Abstract
Background
Vaccination has proven to be effective in preventing SARS-CoV-2 transmission and severe disease courses. However, immunocompromised patients have not been included in clinical trials and real-world clinical data point to an attenuated immune response to SARS-CoV-2 vaccines among patients with multiple sclerosis (MS) receiving immunomodulatory therapies.
Methods
We performed a retrospective study including 59 ocrelizumab (OCR)-treated patients with MS who received SARS-CoV-2 vaccination. Anti-SARS-CoV-2-antibody titres, routine blood parameters and peripheral immune cell profiles were measured prior to the first (baseline) and at a median of 4 weeks after the second vaccine dose (follow-up). Moreover, the SARS-CoV-2-specific T cell response and peripheral B cell subsets were analysed at follow-up. Finally, vaccination-related adverse events were assessed.
Results
After vaccination, we found anti-SARS-CoV-2(S) antibodies in 27.1% and a SARS-CoV-2-specific T cell response in 92.7% of MS cases. T cell-mediated interferon (IFN)-γ release was more pronounced in patients without anti-SARS-CoV-2(S) antibodies. Antibody titres positively correlated with peripheral B cell counts, time since last infusion and total IgM levels. They negatively correlated with the number of previous infusion cycles. Peripheral plasma cells were increased in antibody-positive patients. A positive correlation between T cell response and peripheral lymphocyte counts was observed. Moreover, IFN-γ release was negatively correlated with the time since the last infusion.
Conclusion
In OCR-treated patients with MS, the humoral immune response to SARS-CoV-2 vaccination is attenuated while the T cell response is preserved. However, it is still unclear whether T or B cell-mediated immunity is required for effective clinical protection. Nonetheless, given the long-lasting clinical effects of OCR, monitoring of peripheral B cell counts could facilitate individualised treatment regimens and might be used to identify the optimal time to vaccinate.
Keywords: multiple sclerosis
Introduction
COVID-19 is a highly infectious disease caused by the SARS-CoV-2. Rapid spread of SARS-CoV-2 has resulted in a global pandemic, with negative implications for people’s individual lives, healthcare systems and the economy. Vaccination against SARS-CoV-2 is a promising approach to reduce incidence and mortality of COVID-19, potentially curbing the global pandemic. Vaccines first became commercially available at the end of 2020, after clinical trials had shown high efficacy in preventing COVID-19 transmission and severe disease courses.1 2 Mechanistically, the antigenic target for COVID-19 vaccines is the spike (S) protein of SARS-CoV-2, which binds to the ACE 2 receptor on host cells mediating virus-cell fusion.3 Currently, different vaccine approaches are available including mRNA, replication-incompetent vector, recombinant protein and inactivated vaccines.4 Vaccines are administered in one or two intramuscular doses and elicit both a B cell response resulting in the production of binding and neutralising antibodies (abs) and a T cell response.5 However, clinical trials assessing the safety and efficacy of COVID-19 vaccines only included immunocompetent people while excluding patients receiving immunomodulatory therapies.1 2 Ocrelizumab (OCR), a selective monoclonal ab targeting CD20, is approved as a disease-modifying therapy (DMT) for relapsing-remitting and primary progressive multiple sclerosis (RRMS and PPMS, respectively).6 More than 200 000 patients have been treated with OCR globally.7 Mechanistically, OCR selectively depletes CD20-expressing cells by complement-mediated cytolysis and cell-mediated phagocytosis and cytotoxicity.6 While the majority of B cells express CD20, only 3%–5% of T cells are CD20-positive.8 9 B cells are an important component of the adaptive immune response providing protection against pathogens. Through production of various cytokines they shape and promote the T cell response and facilitate lymphoid tissue formation.10 Moreover, by terminal differentiation into plasma cells they are the source of antigen-specific immunoglobulin production.11 Accordingly, previous studies reported an attenuated humoral immune response after vaccination of patients receiving B cell-modulating therapies.12–15 However, particularly for these patients, an adequate immune response to vaccination is of great importance since they might be subject to an increased risk for infection, severe disease course and virus evolution.16–20 Despite their attenuated B cell response after COVID-19 infection or vaccination, the SARS-CoV-2 antigen-specific T cell response seems to be preserved.12 14 These results are consistent with the low expression levels of CD20 on T cells.8 9 However, studies assessing the humoral together with the cellular immune response after COVID-19 vaccination in relation to clinical parameters and peripheral immune cell profiles in patients with multiple sclerosis (MS) receiving OCR are scarce.12 21 In addition, differences in the peripheral B cell compartment and their impact on the SARS-CoV-2-specific immune response have previously not been studied. Here, we provide a comprehensive retrospective study aiming to investigate the humoral and T cell-mediated immune response to mRNA-based or vector-based COVID-19 vaccines in relation to the peripheral B cell profiles and clinical characteristics in 59 patients with MS treated with OCR.
Methods
Study population
Adult patients diagnosed with RRMS (ICD-10 (International Classification of Diseases Version 10) G35.1; n=39 (66.1%)) and PPMS (ICD-10 G35.2; n=20 (33.9%)), according to the 2017 revised McDonald criteria,22 who underwent treatment with OCR at the Department of Neurology of the University Hospital Düsseldorf, Germany, between 1 January 2020 and 30 April 2021 were retrospectively identified from the local database. Administration of OCR was performed according to national and international guidelines as well as to the most recent summary of product characteristics.6 The inclusion and exclusion criteria are summarised in table 1.
Table 1.
Inclusion and exclusion criteria
| Inclusion criteria | Exclusion criteria |
|
|
ab, antibody; COVID-19, Coronavirus disease 2019; MS, multiple sclerosis; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
The study was performed according to the Declaration of Helsinki and was approved by the local Ethics Committee of the Board of Physicians of the Region Nordrhein and of the Heinrich Heine University Düsseldorf, Germany (reference number: 5951R). All patients gave written informed consent to participate in the study.
Routine blood test
Differential blood count was assessed by automatic cell counting. Leucocyte subsets were assessed in a central laboratory (CD19+ B cells, CD3+ T cells, CD3+CD4+ T helper cells, CD3+CD8+ cytotoxic T cells and CD56+CD16+ NK cells) using flow cytometry (FC). Blood samples were prepared using the BD Multitest 6-Colour TBNK Reagent (BD Biosciences) according to the manufacturer’s instructions. A BD Canto (BD Biosciences) was used to acquire and analyse data. Serum IgA, IgM and IgG levels were measured by turbidimetry using a Cobas 8000 (C701, Roche Diagnostics).
Characterisation of B cell subsets
Blood samples were taken during clinical routine workup. Peripheral blood mononuclear cells (PBMCs) were isolated from whole blood by Ficoll gradient with SepMate isolation tubes (StemCell Technologies) and were cryopreserved in liquid nitrogen. For FC, PBMCs were thawed and resuspended in FC buffer (Phosphate Buffered Saline (PBS)/Bovine Serum Albumin (BSA)/EDTA). Samples were centrifuged for 5 min at 1500 rpm and 4°C and supernatant was discarded. FC buffer was added and cells were transferred to a 96-well plate. Centrifugation was repeated and cells were resuspended in FC buffer containing a FcR Blocking Reagent (Miltenyi Biotec). Cells were incubated for 5 min at room temperature. Next, the following staining antibodies, diluted in FC buffer, were added: anti-CD19 (HIB19), anti-IgD (IA6-2), anti-CD24 (ML5), anti-CD38 (HB-7), anti-CD20 (2H7), anti-CD14 (M5E2), anti-CD3 (SK7), anti-CD56 (HCD56), anti-CD138 (DL-101), anti-CD27 (M-T271), anti-CD21 (Bu32), all from BioLegend. Zombie Aqua Fixable Viability Kit (BioLegend) was used as a viability marker. Incubation was performed for 30 min at 4°C. Afterwards, cells were washed, centrifuged and resuspended in FC buffer. A CytoFLEX LX (Beckman Coulter) was used to acquire data. Analysis was performed with the software ‘Kaluza Flow Cytometry Analysis’ V.2.1 (Beckman Coulter) as illustrated in online supplemental figure 1A.
jnnp-2021-328197supp001.pdf (2.2MB, pdf)
Anti-SARS-CoV-2-antibody testing
Anti-SARS-CoV-2-abs in peripheral blood (PB) were analysed as part of the clinical routine workup. Immunoassays for the quantitative determination of abs to the SARS-CoV-2 spike (S) protein and nucleocapsid (N) protein (Elecsys Anti-SARS-CoV-2, Roche Diagnostics) were used. Assays were performed according to the manufacturer’s instructions. A titre≥0.8 (anti-SARS-CoV-2(S)-abs) and ≥1.0 (anti-SARS-CoV-2(N)-abs) was considered positive. Analysis was performed prior to and at a median of (~) 4 (range 2.7–8.1) weeks after the last COVID-19 vaccination (figure 1A).
Figure 1.
Long-lasting effects of ocrelizumab treatment. (A) Study design. (B) CD19 B cells in relation to time since the last infusion. Correlation analysis was performed with the Spearman correlation coefficient. The area in between the dotted lines shows the 95% CI. B cells<1% are depicted on the x-axis. BL, baseline; F/U, follow-up; IFN, interferon; Ig, immunoglobulin; Inj, injection; MS, multiple sclerosis; OCR, ocrelizumab; PPMS, primary progressive multiple sclerosis; RRMS, relapsing-remitting multiple sclerosis; V, visit.
Quantification of T cell response to SARS-CoV-2
The SARS-CoV-2 interferon-gamma release assay (IGRA; Euroimmun) was used to assess the T cell response to SARS-CoV-2 in PB ~4 weeks after the second dose of COVID-19 vaccination. Blood samples were prepared using the SARS-CoV-2-IGRA stimulation kit (Euroimmun) according to manufacturer’s instructions. Briefly, 500 µL of lithium heparin blood was transferred to the reaction tubes: CoV-2 IGRA BLANK (negative control), CoV-2 IGRA TUBE (containing the recombinant S1 subunit of the SARS-CoV-2 spike protein) and CoV-2 IGRA STIM (mitogen-coated tubes). The tubes were inverted six times and incubated for 24 hours at 37°C. Samples were then centrifuged at 12 000×g for 10 min, plasma was removed and transferred to fresh polypropylene reaction tubes. The SARS-CoV-2-IGRA was performed at the Clinical Immunological Laboratory Professor Dr Med. Winfried Stöcker (Lübeck). A SARS-CoV-2-specific T cell response was assumed when an interferon-gamma concentration of >200 mIU/mL was detected.
Data analysis
Data analysis and visualisation was performed with ‘GraphPad Prism’ (V.9.0.0). Data are presented as median with range. D'Agostino & Pearson test was used to test for normality. Correlation analysis was performed with the Spearman correlation coefficient. Differences between two groups were determined using the Mann-Whitney U test. A p value of <0.05 was considered significant.
Results
Retrospective identification of patients and baseline cohort characteristics
In total, 39 RRMS (66.1%) and 20 PPMS (33.9%) patients treated with OCR who received anti-SARS-CoV-2-ab testing prior to (baseline, BL) and ~4 weeks after COVID-19 vaccination (follow-up, F/U) were included (figure 1A). Median age was 45 (23–62) years for the RRMS and 56 (43–65) years for the PPMS cohort. Around half of the patients (45.8%) were female. Median disease duration was 14.0 (2.0–35.0) years (RRMS) and 10.0 (3.0–37.0) years (PPMS), median Expanded Disability Status Scale score (EDSS) at BL was 3.5 (0.0–6.5) (RRMS) and 6.0 (2.0–7.5) (PPMS). Median duration of OCR treatment was 2.5 (0.4–8.3) years for patients with RRMS and 2.4 (0.3–3.6) years for patients with PPMS (table 2). Of these patients, 66.1% received one or more DMT(s) prior to OCR. For 61.0% of patients, comorbidities were noted (online supplemental table 1). Median time between the last OCR infusion and first vaccination was 3.9 (0.1–12.2) months (table 2). Of these patients, 92.7% reported side effects of vaccination, with ‘pain at injection site’, ‘fatigue’, ‘headache’ and ‘muscle and joint pain’ being the most common ones.
Table 2.
Demographics and basic disease characteristics of patients with MS
| RRMS | PPMS | |
| Patients (n (%)) | 39.0 (66.1) | 20.0 (33.9) |
| Age (years) (median (range)) | 45 (23–62) | 56(43–65) |
| Sex (% female) | 45.8 | 46.2 |
| Disease duration (years) (median (range)) | 14.0 (2.0–35.0) | 10.0 (3.0–37.0) |
| Time since first diagnosis (years) (median (range)) | 12.0 (1.0–30.0) | 4.5 (2.0–18.0) |
| Duration of OCR treatment (years) (median (range)) | 2.5 (0.4–8.3) | 2.4 (0.3–3.6) |
| Previous OCR cycles | 5.0 (1.0–15.0) | 5.0 (1.0–9.0) |
| EDSS at BL (median (range)) | 3.5 (0.0–6.5) | 6.0 (2.0–7.5) |
| Relapses since start of OCR | 0 | – |
| EDSS worsening since last infusion (n (%)) | – | 5/13 (38.5%) |
| Number of previous therapies (median (range)) | 2.0 (0.0–4.0) | 0.0 (0.0–2.0) |
| Last infusion to vaccination (months) (median (range)) | 4.0 (1.7–7.1) | 3.7 (0.1–12.2) |
BL, baseline; EDSS, Expanded Disability Status Scale; F/U, follow-up; MS, multiple sclerosis; OCR, ocrelizumab; PPMS, primary progressive multiple sclerosis; RRMS, relapsing-remitting multiple sclerosis.
jnnp-2021-328197supp002.pdf (103.1KB, pdf)
Long-lasting effects of OCR treatment and low probability of adverse events
Long-lasting B cell depletion and stable disease courses among OCR-treated patients with MS were previously reported.23 Recommended intervals between vaccination and OCR infusion should be considered. As a result, extension of dosing intervals may be necessary for some patients with MS receiving OCR. We thus assessed the time from the last treatment cycle, EDSS before the last treatment cycle, EDSS at BL and F/U, relapses since the last infusion and side effects. Median time between BL and last OCR treatment was 4.6 (0.2–12.2) months. Median time between F/U and last OCR treatment was 6.4 (2.5–13.8) months. Regarding the clinical course, none of the patients with RRMS had experienced relapses since the first OCR treatment cycle. Moreover, in 5 out of 13 patients with PPMS (38.5%) with available EDSS prior to the last infusion and at F/U, worsening of EDSS since the last infusion was noted. Two out of 19 patients with PPMS (10.5%) with available EDSS at BL and F/U experienced EDSS worsening during the course of the study (online supplemental table 1). Peripheral B cells were <1% in all but three patients (94.8%) at BL and 72.9% of patients at F/U. The amount of peripheral B cells at BL and F/U positively correlated with the time from the last treatment cycle (figure 1B). 18.5% of patients reported mild adverse events of OCR treatment (online supplemental table 1). Abnormalities in routine laboratory studies were noted in some cases, decreased IgM level being the most common one (40.7% of patients) (online supplemental figure 2). Overall, these data suggest long-lasting suppression of peripheral B cells in patients with MS receiving OCR and a stable clinical disease course even exceeding the duration of maximal B cell depletion. Adverse events rarely occurred and were mild in all cases.
Patients with MS treated with OCR show attenuated antibody response to COVID-19 vaccination
Attenuated humoral immune response after vaccination, including COVID-19, has been reported among patients receiving B cell-modulating therapies.12 We analysed anti-SARS-CoV-2(S)-abs ~4 weeks after the second dose of COVID-19 vaccine as well as peripheral B cell counts at BL and F/U. The majority of patients were vaccinated with the BioNTech vaccine (BioNTech: 55 (94.8%), Astra-Zeneca: 2 (3.4%), BioNTech/Astra-Zeneca: 1 (1.7%)) (online supplemental table 1). Anti-SARS-CoV-2(S)-abs were positive in 27.1% of patients at F/U with titres ranging from 1.0 to 954 U/mL (figure 2A). Anti-SARS-CoV-2(N)-abs were negative in all patients at BL and F/U (figure 2A). Anti-SARS-CoV-2(S)-ab titres positively correlated with peripheral B cell counts at BL and F/U (figure 2B), with the time since the last infusion cycle, and with total IgM levels at BL and F/U (figure 2C, D). In addition, a negative correlation between anti-SARS-CoV-2(S)-abs and the number of previous treatment cycles was noted (figure 2E). No correlations could be detected between ab titres and overall IgG and IgA levels at BL and F/U and the age (online supplemental file 3A-C). In five patients, anti-SARS-CoV-2(S)-abs were detectable despite depleted peripheral B cells. In contrast, four patients with detectable peripheral B cells did not develop anti-SARS-CoV-2(S)-abs. To further elucidate these observations, we performed differential B cell phenotyping in 32 patients at F/U (online supplemental figure 1A). As expected, patients with anti-SARS-CoV-2(S)-abs showed higher amounts of B cells compared with patients without abs (figure 2F). Additionally, the proportion of plasma cells was significantly higher in patients with abs compared with those without (figure 2F). When we compared the five patients with abs despite depleted peripheral B cells with patients without abs and depleted B cells, we detected significantly elevated amounts of plasmablasts in PB (figure 2G). No significant differences in B cell subpopulations were seen between the four patients with detectable peripheral B cells without abs in comparison to patients with detectable peripheral B cells and abs (online supplemental figure 1D). In addition, anti-SARS-CoV-2(S)-abs did not differ between patients with RRMS and PPMS (online supplemental figure 3D). Four patients with negative anti-SARS-CoV-2(S)-abs after two doses received a third vaccination dose. However, an increase in the ab titre from <0.4 to 25.4 U/mL and from <0.4 to 44.4 U/mL, respectively, could only be observed in the patients who had a detectable peripheral B cell count (14 /µL and 47 /µL, respectively) prior to third vaccination. In summary, OCR-treated patients with MS have an attenuated humoral immune response to COVID-19 vaccination and ab titres positively correlated with peripheral B cell counts, time since last infusion, and total IgM levels and negatively correlated with the number of previous OCR cycles.
Figure 2.
Attenuated humoral immune response to SARS-CoV-2 vaccination in patients with MS receiving ocrelizumab. (A) Box plot analysis of anti-SARS-CoV-2(N)-abs and anti-SARS-CoV-2(S)-abs at BL and F/U: Lower quartile, median and upper quartile are illustrated by boxes. Whiskers show the min and max values. Individual values are depicted by black dots. Values<0.1 U/mL (anti-SARS-CoV-2(N)) and <0.4 U/mL (anti-SARS-CoV-2(S)) are depicted on the x-axis. Cut-off values are illustrated by dashed red lines. (B–E) Correlation of anti-SARS-CoV-2(S)-abs with peripheral B cell counts (B), time since last infusion (C), total IgM levels in PB (D), and number of previous treatment cycles (E). Correlation analysis was performed with the Spearman correlation coefficient. The area in between the dotted lines shows the 95% CI. B cell counts <10 /µL are depicted on the y-axis and anti-SARS-CoV-2(S)-abs<0.4 U/mL are shown on the x-axis. (F) Box plot analysis comparing the amount of peripheral CD20 B cells and plasma cells (assessed by mFC) at F/U between patients with and without anti-SARS-CoV-2(S)-abs: Lower quartile, median and upper quartile are illustrated by boxes. Whiskers show the min and max values. Individual values are depicted by black dots. (G) Box plot analysis comparing the amount of peripheral plasmablasts (assessed by mFC) at F/U between patients with anti-SARS-CoV-2(S)-abs despite depleted peripheral B cells and patients without abs and depleted B cells: Lower quartile, median and upper quartile are illustrated by boxes. Whiskers show the min and max values. Individual values are depicted by black dots. ab(s), antibody(ies); BL, baseline; F/U, follow-up; Ig, immunoglobulin; max, maximum; mFC, multidimensional flow cytometry; min, minimum; MS, multiple sclerosis; N, nucleocapsid; neg., negative; OCR, ocrelizumab; PB, peripheral blood; pos., positive; S, spike.
Preserved T cell response to SARS-CoV-2 antigens in patients with MS receiving OCR after vaccination
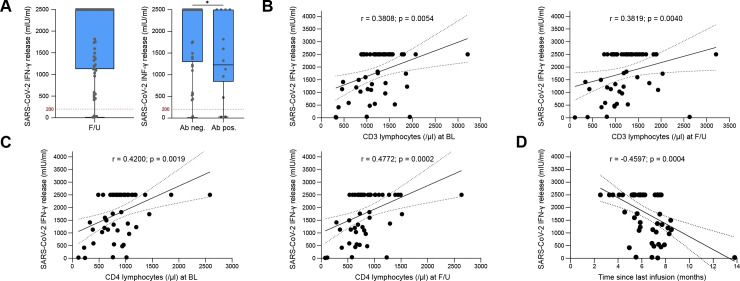
While the majority of B cells express CD20, the amount of T cells expressing CD20 is low.8 9 Accordingly, preserved T cell responses after COVID-19 infection and vaccination have been reported in patient with MSs receiving OCR.12 14 To further corroborate these observations, we assessed the SARS-CoV-2 antigen-specific T cell response of 55 patients with MS receiving OCR at F/U. To this end, we measured the release of interferon (IFN)-γ by T cells following stimulation with the SARS-CoV-2(S) protein. We found a T cell response in 92.7% of patients (figure 3A). No differences could be detected between patients with RRMS and PPMS (online supplemental figure 3E). In four patients, there was no SARS-CoV-2-specific T cell response. Regarding basic clinical characteristics, those patients did not markedly differ from the patients who developed a pronounced T cell response after vaccinations. Positive anti-SARS-CoV-2(S)-abs at F/U were found in two out of four patients (50%). However, total numbers of peripheral lymphocytes (CD3) and lymphocyte subsets (CD4 and CD8) tended to be lower in the patients with negative T cell response but did not reach significance (data not shown). We further divided our MS cohort into two groups depending on the development of anti-SARS-CoV-2(S)-abs. IFN-γ release by SARS-CoV-2-specific T cells was higher in patients with MS without detectable abs (figure 3A). IFN-γ release positively correlated with CD3 and CD4 lymphocyte counts and negatively correlated with the time since last infusion (figure 3B–D). No significant correlations were observed between T cell response and anti-SARS-CoV-2(S)-abs, CD8 lymphocytes and the number of previous treatment cycles (online supplemental figure 3F–H). In summary, we detected a robust T cell-mediated response to SARS-CoV-2 antigens after two doses of vaccination, which was even more pronounced in patients without detectable anti-SARS-CoV-2(S)-abs. T cell response positively correlated with CD3 and CD4 lymphocyte counts and negatively correlated with the time since the last infusion.
Figure 3.
Preserved T cell response to SARS-CoV-2 vaccination in patients with MS receiving ocrelizumab. (A) Box plot analysis of SARS-CoV-2-specific T cell response measured by IFN-γ release at F/U and comparison of SARS-CoV-2-specific T cell response between patients with and without detectable anti-SARS-CoV-2(S)-abs at F/U: Lower quartile, median and upper quartile are illustrated by boxes. Whiskers show the min and max values. Individual values are depicted by black dots. Values>2500 mIU/mL are shown as 2500 mIU/mL. Cut-off values are illustrated by dashed red lines. (B–D) Correlation of IFN-γ release by SARS-CoV-2-specific T cells with CD3 (B) and CD4 lymphocytes at BL and F/U (C) and time since last infusion (D). Correlation analysis was performed with the Spearman correlation coefficient. The area in between the dotted lines shows the 95% CI. Values>2500 mIU/mL are depicted as 2500 mIU/mL. ab(s), antibody(ies); BL, baseline; F/U, follow-up; IFN, interferon; max, maximum; min, minimum; MS, multiple sclerosis; OCR, ocrelizumab; S, spike.
Discussion
Pivotal clinical trials have shown high efficacy of COVID-19 vaccines in preventing virus transmission and severe disease courses.1 2 Accordingly, non-immunocompromised people seem to develop a robust antibody and T cell response after two doses of COVID-19 vaccination.1 2 24 25 However, immunocompromised patients, who can be at increased risk for infections,16–20 have been excluded from those trials. Studies assessing the risk of infections in patients with MS receiving OCR yielded contrasting results. While some reported an increased susceptibility to COVID-19 or a more severe disease course,26–28 others could not link treatment with B cell-depleting therapies to higher infection rates.29–31 Post-approval studies reported an attenuated humoral immune response to COVID-19 vaccination among patients receiving B cell-modulating therapies, while T cell response seemed to be preserved.12–15 Accordingly, we assessed the anti-SARS-CoV-2-abs in a sizeable cohort of 59 patients with MS receiving OCR and could only detect positive anti-SARS-CoV-2(S)-abs in 27.1% of patients. In contrast, anti-SARS-CoV-2-ab response was previously found in the majority of patients with MS on other DMTs (eg, fingolimod 85.7%, IFN-β 96.4% and cladribine 100%).32 As expected, anti-SARS-CoV-2(S)-abs correlated with the amount of peripheral B cells in OCR-treated patients with MS. This is in line with previous observations in patients receiving B cell-modulating therapies.12 15 33 B cells can be divided into subpopulations with unique phenotypes and functions.11 34 Correspondingly, expression of CD20 varies within the B cell compartment. On antigen stimulation, B cells proliferate and differentiate into plasmablasts and plasma cells.11 These cells, which typically do not express CD20 on their surface, produce antigen-specific immunoglobulins.11 35 Thus, impaired generation of plasma cells from CD20-positive B cell progenitors most likely accounts for the attenuated humoral immune response in patients receiving anti-CD20 therapies. This is underscored by our observation that patients with positive anti-SARS-CoV-2(S)-abs have higher amounts of plasma cells in PB compared with patients without abs. Nevertheless, some patients were able to mount an adequate humoral immune response despite depleted peripheral B cells, as previously reported.12 Differential B cell phenotyping revealed higher proportions of peripheral plasmablasts in those patients. Plasmablasts are rapidly generated from B cell progenitors on antigen stimulation; however, their longevity is limited compared with plasma cells.11 Thus, it is tempting to speculate that in patients with depleted B cells, the production of anti-SARS-CoV-2(S)-abs mainly occurs in the short-lived plasmablast compartment in the process of early B cell repopulation after OCR infusion. However, further studies will be needed to phenotype B cell populations at different time points after OCR treatment. This will help to further clarify differences in the humoral immune response in relation to B cell repopulation kinetics among patients treated with OCR. Regarding pharmacokinetics, OCR rapidly depletes B cell numbers and mediates long-lasting B cell depletion. Repopulation of B cells begins about 6 months after the last infusion and B cell counts return to normal numbers at a median of 72 (27–175) weeks.6 36 However, clinical efficacy seems to outlast B cell depletion, which is a still unclarified phenomenon. Our results indicate a variability in B cell repopulation after OCR infusion. In a subgroup of patients, B cells were still fully depleted more than 6 months after the last treatment cycle. Accordingly, no relapses occurred in that subgroup. Interestingly, however, there were no relapses in patients with detectable peripheral B cells, either. This points to a prolonged clinical effect of OCR exceeding actual B cell depletion, in the sense of a long-lasting immunomodulation, which is consistent with previous studies.23 This concept is further corroborated by our observation that anti-SARS-CoV-2(S)-abs negatively correlate with the number of OCR treatment cycles. Given the high variability in the dynamics of B cell repopulation and the long-lasting clinical effects of OCR treatment, B cell monitoring and individual treatment regimens adapted to the peripheral B cell count might be a promising approach warranting further investigations. Patients with delayed B cell-repopulation kinetics might be ‘overtreated’ by receiving OCR every 6 months. Thus, individualised treatment regimens could minimise the time spent in inpatient or outpatient clinics associated with lower healthcare cost and might reduce the risk of infections in a subgroup of patients. Additionally, monitoring the CD19 B cells might be a promising tool to identify the optimal time to vaccinate. This is underscored by our and previous12 observations that peripheral B cell counts, and thus the potential to mount a humoral immune response, correlate with anti-SARS-CoV-2(S)-abs. Based on our results, measuring peripheral plasma cells and plasmablasts could add value to CD19 B cell monitoring. We further analysed anti-SARS-CoV-2(S)-abs in two patients who received a third vaccination dose after partial repopulation of peripheral B cells and found an increase in the anti-SARS-CoV-2(S)-ab titre. This further illustrates the general potential of this vaccination strategy. In this context, it might be a reasonable time to vaccinate when at least 1% of B cells can be detected in PB. However, further longitudinal studies will be necessary to assess the actual benefit of this approach and to determine the appropriate B cell levels to ensure an optimal risk/benefit ratio. In addition to the humoral immune system, adaptive immunity, especially T cells, are essential for the antiviral immune response.37 Besides the well-described acute response in which T cells kill virus-infected cells, memory T cells mediate long-lasting immune competence against viral infections such as SARS-CoV. In contrast, SARS-CoV-specific abs rapidly declined after 4 months post infection.38 39 The importance of T cell-mediated mechanisms in COVID-19 is emphasised by the reduction of peripheral lymphocyte counts (CD4 and CD8 T cells) accompanied by an increase in activation markers and IFN-γ-production in severe cases.40 These cases also frequently featured T cell exhaustion.41 We detected a preserved T cell-mediated immune response against SARS-CoV-2 in OCR-treated patients with MS which is consistent with previous data reporting an increase in T cell response after COVID-19 infection and vaccination.12 14 As opposed to the low percentage of patients who developed anti-SARS-CoV-2-abs after vaccination, the SARS-CoV-2-specific T cell response seems to be higher in OCR-treated patients with MS compared with patients receiving other DMTs (eg, IFN-β 89.3%, cladribine 70% and fingolimod 14.3% of patients).32 Of note, IFN-γ release by SARS-CoV-specific T cells was even more pronounced in OCR-treated patients with MS without anti-SARS-CoV-2-abs which might point towards a compensatory effect for the insufficient humoral immune response. IFN-γ release correlated with peripheral CD3 and CD4 lymphocyte counts, but no correlation with CD8 lymphocytes was observed. It is conceivable that a more prominent IFN-γ response of CD4 compared with CD8 lymphocytes, as previously described,42 might contribute to this observation. IFN-γ release by activated CD4 lymphocytes is crucial for CD8 T cell priming.42 In turn, CD8 T cells are able to eliminate virus-infected cells through T cell receptor-mediated recognition of viral antigens.43 Moreover, a negative correlation between T cell-mediated IFN-γ release and time since last infusion was observed. This might be connected to the fact that B cell-depleting effects of OCR are strongest within the first months after OCR infusion and B cells begin to repopulate over time. In this regard, the negative correlation between T cell response and time since last infusion further supports the concept of T cell compensation for the attenuated B cell response. In four patients, no SARS-CoV-2-specific T cell response could be detected. Lower numbers of peripheral lymphocytes (CD3) and lymphocyte subsets (CD4 and CD8) might contribute to this observation. However, further studies with larger patient cohorts will be necessary to corroborate those findings. Taken together, our results indicate that the T cell arm of the adaptive immunity is able to mount an adequate immune response against SARS-CoV-2. Measuring the SARS-CoV-2-specific T cellular immune response after COVID-19 vaccination might be suitable to assess immunity against SARS-CoV-2 in patients treated with B cell-depleting therapies. However, further studies will be needed to evaluate whether a compromised anti-SARS-CoV-2-ab production impairs immunity against SARS-CoV-2 despite the robust T cell response. Apart from the efficacy of vaccination, the safety profile is another crucial aspect. Analysis of adverse events after SARS-CoV-2 vaccination showed that they were mild in all cases. Furthermore, none of the patients with RRMS reported relapses after vaccination. EDSS worsening between BL and F/U was noted in 2 out of 19 patient with PPMSs, most likely due to the natural progressive clinical disease course. Our data therefore suggest that short-term safety profiles of COVID-19 vaccines seem to be favourable among patients with MS receiving B cell-depleting therapies. This is in line with data from MS centres in Israel and Italy.44 45 However, the long-term safety profile of COVID-19 vaccination in patients with MS will have to be investigated in future studies.
We are aware that our study is limited by its retrospective design. As a result of this design, not all data were available for every patient and BL anti-SARS-CoV-2(S)-ab titres were assessed more than 2 weeks prior to first vaccination in some cases. However, none of the patients reported a history of SARS-CoV-2, and anti-SARS-CoV-2(N)-abs were negative at F/U in all patients. Besides, we did not subclassify anti-SARS-CoV-2(S)-abs into IgM, IgA and IgG. Moreover, time between vaccinations and from second vaccination to F/U was variable, and long-term effects of vaccination as well as long-term safety profiles were not assessed.
In conclusion, our results indicate that patients with MS receiving OCR can mount an adequate T cellular immune response to SARS-CoV-2 vaccination while anti-SARS-CoV-2(S)-abs production seems to be compromised. Measuring T cell effector mechanisms could therefore be suitable to assess vaccine response in immunocompromised patients. Given the long-lasting and heterogeneous effects of OCR, monitoring of peripheral B cells could facilitate individualised treatment regimens and might be a tool to identify the optimal time to vaccinate.
Footnotes
SR and MK contributed equally.
SGM and DK contributed equally.
Contributors: SR, MK, LR, NH, TK and JI identified patients. SR, MK, NH, TM, TK, VD, AMH, KvWL, CBS, CN, LRN, BL and BT performed data acquisition. SR performed data analyses. SGM and DK conceived the study. SGM and DK supervised the study. TR, NM, OA and PA co-supervised the study. SR and DK wrote the manuscript. MB and H-PH gave valuable scientific input to the manuscript. All authors critically revised the manuscript and agreed with its content. DK accepts full responsibility for the finished work and the conduct of the study, had access to the data, and controlled the decision to publish.
Funding: This project was supported by a grant from the DMSG OV Düsseldorf u.U.e.V. (no grant number).
Competing interests: MK received travel grants from Merck Serono and Biogen. LR received travel reimbursements from Merck Serono and Sanofi Genzyme. MP's research is funded by the German Multiple Sclerosis Society North Rhine-Westphalia (DMSG), Novartis and the programme "Innovative Medizinische Forschung" (IMF) of the Medical Faculty of the University of Muenster. MB served on scientific advisory boards for Biogen, Novartis and Genzyme and has received conference travel support from Biogen and Novartis. He serves on steering committees for trials conducted by Novartis. His institution has received research support from Biogen, Merck and Novartis. H-PH has received fees for consulting, speaking and serving on steering committees from Bayer Healthcare, Biogen, GeNeuro, MedImmune, Merck, Novartis, Opexa, Receptos Celgene, Roche, Sanofi Genzyme, CSL Behring, Octapharma and Teva, with approval from the Rector of Heinrich-Heine-University. OA received personal fees from Alexion, Bayer Healthcare, Biogen, Celgene, Merck Serono, MedImmune, Novartis, Roche, Teva and Zambon, and he received research support from the German Science Foundation (DFG) and the German Ministry of Education, Science, Research and Technology (BMBF), outside of the submitted work. PA received compensation for serving on Scientific Advisory Boards and/or speaker honoraria and/or travel support from Novartis, Teva, Biogen, Bristol Meyers Squibb, Celgene, Janssen Cilag, Merz Pharmaceuticals, Ipsen, Allergan, Bayer Healthcare, Esai, UCB and Glaxo Smith Kline, Roche; he received research support from Novartis, Biogen, Celgene, Teva, Merz Pharmaceuticals, Ipsen and Roche. NM has received honoraria for lecturing and travel expenses for attending meetings from Biogen Idec, GlaxoSmith Kline, Teva, Novartis Pharma, Bayer Healthcare, Genzyme, Alexion Pharamceuticals, Fresenius Medical Care, Diamed and BIAL, and has received financial research support from Euroimmun, Fresenius Medical Care, Diamed, Alexion Pharmaceuticals and Novartis Pharma. TR reports grants from German Ministry of Education, Science, Research and Technology, during the conduct of the study; grants and personal fees from Sanofi-Genzyme; personal fees from Biogen; personal fees and non-financial support from Merck Serono; personal fees from Roche; and personal fees from Teva, outside the submitted work. SGM received honoraria for lecturing and travel expenses for attending meetings from Almirall, Amicus Therapeutics Germany, Bayer Health Care, Biogen, Celgene, Diamed, Genzyme, MedDay Pharmaceuticals, Merck Serono, Novartis, Novo Nordisk, ONO Pharma, Roche, Sanofi-Aventis, Chugai Pharma, QuintilesIMS and Teva. His research is funded by the German Ministry for Education and Research (BMBF), Deutsche Forschungsgemeinschaft (DFG), Else Kröner Fresenius Foundation, German Academic Exchange Service, Hertie Foundation, Interdisciplinary Center for Clinical Studies (IZKF) Muenster, German Foundation Neurology, and by Almirall, Amicus Therapeutics Germany, Biogen, Diamed, Fresenius Medical Care, Genzyme, Merck Serono, Novartis, ONO Pharma, Roche and Teva. DK received travel grants from GeNeuro and Merck, refund of congress participation fees from GeNeuro, Merck and Servier, consulting fees from Grifols, payment for lectures from Grifols, support for research projects from Teva and was funded by the Deutsche Forschungsgemeinschaft (DFG).
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request. Further information and requests for resources and anonymised clinical data should be directed to and will be fulfilled by David Kremer (DavidMaxJoe.Kremer@med.uni-duesseldorf.de)
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
This study involves human participants and was approved by local Ethics Committee of the Board of Physicians of the Region Nordrhein and of the Heinrich Heine University Düsseldorf, Germany (reference number: 5951R). Participants gave informed consent to participate in the study before taking part.
References
- 1. Polack FP, Thomas SJ, Kitchin N, et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med 2020;383:2603–15. 10.1056/NEJMoa2034577 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Baden LR, El Sahly HM, Essink B, et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med 2021;384:403–16. 10.1056/NEJMoa2035389 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Huang Y, Yang C, Xu X-F, et al. Structural and functional properties of SARS-CoV-2 spike protein: potential antivirus drug development for COVID-19. Acta Pharmacol Sin 2020;41:1141–9. 10.1038/s41401-020-0485-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Wu Q, Dudley MZ, Chen X, et al. Evaluation of the safety profile of COVID-19 vaccines: a rapid review. BMC Med 2021;19:173. 10.1186/s12916-021-02059-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Walsh EE, Frenck RW, Falsey AR, et al. Safety and immunogenicity of two RNA-based Covid-19 vaccine candidates. N Engl J Med 2020;383:2439–50. 10.1056/NEJMoa2027906 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. European Medicines Agency . Ocrevus - Summary of product characteristics [Internet]. Available: https://ec.europa.eu/health/documents/community-register/2021/20210111150165/anx_150165_en.pdf [Accessed 15 Jun 2021].
- 7. Roche H-L. New data for Roche’s OCREVUS (ocrelizumab) reinforce significant benefit on slowing disease progression in relapsing and primary progressive multiple sclerosis [Internet]. Available: https://www.roche.com/media/releases/med-cor-2021-04-16.htm [Accessed 06 Oct 2021].
- 8. Leandro MJ. B-cell subpopulations in humans and their differential susceptibility to depletion with anti-CD20 monoclonal antibodies. Arthritis Res Ther 2013;15 Suppl 1:S3. 10.1186/ar3908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Chen Q, Yuan S, Sun H, et al. CD3+CD20+ T cells and their roles in human diseases. Hum Immunol 2019;80:191–4. 10.1016/j.humimm.2019.01.001 [DOI] [PubMed] [Google Scholar]
- 10. Shen P, Fillatreau S. Antibody-independent functions of B cells: a focus on cytokines. Nat Rev Immunol 2015;15:441–51. 10.1038/nri3857 [DOI] [PubMed] [Google Scholar]
- 11. Nutt SL, Hodgkin PD, Tarlinton DM, et al. The generation of antibody-secreting plasma cells. Nat Rev Immunol 2015;15:160–71. 10.1038/nri3795 [DOI] [PubMed] [Google Scholar]
- 12. Apostolidis SA, Kakara M, Painter MM, et al. Cellular and humoral immune responses following SARS-CoV-2 mRNA vaccination in patients with multiple sclerosis on anti-CD20 therapy. Nat Med 2021;27:1990–2001. 10.1038/s41591-021-01507-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Monschein T, Hartung H-P, Zrzavy T, et al. Vaccination and multiple sclerosis in the era of the COVID-19 pandemic. J Neurol Neurosurg Psychiatry 2021;92:1033–43. 10.1136/jnnp-2021-326839 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Brill L, Rechtman A, Zveik O, et al. Humoral and T-cell response to SARS-CoV-2 vaccination in patients with multiple sclerosis treated with Ocrelizumab. JAMA Neurol 2021;78:1510. 10.1001/jamaneurol.2021.3599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Disanto G, Sacco R, Bernasconi E, et al. Association of disease-modifying treatment and anti-CD20 infusion timing with humoral response to 2 SARS-CoV-2 vaccines in patients with multiple sclerosis. JAMA Neurol 2021;78:1529. 10.1001/jamaneurol.2021.3609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Hauser SL, Bar-Or A, Comi G, et al. Ocrelizumab versus interferon beta-1a in relapsing multiple sclerosis. N Engl J Med 2017;376:221–34. 10.1056/NEJMoa1601277 [DOI] [PubMed] [Google Scholar]
- 17. Montalban X, Hauser SL, Kappos L, et al. Ocrelizumab versus placebo in primary progressive multiple sclerosis. N Engl J Med 2017;376:209–20. 10.1056/NEJMoa1606468 [DOI] [PubMed] [Google Scholar]
- 18. Ren L, Zhang L, Chang D, et al. The kinetics of humoral response and its relationship with the disease severity in COVID-19. Commun Biol 2020;3:780. 10.1038/s42003-020-01526-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Luna G, Alping P, Burman J, et al. Infection risks among patients with multiple sclerosis treated with fingolimod, natalizumab, rituximab, and injectable therapies. JAMA Neurol 2020;77:184–91. 10.1001/jamaneurol.2019.3365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Corey L, Beyrer C, Cohen MS, et al. SARS-CoV-2 variants in patients with immunosuppression. N Engl J Med 2021;385:562–6. 10.1056/NEJMsb2104756 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ferguson J, Murugesan K, Banaei N. Interferon-Gamma release assay testing to assess COVID-19 vaccination response in a SARS-CoV-2 seronegative patient on rituximab: a case report. Int J Infect Dis 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Thompson AJ, Banwell BL, Barkhof F, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 2018;17:162–73. 10.1016/S1474-4422(17)30470-2 [DOI] [PubMed] [Google Scholar]
- 23. Rolfes L, Pawlitzki M, Pfeuffer S, et al. Ocrelizumab extended interval dosing in multiple sclerosis in times of COVID-19. Neurol Neuroimmunol Neuroinflamm 2021;8. 10.1212/NXI.0000000000001035. [Epub ahead of print: 14 Jul 2021]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Bayram A, Demirbakan H, Günel Karadeniz P, et al. Quantitation of antibodies against SARS-CoV-2 spike protein after two doses of CoronaVac in healthcare workers. J Med Virol 2021;93:5560–7. 10.1002/jmv.27098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Woldemeskel BA, Garliss CC, Blankson JN. SARS-CoV-2 mRNA vaccines induce broad CD4+ T cell responses that recognize SARS-CoV-2 variants and HCoV-NL63. J Clin Invest 2021;131. 10.1172/JCI149335. [Epub ahead of print: 17 May 2021]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Sormani MP, De Rossi N, Schiavetti I, et al. Disease-Modifying therapies and coronavirus disease 2019 severity in multiple sclerosis. Ann Neurol 2021;89:780–9. 10.1002/ana.26028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Sahraian MA, Azimi A, Navardi S, et al. Evaluation of the rate of COVID-19 infection, hospitalization and death among Iranian patients with multiple sclerosis. Mult Scler Relat Disord 2020;46:102472. 10.1016/j.msard.2020.102472 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Safavi F, Nourbakhsh B, Azimi AR. B-cell depleting therapies may affect susceptibility to acute respiratory illness among patients with multiple sclerosis during the early COVID-19 epidemic in Iran. Mult Scler Relat Disord 2020;43:102195. 10.1016/j.msard.2020.102195 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Montero-Escribano P, Matías-Guiu J, Gómez-Iglesias P, et al. Anti-CD20 and COVID-19 in multiple sclerosis and related disorders: a case series of 60 patients from Madrid, Spain. Mult Scler Relat Disord 2020;42:102185. 10.1016/j.msard.2020.102185 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Parrotta E, Kister I, Charvet L, et al. COVID-19 outcomes in MS: observational study of early experience from NYU multiple sclerosis comprehensive care center. Neurol Neuroimmunol Neuroinflamm 2020;7. 10.1212/NXI.0000000000000835. [Epub ahead of print: 09 Jul 2020]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Louapre C, Collongues N, Stankoff B, et al. Clinical characteristics and outcomes in patients with coronavirus disease 2019 and multiple sclerosis. JAMA Neurol 2020;77:1079–88. 10.1001/jamaneurol.2020.2581 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Tortorella C, Aiello A, Gasperini C, et al. Humoral- and T-cell-specific immune responses to SARS-CoV-2 mRNA vaccination in patients with MS using different disease-modifying therapies. Neurology 2022;98:e541–54. 10.1212/WNL.0000000000013108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Achiron A, Mandel M, Dreyer-Alster S, et al. Humoral immune response to COVID-19 mRNA vaccine in patients with multiple sclerosis treated with high-efficacy disease-modifying therapies. Ther Adv Neurol Disord 2021;14:17562864211012836. 10.1177/17562864211012835 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Sanz I, Wei C, Jenks SA, et al. Challenges and opportunities for consistent classification of human B cell and plasma cell populations. Front Immunol 2019;10:2458. 10.3389/fimmu.2019.02458 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Roll P, Palanichamy A, Kneitz C, et al. Regeneration of B cell subsets after transient B cell depletion using anti-CD20 antibodies in rheumatoid arthritis. Arthritis Rheum 2006;54:2377–86. 10.1002/art.22019 [DOI] [PubMed] [Google Scholar]
- 36. Bittner S, Ruck T, Wiendl H, et al. Targeting B cells in relapsing-remitting multiple sclerosis: from pathophysiology to optimal clinical management. Ther Adv Neurol Disord 2017;10:51–66. 10.1177/1756285616666741 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Le Bert N, Tan AT, Kunasegaran K, et al. SARS-CoV-2-specific T cell immunity in cases of COVID-19 and SARS, and uninfected controls. Nature 2020;584:457–62. 10.1038/s41586-020-2550-z [DOI] [PubMed] [Google Scholar]
- 38. Cao W-C, Liu W, Zhang P-H, et al. Disappearance of antibodies to SARS-associated coronavirus after recovery. N Engl J Med 2007;357:1162–3. 10.1056/NEJMc070348 [DOI] [PubMed] [Google Scholar]
- 39. Ng O-W, Chia A, Tan AT, et al. Memory T cell responses targeting the SARS coronavirus persist up to 11 years post-infection. Vaccine 2016;34:2008–14. 10.1016/j.vaccine.2016.02.063 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Wang F, Hou H, Luo Y, et al. The laboratory tests and host immunity of COVID-19 patients with different severity of illness. JCI Insight 2020;5. 10.1172/jci.insight.137799. [Epub ahead of print: 21 May 2020]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Heming M, Li X, Räuber S, et al. Neurological manifestations of COVID-19 feature T cell exhaustion and dedifferentiated monocytes in cerebrospinal fluid. Immunity 2021;54:164–75. 10.1016/j.immuni.2020.12.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Ngai P, McCormick S, Small C, et al. Gamma interferon responses of CD4 and CD8 T-cell subsets are quantitatively different and independent of each other during pulmonary Mycobacterium bovis BCG infection. Infect Immun 2007;75:2244–52. 10.1128/IAI.00024-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Gutierrez L, Beckford J, Alachkar H. Deciphering the TCR repertoire to solve the COVID-19 mystery. Trends Pharmacol Sci 2020;41:518–30. 10.1016/j.tips.2020.06.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Lotan I, Wilf-Yarkoni A, Friedman Y, et al. Safety of the BNT162b2 COVID-19 vaccine in multiple sclerosis (MS): early experience from a tertiary MS center in Israel. Eur J Neurol 2021;28:3742–8. 10.1111/ene.15028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Di Filippo M, Cordioli C, Malucchi S. mRNA COVID-19 vaccines do not increase the short-term risk of clinical relapses in multiple sclerosis. J Neurol Neurosurg Psychiatr 2021. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jnnp-2021-328197supp001.pdf (2.2MB, pdf)
jnnp-2021-328197supp002.pdf (103.1KB, pdf)
Data Availability Statement
Data are available upon reasonable request. Further information and requests for resources and anonymised clinical data should be directed to and will be fulfilled by David Kremer (DavidMaxJoe.Kremer@med.uni-duesseldorf.de)