Abstract
Stenotrophomonas maltophilia is increasingly recognized as a nosocomial bacterial pathogen with a multi-drug resistance profile. In this study, the novel drug gepotidacin, the first compound of the novel triazaacenaphthylene topoisomerase inhibitor antibiotics class, was evaluated on its activity against clinical S. maltophilia isolates. Ninety-nine S. maltophilia isolates plus reference strain K279a (N = 100) were tested on their susceptibility towards gepotidacin in a broth microdilution. Additional susceptibility testing was performed towards the commonly applied combination trimethoprim/sulfamethoxazole (TMP/SXT), moxifloxacin, and levofloxacin. The time–kill kinetic of gepotidacin was observed in a time–kill assay. The greater wax moth Galleria mellonella was used to determine the activity of gepotidacin against S. maltophilia in vivo. Gepotidacin showed minimum inhibitory concentrations (MICs) between 0.25 and 16 mg/L (MIC50: 2 mg/L; MIC90: 8 mg/L), independently of its susceptibility towards TMP/SXT. The five TMP/SXT resistant strains exhibited gepotidacin MICs from 1 to 4 mg/L. The S. maltophilia strains resistant to the assessed fluoroquinolones showed in parts high MICs of gepotidacin. The time–kill assay revealed a time- and strain-dependent killing effect of gepotidacin. In vivo, injection of gepotidacin increased the survival rate of the larvae from 61 % to 90 % after 2 days. This study showed antimicrobial effects of gepotidacin towards S. maltophilia.
Keywords: Stenotrophomonas maltophilia, gepotidacin, triazaacenaphthylene, topoisomerase inhibitor, Galleria mellonella, cystic fibrosis
1. Introduction
Stenotrophomonas maltophilia is a non-fermentative, Gram-negative, opportunistic, ubiquitous bacterium. While it is known to occur in water and soil, it is also known to cause a broad range of infections in humans [1,2,3]. In particular, nosocomial infections with S. maltophilia are linked to a considerable mortality with a rate of 37.5 % [4]. The most frequently occurring infections with S. maltophilia are infections of the respiratory tract [5,6,7] and bacteremia [8,9].
Due to its elevated level of intrinsic resistances towards multiple classes of commonly applied antibiotics, e.g., carbapenems and aminoglycosides, and the description of acquired antimicrobial resistance mechanisms, e.g., against fluoroquinolones, and/or polymyxins, S. maltophilia is considered a human pathogen with a multi-drug resistance profile [1,10,11,12]. In addition to the reports on nosocomial infections in hospitalized and/or immunosuppressed patients, the chronic colonization of the respiratory tract of cystic fibrosis (CF) patients is also reported. With rates of ~10 %, colonization with S. maltophilia is an independent risk factor for pulmonary exacerbation of CF patients, associated with a decline in lung function and a worse outcome [13,14,15,16].
Today, the main therapeutic choice for infections with S. maltophilia is trimethoprim/sulfamethoxazole (co-trimoxazole, TMP/SXT) [17]. When treatment is not possible, e.g., due to resistances towards co-trimoxazole or patient’s intolerance, topoisomerase inhibitors such as ciprofloxacin or moxifloxacin as well as other antibiotics as ceftazidime and ticarcillin/clavulanate, alone or in combination with other antibiotics, may be considered for treatment [17]. However, the application of novel antibiotic classes in times of rising resistances, also towards fluoroquinolones, may become an important topic in the near future.
Gepotidacin (NBTI 5463; GSK2140944) is the first-in-class of the new bacterial topoisomerase inhibitors (NBTIs), known as triazaacenaphthylene bacterial type II topoisomerase inhibitor, showing activity against several bacterial species being resistant to fluoroquinolones [18,19,20]. In vitro data showed promising activity of gepotidacin against (methicillin-resistant) Staphylococcus aureus, Streptococcus pneumoniae, Escherichia coli, and Neisseria gonorrhoeae [21]. An ongoing clinical trial on single-dose treatment of urinary tract infection and uncomplicated urogenital gonorrhea with gepotidacin resulted in a 95 % efficiency against N. gonorrhoeae [22]. Gepotidacin inhibits the bacterial DNA-gyrase as well as the topoisomerase IV, using a distinct mechanism compared to the fluoroquinolones.
S. maltophilia’s multi-drug resistance together with increasing reports on nosocomial infections make S. maltophilia an emerging pathogen [23]. The aim of this study was to investigate the capability of gepotidacin to inhibit S. maltophilia growth in vitro. Additionally, the activity of gepotidacin against S. maltophilia infection was studied in vivo using the alternative infection model Galleria mellonella.
2. Results
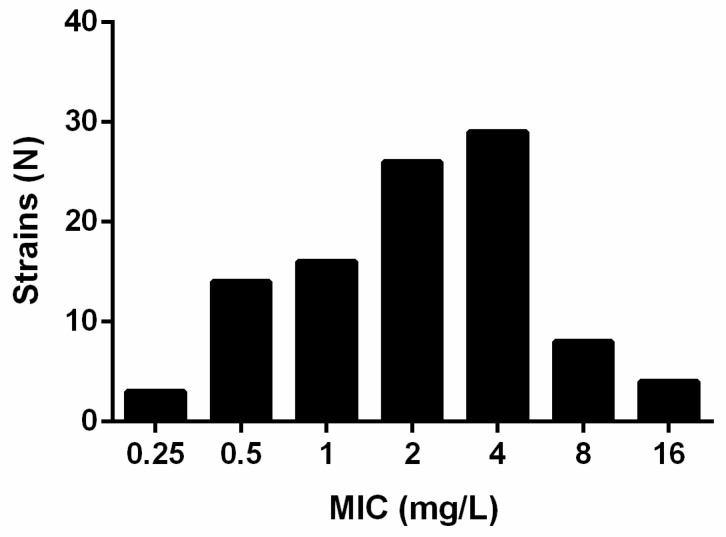
In vitro, gepotidacin showed a strain-dependent activity towards the here included isolates with minimum inhibitory concentrations (MICs) ranging from 0.25 to 16 mg/L (Figure 1).
Figure 1.
MIC (mg/L) distribution of N = 100 Stenotrophomonas maltophilia strains for gepotidacin as found in broth microdilution after EUCAST.
The MIC50 and MIC90 were determined to be 2 and 8 mg/L, respectively. The MIC90 of the fluoroquinolones (N = 100) were 4 mg/L for levofloxacin and 2 mg/L for moxifloxacin (Table 1).
Table 1.
Determined minimum inhibitory concentrations (MICs) of gepotidacin, levofloxacin, moxifloxacin, and co-trimoxazole against S. maltophilia.
| Agent | N | Method | MIC (mg/L) | ||
|---|---|---|---|---|---|
| Range | MIC50 | MIC90 | |||
| Gepotidacin | 100 | Microdilution | 0.25–16 | 2 | 8 |
| Levofloxacin | 100 | Microdilution | 0.25–>16 | 1 | 4 |
| Moxifloxacin | 100 | Microdilution | ≤0.003–>16 | 0.5 | 2 |
| Co-trimoxazole | 100 | MIC test strips | 0.016–>32 | 0.19 | >32 |
Susceptibility testing by MIC test strips revealed 15 (15 %) co-trimoxazole resistant strains. MICs ranged from 0.016 to >32 mg/L and the determined MIC50 and MIC90 were 0.19 and >32 mg/L, respectively.
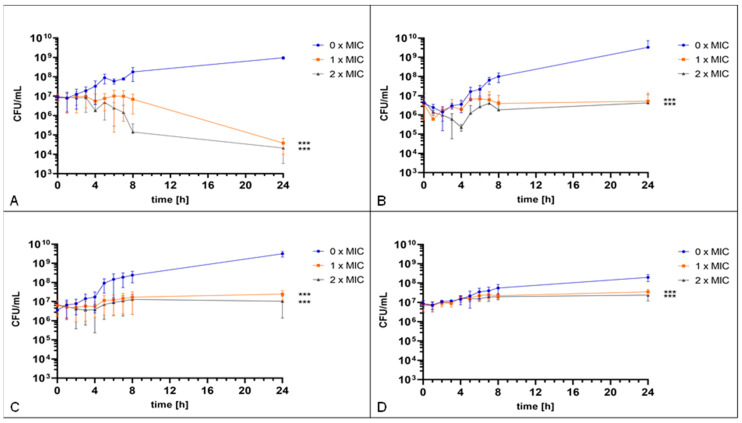
For further analysis, four strains (two wild type (K279a, Sm 538) and two co-trimoxazole resistant isolates (Sm 290, Sm 1222)) were included in a time–kill assay, determining the antimicrobial effect of gepotidacin against S. maltophilia over time. A significant growth inhibiting effect of two-fold of the MIC has been observed for each of the strains. However, the impact on viability of S. maltophilia varied between the different strains. For reference strain K279a, a significant reduction of ~4.5 log colony forming units (CFU)/mL was detected after 24 h of incubation compared to the non-treated control (Figure 2A). For two other strains, Sm 290 and Sm 538, after a reduction of 1.7 log and 2.5 log CFU/mL, respectively, compared to the non-treated control, a regrowth effect could be detected (Figure 2B,C). For the fourth strain, Sm 1222, characterized by slow growing behavior, the reduction in CFU was less with only 0.9 log CFU/mL after 24 h of incubation with gepotidacin when compared to the growth control (Figure 2D).
Figure 2.
Time–kill assay. The reference strain K279a (A) and Sm 538 (C) were incubated with 0 mg/L, 4 mg/L, and 8 mg/L gepotidacin. The strain Sm 290 (B) was incubated with 0 mg/L, 1 mg/L, and 2 mg/L gepotidacin, while the strain Sm 1222 (D) was incubated with 0 mg/L, 2 mg/L, and 4 mg/L gepotidacin. CFU/mL were determined hourly over the first eight hours and after 24 h. Observed growth after incubation without gepotidacin (blue), with one-fold MIC (orange) and two-fold (grey) gepotidacin. Significance levels are shown by asterisks: ***: p < 0.001 and determined via two-way ANOVA using GraphPad Prism.
These in vitro results suggest an antimicrobial effect of gepotidacin against S. maltophilia in a strain-dependent manner.
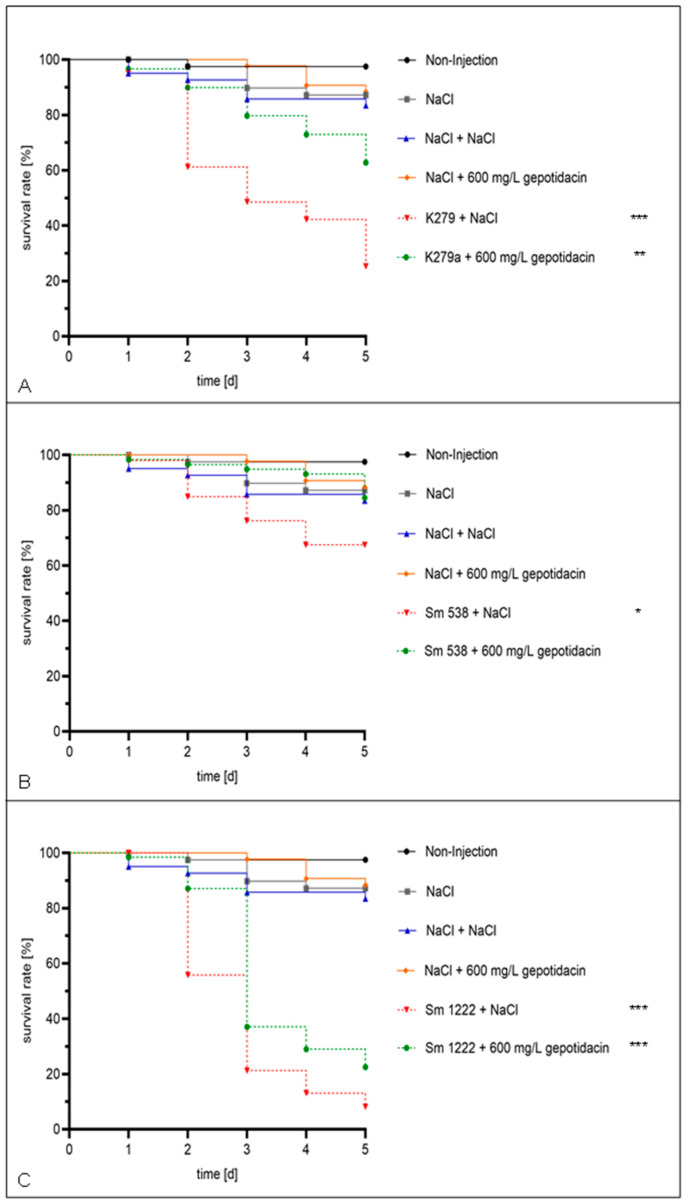
A first evaluation of gepotidacin toxicity in G. mellonella revealed no death larvae when injected in concentrations of up to 32 mg/L. Additionally, the DMSO used for the solution of gepotidacin showed no toxicity in the tested concentrations of up to 10 % (data not shown). Virulence analysis revealed a strain specific virulence, with comparable high virulence of strain Sm 1222, moderate virulence of K279a and Sm 538, as well as low virulence of strain Sm 290. In the following, the latter was excluded from further in vivo antimicrobial activity testing with gepotidacin. Gepotidacin injection of the larvae four hours after infection with S. maltophilia in a concentration of 600 mg/L, equaling a dose of 20 mg/Kg body weight, resulted in an overall survival rate of 89.9 % after 2 d in comparison to the NaCl control group with 60.6 % (p = 0.0604). The effect of gepotidacin is strain dependent and more significant in the early phase of infection. After a monitoring period of 5 days, 53.1 % of the infected and treated larvae survived in comparison to the infected NaCl-control which resulted in 28.4 % survival (p = 0.073). Strain specific survival curves are depicted in Figure 3.
Figure 3.
Single dose effect. The larvae were injected with a concentration of 105 cells/mL and treated after 4 h with NaCl or 600 mg/L gepotidacin, respectively. Double-treatment controls with double injection of NaCl or gepotidacin were set up. The larvae were followed-up for five days. Showing the survival rate of the larvae, which were infected with K279a (A), Sm 538 (B), and Sm 1222 (C). Significance levels are shown by asterisks: *: p < 0.05, **: p < 0.005, ***: p < 0.001 and were determined via logrank test using GraphPad Prism.
3. Discussion
We here demonstrated, for the first time, the antimicrobial activity of gepotidacin against S. maltophilia in an in vitro and in vivo study.
The here determined MICs of gepotidacin towards the tested S. maltophilia strains are comparable to those reported previously for Gram-negative bacteria with a MIC50 of 2 mg/L [21].
Comparing the MICs of gepotidacin against wild-type and co-trimoxazole resistant strains, higher MICs were found for those strains which were resistant towards co-trimoxazole. While, for co-trimoxazole wild-type strains, the MIC50 was 1 mg/L and the MIC90 4 mg/L, the MIC50 and MIC90 of the co-trimoxazole resistant strains were higher with 2 and 16 mg/L, respectively. However, a correlation of these data could not be identified. Comparable results have been shown for strains resistant to at least one of the tested fluoroquinolones (MIC90: 16 mg/L), exhibiting higher MICs compared to fluroquinolone susceptible strains (MIC90: 2 mg/L). These findings are congruent to data from Biedenbach et al. showing distinct MICs of gepotidacin between levofloxacin-susceptible and resistant E. coli strains [24]. In another study, no association could be made between resistances towards different antibiotics including quinolones and increased MICs of gepotidacin [21].
While co-trimoxazole inhibits the synthesis of folic acid, fluoroquinolones target the bacterial topoisomerase II. Gepotidacin also affects the bacterial topoisomerases; thus, a link between co-trimoxazole resistance and high MICs of gepotidacin was not expected, whereas a link between elevated levofloxacin MICs and high gepotidacin MICs can be assumed.
Subsequently performed time–kill assays revealed an antibacterial activity of gepotidacin. Both, the MIC as well as the two-fold MIC showed a growth inhibiting effect over time on each of the tested strains. However, a strain-dependent killing with some strains showing a regrowth effect has been detected. Comparing strain K279a and strain Sm 538, both with similar MICs towards each of the tested agents, the antimicrobial effect of gepotidacin on K279a was stronger. A link between the susceptibility pattern and the strength of gepotidacin effect was thus excluded here. Strain-dependent differences in growth have also been reported for N. gonorrhoeae treated with gepotidacin in other studies [22].
The regrowth effect documented here for strain Sm 290 has also been described previously for S. aureus treated with gepotidacin, a decrease in viable cell count was followed by an increase, resulting in cell counts after 24 h like the concentration of time point at inoculation [21,25]. Lahiri et al. found a spontaneous mutation of the gyrase-subunit, enabling the regrowth of S. aureus after treatment with gepotidacin [25]. It is not to be excluded that a similar mutation might occur as well in some of the here assessed S. maltophilia isolates.
One of the here observed strains (Sm 1222) was characterized as slow growing. This strain revealed the least reduction in viable cell count compared to the other assessed strains with a reduction of 0.45 log CFU/mL after 24 h treatment with one-fold MIC (2 mg/L) in comparison to the non-treated control. It is suggested that the slow growth is indicating a less active metabolism which is responsible for the detected poor activity of gepotidacin in this time-kill assay. Each of the tested strains was not fully eradicated in the observed time period by giving the MIC or the two-fold MIC of gepotidacin.
We here demonstrated an activity of gepotidacin in vivo using the invertebrate infection model G. mellonella. A first test on virulence of the analyzed strains revealed a strain-dependent killing effect in the larvae. Strain Sm 1222 was characterized with a slow growing behavior, but virulence of this strain was high compared to the other strains, resulting in 0 % survival 6 days post inoculation. Gepotidacin showed no toxicity in G. mellonella. The tested concentration was previously safely examined in a rat pyelonephritis model [26]. Furthermore, Barth et al. assessed the safety and systemic exposure of gepotidacin in healthy adults as well as in adolescents. Their results promote an acceptable risk–safety profile in humans for gepotidacin [27].
In this in vivo model, larvae were infected with S. maltophilia and gepotidacin was injected 4 h post infection in a concentration equaling 20 mg/kg bodyweight. Larvae infected with each of the strains and subsequent injection of gepotidacin showed a significant higher survival compared to the control group. Even though G. mellonella is a suitable model for drug testing, further in vivo studies to determine gepotidacin activity against S. maltophilia should be performed, e.g., potentially in murine models.
4. Materials and Methods
4.1. Strains
All clinical samples were subject to conventional microbiological diagnosis before use. The study did not use demographic data about patients, nor did it result in any additional constraints for the patients. Because of the retrospective nature of the study, all data were anonymously analyzed without the need for patient consent. All procedures and methods were carried out in accordance with approved guidelines.
This study involved a total of 100 S. maltophilia strains; among them were 91 clinical and eight environmental strains plus the reference strain K279a. The clinical strains were isolated from respiratory specimens of CF patients (N = 70) and non-CF patients (N = 21). Species were identified by MALDI-TOF-MS (VITEK MS, bioMérieux, Nürtingen, Germany).
4.2. Susceptibility Testing
Susceptibility testing was performed for each included strain in a broth microdilution after EUCAST (European Committee on Antimicrobial Susceptibility Testing) guidelines, determining the MIC of gepotidacin towards each strain. Additionally, MICs of 100 strains towards levofloxacin and moxifloxacin were assessed in a broth microdilution. The anti-infective agents were diluted in solvent (dimethyl sulfoxide, DMSO) and Mueller Hinton Bouillon 2 for the preparation of stock solutions. Working solutions were prepared by dilution in Mueller Hinton Bouillon 2 and all solutions were stored at −20°C. Broth microdilution with gepotidacin, levofloxacin and moxifloxacin (MedChemExpress LLC, Monmouth, NJ, USA) was performed in concentrations between 0.03 and 16 mg/L. Additionally, MIC strip tests (Liofilchem srl, Roseto degli Abruzzi, Italy) were performed for co-trimoxazole for a total of 100 strains.
4.3. Time–Kill Assay
To determine the antimicrobial activity of gepotidacin in dependence to incubation time, a time–kill assay including four clinical strains, among them K279a, was performed. Viability of S. maltophilia was assessed hourly over 8 h and additionally checked after 24 h when incubated with gepotidacin in concentrations of previously identified MIC as well as the two-fold MIC. A respected growth control, not treated with gepotidacin, accompanied the tests. Incubation was performed in a microtiter plate and an inoculum of 1 × 107 cells/mL in Mueller Hinton Broth 2 was prepared. Viability was determined by estimation of CFU/mL. Therefore, appropriate dilutions of the cell suspension were plated on Columbia Sheep Blood agar plates and incubated at 36 °C, until countable colonies were visible.
4.4. In Vivo Infection Assay
For the determination of gepotidacin activity against S. maltophilia infection in vivo, we infected the larvae of the greater wax moth G. mellonella with four different S. maltophilia strains. Infection was performed as with an inoculum of 1 × 105 cells/mL in NaCl. In each run, each sample was injected into 15 larvae. As controls, a non-injection as well as a NaCl control was run additionally. After infection, larvae were incubated at 37 °C for 4 h before the animals were injected with 600 mg/L gepotidacin, equaling 20 mg/kg body weight according to the clinical trials [28]. Larvae were assessed daily on their survival. Dead larvae as well as pupae were noted respectively and removed from the experiment. Survival analysis after Kaplan–Meier was processed using GraphPad Prism8 (GraphPad Inc., La Jolla, CA, USA).
4.5. Statistics
All experiments were performed in triplicates. Statistical analysis was done in GraphPad Prism8. Significance levels are shown by asterisks: *: p < 0.05, **: p < 0.005, ***: p < 0.001. The applied statistical test is indicated in the corresponding figure legends.
5. Conclusions
In conclusion, we here demonstrated for the first time an antimicrobial activity of the novel drug gepotidacin against clinical S. maltophilia isolates in an in vitro and an in vivo study. In vitro, MICs varied between 0.25 and 16 mg/L. The time–kill assay revealed a time- and strain-dependent killing effect of gepotidacin. In vivo, injection of gepotidacin increased the survival rate of the larvae from 61 % to 90 % after 2 days.
Acknowledgments
We thank Medicines for Malaria Venture (MMV) for kindly providing us the pathogen response box. We acknowledge support by the Open Access Publication Fund of the University of Duisburg-Essen.
Author Contributions
L.K. and H.L.V. conceived and designed the experiments. E.A. and M.I.S. established the time–kill assay; M.I.S. established the broth microdilution, MIC tests, and in vivo experiments. L.K., H.L.V., and P.-M.R. supervised the work. J.B. and J.S. contributed reagents/materials/analysis tools. M.I.S. and L.K. wrote the main manuscript text and M.I.S. prepared all figures. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Brooke J.S. Stenotrophomonas maltophilia: An emerging global opportunistic pathogen. Clin. Microbiol. Rev. 2012;25:2–41. doi: 10.1128/CMR.00019-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Denton M., Todd N.J., Kerr K.G., Hawkey P.M., Littlewood J.M. Molecular epidemiology of Stenotrophomonas maltophilia isolated from clinical specimens from patients with cystic fibrosis and associated environmental samples. J. Clin. Microbiol. 1998;36:1953–1958. doi: 10.1128/JCM.36.7.1953-1958.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ryan R.P., Fouhy Y., Garcia B.F., Watt S.A., Niehaus K., Yang L., Tolker-Nielsen T., Dow J.M. Interspecies signaling via the Stenotrophomonas maltophilia diffusible signal factor influences biofilm formation and polymyxin tolerance in Pseudomonas aeruginosa. Mol. Microbiol. 2008;68:75–86. doi: 10.1111/j.1365-2958.2008.06132.x. [DOI] [PubMed] [Google Scholar]
- 4.Falagas M.E., Kastoris A.C., Vouloumanou E.K., Rafailidis P.I., Kapaskelis A.M., Dimopoulos G. Attributable mortality of Stenotrophomonas maltophilia infections: A systematic review of the literature. Future Microbiol. 2009;4:1103–1109. doi: 10.2217/fmb.09.84. [DOI] [PubMed] [Google Scholar]
- 5.Nicodemo A.C., Paez J.I.G. Antimicrobial therapy for Stenotrophomonas maltophilia infections. Eur. J. Clin. Microbiol. Infect. Dis. 2007;26:229–237. doi: 10.1007/s10096-007-0279-3. [DOI] [PubMed] [Google Scholar]
- 6.Nseir S., Di Pompeo C., Cavestri B., Jozefowicz E., Nyunga M., Soubrier S., Roussel-Delvallez M., Saulnier F., Mathieu D., Durocher A. Multiple-drug-resistant bacteria in patients with severe acute exacerbation of chronic obstructive pulmonary disease: Prevalence, risk factors, and outcome. Crit. Care Med. 2006;34:2959–2966. doi: 10.1097/01.CCM.0000245666.28867.C6. [DOI] [PubMed] [Google Scholar]
- 7.Weber D.J., Rutala W.A., Sickbert-Bennett E.E., Samsa G.P., Brown V., Niederman M.S. Microbiology of ventilator–associated pneumonia compared with that of hospital-acquired pneumonia. Infect. Control Hosp. Epidemiol. 2007;28:825–831. doi: 10.1086/518460. [DOI] [PubMed] [Google Scholar]
- 8.Chang Y.-T., Lin C.-Y., Lu P.-L., Lai C.-C., Chen T.-C., Chen C.-Y., Wu D.-C., Wang T.-P., Lin C.-M., Lin W.-R., et al. Stenotrophomonas maltophilia bloodstream infection: Comparison between community-onset and hospital-acquired infections. J. Microbiol. Immunol. Infect. 2014;47:28–35. doi: 10.1016/j.jmii.2012.08.014. [DOI] [PubMed] [Google Scholar]
- 9.Lai C.-H., Chi C.-Y., Chen H.-P., Chen T.-L., Lai C.-J., Fung C.-P., Yu K.-W., Wong W.-W., Liu C.-Y. Clinical characteristics and prognostic factors of patients with Stenotrophomonas maltophilia bacteremia. J. Microbiol. Immunol. Infect. 2004;37:350–358. [PubMed] [Google Scholar]
- 10.Crossman L.C., Gould V.C., Dow J.M., Vernikos G.S., Okazaki A., Sebaihia M., Saunders D., Arrowsmith C., Carver T., Peters N., et al. The complete genome, comparative and functional analysis of Stenotrophomonas maltophilia reveals an organism heavily shielded by drug resistance determinants. Genome Biol. 2008;9:R74. doi: 10.1186/gb-2008-9-4-r74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ryan R.P., Monchy S., Cardinale M., Taghavi S., Crossman L., Avison M.B., Berg G., van der Lelie D., Dow J.M. The versatility and adaptation of bacteria from the genus Stenotrophomonas. Nat. Rev. Microbiol. 2009;7:514–525. doi: 10.1038/nrmicro2163. [DOI] [PubMed] [Google Scholar]
- 12.Gröschel M.I., Meehan C.J., Barilar I., Diricks M., Gonzaga A., Steglich M., Conchillo-Solé O., Scherer I.-C., Mamat U., Luz C.F., et al. The phylogenetic landscape and nosocomial spread of the multidrug-resistant opportunist Stenotrophomonas maltophilia. Nat. Commun. 2020;11:2044. doi: 10.1038/s41467-020-15123-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Vidigal P.G., Schmidt D., Stehling F., Mellies U., Steinmann E., Buer J., Rath P.-M., Steinmann J. Development of a quantitative immunofluorescence assay for detection of Stenotrophomonas maltophilia antibodies in patients with cystic fibrosis. J. Cyst. Fibros. 2013;12:651–654. doi: 10.1016/j.jcf.2013.04.011. [DOI] [PubMed] [Google Scholar]
- 14.Waters V., Yau Y., Prasad S., Lu A., Atenafu E., Crandall I., Tom S., Tullis E., Ratjen F. Stenotrophomonas maltophilia in cystic fibrosis: Serologic response and effect on lung disease. Am. J. Respir. Crit. Care Med. 2011;183:635–640. doi: 10.1164/rccm.201009-1392OC. [DOI] [PubMed] [Google Scholar]
- 15.Waters V., Atenafu E.G., Lu A., Yau Y., Tullis E., Ratjen F. Chronic Stenotrophomonas maltophilia infection and mortality or lung transplantation in cystic fibrosis patients. J. Cyst. Fibros. 2013;12:482–486. doi: 10.1016/j.jcf.2012.12.006. [DOI] [PubMed] [Google Scholar]
- 16.Vidigal P.G., Dittmer S., Steinmann E., Buer J., Rath P.-M., Steinmann J. Adaptation of Stenotrophomonas maltophilia in cystic fibrosis: Molecular diversity, mutation frequency and antibiotic resistance. Int. J. Med. Microbiol. 2014;304:613–619. doi: 10.1016/j.ijmm.2014.04.002. [DOI] [PubMed] [Google Scholar]
- 17.Falagas M.E., Valkimadi P.E., Huang Y.T., Matthaiou D.K., Hsueh P.R. Therapeutic options for Stenotrophomonas maltophilia infections beyond co-trimoxazole: A systematic review. J. Antimicrob. Chemother. 2008;62:889–894. doi: 10.1093/jac/dkn301. [DOI] [PubMed] [Google Scholar]
- 18.Bax B.D., Chan P.F., Eggleston D.S., Fosberry A., Gentry D.R., Gorrec F., Giordano I., Hann M.M., Hennessy A., Hibbs M., et al. Type IIA topoisomerase inhibition by a new class of antibacterial agents. Nature. 2010;466:935–940. doi: 10.1038/nature09197. [DOI] [PubMed] [Google Scholar]
- 19.Gibson E.G., Ashley R.E., Kerns R.J., Osheroff N. Antimicrobial Resistance in the 21st Century. Springer; Cham, Switzerland: 2018. [DOI] [Google Scholar]
- 20.Dougherty T.J., Nayar A., Newman J.V., Hopkins S., Stone G.G., Johnstone M., Shapiro A.B., Cronin M., Reck F., Ehmann D.E. NBTI 5463 is a novel bacterial type ii topoisomerase inhibitor with activity against gram-negative bacteria and in vivo efficacy. Antimicrob. Agents Chemother. 2014;58:2657–2664. doi: 10.1128/AAC.02778-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Flamm R.K., Farrell D.J., Rhomberg P.R., Scangarella-Oman N.E., Sader H.S. Gepotidacin (GSK2140944) in vitro activity against gram-positive and gram-negative bacteria. Antimicrob. Agents Chemother. 2017;61:e00468-17. doi: 10.1128/AAC.00468-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Taylor S.N., Morris D.H., Avery A.K., Workowski K.A., Batteiger B.E., Tiffany C.A., Perry C.R., Raychaudhuri A., Scangarella-Oman N.E., Hossain M., et al. Gepotidacin for the treatment of uncomplicated urogenital gonorrhea: A phase 2, randomized, doseranging, single-oral dose evaluation. Clin. Infect. Dis. 2018;67:504–512. doi: 10.1093/cid/ciy145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brooke J.S., Di Bonaventura G., Berg G., Martinez J.-L. Editorial: A multidisciplinary look at Stenotrophomonas maltophilia: An emerging multi-drug-resistant global opportunistic pathogen. Front. Microbiol. 2017;8:1511. doi: 10.3389/fmicb.2017.01511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Biedenbach D.J., Bouchillon S.K., Hackel M., Miller L.A., Scangarella-Oman N.E., Jakielaszek C., Sahm D.F. In vitro activity of gepotidacin, a novel triazaacenaphthylene bacterial topoisomerase inhibitor, against a broad spectrum of bacterial pathogens. Antimicrob. Agents Chemother. 2016;60:1918–1923. doi: 10.1128/AAC.02820-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lahiri S.D., Kutschke A., McCormack K., Alm R.A. Insights into the mechanism of inhibition of novel bacteria topoisomerase inhibitors from characterization of resistant mutants of Staphylococcus aureus. Antimicrob. Agents Chemother. 2015;59:5278–5287. doi: 10.1128/AAC.00571-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hoover J.L., Singley C.M., Elefante P., Rittenhouse S. Efficacy of human exposures of gepotidacin (GSK2140944) against Escherichia coli in a rat pyelonephritis model. Antimicrob. Agents Chemother. 2019;63:e00086-19. doi: 10.1128/AAC.00086-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Barth A., Hossain M., Brimhall D.B., Perry C.R., Tiffany C.A., Xu S., Dumont E.F. Pharmakokinetics of oral formulations of gepotidacin (GSK2140944), a triazaacenaphthylene bacterial type II topoisomerase inhibitor, in healthy adult and adolescent participants. Antimicrob. Agents Chemother. 2022;66:e0126321. doi: 10.1128/AAC.01263-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.GlaxoSmithKline GSK Starts a Phase III Clinical Programme for a Potential First-in-Class Antibiotic, Gepotidacin. [(accessed on 10 August 2021)]. Available online: https://www.gsk.com/en-gb/media/press-releases/gsk-starts-a-phase-iii-clinical-programme-for-a-potential-first-in-class-antibiotic-gepotidacin/
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.