Highlights
-
•
Focused TTE is a critical diagnostic tool to limit exposure in COVID-19.
-
•
Largest COVID-19 cohort to date showing strong prognostic power of RV parameters.
-
•
RV dilation significantly associated with increased mortality in COVID-19.
-
•
RV dysfunction significantly associated with shock in COVID-19.
-
•
LV abnormalities did not provide prognostic information in COVID-19.
Keywords: Echocardiography, COVID-19, Mortality, Prognosis
Abbreviations: ACE2, angiotensin-converting enzyme 2; ARDS, acute respiratory distress syndrome; ASE, American Society of Echocardiography; BNP, B-type natriuretic peptide; CCE, critical care echocardiography; CI, confidence interval; COVID-19, coronavirus disease 2019; fTTE, focused transthoracic echocardiography; GLS, global longitudinal strain; IQR, interquartile range; IRB, institutional review board; LV, left ventricle / left ventricular; LVEDD, left ventricular end diastolic diameter; LVEF, left ventricular ejection fraction; OR, odds ratio; PA, pulmonary artery; PCR, polymerase chain reaction; PLAX, parasternal long-axis view; POCUS, point-of-care ultrasound; PPE, personal protective equipment; PSAX, parasternal short-axis view; PUI, patient under investigation; RRT, renal replacement therapy; RV, right ventricle / right ventricular; RVLS, right ventricular longitudinal strain; S’, peak lateral tricuspid annular systolic velocity; SARS-CoV-2, novel SARS coronavirus; TAPSE, tricuspid annular plane systolic excursion
Abstract
Background
Focused transthoracic echocardiography (fTTE) has emerged as a critical diagnostic tool during the COVID-19 pandemic, allowing for efficient cardiac imaging while minimizing staff exposure. The utility of fTTE in predicting clinical outcomes in COVID-19 remains under investigation.
Methods
We conducted a retrospective study of 2,266 hospitalized patients at Rush University Medical Center with COVID-19 infection between March and November 2020 who received a fTTE. fTTE data were analyzed for association with primary adverse outcomes (60-day mortality) and with secondary adverse outcomes (need for renal replacement therapy, need for invasive ventilation, shock, and venous thromboembolism).
Results
Of the 427 hospitalized patients who had a fTTE performed (mean 62 years, 43% female), 109 (26%) had died by 60 days. Among patients with an available fTTE measurement, right ventricular (RV) dilation was noted in 34% (106/309), 43% (166/386) had RV dysfunction, and 17% (72/421) had left ventricular (LV) dysfunction. In multivariable models accounting for fTTE data, RV dilation was significantly associated with 60-day mortality (OR 1.93 [CI 1.13–3.3], p = 0.016). LV dysfunction was not significantly associated with 60-day mortality (OR 0.95 [CI: 0.51–1.78], p = 0.87).
Conclusions
Abnormalities in RV echocardiographic parameters are adverse prognosticators in COVID-19 disease. Patients with RV dilation experienced double the risk for 60-day mortality due to COVID-19. To our knowledge, this is the largest study to date that highlights the adverse prognostic implications of RV dilation as determined through fTTE in hospitalized COVID-19 patients.
1. Introduction
Several recent studies have highlighted the various cardiovascular manifestations of the novel SARS coronavirus (SARS-CoV-2) infection, including myocarditis, heart failure, hypertension, arrhythmias, and acute coronary syndrome.[1], [2] Given the heightened risk of cardiovascular complications from coronavirus disease 2019 (COVID-19), especially among at-risk populations such as the elderly, immunocompromised, or those with pre-existing cardiac disease, echocardiography has proven to be an invaluable tool in enabling early detection of cardiovascular involvement and to guide critical decision-making. Rush University System for Health has been at the forefront of pandemic response in the state of Illinois. It served as a regional and referral center, catering to the high case numbers in Chicago and surrounding areas.[3] Thus, in our institution, the pandemic forced a re-evaluation of existing protocols to maximize benefits to patients and minimize risks to healthcare workers, congruent to the guidelines released by the American Society of Echocardiography (ASE).[4], [5] This was implemented through use of an abbreviated focused transthoracic echocardiography (fTTE) protocol, and stringent appropriate use screening, which allowed for the safest and most efficient use of fTTE technology.[3]
Focused TTE is a powerful diagnostic tool in managing patients with COVID-19, especially for those at risk for severe cardiovascular complications. However, there remains limited knowledge on specific echocardiographic findings predictive of adverse outcomes in COVID-19 disease in hospitalized adults. In the ECHOVID-19 study of 214 hospitalized patients in Copenhagen, Denmark, who underwent echocardiographic examination, RV and LV function were significantly impaired compared to matched controls.[6] Reduced global longitudinal strain (GLS), as a marker of left heart function, was also found to be independently associated with mortality from COVID-19.[7] A few smaller studies have also shown a significant association of RV dysfunction with increased mortality. Specifically, an abnormal tricuspid annular plane systolic excursion (TAPSE) and reduced RV longitudinal strain were powerful predictors of adverse outcomes, carrying approximately 20–30% elevated risk of mortality.[6], [7], [8]
This study aimed to evaluate the association of various echocardiographic parameters obtained from a sonographer-performed fTTE with 60-day mortality in COVID-19 disease. We also examined the association of echocardiographic parameters with defined secondary adverse outcomes such as the need for renal replacement therapy (RRT), shock, need for invasive ventilation, and venous thromboembolism. The goal was to determine the echocardiographic abnormalities that are predictive of adverse outcomes in patients who are hospitalized for COVID-19 infection to provide critical insight from risk stratification, prognostic, and possibly, therapeutic standpoints. To our knowledge, this is the largest retrospective study examining the association of multiple echocardiographic abnormalities obtained from fTTE with 60-day adverse clinical outcomes.
2. Methods
2.1. Patient population
We conducted a retrospective study of 2,266 hospitalized adult patients, 18 years or older, at Rush University System with COVID-19 infection between March and November 2020. Rush University System for Health includes the Rush University Medical Center, a 664-bed tertiary academic medical center, and the Rush Oak Park Hospital, a 201-bed affiliated community hospital in Illinois, USA. For patients included multiple times in the registry, only the last hospitalization was included in the analysis.
2.2. Study conduction and data extraction
The study protocol was approved by the Institutional Review Board (IRB). The overall study cohort is comprised of a set of patients identified as Patients Under Investigation (PUIs) by a custom chronic disease registry that Rush implemented to track its COVID-19 patient population from the onset of the outbreak. Clinical data for the patient cohort was obtained from documentation queries executed on clinical data warehouses (Caboodle and Clarity) in Rush Epic electronic medical record application. The cohort was then filtered to include only confirmed COVID-19 positive patients either by polymerase chain reaction (PCR) testing or point-of-care rapid testing.
2.3. Acquisition of fTTEs
Since the time of the COVID-19 outbreak, Rush has been central in piloting the use of point-of-care ultrasound and fTTE to achieve focused and high-yield cardiovascular evaluations. We included all in-patients who received a TTE. TTE orders placed by the inpatient service based on specific clinical indications went through an additional screening process by Cardiology physicians to determine appropriate use.[3] Appropriate use was defined as critically ill cases where echocardiographic knowledge was deemed to affect clinical management. This included but were not limited to cases where there was concern for decompensated heart failure, myocardial infarction, or acute pulmonary embolism. An abbreviated protocol enabled quick bedside evaluations by Cardiology fellows, attendings, or sonographers while reducing the time of exposure and number of personal protective equipment utilized. The Rush fTTE COVID protocol is comprised of the following: (1) parasternal long-axis view (PLAX), (2) parasternal short-axis view (PSAX) at the basal, mid-, and apical views, (3) apical 4-chamber/apical 5-chamber views, (4) apical 3-chamber views, (5) apical 2-chamber view, and (6) subcostal view.[3] Thereafter, all subsequent TTE data were manually reviewed and extracted. Only the first fTTE was analyzed per hospitalization. All TTEs were manually reviewed by two physicians who were blinded to the patients’ clinical history. TTE data might be missing when the image quality did not allow for measurement of the specific parameters or when the views were not acquired by the cardiac sonographer or cardiologist performing the study.
2.4. Outcomes and measures
The primary outcome was 60-day mortality. The secondary outcomes were adverse events that occurred within 60 days of admission, namely the need for renal replacement therapy, shock, need for invasive ventilation and venous thromboembolism. Venous thromboembolism was defined as the occurrence of stroke, pulmonary embolism, or deep venous thrombosis. Abnormal RV and LV size and function definitions were derived from current chamber quantification guidelines set forth by the American Society of Echocardiography.[9] In brief, abnormal RV size was defined as RV basal diameter > 41 mm; abnormal RV function was defined as TAPSE < 17 mm, S’ < 9.5 cm/s, or qualitative RV dysfunction assessed by an experienced attending cardiologist. Abnormal LV ejection fraction (LVEF) was defined as LVEF < 50% and LV dilation as left ventricular end diastolic diameter (LVEDD) > 58.4 mm for males or > 52.2 mm for females.
2.5. Statistical analysis
Categorical variables are represented as numbers and percentages. Continuous variables are represented as medians with interquartile ranges (IQR). An independent Wilcoxon rank sum test was used to compare continuous variables. A Chi-square test of independence without continuity correction was used to compare categorical variables with >5 occurrence per cell and a Fisher exact test to compare categorical variables with <5 occurrence per cells (R package gtsummary).[10] Before logistic regression analyses, missing data for important covariables was imputed using multiple imputation with chain equations (R package MICE) for 10 datasets and 10 iterations.[11] All baseline data listed in Table 1 were used for imputation. Outcome data were used for the imputation but were not themselves imputed as complete data was available for all patients. Convergence of the algorithm and distribution of the imputed data points were verified in diagnostic plots.
Table 1.
Baseline Demographics and Outcomes in All Patients with a TTE Conducted during Admission.
| Characteristic | N | N = 4271 |
|---|---|---|
| Age | 427 | 61.87 (15.03) |
| Sex | 427 | |
| Male | 243 / 427 (57%) | |
| Female | 184 / 427 (43%) | |
| Atrial fibrillation | 421 | 136 / 421 (32%) |
| (Missing) | 6 | |
| CAD | 421 | 183 / 421 (43%) |
| (Missing) | 6 | |
| HTN | 421 | 321 / 421 (76%) |
| (Missing) | 6 | |
| Valvular disease | 421 | 138 / 421 (33%) |
| (Missing) | 6 | |
| CHF | 425 | 81 / 425 (19%) |
| (Missing) | 2 | |
| CKD | 425 | 122 / 425 (29%) |
| (Missing) | 2 | |
| CKD on dialysis | 421 | 82 / 421 (19%) |
| (Missing) | 6 | |
| COPD | 421 | 46 / 421 (11%) |
| (Missing) | 6 | |
| DM | 421 | 238 / 421 (57%) |
| (Missing) | 6 | |
| HLD | 421 | 251 / 421 (60%) |
| (Missing) | 6 | |
| Asthma | 421 | 53 / 421 (13%) |
| (Missing) | 6 | |
| Cancer | 421 | 56 / 421 (13%) |
| (Missing) | 6 | |
| Stroke | 421 | 95 / 421 (23%) |
| (Missing) | 6 | |
| Pacemaker or ICD | 421 | 19 / 421 (4.5%) |
| (Missing) | 6 | |
| RV size | 309 | 38.66 (9.10) |
| (Missing) | 118 | |
| TASPE | 268 | 19.75 (5.46) |
| (Missing) | 159 | |
| Elevated PA pressure | 260 | |
| Normal | 150 / 260 (58%) | |
| Elevated | 110 / 260 (42%) | |
| (Missing) | 167 | |
| PASP (measured) | 192 | 38.58 (15.11) |
| (Missing) | 235 | |
| RAP | 187 | 8.00 (3.00, 15.00) |
| (Missing) | 240 | |
| Dilated RV cavity | 427 | |
| Normal | 360 / 427 (84%) | |
| Dilated | 67 / 427 (16%) | |
| LVEF | 421 | 57.58 (11.56) |
| (Missing) | 6 | |
| LVEDD | 278 | 45.11 (7.74) |
| (Missing) | 149 | |
| Pericardial effusion (%) | 420 | |
| Absent | 416 / 420 (99%) | |
| Present | 4 / 420 (1.0%) | |
| (Missing) | 7 | |
| RV function (Abnormal S' or TAPSE or abnormal by visual estimation. Definition: TAPSE < 17 mm or S’ < 9.5 cm/s) | 386 | |
| Normal | 220 / 386 (57%) | |
| Abnormal | 166 / 386 (43%) | |
| (Missing) | 41 | |
| LVEDD (Definition: LVEDD > 58.4 mm (male) or > 52.2 mm (female)) | 278 | |
| Normal | 256 / 278 (92%) | |
| Abnormal | 22 / 278 (7.9%) | |
| (Missing) | 149 | |
| LVEF (Definition: LVEF < 50%) | 421 | |
| Normal | 349 / 421 (83%) | |
| Abnormal | 72 / 421 (17%) | |
| (Missing) | 6 | |
| RV size (Definition: RV basal diameter > 41 mm) | 309 | |
| Normal | 203 / 309 (66%) | |
| Abnormal | 106 / 309 (34%) | |
| (Missing) | 118 | |
| RV by visual estimation | 378 | |
| Normal | 302 / 378 (80%) | |
| Abnormal | 76 / 378 (20%) | |
| (Missing) | 49 | |
| Wall motion abnormalities | 404 | 56 / 404 (14%) |
| (Missing) | 23 | |
| Takotsubo or stress cardiomyopathy | 406 | 6 / 406 (1.5%) |
| (Missing) | 21 | |
| Composite outcome: Invasive ventilation, death at 60d, shock, thromboembolism or need for RRT | 427 | 249 / 427 (58%) |
| Death at 60 days | 427 | 109 / 427 (26%) |
| Invasive ventilation | 427 | 207 / 427 (48%) |
| RRT | 427 | 85 / 427 (20%) |
| Shock | 427 | 181 / 427 (42%) |
| Thromboembolic event | 427 | 42 / 427 (9.8%) |
| 1Median (p25, p75), mean (sd) or Frequency (%) | ||
Abbreviations: CAD, coronary artery disease; HTN, hypertension; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; DM, diabetes mellitus; HLD, hyperlipidemia; ICD, implantable cardioverter defibrillator; RV, right ventricular; TAPSE, tricuspid annular plane systolic excursion; PA, pulmonary artery; PASP, pulmonary artery systolic pressure; RAP, right atrial pressure; LVEF, left ventricular ejection fraction; LVEDD, left ventricular end diastolic diameter; S’, peak lateral tricuspid annular systolic velocity; RRT, renal replacement therapy.
Following imputation, logistic multivariable analyses were performed to evaluate the relative association of RV and LV parameters with the respective outcomes. The models were multivariable, assessing for RV and LV function and size simultaneously. This model design was chosen in order to assess the relative importance of RV against LV parameters for patient outcomes. Regression analysis was performed on all ten imputed datasets and results were pooled applying Rubin’s rule. The threshold for p-value significance was set to 0.05. All statistical analysis was performed using RStudio version 1.3 (Boston, Massachusetts).
3. Results
From March to November 2020, 2,266 patients were hospitalized with a confirmed COVID-19 infection. A total of 427 patients (18.8%) underwent a TTE during their hospital stay. The mean age was 61 years, and 43% were female. The tables show the baseline characteristics of the patients for the overall cohort (Table 1) and depending on the RV size (Table 2) and function (Supplemental Table 1).
Table 2.
Normal vs. Abnormal Right Ventricular size – Baseline Demographics and Outcomes.
| RV size |
|||||
|---|---|---|---|---|---|
| Variable | N | Overall, N = 3091 | Normal, N = 2031 | Abnormal, N = 1061 | p-value2 |
| Age | 309 | 61.44 (14.66) | 61.22 (15.21) | 61.88 (13.62) | >0.9 |
| Sex | 309 | <0.001 | |||
| Male | 176 / 309 (57%) | 100 / 203 (49%) | 76 / 106 (72%) | ||
| Female | 133 / 309 (43%) | 103 / 203 (51%) | 30 / 106 (28%) | ||
| Atrial fibrillation | 307 | 106 / 307 (35%) | 62 / 201 (31%) | 44 / 106 (42%) | 0.062 |
| (Missing) | 2 | 2 | 0 | ||
| CAD | 307 | 150 / 307 (49%) | 96 / 201 (48%) | 54 / 106 (51%) | 0.6 |
| (Missing) | 2 | 2 | 0 | ||
| HTN | 307 | 239 / 307 (78%) | 157 / 201 (78%) | 82 / 106 (77%) | 0.9 |
| (Missing) | 2 | 2 | 0 | ||
| Valvular disease | 307 | 116 / 307 (38%) | 73 / 201 (36%) | 43 / 106 (41%) | 0.5 |
| (Missing) | 2 | 2 | 0 | ||
| CHF | 308 | 66 / 308 (21%) | 40 / 202 (20%) | 26 / 106 (25%) | 0.3 |
| (Missing) | 1 | 1 | 0 | ||
| CKD | 308 | 93 / 308 (30%) | 57 / 202 (28%) | 36 / 106 (34%) | 0.3 |
| (Missing) | 1 | 1 | 0 | ||
| CKD on dialysis | 307 | 69 / 307 (22%) | 41 / 201 (20%) | 28 / 106 (26%) | 0.2 |
| (Missing) | 2 | 2 | 0 | ||
| COPD | 307 | 33 / 307 (11%) | 25 / 201 (12%) | 8 / 106 (7.5%) | 0.2 |
| (Missing) | 2 | 2 | 0 | ||
| DM | 307 | 176 / 307 (57%) | 120 / 201 (60%) | 56 / 106 (53%) | 0.2 |
| (Missing) | 2 | 2 | 0 | ||
| HLD | 307 | 187 / 307 (61%) | 125 / 201 (62%) | 62 / 106 (58%) | 0.5 |
| (Missing) | 2 | 2 | 0 | ||
| Asthma | 307 | 40 / 307 (13%) | 27 / 201 (13%) | 13 / 106 (12%) | 0.8 |
| (Missing) | 2 | 2 | 0 | ||
| Cancer | 307 | 43 / 307 (14%) | 32 / 201 (16%) | 11 / 106 (10%) | 0.2 |
| (Missing) | 2 | 2 | 0 | ||
| Stroke | 307 | 76 / 307 (25%) | 52 / 201 (26%) | 24 / 106 (23%) | 0.5 |
| (Missing) | 2 | 2 | 0 | ||
| Pacemaker or ICD | 307 | 16 / 307 (5.2%) | 9 / 201 (4.5%) | 7 / 106 (6.6%) | 0.4 |
| (Missing) | 2 | 2 | 0 | ||
| RV size | 309 | 38.66 (9.10) | 33.65 (5.56) | 48.27 (6.45) | <0.001 |
| TASPE | 261 | 19.78 (5.46) | 19.86 (5.27) | 19.66 (5.77) | 0.8 |
| (Missing) | 48 | 42 | 6 | ||
| Elevated PA pressure | 209 | 0.009 | |||
| Normal | 116 / 209 (56%) | 77 / 122 (63%) | 39 / 87 (45%) | ||
| Elevated | 93 / 209 (44%) | 45 / 122 (37%) | 48 / 87 (55%) | ||
| (Missing) | 100 | 81 | 19 | ||
| PASP (measured) | 141 | 40.51 (14.63) | 36.68 (11.53) | 45.40 (16.68) | <0.001 |
| (Missing) | 168 | 124 | 44 | ||
| RAP | 182 | 8.00 (3.00, 15.00) | 3.00 (3.00, 8.00) | 8.00 (3.00, 15.00) | 0.005 |
| (Missing) | 127 | 94 | 33 | ||
| Dilated RV cavity | 309 | <0.001 | |||
| Normal | 250 / 309 (81%) | 186 / 203 (92%) | 64 / 106 (60%) | ||
| Dilated | 59 / 309 (19%) | 17 / 203 (8.4%) | 42 / 106 (40%) | ||
| LVEF | 305 | 58.01 (11.76) | 58.86 (10.46) | 56.43 (13.79) | 0.4 |
| (Missing) | 4 | 4 | 0 | ||
| LVEDD | 219 | 44.68 (7.82) | 43.45 (7.59) | 46.83 (7.79) | 0.008 |
| (Missing) | 90 | 64 | 26 | ||
| Pericardial effusion (%) | 306 | >0.9 | |||
| Absent | 302 / 306 (99%) | 198 / 201 (99%) | 104 / 105 (99%) | ||
| Present | 4 / 306 (1.3%) | 3 / 201 (1.5%) | 1 / 105 (1.0%) | ||
| (Missing) | 3 | 2 | 1 | ||
| RV function (Abnormal S' or TAPSE. Definition: TAPSE < 17 mm or S’ < 9.5 cm/s) | 261 | >0.9 | |||
| Normal | 207 / 261 (79%) | 128 / 161 (80%) | 79 / 100 (79%) | ||
| Abnormal | 54 / 261 (21%) | 33 / 161 (20%) | 21 / 100 (21%) | ||
| (Missing) | 48 | 42 | 6 | ||
| RV function (Abnormal S'. Definiton: S’ < 9.5 cm/s) | 173 | 0.6 | |||
| Normal | 93 / 173 (54%) | 54 / 104 (52%) | 39 / 69 (57%) | ||
| Abnormal | 80 / 173 (46%) | 50 / 104 (48%) | 30 / 69 (43%) | ||
| (Missing) | 136 | 99 | 37 | ||
| RV function (Abnormal TAPSE. Definiton: TAPSE < 17 mm | 261 | >0.9 | |||
| Normal | 188 / 261 (72%) | 116 / 161 (72%) | 72 / 100 (72%) | ||
| Abnormal | 73 / 261 (28%) | 45 / 161 (28%) | 28 / 100 (28%) | ||
| (Missing) | 48 | 42 | 6 | ||
| LVEDD (Definition: LVEDD > 58.4 mm (male) or > 52.2 mm (female)) | 219 | 0.5 | |||
| Normal | 203 / 219 (93%) | 130 / 139 (94%) | 73 / 80 (91%) | ||
| Abnormal | 16 / 219 (7.3%) | 9 / 139 (6.5%) | 7 / 80 (8.8%) | ||
| (Missing) | 90 | 64 | 26 | ||
| LVEF (Definition: LVEF < 50%) | 305 | 0.058 | |||
| Normal | 253 / 305 (83%) | 171 / 199 (86%) | 82 / 106 (77%) | ||
| Abnormal | 52 / 305 (17%) | 28 / 199 (14%) | 24 / 106 (23%) | ||
| (Missing) | 4 | 4 | 0 | ||
| RV by visual estimation | 291 | <0.001 | |||
| Normal | 224 / 291 (77%) | 161 / 185 (87%) | 63 / 106 (59%) | ||
| Abnormal | 67 / 291 (23%) | 24 / 185 (13%) | 43 / 106 (41%) | ||
| (Missing) | 18 | 18 | 0 | ||
| Wall motion abnormalities | 294 | 43 / 294 (15%) | 27 / 192 (14%) | 16 / 102 (16%) | 0.7 |
| (Missing) | 15 | 11 | 4 | ||
| Takotsubo or stress cardiomyopathy | 297 | 3 / 297 (1.0%) | 2 / 196 (1.0%) | 1 / 101 (1.0%) | >0.9 |
| (Missing) | 12 | 7 | 5 | ||
| Composite outcome: Invasive ventilation, death at 60d, shock, thromboembolism or need for RRT | 309 | 194 / 309 (63%) | 119 / 203 (59%) | 75 / 106 (71%) | 0.036 |
| Death at 60 days | 309 | 75 / 309 (24%) | 39 / 203 (19%) | 36 / 106 (34%) | 0.004 |
| Invasive ventilation | 309 | 164 / 309 (53%) | 100 / 203 (49%) | 64 / 106 (60%) | 0.063 |
| RRT | 309 | 72 / 309 (23%) | 34 / 203 (17%) | 38 / 106 (36%) | <0.001 |
| Shock | 309 | 147 / 309 (48%) | 83 / 203 (41%) | 64 / 106 (60%) | 0.001 |
| Thromboembolic event | 309 | 35 / 309 (11%) | 17 / 203 (8.4%) | 18 / 106 (17%) | 0.023 |
| 1Median (p25, p75), mean (sd) or Frequency (%) | |||||
| 2Wilcoxon rank sum test; Pearson's Chi-squared test; Fisher's exact test | |||||
Abbreviations: CAD, coronary artery disease; HTN, hypertension; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; DM, diabetes mellitus; HLD, hyperlipidemia; ICD, implantable cardioverter defibrillator; RV, right ventricular; TAPSE, tricuspid annular plane systolic excursion; PA, pulmonary artery; PASP, pulmonary artery systolic pressure; RAP, right atrial pressure; LVEF, left ventricular ejection fraction; LVEDD, left ventricular end diastolic diameter; S’, peak lateral tricuspid annular systolic velocity; RRT, renal replacement therapy. Bold p-values are statistically significant.
Overall, 249 patients (58%) experienced the composite outcome of invasive ventilation, death at 60 days, shock, thromboembolism or need for RRT. The need for invasive ventilation was the most common component of the composite outcome, accounting for 207 patients (48%).
Of 427 patients, 309 had an RV size measurement (72.3%) recorded. Of these, 106 patients (34%) were documented to have an abnormal RV size (Table 2). These patients were more often males but presented with similar comorbidities as compared with patients with a normal RV size. Those with abnormal RV size also had higher pulmonary artery (PA) pressures but LV function was comparable. With the exception of invasive ventilation, all other analyzed outcomes (death at 60 days, need for RRT, shock, and thromboembolic events) were more prevalent among patients with an abnormal RV size.
For 386/427 patients (90.3%), an assessment of RV function could be made by qualitative or quantitative parameters (Supplemental Table 1). Among these patients, 166 (43.0%) were documented to have abnormal RV function as per quantitative or qualitative evaluation; this subset also more often presented with atrial fibrillation. Those with abnormal RV function also presented with higher PA pressures and with more dilated RVs. Patients with functional RV impairment presented with normal LV function and size. Patients with abnormal RV function more frequently required invasive ventilation and presented with shock more often.
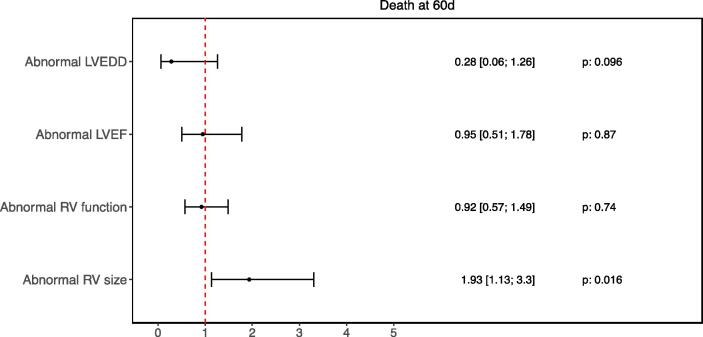
Fig. 1 shows the key findings of the multivariable model accounting for right- and left-sided echocardiographic parameters as they relate to 60-day mortality. Of all parameters tested, only abnormal RV size significantly predicted death at 60 days of follow-up, with OR 1.93 (95% CI 1.13 – 3.3, p = 0.016). Neither LV size, LV function, or RV function were determined to be significantly associated with an increased mortality risk (p = 0.096, p = 0.87, p = 0.016, respectively).
Fig. 1.
Forest Plot – 60-Day Mortality.
Legend: Odds ratios with 95% confidence intervals and corresponding p-values shown, Abnormal LVEDD: > 58.4 mm (male) or > 52.2 mm (female), Abnormal LVEF: < 50%, Abnormal RV Function: TAPSE < 17 mm or S’ < 9.5 cm/s or visual RV dysfunction, Abnormal RV Size: RV basal diameter > 41 mm.
In addition, abnormal RV size significantly predicted almost all clinical outcomes, except the need for mechanical ventilation (Supplemental Figures 1–4). An abnormal RV size not only increased the risk of death; this finding was also associated with increased risk of RRT by 2.35 (95%-CI 1.31–4.21, p = 0.004), shock by 1.81 (95%-CI 1.03–3.18, p = 0.039) and thromboembolism by 2.31 (95%-CI 1.37–3.88, p = 0.002). When assessing RV size, the incremental information provided by RV function did not help with further risk stratification, except for the prediction of shock, where abnormal RV function significantly increased the risk of shock (OR 1.57, 95%-CI 1.04–2.37). Similarly, when already accounting for right-sided parameters, the left-sided parameters were not significant predictors for any of the secondary outcomes.
4. Discussion
Point-of-care ultrasound (POCUS), focused transthoracic echocardiography (fTTE), and critical care echocardiography (CCE) have all played crucial roles in the management of patients with COVID-19 across the spectrum of clinical illness severity.[12] In critically ill patients with COVID-19, echocardiography can readily and rapidly detect cardiovascular complications in various clinical scenarios. For example, echocardiographic assessment of left ventricular (LV) systolic function can help distinguish cardiac vs. non-cardiac dyspnea when elevated cardiac biomarkers are present in patients with concomitant pulmonary disease and cytokine storm. TTE can also provide information about the status of the right ventricle, which is of relevance given the susceptibility of patients with COVID-19 to venous thromboembolism, acute respiratory distress syndrome, pulmonary embolism, and acute cor pulmonale. Lastly, in patients with elevated cardiac biomarkers and concern for myocardial injury, focused evaluation of the left ventricle can assess for wall motion abnormalities and guide timely intervention.[12] This study aimed to identify echocardiographic findings associated with adverse clinical outcomes in hospitalized COVID-19 patients. Specific echocardiographic parameters were identified as providing important prognostic information in this sub-population of hospitalized COVID-19 patients.
COVID-19 causes cardiac injury and manifests in several ways, including heart failure, myocarditis, arrhythmias, acute coronary syndromes, and other conditions.[13] Some reports state that up to 20–25% of patients with COVID-19 experience cardiovascular complications, which is also associated with increased overall mortality.[13] Proposed mechanisms of COVID-19-induced cardiac injury include cytokine-mediated damage, hypoxemia secondary to a misbalance of oxygen supply and demand, microvascular injury leading to ischemia, and direct SARS-CoV-2 invasion of myocardial tissue via ACE2 receptors.[14] While signs of myocardial injury can be evident through elevated cardiac biomarkers such as troponin or brain natriuretic peptide, transthoracic echocardiography can be used to characterize functional and structural abnormalities of the heart.[15] In a recent international study, patients with myocardial injury had more abnormal echocardiography findings than those without myocardial injury. The spectrum of echocardiographic abnormalities described in COVID-19 include LV dysfunction, wall motion abnormalities, RV dysfunction, diastolic dysfunction, and pericardial effusions.[15]
The key findings of our study are as follows. First, RV dilation was an independent prognostic factor associated with increased mortality in our cohort. A third (106/309) had RV dilation, which was significantly associated 60-day mortality (OR 1.93 [CI 1.13–3.3], p = 0.016). This is similar to results from a study by Kim et al., which showed that 172/510 (34%) of study patients had an abnormal RV size.[16] RV dilation in COVID-19 disease is thought to be a compensatory reaction to increased RV afterload due to COVID-19-mediated hypoxia and increased pulmonary circulation requirements. The right ventricle is composed of thin walls that are typically exposed to low pulmonary vascular resistance. In comparison, the left ventricle routinely functions against high systemic resistance.[16] In acute respiratory distress syndrome (ARDS), a common sequela of COVID-19 infection, pulmonary vascular resistance and vasoconstriction in the lungs are pronounced due to hypoxia, hypercapnia, and general pulmonary vascular remodeling. These changes can increase RV afterload and RV wall stress, leading to dilation and dysfunction.[17]
The second key finding of this study is the preponderance of RV dysfunction in the cohort of hospitalized patients with COVID-19. In our study, 166 out of 386 patients (43%) had evidence of RV dysfunction, measured either subjectively or quantitatively. RV dysfunction was significantly associated with shock (OR 1.57 [CI 1.04–2.37], p = 0.031). Several studies have found that RV strain and TAPSE were associated with higher rates of mortality.[7] Li et al. recently conducted a study that included 120 COVID-19 patients and reported similar prognostic findings.[8] They found that patients with reduced RV function as assessed by RV longitudinal strain (RVLS) and TAPSE had a higher risk of dying than those with preserved RV function. Additionally, RVLS was able to predict mortality risk independent of other echo parameters. In another study, decreased RV function was shown to be an independent predictor of death from all causes, with an almost two-fold increase in mortality hazard.[18] While the exact pathophysiologic mechanisms of COVID-19-related RV dysfunction remain obscure, some suggest that myocardial injury from diffuse inflammation triggered by COVID-19 infection plays a significant role.[16]
Another compelling finding of our study was the lack of significance of LV echocardiographic parameters in disease prognostication. Out of 427 echocardiograms in our cohort, abnormal LVEF, defined as LVEF of <50%, was seen in a minority of patients (17%). Our study showed that LV dysfunction was not a significant prognosticator of 60-day mortality with odds ratio 0.95 (CI: 0.51–1.78, p = 0.87). This finding is in line with previous studies, as in a systemic echocardiographic study conducted by Szekely et al., where abnormal LVEF was seen in only 10% of patients with COVID-19 unrelated to disease severity.[13] LV function is typically preserved or hyperdynamic in patients with COVID-19. When LV function is abnormal, it may be secondary to RV volume overload and excess pressure due to ventricular interdependence.[18]
Recent studies from China and New York have suggested that RV parameters such as dilation, longitudinal strain, ejection fraction by 3D echocardiography are correlated with adverse outcomes in COVID-19 infection.[8], [19], [20] These findings, while from relatively small studies of 110–128 patients, are congruent to findings of our study and further support the importance of RV dilation as a marker of severe COVID-19 illness. Currently, robust data on the utility of fTTE in the time of pandemic remains scarce. The higher power offered by our study in a US-based cohort adds to the body of knowledge in the area of fTTE utilization in COVID-19 disease prognostication. To date, our study is the largest retrospective analysis of multiple echocardiographic abnormalities as they relate to 60-day adverse clinical outcomes.
5. Limitations
This study has several limitations. First and foremost is the issue of referral bias. Echocardiograms were obtained according to widely accepted criteria as defined by clinical protocols implemented during the COVID-19 surge at our institution.[3], [21] To limit unnecessary exposure to healthcare personnel, examinations that require close contact, such as echocardiograms, were limited to the acutely ill and, often, those with increased disease severity. TTEs were reserved for patients with high suspicion for significant cardiopulmonary comorbidities such as heart failure, pulmonary embolism, or acute coronary syndrome, according to guidelines provided by ASE.[5] Thus, these factors limit the generalizability of this study to the entire spectrum of patients with COVID-19; the results of this study are most reflective of hospitalized, symptomatic unvaccinated patients with COVID-19 infection.
Second, another limitation of the study is the abbreviated fTTE protocol instituted in Rush University Medical Center. The echocardiograms were performed as abbreviated and focused studies, designed to glean important, clinically relevant information while limiting exposure of frontline staff and sonographers. Imaging views were limited in the COVID-19 cohort to those that would specifically address the clinical question and guide decision-making. Advanced parameters such as RV and LV strain were not performed to decrease exposure times. Consequently, more detailed variables of interest were not performed or available for review and analysis. Further systematic studies of the RV with detailed quantitative assessment using tools such as three-dimensional RV ejection fraction and RV longitudinal strain can be considered.
Third, the quality of imaging that could be obtained from the studied patients was not always optimal. Several patients, particularly in intensive care units, had difficult echocardiographic windows and limited visualization of cardiac structures. We believe that these technical issues are reflective of a real-world scenario with a sicker, hospitalized population.
Fourth, the study was focused on the identification of echocardiographic parameters of most prognostic significance. Potential confounders that were not corrected for are due to the limited power of the study, including age, sex, and hypertension. This should be taken into consideration in the interpretation of this study’s findings.
6. Conclusions
Abnormalities in RV parameters assessed by focused cardiovascular ultrasound were strong adverse prognosticators in COVID-19 disease. Interestingly, LV abnormalities did not provide prognostic information in our study. Patients with RV dilation have almost twice the risk for both in-hospital and 60-day mortality from COVID-19 disease. RV dilation is also significantly associated with an increased incidence of shock and venous thromboembolism, as well as an increased need for RRT.
RV dysfunction, measured either qualitatively or quantitatively with abnormal TAPSE or S’, showed a significant association with shock. While this study did not show a significant correlation between RV dysfunction and mortality, there was a trend to significance towards secondary adverse outcomes such as invasive ventilation.
To our knowledge, this is the largest study to date that validates the strong clinical prognostic power of RV dilation and dysfunction in hospitalized patients with COVID-19.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of Competing Interest
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Volgman – Research support: NIH IND Number 119127; NIH NINR R01NR018443; Novartis CTQJ230A12001. Consulting: MSD/Bayer Virtual Global Advisory Board Member, Bristol Myers Squibb Foundation Diverse Clinical Investigator Career Development Program (DCICDP), National Advisory Committee (NAC), Janssen Health Equity/Diversity Advisory Board, NIH Clinical Trials. Stock ownership: Apple Inc. stock. The remaining authors report no relationships that could be construed as a conflict of interest.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ijcha.2022.100982.
Appendix A. Supplementary material
The following are the Supplementary data to this article:
References
- 1.Akhmerov A., Marbán E. COVID-19 and the heart. Circ. Res. 2020;126(10):1443–1455. doi: 10.1161/CIRCRESAHA.120.317055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bhatraju P.K., Ghassemieh B.J., Nichols M., Kim R., Jerome K.R., Nalla A.K., Greninger A.L., Pipavath S., Wurfel M.M., Evans L., Kritek P.A., West T.E., Luks A., Gerbino A., Dale C.R., Goldman J.D., O’Mahony S., Mikacenic C. Covid-19 in critically ill patients in the seattle region — case series. N. Engl. J. Med. 2020;382(21):2012–2022. doi: 10.1056/NEJMoa2004500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Goldberg A.B., Kyung S., Swearingen S., Rao A. Expecting the unexpected: Echo laboratory preparedness in the time of COVID-19. Echocardiography. 2020;37:1272–1277. doi: 10.1111/ECHO.14763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Picard M.H., Weiner R.B. Echocardiography in the Time of COVID-19. J. Am. Soc. Echocardiogr. 2020;33 doi: 10.1016/j.echo.2020.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kirkpatrick J.N., Mitchell C., Taub C., Kort S., Hung J., Swaminathan M. ASE statement on protection of patients and echocardiography service providers during the 2019 novel coronavirus outbreak: endorsed by the American College of Cardiology. J. Am. Soc. Echocardiogr. 2020;33 doi: 10.1016/j.echo.2020.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.M. Christian Højbjerg Lassen, K. Grundtvig Skaarup, J. Nørgaard Lind, A. Saed Alhakak, M. Sengeløv, A. Bjerg Nielsen, C. Espersen, K. Ravnkilde, R. Hauser, L. Borum Schöps, E. Holt, N. Dyrby Johansen, D. Modin, K. Djernaes, C. Graff, H. Bundgaard, C. Hassager, R. Jabbari, J. Carlsen, A.-M. Lebech, O. Kirk, U. Bodtger, M. Greve Lindholm, G. Joseph, L. Wiese, F. Vinholt Schiødt, O. Peter Kristiansen, E. Schwarz Walsted, O. Wendelboe Nielsen, B. Lindegaard Madsen, N. Tønder, T. Benfield, K. Nielsen Jeschke, C. Suppli Ulrik, F. Knop, M. Lamberts, P. Sivapalan, G. Gislason, J. Louis Marott, R. Møgelvang, G. Jensen, P. Schnohr, P. Søgaard, S.D. Solomon, K. Iversen, J. Ulrik Staehr Jensen, M. Schou, T. Biering-Sørensen, Echocardiographic abnormalities and predictors of mortality in hospitalized COVID-19 patients: the ECHOVID-19 study, (2020). 10.1002/ehf2.13044.
- 7.Martha J.W., Pranata R., Wibowo A., Lim M.A. Tricuspid annular plane systolic excursion (TAPSE) measured by echocardiography and mortality in COVID-19: A systematic review and meta-analysis. Int. J. Infect. Dis. 2021;105 doi: 10.1016/j.ijid.2021.02.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Li Y., Li H., Zhu S., Xie Y., Wang B., He L., Zhang D., Zhang Y., Yuan H., Wu C., Sun W., Zhang Y., Li M., Cui L., Cai Y., Wang J., Yang Y., Lv Q., Zhang L., Xie M. Prognostic value of right ventricular longitudinal strain in patients with COVID-19. JACC: Cardiovasc. Imag. 2020;13 doi: 10.1016/j.jcmg.2020.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lang R.M., Badano L.P., Mor-Avi V., Afilalo J., Armstrong A., Ernande L., Flachskampf F.A., Foster E., Goldstein S.A., Kuznetsova T., Lancellotti P., Muraru D., Picard M.H., Rietzschel E.R., Rudski L., Spencer K.T., Tsang W., Voigt J.-U. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the american society of echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2015;28:1–39.e14. doi: 10.1016/j.echo.2014.10.003. [DOI] [PubMed] [Google Scholar]
- 10.Sjoberg D.D., Whiting K., Curry M., Lavery J.A., Larmarange J. Reproducible summary tables with the gtsummary package. R J. 2021;13 doi: 10.32614/RJ-2021-053. [DOI] [Google Scholar]
- 11.van Buuren S., Groothuis-Oudshoorn K. mice: multivariate imputation by chained equations in R. J. Stat. Softw. 2011;45 doi: 10.18637/jss.v045.i03. [DOI] [Google Scholar]
- 12.Zhang L., Wang B., Zhou J., Kirkpatrick J., Xie M., Johri A.M. Bedside focused cardiac ultrasound in COVID-19 from the Wuhan epicenter: the role of cardiac point-of-care ultrasound, limited transthoracic echocardiography, and critical care echocardiography. J. Am. Soc. Echocardiogr. 2020;33:676–682. doi: 10.1016/j.echo.2020.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Szekely Y., Lichter Y., Taieb P., Banai A., Hochstadt A., Merdler I., Gal Oz A., Rothschild E., Baruch G., Peri Y., Arbel Y., Topilsky Y. Spectrum of cardiac manifestations in COVID-19: a systematic echocardiographic study. Circulation. 2020;142:342–353. doi: 10.1161/CIRCULATIONAHA.120.047971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lala A., Johnson K.W., Januzzi J.L., Russak A.J., Paranjpe I., Richter F., Zhao S., Somani S., Van Vleck T., Vaid A., Chaudhry F., De Freitas J.K., Fayad Z.A., Pinney S.P., Levin M., Charney A., Bagiella E., Narula J., Glicksberg B.S., Nadkarni G., Mancini D.M., Fuster V. Prevalence and impact of myocardial injury in patients hospitalized with COVID-19 infection. J. Am. Coll. Cardiol. 2020;76(5):533–546. doi: 10.1016/j.jacc.2020.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Giustino G., Croft L.B., Stefanini G.G., Bragato R., Silbiger J.J., Vicenzi M., Danilov T., Kukar N., Shaban N., Kini A., Camaj A., Bienstock S.W., Rashed E.R., Rahman K., Oates C.P., Buckley S., Elbaum L.S., Arkonac D., Fiter R., Singh R., Li E., Razuk V., Robinson S.E., Miller M., Bier B., Donghi V., Pisaniello M., Mantovani R., Pinto G., Rota I., Baggio S., Chiarito M., Fazzari F., Cusmano I., Curzi M., Ro R., Malick W., Kamran M., Kohli-Seth R., Bassily-Marcus A.M., Neibart E., Serrao G., Perk G., Mancini D., Reddy V.Y., Pinney S.P., Dangas G., Blasi F., Sharma S.K., Mehran R., Condorelli G., Stone G.W., Fuster V., Lerakis S., Goldman M.E. Characterization of myocardial injury in patients with COVID-19. J. Am. Coll. Cardiol. 2020;76(18):2043–2055. doi: 10.1016/j.jacc.2020.08.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kim J., Volodarskiy A., Sultana R., Pollie M.P., Yum B., Nambiar L., Tafreshi R., Mitlak H.W., RoyChoudhury A., Horn E.M., Hriljac I., Narula N., Kim S., Ndhlovu L., Goyal P., Safford M.M., Shaw L., Devereux R.B., Weinsaft J.W. Prognostic utility of right ventricular remodeling over conventional risk stratification in patients with COVID-19. J. Am. Coll. Cardiol. 2020;76(17):1965–1977. doi: 10.1016/j.jacc.2020.08.066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fayssoil A., Mustafic H., Mansencal N. The right ventricle in COVID-19 patients. J. Cleaner Prod. 2020;130:166. doi: 10.1016/j.amjcard.2020.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Moody W.E., Mahmoud-Elsayed H.M., Senior J., Gul U., Khan-Kheil A.M., Horne S., Banerjee A., Bradlow W.M., Huggett R., Hothi S.S., Shahid M., Steeds R.P. Impact of right ventricular dysfunction on mortality in patients hospitalized with COVID-19. According to Race, CJC Open. 2021;3(1):91–100. doi: 10.1016/j.cjco.2020.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Argulian E., Sud K., Vogel B., Bohra C., Garg V.P., Talebi S., Lerakis S., Narula J. Right ventricular dilation in hospitalized patients with COVID-19 infection. JACC: Cardiovasc. Imag. 2020;13:2459–2461. doi: 10.1016/J.JCMG.2020.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang Y., Sun W., Wu C., Zhang Y., Cui L., Xie Y., Wang B., He L., Yuan H., Zhang Y., Cai Y., Li M., Zhang Y., Yang Y., Li Y., Wang J., Yang Y., Lv Q., Zhang L., Xie M. Prognostic value of right ventricular ejection fraction assessed by 3D echocardiography in COVID-19 patients. Front. Cardiovasc. Med. 2021;8 doi: 10.3389/fcvm.2021.641088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.J.P. Lang, E.A. Bohula, D.A. Morrow, M. Mehra, COVID-19 Cardiology Protocols: Brigham & Women’s Hospital, (2020). https://bwh.covidprotocols.org/protocols/cardiology/.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.