Highlights
-
•
Residual torque enhancement (RTE) is present in voluntary and electrically evoked contractions but differs across muscle groups.
-
•
Greater RTE is observed in electrically evoked compared to voluntary contractions.
-
•
In contrast to in vitro preparations, there is no obvious relationship between RTE and the magnitude of muscle stretching.
-
•
Procedures used to investigate RTE vary considerably, and much of the RTE literature seems to contain great risks of bias.
-
•
There are no studies on RTE for human hip and shoulder muscles.
Keywords: Cross-bridge theory, Eccentric contraction, Force-length relationship, Isometric force, Muscle properties
Abstract
Objective
We reviewed and appraised the existing evidence of in vivo manifestations of residual force enhancement in human skeletal muscles and assessed, through a meta-analysis, the effect of an immediate history of eccentric contraction on the subsequent torque capacity of voluntary and electrically evoked muscle contractions.
Methods
Our search was conducted from database inception to May 2020. Descriptive information was extracted from, and quality was assessed for, 45 studies. Meta-analyses and metaregressions were used to analyze residual torque enhancement and its dependence on the angular amplitude of the preceding eccentric contraction.
Results
Procedures varied across studies with regards to muscle group tested, angular stretch amplitude, randomization of contractions, time window analyzed, and verbal command. Torque capacity in isometric (constant muscle tendon unit length and joint angle) contractions preceded by an eccentric contraction was typically greater compared to purely isometric contractions, and this effect was greater for electrically evoked muscle contractions than voluntary contractions. Residual torque enhancement differed across muscle groups for the voluntary contractions, with a significant enhancement in torque observed for the adductor pollicis, ankle dorsiflexors, ankle plantar flexors, and knee extensors, but not for the elbow and knee flexors. Meta-regressions revealed that the angular amplitude of the eccentric contraction (normalized to the respective joint's full range of motion) was not associated with the residual torque enhancement observed.
Conclusion
There is evidence of residual torque enhancement for most, but not all, muscle groups, and residual torque enhancement is greater for electrically evoked than for voluntary contractions. Contrary to our hypothesis, and contrary to generally accepted findings on isolated muscle preparations, residual torque enhancement in voluntary and electrically evoked contractions does not seem to depend on the angular amplitude of the preceding eccentric contraction.
Graphical abstract
1. Introduction
The force capacity of a muscle is greater and more efficiently delivered (less energy required per unit force) during an eccentric contraction than during an isometric or concentric contraction.1, 2, 3, 4 Force enhancement is observed during active lengthening, and a “residual” part of this enhancement in force persists after the active stretch.5,6 This history-dependent property is called residual force enhancement. It is typically measured as the increase in isometric force capacity that occurs after an active stretch, as compared to a purely isometric contraction at the same muscle fiber length and activation level.7,8
Residual force enhancement has been observed across muscle structural levels, from isolated sarcomeres9, 10, 11 to muscles,7,8,12 and is thought to influence muscle contraction dynamics for a period as long as 20 s. Current research on isolated muscle preparations has been conducted in order to elucidate the mechanisms responsible for residual force enhancement. Currently, the main hypothesis is that titin engages during active muscle stretching and then persists after a stretch in the active and passive muscle.12,13
Possible roles for residual force enhancement in in vivo contractions in humans have been explored.12,14, 15, 16, 17 The most common approach used in human studies is testing whether the isometric steady state (net) torque capacity of a muscle group is greater after an active muscle-tendon unit stretch (i.e., eccentric contraction) in comparison to a reference (i.e., purely isometric contraction) at the same joint angle/muscle–tendon unit length. Residual torque enhancement (RTE) may alter the shape of the torque–angle relationship, and might be relevant in the context of aging, training adaptations, fatigue and injury prevention.14, 15, 16, 17, 18, 19, 20 However, the presence and magnitude of RTE seem to vary considerably across studies in humans, and so do the specific procedures used to evaluate it.
Except for the isolated sarcomere preparation—for which only 1 stretch amplitude has been tested9,10—a dependence of residual force enhancement on stretch amplitude has been observed in isolated muscle preparations at various structural levels.7,11,21 In human in vivo contractions, however, the dependence of RTE on the amplitude of muscle stretch is less clear.8,12,15 There is a decoupling between muscle–tendon unit and fascicle/fiber contractile kinematics,22 which may hinder the association between the angular amplitude of the eccentric contraction and the resulting RTE.23, 24, 25 An increase in joint angular amplitude during an eccentric contraction may not be accompanied by a proportional change in fascicle length change. Additionally it has been shown that the effect of angular stretch amplitude on RTE may differ between voluntary and involuntary, electrically evoked contractions.6
Given the increasing number of studies in humans, summarizing the evidence available with regards to the presence of RTE in voluntary and involuntary contractions is instrumental to future studies in this field and towards an understanding of the overall effect of residual force enhancement in torque capacity across studies. RTE is a topic of increasing interest, and from the date the first systematic review in the area was conducted,8 a large number of studies in humans were published. Additionally, the overall effect of RTE has not been quantitatively investigated through a meta-analysis of the available data in the literature.
In this review and meta-analysis, we performed a study to identify and appraise the existing evidence regarding the presence of RTE in humans. This aim was met via the following objectives: (1) to describe the main outcomes and procedures used to investigate residual force enhancement in human voluntary and electrically evoked contractions; (2) to appraise the existing evidence with respect to potential risk of bias; (3) to assess the effect of active stretch (eccentric contractions) on torque capacity (i.e., RTE) in voluntary and involuntary contractions; and (4) to evaluate the dependence of this effect on the angular amplitude of the eccentric contraction and on the muscle group tested. We hypothesized that an overall positive effect of residual force enhancement on isometric torque generating capacity is observed for in vivo contractions in humans,26 and that this effect depends on the angular amplitude of the eccentric contraction7,11,21 and is greater for electrically evoked contractions than for voluntary contractions.6
Throughout this article, we use the term “isometric” to refer to contractions aimed at a constant muscle–tendon unit length/joint angle and the term “active stretch/eccentric contraction” to refer to an eccentric joint angular movement, in which the target muscle–tendon unit lengthens during contraction. Please note that this definition of isometric contraction, often used in human research, typically allows for some fascicle shortening and angular movement to occur.27, 28, 29, 30
2. Methods
2.1. Protocol and registration
The recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) were followed for this systematic review.31 This study was preregistered in the International Prospective Register of Systematic Reviews (PROSPERO-CRD42020109125).
2.2. Search strategy
We searched the following electronic databases covering the period from inception to May 2020: CINAHL, LILACS, PsycINFO, PubMed, SciELO, Scopus, SPORTDiscus, and Web of Science. The initial search comprised the terms “residual force enhancement” and “force enhancement”. To identify other relevant articles that could meet the criteria, searches were conducted within the reference lists of selected manuscripts. One investigator (DdC) imported the results into Mendeley using the databases search tool and excluded duplicate articles, opinion articles, systematic reviews, narrative reviews, conference abstracts, and case studies. Titles and abstracts of retrieved articles were independently evaluated by 2 investigators (DdC and HdBF) to determine study eligibility. Manuscripts for which abstracts did not provide enough information regarding the inclusion and exclusion criteria were retrieved for full-text evaluation. Disagreements were solved by consensus.
2.3. Eligibility criteria
For the review, we included studies that: (1) were complete; (2) were original; (3) were related to skeletal striated muscle; and (4) investigated residual force enhancement in humans by comparing a purely isometric contraction and an isometric contraction preceded by an active stretch. For the meta-analyses, further inclusion criteria were added: (5) experiments were not torque clamped (i.e., contractions matched in torque through feedback or fixed weights); and (6) mean and standard error (SE) or standard difference (SD) of torque were reported in Newton meters (N·m). Studies that reported an external force, in N, measured with a fixed moment arm across conditions (a proxy for torque) were also included. For studies that met all criteria except Number 6, authors were contacted by email and requested to provide the data.
2.4. Methodological quality assessment
Study quality assessment was independently performed by 2 authors (DdC and HdBF). Disagreements were solved by consensus. The questions deemed relevant for the analysis were determined by the authors through consultation with 2 internationally recognized external experts in the area. The analysis consisted of the evaluation of 16 items related to blinding, description of losses/exclusions, and description of experimental procedures (Supplementary Table 1). Items were answered with “yes”, “no”, or “uncertain” by the authors. The score 0 was assigned to items answered with “no” or “uncertain”, while 1 was given to items answered with “yes”. A “yes” answer was given when the item was met and the information was clearly identified by the authors.
2.5. Data extraction
For the descriptive analysis of studies, we extracted the following information: author/year and study design, population; muscle/muscular group analyzed, muscle excitation method (i.e., voluntary or through electrical stimulation), active stretch features (joint angular amplitude and velocity), and isometric contraction after stretch features (duration of contraction, and joint angle and/or muscle length).
For each condition analyzed in the studies, relative changes in torque and myoelectric activity (electromyography (EMG)) between the purely isometric reference contraction and the isometric contraction preceded by an active stretch were described in terms of percentage change.
2.6. Data analyses
The presence of RTE in humans was first investigated for voluntary and electrically evoked contractions by comparing the torque (N·m) during a purely isometric reference contraction to the isometric contraction torque after the stretch. Hedge's g effect sizes from studies, independent of the tested joint, were therefore combined in 2 distinct meta-analyses (i.e., voluntary contractions or electrically evoked contractions) using a random effects model. Thereafter, studies investigating voluntary contractions were grouped according to the tested muscles, and the random-effects model was used to calculate the standardized mean difference (SMD) for each muscle group. Additionally, subgroups were compared using the fixed effects (plural) model. The Hartung-Knapp-Sidik-Jonkman method was used to estimate the variance of the true effect sizes.32 This estimator (τ2) generally produces more conservative results (i.e., wider confidence intervals), particularly when the number of studies is small and between-study heterogeneity is large.32 For studies reporting different stretching velocity, activation level, or angular stretch amplitudes, data were pooled to generate a single effect size per study with the aim of reducing unit-of-analysis error.33
Heterogeneity was assessed by visual inspection of forest plots and by using χ2 test for heterogeneity (α = 10%). Inconsistency between trials was described using the I2 statistic (Thresholds for the interpretation of the I2 statistic can be misleading, since the importance of inconsistency depends on several factors. A rough guide to interpretation in the context of meta-analyses of randomized trials is as follows: I2 = 0%–40% might not be important; 30%–60% may represent moderate heterogeneity; 50%–90% may represent substantial heterogeneity; and 75%–100%, considerable heterogeneity).34 Small-sample publication bias was assessed by visualizing funnel plots asymmetry and with the Egger's test of the intercept35,36 when including all studies. If asymmetry was confirmed, the trim-and-fill procedure was used to estimate a bias-corrected SMD for the random effects model.37 Egger's test was not conducted for involuntary contractions due to a sample size smaller than 10.36
Meta-regressions were used to determine the effect of angular stretch amplitude (calculated as a percentage of the respective joint's typical full range of motion38) on the SMD in torque between purely isometric reference contractions and the isometric contractions preceded by active stretch.35 Two separate models were run independently for voluntary contractions and electrically evoked contractions; a SMD was considered for each angular stretch amplitude tested in the literature. R Studio (Version 1.3.1056; R Core Team, Vienna, Austria) was used for all analyses using the packages meta,39 metafor,40 dmetar,41 and dplyr and tidyverse.42 We adopted α = 5% for all meta-analyses.
3. Results
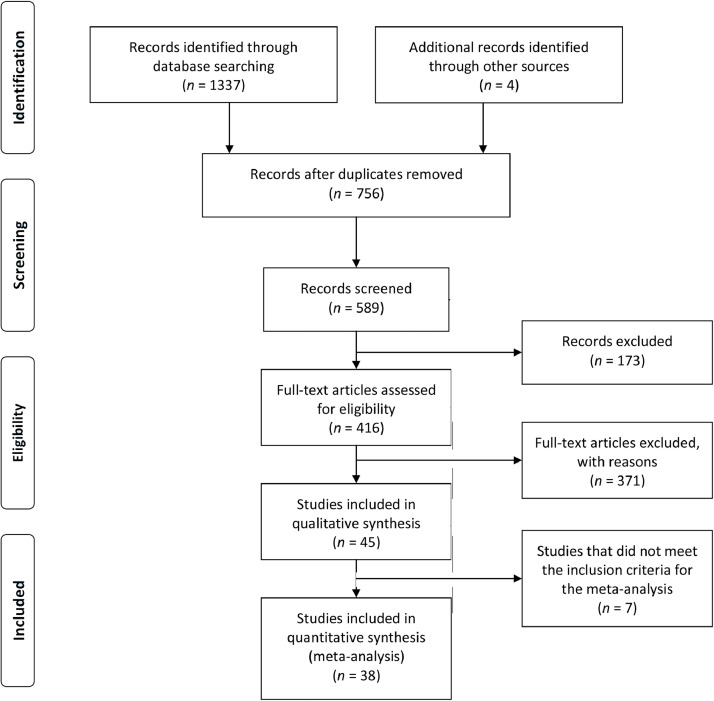
Of the 1341 potentially relevant studies retrieved from electronic databases (CINAHL = 20; LILACS = 0; PubMed = 260; PsycINFO = 11; SciELO = 2; Scopus = 442; SPORTDiscus = 109; and Web of Science = 493) or identified through other sources (1 from reference lists; 3 published after search was ended), 45 original studies met the inclusion criteria. A flow diagram of the search and selection process is shown in Fig. 1. Included studies had a total of 852 participants. Of these, 724 were tested using voluntary contractions, 110 were tested using involuntary contractions elicited by electrical stimulation, and 18 were tested using both voluntary and involuntary contractions. Of the 45 studies, 38 studies were included in the meta-analysis.
Fig. 1.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram of studies search and selection.
Quality assessment showed that procedures varied across studies with regard to randomization of contractions, time window analyzed, standardization of verbal command, and control of confounding variables, such as antagonist coactivation. In Supplementary Table 1, a summary of the quality assessment is shown for all included results. Total study scoring varied 3–11 out of 16 items analyzed. Study blindness, control of fascicle length, and region of the force-length relationship were the items often not accounted for in the literature. Only 3 studies blinded participants to the study hypothesis, and only 1 had the investigator blinded to the hypothesis. Agreement rate between reviewers was κ = 0.96 for quality assessment.
Torque and EMG were the most commonly evaluated outcomes. Selected characteristics of the included studies are described in Supplementary Table 2 along with the observed percentage change in torque and EMG. The majority of studies reported RTE in voluntary contractions (35 of 45), with 2 of them utilizing electrical stimulation in the same study.6,43 Ten studies investigated RTE through electrical stimulation only.44, 45, 46, 47, 48, 49, 50, 51, 52, 53
Eighteen studies analyzed RTE in only maximal contractions,6,15,18,20,23,54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66 and 21 utilized only submaximal contractions.14,17,43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53,67, 68, 69, 70, 71, 72, 73, 74 Six studies reported RTE in both maximal and submaximal levels.16,19,75, 76, 77, 78 RTE has been investigated in the upper limbs for elbow flexors,15,57 for the thumb adductor muscle,6,16,46,47,52,53,70,74,76 and for the first dorsal interosseous muscle.45 Knee extensors have been widely analyzed through monoarticular17,18,23,58,59,65,67,68,71 and multi-joint tasks,14,56,64,73 while only 2 studies assessed the knee flexors.63,78 The ankle joint was tested in order to investigate RTE in dorsiflexors19,20,43,44,51,54,60, 61, 62,66,69,72 and plantar flexors during monoarticular43,48, 49, 50,55,67,75,77 and multi-joint tasks. 14,56,73
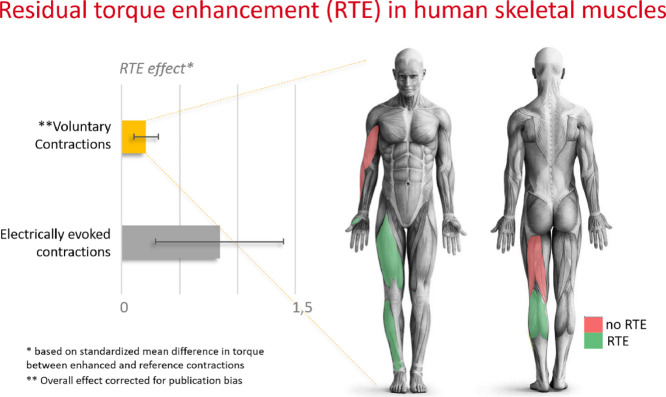
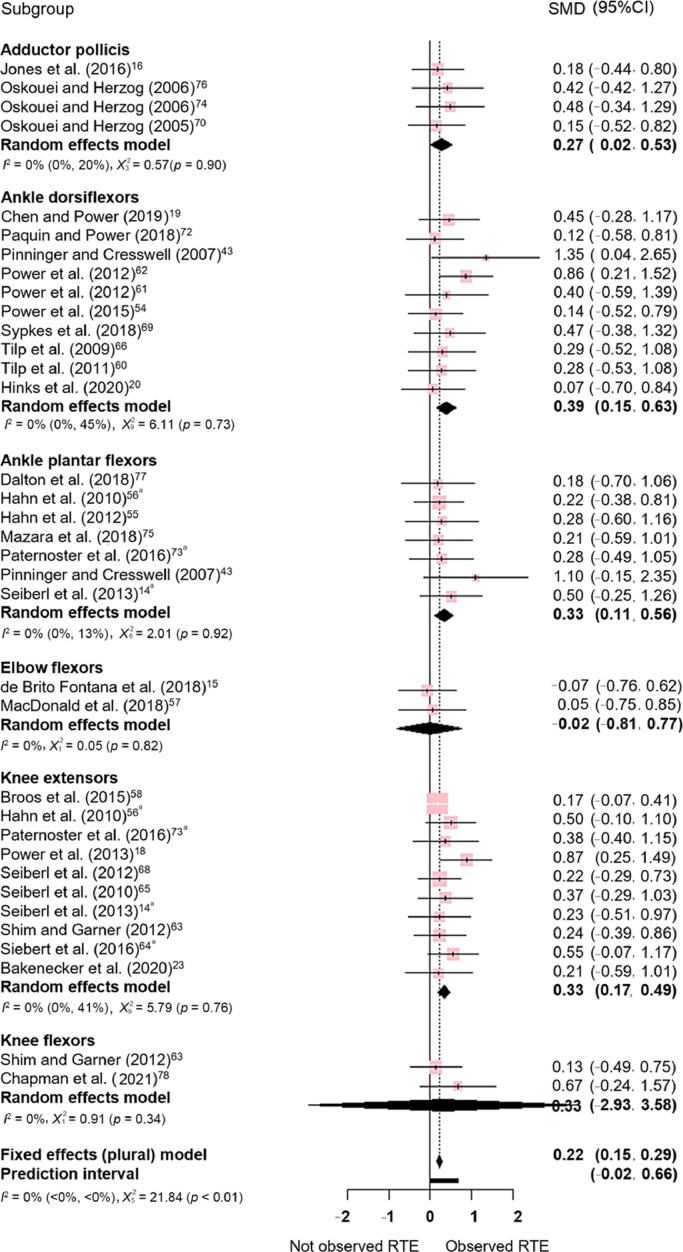
Significant effects favoring torque capacity in isometric contractions preceded by active stretch (compared to purely isometric contractions) were observed for voluntary muscle contraction (SMD = 0.323, 95% confidence interval (95%CI): 0.237–0.410; p < 0.001). For the comparisons within muscle groups tested using voluntary contractions, a significant effect was observed for adductor pollicis (SMD = 0.27, 95%CI: 0.02–0.53), ankle dorsiflexors (SMD = 0.39, 95%CI: 0.15–0.63), ankle plantarflexors (SMD = 0.33, 95%CI: 0.11–0.56), and knee extensors (SMD = 0.33, 95%CI: 0.17–0.49). No effect was observed for elbow flexors (SMD = –0.02, 95%CI: –0.81 to 0.77), or knee flexors (SMD = 0.33, 95%CI: –2.93 to 3.58; Fig. 2).
Fig. 2.
Estimates of differences in torque capacity between purely isometric contractions and isometric contractions preceded by stretching. All comparisons included were for voluntary contractions. Positive values indicate the magnitude of residual torque enhancement (torque is greater in isometric contractions preceded by an active stretch when compared to purely isometric contractions). a indicates the use of multi-joint contractions. 95%CI = 95% confidence interval; RTE = residual torque enhancement; SMD = standardized mean difference.
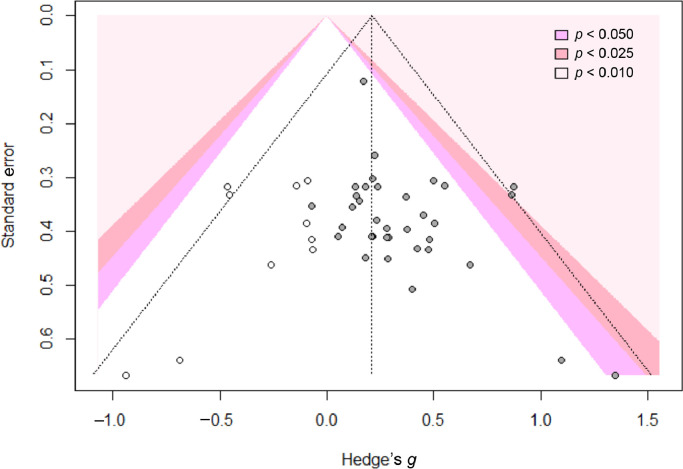
Significant between-group differences were identified (p = 0.006) across muscle groups for the voluntary contractions, with no observed heterogeneity (I2 = 0%; p = 0.992). A significant publication bias was verified for the voluntary contractions (Egger's test of the intercept with p = 0.019). Fig. 3 shows a funnel plot for voluntary contractions. Using the trim-and-fill method, we corrected the random effect model SMD for the voluntary contractions meta-analysis by adding 10 effect sizes (corrected SMD = 0.21, 95%CI: 0.10–0.32; p = 0.003).
Fig. 3.
A significant publication bias was verified for the voluntary contractions (Egger's test of the intercept with p = 0.019). Funnel plots show gray scatter dots, which are studies included in the meta-analysis, and white scatter dots, which are imputed studies mirrored for each trimmed study in order to reach funnel plot symmetry.
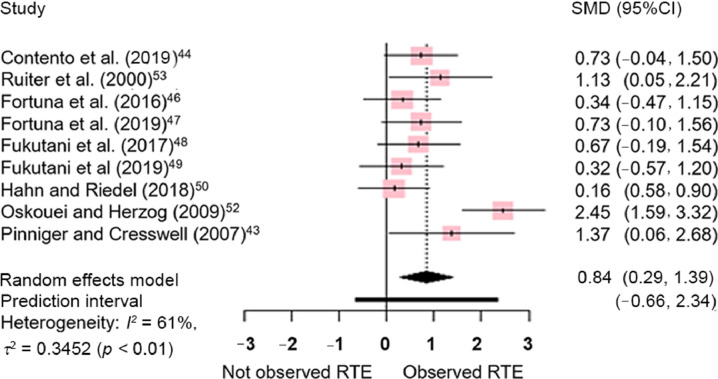
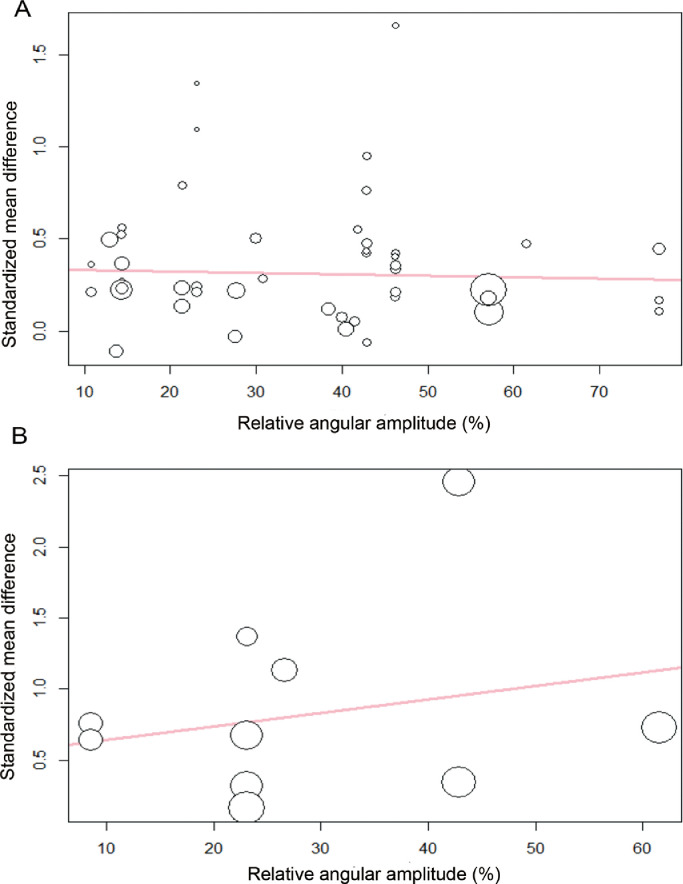
Significant effects favoring torque capacity in isometric contractions preceded by active stretch (compared to purely isometric contractions) were also observed for electrically evoked contractions (SMD = 0.84, 95%CI: 0.29–1.39; p = 0.008; Fig. 4), but with significant heterogeneity (I2 = 61%; p = 0.008). Meta-regressions revealed that the relative angular amplitude of the eccentric contraction was not associated with the magnitude of the effect size for both voluntary (p = 0.752; R2 = 0.00%; Fig. 5A) or electrically evoked contractions (p = 0.529; R2 = 0.00%; Fig. 5B).
Fig. 4.
Estimates of differences in torque produced between purely isometric contractions and isometric contractions preceded by stretching. All comparisons included were for electrically evoked contractions. Positive values indicate the magnitude of residual torque enhancement (torque is greater in isometric contractions preceded by an active stretch when compared to purely isometric contractions). 95%CI = 95% confidence interval; RTE = residual torque enhancement; SMD = standardized mean difference.
Fig. 5.
Bubble plots with fitted meta-regression line indicating the effect of stretching amplitude on the standardized mean difference in torque produced between purely isometric contractions and contractions preceded by stretching, (A) voluntary contractions and (B) electrically evoked contractions. The size of each point is inversely proportional to the variance of the estimated difference.
4. Discussion
The role of residual force enhancement in in vivo contractions has been investigated mostly through the assessment of changes in isometric torque capacity that may result from a previous active stretch (i.e., eccentric contraction) of the muscle–tendon unit. RTE has been evaluated in humans for maximal and submaximal contractions and, in the case of submaximal contractions, a torque-matched, or EMG-matched approach has been used to make contractions comparable. When torque is matched or maximal, a reduction in EMG is often observed (descriptive results, Supplementary Table 2). When EMG is matched or under a maximal voluntary effort, an increase in torque is observed for some muscle groups but not others (Fig. 3, meta-analysis). Interestingly, we found that the overall magnitude of RTE depends on whether the contraction is voluntary or electrically elicited and does not seem to be determined by the relative angular amplitude of the stretch applied.
The results of our meta-analysis suggest RTE is small in voluntary contractions (SMD = 0.21) compared to electrically evoked contractions (SMD = 0.84); and the funnel plot indicates that negative effect sizes in experiments involving voluntary contractions might have been suppressed due to publication bias (Fig. 3) towards positive results. This difference in RTE between voluntary and electrically evoked contractions highlights the dependence of the nervous system modulation of torque production on the history of contraction.44,72
The mechanisms responsible for RTE are thought to emerge during the eccentric contraction. Potential differences between voluntary and electrically evoked eccentric contractions with respect to the activation of mechanoreceptors—such as muscle spindles, Golgi tendon organs, and free nerve endings—and the associated engagement of inhibitory and excitatory circuits might contribute to the different RTE observed between muscle excitation methods.69,79 The specific strategies used by the central nervous system to control isometric contractions after stretch are still controversial and might depend on activation levels (maximal vs. submaximal) or muscle group tested.55,69
RTE in voluntary contractions varies across muscle groups. Knee extensors, ankle plantar flexors, ankle dorsiflexors, and the adductor pollicis were the muscles that showed a clear RTE in our meta-analysis. In contrast, elbow flexors and knee flexors were not affected by the history of contraction. Curiously, despite the functional relevance of hip flexor and extensor muscles, RTE has not been investigated at the hip joint, and no studies regarding the shoulder were found either. Strength assessment in the hip and shoulder may be considered challenging, but with appropriate procedures and familiarization, a good reliability of torque measurements can be achieved.80,81
Despite the overall small RTE observed in voluntary contractions (Fig. 2), a reduction in EMG in the enhanced state was found in most studies investigating voluntary maximal as well as submaximal torque matched contractions (Supplementary Table 2). A shift in the EMG–torque relationship to the right has been described for contractions preceded by an active stretch when compared to the purely isometric reference contractions at the same joint angle.72 This shift leads to a smaller torque/EMG ratio for the enhanced contractions, in comparison to the reference isometric contractions, which might be interpreted in terms of greater neuromuscular economy. For the elbow flexor muscles, which seem to be the muscle group with the smallest effect on torque (Fig. 2), a substantial decrease in EMG (up to 35%) was observed in the enhanced state compared to a reference contraction at the same level of torque.15,72
The mechanisms involved in this economy are not known, but Joumaa and Herzog,82 showed in isolated skinned fiber experiments that the metabolic cost associated with a given level of force production is reduced in isometric contractions when preceded by an active stretch. However, the relevance of such an economic state to the function in vivo is not clear. Seiberl et al17 found that during a long lasting (60 s), fatiguing, purely isometric contraction, a reduction in quadriceps activation occurred when the contraction was preceded by an active stretch. However, this reduction was not observed for all individual quadriceps muscles, lasted about 40 s, and did not influence changes in maximal torque capacity or percentage voluntary activation that occurred with the fatiguing protocol. Additionally, in a study by Paternoster et al,67 no evidence of a reduction in the medial gastrocnemius muscle oxygen consumption (estimated via near-infrared spectroscopy) was observed for submaximal torque matched plantar flexion contractions preceded by an active stretch (compared to purely isometric contractions), even when only “responders” (individuals with activation reduction) were tested in a subgroup analysis.
Our results do not show a dependence of RTE on the angular amplitude of the eccentric contraction. It is generally accepted that residual force enhancement increases with the amplitude of muscle stretching in isolated muscle preparations.5,7 However, it has been shown that force enhancement does not increase unconditionally with increasing stretch magnitude, as is generally accepted, but that increasing force enhancement with stretch depends on which part of the force–length relationship the stretch occurs, favoring the descending limb where a passive force may be present.83,84 Some studies measured the torque–angle relationship.14,15,18,19,23,58 However, the regions of a torque–angle relationship may not necessarily reflect the regions of the force–length curve in which the muscles tested work because changing moment arms have the potential to displace the angle of peak torque occurrence from the angle of peak force occurrence. Two studies experimentally controlled and tested for the different regions of the force–length relationship of the agonist muscle group tested (Supplementary Table 1).15,23 However, it is not possible to state with certainty whether the force–length relationship of the target, EMG, and/or ultrasound-instrumented muscle (biceps brachialis15/vastus lateralis23) was controlled, as the force–length curves may differ between the individual muscles comprising agonist muscle groups.85
Additionally, in voluntary contractions, the interactions between the muscle and the nervous system may influence the relationship between RTE and the angular stretch amplitude. Indeed, the slope of the regression lines in Fig. 5 differ between the voluntary (Fig. 5A) and electrically evoked contractions (Fig. 5B). It is possible that long stretches are accompanied by greater neural inhibition than short stretches, which might partly offset the residual force enhancement dependence on stretch amplitude during voluntary contractions.6 Lee and Herzog6 reported that the effect of angular stretch amplitude on RTE differed between the voluntary and electrically activated adductor pollicis muscle. For the electrically stimulated muscle, they found an increase in RTE along with increasing angular stretch magnitude while, for the voluntary contractions, RTE increased from the smallest to the intermediate angular stretch amplitude, but then decreased from the intermediate to the greatest angular stretch amplitude, leaving the RTE for the shortest and longest stretches the same.
Other sources of variance, such us activation level, muscle groups, and participant age, are present in our regression model, and it is possible that a significant effect of angular stretch amplitude on RTE is only discernible when these factors are controlled. Additionally, due to the decoupling between changes in joint angle and changes in fascicle length, an increase in the angular stretch amplitude might not result in a proportional increase in fascicle/fiber stretch.22 The studies that investigated fascicle length during eccentric contractions23,49,55,60 confirm that fascicles were significantly stretched; however, the relationship between different angular stretch amplitudes and the amount of fascicle stretch has not been systematically evaluated in the context of RTE. Since protocols for the enhanced and reference contractions are based on joint angle control while the fascicles are free to deform upon contraction, shortening of fascicles due to the elongation of the tendon for increasing levels of force might prevent the dependence of residual force enhancement on stretch amplitude from being reflected at the joint level.
In this study, contractions at different activation levels and velocities were pooled into a single effect per study/group of participants. Residual force enhancement following stretch is thought to be independent of stretch velocity,86 a result that is also observed when RTE is tested for different eccentric contraction angular velocities during electrically stimulated and voluntarily activated contractions in vivo.6,54,60,66 Only 4 of the 45 studies included in this review (Supplementary Table 2) tested for the effect of speed, and the results are limited to the dorsiflexor and adductor pollicis muscles; there are no results currently available for plantar flexor and knee extensor muscles—muscles for which we observed significant torque enhancement. The relationship between angular velocity and the contractile kinematics of plantar flexor and knee extensor muscles is greatly affected by series elasticity.22,27,87 RTE, therefore, may be affected differently by variations in stretch speed in these muscles when compared to the dorsiflexor and adductor pollicis muscles.
Previous studies investigating the interaction between RTE and activation suggest that the increase in torque relative to the isometric contraction is similar across maximal and submaximal contractions.72,88 When comparing different activation levels during (not after) the stretch, Oskouei and Herzog74 observed that increasing levels of EMG resulted in an increase in the number of subjects who showed RTE: while only a third of participants showed RTE with an eccentric contraction at 10% of maximal contraction, two thirds showed RTE at 60%, and all participants showed RTE at 100%.
Procedures used to verify RTE varied across studies (Supplementary Table 2), and risk of bias seems to be substantial in the literature regarding RTE in humans. Some studies identified in the systematic review did not report the absolute values of torque for the contractions tested and, therefore, were not included in this meta-analysis. The main sources of bias are associated with the lack of randomization of contraction conditions, the use of subgroup analyses without a strong rationale, and the awareness of participants about the study hypothesis.
Given the interindividual variability regarding the occurrence of RTE, previous studies have grouped individuals into “responders” (subjects that show RTE) and “non-responders” (subjects that do not show RTE) and often exclude the non-responders in the calculation of RTE.67,70,72, 73, 74,88 Using different statistical treatment for responders and non-responders requires a strong rationale and a critical analysis explaining the reliability of group assignment. Individuals that do not show RTE should not be immediately treated as non-responders; rather, consideration should be given to whether the individual response is reliable over repeated trials, preferably on different days.70 The test–retest reliability of RTE in voluntary and electrically evoked contraction has not been statistically evaluated.
Randomization in the order of contractions is rare in studies evaluating RTE in humans. In most studies, the reference contraction is performed first and then followed by the enhanced contractions protocol. The justification is usually based on reducing the effect that fatigue could have in favoring the enhanced contraction. Therefore, a bias towards lower RTE is assumed by first performing the isometric reference contraction and following it with the RTE contraction. While this approach may help to increase confidence over a positive effect, it introduces a systematic bias in the literature. This protocol seems to have been “copied” from the original experiments in isolated preparations in which consecutive trials are known to affect force. In humans, previous research suggests that the use of an interval (arguably a minimum of 2–3 min depending on contraction type) is enough to prevent fatigue between maximal contractions.89
Another aspect of these studies that may deserve critical attention is the command given to subjects. Only 13 studies16,23,52,55,56,59,67,69, 70, 71, 72,74,76 of the 45 studies used standardized verbal commands across contractions. It is known that verbal command may affect torque output in maximal voluntary contractions and that an increase in voice volume, for example, may increase force output during a contraction.90, 91, 92 Blinding subjects and evaluators to the hypothesis of the study and using a specific standardized command might help to overcome this limitation of in vivo torque assessment. The fact that RTE was observed in electrically evoked contractions rules out the possibility that differences in torque originate from verbal command.
In this study, we used the term RTE to refer to the increase in torque capacity of isometric contractions preceded by an eccentric contraction when compared to purely isometric contractions at the same activation level and joint angle. Therefore, we define RTE in a descriptive manner, as is standard in the literature. The mechanisms and factors that result in the presence or absence of RTE are not within the scope of this review, and we do not want to imply any mechanistic cause for how RTE is achieved. The term residual force enhancement, on the other hand, was used when referring specifically to the defined muscle contractile property that results in an increase in isometric steady state muscle tension after the muscle is actively elongated.5,7 While RTE was not observed for some muscle groups, it is possible that the fibers composing these muscles do show residual force enhancement. Residual force enhancement is a reproducible finding in isolated preparations, including a study that tested for residual force enhancement in isolated human muscle fibers.26
5. Conclusion
We conclude that an overall small RTE is present in human voluntary contractions and that this effect differs across muscle groups. A large effect was observed for electrically evoked contractions, but comparisons across muscles were not possible due to the limited number of studies. When studies are pooled, including different muscle groups, activation levels, and phenotypes—such as elderly, female and male adults, trained and untrained individuals—no effect of angular stretch amplitude on the magnitude of RTE was observed. Heterogeneity was not significant for voluntary contractions; however, procedures were not standardized across studies, and sources of bias seem to be significant in the literature regarding the role of residual force enhancement in human in vivo contractions. Future studies should focus on different ways to capture the significance of residual force enhancement to human movement; the inclusion of neuromuscular parameters seems to be relevant. Additionally, investigations of muscles around proximal joints in the limbs, such as the hip and the shoulder, are missing.
Acknowledgments
Authors’ contributions
DdC carried out the search, selected the studies, extracted the information needed for the description and meta-analysis of the literature, and wrote the first draft of the manuscript; LBRO and GST conducted the meta-analysis; WH consulted on the questions used in the risk of bias assessment and contributed to the research questions; HdBF supervised the work; served as a second investigator for the analyses performed by the first author, and wrote the first draft of the manuscript. All authors have read and approved the final version of the manuscript, and agree with the order of presentation of the authors.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Peer review under responsibility of Shanghai University of Sport.
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.jshs.2021.05.006.
Appendix. Supplementary materials
References
- 1.Herzog W. Why are muscles strong, and why do they require little energy in eccentric action? J Sport Heal Sci. 2018;7:255–264. doi: 10.1016/j.jshs.2018.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fenn WO. A quantitative comparison between the energy liberated and the work performed by the isolated sartorius muscle of the frog. J Physiol. 1923;58:175–203. doi: 10.1113/jphysiol.1923.sp002115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hill VA. The Heat of shortening and the dynamic constants of muscle. Proc R Soc B Biol Sci. 1938;126:136–195. [Google Scholar]
- 4.Huxley AF. Muscle structure and theories of contraction. Prog Biophys Biophys Chem. 1957;7:255–318. [PubMed] [Google Scholar]
- 5.Abbott BC, Aubert XM. The force exerted by active striated muscle during and after change of length. J Physiol. 1952;117:77–86. [PMC free article] [PubMed] [Google Scholar]
- 6.Lee DH, Herzog W. Force enhancement following muscle stretch of electrically stimulated and voluntarily activated human adductor pollicis. J Physiol. 2002;545:321–330. doi: 10.1113/jphysiol.2002.018010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Edman KA, Elzinga G, Noble MI. Residual force enhancement after stretch of contracting frog single muscle fibers. J Gen Physiol. 1982;80:769–784. doi: 10.1085/jgp.80.5.769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chapman N, Whitting J, Broadbent S, Crowley-McHattan Z, Meir R. Residual force enhancement in humans: A Systematic review. J Appl Biomech. 2018;34:240–248. doi: 10.1123/jab.2017-0234. [DOI] [PubMed] [Google Scholar]
- 9.Leonard TR, DuVall M, Herzog W. Force enhancement following stretch in a single sarcomere. Am J Physiol Cell Physiol. 2010;299:C1398–C1401. doi: 10.1152/ajpcell.00222.2010. [DOI] [PubMed] [Google Scholar]
- 10.Rassier DE, Pavlov I. Force produced by isolated sarcomeres and half-sarcomeres after an imposed stretch. Am J Physiol Cell Physiol. 2012;302:C240–C248. doi: 10.1152/ajpcell.00208.2011. [DOI] [PubMed] [Google Scholar]
- 11.Joumaa V, Leonard TR, Herzog W. Residual force enhancement in myofibrils and sarcomeres. Proceedings Biol Sci. 2008;275:1411–1419. doi: 10.1098/rspb.2008.0142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Seiberl W, Power GA, Hahn D. Residual force enhancement in humans: Current evidence and unresolved issues. J Electromyogr Kinesiol. 2015;25:571–580. doi: 10.1016/j.jelekin.2015.04.011. [DOI] [PubMed] [Google Scholar]
- 13.Herzog W. The role of titin in eccentric muscle contraction. J Exp Biol. 2014;217:2825–2833. doi: 10.1242/jeb.099127. [DOI] [PubMed] [Google Scholar]
- 14.Seiberl W, Paternoster F, Achatz F, Schwirtz A, Hahn D. On the relevance of residual force enhancement for everyday human movement. J Biomech. 2013;46:1996–2001. doi: 10.1016/j.jbiomech.2013.06.014. [DOI] [PubMed] [Google Scholar]
- 15.de Brito Fontana H, de Campos D, Sakugawa RL. Predictors of residual force enhancement in voluntary contractions of elbow flexors. J Sport Heal Sci. 2018;7:318–325. doi: 10.1016/j.jshs.2018.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jones AA, Power GA, Herzog W. History dependence of the electromyogram: Implications for isometric steady-state EMG parameters following a lengthening or shortening contraction. J Electromyogr Kinesiol. 2016;27:30–38. doi: 10.1016/j.jelekin.2016.01.008. [DOI] [PubMed] [Google Scholar]
- 17.Seiberl W, Hahn D, Paternoster FK. Reduced activation in isometric muscle action after lengthening contractions is not accompanied by reduced performance fatigability. Sci Rep. 2016;6:39052. doi: 10.1038/srep39052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Power GA, Makrakos DP, Rice CL, Vandervoort AA. Enhanced force production in old age is not a far stretch: An investigation of residual force enhancement and muscle architecture. Physiol Rep. 2013;1:e00004. doi: 10.1002/phy2.4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chen J, Power GA. Modifiability of the history dependence of force through chronic eccentric and concentric biased resistance training. J Appl Physiol. 2019;126:647–657. doi: 10.1152/japplphysiol.00928.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hinks A, Davidson B, Akagi R, Power GA. Influence of isometric training at short and long muscle–tendon unit lengths on the history dependence of force. Scand J Med Sci Sport. 2021;31:325–338. doi: 10.1111/sms.13842. [DOI] [PubMed] [Google Scholar]
- 21.Koppes RA, Herzog W, Corr DT. Force enhancement in lengthening contractions of cat soleus muscle in situ: Transient and steady-state aspects. Physiol Rep. 2013;1:e00017. doi: 10.1002/phy2.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.de Brito Fontana H, Roesler H, Herzog W. In vivo vastus lateralis force–velocity relationship at the fascicle and muscle tendon unit level. J Electromyogr Kinesiol. 2014;24:934–940. doi: 10.1016/j.jelekin.2014.06.010. [DOI] [PubMed] [Google Scholar]
- 23.Bakenecker P, Raiteri BJ, Hahn D. Force enhancement in the human vastus lateralis is muscle-length-dependent following stretch but not during stretch. Eur J Appl Physiol. 2020;120:2597–2610. doi: 10.1007/s00421-020-04488-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mahmood S, Sawatsky A, Herzog W. Increased force following muscle stretching and simultaneous fibre shortening: Residual force enhancement or force depression – That is the question? J Biomech. 2021;116 doi: 10.1016/j.jbiomech.2020.110216. [DOI] [PubMed] [Google Scholar]
- 25.Raiteri BJ, Hahn D. A reduction in compliance or activation level reduces residual force depression in human tibialis anterior. Acta Physiol (Oxf) 2018;225:e13198. doi: 10.1111/apha.13198. [DOI] [PubMed] [Google Scholar]
- 26.Pinnell RAM, Mashouri P, Mazara N, Weersink E, Brown SHM, Power GA. Residual force enhancement and force depression in human single muscle fibres. J Biomech. 2019;91:164–169. doi: 10.1016/j.jbiomech.2019.05.025. [DOI] [PubMed] [Google Scholar]
- 27.de Brito Fontana H, Herzog W. Vastus lateralis maximum force-generating potential occurs at optimal fascicle length regardless of activation level. Eur J Appl Physiol. 2016;116:1267–1277. doi: 10.1007/s00421-016-3381-3. [DOI] [PubMed] [Google Scholar]
- 28.Arampatzis A, Karamanidis K, De Monte G, Stafilidis S, Morey-Klapsing G, Brüggemann GP. Differences between measured and resultant joint moments during voluntary and artificially elicited isometric knee extension contractions. Clin Biomech. 2004;19:277–283. doi: 10.1016/j.clinbiomech.2003.11.011. [DOI] [PubMed] [Google Scholar]
- 29.Bakenecker P, Raiteri B, Hahn D. Patella tendon moment arm function considerations for human vastus lateralis force estimates. J Biomech. 2019;86:225–231. doi: 10.1016/j.jbiomech.2019.01.042. [DOI] [PubMed] [Google Scholar]
- 30.Ito M, Kawakami Y, Ichinose Y, Fukashiro S, Fukunaga T. Nonisometric behavior of fascicles during isometric contractions of a human muscle. J Appl Physiol (1985) 1998;85:1230–1235. doi: 10.1152/jappl.1998.85.4.1230. [DOI] [PubMed] [Google Scholar]
- 31.Moher D, Liberati A, Tetzlaff J, Altman DG, Group PRISMA. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Annu Intern Med. 2009;151:264–269. doi: 10.7326/0003-4819-151-4-200908180-00135. [DOI] [PubMed] [Google Scholar]
- 32.Inthout J, Ioannidis JP, Borm GF. The Hartung-Knapp-Sidik-Jonkman method for random effects meta-analysis is straightforward and considerably outperforms the standard DerSimonian-Laird method. BMC Med Res Methodol. 2014;14:25. doi: 10.1186/1471-2288-14-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Higgins JPT, Green S. The Cochrane Collaboration; London: 2008. Cochrane Handbook for Systematic Reviews of Interventions. [Google Scholar]
- 34.Deeks JJ, Higgins JPT, Altman DG. The Cochrane Collaboration; London: 2008. Cochrane Handbook for Systematic Reviews of Interventions. [Google Scholar]
- 35.Borenstein M, Hedges LV, Higgins JPT, Rothstein HR. Introduction to meta-analysis. Available at: https://onlinelibrary.wiley.com/doi/book/10.1002/9780470743386. [accessed 31.10.2020].
- 36.Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–634. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Duval S, Tweedie R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics. 2000;56:455–463. doi: 10.1111/j.0006-341x.2000.00455.x. [DOI] [PubMed] [Google Scholar]
- 38.ACE Gestão de Saúde. Manual of Goniometry measuring the joint angles (Manual de Goniometria medição dos ângulos articulares). Available at: http://acegs.com.br/wp-content/uploads/2016/06/MANUAL-DE-GONIOMETRIA-FINAL.pdf. [accessed 20.02.2021].[in Portuguese].
- 39.Balduzzi S, Rücker G, Schwarzer G. How to perform a meta-analysis with R: A practical tutorial. Evid Based Ment Health. 2019;22:153–160. doi: 10.1136/ebmental-2019-300117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Viechtbauer W. Conducting meta-analyses in R with the metafor. J Stat Softw. 2010;36:1–48. [Google Scholar]
- 41.Harrer M, Cuijpers P, Furukawa T, Ebert DD. Doing meta-analysis in R. Available at: https://dmetar.protectlab.org/. [accessed 21.05.2020].
- 42.Wickham H, Averick M, Bryan J, et al. Welcome to the Tidyverse. J Open Source Softw. 2019;4:1686. doi: 10.21105/joss.01686. [DOI] [Google Scholar]
- 43.Pinniger GJ, Cresswell AG. Residual force enhancement after lengthening is present during submaximal plantar flexion and dorsiflexion actions in humans. J Appl Physiol. 2007;102:18–25. doi: 10.1152/japplphysiol.00565.2006. [DOI] [PubMed] [Google Scholar]
- 44.Contento VS, Dalton BH, Power GA. The inhibitory tendon-evoked reflex is increased in the torque-enhanced state following active lengthening compared to a purely isometric contraction. Brain Sci. 2019;10:13. doi: 10.3390/brainsci10010013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cook CS, McDonagh MJ. Force responses to controlled stretches of electrically stimulated human muscle–tendon complex. Exp Physiol. 1995;80:477–490. doi: 10.1113/expphysiol.1995.sp003862. [DOI] [PubMed] [Google Scholar]
- 46.Fortuna R, Power GA, Mende E, Seiberl W, Herzog W. Residual force enhancement following shortening is speed-dependent. Sci Rep. 2016;6:21513. doi: 10.1038/srep21513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Fortuna R, Goecking T, Seiberl W, Herzog W. Force depression following a stretch-shortening cycle depends on the amount of residual force enhancement established in the initial stretch phase. Physiol Rep. 2019;7:e14188. doi: 10.14814/phy2.14188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Fukutani A, Misaki J, Isaka T. Influence of joint angle on residual force enhancement in human plantar flexors. Front Physiol. 2017;8:234. doi: 10.3389/fphys.2017.00234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Fukutani A, Shimoho K, Isaka T. Isometric preactivation before active lengthening increases residual force enhancement. Scand J Med Sci Sports. 2019;29:1153–1160. doi: 10.1111/sms.13454. [DOI] [PubMed] [Google Scholar]
- 50.Hahn D, Riedel TN. Residual force enhancement contributes to increased performance during stretch-shortening cycles of human plantar flexor muscles in vivo. J Biomech. 2018;77:190–193. doi: 10.1016/j.jbiomech.2018.06.003. [DOI] [PubMed] [Google Scholar]
- 51.Jakobi JM, Kuzyk SL, McNeil CJ, Dalton BH, Power GA. Motor unit contributions to activation reduction and torque steadiness following active lengthening: A study of residual torque enhancement. J Neurophysiol. 2020;123:2209–2216. doi: 10.1152/jn.00394.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Oskouei AE, Herzog W. Activation-induced force enhancement in human adductor pollicis. J Electromyogr Kinesiol. 2009;19:821–828. doi: 10.1016/j.jelekin.2008.02.009. [DOI] [PubMed] [Google Scholar]
- 53.Ruiter CJ, Didden WJ, Jones DA, Haan AD. The force–velocity relationship of human adductor pollicis muscle during stretch and the effects of fatigue. J Physiol. 2000;526:671–681. doi: 10.1111/j.1469-7793.2000.00671.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Power GA, Makrakos DP, Stevens DE, Rice CL, Vandervoort AA. Velocity dependence of eccentric strength in young and old men: The need for speed! Appl Physiol Nutr Metab. 2015;40:703–710. doi: 10.1139/apnm-2014-0543. [DOI] [PubMed] [Google Scholar]
- 55.Hahn D, Hoffman BW, Carroll TJ, Cresswell AG. Cortical and spinal excitability during and after lengthening contractions of the human plantar flexor muscles performed with maximal voluntary effort. PLoS One. 2012;7:e49907. doi: 10.1371/journal.pone.0049907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hahn D, Seiberl W, Schmidt S, Schweizer K, Schwirtz A. Evidence of residual force enhancement for multi-joint leg extension. J Biomech. 2010;43:1503–1508. doi: 10.1016/j.jbiomech.2010.01.041. [DOI] [PubMed] [Google Scholar]
- 57.MacDonald GZ, Mazara N, Herzog W, Power GA. Mitigating the bilateral deficit: Reducing neural deficits through residual force enhancement and activation reduction. Eur J Appl Physiol. 2018;118:1911–1919. doi: 10.1007/s00421-018-3924-x. [DOI] [PubMed] [Google Scholar]
- 58.Broos S, Van Leemputte M, Deldicque L, Thomis MA. History-dependent force, angular velocity and muscular endurance in ACTN3 genotypes. Eur J Appl Physiol. 2015;115:1637–1643. doi: 10.1007/s00421-015-3144-6. [DOI] [PubMed] [Google Scholar]
- 59.Hahn D, Seiberl W, Schwirtz A. Force enhancement during and following muscle stretch of maximal voluntarily activated human quadriceps femoris. Eur J Appl Physiol. 2007;100:701–709. doi: 10.1007/s00421-007-0462-3. [DOI] [PubMed] [Google Scholar]
- 60.Tilp M, Steib S, Schappacher-Tilp G, Herzog W. Changes in fascicle lengths and pennation angles do not contribute to residual force enhancement/depression in voluntary contractions. J Appl Biomech. 2011;27:64–73. doi: 10.1123/jab.27.1.64. [DOI] [PubMed] [Google Scholar]
- 61.Power GA, Rice CL, Vandervoort AA. Residual force enhancement following eccentric induced muscle damage. J Biomech. 2012;45:1835–1841. doi: 10.1016/j.jbiomech.2012.04.006. [DOI] [PubMed] [Google Scholar]
- 62.Power GA, Rice CL, Vandervoort AA. Increased residual force enhancement in older adults is associated with a maintenance of eccentric strength. PLoS One. 2012;7:e48044. doi: 10.1371/journal.pone.0048044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Shim J, Garner B. Residual force enhancement during voluntary contractions of knee extensors and flexors at short and long muscle lengths. J Biomech. 2012;45:913–918. doi: 10.1016/j.jbiomech.2012.01.026. [DOI] [PubMed] [Google Scholar]
- 64.Siebert T, Kurch D, Blickhan R, Stutzig N. Does weightlifting increase residual force enhancement? J Biomech. 2016;49:2047–2052. doi: 10.1016/j.jbiomech.2016.05.017. [DOI] [PubMed] [Google Scholar]
- 65.Seiberl W, Hahn D, Kreuzpointner F, Schwirtz A, Gastmann U. Force enhancement of quadriceps femoris in vivo and its dependence on stretch-induced muscle architectural changes. J Appl Biomech. 2010;26:256–264. doi: 10.1123/jab.26.3.256. [DOI] [PubMed] [Google Scholar]
- 66.Tilp M, Steib S, Herzog W. Force-time history effects in voluntary contractions of human tibialis anterior. Eur J Appl Physiol. 2009;106:159–166. doi: 10.1007/s00421-009-1006-9. [DOI] [PubMed] [Google Scholar]
- 67.Paternoster FK, Hahn D, Stöcker F, Schwirtz A, Seiberl W. Oxygen consumption of gastrocnemius medialis muscle during submaximal voluntary isometric contractions with and without preceding stretch. Sci Rep. 2017;7:4674. doi: 10.1038/s41598-017-04068-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Seiberl W, Hahn D, Herzog W, Schwirtz A. Feedback controlled force enhancement and activation reduction of voluntarily activated quadriceps femoris during sub-maximal muscle action. J Electromyogr Kinesiol. 2012;22:117–123. doi: 10.1016/j.jelekin.2011.10.010. [DOI] [PubMed] [Google Scholar]
- 69.Sypkes CT, Kozlowski BJ, Grant J, Bent LR, McNeil CJ, Power GA. The influence of residual force enhancement on spinal and supraspinal excitability. PeerJ. 2018;6:e5421. doi: 10.7717/peerj.5421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Oskouei AE, Herzog W. Observations on force enhancement in submaximal voluntary contractions of human adductor pollicis muscle. J Appl Physiol(1985) 2005;98:2087–2095. doi: 10.1152/japplphysiol.01217.2004. [DOI] [PubMed] [Google Scholar]
- 71.Altenburg TM, de Ruiter CJ, Verdijk PW, van Mechelen W, de Haan A. Vastus lateralis surface and single motor unit EMG following submaximal shortening and lengthening contractions. Appl Physiol Nutr Metab. 2008;33:1086–1095. doi: 10.1139/H08-092. [DOI] [PubMed] [Google Scholar]
- 72.Paquin J, Power GA. History dependence of the EMG-torque relationship. J Electromyogr Kinesiol. 2018;41:109–115. doi: 10.1016/j.jelekin.2018.05.005. [DOI] [PubMed] [Google Scholar]
- 73.Paternoster FK, Seiberl W, Hahn D, Schwirtz A. Residual force enhancement during multi-joint leg extensions at joint-angle configurations close to natural human motion. J Biomech. 2016;49:773–779. doi: 10.1016/j.jbiomech.2016.02.015. [DOI] [PubMed] [Google Scholar]
- 74.Oskouei AE, Herzog W. Force enhancement at different levels of voluntary contraction in human adductor pollicis. Eur J Appl Physiol. 2006;97:280–287. doi: 10.1007/s00421-006-0167-z. [DOI] [PubMed] [Google Scholar]
- 75.Mazara N, Hess AJ, Chen J, Power GA. Activation reduction following an eccentric contraction impairs torque steadiness in the isometric steady-state. J Sport Heal Sci. 2018;7:310–317. doi: 10.1016/j.jshs.2018.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Oskouei AE, Herzog W. The dependence of force enhancement on activation in human adductor pollicis. Eur J Appl Physiol. 2006;98:22–29. doi: 10.1007/s00421-006-0170-4. [DOI] [PubMed] [Google Scholar]
- 77.Dalton BH, Contento VS, Power GA. Residual force enhancement during submaximal and maximal effort contractions of the plantar flexors across knee angle. J Biomech. 2018;78:70–76. doi: 10.1016/j.jbiomech.2018.07.019. [DOI] [PubMed] [Google Scholar]
- 78.Chapman N, Whitting J, Broadbent S, Crowley-McHattan Z, Meir R. Maximal and submaximal isometric torque is elevated immediately following highly controlled active stretches of the hamstrings. J Electromyogr Kinesiol. 2021;56 doi: 10.1016/j.jelekin.2020.102500. [DOI] [PubMed] [Google Scholar]
- 79.Gruber M, Linnamo V, Strojnik V, Rantalainen T, Avela J. Excitability at the motoneuron pool and motor cortex is specifically modulated in lengthening compared to isometric contractions. J Neurophysiol. 2009;101:2030–2040. doi: 10.1152/jn.91104.2008. [DOI] [PubMed] [Google Scholar]
- 80.Castro MP, Ruschel C, Santos GM, Ferreira T, Pierri CAA, Roesler H. Isokinetic hip muscle strength: A systematic review of normative data. Sport Biomech. 2020;19:26–54. doi: 10.1080/14763141.2018.1464594. [DOI] [PubMed] [Google Scholar]
- 81.Edouard P, Samozino P, Julia M, et al. Reliability of isokinetic assessment of shoulder-rotator strength: A systematic review of the effect of position. J Sport Rehabil. 2011;20:367–383. doi: 10.1123/jsr.20.3.367. [DOI] [PubMed] [Google Scholar]
- 82.Joumaa V, Herzog W. Energy cost of force production is reduced after active stretch in skinned muscle fibres. J Biomech. 2013;46:1135–1139. doi: 10.1016/j.jbiomech.2013.01.008. [DOI] [PubMed] [Google Scholar]
- 83.Bullimore SR, Leonard TR, Rassier DE, Herzog W. History-dependence of isometric muscle force: Effect of prior stretch or shortening amplitude. J Biomech. 2007;40:1518–1524. doi: 10.1016/j.jbiomech.2006.06.014. [DOI] [PubMed] [Google Scholar]
- 84.Hisey B, Leonard TR, Herzog W. Does residual force enhancement increase with increasing stretch magnitudes? J Biomech. 2009;42:1488–1492. doi: 10.1016/j.jbiomech.2009.03.046. [DOI] [PubMed] [Google Scholar]
- 85.de Brito Fontana H, Han SW, Sawatsky A, Herzog W. The mechanics of agonistic muscles. J Biomech. 2018;79:15–20. doi: 10.1016/j.jbiomech.2018.07.007. [DOI] [PubMed] [Google Scholar]
- 86.Edman KA, Elzinga G, Noble MI. Enhancement of mechanical performance by stretch during tetanic contractions of vertebrate skeletal muscle fibres. J Physiol. 1978;281:139–155. doi: 10.1113/jphysiol.1978.sp012413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Farris DJ, Lichtwark GA, Brown NAT, Cresswell AG. The role of human ankle plantar flexor muscle–tendon interaction and architecture in maximal vertical jumping examined in vivo. J Exp Biol. 2015;219:528–534. doi: 10.1242/jeb.126854. [DOI] [PubMed] [Google Scholar]
- 88.Power GA, Hinks A, Mashouri P, Contento VS, Chen J. The long and short of residual force enhancement non-responders. Eur J Appl Physiol. 2020;120:2565–2567. doi: 10.1007/s00421-020-04511-5. [DOI] [PubMed] [Google Scholar]
- 89.Bottaro M, Russo AF, de Oliveira RJ. The effects of rest interval on quadriceps torque during an isokinetic testing protocol in elderly. J Sport Sci Med. 2005;4:285–290. [PMC free article] [PubMed] [Google Scholar]
- 90.Campenella B, Mattacola CG, Kimura IF. Effect of visual feedback and verbal encouragement on concentric quadriceps and hamstrings peak torque of males and females. Isokinet Exerc Sci. 2000;8:1–6. [Google Scholar]
- 91.McNair PJ, Depledge J, Brettkelly M, Stanley SN. Verbal encouragement: Effects on maximum effort voluntary muscle action. Br J Sports Med. 1996;30:243–245. doi: 10.1136/bjsm.30.3.243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Johansson CA, Kent BE, Shepard KF. Relationship between verbal command volume and magnitude of muscle contraction. Phys Ther. 1983;63:1260–1265. doi: 10.1093/ptj/63.8.1260. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.