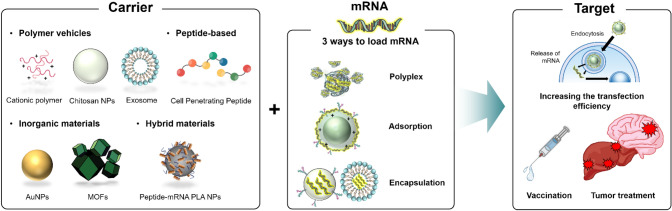
Abstract
RNA therapeutics, including messenger RNA (mRNA) and small interfering RNA (siRNA), are genetic materials that mediate the translation of genetic direction from genes to induce or inhibit specific protein production. Although the interest in RNA therapeutics is rising globally, the absence of an effective delivery system is an obstacle to the clinical application of RNA therapeutics. Additionally, immunogenicity, short duration of protein expression, unwanted enzymatic degradation, and insufficient cellular uptake could limit the therapeutic efficacy of RNA therapeutics. In this regard, novel platforms based on nanoparticles are crucial for delivering RNAs to the targeted site to increase efficiency without toxicity. In this review, the most recent status of nanoparticles as RNA delivery vectors, with a focus on polymeric nanoparticles, peptide-derived nanoparticles, inorganic nanoparticles, and hybrid nanoparticles, is discussed. These nanoparticular platforms can be utilized for safe and effective RNA delivery to augment therapeutic effects. Ultimately, RNA therapeutics encapsulated in nanoparticle-based carriers will be used to treat many diseases and save lives.
Keywords: RNA delivery, Nanoparticles, Nanomedicine, Gene therapy, Biomaterials
Introduction
Nucleic acid acts as a potent therapeutic agent by manipulating gene expression to treat various diseases [1, 2]. Messenger RNA (mRNA) can be used as a protein replacement therapy for cystic fibrosis and lung cancer [3–5]. Recently, mRNA has been utilized to develop the COVID-19 vaccine; it encodes the spike protein of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), making it resistant to infection through acquired immunity. As mRNA is not inserted into the genome but translated into the cytoplasm, there is no risk of insertional mutation. Furthermore, mRNA has shown substantial potential in a range of therapeutic applications, including vaccines for various infectious diseases and cancer immunotherapies. To achieve therapeutic effects, mRNA molecules must reach specific target cells and produce a sufficient level of proteins. However, naked mRNA molecules are susceptible to enzymatic degradation and are not easily taken up by cells because they are intrinsically unstable and large in size [6]. Thus, to develop effective mRNA therapeutics, it is crucial to improve their stability in the body and effectively deliver them to the target cells. According to recent studies, mRNA-loaded nanoparticle-based delivery systems showed higher protein expression than naked mRNA [7–9].
mRNA delivery nanotechnology can be classified into three categories. The first is ionic nanocomplexes, which are produced by mixing cationic delivery carriers and anionic mRNA. As mRNA has a negative charge, nanosized particles can be achieved by electrostatic force with a cationic polymer. Its most significant advantage is that it can be produced in a short time simply by a mixing process. The second method of mRNA delivery is the modification of the nanoparticle surface by a cationic polymer or by carrying out an exchange reaction with cationic functional groups, which generates a positive surface charge. In this case, mRNA can be readily absorbed by electrostatic interactions. The last method involves the encapsulation of mRNA by the nanoparticles, which has the advantage of safely delivering mRNA to the target site by preventing cargo degradation via endogenous ribonuclease (RNase) (Table 1).
Table 1.
Polymeric vehicles based on naturally derived polymers and synthetic polymers for RNA therapeutics to enhance delivery efficiency and treat cancer and other diseases
| Type | Delivery vehicle | Therapeutic modality | Target | Ref |
|---|---|---|---|---|
| Natural polymer | CS deacetylation, HA | mRNA | Promoting in vitro transfection efficiency | [25] |
| CS NP | miRNA | Efficient delivery of miRNA to macrophages/can be used to target atherosclerotic lesions | [27] | |
| CS-Heparin NP | siRNA | Improvement of pH-sensitive tranfection efficiency | [28] | |
| CS-HAD NP | CD44 targeting for bladder cancer therapy | [30] | ||
| CuVa NP | Gene knockdown of in vitro melanoma cell line B16 | [31] | ||
| Milk exosome | Anti-tumor activity against lung tumor xenograft | [34] | ||
| Exosome | mRNA | Tumor-suppressing function in glioma model | [35] | |
| Attenuation of in vivo models of Parkinson’s disease | [36] | |||
| Treatment of genetic disease, familial hypercholesterolemia | [37] | |||
| CS NP | mRNA | Transcript therapy for the treatment of cystic fibrosis | [38] | |
| Synthetic polymer | PEG-PGBA | mRNA | Promoting gene expression levels by modulating the stiffness | [39] |
| hPBAE | Inhaled delivery to the lung epithelium | [40] | ||
| ω-Cholesteryl-PEG-Pasp(DET) | Efficient mRNA introduction into the mouse lung | [41] | ||
| PEG-Plys | Efficient protein expression in mouse brain | [42] | ||
| PEG-Pasp(TEP) | Induction of mRNA to the mouse brain | [43] | ||
| Pasp(DET/CHE) | Targeting the brain for Alzheimer’s disease, Huntington’s disease | [44] | ||
| PAsp(DET) | Genome editing in the brain | [45] | ||
| fluorinated PEI | siRNA | Promotion of siRNA delivery to the lung | [46] | |
| PACE-PEG | mRNA | Increased inhalation delivery to the lung | [47] | |
| PEG-PAsp(DET) | Delivery of mRNA to the kidney | [48] | ||
| PEI-γ-PGA | High protein expression in the liver and spleen | [49] | ||
| CARTs | Anti-tumor activity against lymphoma and colon carcinoma tumors | [52] | ||
| PbAE | Antigen-specific immunity to treat ovarian cancer, melanoma, and glioblastoma | [53] | ||
| tyrosine-modified PEI | siRNA | High knockdown efficacies to treat melanoma | [54] | |
| ECV-modified PEI | Inhibition of tumor growth in prostate carcinoma xenografts | [55] | ||
| PbAE | Considerable knockdown in the glioblastoma cells | [56] | ||
| PbAE | Platform of glioblastoma treatment | [57] | ||
| PGS | mRNA | Vaccine against SARS-CoV-2 delivery system | [59] | |
| PVES | SARS-CoV-2 RBD mRNA vaccine | [60] | ||
| PEG-PAsp(DET/GlcAm) | ATP-responsiveness for selective mRNA release | [61] |
Numerous mRNA-based therapeutics are currently being developed or will be developed for a variety of therapeutic applications, including infectious diseases and cancers [10]. Since et al. 2019 article [11], clinical trials for more than 35 new nanoparticle technologies have commenced. Cancer treatment and imaging applications are among the novel nanoparticle technologies being tested in these new trials [12]. Moderna and AstraZeneca have been conducting phase II clinical research in recent years to evaluate the local administration of vascular endothelial growth factor (VEGF) mRNA for heart regeneration following myocardial infarction. Additionally, Moderna has initiated a phase I trial for the intratumoral administration of mRNA targeting OX40-binding partner, a member of the tumor necrosis factor receptor (TNFR) and tumor necrosis factor (TNF) superfamily expressed on activated CD4 and CD8 T cells [13].
Although RNA therapeutics have been thoroughly investigated and have successfully entered the clinic through lipid nanoparticles (LNP) [14], there are still limitations in delivery efficiency and organ- and tissue-specific targeting. In detail, cationic LNP components have potential toxicity since they can interact with enzymes, such as protein kinase C (PKC), to cause cytotoxicity [15, 16]. Cationic lipids, a component of LNP, interact with RNA to facilitate encapsulation but can also be toxic [17]. Additionally, a high concentration of cationic lipids can disrupt the cellular membrane structures, resulting in cell lysis and necrotic death [15, 16].
To overcome these limitations, various nanoparticle carriers, such as polymer-based nanoparticles (natural and synthetic), inorganic-based nanoparticles, hybrid nanoparticles, and biological-based approaches using exosomes and peptide nanoassemblies have been actively studied worldwide [18]. In fact, nanotechnology has been studied for decades for the treatment and diagnosis of diseases [19]. This review focuses on investigating advanced nanoparticles for the effective delivery of mRNA or small interfering (siRNA) studies over the last 3 years (Table 2).
Table 2.
Peptide-derived nanoparticles for efficient RNA therapeutics transfection and RNA-based cancer therapy
| Type | Delivery vehicle | Therapeutic modality | Target | Ref |
|---|---|---|---|---|
| Peptide-derived NP | HK (histidine-lysine) peptide | mRNA | Increased mRNA transfection in vitro | [64] |
| PEG-KL4 peptide | Inhalable dry powder for mRNA pulmonary delivery | [1] | ||
| OAA with s–s building block | Enhanced transfection efficiency and tolerability | [65] | ||
| CPP PepFect 14 | Induction of specific proteins in ovarian cancer | [67] | ||
| AmPPD | siRNA | Effective therapy in prostate cancer model | [68] | |
| p5RHH NP | Targeting drivers of tumor progression pancreatic adenocarcinoma model | [69] | ||
| chimeric peptide NP | miRNA | Plectin-1 targeted delivery in pancreatic cancer | [70] |
Polymeric Vehicles
Research on polymeric drug delivery has progressed significantly since the 1980s [20]. The major advantages of polymeric delivery systems include high tailoring ability, biodegradability, ease of functionalization, and good drug-release profile [21]. For the delivery of nucleic acids, the gene delivery system has some barriers to overcome. Vectors should increase transfection efficiency to penetrate the cell membrane and protect genes from harsh environments. In this regard, polymeric nanoparticles have many advantages as non-viral vectors for gene delivery. The fate of polymeric vectors is easily determined by structural characteristics such as molecular weight, surface charge, hydrophobicity, and chain compliance to promote delivery efficiency [22, 23]. Therefore, polymeric nanoparticles are promising candidates for gene vectors because their properties can be controlled to meet the eventual targets of the delivery system. Depending on the source material, mRNA/siRNA delivery studies can be divided into either naturally derived polymers or synthetic polymers (Fig. 1).
Fig. 1.
Schematic diagram of mRNA delivery using nanomaterials
Naturally Derived Polymers
Efforts to Improve Delivery Efficiency
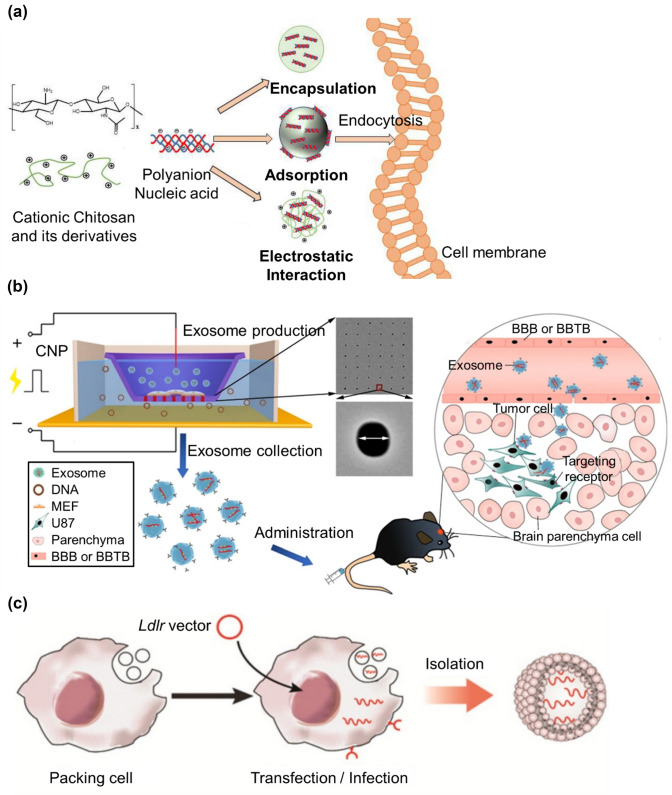
Naturally derived polymers such as hyaluronic acid (HA) or chitosan (CS) are used in drug delivery as mRNA carriers because they are biocompatible and controllable via various modifications. In particular, CS has the potential to act as an mRNA or gene carrier through complexation or surface adsorption owing to its unique advantages, cationic charge, and facile modification (Fig. 2a) [24]. One study showed that CS-based nanoparticles for mRNA delivery achieved enhanced transfection efficiencies by modulating some factors. Specifically, polymer length, degrees of deacetylation and HA, charge densities, and nucleic acids were controlled to optimize formulations reaching 60–65% transfection efficiency in vitro [25].
Fig. 2.
Schematic illustrations of nanoparticles based on naturally derived polymers. a Chitosan structure, fabrication mechanisms of particles based on chitosan–nucleic acid (reproduced with permission from [24]
Copyright 2019, Marine drugs). b Schematic representation of CNP-generated EVs for targeted nucleic acid delivery (reproduced with permission from [35] Copyright 2019, Nature Biomedical Engineering). c Schematic illustration of Ldlr mRNA encapsulation procedure into the exosomes (reproduced with permission from [37] Copyright 2021, Theranostics)
In the case of microRNAs (miRNAs) that have become an ideal class of biomarkers in diverse cancers [26], miRNA mimics are delivered by CS nanoparticles to target macrophages for treating cardiovascular diseases. miRNA/CS complexes can alter the expression of target genes both in vitro and in vivo. Moreover, mice treated with miRNA/CS nanoparticles exhibited reduced reverse cholesterol transport, indicating that this system has the potential to be applied in vivo to target atherosclerotic lesions [27]. The incorporation of heparin into CS nanoparticles is an effective way to increase transfection efficiency. In one study, the oligonucleotide release from heparin-included CS nanoparticles was pH-dependent following polyplex swelling and collapse of the polysaccharide network. The in vitro release studies showed an increased release rate of the oligonucleotide under slightly acidic conditions (pH 4.5); moreover, the efficacy of siRNA silencing vascular endothelial growth factor (VEGF) of ARPE-19 cells was 25% higher with heparin than that of CS nanoparticles without heparin [28] (Table 3).
Table 3.
Inorganic nanoparticles for efficient mRNA delivery and anti-tumor effects
| Type | Delivery vehicle | Therapeutic modality | Target | Ref |
|---|---|---|---|---|
| Inorganic materials | PAMAM-G5D-grafted AuNP | mRNA | Promotion of transfection activity | [72] |
| SeNP | Hepatocellular targeted delivery of mRNA | [74] | ||
| AuNPs-DNA | Inhibition of tumor growth in tumors formed by HeLa cells | [75] | ||
| CS-functionalized SeNP | Potential cancer immunotherapy agent | [76] |
Cancer Therapy
Recently, the importance of appropriate cancer therapy is increasingly emerging since it has become very important to detect and treat cancers in a healthy way [29]. Owing to the feasibility of polymer functionalization, many studies have reported enhanced targeting effects when polymeric nanoparticles are decorated with different peptides or ligands. For the delivery of siRNA targeting CD44, CS-based nanoparticles were conjugated with HA and presented a tumor-specific nanosystem (siRNA@CS-HAD NPs). In vivo experiments showed a significant accumulation of nanoparticles at the tumor site, substantially inhibiting the targeted oncogene and bladder cancer [30]. Another natural polymer, curdlan, which is a water-insoluble linear beta-1,3-glucan, was modified with amine groups, and its alkylation derivatives, denoted as CuVa polymers, exhibited an increase in biocompatibility as well as siRNA delivery efficiency. In vitro results showed that CuVa nanoparticles induced considerable knockdown of a disease-related gene, STAT3, in the mouse melanoma cell line B16 [31].
Exosomes are naturally derived from extracellular vesicles produced in cells and are released to the outside for intercellular communication in eukaryotic cells [32]. They are promising candidates as natural carriers because they are easy to handle, non-cytotoxic, and non-immunogenic [33, 34]. Interestingly, bovine milk exosomes, which are stable and resistant to degradation, were utilized as novel carriers for siRNA delivery. siRNA delivered via milk exosomes could silence key oncogenes, ranging from 2- to 10-fold knockdown in multiple cancers in vitro. In addition, intravenous administration of siRNA-loaded exosomes into the lung tumor xenograft in a nude mouse model induced anti-tumor activity [34]. Exosomes with targeting moieties have also been shown to enhance mRNA delivery in brain tumors by penetrating the blood–brain barrier (BBB). Large-scale exosomes were produced using cellular nanoporation (CNP) biochips, which can stimulate cells to release exosomes loaded with mRNA (Fig. 2b). To achieve glioma targeting, two different peptides were inserted into the N-terminus of CD47 and demonstrated tumor-suppressing function in implanted brain gliomas by inhibiting tumor growth and prolonging animal survival [35].
Applications for Non-cancerous Diseases
As mentioned earlier, exosomes are potent cell-derived nanovesicles (50–150 nm) that can mediate intracellular communication to achieve therapeutic outcomes. Intriguingly, a series of synthetic biology-inspired control devices, denoted as EXOsomal Transfer Into Cells (EXOtic), were investigated to enable efficient exosomal mRNA delivery without concentrating exosomes. In detail, the combination of exosome production boosters encoded in exosome producer cells and a specific mRNA packaging device could increase the delivery of exosomal mRNA into target cells. Exosome producer cells equipped with EXOtic devices have demonstrated their potential in therapeutic applications. Catalase mRNA delivery by exosome producer cells attenuated neurotoxicity and neuroinflammation in both in vitro and in vivo models of Parkinson’s disease [36] (Table 4).
Table 4.
Hybrid nanoparticles for RNA therapeutics delivery to treat cancer and induce immune responses
| Type | Delivery vehicle | Therapeutic modality | Target | Ref |
|---|---|---|---|---|
| Hybrid system | Zr-based MOF | mRNA | Increase of mRNA transfection activities | [80] |
| AMOF | mRNA | Quantitative analysis and in situ imaging of mRNA level | [82] | |
| DOTAP/protamine | High transfection efficiency | [83] | ||
| Zr-based MOF | siRNA | increased therapeutic effect against in vitro ovarian cancer cells | [86] | |
| ZIF-8 | miRNA | Suppression of tumor growth in breast cancer model | [87] | |
| CPP-functionalized DOTAP/mPEG-PCL | mRNA | Anti-cancer effects in colon cancer models | [88] | |
| PLGA/lipid | Strong immune responses for vaccination | [89] | ||
| PLA/CPP | Modulation of DC immune response for mRNA vaccine | [90] |
For the treatment of the genetic disease familial hypercholesterolemia (FH), Ldlr mRNA was encapsulated into exosomes to restore low-density lipoprotein receptor (LDLR) expression (Fig. 2c). Following tail-vein injection into Ldlr−/− mice, reversals of phenotypes such as steatosis, high-LDL cholesterol, and atherosclerosis were demonstrated in vivo. Short-term delivery systems can cause exosomes to accumulate in the liver, restore receptor expression, and treat disorders in Ldlr−/− mice even without targeting moieties. Thus, the exosome-mediated Ldlr mRNA delivery system represents a promising therapeutic approach for the treatment of FH [37]. Another genetic disease, cystic fibrosis (CF), is caused by mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene that requires transcript therapy. CS nanoparticles were used to incorporate capsaicin, then were surface-loaded with wtCFTR-mRNA to restore CFTR function. With increasing transfection efficiency, this mRNA/CS complex effectively restored CFTR function in the CF cell line CFBE41o [38].
Synthetic Polymers
Efforts to Improve Delivery Efficiency
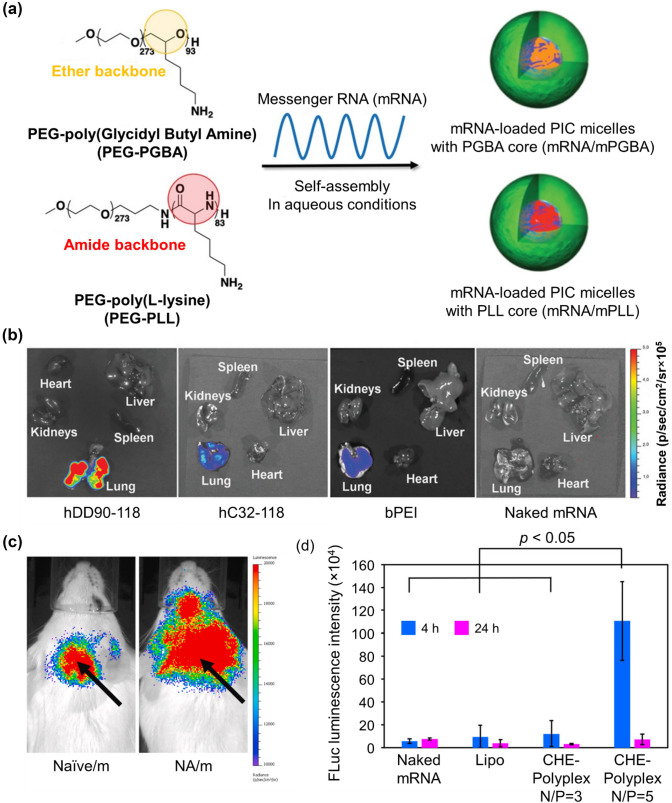
Polycation stiffness plays a significant role in determining mRNA delivery performance. Recently, two block copolymers, poly(ethylene glycol)–poly(glycidyl butylamine) (PEG-PGBA) and PEG-poly(l-lysine) (PEG-PLL), were compared to confirm the influence of additional flexibility (Fig. 3a). Twenty-four hours after the administration of GLuc mRNA/micelles into the mouse lung, the luminescence intensity from mRNA/PEG-PGBA was significantly higher than that from mRNA/PEG-PLL. PEG-PGBA, which has a less rigid segment, enhanced gene expression levels in vivo [39]. As another method to facilitate functional protein production, hyperbranched poly(beta-amino esters) (hPBAEs) were synthesized (hDD90-118) to deliver in vitro transcribed (IVT) mRNA to the lung via inhalation. A vibrating mesh nebulizer was used to deliver the IVT-mRNA-encoding firefly luciferase to mice. Repeated administration of inhaled hPBAE-mRNA resulted in consistent protein production in the lung epithelium (Fig. 3b) [40].
Fig. 3.
Synthetic-polymer-based nanoparticles for improved delivery efficiency. a Preparation of mRNA/m after complexation with PEG-PGBA (mRNA/mPGBA) or PEG-PLL (mRNA/mPLL) (reproduced with permission from [39]
Copyright 2020, Advanced Healthcare Materials). b Bioluminescence 24 h after inhalation of polyplexes; hDD90-118 vectors produced significantly higher radiance localized to the lung compared to hC32-118 and bPEI (p < 0.001, + SD, n = 4) (reproduced with permission from [40] Copyright 2019, Advanced Materials). c Representative images of in vivo luminescence imaging. Arrows represent naive mRNA/ PEG-PLys, NA/PEG-PLys injection site (NA: mRNA nanoassemblies) (reproduced with permission from [42] Copyright 2020, Biomaterials). d Quantitative evaluation of FLuc-mRNA expression level after intracerebroventricular administration of FLuc-mRNA/PAsp(DET/CHE) in mice (reproduced with permission from [44]. Copyright 2019, ACS Central Science)
In another study, mRNA was able to reach the lung by tightening the mRNA into polyplex micelles. To accomplish this, mRNA was pre-hybridized with cholesterol (Chol)-tethered RNA oligonucleotides (Chol (+)-OligoRNA), followed by the combination of ω-cholesteryl (ω-Chol) with PEG-polycation block copolymers. Chol (+)-OligoRNA on mRNA strands played a central role in tightening the mRNA packaging in the polyplex micelles because it attracts the ω-Chol moiety of the block copolymers. After the administration of mRNA into the mouse lung, the remaining amount of pre-hybridized mRNA was much higher than that of non-hybridized mRNA, based on qRT-PCR analysis. Therefore, this approach promotes stabilization of the mRNA delivery system even under harsh in vivo conditions [41]. Similarly, the technique of regulating the mRNA steric structure inside the polyplexes was also used in other studies. The bundled structure of mRNA impacted the physicochemical properties, stability, and functionality of polyplex micelles, resulting in efficient protein expression in cells in vitro and mouse brain in vivo (Fig. 3c). The bundled mRNA in block copolymers consisting of PEG-poly(lysine) (PLys) can improve the stability of polyplex micelles by effectively avoiding polyion exchange reactions and RNase attacks [42].
Several studies have suggested promising carriers for mRNA medicine in brain tissues to cure neuron-related diseases. Specifically, amino ethylene-repeating groups were conjugated to the side chains along the backbone of a polyaspartamide polymer terminated with PEG to enhance its ability to bypass the BBB. This PEG-based nanocomplex encapsulated with luciferase (Luc2) mRNA was infused into mouse brains via intracerebroventricular injection and exhibited the best Luc2 mRNA delivery efficiency [43]. In addition to incorporating PEG chains, the hydrophobic effect is a critical factor in controlling the properties of mRNA-loaded polyplexes. Among the series of amphiphilic polyaspartamide derivatives containing various hydrophobic moieties with cationic diethylenetriamine (DET) in the side chain, the polyaspartamide derivative with 11 residues of 2-cyclohexylethyl (CHE) moieties (PAsp(DET/CHE)) showed the highest mRNA expression both in cultured cells and in the mouse brain (Fig. 3d). These results suggest the possibility of using mRNA technology as a treatment for Alzheimer’s disease and Huntington's disease [44]. As another material used to target the brain to treat many neurological disorders, polyplex micelles are prepared using poly(N′-(N-(2-aminoethyl)-2-aminoethyl) aspartamide and DET (PAsp(DET)). The co-delivery of Cas9 mRNA and single-guide RNA is an efficient strategy for editing the genome in the brain. Furthermore, the presence of Cas9 mRNA and PEG improved diffusion in brain tissues, which could ultimately be efficient for genome editing [45].
In addition, other organs such as the liver, lung, spleen, and kidney can be targeted by a genetic material delivery system. Fluorination was introduced into polyethyleneimine (PEI) to reduce cytotoxicity. In vitro delivery results proved that fluorination promoted siRNA delivery, and thus Cy5.5-labeled siRNA complexed with PEI was significantly accumulated in the lungs after intravenous injection. Notably, fluorination-induced alteration is an excellent strategy for controlling biodistribution [46]. PEGylation, as well as fluorination reduces the toxicity of cationic polymeric vehicles. To investigate the effect of PEGylation on the transfection efficiency, stability, and toxicity, polymers from the poly(amine-co-ester) (PACE) family were blended with PEG-conjugated PACE at different ratios. It was essential to optimize the PEGylation because the addition of low concentrations of PACE-PEG to PACE improved polyplex stability but prohibited transfection in vitro. By contrast, the effects of PACE-PEG in vivo were different depending on the delivery route, and low concentrations of PACE-PEG significantly increased inhalation delivery to the lungs [47]. Interestingly, in an attempt to deliver mRNA to the kidneys, nanomicelles were formed by the self-assembly of mRNA and PEG-polyamino acid PAsp(DET) block copolymers. After the administration of mRNA into mouse kidneys by renal pelvic injection, most of the injection bolus remained at the injection site without diffusing into circulation [48]. Besides cationic polymers, an anionic complex containing PEI, γ-polyglutamic acid, and mRNA exhibited stable and high in vitro protein expression without cytotoxicity. Notably, after intravenous administration of the anionic complex in vivo, high protein expression was observed mainly in the liver and spleen [49].
Applications for Cancer Therapy
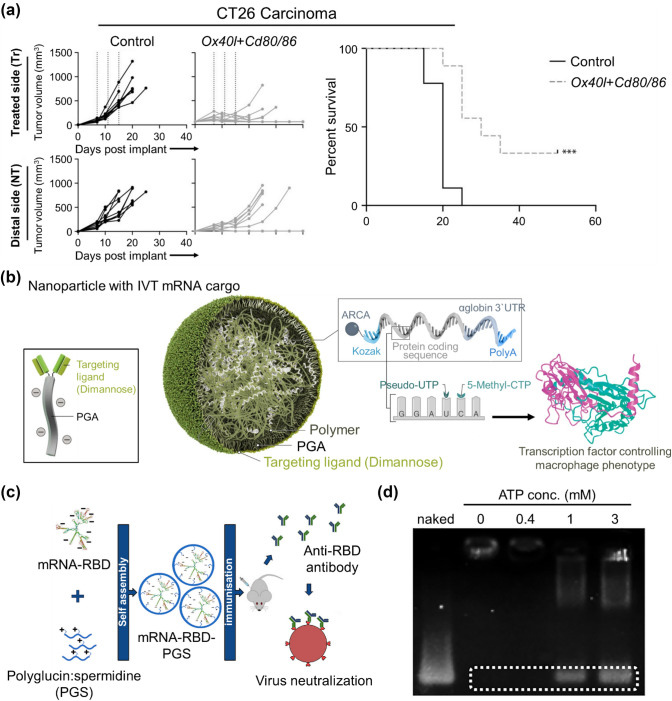
Gene therapy is a powerful tool for cancer treatment because the delivery of certain genes can construct specific protein expressions or inhibit certain genes [50]. Numerous gene carriers have been studied for efficient and safe delivery of nucleic acids for cancer therapy. Specifically, charge-altering releasable transporters (CARTs) initially serve as oligo(α-amino ester) cations that can protect and deliver mRNA, then switch their properties during intramolecular rearrangement to promote the intracellular release of functional mRNA [51]. CARTs have been utilized to carry mRNA-encoding Ox40l-, Cd80-, and Cd866, which are well-known immunomodulatory molecules. In the A20 and CT26 models, CART–mRNA complexes showed local upregulation of proinflammatory cytokines, T-cell activation, and migration of immune cells, leading to delayed tumor growth and enhanced tumor regression (Fig. 4a) [52]. Recently, mRNA-encoding interferon regulatory factor 5 (IRF5), combined with its activating kinase IKKβ, formed complexes with cationic poly(β-amino ester) (PbAE) (Fig. 4b). In animal models of ovarian cancer, melanoma, and gliblastoma (GBM), M1-polarizing transcription factor 5 (IRF5) can reprogram immune-suppressive M2 macrophages into immune-stimulatory M1 macrophages. Therefore, the induction of antigen-specific immunity via mRNA/polymeric nanoparticles is a prospective approach to treat cancer [53].
Fig. 4.
Synthetic-polymer-based nanoparticles for cancer therapy and vaccination. a Mice harboring two CT26 colon carcinoma tumors were treated intratumorally in one of the tumors with either Ox40l + Cd80/86 mRNA-CARTs or control mRNA-CARTs (Reproduced with permission from [52]
Copyright 2019, Cancer Research). b Design of macrophage-targeted polymeric NPs based on poly(beta-amino ester) formulated with mRNAs encoding key regulators of macrophage polarization (Reproduced with permission from [53] Copyright 2019, Nature Communication). c Graphical abstract of polyglucin:spermidine conjugate as a carrier of an mRNA-RBD vaccine encoding the receptor-binding domain (RBD) of the SARS-CoV-2 spike protein (reproduced with permission from [59] Copyright 2021, Vaccines). d Responsivity of PMs (PEG-PAsp(DET/GlcAm) to ATP. Gel electrophoresis was performed after incubation of PMs with several concentrations of ATP (reproduced with permission from [61] Copyright 2021, Journal of Controlled Release)
In prostate carcinoma xenograft mouse model, some modified PEIs have been used in cancer-medicine delivery systems. Specifically, tyrosine-modified PEIs were explored with low or very low molecular weights for siRNA delivery. Tumor xenografts were established by injecting PC3 cells, followed by repeated systemic administration of siRNA complexes via intraperitoneal injection. Importantly, siRNA complexes led to the knockdown of a target gene in tumors [54]. Similarly, the combination of PEI-based nanoparticles with naturally occurring extracellular vesicles (ECVs) showed an increased ability to inhibit tumor cells. ECVs are beneficial in gene delivery systems because they protect their payload from degradation or renal clearance and deliver their cargo directly into the cytosol. In mice bearing PC3 prostate carcinoma xenografts, ECV-modified PEI/siRNA complexes significantly inhibited tumor growth [55].
The treatment of tumor cells with GBM-delivering siRNA is an approach recently receiving great interest. In a recent study, biodegradable PbAE incorporated with disulfide bonds successfully delivered siRNA to the cytosol by environmentally triggered degradation and demonstrated therapeutic efficiency both in vitro and in vivo. The polymeric nanoparticles showed the highest in vitro permeability and siRNA-knockdown in patient-derived GBM cells. Furthermore, in vivo analysis in mice inoculated with GBM1A cells verified significant knockdown in the inoculated GBM cells after crossing the BBB [56]. Likewise, five anti-GBM genes (Robo1, YAP1, NKCC1, EGFR, and survivine) were delivered via bioreducible PbAE-based nanoparticles as a platform for GBM treatment. In mice bearing luciferase-positive GBM1A cells, the in vivo imaging system (IVIS) results showed that tumors treated with co-delivery of five siRNA sequences were less luminescent than the controls [57].
Applications for Vaccination
In recent years, mRNA vaccines have played a major role in combating infectious pathogens. Therefore, the optimization of various mRNA vaccine delivery materials is crucial for safer and more efficient deliveries [58]. Besides lipid-based vehicles, other materials are being developed that can promote cargo storage and transportation.
As the mRNA vaccine targeted toward the SARS-CoV-2 delivery system, a polycationic polymer, polyglucin:spermidine conjugate (PGS), was used as a carrier (Fig. 4c). Moreover, mRNA was encoded with the receptor-binding domain (RBD) of the SARS-CoV-2 spike protein because the main virus-neutralizing epitopes are concentrated in that domain. Mice were immunized with an mRNA-RBD-PGS vaccine to confirm their immunogenicity in vivo. Serum analysis revealed increased uptake by antigen-presenting cells and heightened immune responses with virus-specific antibodies [59].
Copolymers forming polyplex micelles have been studied as efficient and safe mRNA delivery systems. Interestingly, vitamin-E-succinate-modified polyethyleneimine copolymer (PVES) was synthesized for potential mRNA vaccine delivery. The in vitro results proved that modification with vitamin E significantly reduced the cytotoxicity of naïve PEI. Moreover, this self-assembled polymeric micelle delivery system containing SARS-CoV-2 RBD mRNA showed an increased amount of antigen-specific antibody IgG and substantial T-cell response against SARS-CoV-2 RBD antigen after administration in vivo [60]. In another study, the difference between extracellular and intracellular ATP concentration was utilized to achieve efficient cargo release from polyplex micelles. In particular, a PEG-polycation block copolymer consisting of PEG and poly(β-benzyl-l-aspartate) was synthesized with 4-carboxy-3-fluorophenylboronic acid (FPBA) (PEG-PAsp(DET/GlcAm) to obtain ATP responsiveness (Fig. 4d). As the formulation was stabilized by installing cholesterol moieties into the mRNA and ω-end of the block copolymers, in vivo measurement of the blood circulation profile in mice indicated upregulation of intact mRNA in the blood circulation [61].
Peptide-Derived Nanoparticles
Cell-penetrating peptides (CPPs) are short cationic peptide sequences that mediate the intracellular delivery of numerous biological cargos [62]. It is difficult for large protein complexes, genetic materials, and other molecules to penetrate cellular membranes; however, CPPs, a class of 5–30 residue peptides, can pass through membranes in an energy-dependent or independent manner [63]. In this regard, peptide-based delivery platforms have been studied as mRNA carriers of genetic materials for intracellular delivery.
Efforts to Improve Delivery Efficiency
To facilitate mRNA transfection efficiency, a single histidine was added to four branched histidine–lysine (HK) peptides to form H3K(+H)4b peptides. To assess the ability of these peptides to carry mRNA-expressing luciferase to MDA-MB-231 cells, researchers transfected the cells with H3K(+H)4b-mRNA polyplexes, which resulted in luciferase expression levels tenfold greater than those of controls. Stability and intracellular uptake, as well as transfection efficiency, also increased. Taken together, these histidine-rich H3K(+H)4b peptides played an essential role in enhancing mRNA delivery in vitro [64].
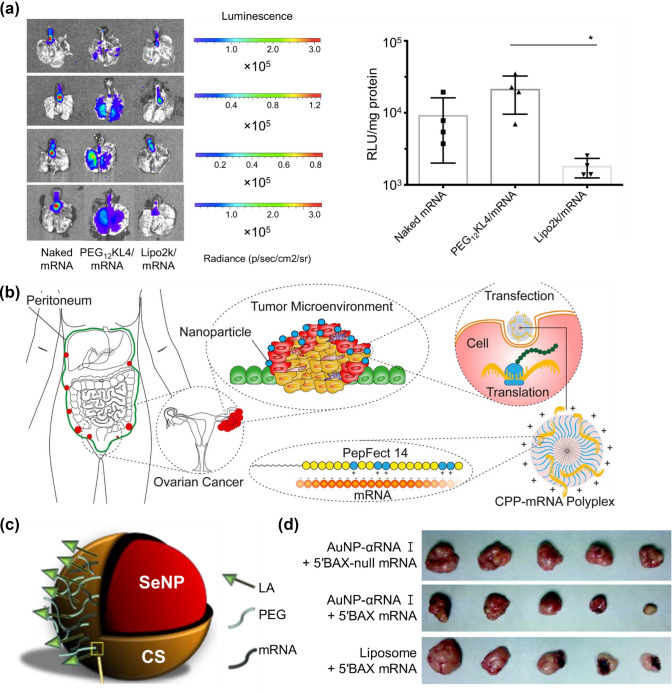
Imitating the structure of peptides is another format for peptide-based delivery systems. For example, the KL4 peptide, which mimics pulmonary surfactant protein B (SP-B), is utilized as the mRNA carrier with a monodisperse linear PEG attachment. Indeed, PEG-KL4/mRNA complexes showed better transfection efficiency than naked mRNA or lipoplexes in the lung region of mice after intratracheal administration (Fig. 5a). Importantly, this synthetic KL4 peptide provides a novel formulation of inhalable dry powder for mRNA pulmonary delivery [1]. In another research, a library of peptide-like oligoaminoamides composed of synthetic amino acids with the beneficial properties of PEI was investigated. Furthermore, various modifications, such as histidine and bioreducible disulfide blocks, were controlled to promote mRNA transfection efficiency [65].
Fig. 5.
mRNA delivery systems formulated with peptide-derived and inorganic nanoparticles. a Pulmonary delivery of mRNA formulations with different transfection agents (naked mRNA, PEG-KL4/mRNA, and lipoplex/mRNA). At 24 h post-administration, the lungs were isolated for bioluminescence imaging, and luciferase protein expression of lung tissues were measured (Reproduced with permission from [1]
Copyright 2019, Journal of Controlled Release). b Graphical abstract of cell-penetrating peptide (CPP) PepFect 14 (PF14)-mRNA nanoparticles in models of ovarian cancer (Reproduced with permission from [67] Copyright 2019, European Journal of Pharmaceutics and Biopharmaceutics). c Illustration of the fully functionalized SeNP (Reproduced with permission from [74] Copyright 2021, Pharmaceutics). d Actual sizes of representative tumors after xenograft implantation followed by injection of 5 nM AuNP-αRNA I-5’BAX mRNA in mice (reproduced with permission from [75] Copyright 2013, PLOS ONE)
Applications for Cancer Therapy
In a recent study, mRNA delivery systems based on CPP PepFect 14 (PF14) nanoparticles and LNPs were compared to measure detectable reporter protein expression in a xenograft model of ovarian cancer which is one of the most common cancers in women [66]. mRNA-PF14 nanoparticles could endure the in vivo environment of the abdominal cavity where ovarian cancer is usually confined, whereas LNPs could not. Therefore, this study demonstrates the potential of a CPP-based delivery system by intraperitoneal injection to induce specific proteins in ovarian cancer (Fig. 5b) [67]. In an effort to treat castration-resistant prostate cancer, amphiphilic phospholipid peptide dendrimers (AmPPDs) were formulated as an effective siRNA delivery system. Notably, DSPE–KK2 AmPPD bearing a less hydrophilic moiety facilitated efficient intracellular uptake and endosome release of siRNA complexes. Furthermore, these complexes caused a more potent gene knockdown and exhibited anti-cancer effects both in vitro and in vivo [68].
Most pancreatic adenocarcinomas (PDACs) are driven by the activation of KRAS, a proto-oncogene family of RAS proteins. In an attempt to target KRAS, a peptide-based p5RHH nanoparticle-condensing oligonucleotide was formed to deliver KRAS-specific siRNA. Delivery of the siRNA/p5RHH system to an in vivo model containing KPC-1 pancreatic cancer cells significantly reduced KRAS expression and inhibited pancreatic cancer growth [69]. Furthermore, a novel chimeric peptide supramolecular nanoparticle delivery system was also developed to deliver plectin-1 (PL-1)-targeted PDAC-specific miR-9. In detail, PL-1/miR-9 nanocomplexes could increase the anti-cancer effect of doxorubicin, giving rise to the chemosensitivity of PDAC cells to doxorubicin. PL-1/miR-9 nanoparticles significantly inhibited autophagy and induced apoptosis in PDAC cells in a pancreatic tumor model established from patient-derived xenografts, highlighting the possibility of synergic therapies against PDACs in the near future [70].
Inorganic Nanoparticles
Inorganic materials uniquely possess ideal characteristics such as non-toxicity, hydrophilicity, biocompatibility, and high stability. Gold nanoparticles (AuNPs) and selenium nanoparticles (SeNPs), in particular, have been widely explored for diagnosis, imaging, and drug delivery because they are biocompatible and chemically inert [71]. Unfortunately, neither inorganic nanomaterials for mRNA delivery nor lipid- or polymer-based vehicles are well developed, and yet requires much progress. However, these underdeveloped fields have shown promising results in recent years.
Efforts to Improve Delivery Efficiency
To improve transfection efficacy, cationic materials such as PEI or polyamidoamine (PAMAM) dendrimers were employed to coat the surface of inorganic nanoparticles with mRNA. PEI- and PAMAM-dendrimer-coated NPs showed high transfection activities owing to their branched structures and multiple amine groups [72, 73]. Another attractive material, SeNPs possess lower toxicity, higher bioavailability, biodegradability, and antioxidant activity. One study explored the novel use of SeNPs as liver-targeted mRNA-based nanocarriers by functionalizing SeNPs with CS, PEG, and lactobionic acid (LA) (Fig. 5c). Among LA-CS-SeNPs, PEG-CS-SeNPs, and dual-targeted PEG-LA-CS-SeNPs, LA-targeted nanocomplexes were primarily taken up by HepG2 cells. Therefore, functionalized SeNPs suggest the possibility of future liver-targeted immunotherapy [74].
Applications for Cancer Therapy
Interestingly, DNA oligonucleotides were conjugated to gold nanoparticles to form AuNP-DNA, which served as efficient vehicles for mRNA delivery. mRNA molecules hybridized to AuNP-DNA conjugates could be protected from attack by cellular RNase, which is why mRNA delivered by the AuNP–DNA complex exhibited greater longevity than that delivered by the liposome-based agent. BAX mRNA loaded onto AuNPs–DNA was injected into xenograft tumors in mice, resulting in the synthesis of biologically functional BAX protein, which could ultimately inhibit tumor growth by inducing apoptosis (Fig. 5d) [75]. In another study, among core–shell type biphasic materials, SeNPs were selected as drug carriers owing to their antioxidant and biocompatible properties. SeNPs formed core–shell structures coated with CS, mRNA, and a folate-targeting moiety. The functionalized surface was able to safely and stably deliver mRNA cargo in vitro and showed significant uptake in folate receptor cells. Therefore, CS-coated SeNPs have the potential for application in tumor vaccination and immunotherapy [76].
Hybrid Nanoparticles
Many efforts have been made to construct an ideal gene delivery platform. Focusing on only one type of component and more than two types of materials can maximize delivery efficiency. Specifically, hybrid nanoparticles using polymers, peptides, lipids, or inorganic materials are currently employed as gene carriers to promote biocompatibility and efficiency and reduce toxicity.
Efforts to Improve Delivery Efficiency
Metal–organic frameworks (MOFs) are extremely porous crystalline hybrid compounds that connect with metal ions and organic ligands to form 3D structures. MOFs possess a networking structure with organic ligands, containing voids and a wide range of surface areas, making them ideal candidates for biomedical applications [77]. These networks can be modulated to incorporate various ligands and metal ions [78, 79].
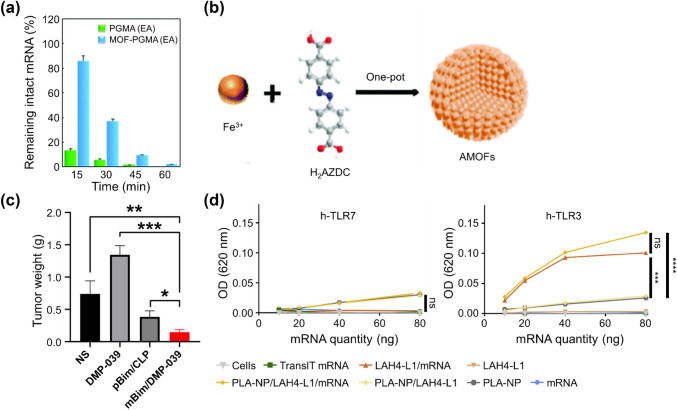
In a recent study, zirconium (Zr)-based MOFs coated with cationic polymers promoted the condensation of mRNA and improved colloidal stability and protection of encapsulated mRNA payloads from enzymatic degradation (Fig. 6a) [80]. In the case of delivering molecular beacons via Zr-based MOFs, selectively lighting up endogenous RNA targets have been shown by double-layer MOFs even without specific treatments [81]. Similarly, Liu et al. investigated VEGF mRNA by monitoring MOFs under hypoxia in living cells. This novel fluorescent nanoprobe system based on azoreductase-responsive functional MOFs (AMOFs) was engineered to visualize VEGF mRNA (Fig. 6b). Owing to a specific reduction reaction in the ligand moiety, the nanoprobe was released from the MOFs and combined with VEGF mRNA in its presence [82].
Fig. 6.
Efficient mRNA delivery by hybrid nanoparticles. a The tolerability of the mRNA encapsulated in MOF-PGMA(EA) or PGMA(EA) complexes (at an N/P ratio of 1.5) in the presence of 10% FBS (reproduced with permission from [80]
Copyright 2018, Chemical Communication). b Synthesis of the azobenzene-unit-containing metal–organic-framework-based nanoprobe (AMOF@MBs) (reproduced with permission from [82] Copyright 2019, Analyst). c mBim/DMP-039 complex suppressing the C26 subcutaneous xenograft model in vivo (reproduced with permission from [88] Copyright 2021, International Journal of Nanomedicine). d mRNA complexes stimulate innate immunity through PRR activation. Evaluation of PRR activation via the TLR7 and TLR3 pathways in HEK-Blue hTLR7 and HEK-Blue hTLR3 cells with mRNA formulations (reproduced with permission from [90] Copyright 2019, Biomaterials)
Polymers and lipids commonly form hybrid nanoparticles for mRNA delivery. Similarly, cationic lipids, 1,2-dioleoyl-3-trimethylammonium-propane (DOTAP), and cationic biopolymer protamine can be combined to form an mRNA delivery platform. As proven in both in vitro and in vivo studies, the most beneficial aspect of combining polymers and lipids is that much higher transfection can be achieved compared to when using a sole carrier [83]. This is due to the fact that hybrid systems based on polymer-lipid conjugates can improve circulatory half-lives dramatically [17]. Thus, the integration of a polymer into a lipid enhances structural stability, allows for controlled release, and prolongs protein expression [84, 85].
Applications for Cancer Therapy
Even though there are few papers that directly address mRNA delivery using MOFs, MOFs carriers have shown to exhibit remarkable cell uptake and lysosomal escape effect for encapsulated or adsorbed cargo. With specific post-modification and a precise understanding of the disease mechanism, MOF-based delivery systems are promising for both RNA delivery and combination tumor therapy. Chunbi et al. found that Zr-based MOFs that protect siRNA from nuclease degradation can enhance siRNA cellular uptake and promote siRNA escape from endosomes to silence MDR genes, increasing the effect of combination therapy against in vitro ovarian cancer cells [86]. MOFs created from another metal source, zinc (Zn)-based MOFs, have also demonstrated the possibility of being developed into potent delivery nanomaterials. The miRNA, which is known to have low stability and delivery efficacy in vivo, when adsorbed onto Zn-based MOFs, was able to effectively suppress tumor growth in a triple-negative breast-cancer mouse model after being successfully released in the cytoplasm [87].
CPP can enhance membrane penetration and cellular internalization. As an example of a peptide-functionalized hybrid delivery platform, the DMP nano-backbone was synthesized via the self-assembly of a cationic lipid (i.e., DOTAP) and copolymer, methoxy PEG-poly(ε-carprolactone) (mPEG-PCL). To promote cellular uptake, they fused two CPPs of R9 with cRGD and introduced them to the DMP backbone, producing DMP-R9-cRGD. As a result, modification of the cRGD-R9 peptide significantly promoted mRNA internalization into cancer cells and anti-cancer solid effects in numerous colon-cancer models (Fig. 6c) [88].
Applications for Vaccination
In one hybrid carrier designed for the co-delivery of mRNA and adjuvant, poly(lactic-co-glycolic acid) (PLGA) composed the core for loading of toll-like receptor (TLR)7 adjuvant, while lipids composed the shell for conjugation with mRNA. In vitro results demonstrated enhanced antigen expression and dendritic cell (DC) maturation. After intravenous administration of the hybrid nanovaccine, the expression levels of OVA-mRNA were high in the spleen, and strong immune responses were caused by dense antigen-presenting cells in vivo. Specifically, adjuvant-loaded hybrid mRNA stimulated a stronger OVA-specific T-cell immune response. Importantly, the co-delivery of antigens and adjuvants via a hybrid delivery platform can be used as a nanovaccine [89].
DCs have been used as targets for mRNA-based vaccine strategies because they connect innate and adaptive immune responses and are major regulators of cytotoxic and humoral adaptive responses. In this regard, poly(lactic acid) (PLA) and cationic CPP, which are membrane-active and efficient at mRNA translation in the cytosol, were synthesized together to deliver antigen-coding mRNAs into the DC cytosol. CPP was able to successfully condense mRNA and associate it with PLA nanoparticles. In all, in vitro results showed strong protein expression in DCs and the promotion of innate immunity activation (Fig. 6d) [90].
Conclusion and Perspectives
In this brief overview of RNA delivery systems, naturally derived carriers, synthetic polymer-based nanoparticles for numerous purposes, inorganic materials, and hybrid nanoparticles were introduced. The most crucial aspect of mRNA or siRNA delivery for biomedical applications is that genetic materials should reach the targeted site as efficiently and safely as possible. To promote delivery efficiency, numerous techniques for encapsulating genetic materials have been investigated. These carriers can protect the cargo from harsh in vivo environments, such as enzymatic degradation and recognition by the innate immune system with biocompatibility and long-term stability [67]. Ideal carriers should also induce efficient transfection into cells and produce high levels of specific proteins. In this regard, the nanoparticle has been attractive in the biomedical field due to their useful physiochemical properties [91].
Currently, LNPs carrying mRNA have received considerable attention owing to the COVID-19 pandemic. However, they have faced problems related to the potential toxicity of the components and their decreased interaction with endosomal membranes [92]. Up to this point, the feasibility of polymers or low-cytotoxicity of naturally derived materials and peptides has been advantageous for RNA delivery. Controlling various parameters such as stiffness, the level of branching, hydrophobicity, surface modification, and the steric structures of the cargo can affect delivery efficiency. Moreover, other therapeutic purposes, as well as vaccination, can be achieved through mRNA therapy. For example, numerous tumor models, such as ovarian cancer, glioma, and prostate cancer, have been tested using gene therapy. Gene therapy has many potential applications in protein replacement therapy and gene-editing techniques.
Applications in cancer immunotherapy, gene editing, and cell reprograming require protein expression for only a desired duration as well as high carrier stability, moderate binding force, efficient cellular uptake, and endosomal escape [93, 94]. Studies related to gene delivery systems have attempted to solve immunogenicity problems, the short duration of protein expression, rapid enzymatic degradation, and limited cellular uptake [95]. However, there still remains challenges to be addressed. First, detailed mechanisms of the nanoparticle-mediated delivery processes need more study to improve endosomal escape and transfection efficiency. Next, different nucleic acid medicines should achieve in vivo targeted delivery to the same immune cell of interest. Although the medicines were effective in animal studies, they may not be applicable to humans due to the differences in the immune systems between human and animal models [16].
In this regard, future research should concentrate on better understanding many aspects related to cellular internalization, intracellular trafficking pathways, and formulation design. Collaboration between biologists, biomaterial scientists, and doctors are critical in this endeavor [96]. Interestingly, there has been extensive focus on combining nanotechnology, chemistry, physics, and biology to develop biomaterials [97]. In conclusion, in addressing mentioned challenges, mRNA nanomedicines will solve various medical problems that are intractable using conventional approaches and constitute a promising strategy in biomedical research to treat serious diseases [95, 98].
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) Grant funded by the Korean government (MSIT) (NRF-2019R1A4A1028700 and NRF-2019R1C1C1006300). This work was also supported by the Korea Medical Device Development Fund grant funded by the Korean government (the Ministry of Science and ICT, the Ministry of Trade, Industry and Energy, the Ministry of Health and Welfare, the Ministry of Food and Drug Safety) (Project Number: NRF- 2021M3E5E5096420) and "Regional Innovation Strategy (RIS)" through the National Research Foundation of Korea(NRF) funded by the Ministry of Education(MOE) (2021RIS-001).
Declarations
Conflict of interest
The authors declare that they have no known competing financial interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Min Ji Byun and Jaesung Lim contributed equally to this work.
Contributor Information
Wooram Park, Email: parkwr@skku.edu.
Chun Gwon Park, Email: chunpark@skku.edu.
References
- 1.Qiu Y, Man RCH, Liao Q, Kung KLK, Chow MYT, et al. Effective mRNA pulmonary delivery by dry powder formulation of PEGylated synthetic KL4 peptide. J. Control Release. 2019;314:102–115. doi: 10.1016/j.jconrel.2019.10.026. [DOI] [PubMed] [Google Scholar]
- 2.Sahin U, Kariko K, Tureci O. mRNA-based therapeutics–developing a new class of drugs. Nat. Rev. Drug Discov. 2014;13:759–780. doi: 10.1038/nrd4278. [DOI] [PubMed] [Google Scholar]
- 3.Kormann MSD, Hasenpusch G, Aneja MK, Nica G, Flemmer AW, et al. Expression of therapeutic proteins after delivery of chemically modified mRNA in mice. Nat. Biotechnol. 2011;29:154–U196. doi: 10.1038/nbt.1733. [DOI] [PubMed] [Google Scholar]
- 4.Mays LE, Ammon-Treiber S, Mothes B, Alkhaled M, Rottenberger J, et al. Modified Foxp3 mRNA protects against asthma through an IL-10 dependent mechanism. J. Clin. Invest. 2013;123:1216–1228. doi: 10.1172/Jci65351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sahu I, Haque A, Weidensee B, Weinmann P, Kormann MSD. Recent developments in mRNA-based protein supplementation therapy to target lung diseases. Mol. Ther. 2019;27:803–823. doi: 10.1016/j.ymthe.2019.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Meng CY, Chen Z, Li G, Welte T, Shen HF. Nanoplatforms for mRNA therapeutics. Adv. Ther. Germ. 2021 doi: 10.1002/adtp.202000099. [DOI] [Google Scholar]
- 7.Probst J, Weide B, Scheel B, Pichler BJ, Hoerr I, et al. Spontaneous cellular uptake of exogenous messenger RNA in vivo is nucleic acid-specific, saturable and ion dependent. Gene Ther. 2007;14:1175–1180. doi: 10.1038/sj.gt.3302964. [DOI] [PubMed] [Google Scholar]
- 8.Van Lint S, Goyvaerts C, Maenhout S, Goethals L, Disy A, et al. Preclinical evaluation of TriMix and antigen mRNA-based antitumor therapy. Cancer Res. 2012;72:1661–1671. doi: 10.1158/0008-5472.CAN-11-2957. [DOI] [PubMed] [Google Scholar]
- 9.Phua KK, Leong KW, Nair SK. Transfection efficiency and transgene expression kinetics of mRNA delivered in naked and nanoparticle format. J. Control Release. 2013;166:227–233. doi: 10.1016/j.jconrel.2012.12.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Li B, Zhang X, Dong Y. Nanoscale platforms for messenger RNA delivery. Wiley Interdiscip. Rev Nanomed. Nanobiotechnol. 2019;11:e1530. doi: 10.1002/wnan.1530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Anselmo AC, Mitragotri S. Nanoparticles in the clinic: an update. Bioeng. Transl. Med. 2019;4:e10143. doi: 10.1002/btm2.10143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Anselmo AC, Mitragotri S. Nanoparticles in the clinic: an update post COVID-19 vaccines. Bioeng. Transl. Med. 2021 doi: 10.1002/btm2.10246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kowalski PS, Rudra A, Miao L, Anderson DG. Delivering the messenger: advances in technologies for therapeutic mRNA delivery. Mol. Ther. 2019;27:710–728. doi: 10.1016/j.ymthe.2019.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hou X, Zaks T, Langer R, Dong Y. Lipid nanoparticles for mRNA delivery. Nat. Rev. Mater. 2021 doi: 10.1038/s41578-021-00358-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Xue HY, Liu S, Wong HL. Nanotoxicity: a key obstacle to clinical translation of siRNA-based nanomedicine. Nanomedicine (London) 2014;9:295–312. doi: 10.2217/nnm.13.204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ho W, Gao M, Li F, Li Z, Zhang XQ, et al. Next-generation vaccines: nanoparticle-mediated DNA and mRNA delivery. Adv. Healthc. Mater. 2021;10:e2001812. doi: 10.1002/adhm.202001812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tenchov R, Bird R, Curtze AE, Zhou Q. Lipid nanoparticles-from liposomes to mRNA vaccine delivery, a landscape of research diversity and advancement. ACS Nano. 2021 doi: 10.1021/acsnano.1c04996. [DOI] [PubMed] [Google Scholar]
- 18.Smith SA, Selby LI, Johnston APR, Such GK. The endosomal escape of nanoparticles: toward more efficient cellular delivery. Bioconjug. Chem. 2019;30:263–272. doi: 10.1021/acs.bioconjchem.8b00732. [DOI] [PubMed] [Google Scholar]
- 19.Guerrero JM, Aguirre FS, Mota ML, Carrillo A. Advances for the development of in vitro immunosensors for multiple sclerosis diagnosis. Biochip J. 2021;15:205–215. doi: 10.1007/s13206-021-00018-z. [DOI] [Google Scholar]
- 20.Sung YK, Kim SW. Recent advances in polymeric drug delivery systems. Biomater. Res. 2020;24:12. doi: 10.1186/s40824-020-00190-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kamaly N, Xiao ZY, Valencia PM, Radovic-Moreno AF, Farokhzad OC. Targeted polymeric therapeutic nanoparticles: design, development and clinical translation. Chem. Soc. Rev. 2012;41:2971–3010. doi: 10.1039/c2cs15344k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ke LJ, Cai PQ, Wu YL, Chen XD. Polymeric nonviral gene delivery systems for cancer immunotherapy. Adv. Ther. Germ. 2020 doi: 10.1002/adtp.201900213. [DOI] [Google Scholar]
- 23.Rai R, Alwani S, Badea I. Polymeric nanoparticles in gene therapy: new avenues of design and optimization for delivery applications. Polym. Basel. 2019 doi: 10.3390/polym11040745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cao Y, Tan YF, Wong YS, Liew MWJ, Venkatraman S. Recent advances in chitosan-based carriers for gene delivery. Mar. Drugs. 2019 doi: 10.3390/md17060381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Soliman OY, Alameh MG, De Cresenzo G, Buschmann MD, Lavertu M. Efficiency of chitosan/hyaluronan-based mRNA delivery systems in vitro: influence of composition and structure. J. Pharm. Sci. 2020;109:1581–1593. doi: 10.1016/j.xphs.2019.12.020. [DOI] [PubMed] [Google Scholar]
- 26.Lee WJ, Kim KJ, Hossain MK, Cho HY, Choi JW. DNA-gold nanoparticle conjugates for intracellular miRNA detection using surface-enhanced raman spectroscopy. Biochip J. 2022 doi: 10.1007/s13206-021-00042-z. [DOI] [Google Scholar]
- 27.Nguyen MA, Wyatt H, Susser L, Geoffrion M, Rasheed A, et al. Delivery of microRNAs by chitosan nanoparticles to functionally alter macrophage cholesterol efflux in vitro and in vivo. ACS Nano. 2019;13:6491–6505. doi: 10.1021/acsnano.8b09679. [DOI] [PubMed] [Google Scholar]
- 28.Pilipenko I, Korzhikov-Vlakh V, Sharoyko V, Zhang N, Schafer-Korting M, et al. pH-sensitive chitosan-heparin nanoparticles for effective delivery of genetic drugs into epithelial cells. Pharmaceutics. 2019 doi: 10.3390/pharmaceutics11070317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bae J, Park SJ, Shin DS, Lee J, Park S, et al. A dual functional conductive hydrogel containing titania@polypyrrole-cyclodextrin hybrid nanotubes for capture and degradation of toxic chemical. Biochip J. 2021;15:162–170. doi: 10.1007/s13206-021-00015-2. [DOI] [Google Scholar]
- 30.Liang Y, Wang Y, Wang L, Liang Z, Li D, et al. Self-crosslinkable chitosan-hyaluronic acid dialdehyde nanoparticles for CD44-targeted siRNA delivery to treat bladder cancer. Bioact. Mater. 2021;6:433–446. doi: 10.1016/j.bioactmat.2020.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Erdene-Ochir T, Ganbold T, Zandan J, Han S, Borjihan G, et al. Alkylation enhances biocompatibility and siRNA delivery efficiency of cationic curdlan nanoparticles. Int. J. Biol. Macromol. 2020;143:118–125. doi: 10.1016/j.ijbiomac.2019.12.048. [DOI] [PubMed] [Google Scholar]
- 32.O'Brien K, Breyne K, Ughetto S, Laurent LC, Breakefield XO. RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat. Rev. Mol. Cell Biol. 2020;21:585–606. doi: 10.1038/s41580-020-0251-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Luan X, Sansanaphongpricha K, Myers I, Chen HW, Yuan HB, et al. Engineering exosomes as refined biological nanoplatforms for drug delivery. Acta Pharmacol. Sin. 2017;38:754–763. doi: 10.1038/aps.2017.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Aqil F, Munagala R, Jeyabalan J, Agrawal AK, Kyakulaga AH, et al. Milk exosomes—natural nanoparticles for siRNA delivery. Cancer Lett. 2019;449:186–195. doi: 10.1016/j.canlet.2019.02.011. [DOI] [PubMed] [Google Scholar]
- 35.Yang Z, Shi J, Xie J, Wang Y, Sun J, et al. Large-scale generation of functional mRNA-encapsulating exosomes via cellular nanoporation. Nat Biomed Eng. 2020;4:69–83. doi: 10.1038/s41551-019-0485-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kojima R, Bojar D, Rizzi G, Hamri GC, El-Baba MD, et al. Designer exosomes produced by implanted cells intracerebrally deliver therapeutic cargo for Parkinson's disease treatment. Nat. Commun. 2018;9:1305. doi: 10.1038/s41467-018-03733-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li Z, Zhao P, Zhang Y, Wang J, Wang C, et al. Exosome-based Ldlr gene therapy for familial hypercholesterolemia in a mouse model. Theranostics. 2021;11:2953–2965. doi: 10.7150/thno.49874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kolonko AK, Efing J, Gonzalez-Espinosa Y, Bangel-Ruland N, van Driessche W, et al. Capsaicin-loaded chitosan nanocapsules for wtCFTR-mRNA delivery to a cystic fibrosis cell line. Biomedicines. 2020 doi: 10.3390/biomedicines8090364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Miyazaki T, Uchida S, Nagatoishi S, Koji K, Hong T, et al. Polymeric nanocarriers with controlled chain flexibility boost mRNA delivery in vivo through enhanced structural fastening. Adv. Healthc. Mater. 2020 doi: 10.1002/adhm.202000538. [DOI] [PubMed] [Google Scholar]
- 40.Patel AK, Kaczmarek JC, Bose S, Kauffman KJ, Mir F, et al. Inhaled nanoformulated mRNA polyplexes for protein production in lung epithelium. Adv. Mater. 2019;31:e1805116. doi: 10.1002/adma.201805116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yoshinaga N, Uchida S, Naito M, Osada K, Cabral H, et al. Induced packaging of mRNA into polyplex micelles by regulated hybridization with a small number of cholesteryl RNA oligonucleotides directed enhanced in vivo transfection. Biomaterials. 2019;197:255–267. doi: 10.1016/j.biomaterials.2019.01.023. [DOI] [PubMed] [Google Scholar]
- 42.Koji K, Yoshinaga N, Mochida Y, Hong T, Miyazaki T, et al. Bundling of mRNA strands inside polyion complexes improves mRNA delivery efficiency in vitro and in vivo. Biomaterials. 2020;261:120332. doi: 10.1016/j.biomaterials.2020.120332. [DOI] [PubMed] [Google Scholar]
- 43.Chan LY, Khung YL, Lin CY. Preparation of messenger RNA nanomicelles via non-cytotoxic PEG-polyamine nanocomplex for intracerebroventicular delivery: a proof-of-concept study in mouse models. Nanomaterials (Basel) 2019 doi: 10.3390/nano9010067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kim HJ, Ogura S, Otabe T, Kamegawa R, Sato M, et al. Fine-tuning of hydrophobicity in amphiphilic polyaspartamide derivatives for rapid and transient expression of messenger RNA directed toward genome engineering in brain. ACS Cent. Sci. 2019;5:1866–1875. doi: 10.1021/acscentsci.9b00843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Abbasi S, Uchida S, Toh K, Tockary TA, Dirisala A, et al. Co-encapsulation of Cas9 mRNA and guide RNA in polyplex micelles enables genome editing in mouse brain. J. Control Release. 2021;332:260–268. doi: 10.1016/j.jconrel.2021.02.026. [DOI] [PubMed] [Google Scholar]
- 46.Xue L, Yan Y, Kos P, Chen X, Siegwart DJ. PEI fluorination reduces toxicity and promotes liver-targeted siRNA delivery. Drug Deliv. Transl. Res. 2021;11:255–260. doi: 10.1007/s13346-020-00790-9. [DOI] [PubMed] [Google Scholar]
- 47.Grun MK, Suberi A, Shin K, Lee T, Gomerdinger V, et al. PEGylation of poly(amine-co-ester) polyplexes for tunable gene delivery. Biomaterials. 2021;272:120780. doi: 10.1016/j.biomaterials.2021.120780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Oyama, N.K., Itaka, M.K.; Kawakami, S.: Efficient messenger RNA delivery to the kidney using renal pelvis injection in mice. Pharmaceutics 13, 1810 (2021). 10.3390/pharmaceutics13111810 [DOI] [PMC free article] [PubMed]
- 49.Hamada E, Kurosaki T, Hashizume J, Harasawa H, Nakagawa H, et al. Anionic complex with efficient expression and good safety profile for mRNA delivery. Pharmaceutics. 2021 doi: 10.3390/pharmaceutics13010126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mohammadinejad R, Dehshahri A, Sagar Madamsetty V, Zahmatkeshan M, Tavakol S, et al. In vivo gene delivery mediated by non-viral vectors for cancer therapy. J. Control Release. 2020;325:249–275. doi: 10.1016/j.jconrel.2020.06.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.McKinlay CJ, Vargas JR, Blake TR, Hardy JW, Kanada M, et al. Charge-altering releasable transporters (CARTs) for the delivery and release of mRNA in living animals. Proc. Natl. Acad. Sci. USA. 2017;114:E448–E456. doi: 10.1073/pnas.1614193114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Haabeth OAW, Blake TR, McKinlay CJ, Tveita AA, Sallets A, et al. Local delivery of Ox40l, Cd80, and Cd86 mRNA kindles global anticancer immunity. Cancer Res. 2019;79:1624–1634. doi: 10.1158/0008-5472.CAN-18-2867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhang F, Parayath N, Coon M, Stephan S, Stephan M. Genetic programming of macrophages to perform anti-tumor functions using targeted mRNA nanocarriers. Nat. Commun. 2019;10:3974. doi: 10.1158/0008-5472.CAN-18-2867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ewe A, Noske S, Karimov M, Aigner A. Polymeric nanoparticles based on tyrosine-modified, low molecular weight polyethylenimines for siRNA delivery. Pharmaceutics. 2019 doi: 10.3390/pharmaceutics11110600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zhupanyn P, Ewe A, Buch T, Malek A, Rademacher P, et al. Extracellular vesicle (ECV)-modified polyethylenimine (PEI) complexes for enhanced siRNA delivery in vitro and in vivo. J. Control Release. 2020;319:63–76. doi: 10.1016/j.jconrel.2019.12.032. [DOI] [PubMed] [Google Scholar]
- 56.Karlsson J, Rui Y, Kozielski KL, Placone AL, Choi O, et al. Engineered nanoparticles for systemic siRNA delivery to malignant brain tumours. Nanoscale. 2019;11:20045–20057. doi: 10.1039/c9nr04795f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kozielski KL, Ruiz-Valls A, Tzeng SY, Guerrero-Cazares H, Rui Y, et al. Cancer-selective nanoparticles for combinatorial siRNA delivery to primary human GBM in vitro and in vivo. Biomaterials. 2019;209:79–87. doi: 10.1016/j.biomaterials.2019.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Pardi N, Hogan MJ, Weissman D. Recent advances in mRNA vaccine technology. Curr. Opin. Immunol. 2020;65:14–20. doi: 10.1016/j.coi.2020.01.008. [DOI] [PubMed] [Google Scholar]
- 59.Karpenko LI, Rudometov AP, Sharabrin SV, Shcherbakov DN, Borgoyakova MB, et al. Delivery of mRNA vaccine against SARS-CoV-2 using a polyglucin: spermidine conjugate. Vac. Basel. 2021 doi: 10.3390/vaccines9020076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ren J, Cao YM, Li L, Wang X, Lu HT, et al. Self-assembled polymeric micelle as a novel mRNA delivery carrier. J. Control. Release. 2021;338:537–547. doi: 10.1016/j.jconrel.2021.08.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Yoshinaga N, Uchida S, Dirisala A, Naito M, Osada K, et al. mRNA loading into ATP-responsive polyplex micelles with optimal density of phenylboronate ester crosslinking to balance robustness in the biological milieu and intracellular translational efficiency. J. Control. Release. 2021;330:317–328. doi: 10.1016/j.jconrel.2020.12.033. [DOI] [PubMed] [Google Scholar]
- 62.Jones SW, Christison R, Bundell K, Voyce CJ, Brockbank SM, et al. Characterisation of cell-penetrating peptide-mediated peptide delivery. Br. J. Pharmacol. 2005;145:1093–1102. doi: 10.1038/sj.bjp.0706279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Shoari A, Tooyserkani R, Tahmasebi M, Lowik DWPM. Delivery of various cargos into cancer cells and tissues via cell-penetrating peptides: a review of the last decade. Pharmaceutics. 2021 doi: 10.3390/pharmaceutics13091391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.He JX, Xu SH, Leng QX, Mixson AJ. Location of a single histidine within peptide carriers increases mRNA delivery. J. Gene Med. 2021 doi: 10.1002/jgm.3295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Krhac-Levacic A, Berger S, Muller J, Wegner A, Lachelt U, et al. Dynamic mRNA polyplexes benefit from bioreducible cleavage sites for in vitro and in vivo transfer. J. Control Release. 2021;339:27–40. doi: 10.1016/j.jconrel.2021.09.016. [DOI] [PubMed] [Google Scholar]
- 66.Eom G, Hwang A, Kim H, Moon J, Kang H, et al. Ultrasensitive detection of ovarian cancer biomarker using Au nanoplate SERS immunoassay. Biochip J. 2021;15:348–355. doi: 10.1007/s13206-021-00031-2. [DOI] [Google Scholar]
- 67.van den Brand D, Gorris MAJ, van Asbeck AH, Palmen E, Ebisch I, et al. Peptide-mediated delivery of therapeutic mRNA in ovarian cancer. Eur. J. Pharm. Biopharm. 2019;141:180–190. doi: 10.1016/j.ejpb.2019.05.014. [DOI] [PubMed] [Google Scholar]
- 68.Dong YW, Chen Y, Zhu DD, Shi KJ, Ma C, et al. Self-assembly of amphiphilic phospholipid peptide dendrimer-based nanovectors for effective delivery of siRNA therapeutics in prostate cancer therapy. J. Control. Release. 2020;322:416–425. doi: 10.1016/j.jconrel.2020.04.003. [DOI] [PubMed] [Google Scholar]
- 69.Strand MS, Krasnick BA, Pan H, Zhang X, Bi Y, et al. Precision delivery of RAS-inhibiting siRNA to KRAS driven cancer via peptide-based nanoparticles. Oncotarget. 2019;10:4761–4775. doi: 10.18632/oncotarget.27109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Wu Y, Tang Y, Xie S, Zheng X, Zhang S, et al. Chimeric peptide supramolecular nanoparticles for plectin-1 targeted miRNA-9 delivery in pancreatic cancer. Theranostics. 2020;10:1151–1165. doi: 10.7150/thno.38327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kim R, Nam Y. Fabrication of a nanoplasmonic chip to enhance neuron membrane potential imaging by metal-enhanced fluorescence effect. Biochip J. 2021;15:171–178. doi: 10.1007/s13206-021-00017-0. [DOI] [Google Scholar]
- 72.Mbatha LS, Maiyo F, Daniels A, Singh M. Dendrimer-coated gold nanoparticles for efficient folate-targeted mRNA delivery in vitro. Pharmaceutics. 2021 doi: 10.3390/pharmaceutics13060900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kim KS, Han JH, Park JH, Kim HK, Choi SH, et al. Multifunctional nanoparticles for genetic engineering and bioimaging of natural killer (NK) cell therapeutics. Biomaterials. 2019 doi: 10.1016/j.biomaterials.2019.119418. [DOI] [PubMed] [Google Scholar]
- 74.Singh D, Singh M. Hepatocellular-targeted mRNA delivery using functionalized selenium nanoparticles in vitro. Pharmaceutics. 2021 doi: 10.3390/pharmaceutics13030298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Yeom JH, Ryou SM, Won M, Park M, Bae J, et al. Inhibition of xenograft tumor growth by gold nanoparticle-DNA oligonucleotide conjugates-assisted delivery of BAX mRNA. PLoS ONE. 2013;8:e75369. doi: 10.1371/journal.pone.0075369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Maiyo F, Singh M. Folate-targeted mRNA delivery using chitosan-functionalized selenium nanoparticles: potential in cancer immunotherapy. Pharmaceuticals Basel. 2019 doi: 10.3390/ph12040164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Singh N, Qutub S, Khashab NM. Biocompatibility and biodegradability of metal organic frameworks for biomedical applications. J. Mater. Chem. B. 2021;9:5925–5934. doi: 10.1039/d1tb01044a. [DOI] [PubMed] [Google Scholar]
- 78.Yang H, Peng F, Dang C, Wang Y, Hu D, et al. Ligand charge separation to build highly stable quasi-isomer of MOF-74-Zn. J. Am. Chem. Soc. 2019;141:9808–9812. doi: 10.1021/jacs.9b04432. [DOI] [PubMed] [Google Scholar]
- 79.Peng S, Bie B, Sun Y, Liu M, Cong H, et al. Metal-organic frameworks for precise inclusion of single-stranded DNA and transfection in immune cells. Nat. Commun. 2018;9:1293. doi: 10.1038/s41467-018-03650-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Sun P, Li Z, Wang J, Gao H, Yang X, et al. Transcellular delivery of messenger RNA payloads by a cationic supramolecular MOF platform. Chem. Commun. (Camb.) 2018;54:11304–11307. doi: 10.1039/c8cc07047d. [DOI] [PubMed] [Google Scholar]
- 81.Gao P, Lou R, Liu X, Cui B, Pan W, et al. Rational design of a dual-layered metal-organic framework nanostructure for enhancing the cell imaging of molecular beacons. Anal Chem. 2021;93:5437–5441. doi: 10.1021/acs.analchem.0c05060. [DOI] [PubMed] [Google Scholar]
- 82.Liu N, Zou Z, Liu J, Zhu C, Zheng J, et al. A fluorescent nanoprobe based on azoreductase-responsive metal-organic frameworks for imaging VEGF mRNA under hypoxic conditions. Analyst. 2019;144:6254–6261. doi: 10.1039/c9an01671f. [DOI] [PubMed] [Google Scholar]
- 83.Siewert CD, Haas H, Cornet V, Nogueira SS, Nawroth T, et al. Hybrid biopolymer and lipid nanoparticles with improved transfection efficacy for mRNA. Cells. 2020 doi: 10.3390/cells9092034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Zhao W, Zhang C, Li B, Zhang X, Luo X, et al. Lipid polymer hybrid nanomaterials for mRNA delivery. Cell Mol. Bioeng. 2018;11:397–406. doi: 10.1007/s12195-018-0536-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Dave V, Tak K, Sohgaura A, Gupta A, Sadhu V, et al. Lipid-polymer hybrid nanoparticles: synthesis strategies and biomedical applications. J. Microbiol. Methods. 2019;160:130–142. doi: 10.1016/j.mimet.2019.03.017. [DOI] [PubMed] [Google Scholar]
- 86.He C, Lu K, Liu D, Lin W. Nanoscale metal-organic frameworks for the co-delivery of cisplatin and pooled siRNAs to enhance therapeutic efficacy in drug-resistant ovarian cancer cells. J. Am. Chem. Soc. 2014;136:5181–5184. doi: 10.1021/ja4098862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Zhao HX, Li TT, Yao C, Gu Z, Liu CX, et al. Dual roles of metal-organic frameworks as nanocarriers for miRNA delivery and adjuvants for chemodynamic therapy. ACS Appl. Mater. Interfaces. 2021;13:6034–6042. doi: 10.1021/acsami.0c21006. [DOI] [PubMed] [Google Scholar]
- 88.Gao Y, Men K, Pan CB, Li JM, Wu JP, et al. Functionalized DMP-039 hybrid nanoparticle as a novel mRNA vector for efficient cancer suicide gene therapy. Int. J. Nanomed. 2021;16:5211–5232. doi: 10.2147/Ijn.S319092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Yang J, Arya S, Lung P, Lin Q, Huang J, et al. Hybrid nanovaccine for the co-delivery of the mRNA antigen and adjuvant. Nanoscale. 2019;11:21782–21789. doi: 10.1039/c9nr05475h. [DOI] [PubMed] [Google Scholar]
- 90.Coolen AL, Lacroix C, Mercier-Gouy P, Delaune E, Monge C, et al. Poly(lactic acid) nanoparticles and cell-penetrating peptide potentiate mRNA-based vaccine expression in dendritic cells triggering their activation. Biomaterials. 2019;195:23–37. doi: 10.1016/j.biomaterials.2018.12.019. [DOI] [PubMed] [Google Scholar]
- 91.Nguyen QH, Kim MI. Using nanomaterials in colorimetric toxin detection. Biochip J. 2021;15:123–134. doi: 10.1007/s13206-021-00013-4. [DOI] [Google Scholar]
- 92.Reichmuth AM, Oberli MA, Jaklenec A, Langer R, Blankschtein D. mRNA vaccine delivery using lipid nanoparticles. Ther. Deliv. 2016;7:319–334. doi: 10.4155/tde-2016-0006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Hajj KA, Whitehead KA. Tools for translation: non-viral materials for therapeutic mRNA delivery. Nat. Rev. Mater. 2017 doi: 10.1038/natrevmats.2017.56. [DOI] [Google Scholar]
- 94.Guan S, Rosenecker J. Nanotechnologies in delivery of mRNA therapeutics using nonviral vector-based delivery systems. Gene Ther. 2017;24:133–143. doi: 10.1038/gt.2017.5. [DOI] [PubMed] [Google Scholar]
- 95.Uchida S, Perche F, Pichon C, Cabral H. Nanomedicine-based approaches for mRNA delivery. Mol Pharm. 2020;17:3654–3684. doi: 10.1021/acs.molpharmaceut.0c00618. [DOI] [PubMed] [Google Scholar]
- 96.Wu Z, Li T. Nanoparticle-mediated cytoplasmic delivery of messenger RNA vaccines: challenges and future perspectives. Pharm. Res. 2021;38:473–478. doi: 10.1007/s11095-021-03015-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Cho HH, Heo JH, Jung D, Kim SH, Suh SJ, et al. Portable Au nanoparticle-based colorimetric sensor strip for rapid on-site detection of Cd2+ ions in potable water. Biochip. J. 2021;15:276–286. doi: 10.1007/s13206-021-00029-w. [DOI] [Google Scholar]
- 98.Islam MA, Reesor EK, Xu Y, Zope HR, Zetter BR, et al. Biomaterials for mRNA delivery. Biomater. Sci. 2015;3:1519–1533. doi: 10.1039/c5bm00198f. [DOI] [PMC free article] [PubMed] [Google Scholar]