Abstract
In 2019, the third and updated edition of the Clinical Practice Guideline (CPG) on Prevention and Treatment of Pressure Ulcers/Injuries has been published. In addition to this most up‐to‐date evidence‐based guidance for clinicians, related topics such as pressure ulcers (PUs)/pressure injuries (PIs) aetiology, classification, and future research needs were considered by the teams of experts. To elaborate on these topics, this is the third paper of a series of the CPG articles, which summarises the latest understanding of the aetiology of PUs/PIs with a special focus on the effects of soft tissue deformation. Sustained deformations of soft tissues cause initial cell death and tissue damage that ultimately may result in the formation of PUs/PIs. High tissue deformations result in cell damage on a microscopic level within just a few minutes, although it may take hours of sustained loading for the damage to become clinically visible. Superficial skin damage seems to be primarily caused by excessive shear strain/stress exposures, deeper PUs/PIs predominantly result from high pressures in combination with shear at the surface over bony prominences, or under stiff medical devices. Therefore, primary PU/PI prevention should aim for minimising deformations by either reducing the peak strain/stress values in tissues or decreasing the exposure time.
Keywords: aetiology, pressure injury, pressure ulcer, risk
1. INTRODUCTION
In 2019, the third and updated edition of the Clinical Practice Guideline (CPG) on Prevention and Treatment of Pressure Ulcers/Injuries has been published by the European Pressure Ulcer Advisory Panel (EPUAP), the National Pressure Injury Advisory Panel (NPIAP), and the Pan Pacific Pressure Injury Alliance (PPPIA). 1 Twenty‐eight small working groups (SWGs) and the Guideline Governance Group (GGG) supported by a methodologist reviewed, summarised, and appraised the current evidence regarding pressure ulcer (PU)/pressure injury (PI) prevention and treatment. In addition to this most up‐to‐date and comprehensive evidence‐based guidance for clinicians, related topics such as PU/PI aetiology, classification, and future research needs were considered by the SWGs and GGG. 2 In order to elaborate on these important topics that provide the pathophysiological and research contexts to the practical clinical guidance, a series of journal papers are being developed. Specifically, after describing the CPG 2019 development methods 3 , 4 and discussing PU/PI classification, 5 this is the third paper in a series of CPG articles. The objective of this compilation of the Aetiology Chapter of the 2019 CPG 2 and relevant literature published since is to make this aetiological information accessible and updated for use by the scientific, medical research, and clinical communities.
A PU/PI has been defined in the 2019 CPG as “localised damage to the skin and/or underlying tissue, as a result of pressure or pressure in combination with shear; PUs/PIs usually occur over a bony prominence but may also be related to a medical device or other object”. 2 The PU/PI can present itself as intact skin or as an open wound and may be painful. The tissue damage occurs as a result of intense and/or prolonged exposure of soft tissues to sustained mechanical loading, that is, deformations in compression, tension, or shear or a combination of these loading modes. Sustained loading (also known as quasi‐static loading, ie, a nearly/almost static loading state) refers to loads that are applied continuously for long durations, such as minutes to hours or even days. The tolerance of soft tissues to sustained deformations differs by the tissue type; it may also be influenced by microclimate, perfusion, age, health status (either chronic or acute), and systemic comorbidities and localised (topical) conditions of the soft tissues, which are affected by the sustained mechanical loading. A number of contributing or confounding factors are also associated with PUs/PIs; the primary of which are impaired mobility/activity and a high potential for exposure to static or dynamic frictional forces. 2 A secondary contributing factor is impaired sensory perception (sensation), 2 which often leads to impaired mobility/activity, as a person does not move because they do not feel the discomfort, and further on, the nociceptive pain that appears as damage forms in cells and tissues. 6 Without the presence of impaired mobility, PUs/PIs are unlikely to occur, the case of device‐related injuries notwithstanding.
2. TERMINOLOGY
Since the first description of the wound referred to earlier as a PU/PI, there has been an ongoing debate regarding terminology. 7 The oldest term is decubitus, which evolved in the 1950s to decubitus ulcers or ischaemic ulcers. None of these terms are accurately descriptive and the latter term used by Kosiak 8 implies an overly limited aetiological pathway that focuses almost exclusively on pressure‐induced deformation and occlusion of capillaries with resultant tissue ischaemia. Although this pathway is well documented in the research literature, it does not account for the aetiological pathways initiated by cellular and tissue deformation that is the primary focus of this article. The term bedsores arose after publication of the Bedsore Biomechanics (1976) edited book that followed the first international conference on pressure ulcer aetiology held in Glasgow in the previous year. This term indicates the association of wounds with a stay in bed, which ignores the potential occurrence of these wounds on any other type of support surfaces. In the 1980s, the term pressure sore became more popular, thus no longer relating the injury to (just) the bed. Since the early 1990s, the term pressure ulcer, referring to an open ulcer at the skin surface that is difficult to heal or fails to heal, has been in common usage. However, this term fails to capture both the deep tissue pressure injury form, an internal soft tissue damage under intact skin, and Category/Stage I in which the skin surface remains intact. 2 Currently, in Europe, the term pressure ulcer is widely used. In the United States, Canada, Australia, New Zealand, and some countries in Southeast Asia, the term pressure injury was widely adopted. However, the aetiological triggering events and driving factors of PUs/PIs are identical, and all terms refer to the same phenomenon. 5 In this article, the terms pressure ulcer (PU) and pressure injury (PI) are used interchangeably.
Several aetiological pathways for PI/PU development have been proposed and supported by laboratory evidence including ischaemia from capillary closure, reperfusion injury, and tissue deformation. This article primarily focuses on more recent evidence regarding the effects of tissue deformation and its implications for clinical practice.
3. MECHANICAL LOADS APPLIED TO SOFT TISSUES
Mechanical loads applied to soft tissues are all the types of forces that can possibly act upon skin and underlying tissues of an individual as a result of contact between the skin and an external surface, object, or medical device. These loads include the bodyweight forces typically transferred through bony structures into soft tissues. External mechanical loads are often characterised as being a normal force (acting perpendicularly to the skin) or a shear force (acting parallel to skin). In real‐world scenarios, external forces always have normal and shear components. Pressure is defined as normal force per unit surface area (of skin or underlying tissue). Shear stress is likewise defined as the shear force (acting in a direction that is parallel to the skin or underlying tissue surface) per unit surface area.
The term friction is used as an abbreviation of the “coefficient of friction” to describe interface properties and the potential for sliding of two surfaces with respect to each other. The friction phenomenon is associated with shear deformations and stresses on the skin surface, within skin layers and subcutaneously, which may be static (if there is no relative movement between the skin and the contacting surface/object/device) or dynamic (when such relative movement occurs). 9 , 10 Continuous rubbing or sliding of the skin against materials such as textiles can result in inflammation, abrasions, or wounds referred to as friction blisters 11 ; these blisters are not considered to be PUs/PIs. 1 , 12
Loaded skin and deeper soft tissues distort and deform in response to mechanical loads, resulting in localised strain (a measure of the relative deformation) and stress (force transferred per unit area) in tissues. Excessive internal strains and stresses or prolonged tissue exposures to sustained strains and stresses will impair transport phenomena in cells by causing damage to cell structures such as the cytoskeleton or plasma membrane. 13 , 14 At a mesoscale, strains and stresses may also hinder transport processes within tissues, for example, by reducing blood perfusion, impairing lymphatic function, and affecting transport in interstitial spaces. 1 Cell death, in turn, triggers an inflammatory response, which generates inflammatory oedema 1 , 15 that further increases the mechanical loads on cells and tissues resulting from a rise in the interstitial pressure levels (Figure 1). If an underlying ischaemic condition exists, which lowers the peripheral perfusion pressure to an extent where plasma does not cross the vascular wall even if the wall is abnormally permeable (since the vascular wall resistance to the flow is too high to allow fluid movement across the wall to the interstitial space), then local oedema will not form.
FIGURE 1.
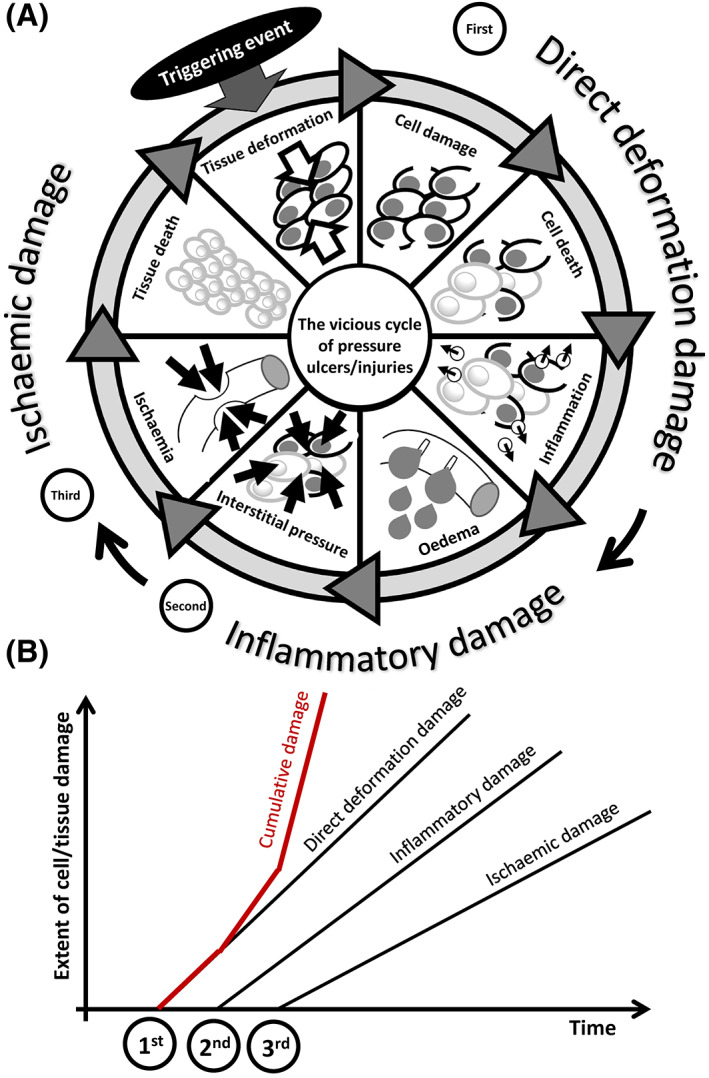
A, A schematic description of the vicious cycle of cell and tissue damage in pressure ulcers/injuries (PUs/PIs), resulting from sustained mechanical deformations (the triggering event), which inflicts the primary, direct deformation damage (first damage event), then leading to secondary inflammatory‐oedema related damage (second damage event), and finally to tertiary ischaemic damage (third damage event). B, Each of these three factors contributes to the cumulative cell and tissue damage, which develops in an escalated manner as a result of the added contributions of the above factors. Of note, while it is likely that the ischaemic damage follows from the inflammatory damage as visualised here, ischaemia per se may also be present prior to the onset of the PU/PI vicious cycle (eg, due to a persistent peripheral vascular disease), or develop independently from, or concurrently with the inflammation (eg, as a result of a thrombotic event in a patient with coronavirus disease 2019). In contrast, localised inflammation may follow from or intensify because of an existing ischaemic condition, especially when reperfusion is allowed (eg, immediately after repositioning). Accordingly, the process of accumulation of tissue damage over time in a forming PU/PI as described here is likely, but is not necessarily a linear one (particularly with regard to the order of the inflammatory and ischaemic damage phases)
The specific ways by which cells and tissues are affected by mechanical loads are complex processes, which depend on anatomical structure and morphology, for example, the sizes and shapes of the different tissue layers and the biophysical and mechanical properties of the tissues involved. Relevant biophysical and mechanical tissue properties are the density and composition, water contents, stiffness, strength, and diffusion coefficients. These properties shape the magnitudes and distributions of the mechanical forces that are applied to the tissues at the regions of contact with a support surface, object or medical device, and also, within the tissue structures. 16 In addition, the morphology, mechanical properties, and tissue tolerance to the loading can all change over time because of ageing, lifestyle, chronic injury, or disease. 16 , 17 , 18 In general, externally applied forces, even of a uniform nature, will lead to highly irregular internal tissue strain/stress responses, that is, different loading intensities at different tissue locations, depending on the internal anatomy, body posture, and properties of tissues of the individual. 19 , 20 , 21 , 22 This can also be referred to as heterogeneous or nonhomogeneous tissue responses to the applied loads. Further complications are introduced because normal forces on the surface of the weight‐bearing body (eg, during lying or sitting) or due to a skin‐contacting device (eg, oxygen mask) will be highly non‐uniform across the supported areas and some shear forces will always exist. For example, while an individual is sitting in a chair, it is common that internal tissue strain levels in muscle can reach values of 50% and above. 19 , 20 , 23 Furthermore, soft tissue stiffness increases under weight‐bearing sitting and may contribute to increasing the potential PU/PI risk in sitting, compared with lying prone, given the stiffer behaviour of tissues observed in sitting postures. 16
Techniques available for assessment of internal tissue deformations are magnetic resonance imaging, elastography, and ultrasound. 16 , 24 These imaging modalities can be used in combination with subject‐specific theoretical computational models such as finite element (FE) models; FE modelling is a method of solving (bio)mechanical problems by means of a capable computer workstation and dedicated software, to estimate deformations, strains, and stresses throughout cell and tissue structures, and thereby predict the risk of cell and tissue damage. However, even the best and most accurate FE models are theoretical assumptions 25 and clinical reality is infinitely complex, PUs/PIs develop as a result of the internal tissue response to the mechanical loading state in the individual. Understanding the aetiology of PUs/PIs therefore relies on knowledge concerning the internal cell and tissue responses to mechanical loads (including the responses of vascular and lymphatic structures) and not on just what is apparent on the outside of the body or at the skin surface. 26 , 27 , 28
4. SOFT TISSUE RESPONSES TO SUSTAINED MECHANICAL LOADS
The primary cause of PUs/PIs is exposure to sustained mechanical loads that are applied to soft tissues, most often near a bony prominence, but not always. These mechanical loads can originate from bodyweight forces (ie, the body mass pulled by gravity) or from the environment, for example, delivered by a medical device such as a ventilation mask or a pulse oximeter, which apply continuous forces and deformations to skin and deeper tissues near the contact region with these devices. Such medical devices are typically stiffer than the skin and underlying soft tissues. The mismatch in mechanical properties between device and skin as well as underlying soft tissues causes focal deformations and mechanical stress concentrations in tissues near the contact sites with the device. 15 , 29 By definition, sustained deformations of skin and deeper tissues, either due to bodyweight loads or exerted from the environment, must be present in order to cause the initial cell death and tissue damage that ultimately result in the formation of a PU/PI (Figure 1). Without timely relief of the localised loads, this initial cell death and tissue damage associated with the aforementioned compromised control over molecular transport at the microscale may occur within short time frames, in the order of minutes. 30 , 31 , 32 , 33 , 34 , 35 , 36 , 37 , 38
A large body of research performed over decades has shown that the magnitude of the internal mechanical loads required to lead to tissue damage depends on the duration of time during which the loads are applied. Both a high‐magnitude load for a short period and a low‐magnitude load applied for a prolonged period can lead to tissue damage. 31 , 33 , 39 , 40 , 41 , 42 , 43 , 44 , 45 , 46 Taken together, these study results indicate that high tissue deformations result in cell damage, which may be visible on a microscopic level within just a few minutes, although it may take hours of sustained loading for the damage to become clinically visible.
By contrast, impact damage, which usually occurs because of an accident or trauma, does not fall under the definition of PUs/PIs (although impact similarly damages cells and tissues by application of mechanical loads). The aetiological difference is essentially the time of exposure to the mechanical loads. For impact damage, an impulse of high mechanical load is applied within a fraction of a second to the tissues and organs. The mass of the impacting objects plays an important role and inertia effects leading to shock, shear and pressure waves in the tissues may cause high external and internal damage in a fraction of a second. 47 Discriminating between traumatic damage and PU/PI requires knowing the circumstances leading to the wound.
The historical threshold function for tissue damage developed by Reswick 42 depended on the interface pressures applied to human skin and the duration of these applied pressures (Figure 2). However, their curve required corrections as it did not accurately reflect the risk of tissue damage at the extremes of the very short and very long loading times. Sufficiently high loads can cause damage within minutes to tissues at a microscopic level. Conversely, very low loads will not lead to damaged tissues even if applied for extended periods of time. 48 , 49 Due to variability in individual anatomies, tissue tolerances, and confounding factors, it is not possible to determine generic quantitative values for tissue damage thresholds as a function of the mechanical loads and exposure time. The absence of time and pressure scales on the graph in Figure 2 is intentional to emphasise the need for individualisation in care. An example of an extrinsic confounding factor that has been shown to have a profound effect on the tissue tolerance to pressure damage is temperature, which is important in aspects of interaction with support surfaces and medical devices. 50 , 51 , 52 Another intrinsic confounding factor may be arteriolar insufficiency and endothelial dysfunction, for example, related to diabetes mellitus. 53 , 54
FIGURE 2.
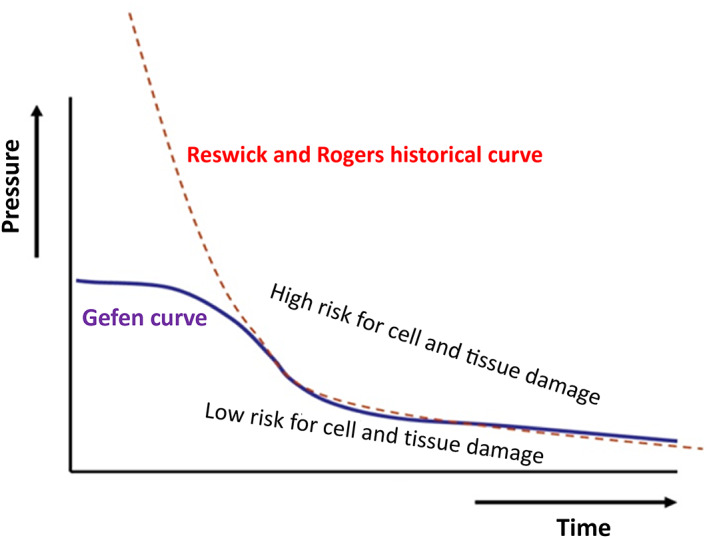
Tolerance behaviour of soft tissues subjected to sustained mechanical loads formulated by Linder‐Ganz et al. 33 and Gefen et al. 32 based on experimental data from animal and tissue‐engineered models, respectively (marked “Gefen curve”), compared with the historical Reswick and Rogers pressure‐based damage threshold proposed for sitting and recumbent patients in the 1970s
Minimising pressure and shear stresses at the interface between the body and a support surface or at a skin‐device contact region are effective clinical interventions for reducing the risk of developing PUs/PIs. 1 , 55 , 56 , 57 However, pressure measurements alone are not reliable measures for the risk of tissue breakdown, given that similar interface pressure magnitudes will translate to considerably different internal tissue loads in different individuals depending on their internal anatomy (curvature of bony prominences, masses and composition of soft tissues, skin roughness characteristics, and soft tissue mechanic properties). Thus, damage thresholds based on interface pressures alone or even exposure to interface pressures over time are not appropriate. 19 , 20 , 26 , 27 , 28 , 31 , 58 , 59 In other words, while clinically, interface pressure measurements are commonly used as a relative guide to repositioning individuals who are at risk of PUs/PIs, for minimising certain areas of high localised interface pressures, these measurements are not appropriate for predicting injury conditions in the examined individuals or more generally, as an absolute measure of tissue damage thresholds.
Elevated shear stresses at the interface between the body and a support surface or medical device can exacerbate the damaging deformations caused by pressures alone. 60 , 61 , 62 , 63 Internal strains and stresses adjacent to bony prominences are substantially higher than those near the surface, and rise with the level of sharpness of the bony prominence, due to stress concentration effects. These stress concentrations have the potential to cause damage in deep tissues before the superficial tissue is damaged and before damage is visible to the unaided eye. 19 , 20 , 55 , 57 , 58 , 64 , 65 , 66 , 67 , 68 , 69 In addition to internal tissue stress concentrations, superficial shear stresses associated with frictional forces that act on skin may disturb the barrier function of the stratum corneum. 51 Accordingly, support surfaces characterised by high coefficient of friction (COF), or for which the COF increases substantially due to wetness (perspiration, exudates and body fluids), represent an extra danger for skin health. Sustained weight‐bearing or application of a skin‐contacting medical device for prolonged periods, per se or in combination with moisture and wetness at the skin‐object interface, may affect the skin microtopography (roughness) features, which in turn affects the COF of skin with the contacting object. 9 , 10 , 59 , 70 , 71 , 72 , 73 , 74
If a PU/PI has been formed due to sustained deformations, soft tissue properties may abnormally change. For example, skeletal muscle may exhibit localised “rigour mortis” sites of stiffening (ie, localised pathological contractions due to destruction of muscle fibre membranes), which adds inhomogeneity to the loading patterns and promotes intramuscular stress concentrations that endanger adjacent tissues. 65 , 75 Likewise, fibrous scar tissues at the skin or internally promote tissue stress concentrations and contribute to the inhomogeneity of the sustained loading, as scars are less effective than healthy tissues in dissipating mechanical loads through elastic deformations, due to their disorganised microstructure. 59 , 76 , 77 This provides a biomechanical explanation for the typical recurrence of a PU/PI at the site of a former, already healed full‐thickness PU/PI.
5. TISSUE DAMAGE DUE TO MECHANICAL LOADING, REPAIR, AND INDIVIDUAL SUSCEPTIBILITY
There seem to be differences between the aetiology of superficial PUs/PIs, which onset as skin damage, versus those PUs/PIs that originate and form in deeper tissue layers. 78 , 79 , 80 , 81 Superficial skin damage seems to be primarily caused by excessive shear strain/stress exposure at the skin surface. Deeper PUs/PIs predominantly result from high pressures in combination with shear at the surface over bony prominences, or under a stiff medical device where tissues are continuously distorted between the device and musculoskeletal structures, such as in the case of facial tissues that are confined between an oxygen mask and the skull. 59 , 72 , 75 , 79 , 82 , 83 , 84 , 85 , 86 , 87 , 88 , 89 , 90 , 91
A possible damage cascade in PIs/PIs, illustrated in Figure 1A, includes the sequential cell and tissue damage associated with direct deformation (first factor), damage associated with the inflammatory response (second factor), and damage induced by ischaemia (third factor). 15 , 41 , 92 The additive nature of these damages (depicted in Figure 1B) highlights the importance of minimisation of the exposure to sustained tissue deformations and early detection of cell and tissue damage for effective PU/PI prevention. In the context of the theoretical framework described in Figure 1, the work of Coleman and colleagues 93 highlighted the factors that characterise the PU/PI risk of an individual. The internal anatomy, including the sharpness of bony prominences, the tissue morphologies, and the mechanical and thermal properties of tissues will altogether dictate the internal tissue deformations, strains, and stresses, as well as the thermodynamic state and transport (diffusional) properties of the distorted tissues. The cell and tissue repair capacity and transport properties at the cell and tissue scales will determine the ability of the body to repair damage at various stages of progression. The progression of damage relative to the progression of repair will constitute the time for a PU/PI to develop (if developed) in the individual and the extent and severity to which the injury will progress, if that individual is immobile/inactive.
Of note, while it is likely that the ischaemic damage follows from the inflammatory damage as visualised in Figure 1, ischaemia per se may also be present prior to the onset of the PU/PI vicious cycle (eg, due to a persistent peripheral vascular disease or prolonged pressure‐induced capillary closure), or develop independently from or concurrently with the inflammation (eg, as a result of a thrombotic event in a patient with coronavirus disease). 94 In contrast, localised inflammation may follow from or intensify due to an existing ischaemic condition, especially when reperfusion and possible reperfusion injury are allowed (eg, immediately after repositioning). 34 , 35 , 95 , 96 , 97 , 98 , 99 , 100 , 101 Accordingly, the process of accumulation of tissue damage over time in a forming PU/PI as described here is likely, but is not necessarily a linear one (particularly with regard to the order of the inflammatory and ischaemic damage phases).
Once the vicious cycle of a PU/PI enters the ischaemic phase in the cellular deformation model (Figure 1A), biochemical stress develops in the affected tissues because of the lack of supply of essential molecules and impaired clearance of metabolic waste products. Specifically, the ischaemia resulting from the inflammatory oedema (through an increase in the interstitial pressure level that distorts the vasculature) may lead to hypoxia, reduced supply of glucose and essential hormones (eg, insulin), and impaired removal of carbon dioxide and nitrogenous compounds (eg, ammonia, urea, uric acid, and creatinine). Lymphatic impediments are also associated with abnormally high interstitial pressures. 102 , 103 Deprivation of nutrients and decrease in the pH of the interstitial fluid towards a more acidic extracellular environment, because of the accumulation of metabolic waste products, will eventually lead to cell death and necrotic tissue damage; however, cells are able to survive for considerable times, on the order of hours, by shifting to an anaerobic metabolism. 39 , 40 , 61 , 104 Prolonged exposure to ischaemic conditions including an acidic extracellular environment (ie, pH < 7.4) has shown to slow collective cell migration, particularly of fibroblasts, in cell culture models, 105 which may compromise the body's attempts to repair microscale damage and hence contribute to an overall accelerated rate of tissue damage in PUs/PIs.
The time duration during which cells and tissues can endure ischaemia without irreversible damage differs for the tissue types (ie, skeletal muscle, adipose, and skin) that are potentially involved in PUs/PIs. Skeletal muscle tissues are more susceptible to PU/PI damage than skin, likely due to the greater capillary and mitochondrial volume density in muscles, which is associated with their greater metabolic demand. 43 , 44 , 61 , 62 , 106 Skin is also considerably stiffer than muscle or adipose tissues and therefore deforms to a lesser extent in most clinically relevant scenarios, which in turn makes it less susceptible to ischaemic damage. In animal experiments, the first signs of ischaemic damage are found in skeletal muscle after 2 to 4 hours of sustained deformations. 33 , 34 , 35 , 39 , 40 , 61 , 69 , 104
Sustained skeletal muscle deformations at strains greater than 50% will almost immediately (within minutes) lead to tissue damage at a microscopic scale. 31 At these strain levels, there is a strong correlation between the magnitude of the strain and the amount of damage inflicted to the muscle cells and fibres. This direct deformation‐inflicted damage to cells is the result of (i) loss of integrity and structural support provided to the cell body by the cytoskeleton; (ii) overstretching of the plasma membrane, which increases when the structural support provided to the membrane by the cytoskeleton diminishes; and (iii) internal signalling pathways related to these excessive cell deformations that cause apoptotic cell death. 13 , 14 , 44 , 45 , 46 , 107 , 108 , 109 , 110 Recent mechanobiology work focusing on the cell scale has further indicated that stimulating cells mechanically, by applying low‐level, non‐damaging mechanical deformations (strains), accelerates collective cell migration into damage sites in laboratory cell cultures. 111 , 112 Given that PUs/PIs form when the rate of cell and tissue death is greater than the corresponding rate of regeneration (ie, through cell proliferation, migration, and differentiation), mechanobiology research has already identified certain features of stimuli to promote repair processes, particularly migration of cells into a damage site at the onset of a PU/PI. 111 , 112
Balance at the interstitial spaces, where transport of nutrients and waste products occurs, is critical for healthy tissue homeostasis. Specifically, diffusion of nutrients and clearance of waste products and hormones that regulate tissue metabolism may be hindered by sustained mechanical loading. 32 , 38 , 113 , 114 Cell culture, tissue engineering, and computational modelling work suggested that the localised sustained large tissue deformations in weight‐bearing body regions under bony prominences translate to large cellular deformations at the microscale, thereby causing distortion of cellular organelles, for example, considerable stretching of cellular plasma membranes. 14 , 91 , 108 , 113 , 114 , 115 , 116 , 117 , 118 , 119 The prolonged exposure to large tensional plasma membrane strains may interfere with normal cellular homeostasis, primarily by affecting transport through the plasma membrane, which becomes more permeable when it is highly stretched. This has been visualised and quantified in cell cultures subjected to physiologically relevant deformations for periods of 2 to 3 hours, using biomolecular fluorescent markers. 13 , 14 , 107 The progression of cell death and tissue necrosis causes gradual local alterations of the mechanical properties of the injured tissues that can, in turn, change the distributions of strains and stresses in forms that are likely to exacerbate the evolving injury, for example, through development of inflammatory oedema and localised rigour mortis in skeletal muscles. 65 , 69 , 75 , 120 , 121 , 122 Localised inflammatory oedema, one of the earliest signs of cell death in PUs/PIs, may be detectable via measurement of a biophysical marker called the biocapacitance of tissues. 15 , 121 , 122 , 123 , 124 , 125 , 126 , 127 , 128 , 129 , 130 , 131 , 132 Reperfusion that follows a period of prolonged ischaemia may further increase the extent of tissue damage as it involves release of damaging oxygen free radicals. 95 , 96 , 97 , 98 , 99 , 100 , 133
The microclimate between the skin and the support surface or any skin‐contacting medical device or object plays an important role in the development of PUs/PIs. 51 , 52 , 134 Microclimate refers to the temporal and spatial temperature, humidity, and airflow in the vicinity of the outer surface of the skin. The characteristics of an optimal microclimate are still a matter of debate and ongoing research; however, with an increase in temperature and humidity, the skin becomes more vulnerable to damage. 51 , 55 , 135 Excessively dry skin may be also undesirable. 136 Wetness of the interface plays an important role in the mechanical interactions between the skin and a support surface, a medical device, clothing, bedsheets, or other objects. 9 , 10 , 52 , 134 , 135 , 137 , 138 Materials with a higher wettability, including skin, generally show a larger increase in COF when exposed to warm and moist conditions. 9 , 139 The body core and skin surface temperatures influence transepidermal water loss 140 and sweating. 141 The evaporation of perspiration from the skin surface depends on the local and ambient humidity. 51 , 135 These microclimate conditions eventually impact the frictional properties of skin, that is, the skin COF with contacting objects. The magnitude of this COF influences the intensity of the frictional forces acting on the body and ultimately, the skin and subdermal tissue deformations resulting from any frictional sliding movements between the skin and a support surface, a medical device, or other objects. 1 , 9 , 10 , 52 , 59 Overall, there are strong connections between microclimate and friction, and hence, surface and internal tissue loads and the exposure of living cells to these mechanical loads strongly depend on the microclimate conditions. 52 , 72 , 73 , 134
6. CONCLUSION: WHAT DOES THIS MEAN FOR CLINICAL PRACTICE AND RESEARCH?
Current aetiological knowledge confirms that PUs/PIs develop due to sustained mechanical loading leading to soft tissue deformation. Therefore, primary PU/PI prevention should aim for minimising deformations by either reducing the peak strain/stress values in tissues (eg, through the use of an appropriate pressure redistribution support surface) or decreasing the continuous exposure time of tissues to the sustained strain/stress state (eg, through repositioning). 1 , 142 Early detection of initial tissue damage through regular skin assessment is a strategy to prevent possible progression (secondary prevention). 1 , 128 Other PI prevention strategies such as nutritional interventions or appropriate use of leave‐on products aim to enhance tissue tolerance. 1
The described recent advances in research also enable various innovative approaches for primary and secondary PU/PI prevention. 53 , 101 , 143 , 144 , 145 , 146 , 147 These innovations are promising and urgently needed because the incidence and prevalence of PUs/PIs remain high across all healthcare settings. 1 Until evidence supports clinical effectiveness of these innovations, evidence‐based PU/PI prevention should be based on the latest guideline recommendations. 1
CONFLICT OF INTEREST
All authors were actively involved and contributed to the International Prevention and Treatment of Pressure Ulcers/Injuries: Clinical Practice Guideline 2019.
ACKNOWLEDGEMENTS
The authors thank all volunteers who participated in the Small Working Groups, Associate Organisations, and stakeholders and consumers who contributed to the development of the Prevention and Treatment of Pressure Ulcers/Injuries: Clinical Practice Guideline 2019.
Gefen A, Brienza DM, Cuddigan J, Haesler E, Kottner J. Our contemporary understanding of the aetiology of pressure ulcers/pressure injuries. Int Wound J. 2022;19(3):692-704. 10.1111/iwj.13667
DATA AVAILABILITY STATEMENT
Data sharing not applicable to this article as no datasets were generated or analysed during the current work.
REFERENCES
- 1. EPUAP, NPIAP, PPPIA . Prevention and treatment of pressure ulcers/injuries: clinical practice guideline. In: Haesler E, ed. European Pressure Ulcer Advisory Panel, National Pressure Ulcer Advisory Panel, Pan Pacific Pressure Injury Alliance. EPUAP, NPIAP, PPPIA; 2019. [Google Scholar]
- 2. Gefen A, Brienza D, Edsberg L, et al. The etiology of pressure injuries. In: Haesler E, ed. Prevention and Treatment of Pressure Ulcers/Injuries: Clinical Practice Guideline: European Pressure Ulcer Advisory Panel. Pan Pacific Pressure Injury Alliance: National Pressure Ulcer Advisory Panel; 2019. [Google Scholar]
- 3. Kottner J, Cuddigan J, Carville K, et al. Prevention and treatment of pressure ulcers/injuries: the protocol for the second update of the international clinical practice guideline 2019. J Tissue Viability. 2019;28(2):51‐58. [DOI] [PubMed] [Google Scholar]
- 4. Kottner J, Cuddigan J, Carville K, et al. A closer look at the 2019 international guideline on the prevention and treatment of pressure ulcers/injuries. J Tissue Viability. 2020;29(4):225‐226. 10.1016/j.jtv.2020.09.003 [DOI] [PubMed] [Google Scholar]
- 5. Kottner J, Cuddigan J, Carville K, et al. Pressure ulcer/injury classification today: an international perspective. J Tissue Viability. 2020;29(3):197‐203. 10.1016/j.jtv.2020.04.003 [DOI] [PubMed] [Google Scholar]
- 6. Gefen A, Soppi E. What is new in our understanding of pressure injuries: the inextricable association between sustained tissue deformations and pain and the role of the support surface. Wound Pract Res. 2020;28(2):58‐65. 10.33235/wpr.28.2.58-65 [DOI] [Google Scholar]
- 7. Kottner J, Sigaudo‐Roussel D, Cuddigan J. From bed sores to skin failure: linguistic and conceptual confusion in the field of skin and tissue integrity. Int J Nurs Stud. 2019;92:58‐59. 10.1016/j.ijnurstu.2019.01.007 [DOI] [PubMed] [Google Scholar]
- 8. Kosiak M. Etiology and pathology of ischemic ulcers. Arch Phys Med Rehabil. 1959;40(2):62‐69. [PubMed] [Google Scholar]
- 9. Schwartz D, Magen YK, Levy A, et al. Effects of humidity on skin friction against medical textiles as related to prevention of pressure injuries. Int Wound J. 2018;15(6):866‐874. 10.1111/iwj.12937 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Shaked E, Gefen A. Modeling the effects of moisture‐related skin‐support friction on the risk for superficial pressure ulcers during patient repositioning in bed. Front Bioeng Biotechnol. 2013;14(1):9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hashmi F, Richards BS, Forghany S, et al. The formation of friction blisters on the foot: the development of a laboratory‐based blister creation model. Skin Res Technol. 2013;19(1):e479‐e489. 10.1111/j.1600-0846.2012.00669.x [DOI] [PubMed] [Google Scholar]
- 12. Berke CT. Pathology and clinical presentation of friction injuries: case series and literature review. J Wound Ostomy Continence Nurs. 2015;42(1):47‐61. 10.1097/won.0000000000000087 [DOI] [PubMed] [Google Scholar]
- 13. Slomka N, Gefen A. Relationship between strain levels and permeability of the plasma membrane in statically stretched myoblasts. Ann Biomed Eng. 2012;40(3):606‐618. 10.1007/s10439-011-0423-1 [DOI] [PubMed] [Google Scholar]
- 14. Leopold E, Gefen A. Changes in permeability of the plasma membrane of myoblasts to fluorescent dyes with different molecular masses under sustained uniaxial stretching. Med Eng Phys. 2013;35(5):601‐607. 10.1016/j.medengphy.2012.07.004 [DOI] [PubMed] [Google Scholar]
- 15. Lustig M, Levy A, Kopplin K, et al. Beware of the toilet: the risk for a deep tissue injury during toilet sitting. J Tissue Viability. 2018;27(1):23‐31. 10.1016/j.jtv.2017.04.005 [DOI] [PubMed] [Google Scholar]
- 16. Mansur R, Peko L, Shabshin N, et al. Ultrasound elastography reveals the relation between body posture and soft‐tissue stiffness which is relevant to the etiology of sitting‐acquired pressure ulcers. Physiol Meas. 2021;41(12):124002. 10.1088/1361-6579/abc66d [DOI] [PubMed] [Google Scholar]
- 17. Gefen A. Tissue changes in patients following spinal cord injury and implications for wheelchair cushions and tissue loading: a literature review. Ostomy Wound Manag. 2014;60(2):34‐45. [PubMed] [Google Scholar]
- 18. Traa WA, van Turnhout MC, Nelissen JL, et al. There is an individual tolerance to mechanical loading in compression induced deep tissue injury. Clin Biomech. 2019;63:153‐160. 10.1016/j.clinbiomech.2019.02.015 [DOI] [PubMed] [Google Scholar]
- 19. Linder‐Ganz E, Shabshin N, Itzchak Y, Gefen A. Assessment of mechanical conditions in sub‐dermal tissues during sitting: a combined experimental‐MRI and finite element approach. J Biomech. 2007;40(7):1443‐1454. 10.1016/j.jbiomech.2006.06.020 [DOI] [PubMed] [Google Scholar]
- 20. Linder‐Ganz E, Shabshin N, Itzchak Y, et al. Strains and stresses in sub‐dermal tissues of the buttocks are greater in paraplegics than in healthy during sitting. J Biomech. 2008;41(3):567‐580. 10.1016/j.jbiomech.2007.10.011 [DOI] [PubMed] [Google Scholar]
- 21. Oomens CW, Broek M, Hemmes B, et al. How does lateral tilting affect the internal strains in the sacral region of bed ridden patients? A contribution to pressure ulcer prevention. Clin Biomech. 2016;35:7‐13. 10.1016/j.clinbiomech.2016.03.009 [DOI] [PubMed] [Google Scholar]
- 22. Sonenblum SE, Sprigle SH. Buttock tissue response to loading in men with spinal cord injury. PLoS One. 2018;13(2):e0191868. 10.1371/journal.pone.0191868 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Reger SI, McGovern TF, Chung KC. Biomechanics of tissue distortion and stiffness by magnetic resonance imaging. In: Bader DL, ed. Pressure Sores: Clinical Practice and Scientific Approach. London: MacMillan; 1990:177‐190. [Google Scholar]
- 24. Swaine JM, Breidahl W, Bader DL, et al. Ultrasonography detects deep tissue injuries in the subcutaneous layers of the buttocks following spinal cord injury. Top Spinal Cord Inj Rehabil. 2018;24(4):371‐378. 10.1310/sci17-00031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Savonnet L, Wang X, Duprey S. Finite element models of the thigh‐buttock complex for assessing static sitting discomfort and pressure sore risk: a literature review. Comput Methods Biomech Biomed Engin. 2018;21(4):379‐388. 10.1080/10255842.2018.1466117 [DOI] [PubMed] [Google Scholar]
- 26. Gefen A, Levine J. The false premise in measuring body‐support interface pressures for preventing serious pressure ulcers. J Med Eng Technol. 2007;31(5):375‐380. 10.1080/03091900601165256 [DOI] [PubMed] [Google Scholar]
- 27. Oomens CW, Loerakker S, Bader DL. The importance of internal strain as opposed to interface pressure in the prevention of pressure related deep tissue injury. J Tissue Viability. 2010;19(2):35‐42. 10.1016/j.jtv.2009.11.002 [DOI] [PubMed] [Google Scholar]
- 28. Reenalda J, Jannink M, Nederhand M, et al. Clinical use of interface pressure to predict pressure ulcer development: a systematic review. Assist Technol. 2009;21(2):76‐85. 10.1080/10400430903050437 [DOI] [PubMed] [Google Scholar]
- 29. Levy A, Kopplin K, Gefen A. Device‐related pressure ulcers from a biomechanical perspective. J Tissue Viability. 2017;26(1):57‐68. 10.1016/j.jtv.2016.02.002 [DOI] [PubMed] [Google Scholar]
- 30. Ceelen KK, Stekelenburg A, Loerakker S, et al. Compression‐induced damage and internal tissue strains are related. J Biomech. 2008;41(16):3399‐3404. 10.1016/j.jbiomech.2008.09.016 [DOI] [PubMed] [Google Scholar]
- 31. Gefen A. The compression intensity index: a practical anatomical estimate of the biomechanical risk for a deep tissue injury. Technol Health Care. 2008;16(2):141‐149. [PubMed] [Google Scholar]
- 32. Gefen A. How much time does it take to get a pressure ulcer? Integrated evidence from human, animal, and in vitro studies. Ostomy Wound Manage. 2008;54(10):26‐35. [PubMed] [Google Scholar]
- 33. Linder‐Ganz E, Engelberg S, Scheinowitz M, et al. Pressure‐time cell death threshold for albino rat skeletal muscles as related to pressure sore biomechanics. J Biomech. 2006;39(14):2725‐2732. 10.1016/j.jbiomech.2005.08.010 [DOI] [PubMed] [Google Scholar]
- 34. Loerakker S, Manders E, Strijkers GJ, et al. The effects of deformation, ischemia, and reperfusion on the development of muscle damage during prolonged loading. J Appl Physiol. 2011;111(4):1168‐1177. 10.1152/japplphysiol.00389.2011 [DOI] [PubMed] [Google Scholar]
- 35. Loerakker S, Oomens CWJ, Manders E, et al. Ischemia‐reperfusion injury in rat skeletal muscle assessed with T‐2‐weighted and dynamic contrast‐enhanced MRI. Magn Reson Med. 2011;66(2):528‐537. [DOI] [PubMed] [Google Scholar]
- 36. Loerakker S, Stekelenburg A, Strijkers GJ, et al. Temporal effects of mechanical loading on deformation‐induced damage in skeletal muscle tissue. Ann Biomed Eng. 2010;38(8):2577‐2587. 10.1007/s10439-010-0002-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Oomens CWJ. A multi‐scale approach to study the aetiology of pressure ulcers. Wound Repair Regen. 2010;18(4):A74. [Google Scholar]
- 38. Gefen A, Cornelissen LH, Gawlitta D, et al. The free diffusion of macromolecules in tissue‐engineered skeletal muscle subjected to large compression strains. J Biomech. 2008;41(4):845‐853. 10.1016/j.jbiomech.2007.10.023 [DOI] [PubMed] [Google Scholar]
- 39. Gawlitta D, Li W, Oomens CW, et al. The relative contributions of compression and hypoxia to development of muscle tissue damage: an in vitro study. Ann Biomedl Eng. 2007;35(2):273‐284. 10.1007/s10439-006-9222-5 [DOI] [PubMed] [Google Scholar]
- 40. Gawlitta D, Oomens CW, Bader DL, et al. Temporal differences in the influence of ischemic factors and deformation on the metabolism of engineered skeletal muscle. J Appl Physiol. 2007;103(2):464‐473. 10.1152/japplphysiol.01374.2006 [DOI] [PubMed] [Google Scholar]
- 41. Oomens CW, Bader DL, Loerakker S, et al. Pressure induced deep tissue injury explained. Ann Biomed Eng. 2015;43(2):297‐305. 10.1007/s10439-014-1202-6 [DOI] [PubMed] [Google Scholar]
- 42. Reswick JB, Rogers JE. Experience at rancho Los amigos hospital with devices and techniques that prevent pressure sores. In: Kenedi RM, Cowden JM, eds. Bedsore Biomechanics. 1st ed. London, UK: The Macmillan Press; 1976:301‐310. [Google Scholar]
- 43. Salcido R, Donofrio JC, Fisher SB, et al. Histopathology of pressure ulcers as a result of sequential computer‐controlled pressure sessions in a fuzzy rat model. Adv Wound Care. 1994;7(5):23‐24. [PubMed] [Google Scholar]
- 44. Stekelenburg A, Oomens CW, Strijkers GJ, et al. Compression‐induced deep tissue injury examined with magnetic resonance imaging and histology. J App Physiol. 2006;100(6):1946‐1954. [DOI] [PubMed] [Google Scholar]
- 45. Breuls RG, Bouten CV, Oomens CW, et al. Compression induced cell damage in engineered muscle tissue: an in vitro model to study pressure ulcer aetiology. Ann Biomed Eng. 2003;31(11):1357‐1364. 10.1114/1.1624602 [DOI] [PubMed] [Google Scholar]
- 46. Breuls RG, Bouten CV, Oomens CW, et al. A theoretical analysis of damage evolution in skeletal muscle tissue with reference to pressure ulcer development. J Biomech Eng. 2003;125(6):902‐909. 10.1115/1.1634287 [DOI] [PubMed] [Google Scholar]
- 47. Friedman R, Haimy A, Epstein Y, Gefen A. Evaluation of helmet and goggle designs by modeling non‐penetrating projectile impacts. Comput Methods Biomech Biomed Engin. 2019;22(3):229‐242. [DOI] [PubMed] [Google Scholar]
- 48. Gefen A. Reswick and Rogers pressure‐time curve for pressure ulcer risk. Part 1. Nurs Stand. 2009;23(45):64‐68. 10.7748/ns2009.07.23.45.64.c7115 [DOI] [PubMed] [Google Scholar]
- 49. Gefen A. Reswick and Rogers pressure‐time curve for pressure ulcer risk. Part 2. Nurs Stand. 2009;23(46):40‐44. 10.7748/ns2009.07.23.46.40.c7169 [DOI] [PubMed] [Google Scholar]
- 50. Lachenbruch C, Tzen Y, Brienza D, et al. The relative effects of interface pressure, shear stress, and temperature on tissue ischemia: a cross‐sectional pilot study. Ostomy Wound Manage. 2013;59(3):25‐34. [PubMed] [Google Scholar]
- 51. Kottner J, Black J, Call E, et al. Microclimate: a critical review in the context of pressure ulcer prevention. Clin Biomech. 2018;59:62‐70. 10.1016/j.clinbiomech.2018.09.010 [DOI] [PubMed] [Google Scholar]
- 52. Zeevi T, Levy A, Brauner N, et al. Effects of ambient conditions on the risk of pressure injuries in bedridden patients‐multi‐physics modelling of microclimate. Int Wound J. 2018;15(3):402‐416. 10.1111/iwj.12877 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Levy A, Kottner J, Gefen A. Release of sodium pyruvate from sacral prophylactic dressings: a computational model. Int Wound J. 2019;16(4):1000‐1008. 10.1111/iwj.13137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Zwanenburg PR, Backer SFM, Obdeijn MC, et al. A systematic review and meta‐analysis of the pressure‐induced vasodilation phenomenon and its role in the pathophysiology of ulcers. Plast Reconstr Surg. 2019;144(4):669e‐681e. 10.1097/PRS.0000000000006090 [DOI] [PubMed] [Google Scholar]
- 55. Amrani G, Gefen A. Which endotracheal tube location minimises the device‐related pressure ulcer risk: the Centre or a corner of the mouth? Int Wound J. 2020;17(2):268‐276. 10.1111/iwj.13267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Brienza DM, Karg PE, Geyer MJ, et al. The relationship between pressure ulcer incidence and buttock‐seat cushion interface pressure in at‐risk elderly wheelchair users. Arch Phys Med Rehabil. 2001;82(4):529‐533. 10.1053/apmr.2001.21854 [DOI] [PubMed] [Google Scholar]
- 57. Lustig M, Wiggermann N, Gefen A. How patient migration in bed affects the sacral soft tissue loading and thereby the risk for a hospital‐acquired pressure injury. Int Wound J. 2020;17(3):631‐640. 10.1111/iwj.13316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Brienza D, Vallely J, Karg P, et al. An MRI investigation of the effects of user anatomy and wheelchair cushion type on tissue deformation. J Tissue Viability. 2018;27(1):42‐53. 10.1016/j.jtv.2017.04.001 [DOI] [PubMed] [Google Scholar]
- 59. Sopher R, Nixon J, Gorecki C, et al. Effects of intramuscular fat infiltration, scarring, and spasticity on the risk for sitting‐acquired deep tissue injury in spinal cord injury patients. J Biomech Eng. 2011;133(2):021011. 10.1115/1.4003325 [DOI] [PubMed] [Google Scholar]
- 60. Knight SL, Taylor RP, Polliack AA, et al. Establishing predictive indicators for the status of loaded soft tissues. J App Physiol. 2001;90(6):2231‐2237. [DOI] [PubMed] [Google Scholar]
- 61. Linder‐Ganz E, Gefen A. The effects of pressure and shear on capillary closure in the microstructure of skeletal muscles. Ann Biomed Eng. 2007;35(12):2095‐2107. 10.1007/s10439-007-9384-9 [DOI] [PubMed] [Google Scholar]
- 62. Shilo M, Gefen A. Identification of capillary blood pressure levels at which capillary collapse is likely in a tissue subjected to large compressive and shear deformations. Comput Methods Biomech Biomed Engin. 2012;15(1):59‐71. 10.1080/10255842.2010.539208 [DOI] [PubMed] [Google Scholar]
- 63. de Wert LA, Geerts M, van der Brug S, et al. The effect of shear force on skin viability in patients with type 2 diabetes. J Diabetes Res. 2019;2019:1973704. 10.1155/2019/1973704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Akins JS, Vallely JJ, Karg PE, et al. Feasibility of freehand ultrasound to measure anatomical features associated with deep tissue injury risk. Med Eng Phys. 2016;38(9):839‐844. 10.1016/j.medengphy.2016.04.026 [DOI] [PubMed] [Google Scholar]
- 65. Gefen A, Gefen N, Linder‐Ganz E, Margulies SS. In vivo muscle stiffening under bone compression promotes deep pressure sores. J Biomech Eng. 2005;127:512‐524. [DOI] [PubMed] [Google Scholar]
- 66. Oomens CW, Bressers OF , Bosboom EM, et al. Can loaded interface characteristics influence strain distributions in muscle adjacent to bony prominences? Comput Methods Biomech Biomed Engin. 2003;6(3):171‐180. 10.1080/1025584031000121034 [DOI] [PubMed] [Google Scholar]
- 67. Peko L, Barakat‐Johnson M, Gefen A. Protecting prone positioned patients from facial pressure ulcers using prophylactic dressings: a timely biomechanical analysis in the context of the COVID‐19 pandemic. Int Wound J. 2020;17(6):1595‐1606. 10.1111/iwj.13435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Todd BA, Thacker JG. Three‐dimensional computer model of the human buttocks, in vivo. J Rehabil Res Dev. 1994;31(2):111‐119. [PubMed] [Google Scholar]
- 69. Linder‐Ganz E, Gefen A. Mechanical compression‐induced pressure sores in rat hindlimb: muscle stiffness, histology, and computational models. J App Physiol. 2004;96(6):2034‐2049. 10.1152/japplphysiol.00888.2003 [DOI] [PubMed] [Google Scholar]
- 70. Dobos G, Gefen A, Blume‐Peytavi U, Kottner J. Weight‐bearing‐induced changes in the microtopography and structural stiffness of human skin in vivo following immobility periods. Wound Repair Regen. 2015;23(1):37‐43. 10.1111/wrr.12259 [DOI] [PubMed] [Google Scholar]
- 71. Gefen A. The bioengineering theory of the key modes of action of a cyanoacrylate liquid skin protectant. Int Wound J. 2020;17(5):1396‐1404. 10.1111/iwj.13401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Gefen A, Alves P, Ciprandi G, et al. Device‐related pressure ulcers: SECURE prevention. J Wound Care. 2020;29(Sup2b):S1‐S52. 10.12968/jowc.2020.29.Sup2a.S1 [DOI] [PubMed] [Google Scholar]
- 73. Gefen A, Ousey K. Update to device‐related pressure ulcers: SECURE prevention. COVID‐19, face masks and skin damage. J Wound Care. 2020;29(5):245‐259. 10.12968/jowc.2020.29.5.245 [DOI] [PubMed] [Google Scholar]
- 74. Tomova‐Simitchieva T, Lichterfeld‐Kottner A, Blume‐Peytavi U, et al. Comparing the effects of 3 different pressure ulcer prevention support surfaces on the structure and function of heel and sacral skin: an exploratory cross‐over trial. Int Wound J. 2018;15(3):429‐437. 10.1111/iwj.12883 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Gefen A. Deep tissue injury from a bioengineering point of view. Ostomy Wound Manag. 2009;55(4):26‐36. [PubMed] [Google Scholar]
- 76. Levy A, Kopplin K, Gefen A. Simulations of skin and subcutaneous tissue loading in the buttocks while regaining weight‐bearing after a push‐up in wheelchair users. J Mechanical Behav Biomed Mater. 2013;28:436‐447. 10.1016/j.jmbbm.2013.04.015 [DOI] [PubMed] [Google Scholar]
- 77. Levy A, Kopplin K, Gefen A. Computer simulations of efficacy of air‐cell‐based cushions in protecting against reoccurrence of pressure ulcers. J Rehabil Res Dev. 2014;51(8):1297‐1319. 10.1682/jrrd.2014.02.0048 [DOI] [PubMed] [Google Scholar]
- 78. Black JM, Berke CT. Deep tissue pressure injuries: identification, treatment, and outcomes among critical care patients. Crit Care Nurs Clin North Am. 2020;32(4):563‐572. 10.1016/j.cnc.2020.08.006 [DOI] [PubMed] [Google Scholar]
- 79. Agam L, Gefen A. Pressure ulcers and deep tissue injury: a bioengineering perspective. J Wound Care. 2007;16(8):336‐342. 10.12968/jowc.2007.16.8.27854 [DOI] [PubMed] [Google Scholar]
- 80. Kottner J, Gefen A, Lahmann N. Weight and pressure ulcer occurrence: a secondary data analysis. Int J Nurs Stud. 2011;48(11):1339‐1348. 10.1016/j.ijnurstu.2011.04.011 [DOI] [PubMed] [Google Scholar]
- 81. Kayser SA, VanGilder CA, Lachenbruch C. Predictors of superficial and severe hospital‐acquired pressure injuries: a cross‐sectional study using the international pressure ulcer prevalence survey. Int J Nurs Stud. 2019;89:46‐52. [DOI] [PubMed] [Google Scholar]
- 82. Gefen A. Risk factors for a pressure‐related deep tissue injury: a theoretical model. Med Biolog Eng Comput. 2007;45(6):563‐573. 10.1007/s11517-007-0187-9 [DOI] [PubMed] [Google Scholar]
- 83. Gefen A. The biomechanics of sitting‐acquired pressure ulcers in patients with spinal cord injury or lesions. Int Wound J. 2007;4(3):222‐231. 10.1111/j.1742-481X.2007.00330.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Gefen A. Bioengineering models of deep tissue injury. Adv Skin Wound Care. 2008;21(1):30‐36. 10.1097/01.ASW.0000305403.89737.6c [DOI] [PubMed] [Google Scholar]
- 85. Gefen A, Farid KJ, Shaywitz I. A review of deep tissue injury development, detection, and prevention: shear savvy. Ostomy Wound Manage. 2013;59(2):26‐35. [PubMed] [Google Scholar]
- 86. Lahmann NA, Kottner J. Relation between pressure, friction and pressure ulcer categories: a secondary data analysis of hospital patients using CHAID methods. Int J Nurs Stud. 2011;48(12):1487‐1494. 10.1016/j.ijnurstu.2011.07.004 [DOI] [PubMed] [Google Scholar]
- 87. Linder‐Ganz E, Gefen A. Stress analyses coupled with damage laws to determine biomechanical risk factors for deep tissue injury during sitting. J Biomech Eng. 2009;131(1):011003. 10.1115/1.3005195 [DOI] [PubMed] [Google Scholar]
- 88. Linder‐Ganz E, Yarnitzky G, Yizhar Z, et al. Real‐time finite element monitoring of sub‐dermal tissue stresses in individuals with spinal cord injury: toward prevention of pressure ulcers. Ann Biomed Eng. 2009;37(2):387‐400. 10.1007/s10439-008-9607-8 [DOI] [PubMed] [Google Scholar]
- 89. Peko Cohen L, Levy A, Shabshin N, Neeman Z, Gefen A. Sacral soft tissue deformations when using a prophylactic multilayer dressing and positioning system. J Wound Ostomy Continence Nurs. 2018;45(5):432‐437. [DOI] [PubMed] [Google Scholar]
- 90. Shabshin N, Ougortsin V, Zoizner G, et al. Evaluation of the effect of trunk tilt on compressive soft tissue deformations under the ischial tuberosities using weight‐bearing MRI. Clin Biomech. 2010;25(5):402‐408. 10.1016/j.clinbiomech.2010.01.019 [DOI] [PubMed] [Google Scholar]
- 91. Shoham N, Gefen A. Deformations, mechanical strains and stresses across the different hierarchical scales in weight‐bearing soft tissues. J Tissue Viability. 2012;21(2):39‐46. 10.1016/j.jtv.2012.03.001 [DOI] [PubMed] [Google Scholar]
- 92. Van Damme N, Van Hecke A, Remue E, et al. Physiological processes of inflammation and edema initiated by sustained mechanical loading in subcutaneous tissues: a scoping review. Wound Repair Regen. 2020;28(2):242‐265. 10.1111/wrr.12777 [DOI] [PubMed] [Google Scholar]
- 93. Coleman S, Nixon J, Keen J, et al. A new pressure ulcer conceptual framework. J Adv Nurs. 2014;70(10):2222‐2234. 10.1111/jan.12405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Black J, Cuddigan J, National Pressure Injury Advisory Panel (NPIAP) Board of Directors . Skin manifestations with COVID‐19: the purple skin and toes that you are seeing may not be deep tissue pressure injury. WCET Journal. 2020;40(2):18‐21. 10.33235/wcet.40.2.18-21 [DOI] [Google Scholar]
- 95. Houwing R, Overgoor M, Kon M, et al. Pressure‐induced skin lesions in pigs: reperfusion injury and the effects of vitamin E. J Wound Care. 2000;9(1):36‐40. 10.12968/jowc.2000.9.1.25939 [DOI] [PubMed] [Google Scholar]
- 96. Ikebe K, Kato T, Yamaga M, et al. Increased ischemia‐reperfusion blood flow impairs the skeletal muscle contractile function. J Surg Res. 2001;99(1):1‐6. 10.1006/jsre.2001.6134 [DOI] [PubMed] [Google Scholar]
- 97. Peirce SM, Skalak TC, Rodeheaver GT. Ischemia‐reperfusion injury in chronic pressure ulcer formation: a skin model in the rat. Wound Repair Regen. 2000;8(1):68‐76. 10.1046/j.1524-475x.2000.00068.x [DOI] [PubMed] [Google Scholar]
- 98. Reid RR, Sull AC, Mogford JE, et al. A novel murine model of cyclical cutaneous ischemia‐reperfusion injury. J Surg Res. 2004;116(1):172‐180. 10.1016/S0022-4804(03)00227-0 [DOI] [PubMed] [Google Scholar]
- 99. Tsuji S, Ichioka S, Sekiya N, et al. Analysis of ischemia‐reperfusion injury in a microcirculatory model of pressure ulcers. Wound Repair Regen. 2005;13(2):209‐215. 10.1111/j.1067-1927.2005.130213.x [DOI] [PubMed] [Google Scholar]
- 100. Unal S, Ozmen S, DemIr Y, et al. The effect of gradually increased blood flow on ischemia‐reperfusion injury. Ann Plast Surg. 2001;47(4):412‐416. 10.1097/00000637-200110000-00009 [DOI] [PubMed] [Google Scholar]
- 101. Yamazaki S, Sekiguchi A, Uchiyama A, et al. Apelin/APJ signaling suppresses the pressure ulcer formation in cutaneous ischemia‐reperfusion injury mouse model. Sci Rep. 2020;10(1):1349. 10.1038/s41598-020-58452-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Gray RJ, Voegeli D, Bader DL. Features of lymphatic dysfunction in compressed skin tissues ‐ implications in pressure ulcer aetiology. J Tissue Viability. 2016;25(1):26‐31. [DOI] [PubMed] [Google Scholar]
- 103. Gray RJ, Worsley PR, Voegeli D, et al. Monitoring contractile dermal lymphatic activity following uniaxial mechanical loading. Med Eng Phys. 2016;38(9):895‐903. 10.1016/j.medengphy.2016.04.020 [DOI] [PubMed] [Google Scholar]
- 104. Bader DL, Barnhill RL, Ryan TJ. Effect of externally applied skin surface forces on tissue vasculature. Arch Phys Med Rehabil. 1986;67(11):807‐811. [PubMed] [Google Scholar]
- 105. Topman G, Lin FH, Gefen A. The influence of ischemic factors on the migration rates of cell types involved in cutaneous and subcutaneous pressure ulcers. Ann Biomed Eng. 2012;40:1929‐1939. [DOI] [PubMed] [Google Scholar]
- 106. Nelissen JL, Traa WA, de Boer HH, et al. An advanced magnetic resonance imaging perspective on the etiology of deep tissue injury. J Appl Physiol. 2018;124(6):1580‐1596. 10.1152/japplphysiol.00891.2017 [DOI] [PubMed] [Google Scholar]
- 107. Gefen A, Weihs D. Cytoskeleton and plasma‐membrane damage resulting from exposure to sustained deformations: a review of the mechanobiology of chronic wounds. Med Eng Phys. 2016;38(9):828‐833. 10.1016/j.medengphy.2016.05.014 [DOI] [PubMed] [Google Scholar]
- 108. Slomka N, Gefen A. Cell‐to‐cell variability in deformations across compressed myoblasts. J Biomech Eng. 2011;133(8):081007. 10.1115/1.4004864 [DOI] [PubMed] [Google Scholar]
- 109. Stekelenburg A, Gawlitta D, Bader DL, et al. Deep tissue injury: how deep is our understanding? Arch Phys Med Rehabil. 2008;89(7):1410‐1413. 10.1016/j.apmr.2008.01.012 [DOI] [PubMed] [Google Scholar]
- 110. Stekelenburg A, Strijkers GJ, Parusel H, et al. Role of ischemia and deformation in the onset of compression‐induced deep tissue injury: MRI‐based studies in a rat model. J App Physiol. 2007;102(5):2002‐2011. [DOI] [PubMed] [Google Scholar]
- 111. Katzengold R, Orlov A, Gefen A. A novel system for dynamic stretching of cell cultures reveals the mechanobiology for delivering better negative pressure wound therapy. Biomech Model Mechanobiol. 2021;20(1):193‐204. 10.1007/s10237-020-01377-6 [DOI] [PubMed] [Google Scholar]
- 112. Toume S, Gefen A, Weihs D. Low‐level stretching accelerates cell migration into a gap. Int Wound J. 2017;14(4):698‐703. 10.1111/iwj.12679 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Ruschkewitz Y, Gefen A. Cell‐level temperature distributions in skeletal muscle post spinal cord injury as related to deep tissue injury. Med Biolog Eng Comput. 2010;48(2):113‐122. 10.1007/s11517-009-0566-5 [DOI] [PubMed] [Google Scholar]
- 114. Ruschkewitz Y, Gefen A. Cellular‐scale transport in deformed skeletal muscle following spinal cord injury. Comput Methods Biomech Biomed Engin. 2011;14(5):411‐424. [DOI] [PubMed] [Google Scholar]
- 115. Leopold E, Gefen A. Stretching affects intracellular oxygen levels: three‐dimensional multiphysics studies. J Biomech Eng. 2012;134(6):064501. 10.1115/1.4006813 [DOI] [PubMed] [Google Scholar]
- 116. Leopold E, Gefen A. A simple stochastic model to explain the sigmoid nature of the strain‐time cellular tolerance curve. J Tissue Viability. 2012;21(1):27‐36. [DOI] [PubMed] [Google Scholar]
- 117. Leopold E, Sopher R, Gefen A. The effect of compressive deformations on the rate of build‐up of oxygen in isolated skeletal muscle cells. Med Eng Phys. 2011;33(9):1072‐1078. 10.1016/j.medengphy.2011.04.008 [DOI] [PubMed] [Google Scholar]
- 118. Slomka N, Gefen A. Confocal microscopy‐based three‐dimensional cell‐specific modeling for large deformation analyses in cellular mechanics. J Biomech Eng. 2010;43(9):1806‐1816. [DOI] [PubMed] [Google Scholar]
- 119. Slomka N, Or‐Tzadikario S, Sassun D, Gefen A. Membrane‐stretch‐induced cell death in deep tissue injury: computer model studies. Cell Mol Bioeng. 2009;2:118‐132. [Google Scholar]
- 120. Edsberg LE, Cutway R, Anain S, et al. Microstructural and mechanical characterization of human tissue at and adjacent to pressure ulcers. J Rehabil Res Dev. 2000;37(4):463‐471. [PubMed] [Google Scholar]
- 121. Gefen A. The sub‐epidermal moisture scanner: the principles of pressure injury prevention using novel early detection technology. Wounds Int. 2018;9(3):10‐15. [Google Scholar]
- 122. Call E, Tanner L, Cheney A, et al. Results of laboratory testing for immersion, envelopment, and horizontal stiffness on turn and position devices to manage pressure injury. Adv Skin Wound Care. 2020;33(10S Suppl 1):S11‐S22. 10.1097/01.ASW.0000696412.04000.98 [DOI] [PubMed] [Google Scholar]
- 123. Bates‐Jensen BM, McCreath HE, Nakagami G, et al. Subepidermal moisture detection of heel pressure injury: the pressure ulcer detection study outcomes. Int Wound J. 2018;15(2):297‐309. 10.1111/iwj.12869 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Bates‐Jensen BM, McCreath HE, Patlan A. Subepidermal moisture detection of pressure induced tissue damage on the trunk: the pressure ulcer detection study outcomes. Wound Repair Regen. 2017;25(3):502‐511. 10.1111/wrr.12548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Bates‐Jensen BM, McCreath HE, Pongquan V, et al. Subepidermal moisture differentiates erythema and stage I pressure ulcers in nursing home residents. Wound Repair Regen. 2008;16(2):189‐197. [DOI] [PubMed] [Google Scholar]
- 126. Gefen A, Ross G. The subepidermal moisture scanner: the technology explained. J Wound Care. 2020;29(Sup2c):S10‐S16. 10.12968/jowc.2020.29.Sup2c.S10 [DOI] [PubMed] [Google Scholar]
- 127. Gershon S. Using subepidermal moisture level as an indicator of early pressure damage to local skin and tissue. Adv Skin Wound Care. 2020;33(9):469‐475. 10.1097/01.ASW.0000655380.86380.7b [DOI] [PubMed] [Google Scholar]
- 128. Okonkwo H, Bryant R, Milne J, et al. A blinded clinical study using a subepidermal moisture biocapacitance measurement device for early detection of pressure injuries. Wound Repair Regen. 2020;28(3):364‐374. 10.1111/wrr.12790 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Peko Cohen L, Gefen A. Phantom testing of the sensitivity and precision of a sub‐epidermal moisture scanner. Int Wound J. 2019;16(4):979‐988. 10.1111/iwj.13132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. Peko L, Gefen A. Sensitivity and laboratory performances of a second‐generation sub‐epidermal moisture measurement device. Int Wound J. 2020;17(3):864‐867. 10.1111/iwj.13339 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Ross G, Gefen A. Assessment of sub‐epidermal moisture by direct measurement of tissue biocapacitance. Med Eng Phys. 2019;73:92‐99. 10.1016/j.medengphy.2019.07.011 [DOI] [PubMed] [Google Scholar]
- 132. Budri AMV, Moore Z, Patton D, O'Connor T, Nugent L, Avsar P. Sub‐epidermal moisture measurement: an evidence‐based approach to the assessment for early evidence of pressure ulcer presence. Int Wound J. 2020;17(6):1615‐1623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Bullkich E, Kimmel E, Golan S. A novel ischemia reperfusion injury hereditary tissue model for pressure ulcers progression. Biomech Model Mechanobiol. 2019;18(6):1847‐1866. 10.1007/s10237-019-01181-x [DOI] [PubMed] [Google Scholar]
- 134. Schwartz D, Gefen A. An integrated experimental‐computational study of the microclimate under dressings applied to intact weight‐bearing skin. Int Wound J. 2020;17(3):562‐577. 10.1111/iwj.13309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Gefen A. How do microclimate factors affect the risk for superficial pressure ulcers: a mathematical modeling study. J Tissue Viability. 2011;20(3):81‐88. 10.1016/j.jtv.2010.10.002 [DOI] [PubMed] [Google Scholar]
- 136. Lechner A, Lahmann N, Neumann K, et al. Dry skin and pressure ulcer risk: a multi‐center cross‐sectional prevalence study in German hospitals and nursing homes. Int J Nurs Stud. 2017;73:63‐69. 10.1016/j.ijnurstu.2017.05.011 [DOI] [PubMed] [Google Scholar]
- 137. Gerhardt LC, Strässle V, Lenz A, et al. Influence of epidermal hydration on the friction of human skin against textiles. J Royal Soc Interface. 2008;5(28):1317‐1328. 10.1098/rsif.2008.0034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Sergio N, Jo‐Ann C, David Y, Eugene HG. Skin friction coefficient: changes induced by skin hydration and emollient application and correlation with perceived skin feel. Soc. Cosmet. Chem. 1981;32:55‐65. [Google Scholar]
- 139. Klaassen M, de Vries EG, Masen MA. The static friction response of non‐glabrous skin as a function of surface energy and environmental conditions. Biotribology. 2017;11:124‐131. 10.1016/j.biotri.2017.05.004 [DOI] [Google Scholar]
- 140. Akdeniz M, Gabriel S, Lichterfeld‐Kottner A, et al. Transepidermal water loss in healthy adults: a systematic review and meta‐analysis update. Br J Dermatol. 2018;179(5):1049‐1055. 10.1111/bjd.17025 [DOI] [PubMed] [Google Scholar]
- 141. DiPasquale DM, Buono MJ, Kolkhorst FW. Effect of skin temperature on the cholinergic sensitivity of the human eccrine sweat gland. Jpn J Physiol. 2003;53(6):427‐430. 10.2170/jjphysiol.53.427 [DOI] [PubMed] [Google Scholar]
- 142. Lechner A, Kottner J, Coleman S, et al. Outcomes for pressure ulcer trials (OUTPUTs) project: review and classification of outcomes reported in pressure ulcer prevention research. Br J Dermatol. 2021;184(4):617‐626. 10.1111/bjd.19304 [DOI] [PubMed] [Google Scholar]
- 143. Traa WA, Strijkers GJ, Bader DL, et al. Myoglobin and troponin concentrations are increased in early stage deep tissue injury. J Mech Behav Biomed Mater. 2019;92:50‐57. 10.1016/j.jmbbm.2018.12.026 [DOI] [PubMed] [Google Scholar]
- 144. Bader DL, Worsley PR. Technologies to monitor the health of loaded skin tissues. Biomed Eng Online. 2018;17(1):40. 10.1186/s12938-018-0470-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Aloweni FAB, Ang SY, Chang YY, et al. Evaluation of infrared technology to detect category I and suspected deep tissue injury in hospitalised patients. J Wound Care. 2019;28(Supplement 12):S9‐S16. 10.12968/jowc.2019.28.Sup12.S9 [DOI] [PubMed] [Google Scholar]
- 146. Amrani G, Peko L, Hoffer O, et al. The microclimate under dressings applied to intact weight‐bearing skin: infrared thermography studies. Clin Biomech. 2020;75:104994. 10.1016/j.clinbiomech.2020.104994 [DOI] [PubMed] [Google Scholar]
- 147. Bhargava A, Chanmugam A, Herman C. Heat transfer model for deep tissue injury: a step towards an early thermographic diagnostic capability. Diagn Pathol. 2014;9:36. 10.1186/1746-1596-9-36 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analysed during the current work.


