Abstract
Enteric viruses, particularly rotaviruses and noroviruses, are a leading cause of gastroenteritis worldwide. Rotaviruses primarily affect young children, accounting for almost 40% of hospital admissions for diarrhoea and 200 000 deaths worldwide, with the majority of deaths occurring in developing countries. Two vaccines against rotavirus were licensed in 2006 and have been implemented in 95 countries as of April, 2018. Data from eight high-income and middle-income countries showed a 49–89% decline in rotavirus-associated hospital admissions and a 17–55% decline in all-cause gastroenteritis-associated hospital admissions among children younger than 5 years, within 2 years of vaccine introduction. Noroviruses affect people of all ages, and are a leading cause of foodborne disease and outbreaks of gastroenteritis worldwide. Prevention of norovirus infection relies on frequent hand hygiene, limiting contact with people who are infected with the virus, and disinfection of contaminated environmental surfaces. Norovirus vaccine candidates are in clinical trials; whether vaccines will provide durable protection against the range of genetically and antigenically diverse norovirus strains remains unknown. Treatment of viral gastroenteritis is based primarily on replacement of fluid and electrolytes.
Introduction
Acute gastroenteritis is a common illness that affects people of all ages, with potentially severe complications in young children and elderly people who are vulnerable to dehydration.1–4 Since their identification in the early 1970s, enteric viruses have been recognised as a leading cause of gastroenteritis worldwide. Of the major enteric viruses, rotaviruses have a predilection for young children, whereas noroviruses affect people of all ages. Viral gastroenteritis manifests abruptly with vomiting and watery diarrhoea, often accompanied by low-grade fever and abdominal cramps. To differentiate viral gastroenteritis from gastroenteritis caused by bacterial agents on the basis of clinical manifestations alone is often difficult, and laboratory tests are required to make a specific diagnosis. Viral gastroenteritis is generally self-limited with recovery within 2–5 days, and treatment efforts focus on maintaining adequate hydration.1–4 In this Seminar, we highlight advances in our understanding of viral gastroenteritis and its prevention and control.
Aetiology
Rotavirus
Rotaviruses belong to the Reoviridae family, and are characterised by a 70–75 nm non-enveloped multilayered virion with 11-segmented double-stranded RNA. Nine rotavirus species have been identified (referred to as rotavirus A to rotavirus I) and a tentative tenth species—rotavirus J—has been described.5–7 Humans are susceptible to infection with rotavirus A, B, C, and H; however, rotavirus A is by far the most medically important species worldwide.8 The rotavirus genome is approximately 18·5 kb pairs in length, and encodes 11 or 12 proteins, including six structural viral proteins (VP1 to VP4, VP6, and VP7) and five or six non-structural proteins (NSP1 to NSP5 or NSP6).8 VP7 (G antigen, glycoprotein) and VP4 (P antigen, protease sensitive) are located on the virus surface, functioning as cell-attachment proteins and independently eliciting neutralising antibodies. Genetically, VP7 is classified into G types and VP4 into P types. Rotavirus-type specificities show complex patterns of host-species distribution. In human beings, at least 14 G types and 17 P types of rotavirus A strains have been identified in more than 80 different combinations, but only six combinations (G1P[8], G2P[4], G3P[8], G4P[8], G9P[8], and G12P[8]) account for about 90% of severe infections requiring medical attention.9,10 These antigen combinations are expressed on two conserved viral backbone gene constellations—Wa-like and DS1-like. Most strains with G1P[8], G3P[8], G4P[8], G9P[8], and G12P[8] antigen combinations are expressed on the Wa-like genetic backbone, whereas the G2P[4] antigen combination tends to be associated with the DS1-like genetic backbone.8 However, some studies report an inverse association between the abovementioned backbone gene constellations and their preferably expressed neutralisation-antigen combinations. The sporadic detection of animal (mainly porcine, bovine, canine, feline, and lapine) rotavirus strains in humans suggests that host-specific barriers are permeable to some extent. In fact, some of the globally or regionally dispersed human strains (including G5, G8, G9, and G12) are thought to have originated from an animal host.8
Search strategy and selection criteria.
We searched PubMed, MEDLINE, and Embase for articles up until April 17, 2018, without restriction of language using the terms “epidemiology”, “disease burden”, “pathogenesis”, “immunity”, “genetics”, “clinical”, “diagnosis”, “therapy”, “prevention and control”, “virus discovery”, and “novel virus” in combination with each of the following terms: “viral gastroenteritis”, “viral diarrhea”, “gastroenteritis virus”, “rotavirus”, and “norovirus”. The primary approach we used was to review recently published titles and abstracts and then select the most relevant articles for inclusion into this Seminar. Additional references were also included from the selected articles if not identified through the main search strategy. When multiple articles discussed a particular topic or illustrated a specific point, typically, the most representative article was chosen.
Norovirus
Noroviruses belong to the Caliciviridae family and have a non-enveloped particle, which is 27–40 nm in diameter. The 7·5–7·7 kb positive-stranded RNA genome contains three open reading frames (ORFs): one ORF encodes a polyprotein that is post-translationally cleaved into non-structural proteins (NS1 to NS7), and the other two ORFs have a role in virion formation.11 The major capsid protein, VP1, with two domains (shell S and protruding P) forms the virion with the minor capsid protein VP2. Receptor binding sites are thought to be localised on the P domain. Genetic classification is based on VP1 and the RNA-dependent RNA polymerase (NS7).12 Human noroviruses can be classified into three genogroups and numerous genotypes within each genogroup (ie, nine GI, 22 GII, and one GIV). Further subclassification refers to new variants within a genotype; for example, at least seven variants of the most common norovirus genotype—GII.4—have been identified, all of which emerged after marked antigenic drift, and have caused pandemics over the past two decades with a typical interval of 2–3 years.13 A new globally emerging genotype—GII.17—has been described.14 Further diversification of noroviruses comes from recombination events that lead to new combinations of capsid and RNA-dependent RNA-polymerase types; these norovirus strains with recombinant genomes might have increased fitness and transmissibility over parental strains with shared capsid types.15 Typically, genotype specificity among human and animal noroviruses is not shared, and zoonotic transmission has not yet been firmly shown to occur.
Other viruses
In addition to rotaviruses and noroviruses, sapoviruses of the Caliciviridae family (with seven genotypes within genogroup GI, seven within GII, one within GIV, and two within GV), human astroviruses of the Astroviridae family (with eight serotypes, human astrovirus types 1–8), and human enteric adenoviruses of the Adenoviridae family (with two serotypes, HAdV40 and HAdV41) are among the medically important viruses causing gastroenteritis. Together, these agents account for about 10–20% of community diarrhoea cases in children, and are sometimes associated with outbreaks in semiclosed communities in all age groups. Additionally, if patients who are immunocompromised contract any of these viruses, they can develop severe, long-lasting diarrhoea that can occasionally be fatal.1,3,4
Depending on the setting and patient population, in up to 50% of acute gastroenteritis cases the causative agent can remain undiagnosed with laboratory methods.16,17 This low diagnostic proportion has prompted efforts towards pathogen discovery using tools such as broad-range PCR assays, pan-viral microarrays, and unbiased viral metagenomics.18 Novel members of the Picornaviridae (eg, salivirus, cosavirus, and Saffold virus), Astroviridae (commonly referred to as VA-like and MLB-like astroviruses), and Parvoviridae families (such as bocaviruses, tusaviruses, and bufaviruses) have been identified in patients with gastroenteritis using these methods. However, similarly to other putative enteric viruses that were identified a long time ago (eg, toroviruses, kobuviruses, and picobirnaviruses), the association between these viruses and gastroenteritis in humans has not been unequivocally confirmed.18
Disease burden and epidemiology
Rotavirus
In countries without rotavirus vaccination programmes, almost all children have at least one rotavirus infection by age 5 years. Infections in the first 3 months of life are generally milder, probably because of protection from maternal antibodies. Children aged 4–23 months are at greatest risk for severe rotavirus disease, which can include hospital admission. Immunity is incomplete at this age, and repeated infections frequently occur. For example, about 42% of Mexican children who were followed from birth had three or more rotavirus infections by age 2 years.19 However, immunity progressively increases with each subsequent infection, and thus symptoms are milder upon reinfection.19,20 Consequently, although adults can be infected with rotavirus, the disease is usually subclinical or mild.
Rotavirus causes more severe gastroenteritis than most other enteric pathogens. Therefore, the proportion of rotavirus infections that are detected in patients increases with increasing severity of gastroenteritis; these proportions range from 8–10% of diarrhoea episodes of all severity to almost 35–40% of diarrhoea episodes requiring hospital admission worldwide.21,22 Although infection and severe disease from rotavirus are prevalent worldwide, more than 90% of the approximate 200 000 annual deaths from rotavirus globally occur in developing countries, where access to rehydration therapy is poorer than in high-income countries.
Rotavirus gastroenteritis shows prominent winter seasonality in many developed countries, with the prevalence of infection substantially decreasing during summer months.23 In developing countries, however, rotavirus disease is less seasonal than in high-income countries, perhaps reflecting the greater intensity of exposure to rotavirus in these settings.
Norovirus
Noroviruses are the leading cause of acute gastroenteritis in people of all ages worldwide, and are estimated to cause 12–24% of community-based or clinic-based cases of acute gastroenteritis, 11–17% of emergency room or hospital cases, and approximately 70 000–200 000 deaths annually.24,25 In the USA alone, noroviruses cause on average 19–21 million illnesses, 56 000–71 000 hospital admissions, and 570–800 deaths annually (figure 1).26
Figure 1: Estimates of the annual burden and individual lifetime risks for norovirus disease across all age groups in the USA.

Ranges represent point estimates from different studies, not uncertainty bounds. Reproduced from Hall and colleagues.26
The incidence of norovirus gastroenteritis is greatest among young children. For example, in a study from England, the incidence of norovirus gastroenteritis was 21·4 episodes per 100 person-years among children younger than 5 years, compared with 3·3 episodes per 100 person-years among those older than 5 years.27 Notably, in some countries such as the USA and Finland, following the decline in rotavirus disease after rotavirus vaccine implementation, norovirus has emerged as the leading cause of severe paediatric gastroenteritis.28,29
Severe outcomes of norovirus gastroenteritis are also common among adults aged at least 65 years, who account for the majority of fatalities associated with norovirus.30,31 Among nursing-home residents in the USA, the number of hospital admissions and deaths from all causes were shown to be significantly elevated during periods with norovirus outbreaks in these facilities, compared with periods without outbreaks.32 Indeed, in US adults older than 65 years, norovirus is second only to Clostridioides difficile as a cause of death from gastroenteritis, with approximately 800 deaths reported annually.30 Patients who are immunocompromised following a transplant can become chronically infected, and this infection is difficult to clear before restoration of the immune system.
Noroviruses are shed in the faeces of people who are infected, and transmission is faecal–oral, either directly through contaminated hands or by ingestion of food and water that is contaminated with faecal matter. Several characteristics of noroviruses make them highly contagious, including their low infectious dose (around 18–1000 viral particles),33 the occurrence of viral shedding before the onset of clinical disease in about a third of patients, the extended duration of shedding following clinical illness,34,35 the stability of the virus in the environment across a wide range of temperatures (from freezing to 60°C) and their persistence in the environment for days,36 the absence of complete cross-protection against the range of genetically diverse strains, and the constant evolution of the virus leading to the emergence of new strains.
Noroviruses are the leading cause of outbreaks of acute gastroenteritis, including foodborne outbreaks. In developed countries, outbreak activity tends to increase during the cooler months of the year.25,37–39 Outbreaks are frequently reported in institutional settings such as hospitals, long-term care facilities, and schools because of the close contact among infected individuals.37 Outbreaks have been associated with food items contaminated with faecal matter at their source, especially consumption of oysters that can concentrate viruses from contaminated waters.40–42 However, food contamination often occurs from infected food handlers at the point of service, and ready-to-eat foods such as salads and sandwiches are particularly implicated in this type of transmission.43 Cyclical increases in norovirus outbreaks occur every few years, and often these increases result from the emergence of genetically distinct viral strains, against which population immunity is inadequate.44,45
Clinical presentation
Clinical presentation of viral gastroenteritis ranges from an asymptomatic infection to severe dehydrating diarrhoea. Asymptomatic viral carriage can be as high as 50% in some populations. In symptomatic infections, the incubation period is 24–72 h.1,2,46–48 The illness is most typically self-limited with recovery within 2–5 days in individuals who are immunocompetent; in those who are immunocompromised, viral gastroenteritis can last several weeks to months, or even years, irrespectively of the viral cause of infection. Resolution of chronic infection and disease in patients who have undergone a transplant and are immunosuppressed is associated with reconstitution of the immune system and the detection of serum antibodies that can block viral binding to histoblood antigens.49,50
The main features of viral gastroenteritis include non-bloody diarrhoea and vomiting. These symptoms can be accompanied by nausea, abdominal cramps, and fever. Typically, the duration of illness is shorter for a norovirus infection than a rotavirus infection. Nonetheless, viral gastroenteritis is often difficult to differentiate from gastroenteritis caused by enteric bacteria on the basis of clinical presentation alone; therefore, laboratory tests are required for adequate specific diagnosis.1 Dehydration is a severe complication that can lead to hypovolaemic shock, coma, and death. Disease severity depends on several factors, including viral pathogenicity, as well as the host’s genetics, comorbidities, and immune status.48 For example, outbreaks caused by GII.4 norovirus seem to cause more severe illness than those caused by non-GII.4 noroviruses. Additionally, patients who are immunocompromised or elderly develop more severe norovirus disease with greater risk of death. On the contrary, rotavirus infections that are of animal origin can go undetected in children because of their reduced virulence, and newborn babies are known to commonly undergo asymptomatic rotavirus infections in neonatal hospital units.47,51
Extraintestinal symptoms, such as respiratory symptoms or central nervous system manifestations in rotavirus infection, or headache, myalgia, and malaise in norovirus infection, are not unusual, but are difficult to interpret without a full understanding of the patient’s immune status, genetics, and comorbidities.
Pathogenesis
Rotavirus
Rotavirus infects intestinal villus enterocytes and enteroendocrine cells; crypt cells are spared. Rotavirus infection-associated diarrhoea is non-inflammatory and involves both malabsorptive and secretory mechanisms. Malabsorption occurs from virus-mediated destruction of absorptive enterocytes, downregulation of the expression of absorptive enzymes, and functional changes in tight junctions between enterocytes that lead to paracellular leakage. A secretory component of rotavirus infection-associated diarrhoea involves elevated intracellular Ca2+ concentration, which is important for the stimulation of Cl− secretion.52,53 Rotavirus replication in enterocytes and enteroendocrine cells stimulates serotonin release from human enterochromaffin cells, and activates brain regions associated with nausea and vomiting.54 Altered plasma-protein function and decreased Na+ absorption by inhibition of sodium–hydrogen exchanger activity also contribute to diarrhoea.55,56
A short period of viraemia can occur in a large proportion of children with severe rotavirus gastroenteritis, and virus can be detected in the non-intestinal tissues of those who are immunocompetent.57,58 The clinical consequences of systemic infection in children remain unclear, but could explain putative associations with conditions such a central nervous system disease, meningitis, and biliary atresia.
Norovirus
Information about norovirus pathogenesis, pathology, and immunity has been obtained primarily from volunteer studies, because human noroviruses remained non-cultivatable in intestinal epithelial cells until 2016, and no animal model fully recapitulates human disease after oral inoculation.
Histological changes are present in jejunal biopsy samples.34,35,59,60 Symptomatic illness correlates with broadening and blunting of intestinal villi, crypt-cell hyperplasia, cytoplasmic vacuolisation, and infiltration of polymorphonuclear and mononuclear cells into the lamina propria, with the mucosa remaining intact. The extent of small intestinal involvement remains unknown. Intestinal biopsy samples from children chronically infected with norovirus after small intestinal transplants show increased enterocyte apoptosis and inflammation that is difficult to distinguish from allograft rejection.61 Additionally, human norovirus antigen has been detected in villus enterocytes but not crypt cells of immunocompromised patients with chronic norovirus infection who had undergone haematopoietic-stem-cell or small-bowel transplantation.62 Norovirus infection is also associated with epithelial barrier dysfunction.63 Although noroviruses exhibit high species specificity, gnotobiotic piglets and calves can be infected orally with GII.4 human norovirus; once infected, these animals have mild diarrhoea and patchy infection of the intestine, primarily of the proximal small intestine (duodenal and jejunal) enterocytes.64
Experimental human infections show that small-intestinal brush-border enzymatic activities (alkaline phosphatase, sucrase, and trehalase) are decreased, resulting in mild steatorrhoea and transient carbohydrate malabsorption.59 Jejunal adenylate cyclase activity is not elevated, and transient gastric secretion of hydrochloric acid, pepsin, and intrinsic factor occurs.65 Gastric emptying is delayed and this, or the transient structural damage to the intestinal villi, might contribute to nausea and vomiting.66 Symptomatic illness might also be due to the host’s innate immune response to the viral infection, including responses to induced cytokines.67
Further understanding of human norovirus infection and pathogenesis is expected with the successful cultivation of these pathogens in novel human intestinal stem-cell-derived cultures called human intestinal enteroids.68 These non-transformed epithelial-cell cultures from different segments of the small intestine are multicellular, and enterocytes in these cultures support the replication of numerous viral strains; replication is strain dependent and replication of individual strains either requires or is dependent on the addition of bile to the cultures. This system can be used to evaluate virus inactivation and measure the amount of neutralising antibodies, and can allow a detailed understanding of virus–host interactions in these physiologically active cultures.
Immunity
Rotavirus
The first level of immunity to human rotaviruses is an innate susceptibility or resistance to infection based partially on an individual’s fucosyltransferase 2 (FUT2) genotype. FUT2 controls fucosylation and the expression of ABH histo-blood group antigens (HBGAs) at the intestinal mucosa. These carbohydrates function as binding ligands and are thought to be the initial receptors required for rotavirus binding to host cells. Susceptibility or resistance to a particular rotavirus strain depends on whether the cell surface in the gut expresses the appropriate binding glycan. Human rotaviruses bind to HBGAs, whereas most animal virus strains bind to sialoglycans. The distribution of HBGA types in different human populations affects the prevalence of common rotavirus genotypes across the globe, and susceptibility to some strains of animal origin.69–71 Innate immunity that regulates interferon and the induction of interferon-stimulated genes also has an important role in modulating rotavirus infection in vitro and in animal models,72 but its role in humans remains to be fully understood.
Additionally, acquired immunity is important to prevent both recurrent disease, and to a lesser extent, reinfection following primary infection.73 Protection generally positively correlates with markers of mucosal immunity, including concentrations of anti-rotavirus intestinal immunoglobulin (Ig)A, enteric rotavirus reactive antibody-secreting cells, and IgA memory cells. Studies in mice have shown that B cells act as the primary protection from reinfection, whereas CD8 T cells are responsible for shortening the course of primary infection.74 CD4 T cells are generally involved in supplying help to CD8 T cells and B cells, and they can also mediate active protection via an interferon-γ-dependent pathway after immunisation with recombinant VP6, the major structural protein in the virus.75 Lymphocyte homing also has a prominent role in regulating rotavirus immunity, and B-cell trafficking to the intestine seems important to generate optimal protection in a chronically infected mouse model.76 Neutralising antibody targeting VP4 or VP7 can transfer either homotypic or heterotypic protection. Non-neutralising IgA antibodies targeting VP6 can also mediate protection, apparently via an intracellular antiviral effect that occurs during transcytosis.77 Viral-induced diarrhoea is reduced in mouse pups born to dams immunised with the enterotoxin NSP4.52,78 The role, if any, of these protective mechanisms in human immunity is unknown, and a correlate of protection remains to be established.
Norovirus
Immunity to human noroviruses is first based on an innate genetic susceptibility or resistance to infection based on the presence of host HBGAs that serve as cellular attachment factors required to establish infection and clinical disease.79 Many human noroviruses bind to HBGAs present on the surface of epithelial cells, but noroviruses cannot bind to the cells of individuals in whom these antigens are absent (ie, non-secretors).80 The absent expression of secretor-type HBGAs on the gut epithelium is associated with resistance to infection with GI.1 and most GII.4 strains.81–83 HBGAs bind noroviruses in a strain-specific manner.84
The contribution of adaptive immunity to protection began to be understood once genetic resistance was recognised. Serum antibody concentrations measured by ELISA do not reliably predict susceptibility or resistance to illness, because these assays measure antibodies that simply bind to particles. Instead, higher concentrations of pre-existing serum antibody that block viral binding to HBGAs are associated with a decreased risk of illness in individuals who are secretor positive, and who have been exposed to GI.1 or GII.4 virus in epidemiological studies.85–87 HBGA-blocking assays are considered to measure functional antibody, which is a surrogate for neutralising antibody and a correlate of immunity. Having the ability to measure functional antibody that correlates with protection facilitates the evaluation of norovirus vaccines. Norovirus infection during adulthood leads to an increase in circulating, virus-specific, IgG-producing and IgA-producing plasmablasts 1 week after infection, and virus-specific memory B-cell concentrations then peak at 2 weeks. Pre-existing serum IgA and IgG and high concentrations of IgG memory B cells at the time of virus exposure are also associated with a lower risk of illness.88 Maternal antibodies help protect children from norovirus infection, and children subsequently develop antibodies with high blocking activity.89 Norovirus-blocking antibodies persist for at least 6 months in adults and children.87,89 T-cell responses occur after infection, but their relative importance is less well characterised than that of B cells and antibodies.90 The contribution of past infection with heterologous strains to protection from infection from new strains remains to be fully understood; heterotypic HBGA-blocking responses can be observed in both adults and young children, but some evidence suggests primary infection of children might not result in protection from infection with other strains.86,90,91
Laboratory diagnosis
Routine aetiological testing of patients presenting for treatment of acute gastroenteritis is not required. Testing could help guide specific treatment in patients with severe gastroenteritis, particularly those with bloody stools, illness associated with travel, prolonged illness, or in patients with underlying chronic conditions.
Rotavirus
Laboratory diagnosis of rotavirus infection relies on direct viral detection from faecal specimens, which can contain up to 1 × 1010 viral particles per gram of stool. Commercially available enzyme immunoassay tests show high specificity and sensitivity (>90% each), and are the most commonly used assay format in clinical and public health laboratories. Other antigen detection methods, such as latex agglutination and lateral flow immunoassays are available for point-of-care diagnosis.46 Importantly, detection of rotavirus antigen correlates better with the presence of illness than with the detection of viral nucleic acids (eg, by RT-PCR). When viral nucleic acid detection is used, subclinical infections are also identified.
New-generation molecular detection methods that rely on expensive laboratory equipment and target multiple gastroenteritis pathogens simultaneously show high sensitivity and specificity for rotaviruses (>90% each).92–94
Norovirus
Direct virus detection, in particular viral-RNA detection methods, are primarily used for diagnosis of norovirus infection. End-point RT-PCR and real-time RT-PCR assays that target the genomic region bridging the RNA polymerase and major capsid protein-coding region are among the most broadly used detection methods. Sequencing of this short amplified region provides information about genotypes that is useful in molecular epidemiological studies.95 Norovirus also serves as a target in multiplexed molecular detection assays.93,94
Antigen-detection assays usually contain a mixture of norovirus-specific antibodies and show some variation in sensitivity because of the large number of antigen types. Rapid antigen-detection tests have been commercialised for point-of-care diagnosis. Additionally, enzyme immunoassays are available and show similar specificity and sensitivity with immunochromatography tests.95 Although the overall sensitivity is low (up to 52% for genogroup GI norovirus and up to 78% for the prevailing GII.4 noroviruses), in outbreak settings where multiple samples are available for testing, this low sensitivity could be acceptable.
Treatment
For treatment of dehydration caused by gastroenteritis viruses, the administration of reduced-osmolality oral-rehydration solutions (50–60 mmol/L sodium) is the first line of therapy. Profuse vomiting, worsening dehydration with altered consciousness, or severe acidosis, hypovolaemic shock, abdominal distension, and ileus are indications for intravenous rehydration.96
Probiotics (eg, Lactobacillus rhamnosus strain GG, Lactobacillus reuteri, and Saccharomyces boulardii) reduce the duration of diarrhoea and can be considered as an adjunct to rehydration therapy.97 Breastfeeding should continue in infants, and early resumption of feeding after rehydration is recommended. Zinc supplementation is recommended for children younger than 6 months in developing countries, but its value in developed countries, where zinc deficiencies are rare, is unknown.98
Antimotility drugs are not recommended. Antisecretory drugs and some drugs with broad antiviral effects have been shown to reduce the duration of diarrhoea in some studies; however, the data are either non-conclusive or not sufficient to recommend the routine use of these drugs in children.99–102 Additionally, selected antiemetics that reduce vomiting are available but are not universally recommended, because they might increase the frequency of diarrhoea and have other side-effects.
Oral administration of antibody preparations has shown some benefit in patients who have been admitted to hospital with rotavirus and norovirus, including patients who are immunocompromised, but are not routinely used.103–108
Prevention
Rotavirus
Since 2006, two rotavirus vaccines with similar efficacy and safety have been licensed—a pentavalent bovine–human reassortant vaccine (RotaTeq, Merck, West Point, PA, USA) and a monovalent human vaccine (Rotarix, GlaxoSmithKline Biologicals, Rixensart, Belgium; table 1). As of April, 2018, routine vaccination of infants against rotavirus has been implemented in 95 countries globally (figure 2).109,110 After vaccine implementation, substantial declines in the incidence of severe rotavirus gastroenteritis have been documented in many countries.111,112 For example, data obtained over nearly a decade following vaccine implementation in the USA show delayed, shorter rotavirus seasons with markedly fewer cases compared with the years before the vaccine programme was implemented (figure 3).111 In several countries, reductions in severe gastroenteritis have been documented in children who are unvaccinated, and even older adults, suggesting that vaccination of young infants might be indirectly protecting these groups by reducing rotavirus transmission in the community.114 Most importantly, declines in childhood deaths from diarrhoea have been documented in several countries after rotavirus vaccine introduction, reaffirming the life-saving potential of these vaccines.115,116
Table 1:
Globally and nationally licensed live oral rotavirus vaccines
| Number of doses | Vaccine strains (genotypes) | Year of introduction | Availability | Efficacy in phase 3 vaccine trials | Comments | |
|---|---|---|---|---|---|---|
|
| ||||||
| RotaTeq (Merck, West Point, PA, USA) | Three doses | Pentavalent bovine-human reassortant strains: WC3 × WI79 (G1P[5]); WC3 × SC-2 (G2P[5]); WC3 × WI78 (G3P[5]); WC3 × BrB (G4P[5]); and WC3 × WI79 (G6P[8]) | 2006 | Worldwide | 51–64% in developing countries and 94-100% in developed countries | Available in more than 100 countries, and recommended by WHO for global use |
| Rotarix (GlaxoSmithKline, Rixensart, Belgium) | Two doses | Monovalent attenuated human strain: 89–12 (G1P[8]) | 2006 | Worldwide | 49–72% in developing countries and 72–97% in developed countries | Available in more than 100 countries, and recommended by WHO for global use |
| Lanzhou lamb rotavirus vaccine (Lanzhou Institute of Biological Products, Lanzhou, China) | Multiple doses | Monovalent ovine rotavirus strain: LLR-85 (G10P[12]) | 2000 | China | Not applicable | Vaccine licensed based on immunogenicity data from phase 2 trials; post-licensure studies show effectiveness of 35–77%; a trivalent reassortant vaccine candidate that uses the vaccine strain is currently in a phase 3 trial |
| Rotavin-M1 (PolyVac, Hanoi, Vietnam) | Two doses | Monovalent attenuated human strain: KH0118–2003 (G1P[8]) | 2012 | Vietnam | Not applicable | Vaccine licensed based on immunogenicity data from phase 2 trials |
| ROTAVAC (Bharat Biotech International, Hyderabad, India) | Three doses | Monovalent neonatal strain: 116E (G9P[11]) | 2014 | Low-income countries and some middle-income countries worldwide | 56% in India | Vaccine has been implemented in routine immunisation of nine Indian states, which constitute about 50% of the birth cohort; WHO prequalified for global use in 2018 |
| ROTASIIL (Serum Institute of India, Pune, India) | Three doses | Pentavalent bovine-human reassortant strains: UK × D (G1P[5]); UK × DS1 (G2P[5]); UK × P (G3P[5]); UK × ST3 (G4P[5]); and UK × AU32 (G9P[5]) | 2017 | India | 66% in Niger and 39% in India | Vaccine administered out of the cold chain in clinical trial; under consideration by WHO for global recommendation |
Figure 2: Current rotavirus vaccine introduction map.

As of April, 2018, 95 countries have introduced rotavirus vaccines; this includes 88 national introductions, three ongoing phased introductions (Pakistan, India, and Philippines), and four pilot or sub-national introductions (Canada, Italy, Sweden, and Thailand). Data are from the ROTA council.109
Figure 3: Percentage of rotavirus tests with positive results for the different weeks of the year starting in July and finishing in June, USA, 2000–15.
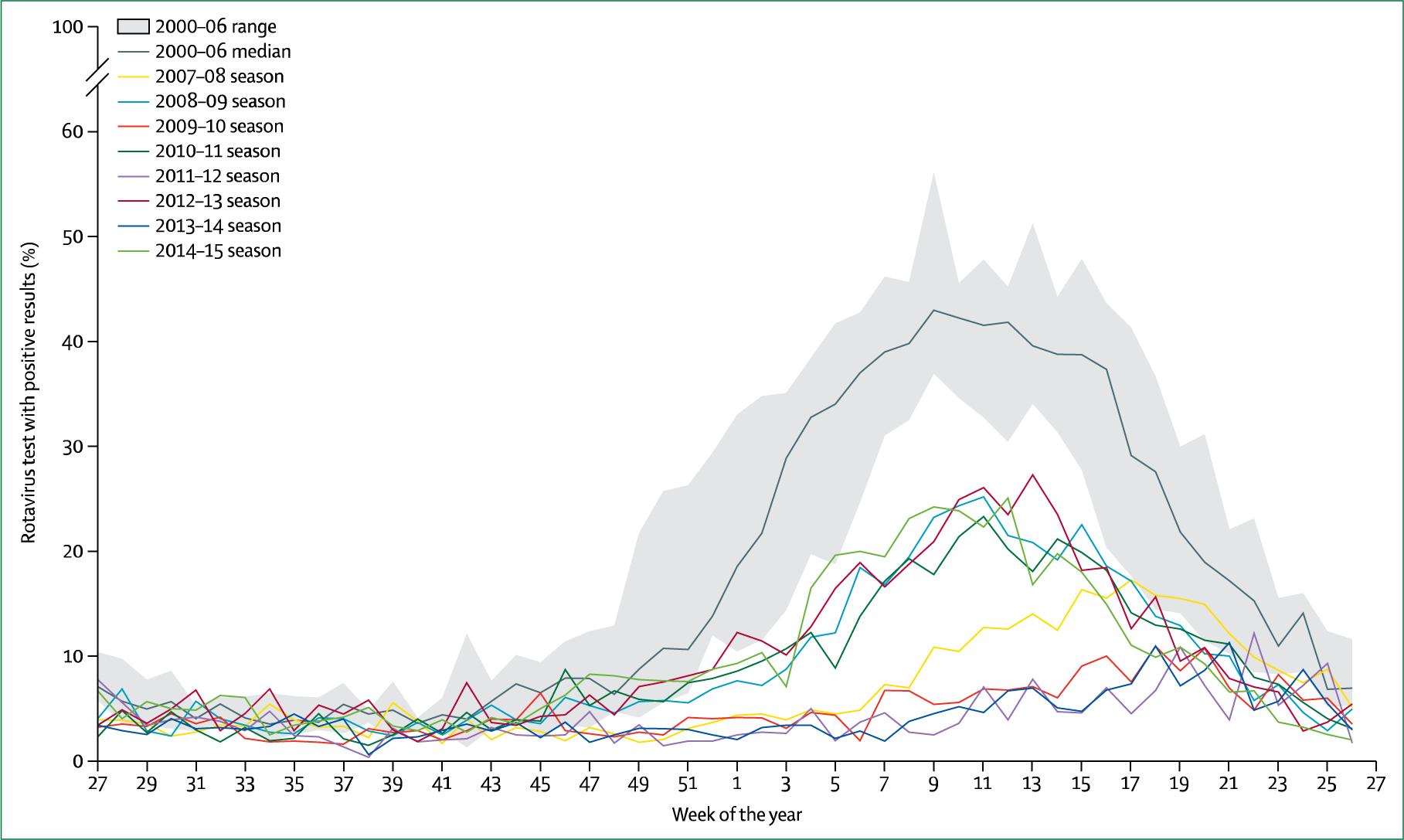
The maximum and minimum percentage of rotavirus-positive tests for 2000–06 could have occurred during any of these six baseline seasons. Since the introduction of a rotavirus vaccine in the USA, epidemics have occurred every other year (ie, 2008–09, 2010–11, 2012–13, and 2014–15). Data were obtained from the US Centers for Disease Control and Prevention National Respiratory and Enteric Virus Surveillance System.113
Because rotavirus vaccines are orally administered, concerns were raised as to whether factors such as co-infection with other enteric pathogens, concurrent illnesses, or malnutrition might adversely effect the performance of rotavirus vaccines in developing countries.117 Because of these concerns, global recommendations for rotavirus vaccine use were only issued in 2009, after randomised trials had shown their efficacy in developing countries.118–120 Although rotavirus vaccines showed lower vaccine efficacy in developing countries than developed countries (50–64% vs 85–98%), the burden of disease prevented by vaccination is greater in developing countries, because of the markedly greater baseline incidence of severe rotavirus disease. Initial results from African countries that have implemented routine rotavirus vaccination have reaffirmed the public health value of rotavirus vaccines.121,122
Several high-income and middle-income countries have identified a low risk of intussusception—a form of bowel obstruction—with rotavirus vaccination, which occurs at a rate of about one to five excess cases per 100 000 vaccinated infants.123–128 Because this risk is relatively low compared with the numerous health benefits of vaccination, global-level and country-level policy makers have not recommended any changes to the rotavirus vaccine policy.123,124,129
In addition to the two globally licensed vaccines, several vaccines are being developed by emerging-market manufacturers (table 1). An indigenously manufactured rotavirus vaccine was licensed in India in 2014 after showing 56% efficacy in a trial (ROTAVAC, Bharat Biotech, Hyderabad, India).130,131 As of September, 2017, Rotavac has been implemented in nine states in India, which account for about 50% of the birth cohort. Another indigenously manufactured rotavirus vaccine licensed in India (Rotasiil, Serum Institute of India, Pune, India) showed 66% efficacy in a clinical trial in Niger.132 In early 2018, Rotavac was prequalified by WHO, allowing its procurement by UNICEF for global use; Rotasiil is also being reviewed for prequalification by WHO. Additional vaccine candidates, including both live-attenuated and subunit vaccines are being tested (table 2), and the plasmid-based reverse-genetics system offers extended opportunities to design new generations of rotaviruses.133,135
Table 2:
| Name, design, and target neutralisation antigens | Vaccination route (features) | Developer or manufacturer | |
|---|---|---|---|
|
| |||
| Rotavirus | |||
| Clinical phase 3 | Lanzhou lamb rotavirus reassortants; lamb rotavirus-based reassortants carrying human rotavirus antigens; and G2 to G4 specificity | Oral (live attenuated) | Lanzhou Institute of Biological Products, Lanzhou, China |
| Clinical phase 3 | RotaShield; rhesus-human reassortant strains and tetravalent; and G1 to G4 specificity | Oral (live attenuated) | International Medica Foundation, Rochester, MN, USA |
| Clinical phase 3 | UK-BRV tetravalent; and G1 to G4 specificity | Oral (live attenuated) | Shantha Biotech, Hyderabad, India |
| Clinical phase 2b | RV3 neonatal strain; and G3P6 specificity | Oral (live attenuated) | Murdoch Children’s Research Institute, Parkville, VIC, Australia and PT Biofarma, Bandung, Indonesia |
| Clinical phase 2 | Subunit; truncated VP4 protein; and P4, P6, and P8 specificity | Intramuscular (subunit) | PATH, Seattle, WA, USA |
| Clinical phase 1 | UK-BRV reassortant; and G1 to G4, and G9 specificity | Oral (live attenuated) | Instituto Butantan, Sao Paulo, Brazil |
| Clinical phase 1 | UK-BRV reassortant; and G1 to G4, G8, and G9 specificity | Oral (live attenuated) | Wuhan Institute of Biological Products, Wuhan, China and PATH, Seattle, WA, USA |
| Norovirus | |||
| Clinical phase 2b | Norovirus virus-like particles; and GI.1 and GII.4 | Intramuscular | Takeda Vaccines, Cambridge, MA, USA |
| Clinical phase 1 | Recombinant adenovirus expressing norovirus VP1; and GI.l | Oral | Vaxart, San Fransisco, CA, USA |
Norovirus
Limiting contact with infected people during illness and for 1–2 days after illness resolution and frequent handwashing diminishes the transmission of norovirus.136,137 Exclusion from work during illness and for at least 48 h after illness resolution is recommended for individuals with a high propensity to transmit the virus, such as ill staff in health-care facilities, and people involved in food handling. Thorough disinfection of contaminated surfaces with a chlorine bleach solution at a concentration of 1000–5000 ppm (1:50–1:10 dilution of household bleach [5·25%]) or another approved disinfectant decreases the environmental spread of the virus.138
The high burden of norovirus gastroenteritis, particularly associated with severe outcomes in both children and elderly people, has led to efforts to develop vaccines for the prevention of endemic norovirus disease. Groups with a high propensity to transmit norovirus, such as food handlers and health-care workers, might also be candidates for a vaccine. Vaccines based on virus-like particles (VLPs) are in phase 2 clinical trials, and vaccines based on recombinant adenoviruses expressing norovirus VP1 protein are in phase 1 clinical trials (table 2).134
Volunteers who were intranasally administered a monovalent GI.1 VLP vaccine and challenged with a homologous strain showed that vaccine efficacy was 47%, and the vaccine reduced the severity of illness compared with a placebo.139 Volunteers who were intramuscularly administered a bivalent GI.1 and GII.4 VLP vaccine,140 with the GII.4 component including a consensus sequence from three different GII.4 variants, showed a reduction in disease severity when subsequently challenged with a GII.4 norovirus strain. Although these results are promising, many key issues require further study, including the understanding of the immune correlates of protection, assessment of the duration of protection from vaccination, and the amount of heterotypic protection against antigenically distinct strains, particularly given the continuing and rapid viral evolution leading to the emergence of novel norovirus strains.
Conclusions
Viral gastroenteritis remains a major public health concern because of its global societal and economic burden. Rotaviruses and noroviruses are the most medically important gastroenteritis viruses in childhood. Additionally, norovirus is commonly implicated in gastroenteritis outbreaks in semiclosed communities, and is the leading foodborne gastroenteritis virus globally. The high disease burden caused by these viruses has led to vaccine development, and after three decades of clinical trials, rotavirus vaccines have become a reality, and are available for use in more than 100 countries worldwide. Norovirus vaccine candidates are in clinical trials, but preliminary data remind us that genetic and antigenic diversity within noroviruses could pose challenges for the development of a norovirus vaccine that will elicit cross-protective clinical immunity.
Acknowledgments
KB is supported by the Momentum Program awarded by the Hungarian Academy of Sciences. MKE is supported by public-health service grants (AI080656, AI105101, and AI057788) from the National Institutes of Health. VM is supported by a grant from the University of Bari (2017–University of Bari–PRIN 2015). The findings and conclusions in this report are our own and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Footnotes
Declaration of interests
MKE is named as an inventor on patents related to cloning of the Norwalk virus genome, and has received consultant fees from Takeda Vaccines. KB, VM, and UDP declare no competing interests.
Contributor Information
Krisztián Bányai, Institute for Veterinary Medical Research, Centre for Agricultural Research, Hungarian Academy of Sciences, Budapest, Hungary.
Mary K Estes, Department of Molecular Virology and Microbiology, Baylor College of Medicine, Houston, TX, USA.
Vito Martella, Department of Veterinary Medicine, University Aldo Moro of Bari, Provincial Road to Casamassima, Valenzano, Italy.
Umesh D Parashar, Viral Gastroenteritis Branch, Division of Viral Diseases, Centers for Disease Control and Prevention, Atlanta, GA, USA.
References
- 1.Blacklow NR, Greenberg HB. Viral gastroenteritis. N Engl J Med 1991; 325: 252–64. [DOI] [PubMed] [Google Scholar]
- 2.Glass RI, Parashar UD, Estes MK. Norovirus gastroenteritis. N Engl J Med 2009; 361: 1776–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bosch A, Pintó RM, Guix S. Human astroviruses. Clin Microbiol Rev 2014; 27: 1048–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Oka T, Wang Q, Katayama K, Saif LJ. Comprehensive review of human sapoviruses. Clin Microbiol Rev 2015; 28: 32–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Matthijnssens J, Otto PH, Ciarlet M, Desselberger U, Van Ranst M, Johne R. VP6-sequence-based cutoff values as a criterion for rotavirus species demarcation. Arch Virol 2012; 157: 1177–82. [DOI] [PubMed] [Google Scholar]
- 6.Mihalov-Kovács E, Gellért Á, Marton S, et al. Candidate new rotavirus species in sheltered dogs, Hungary. Emerg Infect Dis 2015; 21: 660–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bányai K, Kemenesi G, Budinski I, et al. Candidate new rotavirus species in Schreiber’s bats, Serbia. Infect Genet Evol 2017; 48: 19–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dóró R, Farkas SL, Martella V, Bányai K. Zoonotic transmission of rotavirus: surveillance and control. Expert Rev Anti Infect Ther 2015; 13: 1337–50. [DOI] [PubMed] [Google Scholar]
- 9.Bányai K, László B, Duque J, et al. Systematic review of regional and temporal trends in global rotavirus strain diversity in the pre rotavirus vaccine era: insights for understanding the impact of rotavirus vaccination programs. Vaccine 2012; 30 (suppl 1): A122–30. [DOI] [PubMed] [Google Scholar]
- 10.Dóró R, László B, Martella V, et al. Review of global rotavirus strain prevalence data from six years post vaccine licensure surveillance: is there evidence of strain selection from vaccine pressure? Infect Genet Evol 2014; 28: 446–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Thorne LG, Goodfellow IG. Norovirus gene expression and replication. J Gen Virol 2014; 95: 278–91. [DOI] [PubMed] [Google Scholar]
- 12.Kroneman A, Vega E, Vennema H, et al. Proposal for a unified norovirus nomenclature and genotyping. Arch Virol 2013; 158: 2059–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Karst SM, Baric RS. What is the reservoir of emergent human norovirus strains? J Virol 2015; 89: 5756–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.de Graaf M, van Beek J, Vennema H, et al. Emergence of a novel GII.17 norovirus—end of the GII.4 era? Euro Surveill 2015; 20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.de Graaf M, van Beek J, Koopmans MP. Human norovirus transmission and evolution in a changing world. Nat Rev Microbiol 2016; 14: 421–33. [DOI] [PubMed] [Google Scholar]
- 16.Simpson R, Aliyu S, Iturriza-Gómara M, Desselberger U, Gray J. Infantile viral gastroenteritis: on the way to closing the diagnostic gap. J Med Virol 2003; 70: 258–62. [DOI] [PubMed] [Google Scholar]
- 17.Vocale C, Rimoldi SG, Pagani C, et al. Comparative evaluation of the new xTAG GPP multiplex assay in the laboratory diagnosis of acute gastroenteritis. Clinical assessment and potential application from a multicentre Italian study. Int J Infect Dis 2015; 34: 33–37. [DOI] [PubMed] [Google Scholar]
- 18.Oude Munnink BB, van der Hoek L. Viruses causing gastroenteritis: The known, the new and those beyond. Viruses 2016; 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Velazquez FR, Matson DO, Calva JJ, et al. Rotavirus infections in infants as protection against subsequent infections. N Engl J Med 1996; 335: 1022–28. [DOI] [PubMed] [Google Scholar]
- 20.Gladstone BP, Ramani S, Mukhopadhya I, et al. Protective effect of natural rotavirus infection in an Indian birth cohort. N Engl J Med 2011; 365: 337–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tate JE, Burton AH, Boschi-Pinto C, et al. 2008. estimate of worldwide rotavirus-associated mortality in children younger than 5 years before the introduction of universal rotavirus vaccination programmes: a systematic review and meta-analysis. Lancet Infect Dis 2012; 12: 136–41. [DOI] [PubMed] [Google Scholar]
- 22.Lanata CF, Fischer-Walker CL, Olascoaga AC, et al. Global causes of diarrheal disease mortality in children <5 years of age: a systematic review. PLoS One 2013; 8: e72788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Patel MM, Pitzer VE, Alonso WJ, et al. Global seasonality of rotavirus disease. Pediatr Infect Dis J 2013; 32: e134–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ahmed SM, Hall AJ, Robinson AE, et al. Global prevalence of norovirus in cases of gastroenteritis: a systematic review and meta-analysis. Lancet Infect Dis 2014; 14: 725–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Patel MM, Hall AJ, Vinje J, Parashar UD. Noroviruses: a comprehensive review. J Clin Virol 2009; 44: 1–8. [DOI] [PubMed] [Google Scholar]
- 26.Hall AJ, Lopman BA, Payne DC, et al. Norovirus disease in the United States. Emerg Infect Dis 2013; 19: 1198–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Phillips G, Tam CC, Conti S, et al. Community incidence of norovirus-associated infectious intestinal disease in England: improved estimates using viral load for norovirus diagnosis. Am J Epidemiol 2010; 171: 1014–22. [DOI] [PubMed] [Google Scholar]
- 28.Payne DC, Vinje J, Szilagyi PG, et al. Norovirus and medically attended gastroenteritis in U.S. children. N Engl J Med 2013; 368: 1121–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hemming M, Rasanen S, Huhti L, Paloniemi M, Salminen M, Vesikari T. Major reduction of rotavirus, but not norovirus, gastroenteritis in children seen in hospital after the introduction of RotaTeq vaccine into the National Immunization Programme in Finland. Eur J Pediatr 2013; 172: 739–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hall AJ, Curns AT, McDonald LC, Parashar UD, Lopman BA. The roles of clostridium difficile and norovirus among gastroenteritis-associated deaths in the United States, 1999–2007. Clin Infect Dis 2012; 55: 216–23. [DOI] [PubMed] [Google Scholar]
- 31.Harris JP, Edmunds JW, Pebody RG, Brown DW, Lopman BA. Deaths from norovirus among the elderly, England and Wales. Emerg Infect Dis 2008; 14: 1546–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Trivedi TK, DeSalvo T, Lee L, et al. Hospitalizations and mortality associated with norovirus outbreaks in nursing homes, 2009–2010. J Am Med Assoc 2012; 308: 1668–75. [DOI] [PubMed] [Google Scholar]
- 33.Teunis PF, Moe CL, Liu P, et al. Norwalk virus: How infectious is it? J Med Virol 2008; 80: 1468–76. [DOI] [PubMed] [Google Scholar]
- 34.Graham DY, Jiang X, Tanaka T, Opekun AR, Madore HP, Estes MK. Norwalk virus infection of volunteers: new insights based on improved assays. J Infect Dis 1994; 170: 34–43. [DOI] [PubMed] [Google Scholar]
- 35.Atmar RL, Opekun AR, Gilger MA, et al. Norwalk virus shedding after experimental human infection. Emerg Infect Dis 2008; 14: 1553–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Duizer E, Bijkerk P, Rockx B, De Groot A, Twisk F, Koopmans M. Inactivation of caliciviruses. Appl Environ Microbiol 2004; 70: 4538–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hall AJ, Wikswo ME, Manikonda K, Roberts VA, Yoder JS, Gould LH. Acute gastroenteritis surveillance through the National Outbreak Reporting System, United States. Emerg Infect Dis 2013; 19: 1305–09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ahmed SM, Lopman BA, Levy K. A systematic review and meta-analysis of the global seasonality of norovirus. PLoS One 2013; 8: e75922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hall AJ, Wikswo ME, Pringle K, Gould LH, Parashar UD. Vital signs: foodborne norovirus outbreaks—United States, 2009–2012. Morb Mortal Wkly Rep 2014; 63: 491–95. [PMC free article] [PubMed] [Google Scholar]
- 40.Westrell T, Dusch V, Ethelberg S, et al. Norovirus outbreaks linked to oyster consumption in the United Kingdom, Norway, France, Sweden and Denmark, 2010. Euro Surveill 2010; 15: 19524. [PubMed] [Google Scholar]
- 41.Falkenhorst G, Krusell L, Lisby M, Madsen SB, Bottiger B, Molbak K. Imported frozen raspberries cause a series of norovirus outbreaks in Denmark, 2005. Euro Surveill 2005; 10: E050922.2. [DOI] [PubMed] [Google Scholar]
- 42.Malek M, Barzilay E, Kramer A, et al. Outbreak of norovirus infection among river rafters associated with packaged delicatessen meat, Grand Canyon, 2005. Clin Infect Dis 2009; 48: 31–37. [DOI] [PubMed] [Google Scholar]
- 43.Hall AJ, Eisenbart VG, Etingue AL, Gould LH, Lopman BA, Parashar UD. Epidemiology of foodborne norovirus outbreaks, United States, 2001–2008. Emerg Infect Dis 2012; 18: 1566–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zheng DP, Widdowson MA, Glass RI, Vinje J. Molecular epidemiology of genogroup II-genotype 4 noroviruses in the United States between 1994 and 2006. J Clin Microbiol 2010; 48: 168–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cannon JL, Lindesmith LC, Donaldson EF, Saxe L, Baric RS, Vinje J. Herd immunity to GII.4 noroviruses is supported by outbreak patient sera. J Virol 2009; 83: 5363–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Esona MD, Gautam R. Rotavirus. Clin Lab Med 2015; 35: 363–91. [DOI] [PubMed] [Google Scholar]
- 47.Robilotti E, Deresinski S, Pinsky BA. Norovirus. Clin Microbiol Rev 2015; 28: 134–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Desselbrerger U Viral gastroenteritis. Medicine 2017; 45: 690–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Knoll BM, Lindesmith LC, Yount BL, Baric RS, Marty FM. Resolution of diarrhea in an immunocompromised patient with chronic norovirus gastroenteritis correlates with constitution of specific antibody blockade titer. Infection 2016; 44: 551–54. [DOI] [PubMed] [Google Scholar]
- 50.Saif MA, Bonney DK, Bigger B, et al. Chronic norovirus infection in pediatric hematopoietic stem cell transplant recipients: a cause of prolonged intestinal failure requiring intensive nutritional support. Pediatr Transplant 2011; 15: 505–09. [DOI] [PubMed] [Google Scholar]
- 51.Gentsch JR, Laird AR, Bielfelt B, et al. Serotype diversity and reassortment between human and animal rotavirus strains: implications for rotavirus vaccine programs. J Infect Dis 2005; 192 (suppl 1): S146–59. [DOI] [PubMed] [Google Scholar]
- 52.Ball JM, Tian P, Zeng CQ, Morris AP, Estes MK. Age-dependent diarrhea induced by a rotaviral nonstructural glycoprotein. Science 1996; 272: 101–04. [DOI] [PubMed] [Google Scholar]
- 53.Lorrot M, Vasseur M. How do the rotavirus NSP4 and bacterial enterotoxins lead differently to diarrhea? Virol J 2007; 4: 31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hagbom M, Istrate C, Engblom D, et al. Rotavirus stimulates release of serotonin (5-HT) from human enterochromaffin cells and activates brain structures involved in nausea and vomiting. PLoS Pathog 2011; 7: e1002115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hagbom M, Sharma S, Lundgren O, Svensson L. Towards a human rotavirus disease model. Curr Opin Virol 2012; 2: 408–18. [DOI] [PubMed] [Google Scholar]
- 56.Zachos NC, Foulke-Abel J, Biswas RS, et al. Rotavirus inhibits NHE3 activity via clathrin-independent endocytosis resulting in increased NHE3 degradation. Gastroenterology 2014; 146. [Google Scholar]
- 57.Blutt SE, Kirkwood CD, Parreño V, et al. Rotavirus antigenaemia and viraemia: a common event? Lancet 2003; 362: 1445–49. [DOI] [PubMed] [Google Scholar]
- 58.Blutt SE, Conner ME. Rotavirus: to the gut and beyond! Curr Opin Gastroenterol 2007; 23: 39–43. [DOI] [PubMed] [Google Scholar]
- 59.Agus SG, Dolin R, Wyatt RG, Tousimis AJ, Northrup RS. Acute infectious nonbacterial gastroenteritis: intestinal histopathology. Histologic and enzymatic alterations during illness produced by the Norwalk agent in man. Ann Intern Med 1973; 79: 18–25. [DOI] [PubMed] [Google Scholar]
- 60.Widerlite L, Trier JS, Blacklow NR, Schreiber DS. Structure of the gastric mucosa in acute infectious bacterial gastroenteritis. Gastroenterology 1975; 68: 425–30. [PubMed] [Google Scholar]
- 61.Kaufman SS, Chatterjee NK, Fuschino ME, et al. Characteristics of human calicivirus enteritis in intestinal transplant recipients. J Pediatr Gastroenterol Nutr 2005; 40: 328–33. [DOI] [PubMed] [Google Scholar]
- 62.Karandikar UC, Crawford SE, Ajami NJ, et al. Detection of human norovirus in intestinal biopsies from immunocompromised transplant patients. J Gen Virol 2016; 97: 2291–300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Troeger H, Loddenkemper C, Schneider T, et al. Structural and functional changes of the duodenum in human norovirus infection. Gut 2009; 58: 1070–77. [DOI] [PubMed] [Google Scholar]
- 64.Cheetham S, Souza M, Meulia T, Grimes S, Han MG, Saif LJ. Pathogenesis of a genogroup II human norovirus in gnotobiotic pigs. J Virol 2006; 80: 10372–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Levy AG, Widerlite L, Schwartz CJ, et al. Jejunal adenylate cyclase activity in human subjects during viral gastroenteritis. Gastroenterology 1976; 70: 321–25. [PubMed] [Google Scholar]
- 66.Meeroff JC, Schreiber DS, Trier JS, Blacklow NR. Abnormal gastric motor function in viral gastroenteritis. Ann Intern Med 1980; 92: 370–73. [DOI] [PubMed] [Google Scholar]
- 67.Newman KL, Moe CL, Kirby AE, Flanders WD, Parkos CA, Leon JS. Norovirus in symptomatic and asymptomatic individuals: cytokines and viral shedding. Clin Exp Immunol 2016; 184: 347–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ettayebi K, Crawford SE, Murakami K, et al. Replication of human noroviruses in stem cell-derived human enteroids. Science 2016; 353: 1387–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hu L, Crawford SE, Czako R, et al. Cell attachment protein VP8* of a human rotavirus specifically interacts with A-type histo-blood group antigen. Nature 2012; 485: 256–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Nordgren J, Sharma S, Bucardo F, et al. Both Lewis and secretor status mediate susceptibility to rotavirus infections in a rotavirus genotype-dependent manner. Clin Infect Dis 2014; 59: 1567–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Böhm R, Fleming FE, Maggioni A, et al. Revisiting the role of histo-blood group antigens in rotavirus host-cell invasion. Nat Commun 2015; 6: 5907. [DOI] [PubMed] [Google Scholar]
- 72.Lin JD, Feng N, Sen A, et al. Distinct roles of type I and type III interferons in intestinal immunity to homologous and heterologous rotavirus infections. PLoS Pathog 2016; 12: e1005600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Greenberg HB, Estes MK. Rotaviruses: from pathogenesis to vaccination. Gastroenterology 2009; 136: 1939–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Franco MA, Greenberg HB. Challenges for rotavirus vaccines. Virology 2001; 281: 153–55. [DOI] [PubMed] [Google Scholar]
- 75.McNeal MM, Basu M, Bean JA, Clements JD, Choi AH, Ward RL. Identification of an immunodominant CD4+ T cell epitope in the VP6 protein of rotavirus following intranasal immunization of BALB/c mice. Virology 2007; 363: 410–18. [DOI] [PubMed] [Google Scholar]
- 76.Kuklin NA, Rott L, Feng N, et al. Protective intestinal anti-rotavirus B cell immunity is dependent on alpha 4 beta 7 integrin expression but does not require IgA antibody production. J Immunol 2001; 166: 1894–902. [DOI] [PubMed] [Google Scholar]
- 77.Corthesy B, Benureau Y, Perrier C, et al. Rotavirus anti-VP6 secretory immunoglobulin A contributes to protection via intracellular neutralization but not via immune exclusion. J Virol 2006; 80: 10692–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Choi NW, Estes MK, Langridge WH. Oral immunization with a shiga toxin B subunit: rotavirus NSP4(90) fusion protein protects mice against gastroenteritis. Vaccine 2005; 23: 5168–76. [DOI] [PubMed] [Google Scholar]
- 79.Parrino TA, Schreiber DS, Trier JS, Kapikian AZ, Blacklow NR. Clinical immunity in acute gastroenteritis caused by Norwalk agent. N Engl J Med 1977; 297: 86–89. [DOI] [PubMed] [Google Scholar]
- 80.Marionneau S, Ruvoen N, Le Moullac-Vaidye B, et al. Norwalk virus binds to histo-blood group antigens present on gastroduodenal epithelial cells of secretor individuals. Gastroenterology 2002; 122: 1967–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Frenck R, Bernstein DI, Xia M, et al. Predicting susceptibility to norovirus GII.4 by use of a challenge model involving humans. J Infect Dis 2012; 206: 1386–93. [DOI] [PubMed] [Google Scholar]
- 82.Hutson AM, Airaud F, LePendu J, Estes MK, Atmar RL. Norwalk virus infection associates with secretor status genotyped from sera. J Med Virol 2005; 77: 116–20. [DOI] [PubMed] [Google Scholar]
- 83.Lindesmith L, Moe C, Marionneau S, et al. Human susceptibility and resistance to Norwalk virus infection. Nat Med 2003; 9: 548–53. [DOI] [PubMed] [Google Scholar]
- 84.Huang P, Farkas T, Zhong W, et al. Norovirus and histo-blood group antigens: demonstration of a wide spectrum of strain specificities and classification of two major binding groups among multiple binding patterns. J Virol 2005; 79: 6714–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Atmar RL, Bernstein DI, Lyon GM, et al. Serological correlates of protection against a GII.4 norovirus. Clin Vaccine Immunol 2015; 22: 923–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Malm M, Uusi-Kerttula H, Vesikari T, Blazevic V. High serum levels of norovirus genotype-specific blocking antibodies correlate with protection from infection in children. J Infect Dis 2014; 210: 1755–62. [DOI] [PubMed] [Google Scholar]
- 87.Reeck A, Kavanagh O, Estes MK, et al. Serological correlate of protection against norovirus-induced gastroenteritis. J Infect Dis 2010; 202: 1212–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ramani S, Neill FH, Opekun AR, et al. Mucosal and cellular immune responses to Norwalk virus. J Infect Dis 2015; 212: 397–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Blazevic V, Malm M, Honkanen H, Knip M, Hyoty H, Vesikari T. Development and maturation of norovirus antibodies in childhood. Microbes Infect 2016; 18: 263–69. [DOI] [PubMed] [Google Scholar]
- 90.Lindesmith LC, Donaldson E, Leon J, et al. Heterotypic humoral and cellular immune responses following Norwalk virus infection. J Virol 2010; 84: 1800–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Czako R, Atmar RL, Opekun AR, Gilger MA, Graham DY, Estes MK. Experimental human infection with Norwalk virus elicits a surrogate neutralizing antibody response with cross-genogroup activity. Clin Vaccine Immunol 2015; 22: 221–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Reddington K, Tuite N, Minogue E, Barry T. A current overview of commercially available nucleic acid diagnostics approaches to detect and identify human gastroenteritis pathogens. Biomol Detect Quantif 2014; 1: 3–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Deng J, Luo X, Wang R, et al. A comparison of Luminex xTAG® Gastrointestinal Pathogen Panel (xTAG GPP) and routine tests for the detection of enteropathogens circulating in Southern China. Diagn Microbiol Infect Dis 2015; 83: 325–30. [DOI] [PubMed] [Google Scholar]
- 94.Buss SN, Leber A, Chapin K, et al. Multicenter evaluation of the BioFire FilmArray gastrointestinal panel for etiologic diagnosis of infectious gastroenteritis. J Clin Microbiol 2015; 53: 915–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Vinjé J Advances in laboratory methods for detection and typing of norovirus. J Clin Microbiol 2015; 53: 373–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Guarino A, Albano F, Ashkenazi S, et al. European Society for Paediatric Gastroenterology, Hepatology, and Nutrition/European Society for Paediatric Infectious Diseases evidence-based guidelines for the management of acute gastroenteritis in children in Europe. J Pediatr Gastroenterol Nutr 2008; 46 (suppl 2): S81–122. [DOI] [PubMed] [Google Scholar]
- 97.Szajewska H, Mrukowicz JZ. Probiotics in the treatment and prevention of acute infectious diarrhea in infants and children: a systematic review of published randomized, double-blind, placebo-controlled trials. J Pediatr Gastroenterol Nutr 2001; 33 (suppl 2): S17–25. [DOI] [PubMed] [Google Scholar]
- 98.WHO, UNICEF. WHO/UNICEF joint statement. Clinical management of acute diarrhoea. New York/Geneva: The United Nations Children’s Fund/World Health Organization, 2004. [Google Scholar]
- 99.Salazar-Lindo E, Santisteban-Ponce J, Chea-Woo E, Gutierrez M. Racecadotril in the treatment of acute watery diarrhea in children. N Engl J Med 2000; 343: 463–67. [DOI] [PubMed] [Google Scholar]
- 100.Rossignol JF, Abu-Zekry M, Hussein A, Santoro MG. Effect of nitazoxanide for treatment of severe rotavirus diarrhoea: randomised double-blind placebo-controlled trial. Lancet 2006; 368: 124–29. [DOI] [PubMed] [Google Scholar]
- 101.Morris J, Brown W, Morris CL. Nitazoxanide is effective therapy for norovirus gastroenteritis after chemotherapy and hematopoietic stem cell transplantation (HSCT). Blood 2013; 122: 4581. [Google Scholar]
- 102.Rossignol JF, El-Gohary YM. Nitazoxanide in the treatment of viral gastroenteritis: a randomized double-blind placebo-controlled clinical trial. Aliment Pharmacol Ther 2006; 24: 1423–30. [DOI] [PubMed] [Google Scholar]
- 103.Sarker SA, Casswall TH, Juneja LR, et al. Randomized, placebo-controlled, clinical trial of hyperimmunized chicken egg yolk immunoglobulin in children with rotavirus diarrhea. J Pediatr Gastroenterol Nutr 2001; 32: 19–25. [DOI] [PubMed] [Google Scholar]
- 104.Sarker SA, Casswall TH, Mahalanabis D, et al. Successful treatment of rotavirus diarrhea in children with immunoglobulin from immunized bovine colostrum. Pediatr Infect Dis J 1998; 17: 1149–54. [DOI] [PubMed] [Google Scholar]
- 105.Vega CG, Bok M, Vlasova AN, et al. Recombinant monovalent llama-derived antibody fragments (VHH) to rotavirus VP6 protect neonatal gnotobiotic piglets against human rotavirus-induced diarrhea. PLoS Pathog 2013; 9: e1003334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Tokuhara D, Álvarez B, Mejima M, et al. Rice-based oral antibody fragment prophylaxis and therapy against rotavirus infection. J Clin Invest 2013; 123: 3829–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Florescu DF, Hill LA, McCartan MA, Grant W. Two cases of Norwalk virus enteritis following small bowel transplantation treated with oral human serum immunoglobulin. Pediatr Transplant 2008; 12: 372–75. [DOI] [PubMed] [Google Scholar]
- 108.Garaicoechea L, Aguilar A, Parra GI, et al. Llama nanoantibodies with therapeutic potential against human norovirus diarrhea. PLoS One 2015; 10: e0133665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.ROTA Council. Global introduction status. http://rotacouncil.org/vaccine-introduction/global-introduction-status/ (accessed April 10, 2018).
- 110.PATH. Rotavirus vaccine advocacy resources. 2017. http://sites.path.org/rotavirusvaccine/country-introduction-maps-and-spreadsheet/ (accessed April 10, 2018).
- 111.Aliabadi N, Tate JE, Haynes AK, Parashar UD. Sustained decrease in laboratory detection of rotavirus after implementation of routine vaccination—United States, 2000–2014. Morb Mortal Wkly Rep 2015; 64: 337–42. [PMC free article] [PubMed] [Google Scholar]
- 112.Patel MM, Glass R, Desai R, Tate JE, Parashar UD. Fulfilling the promise of rotavirus vaccines: how far have we come since licensure? Lancet Infect Dis 2012; 12: 561–70. [DOI] [PubMed] [Google Scholar]
- 113.US Centers for Disease Control and Prevention. The National Respiratory and Enteric Virus Surveillance System (NREVSS). 2018. https://www.cdc.gov/surveillance/nrevss/index.html (accessed April 10, 2018). [Google Scholar]
- 114.Gastañaduy PA, Curns AT, Parashar UD, Lopman BA. Gastroenteritis hospitalizations in older children and adults in the United States before and after implementation of infant rotavirus vaccination. J Am Med Assoc 2013; 310: 851–53. [DOI] [PubMed] [Google Scholar]
- 115.do Carmo GM, Yen C, Cortes J, et al. Decline in diarrhea mortality and admissions after routine childhood rotavirus immunization in Brazil: a time-series analysis. PLoS Med 2011; 8: e1001024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Richardson V, Hernandez-Pichardo J, Quintanar-Solares M, et al. Effect of rotavirus vaccination on death from childhood diarrhea in Mexico. N Engl J Med 2010; 362: 299–305. [DOI] [PubMed] [Google Scholar]
- 117.Glass RI, Parashar UD, Bresee JS, et al. Rotavirus vaccines: current prospects and future challenges. Lancet 2006; 368: 323–32. [DOI] [PubMed] [Google Scholar]
- 118.Armah GE, Sow SO, Breiman RF, et al. Efficacy of pentavalent rotavirus vaccine against severe rotavirus gastroenteritis in infants in developing countries in sub-Saharan Africa: a randomised, double-blind, placebo-controlled trial. Lancet 2010; 376: 606–14. [DOI] [PubMed] [Google Scholar]
- 119.Zaman K, Dang DA, Victor JC, et al. Efficacy of pentavalent rotavirus vaccine against severe rotavirus gastroenteritis in infants in developing countries in Asia: a randomised, double-blind, placebo-controlled trial. Lancet 2010; 376: 615–23. [DOI] [PubMed] [Google Scholar]
- 120.Madhi SA, Cunliffe NA, Steele D, et al. Effect of human rotavirus vaccine on severe diarrhea in African infants. N Engl J Med 2010; 362: 289–98. [DOI] [PubMed] [Google Scholar]
- 121.Bar-Zeev N, Kapanda L, Tate JE, et al. Effectiveness of a monovalent rotavirus vaccine in infants in Malawi after programmatic roll-out: an observational and case-control study. Lancet Infect Dis 2015; 15: 422–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Groome MJ, Page N, Cortese MM, et al. Effectiveness of monovalent human rotavirus vaccine against admission to hospital for acute rotavirus diarrhoea in South African children: a case-control study. Lancet Infect Dis 2014; 14: 1096–104. [DOI] [PubMed] [Google Scholar]
- 123.Carlin JB, Macartney K, Lee KJ, et al. Intussusception risk and disease prevention associated with rotavirus vaccines in Australia’s national immunisation program. Clin Infect Dis 2013; 57: 1427–34. [DOI] [PubMed] [Google Scholar]
- 124.Patel M, López-Collada V, Bulhões M, et al. Intussusception risk and health benefits of rotavirus vaccination in Mexico and Brazil. N Engl J Med 2011; 364: 2283–92. [DOI] [PubMed] [Google Scholar]
- 125.Velazquez FR, Colindres RE, Grajales C, et al. Postmarketing surveillance of intussusception following mass introduction of the attenuated human rotavirus vaccine in Mexico. Pediatr Infect Dis J 2012; 31: 736–44. [DOI] [PubMed] [Google Scholar]
- 126.Shui I, Baggs J, Patel M, et al. Risk of intussusception following administration of a pentavalent rotavirus vaccine in US Infants. J Am Med Assoc 2012; 307: 598–604. [DOI] [PubMed] [Google Scholar]
- 127.Weintraub ES, Baggs J, Duffy J, et al. Risk of intussusception after monovalent rotavirus vaccination. N Engl J Med 2014; 370: 513–19. [DOI] [PubMed] [Google Scholar]
- 128.Yih WK, Lieu TA, Kulldorff M, et al. Intussusception risk after rotavirus vaccination in U.S. infants. N Engl J Med 2014; 370: 503–12. [DOI] [PubMed] [Google Scholar]
- 129.Desai R, Cortese MM, Meltzer MI, et al. Potential intussusception risk versus benefits of rotavirus vaccination in the United States. Pediatr Infect Dis J 2013; 32: 1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Bhandari N, Rongsen-Chandola T, Bavdekar A, et al. India Rotavirus Vaccine Group. Efficacy of a monovalent human-bovine (116E) rotavirus vaccine in Indian infants: a randomised, double-blind, placebo-controlled trial. Lancet 2014; 383: 2136–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Bhandari N, Rongsen-Chandola T, Bavdekar A, et al. India Rotavirus Vaccine Group. Efficacy of a monovalent human-bovine (116E) rotavirus vaccine in Indian children in the second year of life. Vaccine 2014; 32 (suppl 1): A110–16. [DOI] [PubMed] [Google Scholar]
- 132.Isanaka S, Guindo O, Langendorf C, et al. Efficacy of a low-cost, heat-stable oral rotavirus vaccine in Niger. N Engl J Med 2017; 376: 1121–30. [DOI] [PubMed] [Google Scholar]
- 133.Burnett E, Parashar U, Tate J. Rotavirus vaccines: effectiveness, safety, and future directions. Paediatr Drugs 2018; 20: 223–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Cortes-Penfield NW, Ramani S, Estes MK, Atmar RL. Prospects and challenges in the development of a norovirus vaccine. Clin Ther 2017; 39: 1537–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Kanai Y, Komoto S, Kawagishi T, et al. Entirely plasmid-based reverse genetics system for rotaviruses. Proc Natl Acad Sci USA 2017; 114: 2349–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Hall AJ, Vinje J, Lopman B, et al. Updated norovirus outbreak management and disease prevention guidelines. MMWR Recomm Rep 2011; 60: 1–20. [PubMed] [Google Scholar]
- 137.MacCannell T, Umscheid CA, Agarwal RK, Lee I, Kuntz G, Stevenson KB. Guideline for the prevention and control of norovirus gastroenteritis outbreaks in healthcare settings. Infect Control Hosp Epidemiol 2011; 32: 939–69. [DOI] [PubMed] [Google Scholar]
- 138.United States Environmental Protection Agency. List G: EPA’s registered antimicrobial products effective against norovirus (Norwalk-like virus). 2009. https://www.epa.gov/sites/production/files/2018-04/documents/list_g_disinfectant_list_3_15_18.pdf (accessed April 10, 2018).
- 139.Atmar RL, Bernstein DI, Harro CD, et al. Norovirus vaccine against experimental human Norwalk virus illness. N Engl J Med 2011; 365: 2178–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Bernstein DI, Atmar RL, Lyon GM, et al. Norovirus vaccine against experimental human GII.4 virus illness: a challenge study in healthy adults. J Infect Dis 2015; 211: 870–78. [DOI] [PMC free article] [PubMed] [Google Scholar]


