Abstract
Coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), remains a significant global health emergency with new variants in some cases evading current therapies and approved vaccines. COVID-19 presents with a broad spectrum of acute and long-term manifestations. Severe COVID-19 is characterized by dysregulated cytokine release profile, dysfunctional immune responses, and hypercoagulation with a high risk of progression to multi-organ failure and death. Unraveling the fundamental immunological processes underlying the clinical manifestations of COVID-19 is vital for the identification and design of more effective therapeutic interventions for individuals at the highest risk of severe outcomes. Caspases are expressed in both immune and non-immune cells and mediate inflammation and cell death, including apoptosis and pyroptosis. Here we review accumulating evidence defining the importance of the expression and activity of caspase family members following SARS-CoV-2 infection and disease. Research suggests SARS-CoV-2 infection is linked to the function of multiple caspases, both mechanistically in vitro as well as in observational studies of individuals with severe COVID-19, which may further the impact on disease severity. We also highlight immunological mechanisms that occur in severe COVID-19 pathology upstream and downstream of activated caspase pathways, including innate recognition receptor signaling, inflammasomes, and other multiprotein complex assembly, inflammatory mediators IL-1β and IL-18, and apoptotic and pyroptotic cell death. Finally, we illuminate discriminate and indiscriminate caspase inhibitors that have been identified for clinical use that could emerge as potential therapeutic interventions that may benefit clinical efforts to prevent or ameliorate severe COVID-19.
Keywords: COVID-19, caspase, inflammation, apoptosis, therapeutic
Introduction
According to the World Health Organization (WHO), as of December 2021, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic and the cause of Coronavirus disease 2019 (COVID-19) has led to over 244 million infections and ~5 million deaths globally since the virus outbreak was first reported in 2019. SARS-CoV-2 infection we know now can result in a vast range of clinical pulmonary manifestations, from no symptoms to critical illness, which the latter could lead to extrapulmonary complications, including neurological, thromboembolic, cardiovascular, renal, gastrointestinal, hepatobiliary, endocrinologic, and dermatologic manifestations (1–3). Furthermore, unlike any other respiratory viruses, many individuals who recover from COVID-19 report lingering short term and long term persistent symptoms referred to as long-COVID or post-acute sequelae SARS-CoV-2 infection (PASC). Long-COVID can persist beyond 6 months after symptom onset and present with neurological, psychosocial, cardiothoracic, pulmonary, gastrointestinal, hematologic, and/or renal issues (4–7). The complexity of COVID-19 has been contentious in the area of therapies to combat the infection (8). Current FDA approved treatment for adults and children with COVID-19 include VEKLURY (remdesivir) and several emergency use authorizations (EUA) have been issued for several monoclonal antibodies, molnupiravir, and paxlovid (9–11). Treatment options targeting both the virus and/or host factors for the various stages and presentations of COVID-19 continue to expand and remain an area of critical need in an attempt to reduce the risk of hospitalization or death. With the advent of highly protective vaccines against SARS-Cov-2 infection the spread, disease severity, and mortality has been altered, though protection against novel variants of concern (VOCs) is proving an ongoing challenge.
Caspases are a highly conserved family of intracellular cysteine-dependent aspartate-specific proteases that primarily mediate cell death and inflammation (12–14). All caspases are constitutively expressed during homeostasis in both immune and non-immune cells as catalytically inactive zymogens that require appropriate signals to activate c-terminal protease domain (15). Caspases contain common highly conserved protein domains, such as caspase-associated recruitment domains (CARDS) and death effector domains (DEDs). Caspases have been functionally classified according to their involvement in either apoptosis or inflammation. Apoptosis is an immunologically silent and coordinated non-lytic process of dismantling and removing of damaged, infected, and aging cells. Host cellular apoptosis is thought to be a common viral infection response mechanism for restricting viral expansion. Much like apoptosis, inflammation is another initial host cell response to viral infection. Caspases that mediate inflammation facilitate the maturation of pro-interleukins by cleaving and activating their zymogen forms as well as promoting an inflammatory form of cell death called pyroptosis (16, 17). While there are also ‘outlier’ caspases defined by their role in the cell cycle and cell differentiation (18, 19), they are currently not known to be of significance in SARS-CoV-2 infection.
Evidence demonstrates that COVID-19 is an inflammatory disease mediated by a hyperactive immune response. Conventionally, SARS-CoV-2 gains cellular entry through the interaction of the spike protein receptor binding domain and host angiotensin-converting enzyme 2 (ACE2) receptor through endosomal mechanisms or TMPRSS2-mediated membrane fusion at the cell surface (20, 21), but noncanonical routes have also been identified (22–25). Nonetheless of entry mechanisms, uncoated viral RNA is released into the cell cytosol for damage recognition by host cell pattern recognition receptors (PRRs), such as Toll-like receptors (TLRs) and RIG-I like Receptors (RLRs), which can elicit a robust immune response. While SARS-CoV-2 viral RNA interactions with endosomal TLRs and RLRS can lead to the production of NF-κB pro-inflammatory cytokines (i.e. IL-6, IL-1β) and type I and III interferons (26), SARS-CoV-2 proteins can also activate host TLR2 pathways to induce pro-inflammatory cytokine production (27, 28). However, caspase activity is also a significant contributor to the pronounced cellular death and inflammatory characteristics of COVID-19. Unraveling caspase-related immunological processes contributing to COVID-19 sequelae is vital to identify and design effective host targeted therapeutic interventions for individuals at the highest risk of severe outcomes. This review, will focus on updates on the role of caspases and COVID-19 in disease pathogenesis and targeted therapies being considered to ameliorate disease outcomes.
Caspase Pathways in Inflammation and During SARS-CoV-2 Infection
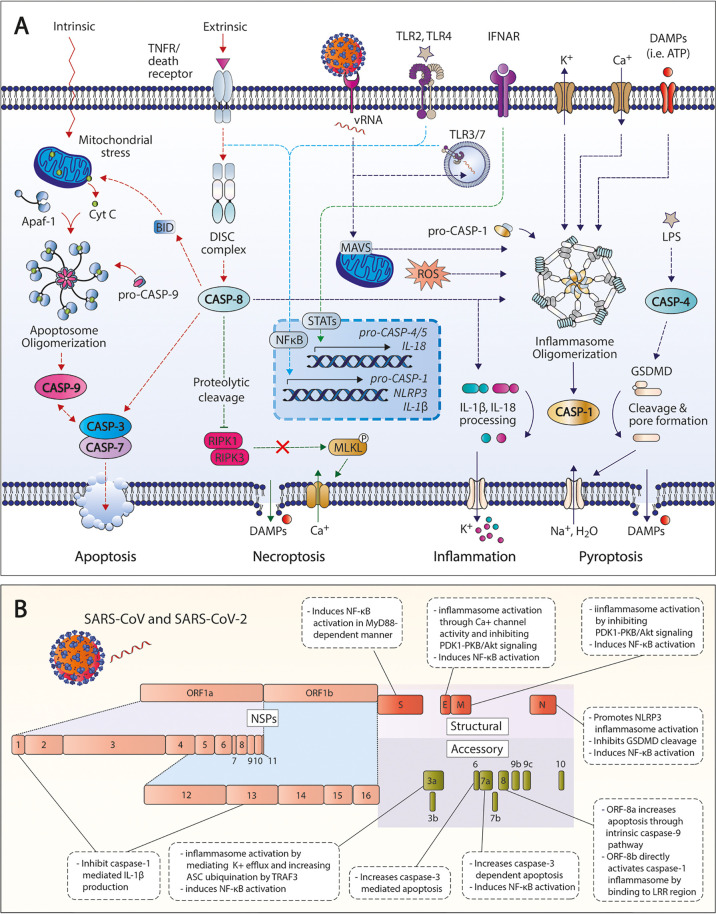
Previous preclinical studies have suggested the role of caspases primarily as inflammatory and apoptotic mediators in various pathologies, including Inflammatory, neurological and metabolic diseases, and cancer. Accumulating evidence reveal new insights on the importance of caspase-mediated inflammatory and apoptotic pathways during SARS-CoV-2 infection ( Figure 1A ). Cell death and dysregulated caspase activation has been associated with hematological and immunological findings in patients with COVID-19 (29, 30). Like other members of the Coronaviridae family, SARS-CoV-2 is an enveloped single-stranded positive-sense RNA virus comprised of four structural proteins: nucleocapsid (N), membrane (M), envelope (E), and spike (S) proteins (31). Additionally, SARS-CoV-2 open reading frames (ORFs) also encode for various non-structural proteins (NSPs) and accessory proteins that can be involved in viral RNA transcription and replication, and/or controlling the production of other viral proteins (32). These encoded structural, non-structural, and accessory proteins can target crucial immune pathways that contribute to host immune dysregulation and active viral evasion. SARS-CoV and SARS-CoV-2 proteins shown to actively modulate the induction and/or signaling of caspase-mediated pathways are summarized in Figure 1B .
Figure 1.
Activated caspase pathways in SARS-CoV-2. (A) Apoptotic and inflammatory pathways associated with caspases in SARS-CoV-2 infection and COVID-19. (B) Structural, non-structural, and accessory proteins of SARS-CoV and SARS-CoV-2 that modulate caspase-related pathways. TNFR, tumor necrosis factor receptor; CASP, caspase; DISC, death-inducing signaling complex; GSDMD, gasdermin D; ROS, reactive oxygen species; LDH, lactate dehydrogenase; DAMPs, danger-associated molecular patterns; TNFR, tumor necrosis factor receptor; IFNAR, interferon α/β receptor; LPS, lipopolysaccharide; RIPK, receptor-interacting serine/threonine, protein kinase; MLKL, mixed lineage kinase domain-like protein; NSP, non-structural protein.
Caspase-Mediated Apoptotic Pathways
Caspases that execute apoptosis either function in initiator (caspases 8, 9, and 10) or effector (caspases 3, 6, and 7) roles depending on their position in the signaling cascade (16, 17). Initiator caspases are recruited into multiprotein complexes, such as the apoptosome and death-inducing signaling complex (DISC), driven by a local increase of concentration that must be first triggered by either intrinsic or extrinsic processes. In the intrinsic pathway, intracellular stress signals lead to the release of cytochrome c (cyt c) from the mitochondria, which induces the formation of the apoptosome (33, 34). The apoptosome, consisting of cyt c and apoptotic protease-activating factor-1 (Apaf-1), recruits pro-caspase-9 via its N-terminal CARD. The extrinsic apoptotic pathway is mediated through the engagement of certain death receptors of the tumor necrosis factor (TNF) family (i.e. Fas), leading to the recruitment of adaptor proteins and caspases-8 or -10 into DISC via DED-mediated interactions (35, 36). Once recruited to multiprotein complexes, initiator caspases will dimerize to undergo proximity-induced autoactivation and act as proteolytic signal amplifiers to activate effector caspases (caspases 3, 6, and 7). However, caspase-8 can also cleave the pro-death BCL-2 family protein Bid to its truncated form (tBid) to induce cyt c release from the mitochondria and propagate the apoptotic pathway (37). Once effector caspases are activated, they induce the proteolytic degradation of multiple specific cellular substrates that facilitate the dismantling of the cell, including those that drive membrane blebbing, fragmentation of chromosomal DNA, and apoptotic body formation. Apoptosis is canonically thought of as an immunologically silent form of cell death; however, Fas-mediated apoptosis has been shown to result in the production of monocyte chemoattractant protein-1 (MCP-1), IL-6, and IL-8 (38). Furthermore, while apoptosis is considered an efficient antiviral defense to eliminate infected and damaged cells and dampen inflammation via the cleavage and inactivation of proinflammatory cellular signals (39), pathogen-induced apoptosis may increase infection and viral pathogenicity (40).
Several apoptotic caspases are shown to be active with SARS-CoV-2 infection. In vitro models using the human lung cancer line, Calu-3, found that caspases 3, 8, and 9 were cleaved into their activated forms in SARS-CoV-2-infected cells (41). Furthermore, active caspase-3 was also increased in SARS-CoV-2 infected human cortical organoids and glial cells indicating a strong link with SARS-CoV-2 inducing apoptosis (42). In COVID-19 patients, caspase-3/7 activity in red blood cells is upregulated compared to healthy individuals (43). Caspase-3 is also thought to play a role in the programmed cell death of platelets with SARS-CoV-2 infection. The internalization of SARS-CoV-2 by platelets, either in vitro or in COVID-19 patients, results in the colocalization of SARS-CoV-2 with phosphorylated mixed lineage kinase domain-like protein (phospho-MLKL), a mediator of necroptosis, and caspase-3 on nonpermeabilized platelets (44). This caspase-3 activity is suggested to be a potential contributor to thrombotic events observed in severe COVID-19 (45). Specific viral components of SARS-CoV-2 have been identified to modulate apoptosis via several mechanisms. SARS-CoV-2 accessory protein, ORF3a, was shown to induce apoptosis in Vero E6, HEK293T, and HepG2 cells via the extrinsic pathway, through activated caspase-8 cleavage of Bid to tBid (46). ORF-3a of SARS-CoV was previously identified to induce apoptosis through both death receptor- and mitochondria-mediated pathways, propagated through caspase 8 and 9 pathways, respectively (47–49); however, it’s pro-apoptotic capacity is shown to be greater than that of the ORF3a of SARS-CoV-2 (46). Beyond ORF-3a, SARS-CoV ORF-6, -7a, and 8a, all have been previously shown to trigger cellular apoptosis. ORF-6 induces apoptosis via caspase-3 mediated ER stress and JNK-dependent pathways (50), whereas ORF-8a is through a mitochondria-dependent pathway (51). While the mechanism of activation for ORF7a in promoting caspase-associated inflammation is unclear, the overexpression of ORF7a induces apoptosis in a caspase-3-dependent manner (52, 53). Finally, membrane glycoprotein M in conjunction with the N protein is also shown to trigger caspase-dependent apoptosis via inhibiting the activation of PDK1-PKB/Akt signaling (54).
Caspase-Mediated Inflammation and Pyroptosis
Inflammatory caspases are recruited to their cognate activation complexes called inflammasomes, protein platforms that aggregate in the cytosol in response to different stimuli (55). However, an initial priming step is generally required mediated by NF-κB through the engagement of PPRs that recognize pathogen associated molecular patterns (PAMPs) or host-derived damage associate molecular patterns (DAMPs), such as ATP or mitochondrial DNA. The most studied inflammatory caspase, caspase-1, is engaged by inflammasomes, including the NLRP, AIM2, and IFI16 inflammasomes. Activated caspase-1 then mediates the processing and secretion of the proinflammatory cytokines IL-1β and IL-18 (56). These cytokines have multiple roles in innate immunity and in bridging adaptive immune responses. IL-18 induces downstream IFN-γ responses, while IL-1β plays roles in neutrophil influx and activation, T and B-cell activation, cytokine and antibody production, and Th17 differentiation (57–60). On the other hand, inflammatory caspases 4 and 5 directly recognize intracellular lipopolysaccharide (LPS) (61), but require an initial step through the signaling of IFNAR and subsequent members of the signal transducer and activation of transcription (STAT) protein family. Another outcome of the activation of inflammatory caspases is pyroptosis, an inflammatory-related nonprogrammed cell death driven primarily by inflammasome and caspase-4/5 mediated cleavage of the pyroptotic executor cytosolic protein gasdermin D (GSDMD) (62–64). As caspase-1-dependent cytokines and DAMPs lack secretion signals, pyroptosis is thought to be one of the prime mechanisms mediating their cellular release (65–69). Although the conventional idea that inflammatory caspase activation would be protective by enhancing immunity against SARS-CoV-2 through the removal of infected cells and recruitment of monocytes to injury sites, concomitant pyroptosis exacerbating inflammation due to cellular release of DAMPs could lead to tissue death, organ failure, and septic shock (70, 71). While caspase-8 is known predominately as a mediator of apoptosis, it is also a master regulator of pyroptosis and necroptosis (72) and is capable of processing pro-IL-1β and pro-IL-18 into their functional cytokine forms (73–75). Caspase-8 can regulate necroptosis, unregulated cell death, by preventing the phosphorylation of MLKL into its active form, phospho-MLKL, by inactivating RIPK1 and RIPK3 by proteolytic cleavage (76–78).
Excessive inflammation is central to poor clinical outcomes in COVID-19, with data suggesting caspase-mediated inflammation being an important feature. Higher levels of active caspase-1 (Casp1p20) in the sera of COVID-19 patients are associated with severe disease and poor clinical outcomes (79). Caspase-1 activity is also upregulated in CD4+ T cells of COVID-19 patients that were hospitalized, those with liver disease, and long-haulers (43, 80). Human caspase-4 in infected individuals and its mouse homologue caspase-11 in SARS-CoV-2 murine models were recently found to be upregulated in lung tissue histologically and promote COVID-19-associated inflammation and coagulopathy (81). SARS-CoV-2 infection activates caspase-8, which triggers inflammatory cytokine processing of pro-IL-1β in lung epithelial cells and lung cells of SARS-CoV-2-infected HFH4-hACE2 transgenic mice (41). Inflammatory mediators IL-1β and IL-18, the main cytokine products of caspase-1 activation, are observed to be increased in the lungs and sera of patients with symptomatic COVID-19 compared to asymptomatic patients and healthy individuals (82–84). IL-18 levels are also shown to correlate with other inflammatory markers in SARS-CoV-2 individuals (83). Interestingly, IL-18 can contribute to the pathology of COVID-19 by altering MAIT cell function (85). In human monocytes, caspase-1 activation along with IL-1β production and pyroptosis is observed in both SARS-CoV-2 infected ex vivo and from infected ICU patients (86). RNH1 protein, an inhibitor of inflammasome activation through proteasome-mediated degradation of caspase-1, is increased in the blood and lung biopsies from individuals with COVID-19 and is negatively associated with SARS-CoV-2-mediated inflammation and adverse clinical outcomes (87). In vitro, SARS-CoV-2 infected human monocytes demonstrate pyroptotic activity, which was associated with caspase-1 activation, IL-1β production, GSDMD cleavage, and enhanced pro-inflammatory cytokine levels (86). High serum levels of lactate dehydrogenase (LDH), an indicator of pyroptosis, is also shown to associate with poor prognosis and the extent of lung damage and disease severity in individuals with COVID-19 (88–90) and has been proposed as a potentially useful marker for monitoring treatment response in COVID-19-associated pneumonia (91).
Mechanistically, SARS-CoV-2 N protein has been shown to promote NLRP3 inflammasome activation to induce caspase-mediated inflammatory milieu (IL-1β, IL-18) and pyroptotic cell death (88, 92–94). However, N protein is also shown to inhibit the cleavage of GSDMD by caspase-1 in monocytes in vitro (95). In previous studies of SARS-CoV, several accessory proteins have been shown to modulate inflammasome activation. The accessory protein ORF3a is shown to act as a K+ channel to induce NLRP3 inflammasome activation (96). However, another study indicates ORF3a can promote NLRP3 inflammasome activation by enhancing the ability of TNF receptor-associated factor 3 (TRAF3) in ubiquinating the inflammasome adapter ASC (97). In macrophages, SARS-CoV ORF8b was found to directly bind the LRR of NLRP3 inflammasomes to propagate caspase-1 activation (98). However, two SARS-CoV-2 NSPs, NSP1 and NSP13, are shown to inhibit NLRP3 inflammasome caspase-1-mediated IL-1β production in the monocytic cell line THP-1 (99). The E glycoprotein of SARS-CoV is also involved in inflammasome activation, as in mouse models show that viruses lacking E protein induced lower levels of inflammasome-activated IL-1β (100) by possessing calcium ion channel activity (101). Finally, many SARS-CoV encoded proteins are shown to induce NF-κB activation in vitro, including ORF3a, ORF7a, M, and N proteins (102, 103).
Therapeutic Potential of Targeting Caspase Pathways for COVID-19
The COVID-19 pandemic is going on its third year, and efforts are still converging globally to effectively distribute SARS-CoV-2 vaccinations. Global vaccination efforts have not proceeded at a similar pace worldwide and vaccine hesitancy persists in the public. Furthermore, the continuous evolution of SARS-CoV-2 could lead to new VOCs, such as the recently emerged and rapidly disseminating Omicron variant. These new VOCs could impact the efficacy of neutralizing antibodies, monoclonal or vaccine-induced, and exhibit potential for increased transmissibility, as observed with Omicron (104, 105). While vaccination and previous infection by SARS-CoV-2 so far have shown to provide protection, particularly regarding the prevention of serious disease and mortality, therapeutics are still an urgent need to attenuate severe disease and are highly investigated due to the persistent unvaccinated population, breakthrough cases, and the potential emergence of immunoevasive VOCs. Therapeutics recommended by the WHO for severe and critical COVID-19 mainly aim at disrupting the viral life cycle to limit the spread of infection, such as the use of neutralizing monoclonal antibodies (i.e. casirivimab) and the protease inhibitor Paxlovid, or to hinder the development of severe disease, including the use of systemic corticosteroids (i.e. dexamethasone). For the latter, targeting inflammatory innate immune pathways are a viable target, given the therapeutic promise of IL-6 receptor blockers, such as toclilizumab or sarilumab, in reducing severe outcomes in COVID-19 (106–109). Given the role of caspases in SARS-CoV-2, targeting related pathways could emerge as a potential therapeutic strategy that may benefit clinical efforts to prevent or ameliorate severe COVID-19.
Therapeutics for caspase-associated inflammation and cell death can be through the modulation of caspase activity directly, the targeting of upstream signaling complexes (i.e. inflammasomes), or the neutralization of caspase substrates (i.e. IL-1β). Regarding caspase targeting agents, the pan-caspase inhibitor Emricasan (EMR) was shown to attenuate caspase-1 hyperactivity in CD4+ T cells from COVID-19 patients ex vivo (43) and the caspase-8 inhibitor Z-IETD-FMK subdued SARS-CoV-2-induced BID cleavage and caspase-3 activation (41). However, direct caspase-1 inhibition did not affect SARS-CoV-2-induced IL-1β processing and secretion (41). Interestingly, several caspase inhibitors were shown to target the main protease of SARS-CoV-2 Mpro, including pan-caspase inhibition with Z-VAD(OMe)-FMK and discriminate inhibitors Z-DEVD-FMK and Z-IETD-FMK, for caspase-3 and caspase-8, respectively (110). Furthermore, among ~6,070 drugs screened, EMR was identified to inhibit the activity of Mpro in vitro and through computation screening shown to bind to ACE2 (111, 112). Nonetheless, while several targeted and indiscriminate caspase inhibitors have been identified and developed with intended therapeutic use, only few have advanced into clinical trials, and none are used clinically. However, therapeutics targeting the downstream effects of caspase-mediated inflammation and pyroptosis are making progress. The use of IL-1 receptor antagonist anakinra in COVID-19 patients showed significant decreases in oxygen requirements, increased duration without invasive mechanical ventilation, and decreases of fever and C-reactive protein, indicating early IL-1 receptor blockade could hold therapeutic value in acute hyperinflammatory respiratory failure (113). The anti-IL-1β antibody inhibitor canakinumab was also suggested as a viable therapeutic for COVID-19 patients (114); however, a recent clinical trial investigating its use showed that it did not significantly increase survival without invasive mechanical ventilation (115). NLRP3 inflammasome inhibition with MCC950 reduced lung inflammation and COVID-19-like pathology in human ACE2 transgenic mice infected with SARS-CoV-2 (116). Finally, Disulfiram, the GSDMD inhibitor that covalently modifies GSDMD to block pyroptotic pore formation, was shown to associate with a lower incidence of COVID-19 in a retrospective study (117).
Conclusion
This review highlights multiple caspases implicated in SARS-CoV-2 infection and disease severity. Although targeting caspases and related pathways may be a promising intervention, caspase signaling may still be paramount for functional and balanced immune activity against SARS-CoV-2 infection. Further understanding the roles caspase pathways play during the progression of infection and disease including PASC is crucial for further therapeutic development or the repurposing of drugs, combination therapies to curtail inflammation and cell death in COVID-19 and limit disease severity and death in all age and risk groups following SARS-CoV-2 infection.
Author Contributions
TP and LN drafted the manuscript. TP developed the graphical figures. All authors contributed to the discussion and approved the submitted version.
Conflict of Interest
Author OA was employed by company Amerimmune.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
Graphical figures were created with BioRender.com and Adobe Illustrator v25.2.
References
- 1. Gupta A, Madhavan MV, Sehgal K, Nair N, Mahajan S, Sehrawat TS, et al. Extrapulmonary Manifestations of COVID-19. Nat Med (2020) 26:1017–32. doi: 10.1038/s41591-020-0968-3 [DOI] [PubMed] [Google Scholar]
- 2. Ram-Mohan N, Kim D, Zudock EJ, Hashemi MM, Tjandra KC, Rogers AJ, et al. SARS-CoV-2 RNAemia Predicts Clinical Deterioration and Extrapulmonary Complications From COVID-19. Clin Infect Dis (2022) 74(2):218–26. doi: 10.1093/cid/ciab394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical Course and Risk Factors for Mortality of Adult Inpatients With COVID-19 in Wuhan, China: A Retrospective Cohort Study. Lancet (2020) 395:1054–62. doi: 10.1016/S0140-6736(20)30566-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Lopez-Leon S, Wegman-Ostrosky T, Perelman C, Sepulveda R, Rebolledo PA, Cuapio A, et al. More Than 50 Long-Term Effects of COVID-19: A Systematic Review and Meta-Analysis. Sci Rep (2021) 11:16144. doi: 10.1038/s41598-021-95565-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Nalbandian A, Sehgal K, Gupta A, Madhavan MV, McGroder C, Stevens JS, et al. Post-Acute COVID-19 Syndrome. Nat Med (2021) 27:601–15. doi: 10.1038/s41591-021-01283-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. O'Donnell JS, Chappell KJ. Chronic SARS-CoV-2, a Cause of Post-Acute COVID-19 Sequelae (Long-COVID)? Front Microbiol (2021) 12:724654. doi: 10.3389/fmicb.2021.724654 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Groff D, Sun A, Ssentongo AE, Ba DM, Parsons N, Poudel GR, et al. Short-Term and Long-Term Rates of Postacute Sequelae of SARS-CoV-2 Infection: A Systematic Review. JAMA Netw Open (2021) 4:e2128568. doi: 10.1001/jamanetworkopen.2021.28568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. COVID Research: A Year of Scientific Milestones. (2021). Available at: https://www.nature.com/articles/d41586-020-00502-w (Accessed February 10, 2022). [DOI] [PubMed] [Google Scholar]
- 9. Jayk Bernal A, Gomes da Silva MM, Musungaie DB, Kovalchuk E, Gonzalez A, Delos Reyes V, et al. Molnupiravir for Oral Treatment of Covid-19 in Nonhospitalized Patients. N Engl J Med (2022) 386:509–20. doi: 10.1056/NEJMoa2116044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Health, N. I. O . COVID-19 Treatment Guidelines. (2021). Available at: https://www.covid19treatmentguidelines.nih.gov/about-the-guidelines/whats-new/.
- 11. Ledford H. COVID Antiviral Pills: What Scientists Still Want to Know. Nature (2021) 599:358–9. doi: 10.1038/d41586-021-03074-5 [DOI] [PubMed] [Google Scholar]
- 12. Alnemri ES, Livingston DJ, Nicholson DW, Salvesen G, Thornberry NA, Wong WW, et al. Human ICE/CED-3 Protease Nomenclature. Cell (1996) 87:171. doi: 10.1016/s0092-8674(00)81334-3 [DOI] [PubMed] [Google Scholar]
- 13. Man SM, Kanneganti TD. Converging Roles of Caspases in Inflammasome Activation, Cell Death and Innate Immunity. Nat Rev Immunol (2016) 16:7–21. doi: 10.1038/nri.2015.7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Lamkanfi M, Declercq W, Kalai M, Saelens X, Vandenabeele P. Alice in Caspase Land. A Phylogenetic Analysis of Caspases From Worm to Man. Cell Death Differ (2002) 9:358–61. doi: 10.1038/sj.cdd.4400989 [DOI] [PubMed] [Google Scholar]
- 15. Becker RC. COVID-19 Update: Covid-19-Associated Coagulopathy. J Thromb Thrombolysis (2020) 50:54–67. doi: 10.1007/s11239-020-02134-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Kesavardhana S, Kanneganti TD. Mechanisms Governing Inflammasome Activation, Assembly and Pyroptosis Induction. Int Immunol (2017) 29:201–10. doi: 10.1093/intimm/dxx018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Man SM, Karki R, Kanneganti TD. Molecular Mechanisms and Functions of Pyroptosis, Inflammatory Caspases and Inflammasomes in Infectious Diseases. Immunol Rev (2017) 277:61–75. doi: 10.1111/imr.12534 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Denecker G, Ovaere P, Vandenabeele P, Declercq W. Caspase-14 Reveals Its Secrets. J Cell Biol (2008) 180:451–8. doi: 10.1083/jcb.200709098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Lim Y, Dorstyn L, Kumar S. The P53-Caspase-2 Axis in the Cell Cycle and DNA Damage Response. Exp Mol Med (2021) 53:517–27. doi: 10.1038/s12276-021-00590-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Hoffmann M, Kleine-Weber H, Schroeder S, Kruger N, Herrler T, Erichsen S, et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell (2020) 181:271–80.e278. doi: 10.1016/j.cell.2020.02.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, Veesler D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell (2020) 181:281–92.e286. doi: 10.1016/j.cell.2020.02.058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Wang K, Chen W, Zhang Z, Deng Y, Lian JQ, Du P, et al. CD147-Spike Protein Is a Novel Route for SARS-CoV-2 Infection to Host Cells. Signal Transduct Target Ther (2020) 5:283. doi: 10.1038/s41392-020-00426-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Perez-Zsolt D, Munoz-Basagoiti J, Rodon J, Elosua-Bayes M, Raich-Regue D, Risco C, et al. SARS-CoV-2 Interaction With Siglec-1 Mediates Trans-Infection by Dendritic Cells. Cell Mol Immunol (2021) 18:2676–8. doi: 10.1038/s41423-021-00794-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Cantuti-Castelvetri L, Ojha R, Pedro LD, Djannatian M, Franz J, Kuivanen S, et al. Neuropilin-1 Facilitates SARS-CoV-2 Cell Entry and Infectivity. Science (2020) 370:856–60. doi: 10.1126/science.abd2985 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Clausen TM, Sandoval DR, Spliid CB, Pihl J, Perrett HR, Painter CD, et al. SARS-CoV-2 Infection Depends on Cellular Heparan Sulfate and ACE2. Cell (2020) 183:1043–57.e1015. doi: 10.1016/j.cell.2020.09.033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Kasuga Y, Zhu B, Jang KJ, Yoo JS. Innate Immune Sensing of Coronavirus and Viral Evasion Strategies. Exp Mol Med (2021) 53:723–36. doi: 10.1038/s12276-021-00602-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Zheng M, Karki R, Williams EP, Yang D, Fitzpatrick E, Vogel P, et al. TLR2 Senses the SARS-CoV-2 Envelope Protein to Produce Inflammatory Cytokines. Nat Immunol (2021) 22:829–38. doi: 10.1038/s41590-021-00937-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Khan SS, Shafiei M, Longoria C, Schoggins J, Savani RC, Zaki H. SARS-CoV-2 Spike Protein Induces Inflammation via TLR2-Dependent Activation of the NF-kB Pathway. eLife (2021) 10:e68563. doi: 10.1101/2021.03.16.435700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Terpos E, Ntanasis-Stathopoulos I, Elalamy I, Kastritis E, Sergentanis TN, Politou M, et al. Hematological Findings and Complications of COVID-19. Am J Hematol (2020) 95:834–47. doi: 10.1002/ajh.25829 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Yap JKY, Moriyama M, Iwasaki A. Inflammasomes and Pyroptosis as Therapeutic Targets for COVID-19. J Immunol (2020) 205:307–12. doi: 10.4049/jimmunol.2000513 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Coronaviridae Study Group of the International Committee on Taxonomy of V . The Species Severe Acute Respiratory Syndrome-Related Coronavirus: Classifying 2019-Ncov and Naming It SARS-CoV-2. Nat Microbiol (2020) 5:536–44. doi: 10.1038/s41564-020-0695-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Redondo N, Zaldivar-Lopez S, Garrido JJ, Montoya M. SARS-CoV-2 Accessory Proteins in Viral Pathogenesis: Knowns and Unknowns. Front Immunol (2021) 12:708264. doi: 10.3389/fimmu.2021.708264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Boatright KM, Renatus M, Scott FL, Sperandio S, Shin H, Pedersen IM, et al. A Unified Model for Apical Caspase Activation. Mol Cell (2003) 11:529–41. doi: 10.1016/s1097-2765(03)00051-0 [DOI] [PubMed] [Google Scholar]
- 34. Acehan D, Jiang X, Morgan DG, Heuser JE, Wang X, Akey CW. Three-Dimensional Structure of the Apoptosome: Implications for Assembly, Procaspase-9 Binding, and Activation. Mol Cell (2002) 9:423–32. doi: 10.1016/s1097-2765(02)00442-2 [DOI] [PubMed] [Google Scholar]
- 35. Kischkel FC, Hellbardt S, Behrmann I, Germer M, Pawlita M, Krammer PH, et al. Cytotoxicity-Dependent APO-1 (Fas/CD95)-Associated Proteins Form a Death-Inducing Signaling Complex (DISC) With the Receptor. EMBO J (1995) 14:5579–88. doi: 10.1002/j.1460-2075.1995.tb00245.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Wachmann K, Pop C, van Raam BJ, Drag M, Mace PD, Snipas SJ, et al. Activation and Specificity of Human Caspase-10. Biochemistry (2010) 49:8307–15. doi: 10.1021/bi100968m [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Wang Y, Tjandra N. Structural Insights of Tbid, the Caspase-8-Activated Bid, and Its BH3 Domain. J Biol Chem (2013) 288:35840–51. doi: 10.1074/jbc.M113.503680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Cullen SP, Henry CM, Kearney CJ, Logue SE, Feoktistova M, Tynan GA, et al. Fas/CD95-Induced Chemokines can Serve as "Find-Me" Signals for Apoptotic Cells. Mol Cell (2013) 49:1034–48. doi: 10.1016/j.molcel.2013.01.025 [DOI] [PubMed] [Google Scholar]
- 39. Martin SJ, Henry CM, Cullen SP. A Perspective on Mammalian Caspases as Positive and Negative Regulators of Inflammation. Mol Cell (2012) 46:387–97. doi: 10.1016/j.molcel.2012.04.026 [DOI] [PubMed] [Google Scholar]
- 40. Benedict CA, Norris PS, Ware CF. To Kill or be Killed: Viral Evasion of Apoptosis. Nat Immunol (2002) 3:1013–8. doi: 10.1038/ni1102-1013 [DOI] [PubMed] [Google Scholar]
- 41. Alijotas-Reig J, Esteve-Valverde E, Belizna C, Selva-O'Callaghan A, Pardos-Gea J, Quintana A, et al. Immunomodulatory Therapy for the Management of Severe COVID-19. Beyond the Anti-Viral Therapy: A Comprehensive Review. Autoimmun Rev (2020) 19:102569. doi: 10.1016/j.autrev.2020.102569 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. McMahon CL, Staples H, Gazi M, Carrion R, Hsieh J. SARS-CoV-2 Targets Glial Cells in Human Cortical Organoids. Stem Cell Rep (2021) 16:1156–64. doi: 10.1016/j.stemcr.2021.01.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Plassmeyer M, Alpan O, Corley MJ, Premeaux TA, Lillard K, Coatney P, et al. Caspases and Therapeutic Potential of Caspase Inhibitors in Moderate-Severe SARS CoV2 Infection and Long COVID. Allergy (2022) 77(1):118–29. doi: 10.1111/all.14907 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Koupenova M, Corkrey HA, Vitseva O, Tanriverdi K, Somasundaran M, Liu P, et al. SARS-CoV-2 Initiates Programmed Cell Death in Platelets. Circ Res (2021) 129:631–46. doi: 10.1161/CIRCRESAHA.121.319117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Malas MB, Naazie IN, Elsayed N, Mathlouthi A, Marmor R, Clary B. Thromboembolism Risk of COVID-19 Is High and Associated With a Higher Risk of Mortality: A Systematic Review and Meta-Analysis. EClinicalMedicine (2020) 29:100639. doi: 10.1016/j.eclinm.2020.100639 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Ren Y, Shu T, Wu D, Mu J, Wang C, Huang M, et al. The ORF3a Protein of SARS-CoV-2 Induces Apoptosis in Cells. Cell Mol Immunol (2020) 17:881–3. doi: 10.1038/s41423-020-0485-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Padhan K, Tanwar C, Hussain A, Hui PY, Lee MY, Cheung CY, et al. Severe Acute Respiratory Syndrome Coronavirus Orf3a Protein Interacts With Caveolin. J Gen Virol (2007) 88:3067–77. doi: 10.1099/vir.0.82856-0 [DOI] [PubMed] [Google Scholar]
- 48. Chan CM, Tsoi H, Chan WM, Zhai S, Wong CO, Yao X, et al. The Ion Channel Activity of the SARS-Coronavirus 3a Protein Is Linked to Its Pro-Apoptotic Function. Int J Biochem Cell Biol (2009) 41:2232–9. doi: 10.1016/j.biocel.2009.04.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Law PTW, Wong CH, Au TCC, Chuck CP, Kong SK, Chan PKS, et al. The 3a Protein of Severe Acute Respiratory Syndrome-Associated Coronavirus Induces Apoptosis in Vero E6 Cells. J Gen Virol (2005) 86:1921–30. doi: 10.1099/vir.0.80813-0 [DOI] [PubMed] [Google Scholar]
- 50. Ye Z, Wong CK, Li P, Xie Y. A SARS-CoV Protein, ORF-6, Induces Caspase-3 Mediated, ER Stress and JNK-Dependent Apoptosis. Biochim Biophys Acta (2008) 1780:1383–7. doi: 10.1016/j.bbagen.2008.07.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Chen CY, Ping YH, Lee HC, Chen KH, Lee YM, Chan YJ, et al. Open Reading Frame 8a of the Human Severe Acute Respiratory Syndrome Coronavirus Not Only Promotes Viral Replication But Also Induces Apoptosis. J Infect Dis (2007) 196:405–15. doi: 10.1086/519166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Tan YX, Tan TH, Lee MJ, Tham PY, Gunalan V, Druce J, et al. Induction of Apoptosis by the Severe Acute Respiratory Syndrome Coronavirus 7a Protein Is Dependent on Its Interaction With the Bcl-XL Protein. J Virol (2007) 81:6346–55. doi: 10.1128/JVI.00090-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Tan YJ, Fielding BC, Goh PY, Shen S, Tan TH, Lim SG, et al. Overexpression of 7a, a Protein Specifically Encoded by the Severe Acute Respiratory Syndrome Coronavirus, Induces Apoptosis via a Caspase-Dependent Pathway. J Virol (2004) 78:14043–7. doi: 10.1128/JVI.78.24.14043-14047.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Ren Y, Wang A, Fang Y, Shu T, Wu D, Wang C, et al. SARS-CoV-2 Membrane Glycoprotein M Triggers Apoptosis With the Assistance of Nucleocapsid Protein N in Cells. Front Cell Infect Microbiol (2021) 11:706252. doi: 10.3389/fcimb.2021.706252 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Man SM, Hopkins LJ, Nugent E, Cox S, Gluck IM, Tourlomousis P, et al. Inflammasome Activation Causes Dual Recruitment of NLRC4 and NLRP3 to the Same Macromolecular Complex. Proc Natl Acad Sci USA (2014) 111:7403–8. doi: 10.1073/pnas.1402911111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Broz P, Dixit VM. Inflammasomes: Mechanism of Assembly, Regulation and Signalling. Nat Rev Immunol (2016) 16:407–20. doi: 10.1038/nri.2016.58 [DOI] [PubMed] [Google Scholar]
- 57. Akdis M, Aab A, Altunbulakli C, Azkur K, Costa RA, Crameri R, et al. Interleukins (From IL-1 to IL-38), Interferons, Transforming Growth Factor Beta, and TNF-Alpha: Receptors, Functions, and Roles in Diseases. J Allergy Clin Immunol (2016) 138:984–1010. doi: 10.1016/j.jaci.2016.06.033 [DOI] [PubMed] [Google Scholar]
- 58. Arend WP, Palmer G, Gabay C. IL-1, IL-18, and IL-33 Families of Cytokines. Immunol Rev (2008) 223:20–38. doi: 10.1111/j.1600-065X.2008.00624.x [DOI] [PubMed] [Google Scholar]
- 59. He Y, Hara H, Nunez G. Mechanism and Regulation of NLRP3 Inflammasome Activation. Trends Biochem Sci (2016) 41:1012–21. doi: 10.1016/j.tibs.2016.09.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Latz E, Xiao TS, Stutz A. Activation and Regulation of the Inflammasomes. Nat Rev Immunol (2013) 13:397–411. doi: 10.1038/nri3452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Vigano E, Diamond CE, Spreafico R, Balachander A, Sobota RM, Mortellaro A. Human Caspase-4 and Caspase-5 Regulate the One-Step Non-Canonical Inflammasome Activation in Monocytes. Nat Commun (2015) 6:8761. doi: 10.1038/ncomms9761 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Liu X, Zhang Z, Ruan J, Pan Y, Magupalli VG, Wu H, et al. Inflammasome-Activated Gasdermin D Causes Pyroptosis by Forming Membrane Pores. Nature (2016) 535:153–8. doi: 10.1038/nature18629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Vande Walle L, Lamkanfi M. Pyroptosis. Curr Biol (2016) 26:R568–72. doi: 10.1016/j.cub.2016.02.019 [DOI] [PubMed] [Google Scholar]
- 64. Shi J, Zhao Y, Wang K, Shi X, Wang Y, Huang H, et al. Cleavage of GSDMD by Inflammatory Caspases Determines Pyroptotic Cell Death. Nature (2015) 526:660–5. doi: 10.1038/nature15514 [DOI] [PubMed] [Google Scholar]
- 65. Lamkanfi M, Sarkar A, Vande Walle L, Vitari AC, Amer AO, Wewers MD, et al. Inflammasome-Dependent Release of the Alarmin HMGB1 in Endotoxemia. J Immunol (2010) 185:4385–92. doi: 10.4049/jimmunol.1000803 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Kayagaki N, Stowe IB, Lee BL, O'Rourke K, Anderson K, Warming S, et al. Caspase-11 Cleaves Gasdermin D for Non-Canonical Inflammasome Signalling. Nature (2015) 526:666–71. doi: 10.1038/nature15541 [DOI] [PubMed] [Google Scholar]
- 67. He WT, Wan H, Hu L, Chen P, Wang X, Huang Z, et al. Gasdermin D Is an Executor of Pyroptosis and Required for Interleukin-1beta Secretion. Cell Res (2015) 25:1285–98. doi: 10.1038/cr.2015.139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Sutterwala FS, Haasken S, Cassel SL. Mechanism of NLRP3 Inflammasome Activation. Ann NY Acad Sci (2014) 1319:82–95. doi: 10.1111/nyas.12458 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Moretti J, Blander JM. Increasing Complexity of NLRP3 Inflammasome Regulation. J Leukoc Biol (2021) 109(3):561–71. doi: 10.1002/JLB.3MR0520-104RR [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Bergsbaken T, Fink SL, Cookson BT. Pyroptosis: Host Cell Death and Inflammation. Nat Rev Microbiol (2009) 7:99–109. doi: 10.1038/nrmicro2070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Aglietti RA, Dueber EC. Recent Insights Into the Molecular Mechanisms Underlying Pyroptosis and Gasdermin Family Functions. Trends Immunol (2017) 38:261–71. doi: 10.1016/j.it.2017.01.003 [DOI] [PubMed] [Google Scholar]
- 72. Amaral MP, Bortoluci KR. Caspase-8 and FADD: Where Cell Death and Inflammation Collide. Immunity (2020) 52:890–2. doi: 10.1016/j.immuni.2020.05.008 [DOI] [PubMed] [Google Scholar]
- 73. Mahib MR, Hosojima S, Kushiyama H, Kinoshita T, Shiroishi T, Suda T, et al. Caspase-7 Mediates Caspase-1-Induced Apoptosis Independently of Bid. Microbiol Immunol (2020) 64:143–52. doi: 10.1111/1348-0421.12756 [DOI] [PubMed] [Google Scholar]
- 74. Tsuchiya K, Nakajima S, Hosojima S, Thi Nguyen D, Hattori T, Manh Le T, et al. Caspase-1 Initiates Apoptosis in the Absence of Gasdermin D. Nat Commun (2019) 10:2091. doi: 10.1038/s41467-019-09753-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Tummers B, Green DR. Caspase-8: Regulating Life and Death. Immunol Rev (2017) 277:76–89. doi: 10.1111/imr.12541 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Vanlangenakker N, Bertrand MJ, Bogaert P, Vandenabeele P, Vanden Berghe T. TNF-Induced Necroptosis in L929 Cells Is Tightly Regulated by Multiple TNFR1 Complex I and II Members. Cell Death Dis (2011) 2:e230. doi: 10.1038/cddis.2011.111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Sun L, Wang H, Wang Z, He S, Chen S, Liao D, et al. Mixed Lineage Kinase Domain-Like Protein Mediates Necrosis Signaling Downstream of RIP3 Kinase. Cell (2012) 148:213–27. doi: 10.1016/j.cell.2011.11.031 [DOI] [PubMed] [Google Scholar]
- 78. Li J, McQuade T, Siemer AB, Napetschnig J, Moriwaki K, Hsiao YS, et al. The RIP1/RIP3 Necrosome Forms a Functional Amyloid Signaling Complex Required for Programmed Necrosis. Cell (2012) 150:339–50. doi: 10.1016/j.cell.2012.06.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Rodrigues TS, de Sa KSG, Ishimoto AY, Becerra A, Oliveira S, Almeida L, et al. Inflammasomes Are Activated in Response to SARS-CoV-2 Infection and Are Associated With COVID-19 Severity in Patients. J Exp Med (2021) 218(3):e20201707. doi: 10.1084/jem.20201707 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Kroemer A, Khan K, Plassmeyer M, Alpan O, Haseeb MA, Gupta R, et al. Inflammasome Activation and Pyroptosis in Lymphopenic Liver Patients With COVID-19. J Hepatol (2020) 73:1258–62. doi: 10.1016/j.jhep.2020.06.034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Eltobgy M, Zani ,A, Kenney AD, Estfanous S, Kim E, Badr A, et al. Caspase-4/11 Exacerbates Disease Severity in SARS-CoV-2 Infection by Promoting Inflammation and Thrombosis. bioRxiv (2021). doi: 10.1101/2021.09.24.461743 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Zhou Z, Ren L, Zhang L, Zhong J, Xiao Y, Jia Z, et al. Heightened Innate Immune Responses in the Respiratory Tract of COVID-19 Patients. Cell Host Microbe (2020) 27:883–890 e882. doi: 10.1016/j.chom.2020.04.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Satis H, Ozger HS, Aysert Yildiz P, Hizel K, Gulbahar O, Erbas G, et al. Prognostic Value of Interleukin-18 and Its Association With Other Inflammatory Markers and Disease Severity in COVID-19. Cytokine (2021) 137:155302. doi: 10.1016/j.cyto.2020.155302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Tjan LH, Furukawa K, Nagano T, Kiriu T, Nishimura M, Arii J, et al. Early Differences in Cytokine Production by Severity of Coronavirus Disease 2019. J Infect Dis (2021) 223:1145–9. doi: 10.1093/infdis/jiab005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Flament H, Rouland M, Beaudoin L, Toubal A, Bertrand L, Lebourgeois S, et al. Outcome of SARS-CoV-2 Infection Is Linked to MAIT Cell Activation and Cytotoxicity. Nat Immunol (2021) 22:322–35. doi: 10.1038/s41590-021-00870-z [DOI] [PubMed] [Google Scholar]
- 86. Ferreira AC, Soares VC, de Azevedo-Quintanilha IG, Dias S, Fintelman-Rodrigues N, Sacramento CQ, et al. SARS-CoV-2 Engages Inflammasome and Pyroptosis in Human Primary Monocytes. Cell Death Discov (2021) 7:43. doi: 10.1038/s41420-021-00428-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Bombaci G, Sarangdhar MA, Andina N, Tardivel A, Yu EC, Mackie GM, et al. LRR Protein RNH1 Dampens the Inflammasome Activation and Is Associated With Adverse Clinical Outcomes in COVID-19 Patients. bioRxiv (2021). doi: 10.1101/2021.04.12.438219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Han Y, Zhang H, Mu S, Wei W, Jin C, Tong C, et al. Lactate Dehydrogenase, an Independent Risk Factor of Severe COVID-19 Patients: A Retrospective and Observational Study. Aging (Albany NY) (2020) 12:11245–58. doi: 10.18632/aging.103372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med (2020) 382:1708–20. doi: 10.1056/NEJMoa2002032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Martha JW, Wibowo A, Pranata R. Prognostic Value of Elevated Lactate Dehydrogenase in Patients With COVID-19: A Systematic Review and Meta-Analysis. Postgrad Med J (2021) 1–6. doi: 10.1136/postgradmedj-2020-139542 [DOI] [PubMed] [Google Scholar]
- 91. Wu MY, Yao L, Wang Y, Zhu XY, Wang XF, Tang PJ, et al. Clinical Evaluation of Potential Usefulness of Serum Lactate Dehydrogenase (LDH) in 2019 Novel Coronavirus (COVID-19) Pneumonia. Respir Res (2020) 21:171. doi: 10.1186/s12931-020-01427-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Pan P, Shen M, Yu Z, Ge W, Chen K, Tian M, et al. SARS-CoV-2 N Protein Promotes NLRP3 Inflammasome Activation to Induce Hyperinflammation. Nat Commun (2021) 12:4664. doi: 10.1038/s41467-021-25015-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical Features of Patients Infected With 2019 Novel Coronavirus in Wuhan, China. Lancet (2020) 395:497–506. doi: 10.1016/S0140-6736(20)30183-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Chen G, Wu D, Guo W, Cao Y, Huang D, Wang H, et al. Clinical and Immunological Features of Severe and Moderate Coronavirus Disease 2019. J Clin Invest (2020) 130:2620–9. doi: 10.1172/JCI137244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Ma J, Zhu F, Zhao M, Shao F, Yu D, Ma J, et al. SARS-CoV-2 Nucleocapsid Suppresses Host Pyroptosis by Blocking Gasdermin D Cleavage. EMBO J (2021) 40:e108249. doi: 10.15252/embj.2021108249 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Chen IY, Moriyama M, Chang MF, Ichinohe T. Severe Acute Respiratory Syndrome Coronavirus Viroporin 3a Activates the NLRP3 Inflammasome. Front Microbiol (2019) 10:50. doi: 10.3389/fmicb.2019.00050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Siu KL, Yuen KS, Castano-Rodriguez C, Ye ZW, Yeung ML, Fung SY, et al. Severe Acute Respiratory Syndrome Coronavirus ORF3a Protein Activates the NLRP3 Inflammasome by Promoting TRAF3-Dependent Ubiquitination of ASC. FASEB J (2019) 33:8865–77. doi: 10.1096/fj.201802418R [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Shi CS, Nabar NR, Huang NN, Kehrl JH. SARS-Coronavirus Open Reading Frame-8b Triggers Intracellular Stress Pathways and Activates NLRP3 Inflammasomes. Cell Death Discov (2019) 5:101. doi: 10.1038/s41420-019-0181-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Kim NE, Kim DK, Song YJ. SARS-CoV-2 Nonstructural Proteins 1 and 13 Suppress Caspase-1 and the NLRP3 Inflammasome Activation. Microorganisms (2021) 9(3):494. doi: 10.3390/microorganisms9030494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Nieto-Torres JL, DeDiego ML, Verdia-Baguena C, Jimenez-Guardeno JM, Regla-Nava JA, Fernandez-Delgado R, et al. Severe Acute Respiratory Syndrome Coronavirus Envelope Protein Ion Channel Activity Promotes Virus Fitness and Pathogenesis. PloS Pathog (2014) 10:e1004077. doi: 10.1371/journal.ppat.1004077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Nieto-Torres JL, Verdia-Baguena C, Jimenez-Guardeno JM, Regla-Nava JA, Castano-Rodriguez C, Fernandez-Delgado R, et al. Severe Acute Respiratory Syndrome Coronavirus E Protein Transports Calcium Ions and Activates the NLRP3 Inflammasome. Virology (2015) 485:330–9. doi: 10.1016/j.virol.2015.08.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Wu Y, Ma L, Cai S, Zhuang Z, Zhao Z, Jin S, et al. RNA-Induced Liquid Phase Separation of SARS-CoV-2 Nucleocapsid Protein Facilitates NF-kappaB Hyper-Activation and Inflammation. Signal Transduct Target Ther (2021) 6:167. doi: 10.1038/s41392-021-00575-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Su CM, Wang L, Yoo D. Activation of NF-kappaB and Induction of Proinflammatory Cytokine Expressions Mediated by ORF7a Protein of SARS-CoV-2. Sci Rep (2021) 11:13464. doi: 10.1038/s41598-021-92941-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. VanBlargan LA, Errico JM, Halfmann PJ, Zost SJ, Crowe JE, Jr, Purcell LA, et al. An Infectious SARS-CoV-2 B.1.1.529 Omicron Virus Escapes Neutralization by Therapeutic Monoclonal Antibodies. Nat Med (2022), 1–6. doi: 10.1038/s41591-021-01678-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Schmidt F, Muecksch F, Weisblum Y, Da Silva J, Bednarski E, Cho A, et al. Plasma Neutralization of the SARS-CoV-2 Omicron Variant. N Engl J Med (2022) 386:599–601. doi: 10.1056/NEJMc2119641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Group RC, Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, et al. Dexamethasone in Hospitalized Patients With Covid-19 - Preliminary Report. N Engl J Med (2021) 384:693–704. doi: 10.1056/NEJMoa2021436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Xu X, Han M, Li T, Sun W, Wang D, Fu B, et al. Effective Treatment of Severe COVID-19 Patients With Tocilizumab. Proc Natl Acad Sci USA (2020) 117:10970–5. doi: 10.1073/pnas.2005615117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Spinner CD, Gottlieb RL, Criner GJ, Arribas Lopez JR, Cattelan AM, Soriano Viladomiu A, et al. Effect of Remdesivir vs Standard Care on Clinical Status at 11 Days in Patients With Moderate COVID-19: A Randomized Clinical Trial. JAMA (2020) 324:1048–57. doi: 10.1001/jama.2020.16349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. White KM, Rosales R, Yildiz S, Kehrer T, Miorin L, Moreno E, et al. Plitidepsin has Potent Preclinical Efficacy Against SARS-CoV-2 by Targeting the Host Protein Eef1a. Science (2021) 371(6532):926–31. doi: 10.1126/science.abf4058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Wang Z, Zhao Y, Wang Q, Xing Y, Feng L, Kong J, et al. Identification of Proteasome and Caspase Inhibitors Targeting SARS-CoV-2 M(pro). Signal Transduct Target Ther (2021) 6:214. doi: 10.1038/s41392-021-00639-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Baker JD, Uhrich RL, Kraemer GC, Love JE, Kraemer BC. A Drug Repurposing Screen Identifies Hepatitis C Antivirals as Inhibitors of the SARS-CoV-2 Main 1 Protease. PLoS ONE (2020) 16(2):e0245962. doi: 10.1101/2020.07.10.197889 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Kim J, Zhang J, Cha Y, Kolitz S, Funt J, Escalante Chong R, et al. Advanced Bioinformatics Rapidly Identifies Existing Therapeutics for Patients With Coronavirus Disease-2019 (COVID-19). J Transl Med (2020) 18:257. doi: 10.1186/s12967-020-02430-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Cauchois R, Koubi M, Delarbre D, Manet C, Carvelli J, Blasco VB, et al. Early IL-1 Receptor Blockade in Severe Inflammatory Respiratory Failure Complicating COVID-19. Proc Natl Acad Sci USA (2020) 117:18951–3. doi: 10.1073/pnas.2009017117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Landi L, Ravaglia C, Russo E, Cataleta P, Fusari M, Boschi A, et al. Blockage of Interleukin-1beta With Canakinumab in Patients With Covid-19. Sci Rep (2020) 10:21775. doi: 10.1038/s41598-020-78492-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Caricchio R, Abbate A, Gordeev I, Meng J, Hsue PY, Neogi T, et al. Effect of Canakinumab vs Placebo on Survival Without Invasive Mechanical Ventilation in Patients Hospitalized With Severe COVID-19: A Randomized Clinical Trial. JAMA (2021) 326:230–9. doi: 10.1001/jama.2021.9508 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Zeng J, Xie X, Feng XL, Xu L, Han JB, Yu D, et al. Specific Inhibition of the NLRP3 Inflammasome Suppresses Immune Overactivation and Alleviates COVID-19 Like Pathology in Mice. EBioMedicine (2022) 75:103803. doi: 10.1016/j.ebiom.2021.103803 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Fillmore N, Bell S, Shen C, Nguyen V, La J, Dubreuil M, et al. Disulfiram Use Is Associated With Lower Risk of COVID-19: A Retrospective Cohort Study. PloS One (2021) 16:e0259061. doi: 10.1371/journal.pone.0259061 [DOI] [PMC free article] [PubMed] [Google Scholar]