Abstract
Purpose of the Review
Inflammation is a key component in the pathogenesis of cerebrovascular diseases. In the past few years, the role of systemic infection and gut dysbiosis in modulating inflammation and stroke risk has been increasingly acknowledged. In this review, we synthesize contemporary literature on the effects of infection and inflammation on stroke risk and outcomes, with a focus on periodontal disease, COVID-19 infection, and gut dysbiosis.
Recent Findings
Chronic and acute infections such as periodontitis and COVID-19 induce systemic inflammation that cause atherogenesis and increase cardiac injury and arrhythmias. These infections also directly injure the endothelium leading to worsened secondary inflammation after stroke. Gut dysbiosis engenders a pro-inflammatory state by modulating intestinal lymphocyte populations that can traffic directly to the brain. Additionally, post-stroke immune dysregulation creates a compounding feedback loop of further infections and gut dysbiosis that worsen outcomes.
Summary
Recent advances in understanding the pathophysiology of how infection and dysbiosis affect the progression of stroke, as well as long-term recovery, have revealed tantalizing glimpses at potential therapeutic targets. We discuss the multidirectional relationship between stroke, infection, and gut dysbiosis, and identify areas for future research to further explore therapeutic opportunities.
Supplementary Information
The online version contains supplementary material available at 10.1007/s11910-022-01179-6.
Keywords: Infection, Inflammation, COVID-19, Periodontitis, Dysbiosis, Stroke
Introduction
Inflammation has long been implicated in the pathogenesis of cerebrovascular disease. Conditions related to stroke, including infection, gut dysbiosis, and autoimmune disease, in addition to lifestyle factors such as tobacco use and alcohol consumption, are all linked to increased inflammation and cerebrovascular disease [1, 2•, 3]. There is growing evidence that targeting inflammation and immune responses during both the acute and chronic phases of stroke may have preventative and therapeutic implications [4, 5]. In this review, we synthesize the contemporary literature on the effects of infection and inflammation on stroke risk and outcomes, drawing on new data on periodontal disease, COVID-19 infection, and gut dysbiosis. We also discuss how post-stroke inflammation and immune dysregulation can create a compounding feedback loop of further infections and gut dysbiosis that may worsen outcomes.
Systemic Infection and Stroke Risk
The relationship between infection, inflammation, and stroke has been well studied, and this topic has seen increased interest with the association of severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) — COVID-19 — with acute cerebrovascular disease [2•]. In this section, we update the established paradigms in the field by focusing on emerging data on periodontal disease, as an example of a chronic condition, and COVID-19 infection, as an example of an acute infection. We first review the epidemiological literature and then review mechanistic data.
Chronic infectious burden, or the cumulative number of lifetime infections, increases long-term risk of atherosclerotic disease and stroke. A landmark analysis from the Northern Manhattan study found that the burden of common infections (Chlamydia pneumoniae, Helicobacter pylori, cytomegalovirus, and herpes simplex virus 1 and 2) was associated with a 1.4-fold increased risk of all strokes after adjusting for demographics and risk factors [6]. A similar effect was seen with chronic viral infections such as HIV, which is thought to cause arterial inflammation and cerebral vasculopathy [7]. A recent meta-analysis showed that people living with HIV have an increased incidence rate of any stroke, with a 1.6-fold increase in ischemic stroke and a 2.2-fold increase in intracranial hemorrhage. Foundational data on the topic of chronic infectious disease burden and stroke has been synthesized elsewhere [8, 9].
Recently, periodontal disease has emerged as a prototypical chronic infectious-inflammatory condition that may impact cerebrovascular health. Periodontitis is a chronic inflammatory disorder caused by polybacterial colonization and repeated low-grade exposure to gram-negative bacteria, leading to an increase in systemic inflammation [10•]. A meta-analysis of 30 longitudinal cohort studies showed a modest but consistently increased risk of stroke by 1.24-fold in populations with periodontal disease [10•]. In an Atherosclerosis Risk in Communities analysis, periodontal disease was associated particularly with cardioembolic and thrombotic stroke subtypes [11]. Interestingly, this study showed that groups with regular dental care exhibited a lower burden of periodontal disease, and that they had a lower risk of incident ischemic stroke [11]. Additionally, IgA seropositivity to common periodontal microbial agents has been found to be associated with an increased risk of lacunar type ischemic stroke [12]. Taken together, these data suggest that a common and chronic low-grade infectious process may contribute to the burden of cerebrovascular disease, providing an exciting preventive opportunity, for example through increased access to dental care among at-risk groups. Future studies with optimal measures of chronic infectious burden are required to confirm its effect on stroke risk and determine which organisms and conditions warrant intervention [13•].
Apart from the impact of chronic infections as a stroke risk factor, several studies indicate that acute systemic infections may trigger stroke. A seminal study of over 19,000 cases of first stroke found that risk for stroke was increased by over threefold after a diagnosis of respiratory tract infections. The risk was especially high during the first 3 days then gradually decreased during the following weeks [14]. Similarly, sepsis has been associated with a greater than 28-fold increase in ischemic stroke and greater than 12-fold increase in hemorrhagic stroke within 15 days, with the risk remaining elevated for as long as 1 year after hospitalization [15]. A Cardiovascular Health Study analysis provided supportive data, showing that hospitalization for infection was over 3 times as likely during periods before incident ischemic stroke compared to during control time periods in prior years [16]. This relationship was also seen in the Atherosclerosis Risk in Communities cohort, in which this relationship was stronger among those at lower overall cardiovascular disease risk based on traditional risk factors [17]. Further investigation has shown that many infection types in different organ systems are associated with a heightened risk of ischemic stroke [18].
The high incidence of COVID-19 during the ongoing pandemic has provided vital new information about the impact of acute viral infections on stroke. Previously, it was known from a large study of over 36,000 hospitalized patients that the risk of ischemic stroke is increased nearly threefold in the 15 days after an influenza-like illness [19]. These data foretold what has been seen in the past 2 years during the COVID-19 pandemic. Research on stroke in COVID-19 was set in motion with a case series of large vessel occlusion strokes seen in young patients with COVID-19 [20]. Several rigorous epidemiological studies followed, with one study showing that the rate of acute ischemic stroke was eightfold higher in COVID-19 infection than in influenza infection [21•]. Several retrospective studies of patients hospitalized with COVID-19 infection have reported stroke incidence rates ranging from 1 to 3%, with rates as high as 6% in critically ill patients [21•, 22, 23]. Two recent meta-analyses found that cryptogenic stroke is particularly overrepresented in patients with COVID-19 infection [24•, 25•]. Additionally, patients with COVID-19 infection were younger, with greater stroke severity, and higher frequency of large vessel occlusion compared to uninfected patients with stroke [24•, 25•]. Overall, these data support the paradigm whereby acute viral infections can trigger stroke. Further, overrepresentation of cryptogenic stroke and stroke in young people otherwise at low risk of stroke suggests virus-specific mechanisms may be present.
The mechanisms by which these chronic and acute infections may cause stroke are manifold. Proposed etiologies include pro-inflammatory states that lead to prothrombotic states, endothelial dysfunction, and vascular inflammation leading to increased progression of carotid and cerebral atherosclerosis, as well as direct pathogenic invasion of the vessel wall leading to vascular injury. Individual mechanisms for specific pathogens were reviewed comprehensively elsewhere [13•, 26, 27]. Here, we focus on mechanisms specific to two conditions: periodontitis and COVID-19 infection.
Periodontitis induces a chronic low-grade bacteremia and pro-inflammatory state, characterized by an increase in acute phase reactants such as C-reactive protein (CRP), interleukin 6 (IL-6), and tumor necrosis factor (TNF) [28•]. Periodontal microorganisms may also cause direct vascular injury, and viable periodontal pathogens have been found in the vascular endothelium of major arteries [29]. Systemic inflammation caused by periodontitis is hypothesized to contribute to endothelial dysfunction and atherogenesis. Periodontitis has accordingly been associated with atherosclerosis in carotid vasculature, and linked with increased thickness and calcification of aortic arch atheroma, a prevalent risk factor for embolic stroke [30]. Furthermore, systemic inflammation linked to periodontitis is thought to play a role in the initiation and persistence of atrial fibrillation. This may explain the association between periodontal disease and atrial fibrillation, the most common cause of cardioembolic ischemic stroke [28•].
COVID-19 has been linked with immune dysregulation as well as direct vascular damage, in addition to unique prothrombotic mechanisms. Through modulating the adrenergic system and the hypothalamic-pituitary-adrenocortical axis, COVID-19 induces initial hyperactivity of innate immunity that causes neutrophilia and increases production of cytokines such as IL-6, IL-1β, and TNF. This is followed by immunosuppression and lymphopenia [31•]. Markers of systemic inflammation, such as erythrocyte sedimentation rate (ESR), IL-6, and TNF, are elevated in COVID-19 patients and reflect disease severity [32–34]. Apart from immune dysregulation, COVID-19 can also directly infect brain endothelial cells and induce microvascular injury. A viral protease encoded in the SARS-CoV-2 genome was seen to cleave (NF)-κB essential modulator (NEMO); the disruption of this pathway in mouse models induced blood–brain-barrier disruption, neuroinflammation, and a microvascular damage similar to that observed in brains of patients with COVID-19 infection. COVID-19 infection also elicits a particularly hypercoagulable state characterized by increased D-dimer and fibrinogen. This coagulopathy is caused by viral-mediated cytokine release, endothelial injury, and neutrophil and complement activation [31•]. This distinctive pro-coagulant state may explain why ischemic stroke due to large vessel occlusion was seen more commonly in COVID-19 cases [24•, 35, 36]. Last, acute cardiac injury and arrhythmias, such as atrial fibrillation, occur frequently in COVID-19 infection, which would also increase the risk of cardioembolic stroke [37]. Further research is needed to identify which pathway would be most amenable to intervention to mitigate stroke risk. Such discoveries may benefit patients with COVID-19 infection and enrich our understanding of virally mediated ischemic stroke in general.
Pre-stroke Infection and Stroke Outcomes
It is well known that infections can lead to greater morbidity and mortality in stroke, but the temporality and causality of the relationship between pre-stroke infection and subsequent stroke outcomes are not well elucidated [38, 39]. Prior to the COVID-19 pandemic, several studies demonstrated that pre-stroke and post-stroke infections may differently impact morbidity and mortality after stroke. For example, in a study of young patients with first-ever stroke, pre-stroke infections were associated with a nearly threefold increase in unfavorable 3-month functional outcomes, but not increased mortality. This was in contrast to post-stroke infection, which caused an increase in both long-term all-cause mortality and morbidity [39]. The heterogeneity of infections in prior studies precludes drawing confident conclusions about these relationships. The COVID-19 pandemic, characterized by a large burden of a single-agent infection across a large population, has improved our understanding of whether and how infections preceding stroke onset can influence stroke outcomes, and we focus on these data here.
The median duration from COVID-19 symptom onset to stroke diagnosis across several studies was approximately 12–16 days (with a range of 5–28 days) [21•, 40, 41]. Several studies have shown that COVID-19 infection is associated with higher stroke severity, worse functional outcomes, and greater mortality [40, 42]. The clear temporal relationship between COVID-19 symptom onset and acute stroke symptom onset supports the notion that pre-stroke infection does indeed detrimentally impact outcomes after stroke. However, it should be noted that much of these data reflect outcomes during the initial phase of the COVID-19 pandemic and do not fully account for confounding factors such as delays in admission and other factors that may impact outcomes.
The mechanisms through which COVID-19 infection increases stroke risk, described above, may also impact post-stroke outcomes. For example, the vascular damage and acute inflammatory reactants induced by pre-stroke COVID-19 infection may worsen secondary inflammation after stroke onset, especially in the penumbral area of the stroke [39]. Patients with stroke and preceding infection have increased platelet activation, C-reactive protein levels, and fibrinogen levels [39, 43, 44]. Additionally, direct invasion of brain endothelial cells and blood brain barrier disruption may increase vessel permeability, reducing CNS perfusion [45••]. This may increase vulnerability to hypoxic injury, further worsening outcomes. In addition, pre-stroke infections are also associated with an increased risk of peri-procedural adverse events after thrombectomy and thrombolysis such as intracerebral bleeding, which may also be a consequence of pre-stroke inflammation and resulting vascular damage [39]. Last, COVID-19 infection causes an initial hyper-inflammatory state followed by immune suppression and lymphopenia that leads to a greater risk of post-stroke infection, which may further compound injury and worsen overall outcomes [31•].
Post-stroke Infection and Stroke Outcomes
Infection and stroke exhibit a bidirectional relationship. While infection increases stroke risk, stroke can also increase the risk of infection. Urinary tract infections and pneumonia are commonly seen post-stroke infections [46]. Post-stroke pneumonia in particular is associated with poor functional outcomes, with a large meta-analysis showing a 3.6-fold increase in death [46]. Even after adjusting for confounders such as stroke severity, ischemic stroke sub-type, and co-morbidities, pneumonia is associated with longer in-hospital stay, worse functional outcomes, and increased mortality [47].
There is growing understanding of the mechanisms through which post-stroke infections occur and influence outcomes. After stroke, there is initial immune activation marked by neutrophilia, which is soon followed by immunodepression characterized by prolonged peripheral lymphopenia and reduced T cell responsiveness. This immune dysregulation is mediated by an activation of the autonomic nervous system and the hypothalamic-adrenal axis, and predispose patients to post-stroke infections [48•]. Persistent inflammation after stroke, as measured by the neutrophil to lymphocyte ratio, has been associated with a higher incidence of in-hospital pulmonary infection, indicating stroke-induced lymphopenia increases risk of infection [49]. Antigen-dependent autoimmunity is also activated after stroke, due to the abundance of novel brain antigens that are able to reach the circulation and stimulate systemic B and T lymphocytes [48•]. This autoreactive immune response can be harmful or beneficial depending on the polarization of T lymphocytes. The pro-inflammatory insult of an infection can lead to the detrimental T helper cell type 1 (Th1) response against brain antigens such as myelin basic protein and glial fibrillary acidic protein. This is associated with worse functional outcomes and increased cognitive decline in the year after stroke [50, 51].
While the prevention of post-stroke infection seems to be an obvious target to improve outcomes, the solution may not be so simple. A meta-analysis of several trials found that antibiotic therapy during the acute phase of stroke reduced the incidence of infections but did not improve patient functional outcomes [52]. Intriguingly, prophylactic antibiotics reduced the incidence of urinary tract infection but not pneumonia, the latter of which has the greatest effect on post-stroke outcomes [46, 52]. This may indicate the need for a more tailored approach to target stroke-associated pneumonia, by finding optimal antibiotic regimens, choosing specific at-risk patient subgroups, and using biomarker-guided therapy [53]. In experimental models, the inhibition of β-adrenergic receptors using propranolol was shown to reduce bacteremia and lung colonization perhaps by increasing survival rates of circulating lymphocytes, indicating that modulation of the adrenergic system may also be a therapeutic target [54]. Unfortunately, retrospective studies and prospective clinical trials have not borne out this hypothesis, indicating a need for a more focused immunomodulatory approach to abrogate the impact of post-stroke infections on stroke outcomes [55, 56].
Pre-stroke Gut Dysbiosis and Stroke
The gastrointestinal tract is a prime target for immunomodulation. The gastrointestinal tract is a major immune organ, and the largest reservoir of microorganisms in the human body [57•]. The interplay of the brain-gut axis and its role in the pathophysiology of stroke have become an emerging topic of investigation. In this section, we discuss the impact of the pre-stroke gut microbiome dysbiosis on stroke outcomes and the potential mechanisms involved in these interactions.
Studies in mice have shown that alterations of the commensal microbiome can affect stroke outcomes by altering the peripheral immune system, and that these changes are transmissible through fecal transplantation. In one elegant study, antibiotics were used to alter mice gut microbiota prior to stroke induction. The altered microbiota was characterized by a contraction of Clostridiaceae (Firmicutes) and Bacteroidetes with a concomitant increase in Proteobacteria. The mice with the altered microbiota exhibited significant reduction in infarct volume, and improved sensory and motor functions. Moreover, microbiota signature from these mice could be transferred through fecal transplant into another mouse with the same neuroprotective effect [58]. The same group expanded upon their findings to show that combinatorial and singular antibiotic treatment protocols could be used to create targeted modifications in microbiota composition in mice models. Specifically, singular treatments with ampicillin or vancomycin improved stroke outcomes by reducing infarct volumes and sensorimotor functioning [59••]. Age-related changes in the microbiome may also play a role in affecting stroke outcomes. In aged mice, the ratio of Firmicutes to Bacteroidetes (the two bacterial phyla that primarily compose the human and mouse gut microbiome) was found to be altered compared to young mice. In addition, fecal transplants from young mice into aged mice were shown to reduce infarct volume and improve stroke outcomes [60]. In humans, stroke co-morbidities such as diabetes, hypertension, dyslipidemia, or obesity are associated with dysbiosis [61]. Those at high-risk of stroke, defined by the number of co-morbidities, have high prevalence of opportunistic pathogens in their gut microbiota compared to patients without stroke risk factors [62]. While these data allow us to speculate about how pre-stroke gut dysbiosis may affect stroke risk and outcomes, there is a need for prospective cohort studies with careful characterization and functional analyses of gut microbiome to inform this paradigm.
Mechanistically, animal studies have demonstrated the gut microbiome can regulate the neuroinflammatory response to acute brain injury, and clinical studies have shown that gut dysbiosis is associated with systemic inflammation [57•, 63]. One study in mice models has shown that neuroprotective microbiota can prime dendritic cells in the gut to induce an expansion of anti-inflammatory regulatory T cells (Treg). These Treg cells then secrete IL-10 to suppress pro-inflammatory IL-17 + γδT cells which would normally be activated by stroke. Surprisingly, these intestinal T cells were seen to traffic to the meninges after stroke, indicating that immunomodulation in the gut may directly impact the brain [58]. Microbial metabolites such as short-chain fatty acids (SCFAs), tryptophan, and membrane-derived molecules such as LPS may be an additional means by which gut bacteria can impact stroke outcomes [57•]. SCFAs, such as acetate, propionate, and butyrate, have received particular interest for their anti-inflammatory effects, especially their direct role in affecting the blood–brain-barrier and microglial activation [64]. In animal models, young mice transplanted with aged microbiome demonstrated reduced SCFA and an exaggerated systemic inflammatory response [60]. The administration of SCFAs (specifically butyric acid) after ischemic stroke in rats reduced neurological impairment as well as cerebral infarct volume and edema, ameliorated intestinal permeability and leakiness, and enriched the gut microbiota of beneficial bacteria such as Lactobacillus [65•]. Another study showed that augmenting SCFA-producing bacteria in aged mice improved post-stroke sensorimotor outcomes, reduced brain IL-17 + γδT cells, and increased systemic SCFA concentrations [66••]. In human studies, SCFA concentration was found to be lower in stroke patients, especially in those who exhibited poor neurological outcomes [67]. Moreover, acetate was seen to be decreased after stroke in patients and negatively correlated with the levels of glycated hemoglobin and low-density lipoprotein cholesterol, indicating a role in modulating inflammation [63].
Gut Dysbiosis After Stroke
Like the bidirectional relationship between stroke and infection, stroke can also affect the gut microbiome. Up to 50% of stroke patients experience gastrointestinal complications such as dysmotility and GI bleeding which are associated with increased mortality rates [68]. In addition, stroke increases gut permeability causing a “leaky” gut that allows the translocation and seeding of gut bacteria into the lungs, spleen, and liver [69]. This can exacerbate systemic immune responses and lead to sepsis as well as multi-organ dysfunction, worsening post-stroke outcomes [70, 71].
In animal models, severe gastrointestinal paralysis is inducible with acute brain injury and is associated with an overgrowth of intestinal bacteria [72, 73]. Additionally, bacterial diversity in stool is reduced in mice with severe stroke, characterized by a reduction in Firmicutes with a concomitant expansion of Bacteroidetes [73]. When the microbiota from post-stroke mice were transplanted into control mice, they developed significantly larger infarct volumes and worse sensorimotor outcomes, indicating that post-stroke dysbiosis leads to negative outcomes and that it is also transferrable. Intriguingly, when post-stroke mice were transplanted with healthy fecal microbiota, they exhibited reduced lesion sizes and improved stroke outcomes indicating that the negative impact of post-stroke dysbiosis is also reversible [73].
As mentioned above, stroke patients exhibit significantly different microbiomes compared to their healthy counterparts [74]. Few retrospective cohort studies have characterized the microbiome composition in stroke patients compared to a control cohort [74]. Stroke patients often exhibit a decrease in members of the phylum Bacteroidetes and an increase in Lactobacillus (from phylum Firmicutes) [63, 75, 76]. Some studies have correlated an expansion in certain microbial genera with stroke severity and functional outcome; however, the microbial compositions differ from study to study [75–77]. These discrepancies may be due to differences in the local microbiomes of the study populations and study design, including any exposure to antibiotics, sample processing, and analysis. This highlights the need for a consensus on study techniques and taxonomic considerations, as well as the need for large, multi-center cohort studies.
Mechanistically, the catecholaminergic stress response in the gut following stroke may lead to altered intestinal motility, impaired intestinal barrier function, and gut dysbiosis [48•, 72, 73]. Gut dysbiosis affects the balance of intestinal immune cells, specifically T lymphocytes, which play an important role in stroke progression [57•, 78]. A post-stroke dysbiotic microbiome can induce pro-inflammatory T cell populations (such as Th1, Th17, and IL-17 + γδT cells) that can traffic directly from the gut to the brain to worsen inflammation [58, 73]. Conversely, the transfer of healthy microbiome into a post-stroke mouse can induce an increase in neuroprotective Treg cells in the peripheral immune organs and the ischemic brain. Further, this neuroprotection is abrogated in a lymphocyte-deficient mouse, highlighting the key role of lymphocytes in gut microbiota-mediated neuroprotection [73]. SCFAs produced by the gut microbiome are also seen to influence post-stroke T cell polarization and improve outcomes [66••, 79•]. Colonization of post-stroke mice with SCFA-producing microbiota reduced the level of brain pro-inflammatory IL-17 + γδT cells and improved post-stroke recovery [66••]. This indicates that modulating the microbiome and directly supplementing neuroprotective SCFAs are both viable interventions in improving post-stroke recovery.
Conclusion
We have discussed the impact of inflammation caused by chronic infection, acute infection, and gut dysbiosis on stroke risk and outcome (Table 1). Additionally, we considered how stroke can further dysregulate systemic homeostasis and lead to increased infections and gut dysbiosis. Chronic infections, such as periodontal disease, increase stroke risk by inducing chronic inflammation and vascular damage that leads to atherogenesis. This systemic inflammation may also contribute to atrial fibrillation. Similarly, acute infections such as COVID-19 cause direct microvascular injury and induce coagulopathy that increases stroke risk. Also, an increased incidence of acute cardiac injury and arrhythmia is also seen in COVID-19 infection. Both chronic and acute infection can cause endothelial injury that can damage the blood–brain barrier and worsen secondary inflammation after stroke. COVID-19 in particular dysregulates the immune system to cause a lymphopenic state that predisposes to further infection and worse stroke outcomes. Gut dysbiosis is another condition that dysregulates the immune response and shifts the milieu to a pro-inflammatory state, worsening stroke outcomes. Further immune dysregulation and stress responses provoked by stroke can aggravate inflammation, which in turn increases the risk of post-stroke infection and compromises gut homeostasis (Table 2).
Table 1.
Mechanisms by which infection and gut dysbiosis impact stroke
| Condition | Mechanisms for increased stroke risk | Mechanisms for worse stroke outcome |
|---|---|---|
| Chronic infection: Periodontal disease |
• Chronic inflammation • ↑Atherosclerosis • Initiation and persistence of atrial fibrillation |
• Endothelial injury and vascular inflammation • ↑Peri-procedural adverse events |
| Acute infection: COVID-19 |
• Immune dysregulation • Direct microvascular injury • Coagulopathy • ↑Acute cardiac injury and arrhythmia |
• BBB damage • ↑LVO/stroke severity • ↑Secondary inflammation • Lymphopenia |
| Gut dysbiosis* |
• Altered ratio of pro-inflammatory T cells (Th1, Th17, γδ IL-17 +) to anti-inflammatory T cells (Treg) • Decreased levels of SCFA • ↑Intestinal permeability and bacterial translocation |
|
Abbreviations: BBB, blood brain barrier; LVO, large vessel occlusion; SCFA, short-chain fatty acid
*Available mechanistic data regarding gut dysbiosis largely apply to both stroke risk and stroke outcomes
Table 2.
Mechanisms by which stroke leads to infection, inflammation, and gut dysbiosis
| Condition | Mechanisms |
|---|---|
| Infection and inflammation |
• Activation of adrenergic system through the HPA axis leading to a surge of glucocorticoids and catecholamines • Immunosuppression and lymphopenia • Antigen-dependent autoimmunity |
| Gut dysbiosis |
• Catecholaminergic surge • ↑Intestinal permeability • ↑Intestinal dysmotility • Overgrowth of intestinal bacteria |
Abbreviations: HPA, hypothalamus–pituitary–adrenal axis
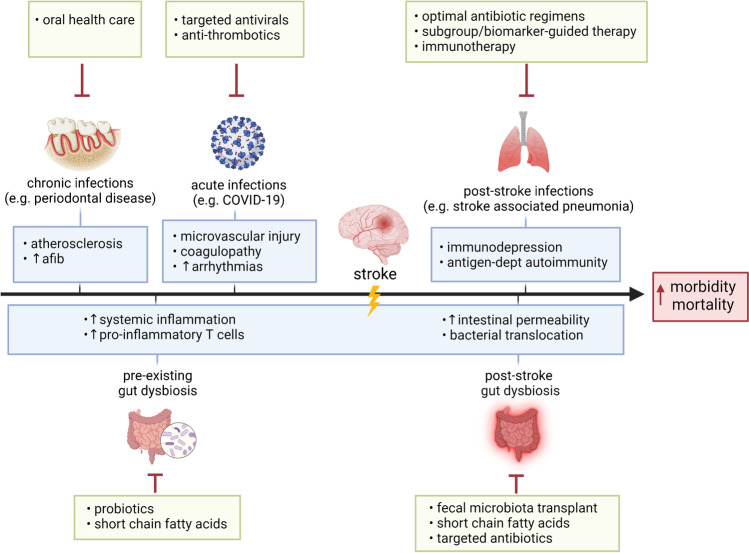
A mechanistic understanding of how infection and gut dysbiosis impact stroke and vice versa has revealed therapeutic opportunities in improving stroke morbidity and mortality (Fig. 1). Primary preventative therapy to reduce chronic infections, targeted therapy to reduce the impact of acute infections, and a focus on optimal gut microbiome composition may decrease stroke risk and improve outcomes. Additionally, interventions such as immunotherapy, targeted antibiotics, and fecal microbiota transplantation may be employed to reduce the impact of stroke on post-stroke infection, inflammation, and gut dysbiosis.
Fig. 1.
Therapeutic opportunities to abrogate the impact of infections and gut dysbiosis on stroke. Interventions, such as oral health care for periodontal disease, or targeted antivirals and anti-thrombotics for COVID-19 and other acute infections may reduce stroke risk and improve stroke outcomes. Probiotics that induce a healthy gut microbiome or supplementation with neuroprotective short-chain fatty acids may also reduce inflammation to improve stroke outcomes. After stroke onset, subgroup/biomarker-guided antibiotic therapies and immunotherapy are examples of interventions that target post-stroke infections. Post-stroke gut dysbiosis may be restored using fecal microbiota transplants, targeted antibiotics, or direct supplementation with short-chain fatty acids to improve stroke outcomes
There is a need for additional epidemiologic and experimental studies to better characterize the relationship between infection and gut dysbiosis with subsequent stroke risk and outcomes. Further, ischemic stroke is the prevalent stroke type represented in the extant literature, highlighting a need for a focus on hemorrhagic strokes as well. Prospective trials utilizing the above therapies and examining their impact on stroke remain to be performed and present an exciting opportunity to mitigate the burden of cerebrovascular disease.
Supplementary Information
Below is the link to the electronic supplementary material.
Author Contribution
Dr. Oh: conceptualization, writing–original draft; Dr. Parikh: conceptualization, writing–review and editing, funding acquisition, supervision.
Funding
Dr. Parikh is supported by the NIH/NIA (K23 AG073524), the Leon Levy Neuroscience Fellowship, and the Florence Gould Endowment for Discovery in Stroke.
Compliance with Ethical Standards
Ethics Approval
Not applicable for review article.
Conflict of Interest
Dr. Parikh has received personal compensation for medicolegal consulting on stroke and the New York State Empire Clinical Research Investigator Program, unrelated to this work.
Footnotes
Topical Collection on Stroke
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
- 1.Anrather J, Iadecola C. Inflammation and stroke: an overview. Neurotherapeutics. 2016;13(4):661–670. doi: 10.1007/s13311-016-0483-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Parikh NS, Merkler AE, Iadecola C. Inflammation, autoimmunity, infection, and stroke: epidemiology and lessons from therapeutic intervention. Stroke. 2020;51(3):711–718. doi: 10.1161/strokeaha.119.024157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Levard D, Buendia I, Lanquetin A, Glavan M, Vivien D, Rubio M. Filling the gaps on stroke research: focus on inflammation and immunity. Brain Behav Immun. 2021;91:649–667. doi: 10.1016/j.bbi.2020.09.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mena H, Cadavid D, Rushing EJ. Human cerebral infarct: a proposed histopathologic classification based on 137 cases. Acta Neuropathol. 2004;108(6):524–530. doi: 10.1007/s00401-004-0918-z. [DOI] [PubMed] [Google Scholar]
- 5.Kelly PJ, Lemmens R, Tsivgoulis G. Inflammation and stroke risk: a new target for prevention. Stroke. 2021;52(8):2697–2706. doi: 10.1161/STROKEAHA.121.034388. [DOI] [PubMed] [Google Scholar]
- 6.Elkind MS, Ramakrishnan P, Moon YP, Boden-Albala B, Liu KM, Spitalnik SL, et al. Infectious burden and risk of stroke: the northern Manhattan study. Arch Neurol. 2010;67(1):33–38. doi: 10.1001/archneurol.2009.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gutierrez J, Menshawy K, Gonzalez M, Goldman J, Elkind MS, Marshall R, et al. Brain large artery inflammation associated with HIV and large artery remodeling. AIDS. 2016;30(3):415–423. doi: 10.1097/QAD.0000000000000927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Miller EC, Elkind MS. Infection and stroke: an update on recent progress. Curr Neurol Neurosci Rep. 2016;16(1):2. doi: 10.1007/s11910-015-0602-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Guiraud V, Amor MB, Mas JL, Touzé E. Triggers of ischemic stroke: a systematic review. Stroke. 2010;41(11):2669–2677. doi: 10.1161/strokeaha.110.597443. [DOI] [PubMed] [Google Scholar]
- 10.Larvin H, Kang J, Aggarwal VR, Pavitt S, Wu J. Risk of incident cardiovascular disease in people with periodontal disease: a systematic review and meta-analysis. Clin Exper Dent Res. 2021;7(1):109–122. doi: 10.1002/cre2.336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sen S, Giamberardino LD, Moss K, Morelli T, Rosamond WD, Gottesman RF, et al. Periodontal disease, regular dental care use, and incident ischemic stroke. Stroke. 2018;49(2):355–362. doi: 10.1161/STROKEAHA.117.018990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Palm F, Pussinen PJ, Aigner A, Becher H, Buggle F, Bauer MF, et al. Association between infectious burden, socioeconomic status, and ischemic stroke. Atherosclerosis. 2016;254:117–123. doi: 10.1016/j.atherosclerosis.2016.10.008. [DOI] [PubMed] [Google Scholar]
- 13.Elkind MSV, Boehme AK, Smith CJ, Meisel A, Buckwalter MS. Infection as a stroke risk factor and determinant of outcome after stroke. Stroke. 2020;51(10):3156–3168. doi: 10.1161/strokeaha.120.030429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Smeeth L, Thomas SL, Hall AJ, Hubbard R, Farrington P, Vallance P. Risk of myocardial infarction and stroke after acute infection or vaccination. N Engl J Med. 2004;351(25):2611–2618. doi: 10.1056/NEJMoa041747. [DOI] [PubMed] [Google Scholar]
- 15.Boehme AK, Ranawat P, Luna J, Kamel H, Elkind MS. Risk of acute stroke after hospitalization for sepsis: a case-crossover study. Stroke. 2017;48(3):574–580. doi: 10.1161/STROKEAHA.116.016162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Elkind MS, Carty CL, O'Meara ES, Lumley T, Lefkowitz D, Kronmal RA, et al. Hospitalization for infection and risk of acute ischemic stroke: the Cardiovascular Health Study. Stroke. 2011;42(7):1851–1856. doi: 10.1161/STROKEAHA.110.608588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cowan LT, Alonso A, Pankow JS, Folsom AR, Rosamond WD, Gottesman RF, et al. Hospitalized infection as a trigger for acute ischemic stroke: the atherosclerosis risk in communities study. Stroke. 2016;47(6):1612–1617. doi: 10.1161/STROKEAHA.116.012890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sebastian S, Stein LK, Dhamoon MS. Infection as a stroke trigger: associations between different organ system infection admissions and stroke subtypes. Stroke. 2019;50(8):2216–2218. doi: 10.1161/STROKEAHA.119.025872. [DOI] [PubMed] [Google Scholar]
- 19.Boehme AK, Luna J, Kulick ER, Kamel H, Elkind MSV. Influenza-like illness as a trigger for ischemic stroke. Ann Clin Transl Neurol. 2018;5(4):456–463. doi: 10.1002/acn3.545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Oxley TJ, Mocco J, Majidi S, Kellner CP, Shoirah H, Singh IP, et al. Large-vessel stroke as a presenting feature of Covid-19 in the young. N Engl J Med. 2020;382(20):e60. doi: 10.1056/NEJMc2009787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Merkler AE, Parikh NS, Mir S, Gupta A, Kamel H, Lin E, et al. Risk of ischemic stroke in patients with coronavirus disease 2019 (COVID-19) vs patients with influenza. JAMA Neurol. 2020;77(11):1366–1372. doi: 10.1001/jamaneurol.2020.2730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yaghi S, Ishida K, Torres J, Mac Grory B, Raz E, Humbert K, et al. SARS-CoV-2 and stroke in a New York healthcare system. Stroke. 2020;51(7):2002–2011. doi: 10.1161/STROKEAHA.120.030335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mao L, Jin H, Wang M, Hu Y, Chen S, He Q, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77(6):683–690. doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nannoni S, de Groot R, Bell S, Markus HS. Stroke in COVID-19: a systematic review and meta-analysis. Int J Stroke. 2021;16(2):137–149. doi: 10.1177/1747493020972922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Katsanos AH, Palaiodimou L, Zand R, Yaghi S, Kamel H, Navi BB, et al. The impact of SARS-CoV-2 on stroke epidemiology and care: a meta-analysis. Ann Neurol. 2021;89(2):380–388. doi: 10.1002/ana.25967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Elkind MS. Infectious burden: a new risk factor and treatment target for atherosclerosis. Infect Disord Drug Targets. 2010;10(2):84–90. doi: 10.2174/187152610790963519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fugate JE, Lyons JL, Thakur KT, Smith BR, Hedley-Whyte ET, Mateen FJ. Infectious causes of stroke. Lancet Infect Dis. 2014;14(9):869–880. doi: 10.1016/S1473-3099(14)70755-8. [DOI] [PubMed] [Google Scholar]
- 28.Sen S, Redd K, Trivedi T, Moss K, Alonso A, Soliman EZ, et al. Periodontal disease, atrial fibrillation and stroke. Am Heart J. 2021;235:36–43. doi: 10.1016/j.ahj.2021.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kebschull A, Demmer R, Papapanou P. “Gum bug, leave my heart alone!”—epidemiologic and mechanistic evidence linking periodontal infections and atherosclerosis. J Dent Res. 2010;89(9):879–902. doi: 10.1177/0022034510375281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sen S, Chung M, Duda V, Giamberardino L, Hinderliter A, Offenbacher S. Periodontal disease associated with aortic arch atheroma in patients with stroke or transient ischemic attack. J Stroke Cerebrovasc Dis. 2017;26(10):2137–2144. doi: 10.1016/j.jstrokecerebrovasdis.2017.04.035. [DOI] [PubMed] [Google Scholar]
- 31.Iadecola C, Anrather J, Kamel H. Effects of COVID-19 on the nervous system. Cell. 2020;183(1):16–27.e1. doi: 10.1016/j.cell.2020.08.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Solomon IH, Normandin E, Bhattacharyya S, Mukerji SS, Keller K, Ali AS, et al. Neuropathological features of Covid-19. N Engl J Med. 2020;383(10):989–992. doi: 10.1056/NEJMc2019373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Diao B, Wang C, Tan Y, Chen X, Liu Y, Ning L, et al. Reduction and functional exhaustion of T cells in patients with coronavirus disease 2019 (COVID-19) Front Immunol. 2020;11:827. doi: 10.3389/fimmu.2020.00827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Helms J, Tacquard C, Severac F, Leonard-Lorant I, Ohana M, Delabranche X, et al. High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study. Intensive Care Med. 2020;46(6):1089–1098. doi: 10.1007/s00134-020-06062-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hess DC, Eldahshan W, Rutkowski E. COVID-19-related stroke. Transl Stroke Res. 2020;11(3):322–325. doi: 10.1007/s12975-020-00818-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kihira S, Schefflein J, Mahmoudi K, Rigney B, Delma BN, Mocco J, et al. Association of coronavirus disease (COVID-19) with large vessel occlusion strokes: a case-control study. AJR Am J Roentgenol. 2021;216(1):150–6. doi: 10.2214/ajr.20.23847. [DOI] [PubMed] [Google Scholar]
- 37.Goyal P, Choi JJ, Pinheiro LC, Schenck EJ, Chen R, Jabri A, et al. Clinical characteristics of Covid-19 in New York city. N Engl J Med. 2020;382(24):2372–2374. doi: 10.1056/NEJMc2010419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR, et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the Global Burden of Disease Study. The Lancet. 2020;395(10219):200–211. doi: 10.1016/S0140-6736(19)32989-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Heikinheimo T, Broman J, Haapaniemi E, Kaste M, Tatlisumak T, Putaala J. Preceding and poststroke infections in young adults with first-ever ischemic stroke: effect on short-term and long-term outcomes. Stroke. 2013;44(12):3331–3337. doi: 10.1161/strokeaha.113.002108. [DOI] [PubMed] [Google Scholar]
- 40.Calmettes J, Peres R, Goncalves B, Varlan D, Turc G, Obadia M, et al. Clinical outcome of acute ischemic strokes in patients with COVID-19. Cerebrovasc Dis. 2021;50(4):412–419. doi: 10.1159/000514562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Valencia-Enciso N, Ortiz-Pereira M, Zafra-Sierra MP, Espinel-Gómez L, Bayona H. Time of stroke onset in coronavirus disease 2019 patients around the globe: a systematic review and analysis. J Stroke Cerebrovasc Dis. 2020;29(12):105325. doi: 10.1016/j.jstrokecerebrovasdis.2020.105325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Martí-Fàbregas J, Guisado-Alonso D, Delgado-Mederos R, Martínez-Domeño A, Prats-Sánchez L, Guasch-Jiménez M, et al. Impact of COVID-19 infection on the outcome of patients with ischemic stroke. Stroke. 2021;52:3908–3917. doi: 10.1161/strokeaha.121.034883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ameriso SF, Wong VL, Quismorio FP, Jr, Fisher M. Immunohematologic characteristics of infection-associated cerebral infarction. Stroke. 1991;22(8):1004–1009. doi: 10.1161/01.str.22.8.1004. [DOI] [PubMed] [Google Scholar]
- 44.Zeller JA, Lenz A, Eschenfelder CC, Zunker P, Deuschl G. Platelet-leukocyte interaction and platelet activation in acute stroke with and without preceding infection. Arterioscler Thromb Vasc Biol. 2005;25(7):1519–1523. doi: 10.1161/01.Atv.0000167524.69092.16. [DOI] [PubMed] [Google Scholar]
- 45.Wenzel J, Lampe J, Müller-Fielitz H, Schuster R, Zille M, Müller K, et al. The SARS-CoV-2 main protease Mpro causes microvascular brain pathology by cleaving NEMO in brain endothelial cells. Nat Neurosci. 2021;24(11):1522–1533. doi: 10.1038/s41593-021-00926-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Westendorp WF, Nederkoorn PJ, Vermeij JD, Dijkgraaf MG, van de Beek D. Post-stroke infection: a systematic review and meta-analysis. BMC Neurol. 2011;11:110. doi: 10.1186/1471-2377-11-110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Finlayson O, Kapral M, Hall R, Asllani E, Selchen D, Saposnik G, et al. Risk factors, inpatient care, and outcomes of pneumonia after ischemic stroke. Neurology. 2011;77(14):1338–1345. doi: 10.1212/WNL.0b013e31823152b1. [DOI] [PubMed] [Google Scholar]
- 48.Iadecola C, Buckwalter MS, Anrather J. Immune responses to stroke: mechanisms, modulation, and therapeutic potential. J Clin Invest. 2020;130(6):2777–2788. doi: 10.1172/jci135530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hou D, Wang C, Ye X, Zhong P, Wu D. Persistent inflammation worsens short-term outcomes in massive stroke patients. BMC Neurol. 2021;21(1):62. doi: 10.1186/s12883-021-02097-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Becker KJ, Kalil AJ, Tanzi P, Zierath DK, Savos AV, Gee JM, et al. Autoimmune responses to the brain after stroke are associated with worse outcome. Stroke. 2011;42(10):2763–2769. doi: 10.1161/strokeaha.111.619593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Becker KJ, Tanzi P, Zierath D, Buckwalter MS. Antibodies to myelin basic protein are associated with cognitive decline after stroke. J Neuroimmunol. 2016;295:9–11. doi: 10.1016/j.jneuroim.2016.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Vermeij JD, Westendorp WF, Dippel DW, van de Beek D, Nederkoorn PJ. Antibiotic therapy for preventing infections in people with acute stroke. Cochrane Database Syst Rev. 2018 doi: 10.1002/14651858.CD008530.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kishore AK, Jeans AR, Garau J, Bustamante A, Kalra L, Langhorne P, et al. Antibiotic treatment for pneumonia complicating stroke: recommendations from the pneumonia in stroke consensus (PISCES) group. Eur Stroke J. 2019;4(4):318–328. doi: 10.1177/2396987319851335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Prass K, Meisel C, Höflich C, Braun J, Halle E, Wolf T, et al. Stroke-induced immunodeficiency promotes spontaneous bacterial infections and is mediated by sympathetic activation reversal by poststroke T helper cell type 1-like immunostimulation. J Exp Med. 2003;198(5):725–736. doi: 10.1084/jem.20021098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Maier IL, Becker JC, Leyhe JR, Schnieder M, Behme D, Psychogios MN, et al. Influence of beta-blocker therapy on the risk of infections and death in patients at high risk for stroke induced immunodepression. PLoS ONE. 2018;13(4):e0196174. doi: 10.1371/journal.pone.0196174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Maier IL, Karch A, Mikolajczyk R, Bähr M, Liman J. Effect of beta-blocker therapy on the risk of infections and death after acute stroke–a historical cohort study. PLoS ONE. 2015;10(2):e0116836. doi: 10.1371/journal.pone.0116836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Benakis C, Martin-Gallausiaux C, Trezzi JP, Melton P, Liesz A, Wilmes P. The microbiome-gut-brain axis in acute and chronic brain diseases. Curr Opin Neurobiol. 2020;61:1–9. doi: 10.1016/j.conb.2019.11.009. [DOI] [PubMed] [Google Scholar]
- 58.Benakis C, Brea D, Caballero S, Faraco G, Moore J, Murphy M, et al. Commensal microbiota affects ischemic stroke outcome by regulating intestinal γδ T cells. Nat Med. 2016;22(5):516–523. doi: 10.1038/nm.4068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Benakis C, Poon C, Lane D, Brea D, Sita G, Moore J, et al. Distinct commensal bacterial signature in the gut is associated with acute and long-term protection from ischemic stroke. Stroke. 2020;51(6):1844–1854. doi: 10.1161/strokeaha.120.029262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Spychala MS, Venna VR, Jandzinski M, Doran SJ, Durgan DJ, Ganesh BP, et al. Age-related changes in the gut microbiota influence systemic inflammation and stroke outcome. Ann Neurol. 2018;84(1):23–36. doi: 10.1002/ana.25250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Battaglini D, Pimentel-Coelho PM, Robba C, dos Santos CC, Cruz FF, Pelosi P, et al. Gut microbiota in acute ischemic stroke: from pathophysiology to therapeutic implications. Front Neurol. 2020;11:598. doi: 10.3389/fneur.2020.00598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zeng X, Gao X, Peng Y, Wu Q, Zhu J, Tan C, et al. Higher risk of stroke is correlated with increased opportunistic pathogen load and reduced levels of butyrate-producing bacteria in the gut. Front Cell Infect Microbiol. 2019;9:4. doi: 10.3389/fcimb.2019.00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Yamashiro K, Tanaka R, Urabe T, Ueno Y, Yamashiro Y, Nomoto K, et al. Gut dysbiosis is associated with metabolism and systemic inflammation in patients with ischemic stroke. PLoS ONE. 2017;12(2):e0171521. doi: 10.1371/journal.pone.0171521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Erny D, de Angelis ALH, Jaitin D, Wieghofer P, Staszewski O, David E, et al. Host microbiota constantly control maturation and function of microglia in the CNS. Nat Neurosci. 2015;18(7):965–977. doi: 10.1038/nn.4030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chen R, Xu Y, Wu P, Zhou H, Lasanajak Y, Fang Y, et al. Transplantation of fecal microbiota rich in short chain fatty acids and butyric acid treat cerebral ischemic stroke by regulating gut microbiota. Pharmacol Res. 2019;148:104403. doi: 10.1016/j.phrs.2019.104403. [DOI] [PubMed] [Google Scholar]
- 66.Lee J, d’Aigle J, Atadja L, Quaicoe V, Honarpisheh P, Ganesh BP, et al. Gut microbiota-derived short-chain fatty acids promote poststroke recovery in aged mice. Circ Res. 2020;127(4):453–465. doi: 10.1161/CIRCRESAHA.119.316448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Tan C, Wu Q, Wang H, Gao X, Xu R, Cui Z, et al. Dysbiosis of gut microbiota and short-chain fatty acids in acute ischemic stroke and the subsequent risk for poor functional outcomes. J Parenter Enter Nutr. 2021;45(3):518–529. doi: 10.1002/jpen.1861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Camara-Lemarroy CR, Ibarra-Yruegas BE, Gongora-Rivera F. Gastrointestinal complications after ischemic stroke. J Neurol Sci. 2014;346(1–2):20–25. doi: 10.1016/j.jns.2014.08.027. [DOI] [PubMed] [Google Scholar]
- 69.Stanley D, Mason LJ, Mackin KE, Srikhanta YN, Lyras D, Prakash MD, et al. Translocation and dissemination of commensal bacteria in post-stroke infection. Nat Med. 2016;22(11):1277–1284. doi: 10.1038/nm.4194. [DOI] [PubMed] [Google Scholar]
- 70.Crapser J, Ritzel R, Verma R, Venna VR, Liu F, Chauhan A, et al. Ischemic stroke induces gut permeability and enhances bacterial translocation leading to sepsis in aged mice. Aging. 2016;8(5):1049–63. doi: 10.18632/aging.100952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.de Jong PR, González-Navajas JM, Jansen NJ. The digestive tract as the origin of systemic inflammation. Crit Care. 2016;20(1):279. doi: 10.1186/s13054-016-1458-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Houlden A, Goldrick M, Brough D, Vizi ES, Lénárt N, Martinecz B, et al. Brain injury induces specific changes in the caecal microbiota of mice via altered autonomic activity and mucoprotein production. Brain Behav Immun. 2016;57:10–20. doi: 10.1016/j.bbi.2016.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Singh V, Roth S, Llovera G, Sadler R, Garzetti D, Stecher B, et al. Microbiota dysbiosis controls the neuroinflammatory response after stroke. J Neurosci. 2016;36(28):7428–7440. doi: 10.1523/jneurosci.1114-16.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Lee YT, Mohd Ismail NI, Wei LK. Microbiome and ischemic stroke: a systematic review. PloS one. 2021;16(1):e0245038. doi: 10.1371/journal.pone.0245038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Yin J, Liao SX, He Y, Wang S, Xia GH, Liu FT, et al. Dysbiosis of gut microbiota with reduced trimethylamine-N-oxide level in patients with large-artery atherosclerotic stroke or transient ischemic attack. J Am Heart Assoc. 2015;4(11):e002699. doi: 10.1161/JAHA.115.002699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Li H, Zhang X, Pan D, Liu Y, Yan X, Tang Y, et al. Dysbiosis characteristics of gut microbiota in cerebral infarction patients. Transl Neurosci. 2020;11(1):124–133. doi: 10.1515/tnsci-2020-0117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Li N, Wang X, Sun C, Wu X, Lu M, Si Y, et al. Change of intestinal microbiota in cerebral ischemic stroke patients. BMC Microbiol. 2019;19(1):191. doi: 10.1186/s12866-019-1552-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Liesz A, Hu X, Kleinschnitz C, Offner H. Functional role of regulatory lymphocytes in stroke: facts and controversies. Stroke. 2015;46(5):1422–1430. doi: 10.1161/strokeaha.114.008608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Sadler R, Cramer JV, Heindl S, Kostidis S, Betz D, Zuurbier KR, et al. Short-chain fatty acids improve poststroke recovery via immunological mechanisms. J Neurosci. 2020;40(5):1162–1173. doi: 10.1523/JNEUROSCI.1359-19.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.