Abstract
Multiple efforts are currently underway to control and treat severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), causing coronavirus disease 2019 (COVID-19) worldwide. Despite all efforts, the virus that emerged in Wuhan city has rapidly spread globally and led to a public health emergency of international concern (PHEIC) due to the lack of approved antiviral therapy. Nevertheless, SARS-CoV-2 has had a significant influence on the evolution of cellular therapeutic approaches. Adoptive immune cell therapy is innovative and offers either promising prophylactic or therapy for patients with moderate-to-severe COVID-19. This approach is aimed at developing safety and providing secure and effective therapy in combination with standard therapy for all COVID-19 infected individuals. Based on the effective results of previous studies on both inflammatory and autoimmune diseases, various immune cell therapies against COVID-19 have been reviewed and discussed. It must be considered that the application of cell therapy for treatment and to eliminate infected respiratory cells could result in excessive inflammation, so this treatment must be used in combination with other treatments, despite its many beneficial efforts.
Keywords: COVID-19, SARS-CoV-2, Immune cell therapy, Treg therapy, NK therapy, MSC therapy, DC therapy, CAR-T cell therapy, Viral specific T cell therapy
1. Introduction
There is a global health emergency on the threat of a rapidly spreading pathogenic SARS-coronavirus 2 (SARS-CoV-2) causing COVID-19, a highly lethal pandemic of a respiratory pathogen with the potential for killing millions of people [1]. The SARS-CoV-2 pandemic presents unparalleled anti-viral treatment challenges as COVID-19 disease is inflammatory in nature and characterized by its hallmark uncontrolled inflammatory response. Therefore, the inefficiency of the immune system, both in terms of over activity and the inability to destroy the virus, can play a crucially significant role in the pathology of the virus [2]. Therefore, it is thought that inflammation reduction therapies can efficiently maintain the patient and prevent death. Based on this idea, anti-inflammatory medications have been used to reduce the severity of inflammation in some centers, including the monoclonal antibodies against inflammatory cytokines [3]. In this regard, we should not neglect the new method such as immune cell therapy to overcome therapeutic barriers. This approach was used to treat various inflammatory and autoimmune diseases such as rheumatoid arthritis (RA), and acceptable results were obtained [4], [5]. Thus, this paper aims to review the potentially life-saving complex therapies, such as the types of immune cells that have therapeutic potential in the management of COVID-19.
2. Immunology & immunopathology of COVID-19
Although there is no exact detection of the pathogenesis of COVID-19, there are plenty of reports on the well-known mechanism (s) that is applied by the virus to the entering of host cells. SARS-CoV-2 binds to target cells through the interaction of viral spike glycoprotein (S) with the angiotensin-converting enzyme 2 (ACE2) [6] a dipeptidyl peptidase 4 (DPP4) expressed on the surface of endothelial cells [7], similar to SARS-CoV. This interaction leads to the entrance of viral RNA into the cytoplasm. Subsequently, it is encapsulated, polyadenylated, and encrypts various structural and non-structural polypeptide genes, and eventually, the novel-produced viruses are released from host cells. The released viruses infect a broad range of cells in several systems, such as respiratory, cardiovascular, renal, neurological, hepatic, biliary, reproductive, gastrointestinal, and integumentary [8]. One of the primary reasons for the prevalent symptoms of COVID-19 is the distribution of ACE2 receptors in different tissues [1]. Cellular entrance of SARS-CoV-2 hinges on the viral spike (S) protein and other viral polypeptide priming by the host cellular transmembrane serine protease 2 (TMPRSS2) in addition to the attachment of viral (S) protein to ACE2 [6]. Recent evidence from single-cell RNA sequencing (scRNA-seq) indicates that other proteases such as cathepsin B and L are more probable to be implicated in the cellular entrance procedure because both ACE2 and TMPRSS2 are not expressed on the same cell [9].
3. Immune response and cytokine storm
Hyper-inflammatory reaction to SARS-CoV-2 infection, known as cytokine release syndrome (CRS) or cytokine storm (CS), arise in some patients in whom acutely elevated serum levels of different pro-inflammatory cytokines such as interleukin (IL)-1β, IL-2, IL-6, IL-10, tumor necrosis factor-α (TNF-α), interferon-gamma (INF-γ), CXCL9, CXCL10 and soluble IL-2 receptor alpha (sIL-2Rα) are observed [1], [10]. Multiple molecular pathways are proposed to better understand the complex molecular events behind the CS response in patients seriously affected by COVID-19. Understanding of the CS process is essential because it causes lymphopenia and lymphocyte dysfunction [11] and the defects in lymphocyte cytolytic activity of natural killer (NK) cells from innate immunity and CD8 cytolytic T cells from adaptive immunity are mentioned as a reason for the progress of CS [12]. However, this defective condition, either genetic or acquired, leads to the incapacity of cytolytic cells to provoke apoptosis in infected and activated antigen-presenting cells. In this sense, many pro-inflammatory cytokines are released as a result of extended and exaggerated interactions between innate and adaptive immune cells, which flow cytometry analysis demonstrates that immune cells numbers, such as CD4+ T cells, CD8+ T cells, NK cells, and B cells are significantly changed in a person with COVID-19 [11].
It should be noted that in the CS, both groups of pro-inflammatory and anti-inflammatory cytokines increased in patients' serum. Particularly, SARS-CoV-2 can trigger Th1 cells to secrete pro-inflammatory cytokines such as granulocyte–macrophage colony-stimulating factor (GM-CSF), IL-6, and TNF-α, which are the principal components in the CS. Subsequently, GM-CSF produces high levels of IL-6, TNF-α, and other cytokines by activating inflammatory monocytes [13]. According to clinical evidence, the massive release of inflammatory cytokines is a potentially lethal immune situation characterized by infiltration, proliferation, and hyper-activation of heterogeneous immune cells, such as macrophages, neutrophils, NK cells, and T cells, inside the inflamed tissue [14]. The non-specific hyperactivity of immune cell infiltration results in adverse effects on tissues such as the lung. Lung failure -a common outcome of CS- causes lung-parenchyma changes, such as diffuse alveolar damage, which can develop acute lung damage or a more severe form, acute respiratory distress syndrome (ARDS), finally leading to pulmonary fibrosis and death [15]. The increase in neutrophil extracellular traps (NETs) and NETosis has been reported as a cause of CS development in COVID-19 [16]. NETs are extracellular fiber networks consisting of activated neutrophil DNA and protect against infections and pathologies of immune-mediated diseases like CS [17]. Based on autopsy findings, it is speculated that viral RNA and pro-inflammatory cytokines may stimulate neutrophils and establish NETs and NETosis. A reason for the unrestrained cytokine release is the viral manipulation of nuclear factor-kB (NF-κB) and mitogen-activated protein kinases (MAPK) signaling pathway via the Host-virus mediated TMPRSS2 and ACE2 interaction [18], [19]. This interaction triggers the hyper-activation of NF-κB, predominantly in non-immune cells, including pulmonary epithelial cells, which in turn produce more cytokines and chemokines.
4. Strategies to immune cell therapy
One useful therapeutic approach may be the application of strategies for reducing the production of inflammatory cytokines. These approaches can be divided into two categories: cell-free and cell-mediated therapy. Fundamentally, the first group is applying medications, including small inhibitors such as Baricitinib, Tofacitinib, monoclonal antibodies, and secretory vesicles (known as exosomes) that contain anti-inflammatory substances. The second group, cell-mediated therapy, as the topic of this article, involves the employment of several immunomodulatory cells, which can be provided from both autologous and allogeneic sources [20]. The allogeneic source has a high priority along with the troubles, costs, and emergency conditions of COVID-19. Several immunotherapeutic approaches are currently being evaluated to combat SARS-CoV-2 infection (Table 1 ). These approaches are particularly focused on targeting inflammatory processes, and many other strategies are being evaluated (Fig. 1 ). However, in view of unpredictable immune responses in severe COVID-19 patients, all strategies based on reducing inflammation and raising the effective immune response against the SARS-CoV-2 infection considered for COVID-19 patient management are summarized in Table 1 and will be covered in more detail.
Table 1.
Overview of cell-based clinical trials for the treatment of COVID-19.
| Strategy | Type | Study Title | Phase | NCT number |
|---|---|---|---|---|
| Treg | – | Regulatory T Cell infusion for Lung Injury Due to COVID-19 pneumonia | 1 | NCT04468971 |
| RAPA-501-Allo Therapy of COVID-19-ARDS | 1 & 2 | NCT04482699 | ||
| NK | NK | Clinical Trial on NK Cells for COVID-19 | 1 | NCT04634370 |
| Natural Killer Cell (CYNK-001) Infusions in Adults With COVID-19 | 1 & 2 | NCT04365101 | ||
| CAR-NK | Universal Off-the-shelf NKG2D-ACE2 CAR-NK Cells for Therapy of COVID-19 | 1 & 2 | NCT04324996 | |
| MSC | Ad-MSCs | Autologous Adipose-derived Stem Cells (AdMSCs) for COVID-19 | 2 | NCT04428801 |
| BM-MSCs | Treatment of Severe COVID-19 Pneumonia With Allogeneic Mesenchymal Stromal Cells (COVID_MSV) | NCT04361942 | ||
| mesenchymal Stem Cell Infusion for COVID-19 Infection | 1 | NCT04444271 | ||
| Mesenchymal Stem Cells Therapy in Patients With COVID-19 Pneumonia | 1 | NCT04713878 | ||
| mesenchymal Stromal Cells for the Treatment of SARS-CoV-2 Induced Acute Respiratory Failure (COVID-19 Disease) | 1 | NCT04345601 | ||
| Study of the Safety of Therapeutic Tx With Immunomodulatory MSC in Adults With COVID-19 Infection Requiring Mechanical Ventilation | 1 | NCT04397796 | ||
| UC-MSCs | Clinical Trial of Allogeneic Mesenchymal Cells From Umbilical Cord Tissue in Patients With COVID-19 | 1 | NCT04366271 | |
| Clinical Research of Human Mesenchymal Stem Cells in the Treatment of COVID-19 Pneumonia | 1 & 2 | NCT04339660 | ||
| Use of Mesenchymal Stem Cells in Acute Respiratory Distress Syndrome Caused by COVID-19 | Early Phase 1 | NCT04456361 | ||
| Treatment With Human Umbilical Cord-derived Mesenchymal Stem Cells for Severe Corona Virus Disease 2019 (COVID-19) | 2 | NCT04288102 | ||
| Umbilical Cord-derived Mesenchymal Stem Cells for COVID-19 Patients With Acute Respiratory Distress Syndrome (ARDS) | 1 & 2 | NCT04355728 | ||
| Cell Therapy Using Umbilical Cord-derived Mesenchymal Stromal Cells in SARS-CoV-2-related ARDS | 1 & 2 | NCT04333368 | ||
| Cellular Immuno-Therapy for COVID-19 Acute Respiratory Distress Syndrome | 1 & 2 | NCT04400032 | ||
| Mesenchymal Stem Cells for the Treatment of COVID-19 | 1 | NCT04573270 | ||
| NHPBSC | study Evaluating the Safety and Efficacy of Autologous Non-Hematopoietic Peripheral Blood Stem Cells in COVID-19 | 1 & 2 | NCT04473170 | |
| DP-MSCs | Novel Coronavirus Induced Severe Pneumonia Treated by Dental Pulp Mesenchymal Stem Cells | 1 | NCT04302519 | |
| Safety and Efficacy Study of Allogeneic Human Dental Pulp Mesenchymal Stem Cells to Treat Severe COVID-19 Patients | 1 & 2 | NCT04336254 | ||
| SBI-101 | A Study of Cell Therapy in COVID-19 Subjects With Acute Kidney Injury Who Are Receiving Renal Replacement Therapy | 1 & 2 | NCT04445220 | |
| HCT-MSCs | hCT-MSCs for COVID19 ARDS | 1 & 2 | NCT04399889 | |
| HB-adMSCs | A Clinical Trial to Determine the Safety and Efficacy of Hope Biosciences Autologous Mesenchymal Stem Cell Therapy (HB-adMSCs) to Provide Protection Against COVID-19 | 2 | NCT04349631 | |
| A Randomized, Double-Blind, Placebo-Controlled Clinical Trial to Determine the Safety and Efficacy of Hope Biosciences Allogeneic Mesenchymal Stem Cell Therapy (HB-adMSCs) to Provide Protection Against COVID-19 | 2 | NCT04348435 | ||
| MD-MSCs | Treatment of Covid-19 Associated Pneumonia With Allogenic Pooled Olfactory Mucosa-derived Mesenchymal Stem Cells | 1 & 2 | NCT04382547 | |
| Therapeutic Study to Evaluate the Safety and Efficacy of DW-MSC in COVID-19 Patients | 1 | NCT04535856 | ||
| Not determined | Clinical Use of Stem Cells for the Treatment of Covid-19 | 1 & 2 | NCT04392778 | |
| Mesenchymal Stromal Cell Therapy For The Treatment Of Acute Respiratory Distress Syndrome | 1 | NCT04447833 | ||
| Safety and Effectiveness of Mesenchymal Stem Cells in the Treatment of Pneumonia of Coronavirus Disease 2019 | Early Phase 1 | NCT04371601 | ||
| DC | – | Dendritic Cell Vaccine to Prevent COVID-19 | 1 & 2 | NCT04685603 |
| Specific T cell | – | Novel Adoptive Cellular Therapy With SARS-CoV-2 Specific T Cells in Patients With Severe COVID-19 | 1 | NCT04351659 |
| Part Two of Novel Adoptive Cellular Therapy With SARS-CoV-2 Specific T Cells in Patients With Severe COVID-19 | 1 &2 | NCT04457726 | ||
| SARS-CoV-2 Specific Cytotoxic T Lymphocytes for the Treatment of COVID-19 in Patients With Cancer | 1 | NCT04742595 | ||
| memory T and B cell | – | COVID-19: SARS-CoV-2 Specific Memory B and T-CD4± Cells | NCT04402892 | |
| Safety Infusion of Natural Killer cells or Memory T Cells as Adoptive Therapy in COVID-19 pneumonia or Lymphopenia | 1 &2 | NCT04578210 | ||
| monocyte | – | The MONACO Cell Therapy Study: Monocytes as an Anti-fibrotic Treatment After COVID-19 | 1 &2 | NCT04805086 |
Dental pulp: DP
Non-hematopoietic peripheral blood stem cells: NHPBSC
Mucosa-derived Mesenchymal Stem Cells: MD-MSCs
Allogeneic hybrid Treg/Th2 cell (RAPA-501-ALLO)
Human cord tissue mesenchymal stromal cells: HC-MSCs
Hope biosciences autologous mesenchymal stem cell: HB-adMSCs
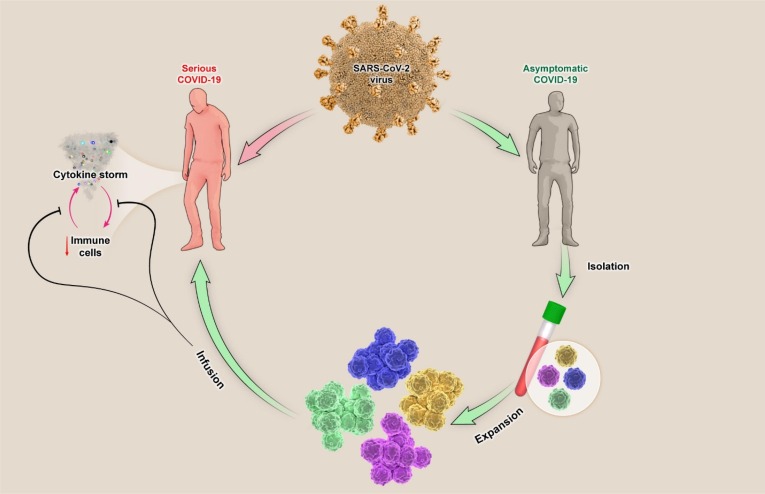
Fig. 1.
Schematically strategy for Immune Cell-Based Immunotherapy 19. In immunotherapy, the first step is to mass-produce functional immune cells that can be from recovered individuals and subsequently re-injected to the severely affected patient after performing of expansion procedures in vitro. Injected immune cells, relying on their functional properties, can suppress the process of cytokine storm and reduction of immune cells that results in improving the patient with severe symptoms.
4.1. T Regulatory cells therapy
Regulatory T (Treg) cells characterized by CD4+CD25highCD127low/-FoxP3+ phenotypic markers, as more precise regulatory mechanisms, are naturally accessible in adjusting the initiation of adaptive immune responses and monitoring the progressive inflammation responses in the body [21]. The honored therapeutic potential of Treg cell subsets in animal models promises to elaborate novel therapeutic approaches for treating of immune-mediated diseases in humans [4]. According to the anti-inflammatory activity of Treg cells in viral pneumonia [22], [23], it has been hypothesized that the disruption of Treg cells, either in quantity or function, may contribute to the severity of COVID-19 pathology [24], [25], [26]. The infiltration of these cells into the inflamed lung tissue to prevent damage can be cited as a potential reason. Decreased IL-2 and increased CD25 levels were detected in bronchoalveolar lavage specimens of patients suffering from severe COVID-19 [27], [28]. Thus, a decline in IL-2 levels would stimulate Tregs apoptosis. Alternatively, COVID-19 patients have increased CD25 levels, perhaps owing to rising proteolytic degradation of CD25 cells relative to inflammation [29]. This soluble CD25 might interfere with bioavailability and signaling pathways and increase Tregs apoptosis. It should be noted that the probability of a direct SARS-CoV-2 effect on the biology of Tregs must not be ruled out because it has formerly been explained that the Middle East Respiratory Syndrome (MERS) coronavirus efficiently decreases T lymphocytes count through induction of both extrinsic and intrinsic apoptosis pathways [30]. Considering the importance of Tregs in immune homeostasis, reduction in Tregs levels could be a reason for the overstimulation of the immune system and lung damage in severe COVID-19 patients. Therefore, the potential of regenerating either the quantity or function of these cells in patients has been proposed [31]. Noted that some factors like obesity or chronic diseases like systemic lupus erythematosus (SLE) and diabetes are the risk factors for COVID-19, and data from these subjects indicate that Tregs quantity or activity reduced compared with those in healthy subjects, and, so, there is a higher state of inflammation and CS.
The revival of Treg cell function has been markedly reported after the administration of biological medication in patients with an inflammatory disease like RA [32], albeit those medications are not primarily designed with the intention of adjusting Treg cells. Hence, multiple approaches have been devised to enhance the quantity and recover the function of Treg cells, such as the expansion of autologous Treg, expansion of antigen-specific Treg, induced Treg from the naïve CD4+ T cell, and FoxP3 gene transduction [33], [34], [35]. The available evidence for Treg cell potential in therapy can be outlined in two sections, covering both in vivo and ex vivo evidence. Adoptive transfusion of Treg cells in respiratory syncytial virus-infected animals reduces immunopathology by regulating the CD8+ effector T cells against the virus [36]. Additionally, to inhibit inflammatory responses, Treg cells can enhance tissue remediation by the expression of amphiregulin, which does not depend on the immunosuppressive activity of Treg cells [37].
4.2. Natural killer cell therapy
Natural killer (NK) cells, as a part of the innate immune system, serve as useful effectors against life-threatening viral infection. The results indicate a correlation between a higher count of NK cells in peripheral blood with negative viral RNA and positive IgG/IgM tests [38]. The assessment of SARS-CoV-2 and SARS-CoV-1 clinical data shows that COVID-19 disease severity is correlated with a greater reduction in the count of NK cells [39], the exhaustion of NK cells, lack of specific maturity, and potent NK cell phenotypes [40]. However, the NK cell lymphocytopenia group had a remarkably worse survival ratio and a long period of viral shedding. Because NK cells play an essential role against SARS-CoV-2-infected cells in several manners, including the direct killing effect through killer-cell immunoglobulin-like receptors (KIR) receptors mediated apoptosis, degranulation, antibody-dependent cell-mediated cytotoxicity (ADCC), active interaction with dendritic cells (DC) in antigen presentation processes and the secretion of specific cytokines.
Notably, these anti-viral properties make NK cells more useful candidates for any pandemic infections and re-emerging viruses, as well as COVID-19 immunotherapy. Several publications have mentioned a potential role of NK cells in fighting various viral infections, as well as COVID-19 with a permanent modification in the NK cell profile toward those more mature and potent phenotypes in recovering patients [41], [42], [43], [44]. Early adoptive transfer of highly activated NK cells at the onset of the disease may encourage innate and adaptive immunity, thereby improving survival and reducing rates of disease progression in SARS-CoV-2 infected patients.
4.3. Mesenchymal stem cells therapy
The therapeutic application of mesenchymal stem cells (MSCs) as a promising prospect has been extensively investigated for various clinical applications in medicine. There are two hypotheses based on their unique characteristics -immunomodulatory and regenerative potencies- that comprise: 1) they can reduce acute lung damage and 2) they can suppress severe cellular inflammatory responses induced by SARS-CoV-2. Therefore, these multipotent cells may be exploited in various branches of therapy, which includes regenerative medicine, immunotherapy, tissue engineering, and cellular / molecular biology [45], [46], [47], [48]. The immunomodulatory activities of MSCs would possibly consist of (a) inhibiting the proliferation and function of T cells, B cells, DCs, and NK cells; (b) polarizing monocytes to anti-inflammatory M2 macrophages; (c) producing IL-10 associate with diminishing the production of TNF-α and IL-12 and (d) inhibiting the production of hydrogen peroxide by stimulated neutrophils [49], [50], [51], [52]. Moreover, MSCs have potent anti-fibrotic effects and decrease lung fibrosis [53], [54], which facilitates the regeneration of damaged pulmonary epithelial cells and promotes alveolar fluid clearance [11]. Hence, lung function is renovated through augmented alveolar air-space volume, reducing alveolar thickening, and inflammation markers [55], [56].
Unlike different cells, MSCs do not substantially express the surface ACE2 receptor and TMPRSS2 receptors (entrance gate of coronavirus into the host cells), which ensures that adoptive MSCs transfer therapy can perform their immunomodulatory effects without being neither infected nor destroyed by the virus [57]. Moreover, intrinsic interferon-stimulated genes (ISGs) play a significant role in MSCs’ resistance to viral infections as compared to their differentiated progenies. In fact, viral infection is prevented by the expression of ISGs [58]. Additionally, the produced Leukemia inhibitory factor (LIF) by MSCs has the capacity to neutralize CS in the lungs during viral pneumonia [59]. Moreover, several studies indicated that some antiviral activities of the MSCs contributed to the expression of indoleamine-pyrrole 2,3-dioxygenase (IDO) by these cells [60], [61], [62]. The apoptosis of activated T cells and the transformation of tryptophan into kynurenine are induced by the secretion of IDO from MSCs, which suppresses the proliferation of effector T cells [63]. MSCs also contribute to the protection of both epithelial and endothelial barrier function in ARDS and sepsis [64], [65]. Due to the pathogenesis of ARDS, the restoration of disrupted alveolar-capillary barrier is promoted by keratinocytes growth factor (KGF) and angiopoietin-1 (Ang-1) secreted by MSCs [66]. It is beneficial for patients who develop pulmonary fibrosis even after surviving the acute phase of the disease [67], [68], [69].
In comparison with other sources of MSCs, umbilical cord mesenchymal stem cells (UC-MSCs) have been predominantly exploited in diverse diseases because of their adequate collection, no ethical issues, little immunogenicity, and rapid proliferation rate [70], [71]. An established route of treatment with UC-MSCs is the intravenous injection of these cells as they are trapped in the lungs, the most affected organ in COVID-19 patients [72], but it is still unknown whether the intratracheal / bronchial administration of MSCs is feasible and efficient or not [73]. The average size of MSCs is about 30 µm (range 16 to 53 µm), which range is in the relatively large cells category [74], [75]. Hence, after intravenous administration, they are merely trapped in the lungs and are beneficial for COVID-19 therapy because viruses, particularly targeting this organ, might be a priority to other therapies [76]. Modulation of immune cells activation, suppression of infiltrating cells, and reduction of edema can contribute to the regulation of the immune system in the lung tissue by these cells [77]. According to reviews of the function of extracellular vesicles (EVs), including microvesicles and exosomes, MSCs may be effective in healing acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) by producing EVs [11], [78]. In a clinical survey of patients infected with the H7N9 influenza virus, which submitted symptoms such as ARDS, lung failure, and severe pneumonia, MSC post-transplant mortality rate has decreased without adverse effects [79]. Since influenza A and COVID-19 share related symptoms (such as ARDS and lung failure) and similar multi-organ dysfunction, MSC-based therapy may be a promising alternative therapy to deal with COVID-19.
4.4. Dendritic cells therapy
Dendritic cells (DCs) with various subpopulations, as well known as conventional dendritic cells (cDCs) and plasmacytoid dendritic cells (pDCs), play a critical role in both innate and adaptive immune responses against pathogens. Simultaneously, it also provides an essential service for effective vaccination. Furthermore, activated DCs could save the adjacent cells from viral attack through the secretion of a considerable level of type one interferons [80]. It should be regarded that in the case of SARS-CoV-2 infection, DCs infected along with endothelial cells like type II alveolar, and infected DCs might assist the induction of CS in patients [81]. Inquiry of DCs in COVID-19 patients has proved depletion and reduction of maturation in both cDC and pDC subsets and a re-localization of activated cDC2 in the lungs [82], [83]. Prior findings stated the inadequacy of T-cell responses to the virus and induction of NK cell cytotoxicity against infected DCs owing to downregulation of MHC-I and II molecules as well as a decreased expression of CD80/CD86 molecules on MERS- infected DCs. [83], [84]. According to the T cell insufficiency in COVID-19 patients and the vital role of DCs in T cell priming, it seems reasonable to reinforce DCs for activating vigorous T cell responses in these patients. It has already been established that antigen-specific T cell responses stimulated by DC-delivered peptides are 100 to 1000 fold more effective than nonspecific stimulation [85]. Therefore, there are great attempts to induce efficient immune responses using DCs presenting specific viral antigens in severe infections such as human immunodeficiency virus (HIV) [5]. For the intent of DC therapy, there are two main cell sources, including CD14+ monocytes from peripheral blood mononuclear cells (PBMCs) and the CD34+ stem cells from the umbilical cord or bone marrow. Although the application of these cells in anti-cancer DC-based therapy has given encouraging results [86], a serious concern in using DCs to treat COVID-19 is the potential of in-vivo differentiation to the inflammatory macrophages. As noted, macrophages are one of the major sources of pro-inflammatory cytokines secretion and pernicious inflammation inside the infected tissue.
The other limitation in the autologous CD14+ cells assembly pathway is the high risk of extracting infected monocytes whose function was formerly impaired [87]. To defeat problems, CD34+ stem cells can be exploited after differentiation into DCs in the presence of GM-CSF in vitro. In this sense, umbilical cord stem cells appear to be preferable because they generate a milder graft- versus-host-disease (GVHD) and require less HLA matching between donor and recipient; however, bone marrow-derived cells possessing immunomodulatory properties provide further gains for the patients suffering from disordered cytokine production [88]. There are other questions that need to be responded in DC-based therapy. To achieve the most advantages of DCs function, they should be loaded with appropriate antigens that could trigger preferentially antigen-specific T cell responses against the viral particles. Obviously, an excellent candidate for this purpose is the spike protein (S) of SARS-CoV. The constructed-S protein was explained to induce both cellular and antibody-mediated responses. Nevertheless, determining the best peptide fragments derived from this huge protein is a subject still under investigation. Several supplied structural epitopes and peptide fragments of SARS-CoV, which are presumed to activate anti-viral cytotoxic T cells, comprise; S436, S525, EP1 (aa 51–71), EP2 (aa 134–208), EP3 (aa 249–273), EP4 (aa 349–422), and N1 from the N protein [89], [90]. S450-650 is another fragment under survey that is believed to provoke appreciable antigen-specific responses; nonetheless, the conformational multiplicity makes it more complex to ascertain a suitable peptide that could induce the production of reliable T-dependent neutralizing antibodies [91]. After choosing the best antigen, the next step is to select the most convenient antigen-loading technique. There are several introduced ways, including; RNA injection by electroporation, lipid-mediated transfection, viral vectors, and antigen nanoparticles [92], [93]. Some investigators have also attempted to differentiate monocytes into DCs using extracorporeal photopheresis (ECP) and prime them with killed viruses to develop potent DCs [94]. Although the antigen loading techniques are continuously improving, some disadvantages such as toxicity, allergenicity, and the possibility of DC phenotype alteration remain to be worked out. Besides all these efforts to generate efficient DCs for inducing T-cell responses against coronavirus, it is worth remarking that generation of tolerogenic DCs with andrographolide, which prevents NF-KB, might help control the excessive immune responses in COVID-19 patients [84].
4.5. Chimeric antigen receptor T cell therapy
Chimeric antigen receptor (CAR) and T cell receptor (TCR) T-cell therapy involve genetically modified patient's T-cells with antigen-specific receptors. This approach, as a revolution in anti-cancer immunotherapy, is a reviving therapy in handling different types of cancer. Based on the ability of CAR-T and TCR-T cells to recognize specific surface and intracellular antigens, respectively, and guide the immune cells to eradicate the targets when infused back into the patients, the harnessing potency of these approaches have been proposed to treat viral infections such as hepatitis B and HIV beyond cancer [95]. Therefore, scientists are investigating the adoptive transfer of SARS-CoV-2 − specific T-cells to prevent and treat COVID-19, based on previous research in which the developed SARS-specific cytotoxic T-cells conducted by specific-TCR gene transfer could recognize SARS antigens [96].
Even though CAR-T or TCR-T therapy has provided an inimitable response, there are several potential disadvantages that must be considered and overcome, including long-lasting cytopenia, CS, neurotoxicity, and elimination of all infected cells that might affect vital organs [97], [98], [99], [100]. One way to address this dilemma is to engineer CAR/TCR T-cells using mRNA electroporation, which can restrict their functional activity duration and inflammatory capability [96]. Furthermore, it is undoubtedly offered that combination therapy - CAR/TCR T-cells combined with an anti-viral medication may prove to be safe and effective. Generally, focused and rigorous research is necessary to refine the immunotherapy approach to treating COVID-19 and other viral diseases.
4.6. Specific T cell therapy
Cellular immunity, particularly specific-memory T cells, has gained a special interest in the induction of sustained immunity against coronaviruses, involving unstable antibody-mediated immunity in convalescent patients and reducing IgM and IgG serum levels against coronavirus antigens [101]. It has already been found that virus-specific T cells could be isolated and expanded. These T cells are able to recognize various viral antigens, especially structural antigens such as membrane proteins [102]. In a recent study, SARS-CoV-2-specific T cells (SARS-CoV-STs) were isolated from PBMCs of convalescent donors and cultured in the presence of IL-4 and IL-7. ELISPOT analysis indicated that these T cells could respond to the membrane, spike, and nucleocapsid proteins. The results also suggest the potential of specific-T cell therapy, either from an autologous or off-the-shelf source, to treat critical COVID-19 patients and gives hope for preventive strategies in case of immunocompromised patients such as those undergoing hematopoietic stem cell transplantation [103]. Currently, two clinical trials are investigating SARS-CoV-STs therapy, which follows three major goals, including the feasibility of rapidly isolating SARS-CoV-STs from the convalescent donors, appropriately using SARS-CoV-2-specific peptides and an automated medical device for the emerging therapy for severe COVID-19.
5. General and fundamental aspects of cell therapy efficacy
The main aim of cell therapy is to reinvigorate the patient's diminished and failed immune system. This aim might be out of achieving or ineffective owing to the production of elevated levels of inflammatory cytokines along with altered expression of immunosuppressive agents. The normal immune system is skilled at maintaining immune tolerance and preventing autoimmunity by expressing immune checkpoint molecules and silencing the effector T cell function [104]. In this regard, plenty of pathogens as well as cancer cells induced the expression of immune checkpoint molecules to escape the host's immune surveillance, which leads to the effector T cell exhaustion [105], [106]. Since immune cell therapy has been alternatively proposed as a high-potential therapy for COVID-19 patients, it is substantial that health care providers have a definite insight into the advantages and disadvantages of cell therapy. Therefore, the application of immune checkpoint inhibitors (ICIs) before or during cell therapy might be considered as an appropriate combination therapy method to treat and/or improve the outcome of chronic and acute infectious diseases treatment [105].
6. The application of immune checkpoint inhibitors
Immune checkpoints express naturally throughout the immune system and are principally responsible for modulating immune response [105]. These molecules are triggered after binding to relevant ligands on the surface of other cells and launch an immunosuppressive message to the immune cell, which ultimately provides the essential equilibrium of both co-stimulatory and co-inhibitory signals in T cell activation [105]. As an escape mechanism and overcoming the host immune response, SARS-CoV-2 uses diverse procedures similar to other infectious agents or cancer cells. A clear example is a SARS-CoV-2 ability to produce severe leukopenia through the production of PLpro and ORF3b, which prevents NF-κB cleavage from IkBα and IRF3 nuclear localization, respectively. SARS-CoV-2 infection, in addition to the reduction of T cells, increases the exhaustion of effector T cells by provoking the expression of inhibitory receptors such as programmable cell death protein-1 (PD-1), T-cell immunoglobulin and mucin domain-3 (TIM-3), and T cell immune receptor with Ig and ITIM domains (TIGIT) on the surface of immune cells, such as T cells, consequently evolving of CS or by reducing the regulatory T cell population [106], [107], [108], [109]. According to the reported results, in vitro blockade of PD-1 improves T cell function and enhances T cell-mediated response to SARS-CoV-2 peptides [110].
The ICIs are monoclonal antibodies that are used to recover cell-mediated immunocompetence [111] through targeting T-cell exhaustion pathways [112], interrupting the binding of immune checkpoint proteins to their ligands [113]. The main classes of ICIs containing anti- PD-1, anti-Programmed death-ligand 1 (PD-L1), and anti-cytotoxic T lymphocyte-associated protein (CTLA-4) antibodies have been potentially planned for managing as immunotherapy medicine in solid tumors [105], [106] can enhance the immune response in COVID-19 patients [111], [113]. Although the application of ICIs-containing therapy regimens may have indicated constructive results in some research, it is essential to remark that a higher rate of hospitalization was observed among patients receiving ICIs-containing regimens. Because of immunotherapy-induced pneumonitis, their application as an effective medicine is in an ambiguous state [112], [114], [115]. Nevertheless, there are currently four clinical trials to evaluate the effectiveness of anti-PD-1 antibody administration for cancer and non-cancer patients with COVID-19 [113].
7. Conclusion
Since the outbreak of COVID-19, we have witnessed a new wave of morbidity and mortality owing to the incidence of COVID-19 in the community continuing to rise. Therefore, given the many impediments to the vaccination process and the mutations that occur in the viral genome, the introduction of an approved treatment method remains an urgent medical need. It appears that the COVID-19 treatment is likely to demand an effective strategy to modulate the over-activation of the immune system while increasing its ability to destroy the virus. Although many ambiguities remain unclear about the application of immune cell therapy, their massive capacity to induce immune homeostasis and biological tolerance makes them a perfect target for any therapeutic application. Hence, we focused on providing insight into the potential benefits of immune-cell therapy in the COVID-19 treatment. Obtained promising findings and technological advances result in improving new therapeutic possibilities, which are favored by both immune system disorders and regenerative medicine. Various immunotherapy approaches that block CS, further lung damage and help to regenerate lung tissue have gained massive attention in the management of patients with severe COVID-19. Meanwhile, the MSC-based approaches appear to be extra attractive to overcome COVID-19, given that it has been the subject of vast clinical studies. Finally, accurate management of immune response in any way that stimulates antiviral immunity while suppressing systemic inflammation could be the key to success in COVID-19 treatment. We therefore, suggest that the combination of cell-based therapy and other therapies is a worthy field for further investigation.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Zavva M., Emadi Kochak H., Abdolmohammadi K., Rashidi N., Mokhtari M., Noorbakhsh F., Azadmanesh K., Shamsi Gooshki E., Fatahi Y., Mokhtari Azad T., Jahangirifard A., Mousavi M.J., Masoumi E., Mirzaei H.R., Gouya M.M., Rezaei F., Nicknam M.H. SARS-Cov-2 and COVID-19, Basic and Clinical Aspects of the Human Pandemic: A Review. Iran J Public Health. 2021 doi: 10.18502/ijph.v50i4.5991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Assadiasl S., Fatahi Y., Zavvar M., Nicknam M.H. COVID-19: Significance of antibodies. Hum Antibodies. 2020;28(4):287–297. doi: 10.3233/HAB-200429. [DOI] [PubMed] [Google Scholar]
- 3.Zhang W., et al. The use of anti-inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): The Perspectives of clinical immunologists from China. Clin Immunol. 2020;214 doi: 10.1016/j.clim.2020.108393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zavvar M., Assadiasl S., Zargaran S., Akhtari M., Poopak B., Dinarvand R., Fatahi Y., Tayebi L., Soleimanifar N., Nicknam M.H. Adoptive Treg cell-based immunotherapy: Frontier therapeutic aspects in rheumatoid arthritis. Immunotherapy. 2020;12(12):933–946. doi: 10.2217/imt-2020-0071. [DOI] [PubMed] [Google Scholar]
- 5.Kundu S.K., Engleman EDGAR, Benike CLAUDIA, Shapero M.H., Dupuis MARC, Van schooten W.C.A., Eibl MARTHA, Merigan T.C. A pilot clinical trial of HIV antigen-pulsed allogeneic and autologous dendritic cell therapy in HIV-infected patients. AIDS research and human retroviruses. 1998;14(7):551–560. doi: 10.1089/aid.1998.14.551. [DOI] [PubMed] [Google Scholar]
- 6.Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.-H., Nitsche A., Müller M.A., Drosten C., Pöhlmann S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181(2):271–280.e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Solerte S.B., Di Sabatino A., Galli M., Fiorina P. Dipeptidyl peptidase-4 (DPP4) inhibition in COVID-19. Acta Diabetol. 2020;57(7):779–783. doi: 10.1007/s00592-020-01539-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.van Eijk L.E., et al. COVID-19: immunopathology, pathophysiological mechanisms, and treatment options. J Pathol. 2021 doi: 10.1002/path.5642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sungnak W., Huang N.i., Bécavin C., Berg M., Queen R., Litvinukova M., Talavera-López C., Maatz H., Reichart D., Sampaziotis F., Worlock K.B., Yoshida M., Barnes J.L. SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nat Med. 2020;26(5):681–687. doi: 10.1038/s41591-020-0868-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Longo D.L., Fajgenbaum D.C., June C.H. Cytokine Storm. N Engl J Med. 2020;383(23):2255–2273. doi: 10.1056/NEJMra2026131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Delshad M., et al. The contributory role of lymphocyte subsets, pathophysiology of lymphopenia and its implication as prognostic and therapeutic opportunity in COVID-19. Int Immunopharmacol. 2021;95 doi: 10.1016/j.intimp.2021.107586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Soy M., Keser G., Atagündüz P., Tabak F., Atagündüz I., Kayhan S. Cytokine storm in COVID-19: pathogenesis and overview of anti-inflammatory agents used in treatment. Clin Rheumatol. 2020;39(7):2085–2094. doi: 10.1007/s10067-020-05190-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hu B., Huang S., Yin L. The cytokine storm and COVID-19. J Med Virol. 2021;93(1):250–256. doi: 10.1002/jmv.26232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sun X., Wang T., Cai D., Hu Z., Chen J., Liao H., Zhi L., Wei H., Zhang Z., Qiu Y., Wang J., Wang A. Cytokine storm intervention in the early stages of COVID-19 pneumonia. Cytokine Growth Factor Rev. 2020;53:38–42. doi: 10.1016/j.cytogfr.2020.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gibson P.G., Qin L., Puah S.H. COVID-19 acute respiratory distress syndrome (ARDS): clinical features and differences from typical pre-COVID-19 ARDS. Med J Aust. 2020;213(2):54–56 e1. doi: 10.5694/mja2.50674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Arcanjo A., et al. The emerging role of neutrophil extracellular traps in severe acute respiratory syndrome coronavirus 2 (COVID-19) Sci Rep. 2020;10(1):19630. doi: 10.1038/s41598-020-76781-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Papayannopoulos V. Neutrophil extracellular traps in immunity and disease. Nat Rev Immunol. 2018;18(2):134–147. doi: 10.1038/nri.2017.105. [DOI] [PubMed] [Google Scholar]
- 18.Goel S., et al. SARS-CoV-2 Switches 'on' MAPK and NFkappaB Signaling via the Reduction of Nuclear DUSP1 and DUSP5 Expression. Front Pharmacol. 2021;12 doi: 10.3389/fphar.2021.631879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhao J., et al. Recent advances on viral manipulation of NF-kappaB signaling pathway. Curr Opin Virol. 2015;15:103–111. doi: 10.1016/j.coviro.2015.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Golchin A. Cell-Based Therapy for Severe COVID-19 Patients: Clinical Trials and Cost-Utility. Stem Cell Rev Rep. 2021;17(1):56–62. doi: 10.1007/s12015-020-10046-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Horwitz D.A., Zheng S.G., Gray J.D. Natural and TGF-beta-induced Foxp3(+)CD4(+) CD25(+) regulatory T cells are not mirror images of each other. Trends Immunol. 2008;29(9):429–435. doi: 10.1016/j.it.2008.06.005. [DOI] [PubMed] [Google Scholar]
- 22.D’Alessio F.R., Tsushima K., Aggarwal N.R., West E.E., Willett M.H., Britos M.F., Pipeling M.R., Brower R.G., Tuder R.M., McDyer J.F., King L.S. CD4+CD25+Foxp3+ Tregs resolve experimental lung injury in mice and are present in humans with acute lung injury. J Clin Invest. 2009;119(10):2898–2913. doi: 10.1172/JCI36498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mangodt T.C., Van Herck M.A., Nullens S., Ramet J., De Dooy J.J., Jorens P.G., De Winter B.Y. The role of Th17 and Treg responses in the pathogenesis of RSV infection. Pediatr Res. 2015;78(5):483–491. doi: 10.1038/pr.2015.143. [DOI] [PubMed] [Google Scholar]
- 24.Harb H., Benamar M., Lai P.S., Contini P., Griffith J.W., Crestani E., Schmitz-Abe K., Chen Q., Fong J., Marri L., Filaci G., Del Zotto G., Pishesha N., Kolifrath S., Broggi A., Ghosh S., Gelmez M.Y., Oktelik F.B., Cetin E.A., Kiykim A., Kose M., Wang Z., Cui Y.e., Yu X.G., Li J.Z., Berra L., Stephen-Victor E., Charbonnier L.-M., Zanoni I., Ploegh H., Deniz G., De Palma R., Chatila T.A. Notch4 signaling limits regulatory T-cell-mediated tissue repair and promotes severe lung inflammation in viral infections. Immunity. 2021;54(6):1186–1199.e7. doi: 10.1016/j.immuni.2021.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wang W., Su B., Pang L., Qiao L., Feng Y., Ouyang Y., Guo X., Shi H., Wei F., Su X., Yin J., Jin R., Chen D. High-dimensional immune profiling by mass cytometry revealed immunosuppression and dysfunction of immunity in COVID-19 patients. Cell Mol Immunol. 2020;17(6):650–652. doi: 10.1038/s41423-020-0447-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Meckiff B.J., Ramírez-Suástegui C., Fajardo V., Chee S.J., Kusnadi A., Simon H., Eschweiler S., Grifoni A., Pelosi E., Weiskopf D., Sette A., Ay F., Seumois G., Ottensmeier C.H., Vijayanand P. Imbalance of Regulatory and Cytotoxic SARS-CoV-2-Reactive CD4(+) T Cells in COVID-19. Cell. 2020;183(5):1340–1353.e16. doi: 10.1016/j.cell.2020.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang Y., Wang X., Li X., Xi D., Mao R., Wu X., Cheng S., Sun X., Yi C., Ling Z., Ma L., Ning Q., Fang Y., Sun B., Wu D.i. Potential contribution of increased soluble IL-2R to lymphopenia in COVID-19 patients. Cell Mol Immunol. 2020;17(8):878–880. doi: 10.1038/s41423-020-0484-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kalfaoglu B., et al. T-Cell Hyperactivation and Paralysis in Severe COVID-19 Infection Revealed by Single-Cell Analysis. Front Immunol. 2020;11 doi: 10.3389/fimmu.2020.589380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chen G., Wu D.i., Guo W., Cao Y., Huang D.a., Wang H., Wang T., Zhang X., Chen H., Yu H., Zhang X., Zhang M., Wu S., Song J., Chen T., Han M., Li S., Luo X., Zhao J., Ning Q. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. 2020;130(5):2620–2629. doi: 10.1172/JCI137244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chu H., Zhou J., Wong B.-Y., Li C., Chan J.-W., Cheng Z.-S., Yang D., Wang D., Lee A.-Y., Li C., Yeung M.-L., Cai J.-P., Chan I.-Y., Ho W.-K., To K.-W., Zheng B.-J., Yao Y., Qin C., Yuen K.-Y. Middle East Respiratory Syndrome Coronavirus Efficiently Infects Human Primary T Lymphocytes and Activates the Extrinsic and Intrinsic Apoptosis Pathways. J Infect Dis. 2016;213(6):904–914. doi: 10.1093/infdis/jiv380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Stephen-Victor E., Das M., Karnam A., Pitard B., Gautier J.-F., Bayry J. Potential of regulatory T-cell-based therapies in the management of severe COVID-19. Eur Respir J. 2020;56(3):2002182. doi: 10.1183/13993003.02182-202010.1183/13993003.02182-2020.Shareable1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Huang Z., et al. Anti-TNF-alpha therapy improves Treg and suppresses Teff in patients with rheumatoid arthritis. Cell Immunol. 2012;279(1):25–29. doi: 10.1016/j.cellimm.2012.09.001. [DOI] [PubMed] [Google Scholar]
- 33.Zavvar M., Abdolmaleki M., Farajifard H., Noorbakhsh F., Azadmanesh K., Vojgani M., Hossein Nicknam M. Collagen II-primed Foxp3 Transduced T Cells Ameliorate Collagen-induced Arthritis in Rats: The Effect of Antigenic Priming on T Regulatory Cell Function. Iran J Allergy Asthma Immunol. 2018 doi: 10.18502/ijaai.v17i4.95. [DOI] [PubMed] [Google Scholar]
- 34.Zavvar M., Assadiasl S., Soleimanifar N., Pakdel F.D., Abdolmohammadi K., Fatahi Y., Abdolmaleki M., Baghdadi H., Tayebi L., Nicknam M.H. Gene therapy in rheumatoid arthritis: Strategies to select therapeutic genes. J Cell Physiol. 2019;234(10):16913–16924. doi: 10.1002/jcp.28392. [DOI] [PubMed] [Google Scholar]
- 35.Liu Y.u., Qi G., Bellanti J.A., Moser R., Ryffel B., Zheng S.G. Regulatory T cells: A potential weapon to combat COVID-19? MedComm (Beijing) 2020;1(2):157–164. doi: 10.1002/mco2.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Josefowicz S.Z., Lu L.-F., Rudensky A.Y. Regulatory T cells: mechanisms of differentiation and function. Annu Rev Immunol. 2012;30(1):531–564. doi: 10.1146/annurev.immunol.25.022106.141623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Collison L.W., Delgoffe G.M., Guy C.S., Vignali K.M., Chaturvedi V., Fairweather DeLisa, Satoskar A.R., Garcia K.C., Hunter C.A., Drake C.G., Murray P.J., Vignali D.A.A. The composition and signaling of the IL-35 receptor are unconventional. Nat Immunol. 2012;13(3):290–299. doi: 10.1038/ni.2227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bao C., Tao X., Cui W., Hao Y., Zheng S., Yi B., Pan T., Young K.H., Qian W. Natural killer cells associated with SARS-CoV-2 viral RNA shedding, antibody response and mortality in COVID-19 patients. Exp Hematol Oncol. 2021;10(1) doi: 10.1186/s40164-021-00199-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zheng M., Gao Y., Wang G., Song G., Liu S., Sun D., Xu Y., Tian Z. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell Mol Immunol. 2020;17(5):533–535. doi: 10.1038/s41423-020-0402-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Osman M., Faridi R.M., Sligl W., Shabani-Rad M.-T., Dharmani-Khan P., Parker A., Kalra A., Tripathi M.B., Storek J., Cohen Tervaert J.W., Khan F.M. Impaired natural killer cell counts and cytolytic activity in patients with severe COVID-19. Blood Adv. 2020;4(20):5035–5039. doi: 10.1182/bloodadvances.2020002650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Björkström N.K., Strunz B., Ljunggren H.-G. Natural killer cells in antiviral immunity. Nat Rev Immunol. 2022;22(2):112–123. doi: 10.1038/s41577-021-00558-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Littwitz E., et al. Distinct roles of NK cells in viral immunity during different phases of acute Friend retrovirus infection. Retrovirology. 2013;10:127. doi: 10.1186/1742-4690-10-127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ahmed F., Jo D.H., Lee S.H. Can Natural Killer Cells Be a Principal Player in Anti-SARS-CoV-2 Immunity? Front Immunol. 2020;11 doi: 10.3389/fimmu.2020.586765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.van Eeden C., Khan L., Osman M.S., Cohen Tervaert J.W. Natural Killer Cell Dysfunction and Its Role in COVID-19. Int J Mol Sci. 2020;21(17):6351. doi: 10.3390/ijms21176351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Coelho A., et al. Mesenchymal Stem Cells (MSCs) as a Potential Therapeutic Strategy in COVID-19 Patients: Literature Research. Front Cell Dev Biol. 2020;8 doi: 10.3389/fcell.2020.602647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Akhondzadeh F., Kadkhodaee M., Seifi B., Ashabi G., Kianian F., Abdolmohammadi K., Izad M., Adelipour M., Ranjbaran M. Adipose-Derived Mesenchymal Stem Cells and Conditioned Medium Attenuate the Memory Retrieval Impairment During Sepsis in Rats. Mol Neurobiol. 2020;57(9):3633–3645. doi: 10.1007/s12035-020-01991-6. [DOI] [PubMed] [Google Scholar]
- 47.Abdolmohammadi K., Mahmoudi T., Nojehdehi S., Tayebi L., Hashemi S.M., Noorbakhsh F., Abdollahi A., Soleimani M., Nikbin B., Nicknam M.H. Effect of Hypoxia Preconditioned Adipose-Derived Mesenchymal Stem Cell Conditioned Medium on Cerulein-Induced Acute Pancreatitis in Mice. Adv Pharm Bull. 2020;10(2):297–306. doi: 10.34172/apb.2020.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mahmoudi T., Abdolmohammadi K., Bashiri H., Mohammadi M., Rezaie M.J., Fathi F., Fakhari S., Rezaee M.A., Jalili A., Rahmani M.R., Tayebi L. Hydrogen Peroxide Preconditioning Promotes Protective Effects of Umbilical Cord Vein Mesenchymal Stem Cells in Experimental Pulmonary Fibrosis. Adv Pharm Bull. 2020;10(1):72–80. doi: 10.15171/apb.2020.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Murphy, M.B., K. Moncivais, and A.I. Caplan, Mesenchymal stem cells: environmentally responsive therapeutics for regenerative medicine. Exp Mol Med, 2013. 45: p. e54. [DOI] [PMC free article] [PubMed]
- 50.Pittenger M.F., et al. Mesenchymal stem cell perspective: cell biology to clinical progress. NPJ Regen Med. 2019;4:22. doi: 10.1038/s41536-019-0083-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chow L., Johnson V., Impastato R., Coy J., Strumpf A., Dow S. Antibacterial activity of human mesenchymal stem cells mediated directly by constitutively secreted factors and indirectly by activation of innate immune effector cells. Stem Cells Transl Med. 2020;9(2):235–249. doi: 10.1002/sctm.19-0092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Galipeau J., Sensébé L. Mesenchymal Stromal Cells: Clinical Challenges and Therapeutic Opportunities. Cell Stem Cell. 2018;22(6):824–833. doi: 10.1016/j.stem.2018.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ni K.e., Liu M., Zheng J., Wen L., Chen Q., Xiang Z., Lam K.-T., Liu Y., Chan G.-F., Lau Y.-L., Tu W. PD-1/PD-L1 Pathway Mediates the Alleviation of Pulmonary Fibrosis by Human Mesenchymal Stem Cells in Humanized Mice. Am J Respir Cell Mol Biol. 2018;58(6):684–695. doi: 10.1165/rcmb.2017-0326OC. [DOI] [PubMed] [Google Scholar]
- 54.Tzouvelekis A., et al. Mesenchymal Stem Cells for the Treatment of Idiopathic Pulmonary Fibrosis. Front Med (Lausanne) 2018;5:142. doi: 10.3389/fmed.2018.00142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Cao X. COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol. 2020;20(5):269–270. doi: 10.1038/s41577-020-0308-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Dominici M., Le Blanc K., Mueller I., Slaper-Cortenbach I., Marini F.C., Krause D.S., Deans R.J., Keating A., Prockop D.J., Horwitz E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315–317. doi: 10.1080/14653240600855905. [DOI] [PubMed] [Google Scholar]
- 57.Leng Z., Zhu R., Hou W., Feng Y., Yang Y., Han Q., Shan G., Meng F., Du D., Wang S., Fan J., Wang W., Deng L., Shi H., Li H., Hu Z., Zhang F., Gao J., Liu H., Li X., Zhao Y., Yin K., He X., Gao Z., Wang Y., Yang B.o., Jin R., Stambler I., Lim L.W., Su H., Moskalev A., Cano A., Chakrabarti S., Min K.-J., Ellison-Hughes G., Caruso C., Jin K., Zhao R.C. Transplantation of ACE2(-) Mesenchymal Stem Cells Improves the Outcome of Patients with COVID-19 Pneumonia. Aging Dis. 2020;11(2):216. doi: 10.14336/AD.2020.0228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wu X., Dao Thi V.L., Huang Y., Billerbeck E., Saha D., Hoffmann H.-H., Wang Y., Silva L.A.V., Sarbanes S., Sun T., Andrus L., Yu Y., Quirk C., Li M., MacDonald M.R., Schneider W.M., An X., Rosenberg B.R., Rice C.M. Intrinsic Immunity Shapes Viral Resistance of Stem Cells. Cell. 2018;172(3):423–438.e25. doi: 10.1016/j.cell.2017.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Nasef A., Mazurier C., Bouchet S., François S., Chapel A., Thierry D., Gorin N.-C., Fouillard L. Leukemia inhibitory factor: Role in human mesenchymal stem cells mediated immunosuppression. Cell Immunol. 2008;253(1-2):16–22. doi: 10.1016/j.cellimm.2008.06.002. [DOI] [PubMed] [Google Scholar]
- 60.Meisel R., Zibert A., Laryea M., Göbel U., Däubener W., Dilloo D. Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase-mediated tryptophan degradation. Blood. 2004;103(12):4619–4621. doi: 10.1182/blood-2003-11-3909. [DOI] [PubMed] [Google Scholar]
- 61.Meisel R., Brockers S., Heseler K., Degistirici Ö., Bülle H., Woite C., Stuhlsatz S., Schwippert W., Jäger M., Sorg R., Henschler R., Seissler J., Dilloo D., Däubener W. Human but not murine multipotent mesenchymal stromal cells exhibit broad-spectrum antimicrobial effector function mediated by indoleamine 2,3-dioxygenase. Leukemia. 2011;25(4):648–654. doi: 10.1038/leu.2010.310. [DOI] [PubMed] [Google Scholar]
- 62.Thanunchai M., Hongeng S., Thitithanyanont A. Mesenchymal Stromal Cells and Viral Infection. Stem Cells Int. 2015;2015 doi: 10.1155/2015/860950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Verma Y.K., Verma R., Tyagi N., Behl A., Kumar S., Gangenahalli G.U. COVID-19 and its Therapeutics: Special Emphasis on Mesenchymal Stem Cells Based Therapy. Stem Cell Rev Rep. 2021;17(1):113–131. doi: 10.1007/s12015-020-10037-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Walter J., Ware L.B., Matthay M.A. Mesenchymal stem cells: mechanisms of potential therapeutic benefit in ARDS and sepsis. Lancet Respir Med. 2014;2(12):1016–1026. doi: 10.1016/S2213-2600(14)70217-6. [DOI] [PubMed] [Google Scholar]
- 65.Moradinasab S., et al. Mesenchymal stromal/stem cells (MSCs) and MSC-derived extracellular vesicles in COVID-19-induced ARDS: Mechanisms of action, research progress, challenges, and opportunities. Int Immunopharmacol. 2021;97 doi: 10.1016/j.intimp.2021.107694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lee J.W., Krasnodembskaya A., McKenna D.H., Song Y., Abbott J., Matthay M.A. Therapeutic effects of human mesenchymal stem cells in ex vivo human lungs injured with live bacteria. Am J Respir Crit Care Med. 2013;187(7):751–760. doi: 10.1164/rccm.201206-0990OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Hu S., Li J., Xu X., Liu A., He H., Xu J., Chen Q., Liu S., Liu L., Qiu H., Yang Y.i. The hepatocyte growth factor-expressing character is required for mesenchymal stem cells to protect the lung injured by lipopolysaccharide in vivo. Stem Cell Res Ther. 2016;7(1) doi: 10.1186/s13287-016-0320-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Yang Y., et al. The Vascular Endothelial Growth Factors-Expressing Character of Mesenchymal Stem Cells Plays a Positive Role in Treatment of Acute Lung Injury In Vivo. Mediators Inflamm. 2016;2016:2347938. doi: 10.1155/2016/2347938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Harrell C.R., et al. Mesenchymal Stem Cell-Based Therapy of Inflammatory Lung Diseases: Current Understanding and Future Perspectives. Stem Cells Int. 2019;2019:4236973. doi: 10.1155/2019/4236973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Esposito M., et al. Differentiation of human umbilical cord-derived mesenchymal stem cells, WJ-MSCs, into chondrogenic cells in the presence of pulsed electromagnetic fields. In Vivo. 2013;27(4):495–500. [PubMed] [Google Scholar]
- 71.Shu Y.i., Yang C., Ji X., Zhang L., Bi Y., Yang K.e., Gong M., Liu X., Guo Q.i., Su Y., Qu X., Nan G., Zhao C., Zeng Z., Yu X., Zhang R., Yan S., Lei J., Wu K.e., Wu Y., An L., Huang S., Gong C., Yuan C., Liu W., Huang B.o., Feng Y., Zhang B.o., Dai Z., Shen Y.i., Luo W., Wang X.i., Haydon R.C., Luu H.H., Reid R.R., Wolf J.M., Lee M.J., He T.-C., Li Y. Reversibly immortalized human umbilical cord-derived mesenchymal stem cells (UC-MSCs) are responsive to BMP9-induced osteogenic and adipogenic differentiation. J Cell Biochem. 2018;119(11):8872–8886. doi: 10.1002/jcb.27140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wang J., Jiang M., Chen X., Montaner L.J. Cytokine storm and leukocyte changes in mild versus severe SARS-CoV-2 infection: Review of 3939 COVID-19 patients in China and emerging pathogenesis and therapy concepts. J Leukoc Biol. 2020;108(1):17–41. doi: 10.1002/JLB.3COVR0520-272R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Song N.a., Wakimoto H., Rossignoli F., Bhere D., Ciccocioppo R., Chen K.-S., Khalsa J.K., Mastrolia I., Samarelli A.V., Dominici M., Shah K. Mesenchymal stem cell immunomodulation: In pursuit of controlling COVID-19 related cytokine storm. Stem Cells. 2021;39(6):707–722. doi: 10.1002/stem.3354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Furlani D., Ugurlucan M., Ong LeeLee, Bieback K., Pittermann E., Westien I., Wang W., Yerebakan C., Li W., Gaebel R., Li R.-k., Vollmar B., Steinhoff G., Ma N. Is the intravascular administration of mesenchymal stem cells safe? Mesenchymal stem cells and intravital microscopy. Microvasc Res. 2009;77(3):370–376. doi: 10.1016/j.mvr.2009.02.001. [DOI] [PubMed] [Google Scholar]
- 75.Leibacher J., Henschler R. Biodistribution, migration and homing of systemically applied mesenchymal stem/stromal cells. Stem Cell Res Ther. 2016;7:7. doi: 10.1186/s13287-015-0271-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wang S.H., et al. Combating COVID-19 With Mesenchymal Stem/Stromal Cell Therapy: Promise and Challenges. Front Cell Dev Biol. 2020;8 doi: 10.3389/fcell.2020.627414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Liu S., Peng D., Qiu H., Yang K.e., Fu Z., Zou L. Mesenchymal stem cells as a potential therapy for COVID-19. Stem Cell Res Ther. 2020;11(1) doi: 10.1186/s13287-020-01678-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Chan J.-W., Choi G.-Y., Tsang A.-L., Tee K.-M., Lam H.-Y., Yip C.-Y., To K.-W., Cheng V.-C., Yeung M.-L., Lau S.-P., Woo P.-Y., Chan K.-H., Tang B.-F., Yuen K.-Y., Loeffelholz M.J. Development and Evaluation of Novel Real-Time Reverse Transcription-PCR Assays with Locked Nucleic Acid Probes Targeting Leader Sequences of Human-Pathogenic Coronaviruses. J Clin Microbiol. 2015;53(8):2722–2726. doi: 10.1128/JCM.01224-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Khatri M., Richardson L.A., Meulia T. Mesenchymal stem cell-derived extracellular vesicles attenuate influenza virus-induced acute lung injury in a pig model. Stem Cell Res Ther. 2018;9(1):17. doi: 10.1186/s13287-018-0774-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Collin M., Ginhoux F. Human dendritic cells. Semin Cell Dev Biol. 2019;86:1–2. doi: 10.1016/j.semcdb.2018.04.015. [DOI] [PubMed] [Google Scholar]
- 81.Cui J., Li F., Shi Z.-L. Origin and evolution of pathogenic coronaviruses. Nature Reviews Microbiology. 2019;17(3):181–192. doi: 10.1038/s41579-018-0118-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Schulte-Schrepping, J., et al., Severe COVID-19 is marked by a dysregulated myeloid cell compartment. Cell, 2020. 182(6): p. 1419-1440. e23. [DOI] [PMC free article] [PubMed]
- 83.Zhou R., To K.-W., Wong Y.-C., Liu L.i., Zhou B., Li X., Huang H., Mo Y., Luk T.-Y., Lau T.-K., Yeung P., Chan W.-M., Wu A.-L., Lung K.-C., Tsang O.-Y., Leung W.-S., Hung I.-N., Yuen K.-Y., Chen Z. Acute SARS-CoV-2 Infection Impairs Dendritic Cell and T Cell Responses. Immunity. 2020;53(4):864–877.e5. doi: 10.1016/j.immuni.2020.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Han J., Sun J., Zhang G., Chen H. DCs-based therapies: potential strategies in severe SARS-CoV-2 infection. Int J Med Sci. 2021;18(2):406–418. doi: 10.7150/ijms.47706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Hawiger D., Inaba K., Dorsett Y., Guo M., Mahnke K., Rivera M., Ravetch J.V., Steinman R.M., Nussenzweig M.C. Dendritic cells induce peripheral T cell unresponsiveness under steady state conditions in vivo. The Journal of experimental medicine. 2001;194(6):769–780. doi: 10.1084/jem.194.6.769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Palucka K., Banchereau J. Dendritic-cell-based therapeutic cancer vaccines. Immunity. 2013;39(1):38–48. doi: 10.1016/j.immuni.2013.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Merad M., Martin J.C. Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nature Reviews Immunology. 2020;20(6):355–362. doi: 10.1038/s41577-020-0331-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Balan S., Kale V.P., Limaye L.S. A simple two-step culture system for the large-scale generation of mature and functional dendritic cells from umbilical cord blood CD34+ cells. Transfusion. 2009;49(10):2109–2121. doi: 10.1111/j.1537-2995.2009.02231.x. [DOI] [PubMed] [Google Scholar]
- 89.Zhao J., Wang W., Wang G.-F., Li Y., Zhuang H., Xu X., Ren F., Zhao Z., Gao X.-M. Development and evaluation of an enzyme-linked immunosorbent assay for detection of antibodies against the spike protein of SARS-coronavirus. Journal of clinical virology : the official publication of the Pan American Society for Clinical Virology. 2005;33(1):12–18. doi: 10.1016/j.jcv.2004.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Zhao J., Wang W., Yuan Z., Jia R., Zhao Z., Xu X., Lv P., Zhang Y., Jiang C., Gao X.-M. A study on antigenicity and receptor-binding ability of fragment 450–650 of the spike protein of SARS coronavirus. Virology. 2007;359(2):362–370. doi: 10.1016/j.virol.2006.09.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Zhao J.-C., Zhao Z.-D., Wang W., Gao X.-M. Prokaryotic expression, refolding, and purification of fragment 450–650 of the spike protein of SARS-coronavirus. Protein expression and purification. 2005;39(2):169–174. doi: 10.1016/j.pep.2004.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Wysocki P.J., et al. Genetically modified dendritic cells–a new, promising cancer treatment strategy? Expert Opin Biol Ther. 2002;2(8):835–845. doi: 10.1517/14712598.2.8.835. [DOI] [PubMed] [Google Scholar]
- 93.Tuyaerts S., Aerts J.L., Corthals J., Neyns B., Heirman C., Breckpot K., Thielemans K., Bonehill A. Current approaches in dendritic cell generation and future implications for cancer immunotherapy. Cancer Immunol Immunother. 2007;56(10):1513–1537. doi: 10.1007/s00262-007-0334-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Ventura A., Vassall A., Robinson E., Filler R., Hanlon D., Meeth K., Ezaldein H., Girardi M., Sobolev O., Bosenberg M.W., Edelson R.L. Extracorporeal Photochemotherapy Drives Monocyte-to-Dendritic Cell Maturation to Induce Anticancer Immunity. Cancer Res. 2018;78(14):4045–4058. doi: 10.1158/0008-5472.CAN-18-0171. [DOI] [PubMed] [Google Scholar]
- 95.Mu W., Carrillo M.A., Kitchen S.G. Engineering CAR T Cells to Target the HIV Reservoir. Front Cell Infect Microbiol. 2020;10:410. doi: 10.3389/fcimb.2020.00410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Oh H.-L.- J., Chia A., Chang C.X.L., Leong H.N., Ling K.L., Grotenbreg G.M., Gehring A.J., Tan Y.J., Bertoletti A. Engineering T cells specific for a dominant severe acute respiratory syndrome coronavirus CD8 T cell epitope. J Virol. 2011;85(20):10464–10471. doi: 10.1128/JVI.05039-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Brudno J.N., Kochenderfer J.N. Toxicities of chimeric antigen receptor T cells: recognition and management. Blood. 2016;127(26):3321–3330. doi: 10.1182/blood-2016-04-703751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Rubin D.B., Danish H.H., Ali A.B., Li K., LaRose S., Monk A.D., Cote D.J., Spendley L., Kim A.H., Robertson M.S., Torre M., Smith T.R., Izzy S., Jacobson C.A., Lee J.W., Vaitkevicius H. Neurological toxicities associated with chimeric antigen receptor T-cell therapy. Brain. 2019;142(5):1334–1348. doi: 10.1093/brain/awz053. [DOI] [PubMed] [Google Scholar]
- 99.Morgan R.A., Yang J.C., Kitano M., Dudley M.E., Laurencot C.M., Rosenberg S.A. Case report of a serious adverse event following the administration of T cells transduced with a chimeric antigen receptor recognizing ERBB2. Mol Ther. 2010;18(4):843–851. doi: 10.1038/mt.2010.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Bachanova V., Perales M.A., Abramson J.S. Modern management of relapsed and refractory aggressive B-cell lymphoma: A perspective on the current treatment landscape and patient selection for CAR T-cell therapy. Blood Rev. 2020;40 doi: 10.1016/j.blre.2019.100640. [DOI] [PubMed] [Google Scholar]
- 101.Ni L., Ye F., Cheng M.-L., Feng Y.u., Deng Y.-Q., Zhao H., Wei P., Ge J., Gou M., Li X., Sun L., Cao T., Wang P., Zhou C., Zhang R., Liang P., Guo H., Wang X., Qin C.-F., Chen F., Dong C. Detection of SARS-CoV-2-Specific Humoral and Cellular Immunity in COVID-19 Convalescent Individuals. Immunity. 2020;52(6):971–977.e3. doi: 10.1016/j.immuni.2020.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Harris K.M., et al. Virus-Specific T Cells: Current and Future Use in Primary Immunodeficiency Disorders. The Journal of Allergy and Clinical Immunology. In Practice. 2019;7(3):809–818. doi: 10.1016/j.jaip.2018.10.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Keller M.D., Harris K.M., Jensen-Wachspress M.A., Kankate V.V., Lang H., Lazarski C.A., Durkee-Shock J., Lee P.-H., Chaudhry K., Webber K., Datar A., Terpilowski M., Reynolds E.K., Stevenson E.M., Val S., Shancer Z., Zhang N., Ulrey R., Ekanem U., Stanojevic M., Geiger A., Liang H., Hoq F., Abraham A.A., Hanley P.J., Cruz C.R., Ferrer K., Dropulic L., Gangler K., Burbelo P.D., Jones R.B., Cohen J.I., Bollard C.M. SARS-CoV-2-specific T cells are rapidly expanded for therapeutic use and target conserved regions of the membrane protein. Blood. 2020;136(25):2905–2917. doi: 10.1182/blood.2020008488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Catakovic K., Klieser E., Neureiter D., Geisberger R. T cell exhaustion: from pathophysiological basics to tumor immunotherapy. Cell Commun Signal. 2017;15(1) doi: 10.1186/s12964-016-0160-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Pezeshki P.S., Rezaei N. Immune checkpoint inhibition in COVID-19: risks and benefits. Expert Opin Biol Ther. 2021;21(9):1173–1179. doi: 10.1080/14712598.2021.1887131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Aghbash P.S., et al. SARS-CoV-2 infection: The role of PD-1/PD-L1 and CTLA-4 axis. Life Sci. 2021;270 doi: 10.1016/j.lfs.2021.119124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Shah V.K., et al. Overview of Immune Response During SARS-CoV-2 Infection: Lessons From the Past. Front Immunol. 2020;11:1949. doi: 10.3389/fimmu.2020.01949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Chamoto K., Al-Habsi M., Honjo T. Role of PD-1 in Immunity and Diseases. Curr Top Microbiol Immunol. 2017;410:75–97. doi: 10.1007/82_2017_67. [DOI] [PubMed] [Google Scholar]
- 109.Sabbatino, F., et al., PD-L1 Dysregulation in COVID-19 Patients. Front Immunol, 2021. 12: p. 695242. [DOI] [PMC free article] [PubMed]
- 110.Loretelli, C., et al., PD-1 blockade counteracts post-COVID-19 immune abnormalities and stimulates the anti-SARS-CoV-2 immune response. JCI Insight, 2021. 6(24). [DOI] [PMC free article] [PubMed]
- 111.Vivarelli, S., et al., Cancer Management during COVID-19 Pandemic: Is Immune Checkpoint Inhibitors-Based Immunotherapy Harmful or Beneficial? Cancers (Basel), 2020. 12(8). [DOI] [PMC free article] [PubMed]
- 112.Qian W., Ye Y., Zuo L., Song T., Xu Q., Wang Y., Qian J., Tian Y. Immune checkpoint inhibitors use and effects on prognosis of COVID-19 infection: a systematic review and meta-analysis. Immunotherapy. 2021;13(15):1271–1282. doi: 10.2217/imt-2021-0007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Vivarelli S., Falzone L., Torino F., Scandurra G., Russo G., Bordonaro R., Pappalardo F., Spandidos D., Raciti G., Libra M. Immune-checkpoint inhibitors from cancer to COVID19: A promising avenue for the treatment of patients with COVID19 (Review) Int J Oncol. 2021;58(2):145–157. doi: 10.3892/ijo.2020.5159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Rossi E., Schinzari G., Tortora G. Pneumonitis from immune checkpoint inhibitors and COVID-19: current concern in cancer treatment. J Immunother Cancer. 2020;8(2) doi: 10.1136/jitc-2020-000952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Sullivan R.J., Johnson D.B., Rini B.I., Neilan T.G., Lovly C.M., Moslehi J.J., Reynolds K.L. COVID-19 and immune checkpoint inhibitors: initial considerations. J Immunother Cancer. 2020;8(1) doi: 10.1136/jitc-2020-000933. [DOI] [PMC free article] [PubMed] [Google Scholar]