Abstract
Some maternal infections, contracted before or during pregnancy, can be transmitted to the fetus, during gestation (congenital infection), during labor and childbirth (perinatal infection) and through breastfeeding (postnatal infection). The agents responsible for these infections can be viruses, bacteria, protozoa, fungi. Among the viruses most frequently responsible for congenital infections are Cytomegalovirus (CMV), Herpes simplex 1–2, Herpes virus 6, Varicella zoster. Moreover Hepatitis B and C virus, HIV, Parvovirus B19 and non-polio Enteroviruses when contracted during pregnancy may involve the fetus or newborn at birth. Recently, new viruses have emerged, SARS-Cov-2 and Zika virus, of which we do not yet fully know the characteristics and pathogenic power when contracted during pregnancy.
Viral infections in pregnancy can damage the fetus (spontaneous abortion, fetal death, intrauterine growth retardation) or the newborn (congenital anomalies, organ diseases with sequelae of different severity). Some risk factors specifically influence the incidence of transmission to the fetus: the timing of the infection in pregnancy, the order of the infection, primary or reinfection or chronic, the duration of membrane rupture, type of delivery, socio-economic conditions and breastfeeding. Frequently infected neonates, symptomatic at birth, have worse outcomes than asymptomatic. Many asymptomatic babies develop long term neurosensory outcomes.
The way in which the virus interacts with the maternal immune system, the maternal-fetal interface and the placenta explain these results and also the differences that are observed from time to time in the fetal‑neonatal outcomes of maternal infections. The maternal immune system undergoes functional adaptation during pregnancy, once thought as physiological immunosuppression. This adaptation, crucial for generating a balance between maternal immunity and fetus, is necessary to promote and support the pregnancy itself and the growth of the fetus. When this adaptation is upset by the viral infection, the balance is broken, and the infection can spread and lead to the adverse outcomes previously described. In this review we will describe the main viral harmful infections in pregnancy and the potential mechanisms of the damages on the fetus and newborn.
Keywords: Cytomegalovirus, Herpes simplex 1-2, Herpes virus 6, Hepatitis B, Hepatitis C, HIV, Parvovirus B19, Enterovirus, Varicella, SARS-Cov-2, Zika, Fetuses, Neonates
1. Introduction
Some maternal infections, contracted before or during pregnancy, can be transmitted to the fetus, during gestation (congenital infection), during labor and childbirth (perinatal infection) and through breastfeeding (postnatal infection). The agents responsible for these infections can be viruses, bacteria, protozoa, fungi.
Among the viruses most frequently responsible for congenital infections are Cytomegalovirus (CMV), Herpes simplex 1-2 (HSV 1-2), Herpes virus 6 (HHV-6), Varicella zoster virus (VZV), Rubella virus (RuV). Moreover, Hepatitis B and C virus (HBV and HCV), Human immunodeficiency virus (HIV), Parvovirus B19 (B19V) and non-polio Enterovirus (EV) when contracted during pregnancy may involve the fetus or newborn at birth.
Recently, new viruses have emerged, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and Zika virus (ZIKV), of which we do not yet fully know the characteristics and pathogenic power when contracted during pregnancy.
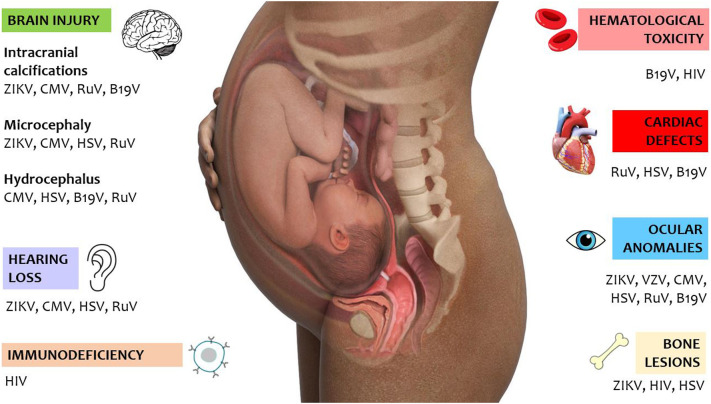
In this review we will describe the main viral harmful infections in pregnancy and the potential mechanisms of the damages on the fetus and newborn (Fig. 1 ).
Fig. 1.
Summary of the potential injuries of viral infections during pregnancy.
2. Search strategy and selection criteria
We searched PubMed using the name of the considered virus (CMV, HSV 1-2, HHV-6, VZV, RuV, HBV, HCV, HIV, B19V, EV, SARS-CoV-2 and ZIKV) in combination with one of the following terms: “pregnancy, vertical transmission, fetus, placenta or birth defect”. We also screened reference lists of identified studies and additional references for this review were identified by each author based on their knowledge on the field. We selected articles published in English from January 2000 to 30th April 2021 that we judged to be the most relevant and pertinent. We focused on articles published in the last 5 years, when possible.
3. Cytomegalovirus
-
1)
Characteristics of the virus
Cytomegalovirus is a DNA virus belonging to the Betaherpesvirinae subfamily of Herpesviridae family. Congenital CMV infection, more frequent in cases of poor socioeconomic status [1], may have serious clinical consequences [2,3]. Considering the relatively high number of CMV seronegative women of reproductive age, the risk of primary CMV infection during gestation is relevant [4]. Moreover, unlike other infectious diseases, the risk of fetal involvement from CMV infection in pregnancy is greater in the population, as the prevalence of serological positivity in women of childbearing age is high. CMV after a first infection can cause reactivations in the pregnant woman, being able to determine fetal infection [5]. In this last situation the probability of CMV transmission to the fetus is lower than in the course of the first maternal infection. The transplacental transmission of human CMV infection during pregnancy is about 20–70% during the primary maternal infections, whilst the risk of transmission/disease is lower as 1–1,5% in case of recurrent infection [6]. Transmission may occur throughout the whole pregnancy period, but mainly in the first trimester [7], coming into contact with body fluids (such as saliva, urine, blood, and genital secretions) from an infected individual.
10–15% of infected neonates become symptomatic early after birth. Symptoms of CMV infection detectable already at birth include intrauterine growth retardation (IUGR), purpura, jaundice, hepatosplenomegaly, microcephaly, hearing impairment, and thrombocytopenia [8]. About 40–60% of cases symptomatic at birth subsequently develop long-term sequelae (neurological disorders, vision and hear loss) [2,3]. In contrast, childhood sequelae developing after an asymptomatic early postnatal period occur in 10–15% of cases, and mostly include progressive hearing loss [9].
-
2)
Mechanisms of fetal damage
A proper understanding of the pathogenetic mechanisms leading to intrauterine CMV infection is auspicable to optimize the prophylactic and/or therapeutic interventions and improve the outcome of the affected children. However, such underlying mechanisms are not completely clear yet [10]. A key issue emerged in the last years is the key role of placental impairment in the pathogenesis of congenital CMV infection. It is known that CMV replicates in cytotrophoblasts, and that CMV infection interferes with cytotrophoblasts differentiation and invasion, inhibits the proper development of new villi and leads to placental edema/fibrosis and impaired transport of oxygen and nutritive substances to the fetus, which may contribute to IUGR [[10], [11], [12], [13], [14], [15]]. This means that although CMV-associated fetal disorders are for sure related to a direct infection of the fetus, such abnormal development and function of the placenta may also strongly contribute to disease development and severity [[10], [11], [12]]. In addition, such CMV-related impairment of placental function may in some cases lead to IUGR even if viral transmission to the fetus does not actually occur [11].
Different molecular mechanisms cooperate to CMV-related placental damage [10,11,13]: a) an impaired development of the extracellular matrix, with decreased expression of integrin molecules, which leads to lower cell adhesion and tissue invasion ability [[16], [17], [18]]; b) IL-10 mediated impairment of matrix metalloproteinases (MMP) activity in the placenta, with subsequent lower expression of HLA molecules on cytotrophoblasts [16,19]; c) activation of the peroxisome proliferator-activated receptor (PPAR), interfering with the biological functions of the cytotrophoblast [20,21]. Interestingly, some of the physiopathologic mechanisms involved in CMV-related poor placentation are similar to those detected in other pregnancy disorders based on placental insufficiency, such as preeclampsia [22]. Considering the crucial involvement of the placental in case of CMV intrauterine infection, it is not surprising that the timing of trophoblast infection may lead to different pregnancy outcomes, with higher incidence of fetal loss after precocious infection and of IUGR or other disorders when infection develops more tardively [23,24].
-
3)
Diagnosis of congenital infection
Unless indicated by specific clinical conditions, such as abnormal ultrasonographic findings (increased periventricular echogenicity, ventriculomegaly, intracranial calcifications and periventricular cysts), no laboratory testing for CMV is officially recommended during pregnancy, although this remains a controversial issue [25]. However, obstetricians often suggest CMV diagnostics during the first trimester of gestation. A combined assessment of serological testing for CMV-specific IgM, IgG and IgG avidity may be of help in the distinction between primary and secondary maternal infections [4]. However, the serological diagnosis of non-primary CMV infection may be challenging, since IgM might be poorly sensitive, detection may vary significantly and since the assessment of IgG titers has been poorly described [26]. Moreover, the detection of CMV-DNA in maternal body fluids could be reliable only if we have an available serological status detected before pregnancy and requires molecular tests with high sensitivity and specificity to detect viral DNA in different body fluids. When primary maternal CMV infection is confirmed, amniocentesis with CMV polymerase chain reaction (PCR) performed on amniotic fluid performed after 21–22 weeks' gestation may detect if in-utero transmission occurred [4]. Furthermore, current knowledge suggests that placental pathologic examination might be of help in the risk assessment for CMV infection in pregnancy [27].
After birth, a PCR assay of urine and/or saliva of the neonate should also be performed within the first 3 weeks of life [28]. A universal neonatal CMV screening using saliva or urine through PCR assay seemed to be a feasible method to identity high-risk infants, even those born to mothers who were not screened during pregnancy, although its cost-effectiveness ratio is yet to be determined [29,30]. Dried blood spot (DBS) samples that are routinely collected because of neonatal screening program for genetic and congenital disorders, could be used in order to distinguish between a congenital and an acquired CMV infection [31].
The finding of a positive PCR on urine and/or saliva after the first 3 weeks of life does not allow to distinguish with certainty the congenital infection from the perinatal one. The latter, in most cases, is acquired by the newborn through breastfeeding from the infected mother and usually has no proven sensorineural consequences.
-
4)
Prevention strategies
There are currently no universal programs that offer screening for pregnant women and newborns that identify infected mothers and babies early, no vaccines to prevent infection, and no conclusive evidence about optimal therapeutic strategies for CMV infection during pregnancy: hygiene education remains a relevant preventive strategy for congenital CMV.
Possible antenatal interventions include pregnancy termination and administration of CMV hyperimmune globulins, which show promising efficacy but have been poorly investigated [32] and are still not routinely recommended [4,28]. Recently Kagan et al. have reported in literature that biweekly hyperimmune immunoglobulins administration at a dose of 200 IU/kg, higher than those previously studied, seems to significantly prevent maternal–fetal transmission up to 20 weeks' gestation, after a primary maternal CMV infection in the first trimester [33].
The currently available antiviral drugs for CMV treatment (valaciclovir, ganciclovir and valganciclovir) have the ability to inhibit the viral DNA polymerase, although with different pharmacologic features [10]. However, poor evidence also exists about the safety and efficacy profile of antivirals for the treatment of neonatal consequences of CMV [34]. A recent prospective randomized clinical trial sustains that early treatment with Valacyclovir, at a dose of 8 g per day, twice daily, reduces significantly CMV fetal infection after maternal primary infection acquired early in pregnancy, without no adverse events [35].
4. Herpes simplex viruses
-
1)
Characteristics of the virus
Herpes simplex virus type 1 and 2 (HSV-1 and HSV-2) are DNA viruses belonging to Alphaherpesvirinae, a subfamily of the Herpesviridae family. Both of them, highly prevalent human pathogens, are transmitted across epithelial mucosal cells and skin discontinuations, migrate to the nerve tissues, and there are able to persist in a latent state [36]. Although the viruses have different preferential site of infection, HSV-1 predominantly involving the trigeminal ganglia and HSV-2 the lumbosacral ganglia, both viruses are able to infect both the orofacial and the genital areas [36]. HSV infection is a sexually transmitted disease widely diffused in the world [37]. Most cases of recurrent genital herpes are caused by HSV-2. It is estimated that in the United States of America almost 50 million people are infected with this type of virus [37]. Since the highest incidence of HSV infections involves women of reproductive age, the risk of maternal transmission to the fetus/neonate is a major health concern [36,[38], [39], [40], [41]]. The clinical condition characterized by acquisition of HSV-1 or HSV-2 without previous exposure to either virus or hence no preformed antibodies is referred to as “first-episode primary infection”. In contrast, the clinical condition characterized by acquisition of HSV-2 in an individual with previous HSV-1 antibodies or vice versa is referred to as “first-episode nonprimary infection”. In contrast, the term “reactivation” refers to the detection of HSV-1 in a patient who already has HSV-1 antibodies, or of HSV-2 in a patient who already has HSV-2 antibodies [38,42]. Recurrence and reactivation of the infection are much more frequent for HSV-2 than for HSV-1 genital infection and the prognosis and the type of counselling needed depend on the type of genital herpes (HSV-1 or HSV-2) causing the infection. It is therefore advisable that the clinical diagnosis of genital herpes is confirmed by type-specific laboratory tests and that positive subjects also be diagnosed for HIV, considering that genital HSV infection has long been identified as a risk factor in HIV acquisition, with a 2- to 3-fold higher risk, also due to multiple sex partners. The biological basis of increased susceptibility is due to both increased ulceration and increased inflammation (with higher CD4+ cells) present in the skin and mucosa of HSV-2 infected individuals [43].
-
2)
Mechanisms of fetal damage
HSV infection process starts with virions binding to host cell surfaces, which triggers a cascade of multiple steps leading to penetration of viral genome into the nucleus, release of viral DNA for replication, capsid assembly, DNA packaging, envelopment via passage through the trans-Golgi network and host cell is destruction [42,[44], [45]].
Possible risk factors for HSV transmission to neonates include: a) HSV serotype (HSV1 > HSV2); b) type of maternal infection (recurrent HSV genital lesions are associated with lower risk for transmission to exposed neonates than primary HSV genital lesions; c) maternal serostatus; d) delivery mode (vaginal delivery > cesarean section); e) prolonged duration of rupture of membranes; f) impaired fetal cutaneous barriers (instrumentation-related) [38,39,44]. Timing of maternal infection is fundamental to establish: the risk for transmission of HSV infection to the neonate is high (30–50%) when women acquire genital HSV near the time of delivery and low (<1%) when women have prenatal history of recurrent HSV infections or who acquire genital HSV during the first half of pregnancy) [46]. Vertical transmission of viral infection may occur during the intrauterine period (5%), in the peripartum period (85.9%) or postnatally (10%) [38,39,42,44]. HSV-1 prevalence was higher than HSV-2 at both maternal and fetal side of the placenta, evidencing how crossing of maternal-fetal interface is a common strategy used by HSV-1, whereas in the case of HSV-2 infection the hematogenous route is more frequent [47].
Intrauterine HSV infection has been associated with encephalomalacia, hydrocephalus, calcifications and microcephaly [48]: HSV-infected neonates could display significant neurodevelopmental outcomes [49].
-
3)
Diagnosis of congenital infection
From a clinical point of view, HSV congenital infection may be associated with relevant morbidity and mortality and antiviral treatment (acyclovir), with different duration according to clinical presentation, is desirable. Maternal HSV infection during pregnancy increase the risk of spontaneous abortion, premature birth, and stillbirth [41]. Neonatal HSV infection may present with different clinical manifestations, predictive of both morbidity and mortality: a) skin, eyes, and mouth herpes (SEM) disease, without involvement of the central nervous system (CNS)/visceral organs, usually presenting as a vesicular rash occurring at 10–12 days of life; b) CNS disease, with (60–70%) or without skin involvement, with clinical manifestations of encephalitis, potentially starting at any time within the first month of life; c) disseminated disease, a life-threatening condition involving multiple organs including CNS, lungs, liver, adrenal, skin, eye, and/or mouth, with about 40% of infants never developing a vesicular rash [39,42,44,50]. HSV DNA isolation in tissue culture by PCR (swabs from conjunctivae, nasopharynx, mouth, anus; blood; cerebrospinal fluid) is a definitive diagnostic test [39,42,44]. HSV DNA detection is also routinely performed when HSV infection is suspected [42,44]. Plasma HSV DNA levels at the time of diagnosis seem to correlate with clinical classification of neonatal HSV disease, being higher in infants showing disseminated HSV infection [[51], [52]] but further validation of such findings is required. Serologic diagnosis of neonatal HSV is not usually recommended for diagnostic purposes [39].
-
4)
Prevention strategies
Currently, interventions to decrease HSV neonatal transmission remain a challenge [38,39,[53], [54], [55], [56]]. The prevention of neonatal herpes depends on the prevention of maternal infections near delivery and on the prevention of exposure of the newborn to active maternal lesions. Given the highest risk of serious neonatal infection in newborns from women who acquire genital infection in the last trimester of pregnancy, these women must be managed by both obstetricians and infectious diseases specialists. The history of genital herpes should be investigated in all pregnant women and at the onset of labor, all women should be carefully examined for HSV 1–2 lesions. Cesarean delivery, recommended when genital lesions or prodromal symptoms are present at the time of delivery, minimizes neonatal exposure to the virus and therefore decreases HSV transmission, but does not completely prevent it [38,39,53,55]. Antiviral suppressive therapy during pregnancy in women with recurrent genital herpes at 36 weeks of gestation seems to be associated with decreased occurrence of genital lesions at the time of delivery and lower HSV detection by culture/PCR. The recommended regimen of therapy in pregnant women with recurrent genital herpes is Acyclovir 400 mg orally three times a day or Valacyclovir 500 mg orally twice a day, started at 36 weeks of gestation [55]. However, such intervention reduces but does not completely prevent viral transmission to the neonate [55]. To date, no vaccine has proven to be effective to prevent yet the acquisition of HSV-1/HSV-2 [38,39]. Strategies aimed at preventing maternal HSV acquisition during pregnancy, such as screening of all women at 24–28 weeks of gestation with an immunoglobulin G-based assay and screening of all couples for HSV serology at 14–18 weeks of gestation, have been assessed in the last years but still lack a final validation [38,39,56].
5. Herpes virus 6
-
1)
Characteristics of the virus
Human herpesvirus 6 (HHV-6) is a double stranded DNA virus belonging to the Betaherpesvirinae subfamily of Herpesviridae family, with two distinct variants: HHV-6A and HHV-6B. In analogy with other herpesviruses, HHV-6 can establish a latency in immune cells and is capable to reactivate after the first infection [57]. Both HHV-6A and HHV-6B replicate in immune cells (especially CD4+ T cells) but viral entry is based to different receptors: human cluster differentiation 46 (CD46) is the primary receptor for HHV-6A virus, while HHV-6B uses cluster differentiation 134 (CD134) [57].
HHV-6A is sometimes isolated from immunosuppressed patients [58], while HHV-6B infections are most frequent in early childhood and usually occur without clinical severity. Moreover HHV-6 causes pitiriasis rosea, a self-limiting exanthematous disease that in pregnancy may be associated with many negative outcomes, when the infection occurs before 15 weeks of gestation and the blood viral load is higher than 585 copies/mL [59]. Clinical features in children vary from typical exanthem subitum (also known as 6th disease) [60] to afebrile seizures [61], neonatal urticaria multiforme [62] and meningitis [63]. Most infants acquire HHV-6 from the saliva of asymptomatic contacts [64].
-
2)
Mechanisms of fetal damage
HHV-6 is known to integrate itself into human genome (chromosomally integrated HHV-6, ciHHV-6) at specific sites [58], through homologous recombination between telomeric regions of human chromosomes and HHV-6 sequences [65]. The HHV-6 specific U94 gene product could also favor integration [65]. Congenital HHV-6 infections occur in about 1% of neonates through two modalities: in the 86% of cases the virus is integrated in maternal or paternal chromosomes and transmitted through the germ-line; in the remaining 14% of cases transmission occurs by transplacental route [66,67]. Furthermore, integration can occur in somatic cells or germinal cells, the first not transmitting the virus via the germ line while the latter leading to half of gametes carry ciHHV-6 [68].
Hypothetical cellular consequences associated with ciHHV-6 include no viral gene transcription, viral gene expression and replication, anomalies of telomeres and impaired chromosome stability, activation of cellular gene expression after integration, elimination of tissues or cells expressing HHV-6 antigens by immune defense mechanisms [65]. Exposure to viral infection during pregnancy has been associated to preterm delivery [69] and fetal loss, as the presence of HHV-6 DNA was detected both in the placenta of miscarrying women and in the tissues of miscarried fetuses [70]. Indeed, HHV-6A DNA can induce alterations in the endometrial cells and interfere with trophoblast invasion and correct implantation [71] and inherited ciHHV-6 has been confirmed as a risk factor for spontaneous abortion [72].
Furthermore, persistency of viral activity in a developing brain could lead to poor neurodevelopmental outcomes, such as in congenital CMV infection and further studies are thus needed to clarify this association [66].
-
3)
Diagnosis of congenital infection
Congenital infection is usually defined as HHV-6 DNA is detected by PCR in blood samples within 72 h since birth [58].
Transplacentally-acquired infections are those with HHV-6 DNA found in cord blood samples but with lower viral loads (≤1 genomic equivalent copy (gec) per 104 to 105 leukocytes, or <1 gec/μg of HHV-6 DNA) than in chromosomally integrated infections (≥1 gec per leukocyte, or ≥ 1–2 × 105 gec/μg DNA) [67].
A further method to distinguish chromosomally integrated and transplacentally-acquired infections is to examine hair follicle samples for the presence of HHV-6 DNA: when congenital infection is due to ciHHV-6, HHV-6 DNA is detected in hair follicles in both the infant and at least one parent [67].
Infants reported by Hall and colleagues were all asymptomatic: one-third of congenital infections were due to HHV6-A, whereas in all postnatal infections HHV-6B was detected [64].
Increasing use of PCR-based pathogen detection panels in newborns may lead to more always frequent diagnoses of HHV-6 and interpretation of this finding could be challenging to interpret [73].
-
4)
Prevention strategies
Maternally acquired HHV-6 antibodies are usually found in neonates, reflecting the high prevalence in adults [58], with a protective effect on their still immature immune system [74]. To date, no vaccine is available to prevent the acquisition of HHV-6.
Intravenous ganciclovir is feasible to decrease viral load but treatment for HHV-6 is appropriate only in patients who are either immunocompromised, have a high viral load (without a ciHHV-6), or present with a severe disease [73].
6. Varicella zoster virus
-
1)
Characteristics of the virus
The Varicella Zoster virus (VZV), also known as the human herpes virus 3, is an exclusively human neurotropic virus that belongs to the Alpha-herpesvirinae family. Its double-stranded DNA genome (of about 125 kb) shares much homology with the HSV genome. The virus is enveloped, spherical and 200 nm in diameter. It has an icosahedral symmetry and the capsid consist of 162 capsomers. Glycoproteins are embedded in the lipid envelope. VZV has no animal reservoir; its main targets are T lymphocytes, epithelial cells and ganglia. Initial infection results in varicella (chickenpox), most commonly affecting children. The disease is highly infective and is characterized by a skin rash that forms small, itchy blisters that can be associated with fever. Symptoms appear 10 to 21 days after exposure to the virus and usually last five to seven days. Primary infection usually confers lifelong immunity to this disease [75].
Whereas Varicella in children is usually self-limiting, primary infection in adults is usually more severe and can cause interstitial pneumonia. During the primary infection VZV becomes latent in ganglionic neurons and can reactivate causing Zoster (shingles), due to a decline in cell-mediated immunity. Zoster can be complicated by a wide range of neurologic diseases (postherpetic neuralgia, myelopathy, retinal necrosis, vasculopathy and meningoencephalitis and cerebellitis) as well as visceral involvement (hepatitis and pancreatitis) [76].
Transmission of Varicella usually occurs through respiratory droplets from 48 h prior the onset of the rash until all lesions have crusted over but can also occur through contact with the blisters and to the fetus through placenta.
-
2)
Mechanisms of fetal damage
Vertical transmission of viral infection may occur during pregnancy. The risk of contracting varicella during pregnancy is low, because women of childbearing age have a high seroprevalence state. A survey conducted in the US showed that only 5% of adults aged 20–29 years were susceptible to contract varicella, and only 1.1% of those aged 30–39 years were still susceptible [77].
Maternal infection is associated with still birth and later with premature delivery. The risk of developing Congenital Varicella Syndrome (CVS) if a pregnant woman develops varicella is 0.91%; it is low during the first 12 weeks (0.4%) and highest during weeks 13 to 20 (2%). On the contrary, the risk for adverse effects for the mother is greatest in the third trimester of pregnancy. Although rare, CVS can range in severity from segmental lesions including scar and limb deformities [78] as well as ocular abnormalities (chorioretinitis, chorioretinal scarring, cataracts), brain abnormalities (microcephaly, encephalitis, hydrocephaly) and mental retardation [79]. Children born to mothers with varicella during pregnancy can also develop Herpes Zoster during the first year of life.
The pathogenesis of CVS reflects intrauterine dissemination of infection and the failure of the fetal immune system to determine the virus-latency, which normally occurs in postnatal VZV infection. Since VZV is a lymphotropic virus, it has the potential to spread to all fetal organs by the hematogenous route. Examining fetuses infected with maternal varicella, VZV has been shown to be distributed throughout the fetal tissues. This finding supports the hypothesis that VZV causes viremia, in the absence of an adequate immune response of the fetus [80]. Given that VZV is a neurotropic virus, many of the CVS defects are the result of infection of the spinal cord and ganglia, with destruction of the plexus during embryogenesis and denervation of the limb bud and subsequent hypoplasia. Failure of muscle development also causes damage to the bone formation of the limbs. Skin lesions may reflect sensory nerve involvement. Infection of optic tract cells also explains optic atrophy and chorioretinitis. Some skin lesions, distributed according to dermatomers, may be caused by the reactivation of the VZV in utero. The short latency phase, if latency is established, can be explained by the poor cell-mediated immune response in the fetus [81]. Neonates born to mothers in whom varicella develops between 5 days before delivery to 2 days after delivery have a 50% risk of developing a “neonatal varicella”. In 23% of cases the infection can be severe or even life threatening, therefore these neonates must receive varicella zoster immunoglobulins and antiviral therapy immediately after birth. Shingles in pregnancy is not associated with viremia and no adverse fetal sequelae have been described [82].
-
3)
Diagnosis of congenital infection
When a pregnant woman has a suspicious varicella contact, a serology assay should be performed to check her immune status against varicella. In case of seronegativity, prevention should be carried out using specific immunoglobulins. Contrarywise, clinical varicella does not require virology confirmation but requires immediate treatment with immunoglobulins. Recently valacyclovir has been proposed to treat varicella in pregnant women [83]. If CVS is suspected invasive testing such as amniocentesis or cordocentesis can be performed to confirm fetal infection by detecting VZV DNA. In contrast to other viral illnesses, VZV IgM are hard to detect in fetal blood and are not routinely tested. Nevertheless, the demonstration of VZV DNA in cord blood is not synonymous of CVS. In fact, congenitally infected fetuses without any anatomical abnormalities have a good prognosis and no neurological impairment. Therefore, the diagnostic modality of choice to diagnose CVS remains fetal ultrasound, that can detect if there are some CVS associated abnormality [84]. When a neonate with CVS is born, it is important to determine if the disease is active. If the baby has skin lesions and positive molecular or IgM serological testing for VZV, then he should be isolated to prevent viral spread. Neonates born to mothers in whom varicella developed 2 days before to 5 days after delivery should always be isolated because potentially infected.
-
4)
Prevention strategies
Prevention strategies are crucial to prevent the risk of CVS as, although rare, CVS can have serious adverse outcomes for the affected child. As a live attenuated varicella vaccine is available, vaccination of all the women in childbearing age with a negative varicella history, confirmed by negative VZV IgG antibody test, should be encouraged. Pregnancy should be avoided for at least one month after vaccination [79].
If a susceptible pregnant woman is exposed to a person with varicella, CDC recommends administration of varicella zoster immunoglobulins (VariZIG) as soon as possible after exposure to varicella-zoster virus and within 10 days after contact [85].
Even though varicella infection can be prevented in <50% of cases, immunoglobulins can reduce the severity of the disease [86]. Recently valacyclovir has been proposed to treat pregnant women who develop CMV or varicella infection early in pregnancy [35].
7. Rubella virus
-
1)
Characteristics of the virus
Rubella virus (RuV) is a member of the genus Rubivirus, belonging to the family of Matonaviridae. Its genome of single-stranded RNA is enclosed by an icosahedral capsid. The viral genome codes for the two non-structural (p90, p150) and three structural proteins: viral capsid protein (C) and surface glycoproteins (E1 and E2). Human beings are the only known host. Rubella infection, also known as three-day measles or German measles (because firstly described by two German physicians), is typically mild and occurs mostly in childhood. The exanthem for Rubella is a maculopapular rash on the face which spreads to the trunk an limbs and usually vanishes within three days, while the typical exanthem (Forchheimer's sign) consists of petechiae on the soft palate: fever, lymphadenopathy, myalgias, and arthralgias may also occur [87].
Transmission usually occurs through respiratory droplets but can also occur to the fetus through placenta. Vertical transmission of rubella is higher in the first weeks of gestation, leading to miscarriage, stillbirth, or a chronic injury of several organs in the fetus, well-known as Congenital rubella syndrome (CRS), with the classical fetal Gregg's triad of congenital cataracts, deafness, and cardiac defects [88].
-
2)
Mechanisms of fetal damage
During maternal viremia, RuV may cross the placental barrier and enter into the fetal circulation, causing a necrotizing vasculitis. The virus is probably transported as infected endothelial cell emboli, resulting in a chronic injury [89]. The exact mechanisms at the basis of CRS are still not completely understood, considering the near eradication of rubella and the absence of animal models to study.
Gestational age (GA) at the time of maternal infection plays a key role in causing fetal damage. The risk of CRS is higher during the first 11–12 weeks of GA and then clearly decreases [90].
RuV antigen was detected in several organs (lung, heart, aorta and pulmonary artery, brain, placenta) of cases with a poor prognosis [90]. Apoptosis pathways have been showed to involve RuV-infected adult cells, whereas apoptosis in fetal cells was not supported and this could promote viral persistence in developing cells [91].
Furthermore, RuV infection could play an indirect role in impairing growth and differentiation mechanisms through secretion of interferon and other chemokines [92].
Chronic induced injury has been confirmed by the observed increase of inflammatory chemokines (CCL5, CXCL10 and CXCL11) in case of RuV-infected endothelial cells [93]. Interestingly, the chemokine CCL14 that promotes embryo implantation was down-regulated, as in parallel a set of genes involved in eye and ear development has also been shown as down-regulated in a model with RuV-infected fetal endothelial cells [93].
CRS includes a spectrum of several defects: eye defects (such as cataracts, microphthalmia, chorioretinitis, corneal opacity and glaucoma), sensorineural hearing loss, cardiovascular anomalies (most commonly patent ductus arteriosus, peripheral pulmonary artery stenosis or coarctation of aorta), brain injury (like microcephaly, hydrocephalus and cerebral calcifications) [94].
Low birth weight was observed in 9% of CRS cases [95].
-
3)
Diagnosis of congenital infection
A confirmed case of CRS has been defined as an infant with at least one of the previously described clinical features, and a laboratory evidence of RuV by polymerase chain reaction (PCR) in a specimen at birth (nasopharyngeal swab, urine, cerebrospinal fluid or blood) or positive RuV-specific IgM antibodies at birth to 3 months or a RuV antibody level that persists more than expected from a passive transfer of maternal antibodies [96]. Avidity testing of IgG could also help diagnose recent infections.
Despite the absence of clinical signs of CRS in the second part of pregnancy where the development of most organs is already completed, such cases are reported as Congenital Rubella Infection (CRI) if laboratory confirmed.
Perinatal infection could be accompanied by low birthweight and non-specific features such as the purpuric “blue-berry muffin” rash, hepatosplenomegaly, and thrombocytopenia [94].
A follow-up study showed that most infants with CRS later reported sensory defects and developmental delay [97]: this explains the importance of an early identification and an adequate follow-up.
-
4)
Prevention strategies
Congenital Rubella syndrome is nowadays rare in countries with a well-established immunization program against this virus and it represents an important model in order to design future public health strategies against prenatal infections [96], although screening for rubella serostatus is still recommended for all pregnant women to promptly intervene and carry out the appropriate strategies in case of a positive result [98]. Immunity to rubella virus is commonly determined by measuring rubella-specific IgG, despite a lack of standardization of available assays could influence interpretation of results [99].
Rubella is a vaccine-preventable viral self-limiting infection, without a specific treatment. Live-virus vaccines, such as mumps, measles and rubella containing vaccine (MMR), are contraindicated during pregnancy and women should be advised to avoid pregnancy for one month after receiving RuV-vaccine [100]. However, there is still no evidence of CRS in infants born to mothers who had received a RuV-vaccine during pregnancy and an inadvertent vaccination should not be an indication for termination of pregnancy [101].
In case of exposure to the virus during pregnancy, polyclonal immunoglobulins administered up to five days after seem to be of benefit for preventing rubella [102].
8. Hepatitis b and C viruses
-
1)
Characteristics of the virus
Hepatitis B virus (HBV) is a partially double-stranded DNA virus, belonging to the Hepadnaviridae family of viruses [103], whereas hepatitis C virus (HCV) is a small, enveloped, positive-sense single-stranded RNA virus of the Flaviviridae family [104]. They are both transmitted via blood or non-sterile needle use (through drug injections, tattoos or medical procedures) or via sexual contacts.
Hepatitis B surface antigen (HBsAg) is a particle produced in excess during the life cycle of the virus: HBsAg is the first detectable viral antigen to appear during infection but could also be later found, whereas core antigen (HBcAg) and envelope antigen (HBeAg) are indicators of active viral replication. Anti-HBs antibodies can be detected after seroconversion or after a successful anti-HBV immunization, while HBV-DNA is a feasible marker to monitor viral load [105]. The risk of Mother-to-Child transmission (MTCT) of HBV is dependent on the timing of exposure: in the case of a HBV-infected mother in the third trimester, the risk is higher and vertical transmission will occur in 80% to 90% [106].
The presence of HCV-RNA indicates active infection: the risk of vertical transmission of HCV is reduced in women with an undetectable HCV-RNA, although an intermittent viral load could lead to vertical transmission [107]. Approximately HCV infection occurs in 6 to 11% of infants born to HCV-infected mothers [104]; the risk is greater when mothers are HIV co-infected [108].
-
2)
Mechanisms of fetal damage
Transplacental leakage (related to immature placenta or uterine contractions), placental infection, and infected maternal peripheral blood mononuclear cells (that turn out to be HBV carriers from mother to fetus) have been described as potential mechanisms for HBV intrauterine transmission, considering that only HBeAg is small enough to cross placenta [109]. During pregnancy, significant changes in maternal immune system usually occur to avoid rejection of the semiallogenic fetus. The natural course of HBV infection is influenced by a depressed maternal cell-mediated immunity [110], that could stimulate MTCT and the development of an immune-tolerant HBV infection in the fetus [111]. Although acute HBV infection during pregnancy is not associated with increased fetal mortality or teratogenicity [105], a higher risk of gestational diabetes, postpartum hemorrhage, preterm birth, and low birthweights was reported among complications of HBV infected pregnant women [110,112]. Vertically HBV-infected infants are usually asymptomatic and anicteric, but they are likely to have a chronic infection afterwards in 90% of cases [113].
As in other viral infections, human trophoblast cells express cellular receptors (Occludin, Claudin-1 and low-density lipoproteins receptor) that HCV use to enter into host cells. This leads to a strong antiviral response that alters innate immunity at the maternal-fetal interface. Furthermore, immunologic changes trigger apoptotic pathway in extra-villous trophoblast cells, altering placental morphology [114]: this probably plays a key role in pregnancy-related complications: women with HCV infection are more likely to have stillborn infants [115] or infants born preterm, with low birthweight or birth defects [116].
-
3)
Diagnosis of congenital infection
All pregnant women should be routinely screened for HBsAg: if HBsAg tested positive, a complete assessment including HBV-DNA, HBV serologic markers and liver function tests should be performed, in order to identify if any antiviral therapy should be started or not [117]. If HBsAg tested negative and the woman has a high risk to acquire HBV infection (e.g. having an HBsAg-positive sex partner), vaccination should be performed since pregnancy is not a contraindication [105].
At birth, perinatally acquired HBV infection could be confirmed detecting HBV-DNA in neonatal serum. HBsAg and HBeAg could be also detected, whereas serum aminotransferases are normal or only slightly elevated [113].
Similarly, infants born to HCV-positive mothers should be tested via HCV-RNA PCR and regularly followed up to 18 months (when clearance of maternal antibodies is expected) [118]. They should be retested after 3 months of age because of possible false-negative results in case of a sole earlier testing [119].
-
4)
Prevention strategies
All pregnant women should be tested not only for HBV, but also for HCV [120]. Prenatal invasive procedures should be minimized in pregnant women with hepatitis when possible, whereas cesarean section should not be performed for the sole aim to reduce risk of HBV and HCV vertical transmission [121].
The risk of HBV perinatal transmission is higher when HBV-DNA viral load exceeds the threshold of 200,000 IU/ml (>106 copies/mL) and could be reduced with any antiretroviral therapy (lamivudine, telbivudine, and tenofovir), started at 28–32 weeks of gestation [122]. There is no increased risk of adverse maternal or fetal outcomes, although safety data are limited [123]. The use of antiretroviral therapy is not recommended when HBV-DNA are lower than 200,000 IU/ml [122].
Maternal administration of HBV-specific immunoglobulin can have an effect on preventing MTCT of HBV, although with a limited evidence [124]. Infants born to HBsAg-positive women should receive post-exposure prophylaxis (PEP: HBV vaccination and HBV-specific immunoglobulin) within 12 h of life. The risk of transmission to the infant born to a HBsAg-positive/HBeAg-negative mother is low (about 10%) if PEP is correctly given at birth. Conversely, the risk increases up to 90% in infants born to HBsAg-positive/HBeAg-positive mothers if not given PEP [110]. Infants born to HBsAg-positive mothers should be followed up for completion of HBV vaccination and postvaccination serologic testing (PVST), to evaluate effects of immunization and eventually booster immune response in low-responding infants with further doses [117].
The risk of HCV perinatal transmission is increased when HCV-RNA viral load exceeds the threshold of 615 copies/mL and rupture of membrane is longer than 6 h [107]. Ribavirin and interferon are not used in pregnancy because of their teratogenic effects. Direct-acting antivirals (DAAs) have not yet approved for use during pregnancy, although no teratogenic effects have been reported in animals [125]. A phase 1 pharmacokinetic and safety trial (ClinicalTrials.gov number NCT02683005) evaluated ledipasvir-sofosbuvir use in pregnancy [126]: recruitment was completed in March 2020 but results were not yet published at time of writing. In the absence of an available vaccine against hepatitis C, an accurate screening of infants HCV-exposed during pregnancy is to be pursued to avoid that many pediatric infections remain undetected [127], providing a prompt treatment and preventing thus an early-onset cirrhosis [128].
9. Human immunodeficiency virus
-
1)
Characteristics of the virus
Human immunodeficiency virus (HIV) is a member of the genus Lentivirus (within the family of Retroviridae). It has a single-stranded, positive-sense, enveloped RNA. The virus targets CD4+ lymphocytes, where it embeds itself into the host cell genome. After entry into the lymphocytes, the viral RNA is converted into double-stranded DNA by a reverse transcriptase, and integrates itself in the host genome, progressively leading to acquired immunodeficiency syndrome (AIDS) and death. Even with the most potent antiretroviral therapy, the virus has been shown to remain latent in peripheral blood monocytes. The infection is therefore permanent. Two types of HIV have been identified: HIV-1, more virulent and globally spread, and HIV-2, lower virulent and confined to West Africa [129]. In most cases, HIV is a sexually transmitted infection, through blood, semen, and vaginal fluids. Mother-to-Child transmission (MTCT) could occur during pregnancy, and not only at delivery, but also in the postpartum period through breastfeeding [130]. Evidence of in-utero transmission of HIV has been demonstrated on the syncytiothrophoblast by the presence of cluster of differentiation 4 (CD4) and CC chemokine receptor type 5 (CCR5) receptors, that the virus uses as coreceptors to enter into cells [131]. HIV was found in fetal circulation regardless of maternal Highly Active Anti-Retroviral Therapy (HAART) [131].
-
2)
Mechanisms of fetal damage
Systemic immune activation begins early after HIV infection, causing a chronic inflammation [132], but pregnancy seems to counterbalance it: Pereira et al. recently described how the placenta keeps the ability to stimulate an intense antiviral network mediated by a pronounced type I interferon response, which could limit the vertical transmission at the maternal-fetal interface, regardless of HAART, but also lead to immunological dysfunction of the newborn [133]. Placental macrophages (Hofbauer cells) seem to be pivotal in in-utero HIV transmission through constitutive expression of regulatory cytokines, which have been demonstrated to inhibit HIV-1 replication in vitro [134]. T regulatory cells (Treg) could also have a role in contrasting MTCT, probably suppressing immune activation in fetuses and infants that has been found more pronounced in HIV-infected infants [135].
The effect of HAART on maternal HIV-induced inflammation is complex: it depends on immunodeficiency and the viral reservoir at the start of the treatment [136]. Adherence to HAART is crucial in pregnancy, although one of the barriers is the fear around side-effects of these drugs for pregnant women and their fetuses. The association between HAART and adverse birth outcomes is still debated [137]. Specific congenital defects that were significantly associated with specific drug exposures were male genital (zidovudine and lamivudine), musculoskeletal (atazanavir, ritonavir, and didanosine associated to stavudine), cardiovascular (atazanavir and ritonavir) and skin (atazanavir) anomalies [138] and neural tube defects (dolutegravir) [139].
It is now widely accepted that HAART in pregnancy is associated with increased risk of preterm delivery, but questions remain about the exact nature of this association, for example how timing of HAART initiation (pre- or post- conception) and maternal immunological status (e.g. CD4+ T-cell count) are involved [136]. Low birthweight could be also a proxy for preterm birth, but there is a little evidence that HAART exposure during pregnancy influences placental vascular changes and consequently may drive to fetal growth restriction [140].
On the other hand, untreated maternal HIV infection is equally correlated with an increased risk of adverse birth outcomes, such as premature delivery, low birthweight/small for gestational age neonates and stillbirth, especially in women with advanced HIV disease [137]. The evidence for an association of maternal HIV infection with adverse perinatal outcomes was still strong in sub-Saharan Africa, where a greater maternal HIV-1 burden occurs [141].
Before HAART, cardiovascular complications occurred in up to 25% of HIV-infected children, with an earlier myocardial dysfunction [142]. Neurodevelopmental outcomes have been reported as improved if HAART is offered earlier to HIV-infected infants [143], reducing the effects of HIV replication on their immature brain and consequently future HIV-associated neurocognitive disorders (HAND).
-
3)
Diagnosis of congenital infection
Serologic HIV testing methods are not useful in infants during the first 18 months because of placental transferring of maternal immunoglobulin IgG: even HIV-exposed uninfected (HEU) could test positive. They are generally informative only if the test result is negative [144].
Virologic assays, including HIV-1 DNA or RNA PCR assays, represent the gold standard to diagnose HIV infection in infants [145].
A positive HIV-DNA PCR by 48 h of age is the most reliable marker of in-utero infection: a considerable limitation of this method is the possibility of false negatives in case of non-B-subtype HIV-1 infections. HIV-RNA PCR assay is better to use in case of non-B-subtype HIV-1 infections, although the risk of a false negative test should be considered with antiretroviral prophylaxis [145].
After birth, infants should thus be tested within first days of life and then again .at 2–3 weeks of age, at 1–2 months of age and at 4–6 months of age [146].
All pregnant mothers should be screened for HIV in the first trimester: for infants born to mothers with unknown HIV status, a rapid HIV screening of mothers and/or infants should be performed as soon as possible, either during labor or after delivery, starting immediately the appropriate neonatal postexposure prophylaxis (PEP) if the test is positive [147].
Challenges in early diagnosis of HIV include missed or delayed testing, poor access to care, loss to follow-up but especially mortality peak in HIV-infected infants. Point-of-care HIV testing of infants with heel-prick sampling could be a solution for some of these problems, in particular in low-resources settings [148].
Children with congenital HIV infection are usually asymptomatic at birth: symptoms usually occur after 3 months and within 10 years, with lymphadenopathy, weight of loss, recurring infections and a progressive developmental delay [149,150].
-
4)
Prevention strategies
One of the greatest successes in the field of HIV prevention has been the prevention of MTCT, using HAART [132]. As mentioned, the virus can be detected in peripheral monocytes although not detectable in plasma, therefore the CDC recommends this regimen also for pregnant women with HIV RNA levels below 1000 copies/ml. All pregnant women with HIV should be treated with HAART regardless of their CD4+ count to decrease vertical transmission [146]. Elective cesarean delivery in HIV-infected women who have not received HAART, when performed prior to rupture of membranes and the onset of labor, reduces the vertical transmission rate by 50%. If HIV infection is untreated, the risk of vertical transmission is as high as up to 25%, while is virtually zero in mothers on well-assumed HAART before conception and a suppressed plasma viral load [151].
Usually HIV-exposed infants receive since birth a zidovudine (ZDV) PEP, that should be started as earlier as possible, better within 12 h of delivery, even if mothers correctly took antiretrovirals. Neonatal PEP with ZDV when infants are born to women with undetectable viral load could be shortened to 4 weeks (instead of 6) [152]. If risk of MTCT is higher, combination antiretrovirals regimens with two or more drugs are increasingly preferred without further side effects compared with ZDV-monotherapy prophylaxis [153].
However, there are still many concerns regarding also HEU infants of mothers who correctly received antiretrovirals. HEU infants receiving neonatal PEP with ZDV showed a lower trend (although not significant) in CD4+ and CD8+ T-cells and CD19+ B-cells than those who did not, suggesting a possible toxicity that could impact their global health [154]. Growth impairment in infants exposed to any antiretroviral regimen should be carefully followed [155].
Anyway, we should keep in mind that, without HAART, 50% of HIV-infected infants in Africa would die within 2 years of life, compared to a median survival time of 11 years from time of infection in adults without treatment [156].
To date, besides HAART, obstetrical interventions (such as scheduled cesarean section before labor onset where appropriate) formula feeding and intrapartum maternal prophylaxis are still the sole viable options to prevent HIV infection in perinatally exposed HIV-infants. None of the developed HIV vaccines has been approved so far: the calling for the development of an effective and safe vaccine to prevent the infection and end the AIDS pandemic remains therefore still open.
10. Parvovirus B19
-
1)
Characteristics of the virus
Human parvovirus B19 (B19V) is a member of the genus Erythroparvovirus (within the family of Parvoviridae) and three genotypes have been identified, of whom genotype 1 is the most prevalent and ubiquitous worldwide. It is a non-enveloped, icosahedral virus that contains a single-stranded linear DNA. The capsid surface with its two main structural proteins (VP1 and VP2) is involved in many functions in the virus life cycle: specific binding to host cell receptors, internalization mediated by its phospholipase A2 (PLA2) activity, nuclear entry, and recognition and avoidance of the host immune response [157]. In most cases, B19V is spread through respiratory droplets [158] but could also be transmitted vertically through the placenta to the fetus and through transfusion of blood products [159]. The target cell of B19V is the erythroid progenitor in the bone marrow, through globoside or P blood group antigen, that acts as a cellular receptor, inducing apoptosis and a transient arrest of erythropoiesis [157].
The common clinical presentation of B19V infection is “Fifth Disease”, also known as erythema infectiosum, which occurs mostly in healthy children: the typical “slapped cheek” rash implies the clearance of viremia through immune complex formation. Considering the adequate red cell lifespan of healthy subjects, no clinical anemia is usually observed. On the contrary, in patients with chronic hemolytic or sickle cell disease (where red cells' lifespan is shorter), B19V infection could present with aplastic crises, while pure red cell aplasia (PRCA) occurs in immunocompromised hosts (i.e. HIV/AIDS or after chemotherapy) [160].
Extra-hematological manifestations related to B19V infection have also been described in adults with arthropathy, glomerulonephritis, peripheral nervous system and muscle impairment [161].
-
2)
Mechanisms of fetal damage
Parvovirus B19 vertical transmission occurs in about 30% of cases of maternal infection [162].
Differentiating syncytiothrophoblast and cytotrophoblast cells have a key role in B19V vertical transmission, because of a strong expression of globoside-receptor on their surfaces. Although they are not permissive to viral infection, a receptor-mediated transcytosis process leads to release of the virus into fetal circulation [14]. Fetal capillary endothelium in placental villi could also be an additional target of B19V, leading to a structural and functional injury concurring to anomalous maternal-fetal blood exchanges and to the dissemination of the virus [162]. Furthermore, B19V infection of EPCs generates a DNA damage response by interfering with repair pathways and thus promoting viral DNA replication [163].
Once in the fetal circulation, the virus may then spread to fetal erythroid precursor cells (EPCs) in the liver and/or in bone marrow, and eventually cardiomyocytes, in the absence of an effective fetal immune response. The fetus is also more vulnerable considering that the half-life of its red blood cells is short (50–75 days) [164].
The induced cytotoxic effects and apoptotic mechanisms lead to an arrest of the marked erythropoiesis of the fetus, resulting in anemia, tissue hypoxia, myocarditis, cardiomegaly and pericardial effusion, and hydropic or non-hydropic Intra-Uterine Fetal Death (IUFD) [165].
The peak incidence of B19V-related fetal death occurs early during pregnancy (within the 20th week) [166], with a lower risk in the second half of pregnancy. On the contrary, fetal hydrops, defined as an accumulation of fluids in at least two fetal compartments (subcutaneous, pericardial, pleural, and abdominal), occurs when maternal infection happens later [165]. Hydrops may eventually lead to IUFD, but frequently the fetus can recover without a poor neurodevelopmental outcome. After birth, mortality associated with hydrops remains high despite advances in treatment, especially in preterm infants [167].
Thrombocytopenia may accompany severe anemia, as a result of megakaryocyte destruction: fetal hemorrhage was not seen as a complication of thrombocytopenia [168].
B19V seems to be not teratogenic. However, further studies are needed to conclude if other fetal abnormalities associated with B19V infection (meconium peritonitis, ocular injuries, brain lesions such as parenchymal calcifications, arterial infarction, cerebellar hemorrhage, hydrocephalus and polymicrogyria) are rare or not [3,164].
-
3)
Diagnosis of congenital infection
In all infants with fetal hydrops, a B19V infection should be always ruled out. Assessment of Parvovirus B19 infection in pregnant women relies firstly on serology through testing for both B19V specific IgG and IgM antibodies. In general, IgM to B19V appears 7 to 10 days after infection, while IgG appear a few days after IgM and then persist for months or even longer [169].
After a recent exposure, the presence of IgG and the absence of IgM suggest immunity, without any consequence in pregnancy. If IgM is present, a possible recent infection should be considered: an accurate counselling and serial ultrasounds should be performed, in order to rule out fetal hydrops and predict fetal anemia with Doppler measurement of the middle cerebral artery (MCA) peak systolic velocity [170].
However, a negative IgM result should also be cautiously interpreted, considering the risk of false-negative results or a future maternal seroconversion [171].
Furthermore, several commercial assays have been developed to detect specific B19V antibodies, not always with the same sensitivity and specificity [172].
Polymerase chain reaction (PCR) on plasma represent the standard method for the molecular detection of B19V DNA and may help to determine the stage of infection [172]: viremia occurs as early as 5 to 10 days after exposure, before any change in serology is detected [171]. Nonetheless, low viral loads may sometimes persist for a long time.
Invasive prenatal diagnosis of fetal infection involves the use of amniocentesis: the concentration of B19V DNA has been detected to be 100 to 5000 times higher in amniotic fluid than in maternal serum and corresponded to the matching fetal serum [173]. However, diagnosis is challenging because the presence of viral particles can only be found during the viremic stage. Furthermore, the detection of B19V IgM in fetal blood cannot be used to early diagnose a congenital infection, because the fetus does not begin to make its own IgM until 22 weeks of gestational age [174].
-
4)
Prevention strategies
Considering that B19V-related fetal damage is primarily due to fetal anemia, Intra-Uterine blood Transfusion (IUT) represents a life-saving procedure in prenatal management of the infection [175].
The overall survival rate in fetuses with severe anemia who required IUT is increasing, particularly I the absence of hydrops and if the first transfusion occurs after a gestational age of >22 weeks [176].
However, neurodevelopmental impairment among survivors may occur, even if adequately managed with IUT. Up to 26% survivors presented with prenatal abnormal cerebral lesions: an high viral load in fetal blood samples resulted as associated with brain lesions [177].
Mechanisms underlying this damage are still unknown: B19V DNA has been detected in several cerebral areas [178,179] but an hypoxic ischemic encephalopathy related to fetal anemia has been hypothesized [175].
To date, there is still a gap in the development of antiviral agents against B19V and although the relatively simple structure of its capsid, a vaccine is not available [180].
11. Non-polio enteroviruses
-
1)
Characteristics of the virus
Enterovirus (EV) is a genus of positive-sense single-stranded RNA viruses, belonging to Picornaviridae family. Historically, poliomyelitis was the most worrying disease caused by an enterovirus, namely poliovirus (types 1 to 3), become exceedingly rare in most developed countries as a result of routine immunization programs. There are 81 non-polio human enteroviruses (EVs): 22 Coxsackie A viruses (CV A1 to A21, A24), 6 Coxsackie B viruses (CV B1 to B6), 28 echoviruses (ECV 1-7, 9, 11-21, 24-27, 29-33), and 25 other enteroviruses (EV 68-71, 73-91, 100-101) [181].
EVs infections in the neonate are associated with a wide spectrum of signs and symptoms, which range from a non-specific febrile illness to potentially fatal multisystem disease, frequently referred to as ‘neonatal enterovirus sepsis’ or ‘enteroviral sepsis syndrome’ [182]. Common findings include fever or hypothermia, irritability, lethargy, anorexia, rash (“hand, foot and mouth disease”), jaundice, respiratory symptoms, apnea, hepatomegaly, abdominal distension, emesis, diarrhea, and decreased perfusion [183]. Most EVs cases have a benign course, whereas some have a severe presentation, including sepsis, meningoencephalitis, myocarditis, hepatitis, coagulopathy, and pneumonitis [183,184]. Neurotropism of EVs still represents a continuous threat to public health, with aseptic meningitis, encephalitis, acute flaccid paralysis [185].
-
2)
Mechanisms of fetal damage
Evidence suggests that EVs infections can be acquired antenatally, intrapartum and postnatally. In-utero transmission in late gestation has been demonstrated in animal models [186], despite the predominant mode of transmission of neonatal infection (63%) is intrapartum, at the time of delivery through contact with maternal blood, stool, amniotic fluid, or vaginal or cervical secretions [183]. Postnatal transmission after exposure to oral secretions of mother and other family members is relatively common. Liu et al. explained the role played by specific host cell surfaces receptors, called Coxsackie-Adenovirus Receptors (CARs), that seemed to be necessary for coxsackieviruses attachment and entry into cells, and accounts for organ and age-specific susceptibility to infection. It has been postulated that the lack of expression of these receptors in the placenta may help to prevent vertical transmission of the virus to the fetus [187]. The role of a co-factor such as Decay-accelerating factor molecule has been studied for interaction with CV-B3: this cofactor blocks the CAR binding site and causing the two receptors to complete themselves [188].
Regarding Enterovirus 71 (EV-A71) infection, Feng et al. demonstrated in a mouse model that the CNS damage in neonatal cases might be caused by an activated fetal cerebral immune response to the virus, including the disruption of brainstem function through increased levels of cytokines and neurotransmitters, rather than just a cytopathic effect of viral infection [189].
-
3)
Diagnosis of congenital infection
RT-PCR has been shown to be far more sensitive than cell culture for detection of EVs in clinical samples. Although specific IgM and IgG assays for EVs have been described and are available, their clinical utility is limited due to the cross-reactivity of the antigens used among the different serotypes [190]. Most EVs cases occur from 3rd to 5th day of life, suggesting that the timing is likely related to the immediate perinatal period. Risk factors include maternal illness within a week of delivery, with acute fever and severe lower abdominal pain, often misdiagnosed as placental abruption or appendicitis [182], prematurity and absence of specific antibodies to the infecting serotype in the neonate [183]. The timing of infection and the type of virus influence the clinical presentation, considering the typical tissue tropism of certain EV. Disseminated infections with CVs and ECVs could frequently involve the CNS [191], perhaps due to viremia. CVs and ECVs can also cause myocarditis, and therefore, intrauterine infection with enteroviruses should be considered in the differential diagnosis of non-immune hydrops fetalis [192].
-
4)
Prevention strategies
No specific intervention is available for pregnant women, but a history of relevant symptoms with fever and diarrhea may help clinicians to early identify the cause of severe neonatal infections.
There are still no available vaccines: potential vaccines still remain in the initial stages of development [193]. Recently, Wei et al. studied the transfer and decay of maternal antibodies against EV-A71: antibodies were efficiently transferred to neonates but declined quickly to below the protective threshold. Maternal vaccination could be explored to provide neonatal protection against EV-A71 through maternal antibodies, whereas catch-up vaccination between ages 6 months to 5 years could provide protection afterwards [194]. A phase III clinical trial has been planned to evaluate the efficacy, safety, and immunogenicity of a vaccine against EV-A71 at this age, but it is still not recruiting (ClinicalTrials.gov number NCT03865238).
12. Severe acute respiratory syndrome coronavirus 2
-
1)
Characteristics of the virus
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the virus that causes coronavirus disease 2019 (COVID-19), the respiratory illness responsible for the ongoing pandemic. It is a newly discovered β-Coronavirus with a positive-sense single-stranded RNA virus, belonging to Coronaviridae family. The first step in SARS-CoV-2 infection is the invasion of a host cell, a process that is mediated by the spike (S) glycoprotein. SARS-CoV-2 structural proteins also include the nucleocapsid protein (N), the membrane protein (M) and the envelope protein (E) [195].
This novel virus has a strong capacity to spread, mainly via droplets and aerosols during close unprotected contacts, with an incubation period ranging from 2 to 14 days. It can be easily detected by Real Time Polymerase Chain Reaction (RT-PCR) in nasopharyngeal swabs [196].
Most infections are uncomplicated, but some patients could require admission to intensive care unit because of respiratory and multiorgan failure. Risk factors for complicated disease are advanced age (>50 years), male sex and comorbidities (especially hypertension, diabetes, cardiovascular disease, chronic pulmonary disease, acute kidney injury) [197]. Neonatal and pediatric cases are principally family cluster cases [198]; most of them have epidemiological links to adult subjects, and show only milder clinical symptoms [199].
-
2)
Mechanisms of fetal damage
The SARS-CoV-2 virus links itself through its spike protein to the receptor of the angiotensin-2 converting enzyme (ACE2), that is expressed on the membrane of many host cells, including placental ones. It is widely expressed, in particular on syncytiothrophoblasts, hypothetically being able to contribute to the transplacental transmission of the virus to the fetus during maternal infection [200]. In addition, the co-expression of transmembrane protease serine 2 (TMPRSS2) can foster the entry by stimulating the fusion of viral particles with the host cell-membrane and related viral replication [201]. Similar to other RNA viral infections in pregnancy, the presence of placental lesions due to SARS-CoV-2 infection has been described by a large study, with fetal and/or maternal vascular malperfusion and inflammation signs, although all infants of infected mothers tested negative on RT-PCR and were asymptomatic [202]. Furthermore, the virus can be present in the placenta with a viral load at least two-fold higher that of the maternal blood and nasopharynx, increasing the eventual risk of intrapartum transmission [203].
The theoretical risk of vertical transmission has therefore a biological plausibility, but a caution is recommended in the interpretation of data, due to lack of collection of appropriate specimens from all tissues of both mother and infant at appropriate times and data still related more often to case reports and series rather than to large multicenter studies.
Most cases are related to infection contracted in the third trimester or in the peripartum, when maturation of most organs is already completed, whereas no cases of infections contracted early in pregnancy with confirmed teratogenic effects have been reported [204]. Overall rate of malformations in women with SARS-CoV-2 infection resulted to be similar to non-infected women, according to data from a Spanish multicenter study [205].
In a systematic review about effect of COVID-19 on pregnancy and perinatal outcomes, only four cases of spontaneous miscarriage or abortion (1.4%) were reported [206].
Transcriptome models showed that ACE2 expression changes over time in placental, fetal, and neonatal tissues increasing between the end of gestation and the first days of postnatal life [207]. This would justify the lack of reports describing negative outcomes of maternal infection in the early stages of gestation, although we cannot yet exclude that they exist. However, it would be argued that transplacental transmission is possible or more likely in the last weeks of pregnancy. Vivanti's recent report seems to have demonstrated neonatal viremia, following placental infection: the placenta showed signs of acute and chronic intervillous inflammation consistent with the severe systemic maternal inflammatory status triggered by SARS-CoV-2 infection. Both maternal and neonatal blood samples were positive, and the newborn presented symptoms similar to infected adults [208].
-
3)
Diagnosis of congenital infection
In a recent systematic review and meta-analysis of 176 published cases of neonatal SARS-CoV-2 infections, only 5.7% of cases were classified as confirmed congenital infections [209].
Recently, a panel of the World Health Organization (WHO) including experts of all involved fields proposed a consensus about classification system of vertical transmission of SARS-CoV-2, to allow comparison of data from different studies and better understand clinical consequences for the neonates born to infected mothers [210].
Firstly, they classified the timing of vertical transmission (in-utero, intrapartum and early postnatal) in mutually exclusive categories, as follows: (a) confirmed; (b) possible (evidence is suggestive but not confirmatory for infection); (c) unlikely (little support for diagnosis but infection cannot be completely ruled out); and (d) indeterminate (when tests required to define classification have not been performed). An in-utero infection can be defined as confirmed if there is 1) “evidence of maternal infection” anytime during pregnancy and 2) “in-utero fetal exposure” (when at least one neonatal sample tests positive for SARS-CoV-2 within 24 h of life) and 3) “SARS-CoV-2 persistence or immune response in the neonate” (at least one neonatal sample test positive again at 24–48 h of life).
The presence of the criterion of viral persistence/immune response in the neonate after 24 h of life is fundamental because a single positive RT-PCR early obtained on a neonatal respiratory sample may indicate either active viral replication, viral fragments intrapartum or postnatally acquired, or just a contamination.
Conversely, most of neonatal SARS-CoV-2 cases are postnatally acquired: infants may be exposed to the virus from their mother, healthcare workers or other family members, making the source of infection difficult to assess. Despite the finding of viral particle in human milk, breastfeeding resulted as not associated with SARS-CoV-2 infection [209] and this suggests that viral transmission through the milk, if any, should be rare. Therefore, mothers with suspected or confirmed COVID-19, if they are in good clinical conditions, can breastfeed their babies, by mandatorily applying all correct hygiene rules [211].
-
4)
Prevention strategies
At time of writing, available vaccines have given us a first hope, but the fight against COVID-19 is not over yet. The emergence of new variants with a higher spreading capability (such as B.1.1.7 from United Kingdom, B.1.351 from South Africa, P.1 from Brazil and B.1.617 from India) is an expected occurrence, considering that RNA viruses usually have an higher incidence of mutations than DNA viruses [212]. This emphasizes how international strategies of surveillance and tracking could tackle this moving target [213].
Everyday preventive actions to reduce the spread of COVID-19, including social distancing, protective masks, and hands hygiene still remain key elements of this struggle.
Our hope is that always more vaccine doses would be available and administered as soon as possible; furthermore, we hope that current vaccines could provide an effective prolonged protection, beyond a few changes at antigenic sites in SARS-CoV-2 new variants.
Data on the safety of COVID-19 vaccines in pregnant women and during breastfeeding are still limited. However, many pregnant women have decided to accept to be vaccinated [214]. By monitoring the outcomes for these women and their infants, first data reported a maternal immune response and transfer of maternal antibodies to confer passive immunity against SARS-CoV-2 in neonates after maternal vaccination with mRNA-based vaccines (BNT162b2 Pfizer/BioNTech or mRNA-1273 Moderna/NIH) [215,216].
13. ZIKA virus
-
1)
Characteristics of the virus
Zika virus (ZIKV) is a member of the genus Flavivirus (within the family of Flaviviridae), related to dengue virus (DENV), yellow fever virus (YFV), Japanese encephalitis virus (JEV), Chikungunya virus (CHIKV) and West Nile virus (WNV). It is a enveloped and icosahedral virus that contains a single-stranded, positive-sense RNA; it is spread by daytime-active Aedes mosquitoes (such as A. aegypti and A. albopictus), sexual contacts, blood transfusions, and vertical transmission [217]. Two receptors, AXL tyrosine-protein kinase receptor and T-cell immunoglobulin and mucin domain 1 (TIM1) receptor, were suggested as candidates for ZIKV entry in vitro, while their role in vivo is still to fully explain [218].
In most of the cases, ZIKV infection is asymptomatic (up to 80%) [219]; after an incubation period of 3 to 14 days, sometimes patients experience a maculopapular rash, low-grave fever, myalgia/arthralgia, headache, and conjunctivitis [220]. In adults, ZIKV infection has been associated with neurological disorders (such as Guillain-Barré syndrome and acute myelitis), ocular disorders (hypertensive iridocyclitis, maculopathy, uveitis), thrombocytopenic purpura and transient myocarditis [221].
Conversely, the 2015–16 outbreak of Zika virus brought the world's attention to links between ZIKV infection and microcephaly in fetuses and neonates born to women infected during pregnancy [222].
Among exposed fetuses, fetal loss occurred in 14% and severe complications compatible with Congenital Zika Syndrome (CZS) occurred in 21% [221]. To date, although ZIKV particles have been detected in human milk, breastfeeding has not been confirmed as a transmission route and mothers with possible or confirmed ZIKV infection are recommended to keep breastfeeding their babies [223].
-
2)
Mechanisms of fetal damage
Maternal ZIKV exposure induces to a diffuse placental injury, with trophoblast hyperplasia, focal regions of necrosis, and loss of embryonic blood vessels: these changes probably promote adverse fetal outcomes, even in the absence of fetal infection [224]. An increase in the number of macrophages and Hofbauer cells has been observed, promoting the production of matrix metalloproteinases that degrade the collagen (as well as TNF-α) and activate immune cell trafficking. CD68+ and T CD8+ cells were largely detected in infected placentae, with an high expression of cytokines (IFN-γ and TNF-α) and other immunological mediators (RANTES/CCL5 and VEGFR-2) that confirmed excessive inflammation and vascular permeability dysfunction [225]. Furthermore, in the presence of ZIKV infection, Bcl-2 protein has been showed to be overexpressed in the third trimester syncytiothrophoblast cells, leading to a higher cellular apoptosis and persistence of viral particles in placenta [226].
After breaching the maternal-fetal interface, the virus reaches the developing brain by hematogenous route or via the cerebrospinal fluid, where increased specific immunoglobulins levels have been recently demonstrated in neonates exposed to Zika during fetal life [227].
AXL receptor seems to be the primary ZIKV entry cofactor on human umbilical vein endothelial cells (HUVECs), allowing to the virus to enter the fetal bloodstream to gain access to other fetal tissues [228]. However, other receptors may also contribute to the entry of the virus, considering that AXL resulted to be not fundamental for vertical transmission in mice [229].
AXL mRNA has also been found in human neural progenitor cells (NPCs) and other brain cells (such as radial glial cells, microglial cells and astrocytes), suggesting a susceptibility of these cells and explaining related brain abnormalities [230].
Pro-inflammatory state in the fetal brain has been recently confirmed by increased immunoglobulins levels in cerebrospinal fluid in neonates.
-
3)
Diagnosis of congenital infection
Pregnant women with ZIKV infection should be serially screened, although prenatal ultrasound examination cannot always detect CZS abnormalities [231]. Specific IgM can be detected in the mother as early as 4–5 days post-infections and for up to 12 weeks; after exposure, a negative IgM ELISA test is strongly related to the absence of a recent infection [232]. While amniocentesis is useful for diagnosing other fetal viral infection, the need of testing amniotic fluid to detect ZIKV RNA is still debated [233].
Key features of CZS include both structural lesions and functional anomalies. A fetal brain disruption sequence is typically observed, including severe microcephaly, premature closure of the fontanels and partial collapse of the skull. Brain abnormalities include cortical thinning, hypoplasia of the cerebellum vermis, dysgenesis of the corpus callosum and gyral anomalies, linear calcifications (mainly subcortical), ventriculomegaly and increased pericerebral spaces [221]. Ocular abnormalities should also be ruled out: posterior pole focal pigment mottling of retina, chorioretinal atrophy in the macular area, optic nerve abnormalities (such as optic nerve hypoplasia and severe optic disc cupping) were the prevalent lesions observed in Brazil [234]. A typical “femur-sparing” profile of intrauterine growth restriction (IUGR) could be observed in infected fetus at ultrasound scans, with an arrest of fetal head growth while the long bones (i.e. femur) continue to grow normally [235].
The absence of clinical and neuroradiological CZS abnormalities at birth does not exclude future functional implications for both the affected infant as well as its family, such as seizures, hearing loss, abnormal visual function, dysphagia, sleep disorders, and neurodevelopmental impairment [236,237].
-
4)
Prevention strategies
There is currently no licensed vaccine or specific drug to prevent or treat ZIKV infection. Primary prevention should focus on educating pregnant women. All non-essential travels to destinations with risk of Zika should be avoided. If travels could not be avoided or if women live in at-risk areas, appropriate protective measures must be taken. Mosquito bites could be prevented by covering exposed skin and using appropriate insect repellents, such as picaridin or diethyltoluamide (DEET). It is better to stay in air-conditioned rooms and sleep under a mosquito bed net. Women who traveled in high-risk areas should wait at least 2 months before trying to become pregnant, regardless of symptoms [238]. Men should wait at least 6 months, due to the persistence of viral particles in male reproductive organs [232]. Conversely, condoms should be used to avoid getting ZIV through sexual contacts, if partners have recently traveled to a Zika-affected area, although is an unpopular choice in stable couples [239].
14. Conclusion
The placenta is a immune-tolerant environment, in continuous development during gestation that allows viruses to replicate. The infectious power of viruses varies in relation to the characteristics of the developmental stage of the placenta, generating fetal damage of different severity or no damage. The mechanisms of this placental barrier function are not fully understood, for both known and emerging viruses. Oxygen levels certainly constitute an up-regulation factor of adhesion receptors, which are cofactors of infection. The vascular bed is forming, and the development of new vessels allows lymphocytes to enter, carrying viruses. Furthermore, the placental is a low resistance vascular area and this facilitates the entry of viruses into the intervillous space.
The problem is complex, but the greater the knowledge, the more effect the prevention strategies of neonatal damage will have, as has been shown by the history of HIV, the better the weapons will be to face the challenges of new viruses, whose probability of emergence will be proportional to the climatic damages, that we will bring to the environment around us.
CRediT authorship contribution statement
CA and DUDR drafted the section on SARS-CoV-2 and ZIKV; DUDR drafted the section on RuV, HIV and B19V; DUDR and AS drafted the section on HBV and HCV; FS drafted the section on VZV; IB drafted the section on CMV; IB and LM drafted the section on HSV; DUDR and MPR drafted the section on HHV-6; MPR drafted the section on EV.
CA integrated all authors' contributions: the draft was then reviewed and critically revised by all authors.
Funding
None.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Colugnati F.A., Staras S.A., Dollard S.C., Cannon M.J. Incidence of cytomegalovirus infection among the general population and pregnant women in the United States. BMC Infect. Dis. 2007;7:1–10. doi: 10.1186/1471-2334-7-71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dreher A.M., Arora N., Fowler K.B., Novak Z., Britt W.J., Boppana S.B., et al. Spectrum of disease and outcome in children with symptomatic congenital Cytomegalovirus infection. J. Pediatr. 2014;164:855–859. doi: 10.1016/j.jpeds.2013.12.007.Spectrum. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.de Vries L.S. Viral infections and the neonatal brain. Semin. Pediatr. Neurol. 2020;32 doi: 10.1016/j.spen.2019.08.005. [DOI] [PubMed] [Google Scholar]
- 4.Naing Z.W., Scott G.M., Shand A., Hamilton S.T., van Zuylen W.J., Basha J., et al. Congenital cytomegalovirus infection in pregnancy: a review of prevalence, clinical features, diagnosis and prevention. Aust. New Zeal. J. Obstet. Gynaecol. 2016;56:9–18. doi: 10.1111/ajo.12408. [DOI] [PubMed] [Google Scholar]
- 5.Britt W. Controversies in the natural history of congenital human cytomegalovirus infection: the paradox of infection and disease in offspring of women with immunity prior to pregnancy. Med. Microbiol. Immunol. 2015;204:263–271. doi: 10.1007/s00430-015-0399-9. [DOI] [PubMed] [Google Scholar]
- 6.Britt W.J. Maternal immunity and the natural history of congenital human Cytomegalovirus infection. Viruses. 2018;10:405. doi: 10.3390/v10080405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Enders G., Daiminger A., Bäder U., Exler S., Enders M. Intrauterine transmission and clinical outcome of 248 pregnancies with primary cytomegalovirus infection in relation to gestational age. J. Clin. Virol. 2011;52:244–246. doi: 10.1016/j.jcv.2011.07.005. [DOI] [PubMed] [Google Scholar]
- 8.Lim Y., Lyall H. Congenital cytomegalovirus − who, when, what-with and why to treat. J. Inf. Secur. 2017;74:S89–S94. doi: 10.1016/S0163-4453(17)30197-4. [DOI] [PubMed] [Google Scholar]
- 9.Boppana S.B., Fowler K.B., Pass R.F., Rivera L.B., Bradford R.D., Lakeman F.D., et al. Congenital cytomegalovirus infection: association between virus burden in infancy and hearing loss. J. Pediatr. 2005;146:817–823. doi: 10.1016/j.jpeds.2005.01.059. [DOI] [PubMed] [Google Scholar]
- 10.Cheeran M.C., Lokensgard J.R., Schleiss M.R. Neuropathogenesis of congenital Cytomegalovirus infection: disease mechanisms and prospects for intervention. Clin. Microbiol. Rev. 2009;22:99–126. doi: 10.1128/CMR.00023-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pereira L., Petitt M., Tabata T. Cytomegalovirus infection and antibody protection of the developing placenta. Clin. Infect. Dis. 2013;57:174–177. doi: 10.1093/cid/cit583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Adler S.P., Nigro G., Pereira L. Recent advances in the prevention and treatment of congenital Cytomegalovirus infections. Semin. Perinatol. 2007;31:10–18. doi: 10.1053/j.semperi.2007.01.002. [DOI] [PubMed] [Google Scholar]
- 13.Tabata T., Petitt M., Zydek M., Fang-Hoover J., Larocque N., Tsuge M., et al. Human Cytomegalovirus infection interferes with the maintenance and differentiation of trophoblast progenitor cells of the human. J. Virol. 2015;89:5134–5147. doi: 10.1128/JVI.03674-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pereira L. Congenital viral infection: traversing the uterine-placental Interface. Annu. Rev. Virol. 2018;5:273–299. doi: 10.1146/annurev-virology-092917-043236. [DOI] [PubMed] [Google Scholar]
- 15.Weisblum Y., Panet A., Haimov-Kochman R., Wolf D.G. Models of vertical cytomegalovirus (CMV) transmission and pathogenesis. Semin. Immunopathol. 2014;36:615–625. doi: 10.1007/s00281-014-0449-1. [DOI] [PubMed] [Google Scholar]
- 16.Fisher S., Genbacev O., Maidji E., Pereira L. Human Cytomegalovirus infection of placental cytotrophoblasts in vitro and in utero: implications for transmission and pathogenesis. J. Virol. 2000;74:6808–6820. doi: 10.1128/jvi.74.15.6808-6820.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schleiss M.R., Aronow B.J., Handwerger S. Cytomegalovirus infection of human syncytiotrophoblast cells strongly interferes with expression of genes involved in placental differentiation and tissue integrity. Pediatr. Res. 2007;61:565–571. doi: 10.1203/pdr.0b013e318045be6d. [DOI] [PubMed] [Google Scholar]
- 18.Tabata T., McDonagh S., Kawakatsu H., Pereira L. Cytotrophoblasts infected with a pathogenic human Cytomegalovirus strain dysregulate cell-matrix and cell-cell adhesion molecules: a quantitative analysis. Placenta. 2007;28:527–537. doi: 10.1016/j.placenta.2006.05.006. [DOI] [PubMed] [Google Scholar]
- 19.Yamamoto-Tabata T., McConagh S., Chang H., Fisher S., Pereira L. Human Cytomegalovirus Interleukin-10 downregulates metalloproteinase activity and impairs endothelial cell migration and placental Cytotrophoblast invasiveness in vitro. J. Virol. 2004;78:2831–2840. doi: 10.1128/JVI.78.6.2831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rauwel B., Mariamé B., Martin H., Nielsen R., Allart S., Pipy B., et al. Activation of peroxisome proliferator-activated receptor gamma by human Cytomegalovirus for De Novo replication impairs migration and invasiveness of cytotrophoblasts from early placentas. J. Virol. 2010;84:2946–2954. doi: 10.1128/JVI.01779-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fournier T., Guibourdenche J., Handschuh K., Tsatsaris V., Rauwel B., Davrinche C., et al. PPARγ and human trophoblast differentiation. J. Reprod. Immunol. 2011;90:41–49. doi: 10.1016/j.jri.2011.05.003. [DOI] [PubMed] [Google Scholar]
- 22.Fisher S.J. Why is placentation abnormal in preeclampsia? Am. J. Obstet. Gynecol. 2015;213:S115–S122. doi: 10.1016/j.ajog.2015.08.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pereira L., Maidji E., McDonagh S., Genbacev O., Fisher S. Human Cytomegalovirus transmission from the uterus to the placenta correlates with the presence of pathogenic bacteria and maternal immunity. J. Virol. 2003;77:13301–13314. doi: 10.1128/JVI.77.24.13301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.La Torre R, La Torre R., Nigro G., Mazzocco M., Best A.M., Adler S.P. Placental enlargement in women with primary maternal Cytomegalovirus infection is associated with fetal and neonatal disease. Clin. Infect. Dis. 2006;43:994–1000. doi: 10.1086/507634. [DOI] [PubMed] [Google Scholar]
- 25.Walker S.P., Palma-Dias R., Wood E.M., Shekleton P., Giles M.L. Cytomegalovirus in pregnancy: to screen or not to screen. BMC Pregnancy Childbirth. 2013;13:1–8. doi: 10.1186/1471-2393-13-96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lazzarotto T., Gabrielli L., Lanari M., Guerra B., Bellucci T., Sassi M., et al. Congenital Cytomegalovirus infection: recent advances in the diagnosis of maternal infection. Hum. Immunol. 2004;65:410–415. doi: 10.1016/j.humimm.2004.02.006. [DOI] [PubMed] [Google Scholar]
- 27.Tsuge M., Hida A.I., Minematsu T., Honda N., Oshiro Y., Yokoyama M., et al. Prospective cohort study of congenital Cytomegalovirus infection during pregnancy with fetal growth restriction: serologic analysis and placental pathology. J. Pediatr. 2019;206 doi: 10.1016/j.jpeds.2018.10.003. (42-48.e2) [DOI] [PubMed] [Google Scholar]
- 28.Rawlinson W.D., Boppana S.B., Fowler K.B., Kimberlin D.W., Lazzarotto T., Alain S., et al. Congenital cytomegalovirus infection in pregnancy and the neonate: consensus recommendations for prevention, diagnosis, and therapy. Lancet Infect. Dis. 2017;17:1–12. doi: 10.1016/S1473-3099(17)30143-3. [DOI] [PubMed] [Google Scholar]
- 29.Barkai G., Ari-Even Roth D., Barzilai A., Tepperberg-Oikawa M., Mendelson E., Hildesheimer M., et al. Universal neonatal cytomegalovirus screening using saliva – report of clinical experience. J. Clin. Virol. 2014;60:361–366. doi: 10.1016/j.jcv.2014.04.024. [DOI] [PubMed] [Google Scholar]
- 30.Yamada H., Tanimura K., Fukushima S., Fujioka K., Deguchi M., Sasagawa Y., et al. A cohort study of the universal neonatal urine screening for congenital cytomegalovirus infection. J. Infect. Chemother. 2020;26:790–794. doi: 10.1016/j.jiac.2020.03.009. [DOI] [PubMed] [Google Scholar]
- 31.Pellegrinelli L., Alberti L., Pariani E., Barbi M., Binda S. Diagnosing congenital Cytomegalovirus infection: don’t get rid of dried blood spots. BMC Infect. Dis. 2020;20:1–5. doi: 10.1186/s12879-020-4941-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Revello M.G., Lazzarotto T., Guerra B., Spinillo A., Ferrazzi E., Kustermann A., et al. A randomized trial of Hyperimmune globulin to prevent congenital Cytomegalovirus. N. Engl. J. Med. 2014;370:1316–1326. doi: 10.1056/NEJMoa1310214. [DOI] [PubMed] [Google Scholar]
- 33.Kagan K.O., Enders M., Schampera M.S., Baeumel E., Hoopmann M., Geipel A., et al. Prevention of maternal-fetal transmission of cytomegalovirus after primary maternal infection in the first trimester by biweekly hyperimmunoglobulin administration. Ultrasound Obstet. Gynecol. 2019;53:383–389. doi: 10.1002/uog.19164. [DOI] [PubMed] [Google Scholar]
- 34.Hamilton S.T., van Zuylen W., Shand A., Scott G.M., Naing Z., Hall B., et al. Prevention of congenital cytomegalovirus complications by maternal and neonatal treatments: a systematic review. Rev. Med. Virol. 2014;24:420–433. doi: 10.1002/rmv. [DOI] [PubMed] [Google Scholar]
- 35.Shahar-Nissan K., Pardo J., Peled O., Krause I., Bilavsky E., Wiznitzer A., et al. Valaciclovir to prevent vertical transmission of cytomegalovirus after maternal primary infection during pregnancy: a randomised, double-blind, placebo-controlled trial. Lancet. 2020;396:779–785. doi: 10.1016/S0140-6736(20)31868-7. [DOI] [PubMed] [Google Scholar]
- 36.Kriebs J.M. Understanding herpes simplex virus: transmission, diagnosis, and considerations in pregnancy management. J. Midwifery Womens Health. 2008;53:202–208. doi: 10.1016/j.jmwh.2008.01.010. [DOI] [PubMed] [Google Scholar]
- 37.Bradley H., Markowitz L.E., Gibson T., McQuillan G.M. Seroprevalence of herpes simplex virus types 1 and 2 — United States, 1999 – 2010. J. Infect. Dis. 2014;209:325–333. doi: 10.1093/infdis/jit458. [DOI] [PubMed] [Google Scholar]
- 38.Pinninti S.G., Kimberlin D.W. Preventing herpes simplex virus in the newborn. Clin. Perinatol. 2014;41:945–955. doi: 10.1016/j.clp.2014.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pinninti S.G., Kimberlin D.W. Neonatal herpes simplex virus infections. Semin. Perinatol. 2018;42:168–175. doi: 10.1053/j.semperi.2018.02.004. [DOI] [PubMed] [Google Scholar]
- 40.Veal G.J., Errington J., Hayden J., Hobin D., Murphy D., Dommett R.M., et al. Carboplatin therapeutic monitoring in preterm and full-term neonates. Eur. J. Cancer. 2015;51:2022–2030. doi: 10.1016/j.ejca.2015.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Shi T., Huang L., Xiong Y., Zhong Y., Yang J., Fu T., et al. The risk of Herpes Simplex Virus and human Cytomegalovirus infection during pregnancy upon adverse pregnancy outcomes: A meta-analysis. J. Clin. Virol. 2018;104:48–55. doi: 10.1016/j.jcv.2018.04.016. [DOI] [PubMed] [Google Scholar]
- 42.James S.H., Kimberlin D.W. Neonatal herpes simplex virus infection - epidemiology and treatment. Clin. Perinatol. 2015;42:47–59. doi: 10.1016/j.clp.2014.10.005. [DOI] [PubMed] [Google Scholar]
- 43.Johnston C., Corey L. Current concepts for genital herpes simplex virus infection: diagnostics and pathogenesis of genital tract shedding. Clin. Microbiol. Rev. 2016;29:149–161. doi: 10.1128/CMR.00043-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.James S.H., Kimberlin D.W. Neonatal herpes simplex virus infection. Infect. Dis. Clin. N. Am. 2015;29:391–400. doi: 10.1016/j.idc.2015.05.001. [DOI] [PubMed] [Google Scholar]
- 45.Akhtar J., Shukla D. Viral entry mechanisms: cellular and viral mediators of herpes simplex virus entry. FEBS J. 2009;276:7228–7236. doi: 10.1111/j.1742-4658.2009.07402.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Brown Z.A., Wald A., Morrow R.A., Selke S., Zeh J., Corey L. Effect of serologic status and cesarean delivery on transmission rates of herpes simplex virus from mother to infant. JAMA. 2003;289:203–209. doi: 10.1001/jama.289.2.203. [DOI] [PubMed] [Google Scholar]
- 47.Finger-Jardim F., Avila E.C., Vanusa P., Gonçalves C V., de Martinez A.M.B., Soares M.A. Prevalence of herpes simplex virus types 1 and 2 at maternal and fetal sides of the placenta in asymptomatic pregnant women. Am. J. Reprod. Immunol. 2017;78:1–12. doi: 10.1111/aji.12689. [DOI] [PubMed] [Google Scholar]
- 48.Neuberger I., Garcia J., Meyers M.L., Feygin T., Bulas D.I., Mirsky D.M. Imaging of congenital central nervous system infections. Pediatr. Radiol. 2018;48:513–523. doi: 10.1007/s00247-018-4092-1. [DOI] [PubMed] [Google Scholar]
- 49.Silasi M., Cardenas I., Racicot K., Kwon J.-Y., Aldo P., Mor G. Viral infections during pregnancy. Am. J. Reprod. Immunol. 2015;73:199–213. doi: 10.1111/aji.12355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kimberlin D.W., Lin C.-Y., Jacobs R.F., Powell D.A., Frenkel L.M., Gruber W.C., et al. Natural history of neonatal herpes simplex virus infections in the acyclovir era. Pediatrics. 2001;108:223–229. doi: 10.1542/peds.108.2.223. [DOI] [PubMed] [Google Scholar]
- 51.Melvin A.J., Mohan K.M., Schiffer J.T., Drolette L.M., Magaret A., Corey L., et al. Plasma and cerebrospinal fluid herpes simplex virus levels at diagnosis and outcome of neonatal infection. J. Pediatr. 2015;166:827–833. doi: 10.1016/j.jpeds.2014.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.James S.H., Kimberlin D.W. Quantitative herpes simplex virus concentrations in neonatal infection. J. Pediatr. 2015;166:793–795. doi: 10.1016/j.jpeds.2014.12.077. [DOI] [PubMed] [Google Scholar]
- 53.Anzivino E., Fioriti D., Mischitelli M., Bellizzi A., Barucca V., Chiarini F., et al. Herpes simplex virus infection in pregnancy and in neonate: status of art of epidemiology, diagnosis, therapy and prevention. Virol. J. 2009;6:1–11. doi: 10.1186/1743-422X-6-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Roberts S. Herpes simplex virus: incidence of neonatal herpes simplex virus, maternal screening, management during pregnancy, and HIV. Curr. Opin. Obstet. Gynecol. 2009;21:124–130. doi: 10.1097/GCO.0b013e3283294840. [DOI] [PubMed] [Google Scholar]
- 55.American College of Obstetricians and Gynecologists Management of herpes in pregnancy. ACOG practice bulletin no. 82. Obstet. Gynecol. 2007;109:1489–1498. doi: 10.1097/01.aog.0000263902.31953.3e. [DOI] [PubMed] [Google Scholar]
- 56.Corey L., Wald A. Maternal and neonatal herpes simplex virus infections. N. Engl. J. Med. 2009;361:1376–1385. doi: 10.1056/NEJMra0807633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Agut H., Bonnafous P., Gautheret-Dejean A. Human Herpesviruses 6A, 6B, and 7. Microbiol. Spectr. 2015;4:1–18. doi: 10.1128/microbiolspec.DMIH2-0007-2015. [DOI] [PubMed] [Google Scholar]
- 58.Ward K.N. Human herpesviruses-6 and -7 infections. Curr. Opin. Infect. Dis. 2005;18:247–252. doi: 10.1097/01.qco.0000168386.16519.93. [DOI] [PubMed] [Google Scholar]
- 59.Drago F., Ciccarese G., Herzum A., Rebora A., Parodi A. Pityriasis Rosea during pregnancy: major and minor alarming signs. Dermatology. 2018;234:31–36. doi: 10.1159/000489879. [DOI] [PubMed] [Google Scholar]
- 60.Kawaguchi S., Suga S., Kozawa T., Nakashima T., Yoshikawa T., Asano Y. Primary human herpesvirus-6 infection (exanthem subitum) in the newborn. Pediatrics. 1992;90:628–630. [PubMed] [Google Scholar]
- 61.Zerr D.M., Yeung L.C., Obrigewitch R.M., Huang M., Frenkel L.M., Corey L. Case report: primary human Herpesvirus-6 associated with an afebrile seizure in a 3-week-old infant. J. Med. Virol. 2002;66:384–387. doi: 10.1002/jmv.2156. [DOI] [PubMed] [Google Scholar]
- 62.Borghesi A., Cipelletti P., Maragliano R., Manzoni P., Stronati M. Human herpesvirus-6 associated neonatal urticaria multiforme. Arch. Dis. Child. Fetal Neonatal Ed. 2013;98:F450. doi: 10.1136/archdischild-2012-301995. [DOI] [PubMed] [Google Scholar]
- 63.Kim F., Reichman V., Hooven T.A. Human Herpesvirus-6 meningitis in a premature infant with fevers: a case and literature review. Clin. Med. Insights Case Reports. 2020;13:1–6. doi: 10.1177/1179547620912952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hall C.B., Caserta M.T., Schnabel K.C., Boettrich C., McDermott M.P., Lofthus G.K., et al. Congenital infections with human herpesvirus 6 (HHV6) and human herpesvirus 7 (HHV7) J. Pediatr. 2004;6:472–477. doi: 10.1016/j.jpeds.2004.06.017. [DOI] [PubMed] [Google Scholar]
- 65.Morissette G., Flamand L. Herpesviruses and chromosomal integration. J. Virol. 2010;84:12100–12109. doi: 10.1128/JVI.01169-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Hall C.B., Caserta M.T., Schnabel K., Shelley L.M., Marino S., Carnahan J.A., et al. Chromosomal integration of human herpesvirus 6 is the major mode of congenital human herpesvirus. Pediatrics. 2008;122:513–520. doi: 10.1542/peds.2007-2838. [DOI] [PubMed] [Google Scholar]
- 67.Hall C.B., Caserta M.T., Schnabel K.C., Shelley L.M., Carnahan J.A., Marino A.S., et al. Transplacental Human Herpesvirus 6 (HHV-6) congenital infection caused by maternal chromosomally integrated virus. J. Infect. Dis. 2010;201:505–507. doi: 10.1086/650495.Transplacental. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kaufer B.B., Flamand L. Impact on virus, cell and organismal biology. Curr. Opin. Virol. 2014;9:111–118. doi: 10.1016/j.coviro.2014.09.010. [DOI] [PubMed] [Google Scholar]
- 69.Gibson C., Goldwater P., MacLennan A., Haan E., Priest K., Dekker G., et al. Fetal exposure to herpesviruses may be associated with pregnancy-induced hypertensive disorders and preterm birth in a Caucasian population. BJOG An. Int. J. Obstet. Gynaecol. 2008;115:492–500. doi: 10.1111/j.1471-0528.2007.01653.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Rebora A., Ciccarese G., Herzum A., Parodi A., Drago F. Pityriasis rosea and other infectious eruptions during pregnancy: possible life-threatening health conditions for the fetus. Clin. Dermatol. 2020;38:105–112. doi: 10.1016/j.clindermatol.2019.10.020. [DOI] [PubMed] [Google Scholar]
- 71.Bortolotti D., Soffritti I., D’Accolti M., Gentili V., Di Luca D., Rizzo R., et al. HHV-6A infection of endometrial epithelial cells affects miRNA expression and trophoblast cell attachment. Reprod. Sci. 2020;27:779–786. doi: 10.1007/s43032-019-00102-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Miura H., Kawamura Y., Ohye T., Hattori F., Kozawa K., Ihira M., et al. Inherited chromosomally integrated human herpesvirus 6 is a risk factor for spontaneous abortion. J. Infect. Dis. 2020 Sep 28 doi: 10.1093/infdis/jiaa606. [DOI] [PubMed] [Google Scholar]
- 73.Dantuluri K.L., Konvinse K.C., Crook J., Thomsen I.P.T., Banerjee R. Human herpesvirus 6 detection during the evaluation of sepsis in infants using the FilmArray meningitis/encephalitis panel. J. Pediatr. 2020 Aug doi: 10.1016/j.jpeds.2020.03.023. (204-206.e1) [DOI] [PubMed] [Google Scholar]
- 74.Sugimoto T., Tanaka-taya K., Ono J., Miyoshi H., Okada S., Yamanishi K. Human herpesvirus-6 infection in neonates: not protected by only humoral immunity. Pediatr. Int. 2002;44:281–285. doi: 10.1046/j.1442-200x.2002.01562.x. [DOI] [PubMed] [Google Scholar]
- 75.Kennedy P.G.E., Gershon A.A. Clinical features of varicella-zoster virus infection. Viruses. 2018;10:1–11. doi: 10.3390/v10110609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Gilden D., Cohrs R.J., Mahalingam R., Nagel M.A. Neurological disease produced by varicella zoster virus reactivation without rash. Curr. Top. Microbiol. Immunol. 2010;342:243–253. doi: 10.1007/82_2009_3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Arvin A.M., Gilden D. Fields Virol. 7th. Vol. 2. Lippincott Williams & Wilkins Press; Philadelphia, PA: 2013. Varicella-zoster virus; pp. 2015–2057. [Google Scholar]
- 78.Auriti C., Piersigilli F., De Gasperis M.R., Seganti G. Congenital varicella syndrome: still a problem? Fetal Diagn. Ther. 2009;25:224–229. doi: 10.1159/000220602. [DOI] [PubMed] [Google Scholar]
- 79.Tan M.P., Koren G. Chickenpox in pregnancy: revisited. Reprod. Toxicol. 2006;21:410–420. doi: 10.1016/j.reprotox.2005.04.011. [DOI] [PubMed] [Google Scholar]
- 80.Nikkels A.F., Delbecque K., Pierard G.E., Wienkotter B., Schalasta G., Enders M. Distribution of varicella-zoster virus DNA and gene products in tissues of a first-trimester varicella-infected fetus. J. Infect. Dis. 2005;191:540–545. doi: 10.1086/426942. [DOI] [PubMed] [Google Scholar]
- 81.Smith C.K., Arvin A.M. Varicella in the fetus and newborn. Semin. Fetal Neonatal Med. 2009;14:209–217. doi: 10.1016/j.siny.2008.11.008. [DOI] [PubMed] [Google Scholar]
- 82.Lamont R.F., Sobel J.D., Carrington D., Mazaki-Tovi S., Kusanovic J.P., Vaisbuch E., et al. Varicella-zoster virus (chickenpox) infection in pregnancy. BJOG An. Int. J. Obstet. Gynaecol. 2011;118:1155–1162. doi: 10.1111/j.1471-0528.2011.02983.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Gaymard A., Pichon M., Bal A., Massoud M., Buenerd A., Massardier J., et al. How to manage chickenpox during pregnancy: case reports. Ann. Biol. Clin. 2018;76:669–674. doi: 10.1684/abc.2018.1385. [DOI] [PubMed] [Google Scholar]
- 84.Mattson S.N., Jones K.L., Gramling L.J., Schonfeld A.M., Riley E.P., Harris J.A., et al. Neurodevelopmental follow-up of children of women infected with varicella during pregnancy: a prospective study. Pediatr. Infect. Dis. J. 2003;22:819–823. doi: 10.1097/01.inf.0000086402.18710.54. [DOI] [PubMed] [Google Scholar]
- 85.Centers for Disease Control and Prevention (CDC). Updated recommendations for use of VariZIG - United States, 2013. MMWR Recomm. Rep. 2013;62:574–576. [PMC free article] [PubMed] [Google Scholar]
- 86.Morgan-Capner P., Crowcroft N. Guidelines on the management of, and exposure to, rash illness in pregnancy (including consideration of relevant antibody screening programmes in pregnancy) Commun. Dis. Public Health. 2002;5:59–71. [PubMed] [Google Scholar]
- 87.Muzumdar S., Rothe M.J., Grant-Kels J.M. The rash with maculopapules and fever in adults. Clin. Dermatol. 2019;37:109–118. doi: 10.1016/j.clindermatol.2018.12.004. [DOI] [PubMed] [Google Scholar]
- 88.Woyessa A.B., Ali M.S., Korkpor T.K., Ii R.T., Kohar H.T., Dogba J., et al. Rubella transmission and the risk of congenital rubella syndrome in Liberia: a need to introduce rubella-containing vaccine in the routine immunization program. BMC Infect. Dis. 2019;19:813. doi: 10.1186/s12879-019-4464-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Banatvala J., Brown D. Rubella. Lancet. 2004;363:1127–1137. doi: 10.1016/S0140-6736(04)15897-2. [DOI] [PubMed] [Google Scholar]
- 90.Lazar M., Perelygina L., Martines R., Greer P., Paddock C.D., Peltecu G., et al. Immunolocalization and distribution of rubella antigen in fatal congenital rubella syndrome. EBioMedicine. 2016;3:86–92. doi: 10.1016/j.ebiom.2015.11.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.George S., Viswanathan R., Sapkal G.N. Molecular aspects of the teratogenesis of rubella virus. Biol. Res. 2019;52:1–8. doi: 10.1186/s40659-019-0254-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Adamo M.P., Zapata M., Frey T.K. Analysis of gene expression in fetal and adult cells infected with rubella virus. Virology. 2008;370:1–11. doi: 10.1016/j.virol.2007.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Geyer H., Bauer M., Neumann J., Lüdde A., Rennert P., Friedrich N., et al. Gene expression profiling of rubella virus infected primary endothelial cells of fetal and adult origin. Virol. J. 2016;13:1–17. doi: 10.1186/s12985-016-0475-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Neu N., Duchon J., Zachariah P. TORCH infections. Clin. Perinatol. 2015;42:77–103. doi: 10.1016/j.clp.2014.11.001. [DOI] [PubMed] [Google Scholar]
- 95.Yazigi A., De Pecoulas A.E., Vauloup-Fellous C., Grangeot-Keros L., Ayoubi J.-M., Picone O. Fetal and neonatal abnormalities due to congenital rubella syndrome: a review of literature. J. Matern. Neonatal. Med. 2017;30:274–278. doi: 10.3109/14767058.2016.1169526. [DOI] [PubMed] [Google Scholar]
- 96.Gordon-Lipkin E., Hoon A., Pardo C.A. Prenatal cytomegalovirus, rubella, and Zika virus infections associated with developmental disabilities: past, present, and future. Dev. Med. Child Neurol. 2020:1–9. doi: 10.1111/dmcn.14682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Toizumi M., Nguyen G.T., Motomura H., Nguyen T.H., Pham E., Kaneko K., et al. Sensory defects and developmental delay among children with congenital rubella syndrome. Sci. Rep. 2017;7:1–11. doi: 10.1038/srep46483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Boucoiran I., Castillo E. Rubella in pregnancy. J. Obstet. Gynaecol. Can. 2018;40:1646–1656. doi: 10.1016/j.jogc.2018.07.003. [DOI] [PubMed] [Google Scholar]
- 99.Vauloup-Fellous C. Standardization of rubella immunoassays. J. Clin. Virol. 2018;102:34–38. doi: 10.1016/j.jcv.2018.02.006. [DOI] [PubMed] [Google Scholar]
- 100.De Martino M. Dismantling the taboo against vaccines in pregnancy. Int. J. Mol. Sci. 17 (2016) 1–8. doi: 10.3390/ijms17060894. [DOI] [PMC free article] [PubMed]
- 101.Mangtani P., Evans S.J.W., Lange B., Oberle D., Smith J., Drechsel-Baeuerle U., et al. Safety profile of rubella vaccine administered to pregnant women: A systematic review of pregnancy related adverse events following immunisation, including congenital rubella syndrome and congenital rubella infection in the foetus or infant. Vaccine. 2020;38:963–978. doi: 10.1016/j.vaccine.2019.11.070. [DOI] [PubMed] [Google Scholar]
- 102.Young M, Cripps A, Nimmo G, van Driel M. Post-exposure passive immunisation for preventing rubella and congenital rubella syndrome. Cochrane Database Syst. Rev. (2015 Sep 9) CD010586. doi: 10.1002/14651858.CD010586.pub2.www.cochranelibrary.com. [DOI] [PMC free article] [PubMed]
- 103.Magnius L., Mason W.S., Taylor J., Kann M., Glebe D., Dény P., et al. ICTV virus taxonomy profile: Hepadnaviridae. J. Gen. Virol. 2020;101:571–572. doi: 10.1099/jgv.0.001415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Nwaohiri A., Schillie S., Bulterys M., Kourtis A.P. Hepatitis C virus infection in children: how do prevent it and how do we treat it? Expert Rev. Anti-Infect. Ther. 2018;16:689–694. doi: 10.1080/14787210.2018.1509707.Hepatitis. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Castillo E., Murphy K., van Schalkwyk J. Hepatitis B and pregnancy. J. Obstet. Gynaecol. Can. 2017;39:181–190. doi: 10.1016/j.jogc.2016.11.001. [DOI] [PubMed] [Google Scholar]
- 106.Bartholomew M.L., Lee M.-J. Management of hepatitis B infection in pregnancy. Clin. Obstet. Gynecol. 2018;61:137–145. doi: 10.1097/GRF.0000000000000331. [DOI] [PubMed] [Google Scholar]
- 107.Seto M.T., Cheung K.W., IFN Hung. Management of viral hepatitis A, C, D and E in pregnancy. Best Pract res Clin Obstet Gynaecol. 2020;68:44–53. doi: 10.1016/j.bpobgyn.2020.03.009. [DOI] [PubMed] [Google Scholar]
- 108.Benova L., Mohamoud Y.A., Clavert C., Abu-Raddad L.J. Vertical transmission of hepatitis C virus: systematic review and meta-analysis. Clin. Infect. Dis. 2014;59:765–773. doi: 10.1093/cid/ciu447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Mavilia M.G., Wu G.Y. Mechanisms and prevention of vertical transmission in chronic viral hepatitis. J. Clin. Transl. Hepatol. 2017;5:119–129. doi: 10.14218/JCTH.2016.00067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Cryer A.M., Imperial J.C. Hepatitis B in pregnant women and their infants. Clin. Liver Dis. 2019;23:451–462. doi: 10.1016/j.cld.2019.04.007. [DOI] [PubMed] [Google Scholar]
- 111.Cheung K.W., Seto M.T.Y., Lao T.T.-H. Prevention of perinatal hepatitis B virus transmission. Arch. Gynecol. Obstet. 2019;300:251–259. doi: 10.1007/s00404-019-05190-0. [DOI] [PubMed] [Google Scholar]
- 112.Liu J., Zhang S., Liu M., Wang Q., Shen H., Zhang Y. Maternal pre-pregnancy infection with hepatitis B virus and the risk of preterm birth: a population-based cohort study. Lancet Glob. Health. 2017;5:e624–e632. doi: 10.1016/S2214-109X(17)30142-0. [DOI] [PubMed] [Google Scholar]
- 113.Indolfi G., Easterbrook P., Dusheiko G., Siberry G., Chang M., Thorne C., et al. Hepatitis B virus infection in children and adolescents. Lancet Gastroenterol. Hepatol. 2019;4:466–476. doi: 10.1016/S2468-1253(19)30042-1. [DOI] [PubMed] [Google Scholar]
- 114.Giugliano S., Petroff M.G., Warren B.D., Jasti S., Linscheid C., Ward A., et al. Hepatitis C virus sensing by human trophoblasts induces innate immune responses and recruitment of maternal NK cells: potential implications for limiting vertical transmission. J. Immunol. 2015;195:3737–3747. doi: 10.4049/jimmunol.1500409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Piffer S., Mazza A., Dell’Anna L. Serological screening for hepatitis C during pregnancy: seroprevalence and maternal and neonatal outcomes in 45,000 pregnant women. Eur. J. Obstet. Gynecol. 2020;254:195–199. doi: 10.1016/j.ejogrb.2020.09.023. [DOI] [PubMed] [Google Scholar]
- 116.Connell L.E., Salihu H.M., Salemi J.L., August E.M., Weldeselasse H., Mbah A.K. Maternal hepatitis B and hepatitis C carrier status and perinatal outcomes. Liver Int. 2011;31:1163–1170. doi: 10.1111/j.1478-3231.2011.02556.x. [DOI] [PubMed] [Google Scholar]
- 117.Hou J., Cui F., Ding Y., Dou X., Duan Z., Han G., et al. Management algorithm for interrupting mother-to-child transmission of hepatitis B virus. Clin. Gastroenterol. Hepatol. 2019;17 doi: 10.1016/j.cgh.2018.10.007. (1929-1936.e1) [DOI] [PubMed] [Google Scholar]
- 118.Kushner T., Reau N. Changing epidemiology, implications, and recommendations for hepatitis C in women of childbearing age and during pregnancy. J. Hepatol. 2020;S0168-8278:33814–33819. doi: 10.1016/j.jhep.2020.11.027. [DOI] [PubMed] [Google Scholar]
- 119.Post J.J. Update on hepatitis C and implications for pregnancy. Obstet. Med. 2017;10:157–160. doi: 10.1177/1753495X17708093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Lopata S.M., McNeer E., Dudley J.A., Wester C., Cooper W.O., Carlucci J.G., et al. Hepatitis C testing among perinatally exposed infants. Pediatrics. 2020;145 doi: 10.1542/peds.2019-2482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Tran T.T., Ahn J., Reau N.S. ACG clinical guideline: liver disease and pregnancy. Am. J. Gastroenterol. 2016;111:176–194. doi: 10.1038/ajg.2015.430. [DOI] [PubMed] [Google Scholar]
- 122.Terrault N.A., Bzowej N.H., Chang K.-M., Hwang J.P., Jonas M.M., Murad M.H. AASLD guidelines for treatment of chronic hepatitis B. Hepatology. 2016;63:261–283. doi: 10.1002/hep.28156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Brown R.S., McMahon B.J., Lok A.S.F., Wong J.B., Ahmed A.T., Mouchli M.A., et al. Antiviral therapy in chronic hepatitis B viral infection during pregnancy: A systematic review and meta-analysis. Hepatology. 2016;63:319–333. doi: 10.1002/hep.28302. [DOI] [PubMed] [Google Scholar]
- 124.Eke A., Eleje G., Eke U., Xia Y., Liu J. Hepatitis B immunoglobulin during pregnancy for prevention of mother-to-child transmission of hepatitis B virus. Cochrane Database Syst. Rev. 2017;2 doi: 10.1002/14651858.CD008545.pub2.www.cochranelibrary.com. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Bernstein H.B., Dunkelberg J.C., Leslie K.K. Hepatitis C in pregnancy in the era of direct-acting antiviral treatment: potential benefits of universal screening and antepartum therapy. Clin. Obstet. Gynecol. 2018;61:146–156. doi: 10.1097/GRF.0000000000000345. [DOI] [PubMed] [Google Scholar]
- 126.Indolfi G., Easterbrook P., Dusheiko G., El-Sayed M.H., Jonas M.M., Thorne C., et al. Hepatitis C virus infection in children and adolescents. Lancet Gastroenterol. Hepatol. 2019;4:477–487. doi: 10.1016/S2468-1253(19)30046-9. [DOI] [PubMed] [Google Scholar]
- 127.Chappell C.A., Hillier S.L., Crowe D., Meyn L.A., Bogen D.L., Krans E.E. Hepatitis C virus screening among children exposed during pregnancy. Pediatrics. 2018;141 doi: 10.1542/peds.2017-3273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Ragusa R., Corsaro L.S., Frazzetto E., Bertino E., Bellia M.A., Bertino G. Hepatitis C virus infection in children and pregnant women: an updated review of the literature on screening and treatments. Am. J. Perinatol. Rep. 2020;10:e121–e127. doi: 10.1055/s-0040-1709185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Capriotti T. HIV/AIDS: an update for home healthcare clinicians. Home Healthc. Now. 2018;36:348–355. doi: 10.1097/NHH.0000000000000706. [DOI] [PubMed] [Google Scholar]
- 130.Faure-Bardon V., Ville Y. Maternal infections: revisiting the need for screening in pregnancy. BJOG An. Int. J. Obstet. Gynaecol. 2020 Sep 16 doi: 10.1111/1471-0528.16509. [DOI] [PubMed] [Google Scholar]
- 131.Dorsamy V., Vallen C., Haffejee F., Moodley J., Naicker T. The role of trophoblast cell receptor expression in HIV-1 passage across the placenta in pre-eclampsia: an observational study. BJOG An. Int. J. Obstet. Gynaecol. 2017;124:920–928. doi: 10.1111/1471-0528.14311. [DOI] [PubMed] [Google Scholar]
- 132.Ghosn J., Taiwo B., Seedat S., Autran B., Katlama C. HIV. Lancet. 2018;392 doi: 10.1016/S0140-6736(18)31311-4. (685–69) [DOI] [PubMed] [Google Scholar]
- 133.Pereira N.Z., Branco A.C.C.C., Manfrere K.C.G., de Lima J.F., Yoshikawa F.S.Y., Milanez H.M.B.P.M., et al. Increased expression on innate immune factors in placentas from HIV-infected mothers concurs with dampened systemic immune activation. Front. Immunol. 2020;11:1–10. doi: 10.3389/fimmu.2020.01822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Johnson E.L., Chakraborty R. HIV-1 at the placenta: immune correlates of protection and infection. Curr. Opin. Infect. Dis. 2016;29:248–255. doi: 10.1097/QCO.0000000000000267. [DOI] [PubMed] [Google Scholar]
- 135.Kessler P.A. Potential role of regulatory T cells in mother-to-child transmission of HIV. Curr. HIV Res. 2018;16:396–403. doi: 10.2174/1570162X17666190213094624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Mandelbrot L., Sibiude J. A link between antiretrovirals and perinatal outcomes? Lancet HIV. 2016;4:e3–e5. doi: 10.1016/S2352-3018(16)30188-6. [DOI] [PubMed] [Google Scholar]
- 137.Bailey H., Zash R., Rasi V., Thorne C. HIV treatment in pregnancy. Lancet HIV. 2018;5:e457–e467. doi: 10.1016/S2352-3018(18)30059-6. [DOI] [PubMed] [Google Scholar]
- 138.Van Dyke RB, Chadwick EG, Hazra R, Williams PL, Seage GRI. The PHACS SMARTT study: assessment of the safety of in utero exposure to antiretroviral drugs. Front. Immunol. 7 (2016) 1–7. doi: 10.3389/fimmu.2016.00199. [DOI] [PMC free article] [PubMed]
- 139.Zash R., Holmes L., Diseko M., Jacobson D.L., Brummel S., Mayondi G., et al. Neural-tube defects and antiretroviral treatment regimens in Botswana. N. Engl. J. Med. 2019;381:827–840. doi: 10.1056/NEJMoa1905230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Mohammadi H, Papp E, Cahill L, Rennie M, Banko N, Pinnaduwage L, et al. HIV antiretroviral exposure in pregnancy induces detrimental placenta vascular changes that are rescued by progesterone supplementation. Sci. Rep. 6552 (2018) 1–14. doi: 10.1038/s41598-018-24680-w. [DOI] [PMC free article] [PubMed]
- 141.Wedi C.O.O., Kirtley S., Hopewell S., Corrigan R., Kennedy S.H., Hemelaar J. Perinatal outcomes associated with maternal HIV infection: a systematic review and meta-analysis. Lancet HIV. 2016;3:e33–e48. doi: 10.1016/S2352-3018(15)00207-6. [DOI] [PubMed] [Google Scholar]
- 142.Sims A., Hadigan C. Cardiovascular complications in children with HIV infection. Curr HIV/AIDS Rep. 2011;8:209–214. doi: 10.1007/s11904-011-0081-9. [DOI] [PubMed] [Google Scholar]
- 143.Muller W.J. Treatment of perinatal viral infections to improve neurologic outcomes. Pediatr. Res. 2017;81:162–169. doi: 10.1038/pr.2016.191. [DOI] [PubMed] [Google Scholar]
- 144.Read J.S., Committee on Pediatric AIDS, American Academy of Pediatrics Diagnosis of HIV-1 infection in children younger than 18 months in the United States. Pediatrics. 2007;120:e1547–e1562. doi: 10.1542/peds.2007-2951. [DOI] [PubMed] [Google Scholar]
- 145.Gordon Lynch N., Kesler Johnson A. Congenital HIV. Prevention of maternal to child transmission. Adv. Neonatal. Care. 2018;18:330–340. doi: 10.1097/ANC.0000000000000559. [DOI] [PubMed] [Google Scholar]
- 146.Leeper C., Lutzkanin A.I. Infections during pregnancy. Prim. Care. 2018;45:567–586. doi: 10.1016/j.pop.2018.05.013. [DOI] [PubMed] [Google Scholar]
- 147.Lumaca A., Galli L., De Martino M., Chiappini E. Paediatric HIV-1 infection: updated strategies of prevention mother-to-child transmission. J. Chemother. 2018;30:193–202. doi: 10.1080/1120009X.2018.1451030. [DOI] [PubMed] [Google Scholar]
- 148.Spooner E., Govender K., Reddy T., Ramjee G., Mbadi N., Singh S., et al. Point-of-care HIV testing best practice for early infant diagnosis: an implementation study. BMC Public Health. 2019;19:1–14. doi: 10.1186/s12889-019-6990-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Van Rie A Van, Harrington P.R., Dow A., Robertson K. Neurologic and neurodevelopmental manifestations of pediatric HIV/AIDS: a global perspective. Eur. J. Pediatr. Neurol. 2007;11:1–9. doi: 10.1016/j.ejpn.2006.10.006. [DOI] [PubMed] [Google Scholar]
- 150.Nielsen-Saines K. Perinatal HIV as an infectious cause of developmental regression. Neurosci. Biobehav. Rev. 2019;102:417–423. doi: 10.1016/j.neubiorev.2019.05.011. [DOI] [PubMed] [Google Scholar]
- 151.Mandelbrot L., Tubiana R., LeChenadec J., Dollfus C., Faye A., Pannier E., et al. No perinatal transmission of HIV-1 from women with effective antiretroviral therapy starting before conception. Clin. Infect. Dis. 2015;61:1715–1725. doi: 10.1093/cid/civ578. [DOI] [PubMed] [Google Scholar]
- 152.Parigi S., Licari A., Marseglia G.L., Galli L., Chiappini E. What the paediatrician needs to know about HIV-1 infection. Pediatr. Allergy Immunol. 2020;31:28–31. doi: 10.1111/pai.13155. [DOI] [PubMed] [Google Scholar]
- 153.Anugulruengkitt S., Suntarattiwong P., Ounchanum P., Srirompotong U., Jantarabenjakul W., Sophonphan J., et al. Safety of 6-week neonatal triple-combination antiretroviral postexposure prophylaxis in high-risk HIV-exposed infants. Pediatr. Infect. Dis. J. 2019;38:1045–1050. doi: 10.1097/INF.0000000000002426. [DOI] [PubMed] [Google Scholar]
- 154.Greiter B.M., Kahlert C.R., Eberhard N., Sultan-Beyer L., Berger C., Paioni P., et al. Lymphocyte subsets in HIV-exposed uninfected infants: the impact of neonatal postexposure prophylaxis with zidovudine. Open Forum Infect Dis. 2020;7:1–4. doi: 10.1093/ofid/ofaa108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Powis K.M., Smeaton L., Hughes M.D., Tumbare E.A., Sajini S., Jao J., et al. In-utero triple antiretroviral exposure associated with decreased growth among HIV-exposed uninfected infants in Botswana. AIDS. 2016;30:211–220. doi: 10.1097/QAD.0000000000000895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Pasvol T.J., Foster C., Fidler S. Novel therapies/hopes for HIV cure in perinatally acquired HIV positive adolescents. Curr. Opin. HIV AIDS. 2018;13:281–287. doi: 10.1097/COH.0000000000000455. [DOI] [PubMed] [Google Scholar]
- 157.Qiu J., Söderlund-Venermo M., Young N.S. Human Parvoviruses. Clin. Microbiol. Rev. 2017;30:43–113. doi: 10.1128/CMR.00040-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Attard L., Tadolini M., De Rose D.U., Cattalini M. Overview of fever of unknown origin in adult and paediatric patients. Clin. Exp. Rheumatol. 2018;36 [PubMed] [Google Scholar]
- 159.Young N.S., Brown K.E. Parvovirus B19. N. Engl. J. Med. 2004;350:586–597. doi: 10.1056/NEJMra030840. [DOI] [PubMed] [Google Scholar]
- 160.Sim J.Y., Chang L.-Y., Chen J.-M., Lee P.-I., Huang L.-M., Lu C.-Y. Human parvovirus B19 infection in patients with or without underlying diseases. J. Microbiol. Immunol. Infect. 2019;52:534–541. doi: 10.1016/j.jmii.2019.05.009. [DOI] [PubMed] [Google Scholar]
- 161.Dollat M., Chaigne B., Cormier G., Costedoat-chalumeau N., Lifermann F., Deroux A., et al. Extra-haematological manifestations related to human parvovirus B19 infection: retrospective study in 25 adults. BMC Infect. Dis. 2018;18:1–10. doi: 10.1186/s12879-018-3227-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Pasquinelli G., Bonvicini F., Foroni L., Salfi N., Gallinella G. Placental endothelial cells can be productively infected by Parvovirus B19. J. Clin. Virol. 2009;44:33–38. doi: 10.1016/j.jcv.2008.10.008. [DOI] [PubMed] [Google Scholar]
- 163.Luo Y., Lou S., Deng X., Liu Z., Li Y., Kleiboeker S., et al. Parvovirus B19 infection of human primary erythroid progenitor cells triggers ATR-Chk1 signaling, which promotes B19 virus replication. J. Virol. 2011;85:8046–8055. doi: 10.1128/JVI.00831-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Ornoy A., Ergaz Z. Parvovirus B19 infection during pregnancy and risks to the fetus. Birth Defects. 2017;Res.109:311–323. doi: 10.1002/bdra.23588. [DOI] [PubMed] [Google Scholar]
- 165.Bonvicini F., Bua G., Gallinella G. Parvovirus B19 infection in pregnancy — awareness and opportunities. Curr. Opin. Virol. 2017;27:8–14. doi: 10.1016/j.coviro.2017.10.003. [DOI] [PubMed] [Google Scholar]
- 166.Enders M., Weidner A., Zoellner I., Searle K., Enders G. Fetal morbidity and mortality after acute human parvovirus B19 infection in pregnancy: prospective evaluation of 1018 cases. Prenat. Diagn. 2004;24:513–518. doi: 10.1002/pd.940. [DOI] [PubMed] [Google Scholar]
- 167.Gilby D.M., Mee J.B., Kamlin C.O.F., Kornman L.H., Davis P.G., Manley B.J. Outcomes following antenatal identification of hydrops fetalis: a single-centre experience from 2001 to 2012. Arch. Dis. Child. Fetal Neonatal Ed. 2019;104:F253–F258. doi: 10.1136/archdischild-2017-313604. [DOI] [PubMed] [Google Scholar]
- 168.de Haan T., van den Akker E., Porcelijn L., Oepkes D., Kroes A., Walther F. Thrombocytopenia in hydropic fetuses with parvovirus B19 infection: incidence, treatment and correlation with fetal B19 viral load. BJOG An. Int. J. Obstet. Gynaecol. 2008;115:76–81. doi: 10.1111/j.1471-0528.2007.01555.x. [DOI] [PubMed] [Google Scholar]
- 169.Landry M.L. Parvovirus B19. Microbiol. Spectr. 2016;4:1–13. doi: 10.1128/microbiolspec.DMIH2-0008-2015.Correspondence. [DOI] [PubMed] [Google Scholar]
- 170.Delle Chiaie L., Buck G., Grab D., Terinde R. Prediction of fetal anemia with Doppler measurement of the middle cerebral artery peak systolic velocity in pregnancies complicated by maternal blood group alloimmunization or parvovirus B19 infection. Ultrasound Obstet. Gynecol. 2001;18:232–236. doi: 10.1046/j.0960-7692.2001.00540.x. [DOI] [PubMed] [Google Scholar]
- 171.Attwood L.O., Holmes N.E., Hui L. Identification and management of congenital parvovirus B19 infection. Prenat. Diagn. 2020:1–10. doi: 10.1002/pd.5819. [DOI] [PubMed] [Google Scholar]
- 172.Gallinella G. The clinical use of parvovirus B19 assays: recent advances. Expert. Rev. Mol. Diagn. 2018;18:821–832. doi: 10.1080/14737159.2018.1503537. [DOI] [PubMed] [Google Scholar]
- 173.Ishikawa A., Yoto Y., Asakura H., Tsutsumi H. Quantitative analysis of human parvovirus B19 DNA in maternal and fetal serum, and amniotic fluid during an early stage of pregnancy. J. Med. Virol. 2015;87:683–685. doi: 10.1002/jmv. [DOI] [PubMed] [Google Scholar]
- 174.Crane J., Mundle W., Boucoiran I., Maternal Fetal Medicine Committee Parvovirus B19 infection in pregnancy. J. Obstet. Gynaecol. Can. 2014;36:1107–1116. doi: 10.1016/S1701-2163(15)30390-X. [DOI] [PubMed] [Google Scholar]
- 175.Bascietto F., Liberati M., Murgano D., Buca D., Iacovelli A., Flacco M., et al. Outcome of fetuses with congenital parvovirus B19 infection: systematic review and meta-analysis. Ultrasound Obstet. Gynecol. 2018;52:569–576. doi: 10.1002/uog.19092. [DOI] [PubMed] [Google Scholar]
- 176.Sanchez-Duran M.A., Higueras M.T., Avilés-Garcia M., Maiz N., Rodriguez-Aliberas M., Arévalo S., et al. Perinatal outcomes and central nervous system abnormalities following intrauterine fetal transfusion: 17 years’ experience in a tertiary center. Transfusion. 2020;60(11):2557–2564. doi: 10.1111/trf.16087. [DOI] [PubMed] [Google Scholar]
- 177.Maisonneuve E., Friszer S. Fetal brain injury associated with parvovirus B19 congenital infection requiring intrauterine transfusion. Fetal Diagn. Ther. 2019;46 doi: 10.1159/000489881. [DOI] [PubMed] [Google Scholar]
- 178.Hobbs J.A. Detection of adeno-associated virus 2 and parvovirus B19 in the human dorsolateral prefrontal cortex. J. Neuro-Oncol. 2006;12:190–199. doi: 10.1080/13550280600827351. [DOI] [PubMed] [Google Scholar]
- 179.Grant J.K., Yin N.C., Zaytoun A.M., Waseem H., Hobbs J.A. Persistent adeno-associated virus 2 and parvovirus B19 sequences in post-mortem human cerebellum. Cerebellum. 2009;8:490–498. doi: 10.1007/s12311-009-0126-4. [DOI] [PubMed] [Google Scholar]
- 180.Manaresi E., Gallinella G. Advances in the development of antiviral strategies against parvovirus B19. Viruses. 2019;11:659. doi: 10.3390/v11070659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Majer A., McGreevy A., Booth T.F. Molecular pathogenicity of enteroviruses causing neurological disease. Front. Microbiol. 2020;11:1–22. doi: 10.3389/fmicb.2020.00540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Daley A.J., Isaacs D., Dwyer D.E., Gilbert G.L. A cluster of cases of neonatal coxsackievirus B meningitis and myocarditis. J. Paediatr. Child Health. 1998;34:196–198. doi: 10.1046/j.1440-1754.1998.00176.x. [DOI] [PubMed] [Google Scholar]
- 183.Chuang Y.-Y., Huang Y.-C. Enteroviral infection in neonates. J. Microbiol. Immunol. Infect. 2019;52:851–857. doi: 10.1016/j.jmii.2019.08.018. [DOI] [PubMed] [Google Scholar]
- 184.Brown D.M., Zhang Y., Scheuermann R.H. Epidemiology and sequence-based evolutionary analysis of circulating non-polio enteroviruses. Microorganisms. 2020;8:1–23. doi: 10.3390/microorganisms8121856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Peters C.E., Carette J.E. Return of the neurotropic enteroviruses: co-opting cellular pathways for infection. Viruses. 2021;13:1–23. doi: 10.3390/v13020166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Soike K. Coxsackie B-3 virus infection in the pregnant mouse. J. Infect. Dis. 1961;117:203–208. doi: 10.1093/infdis/117.3.203. [DOI] [PubMed] [Google Scholar]
- 187.Liu J.-Y., Wang S.-M., Chen I.-C., Yu C.-K., Liu C.-C. Hepatic damage caused by coxsackievirus B3 is dependent on age-related tissue tropisms associated with the coxsackievirus-adenovirus receptor. Pathog. Dis. 2013;68:52–60. doi: 10.1111/2049-632X.12044. [DOI] [PubMed] [Google Scholar]
- 188.Hafenstein S., Bowman V.D., Chipman P.R., Kelly C.M.B., Lin F., Medof M.E., et al. Interaction of decay-accelerating factor with Coxsackievirus B3. J. Virol. 2007;81:12927–12935. doi: 10.1128/JVI.00931-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Feng M., Liao Y., Gao Y., Jiang G., Wang L., Zhang Y., et al. Mechanism for the lethal effect of enterovirus A71 intracerebral injection in neonatal mice. Lab. Investig. 2020;100:596–605. doi: 10.1038/s41374-019-0351-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Harvala H., Broberg E., Benschop K., Berginc N., Ladhani S., Susi P., et al. Recommendations for enterovirus diagnostics and characterisation within and beyond Europe. J. Clin. Virol. 2018;101:11–17. doi: 10.1016/j.jcv.2018.01.008. [DOI] [PubMed] [Google Scholar]
- 191.Muehlenbachs A., Bhatnagar J., Zaki S.R. Tissue tropism, pathology and pathogenesis of enterovirus infection. J. Pathol. 2015;235:217–228. doi: 10.1002/path.4438. [DOI] [PubMed] [Google Scholar]
- 192.Ouellet A., Sherlock R., Toye B., Fung K.F.K. Antenatal diagnosis of intrauterine infection with coxsackievirus B3 associated with live birth. Infect. Dis. Obstet. Gynecol. 2004;12:23–26. doi: 10.1080/1064744042000210357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Lin J.-Y., Kung Y.-A., Shih S.-R. Antivirals and vaccines for Enterovirus A71. J. Biomed. Sci. 2019;26:1–10. doi: 10.1186/s12929-019-0560-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Wei X., Yang J., Gao L., Wang L., Liao Q., Qiu Q., et al. The transfer and decay of maternal antibodies against enterovirus A71, and dynamics of antibodies due to later natural infections in Chinese infants: a longitudinal, paired mother – neonate cohort study. Lancet Infect. Dis. 2020;S1473-3099:30480–30481. doi: 10.1016/S1473-3099(20)30480-1. [DOI] [PubMed] [Google Scholar]
- 195.Mariano G., Farthing R.J., Lale-Farjat S.L.M., Bergeron J.R.C. Structural characterization of SARS-CoV-2: where we are, and where we need to be. Front. Mol. Biosci. 2020;7:1–28. doi: 10.3389/fmolb.2020.605236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Salzberger B., Buder F., Lampl B., Ehrenstein B., Hitzenbichler F., Holzmann T., et al. Epidemiology of SARS-CoV-2. Infection. 2020 Oct;8:1–7. doi: 10.1007/s15010-020-01531-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Figliozzi S., Masci P., Ahmadi N., Tondi L., Koutli E., Aimo A., et al. Predictors of adverse prognosis in Covid-19: a systematic review and meta-analysis. Eur. J. Clin. Investig. 2020;50 doi: 10.1111/eci.13362. [DOI] [PubMed] [Google Scholar]
- 198.Olivini N., Calò Carducci F.I., Santilli V., De Ioris M.A., Scarselli A., Alario D., et al. A neonatal cluster of novel coronavirus disease 2019: clinical management and considerations. Ital. J. Pediatr. 2020;46:1–9. doi: 10.1186/s13052-020-00947-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Garazzino S., Montagnani C., Donà D., Meini A., Felici E., Vergine G., et al. Multicentre Italian study of SARS-CoV-2 infection in children and adolescents, preliminary data as at 10 April 2020. Eur. Surveill. 2020;25 doi: 10.2807/1560-7917.ES.2020.25.18.2000600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Jing Y., Run-Qian L., Hao-Ran W., Hao-Ran C., Ya-Bin L., Yang G., et al. Potential influence of COVID-19/ACE2 on the female reproductive system. Mol. Hum. Reprod. 2020 doi: 10.1093/molehr/gaaa030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Beeraka N.M., Sadhu S.P., Madhunapantula S.V., Pragada R.R., Svistunov A.A., Nikolenko V.N., et al. Strategies for targeting SARS CoV-2: small molecule inhibitors — the current status. Front. Immunol. 2020;11:1–22. doi: 10.3389/fimmu.2020.552925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Patberg E.T., Adams T., Rekawek P., Vahanian S.A., Akerman M., Hernandez A., et al. Coronavirus disease 2019 infection and placental histopathology in women delivering at term. Am. J. Obstet. Gynecol. 2020;S0002-9378:31194–31197. doi: 10.1016/j.ajog.2020.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Algarroba G.N., Rekawek P., Vahanian S.A., Khullar P., Palaia T., Peltier M.R., et al. Visualization of severe acute respiratory syndrome coronavirus 2 invading the human placenta using electron microscopy. Am. J. Obstet. Gynecol. 2020;223:275–278. doi: 10.1016/j.ajog.2020.05.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.De Rose DU, Piersigilli F, Ronchetti MP, Santisi A, Bersani I, Dotta A, Danhaive O, Auriti C. Novel Coronavirus disease (COVID-19) in newborns and infants: what we know so far. Ital. J. Pediatr. 2020;46:1–8. [DOI] [PMC free article] [PubMed]
- 205.Crovetto F., Crispi F., Liurba E., Pascal R., Larroya M., Trilla C., et al. Impact of SARS-CoV-2 infection on pregnancy outcomes: a population-based study. Clin. Infect. Dis. 2021 Feb;8:1–25. doi: 10.1093/cid/ciab104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Juan J., Gil M., Rong Z., Zhang Y., Yang H., Poon L. Effects of coronavirus disease 2019 (COVID-19) on maternal, perinatal and neonatal outcomes: a systematic review. Ultrasound Obstet. Gynecol. 2020 doi: 10.1002/uog.22088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Li M., Chen L., Zhang J., Xiong C., Li X. The SARS-CoV-2 receptor ACE2 expression of maternal-fetal interface and fetal organs by single-cell transcriptome study. PLoS One. 2020;15:1–12. doi: 10.1371/journal.pone.0230295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Vivanti A.J., Vauloup-Fellous C., Prevot S., Zupan V., Suffee C., Do Cao J., et al. Transplacental transmission of SARS-CoV-2 infection. Nat. Commun. 2020;11:3572. doi: 10.1038/s41467-020-17436-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Raschetti R., Vivanti A.J., Vauloup-Fellous C., Barbara L., Benachi A., De Luca D. Synthesis and systematic review of reported neonatal SARS-CoV-2 infections. Nat. Commun. 2020;11:5164. doi: 10.1038/s41467-020-18982-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.WHO COVID-19 LENS (Living Evidence Synthesis) working group . 2021. Definition and Categorization of the Timing of Mother-to-child Transmission of SARS-CoV-2; https://www.who.int/publications/i/item/WHO-2019-nCoV-mother-to-child-transmission-2021.1pp. 1–14. [Google Scholar]
- 211.World Health Organization COVID-19: Clinical Management. https://www.who.int/publications/i/item/WHO-2019-nCoV-clinical-2021-1
- 212.Lauring A.S., Hodcroft E.B. Genetic variants of SARS-CoV-2 - what do the mean? JAMA. 2021;325:529–531. doi: 10.1001/jama.2020.27124. [DOI] [PubMed] [Google Scholar]
- 213.Mascola J.R., Graham B.S., Fauci A.S. SARS-CoV-2 viral variants - tackling a moving target. JAMA. 2021 doi: 10.1001/jama.2021.2088. [DOI] [PubMed] [Google Scholar]
- 214.Male V. Are COVID-19 vaccines safe in pregnancy? Nat. Rev. Immunol. 2021;21:200–201. doi: 10.1038/s41577-021-00525-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Rottenstreich A., Zarbiv G., Oiknine-Djian E., Zigron R., Wolf D.G., Porat S. Efficient maternofetal transplacental transfer of anti- SARS-CoV-2 spike antibodies after antenatal SARS-CoV-2 BNT162b2 mRNA vaccination. Clin. Infect. Dis. 2021 doi: 10.1093/cid/ciab266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Gray K.J., Bordt E.A., Atyeo C., Deriso E., Akinwunmi B., Young N., Medina Baez A., Shook L.L., Cvrk D., James K., De Guzman R., Brigida S., Diouf K., Goldfarb I., Bebell L.M., Yonker L.M., Fasano A., Rabi S.A., Elovitz M.A., Alter G., Edlow A.G. COVID-19 vaccine response in pregnant and lactating women: a cohort study. Am. J. Obstet. Gynecol. 2021;S0002-9378 doi: 10.1016/j.ajog.2021.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Oyarzún-Arrau A., Alonso-Palomares L., Valiente-Echeverría F., Osorio F., Soto-Rifo R. Crosstalk between RNA metabolism and cellular stress responses during Zika virus replication. Pathogens. 2020;9:1–19. doi: 10.3390/pathogens9030158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Khaiboullina S.F., Ribeiro F.M., Uppal T., Martynova E.V., Rizvanov A.A., Verma S.C. Zika virus transmission through blood tissue barriers. Front. Microbiol. 2019;10:1–13. doi: 10.3389/fmicb.2019.01465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Masmejan S., Musso D., Vouga M., Pomar L., Dashraath P., Stojanov M., et al. Zika Virus. Pathogens. 2020;9:1–14. doi: 10.3390/pathogens9110898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Arora H.S. A to Z of Zika virus: A comprehensive review for clinicians. Glob. Pediatr. Heal. 2020;7:1–14. doi: 10.1177/2333794X20919595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Musso D., Ko A.I., Baud D. Zika virus infection — after the pandemic. N. Engl. J. Med. 2019;381:1444–1457. doi: 10.1056/NEJMra1808246. [DOI] [PubMed] [Google Scholar]
- 222.Devakumar D., Bamford A., Ferreira M.U., Broad J., Rosch R.E., Groce N., et al. Infectious causes of microcephaly: epidemiology, pathogenesis, diagnosis, and management. Lancet Infect. Dis. 2017;18:e1–13. doi: 10.1016/S1473-3099(17)30398-5. [DOI] [PubMed] [Google Scholar]
- 223.Centeno-Tablante E., Medina-Rivera M., Finkelstein J.L., Herman H.S., Rayco-Solon P., Garcia-Casal M.N., et al. Update on the transmission of Zika virus through breast Milk and breastfeeding: a systematic review of the evidence. Viruses. 2021;13:123. doi: 10.3390/v13010123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Szaba F.M., Tighe M., Kummer L.W., Lanzer K.G., Ward J.M., Lanthier P., et al. Zika virus infection in immunocompetent pregnant mice causes fetal damage and placental pathology in the absence of fetal infection. PLoS Pathog. 2018;14 doi: 10.1371/journal.ppat.1006994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Rabelo K., de Souza L.J., Salomão N.G., Machado L.N., Pereira P.G., Portari E.A., et al. Zika induces human placental damage and inflammation. Front. Immunol. 2020;11:1–15. doi: 10.3389/fimmu.2020.02146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Santos G.R., Pinto C.A.L., Prudente R.C.S., Bevilacqua E.M.A.F., Witkin S.S. Histopathologic changes in placental tissue associated with vertical transmission of Zika virus. Int. J. Gynecol. Pathol. 2020;39:157–162. doi: 10.1097/PGP.0000000000000586. [DOI] [PubMed] [Google Scholar]
- 227.Nascimento-Carvalho G.C., Nascimento-Carvalho E.C., VanDuijn M.M., Ramos C.L., Vilas-Boas A.-L., Moreno-Carvalho O.A., et al. Cerebrospinal immunoglobulins are increased in neonates exposed to Zika virus during foetal life. J. Inf. Secur. 2020;80:419–425. doi: 10.1016/j.jinf.2020.01.006. [DOI] [PubMed] [Google Scholar]
- 228.Richard A.S., Shim B.-S., Kwon Y.-C., Zhang R., Otsuka Y., Schmitt K., et al. AXL-dependent infection of human fetal endothelial cells distinguishes Zika virus from other pathogenic flaviviruses. PNAS. 2017;114:2024–2029. doi: 10.1073/pnas.1620558114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Hastings A.K., Yockey L.J., Jagger B.W., Hwang J., Uraki R., Gaitsch H.F., et al. TAM receptors are not required for Zika virus infection in mice article. Cell Rep. 2017;19:558–568. doi: 10.1016/j.celrep.2017.03.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Rombi F., Bayliss R., Tuplin A., Yeoh S. The journey of Zika to the developing brain. Mol. Biol. Rep. 2020;47:3097–3115. doi: 10.1007/s11033-020-05349-y. [DOI] [PubMed] [Google Scholar]
- 231.Viens L.J., Fleck-Derderian S., Baez-Santiago M.A., Oduyebo T., Broussard C.S., Khan S., et al. Role of prenatal ultrasonography and amniocentesis in the diagnosis of congenital Zika syndrome. Obstet. Gynecol. 2020;135:1185–1197. doi: 10.1097/AOG.0000000000003829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Vouga M., Chiu Y.-C., Pomar L., de Meyer S.V., Masmejan S., Genton B., et al. Dengue, Zika and chikungunya during pregnancy: pre- and post-travel advice and clinical management. J. Travel Med. 2019;26:1–13. doi: 10.1093/jtm/taz077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Mercado Reyes M, Ailes E, Daza M, Tong VT, Osorio J, Valencia D, et al. Zika virus detection in amniotic fluid and Zika-associated birth defects. Am. J. Obstet. Gynecol. 222 (2020) 610.e1–610.e13. doi: 10.1016/j.ajog.2020.01.009. [DOI] [PMC free article] [PubMed]
- 234.de Paula Freitas B., de Oliveira Dias J.R., Prazeres J., Almeida Sacramento G., Icksang Ko A., Maia M., et al. Ocular findings in infants with microcephaly associated with presumed Zika virus congenital infection in Salvador, Brazil. JAMA Ophthalmol. 2016;134:529–535. doi: 10.1001/jamaophthalmol.2016.0267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Walker C.L., Ehinger N., Mason B., Oler E., Little M.-T.E., Ohuma E.O., et al. Ultrasound prediction of Zika virus-associated congenital injury using the profile of fetal growth. PLoS One. 2020;15 doi: 10.1371/journal.pone.0233023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Freitas D.A., Souza-Santos R., Carvalho L.M.A., Barros W.B., Neves L.M., Brasil P., et al. Congenital Zika syndrome: a systematic review. PLoS One. 2020;15:1–27. doi: 10.1371/journal.pone.0242367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Grant R., Fléchelles O., Tressières B., Dialo M., Elenga N., Mediamolle N., et al. In utero Zika virus exposure and neurodevelopment at 24 months in toddlers normocephalic at birth: a cohort study. BMC Med. 2021;19:1–11. doi: 10.1186/s12916-020-01888-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Holtzman M., Golden W.C., Sheffield J.S. Zika virus infection in the pregnant woman. Clin. Obstet. Gynecol. 2018;61:177–185. doi: 10.1097/GRF.0000000000000343. [DOI] [PubMed] [Google Scholar]
- 239.Pires L.C., Dantas L.R., Witkin S.S., Bertozzi A.P.A.P., de CAB Dezena R., Rodrigues M.M.D., et al. Knowledge of Zika virus transmission and its prevention among high-risk pregnant women in Brazil. Viruses. 2021;13:1–9. doi: 10.3390/v13020242. [DOI] [PMC free article] [PubMed] [Google Scholar]