scholar]
- 147.Vousden N., et al. Severity of maternal infection and perinatal outcomes during periods of SARS-CoV-2 wildtype, alpha, and delta variant dominance in the UK: prospective cohort study. BMJ Medicine. 2022 doi: 10.1136/bmjmed-2021-000053. Published online Feb 01, 2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Liu Y., et al. Delta spike P681R mutation enhances SARS-CoV-2 fitness over Alpha variant. bioRxiv. 2021 doi: 10.1101/2021.08.12.456173. Published online September 5, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Takeda M. Proteolytic activation of SARS-CoV-2 spike protein. Microbiol. Immunol. 2022;66:15–23. doi: 10.1111/1348-0421.12945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Ashary N., et al. Single-cell RNA-seq identifies cell subsets in human placenta that highly expresses factors driving pathogenesis of SARS-CoV-2. Front. Cell Dev. Biol. 2020;8:783. doi: 10.3389/fcell.2020.00783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Ouyang Y., et al. Term human placental trophoblasts express SARS-CoV-2 entry factors ACE2, TMPRSS2, and Furin. mSphere. 2021;6 doi: 10.1128/mSphere.00250-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Shanes E.D., et al. Placental pathology in COVID-19. Am. J. Clin. Pathol. 2020;154:23–32. doi: 10.1093/ajcp/aqaa089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Bertero L., et al. Placenta histopathology in SARS-CoV-2 infection: analysis of a consecutive series and comparison with control cohorts. Virchows Arch. 2021;479:715–728. doi: 10.1007/s00428-021-03097-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Shook L.L., et al. SARS-CoV-2 placentitis associated with B.1.617.2 (Delta) variant and fetal distress or demise. J. Infect. Dis. 2022;225:754–758. doi: 10.1093/infdis/jiac008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Ahlberg M., et al. Association of SARS-CoV-2 test status and pregnancy outcomes. JAMA. 2020;324:1782–1785. doi: 10.1001/jama.2020.19124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Mendoza M., et al. Pre-eclampsia-like syndrome induced by severe COVID-19: a prospective observational study. BJOG. 2020;127:1374–1380. doi: 10.1111/1471-0528.16339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Metz T.D., et al. Disease severity and perinatal outcomes of pregnant patients with coronavirus disease 2019 (COVID-19) Obstet. Gynecol. 2021;137:571–580. doi: 10.1097/AOG.0000000000004339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Villar J., et al. Maternal and neonatal morbidity and mortality among pregnant women with and without COVID-19 infection: the INTERCOVID Multinational Cohort Study. JAMA Pediatr. 2021;175:817–826. doi: 10.1001/jamapediatrics.2021.1050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Sun B.Z., et al. Association of preeclampsia in term births with neurodevelopmental disorders in offspring. JAMA Psychiatry. 2020;77:823–829. doi: 10.1001/jamapsychiatry.2020.0306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Pierrat V., et al. Neurodevelopmental outcome at 2 years for preterm children born at 22 to 34 weeks’ gestation in France in 2011: EPIPAGE-2 cohort study. BMJ. 2017;358 doi: 10.1136/bmj.j3448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Sellgren C.M., et al. Increased synapse elimination by microglia in schizophrenia patient-derived models of synaptic pruning. Nat. Neurosci. 2019;22:374–385. doi: 10.1038/s41593-018-0334-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Sheridan S.D., et al. Umbilical cord blood-derived microglia-like cells to model COVID-19 exposure. Transl. Psychiatry. 2021;11:179. doi: 10.1038/s41398-021-01287-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Ceasrine A.M., et al. Single cell profiling of Hofbauer cells and fetal brain microglia reveals shared programs and functions. bioRxiv. 2021 doi: 10.1101/2021.12.03.471177. Published online December 4, 2021. [DOI] [Google Scholar]
- 164.Sheridan S.D.H., et al. Patient-derived in vitro models of microglial function and synaptic engulfment in schizophrenia. Biol. Psychiatry. 2022 doi: 10.1016/j.biopsych.2022.01.004. Published online January 19, 2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Ginhoux F., et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science. 2010;330:841–845. doi: 10.1126/science.1194637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Gomez Perdiguero E., et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature. 2015;518:547–551. doi: 10.1038/nature13989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Gomez Perdiguero E., et al. Development and homeostasis of “resident” myeloid cells: the case of the microglia. Glia. 2013;61:112–120. doi: 10.1002/glia.22393. [DOI] [PubMed] [Google Scholar]
- 168.Schwartz D.A., et al. Placental tissue destruction and insufficiency from COVID-19 causes stillbirth and neonatal death from hypoxic-ischemic injury: a study of 68 cases with SARS-CoV-2 placentitis from 12 countries. Arch. Pathol. Lab. Med. 2022 doi: 10.5858/arpa.202-0029-SA. Published online February 10, 2022. [DOI] [PubMed] [Google Scholar]