Abstract
Symptoms following acute COVID-19 infection are common, but their relationship to initial COVID-19 severity is unclear. We hypothesize that residual symptoms are related to disease severity, and severe acute COVID-19 infection is more likely to cause residual pulmonary damage. This study aims to evaluate symptoms, lung function, and abnormal imaging within 3 months following COVID-19 infection, and to determine whether they are related to initial disease severity. A cross-sectional study was carried out at a designated post-COVID clinic in Hadassah Medical Center, Jerusalem, Israel. Patients with PCR-confirmed SARS-CoV-2 infection were evaluated within 12 weeks following infection and included both admitted and non-admitted subjects. All study participants underwent assessment of symptoms, quality of life (SGRQ), pulmonary function tests, and imaging. A total of 208 patients (age 49.3 ± 16 years) were included in the study. Initial disease severity was mild in 86, moderate in 49, and severe in 73 patients. At the time of follow-up, there were no differences in frequency of residual symptoms or in SGRQ score between groups. Patients with severe COVID-19 were more likely to have residual dyspnea (p = 0.04), lower oxygen saturation (p < 0.01), lower FVC and TLC (p < 0.001, p = 0.03 respectively), abnormal CXR (p < 0.01), and abnormal CT scan (p < 0.01) compared to other groups.Frequency of symptoms and impairment of quality of life at 12 week follow-up are common and are not related to severity of initial COVID-19 disease. In contrast, reduced lung function and abnormal pulmonary imaging are more common in patients with more severe acute COVID-19 infection.
Keywords: COVID-19, Post-COVID, Dyspnea, Pulmonary function, FVC
Background
The severe acute respiratory syndrome coronavirus 2 (SARS-Cov-2) outbreak began in China in December 2019 [1, 2]. Since then, the virus has caused a global pandemic with over a 150 million persons infected and over 3 million deaths. The clinical spectrum of acute coronavirus-induced disease (COVID-19) is wide, and ranges from asymptomatic infection, mild self-limiting disease, to acute life-threatening respiratory failure [2, 3]. Furthermore, the acute infection is not limited to the respiratory system, and may lead to multisystem involvement, including neurological, gastrointestinal, thromboembolic, and cardiovascular disease [4].
In addition to the acute illness, long-term effects of COVID-19 have also become apparent, and patients may suffer from persistent symptoms, impaired lung function, and both pulmonary and extra-pulmonary complications [5]. The increasing number of patients suffering from long-term effects has led to the implementation of terms such as “long-COVID” or “post-COVID” [5, 6], in which patients have persistent multisystem symptoms and complications. Currently, post-COVID is defined as the presence of symptoms extending beyond 3 weeks of acute COVID-19 initial presentation, and post-COVID may even extend beyond 12 weeks [6–8].
Previous reports have detailed residual symptoms following SARS-CoV-2 infection. Persistent symptoms include dyspnea, fatigue, chest pain, and cognitive dysfunction, and were seen in 87.4% of recovered patients [9]. Dyspnea is the most common persistent symptom, present in 42% to 66% of patients at 60–100 days of follow-up [5, 9, 10]. Additional studies have also shown persistent symptoms and functional impairment after COVID-19, particularly in hospitalized patients with severe disease, but also in those with mild disease [5, 11–15].
Chest imaging and lung function may also be impaired following SARS-CoV-2 infection. In 55 patients who recovered from COVID-19, abnormal chest computerized tomography (CT) scan was found in 39 of them, with impaired lung function in 14 patients [16]. Other studies evaluating radiological abnormalities showed that up to 56% of patients have persistent radiographic abnormalities after 12 weeks of follow-up [17]. In several cohorts that assessed lung function in patients who survived COVID-19 hospitalization, restrictive lung abnormality and diffusion limitation demonstrated in 22–38% and 24–71.7% [18, 19], respectively, especially in patients with severe COVID-19. In a large Chinese cohort that included 1733 patients, Huang et al. showed diffusion impairment at follow-up in 56% in severely ill patients, and abnormal CT pattern in 50% of 349 patients at 6 months [20].
There are little data analyzing the long-term residual effects in COVID-19 survivors in relation to initial disease severity. Most published studies in post-COVID-19 patients have focused on those with severe initial disease or hospitalized patients. A recent study of 63 COVID-19 survivors compared both hospitalized and non-hospitalized patients and found persistent dyspnea and fatigue in both groups. Hospitalized patients had lower total lung and diffusion capacity [21]. Another study that evaluated mainly outpatients showed that symptomatic patients have significantly reduced lung function, most notably impaired gas transfer [22].
We hypothesize that residual symptoms in COVID-19 survivors are related to disease severity, and patients with severe disease are more likely to develop residual symptoms, and pulmonary damage as assessed by impaired lung function or abnormal imaging. This study aims to evaluate residual symptoms, lung function, and imaging findings following SARS-CoV-2 infection.
Methods
Study design and participants
We conducted a cross-sectional cohort analysis which was carried out at the post-COVID clinic in Hadassah Medical Center, Jerusalem, Israel. The study protocol was approved by the ethics committee of Hadassah—Hebrew University Medical Center (number HMO-0294–20) and informed consent was obtained from all participants in the study. Post-COVID-19 patients were invited to the clinic by direct phone calls following hospital discharge from COVID wards, addition of written instructions in hospital discharge summaries as to post-COVID clinic details and advertisement in social media targeting outpatients or those who were admitted in other hospitals. After their presentation to the clinic, participants were asked to participate in the study by a research physician.
Between August 2020 and February 2021, we recruited participants who presented to the post-COVID outpatient clinic. Patients were > 18 years of age and diagnosed with SARS-CoV-2 infection by RT-PCR 12 weeks before enrollment. We included both patients that were initially admitted with acute COVID as well as outpatients initially managed at home. No data were collected on patients who did not attend the post-COVID clinic.
Clinical assessment
All data were obtained during the clinic visit. Routine demographic information was collected, as well as details of date of COVID-19 diagnosis, comorbidities, smoking history, symptoms, and clinical features of acute COVID-19. Inpatient data were extracted through review of patient’s electronic medical records. Data relating to acute COVID-19 included peak oxygen requirements, oxygen saturation at presentation (if available), chest X-ray findings, and treatment during admission. Imaging during admission was considered as normal or abnormal.
Participants were divided into three major groups of initial COVID-19 severity, according to the national health institute COVID-19 treatment guidelines [3]: mild, moderate, and severe. Patients with signs and symptoms of COVID-19, but without shortness of breath or abnormal chest imaging were considered “mild”. Moderate disease refers to individuals who show evidence of lower respiratory disease during clinical assessment or imaging and have oxygen saturation of 94% or more breathing room air at sea level. Patients with oxygen saturation less than 94% on room air, respiratory frequency > 30 breaths/min, or lung infiltrates > 50% were categorized as "severe" disease. In addition, patients were compared as inpatients and outpatients’ groups.
During post-COVID clinic assessment, all participants were asked to fill out Saint George respiratory questionnaire (SGRQ) for measurement of impact on overall health, daily life, and perceived well-being [23, 24].
All participants underwent physical examination, oxygen saturation both at rest and exercise, spirometry, and CXR during their visit. Further investigation was performed according to initial disease severity. No further investigation was performed in participants with mild disease. Chest CT, body plethysmography, and diffusing capacity of carbon monoxide (DLCO) were performed in those with severe disease or according to physician judgment.
Spirometry and pulmonary function tests were performed according to the American Thoracic Society/European Respiratory Society standards [25, 26]. Data included forced expiratory volume at 1 s (FEV1), forced vital capacity (FVC), the ratio between the two parameters: FEV1/FVC, static lung volumes, and DLCO using the single breath technique. All data presented as percentage of predicted normal values.
Data analysis
The data were analyzed using SPSS software version 25. Statistical significance was considered for p value lower than 0.05.
Descriptive statistics were performed using means, standard deviations and ranges for the continuous variables, and frequencies for the discrete variables. Differences between patient groups (according to initial COVID-19 severity) were assessed using Chi-square tests for the discrete variables, and Mann–Whitney and Kruskal–Wallis tests for the continuous variables. Post hoc analyses were conducted using Bonferroni corrections for multiple comparisons. To analyze the impact of clinical parameters on persistent symptoms and respiratory outcomes, we used a multivariate analysis which were conducted using Logistic regressions. Separate Logistic regressions were conducted for assessing the probability to have persistent symptoms, reduced FVC, abnormal CXR, and abnormal CT at the time of study participation.
Results
Baseline characteristics
A total of 208 patients (age 49.3 ± 16 years) were included with a mean follow-up of 80 ± 50 days after acute COVID-19 infection (Table 1). 56.3% of patients were male and 43.7% were female. Hypertension (n = 41, 19.7%) and hyperlipidemia (n = 40, 19.2%) were the most frequently reported comorbidities. 83.7% of the patients were non-smokers (n = 174), 7.2% were current smokers, and 9.1% were past smokers. Patients with severe COVID-19 were older and had more comorbidities compared to those with mild and moderate disease.
Table 1.
Baseline patients’ clinical characteristics by initial disease severity
| N (%) or Mean ± SD | p value | ||||
|---|---|---|---|---|---|
| Total (n = 208) |
Mild (n = 86) |
Moderate (n = 49) |
Severe (n = 73) |
||
| Age, years | 49.3 ± 16.1 | 42.3 ± 15.2 | 48.5 ± 15.9 | 58 ± 13.1 | < 0.001 |
| Male | 117 (56.3) | 38 (44.2) | 29 (59.2) | 50 (68.5) | 0.007 |
| Female | 91 (43.8) | 48 (55.8) | 20 (40.8) | 23 (31.5) | |
| Comorbidities | |||||
| Hypertension | 41 (19.7) | 10 (11.6) | 7 (14.3) | 24 (32.9) | < 0.001 |
| Diabetes mellitus | 29 (13.9) | 7 (8.1) | 6 (12.2) | 16 (21.9) | 0.04 |
| Hyperlipidemia | 40 (19.2) | 8 (9.3) | 6 (12.2) | 26 (35.6) | < 0.001 |
| IHD | 12 (5.8) | 3 (3.5) | 0 (0) | 9 (12.3) | – |
| Cancer | 16 (7.7) | 5 (5.8) | 4 (8.2) | 7 (9.6) | 0.67 |
| Asthma/COPD | 10 (4.8) | 4 (4.6) | 3 (6.1) | 3 (4.1) | 0.56 |
| AF | 3 (1.4) | 0 (0) | – | 3 (4.1) | – |
| Heart failure | 3 (1.4) | 1 (1.1) | – | 2 (2.7) | – |
| Cirrhosis | 1 (0.5) | – | – | 1 (1.4) | – |
| Other | 46 (22.1) | 15 (17.4) | 11 (12.8) | 20 (23.3) | 0.32 |
| Smoking | |||||
| Never smoker | 174 (83.7) | 69 (80.2) | 44 (89.8) | 61 (83.6) | 0.35 |
| Active smoker | 15 (7.2) | 9 (10.5) | 4 (8.1) | 2 (2.7) | |
| Past smoker | 19 (9.1) | 8 (9.3) | 1 (2.1) | 10 (13.7) | |
| COVID-19 illness | |||||
| Cough | 147 (70.7) | 48 (55.8) | 37 (75.5) | 62 (84.7) | < 0.001 |
| Dyspnea | 88 (42.3) | 7 (8.1) | 29 (59.2) | 52 (71.2) | < 0.001 |
| Sao2 | 92.2 ± 6.09 | 96.5 ± 1.7 | 95.2 ± 3 | 88 ± 6.1 | < 0.001 |
| Oxygen requirements | |||||
| No oxygen | 142 (68.3) | 86 (100) | 43 (87.7) | 13 (17.8) | < 0.001 |
| Supplemental oxygen | 43 (20.7) | – | 6 (12.3) | 37 (50.7) | < 0.001 |
| HFNC | 14 (6.7) | – | – | 14 (19.2) | < 0.001 |
| Mechanical ventilation | 9 (4.3) | – | – | 9 (12.3) | < 0.001 |
| Chest X-ray | |||||
| Not performed | 92 (44.2) | 66 (76.7) | 16 (32.7) | 10 (13.7) | < 0.001 |
| Normal | 38 (18.3) | 20 (23.3) | 12 (24.5) | 6 (8.2) | |
| Abnormal | 78 (37.5) | – | 21 (42.8) | 57 (78.1) | |
| Chest CT | |||||
| Not performed | 177 (85.1) | 86 | 44 (89.9) | 47 (64.4) | < 0.001 |
| Normal | – | – | – | – | |
| Abnormal | 31 (14.9) | – | 5 (10.1) | 26 (35.6) | |
| Therapy (at infection) | |||||
| Antibiotics | 27 (13) | 1 (1.2) | 3 (6.1) | 23 (31.5) | < 0.001 |
| Remdesivir | 41 (19.7) | – | – | 41 (56.2) | < 0.001 |
| Dexamethasone | 61 (29.3) | 1 (1.2) | 5 (10.2) | 55 (75.3) | < 0.001 |
| Anticoagulation | 67 (32.3) | 2 (2.3) | 9 (18.8) | 56 (76.6) | < 0.001 |
| Other | 22 (10.5) | – | 3 (6.1) | 10 (13.7) | < 0.001 |
| Length of stay, days | 8.92 ± 9.64 | 2.7 ± 2.8 | 3.9 ± 3.6 | 13.5 ± 10.8 | < 0.001 |
IHD Ischemic heart disease, COPD Chronic obstructive pulmonary disease, SaO2 Oxygen saturation at room air, HFNC High Flow Nasal Cannula, CT computerized tomography; other therapy includes convalescent plasma, anti-IL-6, lopinavir–ritonavir, and hydroxychloroquine.
*p < 0.05
During acute COVID-19 illness, 70.7% of patients had cough, 42.3% had dyspnea, and 89.4% had general weakness. Patients in the severe group had lower oxygen saturation on room air at presentation (Table 1). 68.3% of the patients did not need respiratory support, and only 4.3% were mechanically ventilated, all in the severe group. 20.7% were treated with supplemental oxygen and 6.7% required high flow nasal cannula.
116 (55.8%) patients underwent CXR during the acute disease phase and was abnormal in 67.2% (78/116). CXR was not performed in patients with mild disease, and most of those who underwent CXR were in the severe group. Similarly, chest CT was performed in 31 (14.9%) patients and was abnormal in all cases. Therapy during admission and length of stay is presented in Table 1.
Follow-up visit
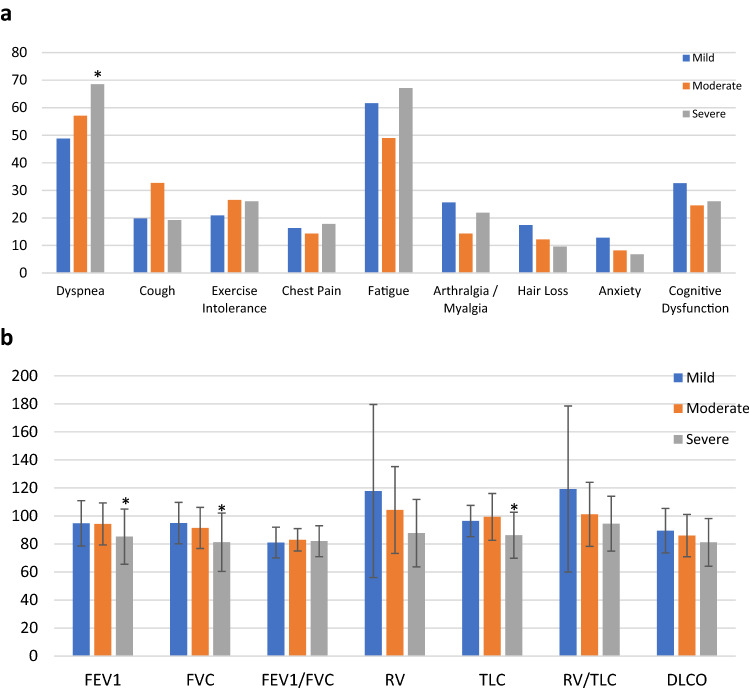
Mean follow-up period was 81 ± 50 days, and was similar in the three groups. 94.2% patients had at least one residual symptom, and only 5.8% (n = 12) were asymptomatic. There were no significant differences in the frequency of pulmonary or extra-pulmonary symptoms between patients with different initial disease severity (Table 2). The most common symptom at follow-up was fatigue [61.6%, 49%, 67.1% in mild, moderate, and severe COVID-19, respectively, (p = 0.13)] (Fig. 1a). Only dyspnea was more common in patients with severe vs. mild and moderate COVID-19 [68.5 vs 48.8% and 57.1%, (p = 0.04)]. Other residual symptoms and their frequencies are reported in Table 2 and Fig. 1. Quality of life as measured by SGRQ score was significantly reduced in all patients, without significant difference between mild, moderate, and severe COVID-19 survivors. Patients with severe disease have significantly lower oxygen saturation compared to patients with mild and moderate disease, both at rest (95.3 ± 2.5 vs 96.8 ± 1.5, and 96.4 ± 1.5, p < 0.001) and exercise (93.3 ± 4 vs. 96.2 ± 1.7 and 95.8 ± 1.7, p < 0.001).
Table 2.
Residual symptoms, SGRQ score, lung function, and imaging findings on follow-up, by initial disease severity
| N (%) or Mean ± SD | p Value | ||||
|---|---|---|---|---|---|
| Total (n = 208) |
Mild (n = 86) |
Moderate (n = 49) |
Severe (n = 73) |
||
| Follow-up, days | 81 ± 50 | 85 ± 55 | 80 ± 48 | 74 ± 45 | 0.48 |
| Residual symptoms | |||||
| Asymptomatic | 12 (5.8) | 3 (3) | 7 (14.2) | 2 (2) | 0.52 |
| Pulmonary | 40 (9.2) | 13 (15.7) | 9 (21.4) | 18 (25.4) | |
| Extrapulmonary | 116 (55.8) | 20 (24.1) | 9 (21.4) | 11 (15.5) | |
| Both | 120 (57.7) | 50 (60.2) | 24 (57.1) | 42 (59.2) | |
| Dyspnea | 120 (57.7) | 42 (48.8) | 28 (57.1) | 50 (68.5) | 0.04 |
| Cough | 47 (22.6) | 17 (19.8) | 16 (32.7) | 14 (19.2) | 0.16 |
| Exercise intolerance | 50 (24) | 18 (20.9) | 13 (26.5) | 19 (26.0) | 0.68 |
| Chest pain | 34 (16.3) | 14 (16.3) | 7 (14.3) | 13 (17.8) | 0.87 |
| Fatigue | 126 (60.6) | 53 (61.6) | 24 (49.0) | 49 (67.1) | 0.13 |
| Arthralgia/myalgia | 45 (21.6) | 22 (25.6) | 7 (14.3) | 16 (21.9) | 0.31 |
| Hair Loss | 28 (13.5) | 15 (17.4) | 6 (12.2) | 7 (9.6) | 0.34 |
| Anxiety | 20 (9.6) | 11 (12.8) | 4 (8.2) | 5 (6.8) | 0.41 |
| Cognitive dysfunction | 59 (28.4) | 28 (32.6) | 12 (24.5) | 19 (26.0) | 0.52 |
| Anosmia/ageusia | 16 (7.7) | 10 (11.6) | 3 (6) | 3 (4) | 0.18 |
| SGRQ score | 39.6 ± 25.6 | 37 ± 24.8 | 42.8 ± 25.6 | 40.9 ± 26.7 | 0.67 |
| SaO2 | |||||
| Rest | 96.2 ± 2.05 | 96.8 ± 1.5 | 96.4 ± 1.5 | 95.3 ± 2.5 | < 0.001 |
| Exercise | 95.0 ± 3.0 | 96.2 ± 1.7 | 95.8 ± 1.7 | 93.3 ± 4.0 | < 0.001 |
| Spirometry | |||||
| FEV1, % predicted | 91.1 ± 17.8 | 94.7 ± 16.2 | 94.3 ± 15 | 85.2 ± 19.7 | < 0.001 |
| FVC, % predicted | 89.0 ± 18.3 | 94.9 ± 14.8 | 91.44 ± 14.7 | 81.3 ± 20.8 | < 0.001 |
| FEV1/FVC, % predicted | 82 ± 18.3 | 81 ± 11 | 83 ± 8 | 82 ± 10 | 0.87 |
| Lung volumes | |||||
| RV, % predicted | 98.2 ± 38.6 | 117.8 ± 61.8 | 104.2 ± 31 | 87.7 ± 24 | 0.13 |
| TLC, % predicted | 91.5 ± 16.3 | 96.4 ± 11.1 | 99.4 ± 16.7 | 86.3 ± 16.4 | 0.03 |
| RV/TLC, % predicted | 101.6 ± 34.1 | 119.2 ± 59.2 | 101.1 ± 22.8 | 94.5 ± 19.6 | 0.60 |
| Diffusing capacity | |||||
| DLCO, % predicted | 84.2 ± 14.5 | 89.5 ± 15.9 | 86 ± 15.1 | 81.1 ± 17.0 | 0.28 |
| CXR | |||||
| Not performed | 19 (9.2) | 8 (9.3) | 9 (18.3) | 2 (2.7) | < 0.001 |
| Normal | 130 (62.8) | 68 (79) | 32 (65.3) | 30 (41.1) | |
| Abnormal | 58 (28) | 10 (11.7) | 8 (16.3) | 41 (56.2) | |
| CT scan | |||||
| Not performed | 141 (67.8) | 74 (86) | 35 (71.4) | 32 (43.8) | 0.26 |
| Normal | 28 (13.5) | 6 (7) | 8 (16.3) | 14 (19.2) | |
| Abnormal | 39 (18.8) | 6 (7) | 6 (12.2) | 27 (37) | |
| Abnormal CT findings | 39 (18.8) | 6 (7) | 6 (12.2) | 27 (37) | < 0.001 |
Pulmonary symptoms include dyspnea, cough, chest pain, and exercise intolerance; SGRQ Saint George Respiratory Questionnaire, SaO2 Oxygen saturation at room air, FEV1 Forced expiratory volume at 1 s, FVC Forced vital capacity, RV Residual volume, TLC Total lung capacity, DLCO diffusion capacity for carbon monoxide, CXR chest X-ray, CT computerized tomography
*p < 0.05
Fig. 1.
a: Residual symptoms frequency comparison between patients according to initial diseases severity: mild, moderate, and severe COVID-19. *p < 0.05; b: pulmonary function tests parameters on follow-up according to initial diseases severity: mild, moderate, and severe COVID-19
Lung function and imaging findings
At follow-up, spirometry was performed in total of 183 patients: 73, 41, and 68 patients in the mild, moderate, and severe group, respectively. Mean FVC (% predicted) at follow-up was 89 ± 18.3. Patients in the severe group has significantly lower FEV1 and FVC (Table 2, Fig. 1b). Absolute lung volumes and diffusion capacity were available in 62 patients: 14 in the mild, 14 in the moderate, and 33 in the severe group. TLC was significantly reduced in the severe group compared to mild and moderate group (86.3 ± 16.4 vs 96.2 ± 11.1 and 99.4 ± 16.7, p = 0.03). DLCO was reduced in the severe group compared to other groups, although the difference did not reach statistical significance.
Patients in the severe group were more likely to have abnormal CXR at follow-up compared to mild and moderate groups (56.2 vs. 11.7% and 16.3%, p < 0.001). When comparing abnormal CT findings, patients in the severe group had significantly higher abnormal CT imaging (Table 2). The pattern of injury seen on CT scan was residual ground glass opacities in 32 patients, and reticular and interstitial abnormalities in 3 patients. One patient had pulmonary embolism. The remaining 3 patients had either incidental emphysema or mosaic pattern of attenuation.
Patients requiring admission vs outpatients
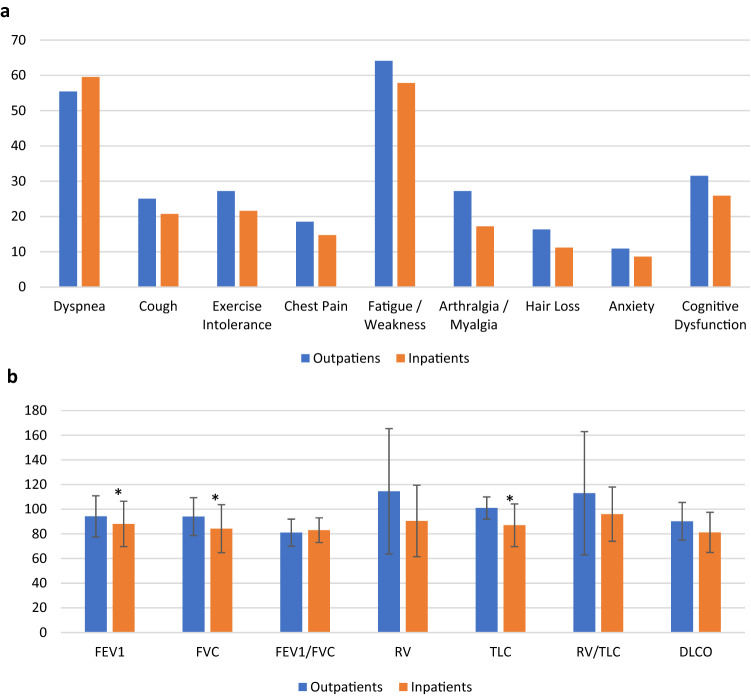
We performed a comparison between subjects requiring hospital admission vs those treated as outpatients for the acute COVID-19 illness (Table 3). 55.8% (116) of the patients were hospitalized for an average of 8.92 days (SD = 9.64). Inpatients were older, had more comorbidities, and were more symptomatic during acute COVID-19 illness. There was a significant difference in in oxygen requirement (51.8% of inpatients, 6.6% of outpatients, p < 0.001). In addition, inpatients were more likely to have abnormal CXR (65.5 vs 2.2%, p = 0.09), abnormal CT scan (26.7 vs 0, p < 0.001), and received therapy during their acute illness (Table 3). On follow-up visit, there was no significant difference in residual symptoms or SGRQ score between outpatients and inpatients. Inpatients had significantly decreased SaO2, both at rest (95.7 ± 2.2 vs 96.7 ± 1.6 in outpatients, p < 0.001) and exercise (94.3 ± 3.5 vs. 96 ± 2 in outpatients, p < 0.001). The following lung functions parameters were significantly reduced in inpatients vs outpatients: FEV1, FVC, and TLC (Table 3, Fig. 2b). DLCO was also lower in inpatients vs outpatients, but this did not reach statistical significance (81.2 ± 16.3 vs. 90.2 ± 15.3, p = 0.28). When compared to outpatient, the inpatient group was more likely to have abnormal CXR (37 vs 16.3%, p < 0.001), and abnormal CT scan (28.5 vs 6.5%, p < 0.01).
Table 3.
Comparison of outpatients Vs inpatients
| N (%) or Mean ± SD | p Value | ||
|---|---|---|---|
| Outpatients N = 92 |
Inpatients N = 116 |
||
| Age, years | 44.8 ± 115.8 | 52.8 ± 15.6 | < 0.001 |
| Male | 50 (54.3) | 67 (57.7) | 0.62 |
| Female | 42 (45.7) | 49 (42.3) | |
| Hypertension | 13 (14.1) | 28 (24.1) | 0.07 |
| Diabetes mellitus | 7 (7.6) | 22 (19) | 0.02 |
| Hyperlipidemia | 9 (9.8) | 31 (26.7) | < 0.001 |
| Cancer | 4 (4.3) | 12 (10.3) | 0.11 |
| Current/past smoking | 18 (19.6) | 16 (13.8) | 0.26 |
| COVID-19 illness | |||
| Cough | 55 (59.8) | 92 (79.3) | < 0.001 |
| Dyspnea | 26 (28.3) | 62 (53.4) | < 0.001 |
| Fatigue/Weakness | 83 (90.2) | 103 (88.8) | 0.74 |
| Sao2 | 91.7 ± 5.7 | 92.3 ± 6.2 | 0.31 |
| Oxygen requirements | |||
| No oxygen | 86 (93.4) | 56 (48.2) | < 0.001 |
| Supplemental oxygen | 6 (6.6) | 37 (31.8) | |
| HFNC | 0 (0) | 14 (12) | |
| Mechanical ventilation | 0 (0) | 9 (8) | |
| CXR | |||
| Not performed | 86 (93.5) | 6 (5.2) | 0.09 |
| Normal | 4 (4.3) | 34 (29.3) | |
| Abnormal | 2 (2.2) | 76 (65.5) | |
| CT | |||
| Not performed | 92 (100) | 85 (73.3) | < 0.001 |
| Normal | 0 | 0 (0) | |
| Abnormal | 0 | 31 (26.7) | |
| Therapy (at infection) | |||
| Antibiotics | 2 (2.2) | 25 (21.6) | < 0.001 |
| Remdesivir | 0 | 41 (35.3) | |
| Dexamethasone | 2 (2.2) | 59 (50.9) | |
| Anticoagulation | 1 (1.1) | 66 (57.4) | |
| Other | 1 (1.1) | 12 (10.3) | |
| Length of stay | 0 | 9 ± 9.6 | – |
| Follow-up visit | |||
| Follow-up, days | 77.6 ± 50 | 82.2 ± 50 | 0.48 |
| Residual symptoms | |||
| Asymptomatic | 1 (1) | 11 (9.5) | 0.8 |
| Pulmonary | 17 (18.4) | 23 (19.8) | |
| Extrapulmonary | 20 (21.7) | 20 (17.2) | |
| Both | 54 (58.6) | 62 (53.4) | |
| Dyspnea | 51 (55.4) | 69 (59.5) | 0.56 |
| Cough | 23 (25.0) | 24 (20.7) | 0.46 |
| Exercise intolerance | 25 (27.2) | 25 (21.6) | 0.35 |
| Chest pain | 17 (18.5) | 17 (14.7) | 0.46 |
| Fatigue/weakness | 59 (64.1) | 67 (57.8) | 0.35 |
| Arthralgia/myalgia | 25 (27.2) | 20 (17.2) | 0.08 |
| Hair Loss | 15 (16.3) | 13 (11.2) | 0.28 |
| Anxiety | 10 (10.9) | 10 (8.6) | 0.58 |
| Cognitive dysfunction | 29 (31.5) | 30 (25.9) | 0.37 |
| Anosmia/ageusia | 12 (13) | 4 (3.4) | 0.009 |
| SGRQ | 40 ± 25.7 | 39.2 ± 25.7 | 0.67 |
| SaO2 | |||
| Rest | 96.7 ± 1.6 | 95.7 ± 2.2 | < 0.001 |
| Exercise | 96 ± 2 | 94.3 ± 3.5 | < 0.001 |
| FEV1, % predicted | 94.2 ± 16.7 | 88.1 ± 18.4 | < 0.001 |
| FVC, % predicted | 94 ± 15.4 | 84.2 ± 19.5 | < 0.001 |
| FEV1/FVC, % predicted | 81 ± 11 | 83 ± 10 | 0.87 |
| RV, % predicted | 114.5 ± 50.9 | 90.5 ± 29 | 0.13 |
| TLC, % predicted | 101 ± 9 | 87 ± 17.3 | 0.03 |
| RV/TLC, % predicted | 113 ± 50 | 96 ± 22 | 0.60 |
| DLCO, % predicted | 90.2 ± 15.3 | 81.2 ± 16.3 | 0.28 |
| CXR | |||
| Not performed | 6 (6.5) | 14 (12) | < 0.001 |
| Normal | 71 (77.2) | 59 (51) | |
| Abnormal | 15 (16.3) | 43 (37) | |
| CT | |||
| Not performed | 78 (84.9) | 63 (54.3) | 0.19 |
| Normal | 8 (8.6) | 20 (17.2) | |
| Abnormal | 6 (6.5) | 33 (28.5) | |
| Abnormal CT findings | 6 (6.5) | 33 (28.5) | < 0.001 |
Major baseline clinical characteristics, residual symptoms, SGRQ score, lung function and imaging findings at follow-up
IHD Ischemic heart disease, SaO2 Oxygen saturation at room air, HFNC High Flow Nasal Cannula, CT computerized tomography, Other therapy include convalescent plasma, anti-IL-6, lopinavir–ritonavir, and hydroxychloroquine, Pulmonary symptoms include: dyspnea, cough, chest pain, and exercise intolerance, SGRQ Saint George Respiratory Questionnaire, SaO2 Oxygen saturation at room air, FEV1 Forced expiratory volume at 1 s, FVC Forced vital capacity, RV Residual volume, TLC Total lung capacity, DLCO diffusion capacity for carbon monoxide, CXR chest X-ray, CT computerized tomography
*p < 0.05
Fig. 2.
a: Residual symptoms frequency comparison between inpatients and outpatients. *p < 0.05; b: pulmonary function tests parameters on follow-up compared between inpatients and outpatients. *p < 0.05
Predictors of respiratory outcomes
The results are presented in Table 4. The regression model shows that age, SaO2 during acute COVID-19, smoking, and treatment with remdesivir and anticoagulation are associated with an increased probability of having persistent symptoms.
Table 4.
Odds ratio and confidence intervals for predictors of residual symptoms, reduced FVC, abnormal CXR, and CT findings
| Residual Symptoms | Reduced FVC | Abnormal CXR | Abnormal CT | |
|---|---|---|---|---|
| Age |
0.95** [0.91, 0.99] |
1.03 [0.99, 1.07] |
1.11** [1.05, 1.18] |
1.11** [1.04, 1.18] |
| Gender (male) |
1.62 [0.54. 4.84] |
1.08 [0.35, 3.31] |
6.12* [1.36, 27.57] |
2.52 [0.58, 10.92] |
| Any medical history |
3.08 [0.93, 10.20] |
1.24 [0.35, 4.33] |
0.63 [0.14, 2.72] |
0.53 [0.12, 2.35] |
| Cancer |
2.52 [0.40, 15.76] |
1.29 [0.22, 7.46] |
1.23 [0.14, 11.19] |
0.13 [0.01, 2.56] |
| Smoker/past smoker |
0.20* [0.04, 0.95] |
1.37 [0.35, 5.41] |
1.17 [0.15, 9.32] |
0.62 [0.10, 4.01] |
| Cough on presentation |
0.26 [0.06, 1.12] |
0.67 [0.18, 2.45] |
0.63 [0.10, 3.89] |
3.98 [0.62, 25.69] |
| Dyspnea on presentation |
0.60 [0.18, 2.05] |
2.74 [0.77, 9.67] |
8.93** [1.71, 46.58] |
3.80 [0.46, 31.49] |
| Fatigue on presentation |
0.74 [0.11, 5.09] |
1.29 [0.25, 6.70] |
1.89 [0.26, 13.72] |
3.40 [0.30, 38.40] |
| SaO2 on presentation |
1.16* [1.03, 1.31] |
0.91 [0.81, 1.03] |
0.93 [0.78, 1.11] |
0.93 [0.83, 1.05] |
| Respiratory support |
0.58 [0.08, 4.11] |
1.19 [0.12, 11.66] |
4.03 [0.28, 58.47] |
2.33 [0.11, 48.09] |
| Antibiotics treatment |
2.33 [0.61, 8.95] |
0.20 [0.03, 1.18] |
2.74 [0.59, 12.82] |
2.11 [0.50, 8.93] |
| Remdesivir treatment |
6.56* [1.21, 35.51] |
0.39 [0.06, 2.33] |
2.94 [0.39, 22.38] |
0.81 [0.11, 6.27] |
| Dexamethasone treatment |
0.45 [0.05, 4.40] |
3.37 [0.25, 53.77] |
0.18 [0.01, 5.39] |
0.04 [0.00, 1.12] |
| Anticoagulation treatment |
7.43* [1.01, 54.47] |
0.13 [0.01, 1.33] |
1.02 0.08, 13.33] |
10.02 [0.84, 119.12] |
SaO2 oxygen saturation on room air
*p < 0.05
Respiratory outcomes included reduced FVC, abnormal CXR, and abnormal CT findings at follow-up. Older age, male gender, and dyspnea during acute illness were associated with a higher likelihood of abnormal CXR at follow-up (OR = 1.11, 6.12, and 8.93, respectively). Older age was associated with a higher likelihood of abnormal CT findings at follow-up (OR = 1.11).
Discussion
In this study, we found that survivors of COVID-19 continue to suffer from reduced quality of life and residual symptoms, particularly shortness of breath and fatigue, 12 weeks after acute disease. Prolonged symptoms are present regardless of initial disease severity. Patients with severe initial COVID-19 have greater impairment of lung function and more frequent abnormal imaging findings at 3 months as compared to patients with mild or moderate diseases.
To our knowledge, only a few studies with a small number of subjects have compared the frequency of prolonged symptoms in COVID-19 survivors with different disease severity [14, 21]. Symptoms following acute COVID-19 are common and have been well documented. Our findings are consistent with previous studies that report that fatigue and shortness of breath are the most common residual symptoms in survivors of COVID-19 [11–15, 18–22]. Persistent symptoms are extremely common, even in patients with mild acute disease. This group accounts for the large majority of COVID-19-infected patients, most of whom are outpatients. Abdallah et al. showed persistent fatigue and exertional breathlessness in 81.6% of non-hospitalized COVID-19 survivors, but without impairment of lung function or cardiopulmonary exercise test [21]. Logue et al. reported persistent symptoms in 65.3% of 150 outpatients with mild disease [11]. Other studies show similar findings [12, 14, 15, 22]. Our study and others clearly demonstrate that even patients who suffered from mild acute COVID-19 disease frequently develop a wide range of residual symptoms with ongoing disability and impaired quality of life, but without abnormalities in pulmonary function tests or imaging studies.
In contrast to mild disease, survivors of severe COVID-19 had significant pulmonary sequelae, with evidence of reduced FEV1, FVC, and TLC, and abnormal chest imaging (CXR or CT) at 3 months of follow-up. Our findings are consistent with previous reports that focused on the evaluation of severe COVID-19 survivors [17–19, 28]. A reduction in diffusion capacity is the most commonly reported physiologic impairment in these patients [5], although in our study, the difference in DLCO did not reach statistical significance.
Persistent morbidity/disability occurring during follow-up in patients with severe COVID-19 is usually relatively easy to identify on pulmonary function tests and/or imaging. Conversely, in mild cases, symptoms at follow-up seem to occur independent of pulmonary function or imaging abnormalities. Our findings are consistent with previous studies which show that persistent symptoms and poor health following COVID-19 infection are not related to initial disease severity [12, 14]. Although the exact mechanism of residual symptoms is not fully clear, prolonged inflammation, immune-mediated vascular dysfunction, and thromboembolism are among the major explanations [29]. Havervall et al. [12] reported the presence of moderate-to-severe symptom lasting at least 2 months in 26% of participants who were seropositive to anti-spike IgG vs. 9% in seronegative participants. These findings might suggest that positive anti-spike IgG may play a role in the occurrence of residual symptoms.
Our findings highlight the fact that inpatients and survivors of severe COVID-19 should be the main focus of follow-up and may benefit from a systematic approach to evaluate for the presence of residual pulmonary injury. Given the significant morbidity that SARS-CoV-2 continue to cause, more survivors of severe COVID-19 are expected to present to post-COVID outpatient clinics or continue to be managed into medical departments. Thus, there is a need for early identification of those patients, rigorous follow-up protocol, and early treatment of their pulmonary sequelae. More accurate predictors for early identifications such as molecular biomarkers may help in identifications of these patients and preventing persistent pulmonary damage.
The management of post-COVID is still an area of active research, and treatment of a subset of pulmonary residual damage with corticosteroids may be beneficial. Our study was not designed to address this question, but findings show that majority of the patients with post-COVID residual pulmonary damage had inflammatory lung disease consistent with organizing pneumonia or ground glass opacities on CT scan. A majority of our severe patient group were treated with corticosteroids. One small UK cohort demonstrated significant symptomatic and radiological improvement of similar patients with corticosteroids [29]. Thus, corticosteroids should be considered as a treatment option for a subset of post-COVID patients with inflammatory pulmonary complications.
Our study has several limitations. This is a single center cohort analysis, follow-up was evaluated within 12 weeks from initial disease, and longer term data are not yet available. Patient selection for our cohort is subject to bias as we included “all comers” to the hospital post-COVID clinic. There are many factors which may have influenced which patients arrived for follow-up, with symptomatic patients more likely to come for follow-up visits than those who have no residual manifestations. We are thus likely to have overestimated poor health and residual symptoms in our cohort, particularly in the mild patient group. Not all patients agreed to perform all follow-up tests, such as X-ray and pulmonary function tests, but this is unlikely to have significantly influenced overall findings. Finally, we did not assess anxiety and depression using a valid tool, and relied on patient reporting only.
Conclusion
We interpret our study findings in the following way: post-COVID-19 symptoms should be considered as following two different trajectories, which certainly overlap and often coexist, but require different approaches for treatment and follow-up. First, non-specific ill-defined symptoms are extremely common and seem to be as frequent following mild as following more severe acute COVID disease. This group of patients is no-doubt very diverse and is likely to include both inpatients and outpatients. The pathophysiological basis for these symptoms is unclear, as is treatment. For many of these patients, health advice and reassurance may suffice. The second trajectory includes patients with evidence of end-organ injury, particularly respiratory impairment, but also cardiac, neurological, and other pathologies. Extensive laboratory evaluation such as full pulmonary function tests, imaging, and echocardiography, etc. should be limited to the latter group of patients.
Acknowledgements
Not applicable.
Abbreviations
- COVID
Coronavirus induces disease
- SARS-Cov-2
Severe acute respiratory syndrome coronavirus 2
- FEV1
Forced expiratory volume in second 1
- FVC
Forced vital capacity
- TLC
Total lung capacity
- RV
Residual volume
- Dlco
Diffusion capacity of carbon monoxide
- CT
Computerized tomography
- PFT
Pulmonary function tests
- SaO2
Oxygen saturation
- SGRQ
Saint George respiratory questionnaire
- CXR
Chest X-ray
- HFNC
High flow nasal cannula
Author contributions
FD: is the guarantor of the article and takes responsibility for the integrity of the work, including data and analysis of the article. FD and NB: conceptualized the research aims and planned the analyses. AAR, KK, DP, UL, ZF, RK, and PGC: collected the clinical data. FD: participated in processing data and performing the statistical analysis. RK and NB: wrote part of the manuscript. FD: wrote the manuscript. All authors contributed to the acquisition, analysis, or interpretation of data. All authors revised the report and approved the final version before submission.
Funding
No financial grants or funding sources.
Data availability
The data will not be shared to reserve patient privacy and not signing them on this issue as a part of the informed consent.
Declarations
Conflict of interests
The authors declare that they have no competing interests.
Ethical approval
The study protocol was approved by the ethics committee of Hadassah—Hebrew University Medical Center (number HMO-0294–20) and informed consent was obtained from all participants in the study.
Consent for publication
Informed consent was obtained from all participants in the study.
Human and animal rights statement
As mentioned in methods and declarations, all patients signed on informed consent before entering the study. No animals included in the study.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020 doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Guan W-J, Yi NZ, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020 doi: 10.1056/nejmoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.NIH (2020) Coronavirus disease 2019 (COVID-19) treatment guidelines. Disponible en: https://www.covid19treatmentguidelines.nih.gov/overview/. Nih. 2019:130. Accessed 19 Oct 2021
- 4.Gupta A, Madhavan MV, Sehgal K, et al. Extrapulmonary manifestations of COVID-19. Nat Med. 2020 doi: 10.1038/s41591-020-0968-3. [DOI] [PubMed] [Google Scholar]
- 5.Nalbandian A, Sehgal K, Gupta A, et al. Post-acute COVID-19 syndrome. Nat Med. 2021 doi: 10.1038/s41591-021-01283-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Greenhalgh T, Knight M, A’Court C, Buxton M, Husain L. Management of post-acute covid-19 in primary care. BMJ. 2020 doi: 10.1136/bmj.m3026. [DOI] [PubMed] [Google Scholar]
- 7.British Thoracic Society (2021). British thoracic society guidance on respiratory follow up of patients with a clinico-radiological diagnosis of COVID-19 pneumonia. https://www.brit-thoracic.org.uk/document-library/quality-improvement/covid-19/resp-follow-up-guidance-post-covid-pneumonia/. Accessed 10 Oct 2021
- 8.NICE (2020). COVID-19 rapid guideline: managing the long-term effects of COVID-19. NICE guidelines. https://www.nice.org.uk/guidance/ng188. Accessed 15 Dec 2021 [PubMed]
- 9.Carfì A, Bernabei R, Landi F. Persistent symptoms in patients after acute COVID-19. JAMA. 2020 doi: 10.1001/jama.2020.12603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mandal S, Barnett J, Brill SE, et al. “Long-COVID”: a cross-sectional study of persisting symptoms, biomarker and imaging abnormalities following hospitalisation for COVID-19. Thorax. 2020 doi: 10.1136/thoraxjnl-2020-215818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Logue JK, Franko NM, McCulloch DJ, et al. Sequelae in adults at 6 months after COVID-19 infection. JAMA Netw Open. 2021 doi: 10.1001/jamanetworkopen.2021.0830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Havervall S, Rosell A, Phillipson M, et al. Symptoms and functional impairment assessed 8 months after mild COVID-19 among health care workers. JAMA. 2021 doi: 10.1001/jama.2021.5612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Morin L, Savale L, Pham T, et al. Four-Month clinical status of a cohort of patients after hospitalization for COVID-19. JAMA. 2021 doi: 10.1001/jama.2021.3331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Townsend L, Dowds J, O’Brien K, et al. Persistent poor health post-COVID-19 is not associated with respiratory complications or initial disease severity. Ann Am Thorac Soc. 2021 doi: 10.1513/annalsats.202009-1175oc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Davis HE, Assaf GS, McCorkell L, et al. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. medRxiv. 2020 doi: 10.1101/2020.12.24.20248802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhao Y-M, Shang Y-M, Song W-B, et al. Follow-up study of the pulmonary function and related physiological characteristics of COVID-19 survivors three months after recovery. EClinicalMedicine. 2020 doi: 10.1016/j.eclinm.2020.100463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.van der Brugge S, Talman S, Boonman-de Winter LJM, et al. Pulmonary function and health-related quality of life after COVID-19 pneumonia. Respir Med. 2021 doi: 10.1016/j.rmed.2020.106272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Smet J, Stylemans D, Hanon S, Verbanck S, Vanderhelst E, Ilsen B. Clinical status and lung function 10 weeks after severe SARS-CoV-2 infection. Respir Med. 2021 doi: 10.1016/j.rmed.2020.106276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Daher A, Balfanz P, Cornelissen C, et al. Follow up of patients with severe coronavirus disease 2019 (COVID-19): pulmonary and extrapulmonary disease sequelae. Respir Med. 2020 doi: 10.1016/j.rmed.2020.106197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Huang C, Huang L, Wang Y, et al. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet. 2021 doi: 10.1016/S0140-6736(20)32656-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Abdallah SJ, Voduc N, Corrales-Medina VF, et al. Symptoms, pulmonary function and functional capacity four months after COVID-19. Ann Am Thorac Soc. 2021 doi: 10.1513/AnnalsATS.202012-1489RL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Trinkmann F, Müller M, Reif A, et al. Residual symptoms and lower lung function in patients recovering from SARS-CoV-2 infection. Eur Respir J. 2021 doi: 10.1183/13993003.03002-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.St. George’s Respiratory Questionnaire (SGRQ). Accessed 29 April 2021. https://www.thoracic.org/members/assemblies/assemblies/srn/questionaires/sgrq.php
- 24.Santus P, Tursi F, Croce G, et al. Changes in quality of life and dyspnoea after hospitalization in COVID-19 patients discharged at home. Multidiscip Respir Med. 2020 doi: 10.4081/mrm.2020.713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Graham BL, Steenbruggen I, Barjaktarevic IZ, et al. Standardization of spirometry 2019 update an official American Thoracic Society and European Respiratory Society technical statement. Am J Respir Crit Care Med. 2019 doi: 10.1164/rccm.201908-1590ST. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pellegrino R, Viegi G, Brusasco V, et al. “ATS/ERS task force: standardisation of lung function testing.” Interpretative strategies for lung function tests. Eur Respir J. 2005 doi: 10.3174/ajnr.A1614. [DOI] [PubMed] [Google Scholar]
- 27.van Gassel RJJ, Bels JLM, Raafs A, et al. High prevalence of pulmonary sequelae at 3 months after hospital discharge in mechanically ventilated survivors of COVID-19. Am J Respir Crit Care Med. 2021;203(3):371–374. doi: 10.1164/rccm.202010-3823LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Maltezou HC, Pavli A, Tsakris A. Post-COVID syndrome: An insight on its pathogenesis. Vaccines. 2021 doi: 10.3390/vaccines9050497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Myall KJ, Mukherjee B, Castanheira AM, et al. Persistent post-COVID-19 inflammatory interstitial lung disease: an observational study of corticosteroid treatment. Ann Am Thorac Soc. 2021 doi: 10.1513/annalsats.202008-1002oc. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data will not be shared to reserve patient privacy and not signing them on this issue as a part of the informed consent.