Abstract
Introduction
To evaluate the effects of the global coronavirus disease 2019 (COVID-19) pandemic on lung cancer trials, we surveyed investigators and collected aggregate enrollment data for lung cancer trials across the world before and during the pandemic.
Methods
A Data Collection Survey collected aggregate monthly enrollment numbers from 294 global lung cancer trials for 2019 to 2020. A 64-question Action Survey evaluated the impact of COVID-19 on clinical trials and identified mitigation strategies implemented.
Results
Clinical trial enrollment declined from 2019 to 2020 by 14% globally. Most reductions in enrollment occurred in April to June where we found significant decreases in individual site enrollment (p = 0.0309). Enrollment was not significantly different in October 2019 to December of 2019 versus 2020 (p = 0.25). The most frequent challenges identified by the Action Survey (N = 172) were fewer eligible patients (63%), decrease in protocol compliance (56%), and suspension of trials (54%). Patient-specific challenges included access to trial site (49%), ability to travel (54%), and willingness to visit the site (59%). The most frequent mitigation strategies included modified monitoring requirements (47%), telehealth visits (45%), modified required visits (25%), mail-order medications (25%), and laboratory (27%) and radiology (21%) tests at nonstudy facilities. Sites that felt the most effective mitigation strategies were telehealth visits (85%), remote patient-reported symptom collection (85%), off-site procedures (85%), and remote consenting (89%).
Conclusions
The COVID-19 pandemic created many challenges for lung cancer clinical trials conduct and enrollment. Mitigation strategies were used and, although the pandemic worsened, trial enrollment improved. A more flexible approach may improve enrollment and access to clinical trials, even beyond the pandemic.
Keywords: Clinical trials, COVID-19, Lung cancer, Telehealth
Introduction
Lung cancer remains the leading worldwide cause of cancer deaths with more than 1.8 million deaths annually.1 , 2 We recently observed a reversal of the trend in lung cancer-specific mortality as a consequence of major advances in lung cancer early detection and extensive use of targeted therapies and immunotherapy leading to increased survival.3 , 4 These improvements are the end result of a multitude of clinical trials.
Patients with lung cancer have been adversely affected by the coronavirus disease 2019 (COVID-19) pandemic in several important ways. First, the severity of the disease and the mortality rates after COVID-19 infection in patients with lung cancer have been quite high in nearly all reports.5, 6, 7, 8, 9, 10, 11 Mortality rates have averaged more than 30% in most of these studies. Patients have experienced delays in diagnostic procedures and initiation and continuation of treatment.11 Furthermore, the pandemic led to a decrease in lung cancer screening rates, a critical area as screening has been found to increase cure rates and save lives.11 Despite the availability of several highly effective vaccines that induce neutralizing antibody levels in most patients with cancer, some reports suggest a small but important few fail to mount normal antibody responses making them susceptible to COVID-19 infection.12, 13, 14, 15, 16
The worldwide COVID-19 pandemic produced a major decline in accrual to clinical trials.13 , 17 , 18 As an example, during the pandemic, actual enrollments to SWOG clinical trials in 2020 were 77.3% of expected enrollments overall and 54.0% of expected enrollments for Cancer Prevention and Control trials.13 Several sponsors of clinical trials have documented the magnitude of the decline in trial accruals and others have documented the effectiveness of some measures taken to mitigate the declines.19, 20, 21 There have been recent reports urging regulatory bodies to incorporate some of the mitigation strategies into permanent changes as a means to overcome low accrual rates.22
The International Association for the Study of Lung Cancer (IASLC) is the largest international organization dedicated to reducing the worldwide burden of lung cancer. The IASLC undertook a worldwide survey of lung cancer-specific clinical trials to understand the effects of the COVID-19 pandemic on accrual to lung cancer trials and document strategies that might be adopted to permanently increase future clinical trial participation. The data presented in this report indicate that the decline in clinical trial enrollment was international in scope and that many of the mitigation strategies were adopted on a worldwide scale. Nonetheless, clinical trial accrual has struggled to reach prepandemic levels. Data from this report emphasize the potential effectiveness of numerous mitigation strategies, which could be carried forward beyond the pandemic.
Materials and Methods
In accomplishing its mission, the IASLC conducted surveys to determine the impact of the COVID-19 pandemic on enrollment to worldwide lung cancer clinical trials and to investigate if any mitigation strategies were effective in improving clinical trial enrollment. An ad hoc executive committee was established and met two to four times per month from September 2020 to March 2021 (Supplementary Data 1). The executive committee identified key global leaders and sites conducting lung cancer clinical trials and created surveys to quantitate trial enrollment and identify mitigation strategies. We implemented a two-pronged approach with a trial enrollment survey and an action mitigation survey to evaluate the experiences of clinical trial sites across the world.
Data Collection Survey
The Data Collection Survey looked at aggregate clinical trial enrollment data, per trial, for each month of 2020, the first year of the pandemic, with 2019, the year before the pandemic, as a control. Data sources included government or regulatory agencies, industry sponsors, and study principal investigators. We included all lung cancer trials open to enrollment at any time in 2019 or 2020, with trial site as the unit of analysis. For multisite and multinational clinical trials, we used site-level data when available and grouped on the basis of country of enrolling site. In some cases, only multiple-site trial data were available. Therefore, we stratified results on the basis of single- versus multiple-site trial data (on the basis of data availability, not the trial design) when comparing quarterly enrollment between 2019 and 2020. We collected the start date and stop date for each trial, and in per trial analyses, trials did not contribute to the denominator in periods before the start date or after the stop date.
A custom REDCap database was developed to house the data from this project at the University of Memphis. Clinical sites had two options to provide data for this project. First, a link was provided to upload data directly to the REDCap database by secure hypertext transfer protocol (http). Second, sites have the option to provide data in an Excel spreadsheet or csv file, using a standardized template uploaded by a custom-secure File Transfer Protocol site. Data were collected in accordance with the IASLC Privacy Policy (https://www.iaslc.org/privacy-policy) with oversight from the University of Memphis Institutional Review Board.
We estimated the number of COVID-19 cases diagnosed each month in 2020 using data downloaded from Our World Data (Global Change Data Laboratory, Our World in Data). Countries were categorized by IASLC region (Asia, Europe, Latin America, North America, and the rest of the world).
Action Mitigation Survey
The Action Survey included 64 questions designed to evaluate the impact of COVID-19 on the conduct of clinical trials in 2020 and to identify mitigation strategies used to combat these impacts. The target respondents were lung cancer clinical trial sites across the world, with site as the unit of analysis (one response per site). The sampling frame for the Action Survey was constructed using a multistage referral process from the executive committee and an ad hoc Steering Committee that was comprised of an additional 21 leaders around the world (Supplementary Data 1). After identification of key site investigators worldwide, we evaluated the list to ensure adequate representation across global regions and expanded as necessary. In total, we identified and contacted 429 potential sites from the Executive and Steering Committee recommendation and 804 principal investigators identified by industry partners. We distributed the survey (Supplementary Data 2) by e-mail, with personal reminders approximately every 2 weeks during the data collection period. This purposive sampling strategy allowed for representation from a diverse group of individuals and institutions globally. Survey results were collected by the IASLC using Survey Monkey.
Statistical Analysis
Data are summarized overall and by region of the world with means and SD or frequencies and percentages. Some analyses collapse categories from a five-point or six-point scale to a binary variable (as described in the Results section). Statistical comparisons across regions used chi-square tests. We evaluated monthly trial enrollment data and compared differences in average quarterly enrollment by year using the Kruskal-Wallis test owing to the distribution of the data. Analyses were conducted with SAS Version 9.4 (Cary, NC), and the threshold for statistical significance was set at 0.05.
Results
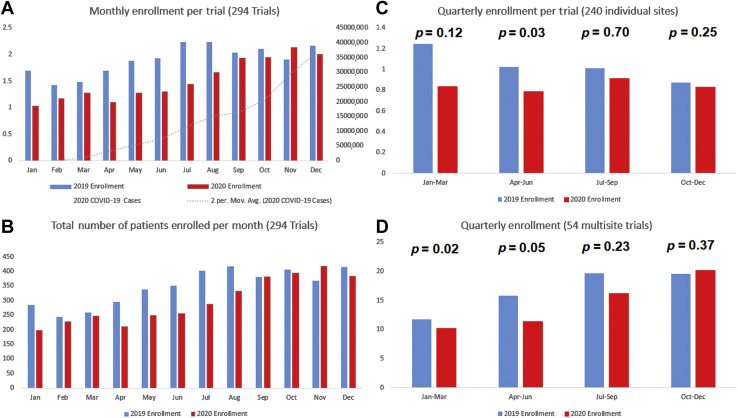
We evaluated study enrollment data from 294 lung cancer trials across 26 countries. This included 114 (39%) from North America, 55 (19%) from Asia, 26 (9%) from Latin America, 79 (27%) from Europe, and 20 (7%) from the rest of the world. These 294 trials enrolled a total of 4163 patients in 2019 compared with 3590 in 2020, a 14% decrease. We evaluated per trial enrollment (Fig. 1 A) and total number of patients enrolled (Fig. 1 B) per month between 2019 and 2020. Quarterly enrollment per trial was compared between 2019 and 2020 within single-institution trial data (Fig. 1 C) and within multiple-institution trial data (Fig. 1 D). Most reductions in enrollment occurred in quarter 2 (April–June) where we found significant decreases from 2019 to 2020 in single-site trial data (p = 0.0309; Fig. 1 C) and marginally significant differences in multiple-site trial data (p = 0.0541; Fig. 1 D). Enrollment in multiple-site trials was also lower in the first quarter (January–March) of 2020 compared with 2019 (p = 0.0185; Fig. 1 D). Nevertheless, there was no meaningful difference in enrollment between 2019 and 2020 in quarter 4 (October–December) in single-site trial data (p = 0.25; Fig. 1 C) or multiple-site trial data (p = 0.37; Fig. 1 D). Despite the apparent rebounding of trial enrollment numbers in the last quarter of 2020, we observed increasing numbers of COVID-19 cases throughout 2020. Specifically, the total numbers of COVID-19 cases diagnosed per month around the world increased consistently from approximately 2,000,000 in February to more than 35,000,000 in December of 2020 (p value for trend < 0.0001; Fig. 1 A).
Figure 1.
(A) The average enrollment in global clinical trials by month for 2019 (the year before the pandemic) and 2020 (the first year of the pandemic). The dotted line reveals the monthly COVID-19 cases diagnosed globally for each month of 2020. (B) Total numbers of patients enrolled in global clinical trials by month for 2019 (the year before the pandemic) and 2020 (the first year of the pandemic). (C) Quarterly enrollment per site for 240 individual sites included in this study, compared between 2019 and 2020. (D) Quarterly enrollment per site for 54 multiple sites included in this study, compared between 2019 and 2020. Apr, April; Aug, August; Avg., Average; COVID-19, coronavirus disease 2019; Dec, December; Feb, February; Jan, January; Jul, July; Jun, June; Mar, March; Mov., moving; Nov, November; Oct, October; Sep, September.
Differences in total enrollment from 2019 to 2020 varied significantly across global regions (p < 0.0001). On the basis of the trials available in our study, we found a 35% decrease in total number of patients enrolled in 2019 compared with 2020 in Europe (from 217 to 142), 10% decrease in Asia (from 177 to 160), 13% decrease in North America (from 3658 to 3195), 88% decrease in Latin America (from 26 to three), and a 5% increase in the rest of the world (from 85 to 89). Quarterly enrollment patterns for each region varied on the basis of the numbers of trials available and are found in Supplementary Data 3.
Action Survey
We received responses to our Action Survey from 172 clinical sites across 45 countries. This included 35 (21%) from North America, 35 (21%) from Asia, 19 (11%) from Latin America, 50 (30%) from Europe, 27 (16%) from the rest of the world, and six with missing information. The primary respondents were 94% (n = 159) physician investigators, 2% (n = 3) research nurses, 2% (n = 4) research coordinators, with the remainder as other or unspecified. Of the responses, 117 (68%) were from academic research centers, 27 (16%) from nonacademic practice or network, 20 (12%) from community practice or health systems, and 7 (4%) other.
Challenges
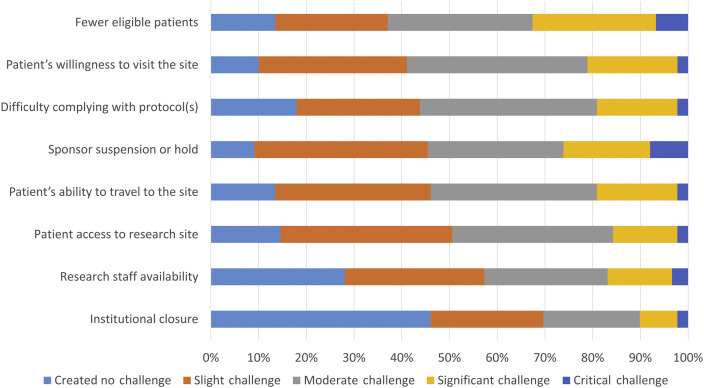
A total of 90 sites provided information in the Action Survey on the greatest challenges their lung cancer clinical trials program faced from the pandemic (Fig. 2 ). We evaluated the proportion of sites that considered each challenge to be moderate, significant, or critical. The most frequent institutional challenges (% moderate or greater) identified were fewer eligible patients (63%), protocol compliance (56%), and suspension of trials (55%; Fig. 2). When asked which months the challenges were first apparent, March (60%), April (82%), and May (77%) of 2020 were the most frequent answers.
Figure 2.
Site reported challenges to enrollment in lung cancer clinical trials by perceived severity.
Critical institutional barriers in Europe (n = 21 sites) were fewer eligible patients (81%), sponsors suspending or placing recruiting on hold (65%), and difficulty complying with protocols (62%). These were also critical in North America (n = 20), with fewer eligible patients (70%), sponsors suspending or placing recruiting on hold (75%), and difficulty complying with protocols (80%). Compared with those in Europe and North America, sites from Asia (n = 20) reported institutional barriers less frequently, with the fewer eligible patients (40%, p = 0.0475), sponsors suspending or placing recruiting on hold (35%, p = 0.0615), and difficulty complying with protocols (45%, p = 0.0596). The most frequent institutional barriers from the rest of the world were fewer eligible patients (71%), difficulty complying with protocols (47%), and sponsors suspending or placing recruiting on hold (41%). Data on institutional barriers (n = 8) from Latin America were too sparse for regional comparison.
Overall, 24% of sites reported disruptions from trial participants owing to COVID-19 infection and 40% from exposure-related quarantine. Patient-specific challenges included access to trial site (49%), ability to travel (54%), and willingness to visit site (59%; Fig. 2). In addition, patient-specific concerns included fear of COVID-19 infection (83%), securing transportation (38%), travel restrictions (50%), and laboratory or radiology access (16%).
Patient willingness to visit the site was a consistent barrier reported across Europe, North America, and Asia. In North America, patient ability to visit the site was seen as less of a barrier, though not significantly different (p = 0.1366). Specifically, patients’ willingness to visit the site (62%) and patients’ ability to visit the site (62%) were recognized as major barriers in Europe, whereas they were at 70% and 35% in North America, 65% and 50% in Asia, and 59% and 76% in the rest of the world, respectively. Data on patient-specific barriers from Latin America were too sparse (n = 8) to make regional comparisons. International travel restrictions were a barrier for patient participation in sites that recruited patients from other countries (22% of sites surveyed; Supplementary Data 4).
The impact on lung cancer trials was substantial, with more than 50% of sites experiencing moderate or greater impacts across phase 1 to phase 3 trials, most frequently affecting chemotherapy (52% of sites), immunotherapy (51% of sites), and targeted therapy trials (38% of sites). As anticipated, trials with investigational agents that required infusion seem to be more affected than those with targeted therapies (Supplementary Data 4). We found little impact from COVID-19 vaccine trials, with 80% of sites not enrolling patients with lung cancer and 83% reporting the vaccine trials had no impact on lung cancer trial enrollment. Only 33% of the sites planned to discuss the choice of COVID-19 vaccine with lung cancer clinical trial patients.
Mitigation Strategies
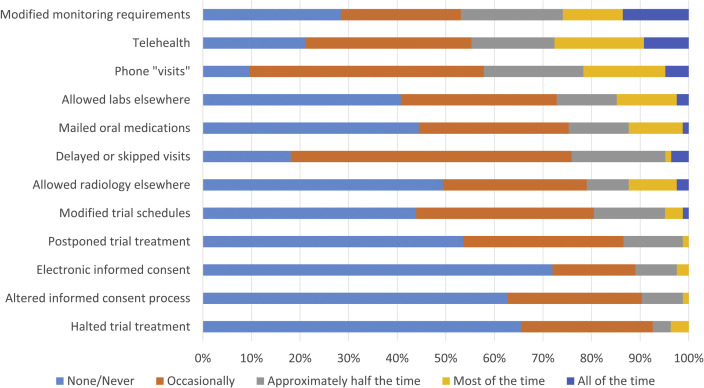
Sites implemented mitigation strategies to battle the barriers to clinical trial enrollment they faced from the pandemic. Mitigation strategies that sites implemented, by the percent using at least half, most, or all the time, were modified monitoring requirements (47%), telehealth visits (45%), phone visits (42%), and mail-order medications (25%; Fig. 3 ). Some sites allowed laboratory (27%) and radiology (21%) tests at nonstudy facilities, and a few implemented altered (10%) or electronic (11%) consent processes (Fig. 3).
Figure 3.
Site reported mitigation strategies to address challenges to enrollment in lung cancer clinical trials.
Telehealth (38%–50%) and phone visits (37%–50%) were among the top mitigation strategies in North America, Europe, and Asia. Mailed oral medications were more frequent in Europe compared with Asia or North America, though this was not statistically significant (39% versus 25% and 17%, p = 0.47). In North America, modified monitoring requirements were more frequent than in Europe or Asia (76% versus 39% and 24%, p = 0.0171). Sites in Asia were significantly more likely to use electronic consent (28% versus 6% and 0%, p = 0.0182) and may have allowed delayed or skipped visits more frequently (33% versus 17% and 11%, p = 0.27) than Europe or North America. Percentages of sites implementing mitigation strategies from Latin America and the rest of the world were generally similar to global percentages, although not compared statistically.
We asked sites to identify the most effective mitigation strategies used to fight the impact of the pandemic on clinical trials. Of the sites, 80% who used telehealth visits reported that it was an effective strategy, 85% reported remote collection of patient-reported symptoms was effective, and 85% reported off-site diagnostic or monitoring procedures (clinical evaluations, blood draws, imaging) was an effective strategy (Fig. 4 ). In addition, 89% felt remote consenting and 81% felt electronic signatures were effective when used. Delayed assessments (81%), institutional review board changes (89%), and use of liquid biopsy in lieu of tissue biopsy (83%) were also considered effective when implemented. We also asked the sites whether they would like to continue any of the adjustments to the lung cancer clinical trial processes after the pandemic. Telehealth visits (52%) were considered the most frequent adjustment that sites would like to continue, followed by remote monitoring (49%), electronic signature (47%), and remote patient-reported symptom collection (35%).
Figure 4.
Mitigation strategies sites identified as the most effective. IRB, Institutional Review Board.
Discussion
The IASLC international survey found significant declines in enrollment in lung cancer clinical trials across the world in the beginning of the COVID-19 pandemic. The magnitude of the declines varied across the globe with 13% decline in North America, 35% decline in Europe, 10% decline in Asia, and an apparent 88% decline in Latin America, although the sample size was smaller. Although the pandemic worsened throughout 2020, most enrollment declines were in the early months of the pandemic with few differences in enrollment in the past 3 months of 2020, likely owing to mitigation strategies.
The challenges at the institutional level centered on the ability to keep sites open, keep staff available, and keep staff and patients safe. This affected the delivery of standard care for patients with lung cancer.21 , 23, 24, 25, 26, 27 Patients with lung cancer had decreased use of outpatient services, hospital admissions, and surgical procedures during the first months of the pandemic.27 A large allocation of health resources toward caring for patients sick with COVID-19 made it more difficult for many institutions to maintain standard care.27 These strains on the system made allocation of time and resources for rigorous clinical trial monitoring, and ultimately, recruiting new patients to clinical trials less feasible. Previously described deficits in clinical trial screening processes for eligible patients were likely magnified at some sites owing to the reported staffing issues in this study.28 An overall reduction in screening and diagnoses may partially explain why sites in this study reported fewer identified patients who may have been trial eligible. We found no evidence that COVID-19 vaccine trials interfered with lung cancer trials, and two-thirds of the respondents did not plan to discuss vaccination with patients with lung cancer. Ensuring access to COVID-19 vaccination for patients and providers involved with lung cancer trials could provide additional mitigation against some of the barriers identified.
Patient-level challenges and concerns included COVID-19 infection or quarantine, fear of exposure to severe acute respiratory syndrome coronavirus 2, transportation to sites, and willingness to travel. Although some of these issues are specific to a time of crisis, transportation and the time it takes to fulfill study visits are well-established barriers to optimal trial enrollment.29 Even before the pandemic, efforts have been underway to provide equitable access to lung cancer clinical trials.30, 31, 32 Less than 5% of adults with cancer currently participate in clinical trials across the world, with noted disparities in enrollment by demographic factors.33
Across the world, the impact of the pandemic on health care delivery was influenced by differences in hospital capacity, intensive care unit capacity, national public health policies, and patient-level factors, such as transportation modality and availability.34 , 35 Nevertheless, we found that many of the challenges faced from the COVID-19 pandemic were similar for different geographic regions. Although the magnitude of these challenges varied somewhat by region, the barriers we have discussed were typically faced by 40% or more of sites in each region. We found that some mitigation strategies were used in similar proportions across regions, such as telehealth and phone visits, whereas the use of others such as modified monitoring requirements and electronic consent was variable by region. Observed differences could reflect variability in the challenges faced, opportunities available on the basis of specific regulatory requirements, or could be an artifact of regional response patterns to our survey. Overall, these data suggest that we face many of the same issues across the world, some solutions may be broadly applicable across regions, and others may benefit from tailoring to the population and setting.
The mitigation strategies that sites identified as most effective in reducing the impact of the COVID-19 pandemic were focused on greater flexibility. Sites used technology to provide more flexibility in “place” by implementing telehealth visits, remote monitoring, and remote diagnostics. Although not novel strategies per se, these were not frequently implemented before the pandemic. Telehealth was a top migration strategy, in both use and perceived effectiveness, across global regions. Historically, symptoms and toxicity data have been collected at in-person visits by physicians and research staff. Shifting interactions to an electronic format became a need that could be continued in places where it has been successfully implemented. The capacity to obtain consent remotely also seemed effective where implemented. Telehealth was adopted early in the COVID-19 crisis and has been strongly supported by the European Society for Medical Oncology recommendations for the management of lung cancer and the American Society for Clinical Oncology, among others.36 , 37 There is growing consensus across medical disciplines that the increased use of telehealth is here to stay.38 Nevertheless, reimbursement barriers that impede clinicians’ ability to conduct telehealth are still in place in some countries and should be addressed at the policy level.
Collection of laboratory and imaging data was a major challenge met during the pandemic with loosening of regulations allowing for use of local laboratories and imaging centers. This strategy could be continued in future trials, including home collection of blood tests. Certification and billing at remote sites are global issues that will require future attention. Re-assessment of the need for the extensive laboratory and imaging end points built into many trials may also be considered to improve trial feasibility.
Allowing mail-order medications was also identified as an effective strategy for oral therapies, which was used most frequently at sites in Europe. This may explain why infusion-related trials were more affected as such trials require travel to the trial site. In addition, mitigation strategies that provided more flexibility in “time,” including delaying study visits and assessments, were found as highly effective. These strategies leveraged modern technology to address barriers to health care delivery, highlighting the need for continual modernization of processes of care delivery. Electronic health records and standardized electronic treatment plans were used to reduce research staff time and reduce article records. Digital health technology such as remote vital sign monitoring, electrocardiogram monitoring, and pulse oximetry monitoring was used in some institutions. The pandemic era has highlighted the need to improve our use of technologic solutions to improve health care.
Agencies including the National Cancer Institute Cancer Therapy Evaluation Program, U.S. Food and Drug Administration, The European Commission, the European Medicines Agency, and national Head of Medicines Agencies, among others, have provided continued guidance on the management of clinical trials during the pandemic.39, 40, 41 Moving forward, the American Association for Cancer Research has called for better use of technology to improve clinical trials across the cancer continuum and incorporate COVID-19 guidance beyond the current crisis.22 The barriers and mitigation strategies we identified in this survey support this call. Moving forward, they suggest a greater role for the integration of electronic medical records and electronic data collection and an overall streamlining of cancer clinical trials.22 Our survey illustrates that these approaches are consistent with the needs of lung cancer investigators and patients across the world.
Trial sponsors, trial sites, and regulatory bodies should consider using trials with less collection of marginally important data, more flexibility in clinical trials (such as allowing for remote monitoring for late-phase trials), while maintaining the necessary scientific rigor that is a hallmark of clinical trials. Well-applied strategies could improve flexibility and maintain scientific rigor. Some changes may be appropriate globally whereas others may require tailoring to the region or country. The future of lung cancer advances continues to rely on good clinical trials, and we should redouble our efforts to increase accrual both during and after the pandemic.
This study achieved many of the goals determined at the onset, but has limitations. We were not able to attain complete information from every site requested, which led to missing data in the surveys. The amount of information requested from sites, differing global regulatory standards for providing data, and clinical challenges during the pandemic likely contributed to this limitation. The purposive sample scheme was used in favor of probabilistic sampling to allow more targeted recruitment of sites in under-represented areas, while not excluding any willing participants from providing potentially useful information. Nevertheless, we attained valuable quantitative and qualitative information on the impact of the pandemic from many sites representing multiple countries. To our knowledge, this is the largest study of the impact of COVID-19 on lung cancer clinical trials.
In conclusion, the COVID-19 pandemic had a substantial impact on enrollment in lung cancer clinical trials across the world. Trial sites and regulatory bodies adapted with mitigation strategies largely aimed to provide more flexibility and leverage modern technology. Despite the acceleration of COVID-19 cases in the later months of 2020, enrollment numbers in lung cancer clinical trials started to rebound. A more flexible approach to clinical trials, removing unnecessary barriers and leveraging technology, may improve enrollment and access to clinical trials, even beyond the COVID-19 pandemic.
CRediT Authorship Contribution Statement
Matthew Smeltzer: Writing - original draft, Conceptualization, Methodology, Formal analysis.
Paul Bunn: Writing - original draft, Conceptualization, Methodology.
Giorgio Scagliotti, Heather Wakelee, Tetsuya Mitsudomi, Fred Hirsch: Writing - review and editing, Conceptualization, Methodology, Data curation.
Upal Roy, Russell Clark, Renee Arndt: Writing - review & editing, Conceptualization, Methodology.
Clayton Pruett: Conceptualization.
Karen Kelly, Peter Ujhazy, Melissa Johnson, Carlos Barrios, Fabrice Barlesi, Yesim Eralp: Writing - review and editing, Data Curation.
Acknowledgments
The authors thank all the investigators and regional champions who supported this project through recruitment of respondents, supplying trial data, or completing the Action Survey. Dr. Pruett passed away on July 24, 2021.
Footnotes
Disclosure: The International Association for the Study of Lung Cancer received funding for this project from Amgen, AstraZeneca, Daiichi Sankyo, Foundation Medicine, Novartis, and Regeneron. Dr. Bunn has received consulting income from AstraZeneca, Ascentage, Bristol-Myers Squibb, C-Stone, Imidex, Merck, and Verastem not related to this project. Dr. Smeltzer has worked as a paid research consultant for the Association of Community Cancer Centers. Dr. Scagliotti received honoraria from AstraZeneca, Eli Lilly, Merck Sharp & Dohme, Pfizer, Roche, Johnson & Johnson, and Takeda; consulting or advisory role for Eli Lilly, Beigene, and AstraZeneca; received institutional research funding from Eli Lilly and Merck Sharp & Dohme; and received travel, accommodations from Bayer, all not related to the current project. Dr. Roy received institutional research funding from AstraZeneca, Eli Lilly, Merck, Blueprint Medicine, Genentech, G1 Therapeutics, Takeda, Bristol-Myers Squibb, and Janssen not related to the current project. Dr. Barlesi received honoraria from AstraZeneca, Bayer, Bristol-Myers Squibb, Boehringer Ingelheim, Eli Lilly Oncology, F. Hoffmann–La Roche Ltd., Novartis, Merck, Mirati, Merck Sharp & Dohme, Pierre Fabre, Pfizer, Seattle Genetics, and Takeda. Dr. Kelly has received honoraria from AbbVie, Amgen, AstraZeneca, Debiopharm, Daiichi Sankyo, EMD Serono, Genentech, Genmab, Janssen, Novartis, Regeneron, and Sanofi/Genzyme. Dr. Mitsudomi received advisory/lecture fees from AstraZeneca, Novartis, Chugai, Boehringer Ingelheim, Pfizer, Roche, Merck Sharp & Dohme, Thermo Fisher, Roche Diagnostics, Bristol-Myers Squibb, Ono Pharmaceuticals, Taiho, Takeda, Amgen, Johnson and Johnson, Janssen Pharma, Puma, Beigene, Bridge Biotherapeutics, Eli Lilly, Daiichi Sankyo, and Thermo Fisher and research funding from AstraZeneca, Chugai, Boehringer Ingelheim, Pfizer, Taiho, Ono, Merck Sharp & Dohme, Daiichi Sankyo, Eli Lilly, Ethicon, and Bridge Biotherapeutics. Dr. Johnson received institutional research funding from AbbVie, Acerta, Adaptimmune, Amgen, Apexigen, Arcus Biosciences, Array BioPharma, Artios Pharma, AstraZeneca, Atreca, BeiGene, BerGenBio, Boehringer Ingelheim, Calithera Biosciences, Checkpoint Therapeutics, Corvus Pharmaceuticals, Curis, CytomX, Daiichi Sankyo, Dracen Pharmaceuticals, Dynavax, Eli Lilly, EMD Serono, Genentech/Roche, Genmab, Genocea Biosciences, GlaxoSmithKline, Gritstone Oncology, Guardant Health, Harpoon, Hengrui Therapeutics, Immunocore, Incyte, Janssen, Jounce Therapeutics, Kadmon Pharmaceuticals, Loxo Oncology, Lycera, Merck, Mirati Therapeutics, Neovia Oncology, Novartis, OncoMed Pharmaceuticals, Pfizer, PMV Pharmaceuticals, Regeneron Pharmaceuticals, Ribon Therapeutics, Rubius Therapeutics, Sanofi, Seven and Eight Biopharmaceuticals/Birdie Biopharmaceuticals, Shattuck Labs, Silicon Therapeutics, Stem CentRx, Syndax Pharmaceuticals, Takeda Pharmaceuticals, Tarveda, TCR2 Therapeutics, TMUNITY Therapeutics, University of Michigan, and WindMIL; consulting or advisory role for AbbVie, Achilles Therapeutics, Amgen, AstraZeneca, Atreca, Boehringer Ingelheim, Bristol-Myers Squibb, Calithera Biosciences, Checkpoint Therapeutics, CytomX Therapeutics, Daiichi Sankyo, Editas Medicine, Eisai, EMD Serono, G1 Therapeutics, Genentech/Roche, GlaxoSmithKline, Gritstone Oncology, Guardant Health, Ideaya Biosciences, Incyte, Janssen, Eli Lilly, Loxo Oncology, Merck, Mirati Therapeutics, Novartis, Pfizer, Ribon Therapeutics, Sanofi, Association of Community Cancer Centers, and WindMIL. The remaining authors declare no conflict of interest.
Note: To access the supplementary material accompanying this article, visit the online version of the Journal of Thoracic Oncology at www.jto.org and at https://doi.org/10.1016/j.jtho.2022.01.017
Supplementary Data
References
- 1.Sung H., Ferlay J., Siegel R.L., et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Zhang Y., Luo G., Etxeberria J., Hao Y. Global patterns and trends in lung cancer incidence: a population-based study. J Thorac Oncol. 2021;16:933–944. doi: 10.1016/j.jtho.2021.01.1626. [DOI] [PubMed] [Google Scholar]
- 3.Schenk E.L., Patil T., Pacheco J., Bunn P.A., Jr. Innovation-based optimism for lung cancer outcomes. Oncologist. 2021;26:e454–e472. doi: 10.1002/onco.13590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Howlader N., Forjaz G., Mooradian M.J., et al. The effect of advances in lung-cancer treatment on population mortality. N Engl J Med. 2020;383:640–649. doi: 10.1056/NEJMoa1916623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Provencio M., Mazarico Gallego J.M., Calles A., et al. Lung cancer patients with COVID-19 in Spain: GRAVID study. Lung Cancer. 2021;157:109–115. doi: 10.1016/j.lungcan.2021.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lei H., Yang Y., Zhou W., et al. Higher mortality in lung cancer patients with COVID-19? A systematic review and meta-analysis. Lung Cancer. 2021;157:60–65. doi: 10.1016/j.lungcan.2021.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yang B., Choi H., Lee S.K., et al. Risk of coronavirus disease 2019 occurrence, severe presentation, and mortality in patients with lung cancer. Cancer Res Treat. 2021;53:678–684. doi: 10.4143/crt.2020.1242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Passaro A., Bestvina C., Velez Velez M., Garassino M.C., Garon E., Peters S. Severity of COVID-19 in patients with lung cancer: evidence and challenges. J Immunother Cancer. 2021;9 doi: 10.1136/jitc-2020-002266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Garassino M.C., Whisenant J.G., Huang L.C., et al. COVID-19 in patients with thoracic malignancies (TERAVOLT): first results of an international, registry-based, cohort study. Lancet Oncol. 2020;21:914–922. doi: 10.1016/S1470-2045(20)30314-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tian J., Yuan X., Xiao J., et al. Clinical characteristics and risk factors associated with COVID-19 disease severity in patients with cancer in Wuhan, China: a multicentre, retrospective, cohort study. Lancet Oncol. 2020;21:893–903. doi: 10.1016/S1470-2045(20)30309-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Greenwood E., Swanton C. Consequences of COVID-19 for cancer care—a CRUK perspective. Nat Rev Clin Oncol. 2021;18:3–4. doi: 10.1038/s41571-020-00446-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bilich T., Roerden M., Maringer Y., et al. Preexisting and post-COVID-19 immune responses to SARS-CoV-2 in patients with cancer. Cancer Discov. 2021;11:1982–1995. doi: 10.1158/2159-8290.CD-21-0191. [DOI] [PubMed] [Google Scholar]
- 13.Unger J.M., Xiao H., LeBlanc M., Hershman D.L., Blanke C.D. Cancer clinical trial participation at the 1-year anniversary of the outbreak of the COVID-19 pandemic. JAMA Netw Open. 2021;4 doi: 10.1001/jamanetworkopen.2021.18433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gomez J.E. OAO1.01 Analysis of lung cancer patients receiving SARS-CoV-2 vaccines revealed a minority subset with poor antibody responses relative to controls. J Thorac Oncol. 2021;16(suppl):S848. [Google Scholar]
- 15.Krammer F., Srivastava K., Alshammary H., et al. Antibody responses in seropositive persons after a single dose of SARS-CoV-2 mRNA vaccine. N Engl J Med. 2021;384:1372–1374. doi: 10.1056/NEJMc2101667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Thakkar A., Gonzalez-Lugo J.D., Goradia N., et al. Seroconversion rates following COVID-19 vaccination among patients with cancer. Cancer Cell. 2021;39:1081–1090.e2. doi: 10.1016/j.ccell.2021.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Unger J.M., Blanke C.D., LeBlanc M., Hershman D.L. Association of the coronavirus disease 2019 (COVID-19) outbreak with enrollment in cancer clinical trials. JAMA Netw Open. 2020;3 doi: 10.1001/jamanetworkopen.2020.10651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fleury M.E., Farner A.M., Unger J.M. Association of the COVID-19 outbreak with patient willingness to enroll in cancer clinical trials. JAMA Oncol. 2021;7:131–132. doi: 10.1001/jamaoncol.2020.5748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tan A.C., Ashley D.M., Khasraw M. Adapting to a pandemic—conducting oncology trials during the SARS-CoV-2 pandemic. Clin Cancer Res. 2020;26:3100–3103. doi: 10.1158/1078-0432.CCR-20-1364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Eng C., Chen E.Y., Rogers J., et al. Moving beyond the momentum: innovative approaches to clinical trial implementation. JCO Oncol Pract. 2021;17:607–614. doi: 10.1200/OP.20.00701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.The Cancer Letter NCTN Group Chairs: cancer trials take backseat to clinical care amid COVID-19 pandemic. https://cancerletter.com/articles/20200410_1/
- 22.Flaherty K.T., Doroshow J.H., Galbraith S., et al. Rethinking cancer clinical trial conduct induced by COVID-19: an academic center, industry, government, and regulatory agency perspective. Cancer Discov. 2021;11:1881–1885. doi: 10.1158/2159-8290.CD-21-0850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Singh N. Management of lung cancer during the COVID-19 pandemic: practical solutions for resource-constrained settings from adaptions of an international consensus. Lung India. 2020;37:381–383. doi: 10.4103/lungindia.lungindia_379_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bertaglia V., Reale M.L., Bironzo P., et al. Italian survey on the clinical management of non-small cell lung cancer patients during the COVID-19 pandemic: a lesson for the second wave. Crit Rev Oncol Hematol. 2021;157 doi: 10.1016/j.critrevonc.2020.103189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bakhribah H., Alfayea T. Immunotherapy and coronavirus disease 2019: challenges and possibilities. J Thorac Oncol. 2020;15:1709–1710. doi: 10.1016/j.jtho.2020.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Whisenant J.G., Trama A., Torri V., et al. TERAVOLT: thoracic cancers international COVID-19 collaboration. Cancer Cell. 2020;37:742–745. doi: 10.1016/j.ccell.2020.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fu R., Wu L., Zhang C., et al. Real-world scenario of patients with lung cancer amid the coronavirus Disease 2019 pandemic in the People’s Republic of China. JTO Clin Res Rep. 2020;1 doi: 10.1016/j.jtocrr.2020.100053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Campillo-Gimenez B., Buscail C., Zekri O., et al. Improving the pre-screening of eligible patients in order to increase enrollment in cancer clinical trials. Trials. 2015;16:15. doi: 10.1186/s13063-014-0535-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ford J.G., Howerton M.W., Lai G.Y., et al. Barriers to recruiting underrepresented populations to cancer clinical trials: a systematic review. Cancer. 2008;112:228–242. doi: 10.1002/cncr.23157. [DOI] [PubMed] [Google Scholar]
- 30.Garcia S., Bisen A., Yan J., et al. Thoracic oncology clinical trial eligibility criteria and requirements continue to increase in number and complexity. J Thorac Oncol. 2017;12:1489–1495. doi: 10.1016/j.jtho.2017.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Forde P.M., Bonomi P., Shaw A., et al. Expanding access to lung cancer clinical trials by reducing the use of restrictive exclusion criteria: perspectives of a multistakeholder working group. Clin Lung Cancer. 2020;21:295–307. doi: 10.1016/j.cllc.2020.02.008. [DOI] [PubMed] [Google Scholar]
- 32.Chino F., Zafar S.Y. Financial toxicity and equitable access to clinical trials. Am Soc Clin Oncol Educ Book. 2019;39:11–18. doi: 10.1200/EDBK_100019. [DOI] [PubMed] [Google Scholar]
- 33.Institute of Medicine (US) Forum on Drug Discovery, Development, and Translation . The National Academies Press; Washington, DC: 2010. Transforming Clinical Research in the United States: Challenges and Opportunities: Workshop Summary. [PubMed] [Google Scholar]
- 34.Sen-Crowe B., Sutherland M., McKenney M., Elkbuli A. A closer look into global hospital beds capacity and resource shortages during the COVID-19 pandemic. J Surg Res. 2021;260:56–63. doi: 10.1016/j.jss.2020.11.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.COVIDSurg Collaborative Effect of COVID-19 pandemic lockdowns on planned cancer surgery for 15 tumour types in 61 countries: an international, prospective, cohort study. Lancet Oncol. 2021;22:1507–1517. doi: 10.1016/S1470-2045(21)00493-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Passaro A., Addeo A., Von Garnier C., et al. ESMO Management and treatment adapted recommendations in the COVID-19 era: lung cancer. ESMO Open. 2020;5(suppl 3) doi: 10.1136/esmoopen-2020-000820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zon R.T., Kennedy E.B., Adelson K., et al. Telehealth in oncology: ASCO standards and practice recommendations. JCO Oncol Pract. 2021;17:546–564. doi: 10.1200/OP.21.00438. [DOI] [PubMed] [Google Scholar]
- 38.Krueger C.A., Mont M.A., Backstein D.J., et al. COVID will end but telemedicine may be here to stay. J Arthroplasty. 2021;36:789–790. doi: 10.1016/j.arth.2021.01.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.US Food and Drug Administration FDA guidance on conduct of clinical trials of medical products during the COVID-19 public health emergency. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/fda-guidance-conduct-clinical-trials-medical-products-during-covid-19-public-health-emergency
- 40.Mooney MM, McCaskill-Stevens WM. Interim Guidance for Patients on Clinical Trials Supported by the NCI Cancer Therapy Evaluation Program and the NCI Community Oncology Research Program (NCORP). https://ctep.cancer.gov/content/docs/Memorandum_on_Interim_Guidance_for_Clinical_Trial_Activities_Affected_by_the_Novel_Coronavirus-3-13-2020.pdf. Accessed September 17, 2021.
- 41.European Commission. Commission recommendation. https://ec.europa.eu/info/sites/default/files/recommendation_on_apps_for_contact_tracing_4.pdf Accessed September 17, 2021.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.