Abstract
Chronic diseases, as stated by the WHO, are a threat to human health which kill 3 out of every 5 people worldwide. Therapeutics for such illnesses can be developed using traditional medicine. However, it is not an easy path from natural products to Western pharmacological and pharmaceutical methods. For several decades, chronic inflammatory disorders, especially in Westernized countries, have increased incidence and prevalence. Several NSAIDs are used to decrease inflammation and pain; however, there are numerous negative consequences of these anti-inflammatory medications, whereas plant-based natural products have anti-inflammatory therapeutic benefits that have little or no adverse effects. Nanoparticles are a new type of drug delivery device that may be designed to provide excellent target selectivity for certain cells and tissues while also having a high drug loading capacity, resulting in better pharmacokinetics, pharmacodynamics (PKPD), and therapeutic bioavailability. The size and polarity of phytochemical compounds make it hard to pass the blood–brain barrier (BBB), blood-vessel endothelial lining, gastrointestinal tract and mucosa. In addition, the gastrointestinal system is enzymatically destroyed. Therefore, nanoparticles or nanocrystals might also be used for encapsulation or conjugation of these chemicals as a method to improve their organic effectiveness through their gastrointestinal stability, absorption rate and dispersion. The therapy of numerous inflammatory illnesses, including arthritis, gastritis, Nephritis, Hepatitis (Type A, B &C), ulcerative colitis, Alzheimer's disease, atherosclerosis, allergic responses (asthma, eczema) or autoimmune disorders, is characterised by nanoparticles. This review paper provides information on the numerous nanosystem described with their probable mechanism to treat chronic inflammatory diseases.
Keywords: Nanoparticles, Chronic inflammatory disorder, Severe acute respiratory syndrome, Interferon, Reactive oxygen species
Introduction
Inflammation is a complicated, stereotypic series of reactions, where the body protects against the invasion of foreign organisms (bacteria, viruses, funguses), such as thorn, irritating or microbial organisms and repairs wound cells to tissue. First time in history, it has been explained by Celsus in the first century AD and described four fundamental symbols in the form of "rubor et tumour cum calore et dolore” (redness, swelling, heat, and pain), while in 1858, another scientist Virchow introduced "functio laesa” (disturbance of function) as a fifth cardinal sign of inflammation (Brusini et al. 2020). It acts as a burden and affects the world population by 3–5% (Wang et al. 2015). Three out of five people globally die from chronic inflammatory illnesses, such as stroke, chronic respiratory illnesses, heart illnesses, obesity, and diabetes according to the World Trade Organization (WHO). While no inflammatory disorders appear to be connected with atherosclerosis, glomerulonephritis, hepatitis, diabetes, or different cardiovascular conditions it is possible to take part among illnesses caused by chronic inflammation (Pahwa et al. 2019).
The patient is affected not only by a specific area of the body, but also by a variety of physical responses triggered by the immune system (Nordqvist 2017) and inflammation manifested in the form of redness, rashes, tenderness, heat, long term soreness, swelling, pain, and mobility (loss of function) of a body part, etc. (Shukla et al. 2019). The most important features include oedema formation, fibrin deposition, and the presence of neutrophils in the injured tissue.
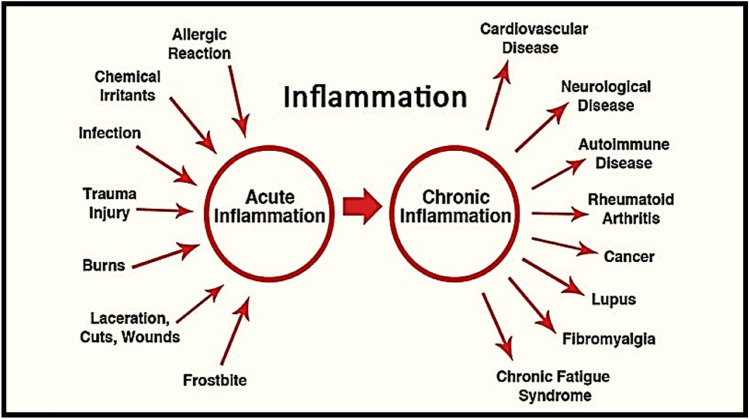
More than 100 different types of inflammatory disease are observed globally (Fig. 1) viz. rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), inflammatory bowel disease (IBD), chronic obstructive pulmonary disease (COPD), asthma, psoriasis, idiopathic pulmonary fibrosis (IPF), type 1 diabetes (T1D) and multiple sclerosis (MS) (Zheng et al. 2021). Inflammation is initiated due to the production of chemical messengers including vasoactive amines or eicosanoids, products of multi-enzyme systems of blood plasma (complement, hemocoagulation, fibrinolytic, and kinin), various hormones, neurotransmitters, and neuropeptides (Malone 2016).
Fig. 1.
Schematic representation of different types of acute and chronic inflammation
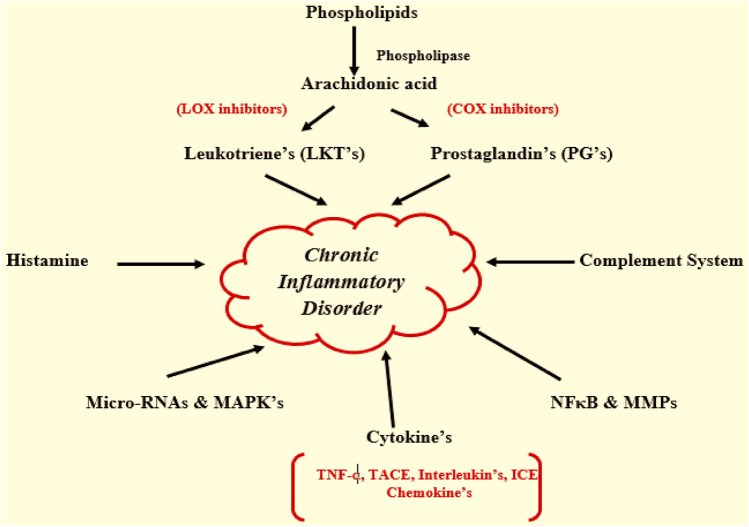
Figure 2 shows several chemical messengers are involved during the inflammation phenomena, such as lipid mediators—Eicosanoids (prostanoids, leukotrienes, thromboxanes, platelet-activating factors—(PAF) (Diegelmann 2016), vasodilators (NO, PGI2, PGE2, PGD2), vasoconstrictors (endothelins, TXA2, PGG2, PGH2), vascular endothelial products, chemotactic factors (C5a), Mast cells (histamine), bradykinins, and prostaglandins (Burini et al. 2020). Inflammatory mediators are solutions that release diffuses chemicals from inflammatory or other cells. They operate both locally at tissue injury and infection sites and anatomically far away (Miyasaka and Takatsu 2016).
Fig. 2.
Chemical messengers involved during the inflammation phenomena (Bhavya and Haridas, 2017)
The mediators (Ley 2018) are divided into:
Exogenous: (bacterial products and toxic substances mainly (e.g., lipopolysack-like Gram-negative bacteria)
Endogenous (activity-like, complementary, hemocoagulatory, fibrinolytic, and kininetic systems, inflammation regulate and homeostasis)
Kinds of inflammation
Inflammation can be classified according to whether it is induced by an external substance or an endogenous aberrant reaction. Inflammation can be acute or chronic depending on how long it lasts (Xiao 2017).
Acute inflammation
Acute inflammation symptoms often last a few weeks, but sub-acute inflammation symptoms later 2–6 weeks. The body doubles the number of leukocytes while in an acute inflammatory reaction (i.e., White blood cells or granular cells) in the affected tissue site. These are the main cells involved in the acute inflammation process containing eosinophil’s, mononuclear cells (monocytes and macrophages) and primary neutrophils, serum proteins (e.g., complement, C-reactive protein), cell receptors (e.g., Toll-like receptors activation), granulocyte–monocyte colony-stimulating factor (GM-CSF), interferon-gamma (IFN-g) from pathogen incursion (Wynn et al. 2013) and cells that discharge cytokines and inflammatory markers (i.e., macrophages, mast cells, natural killer cells) (Cronkite and Strutt 2018). White blood cells (leukocytes) are drawn to the location to help the elimination of wreckages (Shi et al. 2012). Thus, the objective of inflammation is to clean up the damage and begin the healing process, which is then followed by several phases that continue to involve the immune system until the injury or infection returns to equilibrium (Schauss 2013). Above mentioned symptoms and signs are sometimes not observed in the patient, such type of inflammation is known as "silent inflammation."
Chronic inflammation
Chronic inflammation is defined as chronic, low-level inflammation that might last for months, years, or even a lifetime. Constant tiredness, mouth sores, joint, chest, or stomach discomfort, rash, and fever are symptoms and indications of this kind of inflammation. On the other hand, the main cause of chronic inflammation is a longer period of perceptible of the harmful chemical or pathogen. Sometimes, defence mechanisms cannot be controlled easily and hence it damages utmost body parts viz. nervous and musculoskeletal systems, and blood vessels (Pahwa and Jilal 2018) and produces sepsis, asthma, obesity, type 2 diabetes, neurodegenerative diseases (Alzheimer's disease), cardiovascular diseases (e.g., atherosclerosis, cardiac ischemia/reperfusion), cancer, bowel disease, and few scientific journals also have reported Crohn disease, rheumatoid polyarthritis (Molinaro et al. 2016). It has been linked with certain other types of diseases, such as autoimmune diseases (e.g., allergies or hypersensitivity reactions, atopic dermatitis, psoriasis, asthma, chronic obstructive pulmonary disease(COPD), arthritis (osteoarthritis, rheumatoid arthritis), inflammatory bowel diseases (ulcerative colitis), celiac disease, auto inflammatory syndrome, or inflammation accompanying transplant rejection), diabetes, cardiovascular disease (CVD), etc. (Abdulkhaleq et al. 2018).
It is most likely happens due to the continued production of inflammatory cytokines (i.e., interferon-gamma (IFN-γ), IL-2, IL-4, IL-6, IL-15, and IL-17) and fabrication of reactive oxygen species (ROS) forms oxidative stress and (hypoxia) metabolic stress (Schauss 2013). The pathophysiology of pain is influenced by pro-inflammatory cytokines such as IL-1, IL-6, and TNF. Interleukin (IL-6) is important in the neural response to microglial, astrocytic activation, nerve injury, and the regulation of a neuronal neuropeptides phase. TNFR1 and TNFR2 are two key cell surface receptors that regulate NF-β activation, inflammation, and the activation of stress-activated protein kinases (SAPKs) and apoptotic pathways. TNF-α, another inflammatory cytokine, is required for innate and adaptive immunity, proinflammatory characteristics, cell proliferation, and inflammatory and neuropathic hyperalgesia (Tanaka et al. 2014).
Comprising the pathophysiology of inflammation, proteins produced (Zarrin et al. 2021) by particular cells during inflammation may be identified (e.g., ICAM, VCAM, and selectins). Small compounds can influence three different types of protein targets (Durymanov et al. 2017).
Inflammatory mediators' synthesis is regulated by enzyme kinases.
In nucleic acid sensing pathways, receptors and critical signalling molecules.
Soluble ligands (i.e., cytokines and receptors on the cell surface).
Therapeutic strategies for inflammation
High dosages of medicines that might sometimes generate adverse effects on healthy tissue are needed to accomplish the intended therapeutic effects on inflammatory cells. In addition, therapies are categorized in such a way (Whalen et al. 2019).
Non-drug (rehabilitation, physiatry, balneology)
Non-steroidal anti-inflammatory (Trevor et al. 2019) drugs, such as (indomethacin, diclofenac, ibuprofen, piroxicam, tenoxicam, meloxicam, nimesulide, celecoxib, parecoxib, lumiracoxib, aspirin, and naproxen etc.).
Glucocorticoids (hydrocortisone, prednisolone, methylprednisolone, triamcinolone, dexamethasone, betamethasone, etc.).
Immunosuppressant drugs, i.e., Cyclosporin A, Tacrolimus, Pimecrolimus (Ghasemian and Owlia 2015)
Nowadays, the treatment of acute and chronic inflammation has also been done using steroidal (betamethasone, prednisone, and dexamethasone). In the case of NSAIDs, lower doses are mostly accessible for treating inflammatory pain, but greater dosages of NSAIDs and steroidal drugs can be used as prescription drugs (Nordqvist 2017). Both are advantageous to treat chronic and acute inflammation through inhibition of cyclooxygenase (COX); however, excess use can produce some side effects in the tissue or cell. Furthermore, autoimmune diseases such as rheumatoid arthritis (RA) can be treated by anti-rheumatic agents (auranofin, aurothiomalate, d-penicillamine), aminosalicylates (sulfasalazine) (Mayoclinic 2020), and others are disease-modifying anti-rheumatic agents (DMARDs) such as methotrexate (MTX) are effective to slow down the RA development. A combined form of the drug (DMARDs with Glucocorticoids) may be helpful for the prevention and management of anti-inflammatory action and remove the complications that arise due to dose enhancement (Wailoo et al. 2014). In addition, asthmatic disorder and chronic obstructive pulmonary disorder (COPD) can be prevented or treated using combinational therapy (β2-mimetics, anticholinergic agent, phosphodiesterase-4 antagonist, and long-acting theophylline–methylxanthines) (Whalen et al. 2019).
Recently, two additional therapies were introduced in the market which may be breakthroughs in the helpful to indulgence chronic and acute inflammation for example the use of anti-leukotrienes (LTs) that constrain the necessity of inflammatory cells (anti-TNF-α or anti-IL-1 monoclonal antibodies) and proinflammatory cytokine inhibitors (Tabas 2013). However, sometimes treatment is not suitable to obtain up to the mark pharmacological activity and non-specific bio-distribution, poor bioavailability, and/or limited shelf life are the major demerits with the use of the drug.
The quest for novel anti-inflammatory drugs is becoming popular to minimize side impacts and pharmacological restrictions, to provide improved safety, effectiveness, and more economic treatment of inflammation.
Medicinal plant and their phytochemicals for anti-inflammation therapy
The utilization of natural ingredients as anti-inflammatory drugs has recently sparked increased interest in the pharmaceutical industry. The utilization of natural ingredients as anti-inflammatory and anticancer drugs has recently sparked increased interest in the pharmaceutical industry. The natural product has long been used for treating human diseases as inflammatory disorders, i.e., Crohn's disease, and ulcerative colitis. Inflammatory bowel disease (IBD) (chronic inflammation in the GIT zone or Intestinal tract). Secondary metabolites of the plants (Alkaloids, glycosides, flavonoids, phenolics and polyphenols, saponins, tannins, terpenes, anthraquinones, essential oils, and steroids) are only some of the bioactive molecules that may be employed to make pharmacological derivatives to fight inflammation (Salibay et al. 2021). The primary sources of natural agents that are potential bioactive natural chemicals in the pharmaceutical industry include plants, animals, microorganisms and aquatic animals. The phytoactive ingredients of medicinal plants are widely utilised for many therapeutic purposes and, owing to their accessibility and affordability, provide an appropriate alternative to conventional medicine.
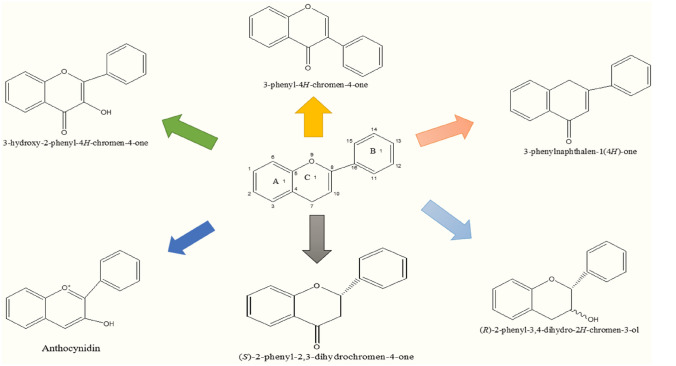
Phenolic compounds are the secondary metabolite and are structurally different from the hydroxyl group-containing cyclic aromatic rings (i.e., Tannin and lignin). They perform a strong antioxidant and anti-inflammatory activity at the broad spectrum level (Davatgaran-Taghipour et al. 2017). These derivatives are divided into main classes depending on the amount and arrangement of their carbon units: Flavonoids and Non-Flavonoids. More than 6000 phenolic constituents are renowned for their antioxidant activity. Flavonoids are further classified depending on chemical structure and number of hydroxyl sets and degree of alkylation or glycosylation. Subgroups of flavonoids are shown in Fig. 3: Flavones (i.e., apigenin and luteolin), flavanol (i.e., epigallocatechin-3-gallate—EGCG) (Singh et al. 2016), flavonols (i.e., QC & kaempferol) (Barreca et al. 2016), isoflavones (i.e., daidzein and genistein) (Aras et al. 2015), flavanones (i.e., naringenin & hesperetin) (Cirmi et al. 2016), and anthocyanins (i.e., cyanidin & delphinidin) (Strathearn et al. 2014) are the best-known flavonoids which are helpful in various aliments (i.e., anti-inflammatory, neurological diseases, and cardiovascular disorder).
Fig. 3.
Basic skeleton structure of flavonoids and their classes
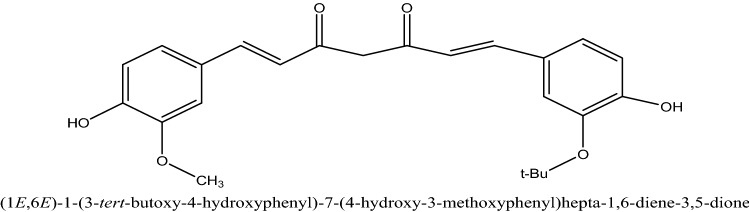
Bioactive compound Curcumin (1E, 6E)-1, 7-bis (4-hydroxy-methoxyphenyl)-1, 6 heptadiene-3, 5-dioneI) which is extracted from Curcuma longa roots and rhizome used in anti-inflammatory activity. Recent scientific reports suggest that nanoformulation of Nanocurcumin regulates the inflammatory response by inhibiting the activity of inducible nitric oxide synthase (iNOS), lipoxygenase (LOX), phospholipases A2 (PLA2s), and cyclooxygenase-2 (COX-2) enzyme pathway, which obstructs the synthesis of prostaglandin, pro-inflammatory leukotrienes an essential inflammatory response mediators (Farhood et al. 2019). Its inflammatory response is linked to an arachidonic acid production route for eicosanoid formation (Lee et al. 2020) (see Fig. 4).
Fig. 4.
Chemical structure of Curcumin
Glycyrrhizin is a bioactive component of liquorice derived from the root of Glycyrrhiza glabra that contains pentacyclic triterpenoid and glycosidic saponin shows remarkable anti-inflammatory activity by inhibition of nuclear transcription factor (NF-kB) which play a vivacious role to regulate inflammation in human beings (Wang et al. 2020) and inhibiting TNF, MMPs, PGE2 and free radicals (Yang et al. 2017). Other plant-derived bioactive natural products resveratrol and stilbenes derivatives (Yuccaols A, B, C, D, and E) obtained from Yucca schidigera extract have shown potent anti-inflammatory activity through inhibition of the NF-kB, which stimulates the synthesis of NOS2 (Reinisalo et al. 2015) (see Fig. 5).
Fig. 5.
Chemical structure of Glycyrrhizin
Nanoparticles
Nanoparticles (NPs) are the nanocarriers or natural colloids/synthetic colloids types of NPs which are available in different forms of colloids carriers, such as liposomes, micelles, dendrimers, inorganic or metallic nanoparticles, and solid–lipid nanoparticles (SLNs). Furthermore, Nanomedicine provides novel treatment possibilities for many drugs which cannot efficiently be employed as traditional formulations (Saraiva et al. 2016). In addition, other drug delivery system includes polymeric nanoparticles, carbon nanotubes, quantum dots, gold nanoparticles, magnetic nanoparticles, polymeric micelles, and viral vectors (Tekade et al. 2017). It is found in various shapes and sizes in the range (1×10-9) have attached by covalent bonds or adsorbed to the surface of a particle or enclosed within a structure, such as liposomes and dendrimers (Landriscina et al. 2015). The desire for better treatments for inflammatory, developmental, viral, and degenerative nervous system diseases has driven its fast growth (Gendelman et al. 2015). In anti-inflammatory therapy, various hybrid NPs have recently been explored. According to current research, one or even more materials are formed into nanostructures, and the properties of every material are extensively used for enhanced biocompatibility and targeting ability. (Agrahari et al. 2019). Current literature shows that the size, shape, biocompatibility, and biodegradability of the component, the carrier and drug stability of many parameters impact the targeting of nanoparticles, the zeta and hydrophobic potential, and surface functionality in case of neurodegenerative illnesses. In this sense, poly (lactic-co-glycolic acid (PLGA) is the most promising hydrophobic synthetic polymer (Mir et al. 2017) utilized as the medication carrier to treat neurodegenerative illnesses (Alzheimer's disease, and Parkinson's disease) by the inhibition of galectin-3 and caspase-8 (cause of neuroinflammation). Therefore, several Nanomedicines were manufactured with cancer (Iqbal et al. 2018), cardiovascular pathology (Dormont and Couverur 2018), autoimmune diseases (Gharagozloo et al. 2015), metabolic syndrome (Veiseh et al. 2015), and neurodegenerative diseases (Goldsmith et al. 2014).
Recent advancement of nanoparticles system for the treatment of inflammation
Nanocrystals are a new nanotechnology field that pertains to the BCS class II medications. According to Nanotechnology in Drug Delivery-Global Market Trajectory & Analytics, it is predicted at US$ 124.7 billion in 2027, with drug nano crystal forecast to reach US$ 83.1 billion by the conclusion of the analysis period, despite the coronavirus pandemic crisis and resulting economic slowdown (Research and Markets 2020). Reducing drug particle size improves the bioavailability of BCS class II medications and may help BCS class IV pharmaceuticals with low solubility and permeation, since increasing drug water solubility raises the concentration gradient and improves drug permeability (Peltonen and Hirvonen 2018). The nanocrystal approach, which is based on the diffusion of nanoparticles through an unstirred water layer, can be used to improve drug permeability (Wood et al. 2018). Furthermore, the mucus layer, which includes mucin glycoproteins, is thought to be a biological barrier to medication absorption; lipids, inorganic salts, and water were chosen based on quick diffusion (Liu et al. 2020). The main advantages of nanocrystals are: increased dissolving rate, increased saturation solubility, enhanced mucoadhesive, and reduced fed/fasted state variance. Furthermore, the increased bioavailability given by nanocrystal technology results in a faster beginning of the action, improving the quality of life in patients receiving anti-inflammatory medication therapy for pain relief. These characteristics enhance the effectiveness and tolerability of anti-inflammatory medicines. They are an appealing method that leads to a significant change in the creation of novel items for various administration routes. The ability to target can reduce side effects and enhance efficacy in the treatment of inflammatory disorders. Bottom-up and top-down technologies, as well as mixtures of the two, are used in the manufacture of drug nanocrystals. The bottom-up process involves the formation of tiny particles from individual molecules. Because drug particles are precipitated from a supersaturated solution, this procedure is also known as nano-precipitation (Sinha et al. 2013). The driving force for crystal development is supersaturation, which is accomplished by cooling down or adding an anti-solvent (De Waard et al. 2011). The most significant industrial technique in micronization is the top-down method. These possible recommendations from the bottom-up in that it does not use organic solvents has better control over particle form and size and is easier to scale up (Salazar et al. 2012). It is a high-energy method in which micron-sized pharmaceuticals are suspended in an aqueous or non-aqueous dispersion media including stabilisers and reduced to the nanoscale by mechanical attrition (wet bead milling or high-pressure homogenization) (Tuomela et al. 2016b). In our findings, it is also the most commonly reported anti-inflammatory medication nano crystal technique. Various nanocrystals have recently been produced for usage in a variety of delivery routes, including oral, topical, parenteral, and ophthalmic. Table 1 shows anti-inflammatory medication nano crystals that can be made in a variety of ways and used in a variety of ways.
Table 1.
Anti-inflammatory drug nano crystal using different preparation methods
| Drug | Route of administration | Method of preparation | References |
|---|---|---|---|
| Indomethacin | Oral | Wet bead milling | Kuroiwa et al. (2018), Liu et al. 2013, 2015) |
| Meloxicam | Liu et al. (2018), Ochi et al. (2014) | ||
| Acelofenac | Narayan et al. (2017) | ||
| Naproxan | Kumar and Burgess (2014) | ||
| Nimesulide | Gulsun et al. (2013) | ||
| Dexibuprofen | Bottom-up technology | Ullah et al. (2018) | |
| Hydrocortisone acetate | High-pressure homogenization | Moschwitzer (2013) | |
| Ibuprofen | Fernandes et al. (2017) | ||
| Celecoxib | He et al. (2017) | ||
| Dexamethasone | Topical | Wet bead milling | Doge et al. (2016) |
| Sodium diclofenac acid | Pireddu et al. (2015) | ||
| Flurbiprofen | High-pressure homogenization | Oktay et al. (2018) | |
| Beclomethasone dipropionate | Bottom-up technology | Assem et al., 2019 | |
| Meloxicam | Yu et al. (2018) | ||
| Methylprednisolone | Karabey-Akyurek et al. (2017) | ||
| Hyaluronic acid/chitosan | Parenteral | Negut and Grumezescu (2021) | |
| Squalene–adenosine NPs | Flavio Dormont et al. (2020) | ||
| Etopozide | Solvent evaporation method | Yadav and Sawant (2010) |
Nanoparticles (NP) may adsorb proteins from biological fluids and produce a protein layer known as a protein corona. The physicochemical features of NPs, such as size, shape, and surface chemistry, influence the protein corona composition (Schmid et al. 2017). Protein nanoparticles have various advantages as a drug delivery technology, including biodegradability, stability, particle surface modification, ease of particle size management, and fewer toxicity concerns, such as immunogenicity (Chu et al. 2017). When nanomaterial’s are administered intravenously (i.v.), they are instantly coated by a layer of blood proteins known as the protein corona or bimolecular corona (Oh et al. 2018). The amount and kind of protein corona vary a lot depending on the physicochemical properties of nanoparticles, which consequently changes the nano-biological interfaces including targeting effectiveness, immunogenicity, and intracellular toxicity (Walkey et al. 2012). To reduce the problems produced by the protein corona and prevent clearance by the mononuclear phagocyte system (MPS), generally, pro coatings of nanoparticles such as poly (ethylene glycol) decorating were first utilised (Tenzer et al. 2013). According to recent research, the deliberate adsorption of specific types of proteins, particularly dysopsonins, can change the character of nanomaterial’s (Dai et al. 2014).
For the treatment of inflammation, many kinds of nanoparticles (liposomes, polymer nanoparticles, micelles, dendrimers, and hydrogel-based formulations) have been created or are presently in pre-clinical research. The first closed system for bilayer phospholipids was the liposomes originally provided as nanomedicines and is used for different types of cancer in clinical studies. Liposomal nanoparticles that have anti-inflammatory properties are currently being investigated (Chang et al. 2020). Furthermore, the use of nanocarriers to the specific target effector cells particularly antigen-presenting cells can be of great benefit in supporting cell response because of their module capacity, which allows them passively to optimise the size and/or surface load of nanoparticles or activate the nanoparticles for antimicrobial studies (Dacoba et al. 2017).To avoid the adverse effects found in traditional treatment, much emphasis has been given to developing more effective anti-inflammatory nanomedicines. Table 2 includes many examples of new revolutionary nanomedicines systems for pre-clinical treatment for inflammatory diseases. To present, FDA has authorized just a few tenths of the nanomedicines and several hundred new clinical trials. Three additional Patisiran/ONPATTRO, VYXEOS, and NBTXR3/Hensify nanomedicines have recently been authorized. However, few anti-inflammatory nanoparticles were able to carry out clinical investigations to our knowledge. Table 3 provides instances of nanomedicines in clinical trials.
Table 2.
Some examples of innovative pre-clinical nanomedicines system for chronic inflammatory diseases
| Nanoparticles (Nps) | Main Compound | Inflammatory lesions | References |
|---|---|---|---|
| N-(2-hydroxypropyl) methacrylamide (HPMA) copolymer NPs | Dexamethasone | Arthritis | Jia et al. (2020) |
| Nanoemulsion formulation of cationic lipid DOTAP | TNF-α siRNA | Parkinson’s disease | Yadav et al. (2019) |
| Omega-3 fatty acid-rich flaxseed oil-based nanoemulsion system | Cyclosporin A | neuroinflammation | |
| Pegylated liposomes | Prednisolone | Renal ischemia and reperfusion injury | Van Alem et al. (2018) |
| Hyaluronic acid/chitosan NPs | Cytokine response modifier A pDNA | Osteoarthritis | Zhou et al. (2018a, b) |
| Hyaluronic acid-coated solid–lipid NPs | Prednisolone | Arthritis | |
| Spherical polymeric Nano constructs | Dexamethasone | Lee et al. (2020) | |
| Solid–lipid NPs | Dexamethasone and Butyrate | Dianzani et al. (2017) |
Table 3.
Nanomedicines under clinical trials
| Nanocarrier/Nanoparticles (NPs) | Principle compound | Disorder | References |
|---|---|---|---|
| Nanoemulsion (oil in water emulsion) | Tretinoin | Acne vulgaris | Sabouri et al. (2018) |
| Liposome | Cyclosporine (lipogel or cream) | Psoriasis | Kumar et al. (2016) |
| PEGylated liposomes | Prednisolone | Ulcerative colitis | Enceladus Pharmaceuticals. (2016) |
Herbal medicines and natural products nanocarriers explored so far for chronic inflammatory diseases
Herbal medicine is a great source for inflammatory disease prevention and therapy. The current era has included the creation of nanoformulations using numerous natural or phtyocompounds.
The derivatives of phenolic and terpene are removed from the plan and are not stable for a long time owing to environmental conditions so that nanoformulations can be kept. It displays a harsh taste when taken orally, therefore, a high derivative or dose concentration was needed for therapy. Furthermore, to overcome the drawbacks of administering natural products through the use of supramolecular nanotherapeutics, which improve biopharmaceutical characteristics following injection via an oral, local, and systematic route.
For the prevention of inflammation, a variety of synthetic or organic compounds are increasingly being utilized. Organic nanoparticles (NPs) have higher biocompatibility than inorganic nanoparticles, and most carriers are made of biomaterials or polymers (Mir et al. 2017). In comparison, when using PLGA as a carrier, the organic nanoparticles form numerous structures with greater stability. When exosome is produced, they produce enough source and organic targeting impact (Wu et al. 2020). For liposomes, the nature of liposomes is simple, extremely stable, and biodegradable. When silver and gold (Ag/Au) are applied as a transport in the case of inorganic nanoparticles, they are employed to act antibacterial or bacterial. Carbon nanotube (Jia and Wei 2017) utilized as an organic nano parts carrier provides excellent biocompatibility, whereas quantum dot (Kalangi et al. 2018) has the best targeting and the capacity to imagine bio-carbon. Although the advantages for both organic and inorganic nanoparticles may be many, some disadvantages have been discovered. With organic nanoparticles, the structurally homogeneous character of pure exosomes is difficult to produce. They are very unstable when dissolved in water molecules; therefore, the product's shelf life is quite short. Inorganic nanoparticles are very unstable, have significant biological toxicity, and have a poor targeting impact (Yan et al. 2020).
Drug delivery methods based on organic nanoparticles are now commercially available in a variety of forms, such as Lutetium-177-1,4,7,10-tetraazacyclododecane–1,4,7,10-tetraacetic acid–hyaluronic acid–poly (lactic-co-glycolic acid) (Trujillo Nolasco et al. 2019), Dexamethasone (Yan et al. 2020) for Rheumatic arthritis. Among the commercially available inorganic nanoparticle-based drug delivery systems are Monocycte (Shi et al. 2020a) for osteomyelitis, Celecoxib (Kalangi et al. 2018) for oedema, and SAM (sodium 10-amino-2-methoxyundecanoate) (Kumar et al. 2016).
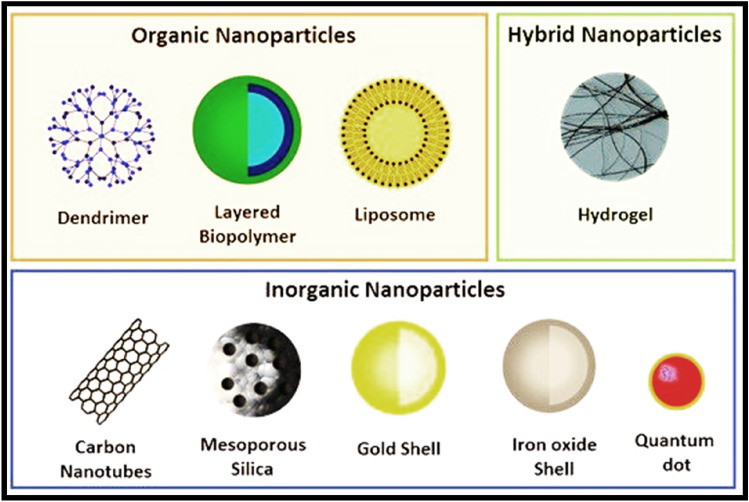
Figure 6 depicts several types of nanoparticles (NPs). Nanoparticles (NCs) or nanoparticles (NPs) can be made from a variety of organic and inorganic constituents (Metallic/metallic oxide NPs, Metallic NPs, and Metallic oxide NPs) including biodegradable and nondegradable polymers (polymeric nanoparticles (PNPs), polymeric conjugates), lipids (solid–lipid nanoparticles (SLNs), liposomes, and Nanoemulsions), dendrimers, micelles, nanocrystals, nanofibers, quantum dots, Nanodiamonds, etc. The natural compound-based nanoparticle formulations reported in Table 4 have aided in the treatment of different inflammatory disorders.
Fig. 6.
Schematic representation of different types of nanoparticles (NPs) divided into organic, hybrid, and inorganic categories (Silva et al. 2019)
Table 4.
Natural compound-based nanoparticle formulations
| Nano formulation | Main Compound | Plant name | References |
|---|---|---|---|
| Liposome | Berberine (BBR) | Berberis thunbergii | Duong et al. (2021) |
| Ethosome | Alpha Phellandrene | Moringa oleifera | Valsalan Soba et al. (2021) |
| Niosomes | Monoammonium Glycyrrhizinate | Glycyrrhiza glabra | Maione et al. (2019) |
| Solid–Lipid Nanoparticles (SLNs) | Curcumin | Curcuma domestica | Ganesan et al. (2019) |
| Nanostructured lipid (NLCs) | Capsaicinoids (Capsaicin-8-methyl-N-vanillyl-6-nonenamide | Capsicum annuum | Nava-Ochoa et al. (2021) |
| Dendrimers | Sinomenine (7,8-didehydro-4-hydroxy3,7-dimethoxy-17-methyl9α, 13α,14α-morphinan-6-one; Sino) | Sinomenium acutum | Sharma et al. (2020) |
| Nanoemulsion | Cordycepin (3′-Deoxyadenosine) | Cordyceps militaris | Rupa et al. (2020) |
|
Polymeric Nanogels and Nanosuspensions |
Aloe-emodin (1, 8-Dihydroxy-3-(hydroxymethyl)-9, 10-anthraquinone) | Aloe vera | Divya et al. (2016) |
| Carbon Nanotubes (CNTs) and Nanofibers | Bis-demethoxy curcumin | Curcuma longa | Francis et al. (2018) |
| Polymeric Nanomicelles | Sesamol | Sesamum indicum | Ning et al. (2021) |
| Polymeric Nanoliposomes | Curcumin | Curcuma longa | Sogut et al. (2020) |
| Phospholipid–nanoparticles (PLN) | Thymoquinone (TQ) | Nigella sativa | Rathore et al. (2019) |
Polymer-based on nanoparticles can efficiently carry medicines, proteins, macromolecules, natural chemicals, hydrophobic and hydrophilic products, enhance their pharmaceutical and biopharmaceuticals characteristics and minimize the adverse effects. Poly (lacticco-glycolic acid) is a synthetic hydrophobic polymer that has been identified as the most effective polymer employed as a vehicle in the transport of medicines and has tremendous potential to target and treat drug incorporation into PLGA and is well researched in the formation of complexes. The polymers are also authorized for pharmaceutical usage by the FDA and the EMA (Anderson and Shive 1997). The polymers based on nanoparticles have two primary structures which rely on raw material collection (Di Marzio et al. 2016): (a) nanospherescore structure: the polymer matrix is distributed and or adsorbed by the Bioactive Compound(s). (b) Nanocapsules: displays water or oil loading medication and a polymer shell.
If NPs reach the outer membrane of a cell, they can interact with plasma or extracellular membrane components and enter the cell mostly by endocytosis. It leads to the absorption and squeezing in membrane invaginations of NPs into endocytic vesicles, which are ultimately transported into specific intracellular trial and trafficking compartments. Endocytosis may be categorized into various kinds, depending on the cell type, proteins, lipids, and other process molecules (Behzadi et al. 2017). Phagocytosis, Clathrin-mediated endocytosis pathway (≤200 nm) Caveolae-dependent pathway (≥ 500 nm) Clathrin/Caveolae-independent endocytosis and macro pinocytosis (0.2–5 μm).
Conclusions
Most inflammatory disorders are chronic thus therapies are needed for their treatment with an adequate therapeutic index. Alongside the advancement in sophisticated nanoscience and significant insights into nanomedicine technology, which helps build translational trust for effective use, the toolkit for clinical efficacy is enriched by innovations in chemical biology and medicinal chemistry.
In short, drug delivery systems based on nanomedicines for the prevention, diagnosis and treatment of chronic inflammatory diseases are offering good possibilities for clinical application which should be a new paradigm in chronic inflammatory pharmacotherapy.
Acknowledgements
I would like to appreciate GD Goenka University, Gurugram-India for the valuable support and encouragement for this manuscript.
Author contributions
TG and VJ wrote the manuscript and tables and figures drawn by MS. VD polished the language and RD supervised the work. The final version of the manuscript has been approved by all authors.
Funding
The authors have not disclosed any funding.
Data availability
Enquiries about data availability should be directed to the authors.
Declarations
Conflict of interest
All authors declare no conflict of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Rohit Dutt, Email: rohitdatt23@rediffmail.com.
Vandana Garg, Email: vandugarg@rediffmail.com.
Tinku, Email: tinkujhajjar87@gmail.com, Email: tinku.gupta@gdgu.org.
References
- Abdulkhaleq LA, Assi MA, Abdullah R, Zamri-Saad M, TaufiqYap YH, Hezmee MNM. The crucial roles of inflammatory mediators in inflammation: a review. Veterinary World. 2018;11(5):627–635. doi: 10.14202/vetworld.2018.627-635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agrahari V, Burnouf PA, Burnouf T. Nanoformulation properties, characterization, and behaviour in complex biological matrices: challenges and opportunities for brain-targeted drug delivery applications and enhanced translational potential. Adv Drug Deliv Rev. 2019;148:146–180. doi: 10.1016/j.addr.2019.02.008. [DOI] [PubMed] [Google Scholar]
- Anderson JM, Shive SS. Biodegradation and biocompatibility of PLA and PLGA microspheres. Adv Drug Deliv Rev. 1997;28:5e24. doi: 10.1016/s0169-409x(97)00048-3. [DOI] [PubMed] [Google Scholar]
- Aras A, Guven M, Akman T, Ozkan A, Sen H, Duz U, et al. Neuroprotective effects of daidzein on focal cerebral ischemia injury in rats. Neural Regen. Res. 2015;10:146–152. doi: 10.4103/1673-5374.150724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Assem M, Khowessah OM, Ghorab D. Nano-crystallization as a tool for the enhancement of beclomethasone dipropionate dermal deposition: Formulation, in vitro characterization and ex vivo study. J Drug Deliv Sci Technol. 2019;54:101318. [Google Scholar]
- Barreca D, Bellocco E, D’onofrio G, Nabavi SF, Daglia M, Rastrelli L, et al. Neuroprotective effects of quercetin: from chemistry to medicine. CNS Neurol Disord Drug Targets. 2016;15:964–975. doi: 10.2174/1871527315666160813175406. [DOI] [PubMed] [Google Scholar]
- Behzadi S, Serpooshan V, Tao W, et al. Cellular uptake of nanoparticles: journey inside the cell. Chem Soc Rev. 2017;46(14):4218–4244. doi: 10.1039/c6cs00636a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhavya BC, Haridas M. Anti-inflammatory molecules: immune system mediators. In: Sugathan S, Pradeep N, Abdulhameed S, editors. Bioresources and bioprocess in biotechnology. Springer Singapore; 2017. pp. 235–268. [Google Scholar]
- Brusini R, Varna M, Couvreur R. Advanced nanomedicines for the treatment of inflammatory diseases. Adv Drug Deliv Rev. 2020;157:161–178. doi: 10.1016/j.addr.2020.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burini RC, Anderson E, Durstine JL, Carson JA. Inflammation, physical activity, and chronic disease: An evolutionary perspective. Sports Med Health Sci. 2020;2(1):1–6. doi: 10.1016/j.smhs.2020.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang MC, Kuo YJ, Hung KH, Peng C-L, Chen K-Y, Yeh L-K. Liposomaldexamethasone-moxifloxacin nanoparticle combinations with collagen/gelatin/alginate hydrogel for cornealinfection treatment and wound healing. Biomed. Mater. 2020;15:055022. doi: 10.1088/1748-605x/ab9510. [DOI] [PubMed] [Google Scholar]
- Chu D, Dong X, Zhao Q, Gu J, Wang Z. Photosensitization priming of tumor microenvironments improves delivery of nanotherapeutics via neutrophil infiltration. Adv Mater. 2017;29(27):1701021. doi: 10.1002/adma.201701021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cirmi S, Ferlazzo N, Lombardo GE, Ventura-Spagnolo E, Gangemi S, Calapai G, et al. Neurodegenerative diseases: might citrus flavonoids play a protective role? Molecules. 2016;21:312. doi: 10.3390/molecules21101312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cronkite DA, Strutt TM. The regulation of inflammation by innate and adaptive lymphocytes. J Immunol Res. 2018;2018:1467538. doi: 10.1155/2018/1467538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dacoba TG, Olivera A, Torres D, Crecente-Campo J, Alonso MJ. Modulating the immune system through nanotechnology. Semin Immunol. 2017;34:78–102. doi: 10.1016/j.smim.2017.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai Q, Walkey C, Chan WC. Polyethylene glycol backfilling mitigates the negative impact of the protein corona on nanoparticle cell targeting. Angew Chem Int Ed Engl. 2014;53:13493. doi: 10.1002/anie.201309464. [DOI] [PubMed] [Google Scholar]
- Davatgaran-Taghipour Y, Masoomzadeh S, Farzaei MH, Bahramsoltani R, Karimi-Soureh Z, Rahimi R, et al. Polyphenol nanoformulations for cancer therapy: experimental evidence and clinical perspective. Intern J Nanomed. 2017;12:2689. doi: 10.2147/IJN.S131973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Waard H, Frijlink HW, Hinrichs WLJ. Bottom-up preparation techniques for nanocrystals of lipophilic drugs. Pharm Res. 2011;28:1220–1223. doi: 10.1007/s11095-010-0323-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Marzio L, Ventura CA, Cosco D, Paolino D, Di Stefano A, Stancanelli R, Tommasini S, Cannava C, Celia C, Fresta M. Nanotherapeutics for anti-inflammatory delivery. J Drug Deliv Sci Technol. 2016;32(B):174–191. [Google Scholar]
- Dianzani C, Foglietta F, Ferrara B, Rosa AC, Muntoni E, Gasco P, Della PC, Canaparo R, Serpe L. Solid lipid nanoparticles delivering anti-inflammatory drugs to treat inflammatory bowel disease: effects in an in vivo model. World J Gastroenterol. 2017;23:4200–4210. doi: 10.3748/wjg.v23.i23.4200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diegelmann RF. Basic biology and clinical Aspects of inflammation; Book Series. Sharjah: Frontiers in Inflammation; Bentham Science Publishers; 2016. [Google Scholar]
- Divya G, Panonnummal R, Gupta S, Jayakumar R, Sabitha M. Acitretin and aloe-emodin loaded chitin nanogel for the treatment of psoriasis. Eur J Pharm Biopharm. 2016;107:97–109. doi: 10.1016/j.ejpb.2016.06.019. [DOI] [PubMed] [Google Scholar]
- Doge N, Honzke S, Schumacher F, Balzus B, Colombo M, Hadam S, Rancan F, Blume- Peytavi U, Schafer-Korting M, Schindler A, Ruhl E, Skov PS, Church MK, Hedtrich S, Kleuser B, Bodmeier R, Vogt A. Ethyl cellulose nanocarriers and nanocrystals differentially deliver dexamethasone into intact, tape-stripped or sodium lauryl sulfate-exposed ex vivo human skin—assessment by intradermal microdialysis and extraction from the different skin layers. J Control Release. 2016;242:25–34. doi: 10.1016/j.jconrel.2016.07.009. [DOI] [PubMed] [Google Scholar]
- Dormont MVF, Couvreur P. Nanoplumbers: biomaterials to fight cardiovascular diseases. Mater Today. 2018;21:122–143. [Google Scholar]
- Duong TT, Isomaki A, Paaver U, Laidmae I, Tonisoo A, Tran YTH, Kogermann K, Raal A, Heinamaki J, Pham T. Nanoformulation and evaluation of oral berberine-loaded liposomes. Molecules. 2021;26(9):2591. doi: 10.3390/molecules26092591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durymanov M, Kamaletdinova T, Lehmann SE, Reineke J. Exploiting passive nanomedicine accumulation at sites of enhanced vascular permeability for noncancerous applications. J Control Release. 2017;261:10–22. doi: 10.1016/j.jconrel.2017.06.013. [DOI] [PubMed] [Google Scholar]
- Enceladus Pharmaceuticals BV Announces Positive Results From a Phase 2a study in Patients With Active Ulcerative Colitis With Nanocort® | Enceladus Pharmaceuticals (2016) A evaluation of the safety of oncocort IV pegylated liposomal dexamethasone phosphate in patients with progressive multiple myeloma – Full Text View, ClinicalTrials.gov 2012
- Farhood B, Mortezaee K, Goradel NH, Khanlarkhani N, Salehi E, Nashtaei MS, Najafi M, Sahebkar A. Curcumin as an anti-inflammatory agent: Implications to radiotherapy and chemotherapy. J Cell Physiol. 2019;234:5728–5740. doi: 10.1002/jcp.27442. [DOI] [PubMed] [Google Scholar]
- Flavio Dormont RB, Cailleau C, Reynaud F, Peramo A, Gendron A, Mougin J, Gaudin F, Varna M, Couvreur P. Squalene-based multidrug nanoparticles for improved mitigation of uncontrolled inflammation. Sci Adv. 2020;6(23):eaaz5466. doi: 10.1126/sciadv.aaz5466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Francis AP, Devasena T, Ganapathy S, Palla VR, Murthy PB, Ramaprabhu S. Multi-walled carbon nanotube-induced inhalation toxicity: Recognizing nano bis-demethoxy curcumin analog as an ameliorating candidate. Nanomedicine. 2018;14(6):1809–1822. doi: 10.1016/j.nano.2018.05.003. [DOI] [PubMed] [Google Scholar]
- Fernandes AR, Ferreira NR, Fangueiro JF, Santos AC, Veiga FJ, Cabral C, Silva AM, Souto EB. Ibuprofen nanocrystals developed by 22 factorial design experiment: a new approach for poorly watersoluble drugs. Saudi Pharm J. 2017;25:1117–1124. doi: 10.1016/j.jsps.2017.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ganesan P, Kim B, Ramalaingam P, Karthivashan G, Revuri V, Park S, Joon KS, Yang KOT, Dong CK. Antineuroinflammatory activities and neurotoxicological assessment of curcumin loaded solid lipid nanoparticles on LPS-stimulated BV-2 microglia cell models. Molecules. 2019;24(6):1170. doi: 10.3390/molecules24061170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gendelman HE, Anantharam V, Bronich T, Ghaisas S, Jin H, Kanthasamy AG, Liu X, McMillan J, Mosley RL, Narasimhan B, Mallapragada SK. Nanoneuromedicines for degenerative, inflammatory, and infectious nervous system diseases. Nanomedicine. 2015;11(3):751–767. doi: 10.1016/j.nano.2014.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gharagozloo M, Majewski S, Foldvari M. Therapeutic applications of nanomedicine in auto immune diseases:from immunosuppression to tolerance induction. Nanomedicine. 2015;11:1003–1018. doi: 10.1016/j.nano.2014.12.003. [DOI] [PubMed] [Google Scholar]
- Ghasemian M, Owlia MB. A different look at pulsed glucocorticoid protocols; is high dose oral prednisolone really necessary just after initiation of pulse therapy? J Case Rep Pract. 2015;3(1):1–3. [Google Scholar]
- Goldsmith M, Abramovitz L, Peer D. Precision nanomedicine in neurodegenerative diseases. ACS Nano. 2014;8:1958–1965. doi: 10.1021/nn501292z. [DOI] [PubMed] [Google Scholar]
- Gulsun T, Budak C, Vural I, Sahin S, Oner L. Preparation and characterization of nimesulide containing nanocrystal formulations. Pharm Dev Technol. 2013;18:653–659. doi: 10.3109/10837450.2012.663390. [DOI] [PubMed] [Google Scholar]
- He J, Han Y, Xu G, Yin L, NgandeuNeubi M, Zhou J, Ding Y. Preparation and evaluation of celecoxib nanosuspensions for bioavailability enhancement. RSC Adv. 2017;7:13053–13064. [Google Scholar]
- Iqbal J, Abbasi BA, Ahmad R, Mahmood T, Ali B, Khalil AT, Kanwal S, Shah SA, Alam MM, Badshah H, Munir A. Nanomedicines for developing cancer nanotherapeutics: from benchtop to bedside and beyond. Appl Microbiol Biotechnol. 2018;102:9449–9470. doi: 10.1007/s00253-018-9352-3. [DOI] [PubMed] [Google Scholar]
- Jia X, Wei F. Advances in production and applications of carbon nanotubes. Top Curr Chem. 2017;375:18. doi: 10.1007/s41061-017-0102-2. [DOI] [PubMed] [Google Scholar]
- Jia Z, Zhao G, Wei X, Kong D, Sun Y, Zhou Y, Lele SM, Fehringer EV, Garvin KL, Goldring SR, Wang D. Structural optimization of HPMA copolymer-based dexamethasone prodrug for improved treatment of inflammatory arthritis. J Control Release. 2020;324:560–573. doi: 10.1016/j.jconrel.2020.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalangi SK, Swarnakar NK, Sathyavathi R, Rao DN, Jain S, Reddanna P. Synthesis, characterization, and biodistribution of quantum Dotcelecoxib conjugate in mouse paw edema model. Oxid Med Cell Longev. 2018;2018:3090517. doi: 10.1155/2018/3090517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karabey-Akyurek Y, Gurcay AG, Gurcan O, Turkoglu OF, Yabanoglu-Ciftci S, Eroglu H, Sargon MF, Bilensoy E, Oner L. Localized delivery of methylprednisolone sodium succinate with polymeric nanoparticles in experimental injured spinal cord model. Pharm Dev Technol. 2017;22:972–981. doi: 10.3109/10837450.2016.1143002. [DOI] [PubMed] [Google Scholar]
- Kumar S, Burgess DJ. Wet milling induced physical and chemical instabilities of naproxen nano-crystalline suspensions. Int J Pharm. 2014;466:223–232. doi: 10.1016/j.ijpharm.2014.03.021. [DOI] [PubMed] [Google Scholar]
- Kumar R, Dogra S, Amarji B, Singh B, Kumar S, Vinay SK, Mahajan R, Katare OP. Efficacy of novel topical liposomal formulation of cyclosporine in mild to moderate stable plaque psoriasis: a randomized clinical trial. JAMA Dermatol. 2016;152:807–815. doi: 10.1001/jamadermatol.2016.0859. [DOI] [PubMed] [Google Scholar]
- Kuroiwa Y, Higashi K, Ueda K, Yamamoto K, Moribe K. Nano-scale and molecular-level understanding of wet-milled indomethacin/poloxamer 407 nanosuspension with TEM, suspended-state NMR, and Raman measurements. Int J Pharm. 2018;537:30–39. doi: 10.1016/j.ijpharm.2017.12.028. [DOI] [PubMed] [Google Scholar]
- Landriscina A, Rosen J, Friedman A. Nanotechnology, inflammation and the skin barrier: Innovative approaches for skin health and cosmesis. Cosmetics [internet] 2015;2(2):177186. [Google Scholar]
- Lee SE, Park HR, Jeon S, Han D, Park YS. Curcumin attenuates acrolein-induced COX-2 expression and prostaglandin production in human umbilical vein endothelial cells. J Lipid Atheroscl. 2020;9:184–194. doi: 10.12997/jla.2020.9.1.184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ley K. Inflammation—fundamental mechanisms. San Diego: La Jolla Institute for Allergy and Immunology; 2018. [Google Scholar]
- Liu P, De Wulf O, Laru J, Heikkilä T, Van Veen B, Kiesvaara J, Hirvonen J, Peltonen L, Laaksonen T. Dissolution studies of poorly soluble drug nanosuspensions in nonsink conditions. AAPS PharmSciTech. 2013;14:748–756. doi: 10.1208/s12249-013-9960-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu P, Viitala T, Kartal-Hodzic A, Liang H, Laaksonen T, Hirvonen J, Peltonen L. Interaction studies between indomethacin nanocrystals and PEO/PPO copolymer stabilizers. Pharm Res. 2015;32:628–639. doi: 10.1007/s11095-014-1491-3. [DOI] [PubMed] [Google Scholar]
- Liu T, Yao G, Zhang X, Zuo X, Wang L, Yin H, Möschwitzer JP. Systematical investigation of different drug nanocrystal technologies to produce fast dissolving meloxicam tablets. AAPS Pharm Sci Tech. 2018;19:783–791. doi: 10.1208/s12249-017-0889-8. [DOI] [PubMed] [Google Scholar]
- Liu J, Tu L, Cheng M, Feng J, Jin Y. Mechanisms for oral absorption enhancement of drugs by nanocrystals. J Drug Deliv Sci Technol. 2020;56:101607. [Google Scholar]
- Maione F, Minosi P, Di Giannuario A, et al. Long-lasting anti-inflammatory and antinociceptive effects of acute ammonium glycyrrhizinate administration: pharmacological, biochemical, and docking studies. Molecules. 2019;24(13):2453. doi: 10.3390/molecules24132453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malone S. Inflamed: discover the root cause of inflammation and personalize a step-by-step plan to create a healthy, vibrant life. Locust Valley: Augustin Publishing; 2016. [Google Scholar]
- Mir M, Ahmed N, Rehman AU. Recent applications of PLGA based nanostructures in drug delivery. Colloids Surf B Biointerfaces. 2017;159:217–231. doi: 10.1016/j.colsurfb.2017.07.038. [DOI] [PubMed] [Google Scholar]
- Miyasaka M, Takatsu K. Chronic inflammation: mechanisms and regulation. Tokyo: Springer; 2016. [Google Scholar]
- Molinaro R, Boada C, Del Rosal GM, Hartman KA, Corbo C, Andrews ED, Toledano-Furman NE, Cooke JP, Tasciotti E. Vascular inflammation: a novel access route for nanomedicine. Methodist Debakey Cardiovasc. 2016;12:169–174. doi: 10.14797/mdcj-12-3-169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moschwitzer JP. Drug nanocrystals in the commercial pharmaceutical development process. Int J Pharm. 2013;453:142–156. doi: 10.1016/j.ijpharm.2012.09.034. [DOI] [PubMed] [Google Scholar]
- Narayan R, Pednekar A, Bhuyan D, Gowda C, Koteshwara KB, Nayak UY. A top-down technique to improve the solubility and bioavailability of aceclofenac: In vitro and in vivo studies. Int J Nanomed. 2017;12:4921–4935. doi: 10.2147/IJN.S141504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nava-Ochoa A, Antunes-RicardoGuajardo-Flores MD. Nano-sized carriers for capsaicinoids with topic analgesic and anti-inflammatory effects. J Biotechnol. 2021;333:77–85. doi: 10.1016/j.jbiotec.2021.04.009. [DOI] [PubMed] [Google Scholar]
- Negut I, Grumezescu V (2021) Hyaluronic acid nanoparticles. Biopolym Nanomater, pp 155–71. 10.1016/B978-0-12-824364-0.00015-0
- Ning W, Hai Y, Qian S, Ping M, Kuo L, Gang B. Sesamol-loaded stearic acid-chitosan nanomicelles mitigate the oxidative stress-stimulated apoptosis and induction of pro-inflammatory cytokines in motor neuronal of the spinal cord through NF-ĸB signaling pathway. Int J Biol Macromol. 2021;186:23–32. doi: 10.1016/j.ijbiomac.2021.06.171. [DOI] [PubMed] [Google Scholar]
- Nordqvist C (2017) Everything you need to know about inflammation. Med News Today [Internet]. Medilexicon
- Ochi M, Kawachi T, Toita E, Hashimoto I, Yuminoki K, Onoue S, Hashimoto N. Development of nanocrystal formulation of meloxicam with improved dissolution and pharmacokinetic behaviors. Int J Pharm. 2014;474:151–156. doi: 10.1016/j.ijpharm.2014.08.022. [DOI] [PubMed] [Google Scholar]
- Oh JY, Kim HS, Palanikumar L, Go EM, Jana B, Park SA, Kim HY, Kim K, Seo JK, Kwak SK, Kim C, Kang S, Ryu JH. Cloaking nanoparticles with protein corona shield for targeted drug delivery. Nat Commun. 2018;9(1):4548. doi: 10.1038/s41467-018-06979-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oktay AN, Karakucuk A, Ilbasmis-Tamer S, Celebi N. Dermal flurbiprofen nanosuspensions: optimization with design of experiment approach and in vitro evaluation. Eur J Pharm Sci. 2018;122:254–263. doi: 10.1016/j.ejps.2018.07.009. [DOI] [PubMed] [Google Scholar]
- Pahwa R, Jialal I. Chronic inflammation. Berlin: StatPearls Publishing; 2018. [PubMed] [Google Scholar]
- Pahwa R, Goyal A, Bansal P, Jialal I. Chronic Inflammation. StatPearls Publishing; Tampa; 2019. [PubMed] [Google Scholar]
- Peltonen L, Hirvonen J. Drug nanocrystals—versatile option for formulation of poorly soluble materials. Int J Pharm. 2018;537:73–83. doi: 10.1016/j.ijpharm.2017.12.005. [DOI] [PubMed] [Google Scholar]
- Pireddu R, Sinico C, Ennas G, Marongiu F, Muzzalupo R, Lai F, Fadda AM. Novel nanosized formulations of two diclofenac acid polymorphs to improve topical bioavailability. Eur J Pharm Sci. 2015;77:208–215. doi: 10.1016/j.ejps.2015.06.006. [DOI] [PubMed] [Google Scholar]
- Rathore C, Upadhyay KN, Sharma A, Uma LR, Raza K, Negi P. Phospholipid nanoformulation of thymoquinone with enhanced bioavailability: Development, characterization and anti-inflammatory activity. Journal of Drug Delivery Science and Technology. 2019;52(19):316–324. [Google Scholar]
- Reinisalo M, Kårlund A, Koskela A, Kaarniranta K, Karjalainen RO. Polyphenol stilbenes: molecular mechanisms of defence against oxidative stress and aging-related diseases. Oxid Med Cell Longev. 2015;2015:1–24. doi: 10.1155/2015/340520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Research and Markets (2020) Nanotechnology in drug delivery—global market trajectory & analytics. https://www.researchandmarkets.com/reports/2769238/nanotechnology_in_drug_delivery_global_market. Accessed Apr 2021
- Rheumatoid Arthritis—Mayo Clinic (2020) Available online: https://www.mayoclinic.org/diseases-conditions/rheumatoidarthritis/diagnosistreatment/drc-20353653. Accessed 17 Dec 2021
- Rupa EJ, Li JF, Arif MH, Yaxi H, Mitra AP, Jong AC, Hoang V, Kaliraj L, Chun DY, Chan SK. Cordyceps militaris fungus extracts-mediated nanoemulsion for improvement antioxidant, antimicrobial, and anti-inflammatory activities. Molecules. 2020;25(23):5733. doi: 10.3390/molecules25235733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sabouri M, Samadi A, Ahmad NS, Farboud ES, Mirrahimi B, Hassanzadeh H, Nassiri KM, Dinarvand R, Firooz A. Tretinoin loaded nanoemulsion for acne vulgaris: fabrication, physicochemical and clinical efficacy assessments. Skin Pharmacol Physiol. 2018;31:316–323. doi: 10.1159/000488993. [DOI] [PubMed] [Google Scholar]
- Salazar J, Ghanem A, Müller RH, Möschwitzer JP. Nanocrystals: comparison of the size reduction effectiveness of a novel combinative method with conventional top-down approaches. Eur J Pharm Biopharm. 2012;81:82–90. doi: 10.1016/j.ejpb.2011.12.015. [DOI] [PubMed] [Google Scholar]
- Salibay CC, Mahboob T, Verma AK, San Sebastian JS, Tabo HA, Raju CS, Nissapatorn V. Inflammation and Natural Products. Academic Press; 2021. Natural product–derived drugs for the treatment of inflammatory bowel diseases (IBD) pp. 235–259. [Google Scholar]
- Saraiva C, Praça C, Ferreira R, Santos T, Ferreira L, Bernardino L. Nanoparticle-mediated brain drug delivery: Overcoming bloodbrain barrier to treat neurodegenerative diseases. J Control Release. 2016;235:34–47. doi: 10.1016/j.jconrel.2016.05.044. [DOI] [PubMed] [Google Scholar]
- Schauss AG. Polyphenols and inflammation. In: Watson RR, Preedy VR, editors. Bioactive food as dietary interventions for arthritis and related inflammatory diseases. San Diego: Academic Press; 2013. pp. 379–392. [Google Scholar]
- Schmid D, Park CG, Hartl CA, Subedi N, Cartwright AN, Puerto RB, Zheng Y, Maiarana J, Freeman GJ, Wucherpfennig KW, Irvine DJ, Goldberg MS. T cell-targeting nanoparticles focus delivery of immunotherapy to improve antitumor immunity. Nat Commun. 2017;8(1):1747. doi: 10.1038/s41467-017-01830-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma R, Kambhampati P, Zhang Z, Sharma A, Chen S, Dua EI, Kannan S, Tso MOM, Kannan RM. Dendrimer mediated targeted delivery of sinomenine for the treatment of acute neuroinflammation in traumatic brain injury. J Control Release. 2020;323:361–375. doi: 10.1016/j.jconrel.2020.04.036. [DOI] [PubMed] [Google Scholar]
- Shi Y, Su J, Roberts AI, Shou P, Rabson AB, Ren G. How mesenchymal stem cells interact with tissue immune responses. Trends Immunol. 2012;33(3):136–143. doi: 10.1016/j.it.2011.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi M, Zhang P, Zhao Q, Shen K, Qiu Y, Xiao Y, et al. Dual functional monocytes modulate bactericidal and anti-inflammation process for severe osteomyelitis treatment. Small. 2020;16:e1905185. doi: 10.1002/smll.201905185. [DOI] [PubMed] [Google Scholar]
- Shukla R, Pandey V, Vadnere G, Lodhi S (2019) Role of Flavonoids in Management of Inflammatory Disorders. In: Bioactive food as dietary interventions for arthritis and related inflammatory diseases, 2nd edition. Academic Press, pp 293–322
- Silva S, Almeida AJ, Vale N. Combination of cell-penetrating peptides with nanoparticles for therapeutic application: a review. Biomolecules. 2019;9:22. doi: 10.3390/biom9010022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh NA, Mandal AKA, Khan ZA. Potential neuroprotective propertiesofepigallocatechin-3-gallate (EGCG) Nutr J. 2016;15:60. doi: 10.1186/s12937-016-0179-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinha B, Müller RH, Möschwitzer JP. Bottom-up approaches for preparing drug nanocrystals: Formulations and factors affecting particle size. Int J Pharm. 2013;453:126–141. doi: 10.1016/j.ijpharm.2013.01.019. [DOI] [PubMed] [Google Scholar]
- Sogut PO, Sezer UA, Sezer S. Liposomal delivery systems for herbal extracts. J Drug Deliv Sci Technol. 2020;61:102147. doi: 10.1016/j.jddst.2020.102147. [DOI] [Google Scholar]
- Strathearn KE, Yousef GG, Grace MH, Roy SL, Tambe MA, Ferruzzi MG, et al. Neuroprotective effects of anthocyanin- and proanthocyanidin-rich extracts in cellular models of Parkinsons disease. Brain Res. 2014;1555:60–77. doi: 10.1016/j.brainres.2014.01.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabas CK. Glass, Anti-inflammatory therapy in chronic disease: challenges and opportunities. Science. 2013;339:166–172. doi: 10.1126/science.1230720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka T, Narazaki M, Kishimoto T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect Biol. 2014;6(10):a016295. doi: 10.1101/cshperspect.a016295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tekade RK, et al. et al. Nanotechnology for the development of nanomedicine. In: Mishra V, et al.et al., editors. Nanotechnology-based approaches for targeting and delivery of drugs and genes. Academic Press; 2017. pp. 3–61. [Google Scholar]
- Tenzer S, Docter D, Kuharev J, Musyanovych A, Fetz V, Hecht R, Schlenk F, Fischer D, Kiouptsi K, Reinhardt C, Landfester K, Schild H, Maskos M, Knauer SK, Stauber RH. Rapid formation of plasma protein corona critically affects nanoparticle pathophysiology. Nat Nanotechnol. 2013;10:772–81. doi: 10.1038/nnano.2013.181. [DOI] [PubMed] [Google Scholar]
- Trevor AJ, Katzung BG, Kruidering Hall M. Katzung & Trevor’s pharmacology. 12. New York: McGraw-Hill; 2019. [Google Scholar]
- Trujillo Nolasco et al. (2019) Drug delivery methods based on organic nanoparticles are now commercially available in a variety of forms such as Lutetium-177-1,4,7,10 tetraazacyclododecane-1,4,7,10-tetraacetic acid-hyaluronic acid-poly (lactic-co-glycolic acid)
- Trujillo-Nolasco RM, Morales-Avila E, Ocampo-Garcia BE, Ferro-Flores G, Gibbens-Bandala BV, Escudero-Castellanos A, et al. Preparation and in vitro evaluation of radiolabeled HA-PLGA nanoparticles as novel MTX delivery system for local treatment of rheumatoid arthritis. Mater Sci Eng C Mater Biol Appl. 2019;103:109766. doi: 10.1016/j.msec.2019.109766. [DOI] [PubMed] [Google Scholar]
- Tuomela A, Saarinen J, Strachan CJ, Hirvonen J, Peltonen L. Production, applications and in vivo fate of drug nanocrystals. J Drug Deliv Sci Technol. 2016;34:21–31. [Google Scholar]
- Ullah N, Khan S, Ahmed S, Govender T, Faidah HS, De Matas M, Shahid M, Minhas MU, Sohail M, Khurram M. Dexibuprofen nanocrystals with improved therapeutic performance: fabrication, characterization, in silico modeling, and in vivo evaluation. Int J Nanomed. 2018;13:1677–1692. doi: 10.2147/IJN.S151597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valsalan Soba S, Babu M, Panonnummal R. Ethosomal gel formulation of alpha phellandrene for the transdermal delivery in gout. Adv Pharm Bull. 2021;11(1):137–149. doi: 10.34172/apb.2021.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Alem CMA, Boonstra M, Prins J, Bezhaeva T, van Essen MF, Ruben JM, Vahrmeijer AL, van der Veer EP, de Fijter JW, Reinders ME, Meijer O, Metselaar JM, van Kooten C, Rotmans JI. Local delivery of liposomal prednisolone leads to an anti-inflammatory profile in renal ischaemia-reperfusion injury in the rat. Nephrol Dial Transplant. 2018;33:44–53. doi: 10.1093/ndt/gfx204. [DOI] [PubMed] [Google Scholar]
- Veiseh O, Tang BC, Whitehead KA, Anderson DG, Langer R. Managing diabetes with nanomedicine: challenges and opportunities. Nat Rev Drug Discov. 2015;14:45–57. doi: 10.1038/nrd4477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wailoo A, Alava MH, Scott IC, Ibrahim F, Scott DL. Cost-effectiveness of treatment strategies using combination disease-modifying anti-rheumatic drugs and glucocorticoids in early rheumatoid arthritis. Rheumatology. 2014;53(10):1773–1777. doi: 10.1093/rheumatology/keu039. [DOI] [PubMed] [Google Scholar]
- Walkey CD, Chan WC. Understanding and controlling the interaction of nanomaterials with proteins in aphysiological environment. Chem Soc Rev. 2012;41(7):2780–99. doi: 10.1039/c1cs15233e. [DOI] [PubMed] [Google Scholar]
- Wang L, Wang FS, Gershwin ME. Human autoimmune diseases: a comprehensive update. J Intern Med. 2015;278(4):369–395. doi: 10.1111/joim.12395. [DOI] [PubMed] [Google Scholar]
- Wang ZF, Liu J, Yang YA, Zhu HL. A review: the anti-inflammatory, anticancer and antibacterial properties of four kinds of licorice flavonoids isolated from licorice. Curr Med Chem. 2020;27(12):1997–2011. doi: 10.2174/0929867325666181001104550. [DOI] [PubMed] [Google Scholar]
- Whalen K, Radhakrishnan R, Field C. Lippincott illustrated reviews: pharmacology. 7. Philadelphia: Wolters Kluwer; 2019. [Google Scholar]
- Wood FL, Houston B, Hallifaz D. Importance of the unstirred water layer and hepatocyte membrane integrity in vitro for quantification of intrinsic metabolic clearance. Drug Metab Dispos. 2018;46(3):268–278. doi: 10.1124/dmd.117.078949. [DOI] [PubMed] [Google Scholar]
- Wu G, Zhang J, Zhao Q, Zhuang W, Ding J, Zhang C, et al. Molecularly engineered macrophage-derived exosomes with inflammation tropism and intrinsic heme biosynthesis for atherosclerosis treatment. Angew Chem Int Ed Engl. 2020;59:4068–4074. doi: 10.1002/anie.201913700. [DOI] [PubMed] [Google Scholar]
- Wynn TA, Chawla A, Pollard JW. Macrophage biology in development, homeostasis and disease. Nature. 2013;496:455e455. doi: 10.1038/nature12034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao TS. Innate immunity and inflammation. Cell Mol Immunol. 2017;14:1–3. doi: 10.1038/cmi.2016.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yadav KS, Sawant KK. Formulation optimization of etoposide loaded PLGA nanoparticles by double factorial design and their evaluation. Curr Drug Deliv. 2010;7:51–64. doi: 10.2174/156720110790396517. [DOI] [PubMed] [Google Scholar]
- Yadav S, Pawar G, Kulkarni P, Ferris C, Amiji M. CNS delivery and anti-inflammatory effects of intranasally administered cyclosporine-A in cationic nanoformulations. J Pharmacol Exp Ther. 2019;370:843–854. doi: 10.1124/jpet.118.254672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan F, Zhong Z, Wang Y, Feng Y, Mei Z, Li H, et al. Exosome-based biomimetic nanoparticles targeted to inflamed joints for enhanced treatment of rheumatoid arthritis. J Nanobiotechnol. 2020;18:115. doi: 10.1186/s12951-020-00675-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang R, Yuan BC, Ma YS, Zhou S, Liu Y. The anti-inflammatory activity of licorice, a widely used Chinese herb. Pharm Biol. 2017;55(1):5–18. doi: 10.1080/13880209.2016.1225775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu Q, Wu X, Zhu Q, Wu W, Chen Z, Li Y, Lu Y. Enhanced transdermal delivery of meloxicam by nanocrystals: preparation, in vitro and in vivo evaluation. Asian J Pharm Sci. 2018;13:518–526. doi: 10.1016/j.ajps.2017.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zarrin AA, Bao K, Lupardus P, Vucic D. Kinase inhibition in autoimmunity and inflammation. Nat Rev Drug Discov. 2021;20:39–63. doi: 10.1038/s41573-020-0082-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng J, Wu J, Ding X, Hung SC, Zou G. Small molecule approaches to treat autoimmune and inflammatory diseases (Part I): Kinase inhibitors. Bioorg Med Chem Lett. 2021;38:1–14. doi: 10.1016/j.bmcl.2021.127862. [DOI] [PubMed] [Google Scholar]
- Zhou M, Hou J, Zhong Z, Hao N, Lin Y, Li C. Targeted delivery of hyaluronic acid-coated solid lipid nanoparticles for rheumatoid arthritis therapy. Drug Deliv. 2018;25:716–722. doi: 10.1080/10717544.2018.1447050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou PH, Qiu B, Deng RH, Li HJ, Xu XF, Shang XF. Chondroprotective effects of hyaluronic acid-chitosan nanoparticles containing plasmid DNA encoding cytokine response modifier A in a rat knee osteoarthritis model. Cell Physiol Biochem. 2018;47:1207–1216. doi: 10.1159/000490217. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Enquiries about data availability should be directed to the authors.