Abstract
SARS-CoV-2 and some other members of Coronaviridae family have recently forced a great deal of health, social, and economic issues globally. To that end, investigations have been oriented towards finding ways for reducing the burden of COVID-19. One of the occurrences which stands in the way of making the treatment of this disease less complicated is the way coronaviruses involve a variety of cells, tissues, organs, and even systems. This action is possible as a result of viral attachment to the angiotensin-converting enzyme 2 or ACE2. Thus, any kind of cell expressing ACE2 is prone to be affected by both SARS-COV and SARS-COV-2. Endocrine system is one of these at-risk systems. In this review, we have considered the relation between coronaviruses and one of the most essential organs of endocrine system: thyroid gland. This relation can be probed from two aspects: how underlying thyroid dysfunction can increase the risk of being infected by these viruses and how these viruses can alter the function of thyroid gland.
Keywords: SARS, COVID-19, Thyroid, Endocrine, Dysfunction, ACE2, Metabolic disease
1. Introduction
COVID-19 is a severe disease clinically manifested by respiratory symptoms which is able to cause the death of 15.2% of total infected individuals after 2–8 weeks of the disease initiation [1]. Recently, expanding the research domains on the effects of SARS-CoV-2 or 2019-nCoV on other tissues, organs, and systems then respiratory system has led to some significant results [[2], [3], [4]]. The importance of these subsidiary-affected tissues, organs, and systems is in complicating the treatment process, decreasing the survival rate, and increasing the global economic burden. Endocrine system is one of the pivotal players of regulating the homeostasis by providing chemical signals between a diversity of cells [5]. Thyroid gland as an essential component of this system is able to regulate the rate of metabolism by secreting a class of hormones including thyroxine [5].
Currently, thyroid dysfunction has attracted a great deal of interest because of two reasons: first, its role in increasing the risk of being infected by SARS-CoV-2 as an underlying disease and second, occurring in individuals as a complication secondary to SARS-CoV-2 infection. In this paper, on one hand, we reviewed a number of evidences manifesting the characteristics of COVID-19 patients in order to find the association between thyroid dysfunction, as a metabolic disease, and the risk of SARS-CoV-2 infection. On the other hand, we have looked into thyroid-related diseases as a side effect of COVID-19. This review might give a new insight on prognostic testing and managing COVID-19 patients for a more qualified after-survival life and a lower chance of mortality.
2. Coronavirus, ACE2, and endocrine system
According to our recent knowledge of coronaviruses, ACE2 takes a critical part in COVID-19 pathogenesis [6]. ACE2 or angiotensin-converting enzyme 2 is capable of binding to the spike protein of this member of Coronaviridae family [7]. In addition to the disease severity, this connection has also another disadvantage for the human body during SARS-CoV-2 infection: decreased amounts of ACE2 on the cellular surface [7,8]. The importance of this action can be understood after discussing the functions of ACE2 in a normal state. ACE2 is a homolog of ACE which has the ability to collate its actions by forming angiotensin (Ang) 1–7 from Ang II [9]. This suggests that ACE2 is actually functioning as a regulator of the renin-angiotensin system or RAS [10]. Being involved in RAS is enabling this enzyme to have both local and systemic impacts, in addition to altering the performance of a diversity of our systems [11,12]. We can classify the roles of ACE2 by the organs which are expressing this protein on their cells (organs including brain, heart, lung, testis, and kidneys [12]):
-
1.
Heart and vasculture: decreasing blood pressure, making some changes in cardiac structure, preventing heart failure, and enhancing cardiac remodeling [[13], [14], [15], [16], [17], [18], [19]].
-
2.
Kidneys: vasodilation, preventing proliferation and inflammation, glomerular injury, decreasing fibrosis [15,20].
-
3.
Lungs: increasing pulmonary blood pressure by Activation of the intrapulmonary RAS, developing fibrosis [11,21,22].
With respect to this expression pattern, any dysregulation in ACE2 expression leads to several metabolic and cardiovascular diseases containing hypertension [23], arrhythmia [18], myocardial infarction [17,24], diabetes [25], glomerulosclerosis [26], albuminuria [15] and etc.
Interestingly, recent investigations have revealed a relation between ACE2 and another ingredient of the endocrine system: thyroid gland [27]. As reported by Li et al. ACE2 is expressed highly in some organs including thyroid gland. Furthermore, they revealed that there is a positive correlation between ACE2 expression and CD8+ T cell enrichment and interferon response in men [28]. Recently, Rotondi et al. has also detected that the mRNA of the ACE2 gene can be found in follicular cells of thyroid sample tissues [29]. Furthermore, a recent pilot study revealed that not only thyroid cells express ACE2 gene but this expression can also be modulated by IFN-γ and TNF-α [30]. On the other hand, thyroid hormones are also capable of activating RAS by influencing the levels of other proteins such as renin and angiotensinogen [31]. Overall, infection with SARS-CoV-2 might be related to metabolic diseases because of ACE2 endocytosis after the viral attachment.
3. COVID-19 patients with underlying thyroid disease
Investigations manifesting the characteristics of COVID-19 patients identified some underlying conditions frequently repeated in these patients. Metabolic diseases seem to be a constant ingredient of these conditions [4,32,33]. However, still the booster impact of thyroid dysfunctions, as important metabolic diseases, on the risk of being infected by SARS-CoV-2 or its mortality rate needs deeper explorations. According to a report by Garg et al. [32] which examined 1482 hospitalized patients, 60 patients had an underlying metabolic disease. Among this number, 15 individuals were suffering from thyroid dysfunction while two of them had also diabetes mellitus beside their disease. In another study [34], after looking into the underlying diseases of 122,653 COVID-19 patients, it was found that only 37 individuals had an existing chronic thyroid disease. In contrast, investigations demonstrate that autoimmune thyroid disease do not put its patients into a higher risk of being infected by 2019-nCoV [35]. Recently, A review of reviews has also confirmed that having an underlying thyroid disease does not increase the risk for SARS-CoV-2 infection [36].
4. SARS-CoV-2 and thyroid gland: a precise look
There are several hypotheses suggested by researchers on how exactly coronaviruses affect thyroid gland and the level of its hormones. Studying the systemic effects of coronavirus is still in its infancy and the limited number of investigations is restricting the borders of our knowledge. In this section, we try to explain the SARS-CoV-2 cellular and molecular effects according to the explorations in this field conducted since December 2019:
Wei et al. [37] examining 5 autopsies of SARS patients concluded that this disease is able to impact follicular epithelial cells and the parafollicular cells and thereby, alter the thyroid function to some degree. They also tested the levels of both thyroid hormones, T3 and T4, and identified a considerable reduction. Furthermore, they confirmed the dual influence of coronavirus on both thyroid and hypothalamus–pituitary axis [37]. As well, Leow et al. [38] found hypothyroidism in SARS patients and suggested that this might be a result of virus-induced thyroiditis or a disturbance in the hypothalamus-piturity-thyroid axis. Furthermore, they implicated that this effect of SARS-CoV-2 is not related to autoantibodies against thyroid [38] and thus, thyroiditis or hypophysitis can define the effects of coronavirus on thyroid hormones better than autoimmune diseases [38]. There is also a paradoxical exploration which provided autopsies of SARS patients and did not detect any viruses in site of thyroid [39]. However, the chosen population in this study was composed of only 4 SARS patients [39]. Similarly, another clinical study on three COVID-19 patients detected no abnormal sign in autopsies obtained from thyroid [40].
The most recent study is conducted by Khoo et al. [41], which is a cohort study containing 621 patients, which concludes that the levels of TSH and FT4 are lower in patients diagnosed with COVID-19 while this reduction is not seen in patients admitted to the hospital without COVID-19. However, they declared that based on their definition of thyrotoxicosis (TSH <0.30 and FT4 > 23.0), “there was no suggestion of a novel COVID-19–related thyroiditis/thyrotoxicosis” [41]. Furthermore, they also suggest that reduced TSH levels might be the consequence of two different effects of SARS-CoV-2: increasing the proinflammatory cytokines such as interleukin-6 and cortisol [41]. They were able to suggest the second mechanism by excluding the patients who were receiving exogenous steroids [41]. In contrast, a retrospective study represented “a high prevalence of overt and subclinical thyrotoxicosis in patients with COVID-19” which is probably correlated with the cytokine storm and IL-6 secretion in COVID-19 patients [42]. Notwithstanding the limitations of this study, they suggested that destructive thyroiditis might be the reason why TSH and FT4 reduced due to SARS-CoV-2 infection [42]. This study disagreed with another previous study relating the low levels of TSH and TT3 to the non-thyroidal illness (NTI) [43]. On the other hand, there is an association between the time of viral nucleic acid cleaning and thyroid dysfunction which might strengthen the idea of direct viral infection [43].
The non-thyroidal illness (NTI) hypothesis indicates that during a systemic illness, a total reduction can be observed in T3, T4, and TSH which is preserving energy in the body [44]. NTI is mediated through a variety of factors including circulating cytokines including IL-6 and TNF-α. Considering the SARS-related cytokine storm, Croce and colleagues suggested that NTI is an explanation for thyroid dysfunction in COVID-19 patients [44]. Additionally, Chen and colleagues examined 50 patients and detected that there is a direct relation between the degree of TSH and TT3 reduced levels and the severity of the disease [45]. Recently, a review of reviews also declared that the most frequent thyroid dysfunction found in COVID-19 patients is non-thyroidal illness syndrome [36].
5. Conclusions
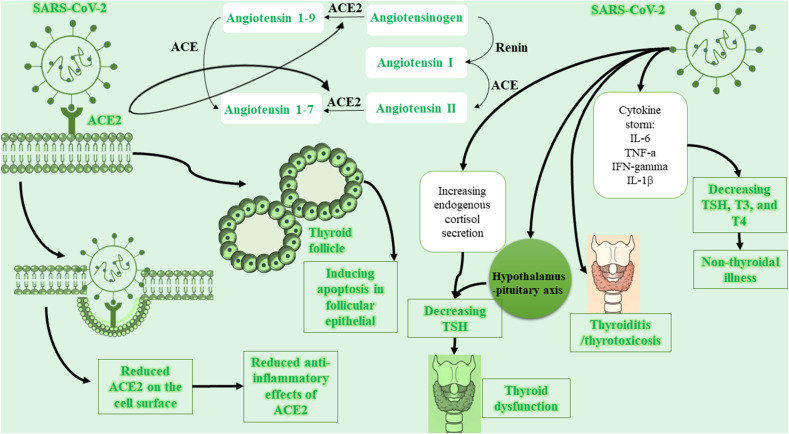
What we have observed while considering SARS-associated studies is that there is no reliable evidence on the neither higher risk of infection due to an underlying thyroid disease nor post-SARS thyroid dysfunction. Trials conducted in this area are commonly done on a limited number of patients and therefore, more investigations are required for confirming their results. Moreover, a majority of studies are using autopsies and more evidence should be established on patients with early stages of the disease. Between the papers we found, SARS-CoV-related evidence have higher number than 2019-nCoV-related evidence. However, due to the receptor which both of these viruses are sharing [46], SARS-related results might be extensible to SARS-CoV-2. We hypothesize that if coronaviruses actually cause thyroid dysfunction, four mechanisms can explain it (represented in Fig. 1 ): first coronavirus might affect the cells of thyroid gland directly and cause thyroiditis and/or apoptosis, second, coronavirus might affect hypothalamus-pituitary-thyroid axis through inducing hypophysitis, third, the cytokine storm initiated during COVID-19 can cause NTI, and fourth, reducing the number of ACE2 on the surface of thyroid cells might be disadvantageous (Fig. 1). Still, these hypotheses need more investigations to be confirmed. Furthermore, the effects of immunotherapy and anti-coagulant drugs used for COVID-19 patients, increased cortisol secretion, and sick euthyroid syndrome [2] should be taken into consideration, as well.
Fig. 1.
Cellular and molecular mechanisms by which SARS-CoV-2 affects thyroid function. ACE, Angiotensin converting enzyme; TSH, Thyroid-stimulating hormone.
Finally, thyroid dysfunctions caused by SARS-CoVs are dynamically changing in conjunction with the course of disease and seem to recover spontaneously and therefore, therapeutic measurements in early stages might not be needed. In advanced patients, on the other hand, thyroid dysfunction is transient and after COVID-19 treatment, TSH levels get back to normal and thus it seems that treatment might be necessary only in patients with specific indicators of a serious thyroid disease.
Availability of data and material
Not applicable.
Author contributions
PMD and ZA contributed in conception, design and drafting of the manuscript.
FS, JH, MAM, BY and MMH contributed in reviewing relevant literature.
All authors approved the final version for submission.
Consent for publication
Not applicable.
Declaration of competing interest
The authors declare no conflict of interest.
Acknowledgements
Not applicable.
References
- 1.Baud D., Qi X., Nielsen-Saines K., Musso D., Pomar L., Favre G. Real estimates of mortality following COVID-19 infection. The Lancet Infect Dis. 2020;20(7):773. doi: 10.1016/S1473-3099(20)30195-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pal R., Banerjee M. COVID-19 and the endocrine system: exploring the unexplored. J Endocrinol Invest. 2020:1. doi: 10.1007/s40618-020-01276-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Clerkin K.J., Fried J.A., Raikhelkar J., Sayer G., Griffin J.M., Masoumi A., et al. COVID-19 and cardiovascular disease. Circulation. 2020;141(20):1648–1655. doi: 10.1161/CIRCULATIONAHA.120.046941. [DOI] [PubMed] [Google Scholar]
- 4.Fang L., Karakiulakis G., Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med. 2020;8(4):e21. doi: 10.1016/S2213-2600(20)30116-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lyons F.M., Meeran K. The physiology of the endocrine system. Int Anesthesiol Clin. 1997;35(4):1–21. doi: 10.1097/00004311-199703540-00002. [DOI] [PubMed] [Google Scholar]
- 6.Li F. Receptor recognition and cross-species infections of SARS coronavirus. Antivir Res. 2013;100(1):246–254. doi: 10.1016/j.antiviral.2013.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Perlman S., Netland J. Coronaviruses post-SARS: update on replication and pathogenesis. Nat Rev Microbiol. 2009;7(6):439–450. doi: 10.1038/nrmicro2147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Perlot T., Penninger J.M. ACE2 - from the renin-angiotensin system to gut microbiota and malnutrition. Microb Infect. 2013;15(13):866–873. doi: 10.1016/j.micinf.2013.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Donoghue M., Hsieh F., Baronas E., Godbout K., Gosselin M., Stagliano N., et al. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1-9. Circ Res. 2000;87(5):E1–E9. doi: 10.1161/01.res.87.5.e1. [DOI] [PubMed] [Google Scholar]
- 10.Santos R.A., Ferreira A.J., Verano-Braga T., Bader M. Angiotensin-converting enzyme 2, angiotensin-(1-7) and Mas: new players of the renin-angiotensin system. J Endocrinol. 2013;216(2):R1–R7. doi: 10.1530/JOE-12-0341. [DOI] [PubMed] [Google Scholar]
- 11.Hamming I., Cooper M.E., Haagmans B.L., Hooper N.M., Korstanje R., Osterhaus A.D., et al. The emerging role of ACE2 in physiology and disease. J Pathol. 2007;212(1):1–11. doi: 10.1002/path.2162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lambert D.W., Clarke N.E., Turner A.J. Not just angiotensinases: new roles for the angiotensin-converting enzymes. Cell Mol Life Sci : CM. 2010;67(1):89–98. doi: 10.1007/s00018-009-0152-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mendoza-Torres E., Oyarzún A., Mondaca-Ruff D., Azocar A., Castro P.F., Jalil J.E., et al. ACE2 and vasoactive peptides: novel players in cardiovascular/renal remodeling and hypertension. Therapeut Adv Cardiovascular Dis. 2015;9(4):217–237. doi: 10.1177/1753944715597623. [DOI] [PubMed] [Google Scholar]
- 14.Crackower M.A., Sarao R., Oudit G.Y., Yagil C., Kozieradzki I., Scanga S.E., et al. Angiotensin-converting enzyme 2 is an essential regulator of heart function. Nature. 2002;417(6891):822–828. doi: 10.1038/nature00786. [DOI] [PubMed] [Google Scholar]
- 15.Anguiano L., Riera M., Pascual J., Soler M.J. Circulating ACE2 in cardiovascular and kidney diseases. Curr Med Chem. 2017;24(30):3231–3241. doi: 10.2174/0929867324666170414162841. [DOI] [PubMed] [Google Scholar]
- 16.Santos R.A., Ferreira A.J., Nadu A.P., Braga A.N., de Almeida A.P., Campagnole-Santos M.J., et al. Expression of an angiotensin-(1-7)-producing fusion protein produces cardioprotective effects in rats. Physiol Genom. 2004;17(3):292–299. doi: 10.1152/physiolgenomics.00227.2003. [DOI] [PubMed] [Google Scholar]
- 17.Yamamoto K., Ohishi M., Katsuya T., Ito N., Ikushima M., Kaibe M., et al. Deletion of angiotensin-converting enzyme 2 accelerates pressure overload-induced cardiac dysfunction by increasing local angiotensin II. Hypertension. 2006;47(4):718–726. doi: 10.1161/01.HYP.0000205833.89478.5b. (Dallas, Tex : 1979) [DOI] [PubMed] [Google Scholar]
- 18.Donoghue M., Wakimoto H., Maguire C.T., Acton S., Hales P., Stagliano N., et al. Heart block, ventricular tachycardia, and sudden death in ACE2 transgenic mice with downregulated connexins. J Mol Cell Cardiol. 2003;35(9):1043–1053. doi: 10.1016/s0022-2828(03)00177-9. [DOI] [PubMed] [Google Scholar]
- 19.Masson R., Nicklin S.A., Craig M.A., McBride M., Gilday K., Gregorevic P., et al. Onset of experimental severe cardiac fibrosis is mediated by overexpression of Angiotensin-converting enzyme 2. Hypertension. 2009;53(4):694–700. doi: 10.1161/HYPERTENSIONAHA.108.122333. (Dallas, Tex : 1979) [DOI] [PubMed] [Google Scholar]
- 20.Oudit G.Y., Herzenberg A.M., Kassiri Z., Wong D., Reich H., Khokha R., et al. Loss of angiotensin-converting enzyme-2 leads to the late development of angiotensin II-dependent glomerulosclerosis. Am J Pathol. 2006;168(6):1808–1820. doi: 10.2353/ajpath.2006.051091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ferreira A.J., Shenoy V., Yamazato Y., Sriramula S., Francis J., Yuan L., et al. Evidence for angiotensin-converting enzyme 2 as a therapeutic target for the prevention of pulmonary hypertension. Am J Respir Crit Care Med. 2009;179(11):1048–1054. doi: 10.1164/rccm.200811-1678OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Li X., Molina-Molina M., Abdul-Hafez A., Uhal V., Xaubet A., Uhal B.D. Angiotensin converting enzyme-2 is protective but downregulated in human and experimental lung fibrosis. Am J Physiol Lung Cell Mol Physiol. 2008;295(1):L178–L185. doi: 10.1152/ajplung.00009.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ferrario C.M., Averill D.B., Brosnihan K.B., Chappell M.C., Iskandar S.S., Dean R.H., et al. Vasopeptidase inhibition and Ang-(1-7) in the spontaneously hypertensive rat. Kidney Int. 2002;62(4):1349–1357. doi: 10.1111/j.1523-1755.2002.kid559.x. [DOI] [PubMed] [Google Scholar]
- 24.Burrell L.M., Risvanis J., Kubota E., Dean R.G., MacDonald P.S., Lu S., et al. Myocardial infarction increases ACE2 expression in rat and humans. Eur Heart J. 2005;26(4):369–375. doi: 10.1093/eurheartj/ehi114. discussion 22-4. [DOI] [PubMed] [Google Scholar]
- 25.Ye M., Wysocki J., Naaz P., Salabat M.R., LaPointe M.S., Batlle D. Increased ACE 2 and decreased ACE protein in renal tubules from diabetic mice: a renoprotective combination? Hypertension. 2004;43(5):1120–1125. doi: 10.1161/01.HYP.0000126192.27644.76. (Dallas, Tex : 1979) [DOI] [PubMed] [Google Scholar]
- 26.Lely A.T., Hamming I., van Goor H., Navis G.J. Renal ACE2 expression in human kidney disease. J Pathol. 2004;204(5):587–593. doi: 10.1002/path.1670. [DOI] [PubMed] [Google Scholar]
- 27.Narayan S.S., Lorenz K., Ukkat J., Hoang-Vu C., Trojanowicz B. Angiotensin converting enzymes ACE and ACE2 in thyroid cancer progression. Neoplasma. 2020;67(2):402–409. doi: 10.4149/neo_2019_190506N405. [DOI] [PubMed] [Google Scholar]
- 28.Li M.-Y., Li L., Zhang Y., Wang X.-S. Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues. Infectious Dis Poverty. 2020;9:1–7. doi: 10.1186/s40249-020-00662-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rotondi M., Coperchini F., Ricci G., Denegri M., Croce L., Ngnitejeu S., et al. Detection of SARS-COV-2 receptor ACE-2 mRNA in thyroid cells: a clue for COVID-19-related subacute thyroiditis. J Endocrinol Invest. 2021;44(5):1085–1090. doi: 10.1007/s40618-020-01436-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Coperchini F., Ricci G., Croce L., Denegri M., Ruggiero R., Villani L., et al. Modulation of ACE-2 mRNA by inflammatory cytokines in human thyroid cells: a pilot study. Endocrine. 2021;74(3):638–645. doi: 10.1007/s12020-021-02807-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Patel S., Rauf A., Khan H., Abu-Izneid T. Renin-angiotensin-aldosterone (RAAS): the ubiquitous system for homeostasis and pathologies. Biomed & Pharmacotherapy = Biomedecine & Pharmacotherapie. 2017;94:317–325. doi: 10.1016/j.biopha.2017.07.091. [DOI] [PubMed] [Google Scholar]
- 32.Garg S. vol. 69. 2020. (Hospitalization rates and characteristics of patients hospitalized with laboratory-confirmed coronavirus disease 2019—COVID-NET, 14 States, March 1–30, 2020. MMWR Morbidity and mortality weekly report). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pal R., Bhansali A. COVID-19, diabetes mellitus and ACE2: the conundrum. Diabetes Res Clin Pract. 2020:162. doi: 10.1016/j.diabres.2020.108132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Covid C., Covid C., Covid C., Chow N., Fleming-Dutra K., Gierke R., et al. Preliminary estimates of the prevalence of selected underlying health conditions among patients with coronavirus disease 2019—United States, February 12–March 28, 2020. MMWR (Morb Mortal Wkly Rep) 2020;69(13):382. doi: 10.15585/mmwr.mm6913e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dworakowska D., Grossman A.B. Thyroid disease in the time of COVID-19. Endocrine. 2020:1–4. doi: 10.1007/s12020-020-02364-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Trimboli P., Camponovo C., Scappaticcio L., Bellastella G., Piccardo A., Rotondi M. Thyroid sequelae of COVID-19: a systematic review of reviews. Rev Endocr Metab Disord. 2021;22(2):485–491. doi: 10.1007/s11154-021-09653-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wei L., Sun S., Xu C-h, Zhang J., Xu Y., Zhu H., et al. Pathology of the thyroid in severe acute respiratory syndrome. Hum Pathol. 2007;38(1):95–102. doi: 10.1016/j.humpath.2006.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Leow M.K.S., Kwek D.S.K., Ng A.W.K., Ong K.C., Kaw G.J.L., Lee L.S.U. Hypocortisolism in survivors of severe acute respiratory syndrome (SARS) Clin Endocrinol. 2005;63(2):197–202. doi: 10.1111/j.1365-2265.2005.02325.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ding Y., He L., Zhang Q., Huang Z., Che X., Hou J., et al. Organ distribution of severe acute respiratory syndrome (SARS) associated coronavirus (SARS-CoV) in SARS patients: implications for pathogenesis and virus transmission pathways. J Pathol: A J Pathol Soc Great Britain and Ireland. 2004;203(2):622–630. doi: 10.1002/path.1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yao X., Li T., He Z., Ping Y., Liu H., Yu S., et al. A pathological report of three COVID-19 cases by minimally invasive autopsies. Zhonghua bing li xue za zhi= Chinese J Pathology. 2020;49:E009. doi: 10.3760/cma.j.cn112151-20200312-00193. [E] [DOI] [PubMed] [Google Scholar]
- 41.Khoo B., Tan T., Clarke S.A., Mills E.G., Patel B., Modi M., et al. Thyroid function before, during, and after COVID-19. J Clin Endocrinol Metabol. 2021;106(2):e803–e811. doi: 10.1210/clinem/dgaa830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lania A., Sandri M.T., Cellini M., Mirani M., Lavezzi E., Mazziotti G. Thyrotoxicosis in patients with COVID-19: the THYRCOV study. Eur J Endocrinol. 2020;183(4):381–387. doi: 10.1530/EJE-20-0335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wang K., Chen Z., Shi J., Feng Y., Yu M., Sun Y., et al. Resveratrol inhibits the tumor migration and invasion by upregulating TET1 and reducing TIMP2/3 methylation in prostate carcinoma cells. Prostate. 2020;80(12):977–985. doi: 10.1002/pros.24029. [DOI] [PubMed] [Google Scholar]
- 44.Croce L., Gangemi D., Ancona G., Liboà F., Bendotti G., Minelli L., et al. The cytokine storm and thyroid hormone changes in COVID-19. J Endocrinol Invest. 2021;44(5):891–904. doi: 10.1007/s40618-021-01506-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chen M., Zhou W., Xu W. Thyroid function analysis in 50 patients with COVID-19: a retrospective study. Thyroid. 2021;31(1):8–11. doi: 10.1089/thy.2020.0363. [DOI] [PubMed] [Google Scholar]
- 46.Li W., Moore M.J., Vasilieva N., Sui J., Wong S.K., Berne M.A., et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426(6965):450–454. doi: 10.1038/nature02145. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.