Abstract
Matrine is a quinazoline alkaloid extracted from Sophora flavescens. The aim of the present study was to determine whether matrine can induce autophagy in the human HeLa and SiHa cervical cancer cell lines in vitro and in vivo. Cell viability assay was used to assess the suppressive effect of matrine and cisplatin on the proliferation of HeLa and SiHa cells. A total of 28 4-week-old female BALB/c nude mice were used for the in vivo study. Autophagy and protein expression were observed via transmission electron microscopy, monodansylcadaverine and immunohistochemical staining and western blotting. The inhibitory effect of matrine on the proliferation of cervical cancer cells was time- and dose-dependent. The combination of matrine and cisplatin synergistically inhibited the proliferation of cervical cancer cells in vitro and in vivo. Transmission electron microscopy showed that after the addition of matrine, numerous autophagosomes and autophagolysosomes were observable in HeLa and SiHa cells, as demonstrated by monodansylcadaverine staining. Western blotting and immunohistochemical staining showed that as the concentration of matrine increased, the expression of the autophagy marker LC3A/B-II also increased significantly in vitro and in vivo. These findings suggested that matrine inhibited the proliferation of cervical cancer cells and induced autophagy by inhibiting the Akt/mTOR signaling pathway. Thus, matrine may represented a potential candidate in combination therapy for cervical cancer as an inducer of autophagy.
Keywords: cervical cancer, matrine, autophagy, Akt/mTOR signaling pathway
Introduction
Cervical cancer was the fourth most frequently diagnosed cancer worldwide in 2018, with an estimated 570,000 new cases. It was also the fourth leading cause of cancer-related deaths in women worldwide, accounting for an estimated 311,000 deaths in 2018 (1). In China, the large size of the population constitutes a great challenge to cervical cancer screening and vaccination programs (2). Despite a current standard of care that includes radiotherapy, chemotherapy and/or surgical resection (3), the 5-year survival of patients with advanced recurrent and metastatic cervical cancer is only 15% (4). To achieve satisfactory therapeutic effectiveness for patients with advanced cervical cancer, improved treatment strategies are needed.
Matrine is a quinazoline alkaloid extracted from the dried roots and fruits of Sophora flavescens (5), and preparations for injection and for use as alkaloid gels have been clinically used in China for several years to treat gynecological inflammation, gynecological tumors, hepatitis and other diseases (6–8). The main component of Sophora flavescens preparations is matrine, which has broad pharmacological properties, including sedation, analgesia (9), antiviral (10), anti-inflammatory (11), antifibrotic (12) antitumor and immunoregulatory activity (13,14), as well as other effects (15). Previous studies have investigated the inhibitory effects of matrine on a variety of tumor cells including cervical cancer (16–20). Matrine combined with chemotherapy drugs, such as cisplatin, docetaxel and adriamycin, may have antitumor activity (21–23). Matrine has also been shown to enhance the levels of immunological parameters such as IgA, IgM, CD8+ and IFN-γ, improve cachexia in patients with advanced cancer, and reduce adverse reactions caused by platinum-based chemotherapy (24).
Autophagy is ubiquitous in tumor cells, and during various stages of the development and progression of cervical cancer, autophagy promotes cell death or cell survival (25). Matrine also has a complex, multifaceted role in tumor development, and can either promote or inhibit autophagy. Matrine-induced autophagy may have opposite effects in different types of tumor cells (26,27). Previous studies have reported that matrine can induce autophagy, promote cell death and enhance the sensitivity of cancer cells to some chemotherapeutic drugs (28,29). Others have reported opposite effects, including reduced autophagy and increased cancer cell survival (30). Our previous observations suggested that matrine could inhibit the growth of cervical cancer by downregulating the p38 signaling pathway (19). However, whether matrine can induce autophagy in cervical cancer remains unclear. In the present study, matrine inhibited the proliferation of cervical cancer cells in vitro and suppressed tumor growth in vivo. With the increased matrine concentration, the expression of the autophagy marker LC3A/B-II increased both in vitro and in vivo, and this effect was mediated by the Akt/mTOR pathway. These findings suggest that matrine-induced autophagy may prove useful for the treatment of cervical cancer.
Materials and methods
Reagents
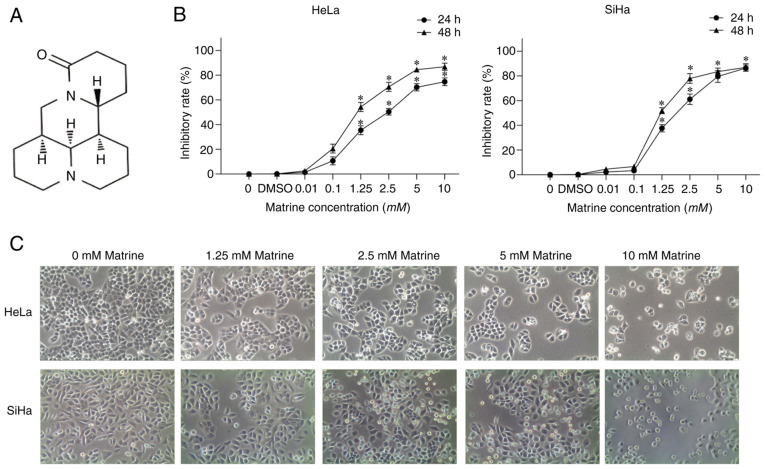
The following reagents were used: Matrine (Xi'an Caogen Biological Engineering), cisplatin (also known as DDP; Qilu Pharmaceutical), DMEM (Hyclone; Cytiva), FBS (Gibco; Thermo Fisher Scientific, Inc.), Cell Counting Kit-8 (CCK-8; Dojindo Laboratories, Inc.) and DMSO (MilliporeSigma). Antibodies specific for LC3A/B (cat. no. 12741t), Akt (cat. no. 4691T), phosphorylated (P)-Akt (Ser473; cat. no. 4060T), mTOR (cat. no. 2983T), P-mTOR (Ser 2448; cat. no. 5536T), p70 ribosomal protein S6 kinase (p70S6; cat. no. 2708T), phospho-p70S6 kinase (Thr 389; cat. no. 9234T) and β-actin (cat. no. 4970T) were obtained from Cell Signaling Technology, Inc. The structure of matrine is shown in Fig. 1A.
Figure 1.
Matrine inhibits the proliferation of cervical cancer cells. (A) Structure of matrine. (B) Cell viability was measured using Cell-Counting Kit-8 assays. The data are presented as the mean ± SD. n=3. *P<0.05 vs. 0 mM matrine. (C) Morphology of cervical cancer cells treated with matrine for 24 h. Magnification, ×200.
Cell culture
The human HeLa and SiHa cervical cancer cell lines were purchased from the American Type Culture Collection and were cultured in DMEM supplemented with 10% FBS, 100 U/ml penicillin and 100 µg/ml streptomycin (Beijing Solarbio Science & Technology Co., Ltd.) at 37°C with 5% CO2.
CCK-8 assay
HeLa and SiHa cells (5×103 cells/well) were seeded in 96-well plates and treated with matrine (0, 0.01, 0.1, 1.25, 2.5, 5 or 10 mM) and cisplatin (0, 0.01, 0.1, 1.25, 2.5, 5 or 10 µg/ml). After 24 or 48 h, cells were incubated for 60 min with CCK-8 solution, and the absorbance at 450 nm was measured using a microplate reader (Bio-Rad Laboratories, Inc.). For morphological observation, HeLa and SiHa cells (1×106 cells/well) were plated in 24-well plates and treated with matrine (0.00, 1.25, 2.50, 5.00 or 10.00 mM). After 24 h, the plates were observed using an inverted microscope.
Synergy determination
Synergy was determined using the Chou-Talalay method (31) with the CompuSyn software (ComboSyn, Inc.; version no. 2005). CompuSyn is based on the median effect principle and its combination index (CI) theorem for pharmacodynamic research and computerized analysis. A CI value <1 indicates a synergistic effect, 1 indicates an additive effect, and >1 indicates an antagonistic effect.
Monodansylcadaverine (MDC) staining
HeLa and SiHa cells (105 cells/well) were seeded in 6-well plates and incubated with matrine (0 or 5 mM) for 24 h. The cells were collected and mixed with MDC for 30 min at room temperature in the dark. Granules were observed using a confocal laser scanning microscope (Leica Microsystems GmbH).
Transmission electron microscopy
Matrine (0 or 5 mM) was added to culture flasks containing 1×106 cells/ml. After incubation for 24 h, the cells were collected and fixed with 2.5% glutaraldehyde and 2% osmium tetroxide in sodium cacodylate buffer (pH 7.2) at 4°C for 2 h. The fixed specimen was postfixed in 1% osmium tetraoxide (OsO4) containing 1.5% potassium ferrocyanide for 30 min at 4°C. The samples were dehydrated using 50, 60, 70, 80, 90 and 100% ethanol series for 20 min each, and embedded in Epon for 12 h at 37°C, for 12 h at 45°C, and for 24 h at 60°C, then sectioned at 50–70 nm with an ultramicrotome. The sections were double stained with 3% uranyl acetate and lead citrate for 30 min at room temperature and observed with a transmission electron microscope.
Tumor xenograft model
The Institutional Animal Care and Treatment Committee of Xi'an Jiaotong University approved the study (approval no. XJTULAC2020-1337). A total of 28 4-week-old female BALB/c nude mice (weight 20 g) were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd. Mice were kept in a specific-pathogen-free laminar air flowbox at 22°C, humidity at 30–50% and light/dark cycle of 14/10 h and were fed with sterile food pellets and water ad libitum. After HeLa cells in logarithmic growth phase were digested with trypsin, a cell suspension was prepared. HeLa cells (1×106) suspended in 0.2 ml PBS were implanted subcutaneously into the right flank of the nude mice. After 7 days, the mice were randomly assigned to the following groups (n=7/group): i) Control, administered 100 µl normal saline; ii) matrine 50 mg/kg; iii) cisplatin 2 mg/kg; and iv) matrine 50 mg/kg + 2 mg/kg cisplatin. The mice were treated by intraperitoneal injection once every 2 days and were sacrificed 21 days later. The length (l) and width (w) of the tumor were measured every 2 days and the volume (V=l × w2/2) was calculated. The mice were sacrificed by cervical dislocation after 21 days, and the tumors were excised and weighed.
Immunohistochemistry
Tumor tissue were fixed in 10% formalin for 24 h at 4°C, paraffin-embedded and sectioned at 5 µm, dewaxed and incubated with 0.01 M sodium citrate at 100°C for 10 min for antigen retrieval. Then the slides were dehydrated with gradient ethanol (100, 95, 90, 80 and 70%) at 1 min each ethanol concentration. After washing in PBS, sections were immersed in 3% hydrogen peroxide for 15 min. After adding 2% bovine serum for 30 min at 37°C to block non-specific antibody binding for 2 h, the sections were washed twice with PBS and incubated overnight with the antibody LC3A/B (1:100, cat. no. 12741t) at 4°C. PBS diluted biotin-labeled rabbit anti-mouse secondary antibody (1:100, cat. no. 7074P2) was added, and the specimens incubated at room temperature for 2 h. Then samples were rinsed quickly with distilled water, and re-stained with hematoxylin for 30 sec at room temperature, dehydrated, cleared and mounted for observation under a light microscope. The expression of LC3A/B-II was assessed using the integral optical density value calculated using ImageProPlus6.0 software.
Western blot assays
Proteins were isolated from cells and tissues in 200 µl RIPA lysis buffer (Beyotime, Shanghai, China) supplemented with 1% phenylmethanesulfonyl fluoride and 1% phosphatase inhibitor. The lysate was centrifuged at 10,000 g and 4°C for 10 min. The protein concentration was determined with a bicinchoninic acid assay kit (Pierce; Thermo Fisher Scientific, Inc.) compared with a standard reference curve. Around 30 mg proteins were separated by 8–12% SDS-PAGE and blotted onto PVDF. Afterwards, electro-rotation was carried out for 60 min under constant current (250–350 mA), and then samples were sealed with 10% skimmed milk powder at room temperature for 1 h. The membranes were incubated overnight at 4°C with primary antibodies (1:1,000, LC3A/B (cat. no. 12741t), Akt (cat. no. 4691T), phosphorylated (P)-Akt (Ser473; cat. no. 4060T), mTOR (cat. no. 2983T), P-mTOR (Ser 2448; cat. no. 5536T), p70 ribosomal protein S6 kinase (p70S6; cat. no. 2708T), phospho-p70S6 kinase (Thr 389; cat. no. 9234T) and β-actin (cat. no. 4970T; CST) and then with horseradish peroxidase-conjugated secondary antibody (1 : 100, cat. no. 7074P2) at 37°C for 1 h. Subsequently, signals were detected using an ECL detection kit (Yeasen). Quantity One software (Bio-Rad Laboratories, Inc.) was used to visualize the proteins and measure their relative expression. Bands were quantified by ImageJ 1.29 software (National Institutes of Health).
Statistical analysis
Statistical analysis was performed using SPSS 19.0 (IBM Corp.). The data are presented as means ± standard deviation. Differences between two groups were compared using Student's t-test. For experiments involving more than two groups, one-way ANOVA was using instead followed by Fisher's least significant difference test (for n<3 groups). or Tukey's post hoc test (for n ≥3). P<0.05 was considered to indicate a statistically significant difference.
Results
Matrine inhibits the proliferation of cervical cancer cells in vitro
The proliferation of HeLa and SiHa cells was assessed using CCK-8 assays. As shown in Fig. 1B, matrine inhibited the proliferation of HeLa and SiHa cells. Indeed, matrine inhibited the proliferation of HeLa and SiHa cells in both a dose- (0–10 mM) and time- (24 and 48 h) manner. Numerous cytoplasmic vacuoles of varying size were present in HeLa and SiHa cells treated with matrine after 24 h. In addition, the number of small, rounded cells increased when the matrine concentration was increased (Fig. 1C).
Matrine induces autophagy in cervical cancer cells in vitro
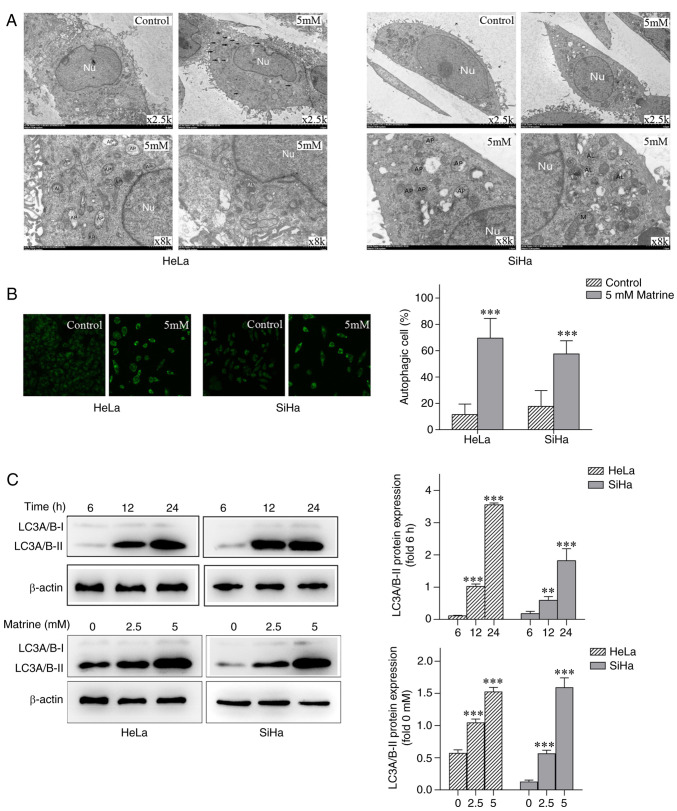
Transmission electron microscopy revealed an abundance of autophagosomes and autophagolysosomes in HeLa and SiHa cells treated with 5 mM matrine (Fig. 2A) compared with untreated control cells. Consistent with the evidence from transmission electron micrographs, MDC staining revealed an abundance of positive particles in HeLa and SiHa cells treated with matrine compared with untreated control cells (Fig. 2B). Western blot assays found that the expression of LC3A/B-II increased significantly as the dose of matrine and the duration of treatment increased (Fig. 2C). These results suggested that matrine induced autophagy in cervical cancer cells in vitro.
Figure 2.
Matrine induces autophagy in cervical cancer cells. (A) Representative electron micrograph of an autophagic cervical cancer cell following matrine treatment. (B) Matrine induced autophagy in cervical cancer cells, as shown by MDC staining and confocal laser scanning microscopy. Magnification, ×400. (C) Western blot analysis of the autophagy marker LC3A/B-II in cervical cancer cells treated with 0, 2.5 or 5 mM matrine for 6, 12 or 24 h. The data are presented as the mean ± SD. n=3. **P<0.01, ***P<0.001 vs. control. Nu, nucleus; AP, autophagosome; M, mitochondria; AL, autophagolysosome.
Matrine initiates autophagy by activating the Akt/mTOR signaling pathway
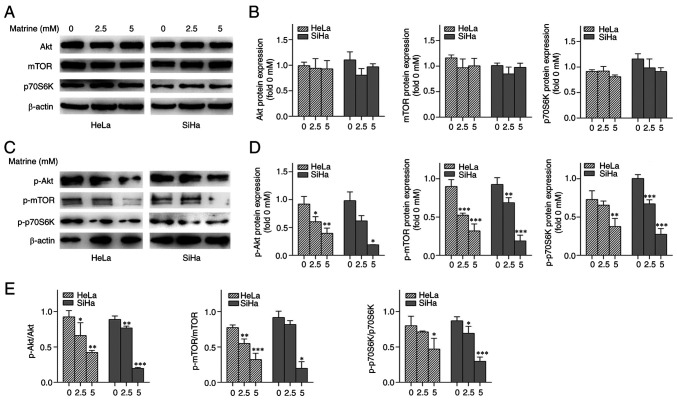
The effect of matrine treatment on Akt/mTOR signaling pathway was assessed by analyzing the expression of Akt, p-Akt, mTOR, p-mTOR and downstream proteins via western blotting. Matrine inhibited the phosphorylation of mTOR-Ser 2448, p70S6K (a downstream target of mTOR) Akt-Ser 473 (an upstream regulator of mTOR) (Fig. 3). These findings suggested that matrine promoted HeLa and SiHa cell autophagy via the Akt/mTOR signaling pathway.
Figure 3.
Effects of matrine on the Akt/mTOR pathway. (A) Western blot images showing the effects of matrine on the expression of proteins involved in Akt/mTOR signaling in cervical cancer cells. (B) Akt (Ser 473), mTOR (Ser 2448), p70S6K (Thr 389) protein expression in cervical cancer cells. (C) Densitometric quantification of the Akt, mTOR, p70S6K. (D) Densitometric quantification of the associated phosphorylated levels of Akt (Ser 473), mTOR (Ser 2448), p70S6K (Thr 389). (E) p-Akt/Akt, p-mTOR/mTOR, p-p70S6K/p70S6K ratio in cervical cancer cells. *P<0.05, **P<0.01, ***P<0.001 vs. 0 mM matrine. p70S6K, p70 ribosomal protein S6 kinase; p-, phosphorylated.
Matrine and cisplatin synergistically inhibit cervical cancer cell proliferation in vitro
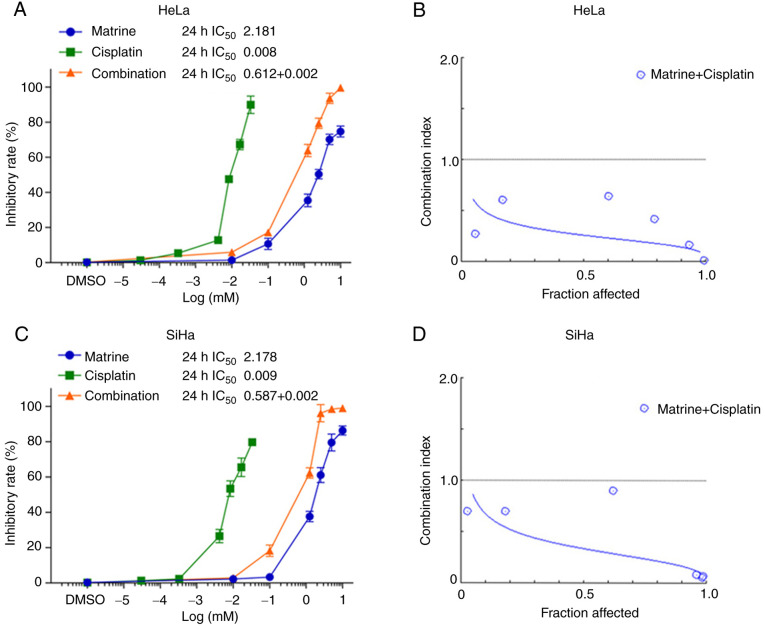
Because matrine alone inhibited the proliferation of cervical cancer cells, the next experiments aimed to determine its effects when combined with cisplatin were investigated. Following treatment for 24 h, the IC50 of matrine was 2.181 mM for HeLa cells and 2.178 mM for SiHa cells. The corresponding IC50 values for cisplatin were 0.008 and 0.009 mM (Fig. 4A and C). When matrine and cisplatin were combined (1:300) for 24 h treatment, the inhibition of HeLa and SiHa cell proliferation was greater than that seen with either drug alone. The corresponding IC50 values for combination group were 0.612+0.002 mM for HeLa cells and 0.587+0.002 mM. An affected fraction (Fa) of 0.5 corresponds to 50% survivability. At Fa=0.5, the CI was 0.26 for HeLa and 0.34 for SiHa cells (Fig. 4B and D). These values suggested that the combination of matrine and cisplatin had a synergistic effect in HeLa and SiHa cells.
Figure 4.
Effects of matrine and cisplatin on the proliferation of cervical cancer cells. (A and C) Drug concentration vs. cell viability. (B and D) Fraction affected vs. combination index graph illustrating the synergistic effects of matrine and cisplatin. The data are presented as the mean ± SD. n=3.
Tumor growth following HeLa cell transplantation into nude mice and treatment with matrine
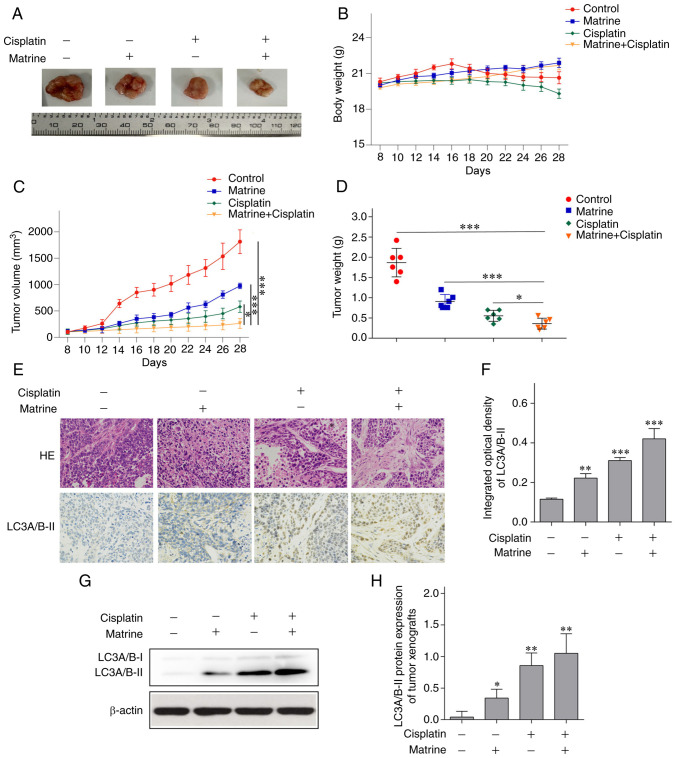
BALB/c nude mice were used to establish an cervical tumor xenograft model and to determine whether matrine induced autophagy in vivo. Matrine (50 mg/kg) + cisplatin (2 mg/kg) significantly suppressed tumor growth (Fig. 5A) without leading to a reduction in body weight (Fig. 5B). The volume (Fig. 5C) and weight (Fig. 5D) of transplanted tumors in nude mice were lower in the matrine + cisplatin group than in the control, matrine, and cisplatin groups. At the end of the experiment, the volumes of the xenografts in the 7 mice in the control group were 1742.91, 1938.40, 1800.63, 1976.20, 1893.55 and 1535.04 mm3. Additionally, the longest diameter of the xenografts of all mice is 16.1 mm. Histological evaluation found less dissemination of disease and destruction of tissue architecture with matrine + cisplatin than in the other treatment groups (Fig. 5E). In addition, immunohistochemical staining (Fig. 5E and F) and western blotting (Fig. 5G and H) suggested that both matrine and matrine + cisplatin upregulated LC3A/B-II in tumor tissue.
Figure 5.
Tumor growth in HeLa cell xenografts transplanted into nude mice treated with matrine. The anti-tumor activity of matrine was dependent on the induction of autophagy. (A) Representative images of dissected tumor tissue. (B) Body weight of mice with cervical cancer tumors. (C) Tumor volumes. (D) Tumor weights. (E) Morphology of tumor tissue stained with hematoxylin and eosin. Immunohistochemical staining of LC3A/B-II in transplanted tumors tissue. Magnification, ×400. (F) Integrated optical density of LC3A/B-II in transplanted tumor tissue. (G) Western blots of LC3A/B-II in transplanted tumor tissue. (H) LC3A/B-II protein expression in tumor tissue. The data are presented as the mean ± SD. n=3. *P<0.05, **P<0.01, ***P<0.001 vs. control.
Discussion
The global prevalence of cervical cancer has dropped from the third place 10 years ago to the fourth place in 2018 (1). The change is associated with the development of medical standards in some countries, increased cervical cancer screening and human papillomavirus vaccination coverage and enhancement of women's health awareness. These interventions seem oncologically safe in the current selection of patients with low-stage and low-grade disease (32). Internationally, the chance for recurrence in early stage cervical cancer is 6–9% (33). However, patients with special types of cervical cancer, such as mid-to-late stage, relapsed, drug-resistant disease, and fertility needs, require multidimensional, multiplan, individualized treatment with few side effects and palliative treatment when necessary (34). Improved understanding of the role and mechanism of matrine in HeLa and SiHa cells may provide new ideas for the treatment of cervical cancer (35).
Cisplatin is the first-line drug in adjuvant combined chemotherapy regimens for cervical cancer (36). Drugs that act synergistically with and reduce the toxicity of cisplatin are important (37). The inhibitory effects of matrine and cisplatin were stronger than those either drug alone (38). When Fa=0.5, isobologram analysis determined that the CI of the drugs in combination was <1.0, indicating that the two drugs had a synergistic effect. The effects of matrine and cisplatin have also been found to be synergistic in urothelial bladder cancer and rhabdomyosarcoma cells (21,39). Matrine may therefore increase the sensitivity of cervical cancer cells to cisplatin. In the present study, matrine combined with cisplatin significantly inhibited tumor volume and weight in vivo. The in vivo antitumor activity of matrine combined with cisplatin has been reported in liver cancer (16), and the combination may be less toxic to normal cells (40).
Sophora flavescens has been widely used clinically in China to treat various diseases including tumors (41). Moreover, matrine suppositories or Sophora flavescens alkaloid gels are used for gynecological diseases such as trichomonal vaginitis, candidal vaginitis and chronic cervicitis (6). Matrine injection is also used for adjuvant treatment of chronic hepatitis (42). Surprisingly, Matrine and Sodium Chloride Injection have a good clinical effect in the treatment of the model mice and 40 patients with coronavirus disease 2019 (COVID-19) (43). At present, the most commonly used anti-tumor matrine drug in clinical practice is Compound Kushen Injection in traditional Chinese medicine (31,44,45). It is a mixture of natural compounds extracted from two medical herbs: Sophora flavescens and Rhizoma smilacis Glabrae (7). Our previous study have suggested that matrine inhibits the proliferation of HeLa and C33A cells (19). Additionally, matrine combined with cisplatin shows antitumor activity in U14 rats with cervical cancer (46). Thus, matrine has the potential for development as therapeutic or adjuvant agent for human cervical cancer. However, matrine as single dosage in the treatment of cervical cancer is still unconfirmed. The reason may be that their ability to inhibit tumor proliferation is limited. In addition, the specific mechanism underlying their potential anti-tumor effects remain unclear. The findings of the present study provide an updated perspective on the relationship between the antitumor property of matrine (either alone or combined with cisplatin) and autophagy, as well as its underlying molecular mechanism.
Autophagy is a double-edged sword in cancer treatment (47–49). Recent studies have shown that the anticancer activity of matrine involves promotion of autophagy (29,50–52). In the present study, both transmission electron microscopy and MDC staining revealed the formation of numerous autophagosomes and autophagolysosomes in cervical cancer cells treated with matrine. In cervical cancer cells, matrine promoted autophagy, as suggested by upregulation of LC3A/B-II in vitro and in vivo. However, the increase in LC3A/B-II cannot be entirely attributed to the increase of autophagy and formation of autophagosomes, but also to decreased digestion of substances in autophagolysosomes. Therefore, it is necessary to determine whether autophagy was inhibited and whether the increase in LC3A/B-II was the result of an increase in autophagy.
The Akt/mTOR signaling pathway is known to be associated with autophagy (53–55). Akt-mediated mTORC1 activation is a key regulator of cell anabolism and growth, and p70S6K is a downstream target of mTORC1 (56). The Akt/mTOR signaling pathway is also active in human papilloma virus (HPV) infection (57). HPV activates the mTORC1 complex (58), increases protein synthesis and reduces host cell autophagy before HPV-16 enters keratinocytes in the early stage to protect the virus entering the body from rapid degradation, thereby prolonging the lifespan of HPV in the host cell (59). In several malignancies, the anticancer activity of matrine depends on inducing autophagy by blocking the Akt/mTOR signal cascade (52,60–62). In the present study, matrine inhibited the phosphorylation of Akt (Ser 473), mTOR (Ser 2448) and the downstream effector molecule p70S6K (Thr 389). Overall, these results are consistent with previous studies on other tumor types (52,61,62).
In conclusion, matrine inhibited the growth of cervical cancer cells by enhancing autophagy in vitro and in vivo. The synergy between matrine and cisplatin provide a theoretical basis for the clinical application of combined treatment of cervical cancer. In addition, matrine-induced autophagy may involve the Akt/mTOR signaling pathway.
Acknowledgements
The authors would like to thank Dr Jennifer C. van Velkinburgh for editing the English language and reviewing the article.
Glossary
Abbreviations
- CCK-8
Cell Counting Kit-8
- CI
combination index
- Fa
fraction affected
- HPV
human papilloma virus
- IC50
half-maximal inhibitory concentration
- MDC
monodansylcadaverine
- p70S6K
p70 ribosomal protein S6 kinase
Funding Statement
This work was supported by The Shaanxi Provincial Key R&D Program (grant no. 2017ZDXM-SF-26-3) and The National Natural Science Foundation of China (grant no. 81702578).
Availability of data and materials
The data sets generated and/or analyzed during the present study are available from the corresponding author on reasonable request.
Authors' contributions
FZ, ML and XW conceived and designed the study. FZ, HZ, WQ, LC and YX performed the in vitro and in vivo experiments and analyzed the data. FZ wrote the manuscript. ML, XW, and LC helped modify the manuscript. FZ revised the manuscript. FZ and HZ confirm the authenticity of all the raw data. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Experiments involving animals were approved by The Biomedical Ethics Committee of The Medical Department of Xi'an Jiaotong University (approval no. XJTULAC2020-1337).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Arbyn M, Weiderpass E, Bruni L, de Sanjose S, Saraiya M, Ferlay J, Bray F. Estimates of incidence and mortality of cervical cancer in 2018: A worldwide analysis. Lancet Glob Health. 2020;8:e191–e203. doi: 10.1016/S2214-109X(19)30482-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhang J, Zhao Y, Dai Y, Dang L, Ma L, Yang C, Li Y, Kong L, Wei L, Zhang S, et al. Effectiveness of High-risk human papillomavirus testing for cervical cancer screening in China: A multicenter, open-label, randomized clinical trial. JAMA Oncol. 2021;7:263–270. doi: 10.1001/jamaoncol.2020.6575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ferrall L, Lin KY, Roden RBS, Hung CF, Wu TC. Cervical cancer immunotherapy: Facts and Hopes. Clin Cancer Res. 2021;27:4953–4973. doi: 10.1158/1078-0432.CCR-20-2833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li Y, Li S, Huang L. Knockdown of Rap2B, a Ras superfamily protein, inhibits proliferation, migration, and invasion in cervical cancer cells via regulating the ERK1/2 signaling pathway. Oncol Res. 2018;26:123–130. doi: 10.3727/096504017X14912172235777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhang H, Chen L, Sun X, Yang Q, Wan L, Guo C. Matrine: A promising natural product with various pharmacological activities. Front Pharmacol. 2020;11:588. doi: 10.3389/fphar.2020.00588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wang X, Chen W, Jin Y, Li J, Hu Y. Effect of Sophora flavescens alkaloid on aerobic vaginitis in gel form for local treatment. J Tradit Chin Med. 2017;37:314–320. doi: 10.1016/S0254-6272(17)30066-3. [DOI] [PubMed] [Google Scholar]
- 7.Wang W, You RL, Qin WJ, Hai LN, Fang MJ, Huang GH, Kang RX, Li MH, Qiao YF, Li JW, et al. Anti-tumor activities of active ingredients in Compound Kushen Injection. Acta Pharmacol Sin. 2015;36:676–679. doi: 10.1038/aps.2015.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang X, Lin H, Zhang R. The clinical efficacy and adverse effects of interferon combined with matrine in chronic hepatitis B: A systematic review and meta-analysis. Phytother Res. 2017;31:849–857. doi: 10.1002/ptr.5808. [DOI] [PubMed] [Google Scholar]
- 9.Oveissi V, Ram M, Bahramsoltani R, Ebrahimi F, Rahimi R, Naseri R, Belwal T, Devkota HP, Abbasabadi Z, Farzaei MH. Medicinal plants and their isolated phytochemicals for the management of chemotherapy-induced neuropathy: Therapeutic targets and clinical perspective. Daru. 2019;27:389–406. doi: 10.1007/s40199-019-00255-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sun N, Zhang H, Sun P, Khan A, Guo J, Zheng X, Sun Y, Fan K, Yin W, Li H. Matrine exhibits antiviral activity in a PRRSV/PCV2 co-infected mouse model. Phytomedicine. 2020;77:153289. doi: 10.1016/j.phymed.2020.153289. [DOI] [PubMed] [Google Scholar]
- 11.Jiang J, Wang G. Matrine protects PC12 cells from lipopolysaccharide-evoked inflammatory injury via upregulation of miR-9. Pharm Biol. 2020;58:314–320. doi: 10.1080/13880209.2020.1719165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhang Y, Cui L, Guan G, Wang J, Qiu C, Yang T, Guo Y, Liu Z. Matrine suppresses cardiac fibrosis by inhibiting the TGFβ/Smad pathway in experimental diabetic cardiomyopathy. Mol Med Rep. 2018;17:1775–1781. doi: 10.3892/mmr.2017.8054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Luo Y, Zhang G, Liu X, Yuan M, Gao Q, Gao H, Ke L, Zhang X, Shi Y, Ma X, et al. Therapeutic and immunoregulatory effects of water-soluble alkaloids E2-a from Sophora moorcroftiana seeds as a novel potential agent against echinococcosis in experimentally protoscolex-infected mice. Vet Res. 2018;49:100. doi: 10.1186/s13567-018-0596-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang L, Zhang H, Zhu Z, Jiang L, Lu X, Zhou M, Sun X, He L, Bai Y, Ma L. Matrine regulates immune functions to inhibit the proliferation of leukemic cells. Int J Clin Exp Med. 2015;8:5591–5600. [PMC free article] [PubMed] [Google Scholar]
- 15.Wang JK, Zhao BS, Wang M, Liu CY, Li YQ, Ma QT, Li PF, Wang TS, Wang CG, Zhou YM. Anti-tumor and phenotypic regulation effect of Matrine on dendritic cells through regulating TLRs pathway. Chin J Integr Med. 2021;27:520–526. doi: 10.1007/s11655-020-3433-8. [DOI] [PubMed] [Google Scholar]
- 16.Hu G, Cao C, Deng Z, Li J, Zhou X, Huang Z, Cen C. Effects of matrine in combination with cisplatin on liver cancer. Oncol Lett. 2021;21:66. doi: 10.3892/ol.2020.12327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gu C, Lu H, Qian Z. Matrine reduces the secretion of exosomal circSLC7A6 from cancer-associated fibroblast to inhibit tumorigenesis of colorectal cancer by regulating CXCR5. Biochem Biophys Res Commun. 2020;527:638–645. doi: 10.1016/j.bbrc.2020.04.142. [DOI] [PubMed] [Google Scholar]
- 18.Liang X, Ju J. Matrine inhibits ovarian cancer cell viability and promotes apoptosis by regulating the ERK/JNK signaling pathway via p38MAPK. Oncol Rep. 2021;45:82. doi: 10.3892/or.2021.8033. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 19.Wu X, Zhou J, Cai D, Li M. Matrine inhibits the metastatic properties of human cervical cancer cells via downregulating the p38 signaling pathway. Oncol Rep. 2017;38:1312–1320. doi: 10.3892/or.2017.5787. [DOI] [PubMed] [Google Scholar]
- 20.Li Y, Zhang J, Ma H, Chen X, Liu T, Jiao Z, He W, Wang F, Liu X, Zeng X. Protective role of autophagy in matrineinduced gastric cancer cell death. Int J Oncol. 2013;42:1417–1426. doi: 10.3892/ijo.2013.1817. [DOI] [PubMed] [Google Scholar]
- 21.Liao XZ, Tao LT, Liu JH, Gu YY, Xie J, Chen Y, Lin MG, Liu TL, Wang DM, Guo HY, Mo SL. Matrine combined with cisplatin synergistically inhibited urothelial bladder cancer cells via down-regulating VEGF/PI3K/Akt signaling pathway. Cancer Cell Int. 2017;17:124. doi: 10.1186/s12935-017-0495-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Li Q, Xu J, He Z, Wen X, Wang F, Zhang P, Li J, Song B, Wang Q, Li R, Huang H. The effects of Matrine in combination with docetaxel on castration-resistant (Androgen-independent) prostate cancer. Cancer Manag Res. 2019;11:10125–10133. doi: 10.2147/CMAR.S213419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhou H, Chen S, Yang Y, Yang C, Chen D, Yao Z, Sun B. Matrine enhances the efficacy of adriamycin chemotherapy in osteosarcoma cells by the STAT3 pathway. Anticancer Drugs. 2019;30:1006–1012. doi: 10.1097/CAD.0000000000000796. [DOI] [PubMed] [Google Scholar]
- 24.Zhang RK, Wang C. Effect of matrine on tumor growth and inflammatory factors and immune function in Wistar rat with breast cancer. Zhongguo Ying Yong Sheng Li Xue Za Zhi. 2018;34:375–378. doi: 10.12047/j.cjap.5657.2018.086. (In Chinese) [DOI] [PubMed] [Google Scholar]
- 25.Sun X, Shu Y, Xu M, Jiang J, Wang L, Wang J, Huang D, Zhang J. ANXA6 suppresses the tumorigenesis of cervical cancer through autophagy induction. Clin Transl Med. 2020;10:e208. doi: 10.1002/ctm2.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wu MY, Wang SF, Cai CZ, Tan JQ, Li M, Lu JJ, Chen XP, Wang YT, Zheng W, Lu JH. Natural autophagy blockers, dauricine (DAC) and daurisoline (DAS), sensitize cancer cells to camptothecin-induced toxicity. Oncotarget. 2017;8:77673–77684. doi: 10.18632/oncotarget.20767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cho YR, Lee JH, Kim JH, Lee SY, Yoo S, Jung MK, Kim SJ, Yoo HJ, Pack CG, Rho JK, Son J. Matrine suppresses KRAS-driven pancreatic cancer growth by inhibiting autophagy-mediated energy metabolism. Mol Oncol. 2018;12:1203–1215. doi: 10.1002/1878-0261.12324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Xie SB, He XX, Yao SK. Matrine-induced autophagy regulated by p53 through AMP-activated protein kinase in human hepatoma cells. Int J Oncol. 2015;47:517–526. doi: 10.3892/ijo.2015.3023. [DOI] [PubMed] [Google Scholar]
- 29.Chi G, Xu D, Zhang B, Yang F. Matrine induces apoptosis and autophagy of glioma cell line U251 by regulation of circRNA-104075/BCL-9. Chem Biol Interact. 2019;308:198–205. doi: 10.1016/j.cbi.2019.05.030. [DOI] [PubMed] [Google Scholar]
- 30.Peng X, Zhou D, Wang X, Hu Z, Yan Y, Huang J. Matrine suppresses proliferation and invasion of SGC7901 cells through inactivation of PI3K/Akt/uPA pathway. Ann Clin Lab Sci. 2016;46:457–462. [PubMed] [Google Scholar]
- 31.Yu L, Zhou Y, Yang Y, Lu F, Fan Y. Efficacy and safety of Compound Kushen Injection on patients with advanced colon cancer: A meta-analysis of randomized controlled trials. Evid Based Complement Alternat Med. 2017;2017:7102514. doi: 10.1155/2017/7102514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Prodromidou A, Iavazzo C, Fotiou A, Psomiadou V, Douligeris A, Vorgias G, Kalinoglou N. Short- and long term outcomes after abdominal radical trachelectomy versus radical hysterectomy for early stage cervical cancer: A systematic review of the literature and meta-analysis. Arch Gynecol Obstet. 2019;300:25–31. doi: 10.1007/s00404-019-05176-y. [DOI] [PubMed] [Google Scholar]
- 33.Schuurman T, Zilver S, Samuels S, Schats W, Amant F, van Trommel N, Lok C. Fertility-sparing surgery in gynecologic cancer: A systematic review. Cancers (Basel) 2021;13:1008. doi: 10.3390/cancers13051008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Naga CP, Gurram L, Chopra S, Mahantshetty U. The management of locally advanced cervical cancer. Curr Opin Oncol. 2018;30:323–329. doi: 10.1097/CCO.0000000000000471. [DOI] [PubMed] [Google Scholar]
- 35.Cohen PA, Jhingran A, Oaknin A, Denny L. Cervical cancer. Lancet. 2019;393:169–182. doi: 10.1016/S0140-6736(18)32470-X. [DOI] [PubMed] [Google Scholar]
- 36.Lorusso D, Petrelli F, Coinu A, Raspagliesi F, Barni S. A systematic review comparing cisplatin and carboplatin plus paclitaxel-based chemotherapy for recurrent or metastatic cervical cancer. Gynecol Oncol. 2014;133:117–123. doi: 10.1016/j.ygyno.2014.01.042. [DOI] [PubMed] [Google Scholar]
- 37.Dasari S, Tchounwou PB. Cisplatin in cancer therapy: Molecular mechanisms of action. Eur J Pharmacol. 2014;740:364–378. doi: 10.1016/j.ejphar.2014.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wang HQ, Jin JJ, Wang J. Matrine induces mitochondrial apoptosis in cisplatin-resistant non-small cell lung cancer cells via suppression of β-catenin/survivin signaling. Oncol Rep. 2015;33:2561–2566. doi: 10.3892/or.2015.3844. [DOI] [PubMed] [Google Scholar]
- 39.Li L, Xue T, Xu W, Zhou B. Effect of matrine combined with cisplatin on the expression of XIAP in human rhabdomyosarcoma RD cells. Oncol Lett. 2016;12:3793–3798. doi: 10.3892/ol.2016.5150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Xia J, Yu X, Song X, Li G, Mao X, Zhang Y. Inhibiting the cytoplasmic location of HMGB1 reverses cisplatin resistance in human cervical cancer cells. Mol Med Rep. 2017;15:488–494. doi: 10.3892/mmr.2016.6003. [DOI] [PubMed] [Google Scholar]
- 41.Chen M, Ding Y, Tong Z. Efficacy and safety of Sophora flavescens (Kushen) based traditional Chinese medicine in the treatment of ulcerative colitis: Clinical evidence and potential mechanisms. Front Pharmacol. 2020;11:603476. doi: 10.3389/fphar.2020.603476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chen Y, Chen LM, Tong Y, You Y. Pharmacological effect and toxicology of Sophorae Tonkinensis Radix et Rhizoma. Zhongguo Zhong Yao Za Zhi. 2017;42:2439–2442. doi: 10.19540/j.cnki.cjcmm.20170609.012. (In Chinese) [DOI] [PubMed] [Google Scholar]
- 43.Yang MW, Chen F, Zhu DJ, Li JZ, Zhu JL, Zeng W, Qu SL, Zhang Y. Clinical efficacy of Matrine and Sodium Chloride Injection in treatment of 40 cases of COVID-19. Zhongguo Zhong Yao Za Zhi. 2020;45:2221–2231. doi: 10.19540/j.cnki.cjcmm.20200323.501. (In Chinese) [DOI] [PubMed] [Google Scholar]
- 44.He R, Ou S, Chen S, Ding S. Network pharmacology-based study on the molecular biological mechanism of action for Compound Kushen Injection in anti-cancer effect. Med Sci Monit. 2020;26:e918520. doi: 10.12659/MSM.918520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ao M, Xiao X, Li Q. Efficacy and safety of compound Kushen injection combined with chemotherapy on postoperative Patients with breast cancer: A meta-analysis of randomized controlled trials. Medicine (Baltimore) 2019;98:e14024. doi: 10.1097/MD.0000000000014024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zhang GL, Jiang L, Yan Q, Liu RH, Zhang L. Anti-tumor effect of matrine combined with cisplatin on rat models of cervical cancer. Asian Pac J Trop Med. 2015;8:1055–1059. doi: 10.1016/j.apjtm.2015.11.005. [DOI] [PubMed] [Google Scholar]
- 47.Onorati AV, Dyczynski M, Ojha R, Amaravadi RK. Targeting autophagy in cancer. Cancer. 2018;124:3307–3318. doi: 10.1002/cncr.31335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Levy JMM, Towers CG, Thorburn A. Targeting autophagy in cancer. Nat Rev Cancer. 2017;17:528–542. doi: 10.1038/nrc.2017.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Amaravadi RK, Kimmelman AC, Debnath J. Targeting autophagy in cancer: Recent advances and future directions. Cancer Discov. 2019;9:1167–1181. doi: 10.1158/2159-8290.CD-19-0292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wan Q, Du Z, Fang Z, Cheng H, Li C, Zhou X. Matrine induces apoptosis and autophagy in human lung adenocarcinoma cells via upregulation of Cavin3 and suppression of PI3K/AKT pathway. J BUON. 2020;25:1512–1516. [PubMed] [Google Scholar]
- 51.Lin S, Zhuang J, Zhu L, Jiang Z. Matrine inhibits cell growth, migration, invasion and promotes autophagy in hepatocellular carcinoma by regulation of circ_0027345/miR-345-5p/HOXD3 axis. Cancer Cell Int. 2020;20:246. doi: 10.1186/s12935-020-01293-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Du J, Li J, Song D, Li Q, Li L, Li B, Li L. Matrine exerts antibreast cancer activity by mediating apoptosis and protective autophagy via the AKT/mTOR pathway in MCF7 cells. Mol Med Rep. 2020;22:3659–3666. doi: 10.3892/mmr.2020.11449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Heras-Sandoval D, Perez-Rojas JM, Hernandez-Damian J, Pedraza-Chaverri J. The role of PI3K/AKT/mTOR pathway in the modulation of autophagy and the clearance of protein aggregates in neurodegeneration. Cell Signal. 2014;26:2694–2701. doi: 10.1016/j.cellsig.2014.08.019. [DOI] [PubMed] [Google Scholar]
- 54.Aoki M, Fujishita T. Oncogenic roles of the PI3K/AKT/mTOR axis. Curr Top Microbiol Immunol. 2017;407:153–189. doi: 10.1007/82_2017_6. [DOI] [PubMed] [Google Scholar]
- 55.Xu Z, Han X, Ou D, Liu T, Li Z, Jiang G, Liu J, Zhang J. Targeting PI3K/AKT/mTOR-mediated autophagy for tumor therapy. Appl Microbiol Biotechnol. 2020;104:575–587. doi: 10.1007/s00253-019-10257-8. [DOI] [PubMed] [Google Scholar]
- 56.Valvezan AJ, Manning BD. Molecular logic of mTORC1 signalling as a metabolic rheostat. Nat Metab. 2019;1:321–333. doi: 10.1038/s42255-019-0038-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Surviladze Z, Sterk RT, DeHaro SA, Ozbun MA. Cellular entry of human papillomavirus type 16 involves activation of the phosphatidylinositol 3-kinase/Akt/mTOR pathway and inhibition of autophagy. J Virol. 2013;87:2508–2517. doi: 10.1128/JVI.02319-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zhang B, Song Y, Sun S, Han R, Hua C, van der Veen S, Cheng H. Human papillomavirus 11 early protein E6 activates autophagy by repressing AKT/mTOR and Erk/mTOR. J Virol. 2019;93:e00172–19. doi: 10.1128/JVI.00172-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Mattoscio D, Medda A, Chiocca S. Human papilloma virus and autophagy. Int J Mol Sci. 2018;19:1775. doi: 10.3390/ijms19061775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zhang X, Hou G, Liu A, Xu H, Guan Y, Wu Y, Deng J, Cao X. Matrine inhibits the development and progression of ovarian cancer by repressing cancer associated phosphorylation signaling pathways. Cell Death Dis. 2019;10:770. doi: 10.1038/s41419-019-2013-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wu J, Hu G, Dong Y, Ma R, Yu Z, Jiang S, Han Y, Yu K, Zhang S. Matrine induces Akt/mTOR signalling inhibition-mediated autophagy and apoptosis in acute myeloid leukaemia cells. J Cell Mol Med. 2017;21:1171–1181. doi: 10.1111/jcmm.13049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hao Y, Yin H, Zhu C, Li F, Zhang Y, Li Y, Wang X, Li D. Matrine inhibits proliferation and promotes autophagy and apoptosis in non-small cell lung cancer cells by deactivating PI3K/AKT/mTOR pathway. Nan Fang Yi Ke Da Xue Xue Bao. 2019;39:760–765. doi: 10.12122/j.issn.1673-4254.2019.07.02. (In Chinese) [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data sets generated and/or analyzed during the present study are available from the corresponding author on reasonable request.