Abstract
The global importance of the hemoparasite Theileria haneyi to equine health was recently shown by its resistance to imidocarb dipropionate (ID) and its interference with T. equi clearance by ID in some co-infected horses. Genetic characterization of T. haneyi revealed marked genomic reduction compared to T. equi, and initial experiments demonstrated reduced clinical severity in spleen-intact horses. Furthermore, in early experiments, splenectomized horses survived T. haneyi infection and progressed to an asymptomatic carrier state, in stark contrast to the high fatality rate of T. equi in splenectomized horses. Thus, we hypothesized that T. haneyi is less virulent than T. equi. To objectively assess virulence, clinical data from nine splenectomized, T. haneyi-infected horses were evaluated and compared to published data on T. equi-infected, splenectomized horses. Seven of eight splenectomized, T. haneyi-infected horses survived. Further, in six horses co-infected with T. equi and T. haneyi, only horses cleared of T. equi by ID survived splenectomy and became asymptomatic carriers. The reduced virulence of T. haneyi in splenectomized horses instructs why T. haneyi was, until recently, undetected. This naturally occurring comparative reduction in virulence in a natural host provides a foundation for defining virulence mechanisms of theileriosis and Apicomplexa in general.
Keywords: Theileria haneyi, Theileria equi, virulence, splenectomy
1. Introduction
Theileria equi and Theileria haneyi are closely related Apicomplexan hemoparasites and known causative agents of equine theileriosis. T. equi has been detected in tropical and subtropical regions around the world, and T. haneyi has been documented on multiple continents, with its distribution anticipated to expand as surveillance for the organism increases [1,2,3,4,5]. T. equi induces disease characterized predominantly by fever, hemolytic anemia, and signs associated with erythrocyte lysis [2].
Recent clinical studies with T. haneyi have characterized clinical disease in spleen-intact horses. These studies suggest that T. haneyi causes minimal morbidity in this population, similar to what is frequently reported for T. equi, in which horses show no overt clinical signs without careful observation. Unfortunately, clinical signs induced experimentally are not always consistent with those observed outside of a controlled environment, especially when animals are used for work and/or athletic purposes [2,6,7]. Indeed, there is significant global variation in disease severity, and the parasite- and host-specific factors associated with variations in virulence are not well understood. The lack of understanding of Theileria pathogenesis, in particular factors leading to significant morbidity and mortality, is a primary reason many countries restrict entry of horses infected with T. equi. Both species are capable of long-term, persistent infection in spleen-intact horses and consequently provide a continual, silent reservoir for transmission [1,2,8].
Beyond infection dynamic studies, comparative genomic analysis of T. equi and T. haneyi has revealed that T. haneyi has undergone genomic reduction. Specifically, the genome of T. haneyi is approximately 2 Mbp smaller than that of T. equi [9]. The consequences of genomic reduction are not yet known, with the exception of the loss of susceptibility to imidocarb diproprionate (ID) [10]. In other organisms, there are mixed data as to whether genomic reduction is associated with an increase or decrease in virulence [11,12].
Virulence is defined as “the relative capacity of a microbe to cause damage”, and in order to characterize virulence for a pathogen, it must be compared to a standard [13]. Previous authors have used rate of mortality, a composite of quantifiable parameters related to morbidity (anemia, fever, and weight loss) or parasite-specific parameters (peak parasitemia, duration of parasitemia, and time to peak) as parameters for comparison [13,14,15,16]. Comparisons are often made between a wild-type strain and a genetically modified strain; however, in this case, genetic modification was provided through evolution of T. haneyi, and T. equi was used as the standard [13]. Even though the two species are phylogenetically distinct, they do share similar pathogeneses, further enabling comparison [1].
Splenectomy is often used within equine babesiosis and theileriosis research to assess the efficacy of chemotherapeutics in eliminating persistent infection [1,10,17,18,19]. This technique is utilized because splenectomy decreases the host’s capacity to control parasitemia, thereby enabling large-scale expansion of previously undetectable parasite populations to detectable, and sometimes clinically significant, levels [20,21]. Within splenectomized horses, both T. equi and T. haneyi induce overt clinical disease, but, unlike T. haneyi, in the absence of intervention, T. equi infection almost always results in fatal hemolytic anemia with perimortem hemoglobinuria and icterus [1,10,22,23,24]. The mortality rate and clinical course of T. haneyi infection of splenectomized horses were not well characterized prior to this study.
For this study, we define virulence as a composite of mortality rate, peak parasitemia, and percent packed cell volume decline in splenectomized horses. The objective of this study was to utilize clinical data from nine splenectomized, T. haneyi-infected horses to determine if T. haneyi is less virulent than T. equi. These data were compared to previous published studies concerning the outcome of splenectomy on T. equi-infected horses [1,10,22,23,24].
2. Results
2.1. Characterization of Experimental T. haneyi Infection of Splenectomized Horses
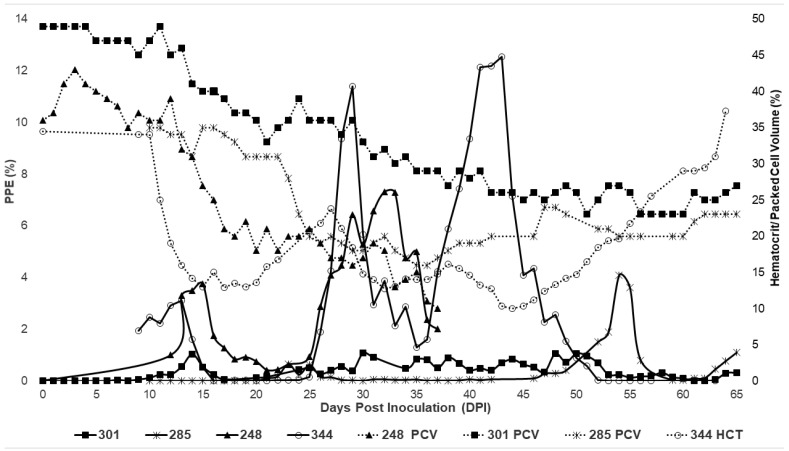
Following inoculation, all horses in Group 1 (Table 1; naïve splenectomized) developed a fever during acute infection, which included the first 60 days post-inoculation. Parasites were first observed via light microscopy between days 7 and 21. Peak fever ranged from 102.4 to 105.8 °F and correlated with rising parasitemia or peak parasitemia. Parasitemia appeared in waves over the course of infection. Horses 248 and 344 had more significant increases in parasitemia than that observed in 285 and 301 (Figure 1).
Table 1.
Inoculation methodology and timing of splenectomy for study horses. PPE = percent parasitized erythrocytes.
| Horse | Group | Inoculation Type | Stabilate Volume and PPE | Splenectomy Pre- or Post-Inoculation | Associated Study |
|---|---|---|---|---|---|
| 248 | 1 | Blood—120 mL | 2 mL/12% (donor) | Pre | [1] |
| 301 | 1 | Blood—120 mL | 2 mL/12% (donor) | Pre | [1] |
| 285 | 1 | Stabilate | 5 mL/8.8% | Pre | This study |
| 344 | 1 | Blood—150 mL | 4 mL/8.8% (donor) | Pre | This study |
| 275 | 2 | Stabilate | 2 mL/12% | Post | [8,10] |
| 277 | 2 | Stabilate | 2 mL/12% | Post | [8,10] |
| 278 | 2 | Stabilate | 2 mL/12% | Post | [8,10] |
| 280 | 2 | Stabilate | 2 mL/12% | Post | [8,10] |
Figure 1.
Changes in parasitemia (solid lines) and hematocrit (dotted lines) during acute T. haneyi infection of splenectomized (Group 1) horses.
For horses inoculated via whole transfusion, the first peak in parasitemia occurred between 12 and 15 days post-inoculation, with the single horse inoculated with stabilates peaking the first time at 23 dpi (Figure 1). The second peak occurred for transfused horses at 29–32 dpi, followed by a third wave in the surviving horses at 43–50 dpi (Table 2). Horse 285 did develop additional parasitemia spikes over the course of infection at 54 and 69 dpi (Table 2, Figure 1). Overall, horses developed significant changes in the packed cell volume/hematocrit (PCV/HCT), with the decline ranging from 33.33 to 72.22% (Table 2). HCT/PCV nadirs were greatest for horses 248 and 344, both of whom were infected via whole-blood transfusion from an acutely infected spleen-intact horse. Within Group 1, only a single horse, 248, was euthanized.
Table 2.
Changes in parasitemia and packed cell volume during acute infection of horses in Group 1. PPE = percent parasitized erythrocytes; PCV = packed cell volume.
| Horse | 248 | 285 | 301 | 344 |
|---|---|---|---|---|
| PPE peaks | 15 dpi—3.75% 32 dpi—7.32% |
23 dpi—0.65% 54 dpi—4.1% 69 dpi—2.34% |
14 dpi—1.03% 30 dpi—1.08% 50 dpi—1.06% |
13 dpi—3.10% 29 dpi—11.40% 43 dpi—12.55% |
| PCV nadirs | 20 dpi—18% 37 dpi—10% |
29 dpi—18% 54 dpi—20% 72 dpi—15% |
21 dpi—33% 38 dpi—27% 51 dpi—23% |
15—13% 32—12.7% 44—10% |
| Maximum% change in PCV # | −72.22% | −57.14% # | −53.06% | −71.01% |
| Anemia duration | 15–37 dpi | 23–87 dpi | 35–67 dpi | 11–62 |
| Survival | Euthanized 37 dpi | survived | survived | Survived |
# PCV change is based upon PCV recorded on day 10, which was the first recorded value for this horse.
2.2. Characterization of Experimental T. haneyi Infection of Spleen-Intact Horses
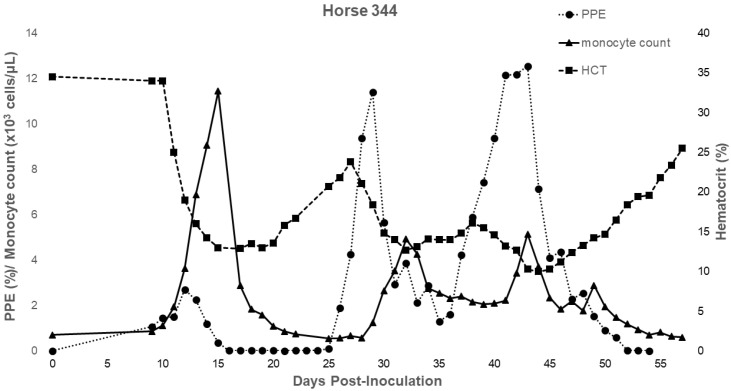
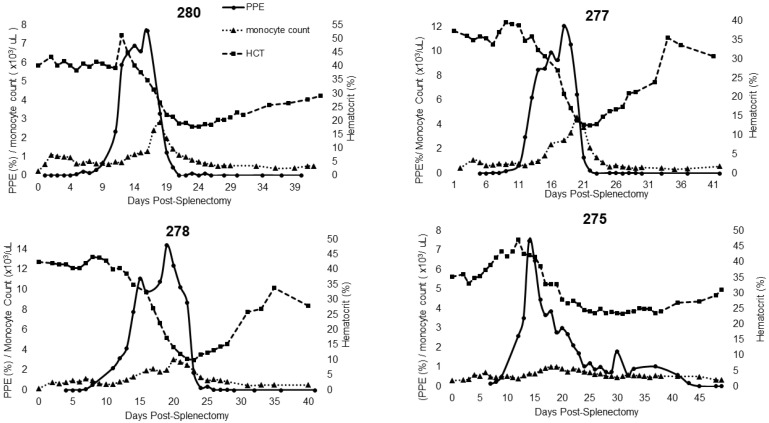
Horses in Group 2 (splenectomized after the development of persistent infection) developed mild, transient clinical signs during acute infection with T. haneyi, as described in [10], and subsequently entered a state of persistent, asymptomatic infection. Following splenectomy, all developed a fever with peak rectal temperature ranging from 101.2 to 103.8 °F. Each horse in this group exhibited a single peak in parasitemia that was equal to or greater than that observed in the horses in Group 1 (Figure 2). All four horses experienced peak percent parasitized erythrocytes (PPE) between days 14 and 19 post-splenectomy, with HCT nadirs at 6–18 days after the peak (Table 3). The percent decline in HCT was similar to that observed in Group 1 and ranged from 33.52 to 76.53%.
Figure 2.
Changes in parasitemia, monocyte count, and hematocrit during acute T. haneyi infection of a splenectomized horse (344) in Group 1.
Table 3.
Hemogram changes in 344 (Group 1) and Group 2 horses post-splenectomy.
| Hemogram Changes | 344 (dpi) | 275 (dps) | 277 (dps) | 278 (dps) | 280 (dps) |
|---|---|---|---|---|---|
| Monocytosis | 11–22, 30–57 | 15–28 | 14–24 | 13–28 | 15–26 |
| Lymphocytosis | 11–67 | None | 15–27, 29 | 19, 21–31 | 20–24,27 |
| Lymphopenia | None | 1–3 | None | None | None |
| Neutropenia | 30–39, 42–51 | 1–3 | None | None | 2 |
| Neutrophilia | 28 | 7, 9–10 | 3, 6–10, 19–25 | 6–29 | 9–11, 13–14, 20–21, 23–30 |
| Anemia duration | 11–62 | 20–42 | 17–32 | 18–34 | 18–43 |
| Anemia nadir | 15, 32, 44 | 31 | 22 | 24 | 24 |
| Maximum% change in HCT | −71.01% | −33.52% | −66.31% | −76.53% | −55.72% |
| Parasitemia peak(s) | 13, 29, 43 | 14 | 17 | 19 | 17 |
2.3. Characterization of Complete Blood Count Parameters in Experimentally Infected Horses
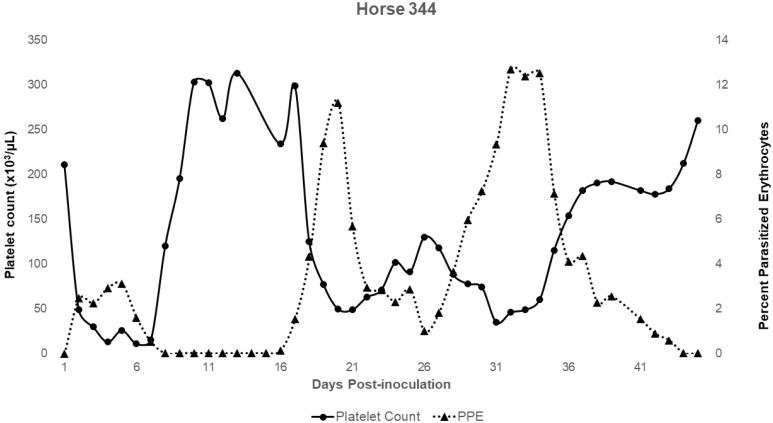
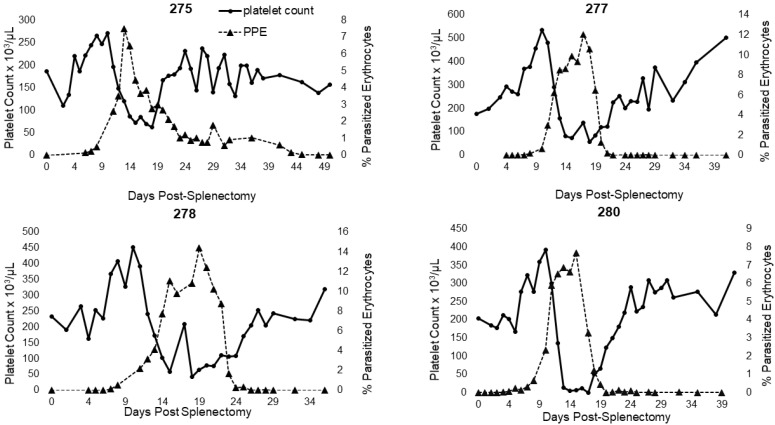
For horse 344 (splenectomized prior to infection; Group 1) and for all horses in Group 2, additional monitoring of CBCs enabled greater assessment of leukocyte and platelet dynamics over the course of acute infection (Table 3). Horse 344 exhibited significant changes in leukocyte and platelet counts during the acute period. The horse developed a lymphocytosis and monocytosis, along with periods of neutropenia and thrombocytopenia (Table 3, Figure 2). For Group 2 horses, only two experienced a brief period of neutropenia within the first 3 days post-splenectomy. All horses in this group developed neutrophilia, monocytosis, and thrombocytopenia (Table 3 and Figure 3). Three out of four horses also developed a lymphocytosis. The monocytosis observed in Group 2 horses was typically present when parasites were observed by light microscopy, with the elevation declining as parasites declined (Figure 3). This trend occurred in 3 out of 4 horses and for 344 (Figure 2 and Figure 3). Horse 275 exhibited only a brief period of monocytosis (Figure 3). All horses, however, did develop monocyte peaks after peak parasitemia was reached and peaks occurred either on the same day or prior to anemia nadirs (Table 3 and Figure 3). Monocyte peaks ranged from 1.03 to 11.44 × 103/µL of whole blood with the normal range being 0.2–0.6 × 103/µL of whole blood (Figure 2 and Figure 3). Each horse also experienced periods of thrombocytopenia around the time(s) of peak parasitemia (Figure 4 and Figure 5). Prolonged clotting times were not appreciated. All Group 2 horses experienced intermittent periods of neutrophilia, whereas 344, the single horse splenectomized prior to infection, (344) experienced periods of neutropenia (Table 3).
Figure 3.
Changes in parasitemia, monocyte count, and hematocrit during acute recrudescence of T. haneyi post-splenetomy in persistently infected (Group 2) horses.
Figure 4.
Changes in platelet count during acute T. haneyi infection of a splenectomized horse (344) in Group 1.
Figure 5.
Changes in platelet count during acute recrudescence of T. haneyi post-splenectomy in persistently infected (Group 2) horses.
2.4. Monitoring of T. haneyi Parasitemia via nPCR and Blood Smear Cytology
Following acute infection, all horses except horse 248 transitioned to state of chronic, asymptomatic infection. Horses were serially monitored by both light microscopy and T. haneyi nPCR to assess whether parasitemia remained detectable. For horses in Group 1, parasites were occasionally detected by light microscopy and horses occasionally became mildly anemic. Two of the three horses that survived acute infection remained nPCR positive. Each of these horses was euthanized due to the development of either chronic renal failure or acute guttural pouch empyema with concurrent severe laminitis. Due to the low number of animals in this study, it is unclear whether the intercurrent diseases that manifested during the persistent phase of infection are related to debilitation caused by infection with T. haneyi. The final surviving horse, 344, remained nPCR positive for approximately 11 months following initial infection. Horse 344 subsequently maintained a parasitemia level below the limits of detection by T. haneyi nPCR for subsequent 40 months. All horses in Group 2 were positive for T. haneyi for a minimum of 106 days post-splenectomy via nPCR, and two of the four horses maintained a parasitemia level below detectable limits of nPCR for the subsequent 10 months (Table 4). Horse 278 was nPCR negative for 6 months prior to testing weak positive and then negative at all subsequent timepoints.
Table 4.
Long-term outcome and associated nPCR results for all horses.
| Horse | Survival Acute Infection | Long-Term Survival | nPCR 6–12 Months Post-Infection/Splenectomy |
|---|---|---|---|
| 248 | N | N | Positive—37 dpi |
| 301 | Y | Euthanized; chronic renal failure | Positive—1402 dpi |
| 285 | Y | Euthanized; guttural pouch empyema and acute, severe laminitis | Positive—556 dpi |
| 344 | Y | Y | No longer detected; last positive 267 days post-inoculation; negative at 350 dpi and last negative at 1565 dpi |
| 275 | Y | Y | Positive; 536 days post-splenectomy |
| 277 | Y | Y | No longer detected after 106 dps (1/17/20), then neg on 3/3, 5/2, and 7/24, faint pos on 9/23, then neg 11/17, and 1/5/21 |
| 278 | Y | Y | No longer detected after 152 dps |
| 280 | Y | Y | No longer detected after 171 dps |
3. Discussion
In this study, clinical data from naïve, splenectomized horses infected with T. haneyi, and from horses persistently infected with T. haneyi that subsequently underwent splenectomy, were retrospectively assessed to generate a clear picture of the pathogenesis of T. haneyi and of its virulence in splenectomized horses compared to T. equi. Previous studies have confirmed the necessary role of the spleen in controlling T. equi parasitemia, and splenic removal invariably leads to uncontrolled parasite replication and usually mortality [2]. The spleen alone is not sufficient to reduce morbidity as studies in foals with severe combined immunodeficiency (SCID) revealed adaptive immunity is also crucial for parasite control [25]. While the exact immunologic mechanism(s) of hemoparasite control in equine theileriosis have not been elucidated, studies in other hemoparasitic diseases have revealed that removal of parasitized erythrocytes by the reticuloendothelial system of the spleen is integral to parasite control. In this, cells of histiocytic lineage remove parasitized erythrocytes, which are either opsonized by antibody or demonstrate reduced deformability, from circulation and destroy them, thereby reducing the circulating parasite burden [26].
Significantly, in this study, the overall survival rate of T. haneyi-infected, splenectomized horses was 87.5%. This is in stark contrast to the high fatality of T. equi in splenectomized horses, which ranges from 75 to 100% [17,18,19,22,23,24,27]. Peak parasitemia for all splenectomized, T. haneyi-infected horses in this study was less than 15% and decline in PCV/HCT ranged from 33.52% to 76.53%. T. equi-infected, splenectomized foals in one study developed peak parasitemias of >37%, and a percent decline in PCV of 73% or greater [23]. In one study using splenectomized donkeys, PPE reached 80%, and all eight donkeys died within 7–9 days [22]. Multiple studies assessing the efficacy of chemotherapeutic drugs also evaluated splenectomized horses. In these studies, untreated splenectomized control horses infected with T. equi developed parasitemias as high as 48%, with PCV declines of >70% of pre-inoculation levels [18,19]. Overall, infection with T. equi leads to a consistently greater peak parasitemia and a greater decline in PCV, resulting in hemoglobinuria, icterus, weakness, and exercise intolerance. None of the T. haneyi-infected, splenectomized horses in this study developed hemoglobinuria or icterus, and the horses experienced only brief, intermittent periods of weakness and exercise intolerance. Thus, objective, clinical comparison of T. haneyi and T. equi infection of splenectomized horses clearly demonstrates that T. haneyi is less virulent.
Interestingly, whereas naïve, splenectomized horses infected with T. haneyi developed multiple parasitemia peaks and associated PCV/HCT nadirs, splenectomy of horses after they had reached the persistent, asymptomatic phase of infection led to recrudescence characterized by only a single parasitemia peak and associated, transient anemia. Chronic infection of horse prior to splenectomy would have enabled the development of robust, adaptive immunity, including responses to antigenic escape variants, should they exist, which may have contributed to the single peak observed. The difference in parasite dynamics is in agreement with previous studies in T. equi that suggest that adaptive immunity is a significant contributing component of immunologic control [25]. Antibody responses to T. equi have been shown to correlate with the transition to persistent infection [28].
This study also revealed that a proportion of splenectomized, T. haneyi-infected horses in both Groups 1 and 2 eventually reached a state of undetectable parasitemia consistent with apparent clearance. This decline is in stark contrast as to what has been documented with T. equi recrudescence following splenectomy of persistently infected horses, wherein splenectomy following infection leads to recrudescence and often death [10,18,19]. Little is yet known regarding how equids transition from acute, symptomatic theileriosis to asymptomatic theileriosis. IgG responses in adult horses appear to correlate with control [28]. In malaria, chemotherapeutic treatment can lead to a period of dormancy [29]; however, the reported period of dormancy is short compared to the duration of undetectable parasitemia observed in these horses following splenectomy (up to 40 months), and undetectable parasitemia was observed in horses that never underwent imidocarb dipropionate treatment. Repeated exposure and resultant boosting of the adaptive immune response to some hemoparasitic diseases likely also enable maintenance of an asymptomatic state [30]. More recently, the concept of disease tolerance has been recognized, wherein a single exposure to an organism leads to host adaptations that minimize the inflammatory response yielding clinical disease [31]. This sort of adaptation could play a role in the observed, reduced disease severity in T. haneyi. Furthermore, it is possible that removal of the spleen sufficiently increased exposure of the T. haneyi parasites to the adaptive immune system, enabling enhanced, targeted control after a period of long-term exposure. To this end, the reduced virulence of T. haneyi is critical in that the infected, splenectomized host equids survived long enough for this degree of immune control to develop. Given that a greater proportion of horses in Group 2 cleared infection than those in Group 1, one additional possibility is that previous exposure to T. equi led to a cross-protective immune response that facilitated clearance of T. haneyi. Additional studies are required to elucidate the definitive mechanism of T. haneyi clearance in splenectomized horses.
Close evaluation of the hematologic changes observed in T. haneyi-infected, splenectomized horses also provided a window into the cellular (leukocyte and platelet) dynamics at play during T. haneyi infection. With each of the peaks of parasitemia for both groups of horses, hematocrit or packed cell volume and platelet counts subsequently declined substantially. For both groups of horses, HCT/PCV declined to nadirs within 0–18 days of the parasitemia peak, with time to decline related to the height of the parasitemia peak, i.e., the larger the peak, the less time to the HCT/PCV nadir. Furthermore, for horses in Group 2, nadirs occurred within 8 days for 3 out of 4 horses. The outlier, horse 275, was mildly dehydrated despite efforts to encourage drinking. Thus, his total protein and HCT were artificially elevated. The decline in HCT/PCV following peak parasitemia, with more rapid decline occurring after higher parasitemia peaks, is similar to that observed in horses infected with T. equi [18,19,23], and is due to both erythrocyte lysis following parasite replication and clearance of infected erythrocytes from circulation [2].
The development of thrombocytopenia overlapped with both significant declines in HCT and periods of peak parasitemia, and is also observed in T. equi-infected horses. The mechanism of thrombocytopenia occurring in Theileria sp. is not yet known; however, based on work in other systems, including Plasmodium sp., several potential mechanisms have been proposed, including immune-mediated platelet destruction, excess consumption following vasculitis and/or endothelial damage, and decreased production [2,32].
In recent years, malaria researchers have ascertained an additional mechanism for thrombocytopenia that is relevant in cases in which the platelet count is low but the patient lacks evidence of coagulopathy (as in T. haneyi and T. equi). Kho et al. found that platelets form complexes with Plasmodium sp.-infected red blood cells (iRBCs) and are subsequently not counted by machines. This study also demonstrated that platelets directly contribute to the elimination/killing of the erythrocytic stage of Plasmodium spp. [33]. This mechanism could explain the apparent normal clotting times for all Group 2 horses and for horse 344 despite profound thrombocytopenia. Furthermore, periods of thrombocytopenia appear to precede nadirs in PCV/HCT of all horses, and correlate with peaks of parasitemia, suggesting possible involvement of platelets with parasite control.
Neutrophilia and lymphocytosis were also observed in the single Group 1 horse for which data were available (344) and for all Group 2 horses. A neutrophilia has also been observed in both T. equi infection of splenectomized and spleen-intact horses [24,34,35]. However, those research groups noted a lymphopenia in conjunction with the neutrophilia in splenectomized horses [24,34]. A study of acute T. equi infection in field cases in Chile did note occasional lymphopenia or lymphocytosis in the cohort of cases evaluated [35]. Neutrophilia and lymphopenia have also been frequently noted in human patients with malaria, and the lymphopenia frequently transitions to a lymphocytosis over time [36,37]. As neutrophils are among the first responders of the innate immune response, elevations in neutrophil count were expected with changes in parasitemia [37]. Lymphopenia may occur in spleen-intact horses due to stress-induced apoptosis and reallocation to sites of inflammation with rebound lymphocytosis due to redistribution of lymphocytes [36]. In our splenectomized cohort, an additional contributing factor to the lymphocytosis may be splenectomy, as multiple studies of splenectomized humans have noted lymphocytosis post-splenectomy, with redistribution to the peripheral circulation being a proposed mechanism [38,39].
The marked monocytosis in T. haneyi-infected, splenectomized horses has not been reported outside of the publication describing the original experiment for which horses in Group 2 were splenectomized [10]. The significant changes in monocyte count immediately follows peak parasitemia, and may be a compensatory response for the removal of iRBCs [40,41]. This presumption stems from what has been elucidated from infection of spleen-intact humans with malaria. Specifically, red pulp macrophages continually remove senescent and less malleable erythrocytes (including P. falciparum-infected erythrocytes) that are incapable of passing through the inter-endothelial slits of the red pulp [42]. Theileria iRBCs are suspected to be more rigid due to biochemical changes induced induced by intraerythrocytic infection, and this increased rigidity has been documented in malaria [22]. These changes in RBC deformability subsequently lead to increased clearance of these damaged RBCs in the spleen [42,43]. However, in splenectomized horses, this major contributor to erythrocyte clearance and resultant decreased severity of disease, is absent. Therefore, with the loss of red pulp macrophages (RPM), other phagocytic cells, such as monocytes and neutrophils, may contribute more to phagocytosis and removal of infected cells from circulation, especially during acute infection [44,45]. Kim specifically showed an increased frequency of monocytes in RPM-deficient, Plasmodium-infected mice that appear to serve a similar role to RPMs in their absence [44]. Sponaas, additionally, found that increased monocyte populations contribute to control of blood-stage malaria [40]. Outside of the experimental setting, monocytosis has also been observed in both symptomatic and asymptomatic acute cases of human malaria [44,46,47,48,49].
An alternative perspective is that monocytes may also contribute to pathology. Other research groups have shown that monocytes contribute to severe anemia and dysregulated cytokine secretion in malaria [50,51]. Our group found that during acute, lethal infection of cattle with the related parasite, Theileria parva, animals that recover from infection exhibit a distinctly different monocyte phenotype than animals that succumb to infection, suggesting that expansion of intermediate and non-classical monocyte phenotypes is a correlate of severe disease in Theileria [52]. Ultimately, additional studies are warranted to assess changes in monocyte subsets and function during acute T. equi and T. haneyi infection to elucidate whether they potentiate protection or pathology.
4. Materials and Methods
4.1. Equine Infection Studies
Retrospective analyses were performed using data from horses previously utilized in equine theileriosis studies between October 2011 and October 2019 [1,8,10]. All experiments were approved by the University of Idaho and/or Washington State University IACUCs (ASAFs 2013-66, 2016-18, 2016-28, and 6241). Records from naïve, splenectomized horses infected with T. haneyi between 2011 and 2016, and from persistently infected horses that were subsequently splenectomized for verification of chemotherapeutic clearance (July–October 2019), were utilized (Table 1) [1,8,10,17,18,19]. Horses were divided into two groups based on the timing of splenectomy. Horses in Group 1 were splenectomized prior to T. haneyi infection and then inoculated 27–392 days later either through whole-blood transfusion or via intravenous inoculation with infected blood erythrocyte stabilate. The inoculum for all horses in this study was developed from the original splenectomized horse used to amplify T. haneyi parasitemia for genome sequencing (horse 208) [1]. Following inoculation, horses were clinically monitored as previously described [9,10]. Two additional horses, 285 and 344, were also included in this evaluation. The horses were inoculated via whole-blood transfusion from a previously infected, spleen-intact horse (344) or via stabilate inoculation (285) (Table 1). Both horses were monitored daily for changes in parasitemia, fever, and PCV/HCT. Horse 344 was additionally monitored through serial complete blood counts (CBCs) and serum biochemical profiles. Group 2 horses were co-infected with T. equi and T. haneyi as described in [8,10]. Briefly, horse 280 was infected with T. haneyi first, and clinically monitored until a state of asymptomatic, persistent infection, characterized by normal complete blood count and physical exam despite positive T. haneyi nPCR, was reached. At that point, 280 was super-infected with T. equi, and again monitored until a state of asymptomatic, persistent infection was reached. Using the same monitoring parameters, horses 275, 277 and 278 were infected with T. equi first, and T. haneyi second. The horses then underwent two rounds of imidocarb dipropionate treatment separated by 2–4 months. Horses were splenectomized 657–743 days later (between 2022 and 2474 days after infection). All horses utilized in this study were negative for T. equi via nPCR at all timepoints post-imidocarb dipriopionate therapy and following splenectomy, but remained T. haneyi positive.
4.2. Blood Collection
Blood was collected via jugular venipuncture daily during the initial acute infection period and serially thereafter for verification of persistent infection. Blood was collected into both ethylenediaminetetraacetic acid (EDTA) and serum separator tubes during both the acute and chronic phases of infection. Blood was processed within two hours of collection with 100 µL aliquots frozen for subsequent DNA isolation. The serum separator tubes were allowed to clot and then subsequently centrifuged to enable serum separation. Serum was then utilized within 2 h for assessment of serum biochemical profiles or aliquoted and stored at −20 °C.
4.3. Light Microscopy
During the acute infection, Giemsa- or Diff-Quik-stained blood smears were evaluated daily for changes in parasitemia. Parasitemia was calculated as previously described [10].
4.4. T. haneyi Nested PCR
DNA was isolated from previously aliquoted whole-blood samples that had been stored at −20 °C. Following thawing at room temperature for 5 min, the DNeasy Blood and Tissue Kit (Qiagen, Inc., Venlo, The Netherlands) was utilized for isolation per the manufacturer’s instructions. DNA was subsequently utilized immediately for PCR or frozen at −20 °C until analysis. [10] The primers utilized amplify a T. haneyi-specific gene that is absent from the T. equi genome and the assay was performed as previously described [8,10].
5. Conclusions
In summary, retrospective assessment of clinical data from T. haneyi-infected, splenectomized horses utilized in previous studies from our lab demonstrates that T. haneyi is less virulent than T. equi, as evidenced by the comparative reduction in mortality and percent PCV/HCT decline in splenectomized, infected horses. This is also the first report of apparent, spontaneous clearance of T. haneyi by splenectomized horses, which, to the author’s knowledge, has not been observed in T. equi. Moreover, this analysis provides insight into the potential role of innate immune effector cells in combatting acute infection that is more readily apparent when the spleen (and its red pulp macrophages) is removed. Splenectomy of persistently infected horses (Group 2) also revealed that long-term exposure to T. haneyi and the resultant memory immune response may enable more rapid control of parasitemia during subsequent periods of recrudescence. Further evaluation is warranted to better characterize this adaptive immune response. Additionally, further evaluation of monocytes and platelets is warranted to determine if and how they are involved in parasite clearance and/or immunopathology during acute and persistent equine theileriosis. In future studies, the discrepant virulence characteristics of these two closely related species of Theileria may be exploited via comparative genomic, transcriptomic, and proteomic analyses during acute and persistent infection to dissect mechanisms of virulence and long-term persistence in Theileria.
Acknowledgments
The authors wish to acknowledge the technical expertise of Shelby Beckner and Nic Durfee, and the animal handling and husbandry expertise of Sara Therrian, Morgan Burke, Emma Karel, and Megan Jacks.
Author Contributions
Conceptualization, K.P.S., L.M.F. and D.P.K.; methodology, K.P.S., D.P.K. and L.M.F.; formal analysis, K.P.S. and L.M.F.; investigation, K.P.S.; resources, D.P.K. and L.M.F.; data curation, K.P.S.; writing—original draft preparation, K.P.S. and L.M.F.; writing—review and editing, D.P.K.; visualization, K.P.S.; supervision, D.P.K. and L.M.F.; project administration, D.P.K. and L.M.F.; funding acquisition, D.P.K. and L.M.F. All authors have read and agreed to the published version of the manuscript.
Funding
The research was funded by USDA-ARS CRIS# #2090-320000-034-00D and in part by the Washington State University, College of Veterinary Medicine Equine Infectious Disease Research Program.
Institutional Review Board Statement
The animal study protocol was approved by the University of Idaho and/or Washington State University Institutional Animal Care and Use Committees (ASAFs 2013-66, 2016-18, 2016-28, and 6241).
Informed Consent Statement
Not applicable.
Data Availability Statement
All data can be found in the manuscript.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Knowles D.P., Kappmeyer L.S., Haney D., Herndon D.R., Fry L.M., Munro J.B., Sears K., Ueti M.W., Wise L.N., Silva M., et al. Discovery of a novel species, Theileria haneyi n. sp., infective to equids, highlights exceptional genomic diversity within the genus Theileria: Implications for apicomplexan parasite surveillance. Int. J. Parasitol. 2018;48:679–690. doi: 10.1016/j.ijpara.2018.03.010. [DOI] [PubMed] [Google Scholar]
- 2.Wise L.N., Kappmeyer L.S., Mealey R.H., Knowles D.P. Review of equine piroplasmosis. J. Vet. Intern. Med. 2013;27:1334–1346. doi: 10.1111/jvim.12168. [DOI] [PubMed] [Google Scholar]
- 3.Mshelia P.W., Kappmeyer L., Johnson W.C., Kudi C.A., Oluyinka O.O., Balogun E.O., Richard E.E., Onoja E., Sears K.P., Ueti M.W. Molecular detection of Theileria species and Babesia caballi from horses in Nigeria. Parasitol. Res. 2020;119:2955–2963. doi: 10.1007/s00436-020-06797-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Coultous R.M., McDonald M., Raftery A.G., Shiels B.R., Sutton D.G.M., Weir W. Analysis of Theileria equi diversity in The Gambia using a novel genotyping method. Transbound. Emerg. Dis. 2019;67:1213–1221. doi: 10.1111/tbed.13454. [DOI] [PubMed] [Google Scholar]
- 5.Bhoora R.V., Collins N.E., Schnittger L., Troskie C., Marumo R., Labuschagne K., Smith R.M., Dalton D.L., Mbizeni S. Molecular genotyping and epidemiology of equine piroplasmids in South Africa. Ticks Tick Borne Dis. 2019;11:101358. doi: 10.1016/j.ttbdis.2019.101358. [DOI] [PubMed] [Google Scholar]
- 6.Zobba R., Ardu M., Niccolini S., Chessa B., Manna L., Cocco R., Pinna Parpaglia M.L. Clinical and Laboratory Findings in Equine Piroplasmosis. J. Equine Vet. Sci. 2008;28:301–308. doi: 10.1016/j.jevs.2008.03.005. [DOI] [Google Scholar]
- 7.Hailat N.Q., Lafi S.Q., Al-Darraji A.M., Al-Ani F.K. Equine babesiosis associated with strenuous exercise: Clinical and pathological studies in Jordan. Vet. Parasitol. 1997;69:1–8. doi: 10.1016/S0304-4017(96)01100-4. [DOI] [PubMed] [Google Scholar]
- 8.Sears K.P., Kappmeyer L.S., Wise L.N., Silva M., Ueti M.W., White S., Reif K.E., Knowles D.P. Infection dynamics of Theileria equi and Theileria haneyi, a newly discovered apicomplexan of the horse. Vet. Parasitol. 2019;271:68–75. doi: 10.1016/j.vetpar.2019.06.009. [DOI] [PubMed] [Google Scholar]
- 9.Wise L.N., Kappmeyer L.S., Silva M.G., White S.N., Grause J.F., Knowles D.P. Verification of post-chemotherapeutic clearance of Theileria equi through concordance of nested PCR and immunoblot. Ticks Tick Borne Dis. 2018;9:135–140. doi: 10.1016/j.ttbdis.2017.08.007. [DOI] [PubMed] [Google Scholar]
- 10.Sears K., Knowles D., Dinkel K., Mshelia P.W., Onzere C., Silva M., Fry L. Imidocarb Dipropionate Lacks Efficacy against Theileria haneyi and Fails to Consistently Clear Theileria equi in Horses Co-Infected with T. haneyi. Pathogens. 2020;9:1035. doi: 10.3390/pathogens9121035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ogier J.C., Pagès S., Bisch G., Chiapello H., Médigue C., Rouy Z., Teyssier C., Vincent S., Tailliez P., Givaudan A., et al. Attenuated virulence and genomic reductive evolution in the entomopathogenic bacterial symbiont species, Xenorhabdus poinarii. Genome Biol. Evol. 2014;6:1495–1513. doi: 10.1093/gbe/evu119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Diop A., Raoult D., Fournier P.E. Rickettsial genomics and the paradigm of genome reduction associated with increased virulence. Microbes Infect. 2018;20:401–409. doi: 10.1016/j.micinf.2017.11.009. [DOI] [PubMed] [Google Scholar]
- 13.Casadevall A. The Pathogenic Potential of a Microbe. mSphere. 2017;2:e15–e17. doi: 10.1128/mSphere.00015-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lover A.A., Coker R.J. Do mixed infections matter? Assessing virulence of mixed-clone infections in experimental human and murine malaria. Infect. Genet. Evol. 2015;36:82–91. doi: 10.1016/j.meegid.2015.08.041. [DOI] [PubMed] [Google Scholar]
- 15.Mackinnon M.J., Read A.F. Virulence in malaria: An evolutionary viewpoint. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2004;359:965–986. doi: 10.1098/rstb.2003.1414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Schmidt L.H., Fradkin R., Sesler C., Squires W., Zeyen P. Attenuation of the virulence of the M strain of Plasmodium cynomolgi during prolonged multiplication in splenectomized rhesus monkeys. Am. J. Trop. Med. Hyg. 1987;37:460–490. doi: 10.4269/ajtmh.1987.37.460. [DOI] [PubMed] [Google Scholar]
- 17.Ueti M.W., Mealey R.H., Kappmeyer L.S., White S.N., Kumpula-McWhirter N., Pelzel A.M., Grause J.F., Bunn T.O., Schwartz A., Traub-Dargatz J.L., et al. Re-Emergence of the apicomplexan Theileria equi in the United States: Elimination of persistent infection and transmission risk. PLoS ONE. 2012;7:e44713. doi: 10.1371/journal.pone.0044713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kuttler K.L., Zaugg J.L., Gipson C.A. Imidocarb and parvaquone in the treatment of piroplasmosis (Babesia equi) in equids. Am. J. Vet. Res. 1987;48:1613–1616. [PubMed] [Google Scholar]
- 19.Zaugg J.L., Lane V.M. Evaluations of buparvaquone as a treatment for equine babesiosis (Babesia equi) Am. J. Vet. Res. 1989;50:782–785. [PubMed] [Google Scholar]
- 20.Bach O., Baier M., Pullwitt A., Fosiko N., Chagaluka G., Kalima M., Pfister W., Straube E., Molyneux M. Falciparum malaria after splenectomy: A prospective controlled study of 33 previously splenectomized Malawian adults. Trans. R. Soc. Trop. Med. Hyg. 2005;99:861–867. doi: 10.1016/j.trstmh.2005.03.008. [DOI] [PubMed] [Google Scholar]
- 21.Buffet P.A., Safeukui I., Deplaine G., Brousse V., Prendki V., Thellier M., Turner G.D., Mercereau-Puijalon O. The pathogenesis of Plasmodium falciparum malaria in humans: Insights from splenic physiology. Blood. 2011;117:381–392. doi: 10.1182/blood-2010-04-202911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ambawat H.K., Malhotra D.V., Kumar S., Dhar S. Erythrocyte associated haemato-biochemical changes in Babesia equi infection experimentally produced in donkeys. Vet. Parasitol. 1999;85:319–324. doi: 10.1016/S0304-4017(99)00110-7. [DOI] [PubMed] [Google Scholar]
- 23.Guimarães A.M., Lima J.D., Tafuri W.L., Ribeiro M.F.B., Sciavicco C.J.S., Botelho A.C.C. Clinical and histopathological aspects of splenectomized foals infected by Babesia equi. J. Equine Vet. Sci. 1997;17:211–216. doi: 10.1016/S0737-0806(97)80563-1. [DOI] [Google Scholar]
- 24.Singh B., Banerjee D.P., Gautam O.P., Gupta R.K.P. Clinicopathological changes in splenectomised donkeys infected with Babesia equi. Indian J. Parasitol. 1980;4:77–80. [Google Scholar]
- 25.Knowles D.P., Jr., Kappmeyer L.S., Perryman L.E. Specific immune responses are required to control parasitemia in Babesia equi infection. Infect. Immun. 1994;62:1909–1913. doi: 10.1128/iai.62.5.1909-1913.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.White N.J. Malaria parasite clearance. Malar. J. 2017;16:88. doi: 10.1186/s12936-017-1731-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kuttler K.L., Gipson C.A., Goff W.L., Johnson L.W. Experimental Babesia equi infection in mature horses. Am. J. Vet. Res. 1986;47:1668–1670. [PubMed] [Google Scholar]
- 28.Cunha C.W., McGuire T.C., Kappmeyer L.S., Hines S.A., Lopez A.M., Dellagostin O.A., Knowles D.P. Development of specific immunoglobulin Ga (IgGa) and IgGb antibodies correlates with control of parasitemia in Babesia equi Infection. Clin. Vaccine Immunol. 2006;13:297–300. doi: 10.1128/CVI.13.2.297-300.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Teuscher F., Gatton M.L., Chen N., Peters J., Kyle D.E., Cheng Q. Artemisinin-Induced Dormancy in Plasmodium falciparum: Duration, Recovery Rates, and Implications in Treatment Failure. J. Infect. Dis. 2010;202:1362–1368. doi: 10.1086/656476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bediako Y., Ngoi J.M., Nyangweso G., Wambua J., Opiyo M., Nduati E.W., Bejon P., Marsh K., Ndungu F.M. The effect of declining exposure on T cell-mediated immunity to Plasmodium falciparum–an epidemiological “natural experiment”. BMC Med. 2016;14:143. doi: 10.1186/s12916-016-0683-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nahrendorf W., Ivens A., Spence P.J. Inducible mechanisms of disease tolerance provide an alternative strategy of acquired immunity to malaria. eLife. 2021;10:e63838. doi: 10.7554/eLife.63838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fry L.M., Schneider D.A., Frevert C.W., Nelson D.D., Morrison W.I., Knowles D.P. East Coast Fever Caused by Theileria parva Is Characterized by Macrophage Activation Associated with Vasculitis and Respiratory Failure. PLoS ONE. 2016;11:e0156004. doi: 10.1371/journal.pone.0156004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kho S., Barber B.E., Johar E., Andries B., Poespoprodjo J.R., Kenangalem E., Piera K.A., Ehmann A., Price R.N., William T., et al. Platelets kill circulating parasites of all major Plasmodium species in human malaria. Blood. 2018;132:1332–1344. doi: 10.1182/blood-2018-05-849307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nafie T.H., El-Allawy T., Mottelib A.A., EL-Amrousi S. Effect of induced equine piroplasmosis on some blood constituents. Assiut Vet. Med. J. 1982;9:123–128. [Google Scholar]
- 35.Rudolph W., Correa J., Zurita L., Manley W. Equine piroplasmosis: Leukocytic response to Babesia equi (Laveran, 1901) infection in Chile. Br. Vet. J. 1975;131:601–609. doi: 10.1016/S0007-1935(17)35194-1. [DOI] [PubMed] [Google Scholar]
- 36.Hviid L., Kemp K. What is the cause of lymphopenia in malaria? Infect. Immun. 2000;68:6087–6089. doi: 10.1128/IAI.68.10.6087-6089.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Aitken E.H., Alemu A., Rogerson S.J. Neutrophils and Malaria. Front. Immunol. 2018;9:3005. doi: 10.3389/fimmu.2018.03005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Juneja S., Januszewicz E., Wolf M., Cooper I. Post-Splenectomy lymphocytosis. Clin. Lab. Haematol. 1995;17:335–337. [PubMed] [Google Scholar]
- 39.Dürig M., Landmann R.M., Harder F. Lymphocyte subsets in human peripheral blood after splenectomy and autotransplantation of splenic tissue. J. Lab. Clin. Med. 1984;104:110–115. [PubMed] [Google Scholar]
- 40.Sponaas A.M., Freitas do Rosario A.P., Voisine C., Mastelic B., Thompson J., Koernig S., Jarra W., Renia L., Mauduit M., Potocnik A.J., et al. Migrating monocytes recruited to the spleen play an important role in control of blood stage malaria. Blood. 2009;114:5522–5531. doi: 10.1182/blood-2009-04-217489. [DOI] [PubMed] [Google Scholar]
- 41.Court R.A., Jackson L.A., Lee R.P. Elevated anti-parasitic activity in peripheral blood monocytes and neutrophils of cattle infected with Babesia bovis. Int. J. Parasitol. 2001;31:29–37. doi: 10.1016/S0020-7519(00)00144-2. [DOI] [PubMed] [Google Scholar]
- 42.Klei T.R., Meinderts S.M., van den Berg T.K., van Bruggen R. From the Cradle to the Grave: The Role of Macrophages in Erythropoiesis and Erythrophagocytosis. Front. Immunol. 2017;8:73. doi: 10.3389/fimmu.2017.00073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Duez J., Holleran J.P., Ndour P.A., Pionneau C., Diakité S., Roussel C., Dussiot M., Amireault P., Avery V.M., Buffet P.A. Mechanical clearance of red blood cells by the human spleen: Potential therapeutic applications of a biomimetic RBC filtration method. Transfus. Clin. Biol. 2015;22:151–157. doi: 10.1016/j.tracli.2015.05.004. [DOI] [PubMed] [Google Scholar]
- 44.Kim C.C., Nelson C.S., Wilson E.B., Hou B., DeFranco A.L., DeRisi J.L. Splenic red pulp macrophages produce type I interferons as early sentinels of malaria infection but are dispensable for control. PLoS ONE. 2012;7:e48126. doi: 10.1371/journal.pone.0048126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Stijlemans B., Cnops J., Naniima P., Vaast A., Bockstal V., De Baetselier P., Magez S. Development of a pHrodo-based assay for the assessment of in vitro and in vivo erythrophagocytosis during experimental trypanosomosis. PLoS Negl. Trop. Dis. 2015;9:e0003561. doi: 10.1371/journal.pntd.0003561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Erhart L.M., Yingyuen K., Chuanak N., Buathong N., Laoboonchai A., Miller R.S., Meshnick S.R., Gasser R.A., Jr., Wongsrichanalai C. Hematologic and clinical indices of malaria in a semi-immune population of western Thailand. Am. J. Trop. Med. Hyg. 2004;70:8–14. doi: 10.4269/ajtmh.2004.70.8. [DOI] [PubMed] [Google Scholar]
- 47.Gérardin P., Rogier C., Ka A.S., Jouvencel P., Brousse V., Imbert P. Prognostic value of thrombocytopenia in African children with falciparum malaria. Am. J. Trop. Med. Hyg. 2002;66:686–691. doi: 10.4269/ajtmh.2002.66.686. [DOI] [PubMed] [Google Scholar]
- 48.Nwaneri D., Oladipo O., Ifebi E., Oviawe O., Asemota O., Ogboghodo B., Israel-Aina Y., Sadoh A. Haematological Parameters and Spleen Rate of Asymptomatic and Malaria Negative Children in Edo South District, Nigeria. Ann. Glob. Health. 2020;86:62. doi: 10.5334/aogh.2458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kini R.G., Chandrashekhar J. Parasite and the Circulating Pool-Characterisation of Leukocyte Number and Morphology in Malaria. J. Clin. Diagn. Res. 2016;10:Ec44–Ec48. doi: 10.7860/JCDR/2016/16425.7887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Dobbs K.R., Embury P., Vulule J., Odada P.S., Rosa B.A., Mitreva M., Kazura J.W., Dent A.E. Monocyte dysregulation and systemic inflammation during pediatric falciparum malaria. JCI Insight. 2017;2:e95352. doi: 10.1172/jci.insight.95352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ortega-Pajares A., Rogerson S.J. The Rough Guide to Monocytes in Malaria Infection. Front. Immunol. 2018;9:2888. doi: 10.3389/fimmu.2018.02888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bastos R.G., Sears K., Dinkel K.D., Knowles D.P., Fry L.M. Changes in the Molecular and Functional Phenotype of Bovine Monocytes during Theileria parva Infection. Infect. Immun. 2019;87:e00703-19. doi: 10.1128/IAI.00703-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data can be found in the manuscript.