Abstract
Modern identification techniques at the genomic level have greatly improved the taxonomic knowledge of mycobacteria. In adjunct to nucleic acid sequences, mycobacterial identification has been endorsed by investigation of the lipidic patterns of unique mycolic acids in such organisms. In the present investigation, the routine use of high-performance liquid chromatography (HPLC) of mycolic acids, followed by the sequencing of the 16S rRNA, allowed us to select 72 mycobacterial strains, out of 1,035 screened, that do not belong to any of the officially recognized mycobacterial species. Most strains (i.e., 47) were isolated from humans, 13 were from the environment, 3 were from animals, and 9 were from unknown sources. The majority of human isolates were grown from the respiratory tract and were therefore most likely not clinically significant. Some, however, were isolated from sterile sites (blood, pleural biopsy, central venous catheter, or pus). Many isolates, including several clusters of two or more strains, mostly slow growers and scotochromogenic, presented unique genetic and lipidic features. We hope the data reported here, including the results of major conventional identification tests, the HPLC profiles of strains isolated several times, and the whole sequences of the 16S rRNA hypervariable regions of all 72 mycobacteria, may encourage reporting of new cases. The taxonomy of the genus Mycobacterium is, in our opinion, still far from being fully elucidated, and the reporting of unusual strains provides the best background for the recognition of new species. Our report also shows the usefulness of the integration of novel technology to routine diagnosis, especially in cases involving slow-growing microorganisms such as mycobacteria.
For many years, the identification of mycobacteria to the species level has relied on investigation of culture features and biochemical characteristics (15). It is well known that, from this approach, phenotypic patterns with features that do not exactly match any established species frequently emerge. Nevertheless, because of the adoption of the “best fit” criterion, all mycobacteria tested are presumptively assigned to a species, with discrepant features being imputed to biological variability. Such an approach was probably responsible for the long period of immobility in the field of mycobacterial taxonomy during the three decades preceding the 1990s.
Now an increasing number of scientists agree that, among strains with overlapping phenotypic patterns, the conventional procedures are unable to distinguish organisms belonging to a certain taxon from other different species (9).
In recent years, the introduction of novel identification methods and the increasing amount of available data have greatly improved the taxonomic knowledge and allowed the differentiation of organisms previously included within grossly defined species. The use of highly discriminatory techniques, such as genetic sequencing (3) and mycolic acid analyses (1, 2, 5), that usually give unique patterns for different species has heightened awareness that a yet undescribed lipidic picture and/or a new characteristic sequence can be suggestive of a new taxon. Despite the huge amount of work done in the last decade, a number of mycobacteria still await to be recognized as new species. In the context described above, the present study presents the genotypic and phenotypic features of a number of unusual mycobacterial strains that were collected over a period of about 7 years in a mycobacterium reference laboratory.
MATERIALS AND METHODS
Mycobacteria other than Mycobacterium tuberculosis (MOTT) investigated in this study were selected by screening 1,035 strains not hybridizing with commercial DNA probes (AccuProbe; GenProbe, San Diego, Calif.). They had been isolated in or referred to the Regional Mycobacteria Reference Center of Florence (RRCM). In case of multiple identical isolates from the same patient, only one was considered.
The panel of tests used for conventional identification (4) included biochemical investigations (niacin, nitrate reduction, catalase at 68°C and semiquantitative catalase, β-glucosidase, Tween hydrolysis, tellurite reduction, arylsulfatase, and urease), cultural investigations (growth rate, pigmentation type, growth at 25, 37, and 45°C, and morphology of colonies), and inhibition investigations (tolerance of p-nitrobenzoic acid, 5% NaCl, thiophenecarboxylic acid hydrazide, tiacetazone, hydroxylamine, isoniazid, oleic acid, and growth on MacConkey agar without crystal violet). The total number of mycobacteria selected for this study, according to the criteria referred in the Results section, included 72 strains, 11 of which were isolated at RRCM and 61 of which were sent by other laboratories for reference purposes.
High-performance liquid chromatography (HPLC) analysis was performed by the standard procedure (1, 11) with bromophenacyl-esterified mycolic acids; a 10-min gradient of methanol and methylene chloride was used on a 126 model System Gold instrumentation (Beckman, Palo Alto, Calif.) with an Ultrasphere-XL column (Beckman) and a 166 model detector (Beckman) set at 254 nm.
Almost the same procedure was followed by two laboratories that performed genetic analysis of mycobacterial isolates. RNA extraction was performed in Tris-HCl (pH 8.0) buffer containing 20% Chelex (Bio-Rad Laboratories, Milan, Italy) at 56°C for 30 min and subsequently at 100°C for 12 min. The 16S rRNA region was then PCR amplified and sequenced basically as described by Rogall et al. (6). Sequencing of the amplicons was carried out with the ALFexpress DNA sequencer (Amersham Pharmacia Biotech, Milan, Italy) by using the Thermo Sequenase fluorescently labeled primer cycle sequencing kit with 7-deaza-dGTP (Amersham Pharmacia Biotech). The sequencing primers included previously described pC, pC′, and pD′ (6). The sequence reaction was performed in a thermal cycler by cycling the temperature to 95°C for 1 min and then to 63°C (primer pC and pC′) or to 53°C (primer pD′) for 1 min and then to 72°C for 2 min for a total of 30 cycles.
Nucleotide sequencing of the complete 16S rRNA gene was performed for the majority of the strains; however, for a few of them, sequencing was limited to a nucleotide stretch covering hypervariable regions of the 16S rRNA gene analogous to the positions in Escherichia coli corresponding to 129 to 212 and 433 to 503.
The BLAST program (version 2.0; European Molecular Biology Laboratory [http://dove.embl-heidelberg.de/Blast2/]) was used to compare the sequences of the study strains with the ones in the databases. The sequences were aligned with the CLUSTALW program (European Bioinformatics Institute [http://www2.ebi.ac.uk/clustalw]) after trimming to embrace the largest available stretch common to all isolates, which included the A and B regions; the same program yielded the phylogenetic file, from which the phylogenetic trees were drawn by the TREEVIEW program (version 1.6.1; Taxonomy and Systematics at Glasgow, Scotland [http://taxonomy.zoology.gla.ac.uk/rod/treeview.html]).
RESULTS
With biochemical tests, an identification, presenting however a variable level of confidence, was reached for all the 1,035 strains screened. The Mycobacterium gordonae-like pattern was the most frequently encountered, but phenotypes resembling Mycobacterium mucogenicum and other species were not rare. For the most frequent such phenotypes, the percentages of positive results are reported for each test in Table 1, which compared with the ones characterizing, in our setting, the corresponding typical species.
TABLE 1.
Test results concerning the most frequent phenotypic patterns detected among 72 unidentifiable mycobacteria
| Characteristic | % Positive/% in typical strainsa
|
||||||
|---|---|---|---|---|---|---|---|
| M. gordonae | M. mucogenicum | M. flavescens | M. interjectum | M. lentiflavum | M. malmoense | M. neoaurum | |
| Niacin accumulation | 0/0 | 0/0 | 0/0 | 0/0 | 0/0 | 0/0 | 0/0 |
| Nitrate reduction | 0/0 | 25/45 | 100/100 | 0/0 | 0/0 | 0/0 | 0/80 |
| β-Glucosidase | 20/0 | 25/Ub | 0/0 | 0/0 | 0/0 | 0/0 | 0/U |
| Catalase at 68°C | 80/100 | 75/12 | 66/100 | 100/99 | 100/100 | 66/66 | 33/75 |
| Catalase, height > 45 mm | 60/96 | 25/15 | 100/100 | 33/U | 0/U | 0/0 | 0/25 |
| Tween 80 hydrolysis | 80/91 | 75/93 | 100/100 | 0/70 | 0/0 | 100/100 | 66/75 |
| Urease | 40/31 | 50/U | 66/72 | 0/50 | 100/65 | 66/71 | 66/100 |
| Tellurite reduction | 0/29 | 50/U | 66/32 | 100/90 | 0/U | 33/74 | 33/75 |
| Arylsulfatase (3 days) | 20/0 | 75/84 | 0/0 | 0/0 | 0/0 | 33/0 | 33/0 |
| Slow growth | 80/100 | 0/0 | 100/50 | 100/100 | 100/100 | 100/100 | 33/0 |
| Scotochromogenicity | 100/100 | 0/0 | 100/100 | 100/100 | 33/100 | 0/0 | 100/100 |
| Photochromogenicity | 0/0 | 0/0 | 0/0 | 0/0 | 33/0 | 0/0 | 0/0 |
| Smooth colonies | 100/100 | 100/100 | 66/100 | 66/100 | 100/100 | 100/100 | 100/100 |
| Growth at 25°C | 100/100 | 100/100 | 100/100 | 66/100 | 100/100 | 100/100 | 100/100 |
| Growth at 45°C | 0/0 | 0/0 | 66/17 | 0/0 | 0/0 | 0/0 | 0/0 |
| MacConkey agar growth | 0/0 | 50/89 | 0/0 | 0/0 | 0/0 | 0/0 | 0/0 |
| NaCl tolerance | 20/0 | 0/0 | 66/62 | 0/0 | 0/U | 0/0 | 33/U |
| TCHb tolerance | 100/100 | 100/100 | 100/100 | 100/100 | 100/100 | 100/100 | 100/100 |
Results are presented as the percent positive test result in the tentatively identified strains / percent positive test result in the typical strains. U, unknown.
TCH, thiophenecarboxylic acid hydrazide.
With HPLC analysis, 80 of the isolates presented a chromatographic pattern not compatible with the tentative conventional identification; such isolates were further investigated by determining the nucleotide sequence of the 16S rRNA gene (3). The majority of them displayed previously unreported nucleotide sequences within hypervariable regions A and B of the 16S rRNA gene; these strains were selected as the object of this study.
From this study were excluded the strains with 16S rRNA sequences that, although not identical to known species, presented a high level of similarity to established taxa. The latter identification was accepted, provided it did not conflict with the corresponding HPLC pattern and was compatible with at least the two major cultural features, pigmentation and growth rate. Such strains were not included in the present investigation.
On the basis of growth characteristics and pigmentation (Table 2), the strains studied could be allocated within the four Runyon groups (8): 33 were rapid growers, and the remaining 39 were slow growers, split among 27 scotochromogenic, 2 photochromogenic, and 10 nonchromogenic strains.
TABLE 2.
Source and phenotypical and genotypical information about 72 unidentified mycobacteria in this study
| Strain(s) | Origina | Isolation (yr) | Source | Growth rateb | Pigmen- tationc | Growth temp (°C) | Closest identification by:
|
||
|---|---|---|---|---|---|---|---|---|---|
| Conventional testsd | HPLCd | Genetic sequencinge | |||||||
| FI-24800 | Prato | 2000 | Sputum | R | N | 25–37 | M. nonchromogenicum | M. parafortuitum | M. nonchromogenicum |
| FI-4696 | FI | 1996 | Bronchial aspirate | R | N | 25–45 | M. fallax | M. terrae | M. terrae complex |
| FI-14797 | Brescia | 1997 | Lymph node (cow) | R | N | 25–45 | Runyon group iv | M. sphagni | M. terrae |
| FI-19295 | Pisa | 1995 | Cutis | S | S | 25–37 | M. gordonae | M. aurum | M. terrae |
| FI-5198 and 5 others | Milan 5-FI | Ante-1981– 1998 | Sputum, unknown 5 | I | Pinkish | 25–37 | M. terrae complex | M. terrae | M. hiberniae |
| FI-2897 | FI | 1996 | Sputum | R | N | 25–45 | M. nonchromogenicum | M. alvei | Mycobacterium sp. strain MCRO16 (9) |
| FI-24198 | Vicenza | 1998 | Sputum | S | S | 25–37 | None | M. phlei | M. terrae |
| FI-22000 | Bergamo | 2000 | Sputum | S | N | 37 | M. terrae | M. parafortuitum | M. terrae |
| FI-8196 and FI-7796 | Milan | 1986–1988 | Urine, unknown | S | S | 25–37 | M. flavescens | Nocardia sp. | M. phlei/M. flavescens |
| FI-32498 | Brescia | 1998 | Urine | I | S | 25–37 | Runyon group iv | M. flavescens | M. novocastrense/M. flavescens |
| FI-15495 and FI-8298 | Vicenza-FI | 1995–1998 | Bronchial aspirate, sputum | I | S | 25–45 | M. flavescens | M. shimoidei | Mycobacterium sp. strain MICRO17 (9)∗ |
| FI-5896 and FI-11096 | Milan | 1991 | Bronchial aspirate, unknown | S | S | 25–37 | None | Nocardia sp. | M. smegmatis |
| FI-17496 | FI | 1996 | Sputum | R | N | 25–37 | M. triviale | M. fortuitum | Rapidly growing thermotolerant mycobacterium |
| FI-28696 | Milan | 1996 | Bronchial aspirate | S | S | 25–37 | M. flavescens | M. mucogenicum | Mycobacterium sp. strain MCRO7 (9) |
| FI-19000 | Milan | 2000 | Sputum | R | P | 37–45 | M. thermoresistibile | M. porcinum | M. smegmatis |
| FI-26099 | Milan | 1999 | Unknown | S | S | 25–45 | M. phlei | M. farcinogenes | M. smegmatis |
| FI-8499 | Vicenza | 1999 | Sputum | R | S | 25–45 | Runyon group iv | M. farcinogenes | M. smegmatis |
| FI-27897 | Florence | 1997 | Water | S | N | 25–37 | None | M. tusciae | Mycobacterium sp. strain MCRO7 (9)∗ |
| FI-22098 | Vicenza | 1998 | Sputum | S | S | 25–45 | None | M. szulgai | M. smegmatis/M. thermoresistibile |
| FI-12796 | Florence | 1996 | Water | S | P | 25–37 | M. kansasii | M. kansasii | M. kansasii |
| FI-2398 and 6 others | India | 1968–1980 | Dust, water | S | S | 25–37 | M. gordonae | M. scrofulaceum | M. bohemicum |
| FI-2400 | Lucca | 2000 | Sputum | S | S | 37 | M. interjectum | M. scrofulaceum | Mycobacterium sp. strain IWGMT90160 (14) |
| FI-23297 | Japan | 1997 | Unknown | S | N | 25–37 | MAC (probe MAC+) | M. avium | M. avium/M. intracellulare |
| FI-4898 | India | 1980 | Soil | S | N | 25–37 | M. malmoense | M. asiaticum | M. asiaticum |
| FI-17499 | Vicenza | 1999 | Sputum | S | S | 37 | None | MCLO | M. gordonae |
| FI-9197 | Florence | 1997 | Water | S | S | 25–37 | M. szulgai | None | M. gordonae |
| FI-14199 | Pordenone | 1999 | Pus | S | S | 25–37 | M. gordonae | M. gordonae | M. marinum/M. ulcerans |
| FI-1500 | Matera | 2000 | Sputum | S | S | 25–37 | M. interjectum | M. conspicuum | M. paraffinicum |
| FI-7499 | FI | 1998 | Urine | S | S | 37–45 | M. xenopi | M. xenopi | M. xenopi |
| FI-7297 | FI | 1997 | Sputum | S | P | 25–37 | M. lentiflavum | None | M. triplex |
| FI-9994 and FI-22995 | Milan- Ancona | 1994–1995 | Sputum, pleural biopsy | S | N | 25–37 | M. simiae | M. lentiflavum | M. simiae |
| FI-23395 | Bergamo | 1995 | Sputum | S | N | 25–37 | M. lentiflavum | M. lentiflavum | M. simiae |
| FI-12100 and FI-25197 | Vicenza- Milan | 1995–2000 | Sputum, blood from AIDS patient | S | S/N | 25–37 | M. lentiflavum | MAC | Mycobacterium sp. strain MCRO33 (7)∗ |
| FI-22895 | FI | 1997 | Sputum | S | S | 25–37 | M. interjectum | MAC | M. paraffinicum |
| FI-24599 | Vicenza | 1999 | Venous central catheter tip | R | S | 25–37 | M. neoaurum | M. neoaurum | Mycobacterium sp. strain IWGMT90161 (14)/M. simiae |
| FI-2389 and 2 others | Milan 2 + FI | 1996–1998 | Urine, gastric juice, sputum | R | S/N | 25–37 | Runyon group iv | M. neoaurum | Mycobacterium sp. strain SRB1151-113f |
| FI-1191 | FI | 1999 | Urine | R | S | 25–37 | M. vaccae | M. fallax | M. neoaurum/M. fallax |
| FI-27599 | Vicenza | 1999 | Bronchial aspirate | S | S | 25–37 | M. gordonae | M. diernhoferi | M. fortuitum/“M. ratisbonense”g |
| FI-7494 | Milan | 1996 | Sputum | R | S | 25–37 | Runyon group iv | M. fortuitum | M. aichiense/M. diernhoferi |
| FI-11097 | Milan | 1997 | Sputum | R | P | 25–37 | Runyon group iv | M. fortuitum | M. peregrinum/M. gilvum |
| FI-17697 | Milan | 1997 | Sputum | R | S | 25–37 | M. neoaurum | M. phlei | M. peregrinum/M. gilvum |
| FI-16194 | FI | 1994 | Bronchial aspirate | R | N | 25–37 | M. mucogenicum | M. fallax | “M. ratisbonense”g |
| FI-100 | Vicenza | 2000 | Sputum | R | N | 25–37 | M. mucogenicum | MCLO | “M. ratisbonense”∗g |
| FI-23299 | Vicenza | 1999 | Water | R | N | 25–45 | M. fallax | M. gordonae | M. fortuitum |
| FI-29396 | Vicenza | 1996 | Sputum | R | S | 25–37 | M. vaccae | M. duvalii | M. gilvum |
| FI-26200 | Vicenza | 2000 | Bronchial aspirate | R | S | 25–37 | M. gordonae | MCLO | M. aurum |
| FI-19499 | Vicenza | 1999 | Bronchial aspirate | R | N | 25–37 | M. mucogenicum | M. aurum | Mycobacterium MO183g/ “M. ratisbonense”g |
| FI-5196 | Florence | 1996 | Water | R | N | 25–37 | M. mucogenicum | M. mucogenicum | M. chelonae |
| FI-19696 | Milan | 1996 | Lymph node (cow) | R | N | 25–37 | Runyon group iv | M. fallax | M. neoaurum |
| FI-4896 | Milan | 1995 | Urine | R | N | 25–45 | Runyon group iv | M. fallax | M. fallax |
| FI-1994 | Milan | 1994 | Fish | S | S | 25–37 | M. neoaurum | Nocardia sp. | M. neoaurum/M. aichiense |
| FI-18799 | Vicenza | 1999 | Sputum | S | N | 25–37 | M. malmoense | M. fallax | Mycobacterium MO183f/ “M. ratisbonense”g |
| FI-2494 & FI-2595 | Milan | 1994–1995 | Sputum 2 | S | N | 25–37 | M. malmoense | M. fallax | M. chlorophenolicum |
Place from which the strains were referred. Unless otherwise noted, the country of origin was Italy. FI, strains isolated at RRCM.
R, rapid; S, slow; I, intermediate.
S, scotochromogenic; P, photochromogenic; N, unpigmented.
MAC, M. avium complex; MCLO, M. chelonae-like organism.
Asterisked strains are identical to, not simply resembling, the stated genotype.
C. Kato, A. T. Bull, and J. A. Calquhoun, unpublished data.
U. Reischl, L. Naumann, S. Emler, H. Melzl, and H. Wolf, unpublished data.
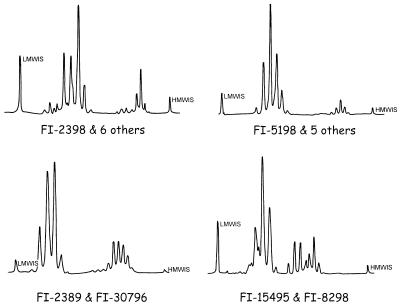
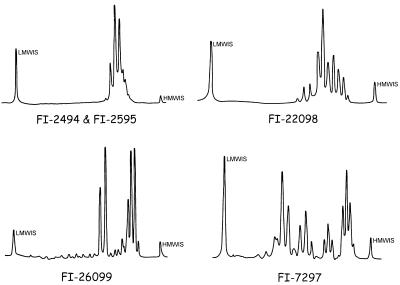
By HPLC analysis of mycolic acids (Fig. 1 and 2), the four most frequent motifs were characterized by one or two clusters of peaks, differentiated in turn by early or delayed retention times. Profiles with three and even four clusters of peaks were also present.
FIG. 1.
Mycolic acid profile in HPLC of several unidentified mycobacteria isolated more than once. LMMIS is the low-molecular-mass internal standard. HMMIS is the high-molecular-mass internal standard.
FIG. 2.
Unusual mycolic acid profiles in HPLC of several unidentified mycobacteria. LMMIS is the low-molecular-mass internal standard. HMMIS is the high-molecular-mass internal standard.
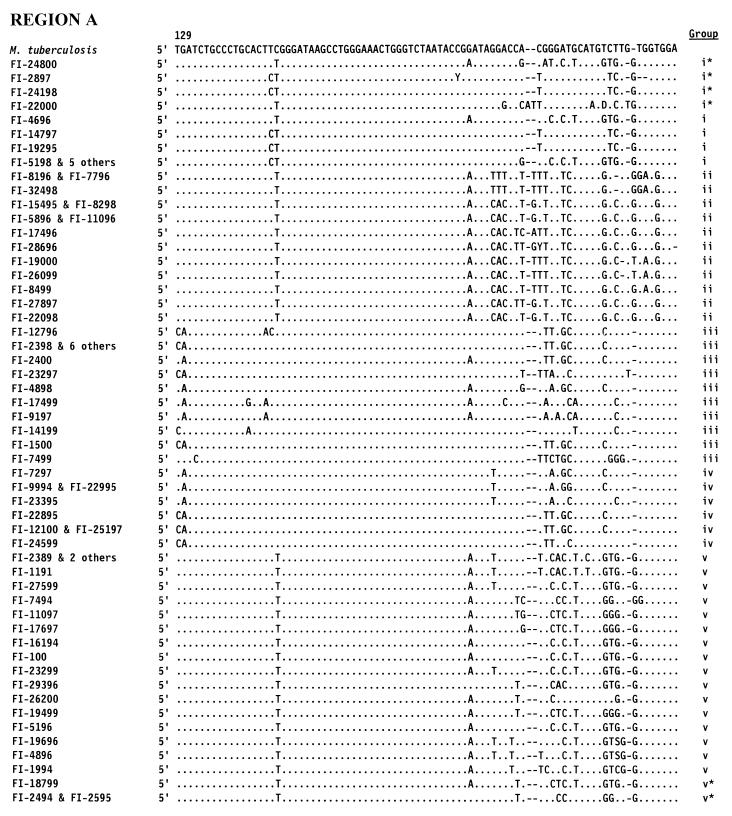
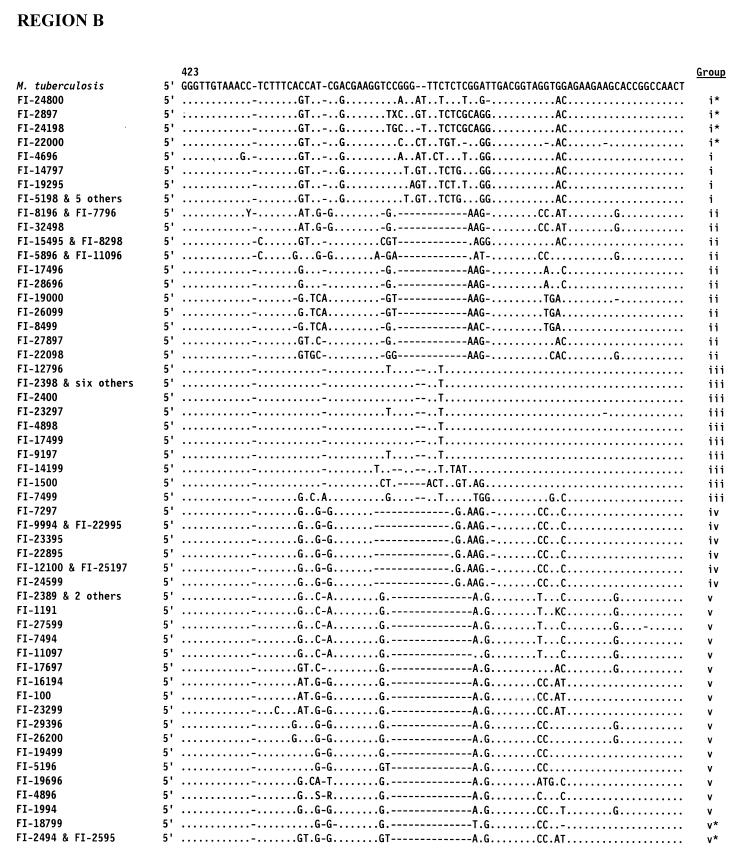
According to the 16S rRNA genetic sequencing (Fig. 3), two major groups were defined on the basis of the length of helix 18 of the 16S rRNA sequence. The majority of the strains (43 strains) lacked 14 nucleotides within the stretch of helix 18, a feature considered typical of rapid growers (10); however, these strains also included 18 slow growers.
FIG. 3.
Sequences of hypervariable regions A and B within the 16S rRNA of 72 unidentified mycobacteria. Positions are indicated by E. coli alignment. Only nucleotides different from M. tuberculosis are shown. Dashes indicate deletions. Groups: i, M. terrae-like; ii, thermotolerant rapid growers; iii, “historical” slow growers; iv, M. simiae-like; v, rapid growers.
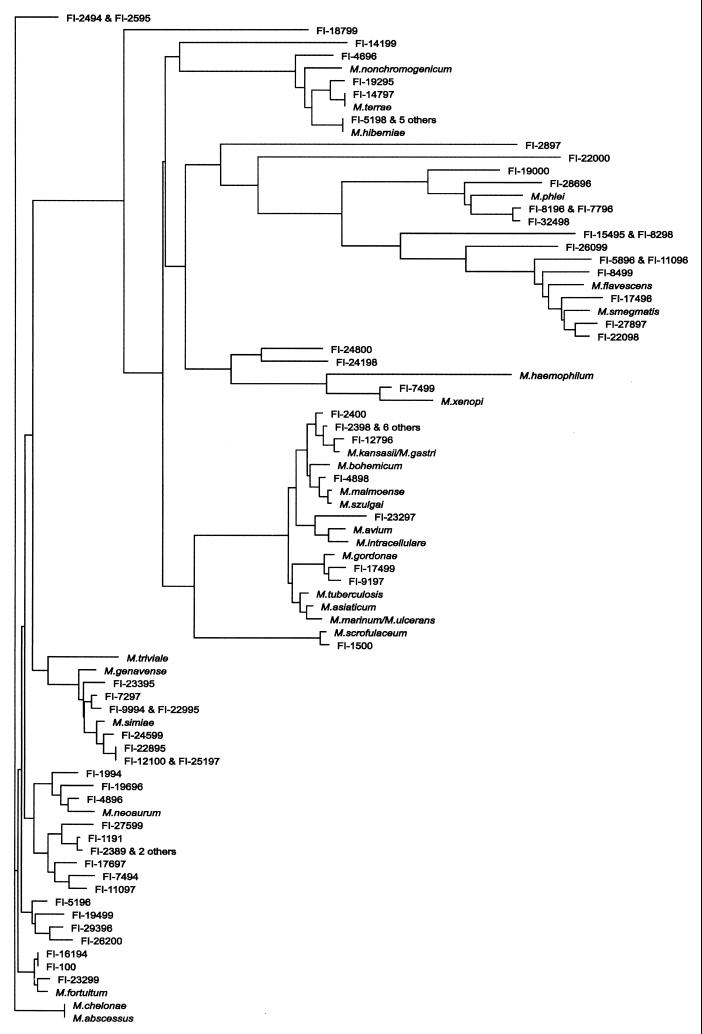
From the phylogenetic arrangement (Fig. 4), five major clusters emerged: (i) the Mycobacterium terrae-related organisms, (ii) the thermotolerant rapid growers, (iii) the ones resembling the “historical” slow growers, (iv) the Mycobacterium simiae-related organisms, and (v) the rapid growers.
FIG. 4.
Phylogenetic tree of 16S rRNAs of the 72 unidentified mycobacteria and of the major known species.
Thirteen isolates clustered with M. terrae; they included both slowly and rapidly growing mycobacteria, as well as scotochromogenic and nonchromogenic organisms. These isolates are characterized by an extended helix 18 in the 16S RNA: i.e., a helix two nucleotides longer than that of any other slow grower (Fig. 3, group i). Among them, however, four strains, although clearly sharing the genetic stigmata of the group, presented as somehow deviant (Fig. 3, asterisked sequences). A group of six identical, although independent, isolates were characterized by an intermediate growth rate and by a pinkish pigmentation. The group of the so-called “thermotolerant rapid growers” included 14 of the unidentified strains: all of them were characterized by a short helix 18 (12 and, in one case, 10 nucleotides shorter than that in M. tuberculosis) in hypervariable region B, and by an insertion of one nucleotide in helix 10 in hypervariable region A (Fig. 3, group ii) at a position corresponding to position 183 of the E. coli sequence, which is considered the genetic marker of thermotolerant rapid growers (3). Interestingly, the isolates belonging to this group were mostly scotochromogenic. Phenotypically, eight of them were slow-growers and only seven of them were able to grow at 45°C.
Sixteen isolates that occurred in a clusters with slowly growing species showed a long helix 18 (Fig. 3, group iii). Phenotypically they all were slow growers and included all pigmentation categories.
Eight strains clustered with M. simiae because of the presence of a short helix 18 (Fig. 3, group iv). Interestingly, this was, from the phenotypic point of view, the most homogeneous group, with seven out of eight isolates being slow growers and with half of them being characterized by a very similar HPLC pattern, close to that of M. simiae.
The largest group included 21 isolates clustering with the classical rapid growers (Runyon group iv); they all had a 12-nucleotide deletion within helix 18 (Fig. 3, group v). Within this group, two isolates with lower genetic homology were present (Fig. 3, asterisked sequences). The large majority of the strains (89%) were rapid growers, and all pigmentation categories were represented, despite a prevalence of scotochromogens.
Among the strains studied, those with identical genetic sequences (the biggest cluster included seven strains) also had overlapping HPLC profiles and quite homogeneous biochemical patterns. In a few instances, organisms presenting very similar HPLC profiles differed in their 16S rRNA sequences as well as phenotypic traits, while in three cases, isolates with similar nucleic acid sequences clearly differed in terms of their chromatographic pattern and conventional tests.
The large majority of unclassifiable strains (65%) were isolated from human clinical samples: 19% were isolated from the environment, and 4% were from animals. The origin of the remaining 12% was unknown. The latter are strains submitted, without background information, from other laboratories during the last two decades. The respiratory tract was the most frequent source, because almost one-half of our mycobacterial strains were isolated from sputa or bronchial aspirates.
DISCUSSION
Paradoxically, the identification of mycobacteria seems to have become still more difficult since the introduction of modern techniques. This feeling is biased by the roughness characterizing speciation based on conventional tests, which, although easily feasible, is substantially inaccurate. In fact, while conventional tests consistently misassign fastidious strains to the species they best resemble among those frequently encountered, both HPLC and genetic profiles, when not overlapping with that of any defined taxon, suggest the possibility of the strain belonging to a species not previously described.
The fact that very few of our strains were isolated before the 1990s and most were isolated after 1994 is not sufficient to jeopardize an increasing trend associated with their isolation. The detection of our strains is biased in fact by a number of factors, the most important of which are the steadily growing attention of mycobacteriologists to MOTT and the enhanced sensitivity of culture methods following the implementation of liquid media.
Our data are of little use in the assessment of the real prevalence of unknown mycobacteria in laboratory isolates, because the majority of the strains examined were referred from other laboratories because of doubts about their identification, which introduced a selection bias. Consideration, however, of only the strains isolated in our laboratory allows us to estimate a prevalence slightly exceeding 1%.
Little is known about the clinical relevance of our isolates for humans; the only ones probably significant are one strain isolated from pus (FI-14199), one from a pleural biopsy (FI-22995), one from an AIDS patient blood culture (FI-12100), and one from the tip of a central venous catheter (FI-24599). Interestingly, three of them are rapid growers. Clinically relevant strains also include the three isolated from animals (two cows and one fish).
Some nucleotide sequences of the hypervariable regions of 16S rRNA presented here have already been reported, but none of them has been assigned so far to any recognized species. This is the case with strains FI-9994 and FI-22955, FI-23395, and FI-22895, corresponding, respectively, to the M. simiae-related genotypes C, D, and E described by Tortoli et al. (13). This is also the case with strains FI-15495 and FI-8298, identical to MCRO17 (9); FI-12100 and FI-25194, identical to MCRO33 (7); FI-100, identical to “Mycobacterium ratisbonense” (U. Reischl, L. Naumann, S. Emler, H. Melzl, and H. Wolf, unpublished data); and FI-27897, identical to MCRO7 (9). The 16S rRNA nucleotide sequence of six strains, among which FI-5198, is identical to that of a mycobacterium isolated in Switzerland (S. Emler, unpublished data). On the other hand, the majority (64%) of the mycobacteria studied in this report are represented by a single isolate, and because they have not been reported elsewhere, we abstain from drawing any conclusion about their taxonomic status, at least until reports of new isolations confirm our findings. The case is different for clustered strains, like the seven identical isolates genetically related to, but different from, Mycobacterium bohemicum, the six related to Mycobacterium hiberniae, and, among the couples of isolates, the one whose sequence is identical to that of MCRO17 (9), as already reported by others. Under these circumstances, it is reasonable to suspect that such organisms belong to new species awaiting recognition, because each cluster shares a consistent genetic sequence, HPLC profile, and biochemical pattern. The complete description of further phenotypic characters is, however, needed for the species definition.
Although none of our isolates exactly fits any of the established species, they presented a variable-level resemblance to some of them. Only in three cases did conventional tests, HPLC, and genetic sequencing agree in terms of the resemblance to a given species. In eight cases, such agreement was limited to conventional tests and HPLC, in eight cases it was limited to sequence and HPLC, and in seven cases it was limited to sequencing and conventional tests.
In contrast to variability within genomic regions, which indeed has phylogenetic relevance, the similarity of mycolic acid patterns does not imply the relatedness of the strains. Genetically related mycobacteria may in fact exhibit clearly different HPLC profiles, while similar mycolic acid patterns may be present in unrelated strains. Several mycobacteria related to M. simiae appear to be the only case in which the relatedness involves both genetic sequence and HPLC profile (13).
Three pairs of phenotypically different strains shared genetic sequences in the hypervariable regions of the 16S rRNA. For FI-8196 and FI-32498, the genetic identity remained uncertain because of two ambiguous bases in FI-8196. FI-16194 and FI-100 presented three discrepancies in the segment between regions A and B. Finally, for FI-22895 and FI-12100, only a short sequence stretch hardly exceeding the hypervariable regions could be compared. Because species sharing an identical nucleic acid sequence in hypervariable regions of 16S rRNA are known (e.g., Mycobacterium kansasii and Mycobacterium gastri), we consider plausible the hypothesis that the isolates of these three couples are independent.
The close similarity of HPLC profiles of several isolates that clearly differed in terms of their genetic sequence and conventional phenotypical features was disregarded, and they were considered to belong to different taxa.
Mycobacteria belonging, from the phenotypic point of view, to slow growers, but genetically clustering with rapid growers, have been already reported elsewhere (12).
Although an estimation of the frequency of unidentifiable mycobacteria is impossible, their number is greater than is generally acknowledged. The exploitation of modern techniques suitable to distinguish such organisms from well-defined species and the reporting of such findings appear to be the most promising tools to widen knowledge, not only of the taxonomy of such mycobacteria but also the epidemiology and their potential pathogenicity for humans and animals.
We hope this study will spur further reporting of isolates like ours and even new ones. The presence of a number of homogeneous strains is, in fact, the main requirement for the recognition of new species and consequently better elucidation of mycobacterial systematics.
ACKNOWLEDGMENTS
Several strains investigated in this study were provided by L. Galassi (Florence, Italy), C. Passerini-Tosi (Bergamo, Italy), L. Bono (Matera, Italy), G. Caroli (Pisa, Italy), B. Casadei (Prato, Italy), P. G. Fiorentino (Florence, Italy), R. Mattei (Lucca, Italy), G. Mucignat (Pordenone, Italy), G. Pinsi (Brescia, Italy), C. Piersimoni (Ancona, Italy), H. Saito (Hiroshima, Japan), and S. Tagliabue (Brescia, Italy).
The work of C.G. and L.R. was supported by funds from the Italian “Istituto Superiore di Sanità,” National Research Program on AIDS, ISS grant no. 50C.11. Thanks are due to GenProbe, Inc., for providing probes used to screen the isolates.
REFERENCES
- 1.Butler W R, Thibert L, Kilburn J O. Identification of Mycobacterium avium complex strains and some similar species by high-performance liquid chromatography. J Clin Microbiol. 1992;30:2698–2704. doi: 10.1128/jcm.30.10.2698-2704.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Guerrant G O, Lambert M A, Moss C W. Gas-chromatographic analysis of mycolic acid cleavage products in mycobacteria. J Clin Microbiol. 1981;13:899–907. doi: 10.1128/jcm.13.5.899-907.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kirschner P, Springer B, Vogel U, Meier A, Wrede A, Kiekenbeck M, Bange F-C, Böttger E C. Genotypic identification of mycobacteria by nucleic acid sequence determination: report of a 2-year experience in a clinical laboratory. J Clin Microbiol. 1993;31:2882–2889. doi: 10.1128/jcm.31.11.2882-2889.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Metchock B G, Nolte F S, Wallace R J., III . Mycobacterium. In: Murray P R, Baron E J, Pfaller M A, Tenover F C, Yolken R H, editors. Manual of clinical microbiology. 7th ed. Washington, D.C.: ASM Press; 1999. pp. 399–437. [Google Scholar]
- 5.Minnikin D E, Minnikin S M, Parlett J M, Goodfellow M, Magnusson M. Mycolic acid patterns of some species of Mycobacterium. Arch Microbiol. 1984;139:225–231. doi: 10.1007/BF00402005. [DOI] [PubMed] [Google Scholar]
- 6.Rogall T, Flohr T, Böttger E C. Differentiation of mycobacterial species by direct sequencing of amplified RNA. J Gen Microbiol. 1990;136:1915–1920. doi: 10.1099/00221287-136-9-1915. [DOI] [PubMed] [Google Scholar]
- 7.Roth A, Reischl U, Streubel A, Naumann L, Kroppenstedt R M, Habicht M, Fischer M, Mauch H. Novel diagnostic algorithm for identification of mycobacteria using genus-specific amplification of the 16S–23S rRNA gene spacer and restriction endonucleases. J Clin Microbiol. 2000;38:1094–1104. doi: 10.1128/jcm.38.3.1094-1104.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Runyon E H. Anonymous mycobacteria in pulmonary disease. Med Clin N Am. 1959;43:273–290. doi: 10.1016/s0025-7125(16)34193-1. [DOI] [PubMed] [Google Scholar]
- 9.Springer B, Stockman L, Teschner K, Roberts G D, Böttger E C. Two-laboratory collaborative study on identification of mycobacteria: molecular versus phenotypic methods. J Clin Microbiol. 1996;34:296–303. doi: 10.1128/jcm.34.2.296-303.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Stahl D A, Urbance J W. The division between fast- and slow-growing species corresponds to natural relationships among the mycobacteria. J Bacteriol. 1990;172:116–124. doi: 10.1128/jb.172.1.116-124.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tortoli E, Bartoloni A. High-performance liquid chromatography and identification of mycobacteria. Rev Med Microbiol. 1996;7:207–219. [Google Scholar]
- 12.Tortoli E, Kroppenstedt R M, Bartoloni A, Caroli G, Jan I, Pawlowsky J, Emler S. Mycobacterium tusciae sp. nov. Int J Syst Bacteriol. 1999;49:1839–1844. doi: 10.1099/00207713-49-4-1839. [DOI] [PubMed] [Google Scholar]
- 13.Tortoli E, Piersimoni C, Kirschner P, Bartoloni A, Burrini C, Lacchini C, Mantella A, Muzzi G, Passerini Tosi C, Penati V, Scarparo C, Simonetti M T, Böttger E C. Characterization of mycobacterial isolates phylogenetically related to, but different from, Mycobacterium simiae. J Clin Microbiol. 1997;35:697–702. doi: 10.1128/jcm.35.3.697-702.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wayne L G, Good R C, Böttger E C, Butler R, Dorsch M, Ezaki T, Gross W, Jonas V, Kilburn J, Kirschner P, Krichevsky M I, Ridell M, Shinnick T M, Springer B, Stackebrandt E, Tarnok I, Tarnok Z, Tasaka H, Vincent V, Warren N G, Knott C A, Johnson R. Semantide- and chemotaxonomy-based analyses of some problematic phenotypic clusters of slowly growing mycobacteria, a cooperative study of the International Working Group on Mycobacterial Taxonomy. Int J Syst Bacteriol. 1996;46:280–297. doi: 10.1099/00207713-46-1-280. [DOI] [PubMed] [Google Scholar]
- 15.Wayne L G, Kubica G P. Family Mycobacteriaceae CHESTER 1897, 63AL. In: Sneath P H A, Mair N S, Sharpe M E, Holt J G, editors. Bergey's manual of systematic bacteriology. Baltimore, Md: The Williams & Wilkins Co.; 1986. pp. 1435–1457. [Google Scholar]