Abstract
Objective The objective of the study was to review the obstetric outcomes of complete hydatidiform molar pregnancies with a coexisting fetus (CHMCF), a rare clinical entity that is not well described.
Materials and Methods We performed a retrospective case series with pathology-confirmed HMCF. The cases were collected via solicitation through a private maternal-fetal medicine physician group on social media. Each contributing institution from across the United States ( n = 9) obtained written informed consent from the patients directly, obtained institutional data transfer agreements as required, and transmitted the data using a Health Insurance Portability and Accountability Act of 1996 (HIPAA) compliant modality. Data collected included maternal, fetal/genetic, placental, and delivery characteristics. For descriptive analysis, continuous variables were reported as median with standard deviation and range.
Results Nine institutions contributed to the 14 cases collected. Nine (64%) cases of CHMCF were a product of assisted reproductive technology and one case was trizygotic. The median gestational age at diagnosis was 12 weeks and 2 days (9 weeks–19 weeks and 4 days), and over half were diagnosed in the first trimester. The median human chorionic gonadotropin (hCG) at diagnosis was 355,494 mIU/mL (49,770–700,486 mIU/mL). Placental mass size universally enlarged over the surveillance period. When invasive testing was performed, insufficient sample or no growth was noted in 40% of the sampled cases. Antenatal complications occurred in all delivered patients, with postpartum hemorrhage (71%) and hypertensive disorders of pregnancy (29%) being the most frequent outcomes. Delivery outcomes were variable. Four patients developed gestational trophoblastic neoplasia.
Conclusion This series is the largest report of obstetric outcomes for CHMCF to date and highlights the need to counsel patients about the severe maternal and fetal complications in continuing pregnancies, including progression to gestational trophoblastic neoplastic disease.
Key Points
CHMCF is a rare obstetric complication and may be associated with the use of assisted reproductive technology.
Universally, patients with CHMCF who elected to manage expectantly developed antenatal complications.
The risk of developing gestational trophoblastic neoplasia after CHMCF is high, and termination of the pregnancy did not decrease this risk.
Keywords: antenatal complications, multiple gestation, twin pregnancy, molar pregnancy, gestational trophoblastic neoplasia, maternal morbidity
Ultrasonographic evidence of an enlarged multicystic placenta with a normal-appearing fetus is an uncommon finding during routine surveillance of pregnancy. The differential diagnoses of these features include partial hydatidiform molar pregnancy with a coexisting fetus (HMCF) or complete HMCF (CHMCF), placental mesenchymal dysplasia (PMD), placental infarcts, chorioangioma, subchorionic hematoma, placental cysts, and placenta accreta spectrum (PAS) disorders. In the context of an otherwise normal-appearing fetus, the obstetrical course and postpartum follow-up of these conditions are vastly different ( Table 1 ).
Table 1. Comparison of the clinical findings of placental mesenchymal dysplasia (PMD), complete hydatidiform mole (CHM), and partial hydatidiform mole (PHM).
| PMD | CHM | PHM | |
|---|---|---|---|
| Ultrasound findings | Enlarged multicystic placenta with anechoic regions (“moth-eaten” appearance) Findings widely distributed, large edematous villi |
||
| Fetus 18 | • Can be unremarkable • FGR (50%) • IUFD or neonatal death (43%) • Consider BWS findings: macroglossia, omphalocele, genitourinary abnormalities, overgrowth, polyhydramnios |
• Coexisting fetus can be unremarkable | • May be structurally abnormal triploid fetus 19 |
| Pathology | • Enlarged stem villi with loose connective tissue and cisternlike formations • Absent trophoblastic changes |
• Hydropic swelling of villi • Diffuse trophoblastic hyperplasia • Diffuse and marked trophoblastic atypia at the molar implantation site |
• Focal trophoblastic hyperplasia • Marked variability in the size and degree of swelling, and cavitation of the villi • Marked scalloping and prominent stromal trophoblastic inclusion in the villi • Focal and mild trophoblastic atypia at molar implantation site |
| Associated maternal morbidities | None identified | • GTN • Preeclampsia • Choriocarcinoma |
1. GTN 2. Preeclampsia 3. Choriocarcinoma |
| Cytogenetics | • Normal karyotype (89%) • 46 XX (78%), 46 XY (22%) • BWS: confirmed or suspected (23%) 20 |
• 46 XX: haploid 23 X sperm duplicates its own chromosomes
21
22
• 46 XY: ova penetrated by 2 sperm (dispermy), 46 XY 23 |
• Triploidy: extra haploid sperm 4 |
| Clinical presentation | No definitive clinical characteristics, but may be associated with preterm labor, secondary to amniotic fluid abnormalities | • Vaginal bleeding • Size greater than dates • Theca lutein cysts • Hyperemesis • Preeclampsia • Hyperthyroidism • Pulmonary edema • Respiratory distress |
• Commonly diagnosed after missed or incomplete abortion, based on pathology |
Abbreviations: BWS, Beckwith–Wiedemann syndrome; CHM, complete hydatidiform mole; FGR, fetal growth restriction; GTN, gestational trophoblastic neoplasia; IUFD, intrauterine fetal demise; PHM, partial hydatidiform mole; PMD, placental mesenchymal dysplasia.
In the case of a CHMCF, it is especially important to have an accurate diagnosis. This rare condition, affecting 20,000 to 100,000 pregnancies, 1 2 is fraught with potential maternal complications, such as hemorrhage, preeclampsia, and preterm delivery of the viable coexisting fetus. Persistent gestational trophoblastic neoplasia (GTN) is also seen more frequently in CHMCF, when compared with a single complete mole, and termination of the pregnancy has not been shown to decrease this risk. 1 3 4
Although there have been large case series reported on CHMCF, they have focused mainly on outcomes as they relate to the GTN associated with this condition. 1 3 4 In these reports, the use of artificial reproductive technology (ART) was either not reported or, when reported, did not account for a majority of cases (13%). An increased use of ART over the past several decades may affect the prevalence of CHMCF and so obstetricians should be cognizant of this condition and its associated ante-, intra-, and postpartum risks. When an isolated complete molar pregnancy is noted, evacuation of the premalignant molar tissue is recommended. However, in the case of a CHMCF, a woman may elect to be managed expectantly to prolong the pregnancy. Here, we provide the first multicenter series of CHMCF reporting detailed accounts of the diagnosis, pregnancy outcomes, and postpartum follow-up, as well as a review of existing literature, to aid in the counseling of this at-risk cohort of pregnant women.
Materials and Methods
A retrospective analysis of women with CHMCF pregnancies was performed. The cases were collected via solicitation through a private maternal-fetal medicine physician group on social media. Each contributing institution from across the United States ( n = 9) obtained written informed consent from the patient(s) directly, obtained institutional data transfer agreements as requested, and transmitted the data using a Health Insurance Portability and Accountability Act of 1996 (HIPAA) compliant modality.
Electronic records were reviewed and the following data were identified: maternal characteristics (age, gravidity, parity, prepregnancy body mass index, race, and prior maternal comorbidities); mode of conception; gestational age at diagnosis; human chorionic gonadotropin (hCG) at diagnosis; zygosity of the pregnancy; screening assessments (including laboratory and imaging); antenatal genetics (procedure type, results, and timing); and size of placental mass as measured by prenatal ultrasonography. Maternal complications including vaginal bleeding, hyperthyroidism, and hypertensive disorders of pregnancy were noted. The timing, mode, and indication for delivery, as well as the estimated blood loss or complications of delivery or procedure were recorded. Postnatal confirmation of genetics and pathology, postpartum follow-up, including hCG trend and time to nadir, diagnosis of GTN, and subsequent treatments were identified.
Fetal and neonatal outcomes recorded included any structural anomalies noted prenatally, intrauterine fetal growth restriction, intrauterine or neonatal fetal demise, and neonatal birthweight.
Statistical Analysis
For descriptive analysis, continuous variables were reported as median with standard deviation and range. Categorical variables were reported as proportions.
Results
After solicitation via social media, nine institutions were able to obtain patient consent and contributed 14 cases in total. Clinical characteristics of the patients are delineated in Table 2 .
Table 2. Patient characteristics and comorbidities.
| Case no. | Age (y) | G/P | Conception | BMI | Race/ethnicity | Comorbidities |
|---|---|---|---|---|---|---|
| 1 | 30 | 2/1001 | OI/GnTP/IUI | 20.8 | Caucasian | None |
| 2 | 27 | 1/0 | OI/CC | 26.7 | Caucasian | PCOS, seizure disorder on Lamictal |
| 3 | 36 | 1/0 | OI/CC/IUI | 30.6 | Caucasian | Lupus on Plaquenil |
| 4 | 32 | 2/1001 | Spontaneous | 23.0 | Caucasian | None |
| 5 | 26 | 2/0010 | Spontaneous | 34.0 | Caucasian/Asian | Anxiety, depression |
| 6 | 29 | 2/1001 | OI/GnTP | 22.6 | Caucasian | Chronic HTN |
| 7 | 27 | 1/0 | OI/CC | 36.0 | Caucasian | None |
| 8 | 35 | 2/1001 | Spontaneous | 31.6 | White | h/o Roux-en-Y, anemia, h/o gestational HTN |
| 9 | 28 | 8/2052 | OI/GnTP | 28.2 | White | Migraine, PCOS with infertility |
| 10 | 28 | 3/1011 | Spontaneous | 19.4 | Arab-American | h/o 2nd trimester IUFD (19 wk) |
| 11 | 32 | 4/2012 | Spontaneous | 21.0 | White | h/o bilateral PE, h/o 2nd trimester IUFD (16 wk) |
| 12 | 38 | 2/1001 | IVF | 22.9 | Asian | seizures on levetiracetam and lamotrigine |
| 13 | 34 | 3/1011 | COH/IUI | 24.0 | Caucasian | None |
| 14 | 33 | 1/0 | IVF | 21.0 | Asian | Asthma |
Abbreviations: BMI, body mass index; CC, clomiphene citrate; COH, controlled ovarian hyperstimulation; GnTP, gonadotropin; h/o, history of; HTN, hypertension; IUI, intrauterine insemination; IUFD, intrauterine fetal demise; IVF, in vitro fertilization; OI, ovulation induction; PCOS, polycystic ovarian syndrome; PE, pulmonary embolism.
Of the cases presented here, 64% were the product of ART: 29% ovulation induction alone, 21% ovulation induction with intrauterine insemination, and 14% in vitro fertilization. Only five cases (36%) were due to spontaneous conception. The median gestational age at diagnosis was 12 weeks and 2 days (9 weeks–19 weeks and 4 days), with 64% ( n = 9) diagnosed in the first trimester and the remaining diagnosed by 20 weeks of gestation. Upon either diagnosis or suspicion of diagnosis, all patients were referred to a maternal-fetal medicine specialist, who was involved in the remainder of the pregnancy. The median hCG at diagnosis was 355,494 mIU/mL (49,770–700,486 mIU/mL). The largest dimension of the placental mass at the time of diagnosis varied, ranging from 3.5 to 12 cm. The size of the placental mass universally enlarged over the antenatal surveillance period. Antenatal genetic analysis was performed in 10 of the 14 cases. Insufficient sample or no growth of the sample from either amniocentesis ( n = 5) or chorionic villous sampling (CVS; n = 5) was a common finding, occurring in 40% of cases sampled ( n = 4).
In the reported dizygotic CHMCF pregnancies, no malformations were identified. The one case of trizygotic CHMCF pregnancy had a complete mole, a coexisting structurally normal fetus, and a partial molar pregnancy with cystic hygroma and complex congenital heart defect.
Antenatal management and complications are described in Table 3 . Universally, patients with CHMCF experienced some form of antenatal complication, including vaginal bleeding (10; 71%), hypertensive disorder of pregnancy (4; 28.9%), pulmonary edema (1; 0.7%), and hyperthyroidism (1; 0.7%). Of the patients with vaginal bleeding, 4 of 10 (40%) required admission and/or transfusion. The case of hyperthyroidism required medical treatment with antithyroid medications and ultimately resulted in termination of pregnancy.
Table 3. Antenatal management and pregnancy outcomes.
| Case no. | Planned management | Complications | GA at delivery | Delivery type | EBL (mL) | Genetics prenatal | hCG trend | Subsequent Dx | Treatment |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Expectant (initially declined termination) Serial growth ultrasounds Termination when HELLP |
SAB of twin B at 14 wk HELLP at 16 wk |
16 wk and 6 d | D&E | 1,000 | 70 XXXY | Plateau at 8 wk PP | Metastatic GTN (FIGO stage 3) lung nodules | IV MTX |
| 2 | Expectant (declined termination) Serial growth ultrasounds |
VB (admission) Anemia Preterm labor |
20 wk and 2 d | SVD | 300 | None | Nadir by 12 wk PP | None | None |
| 3 | Expectant | VB Tachycardia Palpitations |
13 wk and 3 d | D&E | 200 | T22 | Nadir by 13 wk PP | None | None |
| 4 | Expectant (declined termination) | VB Hyperthyroidism (admission) Anemia/transfusion (2 U pRBC) PEC with severe features Hemorrhage with passage of molar tissue Intraoperative transfusion (3 U pRBC) Hysterectomy due to postpartum hemorrhage |
24 wk and 5 d | Emergent classical CD | 2,500 | None | Nadir by 8 wk PP, then increased | Metastatic GTN FIGO stage 4 | IV MTX then IV dactinomycin |
| 5 | Desired termination | Pulmonary edema | 21 wk and 1 d | D&E | 125 | None | Nadir by 7 wk PP | None | None |
| 6 | Expectant Serial laboratories Serial growth ultrasounds |
SAB of twin A VB Superimposed PEC with severe features |
34 wk and 5 d | SVD | 250 | None | Nadir by 4 wk PP | None | None |
| 7 | Expectant Serial laboratories Serial growth ultrasounds |
VB GHTN |
34 wk and 2 d | Classical CD | 1,000 | None | Not available | None | None |
| 8 | Expectant Serial laboratories Serial growth ultrasounds |
VB and anemia PTL Postpartum hemorrhage |
32 wk and 2 d | Urgent classical CD due to funic presentation | 1,500 | None | Nadir by 7 wk PP | None | None |
| 9 | Expectant Serial laboratories Serial growth ultrasounds |
VB PTL HTN Fever and tachycardia (unclear diagnosis) |
28 wk and 3 d | SVD | 350 | None | Nadir by 10 wk PP | None | None |
| 10 | Desired termination | Abnormal TFTs with palpitations (started methimazole) Bilateral ovarian masses (largest 10 × 9 × 8 cm) |
15 wk | D&E | 250 | None | Nadir by 4 wk PP then elevated | Metastatic GTN FIGO stage 3 | IV MTX and leucovorin |
| 11 | Expectant Serial laboratories Serial growth ultrasounds |
VB PTL |
34 wk and 2 d | SVD | 300 | None | Nadir by 6 wk PP | None | None |
| 12 | Desired termination | VB | 16 wk and 6 d | D&E | 250 | None | Nadir by 12 wk PP | None | None |
| 13 | Desired termination | None | 15 wk | D&C | 50 | None | Plateau at 2 wk PP | GTN FIGO stage 1 |
IV MTX |
| 14 | Expectant | Chorioamnionitis Postpartum hemorrhage |
17 wk and 5 d | SVD | 500 | 46 XX | Nadir by 12 wk PP | None | None |
Abbreviations: CD, cesarean delivery; D&C, dilation and curettage; D&E, dilation and evacuation; FIGO, International Federation of Gynecology and Obstetrics; GA, gestational age; GTD, gestational trophoblastic disease; GTN, gestational hypertension; HELLP, hemolysis, elevated liver enzymes, low platelets; HTN, hypertension; IV, intravenous; MTX, methotrexate; PTL, preterm labor; PP, postpartum; SAB, spontaneous abortion; SVD, spontaneous vaginal delivery; VB, vaginal bleeding; PRBC, packed red blood cells.
The majority of patients opted for expectant management (64.3%, n = 9), and the average GA at delivery was 28 weeks and 3 days (16 weeks and 6 days to 34 weeks and 5 days). One patient developed an early-onset HELLP-like syndrome at 16 weeks and 6 days, which prompted treatment with dilation and evacuation (D&E). Another patient experienced persistent vaginal bleeding throughout the pregnancy, resulting in preterm labor and vaginal delivery at 20 weeks and 2 days. A third patient developed hemorrhage and chorioamnionitis and was delivered at 17 weeks and 5 days. Two patients who opted for expectant management also had postpartum hemorrhage, with one of these requiring a hysterectomy due to bleeding after emergent delivery at 24 weeks and 5 days. She subsequently required treatment for metastatic GTN ( Table 3 ).
None of the patients who opted for termination of pregnancy had complications from the procedure, including hemorrhage ( Table 3 ). One of the patients who underwent termination of pregnancy developed pulmonary edema at 20 weeks 0 days at the time of diagnosis.
GTN was diagnosed in 28.6% of patients ( n = 4), with two (2/8; 25%) from the expectant management group and two (2/5; 40%) from the termination group. The two cases of GTN from the termination group were International Federation of Gynecology and Obstetrics (FIGO) stages 1 and 3, while the two cases from the expectant management were FIGO stages 3 and 4. All were treated with intravenous (IV) methotrexate. One patient also received leucovorin, and the patient with FIGO stage 4 disease also received IV dactinomycin. Two of these patients were also noted to have a nadir in their hCG levels by day 56 postdelivery/evacuation.
Discussion
Supplementary Video S1 Dizygotic pregnancy with complete hydatidiform molar tissue and a viable fetus with normal placental tissue.
In this series, we analyzed the patient characteristics, diagnosis, pregnancy complications, and resultant obstetric outcomes of 14 pregnancies complicated by CHMCF. Complete hydatidiform moles (CHM) are generally homozygous 46, XX and result from duplication of the haploid genome of a single sperm following fertilization of an ovum in which the maternal chromosomes are lost during meiosis, or due to postzygotic diploidization in a triploid conception. 5 A multizygotic pregnancy consisting of a partial or complete HMCF is a rare complication of pregnancy, and the available cases series to date focus on GTN risk, instead of obstetrical risk. 6 A multicystic placental mass on ultrasound imaging is typically seen in the first trimester ( Figs. 1 2 3 4 and Supplementary Video S1 ) and should trigger a referral to a maternal-fetal medicine specialist for further imaging and potential diagnostic testing. With improved ultrasound technology and rising rates of ART, 7 HMCF diagnoses may be made earlier and more frequently, highlighting the importance of data accrual on the course and outcome of these pregnancies. 8
Fig. 1.
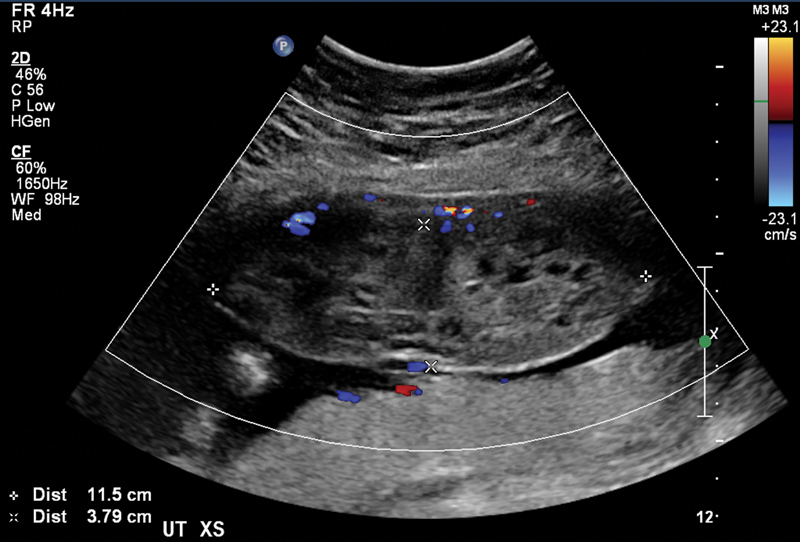
Dizygotic pregnancy with large complete hydatidiform molar tissue and normal placenta.
Fig. 2.
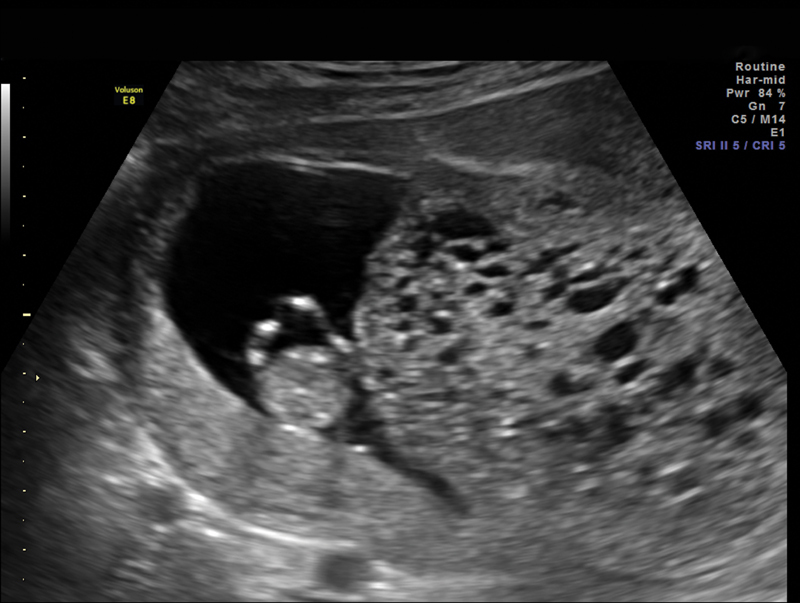
Dizygotic pregnancy at 11 weeks and 4 days with complete hydatidiform molar tissue and viable fetus.
Fig. 3.
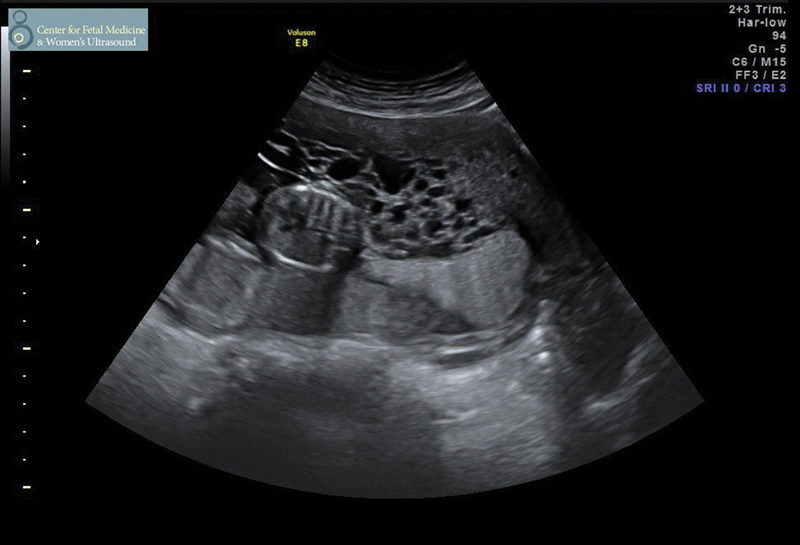
Dizygotic pregnancy with complete hydatidiform molar tissue and abutting normal placenta from a viable fetus.
Fig. 4.
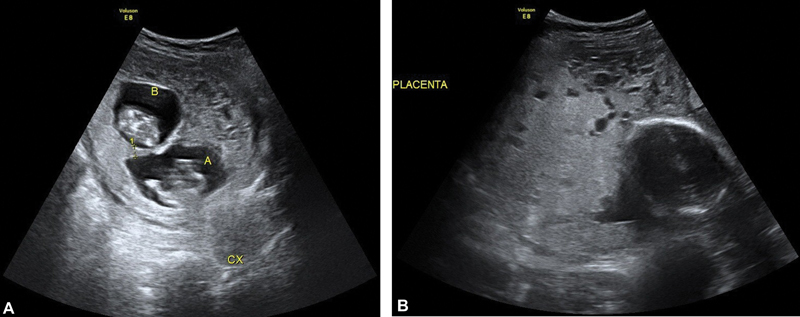
Trizygotic pregnancy at ( A ) 11 weeks and 5 days with complete hydatidiform molar tissue and at ( B ) 24 weeks with the head of twin B and complete hydatidiform molar tissue.
The differential diagnosis of a multicystic placenta with a coexisting fetus can be broad, as a multicystic placenta can represent a hydropic abortus, chromosomal abnormalities, digynic triploid conceptions, PMD, or a molar pregnancy. These distinct diagnoses have varying complications, potential outcomes, and management strategies. The ability to differentiate between these diagnoses is key for optimal counseling and management. Pregnancies with these sonographic findings should be evaluated by and co-managed with a maternal-fetal medicine subspecialist. Maternal serum α-fetoprotein (MSAFP) measurements and β hCG measurements are helpful in confirming the diagnosis. The levels in our case series are comparable to previous case series with β hCG levels greater than 150,000 mIU/mL. Previous cases series have suggested a plateau of β hCG levels in the second trimester and that a failure to reach a plateau was associated with increased risk of adverse pregnancy outcomes. 7
Ultrasound, β hCG, and MSAFP may not provide sufficient data to differentiate between possible diagnoses; thus, invasive diagnostic testing may be necessary for genetic analysis. Amniocentesis can be utilized to evaluate for a triploidy in the coexistent fetus or the placenta as this would be suggestive of a partial hydatidiform mole. Previous literature has suggested CVS of the suspected molar tissue as an alternative via molecular genotyping and segregation analysis of paternal and placental alleles, as absence of maternal alleles can confirm a diandrogenic complete mole. 9 10 11 Our series is the first to report common use of invasive testing in CHMCF, and to show that 40% of invasive procedures may yield no growth or insufficient sample in these cases. Preprocedural counseling regarding invasive testing should include this potential outcome of testing.
Furthermore, CHM is well recognized to have the potential for local invasion and distant spread. It has also been suggested that persistent trophoblastic disease and metastatic GTN are more pervasive with a multifetal gestation with concurrent mole, up to 30% increased risk. 12 β hCG and molar volumes have been used to predict malignant potential, although this is an area where more research is needed. 12
The presence of a CHMCF creates complications for both the mother and the fetus with the clinical course frequently complicated by vaginal bleeding, preeclampsia, hyperemesis gravidarum, hyperthyroidism, and gestational trophoblastic disease. 10 Our case series describes the complication rates in a modern cohort, particularly highlighting the significance of morbid vaginal bleeding and hypertensive disorders of pregnancy in these women. A recent systemic review reported similar findings of a high rate of perinatal morbidities. 13
Including the cases reported in this series, 16 reports of trizygotic pregnancy with two coexisting fetuses and a complete mole have been reported ( Table 4 ). 5 9 10 14 15 16 Of the 16 cases, 87.5% have been pregnancies conceived with ovulation induction medications. The clinical course of these pregnancies shows that vaginal bleeding is very common, presenting in 59% of the cases reported to date.
Table 4. Cases of trizygotic pregnancies consisting of complete mole and two co-existing twins.
| Study | Age (y) | Conception | GA at delivery (wk) | Maternal complications | Pregnancy outcome | GTD | Postpartum therapy | Confirmation of diagnosis |
|---|---|---|---|---|---|---|---|---|
| Sauerbrei et al 14 | 23 | Clomiphene | 22 | VB, PEC with severe features at 22 wk | Spontaneous abortion | No | MTX, ActD (5 cycles) | Postpartum by placental pathology and elevated hCG |
| Ohmichi et al 15 | 34 | hMG-hCG | 17 | VB | Spontaneous abortion | PTT | N/A | Postpartum by placental pathology and elevated hCG |
| Azuma et al 16 | 24 | hMG-hCG | 19 | VB | Spontaneous abortion | No | N/A | Postpartum by placental pathology |
| van de Geijn et al 24 | 31 | GIFT | 24 | VB | PTL, SVD, neonatal deaths of both twins | No | N/A | Antepartum US findings and elevated hCG Confirmed postpartum |
| Shahabi et al 25 | 25 | Clomiphene | 17 | Hyperthyroidism, hyperemesis | Induced abortion due to hyperemesis | Choriocarcinoma, pulmonary metastasis | MTX (2 cycles) | Antepartum US findings and elevated hCG Confirmed postpartum |
| Shozu et al 26 | 31 | IVF-ET | 15 | VB | Induced abortion due to VB | Invasive mole | MTX, ActD (6 cycles) | Postpartum by pathology and DNA polymorphisms in placental tissue |
| Higashino et al 27 | 23 | Clomiphene, hFSH-hCG | 22 | Hyperthyroidism, PEC with severe features, pulmonary edema | Induced abortion due to maternal status | Invasive mole | MTX (7 cycles), etoposide (2 cycles) | Antepartum US findings and elevated hCG Confirmed postpartum |
| Gray-Henry et al 28 | 31 | Metrodin, hCG | 16 | Massive VB | Induced abortion due to life-threatening hemorrhage | No | N/A | Antepartum US findings and elevated hCG Confirmed postpartum |
| Amr et al 29 | 31 | Clomiphene, hCG | 30 | None | PTL, SVD, neonatal death of 1 twin | No | N/A | Postpartum by placental pathology and elevated hCG |
| Rajesh et al 11 | 29 | Spontaneous | 24 | VB | PTL, SVD, neonatal death of both twins | No | N/A | Antepartum US findings and elevated hCG Confirmed postpartum |
| Malhotra et al 12 | 29 | Spontaneous | 21 | VB | Spontaneous abortion | No | N/A | Antepartum US findings and elevated hCG Confirmed postpartum |
| Takagi et al 30 | 37 | hMG, hCG | 28 | Cerclage placed | PTL, CD for malpresentation, survival of both twins | Invasive mole, pulmonary metastases | MTX (6 cycles) | Antepartum US findings and elevated hCG Confirmed postpartum |
| Bovicelli et al 8 | 32 | ICSI | 31 | VB | Emergency CD for nonreassuring fetal testing, IUFD of one twin (fetomaternal hemorrhage) | No | N/A | Antepartum US findings and elevated hCG CVS c/w all paternal genotype Confirmed postpartum |
| Steigrad et al 31 | 40 | IVF | 34 | VB | CD due to VB, survival of both twins | Metastatic GTN, pulmonary metastases | MTX, FA (3 cycles) | Antepartum US findings and elevated hCG Confirmed postpartum |
| Ko et al 32 | 36 | IVF-ET, donor embryo | 33 | PEC with severe features | CD due to PEC, survival of both twins | No | N/A | Antepartum US findings and elevated hCG Confirmed postpartum |
| This study | 30 | GnTp, IUI | 16 | HELLP | SAB of twin B, then induced abortion of twin A due to maternal status | Metastatic GTN, pulmonary metastases | MTX | Antepartum US findings and elevated hCG Confirmed postpartum (twin A unremarkable, twin B partial mole) |
Abbreviations: ActD, actinomycin D; CD, cesarean delivery; EMA-CO, etoposide, methotrexate, actinomycin D, cyclophosphamide, vincristine; ET, embryo transfer; FA, folinic acid; GA, gestational age; GIFT, gamete intrafallopian transfer; GTD, gestational trophoblastic disease; GTN, gestational trophoblastic neoplasia; hCG, human chorionic gonadotropin; HEELP, hemolysis elevated liver enzymes low platelets syndrome; hFSH, human follicle stimulating hormone; hMG, human menopausal gonadotropin; ICSI, intracytoplasmic spermatic injection; IUFD, intrauterine fetal demise; IUI, intrauterine injection; IVF, in vitro fertilization; MTX, methotrexate; PEC, preeclampsia; PT, preterm; PTL, preterm labor; SAB, spontaneous abortion; SVD, spontaneous vaginal delivery; VB, vaginal bleeding.
The risk of GTN is higher in the presence of a complete mole compared with a partial mole (14–20% compared with 1–5%). 5 GTN can include invasive mole, choriocarcinoma, placental site trophoblastic tumor, and epithelioid trophoblastic tumor. The series reported here suggests that the incidence of GTN may be higher in CHMCF than in other molar pregnancies, with 28.6% of patients in this series having GTN. Although the group who opted for termination had a high percentage of GTN, the FIGO stages appeared to be lower. This highlights the importance of counseling regarding the risk of distant metastatic disease with expectant management and need for close patient follow-up postdelivery of patients with CHMCF.
A recent meta-analysis by Albright et al states that the risk of GTN in patients with normalization of β HCG by day 56, or after 8 weeks, is 0.35% for complete mole and 0.03% for partial mole. 17 This is in contrast to our series, where 50% of CHMCF patients who developed GTN had a nadir of β hCG by day 56. More studies and collaborative efforts are warranted to further evaluate the possibility of additional risk of GTN. It is well known that CHMCF carries a much greater risk of pregnancy complication if expectant management is performed, with increased risk of vaginal bleeding, preeclampsia, and preterm labor, but the increased risk of CHMCF may also carry a significantly increased risk of GTN, and this may indicate a longer period of serial β hCG measurements and surveillance and should prompt extensive patient counseling. 1 3 4
One of the greatest strengths of our study is that this is the largest series to date for obstetric data in CHMCF and includes a wide geographic region. Additionally, the use of social media to engage physicians from across the country is a novel approach to transmural collaborations, instead of individual reports of complex cases. Once connected, the physicians were able to use a standardized collection of data across institutions, giving more uniformity to the data for comparison. Although our study has many strengths, it is limited by the potential of selection bias, and given its retrospective recall of cases, the worst cases with the poorest outcomes could have been collected and reviewed. Furthermore, the observational nature of the study cannot truly compare the management protocols, as is often the case with rare disorders.
Conclusion
Overall, our findings demonstrate that it is possible to manage CHMCF expectantly but requires shared decision-making while factoring in maternal antepartum and peripartum risks, as well as increased risk of subsequent metastatic GTN. This case series can serve as a tool for engaging in full counseling of patients about the varied and potentially significant outcomes of CHMCF gestations, which are likely to be on the rise with the increasing use of ART.
Additionally, it is also important to consider innovative methods of extramural collaboration to amplify data accrual for rare disorders, such as CHMCF. This case series demonstrates a novel collaboration, as the idea was initiated in a private social media group of physicians and resulted in a wide collaborative effort from institutions across the United States. These same methods can be used with other rare complications to expand our knowledge base and lead to more meaningful observations from which to draw conclusions.
Footnotes
Conflict of Interest None declared.
References
- 1.Sebire N J, Foskett M, Paradinas F J.Outcome of twin pregnancies with complete hydatidiform mole and healthy co-twin Lancet 2002359(9324):2165–2166. [DOI] [PubMed] [Google Scholar]
- 2.Steller M A, Genest D R, Bernstein M R, Lage J M, Goldstein D P, Berkowitz R S. Natural history of twin pregnancy with complete hydatidiform mole and coexisting fetus. Obstet Gynecol. 1994;83(01):35–42. [PubMed] [Google Scholar]
- 3.Lin L H, Maestá I, Braga A. Multiple pregnancies with complete mole and coexisting normal fetus in North and South America: a retrospective multicenter cohort and literature review. Gynecol Oncol. 2017;145(01):88–95. doi: 10.1016/j.ygyno.2017.01.021. [DOI] [PubMed] [Google Scholar]
- 4.Matsui H, Sekiya S, Hando T, Wake N, Tomoda Y. Hydatidiform mole coexistent with a twin live fetus: a national collaborative study in Japan. Hum Reprod. 2000;15(03):608–611. doi: 10.1093/humrep/15.3.608. [DOI] [PubMed] [Google Scholar]
- 5.Vassilakos P, Riotton G, Kajii T. Hydatidiform mole: two entities. A morphologic and cytogenetic study with some clinical consideration. Am J Obstet Gynecol. 1977;127(02):167–170. doi: 10.1016/s0002-9378(16)33244-6. [DOI] [PubMed] [Google Scholar]
- 6.Fishman D A, Padilla L A, Keh P, Cohen L, Frederiksen M, Lurain J R. Management of twin pregnancies consisting of a complete hydatidiform mole and normal fetus. Obstet Gynecol. 1998;91(04):546–550. doi: 10.1016/s0029-7844(97)00720-5. [DOI] [PubMed] [Google Scholar]
- 7.Lee S W, Kim M Y, Chung J H, Yang J H, Lee Y H, Chun Y K. Clinical findings of multiple pregnancy with a complete hydatidiform mole and coexisting fetus. J Ultrasound Med. 2010;29(02):271–280. doi: 10.7863/jum.2010.29.2.271. [DOI] [PubMed] [Google Scholar]
- 8.Bovicelli L, Ghi T, Pilu G. Prenatal diagnosis of a complete mole coexisting with a dichorionic twin pregnancy: case report. Hum Reprod. 2004;19(05):1231–1234. doi: 10.1093/humrep/deh211. [DOI] [PubMed] [Google Scholar]
- 9.Wax J R, Pinette M G, Chard R, Blackstone J, Cartin A. Prenatal diagnosis by DNA polymorphism analysis of complete mole with coexisting twin. Am J Obstet Gynecol. 2003;188(04):1105–1106. doi: 10.1067/mob.2003.151. [DOI] [PubMed] [Google Scholar]
- 10.Vejerslev L O. Clinical management and diagnostic possibilities in hydatidiform mole with coexistent fetus. Obstet Gynecol Surv. 1991;46(09):577–588. doi: 10.1097/00006254-199109000-00001. [DOI] [PubMed] [Google Scholar]
- 11.Rajesh U, Cohn M R, Foskett M A, Fisher R A, el Zaki D. Triplet pregnancy with a coexisting complete hydatidiform mole of monospermic origin in a spontaneous conception. BJOG. 2000;107(11):1439–1442. doi: 10.1111/j.1471-0528.2000.tb11663.x. [DOI] [PubMed] [Google Scholar]
- 12.Malhotra N, Deka D, Takkar D, Kochar S, Goel S, Sharma M C. Hydatiform mole with coexisting live fetus in dichorionic twin gestation. Eur J Obstet Gynecol Reprod Biol. 2001;94(02):301–303. doi: 10.1016/s0301-2115(00)00338-9. [DOI] [PubMed] [Google Scholar]
- 13.Zilberman Sharon N, Maymon R, Melcer Y, Jauniaux E. Obstetric outcomes of twin pregnancies presenting with a complete hydatidiform mole and coexistent normal fetus: a systematic review and meta-analysis. BJOG. 2020;127(12):1450–1457. doi: 10.1111/1471-0528.16283. [DOI] [PubMed] [Google Scholar]
- 14.Sauerbrei E E, Salem S, Fayle B. Coexistent hydatidiform mole and live fetus in the second trimester: an ultrasound study. Radiology. 1980;135(02):415–417. doi: 10.1148/radiology.135.2.7367637. [DOI] [PubMed] [Google Scholar]
- 15.Ohmichi M, Tasaka K, Suehara N, Miyake A, Tanizawa O. Hydatidiform mole in a triplet pregnancy following gonadotropin therapy. Acta Obstet Gynecol Scand. 1986;65(05):523–524. doi: 10.3109/00016348609157401. [DOI] [PubMed] [Google Scholar]
- 16.Azuma C, Saji F, Takemura M. Triplet pregnancy involving complete hydatidiform mole and two fetuses: genetic analysis by deoxyribonucleic acid fingerprint. Am J Obstet Gynecol. 1992;166(02):664–667. doi: 10.1016/0002-9378(92)91694-6. [DOI] [PubMed] [Google Scholar]
- 17.Albright B B, Shorter J M, Mastroyannis S A, Ko E M, Schreiber C A, Sonalkar S. Gestational trophoblastic neoplasia after human chorionic gonadotropin normalization following molar pregnancy: a systematic review and meta-analysis. Obstet Gynecol. 2020;135(01):12–23. doi: 10.1097/AOG.0000000000003566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nayeri U A, West A B, Grossetta Nardini H K, Copel J A, Sfakianaki A K. Systematic review of sonographic findings of placental mesenchymal dysplasia and subsequent pregnancy outcome. Ultrasound Obstet Gynecol. 2013;41(04):366–374. doi: 10.1002/uog.12359. [DOI] [PubMed] [Google Scholar]
- 19.Jauniaux E, Brown R, Snijders R J, Noble P, Nicolaides K H. Early prenatal diagnosis of triploidy. Am J Obstet Gynecol. 1997;176(03):550–554. doi: 10.1016/s0002-9378(97)70546-5. [DOI] [PubMed] [Google Scholar]
- 20.Cohen M C, Roper E C, Sebire N J, Stanek J, Anumba D O. Placental mesenchymal dysplasia associated with fetal aneuploidy. Prenat Diagn. 2005;25(03):187–192. doi: 10.1002/pd.1103. [DOI] [PubMed] [Google Scholar]
- 21.Szulman A E, Surti U. The syndromes of hydatidiform mole. I. Cytogenetic and morphologic correlations. Am J Obstet Gynecol. 1978;131(06):665–671. doi: 10.1016/0002-9378(78)90829-3. [DOI] [PubMed] [Google Scholar]
- 22.Kajii T, Ohama K.Androgenetic origin of hydatidiform mole Nature 1977268(5621):633–634. [DOI] [PubMed] [Google Scholar]
- 23.Ohama K, Kajii T, Okamoto E.Dispermic origin of XY hydatidiform moles Nature 1981292(5823):551–552. [DOI] [PubMed] [Google Scholar]
- 24.van de Geijn E J, Yedema C A, Hemrika D J, Schutte M F, ten Velden J J. Hydatidiform mole with coexisting twin pregnancy after gamete intra-fallopian transfer. Hum Reprod. 1992;7(04):568–572. doi: 10.1093/oxfordjournals.humrep.a137692. [DOI] [PubMed] [Google Scholar]
- 25.Shahabi S, Naome G, Cobin L. Complete hydatidiform mole and coexisting normal fetuses. A report of two cases with contrasting outcomes. J Reprod Med. 1997;42(11):756–760. [PubMed] [Google Scholar]
- 26.Shozu M, Akimoto K, Kasai T, Inoue M, Michikura Y. Hydatidiform moles associated with multiple gestations after assisted reproduction: diagnosis by analysis of DNA fingerprint. Mol Hum Reprod. 1998;4(09):877–880. doi: 10.1093/molehr/4.9.877. [DOI] [PubMed] [Google Scholar]
- 27.Higashino M, Harada N, Hataya I, Nishimura N, Kato M, Niikawa N. Trizygotic pregnancy consisting of two fetuses and a complete hydatidiform mole with dispermic androgenesis. Am J Med Genet. 1999;82(01):67–69. doi: 10.1002/(sici)1096-8628(19990101)82:1<67::aid-ajmg13>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 28.Gray-Henry D M, Ravindranath N T, Adeghe J H. Triplet pregnancy with complete hydatidiform mole coexisting with two fetuses. J Obstet Gynaecol. 1999;19(01):80–81. doi: 10.1080/01443619966074. [DOI] [PubMed] [Google Scholar]
- 29.Amr M F, Fisher R A, Foskett M A, Paradinas F J. Triplet pregnancy with hydatidiform mole. Int J Gynecol Cancer. 2000;10(01):76–81. doi: 10.1046/j.1525-1438.2000.99064.x. [DOI] [PubMed] [Google Scholar]
- 30.Takagi K, Unno N, Hyodo H E. Complete hydatidiform mole in a triplet pregnancy coexisting two viable fetuses: case report and review of the literature. J Obstet Gynaecol Res. 2003;29(05):330–338. doi: 10.1046/j.1341-8076.2003.00124.x. [DOI] [PubMed] [Google Scholar]
- 31.Steigrad S J, Robertson G, Kaye A L. Serial hCG and ultrasound measurements for predicting malignant potential in multiple pregnancies associated with complete hydatidiform mole: a report of 2 cases. J Reprod Med. 2004;49(07):554–558. [PubMed] [Google Scholar]
- 32.Ko P C, Peng H H, Soong Y K, Chang S D. Triplet pregnancy complicated with one hydatidiform mole and preeclampsia in a 46,XY female with gonadal dysgenesis. Taiwan J Obstet Gynecol. 2007;46(03):276–280. doi: 10.1016/S1028-4559(08)60034-0. [DOI] [PubMed] [Google Scholar]


