Abstract
Prevention of genital herpes simplex virus (HSV) infections is desirable from both a public health standpoint and the patient’s perspective. A key factor in the spread of genital herpes infections is the high proportion of undiagnosed infections. Persons with subclinical or unrecognized infections are best diagnosed by accurate, type-specific antibody tests. Unfortunately, these tests are only now becoming widely available. The use of current, conventional (non-type-specific) serologic tests for diagnosis of herpes infections has resulted in confusion and misdiagnosis of patients. This review provides recent information on the epidemiology of genital herpes infections, describes the importance of subclinical herpes infection and shedding, summarizes the status of HSV type-specific serologic assays being developed, and provides indications for using such assays.
EPIDEMIOLOGY OF GENITAL HERPES
Seroprevalence of HSV-2
Herpes simplex virus type 2 (HSV-2) is the most common cause of genital ulcers in industrialized countries (22). Serosurveys for type-specific antibodies have shown an increase of over 30% in the prevalence of HSV-2 infections over the past two decades (11, 30, 31). Currently, the prevalence of HSV-2 is greater than 20% among adults in the United States (30). Among white persons in the recent National Health and Nutrition Study (NHANES III), 15% of men and 20% of women were HSV-2 seropositive. Among black persons tested in this study, 35% of men and 55% of women were HSV-2 seropositive. HSV-2 seroprevalence can be as high as 50% among women attending sexually transmitted disease (STD) clinics in the United States, the United Kingdom, or Australia (26, 27, 43). Between 60 and 90% of female commercial sex workers worldwide have antibodies to HSV-2 (25).
Recent Changes in HSV-2 Incidence
While studies of HSV-2 incidence suggest that most infections are acquired in the third decade of life (21), the recent seroprevalence studies point to a disturbing shift toward earlier acquisition of HSV-2. For example among teenagers, HSV-2 prevalence is over 5% (4.5% among white persons and 9% among black persons) (30). Serosurveys of a subset of young people, i.e., college students, reveal a similar HSV-2 seroprevalence (1 to 9%) with high yearly seroconversion rates (15, 33). Most troubling, HSV-2 seroprevalence has quintupled in white teenagers and has doubled among young adults in their 20s over the past two decades (30).
Genital HSV-1 Infections
Most recurrent genital herpes outbreaks are caused by HSV-2. However, it is important to note that an increasing proportion of first episodes are caused by HSV-1. In Seattle and areas of the United Kingdom, this proportion is 30% or more (65, 70). While primary HSV-1 genital infections are clinically indistinguishable from primary episodes caused by HSV-2, the recurrence rate of HSV-1 infection appears to be lower (24). Since type-specific serologic assays for HSV do not distinguish between oral and genital infections, HSV-2 seroprevalence data underestimate the total disease burden of genital herpes infections.
CLINICAL IMPACT OF GENITAL HERPES
Immunocompetent Adults
Genital herpes infections can be associated with serious morbidity. For some adults, particularly those without prior antibodies to either HSV-1 or HSV-2, acquisition of primary genital herpes can cause painful ulcerative lesions and systemic manifestations, including headache, malaise, and fever lasting up to 3 weeks. Meningitis accompanies these symptoms in 10% of men and up to 30% of women with primary infections (24). “Nonprimary first episodes” of HSV-2 genital herpes occur in persons with antibodies to HSV-1. This partial immunity results in milder episodes with lower frequency of constitutional signs and symptoms and shorter duration of lesions. Following the initial genital infection, HSV becomes latent in the sacral nerve ganglia and can reactivate to cause recurrent genital lesions. Approximately 85% of women and nearly all men with symptomatic acquisition of genital HSV-2 infection will have a recurrence within the first year at an average rate of 4 to 5 episodes per year (12). While recurrent episodes are shorter and more localized than first episodes, the chronic nature of this disease and the unpredictable occurrence of recurrences result in persistent psychosocial or psychosexual distress for many patients (20, 34).
Immunosuppressed Patients
Patients undergoing chemotherapy, organ or bone marrow transplant recipients, and patients with human immunodeficiency virus (HIV) infection who experience first or recurrent HSV-1 or HSV-2 episodes can develop severe and extensive lesions, which may become difficult to control by standard antiviral therapy (40, 66, 73). In some cases, visceral spread occurs (41, 45, 73).
Neonates
The most severely affected population is neonates, who acquire HSV infection after exposure to the virus during birth (84). This is a relatively rare infection, occurring in about 1 in 3,000 births in the northwestern United States and with a somewhat lower frequency in other areas. However, these infections result in permanent neurological damage for many infants despite appropriate antiviral therapy. The increasing acquisition rate of genital herpes among women of childbearing age (30) suggests that more neonates may be exposed to the virus than in past decades.
HSV and HIV-1 Infection
Genital herpes increases the risk of acquisition of HIV-1 as a result of breaks in the genital mucosal barrier and the recruitment of CD4+ lymphocytes into areas of HSV replication (38, 39, 72). Transmission of HIV-1 to sexual partners may also be aided by the presence of genital ulcers. High levels of HIV-1 have been documented in HSV lesions (68). Furthermore HSV reactivates more frequently in persons with HIV-1 infection (69). These findings indicate a higher risk of HIV-1 exposure for individuals whose sexual partners have both HIV-1 and genital herpes infections.
CORRELATES OF GENITAL HERPES INFECTIONS
Demographic and behavioral factors associated with a higher risk of genital herpes infection may be important in selecting candidates for antibody screening tests as they become available. In general, the risk of HSV-2 infection is correlated with markers of increased sexual exposure: early age of first intercourse, larger numbers of lifetime sexual partners, history of other sexually transmitted infections, and increasing age (27, 57, 82). The age-adjusted risk is higher for persons belonging to high-prevalence demographic groups than for middle-class white persons with equal numbers of partners (30, 82). Black persons, individuals from lower socioeconomic groups, and those with poor educational backgrounds have higher rates of genital herpes infection. Those with prior antibodies to HSV-1 are at lower risk than HSV-seronegative individuals of acquiring HSV-2 genital infections; however, this protection is only partial (53).
ROLE OF UNDIAGNOSED INFECTION AND SUBCLINICAL HSV SHEDDING IN THE HERPES EPIDEMIC
Discordance of History and HSV-2-Seropositive Status
Numerous studies have revealed that the majority of HSV-2 infections are undiagnosed. In the large NHANES survey, only 9% of persons with HSV-2 antibodies had knowledge of their disease (30). Conversely, 22% of those denying a history of genital herpes were found to be HSV-2 seropositive. Our studies have found that only 34% of women with HSV-2 antibodies who presented to the Seattle-King County STD Clinic were aware of having genital herpes (43). Among HSV-2-seropositive men and women in a family practice at the University of Washington, only 26% gave a history of genital herpes (58, 82). In the United Kingdom, only 32% of HSV-2-seropositive STD clinic attenders and 17% of HSV-2-seropositive blood donors reported having genital herpes (26).
Asymptomatic and Unrecognized Genital Herpes
It is estimated that about 20% of patients with HSV-2 antibodies are truly asymptomatic or have lesions only in locations, such as the cervix, that are impossible to observe (23). The remaining 60% of undiagnosed persons with genital HSV-2 infection have symptoms that are not recognized by either physicians or patients as being caused by herpes. Atypical manifestations include vulvar, penile, or perianal fissures, localized erythema, and back pain without genital lesions. Both of these groups are at risk of transmitting the virus. However, the majority of patients with atypical, unrecognized symptoms can learn to recognize their own unique manifestations of recurrent episodes (9, 32, 47).
Subclinical Shedding of HSV
HSV-2 can reactivate from its latent state in sensory ganglia and cause symptomatic episodes of genital lesions. Nearly all patients with clinically recognized first episodes of HSV-2 have a recurrent episode within a year of acquiring the infection (12). HSV-1 also reactivates in the first year but in only about 55% of patients. Reactivation can also result in subclinical excretion or “shedding” of infectious virus from anogenital sites. Recent studies have shown that recurrent episodes as well as subclinical virus shedding vary in frequency among individuals and, over time, within an individual. Subclinical virus shedding is up to three times more frequent in the first 3 months after acquisition of genital herpes than in later months (42). Subclinical HSV-1 shedding occurs with less than one-third the frequency of HSV-2 shedding. Furthermore, women with frequent clinical recurrences have higher rates of subclinical HSV-2 shedding than do those with infrequent symptomatic recurrences (79).
In established infections, subclinical HSV-2 shedding from the genital tract has been estimated to occur on 1 to 6% of days (10, 16, 42, 64, 79). More intensive studies in which women with recently diagnosed genital HSV-2 infections self-sampled cervical, vulvar, and perianal sites daily for a median of 105 days revealed infectious virus in 75% of the women. The virus was isolated in cell culture of samples obtained on 8% of the days covered by the study (83). In this study, amplification and detection of viral DNA by PCR was shown to be 3.5 times more sensitive than viral isolation in demonstrating HSV in the genital tract. By this technique, HSV was demonstrated in genital secretions in 95% of the women. Overall, women were shedding HSV DNA on 28% of the days sampled (83). Lesions consistent with genital herpes were noted on only 40% of days on which HSV DNA was detected; therefore, most of the viral shedding detected in this study was subclinical.
Data from daily sampling studies suggest that one-half to two-thirds of subclinical HSV shedding episodes occur within a week before or after an episode of clinically recognized, symptomatic HSV shedding (79, 80, 82). Therefore, patients who can be taught to recognize subtle symptoms accompanying HSV reactivation can also recognize at least some of the periods during which they are at higher risk of shedding virus without experiencing symptoms.
While most of the subclinical sampling data have been generated from studies of women, men have been shown to shed HSV asymptomatically from genital sites including intact penile skin (74, 76). Semen can also contain infectious virus, especially during primary HSV infection (56). Infectious virus can be isolated from men on 2.2% of the days sampled, a somewhat lower rate than for women (80).
Transmission of Genital Herpes
Unrecognized infections and subclinical viral shedding appear to be major factors in transmission. In a study designed to identify and interview partners of patients with newly diagnosed genital herpes, only 26% of the transmitting partners knew that they were infected (52). The remaining individuals were diagnosed only after they had transmitted herpes to their sexual partner. In a subsequent prospective study of couples where one had genital HSV-2 and the other did not, transmission occurred at an annualized rate of 10%; 70% of the transmission events were during times when the source partner was asymptomatic (53).
Suppressive Antiviral Therapy
Daily therapy with oral acyclovir, famciclovir, or valaciclovir reduces the symptomatic recurrence rate (28, 54, 55, 61, 75). Recent studies also suggest that suppressive acyclovir can reduce the frequency of subclinical HSV shedding from the genital tract by as much as 94%. In a blinded crossover study, virus was isolated on 6% of days when women were taking placebo but on fewer than 0.4% of days when they were taking acyclovir (81). HSV-PCR shows that the frequency of genital tract shedding of HSV is reduced by a median 80% (range, 24 to 91%) during acyclovir treatment compared to periods when the subjects were taking a placebo. Viral shedding resumed within 5 days after acyclovir treatment was discontinued (83).
Suppression of clinical and subclinical viral shedding by antiviral therapy may reduce HSV transmission. However, since viral excretion may still occur at low frequency and titer during antiviral therapy, the extent to which antiviral treatment will interrupt transmission remains to be determined.
DIAGNOSIS OF SYMPTOMATIC GENITAL HERPES
Clinical Diagnosis of Genital Lesions
Clinical presentation, alone, is seldom sufficient to diagnose genital herpes. While some patients present with the classic constellation of bilateral ulcerative lesions, tender inguinal lymphadenopathy, constitutional signs such as fever, and complaints of headache, malaise, and dysuria (24), most do not. Furthermore, genital ulcers or lesions assumed to be caused by herpes may in fact be caused by a number of other infections (syphilis, chancroid, lymphogranuloma venereum, donovanosis, scabies, and candidiasis) or noninfectious conditions (Crohn’s disease, Behçet’s syndrome, trauma, contact dermatitis, erythema multiforme, Reiter’s syndrome, psoriasis, and lichen planus). Microbiologic testing for infectious agents can help ensure that treatable infectious agents are accurately diagnosed.
Equally important, the presenting symptoms of genital herpes are frequently misinterpreted by the patient and by physicians. Skin splits, fissures, minor abrasions, furuncles, erythema, and pain are common manifestations of genital herpes but are often attributed to injury, allergies, insect bites, or other infectious agents (Table 1). Clinicians need to have a high index of suspicion for these lesions (44).
TABLE 1.
Patient-reported etiology of genital lesions from which HSV was isolated
| Women | Men |
|---|---|
| Yeast infection | Folliculitis |
| Vaginitis | Jock itch |
| Urinary tract infection | “Normal” itch |
| Urethral syndrome | Zipper burns |
| Menstrual complaint | Hemorrhoids |
| Hemorrhoids | Allergy to condoms |
| Allergy to: | Irritation from: |
| Condoms | Tight jeans |
| Sperm | Sexual intercourse |
| Spermicide | Bike seat |
| Elastic/pantyhose | Insect or spider bites |
| Irritation or rash from: | |
| Sexual intercourse | |
| Bike seat | |
| Shaving |
However, even the most careful history taken by a practitioner specializing in the care of patients with genital herpes is inadequate to diagnose this disease. In a recent study by Brown et al., a highly experienced clinician conducted a directed interview and then scored the patient as having symptoms “not at all suggestive,” “somewhat suggestive,” or “definitely indicative” of genital herpes (18). Type-specific serology results were then matched with the clinical impression. Consistent with other studies reporting the low sensitivity of history for identifying genital herpes, 37% of those patients with symptoms “not at all suggestive” of herpes were HSV-2 seropositive. Thus, even a highly skilled interview could not elicit a history in a high proportion of infected patients. Moreover, 50% of patients considered to be “highly likely” to be infected were actually HSV-2 seronegative. While a portion of these 50% may have had genital HSV-1 infections, the poor positive predictive value of history for identifying genital herpes confirms the critical role of laboratory methods for diagnosing this disease.
Laboratory Methods for Diagnosis
Virus detection methods should be used for patients presenting with lesions. Antigen detection can be nearly as sensitive as culture methods (7), and the most sensitive strategy is to perform both tests. However, the sensitivity of such tests declines as lesions heal and is lower in subjects with recurrent lesions than in those with first episodes (46). Antibody tests are useful as backup tools, particularly when evidence of seroconversion is being sought by type-specific tests.
Role of Serologic Testing
Definitive diagnosis of genital herpes infections is fundamental to the management of patients and the development of strategies to prevent transmission to partners and neonates. Detection of antibodies allows diagnosis of an infection when virological methods such as culture, antigen detection, or PCR are impractical or yield negative results (44). Because HSV-1 and HSV-2 are closely related antigenically and because both viruses cause lifelong infections with intermittent reactivation, the application of serologic testing is more complicated than for many other human viruses. Tests which can identify HSV antibodies but cannot differentiate between HSV-1 and HSV-2 are widely available. As diagnostic tools, these tests are not helpful. For example, if a serologic test is performed to rule out genital herpes in a patient with mild genital symptoms, a positive test could be due to past HSV-1 oral infection. Therefore, definitive tests for HSV-2 antibodies must be based on HSV-2 type-specific antigens.
Inaccuracy of Commercial Tests Based on Whole-Antigen Preparations
It is important to note that manufacturers of nontyping tests have offered mathematical algorithms to infer the presence of HSV-1 or HSV-2 antibodies on the basis of the relative strength of reactivity against crude antigen preparations from HSV-1 versus HSV-2. This approach results in highly inaccurate inferences of HSV antibody type (Table 2). When we tested three commercial enzyme immunoassay kits from Sigma Diagnostics (St. Louis, Mo.), Whittaker Bioproducts (Walkersville, Md.), and Incstar Corp. (Stillwater, Minn.) on sera from patients with culture-documented first episode infections of 3 to 8 weeks duration, only 33 to 55% of subjects were correctly identified (2). Between 38 and 48% of these patients would have been given a diagnosis of the wrong virus type. The remaining inaccurate results were due to lack of sensitivity of the tests.
TABLE 2.
Performance of a commercial EIA for symptomatic genital herpesa
| Clinical category (no. of patients) | No. (%) of patients with test result for kit:
|
||
|---|---|---|---|
| A | B | C | |
| Primary episode (28) | |||
| Correct diagnosis | 13 (46) | 18 (64) | 21 (75) |
| False negative | 12 (43) | 3 (11) | 1 (4) |
| Positive, wrong type | 3 (11) | 7 (25) | 6 (21) |
| Nonprimary HSV-2 (12) | |||
| Correct diagnosis | 0 (0) | 0 (0) | 1 (8) |
| Incorrect diagnosis | 12 (100) | 12 (100) | 11 (92) |
| Recurrent HSV-2 (45) | |||
| Correct diagnosis | 28 (62) | 44 (98) | 33 (73) |
| False negative | 1 (2) | 0 (0) | 0 (0) |
| Positive, wrong type | 16 (36) | 1 (2) | 12 (27) |
In a study conducted in 1991 (2), sera from patients with culture-documented primary episodes of genital HSV-1 or HSV-2, with nonprimary HSV-2 episodes, or with recurrent HSV-2 episodes were tested with kits from Sigma Diagnostics (A), Whittaker Bioproducts (B), or Incstar Corp. (C). All these kits are still on the market. The Whittaker kits are now available through Carter Wallace and Wampole Laboratories (Cranbury, N.J.). Incstar is now DiaSorin.
For screening purposes, these tests did no better. When the three kits were tested on sera from subjects with subclinical infections, serum antibodies were correctly identified in as few as 5% of HSV-1-seropositive subjects and in only 55 to 75% of those with HSV-2 infections (2). The most consistent problem was in identifying HSV-2 antibodies from patients with prior HSV-1 infections. Because such patients have anamnestic responses to the type common antigens presented by the new HSV-2 infection, the test becomes more strongly reactive against HSV-1. As a result, only one HSV-2 seroconversion was detected in 12 individuals with culture-documented nonprimary HSV-2 in this study (2).
Type-Specific Antibody Tests
Accurate type-specific serologic assays allow identification of silent carriers of HSV-2 infection in patients with or without pre-existing antibodies to HSV-1. Such tests can also provide useful information in symptomatic patients when virological tests such as culture, antigen detection or PCR are not helpful. A number of test formats have been developed to detect type-specific antibodies (5). It is hoped that in the near future truly type-specific tests will supplant those based on crude antigen preparations.
Western blotting.
The Western blot assay for HSV was developed more than 10 years ago, and its performance characteristics were validated against culture in symptomatic patients in the United States and Australia (13, 36, 37). When combined with a step to cross-adsorb antibodies to HSV-1 and HSV-2 antigens, the test is highly accurate in differentiating HSV-1 and HSV-2 antibodies, even in HSV-2 patients with previous HSV-1 infections. However, Western blotting is expensive to perform and requires 2 to 5 days for screening and confirmation by preadsorption. For these reasons, this test has not been developed for commercial distribution.
Tests based on gG.
A number of investigators have developed tests based on the type-specific protein glycoprotein G from HSV-2 (gG-2) and either glycoprotein C (gC-1) or glycoprotein G (gG-1) from HSV-1. Very limited sequence homology exists between gG-1 and gG-2. Type common sequences are found primarily in the leader sequence, which is lost during processing of the proteins after translation, and in the membrane anchor region, which has limited immunogenicity due to its being sequestered in the infected cell membrane (51, 63).
These tests use a variety of formats for immobilizing the gG antigen, including nitrocellulose (48, 49, 60, 67), capture antibodies (35, 77), and plastic microwells (36, 57). While most of these tests use colorimetric end points based on enzyme-substrate reactions (enzyme immunoassays [EIA]), some that involve radioactive (71) or fluorescent (14) signals have also been described. A type-specific monoclonal antibody-blocking assay with native, crude antigen preparations (including gG-2) performed well against Western blotting in clinically characterized cases (71). The specificity of this test is conferred by the monoclonal antibodies whose binding is (or is not) blocked by the test sample.
Another approach to the identification of type-specific antibodies is to apply recombinant gG-1 and gG-2 from a mammalian expression system to nitrocellulose as discrete bands. This strip immunoblot assay developed by Chiron (Emeryville, Calif.) includes bands of the type-common protein gD-2 and an HSV-1 dominant peptide of glycoprotein gB-1. These two proteins elicit early antibody responses and may allow recognition of early seroconversion before gG antibodies are detectable. The resulting assay was tested against Western blotting and was shown to be 95% sensitive for HSV-1 and 98% sensitive for HSV-2 with 99% specificity for both HSV-1 and HSV-2 (4).
Commercial gG-based type-specific tests.
A recently developed gG EIA (Gull Laboratories, Salt Lake City, Utah) may be approved by the Food and Drug Administration (FDA) and on the market in the United States in 1999. This test is based on native gG-1 and gG-2 proteins which have been immunoaffinity purified with monoclonal antibodies. We compared its performance against the Western blot assay on 193 sera submitted to our laboratory for type-specific HSV serologic testing (8). For 172 serum samples which had unequivocal typing results by both assays, the sensitivity of the gG EIA was 95% for HSV-1 and the specificity was 96%; the sensitivity for HSV-2 was 98%, and the specificity was 97%.
Of the 193 serum samples, 13 (7%) had equivocal gG EIA results for HSV-1 or HSV-2. Twelve (2%) of these sera had definitive typing results by Western blotting. Like most EIA formats, the Gull test is rapid (1.5 h) and easy to perform and can be run on any commercially available EIA processor. This test represents a great improvement in accuracy over current EIA tests based on crude antigen preparations. When combined with a second confirmatory test such as Western blotting, the overall sensitivity and specificity approach those of Western blotting.
A second test based on gG-2 that has been purified by lectin chromatography was developed by Diagnology Ltd. (Belfast, Northern Ireland). This test is designed to be performed in a few minutes in clinic or office settings and has been submitted for FDA approval.
Limitations of gG-based tests.
Tests based on recombinant gG from bacterial or baculovirus expression vectors (60, 67) may not detect all the antibodies elicited in humans by exposure to gG. Recombinant gG may lack epitopes that depend upon glycosylation mechanisms available in mammalian cells. Lectin- or immunoaffinity-purified gG is likely to provide a more complete complement of conformational and linear epitopes, and, indeed, tests involving native gG-1 and gG-2 have performed well in terms of sensitivity and specificity for HSV-2 antibodies (6, 48, 49, 59, 78).
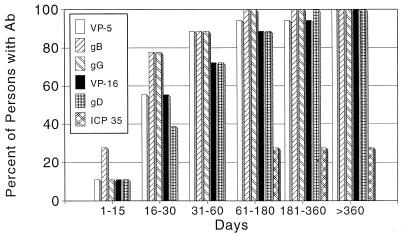
Any test based on antibodies to only one protein is vulnerable to variations in the titer and timing of antibody responses to that protein. By radioimmunoprecipitation (50) and by Western blotting (1), we have shown that antibody to gG arises relatively late, first appearing 2 to 3 months after infection in 60 to 70% of newly infected patients (Fig. 1). The remaining patients can require up to 6 months to seroconvert to gG. Recent studies with a gG capture EIA (35) and with the Gull gG EIA have confirmed the longer times required for detectable immunoglobulin G (IgG) antibodies to gG to develop. The use of IgM tests may decrease the time needed to detect seroconversion (5, 36, 37).
FIG. 1.
Profile of the development of antibodies to HSV-2. Eighteen patients with primary genital HSV-2 were monitored weekly for 8 weeks and then monthly for 12 to 18 months. Sera drawn at these visits were tested, as sets, following adsorption against HSV-1 proteins. Antibodies reacting with HSV-2 proteins were detected by Western blotting. HSV-2 targets of type-specific antibodies were identified by migration characteristics. The cumulative percentage of patients with antibodies (Ab) to glycoproteins gB, gG, and gD, to nucleocapsid proteins VP5 and ICP-35, and to the tegument protein, VP16, were determined for each period (Days) following the onset of genital herpes.
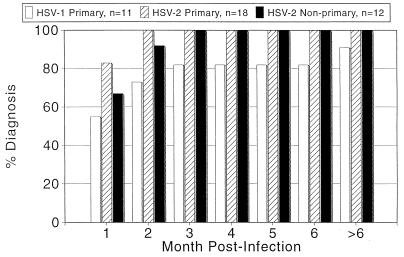
Because of the greater number of antigens available, Western blotting may identify seroconversion more quickly (Fig. 2). In a recent clinical trial, the Chiron strip immunoassay identified approximately 4% of sera that lacked antibody to gG but were reactive against gB and/or gD on the strip (unpublished observation). While it is not known whether these results were due to early seroconversion, the results suggest that inclusion of an early-antibody target may increase the sensitivity of protein-specific tests. In our laboratory, a follow-up Western blot can be used to definitively characterize sera with questionable EIA results (8). Type-specific IgM tests may also be helpful in identifying early seroconversion; such tests are in development.
FIG. 2.
Cumulative seroconversion as shown by Western blotting. Patients with culture-documented first episodes of genital herpes were samples sequentially; 26 were seronegative at the onset of symptoms; of these, 8 had HSV-1 genital herpes (HSV-1 Primary) and 18 had HSV-2 genital herpes (HSV-2 Primary). Twelve were HSV-1 seropositive at onset and had HSV-2 cultured from lesions (HSV-2 Non-primary). The earliest time (Month Post-Infection) at which seroconversion was detected by Western blotting was recorded. The cumulative proportion of patients diagnosed (% Diagnosis) is presented for each period.
RECOMMENDATIONS FOR HSV SEROLOGIC TESTING
Symptomatic Patients
Patients presenting with genital lesions should have samples taken for viral culture or antigen detection (3). This direct testing should always be considered the first choice to definitively establish HSV as the etiologic agent. Typing of HSV isolates from genital lesions should be requested because the natural histories of HSV-1 and HSV-2 differ. However, because virologic tests can be falsely negative when cultures are improperly handled or in patients seen late in the course of primary or nonprimary first episodes, the use of acute- and convalescent-phase sera (best drawn 3 to 4 months later) may be helpful in documenting the acquisition of genital HSV infection. Serologic tests on paired samples alone, is a reasonable backup test when sampling for viral detection is not feasible. While rising titers obtained from nontyping tests may be associated with seroconversion, it is highly recommended that type-specific testing be used if available.
Apparent first episodes of genital symptoms may be, in fact, the first recognized recurrence of a long-standing infection. In our clinic, of more than 200 persons seeking care for first episodes, 5% with HSV-1 and 17% with HSV-2 genital herpes had full Western blot profiles of antibodies to the same type of HSV as was isolated from the genital tract. Rising antibody titers will not be seen in association with such episodes.
Since type-specific serologic test have not been widely used, studies to determine the populations which would most benefit from the identification of HSV serologic status have not been done or are under way. However, the epidemiology and biology of HSV-2 suggest that it may be possible to identify persons at highest risk for serious sequelae of genital herpes. In our opinion, they include asymptomatic patients, patients at risk of HIV infection, and pregnant women. These groups are discussed below.
Asymptomatic Patients
The most significant potential application of serology is to detect silent carriers of HSV-2, especially in high-risk settings such as STD clinics. Screening of semen donors may also be indicated (56). Tests which detect antibodies to type-specific proteins such as gG should be used; those based on crude antigen preparations are too inaccurate to be helpful in most settings. An exception is that nontyping serologic tests are adequate to screen persons who may be undergoing immunosuppressive therapy for organ transplantation or cancer and who may be candidates for antiviral suppressive therapy, regardless of the type and site of HSV infection.
Patients at Risk of HIV Infection
Patients presenting to STD clinics and homosexual men are at higher than normal risk of having genital herpes. Identification of unrecognized HSV-2 infections by type-specific serologic testing may allow these individuals to practice behavioral change to prevent the transmission of HSV-2. The higher risk of acquiring HIV-1 in those with genital herpes suggests that HSV serologic screening may be important in preventing the spread of this infection as well.
Pregnant Women
Type-specific HSV serologic screening has been recommended to identify women at risk of acquiring genital HSV-1 or HSV-2 infections close to term, a setting in which there is high risk (30 to 50%) of neonatal herpes (17, 19, 62). Once rapid, commercial, type-specific tests such gG EIA are widely available, universal screening of pregnant women will become more practical and may be implemented in some settings. Women found to lack HSV-1 or HSV-2 antibodies can be advised of the risks of acquiring genital herpes during late pregnancy and can be provided with focused counseling on behaviors to avoid. In a recent large study of pregnant women, 2.3% of seronegative women seroconverted to HSV-1 and another 1.4% seroconverted to HSV-2 during pregnancy (19). Women whose partners are also tested have the added advantage of understanding the extent of their risk of acquiring genital herpes. The attack rate in HSV-seronegative women with HSV-2-positive partners is 33% over the course of pregnancy, whereas the HSV-1-positive women with an HSV-2-positive partner has a 5% chance of acquiring HSV-2 during pregnancy. The seronegative woman with an HSV-1-positive partner has a 4% chance of acquiring HSV-1; however, not all of these infections are genital (19).
CONCLUSIONS
In conclusion, commercial vendors are developing tests that identify HSV-1 and HSV-2 antibodies from patients with established infections. These tests are considerably more accurate than the kits now on the market. For the next few years, it is likely that the current nontyping tests will remain available. Clinicians and laboratories will have to be especially vigilant to ensure that tests purporting to identify type-specific HSV antibodies are, in fact, based on type-specific proteins or peptide constructs. With that proviso, the widespread availability of low-cost, accurate, type-specific serologic tests may allow the diagnosis of subclinical and unrecognized HSV-2 infections. While serologic diagnosis alone cannot halt the spread of genital herpes, it is an important step in the clinical management and counselling of patients who wish to have full information about the status of their sexual health (29).
REFERENCES
- 1.Ashley R, Mack K, Critchlow C, Shurtleff M, Corey L. Differential effect of systemic acyclovir treatment of genital HSV-2 infections on antibody responses to individual HSV-2 proteins. J Med Virol. 1988;24:309–320. doi: 10.1002/jmv.1890240308. [DOI] [PubMed] [Google Scholar]
- 2.Ashley R, Cent A, Maggs V, Nahmias A, Corey L. Inability of enzyme immunoassays to discriminate between infections with herpes simplex virus types 1 or 2. Ann Intern Med. 1991;115:520–526. doi: 10.7326/0003-4819-115-7-520. [DOI] [PubMed] [Google Scholar]
- 3.Ashley R. Laboratory techniques in the diagnosis of herpes simplex infection. Genitourin Med. 1993;69:174–183. doi: 10.1136/sti.69.3.174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ashley R, Warford A, Gleaves C, Zenilman J, Alexander D, Corey L. Abstracts of the 35th Annual Meeting of the Infectious Disease Society of America 1997. 1997. Evaluation of CHIRON RIBA HSV type-1/type-2 strip immunoblot assay for typing of HSV antibodies, abstr. 29; p. 360. [Google Scholar]
- 5.Ashley R. Type-specific antibodies to HSV-1 and -2: review of methodology. Herpes. 1998;5:33–38. [Google Scholar]
- 6.Ashley R L, Militoni J, Lee F, Nahmias A, Corey L. Comparison of Western blot (immunoblot) and glycoprotein G-specific immunodot enzyme assay for detecting antibodies to herpes simplex virus types 1 and 2 in human sera. J Clin Microbiol. 1988;26:662–667. doi: 10.1128/jcm.26.4.662-667.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ashley R L. Current concepts of laboratory diagnosis of herpes simplex infection. In: Sacks S L, Straus S E, Whitley R J, Griffiths P D, editors. Clinical management of herpes viruses. Washington, D.C: IOS Press; 1995. pp. 137–171. [Google Scholar]
- 8.Ashley R L, Wu L, Pickering J, Tu M, Schnorenberg L. Premarket evaluation of a commercial glycoprotein G-based enzyme immunoassay for herpes simplex virus type-specific antibodies. J Clin Microbiol. 1998;36:294–295. doi: 10.1128/jcm.36.1.294-295.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Barton S E, Wright L K, Link C M, Munday P E. Screening to detect asymptomatic shedding of herpes simplex virus (HSV) in women with recurrent genital HSV infection. Genitourin Med. 1986;62:181–185. doi: 10.1136/sti.62.3.181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Barton S E, Davis J M, Moss V W, Tyms A S, Munday P E. Asymptomatic shedding and subsequent transmission of genital herpes simplex virus. Genitourin Med. 1987;63:102–105. doi: 10.1136/sti.63.2.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Barton S E, Munday P E, Patel R J. Asymptomatic shedding of herpes simplex virus from the genital tract: uncertainly and its consequences for patient management. Intern J Sex Transm Dis AIDS. 1996;7:229–232. doi: 10.1258/0956462961917799. [DOI] [PubMed] [Google Scholar]
- 12.Benedetti J, Corey L, Ashley R. Recurrence rates of genital herpes after symptomatic first-episode infection. Ann Intern Med. 1994;121:847–854. doi: 10.7326/0003-4819-121-11-199412010-00004. [DOI] [PubMed] [Google Scholar]
- 13.Bernstein D I, Lovett M A, Bryson Y J. Serologic analysis of first-episode nonprimary genital herpes simplex virus infection. Am J Med. 1984;77:1055–1060. doi: 10.1016/0002-9343(84)90188-8. [DOI] [PubMed] [Google Scholar]
- 14.Boucher F D, Yasukawa L L, Kerns K, Kastelein M, Arvin A M, Prober C G. Detection of antibodies to herpes simplex virus type 2 with a mammalian cell line expressing glycoprotein gG-2. Clin Diagn Virol. 1993;1:29–38. doi: 10.1016/0928-0197(93)90031-y. [DOI] [PubMed] [Google Scholar]
- 15.Breinig M K, Kingsley L A, Armstrong J A, Freeman D J, Ho M. Epidemiology of genital herpes in Pittsburgh: serologic, sexual, and racial correlates of apparent and inapparent herpes simplex infections. J Infect Dis. 1990;162:299–305. doi: 10.1093/infdis/162.2.299. [DOI] [PubMed] [Google Scholar]
- 16.Brock B V, Selke S, Ashley R L, Corey L. Frequency of asymptomatic shedding of HSV in women with genital herpes. JAMA. 1990;263:418–420. [PubMed] [Google Scholar]
- 17.Brown Z A, Benedetti J, Ashley R, Burchett S, Selke S, Berry S, Vontver L A, Corey L. Neonatal herpes simplex virus infection in relation to asymptomatic maternal infection at the time of labor. N Engl J Med. 1991;324:1247–1252. doi: 10.1056/NEJM199105023241804. [DOI] [PubMed] [Google Scholar]
- 18.Brown Z A, Benedetti J K, Watts D H, Selke S, Berry S, Ashley R, Corey L. A comparison between detailed and simple histories in the diagnosis of genital herpes complicating pregnancy. Am J Obstet Gynecol. 1995;172:1299–1303. doi: 10.1016/0002-9378(95)91496-x. [DOI] [PubMed] [Google Scholar]
- 19.Brown Z A, Selke S, Zeh J, Kopelman J, Maslow A, Ashley R L, Watts D H, Berry S, Herd M, Corey L. The acquisition of herpes simplex virus during pregnancy. N Engl J Med. 1997;337:509–515. doi: 10.1056/NEJM199708213370801. [DOI] [PubMed] [Google Scholar]
- 20.Catotti D N, Clarke P, Catoe K E. Herpes revisited: still a cause for concern. Sex Transm Dis. 1993;20:77–80. [PubMed] [Google Scholar]
- 21.Christenson B, Bottiger M, Svensson A, Jeansson S. A 15-year surveillance study of antibodies to herpes simplex virus types 1 and 2 in a cohort of young girls. J Infect. 1992;25:147–154. doi: 10.1016/0163-4453(92)93943-k. [DOI] [PubMed] [Google Scholar]
- 22.Corey L. Genital herpes. In: Holmes K K, Mardh P A, Sparling P F, Wiesner P J, editors. Sexually transmitted diseases. 2nd ed. New York, N.Y: McGraw Hill Book Co.; 1990. pp. 391–413. [Google Scholar]
- 23.Corey, L. 1994. The current trend in genital herpes. Progress in prevention. Sex. Transm. Dis. 21(Suppl. 2):S38–S44. [PubMed]
- 24.Corey L, Holmes K K. Genital herpes simplex virus infection: current concepts in diagnosis, therapy and prevention. Ann Intern Med. 1983;98:973–983. doi: 10.7326/0003-4819-98-6-973. [DOI] [PubMed] [Google Scholar]
- 25.Corey L, Wald A. New developments in the biology of genital herpes. In: Sacks S L, Straus S E, Whitley R J, Griffiths P D, editors. Clinical management of herpes viruses. New York, N.Y: IOS Press; 1995. pp. 43–53. [Google Scholar]
- 26.Cowan F M, Johnson A M, Ashley R, Corey L, Mindel A. Relationship between antibodies to herpes simplex virus (HSV) and symptoms of HSV infection. J Infect Dis. 1996;174:470–475. doi: 10.1093/infdis/174.3.470. [DOI] [PubMed] [Google Scholar]
- 27.Cunningham A L, Lee F K, Ho D W, Field P R, Law C L H, Packham D R, McCrossin I D, Sjogren-Jansson E, Jeansson S, Nahmias A J. Herpes simplex virus type 2 antibody in patients attending antenatal or STD clinics. Med J Aust. 1993;158:525–528. doi: 10.5694/j.1326-5377.1993.tb121867.x. [DOI] [PubMed] [Google Scholar]
- 28.Douglas J M, Critchlow C, Benedetti J, Mertz G J, Connor J D, Hintz M A, Fahnlander A, Remington M, Winter C, Corey L. A double-blind study of oral acyclovir for suppression of recurrences of genital herpes simplex virus infection. N Engl J Med. 1984;310:1551–1556. doi: 10.1056/NEJM198406143102402. [DOI] [PubMed] [Google Scholar]
- 29.Fairley I, Monteiro E F. Patient attitudes to type-specific serological tests in the diagnosis of genital herpes. Genitourin Med. 1997;73:259–262. doi: 10.1136/sti.73.4.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fleming D T, McQuillan G M, Johnson R E, Nahmias A J, Aral S O, Lee F K, St. Louis M E. Herpes simplex virus type 2 in the United States, 1976 to 1994. N Engl J Med. 1997;337:1105–1111. doi: 10.1056/NEJM199710163371601. [DOI] [PubMed] [Google Scholar]
- 31.Forsgren M, Skoog E, Jeansson S, Olofsson S, Giesecke J. Prevalence of antibodies to herpes simplex virus in pregnant women in Stockholm in 1969, 1983 and 1989: implications for STD epidemiology. Int J Sex Transm Dis AIDS. 1994;5:113–116. doi: 10.1177/095646249400500207. [DOI] [PubMed] [Google Scholar]
- 32.Frenkel L M, Garratty E M, Shen J P, Wheeler N, Clark O, Bryson Y J. Clinical reactivation of herpes simplex virus type 2 infection in seropositive pregnant women with no history of genital herpes. Ann Intern Med. 1993;118:414–418. doi: 10.7326/0003-4819-118-6-199303150-00003. [DOI] [PubMed] [Google Scholar]
- 33.Gibson J J, Hornung C A, Alexander G R, Lee F K, Potts W A, Nahmias A J. A cross-sectional study of herpes simplex virus types 1 and 2 in college students: occurrence and determinants of infection. J Infect Dis. 1990;162:306–312. doi: 10.1093/infdis/162.2.306. [DOI] [PubMed] [Google Scholar]
- 34.Green J, Kocsis A. Psychological factors in recurrent genital herpes. Genitourin Med. 1997;73:253–258. doi: 10.1136/sti.73.4.253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hashido M, Lee F K, Inouye S, Kawana T. Detection of herpes simplex virus type-specific antibodies by an enzyme-linked immunosorbent assay based on glycoprotein G. J Med Virol. 1997;53:319–323. doi: 10.1002/(sici)1096-9071(199712)53:4<319::aid-jmv2>3.0.co;2-a. [DOI] [PubMed] [Google Scholar]
- 36.Ho D W T, Field P R, Sjogren-Jansson E, Jeansson S, Cunningham A L. Indirect ELISA for the detection of HSV-2 specific IgG and IgM antibodies with glycoprotein G (gG-2) J Virol Methods. 1992;36:249–264. doi: 10.1016/0166-0934(92)90056-j. [DOI] [PubMed] [Google Scholar]
- 37.Ho D W T, Field P R, Irving W L, Packham D R, Cunningham A L. Detection of immunoglobulin M antibodies to glycoprotein G-2 by Western blot (immunoblot) for diagnosis of initial herpes simplex virus type 2 genital infections. J Clin Microbiol. 1993;31:3157–3164. doi: 10.1128/jcm.31.12.3157-3164.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Holmberg S D, Stewart J A, Russel G, Byers R H, Lee F K, O’Malley P M, Nahmias A J. Prior herpes simplex virus type 2 infection as a risk factor for HIV infection. JAMA. 1988;259:1048–1050. [PubMed] [Google Scholar]
- 39.Hook E W, Cannon R O, Nahmias A J, Lee F F, Campbell C H, Glasser D, Quinn T C. Herpes simplex virus infection as a risk factor for human immunodeficiency virus infection in heterosexuals. J Infect Dis. 1992;165:251–255. doi: 10.1093/infdis/165.2.251. [DOI] [PubMed] [Google Scholar]
- 40.Johnson L S, Polsky B. Herpes simplex virus infection in the cancer patient. Infect Med. 1993;10:18–52. [Google Scholar]
- 41.Johnson J R, Egaas S, Gleaves C A, Hackman R, Bowden R A. Hepatitis due to herpes simplex virus in marrow-transplant recipients. Clin Infect Dis. 1992;14:38–45. doi: 10.1093/clinids/14.1.38. [DOI] [PubMed] [Google Scholar]
- 42.Koelle D N, Benedetti J, Langenberg A, Corey L. Asymptomatic reactivation of herpes simplex virus in women after first episode of genital herpes. Ann Intern Med. 1992;116:433–437. doi: 10.7326/0003-4819-116-6-433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Koutsky L, Ashley R, Homes K, Stevens C, Critchlow C, Kiviat N, Lipinski C, Wolner-Hanssen P, Corey L. The frequency of unrecognized type 2 herpes simplex virus infection among women: implications for the control of genital herpes. Sex Transm Dis. 1990;17:90–94. doi: 10.1097/00007435-199004000-00009. [DOI] [PubMed] [Google Scholar]
- 44.Koutsky L A, Stevens C E, Holmes K K, Ashley R L, Kiviat N B, Critchlow C W, Corey L. Underdiagnosis of genital herpes by current clinical and viral isolation procedures. N Engl J Med. 1992;326:1533–1539. doi: 10.1056/NEJM199206043262305. [DOI] [PubMed] [Google Scholar]
- 45.Kusne S, Schwartz M, Breinig M, Dummer J S, Lee R E, Selby R, Starzl T E, Simmons R L, Ho M. Herpes simplex virus hepatitis after solid organ transplantation in adults. J Infect Dis. 1991;163:1001–1007. doi: 10.1093/infdis/163.5.1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lafferty W E, Krofft S, Remington M, Giddings R, Winter C, Cent A, Corey L. Diagnosis of herpes simplex virus by direct immunofluorescence and viral isolation from samples of external genital lesions in a high prevalence population. J Clin Microbiol. 1987;25:323–326. doi: 10.1128/jcm.25.2.323-326.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Langenberg A, Benedetti J, Jenkins J, Ashley R L, Winter C, Corey L. Development of clinically recognizable genital lesions among women previously identified as having “asymptomatic” herpes simplex virus type 2 infection. Ann Intern Med. 1989;110:882–887. doi: 10.7326/0003-4819-110-11-882. [DOI] [PubMed] [Google Scholar]
- 48.Lee F K, Coleman R M, Pereira L, Bailey P D, Tatsuno M, Nahmias A J. Detection of herpes simplex virus type 2-specific antibody with glycoprotein G. J Clin Microbiol. 1985;22:641–644. doi: 10.1128/jcm.22.4.641-644.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lee F K, Pereira L, Griffin C, Reid E, Nahmias A. A novel glycoprotein for detection of herpes simplex virus type 1-specific antibodies. J Virol Methods. 1986;14:111–118. doi: 10.1016/0166-0934(86)90041-8. [DOI] [PubMed] [Google Scholar]
- 50.Lopez C, Arvin A, Ashley R. Immunity to herpesvirus infection in humans. In: Roizman B, Whitley R J, Lopez C, editors. The human herpesviruses. New York, N.Y: Raven Press; 1993. pp. 397–425. [Google Scholar]
- 51.McGeoch D J, Moss H W, McNab D, Frame M C. DNA sequence and genetic content of the HindIII region in the short unique component of herpes simplex virus type 2 genome: identification of the gene encoding glycoprotein G, and evolutionary comparisons. J Gen Virol. 1987;68:19–38. doi: 10.1099/0022-1317-68-1-19. [DOI] [PubMed] [Google Scholar]
- 52.Mertz G, Schmidt O, Jourden J L, Guinan M E, Remington M L, Fahnlander A, Winter C, Holmes K K, Corey L. Frequency of acquisition of first-episode genital infection with herpes simplex virus from symptomatic and asymptomatic source contacts. Sex Transm Dis. 1985;12:33–39. doi: 10.1097/00007435-198501000-00007. [DOI] [PubMed] [Google Scholar]
- 53.Mertz G, Benedetti J, Ashley R, Selke S, Corey L. Risk factors for sexual transmission of genital herpes. Ann Intern Med. 1992;116:197–202. doi: 10.7326/0003-4819-116-3-197. [DOI] [PubMed] [Google Scholar]
- 54.Mertz G, Loveless M O, Levin M J, Kraus S J, Fowler S L, Goade D, Trying S K. Oral famciclovir for suppression of recurrent genital herpes simplex virus in women: a multicenter double-blind placebo controlled trial. Arch Intern Med. 1997;157:343–349. [PubMed] [Google Scholar]
- 55.Mindel A, Faherty A, Carney O, Patou G, Freris M, Williams P. Dosage and safety of long-term suppressive acyclovir therapy for recurrent genital herpes. Lancet. 1988;ii:926–928. doi: 10.1016/s0140-6736(88)91725-4. [DOI] [PubMed] [Google Scholar]
- 56.Moore D E, Ashley R L, Zarutskie P W, Coombs R, Soules M R, Corey L. Transmission of genital herpes by artificial insemination with a donor experiencing asymptomatic primary HSV-2 infection. JAMA. 1989;261:3441–3443. [PubMed] [Google Scholar]
- 57.Nahmias A J, Lee F K, Keyserling H L. The epidemiology of genital herpes. In: Stanberry L, editor. Genital and neonatal herpes. New York, N.Y: John Wiley & Sons, Inc.; 1996. pp. 93–108. [Google Scholar]
- 58.Oliver L, Wald A, Kim M, Zeh J, Selke S, Ashley R, Corey L. Seroprevalence of herpes simplex virus infections in a family medicine clinic. Arch Fam Med. 1995;4:228–232. doi: 10.1001/archfami.4.3.228. [DOI] [PubMed] [Google Scholar]
- 59.Olofsson S, Lundstrom M, Marsden H, Jeansson S, Vahine A. Characterization of a herpes simplex virus type 2-specified glycoprotein with affinity for N-acetylgalactosamine-specific lectins and its identification as g92K or gG. J Gen Virol. 1986;67:737–744. doi: 10.1099/0022-1317-67-4-737. [DOI] [PubMed] [Google Scholar]
- 60.Parks D L, Smith C M, Rose J M, Brandis J, Coates S R. Seroreactive recombinant herpes simplex virus type 2-specific glycoprotein G. J Clin Microbiol. 1991;29:778–781. doi: 10.1128/jcm.29.4.778-781.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Patel R, Bodsworth N J, Woolley P, Peters B, Vejlsgaard G, Saari S, Gibb A, Robinson J the International Valaciclovir HSV Study Group. Valaciclovir for the suppression of recurrent genital HSV infection: a placebo controlled study of once daily therapy. Genitourin Med. 1997;73:105–109. doi: 10.1136/sti.73.2.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Prober C G, Corey L, Brown Z A, Hensleigh P A, Frenkel L M, Bryson Y J, Whitley R J, Arvin A M. The management of pregnancies complicated by genital infections with herpes simplex virus. Clin Infect Dis. 1992;15:1031–1038. doi: 10.1093/clind/15.6.1031. [DOI] [PubMed] [Google Scholar]
- 63.Rapoport T A. Protein translocation across and integration into membranes. Crit Rev Biochem. 1986;20:73–137. doi: 10.3109/10409238609115901. [DOI] [PubMed] [Google Scholar]
- 64.Rattray M C, Corey L, Reeves W C, Vontver L A, Holmes K K. Recurrent genital herpes among women: symptomatic vs asymptomatic viral shedding. Br J Vener Dis. 1978;54:262–265. doi: 10.1136/sti.54.4.262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ross J D C, Smith J W, Elton R A. The epidemiology of herpes simplex types 1 and 2 infection of the genital tract in Edinburgh 1978–1991. Genitourin Med. 1993;69:381–383. doi: 10.1136/sti.69.5.381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Safrin, S. 1992. Treatment of acyclovir-resistant herpes simplex virus infections in patients with AIDS. J. Acquired Immune Defic. Syndr. 5(Suppl. 1):S29–S32. [PubMed]
- 67.Sanchez-Martinez D, Schmid D S, Whittington W, Brown D, Reeves W C, Chatterjee S, Whitley R J, Pellet P E. Evaluation of a test based on baculovirus-expressed glycoprotein G for detection of herpes simplex virus type-specific antibodies. J Infect Dis. 1991;164:1196–1199. doi: 10.1093/infdis/164.6.1196. [DOI] [PubMed] [Google Scholar]
- 68.Schacker T, Ryncarz A, Goddard J, Diem K, Shaughnessy M, Corey L. Frequent recovery of HIV-1 from genital herpes simplex virus lesions in HIV-1-infected men. JAMA. 1998;280:61–66. doi: 10.1001/jama.280.1.61. [DOI] [PubMed] [Google Scholar]
- 69.Schacker, T., J. Zeh, H.-L. Hu, E. L. Hill, and L. Corey. Frequency of symptomatic and asymptomatic HSV-2 reactivations among HIV-infected men. J. Infect. Dis., in press. [DOI] [PubMed]
- 70.Scoular A, Leask B G S, Carrington D. Changing trends in genital herpes due to herpes simplex virus type 1 in Glasgow, 1985–88. Genitourin Med. 1990;66:226. doi: 10.1136/sti.66.3.226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Slomka M J, Ashley R L, Cowan R M, Cross A, Brown D W G. Monoclonal antibody blocking testes for the detection of HSV-1 and HSV-2 specific humoral responses: comparison with Western blot assay. J Virol Methods. 1995;55:27–35. doi: 10.1016/0166-0934(95)00042-s. [DOI] [PubMed] [Google Scholar]
- 72.Stamm W E, Handsfield H H, Rompalo A M, Ashley R, Roberts P, Corey L. The association between genital ulcer disease and acquisition of HIV infection in homosexual men. JAMA. 1988;260:1429–1433. [PubMed] [Google Scholar]
- 73.Stewart J A, Reef S E, Pellett P E, Corey L, Whitley R J. Herpesvirus infections in persons infected with human immunodeficiency virus. Clin Infect Dis. 1995;21:S114–S120. doi: 10.1093/clinids/21.supplement_1.s114. [DOI] [PubMed] [Google Scholar]
- 74.Strand A, Vahlne A, Svennerholm B, Wallin J, Lycke E. Asymptomatic virus shedding in men with genital herpes infection. Scand J Infect Dis. 1986;18:195–197. doi: 10.3109/00365548609032327. [DOI] [PubMed] [Google Scholar]
- 75.Straus S E, Takiff H E, Seidlin M, Bachrach S, Lininger L, DiGiovanna J J. Suppression of frequently recurring genital herpes. N Engl J Med. 1984;310:1545–1550. doi: 10.1056/NEJM198406143102401. [DOI] [PubMed] [Google Scholar]
- 76.Straus S E, Seidlin M, Takiff H E, Rooney J F, Felser J M, Smith H A, Roane P, Johnson F, Hallahan C, Ostrove J M, Nusinoff-Lehrman S. Effect of oral acyclovir treatment on symptomatic and asymptomatic virus shedding in recurrent genital herpes. Sex Transm Dis. 1989;16:107–113. doi: 10.1097/00007435-198904000-00013. [DOI] [PubMed] [Google Scholar]
- 77.Sullender W M, Yasukawa L L, Schwartz M, Pereira L, Hensleigh P A, Prober C G, Arvin A M. Type-specific antibodies to herpes simplex virus type 2 (HSV-2) glycoprotein G in pregnant women, infants exposed to maternal HSV-2 infection at delivery, and infants with neonatal herpes. J Infect Dis. 1988;157:164–171. doi: 10.1093/infdis/157.1.164. [DOI] [PubMed] [Google Scholar]
- 78.Svennerholm B, Olofsson S, Jeansson S, Vahlne A, Lycke E. Herpes simplex virus type-selective enzyme-linked immunosorbent assay with Helix pomatia lectin-purified antigens. J Clin Microbiol. 1984;19:235–239. doi: 10.1128/jcm.19.2.235-239.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wald A, Zeh J, Selke S, Ashley R L, Corey L. Virologic characteristics of subclinical and symptomatic genital herpes infections. N Engl J Med. 1995;333:770–775. doi: 10.1056/NEJM199509213331205. [DOI] [PubMed] [Google Scholar]
- 80.Wald A, Taylor L, Warren T, Remington M, Selke S, Zeh J, Corey L. Program and Abstracts of the 35th Interscience Conference on Antimicrobial Agents and Chemotherapy. Washington, D.C: American Society for Microbiology; 1995. Subclinical shedding of herpes simplex virus in men with genital herpes, abstr. H-111; p. 199. [Google Scholar]
- 81.Wald A, Zeh J, Barnum G, Davis L G, Corey L. Suppression of subclinical shedding of herpes simplex virus type 2 with acyclovir. Ann Intern Med. 1996;124:8–15. doi: 10.7326/0003-4819-124-1_part_1-199601010-00002. [DOI] [PubMed] [Google Scholar]
- 82.Wald A, Koutsky L, Ashley R L, Corey L. Genital herpes in a primary care clinic: demographic and sexual correlates of herpes simplex type 2 infections. Sex Transm Dis. 1997;24:149–155. doi: 10.1097/00007435-199703000-00005. [DOI] [PubMed] [Google Scholar]
- 83.Wald A, Corey L, Cone R, Hobson A, Davis G, Zeh J. Frequent genital herpes simplex virus 2 shedding in immunocompetent women: effect of acyclovir treatment. J Clin Invest. 1997;99:1092–1097. doi: 10.1172/JCI119237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Whitley R J. Perinatal herpes simplex virus infections. Rev Med Virol. 1991;1:101–110. [Google Scholar]