Abstract
Heme oxygenase 1 (HO-1), the rate-limiting enzyme in heme degradation, is involved in the maintenance of cellular homeostasis, exerting a cytoprotective role by its antioxidative and anti-inflammatory functions. HO-1 and its end products, biliverdin, carbon monoxide and free iron (Fe2+), confer cytoprotection against inflammatory and oxidative injury. Additionally, HO-1 exerts antiviral properties against a diverse range of viral infections by interfering with replication or activating the interferon (IFN) pathway. Severe cases of coronavirus disease 2019 (COVID-19), an infectious disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), are characterized by systemic hyperinflammation, which, in some cases, leads to severe or fatal symptoms as a consequence of respiratory failure, lung and heart damage, kidney failure, and nervous system complications. This review summarizes the current research on the protective role of HO-1 in inflammatory diseases and against a wide range of viral infections, positioning HO-1 as an attractive target to ameliorate clinical manifestations during COVID-19.
Keywords: heme oxygenase 1, COVID-19, influenza A virus, respiratory syncytial virus, human immunodeficiency virus, Ebola virus, Dengue virus, Zika virus, Hepatitis virus, SARS-CoV-2
1. Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) emerged in late 2019 in Wuhan, China. The World Health Organization (WHO) declared coronavirus disease 2019 (COVID-19) a pandemic health emergency as of 31 January 2020. The treatment goal in COVID-19 patients is to prevent or to decrease the strong virus induced inflammatory stimuli associated with a wide spectrum of poor prognosis clinical manifestations [1]. Heme oxygenase 1 (HO-1) is a microsomal enzyme with a primary antioxidant and anti-inflammatory role involved in heme degradation, generating carbon monoxide (CO), biliverdin (BV), and free iron (Fe2+) [2]. Hence, HO-1 induction is a useful approach for inflammatory diseases treatment [3,4,5,6]. Additionally, HO-1 displays antiviral properties against a wide range of viruses [7]. Hemin, a previously Food and Drug Administration (FDA) and European Medicines Agency (EMA) approved drug for acute intermittent porphyria treatment [8,9], is a well known inducer of HO-1 that increases its plasma concentration in humans. Thus, hemin rises as a promising drug candidate against the replication of different viruses, including SARS-CoV-2. In this review, we summarize the current research on the protective role of HO-1 in inflammatory diseases and in a diverse range of viral infections, positioning this protein as a potential therapeutic target to ameliorate COVID-19′s clinical manifestations.
2. Re-Emergence of the Coronavirus Disease
Coronaviruses (CoVs) are a large family of positive sense, single stranded RNA (+ssRNA) viruses that infect humans, other mammals, and birds, causing respiratory, enteric, hepatic, and neurologic diseases [10,11]. CoVs first became renowned in 2002–2003 during an outbreak of a virus with zoonotic origin; the severe acute respiratory syndrome coronavirus (SARS-CoV) originated in China, with 8096 cases and 774 deaths reported between 2002 and 2003 [12], and a case–fatality ratio of 7.2% [13]. In 2012, the Middle East respiratory syndrome coronavirus (MERS-CoV), another virus with zoonotic origin, emerged in Saudi Arabia and caused 927 fatalities among 2581 registered cases [14,15].
By the end of 2019, the Wuhan Health Commission from China reported a number of pneumonia cases of unknown cause and varying severity in the city of Wuhan, China. High throughput sequencing allowed the quick identification of a novel CoV belonging to the beta-coronavirus family, which was named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), as the causative agent of Coronavirus disease 2019 (COVID-19) [16]. This pathogen rapidly spread globally via travel related cases; constituting a pandemic and setting an immense challenge for public health [16]. Unlike SARS-CoV and MERS-CoV, SARS-CoV-2 can be transmitted among people before the onset of symptoms or from asymptomatic individuals [17], limiting effective control of the spread. As of 30 December 2021, the WHO reports 281,808,270 confirmed cases and 5,411,759 deaths worldwide [15].
Although COVID-19 is primarily a respiratory disease, SARS-CoV-2 has the capacity to infect a broad range of cell types in different organs and systems, including the central nervous system [18]. SARS-CoV-2 infection begins in the proximal airways and could trigger severe and sometimes fatal symptoms when reaching the distal lung [19]. Among the severe respiratory diseases caused by SARS-CoV-2, acute respiratory distress syndrome (ARDS) and acute lung injury (ALI) [20,21] are complications mainly caused by an exacerbated immune response in elders and patients with comorbidities [22]. Cytokine storm contributes to ALI and the development of ARDS in patients with severe pneumonia caused by SARS-CoV-2, as well as SARS-CoV and MERS-CoV [23,24]. Surprisingly, in addition to the respiratory symptoms, patients may suffer from cardiac, hematological, neurological, hepatic, gastrointestinal and kidney complications [25]. COVID-19 may result in long term sequelae characterized by organ injuries that cannot be completely reversed. Several patients, even those with mild cases, may develop lasting symptoms that can have disabling consequences [26,27].
The COVID-19 era is far from being constrained, and the emergence of new viral variants causing future outbreaks remains a threat. As of December 2021, the WHO has defined five variants of concern (Alpha (B.1.1.7), Beta (B.1.351), Gamma (P.1), Delta (B.1.617.2), and Omicron (B.1.1.529)), as well as two variants of interest (Lambda (C.37) and Mu (B.1.621)) [15]. It has been reported that some variants, such as the recently described Omicron [28], have higher transmission rates and are able to escape neutralizing antibodies generated by natural infection and vaccination, or therapeutic antibodies [29,30,31,32]. Incomplete knowledge on the pathogenesis of SARS-CoV-2 and the diverse array of symptoms and manifestations of COVID-19 pose a great challenge for the development of effective treatments that should mainly focus on both, decreasing viral replication and modulating the immune response.
3. Cytokine Storm and Inflammation
Inflammation involves defense mechanisms against infection or injury. It is responsible for activating both innate and adaptive immune responses [33,34]. During infections, innate cells recognize pathogen associated molecular patterns (PAMPs) from the invading agent. In the case of inflammation triggered by tissue damage, trauma or ischemia, innate cells recognize host specific molecules that are released during cell injury or necrotic death, defined as damage associated molecular patterns, such as nucleic acids and adenosine triphosphate (ATP) [33]. During the early stages of inflammation, innate immune cells and endothelial cells (EC) release a diverse set of cytokines: chemotactic cytokines, such as monocyte chemotactic protein-3 (MCP-3) and interferon (IFN) γ-induced protein 10 (IP-10), and recruit other immune cells to the site of infection or inflammation. Proinflammatory cytokines, such as tumor necrosis factor alpha (TNF-α), interleukin-6 (IL-6), and IL-1β [35,36], are also released and trigger the activation of inflammatory pathways, including the mitogen activated protein kinase (MAPK), nuclear factor kappa-B (NF-κB), and Janus kinase (JAK)-signal transducer and activator of transcription (STAT) pathways [34]. Some pathogenic viruses (i.e., highly virulent subtypes of influenza) and bacteria (i.e., Francisella tularensis) can induce the acute dysregulated production of inflammatory cytokines, known as “cytokine storm” or hypercytokinemia [37]. The hypercytokinemia and exacerbated secondary events, such as coagulation, eventually result in widespread necrosis, organ failure and death [33,38].
Once SARS-CoV-2 infects target cells, innate immune cells are recruited to the infection site, where they release cytokines and initiate the activation cascade of adaptive B and T cell immune responses [39]. In most cases, the immune system is able to eliminate virus infected cells and resolve the immune response. However, in some patients, this process is dysfunctional, impairing the effective clearance of infected cells, and causing severe damage to the host [40].
4. The Lead Role of Interferons upon Viral Infection
During viral infections, pattern recognition receptors are stimulated to produce IFN by the innate immune cells. IFNs are crucial for the induction of an antiviral state via autocrine and paracrine signaling. There are three types of IFNs: type I, type II and type III. Type I (IFNα, IFNβ, IFNω, IFNτ, IFNε) and Type III (IFNλ1, IFNλ2/3, IFNλ4) share similar dynamics after binding to its receptor, as cross-phosphorylation between JAK1 and tyrosine kinase 2 (TYK2) occurs [41]. Subsequently, a docking site for STAT1 is exposed, STAT1 is phosphorylated, translocates to the nucleus, and induces the transcription of interferon stimulated genes (ISGs). The IFNs biological effects are wide, including immuno-regulation, antiviral, anti-angiogenic, and pro-apoptotic functions [42]. However, many pathogens have evolved to elude the action mechanisms of these powerful cytokines [43,44,45].
In critically ill COVID-19 patients, a hyperinflammation state prevails. In March 2020, a retrospective study on 150 patients from Wuhan, China, found elevated levels of IL-6 and C-reactive protein (CRP) in SARS-CoV-2 infected patients that died compared with discharged patients [46]. An independent report based on 50 COVID-19 patients with moderate to severe disease, identified IP-10, MCP-3, and IL-1 receptor antagonist (IL-1ra) as independent predictors for disease severity [47]. A longitudinal analysis showcased IL-18 and IFN-α as top biomarkers for predicting mortality. Consequently, higher counts of inflammatory monocytes, plasmablast like neutrophils and eosinophils have been described in patients with severe disease [39].
Blanco-Melo et al. reported an impairment in the response of type I and type III IFNs against SARS-CoV-2 infection [40]. In contrast, a recent study found that severe COVID-19 cases showed an exacerbated expression of type I IFNs, which could lead to augmented inflammation [48]. Several clinical trials evaluating IFNs have been carried out in COVID-19 patients. Two different studies showed a reduction in the mortality rate after IFNβ-1a and IFNβ-1b treatment [49,50]. Another study, using IFNα-2b, reported a decrease in detectable SARS-CoV-2 in the upper respiratory tract associated with lower inflammatory cytokines levels, such as IL-6 and CRP [51]. In addition, peginterferon λ treatment was associated with a reduction in viral RNA [52]. Furthermore, there are several ongoing clinical trials, using recombinant human IFNs or IFNs combined with other drugs [53,54,55]. These evidences highlight IFNs as potential targets for COVID-19 treatment. In the next section we will focus on the stimulation of IFN pathway by HO-1 induction.
5. Understanding the Protective Role of Heme Oxygenase 1
Heme oxygenases (HO) are metabolic enzymes that partake in the degradation of the heme group [2]. To date, three isoforms of this protein have been found: HO-1, which can be induced by external factors (such as hypoxia, oxidative stress, heat shock, reactive oxygen species (ROS), among others) [56]; HO-2, a constitutively expressed isoform; and HO-3, a nonfunctional isoform in humans [57].
In particular, HO-1, encoded by the HMOX1 gene, is involved in the maintenance of cellular homeostasis, exerting a cytoprotective role by its anti-inflammatory, anti-oxidative and anti-apoptotic functions, as revealed in a human case of genetic HO-1 deficiency [58]. This enzyme participates not only in normal physiological processes, but also performs a protective role in inflammatory physiopathological conditions, such as kidney disease [59], cancer [60,61], cardiovascular disease [62], asthma [63] and inflammatory bowel diseases [4,64].
HO-1 is expressed in most cell types and tissues; however, its capacity to counteract inflammation seems to be critically dependent on its specific functions in myeloid cells and in EC [65]. In myeloid cells, HO-1 acts as a key regulator of the TLR4/TLR3/IRF3 induced production of IFN-β and primary IRF3 target genes in macrophages [66] and modulates maturation and specific functions of dendritic cells [67,68]. Moreover, HO-1 over-expression in macrophages negatively regulates the expression of diverse proinflammatory molecules and increases the expression of anti-inflammatory cytokines [69,70,71]. Among HO-1 effects on EC, it is significant to mention its ability to inhibit the expression of pro-inflammatory genes related to EC activation, such as the TNF-α-induced adhesion molecules, E-selectin and VCAM-1, via a mechanism associated with the inhibition of NF-κB activation [72].
HO-1 cleaves the heme group generating BV, CO and Fe2+. Heme is usually bound to a myriad of proteins and it is involved in several homeostatic functions [56]. However, elevated concentrations of heme can cause cell damage because it is a pro-oxidant molecule. It can diffuse through cell membranes and deliver a redox active iron, producing ROS [73]. Excessive amounts of these molecules are toxic and induce oxidative stress that, in turn, generates DNA and protein damage, aggregation and lipid peroxidation, triggering cells permeability and driving cell lysis and death [73].
Several studies highlight heme catabolism end products as potential therapeutic targets in vascular disease, based on their anti-inflammatory and antiproliferative functions [74]. BV and its reduced form, bilirubin (BR), are powerful antioxidants that are able to scavenge ROS and counteract the oxidative stress. BV and BR are critical for the regulation of inflammation by exerting immunosuppressive effects [75], as they have been reported to have potent anti-inflammatory activity against insulin resistance by reducing visceral obesity and adipose tissue inflammation [76].
In addition, CO is considered an anti-apoptotic [77], antiproliferative and anti-inflammatory factor [78]. CO contributes to blood vessel development [79] and promotes angiogenesis, a crucial process involved in tissue reparation after a pathological state [80]. Interestingly, CO also reduces inflammation and inhibits apoptosis by interacting with antigen-presenting cells and suppressing T cell proliferation [81]. Moreover, it has been reported that it downregulates proinflammatory cytokines via the p38/MAPK pathway in RAW 264.7 macrophages and C57BL/6 mice [70], by the c-Jun N-terminal kinase (JNK) pathway in a murine model of sepsis [82] and through the extracellular signal regulated kinase (ERK) signaling pathway in CD4+ T cells [81]. Further, HO-1/CO induced downregulation of the NLRP3 (NOD-, LRR- and pyrin domain-containing protein 3) inflammasome activation has been demonstrated in different models of murine hepatic and lung inflammatory injury [83,84,85].
Moreover, the HO-1 mediated increase in Fe2+ concentration upregulates the expression of ferritin, an iron chelating protein [86]. Ferritin exerts antioxidative and cytoprotective effects [74], as this product scavenges redox active Fe2+, rendering it not harmful for cells and avoiding subsequent production of ROS via Fenton reaction. Fe2+ performs its function by inhibiting IL-2 and IgG production, and downregulating the MAPK and NF-κB signaling pathways [56,75,87].
Therapeutic Potential of HO-1 Induction to Treat Chronic Inflammation
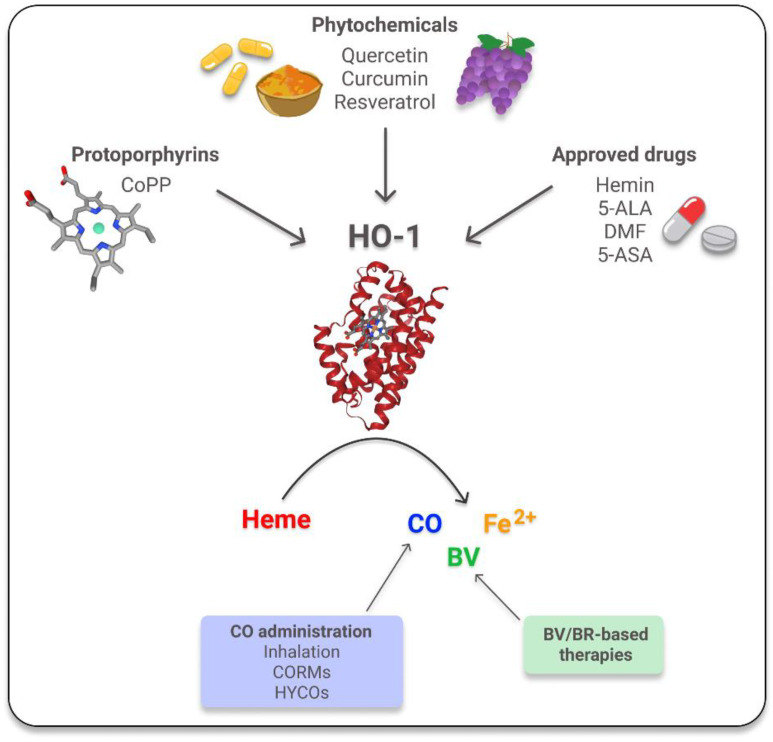
As HO-1 and its reaction products exert protective anti-inflammatory effects in different preclinical models [3,4,5,88,89,90], the induction of the HO-1 system has emerged as a promising potential therapy for chronic inflammatory diseases. Most of the studied strategies are based on the use of the traditional pharmacological inducers: hemin [91,92], an FDA and EMA approved drug, and cobalt protoporphyrin IX (CoPP) [5,93,94]. In addition, many phytochemicals, such as quercetin, curcumin and resveratrol, are currently under investigation, due to their potential as HO-1 alternative inducers to counteract inflammation processes with lower cytotoxic secondary effects [95,96,97]. Alternatively, there are also a few approved drugs, such as 5-aminosalicylic acid (5-ASA), dimethyl fumarate (DMF), and 5-aminolevulinic acid (5-ALA), whose beneficial properties in inflammatory conditions are explained, at least in part, by their capacity to induce HO-1 [96,98,99] (Figure 1). Additionally, another effective option is the use of BV/BR based therapies, which have proven to be effective for these chronic pathologies [100,101,102] and/or the direct administration of CO via inhalation, CO-releasing molecules (CORMs) or hybrid carbon monoxide-releasing molecules (HYCOs). HYCOs are a type of compound where CORMs are combined with DMF, causing a powerful anti-inflammatory action due to its effect on the NRF2/HO-1 pathway [5,100,103,104,105,106] (Figure 1).
Figure 1.
Inducers of HO-1. HO-1 degrades heme producing equimolar amounts of carbon monoxide (CO), biliverdin (BV) and Fe2+. HO-1′s inducers are grouped into protoporphyrins, a type of porphyrins that forms heme; phytochemicals, natural antioxidants compounds contained in plants; and approved drugs, compounds that were previously approved by the FDA. CoPP: cobalt protoporphyrin IX; 5-ALA: 5-aminolevulinic acid; DMF: dimethyl fumarate; 5-ASA: 5-aminosalicylic acid; CORMs: CO-releasing molecules; HYCOs: Hybrid carbon monoxide-releasing molecules; BR: bilirubin. The images of HO-1 and CoPP were taken from RCSB PDB (PDB ID: 1N3U) and The National Center for Biotechnology Information [107,108].
6. HO-1 Mechanism of Action against Inflammatory Lung Diseases
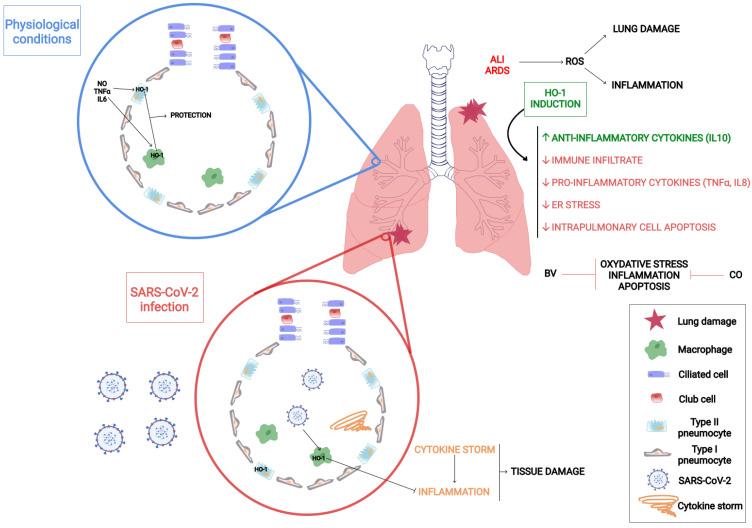
There is extensive literature about the role of HO-1 in lung diseases. This protein is expressed in type II pneumocytes and in alveolar macrophages and contributes to the protection of the lung tissue. The main HO-1 inducers in the lungs are pro-inflammatory cytokines, such as TNF-α and IL-6, the heme group and nitric oxide (NO), as well as hypoxia and hyperoxia conditions [109] (Figure 2). There is sound evidence that states that HO-1 induction is a critical defense factor during acute and chronic lung processes [109,110,111].
Figure 2.
HO-1 and inflammatory lung diseases. HO-1 is expressed in pulmonary cells and confers protection against inflammatory lung diseases such as acute respiratory distress syndrome (ARDS), acute lung injury (ALI), and SARS-CoV-2 infection. Schematic representation displaying HO-1′s reaction and its products’ protective effects in the lung tissue.
As mentioned earlier, during COVID-19 disease, the number of immune cells infiltrating lung tissues and the pro-inflammatory cytokines levels are augmented [112]. Consequently, anti-inflammatory proteins have a crucial role in halting the cytokine storm and the sequelae generated by viral infection [113].
In particular, ALI and ARDS are the most prevalent diseases emerging from an extended diversity of lung injuries [114,115]. ALI and ARDS are pathophysiologically characterized by lung damage, inflammatory infiltration, and an exacerbation of the host immune response [116]. Several reports indicate that ALI and ARDS might be explained by the presence of high ROS levels, where HO-1 acts as a protective factor against oxidative stress under pharmacological induction [117]. HO-1 induction by hemin shows a protective role against ventilator induced lung injury in rabbits with ALI/ARDS, increasing anti-inflammatory cytokine levels, such as IL-10, as well as decreasing the inflammatory infiltrate of immune system cells and the secretion of inflammatory cytokines, such as TNF-α and IL-8 [118] (Figure 2). Furthermore, it has been found that HO-1 confers protection against ischemia-reperfusion injury (LIRI) [119].
HO-1 regulates diverse signaling pathways that are affected during pulmonary diseases. In rats, HO-1 inhibits the PERK/eIF2-α/ATF4/CHOP pathway, which is involved in the endoplasmic reticulum stress (ERS) characteristic in ALI, and also promotes the decrease in intrapulmonary cell apoptosis [120]. It was also reported that the PI3K/Akt pathway attenuates oxidative damage during ALI/ARDS through HO-1 regulation [121]. In pathologies such as silicosis, characterized by excessive ROS production, lung injury is attenuated by HO-1 induction. The mechanism underlying this cytoprotective effect relies on the ERK pathway inhibition by HO-1, CO and BV [122].
Reaction products derived from the HO-1 mediated heme catalysis have protective roles in lung pathologies as well. CO is known to provide protection against ALI and ARDS by reducing cytokine and chemokine levels [105,117,123]. CO decreases EGR-1 (early growth response protein 1), a proinflammatory protein that regulates the expression of TNF-α and IL-2 [124], in mice lungs [123]. Fujita et al. demonstrated that Hmox1 deficient mice had increased mortality after lung ischemia, an effect reverted by CO administration [125]. Furthermore, BV exerted antioxidative, anti-inflammatory and anti-apoptotic effects in a rat model of LIRI [126]. BV administration protected against hemorrhagic shock induced ALI through a decrease in the inflammatory infiltrate and proinflammatory cytokines levels [127].
7. Unveiling How HO-1 Promotes Viral Clearance
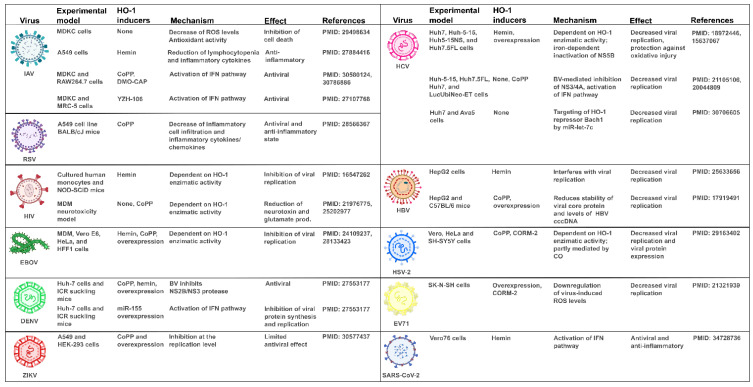
HO-1 has immunomodulatory properties on the innate immune response and there is compelling evidence suggesting that it also plays a central role in the modulation of adaptive immunity. HO-1 displays antiviral properties against a wide range of viruses [7] (Figure 3). Several reports have demonstrated that HO-1 induction is associated with the activation of the IFN pathway. However, the mechanism underlying the antiviral properties of HO-1 exerted by both its classical and noncanonical activities are yet to be fully elucidated.
Figure 3.
HO-1′s induction and its effect on different viral infections. Table containing previously reported studies about HO-1 involvement in influenza A virus (IAV), respiratory syncytial virus (RSV), human immunodeficiency virus (HIV), ebola virus (EBOV), dengue virus (DENV), zika virus (ZIKV), hepatitis C virus (HCV), hepatitis B virus (HBV), herpes simplex virus 2 (HSV-2), enterovirus 71 (EV71) and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infections. The table includes the experimental model, HO-1 inducers, its mechanism of action, its effect and the study’s PMID. CoPP: cobalt protoporphyrin IX, DMO-CAP: 6-demethoxy-4′-O-methylcapillarisin, ROS: Reactive oxygen species, IFN: interferon, MDM: monocyte derived macrophages, BV: Biliverdin, CO: carbon monoxide, CORM-2: CO-releasing molecule-2.
7.1. Respiratory Viruses: IAV and RSV
Influenza A virus (IAV) is a single stranded RNA virus whose infection remains a persistent global health threat with high morbidity and mortality [128]. An estimate of 4.0 to 8.8 deaths per 100,000 individuals with seasonal influenza associated respiratory occur annually (all types of influenza virus) [129]. Considering that the inhibition of virus induced ROS formation impairs IAV replication, proteins such as HO-1 are useful to counteract IAV infections in the host cell [130]. Wang et al. studied the effect of hemin in IAV infections and demonstrated that hemin attenuates the lymphocytopenia caused by IAV infection both in vitro and in vivo [131]. These results suggest that the anti-influenza effect of hemin may be mediated by HO-1′s ability to regulate systemic and local inflammatory responses [131]. Furthermore, Hashiba et al. reported that HO-1 gene transfer is a potential strategy to treat lung injury caused by IAV [132] and Cummins et al. suggested that the therapeutic induction of HO-1 expression may represent a novel adjuvant to enhance influenza vaccine effectiveness [133]. It has also been reported that the HO-1 inducers rupestonic acid derivative YZH-106 and the flavonoid 6-demethoxy-4′-O-methylcapillarisin (DMO-CAP) inhibited IAV replication by activating the HO-1 mediated IFN response [134,135]. Additionally, Ma et al. evaluated the effect of CoPP in IAV infection, focusing on the IFN pathways. The authors demonstrated that HO-1 induction attenuates IAV replication, and the most intriguing finding was that the catalytic function of HO-1 was not essential for the anti-IAV effect of CoPP. Interestingly, they found that HO-1 interacts with IFN regulatory factor 3 (IRF3) promoting its phosphorylation and nuclear translocation, thus activating the IFN pathway. Consequently, CoPP treatment increased the expression of IFITM3, PKR and OAS1, three ISGs markedly involved in the anti-IAV response [128].
Respiratory syncytial virus (RSV) is an RNA virus of the Pneumoviridae family and the most common cause of lower respiratory tract infections in children worldwide [136]. It interacts with host cells’ toll-like receptors in the primary airway epithelium, and promotes the expression and secretion of inflammatory cytokines [137], under the NF-κB pathway’s regulation [138]. Similar to IAV, CoPP HO-1 induction inhibited RSV replication and viral particle production in lung adenocarcinoma (A549) and HEp-2 cells. Most importantly, in vivo assays in BALB/cJ mice treated prophylactically with CoPP also showed a reduction in viral replication and viral particle production, alongside a decrease in inflammatory cell infiltration, and inhibition of proinflammatory cytokine or chemokine secretion during RSV infection [139].
The mentioned reports suggest that HO-1 is involved in host cellular defense mechanisms against IAV and RSV infections. Of note, HO-1’s antiviral effects are mediated by its classical and noncanonical functions.
7.2. Retroviridae: HIV
The human immunodeficiency virus-1 (HIV-1) genome consists of two identical single stranded RNA molecules and it is the causative agent of acquired immune deficiency syndrome (AIDS). HIV infection has become a clinically manageable disease since the development of combination antiretroviral therapy (ART). Globally, 1 million people died from HIV/AIDS in 2016; without ART, more than twice as many people would have died from this disease [140]. In 2020, the United Nations Programme on HIV/AIDS (UNAIDS) reported that of all people with HIV worldwide, 66% were virally suppressed [141,142]. However, emerging drug resistance and limitations in access and adherence to ART impose a threat to controlling the spread of the virus [143]. Current drugs do not eradicate the virus, making lifelong treatment necessary [143].
Several reports from in vitro and in vivo models, and HIV infected subjects, have linked HO-1 with HIV replication and its effects on HIV mediated neurodegeneration. In cultured monocytes and HIV infected mice, hemin efficiently inhibited viral replication; the effect observed in vitro was mediated by HO-1 enzymatic activity [144]. Using an in vitro model of HIV mediated neurotoxicity, HIV infection dysregulated the macrophage antioxidant response and reduced levels of HO-1. Restoration of HO-1 expression in HIV-infected monocyte derived macrophages (MDM) reduced neurotoxin release without altering HIV replication [145]. In HIV infected subjects, HO-1 protein levels were reduced in the dorsolateral prefrontal cortex, which correlated with central nervous system (CNS) viral load markers of immune activation. In a model of human astrocytes treated with IFNγ, an HIV associated CNS immune activator, HO-1 was degraded by the immunoproteasome [146]. Additionally, the use of CoPP reduced HIV-MDM glutamate release and neurotoxicity, suggesting a role for HO-1 in HIV associated neurocognitive disorders pathogenesis [147].
Altogether, these reports propose HO-1 induction as a protective mechanism against HIV infection. In particular, the HO-1′s classical functions mediate its antiviral properties against HIV.
7.3. Filoviridae: EBOV
Ebola virus (EBOV) is a negative-sense RNA virus that belongs to the Filoviridae family [148,149]. From 2013 to 2015 there was an important outbreak in West Africa, which caused >25,000 infections and >10,000 deaths. The average EBOV case fatality rate is ~50% and case fatality rates has ranged from 25% to 90% in past outbreaks [15]. Hemin treatment significantly reduced EBOV replication and delayed pathogenesis in vitro, by stimulating the cellular innate response against the infection [150,151]. In MDM, hemin treatment inhibited EBOV infection in a dose dependent manner. A similar effect was observed in other cell lines, such as HeLa and human foreskin fibroblasts cells, in which hemin also reduced viral replication [150]. Furthermore, it has been reported that the ebola virus protein 35 (VP35) is a critical protein involved in the inhibition of IRF3 phosphorylation as a mechanism that might counteract the antiviral response [152]. Thus, considering that HO-1 promotes IRF3 phosphorylation and activation, its induction may represent a novel therapeutic strategy against EBOV infection.
The cited studies place HO-1 as a novel therapeutic target against EVOB infection. Notably, HO-1′s noncanonical functions are involved in the present example of antiviral action.
7.4. Hepatic Viruses: HCV and HBV
Hepatitis C virus (HCV), a single stranded positive sense RNA virus, is associated with chronic hepatitis, cirrhosis, steatosis and hepatocellular carcinoma [153]. HCV treatment includes a combination of pegylated IFN-α and ribavirin, which has low efficacy and important side effects. With the development of direct-acting antiviral agents, such as sofosbuvir and simeprevir, patient outcomes have greatly improved; however, the disease remains a concern [154]. Since HCV infection generates oxidative damage to hepatocytes, the modulation of HO-1 expression emerges as an attractive therapeutic approach to reduce chronic liver disease. Abdalla et al. observed lower HO-1 mRNA and protein levels in HCV infected patients’ livers, while this alteration was not found in patients with other chronic liver diseases. The authors also reported HO-1 downregulation in hepatocyte cell lines expressing the HCV core protein [155]. Further, the overexpression or hemin-induction of HO-1 in the hepatoma cell line Huh7 decreased HCV replication and conferred protection against oxidative injury [156]. This effect of HO-1 on HCV replication might be explained partly by the iron dependent inactivation of the HCV RNA polymerase NS5B [157], and by the BV mediated inhibition of HCV NS3/4A protease and induction of an antiviral response by IFNα2 and IFNα17 [153,158]. Moreover, overexpression of miR-let-7c, which interferes with the production of proinflammatory cytokines in osteoarthritis and rheumatoid arthritis synovial fibroblasts [159], can reduce HCV replication by targeting HO-1 transcriptional repressor Bach1 [160].
Hepatitis B virus (HBV) is a DNA virus that causes serious liver diseases, representing the most common etiological agent for these pathologies [161]. It has been shown that pharmacological and genetic HO-1 overexpression attenuates viral replication both in vivo and in vitro in HepG2 cells [161,162,163], while also playing a hepatoprotective role [162]. The effect of HO-1 induction using hemin and CoPP mitigated the effects of HBV replication [6,161,162]. On the other hand, blocking HO-1 by siRNA reversed the inhibition of viral replication [6]. Interestingly, Protzer et al. evaluated the effect of HO-1 on HBV core protein by pulse-chase metabolic labeling experiments finding that HO-1 can destabilize structural proteins to prevent the formation of viral capsids, highlighting a direct HO-1 antiviral mechanism rather than limiting its effect to its anti-inflammatory properties [6].
Hence, the summarized reports established the antiviral effects of HO-1 by impairing HCV’s and HBV’s replication.
7.5. Arbovirus: DENV and ZIKV
Dengue virus (DENV) is a single stranded positive sense RNA virus [164] that induces oxidative stress by the activation of inflammatory regulators, such as NF-κB, and leads to the progression and pathogenesis of DENV [165]. In this pro-oxidant scenario, Tseng et al. demonstrated that HO-1 promoter activity and protein synthesis gradually increased during the early stages of DENV infection (6 to 12 h), but they were markedly decreased at later stages (24 to 72 h) [166]. Strikingly, pharmacological and genetic HO-1 induction after infection impaired viral protein synthesis and replication, and reduced DENV mortality. This effect was due to BV but not CO nor Fe2+ production [166]. The authors demonstrated that BV inhibits NS2B/NS3 DENV protease, thus promoting the antiviral IFN response and impairing its blockage by this protease [166]. Accordingly, Su et al. showed the anti-DENV activity of miR-155, which inhibits Bach1, a protein that negatively regulates the expression of many oxidative stress-response genes, including HMOX1 [167]. This, in turn, results in the induction of HO-1, boosting the IFN responses against DENV replication by the activation of interferon induced protein kinase R (PKR), 2’-5’-oligoadenylate synthetase 1 (OAS1), OAS2, and OAS3 expression [167]. Interestingly, the summarized studies demonstrate that HO-1′s antiviral effects against DENV infection involve both, its canonical, in this case mediated by BV, and noncanonical functions.
Zika virus (ZIKV), a single stranded positive sense RNA, is the causative pathogen of Zika fever in humans [168]. Using A549 and embryonic kidney (HEK-293) cell lines, El Kalamouni et al. demonstrated that ZIKV infection downregulated HO-1 expression by triggering endoplasmic-reticulum-associated protein degradation, thus halting its antiviral effects [168]. This report highlights HO-1′s protective role relevance, as it demonstrates that ZIKV infection promotes the decrease in HO-1 expression levels as an evasion mechanism.
7.6. Neurotropic Viruses: HSV-2 and EV71
Herpes simplex virus (HSV) includes HSV-1 and HSV-2, two double stranded DNA viruses that belong to Herpesviridae family. HSV produces recurring lesions in skin and mucosae and can also latently infect neurons of the trigeminal or dorsal root ganglia. HSV infection can result in encephalitis and meningitis [169]. Ibañez et al. demonstrated that pharmacological induction of HO-1 by CoPP hampered HSV-2 propagation in epithelial and neuronal cells. Furthermore, by CORM-2 treatment the authors also showed that the effects of HO-1 were partly mediated by CO [170].
Enterovirus 71 (EV71) is a single stranded positive sense RNA virus that belongs to the Picornaviridae family and is the causative agent of hand foot and mouth disease in children [171]. It was demonstrated that the overexpression of HO-1, as well as CO treatment, decreased viral replication in SK-N-SH cells suggesting that the antiviral effect is mediated by the downregulation of EV71 induced ROS levels [172].
Regarding neurotropic viruses, the summarized reports showcase that HO-1 displays protective effects against HSV-2 and EV71 involving its enzymatic function.
7.7. COVID-19 Causative Agent: SARS-CoV-2
SARS-CoV-2, is the novel beta coronavirus of the Coronaviridae family whose genome is composed of a single stranded RNA molecule [173]. It has been shown that hemin, hemoglobin and protoporphyrin IX bind to SARS-CoV-2 proteins, blocking its adsorption and replication independently from HO-1 induction [174]. However, the current literature regarding HO-1′s antiviral effect against SARS-CoV-2 remains unclear. Maestro et al. showed that hemin does not inhibit SARS-CoV-2 viral replication in vitro [175]. Kidney epithelial Vero-E6 and lung Calu3 cell lines were treated with hemin and results showed that, despite a strong activation of HO-1 in both cell lines, there was no effect on SARS-CoV-2 viral replication, measured by the amplification of the N viral gene by RT-qPCR [175]. However, a more recent report proposed hemin as a potential drug for treating COVID-19 via HO-1 induction [176]. Interestingly, authors observed a reduction in SARS-CoV-2 replication, both when pretreating and after SARS-CoV-2 infection treatment of Vero76 with this drug. Genetic induction or silencing of HO-1 in Vero76 cells demonstrated that the antiviral effect of hemin relies on this protein. Strikingly, this effect was mediated not only by Fe2+ and BV, but also by an HO-1 enzymatic independent mechanism. Further, they showed that hemin induced HO-1 boosted ISG15, OAS1 and MX1 protein expression in SARS-CoV-2 infected cells, highlighting the importance of stimulating the host cell’s IFN response against this virus [176]. Of note, there are reports from our laboratory showcasing that MX1 gene expression is increased in COVID-19 patients. However, MX1 expression is lower in elderly patients, where the disease has been shown to be more severe than in younger people. Additionally, through an in depth proteomics analysis, we described MX1 as a novel HO-1 interactor in prostate cancer (PCa) cell lines [177]. Moreover, genetic and pharmacological HO-1 induction in PCa cells triggered an increase in MX1 at mRNA and protein levels, and altered HO-1 cellular localization, showcasing a clear association between both proteins. Further, MX1 silencing with a specific siRNA significantly decreased the expression of ERS genes (HSPA5, DDIT3 and XBP1), demonstrating the role of MX1 in pro-death events [177].
In summary, the induction of the host infected cells antiviral response appears to be critical for COVID-19 treatment, which could be partly achieved by hemin mediated HO-1 induction, also preventing viral adsorption and replication by binding SARS-CoV-2 proteins. These antiviral effects are mediated by canonical and noncanonical HO-1’s functions.
8. HO-1 Induction as a Strategy against COVID-19
There are mainly two different approaches to develop antiviral therapies: (1) therapies directed against viral factors; or (2) therapies targeting the host immune system. To date, the second strategy has received increasing attention due to the fact that targeting viral factors might cause viruses to mutate, increasing the rate of resistance to antiviral drugs [178]. In contrast to the viral genome, host cells’ DNA does not have a high mutational frequency. Therefore, overpowering viral infection by targeting host factors involved in the antiviral response is conceivably an effective strategy to counteract the severe consequences, while also fighting the infection [179].
During the last two years, several reports have focused on the understanding of the virus–host interaction underlying COVID-19 disease. The worrying situation of the SARS-CoV-2 pandemic and the threat of new variants, such as Omicron, which is spreading across the globe at an unprecedented rate, drive the interest of scientists to seek for new anti-SARS-CoV-2 strategies. Its enhanced transmissibility compared to the Delta variant could be explained in part by its increased rate of replication in human primary airway cultures, higher binding to ACE2, and ability to efficiently enter cells in a TMPRSS2-independent manner [180]. Fortunately, preliminary data of the Omicron variant suggest a lower virus load in both lower and upper respiratory tract, associated to less inflammatory processes in the lungs, using a mouse model of severe disease [181]. However, exceptionally high transmissibility could result in a great burden on healthcare systems across the globe. In this context, HO-1 emerges as a potential target to boost the host’s response to fight the infection and prevent severe COVID-19.
Certainly, HO-1 and its reaction products possess beneficial effects for the host during viral infections: it reduces inflammation and exerts antiviral actions. The most serious COVID-19 complications are: sepsis like inflammation, coagulopathy, and cardiovascular or respiratory complications. Furthermore, respiratory failure triggers hypoxia which, in combination with neuroinflammation, generates neurological complications [182]. When inflammation is not modulated, it turns into hyperinflammation and results in tissue damage or organ failure [183]. Enhancing HO-1 expression might help avoid the severe consequences of this disease. For example, it has been reported that HO-1 induction decreases inflammation, inhibits platelet aggregation, and increases fibrinolysis and phagocytosis, thus preventing tissue damage, thrombosis and sepsis [184]. Additionally, hemin is an activator of neuroglobin, a protein involved in oxygen transport and storage in neurons that increases oxygen’s intracellular partial pressure in neurons, and is crucial to protect neurons from hypoxic injury [185,186,187]. In addition, as mentioned above, HO-1 has a reported antiviral activity against multiple viruses. This effect depends on its classical activity involving its reaction subproducts (BR, BV, CO and Fe2+) and the activation of the IFN pathway; interestingly, its noncanonical activity is also involved in the antiviral effect of HO-1.
HO-1 expression is essentially regulated at the transcriptional level by NRF2. It has been reported that SARS-CoV-2 infection suppresses the NRF2 antioxidant gene expression pathway, and that NRF2 agonists limit viral replication and repress the proinflammatory response of SARS-CoV-2 [188]. This evidence highlights the relevance of the NRF2 signaling pathway on the antiviral response, suggesting that the activation of NRF2 might be a useful strategy against COVID-19 [189].
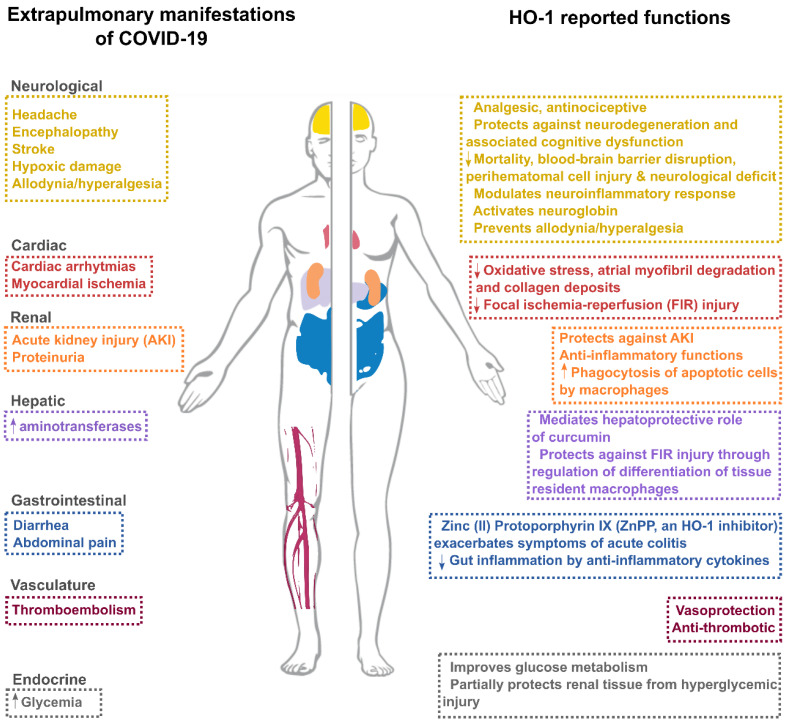
As explained before, clinical complications associated with COVID-19 disease have been described in different organs, including vascular, cardiac, renal, hepatic, endocrine and neurological complications [190] (Figure 4). Interestingly, HO-1 has been reported to be associated with a reduction in tissue damage, mainly through its anti-inflammatory and antioxidative functions in different organs [4,97,147,191,192,193,194,195,196,197,198,199,200,201,202,203,204,205,206,207] (Figure 3). It would be interesting to address HO-1′s vasoprotective and antithrombotic effects for the prevention of thromboembolic events caused by SARS-CoV-2.
Figure 4.
HO-1′s role in different sites that can be affected upon SARS-CoV-2 infection. Extra pulmonary manifestations of COVID-19 are grouped according to their site or body system. HO-1′s reported functions in different experimental conditions or diseases are grouped according to the model or system in which they are studied. The image of the human body has been adapted from Uhlén et al. (Human Protein Atlas, proteinatlas.org) [208].
9. Closing Remarks
Drug repurposing is an attractive proposition, since it involves the use of derisked and previously approved compounds, with lower development costs and shorter development times [209]. Since the onset of the COVID-19 pandemic, we have witnessed a plethora of alternative drugs as potential therapeutic avenues to fight the disease. Thus, hemin, a previously FDA and EMA approved drug for acute intermittent porphyria treatment, rises as a promising drug candidate, inducing HO-1 plasma concentration in humans, and posing a host defense advantage to fight SARS-CoV-2. Further work on optimal drug concentrations, pharmacokinetics and pharmacodynamics should be performed in order to prove hemin’s effectiveness (either alone or in combination with other drugs) to halt infection.
Acknowledgments
We thank Javier Cotignola for his helpful comments and critical revision of this article.
Funding
This work was supported by a grant from AGENCIA-PICT-2019-03215 (ARGENTINA).
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Prete M., Favoino E., Catacchio G., Racanelli V., Perosa F. SARS-CoV-2 Inflammatory Syndrome. Clinical Features and Rationale for Immunological Treatment. Int. J. Mol. Sci. 2020;21:3377. doi: 10.3390/ijms21093377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dunn L.L., Midwinter R.G., Ni J., Hamid H.A., Parish C.R., Stocker R. New insights into intracellular locations and functions of heme oxygenase-1. Antioxid. Redox Signal. 2014;20:1723–1742. doi: 10.1089/ars.2013.5675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Naito Y., Takagi T., Yoshikawa T. Heme oxygenase-1: A new therapeutic target for inflammatory bowel disease. Aliment. Pharmacol. Ther. 2004;20:177–184. doi: 10.1111/j.1365-2036.2004.01992.x. [DOI] [PubMed] [Google Scholar]
- 4.Takagi T., Naito Y., Mizushima K., Hirai Y., Harusato A., Okayama T., Katada K., Kamada K., Uchiyama K., Handa O., et al. Heme oxygenase-1 prevents murine intestinal inflammation. J. Clin. Biochem. Nutr. 2018;63:169–174. doi: 10.3164/jcbn.17-133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chora Â.A., Fontoura P., Cunha A., Pais T.F., Cardoso S., Ho P.P., Lee L.Y., Sobel R.A., Steinman L., Soares M.P. Heme oxygenase-1 and carbon monoxide suppress autoimmune neuroinflammation. J. Clin. Investig. 2007;117:438–447. doi: 10.1172/JCI28844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Protzer U., Seyfried S., Quasdorff M., Sass G., Svorcova M., Webb D., Bohne F., Hösel M., Schirmacher P., Tiegs G. Antiviral Activity and Hepatoprotection by Heme Oxygenase-1 in Hepatitis B Virus Infection. Gastroenterology. 2007;133:1156–1165. doi: 10.1053/j.gastro.2007.07.021. [DOI] [PubMed] [Google Scholar]
- 7.Espinoza J.A., González P.A., Kalergis A.M. Modulation of Antiviral Immunity by Heme Oxygenase-1. Am. J. Pathol. 2017;187:487–493. doi: 10.1016/j.ajpath.2016.11.011. [DOI] [PubMed] [Google Scholar]
- 8.Anderson K.E., Collins S. Open-label study of hemin for acute porphyria: Clinical practice implications. Am. J. Med. 2006;119:801.e19–801.e24. doi: 10.1016/j.amjmed.2006.05.026. [DOI] [PubMed] [Google Scholar]
- 9.European Medicines Agency. [(accessed on 26 January 2022)]. Available online: https://www.ema.europa.eu/en/documents/psusa/human-hemin-list-nationally-authorised-medicinal-products-psusa/00001629/202005_en.pdf.
- 10.Wu D., Wu T., Liu Q., Yang Z. The SARS-CoV-2 outbreak: What we know. Int. J. Infect. Dis. 2020;94:44–48. doi: 10.1016/j.ijid.2020.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hu B., Guo H., Zhou P., Shi Z.L. Characteristics of SARS-CoV-2 and COVID-19. Nat. Rev. Microbiol. 2021;19:141–154. doi: 10.1038/s41579-020-00459-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.WHO Summary of Probable SARS Cases with Onset of Illness from 1 November 2002 to 31 July 2003 (Based on Data as of the 31 December 2003) [(accessed on 6 January 2022)]. Available online: https://www.who.int/publications/m/item/summary-of-probable-sars-cases-with-onset-of-illness-from-1-november-2002-to-31-july-2003%0Ahttps://www.who.int/publications/m/item/summary-of-probable-sars-cases-with-onset-of-illness-from-1-november-2002-to-31-jul.
- 13.Walt G. WHO’s World Health Report 2003. BMJ. 2004;328:6. doi: 10.1136/bmj.328.7430.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Moh Zaki A., Van Boheemen S., Bestebroer T.M., Osterhaus A.D.M.E., Fouchier R.A.M. Isolation of a Novel Coronavirus from a Man with Pneumonia in Saudi Arabia. N. Engl. J. Med. 2012;367:1814–1834. doi: 10.1056/NEJMoa1211721. [DOI] [PubMed] [Google Scholar]
- 15.World Health Organization. [(accessed on 30 December 2021)]. Available online: https://www.who.int/
- 16.Zhu N., Zhang D., Wang W., Li X., Yang B., Song J., Zhao X., Huang B., Shi W., Lu R., et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Johansson M.A., Quandelacy T.M., Kada S., Prasad P.V., Steele M., Brooks J.T., Slayton R.B., Biggerstaff M., Butler J.C. SARS-CoV-2 Transmission from People Without COVID-19 Symptoms. JAMA Netw. Open. 2021;4:e2035057. doi: 10.1001/jamanetworkopen.2020.35057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Song E., Zhang C., Israelow B., Lu-Culligan A., Prado A.V., Skriabine S., Lu P., Weizman O.-E., Liu F., Dai Y., et al. Neuroinvasion of SARS-CoV-2 in human and mouse brain. J. Exp. Med. 2021;218:e20202135. doi: 10.1084/jem.20202135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mulay A., Konda B., Garcia G., Yao C., Beil S., Villalba J., Koziol C., Sen C., Purkayastha A., Kolls J.K., et al. SARS-CoV-2 infection of primary human lung epithelium for COVID-19 modeling and drug discovery. Cell Rep. 2021;35:109055. doi: 10.1016/j.celrep.2021.109055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yaqinuddin A., Kashir J. Novel therapeutic targets for SARS-CoV-2-induced acute lung injury: Targeting a potential IL-1β/neutrophil extracellular traps feedback loop. Med. Hypotheses. 2020;143:109906. doi: 10.1016/j.mehy.2020.109906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xu Z., Shi L., Wang Y., Zhang J., Huang L., Zhang C., Liu S., Zhao P., Liu H., Zhu L., et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet. Respir. Med. 2020;8:420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Soy M., Keser G., Atagündüz P., Tabak F., Atagündüz I., Kayhan S. Cytokine storm in COVID-19: Pathogenesis and overview of anti-inflammatory agents used in treatment. Clin. Rheumatol. 2020;39:2085–2094. doi: 10.1007/s10067-020-05190-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bhaskar S., Sinha A., Banach M., Mittoo S., Weissert R., Kass J.S., Rajagopal S., Pai A.R., Kutty S. Cytokine Storm in COVID-19-Immunopathological Mechanisms, Clinical Considerations, and Therapeutic Approaches: The REPROGRAM Consortium Position Paper. Front. Immunol. 2020;11:1648. doi: 10.3389/fimmu.2020.01648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Channappanavar R., Perlman S. Pathogenic human coronavirus infections: Causes and consequences of cytokine storm and immunopathology. Semin. Immunopathol. 2017;39:529–539. doi: 10.1007/s00281-017-0629-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mokhtari T., Hassani F., Ghaffari N., Ebrahimi B., Yarahmadi A., Hassanzadeh G. COVID-19 and multiorgan failure: A narrative review on potential mechanisms. J. Mol. Histol. 2020;51:613–628. doi: 10.1007/s10735-020-09915-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wang F., Kream R.M., Stefano G.B. Long-Term Respiratory and Neurological Sequelae of COVID-19. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2020;26:e928996. doi: 10.12659/MSM.928996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Al-Aly Z., Xie Y., Bowe B. High-dimensional characterization of post-acute sequelae of COVID-19. Nature. 2021;594:259–264. doi: 10.1038/s41586-021-03553-9. [DOI] [PubMed] [Google Scholar]
- 28.Cao Y., Wang J., Jian F., Xiao T., Song W., Yisimayi A., Huang W., Li Q., Wang P., An R., et al. Omicron escapes the majority of existing SARS-CoV-2 neutralizing antobodies. Res. Briefings. 2021 doi: 10.1038/s41586-021-04385-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lu L., Mok B.W.-Y., Chen L.-L., Chan J.M.-C., Tsang O.T.-Y., Lam B.H.-S., Chuang V.W.-M., Chu A.W.-H., Chan W.-M., Ip J.D., et al. Neutralization of SARS-CoV-2 Omicron variant by sera from BNT162b2 or Coronavac vaccine recipients. Clin. Infect. Dis. 2021:ciab1041. doi: 10.1093/cid/ciab1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cascella M., Rajnik M., Aleem A. Features, Evaluation, and Treatment of Coronavirus (COVID-19) StatPearls; Treasure Island, FL, USA: 2021. [PubMed] [Google Scholar]
- 31.Khailany R.A., Safdar M., Ozaslan M. Genomic characterization of a novel SARS-CoV-2. Gene Rep. 2020;19 doi: 10.1016/j.genrep.2020.100682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Janik E., Niemcewicz M., Podogrocki M., Majsterek I., Bijak M. The Emerging Concern and Interest SARS-CoV-2 Variants. Pathogens. 2021;10:633. doi: 10.3390/pathogens10060633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.D’Elia R.V., Harrison K., Oyston P.C., Lukaszewski R.A., Clark G.C. Targeting the “cytokine storm” for therapeutic benefit. Clin. Vaccine Immunol. 2013;20:319–327. doi: 10.1128/CVI.00636-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen L., Deng H., Cui H., Fang J., Zuo Z., Deng J., Li Y., Wang X., Zhao L. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 2018;9:7204–7218. doi: 10.18632/oncotarget.23208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Medzhitov R. Recognition of microorganisms and activation of the immune response. Nature. 2007;449:819–826. doi: 10.1038/nature06246. [DOI] [PubMed] [Google Scholar]
- 36.Speyer C.L., Ward P.A. Role of endothelial chemokines and their receptors during inflammation. J. Investig. Surg. 2011;24:18–27. doi: 10.3109/08941939.2010.521232. [DOI] [PubMed] [Google Scholar]
- 37.Yang L., Xie X., Tu Z., Fu J., Xu D., Zhou Y. The signal pathways and treatment of cytokine storm in COVID-19. Signal Transduct. Target. Ther. 2021;6:255. doi: 10.1038/s41392-021-00679-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tay M.Z., Poh C.M., Rénia L., MacAry P.A., Ng L.F.P. The trinity of COVID-19: Immunity, inflammation and intervention. Nat. Rev. Immunol. 2020;20:363–374. doi: 10.1038/s41577-020-0311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lucas C., Wong P., Klein J., Castro T.B.R., Silva J., Sundaram M., Ellingson M.K., Mao T., Oh J.E., Israelow B., et al. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature. 2020;584:463. doi: 10.1038/s41586-020-2588-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Blanco-Melo D., Nilsson-Payant B.E., Liu W.C., Uhl S., Hoagland D., Møller R., Jordan T.X., Oishi K., Panis M., Sachs D., et al. Imbalanced Host Response to SARS-CoV-2 Drives Development of COVID-19. Cell. 2020;181:1036–1045.e9. doi: 10.1016/j.cell.2020.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Park A., Iwasaki A. Type I and Type III Interferons—Induction, Signaling, Evasion, and Application to Combat COVID-19. Cell Host Microbe. 2020;27:870–878. doi: 10.1016/j.chom.2020.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hervas-Stubbs S., Perez-Gracia J.L., Rouzaut A., Sanmamed M.F., Le Bon A., Melero I. Direct Effects of Type I Interferons on Cells of the Immune System. Clin. Cancer Res. 2011;17:2619. doi: 10.1158/1078-0432.CCR-10-1114. [DOI] [PubMed] [Google Scholar]
- 43.Levy D.E., García-Sastre A. The Virus Battles: IFN Induction of the Antiviral State and Mechanisms of Viral Evasion. Cytokine Growth Factor Rev. 2001;12:143–156. doi: 10.1016/S1359-6101(00)00027-7. [DOI] [PubMed] [Google Scholar]
- 44.Goodbourn S., Didcock L., Randall R.E. Interferons: Cell signalling, immune modulation, antiviral response and virus countermeasures. J. Gen. Virol. 2000;81:2341–2364. doi: 10.1099/0022-1317-81-10-2341. [DOI] [PubMed] [Google Scholar]
- 45.Cebulla C.M., Miller D.M., Sedmak D.D. Viral inhibition of interferon signal transduction. Intervirology. 1999;42:325–330. doi: 10.1159/000053968. [DOI] [PubMed] [Google Scholar]
- 46.Ruan Q., Yang K., Wang W., Jiang L., Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46:846–848. doi: 10.1007/s00134-020-05991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yang Y., Shen C., Li J., Yuan J., Wei J., Huang F., Wang F., Li G., Li Y., Xing L., et al. Plasma IP-10 and MCP-3 levels are highly associated with disease severity and predict the progression of COVID-19. J. Allergy Clin. Immunol. 2020;146:119–127.e4. doi: 10.1016/j.jaci.2020.04.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lee J.S., Shin E.-C. The type I interferon response in COVID-19: Implications for treatment. Nat. Rev. Immunol. 2020;20:585–586. doi: 10.1038/s41577-020-00429-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Davoudi-Monfared E., Rahmani H., Khalili H., Hajiabdolbaghi M., Salehi M., Abbasian L., Kazemzadeh H., Yekaninejad M.S. A Randomized Clinical Trial of the Efficacy and Safety of Interferon β-1a in Treatment of Severe COVID-19. Antimicrob. Agents Chemother. 2020;64:e01061-20. doi: 10.1128/AAC.01061-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rahmani H., Davoudi-Monfared E., Nourian A., Khalili H., Hajizadeh N., Jalalabadi N.Z., Fazeli M.R., Ghazaeian M., Yekaninejad M.S. Interferon β-1b in treatment of severe COVID-19: A randomized clinical trial. Int. Immunopharmacol. 2020;88:106903. doi: 10.1016/j.intimp.2020.106903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhou Q., Chen V., Shannon C.P., Wei X.S., Xiang X., Wang X., Wang Z.H., Tebbutt S.J., Kollmann T.R., Fish E.N. Interferon-α2b Treatment for COVID-19. Front. Immunol. 2020;11:1061. doi: 10.3389/fimmu.2020.01061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Feld J.J., Kandel C., Biondi M.J., Kozak R.A., Zahoor M.A., Lemieux C., Borgia S.M., Boggild A.K., Powis J., McCready J., et al. Peginterferon lambda for the treatment of outpatients with COVID-19: A phase 2, placebo-controlled randomised trial. Lancet Respir. Med. 2021;9:498–510. doi: 10.1016/S2213-2600(20)30566-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.ClinicalTrials.gov [Internet]. IFN-beta 1b and Remdesivir for COVID19. [(accessed on 7 January 2022)]; Available online: https://clinicaltrials.gov/ct2/show/NCT04647695.
- 54.ClinicalTrials.gov [Internet]. Experimental Trial of rhIFNα Nasal Drops to Prevent 2019-nCOV in Medical Staff; 2021 Nov 30. [(accessed on 7 January 2022)]; Available online: https://clinicaltrials.gov/ct2/show/NCT04320238.
- 55.ClinicalTrials.gov [Internet]. Efficacy and Safety of IFN-α2β in the Treatment of Novel Coronavirus Patients; 2021 Nov 30. [(accessed on 7 January 2022)]; Available online: https://clinicaltrials.gov/ct2/show/NCT04293887.
- 56.Kumar S., Bandyopadhyay U. Free heme toxicity and its detoxification systems in human. Toxicol. Lett. 2005;157:175–188. doi: 10.1016/j.toxlet.2005.03.004. [DOI] [PubMed] [Google Scholar]
- 57.Hayashi S., Omata Y., Sakamoto H., Higashimoto Y., Hara T., Sagara Y., Noguchi M. Characterization of rat heme oxygenase-3 gene. Implication of processed pseudogenes derived from heme oxygenase-2 gene. Gene. 2004;336:241–250. doi: 10.1016/j.gene.2004.04.002. [DOI] [PubMed] [Google Scholar]
- 58.Yachie A., Niida Y., Wada T., Igarashi N., Kaneda H., Toma T., Ohta K., Kasahara Y., Koizumi S. Oxidative stress causes enhanced endothelial cell injury in human heme oxygenase-1 deficiency. J. Clin. Investig. 1999;103:129–135. doi: 10.1172/JCI4165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Le Moine A., Ryter S.W., Jang H.-S., Liu C., Li Y., Ma K., Han Z., Chi M., Sai X., Zhu P., et al. Immunomodulatory Effects of Heme Oxygenase-1 in Kidney Disease. Front. Med. 2021;1:708453. doi: 10.3389/fmed.2021.708453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Chiang K.-C., Chang K.-S., Hsu S.-Y., Sung H.-C., Feng T.-H., Chao M., Juang H.-H. Human Heme Oxygenase-1 Induced by Interleukin-6 via JAK/STAT3 Pathways Is a Tumor Suppressor Gene in Hepatoma Cells. Antioxidants. 2020;9:251. doi: 10.3390/antiox9030251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Gueron G., De Siervi A., Ferrando M., Salierno M., De Luca P., Elguero B., Meiss R., Navone N., Vazquez E.S. Critical Role of Endogenous Heme Oxygenase 1 as a Tuner of the Invasive Potential of Prostate Cancer Cells. Mol. Cancer Res. 2009;7:1745–1755. doi: 10.1158/1541-7786.MCR-08-0325. [DOI] [PubMed] [Google Scholar]
- 62.Immenschuh S., Schröder H. Heme oxygenase-1 and cardiovascular disease. Histol. Histopathol. 2006;21:679–685. doi: 10.14670/HH-21.679. [DOI] [PubMed] [Google Scholar]
- 63.Lin X., Lv J., Ge D., Bai H., Yang Y., Wu J. Heme oxygenase-1 alleviates eosinophilic inflammation by inhibiting STAT3-SOCS3 signaling. Pediatr. Pulmonol. 2020;55:1440–1447. doi: 10.1002/ppul.24759. [DOI] [PubMed] [Google Scholar]
- 64.Sheikh S.Z., Hegazi R.A., Kobayashi T., Onyiah J.C., Russo S.M., Matsuoka K., Sepulveda A.R., Li F., Otterbein L.E., Plevy S.E. An anti-inflammatory role for carbon monoxide and heme oxygenase-1 in chronic Th2-mediated murine colitis. J. Immunol. 2011;186:5506–5513. doi: 10.4049/jimmunol.1002433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Paine A., Eiz-Vesper B., Blasczyk R., Immenschuh S. Signaling to heme oxygenase-1 and its anti-inflammatory therapeutic potential. Biochem. Pharmacol. 2010;80:1895–1903. doi: 10.1016/j.bcp.2010.07.014. [DOI] [PubMed] [Google Scholar]
- 66.Tzima S., Victoratos P., Kranidioti K., Alexiou M., Kollias G. Myeloid heme oxygenase-1 regulates innate immunity and autoimmunity by modulating IFN-β production. J. Exp. Med. 2009;206:1167–1179. doi: 10.1084/jem.20081582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kotsch K., Martins P.N.A., Klemz R., Janssen U., Gerstmayer B., Dernier A., Reutzel-Selke A., Kuckelkorn U., Tullius S.G., Volk H.D. Heme oxygenase-1 ameliorates ischemia/reperfusion injury by targeting dendritic cell maturation and migration. Antioxidants Redox Signal. 2007;9:2049–2063. doi: 10.1089/ars.2007.1801. [DOI] [PubMed] [Google Scholar]
- 68.Chauveau C., Rémy S., Royer P.J., Hill M., Tanguy-Royer S., Hubert F.X., Tesson L., Brion R., Beriou G., Gregoire M., et al. Heme oxygenase-1 expression inhibits dendritic cell maturation and proinflammatory function but conserves IL-10 expression. Blood. 2005;106:1694–1702. doi: 10.1182/blood-2005-02-0494. [DOI] [PubMed] [Google Scholar]
- 69.Kapturczak M.H., Wasserfall C., Brusko T., Campbell-Thompson M., Ellis T.M., Atkinson M.A., Agarwal A. Heme Oxygenase-1 Modulates Early Inflammatory Responses Evidence from the Heme Oxygenase-1-Deficient Mouse. Am. J. Pathol. 2004;165:1045–1053. doi: 10.1016/S0002-9440(10)63365-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Otterbein L.E., Bach F.H., Alam J., Soares M., Tao Lu H., Wysk M., Davis R.J., Flavell R.A., Choi A.M.K. Carbon monoxide has anti-inflammatory effects involving the mitogen-activated protein kinase pathway. Nat. Med. 2000;6:422–428. doi: 10.1038/74680. [DOI] [PubMed] [Google Scholar]
- 71.Lee T.-S., Chau L.-Y. Heme oxygenase-1 mediates the anti-inflammatory effect of interleukin-10 in mice. Nat. Med. 2002;8:240–246. doi: 10.1038/nm0302-240. [DOI] [PubMed] [Google Scholar]
- 72.Bach Tatiana Vassilevskaia F.H., Berberat P.O., Yu J., Miguel Soares T.-Y.P., Seldon M.P., Pombo Gregoire I. Endothelial Cell Activation of Adhesion Molecules Associated with Heme Oxygenase-1 Modulates the Expression. J. Immunol. Ref. 2021;172:3553–3563. doi: 10.4049/jimmunol.172.6.3553. [DOI] [PubMed] [Google Scholar]
- 73.Nakamura T., Naguro I., Ichijo H. Iron homeostasis and iron-regulated ROS in cell death, senescence and human diseases. Biochim. Biophys. Acta Gen. Subj. 2019;1863:1398–1409. doi: 10.1016/j.bbagen.2019.06.010. [DOI] [PubMed] [Google Scholar]
- 74.Ryter S.W., Alam J., Choi A.M.K. Heme oxygenase-1/carbon monoxide: From basic science to therapeutic applications. Physiol. Rev. 2006;86:583–650. doi: 10.1152/physrev.00011.2005. [DOI] [PubMed] [Google Scholar]
- 75.Wu B., Wu Y., Tang W. Heme Catabolic Pathway in Inflammation and Immune Disorders. Front. Pharmacol. 2019;10:825. doi: 10.3389/fphar.2019.00825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Takei R., Inoue T., Sonoda N., Kohjima M., Okamoto M., Sakamoto R., Inoguchi T., Ogawa Y. Bilirubin reduces visceral obesity and insulin resistance by suppression of inflammatory cytokines. PLoS ONE. 2019;14:e0223302. doi: 10.1371/journal.pone.0223302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Brouard S., Otterbein L.E., Anrather J., Tobiasch E., Bach F.H., Choi A.M.K., Soares M.P. Carbon Monoxide Generated by Heme Oxygenase 1 Suppresses Endothelial Cell Apoptosis. J. Exp. Med. 2000;192:1015–1026. doi: 10.1084/jem.192.7.1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Petrache I., Otterbein L.E., Alam J., Wiegand G.W., Choi A.M.K. Heme oxygenase-1 inhibits TNF-α-induced apoptosis in cultured fibroblasts. Am. J. Physiol. Cell. Mol. Physiol. 2000;278:L312–L319. doi: 10.1152/ajplung.2000.278.2.L312. [DOI] [PubMed] [Google Scholar]
- 79.Loboda A., Jazwa A., Grochot-Przeczek A., Rutkowski A.J., Cisowski J., Agarwal A., Jozkowicz A., Dulak J. Heme oxygenase-1 and the vascular bed: From molecular mechanisms to therapeutic opportunities. Antioxidants Redox Signal. 2008;10:1767–1812. doi: 10.1089/ars.2008.2043. [DOI] [PubMed] [Google Scholar]
- 80.Kliche S., Waltenberger J. VEGF Receptor Signaling and Endothelial Function. IUBMB Life. 2001;52:61–66. doi: 10.1080/15216540252774784. [DOI] [PubMed] [Google Scholar]
- 81.Pae H.-O., Oh G.-S., Choi B.-M., Chae S.-C., Kim Y.-M., Chung K.-R., Chung H.-T. Carbon Monoxide Produced by Heme Oxygenase-1 Suppresses T Cell Proliferation via Inhibition of IL-2 Production. J. Immunol. 2004;172:4744–4751. doi: 10.4049/jimmunol.172.8.4744. [DOI] [PubMed] [Google Scholar]
- 82.Morse D., Pischke S.E., Zhou Z., Davis R.J., Flavell R.A., Loop T., Otterbein S.L., Otterbein L.E., Choi A.M.K. Suppression of inflammatory cytokine production by carbon monoxide involves the JNK pathway and AP-1. J. Biol. Chem. 2003;278:36993–36998. doi: 10.1074/jbc.M302942200. [DOI] [PubMed] [Google Scholar]
- 83.Kim S.J., Lee S.M. NLRP3 inflammasome activation in d-galactosamine and lipopolysaccharide- induced acute liver failure: Role of heme oxygenase-1. Free Radic. Biol. Med. 2013;65:997–1004. doi: 10.1016/j.freeradbiomed.2013.08.178. [DOI] [PubMed] [Google Scholar]
- 84.Luo Y.P., Jiang L., Kang K., Fei D.S., Meng X.L., Nan C.C., Pan S.H., Zhao M.R., Zhao M.Y. Hemin inhibits NLRP3 inflammasome activation in sepsis-induced acute lung injury, involving heme oxygenase-1. Int. Immunopharmacol. 2014;20:24–32. doi: 10.1016/j.intimp.2014.02.017. [DOI] [PubMed] [Google Scholar]
- 85.Lv J., Su W., Yu Q., Zhang M., Di C., Lin X., Wu M., Xia Z. Heme oxygenase-1 protects airway epithelium against apoptosis by targeting the proinflammatory NLRP3–RXR axis in asthma. J. Biol. Chem. 2018;293:18454–18465. doi: 10.1074/jbc.RA118.004950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Abraham N.G., Kappas A. Pharmacological and clinical aspects of heme oxygenase. Pharmacol. Rev. 2008;60:79–127. doi: 10.1124/pr.107.07104. [DOI] [PubMed] [Google Scholar]
- 87.Fan Y., Zhang J., Cai L., Wang S., Liu C., Zhang Y., You L., Fu Y., Shi Z., Yin Z., et al. The effect of anti-inflammatory properties of ferritin light chain on lipopolysaccharide-induced inflammatory response in murine macrophages. Biochim. Biophys. Acta Mol. Cell Res. 2014;1843:2775–2783. doi: 10.1016/j.bbamcr.2014.06.015. [DOI] [PubMed] [Google Scholar]
- 88.Sass G., Soares M.C.P., Yamashita K., Seyfried S., Zimmermann W.-H., Eschenhagen T., Kaczmarek E., Ritter T., Volk H.-D., Tiegs G. Heme oxygenase-1 and its reaction product, carbon monoxide, prevent inflammation-related apoptotic liver damage in mice. Hepatology. 2003;38:909–918. doi: 10.1002/hep.1840380417. [DOI] [PubMed] [Google Scholar]
- 89.Chen Z., Zhong H., Wei J., Lin S., Zong Z., Gong F., Huang X., Sun J., Li P., Lin H., et al. Inhibition of Nrf2/HO-1 signaling leads to increased activation of the NLRP3 inflammasome in osteoarthritis. Arthritis Res. Ther. 2019;21:300. doi: 10.1186/s13075-019-2085-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Zhang B., Xie S., Su Z., Song S., Xu H., Chen G., Cao W., Yin S., Gao Q., Wang H. Heme oxygenase-1 induction attenuates imiquimod-induced psoriasiform inflammation by negative regulation of Stat3 signaling. Sci. Rep. 2016;6:21132. doi: 10.1038/srep21132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Liu X., Gao Y., Li M., Geng C., Xu H., Yang Y., Guo Y., Jiao T., Fang F., Chang Y. Sirt1 mediates the effect of the heme oxygenase inducer, cobalt protoporphyrin, on ameliorating liver metabolic damage caused by a high-fat diet. J. Hepatol. 2015;63:713–721. doi: 10.1016/j.jhep.2015.05.018. [DOI] [PubMed] [Google Scholar]
- 92.Zhong W., Xia Z., Hinrichs D., Rosenbaum J.T., Wegmann K.W., Meyrowitz J., Zhang Z. Hemin exerts multiple protective mechanisms and attenuates dextran sulfate sodium-induced colitis. J. Pediatr. Gastroenterol. Nutr. 2010;50:132–139. doi: 10.1097/MPG.0b013e3181c61591. [DOI] [PubMed] [Google Scholar]
- 93.Liu Y., Zhu B., Luo L., Li P., Paty D.W., Cynader M.S. Heme oxygenase-1 plays an important protective role in experimental autoimmune encephalomyelitis. Neuroreport. 2001;12:1841–1845. doi: 10.1097/00001756-200107030-00016. [DOI] [PubMed] [Google Scholar]
- 94.Herfarth H., Paul G., Bataille F., Obermeier F., Bock J., Klebl F., Strauch U., Lochbaum D., Rümmele P., Farkas S., et al. Analysis of intestinal haem-oxygenase-1 (HO-1 ) in clinical and experimental colitis. Clin. Exp. Immunol. 2005;140:547–555. doi: 10.1111/j.1365-2249.2005.02775.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Chen D., Wu C., Qiu Y.B., Chu Q., Sun X.Q., Wang X., Chen J.L., Lu M.D., Chen D.Z., Pang Q. feng Curcumin ameliorates hepatic chronic inflammation induced by bile duct obstruction in mice through the activation of heme oxygenase-1. Int. Immunopharmacol. 2020;78:106054. doi: 10.1016/j.intimp.2019.106054. [DOI] [PubMed] [Google Scholar]
- 96.Foresti R., Bains S.K., Pitchumony T.S., De Castro Brás L.E., Drago F., Dubois-Randé J.L., Bucolo C., Motterlini R. Small molecule activators of the Nrf2-HO-1 antioxidant axis modulate heme metabolism and inflammation in BV2 microglia cells. Pharmacol. Res. 2013;76:132–148. doi: 10.1016/j.phrs.2013.07.010. [DOI] [PubMed] [Google Scholar]
- 97.Selva Nandakumar K., Blank M., Kalergis A.M., Funes S.C., Rios M., Fernández-Fierro A., Covián C., Bueno S.M., Riedel C.A., Mackern-Oberti J.P. Naturally Derived Heme-Oxygenase 1 Inducers and Their Therapeutic Application to Immune-Mediated Diseases. Front. Immunol. 2020;11:1467. doi: 10.3389/fimmu.2020.01467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Kang S., Kim W., Jeong S., Lee Y., Nam J., Lee S., Jung Y. Oxidized 5-aminosalicylic acid activates Nrf2-HO-1 pathway by covalently binding to Keap1: Implication in anti-inflammatory actions of 5-aminosalicylic acid. Free Radic. Biol. Med. 2017;108:715–724. doi: 10.1016/j.freeradbiomed.2017.04.366. [DOI] [PubMed] [Google Scholar]
- 99.Nishio Y., Fujino M., Zhao M., Ishii T., Ishizuka M., Ito H., Takahashi K., Abe F., Nakajima M., Tanaka T., et al. 5-Aminolevulinic acid combined with ferrous iron enhances the expression of heme oxygenase-1. Int. Immunopharmacol. 2014;19:300–307. doi: 10.1016/j.intimp.2014.02.003. [DOI] [PubMed] [Google Scholar]
- 100.Berberat P.O., A-Rahim Y.I., Yamashita K., Warny M.M., Csizmadia E., Robson S.C., Bach F.H. Heme Oxygenase-1-Generated Biliverdin Ameliorates Experimental Murine Colitis. Inflamm. Bowel Dis. 2005;11:350–359. doi: 10.1097/01.MIB.0000164017.06538.8a. [DOI] [PubMed] [Google Scholar]
- 101.Keum H., Kim T.W., Kim Y., Seo C., Son Y., Kim J., Kim D., Jung W., Whang C.H., Jon S. Bilirubin nanomedicine alleviates psoriatic skin inflammation by reducing oxidative stress and suppressing pathogenic signaling. J. Control Release. 2020;325:359–369. doi: 10.1016/j.jconrel.2020.07.015. [DOI] [PubMed] [Google Scholar]
- 102.Rochette L., Zeller M., Cottin Y., Vergely C. Redox Functions of Heme Oxygenase-1 and Biliverdin Reductase in Diabetes. Trends Endocrinol. Metab. 2018;29:74–85. doi: 10.1016/j.tem.2017.11.005. [DOI] [PubMed] [Google Scholar]
- 103.Lin C.-C., Yang C.-C., Hsiao L.-D., Chen S.-Y., Yang C.-M. Heme Oxygenase-1 Induction by Carbon Monoxide Releasing Molecule-3 Suppresses Interleukin-1β-Mediated Neuroinflammation. Front. Mol. Neurosci. 2017;10:387. doi: 10.3389/fnmol.2017.00387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Motterlini R., Mann B.E., Foresti R. Therapeutic applications of carbon monoxide-releasing molecules. Expert Opin. Investig. Drugs. 2005;14:1305–1318. doi: 10.1517/13543784.14.11.1305. [DOI] [PubMed] [Google Scholar]
- 105.Motterlini R., Otterbein L.E. The therapeutic potential of carbon monoxide. Nat. Rev. Drug Discov. 2010;9:728–743. doi: 10.1038/nrd3228. [DOI] [PubMed] [Google Scholar]
- 106.Ali Z.E., Ollivier A., Manin S., Rivard M., Motterlini R., Foresti R. Therapeutic effects of CO-releaser/Nrf2 activator hybrids (HYCOs) in the treatment of skin wound, psoriasis and multiple sclerosis. Redox Biol. 2020;34:101521. doi: 10.1016/j.redox.2020.101521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lad L., Friedman J., Li H., Bhaskar B., Ortiz de Montellano P.R., Poulos T.L. Crystal structure of human heme oxygenase-1 in a complex with biliverdin. Biochemistry. 2004;43:3793–3801. doi: 10.1021/bi035451l. [DOI] [PubMed] [Google Scholar]
- 108.National Center for Biotechnology Information PubChem Compound Summary for CID 4971, Protoporphyrin IX. [(accessed on 4 January 2022)];2022 Available online: https://pubchem.ncbi.nlm.nih.gov/compound/protoporphyrin-IX.
- 109.Fredenburgh L.E., Perrella M.A., Mitsialis S.A. The role of heme oxygenase-1 in pulmonary disease. Am. J. Respir. Cell Mol. Biol. 2007;36:158–165. doi: 10.1165/rcmb.2006-0331TR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Slebos D.-J., Ryter S.W., Choi A.M.K. Heme oxygenase-1 and carbon monoxide in pulmonary medicine. Respir. Res. 2003;4:7. doi: 10.1186/1465-9921-4-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Di Pietro C., Öz H.H., Murray T.S., Bruscia E.M. Targeting the Heme Oxygenase 1/Carbon Monoxide Pathway to Resolve Lung Hyper-Inflammation and Restore a Regulated Immune Response in Cystic Fibrosis. Front. Pharmacol. 2020;11:1059. doi: 10.3389/fphar.2020.01059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Pedersen S.F., Ho Y.-C. SARS-CoV-2: A storm is raging. J. Clin. Investig. 2020;130:2202–2205. doi: 10.1172/JCI137647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Kritas S., Ronconi G., Caraffa A., Gallenga C., Ross R., Conti P. Mast cells contribute to coronavirus-induced inflammation: New anti-inflammatory strategy. J. Biol. Regul. Homeost. Agents. 2019;34:9–14. doi: 10.23812/20-Editorial-Kritas. [DOI] [PubMed] [Google Scholar]
- 114.He Y.Q., Zhou C.C., Yu L.Y., Wang L., Deng J.L., Tao Y.L., Zhang F., Chen W.S. Natural product derived phytochemicals in managing acute lung injury by multiple mechanisms. Pharmacol. Res. 2021;163:105224. doi: 10.1016/j.phrs.2020.105224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Butt Y., Kurdowska A., Timothy A.C. Acute Lung Injury A Clinical and Molecular Review. Arch. Pathol. Lab. Med. 2016;140:345–350. doi: 10.5858/arpa.2015-0519-RA. [DOI] [PubMed] [Google Scholar]
- 116.Huang X., Xiu H., Zhang S., Zhang G. The role of macrophages in the pathogenesis of ali/ards. Mediat. Inflamm. 2018;2018:1–8. doi: 10.1155/2018/1264913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Pereira M.L.M., Marinho C.R.F., Epiphanio S. Could heme oxygenase-1 be a new target for therapeutic intervention in malaria-associated acute lung injury/acute respiratory distress syndrome? Front. Cell. Infect. Microbiol. 2018;8:161. doi: 10.3389/fcimb.2018.00161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.An L., Liu C.T., Qin X.B., Liu Q.H., Liu Y., Yu S.Y. Protective effects of hemin in an experimental model of ventilator-induced lung injury. Eur. J. Pharmacol. 2011;661:102–108. doi: 10.1016/j.ejphar.2011.04.032. [DOI] [PubMed] [Google Scholar]
- 119.Xia Z.Y., Gao J., Ancharaz A.K. Protective effect of ischemic postconditioning on lung ischemia-reperfusion injury in rats and the role of heme oxygenase-1. Chin. J. Traumatol. Engl. Ed. 2009;12:162–166. [PubMed] [Google Scholar]
- 120.Chen X., Wang Y., Xie X., Chen H., Zhu Q., Ge Z., Wei H., Deng J., Xia Z., Lian Q. Heme Oxygenase-1 Reduces Sepsis-Induced Endoplasmic Reticulum Stress and Acute Lung Injury. Mediators Inflamm. 2018;2018:9413876. doi: 10.1155/2018/9413876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Yu J., Wang Y., Li Z., Dong S., Wang D., Gong L., Shi J., Zhang Y., Liu D., Mu R. Effect of Heme Oxygenase-1 on Mitofusin-1 protein in LPS-induced ALI/ARDS in rats. Sci. Rep. 2016;6:36530. doi: 10.1038/srep36530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Nakashima K., Sato T., Shigemori S., Shimosato T., Shinkai M., Kaneko T. Regulatory role of heme oxygenase-1 in silica-induced lung injury. Respir. Res. 2018;19:144. doi: 10.1186/s12931-018-0852-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Ryter S.W. Therapeutic Potential of Heme Oxygenase-1 and Carbon Monoxide in Acute Organ Injury, Critical Illness, and Inflammatory Disorders. Antioxidants. 2020;9:1153. doi: 10.3390/antiox9111153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Shen N., Gong T., Wang J.D., Meng F.L., Qiao L., Yang R.L., Xue B., Pan F.Y., Zhou X.J., Chen H.Q., et al. Cigarette smoke-induced pulmonary inflammatory responses are mediated by EGR-1/GGPPS/MAPK signaling. Am. J. Pathol. 2011;178:110–118. doi: 10.1016/j.ajpath.2010.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Fujita T., Toda K., Karimova A., Yan S.-F., Naka Y., Yet S.-F., Pinsky D.J. Paradoxical rescue from ischemic lung injury by inhaled carbon monoxide driven by derepression of fibrinolysis. Nat. Med. 2001;7:598–604. doi: 10.1038/87929. [DOI] [PubMed] [Google Scholar]
- 126.Tian W.F., Weng P., Sheng Q., Chen J.L., Zhang P., Zhang J.R., Du B., Wu M.C., Pang Q.F., Chu J.J. Biliverdin protects the isolated rat lungs from ischemia-reperfusion injury via antioxidative, anti-inflammatory and anti-apoptotic effects. Chin. Med. J. 2017;130:859–865. doi: 10.4103/0366-6999.202735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Kosaka J., Morimatsu H., Takahashi T., Shimizu H., Kawanishi S. Effects of Biliverdin Administration on Acute Lung Injury Induced by Hemorrhagic Shock and Resuscitation in Rats. PLoS ONE. 2013;8:63606. doi: 10.1371/journal.pone.0063606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Ma L.L., Zhang P., Wang H.Q., Li Y.F., Hu J., Jiang J.D., Li Y.H. heme oxygenase-1 agonist CoPP suppresses influenza virus replication through IRF3-mediated generation of IFN-α/β. Virology. 2019;528:80–88. doi: 10.1016/j.virol.2018.11.016. [DOI] [PubMed] [Google Scholar]
- 129.Iuliano A.D., Roguski K.M., Chang H.H., Muscatello D.J., Palekar R., Tempia S., Cohen C., Gran J.M., Schanzer D., Cowling B.J., et al. Estimates of global seasonal influenza-associated respiratory mortality: A modelling study. Lancet. 2018;391:1285–1300. doi: 10.1016/S0140-6736(17)33293-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Kawser Hossain M., Kumar S., Id S., Dayem A.A., Kim J.-H., Kim K., Yang G.-M., Choi H.Y., Cho S.-G. Bax Inhibitor-1 Acts as an Anti-Influenza Factor by Inhibiting ROS Mediated Cell Death and Augmenting Heme-Oxygenase 1 Expression in Influenza Virus Infected Cells. Int. J. Mol. Sci. Artic. 2018;19:712. doi: 10.3390/ijms19030712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Wang C., Zhang Y., Han L.L., Guo L., Zhong H., Wang J. Hemin ameliorates influenza pneumonia by attenuating lung injury and regulating the immune response. Int. J. Antimicrob. Agents. 2017;49:45–52. doi: 10.1016/j.ijantimicag.2016.09.030. [DOI] [PubMed] [Google Scholar]
- 132.Hashiba T., Suzuki M., Nagashima Y., Suzuki S., Inoue S., Tsuburai T., Matsuse T., Ishigatubo Y. Adenovirus-Mediated Transfer of Heme Oxygenase-1 cDNA Attenuates Severe Lung Injury Induced by the Influenza Virus in Mice. Gene Ther. 2001;8:1499–1507. doi: 10.1038/sj.gt.3301540. [DOI] [PubMed] [Google Scholar]
- 133.Cummins N.W., Weaver E.A., May S.M., Croatt A.J., Foreman O., Kennedy R.B., Poland G.A., Barry M.A., Nath K.A., Badley A.D. Heme oxygenase-1 regulates the immune response to influenza virus infection and vaccination in aged mice. FASEB J. 2012;26:2911–2918. doi: 10.1096/fj.11-190017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Zhong M., Wang H., Ma L., Yan H., Wu S., Gu Z., Li Y. DMO-CAP inhibits influenza virus replication by activating heme oxygenase-1-mediated IFN response. Virol. J. 2019;16:21. doi: 10.1186/s12985-019-1125-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Ma L.-L., Wang H.-Q., Wu P., Hu J., Yin J.-Q., Wu S., Ge M., Sun W.-F., Zhao J.-Y., Aisa H.A., et al. Rupestonic acid derivative YZH-106 suppresses influenza virus replication by activation of heme oxygenase-1-mediated interferon response. Free Radic. Biol. Med. 2016;96:347–361. doi: 10.1016/j.freeradbiomed.2016.04.021. [DOI] [PubMed] [Google Scholar]
- 136.Nair DNB H., Theodoratou E., Rudan I., Nokes D.J., Ngama HND M., Munywoki P.K., Dherani M., Nair H., James Nokes D., Gessner B.D., et al. Global burden of acute lower respiratory infections due to respiratory syncytial virus in young children: A systematic review and meta-analysis. Lancet. 2010;375:1545–1555. doi: 10.1016/S0140-6736(10)60206-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Del Vecchio A., Ferrara T., Maglione M., Capasso L., Raimondi F. New perspectives in Respiratory Syncitial Virus infection. J. Matern. Neonatal Med. 2013;26:55–59. doi: 10.3109/14767058.2013.831282. [DOI] [PubMed] [Google Scholar]
- 138.Yoboua F., Martel A., Duval A., Mukawera E., Grandvaux N. Respiratory Syncytial Virus-Mediated NF-B p65 Phosphorylation at Serine 536 Is Dependent on RIG-I, TRAF6, and IKK. J. Virol. 2010;84:7267–7277. doi: 10.1128/JVI.00142-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Espinoza J.A., León M.A., Céspedes P.F., Gómez R.S., Canedo-Marroquín G., Riquelme S.A., Salazar-Echegarai F.J., Blancou P., Simon T., Anegon I., et al. Heme Oxygenase-1 Modulates Human Respiratory Syncytial Virus Replication and Lung Pathogenesis during Infection. J. Immunol. 2017;199:212. doi: 10.4049/jimmunol.1601414. [DOI] [PubMed] [Google Scholar]
- 140.Antiretroviral Therapy has Saved Millions of Lives from AIDS and Could Save More. [(accessed on 3 January 2022)]. Available online: https://ourworldindata.org/art-lives-saved.
- 141.UNAIDS: Joint United Nations Programme on HIV/AIDS. [(accessed on 31 December 2021)]. Available online: https://www.un.org/youthenvoy/2013/08/unaids-joint-united-nations-programme-on-hivaids/
- 142.Global Statistics. [(accessed on 31 December 2021)]; Available online: https://www.hiv.gov/hiv-basics/overview/data-and-trends/global-statistics.
- 143.Collins D.R., Gaiha G.D., Walker B.D. CD8+ T cells in HIV control, cure and prevention. Nat. Rev. Immunol. 2020;20:471–482. doi: 10.1038/s41577-020-0274-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Devadas K., Dhawan S. Hemin Activation Ameliorates HIV-1 Infection via Heme Oxygenase-1 Induction. J. Immunol. 2006;176:4252–4257. doi: 10.4049/jimmunol.176.7.4252. [DOI] [PubMed] [Google Scholar]
- 145.Cross S.A., Cook D.R., Chi A.W.S., Vance P.J., Kolson L.L., Wong B.J., Jordan-Sciutto K.L., Kolson D.L. Dimethyl fumarate, an immune modulator and inducer of the antioxidant response, suppresses HIV replication and macrophage-mediated neurotoxicity: A novel candidate for HIV neuroprotection. J. Immunol. 2011;187:5015–5025. doi: 10.4049/jimmunol.1101868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Kovacsics C.E., Gill A.J., Ambegaokar S.S., Gelman B.B., Kolson D.L. Degradation of heme oxygenase-1 by the immunoproteasome in astrocytes: A potential interferon-γ-dependent mechanism contributing to HIV neuropathogenesis. Glia. 2017;65:1264–1277. doi: 10.1002/glia.23160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Gill A.J., Kovacsics C.E., Cross S.A., Vance P.J., Kolson L.L., Jordan-Sciutto K.L., Gelman B.B., Kolson D.L. Heme oxygenase-1 deficiency accompanies neuropathogenesis of HIV-associated neurocognitive disorders. J. Clin. Investig. 2014;124:4459–4472. doi: 10.1172/JCI72279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Malvy D., McElroy A.K., de Clerck H., Günther S., van Griensven J. Ebola virus disease. Lancet. 2019;393:936–948. doi: 10.1016/S0140-6736(18)33132-5. [DOI] [PubMed] [Google Scholar]
- 149.Ascenzi P., Bocedi A., Heptonstall J., Capobianchi M.R., Di Caro A., Mastrangelo E., Bolognesi M., Ippolito G. Ebolavirus and Marburgvirus: Insight the Filoviridae family. Mol. Aspects Med. 2008;29:151–185. doi: 10.1016/j.mam.2007.09.005. [DOI] [PubMed] [Google Scholar]
- 150.Huang H., Konduru K., Solovena V., Zhou Z.-H., Kumari N., Takeda K., Nekhai S., Bavari S., Kaplan G.G., Yamada K.M., et al. Therapeutic potential of the heme oxygenase-1 inducer hemin against Ebola virus infection. Curr. Trends Immunol. 2016;17:117–123. [PMC free article] [PubMed] [Google Scholar]
- 151.Hill-Batorski L., Halfmann P., Neumann G., Kawaoka Y. The cytoprotective enzyme heme oxygenase-1 suppresses Ebola virus replication. J. Virol. 2013;87:13795–13802. doi: 10.1128/JVI.02422-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Basler C.F., Mikulasova A., Martinez-Sobrido L., Paragas J., Mühlberger E., Bray M., Klenk H.-D., Palese P., García-Sastre A. The Ebola Virus VP35 Protein Inhibits Activation of Interferon Regulatory Factor 3. J. Virol. 2003;77:7945–7956. doi: 10.1128/JVI.77.14.7945-7956.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Lehmann E., El-Tantawy W.H., Ocker M., Bartenschlager R., Lohmann V., Hashemolhosseini S., Tiegs G., Sass G. The heme oxygenase 1 product biliverdin interferes with hepatitis C virus replication by increasing antiviral interferon response. Hepatology. 2010;51:398–404. doi: 10.1002/hep.23339. [DOI] [PubMed] [Google Scholar]
- 154.Preciado M.V., Valva P., Escobar-Gutierrez A., Rahal P., Ruiz-Tovar K., Yamasaki L., Vazquez-Chacon C., Martinez-Guarneros A., Carpio-Pedroza J.C., Fonseca-Coronado S., et al. Hepatitis C virus molecular evolution: Transmission, disease progression and antiviral therapy. World J. Gastroenterol. 2014;20:15992–16013. doi: 10.3748/wjg.v20.i43.15992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Abdalla M.Y., Britigan B.E., Wen F., Icardi M., McCormick M.L., LaBrecque D.R., Voigt M., Brown K.E., Schmidt W.N. Down-Regulation of Heme Oxygenase-1 by Hepatitis C Virus Infection In Vivo and by the In Vitro Expression of Hepatitis C Core Protein. J. Infect. Dis. 2004;190:1109–1118. doi: 10.1086/423488. [DOI] [PubMed] [Google Scholar]
- 156.Zhu Z., Wilson A.T., Mathahs M.M., Wen F., Brown K.E., Luxon B.A., Schmidt W.N. Heme oxygenase-1 suppresses hepatitis C virus replication and increases resistance of hepatocytes to oxidant injury. Hepatology. 2008;48:1430–1439. doi: 10.1002/hep.22491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Fillebeen C., Rivas-Estilla A.M., Bisaillon M., Ponka P., Muckenthaler M., Hentze M.W., Koromilas A.E., Pantopoulos K. Iron inactivates the RNA polymerase NS5B and suppresses subgenomic replication of hepatitis C virus. J. Biol. Chem. 2005;280:9049–9057. doi: 10.1074/jbc.M412687200. [DOI] [PubMed] [Google Scholar]
- 158.Zhu Z., Wilson A.T., Luxon B.A., Brown K.E., Mathahs M.M., Bandyopadhyay S., McCaffrey A.P., Schmidt W.N. Biliverdin inhibits hepatitis C virus nonstructural 3/4A protease activity: Mechanism for the antiviral effects of heme oxygenase? Hepatology. 2010;52:1897–1905. doi: 10.1002/hep.23921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Law Y.-Y., Lee W.-F., Hsu C.-J., Lin Y.-Y., Tsai C.-H., Huang C.-C., Wu M.-H., Tang C.-H., Liu J.-F. miR-let-7c-5p and miR-149-5p inhibit proinflammatory cytokine production in osteoarthritis and rheumatoid arthritis synovial fibroblasts. Aging. 2021;13:17227–17236. doi: 10.18632/aging.203201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Chen W.C., Wei C.K., Lee J.C. MicroRNA-let-7c suppresses hepatitis C virus replication by targeting Bach1 for induction of haem oxygenase-1 expression. J. Viral Hepat. 2019;26:655–665. doi: 10.1111/jvh.13072. [DOI] [PubMed] [Google Scholar]
- 161.Shen Y.M., Zhang H.L., Wu Y.H., Yu X.P., Hu H.J., Dai L.H. Dynamic correlation between induction of the expression of heme oxygenase-1 and hepatitis B viral replication. Mol. Med. Rep. 2015;11:4706–4712. doi: 10.3892/mmr.2015.3278. [DOI] [PubMed] [Google Scholar]
- 162.Schmidt W.N., Mathahs M.M., Zhu Z. Heme and HO-1 Inhibition of HCV, HBV, and HIV. Front. Pharmacol. 2012;3:129. doi: 10.3389/fphar.2012.00129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Yeh C.-N., Wu R.-C., Cheng C.-T., Tsai C.-Y., Chang Y.-R., Yeh T.-S., Wu T.-H., Lee W.-C., Chiang K.-C. HO-1 is a favorable prognostic factor for HBV-HCC patients who underwent hepatectomy. Cancer Manag. Res. 2018;10:6049–6059. doi: 10.2147/CMAR.S186931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Murugesan A., Manoharan M. Emerging and Reemerging Viral Pathogens: Volume 1: Fundamental and Basic Virology Aspects of Human, Animal and Plant Pathogens. Elsevier; Amsterdam, The Netherlands: 2019. Dengue virus; pp. 281–359. [Google Scholar]
- 165.Olagnier D., Peri S., Steel C., Van Montfoort N., Chiang C. Cellular Oxidative Stress Response Controls the Antiviral and Apoptotic Programs in Dengue Virus-Infected Dendritic Cells. PLoS Pathog. 2014;10:1004566. doi: 10.1371/journal.ppat.1004566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Tseng C.-K., Lin C.-K., Wu Y.-H., Chen Y.-H., Chen W.-C., Young K.-C., Lee J.-C. Human heme oxygenase 1 is a potential host cell factor against dengue virus replication. Sci. Rep. 2016;6:32176. doi: 10.1038/srep32176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Su Y.-C., Huang Y.-F., Wu Y.-W., Chen H.-F., Wu Y.-H., Hsu C.-C., Hsu Y.-C., Lee J.-C. MicroRNA-155 inhibits dengue virus replication by inducing heme oxygenase-1-mediated antiviral interferon responses. FASEB J. 2020;34:7283–7294. doi: 10.1096/fj.201902878R. [DOI] [PubMed] [Google Scholar]
- 168.El Kalamouni C., Frumence E., Bos S., Turpin J., Nativel B., Harrabi W., Wilkinson D.A., Meilhac O., Gadea G., Desprès P., et al. Subversion of the Heme Oxygenase-1 Antiviral Activity by Zika Virus. Viruses. 2018;11:2. doi: 10.3390/v11010002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Suazo P.A., Tognarelli E.I., Kalergis A.M., González P.A. Herpes simplex virus 2 infection: Molecular association with HIV and novel microbicides to prevent disease. Med. Microbiol. Immunol. 2015;204:161–176. doi: 10.1007/s00430-014-0358-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Ibáñez F.J., Farías M.A., Retamal-Díaz A., Espinoza J.A., Kalergis A.M., González P.A. Pharmacological Induction of Heme Oxygenase-1 Impairs Nuclear Accumulation of Herpes Simplex Virus Capsids upon Infection. Front. Microbiol. 2017;8:2108. doi: 10.3389/fmicb.2017.02108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Palacios G., Oberste M.S. Enteroviruses as agents of emerging infectious diseases. J. Neurovirol. 2005;11:424–433. doi: 10.1080/13550280591002531. [DOI] [PubMed] [Google Scholar]
- 172.Tung W.-H., Hsieh H.-L., Lee I.-T., Yang C.-M. Enterovirus 71 induces integrin β1/EGFR-Rac1-dependent oxidative stress in SK-N-SH cells: Role of HO-1/CO in viral replication. J. Cell. Physiol. 2011;226:3316–3329. doi: 10.1002/jcp.22677. [DOI] [PubMed] [Google Scholar]
- 173.Chen Y., Liu Q., Guo D. Emerging coronaviruses: Genome structure, replication, and pathogenesis. J. Med. Virol. 2020;92:418–423. doi: 10.1002/jmv.25681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Lechuga G.C., Souza-Silva F., Sacramento C.Q., Trugilho M.R.O., Valente R.H., Napoleão-Pêgo P., Dias S.S.G., Fintelman-Rodrigues N., Temerozo J.R., Carels N., et al. SARS-CoV-2 Proteins Bind to Hemoglobin and Its Metabolites. Int. J. Mol. Sci. 2021;22:9035. doi: 10.3390/ijms22169035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Maestro S., Córdoba K.M., Olague C., Argemi J., Ávila M.A., González-Aseguinolaza G., Smerdou C., Fontanellas A. Heme oxygenase-1 inducer hemin does not inhibit SARS-CoV-2 virus infection. Biomed. Pharmacother. 2021;137:111384. doi: 10.1016/j.biopha.2021.111384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Kim D.-H., Ahn H.-S., Go H.-J., Kim D.-Y., Kim J.-H., Lee J.-B., Park S.-Y., Song C.-S., Lee S.-W., Ha S.-D., et al. Hemin as a novel candidate for treating COVID-19 via heme oxygenase-1 induction. Sci. Rep. 2021;11:21462. doi: 10.1038/s41598-021-01054-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Ortiz E., Sanchis P., Bizzotto J., Lage-Vickers S., Labanca E., Navone N., Cotignola J., Vazquez E., Gueron G. Myxovirus Resistance Protein 1 (MX1), a Novel HO-1 Interactor, Tilts the Balance of Endoplasmic Reticulum Stress towards Pro-Death Events in Prostate Cancer. Biomolecules. 2020;10:1005. doi: 10.3390/biom10071005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Müller B., Kräusslich H.-G. Antiviral strategies. Handb. Exp. Pharmacol. 2009;189:1–24. doi: 10.1007/978-3-540-79086-0_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Baillie J.K., Digard P. Influenza—Time to Target the Host? N. Engl. J. Med. 2013;369:191–193. doi: 10.1056/NEJMcibr1304414. [DOI] [PubMed] [Google Scholar]
- 180.Brown J., Zhou J., Peacock T.P., Barclay W.S. The SARS-CoV-2 Variant, Omicron, Shows Enhanced Replication in Human Primary Nasal Epithelial Cells. [(accessed on 30 December 2021)]; Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1043652/S1454_Omicron_report_20.12_Imperial.pdf.
- 181.Bentley E.G., Kirby A., Sharma P., Kipar A., Mega D.F., Bramwell C., Penrice-Randal R., Prince T., Brown J.C., Zhou J., et al. SARS-CoV-2 Omicron-B.1.1.529 Variant leads to less severe disease than Pango B and Delta variants strains in a mouse model of severe COVID-19. bioRxiv. 2021 doi: 10.1101/2021.12.26.474085. [DOI] [Google Scholar]
- 182.Sheraton M., Deo N., Kashyap R., Surani S. A Review of Neurological Complications of COVID-19. Cureus. 2020;12:e8192. doi: 10.7759/cureus.8192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Manjili Rose Manjili H.H., Zarei M., Habibi M. COVID-19 as an Acute Inflammatory Disease. J. Immunol Ref. 2021;205:12–19. doi: 10.4049/jimmunol.2000413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Batra N., De Souza C., Batra J., Raetz A.G., Yu A.-M. The HMOX1 Pathway as a Promising Target for the Treatment and Prevention of SARS-CoV-2 of 2019 (COVID-19) Int. J. Mol. Sci. 2020;21:6412. doi: 10.3390/ijms21176412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Zhu Y., Sun Y., Jin K., Greenberg D.A. Hemin induces neuroglobin expression in neural cells. Blood. 2002;100:2494–2498. doi: 10.1182/blood-2002-01-0280. [DOI] [PubMed] [Google Scholar]
- 186.Li H.M., Shi Y.L., Wen D., Luo H.M., Lin X., Xiao F. A novel effective chemical hemin for the treatment of acute carbon monoxide poisoning in mice. Exp. Ther. Med. 2017;14:5186–5192. doi: 10.3892/etm.2017.5157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Lu X., Chen-Roetling J., Regan R.F. Systemic hemin therapy attenuates blood-brain barrier disruption after intracerebral hemorrhage. Neurobiol. Dis. 2014;70:245–251. doi: 10.1016/j.nbd.2014.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Olagnier D., Farahani E., Thyrsted J., Blay-Cadanet J., Herengt A., Idorn M., Hait A., Hernaez B., Knudsen A., Iversen M.B., et al. SARS-CoV2-mediated suppression of NRF2-signaling reveals potent antiviral and anti-inflammatory activity of 4-octyl-itaconate and dimethyl fumarate. Nat. Commun. 2020;11:4938. doi: 10.1038/s41467-020-18764-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Cuadrado A., Pajares M., Benito C., Jiménez-Villegas J., Escoll M., Fernández-Ginés R., Garcia Yagüe A.J., Lastra D., Manda G., Rojo A.I., et al. Can Activation of NRF2 Be a Strategy against COVID-19? Trends Pharmacol. Sci. 2020;41:598–610. doi: 10.1016/j.tips.2020.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Gupta A., Madhavan M.V., Sehgal K., Nair N., Mahajan S., Sehrawat T.S., Bikdeli B., Ahluwalia N., Ausiello J.C., Wan E.Y., et al. Extrapulmonary manifestations of COVID-19. Nat. Med. 2020;26:1017–1032. doi: 10.1038/s41591-020-0968-3. [DOI] [PubMed] [Google Scholar]
- 191.Kaiser S., Frase S., Selzner L., Lieberum J.L., Wollborn J., Niesen W.D., Foit N.A., Heiland D.H., Schallner N. Neuroprotection after hemorrhagic stroke depends on cerebral heme oxygenase-1. Antioxidants. 2019;8:496. doi: 10.3390/antiox8100496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Bijjem K.R.V., Padi S.S.V., lal Sharma P. Pharmacological activation of heme oxygenase (HO)-1/carbon monoxide pathway prevents the development of peripheral neuropathic pain in Wistar rats. Naunyn. Schmiedebergs. Arch. Pharmacol. 2013;386:79–90. doi: 10.1007/s00210-012-0816-1. [DOI] [PubMed] [Google Scholar]
- 193.Singh R.D., Barry M.A., Croatt A.J., Ackerman A.W., Grande J.P., Diaz R.M., Vile R.G., Agarwal A., Nath K.A. The spike protein of SARS-CoV-2 virus induces heme oxygenase-1: Pathophysiologic implications. Biochim. Biophys. Acta Mol. Basis Dis. 2021;1868:166322. doi: 10.1016/j.bbadis.2021.166322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Nath K.A. Heme oxygenase-1 and acute kidney injury. Curr. Opin. Nephrol. Hypertens. 2014;23:17–24. doi: 10.1097/01.mnh.0000437613.88158.d3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Atsaves V., Makri P., Detsika M.G., Tsirogianni A., Lianos E.A. Glomerular epithelial cells-targeted heme oxygenase-1 over expression in the rat: Attenuation of proteinuria in secondary but not primary injury. Nephron. 2016;133:270–278. doi: 10.1159/000445755. [DOI] [PubMed] [Google Scholar]
- 196.Yeh Y.H., Hsu L.A., Chen Y.H., Kuo C.T., Chang G.J., Chen W.J. Protective role of heme oxygenase-1 in atrial remodeling. Basic Res. Cardiol. 2016;111:58. doi: 10.1007/s00395-016-0577-y. [DOI] [PubMed] [Google Scholar]
- 197.Giannini L., Vannacci A., Fabrizi F., Uliva C., Bani D., Masini E., Mannaioni P.F. Protection from cardiac injury by induction of heme oxygenase-1 and nitric oxide synthase in a focal ischaemia-reperfusion model. Cell Mol. Biol. 2005;51:393–401. [PubMed] [Google Scholar]
- 198.Farombi E.O., Shrotriya S., Na H.-K., Kim S.-H., Surh Y.-J. Curcumin attenuates dimethylnitrosamine-induced liver injury in rats through Nrf2-mediated induction of heme oxygenase-1. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2008;46:1279–1287. doi: 10.1016/j.fct.2007.09.095. [DOI] [PubMed] [Google Scholar]
- 199.Devey L., Ferenbach D., Mohr E., Sangster K., Bellamy C.O., Hughes J., Wigmore S.J. Tissue-resident macrophages protect the liver from ischemia reperfusion injury via a heme oxygenase-1-dependent mechanism. Mol. Ther. 2009;17:65–72. doi: 10.1038/mt.2008.237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Ptilovanciv E.O.N., Fernandes G.S., Teixeira L.C., Reis L.A., Pessoa E.A., Convento M.B., Simões M.J., Albertoni G.A., Schor N., Borges F.T. Heme oxygenase 1 improves glucoses metabolism and kidney histological alterations in diabetic rats. Diabetol. Metab. Syndr. 2013;5:3–8. doi: 10.1186/1758-5996-5-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Dolan M.E., Hill D.P., Mukherjee G., McAndrews M.S., Chesler E.J., Blake J.A. Investigation of COVID-19 comorbidities reveals genes and pathways coincident with the SARS-CoV-2 viral disease. Sci. Rep. 2020;10:20848. doi: 10.1038/s41598-020-77632-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Durante W. Targeting heme oxygenase-1 in vascular disease. Curr. Drug Targets. 2010;11:1504–1516. doi: 10.2174/1389450111009011504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Tracz M.J., Juncos J.P., Grande J.P., Croatt A.J., Ackerman A.W., Katusic Z.S., Nath K.A. Induction of heme oxygenase-1 is a beneficial response in a murine model of venous thrombosis. Am. J. Pathol. 2008;173:1882–1890. doi: 10.2353/ajpath.2008.080556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Arngrim N., Schytz H.W., Hauge M.K., Ashina M., Olesen J. Carbon monoxide may be an important molecule in migraine and other headaches. Cephalalgia. 2014;34:1169–1180. doi: 10.1177/0333102414534085. [DOI] [PubMed] [Google Scholar]
- 205.Hervera A., Leánez S., Negrete R., Motterlini R., Pol O. Carbon monoxide reduces neuropathic pain and spinal microglial activation by inhibiting nitric oxide synthesis in mice. PLoS ONE. 2012;7:e43693. doi: 10.1371/journal.pone.0043693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Chen-Roetling J., Song W., Schipper H.M., Regan C.S., Regan R.F. Astrocyte Overexpression of Heme Oxygenase-1 Improves Outcome after Intracerebral Hemorrhage. Stroke. 2015;46:1093–1098. doi: 10.1161/STROKEAHA.115.008686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Lu X., Gu R., Hu W., Sun Z., Wang G., Wang L., Xu Y. Upregulation of heme oxygenase-1 protected against brain damage induced by transient cerebral ischemia-reperfusion injury in rats. Exp. Ther. Med. 2018;15:4629–4636. doi: 10.3892/etm.2018.6049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Uhlén M., Fagerberg L., Hallström B.M., Lindskog C., Oksvold P., Mardinoglu A., Sivertsson Å., Kampf C., Sjöstedt E., Asplund A., et al. Tissue-based map of the human proteome. Science. 2015;347:6220. doi: 10.1126/science.1260419. [DOI] [PubMed] [Google Scholar]
- 209.Pushpakom S., Iorio F., Eyers P.A., Escott K.J., Hopper S., Wells A., Doig A., Guilliams T., Latimer J., McNamee C., et al. Drug repurposing: Progress, challenges and recommendations. Nat. Rev. Drug Discov. 2018;18:41–58. doi: 10.1038/nrd.2018.168. [DOI] [PubMed] [Google Scholar]