Abstract
Licorice (Glycyrrhiza glabra) is a well-known natural herb used to treat different ailments since ancient times. Glycyrrhizin (GL), which is the primary triterpenoid compound of licorice extract, has been known to have broad-spectrum pharmacological effects. GL is cleaved into glucuronide and the aglycone, glycyrrhetinic acid (GA), which exists in two stereoisomeric forms: 18α- and 18β-GA. It is well documented that GL and GA have great potential as anti-inflammatory, anticancer, antiviral, anti-diabetic, antioxidant, and hepatoprotective agents. Studies undertaken during the coronavirus disease 2019 pandemic suggest that GL is effective at inhibiting the viral replication of severe acute respiratory syndrome coronavirus 2. The anticancer effects of GL and GA involve modulating various signaling pathways, such as the phosphatase and tensin homolog/phosphatidylinositol 3-kinase/protein kinase B pathway, the mitogen-activated protein kinase, and the mammalian target of rapamycin/signal transducer and activator of transcription 3, which are mainly involved in regulating cancer cell death, oxidative stress, and inflammation. The potential of GL and GA in preventing cancer development and suppressing the growth and invasion of different cancer types has been reviewed in this paper. This review also provides molecular insights on the mechanism of action for the oncopreventive and oncotherapeutic effects of GL and its derivative, GA, which could help develop more specific forms of these agents for clinical use.
Keywords: Licorice, Glycyrrhizin, Glycyrrhetinic acid, Cancer, HMGB1, COX-2, Inflammation
Graphical Abstract
1. Introduction
Licorice or liquorice is a herbaceous perennial legume copiously cultivated over many places worldwide. The Glycyrrhiza genus consists of about 30 species belonging to the family of Leguminosae. The most common species are Glycyrrhiza uralensis Fisch., G. inflata Bat., G. glabra, G. aspera, G. korshinskyi, and G. eurycarpa [1], [2], [3]. Licorice is widely utilized as a sweetening and flavoring material in the food industry and as a de-bittering agent in the tobacco industry. Over 400 bioactive ingredients and secondary metabolites have been identified in the dried roots and rhizomes of Glycyrrhiza species [2]. There are two major constituents of licorice. The first is the triterpene saponins, which exist in a glucuronide form and aglycone form, known as glycyrrhetinic acid (GA). The aglycone form of oleanane pentacyclic triterpene is characterized by hydrophilic functional groups, such as a hydroxyl group at C-3, a carboxyl group at C-30, and a ketone group at C-11, causing GA to have high water solubility [4], [5]. The second constituent is a group of compounds known as flavonoids. The chemical structure of flavonoids is generally composed of two phenyl rings that bind to a heterocyclic ring with several chemical functional groups. Flavonoids comprise a heterogeneous group of compounds such as flavanones or flavanonols, isoflavones, flavonols, chalcones, dihydrochalcones, and flavan-3-ol. A recently published review article has addressed the molecular mechanisms of licorice flavonoids in cancer [6].
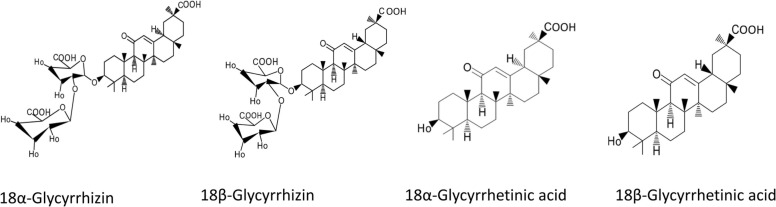
Glycyrrhizin (GL), or glycyrrhizic acid (GZA), is an isomer of 18α-glycyrrhizin and 18β-glycyrrhizin ( Fig. 1). It is the major saponin of a pentacyclic derivative of the β-amyrine type. The dried licorice root extract is made up of around 4–25% of the saponin, of which GL constitutes about 3–13% [7], [8]. When taken orally, GL has low oral bioavailability and can only be detected at very low concentrations after a single dietary ingestion. In humans, the major ingredient of licorice, GZA, is hydrolyzed (pre-systemic hydrolysis) to GA by intestinal bacteria that have a particular ß-glucuronidase enzyme [9]. GL is cleaved by the action of the intestinal bacteria to one molecule of GA and two glucuronide molecules [10]. GA has two pentacyclic stereoisomers, known as 18α- and 18β-GA (Fig. 1) [7]. GL has various pharmacological effects, including anticancer, anti-inflammatory, antioxidant, and antiviral effects [11], [12], [13], [14], [15], [16], [17]. The antiviral activity has been studied previously against hepatitis B virus, hepatitis C virus, and HIV [18], [19], [20]. More recently, researchers have found interesting data suggesting the potential of GL in the treatment of the novel coronavirus [21], [22], [23]. Furthermore, a study found that GL inhibited the viral replication of severe acute respiratory syndrome coronavirus 2 by targeting the main viral protease [21]. In addition, another study found that GL may be useful in reducing lung inflammation by downregulating angiotensin-converting enzyme 2 expression in the lungs [23]. However, further investigation is required for a comprehensive understanding of the mechanism of action of GL in the treatment of COVID-19.
Fig. 1.
Chemical structure of isomeric forms of GL and GA.
Cancer is a heterogeneous disease group characterized by uncontrolled cellular growth and metastasis to a distant site in the body. Cancer is the second leading cause of mortality worldwide and in the USA in 2020. In 2021, the United States is expected to see 1,898,160 new cancer cases and 608,570 cancer deaths [24]. Cancer resistance is a significant problem that demands the usage of multiple targeted treatments [25]. However, the high toxicity profile of synthetic chemical compounds significantly impacts patient survival and lifestyle. Therefore, newer therapies with less toxic profiles and novel targets is warranted and there has been extensive ongoing research on the use of naturally derived compounds from the plant and marine worlds in the treatment of several different cancers [26], [27], [28]. More than 60% of synthetic pharmaceutical drugs come from natural sources; most are derived from plants, marine life, bacteria, and fungi, with plant-derived active compounds accounting for 75% of anticancer medications [29], [30], [31]. One of the advantages of using drugs derived from natural sources over small synthetic molecules is structural diversity, which allows these drugs to exhibit their effects on many molecular targets. In contrast, a small molecule has a limited number of targets [32], [33]. Some of the most widely used chemotherapeutic agents that are derived from natural sources are Etoposide, Topotecan, Irinotecan, Paclitaxel, Vincristine, Vinblastine, Brentuximab vedotin, Clofarabine, Everolimus, and many other drugs [34], [35], [36], [37], [38], [39], [40].
Even though cancer treatment gets a lot of attention and oncoprevention is often overshadowed by it, the importance of oncopreventive research is paramount. Oncopreventive research aims to decrease cancer incidence and prevalence, reducing cancer-associated morbidity and mortality. Although various compounds of licorice have significant pharmacological effects, we will highlight the anticancer properties of GL and its derivatives. To our knowledge, this is the first review to provide an insight into the common mechanistic pathways responsible for the oncopreventive and oncotherapeutic potential of GL and its derivatives.
2. Pharmacokinetics of GZA and GA
Many studies have investigated the pharmacokinetic behavior of GZA and its metabolites after oral and intravenous administration [10], [41], [42]. GZA is detected in the plasma of rats when administered in the dose range of 50–500 mg/kg [43], [44]. In humans, following 100–800 mg oral administration, GZA was not detected in plasma [45]. GL is partly absorbed from the gastrointestinal tract. Oral administration of GL results in pre-systemic metabolism, causing GL to be hydrolyzed to GA by intestinal bacteria [45]. A specialized β-glucuronidase in the bacteria isolated from human intestinal flora, such as Eubacterium sp., Ruminococcus sp., and Clostridium innocuum., showed GL hydrolyzing activity [46], [47]. After oral administration of GL (100 mg), its metabolite GA appeared in plasma (less than 200 ng/mL) [45]. GZA and GA bind plasma albumin extensively in humans and rats [48]. The plasma concentration of GZA and GA after an intravenous administration in rats exhibits a biphasic pattern (distribution phase is followed by a slower elimination phase) [48]. GA is metabolized to glucuronide conjugate in the liver and rapidly excreted into the bile [48].
3. Methodology for literature search and study selection
In this review, we systematically searched the literature using the controlled search terms with various conjunctions: (Glycyrrhizin [TIAB] OR glycyrrhetinic acid [TIAB]) AND (Neoplasia [TIAB] OR Neoplasias [TIAB] OR Neoplasm [TIAB] OR Tumors [TIAB] OR Tumor [TIAB] OR Cancer [TIAB] OR Cancers [TIAB] OR Malignancy [TIAB] OR Malignancies [TIAB] OR “Malignant Neoplasms” [TIAB] OR “Malignant Neoplasm” [TIAB] OR “Neoplasm, Malignant” [TIAB] OR “Neoplasms, Malignant” [TIAB] OR “Benign Neoplasms” [TIAB] OR “Neoplasms, Benign” [TIAB] OR “Benign Neoplasm” [TIAB] OR “Neoplasm, Benign” [TIAB]) through PubMed, Web of Science, Cochrane library, and Scopus databases. Related citations of every included article were also manually examined. We only included articles published in the English language. To meet our purpose for this narrative review, we included all the mechanistic studies and clinical trials during screening and data extraction.
4. Anticancer mechanisms of action of licorice triterpenoids
4.1. Antioxidant effects
Glutathione (GSH) is a non-protein thiol present in high amounts in tissue. Its importance comes from its ability to regulate redox hemostasis in the cells and tissues. It protects the cells from highly reactive oxygen and nitrogen species that cause cellular damage, such as superoxide and nitrogen peroxide [49], [50]. GSH is a crucial signal transduction molecule that regulates many cellular functions, including proliferation, differentiation, and apoptosis. GSH dysregulation has been implicated in tumor initiation, progression, and resistance to treatment [51], [52]. More recently, numerous innovative drugs have been studied to target the GSH antioxidant system [53], [54]. The antioxidant property of GA has been studied in murine skin models using 7,12-dimethyl benz(a)anthracene (DMBA)/TPA to induce skin cancer. The study elaborates that GA inhibits ornithine decarboxylase (ODC), which causes glutathione depletion. This might suggest that the oncopreventive property of GA is a result of its antioxidant properties. GA exerts its effect by inhibiting ornithine decarboxylase, which decreases oxidative stress, ultimately suppressing tumor formation [55].
4.2. Anti-inflammatory and immunomodulatory effects
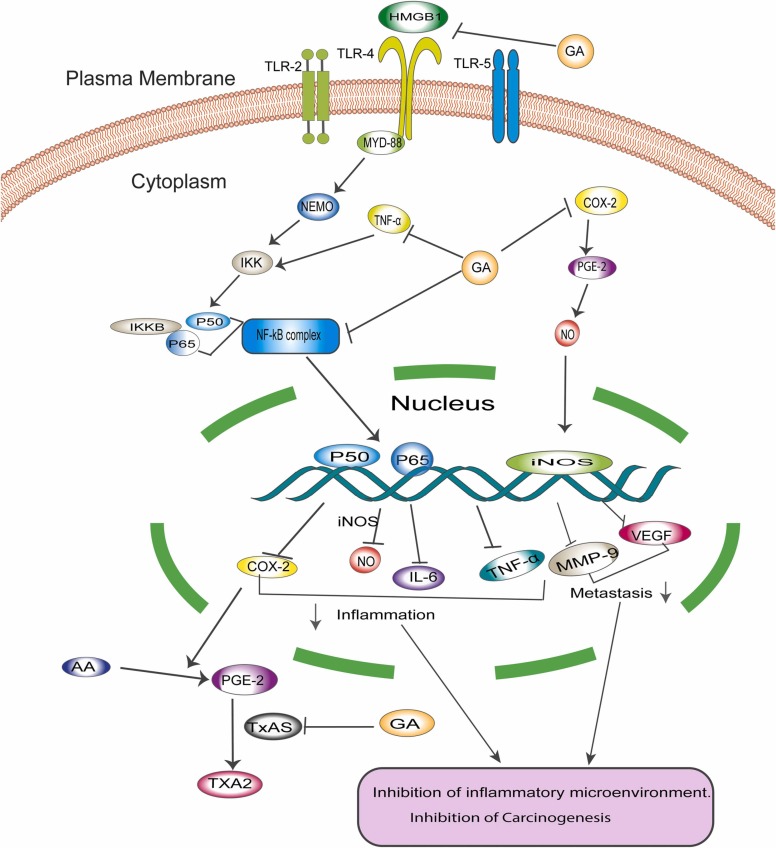
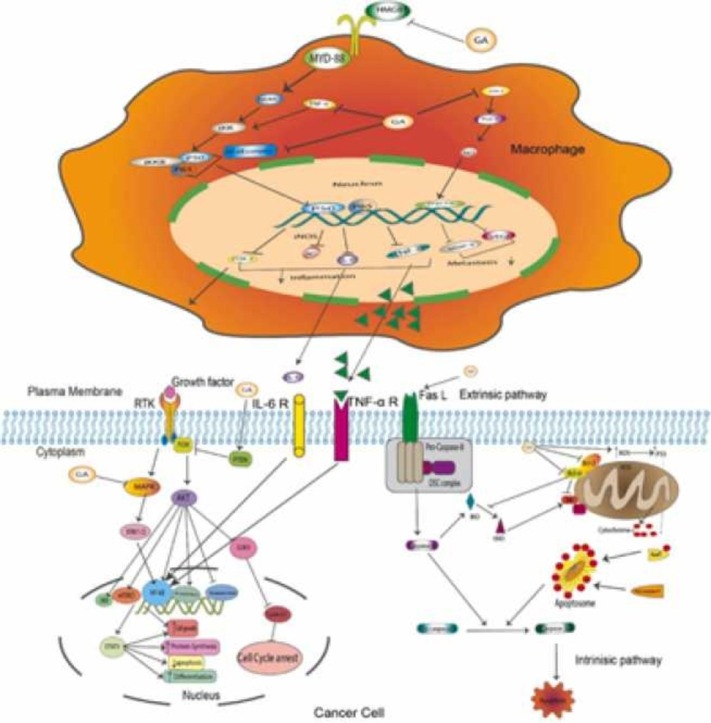
Chronic inflammation is a well-established contributor to cancer progression. Approximately 20% of cancers have been linked to chronic inflammation [56], [57]. In 1863, Virchow posed that tumors could emanate from sites of chronic inflammation, and many mechanistic, clinical, and epidemiological studies demonstrate the correlation between chronic inflammation and cancer progression [58]. Proinflammatory molecules such as cyclooxygenase 2 (COX-2) and its downstream molecule, prostaglandin E2 (PGE2), play a vital role in the inflammatory process by inducing a cell-mediated immune response that causes tissue remodeling and neovascularization ( Fig. 2), consequently facilitating cancer progression [59].
Fig. 2.
Depicts the anti-inflammatory and immunomodulatory role of GA. GA interferes with a different pathway to exhibit anticancer properties by controlling the inflammatory microenvironment that enhances cancer progression and survival. COX-2, its downstream PGE2, and TxAS are key players controlling cancer inflammation. NF-κB affects the expression of many essential molecules in inflammation and the immune response.
COX-2 elevated expression is associated with cancer progression [60]. Cao et al. [61] showed that 18β-glycyrrhetinic acid (GA) inhibits the growth and progression of gastric epithelial mucosa in K19-C2mE transgenic (Tg) mice via inhibition of COX-2 enzyme expression and ameliorates the inflammatory microenvironment [61]. Downstream to COX-2, thromboxane synthase (TxAS) is shown to be overexpressed in non-small cell lung cancer (NSCLC) tissue specimens and is responsible for the poor prognosis of lung cancer [62], [63]. TxAS can synthesize thromboxane-A2 (TxA2) from prostaglandin-H2 [64]. Previous studies have demonstrated that GA reduced both the transcription and protein expression of TxAS in NSCLC cell lines A549 and NCI-H460. GA-mediated inhibition of TxAS further suppresses the activation of ERK and downstream transcriptional factor CREB, which plays a vital role in cancer pathogenesis and promotes cell proliferation by increasing the activity of TxA2 [65], [66], [67]. In addition, TxAS inhibition has been shown to significantly increase the sensitivity of bladder cancer to Cisplatin treatment, suggesting that GA-mediated inhibition of TxAS may enhance sensitivity to conventional treatment and consequently reduce drug resistance [67], [68], [69].
To investigate the anti-inflammatory role of GL and GA in affecting carcinogenesis, Khan et al. [70] explored the therapeutic potential of GZA against the development of colonic precancerous lesions, such as aberrant crypt foci (ACF) and mucin depleted foci (MCF), in Wistar rats exposed to the carcinogen, 1,2-dimethylhydrazine (DMH). GZA, in a dose of 15 mg/kg, was shown to reduce the number of MDF lesions significantly compared to ACF, demonstrating the ability of GZA to prevent the growth of one of the early precancerous events in colon cancer. In addition, GZA has also been shown to attenuate loss of mucin in the colon, which consequently lowers mast cell infiltration to the submucosal layer. Moreover, GZA was found to inhibit nuclear factor kappa B (NF-κB) expression in colonic cells, leading to the deactivation of inflammatory mediators such as COX-2, inducible nitric oxide synthase (iNOS), tumor necrosis factor-alpha (TNF-α), and vascular endothelial growth factor (VEGF) [70]. Additionally, a recent study investigated the role of GL in carcinogenesis after exposing ICR mice to the carcinogen, azoxymethane (AOM)/dextran sodium sulfate (DSS). The study results elaborate that GL decreases the degree of dysplasia and infiltration of inflammatory cells within the colonic mucosa 20 weeks after treatment in comparison to the colonic mucosa of mice treated solely with the carcinogen and inflammatory agent. The study also found that GL reduces the expression of the inflammatory cytokine high-mobility group box 1 (HMGB1) [71]. HMGB1 binds to toll-like receptor 4 (TLR4) on the surface of macrophages and activates the myeloid differentiation 88 and NF-κB essential modulator (MyD88-NEMO) pathway, which ultimately activates NF-κB [72]. NF-κB inhibition results in a decrease in the expression of the proinflammatory genes, including iNOS and COX-2, which attenuates the inflammatory response and carcinogenesis [71].
Elevated expression of COX-2 and its product PGE2 were associated with immune evasion and increased cancer resistance in melanoma [73], [74]. Treatment of B16F10 melanoma cells with GA results in a decrease in the mRNA and protein expression levels of both COX-2 and PGE2, indicates that GA decreases melanoma cell progression and induces apoptosis [75]. Additionally, COX-2 and PGE2 were shown to stimulate induction of T regulatory cells (Treg), which are key mediators of tolerance to the antitumor effect [68], [76]. Therefore, by reducing the expression of COX-2 and PGE2, GA is shown to downregulate the immune suppression of FOXP3+Treg cells, and consequently reduce their production of anti-inflammatory cytokines IL-10 and TGF-β [75]. The D11b+Gr1+Myeloid-derived suppressor cells (MDSCs) are a constituent of immature myeloid cells that induce immunosuppression in melanoma cells [77]. MDSCs that were isolated from mice and treated with GA for 48 h showed significantly reduced PGE2 expression. In addition, pSTAT-3 was also decreased in a significant manner, indicating that GA inhibits the immunosuppression of MDSCs by downregulating STAT-3 phosphorylation [75].
4.3. Anti-estrogenic effects
One study demonstrated a significantly decreased incidence of endometrial adenocarcinoma and atypical endometrial hyperplasia in ovariectomized mice that were fed an estrogen-containing diet and treated with glycyrrhizin compared to mice that were fed the estrogen-containing diet alone [78]. Endometrial hyperplasia is closely associated with the development of endometrial adenocarcinoma. Therefore, the inhibitory action of GL against the incidence of both atypical endometrial hyperplasia and endometrial adenocarcinoma provides promising evidence of glycyrrhizin’s chemopreventive effects against endometrial cancer. Endometrial cancer is also associated with high levels of estrogen in the plasma. Estrogen stimulates cell proliferation in the endometrium, which leads to endometrial hyperplasia and increases the risk of developing cancer [79]. GL has been shown to have anti-estrogenic effects in mice and rats at dose ratios of 500:1 and 1000:1 of glycyrrhizin:estradiol-17β, suggesting that it may exert its chemopreventive effects by acting on the estrogen receptor [80]. There is also evidence that treatment of ovariectomized mice on an estrogen-containing diet with GL significantly reduced the expression of COX-2 mRNA in their uteri compared to mice that were fed the estrogen-containing diet without any additional treatment [78]. These findings indicate that GL could inhibit endometrial carcinogenesis by either competitively inhibiting the binding of estrogen to its receptor or by downregulating the expression of inflammatory enzymes and cytokines. Nevertheless, further studies are required to determine the mechanism of this relationship.
4.4. Induction of cell cycle arrest
Cell cycle control is very critical to ensure that cell division is free from catastrophic error. Cancer mutations perturb cell cycle control, allowing the cancer cell to evade cell cycle checkpoints [81]. While investigating the ability of GL and GA to induce cell cycle arrest, Satomi et al. [82] found that GL with inhibitory concentration IC50 greater than 1200 µM and GA with IC50 equal to 80 µM exhibited antiproliferative effects in a concentration-dependent manner against the hepatoma HepG2 cell line, with GA showing greater efficacy compared to GL [82]. GA is shown to exhibit antiproliferative activity via induction of G1 cell cycle arrest and apoptosis. Conversely, GL is shown to induce G1 cell cycle arrest, but not apoptosis [82].
Moreover, Chang and colleagues [83] demonstrate that GA in combination with curcumin inhibits tumor growth in human hepatocellular carcinoma (HCC) HepG2 cells significantly compared to curcumin alone. Their results show a marked increase of cells in the G1 phase in the combination group compared to GA alone or curcumin alone [83]. A study performed by Wang et al. [84] disclosed that GZA inhibits gastric cancer MGC-803 cell proliferation. In addition, GZA is shown to have a long-lasting inhibitory effect on the proliferation of MGC-803 cells [84]. Cell percentage in the S phase was significantly reduced compared to G0/G1 phase cell percentage. The expression of G1-associated proteins, especially cyclins D1, D2, D3, E1, and E2, was significantly downregulated. Conversely, other proteins were unchanged, confirming that GZA enforces cell cycle arrest in the G1/S phase of the cell cycle [84].
4.5. Induction of cancer cell apoptosis
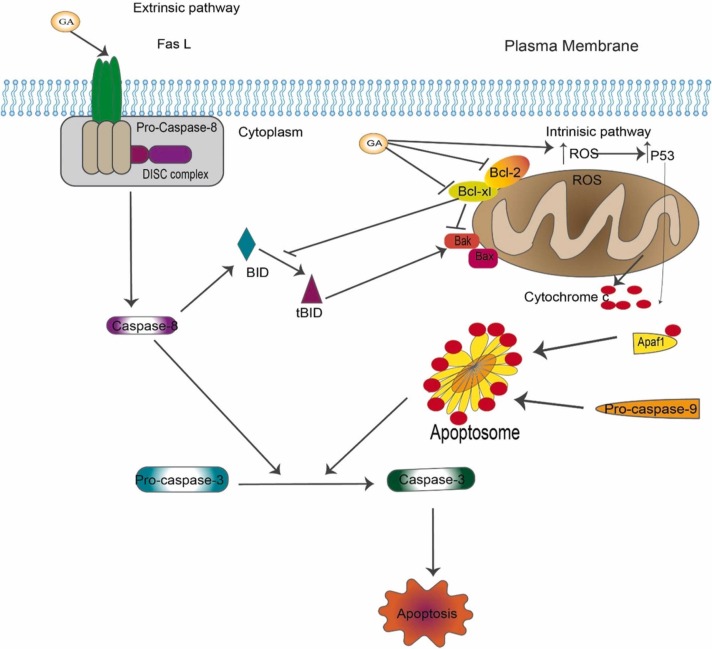
Apoptosis evasion and resisting cell death are among the hallmarks of cancer, as proposed by Hanahan and Weinberg [85] in their seminal review article. Cancer cells are able to escape programmed cell death, a well-established program to prevent cancer progression. One well-known apoptosis mechanism is that cells receive death signals from outside the cell (the extrinsic pathway), while other mechanisms are initiated by signals from inside the cell (the intrinsic pathways). Consequently, both pathways lead to the activation of the activator caspases, mainly caspase-8 for extrinsic pathway and caspase-9 for the intrinsic pathway, that ultimately activate the death executor caspases (mainly caspase-3/7), which induce a cascade of proteolysis involving effector caspases that are responsible for the execution phase of apoptosis [86], [87], [88]. Cancer cells develop strategies to evade apoptosis, such as suppressing the tumor suppressor gene TP53, which is well known to be mutated in cancer. This suppression of TP53 downregulates pro-apoptotic protein expression and activates the anti-apoptotic proteins and survival pathways [89], [90]. GL and GA have been shown to induce apoptosis via both the intrinsic and extrinsic pathways ( Fig. 3).
Fig. 3.
Schematic representation depicts that GA induces cell death. GA can induce cell death via two different mechanisms. The first extrinsic pathway includes the activation of the death receptor followed by a cascade of proteolysis of caspases leading to cells committed to death. A second intrinsic pathway is a mitochondrial-mediated pathway in response to stress. GA activates pro-apoptotic protein Bax and Bak while it inhibits the anti-apoptotic protein Bcl-2 and Bcl-xl.
In human hepatocellular carcinoma cell line HepG2, Satomi et al. [82] demonstrated that high concentrations of GA could induce apoptosis by decreasing the activation of the anti-apoptotic proteins, B-cell lymphoma 2 (Bcl-2) and B-cell lymphoma-extra-large (Bcl-xL), and by cleaving caspase-8 and caspase-9. Interestingly, the expression of the pro-apoptotic proteins Bcl-2-associated X protein (Bax) and Bak remained unchanged [82]. However, GA was unable to induce apoptosis at low concentrations. In melanoma, GA exhibits antiproliferative activity at 30 µg/mL for 24 h. The expression level of the anti-apoptotic protein Bcl-2 was downregulated, whereas the pro-apoptotic protein Bax was upregulated in these cells. In addition, the expression of pro-caspase-3 and pro-caspase-9 was upregulated as well [75].
Wei Lin and colleagues [91] examined the cytotoxicity and antitumor activity of 18β-GA and its synthetic derivative in the human bladder cancer cell line NTUB1 and demonstrated that 18β-GA and its derivative induce apoptotic cell death in a concentration-dependent manner via upregulation of ROS that activate P53 leading to the antitumor activity [91]. Their study also showed that the mitochondrial membrane potential (MMP) decreased dramatically after 24 h and the p-p53 expression increased. Nevertheless, the authors report that neither 18β-GA nor any derivative could induce cell cycle arrest [91].
Furthermore, another study conducted on breast cancer cell line MCF-7 elaborates that 18β-GA exhibits antiproliferative effects against MCF-7 cells in a concentration- and time-dependent manner with maximum inhibitory concentration IC50 of 100 µM after 48 h. To study the mechanistic pathway of apoptosis, the authors used annexin V-PI flow cytometric analysis. In addition, they used the TUNEL assay to test if 18β-GA selectively induces MCF-7 cell apoptosis. Their data revealed that the use of 18β-GA at a concentration of 100 µM increases the binding of annexin-V significantly in a time-dependent manner, decreases the MMP, releases cytochrome c into the cytoplasm, and activates cleavage of caspase-9, which indicates that GA can induce apoptosis in the breast cancer cell line via the intrinsic mitochondrial pathway [92]. Furthermore, GA is shown to increase the Bax:Bcl-2 ratio, as well as markedly increase the BH3 protein, Bim. Bim is shown to be transcriptionally regulated by the forkhead box O 3a (FOXO3a) transcription factor. The activation and localization of FOXO3a to the nucleus is associated with a significant downregulation in Akt kinase. Therefore, Bim, FOXO3a, and downregulation of Akt contribute to the GA-induced apoptosis is observed in the MCF-7 cells [92].
As discussed earlier, GA in combination with curcumin significantly induces apoptosis compared to curcumin or GA alone. Results from the study showed that the expression of the anti-apoptotic protein, Bcl-2, was significantly downregulated, whereas the expression of the pro-apoptotic protein, Bax, was upregulated, suggesting that GA in combination with curcumin induces cell apoptosis via the intrinsic mitochondrial pathway [83]. In the gastric cancer MGC-803 cell line, GZA was shown to induce apoptosis in CICD [84]. CICD implies that apoptosis happens despite the absence of caspases that may occur as a result of the intrinsic pathway or death receptor ligation [93]. Furthermore, the study showed that cells treated with GA show high expression in the pro-apoptotic Bax, cleaved PARP, pro-caspase-3, pro-caspase-8, and pro-caspase-9. Despite this result, the amount of cleaved caspases is still unchanged, indicating that caspases have no role in executing apoptosis, and GA induces apoptosis via the intrinsic mitochondrial pathway in a cancer-type context manner [84]. This finding is supported by evidence from another study by Thirugnanam et al. [94] on prostate cancer. The study demonstrates that GL induces apoptosis in prostate cancer cell lines DU-145 and LNCaP in a caspase-independent pathway [94]. A different study by Lin et al. [95] showed that GA induces cell death and autophagy in the human breast cancer cell line MDA-MB-231 via the ROS-mitochondrial pathway. GA induces MDA-MB-231 cell death by increasing ROS, causing the mitochondrial membrane potential to decrease significantly. Treatment with GA also downregulates the autophagy-associated proteins, p62- and beclin-1, and increases the conversion of LC3 to LC3-II without change in the caspase-associated protein, confirming that GA-derived ROS induces autophagy and apoptosis via the ROS-mitochondrial pathway [95]. Furthermore, 18β-GA was shown to induce autophagy in the osteosarcoma xenograft model [96].
4.6. Inhibition of invasion and metastasis
Cancer metastasis has become one of the major cancer-related causes of death. For cancer cells to metastasize to secondary sites, they undergo many sequential steps, including cell invasion, intravasation, survival in the circulation, extravasation, and colonization to distant sites [97]. Tumor cells endure a phenotypic change leading to the loss of their epithelial characteristics, gain of mesenchymal properties, and activation of specific pathways such as Wnt/β-catenin. This process, called epithelial-to-mesenchymal transition (EMT), is an essential step to cancer cell invasion and metastasis [85], [98].
Jayasooriya and colleagues [99] tested the inhibitory effect of 18β-GA on prostate cancer, bladder cancer, and breast cancer cell lines. At a concentration of 35 μM, 18β-GA exhibited a cytotoxic effect on the prostate cancer cell line, but failed to exhibit its effect on the bladder and breast cancer cell lines. However, GA was shown to inhibit TNF-α activation of matrix metalloproteinase 9 (MMP9) in all three cancer cell lines, leading to a significant reduction in cancer invasion and metastasis [99]. In addition, GA significantly reduced the mRNA expression and protein levels of VEGF [99]. GA seems to inhibit TNF-α via modulation of NF-κB. GA antagonizes TNF-α by downregulating the inhibitory IκBα that prevents the degradation of the NF-κB subunits, p65 and p50, and enhances the nuclear translocation of NF-κB [100]. The p65 and p50 subunits of NF-κB bind to the promoter regions of genes responsible for invasion and metastasis, such as VEGF and MMP9, concluding that GA inhibits invasion and metastasis by interfering with the function of the NF-κB transcription factor [99]. Furthermore, the authors suggest that GA inhibits TNF-α phosphorylation of PI3K/Akt, which in turn suppresses PI3K/Akt signaling pathway modulation of NF-κB. Hence, GA can be used to prevent invasion and metastasis [99]. This finding is supported by another study conducted by Shetty et al. [101] on prostate cancer. The authors demonstrate that 18α-GA inhibits prostate cancer invasion via inhibition of NF-κB-p65, VEGF, and MMP9 [101]. Moreover, a study using BALB/c mice with acute hepatitis induced by a combination of small doses of lipopolysaccharide and D-galactosamine, found that treatment with GL suppressed MMP9 mRNA expression [102]. MMP is a family of calcium- and zinc-dependent proteases involved in the degradation of extracellular matrix produced during liver injury [103]. Thus, GL can inhibit liver injury and subsequent development of HCC via targeting the expression of MMP9. These findings suggest the potential use of GL as an oncoprotective agent in HCC. However, more studies should be conducted for further use of GL in clinics. Interestingly, GL has also been demonstrated to inhibit pre-metastatic niche formation and metastasis in a mouse model via modulating intestinal microbiota [104].
EMT promotes the conversion of immobile epithelial cells into motile mesenchymal cells, enhancing cancer's invasion and metastatic properties [105]. It is considered one of the vital processes of cancer cells that enable the cell to evade conventional treatment and develop resistance [106], [107]. Targeting EMT has been a hot area of research in recent years. Many drugs have been developed to selectively target the upstream molecules in EMT-specific signaling pathways, such as the NOTCH pathway and Wnt pathway [108]. GA inhibits the metastatic potential of SGC-7901 gastric cancer cells by reducing the activity of MMP2 and MMP9, critical enzymes involved in EMT [109]. The intracellular ROS level, PKC-α expression, and the phosphorylation of ERK were significantly decreased after treatment with GA in a concentration-dependent manner. Therefore, GA might inhibit the migration and invasion of gastric cancer via the ROS/PKC-α/ERK signaling pathway [109]. More interestingly, a recent study by Zhang et al. [110] used 40 clinical samples of laryngeal cancer and assessed the activity of Magnesium Isoglycyrrhizinate (MI) in preventing cancer progression and inhibiting epithelial-mesenchymal transition of laryngeal cancer. Results showed that treatment with MI markedly downregulated the expression of Twist [110]. Twist is a marker of EMT that has a poor correlation with overall survival in patients with laryngeal cancer [111], [112]. In addition, MI reduced the expression levels of NF-κB and attenuated EMT in vitro and in vivo. The author concluded that MI significantly inhibited tumor progression and EMT of laryngeal cancer cells in vivo via Twist /NF-κB signaling [110].
High HMGB1 levels are associated with cancer invasion and metastasis [113], [114]. Chang et al. [115] showed that GL inhibits EMT in prostate cancer through modulation of the HMGB1 signaling pathway. Knockdown of HMGB1 resulted in downregulation of the mesenchymal marker, Snail, and upregulation of the epithelial marker, E-cadherin. Interestingly, the study found that using GL in a dose range of 25–200 μM inhibits cell migration. This implies that GL might have a role in inhibiting EMT by targeting HMGB1 [115].
MicroRNAs are a class of non-coding RNAs composed of 18–25 nucleotides in length that regulate gene expression. A recent study showed that licorice extract has the ability to upregulate several tumor suppressor miRNAs while also upregulating some oncogenic miRNAs in liver cancer cells [116]. However, the specific bioactive compounds of licorice extract responsible for the upregulation of these miRNAs have not been demonstrated in this work. Nevertheless, a previous study disclosed that 18β-GA altered miRNA expression profiles in gastric cancer. Results showed that GA specifically upregulates the miR-149–3p that works as a tumor suppressor and inhibits the Wnt-1 gene [61]. Interestingly, miR-149–3p expression was found to be negatively correlated with metastasis in HCC and GC [117], [118]. The Wnt/β-catenin pathway is dysregulated in gastric cancer [119]. Consequently, miR-149–3p, which acts as a tumor suppressor and inhibits Wnt-1, attenuated the inflammatory microenvironment, and inhibited gastric cancer progression and metastasis in transgenic mice via the regulation of miR-149–3p /Wnt-1 axis [61].
4.7. Modulation of NF-κB signaling pathway
Nuclear factor-kappa B (NF-κB) is a well-known transcription factor that mediates many functions in the cytoplasm and nucleus, and its activation regulates gene expression of various inflammatory cytokines, proteins, and growth factors. In addition, NF-κB inflammatory response leads to growth promotion, survival, and vascularization of neoplastic carcinoma cells [120], [121]. The activation of the NF-κB signaling pathway via MyD88 is due to the binding of extracellular HMGB1 to toll-like receptors (TLR), namely TLR-2, TLR-4, and TLR-5 (Fig. 2) [122]. HMGB1 is a vital effector molecule that has been implicated in chronic inflammation and cancer [123]. GL is shown to possess oncopreventive properties in different types of cancer, as discussed before. Previous studies demonstrate that GL inhibits tumor initiation by reducing the levels of HMGB1 in the extracellular matrix and blocking the binding of HMGB1 to TLRs, thereby attenuating the levels of NF-κB expression [124], [125], [126]. This leads to reduced expression of cytokines, growth factors, matrix proteases, chemokines, inflammatory molecules, and survival genes. Interestingly, a study investigating the role of intranuclear HMGB1 on the development of pancreatic ductal adenocarcinoma (PDAC) found that knockout of the HMGB1 gene in mice that had an oncogenic KRAS gene mutation accelerated the development of KRAS-driven PDAC [127]. Notably, treatment of these mice with GL reduced the development of precancerous neoplasms in the pancreas of this mouse model [127]. This may be due to differences in binding targets between extracellular and intranuclear HMGB1. Furthermore, another study showed that 18α−GA downregulates the expression level of HMGB1 in prostate cancer [95].
A gene expression profile study on GL-treated HepG2 cells using oligonucleotide microarray suggested the hepatoprotective effects of GL might be due to the suppression of NF-κB signaling and regulating the genes involved in apoptosis and oxidative stress pathways [128]. TNF-α-stimulated NF-κB activity was dramatically reduced after GA treatment in a concentration-dependent manner. GA prevented IκBα phosphorylation and p65 translocation, lowering iNOS gene expression. It also reduced NO generation and iNOS expression in TNF-α-induced rat primary hepatocytes. These observations demonstrate that GA may confer hepatoprotection against chronic liver inflammation by reducing NF-κB activation and therefore reducing inflammation [129]. Li et al. [130] demonstrated that in the human glioblastoma (GBM) U251 cell line, GZA exerted an anti-proliferative effect in a time- and concentration-dependent manner. GZA also induced apoptosis more effectively compared to the control [130]. Furthermore, GZA treatment resulted in a robust decrease in the expression of NF-κB-p65 in the nucleus, indicating that GZA inhibits cell proliferation and activates apoptosis in the human glioblastoma U251 cell line by influencing the NF-κB mediated pathway [130].
4.8. Modulation of PKC/ERK pathway
Protein kinase C (PKC) is involved in a pro-survival or pro-apoptotic activity which depends on the specific isoforms. PKC δ is a pro-apoptotic mediator, while PKC α/βII promotes the survival of the cells [131], [132]. In vitro study on the NSCLC cell line NCI-H460 suggested that treatment with GA reduced the phosphorylation of PKC α/βII and increased PKC δ, indicating that GA is capable of inducing apoptosis by increasing the pro-apoptotic signal and decreasing the anti-apoptotic signal. Indeed, the study revealed that the expression level of the anti-apoptotic proteins Bcl-xL and Bcl-2 were decreased. In addition, expression levels of the intrinsic executor caspases-3/9 were increased [133]. Furthermore, extracellular activated protein kinase (ERK) phosphorylation was decreased, whereas c-Jun NH2-terminal kinase phosphorylation was increased [133]. PKC δ was shown to activate the MEK-ERK pathway [134]. Therefore, GA decreases the expression of PKC α/βII and increases the expression of PKC δ, which induces apoptosis in NSCLC via modulation of the PKC /ERK signaling pathway [133].
4.9. Modulation of MAPK signaling pathway
The mitogen-activated protein kinase (MAPK) pathway plays a crucial role in cancer. It controls many important cellular events, including survival, dissemination, and resistance to drug therapy [135]. The MAPK pathway is a convergent signaling node that may serve as a tumor suppressor gene or oncogene according to the intensity of the signal and the type of tissue [136]. MAPK receives different signals from inside the cell, such as signals from DNA damage and metabolic pathways, as well as external stimuli, such as growth factor receptor signals [137]. Wang et al. [138] found that GA exhibited a cytotoxic effect, induced apoptosis, and promoted cell cycle arrest at the G0/G1 phase in the rat pituitary adenoma-derived MMQ and GH3 cell lines. The inhibitory concentration IC50 at 24 h was 69.6 µM in GH3 cells and 111.5 µM in MMQ cells. GA is also shown to induce apoptosis via reactive oxygen species (ROS)-dependent mitochondrial dysfunction [138].
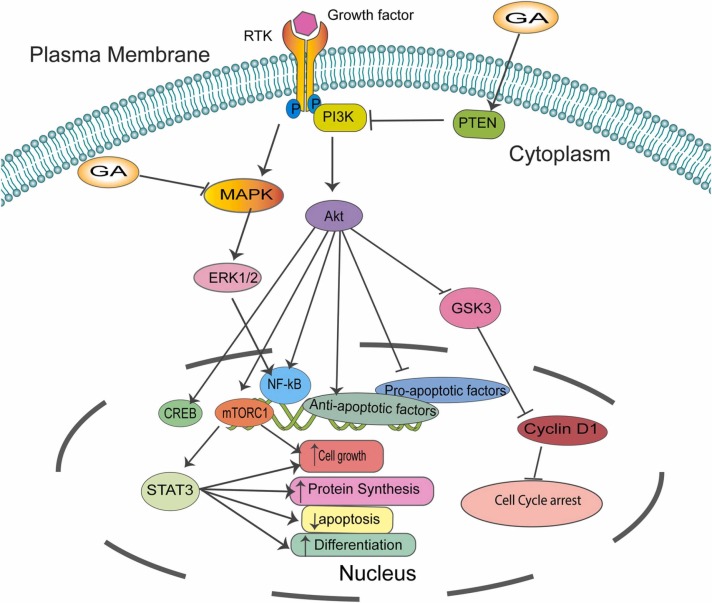
Moreover, the author revealed that GA induces the phosphorylation of c-Jun N-terminal kinases (JNK), P38, and calcium/calmodulin-dependent protein kinase II (CaMKII), which leads to the activation of the MAPK pathway. These results indicate that GA can induce cell cycle arrest and apoptosis via activating ROS that consequently activate the MAPK pathway ( Fig. 4) [138]. The dysregulation of the MAPK pathway is common in cancer, and studies are warranted to increase our understanding of this critical pathway.
Fig. 4.
Schematic representation of common pathways modulated by GA. GA inhibits main pathways critical for cancer growth and metastasis, including PI3K/Akt, MAPK/ERK1/2, and Akt/mTORC1 /STAT3, which are essential for cancer cell growth, differentiation, and senescence.
4.10. Modulation of PI3K/Akt/mTOR signaling pathway
The PI3K/Akt/mTOR signaling axis plays an important role in controlling vital cellular processes such as cell proliferation, cell growth, cell metabolism, apoptosis, and angiogenesis [139], [140]. PI3K/Akt/mTOR upregulation and mutation in many cancers make it an attractive target for cancer therapeutics [141], [142]. In HCC, GA in combination with curcumin activates the tumor suppressor PTEN that controls the PI3K/Akt pathway. Of note, the mRNA expression level of PI3K and Akt was significantly reduced. Additionally, the protein expression level of PI3K, Akt, and p-Akt protein was markedly decreased, concluding that GA works by activating PTEN, which inhibits the PI3K/Akt signaling pathway in HCC [83]. Furthermore, in combination with curcumin, GA also suppressed HCC growth by modulation of the PTEN/PI3K/Akt signaling pathway in a xenograft model. The results show that tumors formed while treated with GA and/or curcumin are less in both size and weight than the control group. Knocking down PTEN reverses the process with increased tumor weight and size, confirming that GA and curcumin inhibit hepatocellular cancer growth via modulation of the PTEN/PI3K/Akt signaling pathway in vitro and in vivo (Fig. 4) [83].
Another study investigated the activity of GZA against gastric cancer, and results showed that GZA induces cell cycle arrest and apoptosis in a caspase-independent cell death (CICD) manner. The expression of phosphorylated PI3K and Akt was significantly downregulated. However, total PI3K and Akt remained unchanged, suggesting that GA inhibits the PI3K/Akt signaling pathway in gastric cancer [84]. In a study using the MCF-7 human breast cancer cell line, GZA inhibited cancer proliferation in a concentration and time-dependent manner. It was shown to induce apoptosis and cell cycle arrest through downregulation of mTOR, p-mTOR, and PI3K/Akt protein expression, indicating that GZA induces breast cancer cell death via inhibition of the PI3K/Akt/mTOR pathway [11].
4.11. Modulation of Akt/mTOR/STAT3 pathway
The Akt/mTOR/STAT3 pathway is a master intracellular signaling pathway that is important in tumor cell growth and migration. Signaling of serine-threonine protein kinase (Akt)/mammalian target of rapamycin (mTOR) and signal transduction and activator of transcription 3 (STAT3) are critical for leukemia cell survival and proliferation [143]. A study published in 2015 showed that GZA markedly inhibited cell proliferation of human leukemia cell line TF-1. This study also demonstrated that GZA could effectively inhibit TF-1 leukemia cell migration and invasion. Moreover, the effect of GZA on TF-1 cells was found to be via targeting the Akt/mTOR/STAT3 pathway (Fig. 4) [143]. These observations demonstrate the potential of GL and GZA in the treatment of AML in vitro; however, further investigation is needed to validate these results and better understand the mechanistic action of GL and GA. Because the response to treatment and overall survival of patients with AML is remarkably variable, and there are several prognostic factors related to patient and tumor characteristics, it is essential to study any possible treatment options, such as GL, which has shown great potential to be used in the treatment of AML.
5. Synergistic action of GA in combination with topoisomerase II α inhibitor in breast cancer
Triple-negative breast cancer (TNBC) is a disease with an unfavorable prognosis. It is characterized by the absence of three key markers: estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 [144], [145]. Topoisomerase II α (TOP ΙΙα) is a subtype of topoisomerase II involved in resolving supercoiled DNA during genetic processes. Previous reports have shown that TOP ΙΙα and HER-2 are over-expressed in breast cancer patients and significantly respond to topoisomerase inhibitors in breast cancer treatment [146]. Etoposide is a widely used drug derived from podophyllotoxin, employed in the treatment of various cancer types since its first approval by the FDA in 1983 [147], [148]. However, resistance to etoposide has been reported in many types of cancer, including breast cancer, lung cancer, and other solid tumors [149], [150], [151]. Interestingly, GA is shown to sensitize topoisomerase IIα to etoposide. In the TNBC MDA-MB-231 cell line, GA in combination with etoposide enhances cytotoxicity and response to treatment by increasing the expression of TOP ΙΙα to 2.4 folds within 12 h, making the enzyme more sensitive to treatment. After 48 h treatment, GA induces apoptosis as well. More interestingly, the study revealed that sensitizing TOP ΙΙα to etoposide is mediated by GA inhibition of GSH depletion, which is consistent with the study that reported on the antioxidant properties of GA [55]. In addition, modulation of the Akt pathway and ERK/JNK/MAPK pathways are involved in the GA-mediated upregulation of TOP ΙΙα expression in TNBC [152].
6. GL inhibition of latency-associated nuclear antigen (LANA) protein in the treatment of Kaposi Sarcoma
Kaposi Sarcoma (KS) is a vascular tumor associated with human herpesvirus 8, also known as Kaposi sarcoma-associated herpesvirus (KSHV) which was initially discovered in tissues derived from patients with acquired immune deficiency syndrome [153]. KSHV is an oncogenic double-stranded DNA virus that is thought to develop a lifelong infection in human B lymphocytes [154]. Detection of the viral protein latency-associated nuclear antigen (LANA) in the biopsy is used in the diagnosis of KS [155]. KSHV gene expression is regulated by binding of the host cell proteins, CTCF and cohesin complex, to the KSHV genome. Cohesin and CTCF also affect the host cells by promoting antiviral defenses [155]. GL treatment on KSHV-positive BCLB1 cells was shown to downregulate LANA mRNA expression and transcripts encoding for LANA and ORF74 [156]. In the same study, potential candidate targets for GL such as SMC3 and SPT5 have been identified, which could account for the action of GL on RNAPII pausing at CTCF-cohesion binding sites [156]. GL has been shown to selectively promote cell death in latent KSHV by restoring p53 function along with its ability to induce apoptosis [157].
7. GL protects against chemotherapy-induced side effects
Conventional chemotherapy has many undesirable side effects. Cisplatin is an alkylating agent used in treatment regimens, either alone or in combination with other agents [158]. Nephrotoxicity is a major side effect of cisplatin treatment [159]. GZA and 18β-GA exhibit a protective effect against cisplatin-induced nephrotoxicity. One study reports that GZA and GA exert their protective effect via activation of nuclear factor E2-related protein (Nrf2) and inhibition of NF-κB in the kidney, suggesting that the protective effect is mediated through the Nrf2/NF-κB pathway. However, further investigations are required to support this study [160]. A randomized control trial of 216 gastric cancer patients on either the FOLFOX chemotherapy regimen (n = 104) or the XELOX chemotherapy regimen (n = 112) investigated the capability of MI to protect against acute drug-induced liver damage from the initial chemotherapy regimen. Out of 216 patients involved in the study, 40 patients developed liver toxicity. The results showed that there is a significant difference between the intervention group taking MI for protection against drug-induced liver damage and the control group, indicating that MI protects against drug-induced liver damage in gastrointestinal tumors [161]. Moreover, a randomized, double-blind, multicenter phase II trial including 174 patients investigated the safety and efficacy of MI injection against antineoplastic drugs in inducing liver toxicity. The study found that MI is effective and safe in the treatment of drug-induced liver damage [162].
8. Drug delivery applications
GA, the metabolite product of GL, is rapidly absorbed from the intestine and transported via specific carrier molecules to the liver, where it is metabolized to either glucuronide or sulfate conjugates that are transported and excreted into the bile and are then circulated to the entero-hepatic circulation [41]. Anticancer therapeutics now include drug delivery systems as an essential integrated part. The development of innovative drug carriers could significantly improve antineoplastic therapy. In this field, GL has much potential as a carrier material. Recent research has found that combining GL with first-line medications has a more substantial therapeutic effect on malignancies [163]. Besides its cancer activity, GA metabolism in the liver greatly benefits targeting hepatocellular carcinoma. GA is used in combination with many polymers to design drug delivery nano-formulations to target HCC. Different types of nano-formulations are being developed, such as the GA-DTX-NGO system, which delivers the chemotherapeutic agent docetaxel (DTX) in nanomaterial composed of GA and nanographene oxide (NGO) to treat liver cancer [164]. Another formulation is DTX/GA-HA-NPs, in which glycyrrhetinic acid-modified hyaluronic acid (GA-HA) is used in combination with the drug DTX [165]. A recent review highlighted the drug carrier potential of GZA and18β-GA in liver cancer [166].
9. Specificity and potential side-effects of GL
The wide range of therapeutic effects of GL may lead to overconsumption of this natural herb. Long-term consumption of licorice could increase mineralocorticoid activity by inhibiting 11βhydroxysteroid dehydrogenase 2 (11βHSD2) and by directly binding to mineralocorticoid receptors, which could cause some adverse effects [167]. GA inhibits 11ßHSD2 200–1000 times more effectively than GZA. Hence its pharmacokinetics are more important after oral administration [168]. Pseudo-hyperaldosteronism is a clinical condition caused by long-term consumption of licorice [169]. However, many preclinical and clinical trials of GL and GA in different disease conditions presented GL with a favorable risk-benefit profile where the benefit outweighs the risk [170], [171], [172].
10. Conclusions and perspectives
GL and its derivative, GA, exhibit a wide range of activities to elicit their oncoprotective and oncotherapeutic effects. The effects range from inducing cell cycle arrest, inducing apoptosis, inhibiting invasion and metastasis, immunomodulation, attenuating the inflammatory microenvironment in cancer to antioxidants, sensitizing the tumor to other drugs, and protecting from conventional therapy side effects. Moreover, GL and GA mediate their action by targeting numerous cancer pathways ( Table 1, Table 2), such as NF-κB, PI3K/Akt, and the MAPK signaling cascade, a characteristic property among plant extracts used in medicine. On the one hand, plant extracts processing multi-targets are considered to be a drawback; on the other hand, it might overcome the resistance accompanied by the usage of target therapy. Using GL and GA extract as adjuvant therapy might offer an elegant solution to cancer resistance. However, caution must be taken with using licorice as an oncoprotective agent against cancer due to side effects. Furthermore, most studies that were conducted to investigate either the oncopreventive or oncotherapeutic activity in vitro or in vivo studies. There were very few clinical trials. Further studies are warranted to evaluate the oncopreventive and oncotherapeutic activity of GL and GA in humans.
Table 1.
In vitro anticancer activities of GL and GA.
| Cancer type | Cells lines | Compound | Conc. | Effects | Mechanisms | References |
|---|---|---|---|---|---|---|
| Bladder cancer | NTUB1 | 18β-GA | 1–50 μM | Induced apoptosis | ↑ROS, ↑p53 | [91] |
| Breast cancer | MCF-7 | 18β-GA | 100–200 µM | Induced apoptosis | ↓Akt, ↑FOXO3a, ↑Bim | [92] |
| Breast cancer | MCF-7 | GZA | 10–100 µM | Induced cell cycle arrest and apoptosis | ↓mTOR, ↓PI3K, ↓Akt | [11] |
| Breast cancer | MDA-MB-231 | GZA | 2.5–20 mM | Induced apoptosis and autophagy | ↑ROS, ↓mitochondrial membrane potential | [95] |
| Breast cancer | MDA-MB-231 | GA | 5–20 μM | Induced apoptosis | ↓GSH, ↑Akt, ↑ERK, ↑JNK, ↑MAPK, ↑TOPO2A | [152] |
| Gastric cancer | SGC-7901 | 18β-GA | 20–120 μM | Inhibited cancer migration and invasion | ↓ROS, ↓PKC-α, ↓ERK | [109] |
| Gastric cancer | MGC-803 | GZA | 0.5–4 mg/mL | Induced cell cycle arrest and apoptosis | ↑Caspase-independent cell death, ↓PI3K, ↓Akt | [84] |
| Hepatocellular carcinoma | HepG2 | Curcumin combined with GA | 5 µM | Induced cell cycle arrest and apoptosis | ↑PTEN, ↓PI3K, ↓Akt | [83] |
| Laryngeal cancer | HEP-2 | MI | 3.22 mg/mL (IC50) | Inhibited cancer progression and EMT | ↓Twist, ↓NF-κB | [110] |
| Leukemia | TF-1 | GZA | 1–40 μM | Induced cell cycle arrest and apoptosis | ↓Akt, ↓mTOR, ↓STAT3 | [143] |
| Lung cancer | A549, NCI-H460 | 18β-GA | 160–320 μM | Inhibited cancer proliferation | ↓TxAS, ↓ERK, ↓CREB | [67] |
| Lung cancer | NCI-H460, A549 | GA | 78 μg/mL for A549 and 62 μg/mL for NCI-H460 (IC50) | Induced cell cycle arrest and apoptosis | ↓PKC α/βII, ↓ERK, ↑JNK | [133] |
| Melanoma | B16F10 | GZA | 30 μg/mL | Induced cell cycle arrest and apoptosis | ↓COX-2, ↓PGE2, ↓pSTAT3 | [75] |
| Pituitary cancer | MMQ, GH3 | 18β-GA | 69.6 µM in GH3 cells and 111.5 µM in MMQ cells (IC50) | Induced cell cycle arrest and apoptosis | ↑ROS, ↑JNK, ↑p38, ↑CaMKII, ↑MAPKs | [138] |
| Prostate cancer | DU145 | GL | 25–200 μM | Inhibited EMT | ↓HMGB1 | [115] |
| Prostate, bladder, and breast cancer | LNCaP, T24, MDA-MB-231 | GA | 5–25 μM | Inhibited cancer invasion and metastasis | ↓TNF-α, ↓MMP9, ↓VEGF, ↓Akt-dependent NF-κB | [99] |
Table 2.
In vivo anticancer activities of GL and GA.
| Cancer type | Animal models | Compound | Dose (route) | Effects | Mechanisms | References |
|---|---|---|---|---|---|---|
| Breast cancer | MDA-MB-231 xenografts in athymic female nude mice | GA | 40 mg/kg (i.p.) | Inhibited tumor growth, invasion, and metastasis | ↓MMP2, ↓MMP9 | [173] |
| Colon cancer | DMH-induced colon carcinogenesis in male Wistar rats | GZA | 15 − 20 mg/kg (p.o.) | Suppressed development of precancerous lesions | ↓NF-κB, ↓TNF-α, ↓COX-2, ↓iNOS, ↓VEGF, ↑p53, ↑connexin-43, ↑caspase-9 | [70] |
| Endometrial cancer | MNU-induced carcinogenesis in the uterine tube in female ICR mice | GL | 0.0625% or 0.625% GL alone or in combination with 5 ppm E2 (p.o.) | Suppressed development of precancerous lesions | ↓COX-2, ↓IL-1α, ↓TNF-α | [78] |
| Gastric cancer | Transgenic mouse tumor model | 18β-GA | 0.05% 18β-GA (drinking water) | Inhibited tumor initiation and progression | ↓COX-2 | [61] |
| Laryngeal cancer | HEP-2 xenografts in BALB/c nude mice | MI | 300 mg/kg (s.c.) | Inhibited tumor progression and EMT | ↓Twist, ↓NF-κB | [110] |
| Leukemia | TF-1 xenografts in female BALB/c mice | GZA | 100 mg/kg(peritumoral) | Inhibited tumor growth | ↓Akt, ↓mTOR, ↓STAT3 | [143] |
| Lung cancer | A549-TPα lung adenocarcinoma in female nude mice | GL alone or in combination with Cisplatin | 15–135 mg/kg(i.p.) | Inhibited tumor growth | ↓TxAS, ↓PCNA | [69] |
| Lung cancer | A549 xenografts in female nude mice | GL | 50 mg/kg(p.o.) | Inhibited tumor growth | ↓TxAS, ↓TxA2 | [174] |
| Lung cancer | HCC827 xenografts in PDX nude mice | GL | 100 mg/kg (i.p.) | Inhibited tumor growth | ↓HMGB1, ↓JAK, ↓STAT3 | [126] |
| Melanoma | B16F10 xenografts in female C57BL/6 mice | GZA | 1–100 mg/kg (i.d.) | Inhibited tumor growth and induced apoptosis | ↓pSTAT3, ↓IL10, ↓TGFβ, ↑IL12, ↑IFNγ, ↑TNFα | [75] |
| Osteosarcoma | HOS xenografts in female BALB/c nude mice | 18β-GA | 2 mg/kg (i.p.) | Inhibited tumor growth and induced cell cycle arrest, apoptosis, and autophagy | ↑Cleaved caspase-3, ↑LC3B-II, ↑p-JNK, ↑cyclin E, ↑CDK4, ↑cyclin D1 | [96] |
| Ovarian cancer | A2780 xenografts in nude mice | GA | 10–30 mg/kg (i.p.) | Inhibited tumor growth/ angiogenesis and induced apoptosis | ↓VEGF | [175] |
| Pituitary cancer | GH3 xenografts in male BALB/c athymic nude mice | 18β-GA | 20 mg/kg (i.p.) | Inhibited tumor growth and induced apoptosis | ↑ROS, ↑MAPK | [138] |
| Skin cancer | DMBA/ TPA-induced papillomas in Swiss albino female mice | 18β-GA | 1.25–2.5 mg (topical) | Inhibited tumor initiation and progression | ↓OLD | [55] |
Funding
This study was partly supported by NIH Grant# R03 CA227218 and Bridge funding (2022), Office of Research, University of Illinois College of Medicine Rockford. Funding for this study was also partly supported by Brovember Inc. Rockford, Illinois.
Credit authorship contribution statement
RJ, MAH, and GM contributed to the conception of the review article. RJ, MAH, SP, CM, and PS contributed to writing and editing. GM provided supervision, reviewed, and edited the draft. All authors read and approved the final manuscript.
Declaration of Competing Interest
Authors declare that they do not have conflicts of interest.
References
- 1.Li X., Sun R., Liu R. Natural products in licorice for the therapy of liver diseases: progress and future opportunities. Pharmacol. Res. 2019;144:210–226. doi: 10.1016/j.phrs.2019.04.025. [DOI] [PubMed] [Google Scholar]
- 2.Zhang Q., Ye M. Chemical analysis of the Chinese herbal medicine Gan-Cao (licorice) J. Chromatogr. A. 2009;1216(11):1954–1969. doi: 10.1016/j.chroma.2008.07.072. [DOI] [PubMed] [Google Scholar]
- 3.Nomura T., Fukai T., Akiyama T. Chemistry of phenolic compounds of licorice (Glycyrrhiza species) and their estrogenic and cytotoxic activities. Pure Appl. Chem. 2002;74(7):1199–1206. [Google Scholar]
- 4.Abe N., Ebina T., Ishida N. Interferon induction by glycyrrhizin and glycyrrhetinic acid in mice. Microbiol. Immunol. 1982;26(6):535–539. doi: 10.1111/j.1348-0421.1982.tb00207.x. [DOI] [PubMed] [Google Scholar]
- 5.Zheng Y.F., Qi L.W., Zhou J.L., Li P. Structural characterization and identification of oleanane-type triterpene saponins in Glycyrrhiza uralensis Fischer by rapid-resolution liquid chromatography coupled with time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 2010;24(22):3261–3270. doi: 10.1002/rcm.4768. [DOI] [PubMed] [Google Scholar]
- 6.Zhang Z., Yang L., Hou J., Tian S., Liu Y. Molecular mechanisms underlying the anticancer activities of licorice flavonoids. J. Ethnopharmacol. 2021;267 doi: 10.1016/j.jep.2020.113635. [DOI] [PubMed] [Google Scholar]
- 7.Wang Z.Y., Nixon D.W. Licorice and cancer. Nutr. Cancer. 2001;39(1):1–11. doi: 10.1207/S15327914nc391_1. [DOI] [PubMed] [Google Scholar]
- 8.Graebin C.S. The pharmacological activities of glycyrrhizinic acid (“glycyrrhizin”) and glycyrrhetinic acid. Sweeteners. 2017:245–261. [Google Scholar]
- 9.Hattori M., Sakamoto T., Yamagishi T., Sakamoto K., Konishi K., Kobashi K., Namba T. Metabolism of glycyrrhizin by human intestinal flora. II. Isolation and characterization of human intestinal bacteria capable of metabolizing glycyrrhizin and related compounds. Chem. Pharm. Bull. 1985;33(1):210–217. doi: 10.1248/cpb.33.210. [DOI] [PubMed] [Google Scholar]
- 10.Krahenbuhl S., Hasler F., Krapf R. Analysis and pharmacokinetics of glycyrrhizic acid and glycyrrhetinic acid in humans and experimental animals. Steroids. 1994;59(2):121–126. doi: 10.1016/0039-128x(94)90088-4. [DOI] [PubMed] [Google Scholar]
- 11.Zhang Z., Feng Y., Li Z.Y., Cao X.Z. Antiproliferative and apoptotic activity of glycyrrhizinic acid in MCF-7 human breast cancer cells and evaluation of its effect on cell cycle, cell migration and m-TOR/PI3K/Akt signalling pathway. Arch. Med. Sci. 2019;15(1):174–182. doi: 10.5114/aoms.2018.79429. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 12.Chueh F.S., Hsiao Y.T., Chang S.J., Wu P.P., Yang J.S., Lin J.J., Chung J.G., Lai T.Y. Glycyrrhizic acid induces apoptosis in WEHI-3 mouse leukemia cells through the caspase- and mitochondria-dependent pathways. Oncol. Rep. 2012;28(6):2069–2076. doi: 10.3892/or.2012.2029. [DOI] [PubMed] [Google Scholar]
- 13.Vaya J., Belinky P.A., Aviram M. Antioxidant constituents from licorice roots: isolation, structure elucidation and antioxidative capacity toward LDL oxidation. Free Radic. Biol. Med. 1997;23(2):302–313. doi: 10.1016/s0891-5849(97)00089-0. [DOI] [PubMed] [Google Scholar]
- 14.Pompei R., Laconi S., Ingianni A. Antiviral properties of glycyrrhizic acid and its semisynthetic derivatives. Mini Rev. Med. Chem. 2009;9(8):996–1001. doi: 10.2174/138955709788681636. [DOI] [PubMed] [Google Scholar]
- 15.Ming L.J., Yin A.C. Therapeutic effects of glycyrrhizic acid. Nat. Prod. Commun. 2013;8(3):415–418. [PubMed] [Google Scholar]
- 16.Wahab S., Annadurai S., Abullais S.S., Das G., Ahmad W., Ahmad M.F., Kandasamy G., Vasudevan R., Ali M.S., Amir M. Glycyrrhiza glabra (Licorice): a comprehensive review on its phytochemistry, biological activities, clinical evidence and toxicology. Plants. 2021;10(12) doi: 10.3390/plants10122751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hasan M.K., Ara I., Mondal M.S.A., Kabir Y. Phytochemistry, pharmacological activity, and potential health benefits of Gly cyrrhiza glabra. Heliyon. 2021;7(6) doi: 10.1016/j.heliyon.2021.e07240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Matsumoto Y., Matsuura T., Aoyagi H., Matsuda M., Hmwe S.S., Date T., Watanabe N., Watashi K., Suzuki R., Ichinose S., Wake K., Suzuki T., Miyamura T., Wakita T., Aizaki H. Antiviral activity of glycyrrhizin against hepatitis C virus in vitro. PLoS One. 2013;8(7) doi: 10.1371/journal.pone.0068992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.van Rossum T.G., Vulto A.G., Hop W.C., Schalm S.W. Pharmacokinetics of intravenous glycyrrhizin after single and multiple doses in patients with chronic hepatitis C infection. Clin. Ther. 1999;21(12):2080–2090. doi: 10.1016/s0149-2918(00)87239-2. [DOI] [PubMed] [Google Scholar]
- 20.Fiore C., Eisenhut M., Krausse R., Ragazzi E., Pellati D., Armanini D., Bielenberg J. Antiviral effects of Glycyrrhiza species. Phytother. Res. 2008;22(2):141–148. doi: 10.1002/ptr.2295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.van de Sand L., Bormann M., Alt M., Schipper L., Heilingloh C.S., Steinmann E., Todt D., Dittmer U., Elsner C., Witzke O., Krawczyk A. Glycyrrhizin effectively inhibits SARS-CoV-2 replication by inhibiting the viral main protease. Viruses. 2021;13(4) doi: 10.3390/v13040609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bailly C., Vergoten G. Glycyrrhizin: an alternative drug for the treatment of COVID-19 infection and the associated respiratory syndrome? Pharmacol. Ther. 2020;214 doi: 10.1016/j.pharmthera.2020.107618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Murck H. Symptomatic protective action of glycyrrhizin (licorice) in COVID-19 infection? Front. Immunol. 2020;11:1239. doi: 10.3389/fimmu.2020.01239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Siegel R.L., Miller K.D., Fuchs H.E., Jemal A. Cancer statistics, 2021. CA Cancer J. Clin. 2021;71(1):7–33. doi: 10.3322/caac.21654. [DOI] [PubMed] [Google Scholar]
- 25.Szakacs G., Paterson J.K., Ludwig J.A., Booth-Genthe C., Gottesman M.M. Targeting multidrug resistance in cancer. Nat. Rev. Drug Discov. 2006;5(3):219–234. doi: 10.1038/nrd1984. [DOI] [PubMed] [Google Scholar]
- 26.Maiuthed A., Chantarawong W., Chanvorachote P. Lung cancer stem cells and cancer stem cell-targeting natural compounds. Anticancer Res. 2018;38(7):3797–3809. doi: 10.21873/anticanres.12663. [DOI] [PubMed] [Google Scholar]
- 27.Mudit M., El Sayed K.A. Cancer control potential of marine natural product scaffolds through inhibition of tumor cell migration and invasion. Drug Discov. Today. 2016;21(11):1745–1760. doi: 10.1016/j.drudis.2016.06.032. [DOI] [PubMed] [Google Scholar]
- 28.Nigam M., Suleria H.A.R., Farzaei M.H., Mishra A.P. Marine anticancer drugs and their relevant targets: a treasure from the ocean. Daru. 2019;27(1):491–515. doi: 10.1007/s40199-019-00273-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Newman D.J., Cragg G.M. Drugs and drug candidates from marine sources: an assessment of the current “state of play”. Planta Med. 2016;82(9–10):775–789. doi: 10.1055/s-0042-101353. [DOI] [PubMed] [Google Scholar]
- 30.Cragg G.M., Newman D.J., Snader K.M. Natural products in drug discovery and development. J. Nat. Prod. 1997;60(1):52–60. doi: 10.1021/np9604893. [DOI] [PubMed] [Google Scholar]
- 31.Atanasov A.G., Zotchev S.B., Dirsch V.M., International Natural Product Sciences T., Supuran C.T. Natural products in drug discovery: advances and opportunities. Nat. Rev. Drug Discov. 2021;20(3):200–216. doi: 10.1038/s41573-020-00114-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Stratton C.F., Newman D.J., Tan D.S. Cheminformatic comparison of approved drugs from natural product versus synthetic origins. Bioorg. Med. Chem. Lett. 2015;25(21):4802–4807. doi: 10.1016/j.bmcl.2015.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sharifi-Rad J., Ozleyen A., Boyunegmez Tumer T., Oluwaseun Adetunji C., El Omari N., Balahbib A., Taheri Y., Bouyahya A., Martorell M., Martins N., Cho W.C. Natural products and synthetic analogs as a source of antitumor drugs. Biomolecules. 2019;9(11) doi: 10.3390/biom9110679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mann J. Natural products in cancer chemotherapy: past, present and future. Nat. Rev. Cancer. 2002;2(2):143–148. doi: 10.1038/nrc723. [DOI] [PubMed] [Google Scholar]
- 35.Cragg G.M., Pezzuto J.M. Natural products as a vital source for the discovery of cancer chemotherapeutic and chemopreventive agents. Med. Princ. Pract. 2016;25(Suppl 2):41–59. doi: 10.1159/000443404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ijaz S., Akhtar N., Khan M.S., Hameed A., Irfan M., Arshad M.A., Ali S., Asrar M. Plant derived anticancer agents: a green approach towards skin cancers. Biomed. Pharmacother. 2018;103:1643–1651. doi: 10.1016/j.biopha.2018.04.113. [DOI] [PubMed] [Google Scholar]
- 37.Younes A., Yasothan U., Kirkpatrick P. Brentuximab vedotin. Nat. Rev. Drug Discov. 2012;11(1):19–20. doi: 10.1038/nrd3629. [DOI] [PubMed] [Google Scholar]
- 38.Lowden P.A.S., Wilkinson B., Böhm G.A., Handa S., Floss H.G., Leadlay P.F., Staunton J. Origin and true nature of the starter unit for the rapamycin polyketide synthase. Angew. Chem. Int. Ed. 2001;40(4):777–779. [PubMed] [Google Scholar]
- 39.Zeng Z., Sarbassov D.D., Samudio I.J., Yee K.W.L., Munsell M.F., Ellen Jackson C., Giles F.J., Sabatini D.M., Andreeff M., Konopleva M. Rapamycin derivatives reduce mTORC2 signaling and inhibit AKT activation in AML. Blood. 2006;109(8):3509–3512. doi: 10.1182/blood-2006-06-030833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nerkar A.G., Chakraborthy G.S., Ukirde R.D. Anti-cancer agents from natural sources: a review. Curr. Trends Pharm. Pharm. Chem. 2021;3(4):38–46. [Google Scholar]
- 41.Ploeger B.A., Meulenbelt J., DeJongh J. Physiologically based pharmacokinetic modeling of glycyrrhizic acid, a compound subject to presystemic metabolism and enterohepatic cycling. Toxicol. Appl. Pharmacol. 2000;162(3):177–188. doi: 10.1006/taap.1999.8843. [DOI] [PubMed] [Google Scholar]
- 42.Nimptsch K., Rohrmann S., Nieters A., Linseisen J. Serum undercarboxylated osteocalcin as biomarker of vitamin K intake and risk of prostate cancer: a nested case-control study in the Heidelberg cohort of the European prospective investigation into cancer and nutrition. Cancer Epidemiol. Biomark. Prev. 2009;18(1):49–56. doi: 10.1158/1055-9965.EPI-08-0554. [DOI] [PubMed] [Google Scholar]
- 43.Yamamura Y., Santa T., Kotaki H., Uchino K., Sawada Y., Iga T. Administration-route dependency of absorption of glycyrrhizin in rats: intraperitoneal administration dramatically enhanced bioavailability. Biol. Pharm. Bull. 1995;18(2):337–341. doi: 10.1248/bpb.18.337. [DOI] [PubMed] [Google Scholar]
- 44.Sakiya Y., Akada Y., Kawano S., Miyauchi Y. Rapid estimation of glycyrrhizin and glycyrrhetinic acid in plasma by high-speed liquid chromatography. Chem. Pharm. Bull. 1979;27(5):1125–1129. doi: 10.1248/cpb.27.1125. [DOI] [PubMed] [Google Scholar]
- 45.Yamamura Y., Kawakami J., Santa T., Kotaki H., Uchino K., Sawada Y., Tanaka N., Iga T. Pharmacokinetic profile of glycyrrhizin in healthy volunteers by a new high-performance liquid chromatographic method. J. Pharm. Sci. 1992;81(10):1042–1046. doi: 10.1002/jps.2600811018. [DOI] [PubMed] [Google Scholar]
- 46.Akao T., Akao T., Kobashi K. Glycyrrhizin beta-D-glucuronidase of Eubacterium sp. from human intestinal flora. Chem. Pharm. Bull. 1987;35(2):705–710. doi: 10.1248/cpb.35.705. [DOI] [PubMed] [Google Scholar]
- 47.Hattori M., Sakamoto T., Kobashi K., Namba T. Metabolism of glycyrrhizin by human intestinal flora. Planta Med. 1983;48(1):38–42. doi: 10.1055/s-2007-969875. [DOI] [PubMed] [Google Scholar]
- 48.Ploeger B., Mensinga T., Sips A., Seinen W., Meulenbelt J., DeJongh J. The pharmacokinetics of glycyrrhizic acid evaluated by physiologically based pharmacokinetic modeling. Drug Metab. Rev. 2001;33(2):125–147. doi: 10.1081/dmr-100104400. [DOI] [PubMed] [Google Scholar]
- 49.Reth M. Hydrogen peroxide as second messenger in lymphocyte activation. Nat. Immunol. 2002;3(12):1129–1134. doi: 10.1038/ni1202-1129. [DOI] [PubMed] [Google Scholar]
- 50.Moldogazieva N.T., Mokhosoev I.M., Feldman N.B., Lutsenko S.V. ROS and RNS signalling: adaptive redox switches through oxidative/nitrosative protein modifications. Free Radic. Res. 2018;52(5):507–543. doi: 10.1080/10715762.2018.1457217. [DOI] [PubMed] [Google Scholar]
- 51.Cichon M.A., Radisky D.C. ROS-induced epithelial-mesenchymal transition in mammary epithelial cells is mediated by NF-kB-dependent activation of Snail. Oncotarget. 2014;5(9):2827–2838. doi: 10.18632/oncotarget.1940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Weinberg F., Hamanaka R., Wheaton W.W., Weinberg S., Joseph J., Lopez M., Kalyanaraman B., Mutlu G.M., Budinger G.R., Chandel N.S. Mitochondrial metabolism and ROS generation are essential for Kras-mediated tumorigenicity. Proc. Natl. Acad. Sci. USA. 2010;107(19):8788–8793. doi: 10.1073/pnas.1003428107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lu H., Samanta D., Xiang L., Zhang H., Hu H., Chen I., Bullen J.W., Semenza G.L. Chemotherapy triggers HIF-1-dependent glutathione synthesis and copper chelation that induces the breast cancer stem cell phenotype. Proc. Natl. Acad. Sci. USA. 2015;112(33):E4600–E4609. doi: 10.1073/pnas.1513433112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kennedy L., Sandhu J.K., Harper M.E., Cuperlovic-Culf M. Role of glutathione in cancer: from mechanisms to therapies. Biomolecules. 2020;10(10) doi: 10.3390/biom10101429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Agarwal M.K., Iqbal M., Athar M. Inhibitory effect of 18beta-glycyrrhetinic acid on 12-O-tetradecanoyl phorbol-13-acetate-induced cutaneous oxidative stress and tumor promotion in mice. Redox Rep. 2005;10(3):151–157. doi: 10.1179/135100005X57346. [DOI] [PubMed] [Google Scholar]
- 56.Crusz S.M., Balkwill F.R. Inflammation and cancer: advances and new agents. Nat. Rev. Clin. Oncol. 2015;12(10):584–596. doi: 10.1038/nrclinonc.2015.105. [DOI] [PubMed] [Google Scholar]
- 57.Eiro N., Vizoso F.J. Inflammation and cancer. World J. Gastrointest. Surg. 2012;4(3):62–72. doi: 10.4240/wjgs.v4.i3.62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Rajput S., Wilber A. Roles of inflammation in cancer initiation, progression, and metastasis. Front Biosci. (Sch. Ed. ) 2010;2:176–183. doi: 10.2741/s55. [DOI] [PubMed] [Google Scholar]
- 59.Dubois R.N. Role of inflammation and inflammatory mediators in colorectal cancer. Trans. Am. Clin. Clim. Assoc. 2014;125:358–372. discussion 372-3. [PMC free article] [PubMed] [Google Scholar]
- 60.Tong B.J., Tan J., Tajeda L., Das S.K., Chapman J.A., DuBois R.N., Dey S.K. Heightened expression of cyclooxygenase-2 and peroxisome proliferator-activated receptor-delta in human endometrial adenocarcinoma. Neoplasia. 2000;2(6):483–490. doi: 10.1038/sj.neo.7900119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cao D., Jia Z., You L., Wu Y., Hou Z., Suo Y., Zhang H., Wen S., Tsukamoto T., Oshima M., Jiang J., Cao X. 18β-glycyrrhetinic acid suppresses gastric cancer by activation of miR-149-3p-Wnt-1 signaling. Oncotarget. 2016;7(44):71960–71973. doi: 10.18632/oncotarget.12443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ermert L., Dierkes C., Ermert M. Immunohistochemical expression of cyclooxygenase isoenzymes and downstream enzymes in human lung tumors. Clin. Cancer Res. 2003;9(5):1604–1610. [PubMed] [Google Scholar]
- 63.Cathcart M.C., Gately K., Cummins R., Kay E., O’Byrne K.J., Pidgeon G.P. Examination of thromboxane synthase as a prognostic factor and therapeutic target in non-small cell lung cancer. Mol. Cancer. 2011;10:25. doi: 10.1186/1476-4598-10-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Huang R.Y., Chen G.G. Cigarette smoking, cyclooxygenase-2 pathway and cancer. Biochim. Biophys. Acta. 2011;1815(2):158–169. doi: 10.1016/j.bbcan.2010.11.005. [DOI] [PubMed] [Google Scholar]
- 65.Wei J., Yan W., Li X., Ding Y., Tai H.H. Thromboxane receptor alpha mediates tumor growth and angiogenesis via induction of vascular endothelial growth factor expression in human lung cancer cells. Lung Cancer. 2010;69(1):26–32. doi: 10.1016/j.lungcan.2009.09.009. [DOI] [PubMed] [Google Scholar]
- 66.Wei J., Yan W., Li X., Chang W.C., Tai H.H. Activation of thromboxane receptor alpha induces expression of cyclooxygenase-2 through multiple signaling pathways in A549 human lung adenocarcinoma cells. Biochem. Pharmacol. 2007;74(5):787–800. doi: 10.1016/j.bcp.2007.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Huang R.Y., Chu Y.L., Huang Q.C., Chen X.M., Jiang Z.B., Zhang X., Zeng X. 18beta-Glycyrrhetinic acid suppresses cell proliferation through inhibiting thromboxane synthase in non-small cell lung cancer. PLoS One. 2014;9(4) doi: 10.1371/journal.pone.0093690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Moussa O., Riker J.M., Klein J., Fraig M., Halushka P.V., Watson D.K. Inhibition of thromboxane synthase activity modulates bladder cancer cell responses to chemotherapeutic agents. Oncogene. 2008;27(1):55–62. doi: 10.1038/sj.onc.1210629. [DOI] [PubMed] [Google Scholar]
- 69.Deng Q.P., Wang M.J., Zeng X., Chen G.G., Huang R.Y. Effects of glycyrrhizin in a mouse model of lung adenocarcinoma. Cell Physiol. Biochem. 2017;41(4):1383–1392. doi: 10.1159/000467897. [DOI] [PubMed] [Google Scholar]
- 70.Khan R., Khan A.Q., Lateef A., Rehman M.U., Tahir M., Ali F., Hamiza O.O., Sultana S. Glycyrrhizic acid suppresses the development of precancerous lesions via regulating the hyperproliferation, inflammation, angiogenesis and apoptosis in the colon of Wistar rats. PLoS One. 2013;8(2) doi: 10.1371/journal.pone.0056020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Wang G., Hiramoto K., Ma N., Yoshikawa N., Ohnishi S., Murata M., Kawanishi S. Glycyrrhizin attenuates carcinogenesis by inhibiting the inflammatory response in a murine model of colorectal cancer. Int. J. Mol. Sci. 2021;22(5) doi: 10.3390/ijms22052609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kang R., Zhang Q., Zeh H.J., 3rd, Lotze M.T., Tang D. HMGB1 in cancer: good, bad, or both? Clin. Cancer Res. 2013;19(15):4046–4057. doi: 10.1158/1078-0432.CCR-13-0495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Liu B., Qu L., Yan S. Cyclooxygenase-2 promotes tumor growth and suppresses tumor immunity. Cancer Cell Int. 2015;15:106. doi: 10.1186/s12935-015-0260-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Becker M.R., Siegelin M.D., Rompel R., Enk A.H., Gaiser T. COX-2 expression in malignant melanoma: a novel prognostic marker? Melanoma Res. 2009;19(1):8–16. doi: 10.1097/CMR.0b013e32831d7f52. [DOI] [PubMed] [Google Scholar]
- 75.Juin S.K., Ghosh S., Majumdar S. Glycyrrhizic acid facilitates anti-tumor immunity by attenuating Tregs and MDSCs: n immunotherapeutiac approach. Int. Immunopharmacol. 2020;88 doi: 10.1016/j.intimp.2020.106932. [DOI] [PubMed] [Google Scholar]
- 76.Mougiakakos D., Johansson C.C., Trocme E., All-Ericsson C., Economou M.A., Larsson O., Seregard S., Kiessling R. Intratumoral forkhead box P3-positive regulatory T cells predict poor survival in cyclooxygenase-2-positive uveal melanoma. Cancer. 2010;116(9):2224–2233. doi: 10.1002/cncr.24999. [DOI] [PubMed] [Google Scholar]
- 77.Blattner C., Fleming V., Weber R., Himmelhan B., Altevogt P., Gebhardt C., Schulze T.J., Razon H., Hawila E., Wildbaum G., Utikal J., Karin N., Umansky V. CCR5(+) myeloid-derived suppressor cells are enriched and activated in melanoma lesions. Cancer Res. 2018;78(1):157–167. doi: 10.1158/0008-5472.CAN-17-0348. [DOI] [PubMed] [Google Scholar]
- 78.Niwa K., Lian Z., Onogi K., Yun W., Tang L., Mori H., Tamaya T. Preventive effects of glycyrrhizin on estrogen-related endometrial carcinogenesis in mice. Oncol. Rep. 2007;17(3):617–622. [PubMed] [Google Scholar]
- 79.Zhang Q., Shen Q., Celestino J., Milam M.R., Westin S.N., Lacour R.A., Meyer L.A., Shipley G.L., Davies P.J., Deng L., McCampbell A.S., Broaddus R.R., Lu K.H. Enhanced estrogen-induced proliferation in obese rat endometrium. Am. J. Obstet. Gynecol. 2009;200(2):186 e1–e8. doi: 10.1016/j.ajog.2008.08.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kumagai A., Nishino K., Shimomura A., Kin T., Yamamura Y. Effect of glycyrrhizin on estrogen action. Endocrinol. Jpn. 1967;14(1):34–38. doi: 10.1507/endocrj1954.14.34. [DOI] [PubMed] [Google Scholar]
- 81.Matthews H.K., Bertoli C., de Bruin R.A.M. Cell cycle control in cancer. Nat. Rev. Mol. Cell Biol. 2022;23(1):74–88. doi: 10.1038/s41580-021-00404-3. [DOI] [PubMed] [Google Scholar]
- 82.Satomi Y., Nishino H., Shibata S. Glycyrrhetinic acid and related compounds induce G1 arrest and apoptosis in human hepatocellular carcinoma HepG2. Anticancer Res. 2005;25(6B):4043–4047. [PubMed] [Google Scholar]
- 83.Chang M., Wu M., Li H. Curcumin combined with glycyrrhetinic acid inhibits the development of hepatocellular carcinoma cells by down-regulating the PTEN/PI3K/AKT signalling pathway. Am. J. Transl. Res. 2017;9(12):5567–5575. [PMC free article] [PubMed] [Google Scholar]
- 84.Wang H., Ge X., Qu H., Wang N., Zhou J., Xu W., Xie J., Zhou Y., Shi L., Qin Z., Jiang Z., Yin W., Xia J. Glycyrrhizic acid inhibits proliferation of gastric cancer cells by inducing cell cycle arrest and apoptosis. Cancer Manag. Res. 2020;12:2853–2861. doi: 10.2147/CMAR.S244481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Hanahan D., Weinberg R.A. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 86.Xu X., Lai Y., Hua Z.C. Apoptosis and apoptotic body: disease message and therapeutic target potentials. Biosci. Rep. 2019;39(1):1930–1943. doi: 10.1042/BSR20180992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Lowe S.W., Cepero E., Evan G. Intrinsic tumour suppression. Nature. 2004;432(7015):307–315. doi: 10.1038/nature03098. [DOI] [PubMed] [Google Scholar]
- 88.Evan G.I., Vousden K.H. Proliferation, cell cycle and apoptosis in cancer. Nature. 2001;411(6835):342–348. doi: 10.1038/35077213. [DOI] [PubMed] [Google Scholar]
- 89.Adams J.M., Cory S. The Bcl-2 apoptotic switch in cancer development and therapy. Oncogene. 2007;26(9):1324–1337. doi: 10.1038/sj.onc.1210220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Junttila M.R., Evan G.I. p53--a Jack of all trades but master of none. Nat. Rev. Cancer. 2009;9(11):821–829. doi: 10.1038/nrc2728. [DOI] [PubMed] [Google Scholar]
- 91.Lin K.W., Huang A.M., Hour T.C., Yang S.C., Pu Y.S., Lin C.N. 18beta-Glycyrrhetinic acid derivatives induced mitochondrial-mediated apoptosis through reactive oxygen species-mediated p53 activation in NTUB1 cells. Bioorg. Med. Chem. 2011;19(14):4274–4285. doi: 10.1016/j.bmc.2011.05.054. [DOI] [PubMed] [Google Scholar]
- 92.Sharma G., Kar S., Palit S., Das P.K. 18β-glycyrrhetinic acid induces apoptosis through modulation of Akt/FOXO3a/Bim pathway in human breast cancer MCF-7 cells. J. Cell Physiol. 2012;227(5):1923–1931. doi: 10.1002/jcp.22920. [DOI] [PubMed] [Google Scholar]
- 93.Tait S.W., Green D.R. Caspase-independent cell death: leaving the set without the final cut. Oncogene. 2008;27(50):6452–6461. doi: 10.1038/onc.2008.311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Thirugnanam S., Xu L., Ramaswamy K., Gnanasekar M. Glycyrrhizin induces apoptosis in prostate cancer cell lines DU-145 and LNCaP. Oncol. Rep. 2008;20(6):1387–1392. [PubMed] [Google Scholar]
- 95.Lin S.C., Chu P.Y., Liao W.T., Wu M.Y., Tsui K.H., Lin L.T., Huang C.H., Chen L.L., Li C.J. Glycyrrhizic acid induces human MDA-MB-231 breast cancer cell death and autophagy via the ROS-mitochondrial pathway. Oncol. Rep. 2018;39(2):703–710. doi: 10.3892/or.2017.6123. [DOI] [PubMed] [Google Scholar]
- 96.Shen S., Zhou M., Huang K., Wu Y., Ma Y., Wang J., Ma J., Fan S. Blocking autophagy enhances the apoptotic effect of 18β-glycyrrhetinic acid on human sarcoma cells via endoplasmic reticulum stress and JNK activation. Cell Death Dis. 2017;8(9) doi: 10.1038/cddis.2017.441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Zeeshan R., Mutahir Z. Cancer metastasis - tricks of the trade. Bosn. J. Basic Med. Sci. 2017;17(3):172–182. doi: 10.17305/bjbms.2017.1908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Robert J. Biology of cancer metastasis. Bull. Cancer. 2013;100(4):333–342. doi: 10.1684/bdc.2013.1724. [DOI] [PubMed] [Google Scholar]
- 99.Jayasooriya R.G., Dilshara M.G., Park S.R., Choi Y.H., Hyun J.W., Chang W.Y., Kim G.Y. 18β-Glycyrrhetinic acid suppresses TNF-α induced matrix metalloproteinase-9 and vascular endothelial growth factor by suppressing the Akt-dependent NF-κB pathway. Toxicol. In Vitro. 2014;28(5):751–758. doi: 10.1016/j.tiv.2014.02.015. [DOI] [PubMed] [Google Scholar]
- 100.Pahl H.L. Activators and target genes of Rel/NF-κB transcription factors. Oncogene. 1999;18(49):6853–6866. doi: 10.1038/sj.onc.1203239. [DOI] [PubMed] [Google Scholar]
- 101.Shetty A.V., Thirugnanam S., Dakshinamoorthy G., Samykutty A., Zheng G., Chen A., Bosland M.C., Kajdacsy-Balla A., Gnanasekar M. 18alpha-glycyrrhetinic acid targets prostate cancer cells by down-regulating inflammation-related genes. Int. J. Oncol. 2011;39(3):635–640. doi: 10.3892/ijo.2011.1061. [DOI] [PubMed] [Google Scholar]
- 102.Abe K., Ikeda T., Wake K., Sato T., Sato T., Inoue H. Glycyrrhizin prevents of lipopolysaccharide/D-galactosamine-induced liver injury through down-regulation of matrix metalloproteinase-9 in mice. J. Pharm. Pharmacol. 2008;60(1):91–97. doi: 10.1211/jpp.60.1.0012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Visse R., Nagase H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: structure, function, and biochemistry. Circ. Res. 2003;92(8):827–839. doi: 10.1161/01.RES.0000070112.80711.3D. [DOI] [PubMed] [Google Scholar]
- 104.Qiu M., Huang K., Liu Y., Yang Y., Tang H., Liu X., Wang C., Chen H., Xiong Y., Zhang J., Yang J. Modulation of intestinal microbiota by glycyrrhizic acid prevents high-fat diet-enhanced pre-metastatic niche formation and metastasis. Mucosal Immunol. 2019;12(4):945–957. doi: 10.1038/s41385-019-0144-6. [DOI] [PubMed] [Google Scholar]
- 105.Baj J., Korona-Glowniak I., Forma A., Maani A., Sitarz E., Rahnama-Hezavah M., Radzikowska E., Portincasa P. Mechanisms of the epithelial-mesenchymal transition and tumor microenvironment in helicobacter pylori-induced gastric cancer. Cells. 2020;9(4) doi: 10.3390/cells9041055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Hay E.D. An overview of epithelio-mesenchymal transformation. Acta Anat. 1995:8–20. doi: 10.1159/000147748. [DOI] [PubMed] [Google Scholar]
- 107.Kalluri R., Weinberg R.A. The basics of epithelial-mesenchymal transition. J. Clin. Investig. 2009;119(6):1420–1428. doi: 10.1172/JCI39104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Du B., Shim J.S. Targeting epithelial-mesenchymal transition (EMT) to overcome drug resistance in cancer. Molecules. 2016;21(7) doi: 10.3390/molecules21070965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Cai H., Chen X., Zhang J., Wang J. 18beta-glycyrrhetinic acid inhibits migration and invasion of human gastric cancer cells via the ROS/PKC-alpha/ERK pathway. J. Nat. Med. 2018;72(1):252–259. doi: 10.1007/s11418-017-1145-y. [DOI] [PubMed] [Google Scholar]
- 110.Zhang J., Zhao R., Xing D., Cao J., Guo Y., Li L., Sun Y., Tian L., Liu M. Magnesium isoglycyrrhizinate induces an inhibitory effect on progression and epithelial-mesenchymal transition of laryngeal cancer via the NF-kappaB/twist signaling. Drug Des. Dev. Ther. 2020;14:5633–5644. doi: 10.2147/DDDT.S272323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Yu L., Li H.Z., Lu S.M., Liu W.W., Li J.F., Wang H.B., Xu W. Alteration in TWIST expression: possible role in paclitaxel-induced apoptosis in human laryngeal carcinoma Hep-2 cell line. Croat. Med. J. 2009;50(6):536–542. doi: 10.3325/cmj.2009.50.536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Cheng G.Z., Zhang W., Wang L.H. Regulation of cancer cell survival, migration, and invasion by Twist: AKT2 comes to interplay. Cancer Res. 2008;68(4):957–960. doi: 10.1158/0008-5472.CAN-07-5067. [DOI] [PubMed] [Google Scholar]
- 113.Sasahira T., Akama Y., Fujii K., Kuniyasu H. Expression of receptor for advanced glycation end products and HMGB1/amphoterin in colorectal adenomas. Virchows Arch. 2005;446(4):411–415. doi: 10.1007/s00428-005-1210-x. [DOI] [PubMed] [Google Scholar]
- 114.Wei F., Yang F., Li J., Zheng Y., Yu W., Yang L., Ren X. Soluble Toll-like receptor 4 is a potential serum biomarker in non-small cell lung cancer. Oncotarget. 2016;7(26):40106–40114. doi: 10.18632/oncotarget.9496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Chang H.Y., Chen S.Y., Wu C.H., Lu C.C., Yen G.C. Glycyrrhizin attenuates the process of epithelial-to-mesenchymal transition by modulating HMGB1 initiated novel signaling pathway in prostate cancer cells. J. Agric. Food Chem. 2019;67(12):3323–3332. doi: 10.1021/acs.jafc.9b00251. [DOI] [PubMed] [Google Scholar]
- 116.Abdel-Wahab A.A., Effat H., Mahrous E.A., Ali M.A., Al-Shafie T.A. A licorice roots extract induces apoptosis and cell cycle arrest and improves metabolism via regulating MiRNAs in liver cancer cells. Nutr. Cancer. 2021;73(6):1047–1058. doi: 10.1080/01635581.2020.1783329. [DOI] [PubMed] [Google Scholar]
- 117.Luo G., Chao Y.L., Tang B., Li B.S., Xiao Y.F., Xie R., Wang S.M., Wu Y.Y., Dong H., Liu X.D., Yang S.M. miR-149 represses metastasis of hepatocellular carcinoma by targeting actin-regulatory proteins PPM1F. Oncotarget. 2015;6(35):37808–37823. doi: 10.18632/oncotarget.5676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Wang Y., Zheng X., Zhang Z., Zhou J., Zhao G., Yang J., Xia L., Wang R., Cai X., Hu H., Zhu C., Nie Y., Wu K., Zhang D., Fan D. MicroRNA-149 inhibits proliferation and cell cycle progression through the targeting of ZBTB2 in human gastric cancer. PLoS One. 2012;7(10) doi: 10.1371/journal.pone.0041693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Chiurillo M.A. Role of the Wnt/beta-catenin pathway in gastric cancer: an in-depth literature review. World J. Exp. Med. 2015;5(2):84–102. doi: 10.5493/wjem.v5.i2.84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Soleimani A., Rahmani F., Ferns G.A., Ryzhikov M., Avan A., Hassanian S.M. Role of the NF-kappaB signaling pathway in the pathogenesis of colorectal cancer. Gene. 2020;726 doi: 10.1016/j.gene.2019.144132. [DOI] [PubMed] [Google Scholar]
- 121.Karin M. Nuclear factor-kappaB in cancer development and progression. Nature. 2006;441(7092):431–436. doi: 10.1038/nature04870. [DOI] [PubMed] [Google Scholar]
- 122.Das N., Dewan V., Grace P.M., Gunn R.J., Tamura R., Tzarum N., Watkins L.R., Wilson I.A., Yin H. HMGB1 activates proinflammatory signaling via TLR5 leading to allodynia. Cell Rep. 2016;17(4):1128–1140. doi: 10.1016/j.celrep.2016.09.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Sims G.P., Rowe D.C., Rietdijk S.T., Herbst R., Coyle A.J. HMGB1 and RAGE in inflammation and cancer. Annu. Rev. Immunol. 2010;28:367–388. doi: 10.1146/annurev.immunol.021908.132603. [DOI] [PubMed] [Google Scholar]
- 124.Shi X., Yu L., Zhang Y., Liu Z., Zhang H., Zhang Y., Liu P., Du P. Glycyrrhetinic acid alleviates hepatic inflammation injury in viral hepatitis disease via a HMGB1-TLR4 signaling pathway. Int. Immunopharmacol. 2020;84 doi: 10.1016/j.intimp.2020.106578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Weng H., Deng Y., Xie Y., Liu H., Gong F. Expression and significance of HMGB1, TLR4 and NF-kappaB p65 in human epidermal tumors. BMC Cancer. 2013;13:311. doi: 10.1186/1471-2407-13-311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Wu X., Wang W., Chen Y., Liu X., Wang J., Qin X., Yuan D., Yu T., Chen G., Mi Y., Mou J., Cui J., Hu A., D. Pei Y.E. Glycyrrhizin suppresses the growth of human NSCLC cell line HCC827 by downregulating HMGB1 level. Biomed. Res. Int. 2018;2018 doi: 10.1155/2018/6916797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Kang R., Xie Y., Zhang Q., Hou W., Jiang Q., Zhu S., Liu J., Zeng D., Wang H., Bartlett D.L., Billiar T.R., Zeh H.J., 3rd, Lotze M.T., Tang D. Intracellular HMGB1 as a novel tumor suppressor of pancreatic cancer. Cell Res. 2017;27(7):916–932. doi: 10.1038/cr.2017.51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Hsiang C.Y., Lin L.J., Kao S.T., Lo H.Y., Chou S.T., Ho T.Y. Glycyrrhizin, silymarin, and ursodeoxycholic acid regulate a common hepatoprotective pathway in HepG2 cells. Phytomedicine. 2015;22(7–8):768–777. doi: 10.1016/j.phymed.2015.05.053. [DOI] [PubMed] [Google Scholar]
- 129.Chen H.J., Kang S.P., Lee I.J., Lin Y.L. Glycyrrhetinic acid suppressed NF-κB activation in TNF-α-induced hepatocytes. J. Agric. Food Chem. 2014;62(3):618–625. doi: 10.1021/jf405352g. [DOI] [PubMed] [Google Scholar]
- 130.Li S., Zhu J.H., Cao L.P., Sun Q., Liu H.D., Li W.D., Li J.S., Hang C.H. Growth inhibitory in vitro effects of glycyrrhizic acid in U251 glioblastoma cell line. Neurol. Sci. 2014;35(7):1115–1120. doi: 10.1007/s10072-014-1661-4. [DOI] [PubMed] [Google Scholar]
- 131.Haughian J.M., Jackson T.A., Koterwas D.M., Bradford A.P. Endometrial cancer cell survival and apoptosis is regulated by protein kinase C alpha and delta. Endocr. Relat. Cancer. 2006;13(4):1251–1267. doi: 10.1677/erc.1.01278. [DOI] [PubMed] [Google Scholar]
- 132.Webb B.L., Hirst S.J., Giembycz M.A. Protein kinase C isoenzymes: a review of their structure, regulation and role in regulating airways smooth muscle tone and mitogenesis. Br. J. Pharmacol. 2000;130(7):1433–1452. doi: 10.1038/sj.bjp.0703452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Song J., Ko H.S., Sohn E.J., Kim B., Kim J.H., Kim H.J., Kim C., Kim J.E., Kim S.H. Inhibition of protein kinase C alpha/betaII and activation of c-Jun NH2-terminal kinase mediate glycyrrhetinic acid induced apoptosis in non-small cell lung cancer NCI-H460 cells. Bioorg. Med. Chem. Lett. 2014;24(4):1188–1191. doi: 10.1016/j.bmcl.2013.12.111. [DOI] [PubMed] [Google Scholar]
- 134.Ueda Y., Hirai S., Osada S., Suzuki A., Mizuno K., Ohno S. Protein kinase C activates the MEK-ERK pathway in a manner independent of Ras and dependent on Raf. J. Biol. Chem. 1996;271(38):23512–23519. doi: 10.1074/jbc.271.38.23512. [DOI] [PubMed] [Google Scholar]
- 135.De Luca A., Maiello M.R., D’Alessio A., Pergameno M., Normanno N. The RAS/RAF/MEK/ERK and the PI3K/AKT signalling pathways: role in cancer pathogenesis and implications for therapeutic approaches. Expert Opin. Ther. Targets. 2012;16(Suppl 2):S17–S27. doi: 10.1517/14728222.2011.639361. [DOI] [PubMed] [Google Scholar]
- 136.Burotto M., Chiou V.L., Lee J.M., Kohn E.C. The MAPK pathway across different malignancies: a new perspective. Cancer. 2014;120(22):3446–3456. doi: 10.1002/cncr.28864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Yang S.H., Sharrocks A.D., Whitmarsh A.J. MAP kinase signalling cascades and transcriptional regulation. Gene. 2013;513(1):1–13. doi: 10.1016/j.gene.2012.10.033. [DOI] [PubMed] [Google Scholar]
- 138.Wang D., Wong H.K., Feng Y.B., Zhang Z.J. 18beta-glycyrrhetinic acid induces apoptosis in pituitary adenoma cells via ROS/MAPKs-mediated pathway. J. Neurooncol. 2014;116(2):221–230. doi: 10.1007/s11060-013-1292-2. [DOI] [PubMed] [Google Scholar]
- 139.Alzahrani A.S. PI3K/Akt/mTOR inhibitors in cancer: at the bench and bedside. Semin. Cancer Biol. 2019;59:125–132. doi: 10.1016/j.semcancer.2019.07.009. [DOI] [PubMed] [Google Scholar]
- 140.Abu-Eid R., Ward F.J. Targeting the PI3K/Akt/mTOR pathway: a therapeutic strategy in COVID-19 patients. Immunol. Lett. 2021;240:1–8. doi: 10.1016/j.imlet.2021.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Iksen S., Pothongsrisit V., Pongrakhananon Targeting the PI3K/AKT/mTOR signaling pathway in lung cancer: an update regarding potential drugs and natural products. Molecules. 2021;26(13) doi: 10.3390/molecules26134100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Miricescu D., Totan A., Stanescu S., II, Badoiu S.C., Stefani C., Greabu M. PI3K/AKT/mTOR signaling pathway in breast cancer: from molecular landscape to clinical aspects. Int. J. Mol. Sci. 2020;22(1) doi: 10.3390/ijms22010173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.He S.Q., Gao M., Fu Y.F., Zhang Y.N. Glycyrrhizic acid inhibits leukemia cell growth and migration via blocking AKT/mTOR/STAT3 signaling. Int. J. Clin. Exp. Pathol. 2015;8(5):5175–5181. [PMC free article] [PubMed] [Google Scholar]
- 144.Foulkes W.D., Smith I.E., Reis-Filho J.S. Triple-negative breast cancer. N. Engl. J. Med. 2010;363(20):1938–1948. doi: 10.1056/NEJMra1001389. [DOI] [PubMed] [Google Scholar]
- 145.Millis S.Z., Gatalica Z., Winkler J., Vranic S., Kimbrough J., Reddy S., O’Shaughnessy J.A. Predictive biomarker profiling of > 6000 breast cancer patients shows heterogeneity in TNBC, with treatment implications. Clin. Breast Cancer. 2015;15(6):473–481 e3. doi: 10.1016/j.clbc.2015.04.008. [DOI] [PubMed] [Google Scholar]
- 146.Mrklic I., Pogorelic Z., Capkun V., Tomic S. Expression of topoisomerase II-alpha in triple negative breast cancer. Appl. Immunohistochem. Mol. Morphol. 2014;22(3):182–187. doi: 10.1097/PAI.0b013e3182910967. [DOI] [PubMed] [Google Scholar]
- 147.Montecucco A., Zanetta F., Biamonti G. Molecular mechanisms of etoposide. EXCLI J. 2015;14:95–108. doi: 10.17179/excli2015-561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Hande K.R. Etoposide: four decades of development of a topoisomerase II inhibitor. Eur. J. Cancer. 1998;34(10):1514–1521. doi: 10.1016/s0959-8049(98)00228-7. [DOI] [PubMed] [Google Scholar]
- 149.Alpsoy A., Yasa S., Gunduz U. Etoposide resistance in MCF-7 breast cancer cell line is marked by multiple mechanisms. Biomed. Pharmacother. 2014;68(3):351–355. doi: 10.1016/j.biopha.2013.09.007. [DOI] [PubMed] [Google Scholar]
- 150.Marengo B., Monti P., Miele M., Menichini P., Ottaggio L., Foggetti G., Pulliero A., Izzotti A., Speciale A., Garbarino O., Traverso N., Fronza G., Domenicotti C. Etoposide-resistance in a neuroblastoma model cell line is associated with 13q14.3 mono-allelic deletion and miRNA-15a/16-1 down-regulation. Sci. Rep. 2018;8(1):13762. doi: 10.1038/s41598-018-32195-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Qiu Z., Lin A., Li K., Lin W., Wang Q., Wei T., Zhu W., Luo P., Zhang J. A novel mutation panel for predicting etoposide resistance in small-cell lung cancer. Drug Des. Dev. Ther. 2019;13:2021–2041. doi: 10.2147/DDDT.S205633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Cai Y., Zhao B., Liang Q., Zhang Y., Cai J., Li G. The selective effect of glycyrrhizin and glycyrrhetinic acid on topoisomerase IIalpha and apoptosis in combination with etoposide on triple negative breast cancer MDA-MB-231 cells. Eur. J. Pharmacol. 2017;809:87–97. doi: 10.1016/j.ejphar.2017.05.026. [DOI] [PubMed] [Google Scholar]
- 153.Katano H. Pathological features of Kaposi’s sarcoma-associated herpesvirus infection. Adv. Exp. Med. Biol. 2018;1045:357–376. doi: 10.1007/978-981-10-7230-7_16. [DOI] [PubMed] [Google Scholar]
- 154.Wen K.W., Damania B. Kaposi sarcoma-associated herpesvirus (KSHV): molecular biology and oncogenesis. Cancer Lett. 2010;289(2):140–150. doi: 10.1016/j.canlet.2009.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Schneider J.W., Dittmer D.P. Diagnosis and treatment of kaposi sarcoma. Am. J. Clin. Dermatol. 2017;18(4):529–539. doi: 10.1007/s40257-017-0270-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Kang H., Lieberman P.M. Mechanism of glycyrrhizic acid inhibition of Kaposi’s sarcoma-associated herpesvirus: disruption of CTCF-cohesin-mediated RNA polymerase II pausing and sister chromatid cohesion. J. Virol. 2011;85(21):11159–11169. doi: 10.1128/JVI.00720-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Curreli F., Friedman-Kien A.E., Flore O. Glycyrrhizic acid alters Kaposi sarcoma-associated herpesvirus latency, triggering p53-mediated apoptosis in transformed B lymphocytes. J. Clin. Investig. 2005;115(3):642–652. doi: 10.1172/JCI23334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Dasari S., Tchounwou P.B. Cisplatin in cancer therapy: molecular mechanisms of action. Eur. J. Pharmacol. 2014;740:364–378. doi: 10.1016/j.ejphar.2014.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.de Jongh F.E., van Veen R.N., Veltman S.J., de Wit R., van der Burg M.E., van den Bent M.J., Planting A.S., Graveland W.J., Stoter G., Verweij J. Weekly high-dose cisplatin is a feasible treatment option: analysis on prognostic factors for toxicity in 400 patients. Br. J. Cancer. 2003;88(8):1199–1206. doi: 10.1038/sj.bjc.6600884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Wu C.H., Chen A.Z., Yen G.C. Protective effects of glycyrrhizic acid and 18beta-glycyrrhetinic acid against cisplatin-induced nephrotoxicity in BALB/c mice. J. Agric. Food Chem. 2015;63(4):1200–1209. doi: 10.1021/jf505471a. [DOI] [PubMed] [Google Scholar]
- 161.Yan Y., Mo Y., Zhang D. Magnesium isoglycyrrhizinate prevention of chemotherapy-induced liver damage during initial treatment of patients with gastrointestinal tumors. Zhonghua Gan Zang Bing. Za Zhi. 2015;23(3):204–208. doi: 10.3760/cma.j.issn.1007-3418.2015.03.010. [DOI] [PubMed] [Google Scholar]
- 162.Wang Y., Wang Z., Gao M., Zhong H., Chen C., Yao Y., Zhang Z., Zhang X., Li F., Zhang J., Gu H.M., Chen Y., Tang J., Zhong W., Zeng M., Mao Y. Efficacy and safety of magnesium isoglycyrrhizinate injection in patients with acute drug‐induced liver injury: a phase II trial. Liver Int. 2019;39(11):2102–2111. doi: 10.1111/liv.14204. [DOI] [PubMed] [Google Scholar]
- 163.Su X., Wu L., Hu M., Dong W., Xu M., Zhang P. Glycyrrhizic acid: a promising carrier material for anticancer therapy. Biomed. Pharmacother. 2017;95:670–678. doi: 10.1016/j.biopha.2017.08.123. [DOI] [PubMed] [Google Scholar]
- 164.Zhu X.L., Wang S.S., Li L.H., Zhang H.J., Xia T., Lyu X.Y. Preparation and characterization of glycyrrhetinic acid-modified nano graphene oxide drug delivery system. Zhongguo Zhong Yao Za Zhi. 2019;44(21):4621–4626. doi: 10.19540/j.cnki.cjcmm.20190804.305. [DOI] [PubMed] [Google Scholar]
- 165.Xue H., Qin L., Zhang L., Li X., Wu F., Wang W., Wang C., Diao W., Jiang B., Lian B., Wu J., Bai J., Sun T., Zhao C., Qu M., Yu W., Wang Y., Gao Z. Preparation of docetaxel-loaded, glycyrrhetinic acid-modified nanoparticles and their liver-targeting and antitumor activity. Exp. Ther. Med. 2021;22(4):1144. doi: 10.3892/etm.2021.10578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Stecanella L.A., Bitencourt A.P.R., Vaz G.R., Quarta E., Silva Júnior J.O.C., Rossi A. Glycyrrhizic acid and its hydrolyzed metabolite 18β-glycyrrhetinic acid as specific ligands for targeting nanosystems in the treatment of liver cancer. Pharmaceutics. 2021;13(11) doi: 10.3390/pharmaceutics13111792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Calo L.A., Zaghetto F., Pagnin E., Davis P.A., De Mozzi P., Sartorato P., Martire G., Fiore C., Armanini D. Effect of aldosterone and glycyrrhetinic acid on the protein expression of PAI-1 and p22(phox) in human mononuclear leukocytes. J. Clin. Endocrinol. Metab. 2004;89(4):1973–1976. doi: 10.1210/jc.2003-031545. [DOI] [PubMed] [Google Scholar]
- 168.Omar H.R., Komarova I., El-Ghonemi M., Fathy A., Rashad R., Abdelmalak H.D., Yerramadha M.R., Ali Y., Helal E., Camporesi E.M. Licorice abuse: time to send a warning message. Ther. Adv. Endocrinol. Metab. 2012;3(4):125–138. doi: 10.1177/2042018812454322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Palermo M., Armanini D., Delitala G. Grapefruit juice inhibits 11beta-hydroxysteroid dehydrogenase in vivo, in man. Clin. Endocrinol. 2003;59(1):143–144. doi: 10.1046/j.1365-2265.2003.01806.x. [DOI] [PubMed] [Google Scholar]
- 170.Isbrucker R.A., Burdock G.A. Risk and safety assessment on the consumption of Licorice root (Glycyrrhiza sp.), its extract and powder as a food ingredient, with emphasis on the pharmacology and toxicology of glycyrrhizin. Regul. Toxicol. Pharmacol. 2006;46(3):167–192. doi: 10.1016/j.yrtph.2006.06.002. [DOI] [PubMed] [Google Scholar]
- 171.Nazari S., Rameshrad M., Hosseinzadeh H. Toxicological effects of Glycyrrhiza glabra (Licorice): a review. Phytother. Res. 2017;31(11):1635–1650. doi: 10.1002/ptr.5893. [DOI] [PubMed] [Google Scholar]
- 172.Kwon Y.J., Son D.H., Chung T.H., Lee Y.J. A review of the pharmacological efficacy and safety of licorice root from corroborative clinical trial findings. J. Med. Food. 2020;23(1):12–20. doi: 10.1089/jmf.2019.4459. [DOI] [PubMed] [Google Scholar]
- 173.Wang X.F., Zhou Q.M., Lu Y.Y., Zhang H., Huang S., Su S.B. Glycyrrhetinic acid potently suppresses breast cancer invasion and metastasis by impairing the p38 MAPK-AP1 signaling axis. Expert Opin. Ther. Targets. 2015;19(5):577–587. doi: 10.1517/14728222.2015.1012156. [DOI] [PubMed] [Google Scholar]
- 174.Huang R.Y., Chu Y.L., Jiang Z.B., Chen X.M., Zhang X., Zeng X. Glycyrrhizin suppresses lung adenocarcinoma cell growth through inhibition of thromboxane synthase. Cell Physiol. Biochem. 2014;33(2):375–388. doi: 10.1159/000356677. [DOI] [PubMed] [Google Scholar]
- 175.Li J., Tang F., Li R., Chen Z., Lee S.M., Fu C., Zhang J., Leung G.P. Dietary compound glycyrrhetinic acid suppresses tumor angiogenesis and growth by modulating antiangiogenic and proapoptotic pathways in vitro and in vivo. J. Nutr. Biochem. 2020;77 doi: 10.1016/j.jnutbio.2019.108268. [DOI] [PubMed] [Google Scholar]