Abstract
Dittrichia viscosa is a perennial Mediterranean plant used in traditional medicine for “calming purposes”, pointing at a possible antidepressant activity of the plant. We conducted chromatographic and bioassay-guided fractionation of D. viscosa root extract to isolate a specific fraction (fraction “K”) with antidepressant-like characteristics in vivo and strong antioxidant properties in vitro. A single dose of “K” reduced immobility time in the forced swim test with a mouse model possessing a depressive-like phenotype. Neurochemical profiling for 5-hydroxytryptamine (5-HT) and its primary metabolite, 5-hydroxyindoleacetic acid (5-HIAA), in prefrontal cortex and hippocampus of “K”-treated mice showed reduction in 5-HIAA, indicative of either serotonin uptake transporter or monoamine oxidase-A inhibition, as well as slight increases in 5-HT content. These neurochemical alterations, as well as the behavioral changes observed, were comparable to the effects of paroxetine. “K” also protected PC12 cells in a H2O2 cytotoxicity assay, thus demonstrating antioxidant properties, yet paroxetine augmented oxidative damage and cell death. Identification of the main compounds in “K” by high-performance liquid chromatography-tandem mass spectrometry (HPLC–MS/MS) indicated that chlorogenic acid and cynarine comprised 87% of the total components. D. viscosa root extract appears to produce antidepressant and cytoprotective effects and may serve as an attractive alternative to standard therapies for depression.
Subject terms: Biochemistry, Drug discovery, Neuroscience, Psychology
Introduction
The global prevalence of depressive disorders is rapidly growing1 and is a leading cause of disability2. However, treatment strategies targeted to depression have a high failure rate and remain limited by their side effects, slow response, and low efficacy3,4. Thus, there is a real need for the development of novel, safe agents with high pharmacological efficacy.
It is estimated that around eighty percent of the global population relies on herbal medicinal products as a primary source of healthcare5. A surge in acceptance and public interest in natural therapies both in developing and developed countries contributes to the development of novel pharmacologically active compounds derived directly or indirectly from plants. In the search for new drug entities, remarkable efforts are spent on chromatographic separation and mass spectrometry techniques allowing single-plant secondary metabolites to be efficiently isolated from their native hosts and their chemical structures to be unequivocally determined. Subsequent bioassay-guided fractionation of plant extracts and high-throughput activity screening using natural product libraries may result in the discovery of a variety of therapeutic agents, many of which are widely used in the clinic today6. Standardization of extracts, their fractions, or single active molecules is another vital issue ensuring qualitative and quantitative values, efficacy, and reproducibility of the pharmacological effect7.
Numerous plant natural products have successfully entered the clinic as complementary and alternative medicines for the treatment of psychiatric disorders, for example, crude and standardized extracts of Hypericum perforatum L.8, Valeriana officinalis L.9, Melissa officinalis L., and Verbena officinalis L.10. Notably, many plant-originated drugs were discovered through their use in traditional medicine. Ethnobotanical research, followed by the use of advanced technological tools, molecular and cellular methodology, and valid animal models, serves as a platform for the development of novel active compounds11,12.
Dittrichia viscosa (L.) Greuter (Asteraceae), commonly known as False Yellowhead or Woody Fleabane, is a herbaceous perennial Mediterranean plant species13 formerly belonging to the genus Inula14. According to ethnopharmacological surveys and reviews of medicinal herbs in Israel, D. viscosa is commonly used in Arabic traditional medicine15–18. The traditional uses of D. viscosa [Arabic name, tayun (sticky) davik] are vast including treatment of skin diseases, rheumatic pains, prevention of infections, reduction of high blood pressure, reduction of high sugar levels, infertility, and other uses18,19. Among different traditional applications of D. viscosa, it is popularly used for “calming purposes”, muscle relaxation, analgesia, and fatigue treatment15,17,18,20. Fatigue is a residual symptom of depression21,22 and many commercially-available antidepressants possess relaxing and analgesic properties23,24, suggesting a possible antidepressant-like potential in D. viscosa.
In this study, we aimed to determine whether an extract of D. viscosa root does indeed have the properties ascribed to it in the ethnobotanical literature, namely antidepressant effects. Further, we aimed to perform a chromatographic separation and bioassay-guided fractionation of the extract, selecting for fractions with increased pharmacological properties, as an initial effort to identify plant compounds of pharmaceutical potential matching the reported effects. We identified one fraction (named fraction “K”) which exhibited cytoprotective qualities as well as antidepressant-like effects in a mouse model with depressive-like phenotype developed in our laboratory25,26. Fraction “K” was evaluated by mass spectral analysis for bioactive compounds and two prominent chemicals were identified: chlorogenic acid and cynarine. Here, we present evidence for the pharmacological potential of D. viscosa root extract as an antidepressant natural product which contains at least two compounds of known pharmacology. Ongoing work in our lab is focused on identifying other molecules of therapeutic potential in fraction “K” of D. viscosa root.
Results
Crude extract did not exert a toxic effect in PC12 cells but failed to protect against oxidative stress
PC12 cells incubated with a broad range (250–5000 μg/mL) of crude root extract concentrations did not present with cytotoxic or proliferative effects (Dunnett’s test: p > 0.05; Fig. 1). However, pretreatment with the crude root extract failed to protect PC12 cells against 400 μM H2O2-induced cytotoxicity and concentration-dependently reduced cell viability by 72.6% (Dunnett’s test [1000–5000 μg/mL]: p < 0.0001). Two-way ANOVA: Crude root extract concentration: F[7,144] = 38.4, p < 0.0001; H2O2 treatment: F[1,144] = 4962, p < 0.0001; interaction: F[7,144] = 26.99, p < 0.0001). Effects of crude root extract with or without the presence of H2O2 differed (Sidak test [A vs. B by concentration], p < 0.0001).
Figure 1.
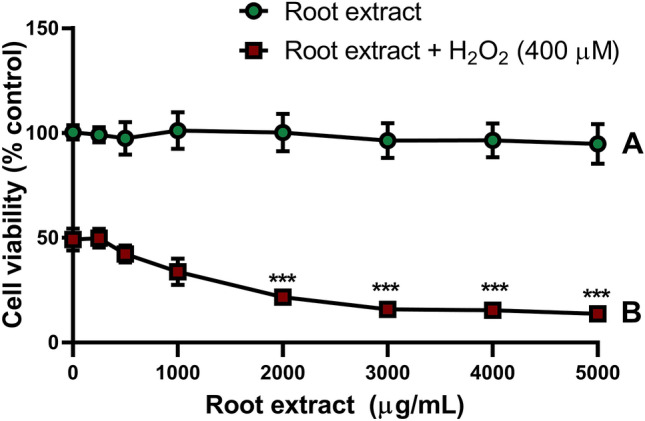
Effects of crude D. viscosa root extract on PC12 cell viability. Mean cell viability (± SD) measured by XTT assay for (A) PC12 cells incubated with concentrations of crude D. viscosa root extract (0–5000 μg/mL) for 14 h or (B) PC12 cells pretreated with concentrations of crude D. viscosa root extract (0–5000 μg/mL) for 2 h followed by incubation for 12 h with H2O2 (400 μM). Two-way ANOVA: crude root extract concentration, p < 0.0001; pretreatment, p < 0.0001; interaction, p < 0.0001. Dunnett’s test: ***, p < 0.0001.
HPLC-guided fractionation resulted in 14 fractions exhibiting diverse properties in a H2O2-induced cytotoxicity model
As shown (Fig. 1B) root extract showed no protective effect against H2O2 cytotoxicity, however we assumed (taking in mind the behavioral effect in FST, Fig. 4) that extract fractionation may result in identification of active compounds with more potent beneficial pharmacological properties. HPLC-guided separation of crude extract (Suppl. Fig. 1A) resulted in 14 fractions. Each fraction contained between 2 to 8 peaks (Suppl. Fig. 1B, representative fraction), with each peak corresponding to at least one compound.
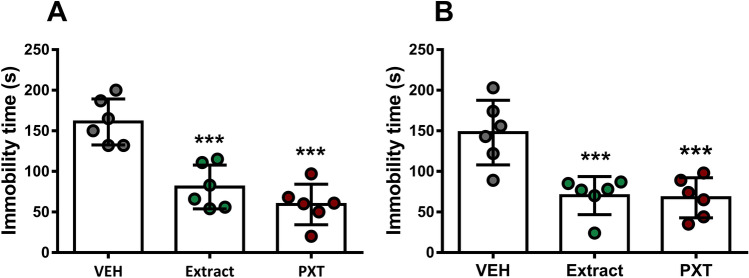
Figure 4.
Effect of crude D. viscosa root extract and PXT on immobility time in FST. (A) Single or (B) sub-chronic (14-day, daily) administration of crude D. viscosa root extract (5 mg/kg i.p.) or PXT (10 mg/kg i.p.) produced reductions in immobility time in FST (One-way ANOVA with Dunnett’s test: A: p < 0.0001; B: p < 0.0001). Data are expressed as mean (± SD). Abbreviations are as follows: VEH, vehicle; Extract, crude D. viscosa root extract; PXT, paroxetine. ***, p < 0.0001.
To identify fractions with beneficial biological activity, we assumed that cytoprotective effects would accompany the FST behavioral effects observed with the crude root extract. Combinations of separated fractions were tested in a concentration range similar to those of the crude extract (0–1000 μg/mL) in the XTT cell viability oxidative stress assay (400 μM H2O2) with PC12 cells (Fig. 2). Our initial assumptions appeared to be correct. Cell viability effects of all fraction combination treatments differed substantially (Two-way ANOVA followed by Tukey’s HSD: Fraction: F[2,135] = 703.5, p < 0.0001; concentration: F[4,135] = 4.897, p = 0.0010; interaction: F[8,135] = 79.81, p < 0.0001; Tukey’s HSD: 250–1000 μg/mL, all fractions, p < 0.0001, ***).
Figure 2.
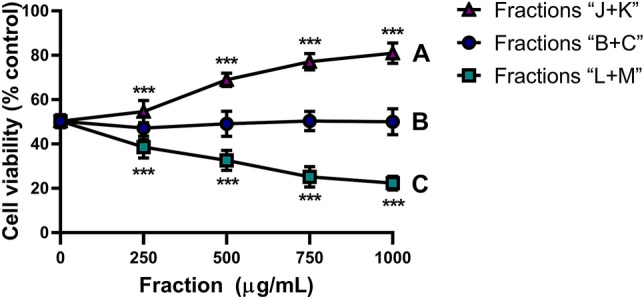
Effect of root extract fraction combinations on PC12 cell viability. Mean cell viability (± SD) measured by XTT assay of PC12 cells pretreated for 2 h with concentrations of combined root extract fractions (0–1000 μg/mL) (A) “J + K”, (B) “B + C”, or (C) “L + M” followed by 12 h of oxidative stress (400 μM H2O2). Two-way ANOVA: fraction, p < 0.0001; concentration, p < 0.0001; interaction, p < 0.0001. Dunnett’s test: p = 0.0001, ***.
Combination of fractions “B + C” and “L + M” were not protective against oxidative stress, with fraction “B + C” conferring no cytoprotective effect and fraction “L + M” concentration-dependently reducing cell viability (Dunnett’s test: p = 0.0001, all concentrations). Fraction “J + K” concentration-dependently increased cell viability by 31% over basal H2O2-induced oxidative stress (Dunnett’s test: p = 0.0001, 500–1000 μg/mL). Thus, fractions “J” and “K” were further separated and evaluated separately against H2O2-evoked cell death.
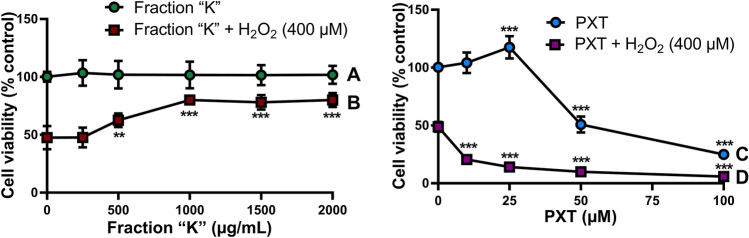
Fraction “K” markedly protected cells against H2O2-induced cytotoxicity
In the XTT cell viability assay, responses between PC12 cells treated with different concentrations of fraction “K” with and without H2O2 differed (Two-way ANOVA with Sidak test: Concentration, F[5,108] = 17.44, p < 0.0001; pretreatment, F[1,108] = 548.2, p < 0.0001; interaction, F[5,108] = 17.76, p < 0.0001; Sidak, all treatments, p < 0.0001; Fig. 3). Fraction “K” did not cause a proliferative effect in PC12 cells, but dose-dependently increased cell survival under H2O2-induced oxidative stress to a maximum of 68.3% at 2000 μg/mL (Dunnett’s test: 500 μg/mL, p = 0.0006; 1000–2000 μg/mL, p < 0.0001). In contrast, fraction “J” produced a deleterious effect on cell viability (Two-way ANOVA: Concentration, F[4,50] = 3.015, p = 0.0264; H2O2 treatment, F[1,50] = 1857, p < 0.0001; interaction, F[4,40] = 5.088, p = 0.0016; Sidak test: p < 0.0001, all treatments; Dunnett’s test: Fraction “J”, p > 0.05; fraction “J” + H2O2, 250 μM, p = 0.0005; 500 μM, p < 0.0001; 1000–2000 μM, p < 0.016; Suppl. Fig. 2). Treatment of PC12 cells with paroxetine (PXT), produced cytoprotective effects at lower concentrations (≤ 25 μM), however increasing concentrations were cytotoxic, reducing cell viability by 88.2% at a 100 μM concentration (Dunnett’s test: 25–100 μM, p < 0.0001). Interestingly, all tested concentrations of PXT, including non-cytotoxic ones, markedly reduced cell survival in the H2O2-induced cytotoxicity model (Dunnett’s test: 1000–2000 μM, p < 0.0001).
Figure 3.
Comparative effects of fraction “K” and PXT on cell viability. Mean cell viability (± SD) measured by XTT assay for (A) PC12 cells incubated with concentrations of fraction “K” (0–2000 μg/mL) for 14 h or (B) PC12 cells pretreated with concentrations of fraction “K” (0–2000 μg/mL) for 2 h followed by incubation for 12 h with H2O2 (400 μM). Mean cell viability (± SD) measured by XTT assay for (C) PC12 cells incubated with concentrations of PXT (0–100 μM) for 14 h or (D) PC12 cells pretreated with concentrations PXT (0–100 μM) for 2 h followed by incubation for 12 h with H2O2 (400 μM). Two-way ANOVA: (A,B) Concentration, p < 0.0001; pretreatment, p < 0.0001; interaction, p < 0.0001. Sidak: all treatments, p < 0.0001; Dunnett’s test: (B) 500 μg/mL, p = 0.0006; 1000–2000 μg/mL, p < 0.0001. (C,D) Concentration, p < 0.0001; pretreatment, p < 0.0001; interaction, p < 0.0001. Sidak: all treatments, p < 0.0001; Dunnett’s test: (C) 25–100 μM, p < 0.0001; (D) all treatments, p < 0.0001. **, p = 0.0006; ***, p < 0.0001.
Acute and sub-chronic D. viscosa root extract administration decreased mouse immobility time in the forced swim test (FST)
The behavioral effect of D. viscosa extracts was examined in submissive (Sub) mice, previously validated as a mouse model of depressive-like behavior26–28. Initial screening of D. viscosa root extract revealed that single and sub-chronic (14 days, daily) 5 mg/kg i.p. injection of root crude extract caused reduction in immobility time in the FST (Single dose: one-way ANOVA: F[2,25] = 23.95, p < 0.0001; sub-chronic dosing: F[2,15] = 13.64, p = 0.0004; Fig. 4). These results were similar to the effect of PXT (10 mg/kg), a widely prescribed selective serotonin reuptake inhibitor (SSRI) that was used as a positive control (Fig. 4). Neither the herbal extract nor PXT produced tachyphylaxis, indicated by similar behavioral responses regardless of single or 14-day daily treatments.
Fraction “K” produced antidepressant-like effects similar to paroxetine
Administration of single doses of fraction “K” to Sub mice subjected to the FST indicated similar reductions of depressive-like behavior comparable to the comparison dose of PXT (10 mg/kg) (One-way ANOVA, F[4,45] = 135.5, p < 0.0001; Fig. 5). Doses of 5 mg/kg or 25 mg/kg fraction “K” reduced mouse immobility times by 55.5% and 71.8%, respectively, which were comparable with the 60.7% reduction in immobility times using PXT. Fraction “K” doses as low as 1.0 mg/kg did not produce any measurable antidepressant-like effect in the FST. In experiments with these doses of fraction “K” in open field assessment, no alterations of locomotory activity were observed (One-way ANOVA F[4,25] = 0.0314, p = 0.998; Suppl. Fig. 3).
Figure 5.
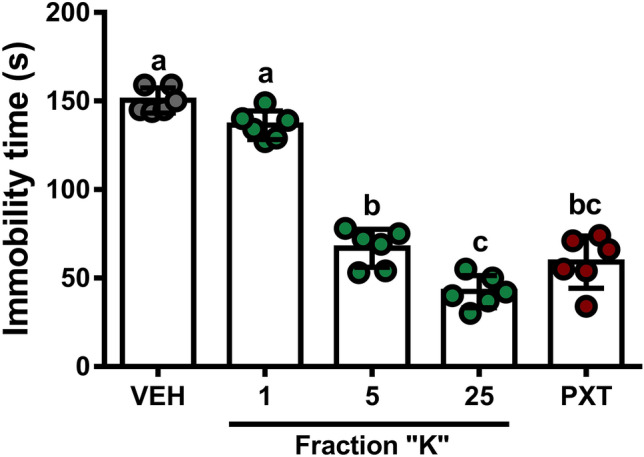
Effect of fraction “K” and PXT on immobility time of mice in the FST. Mean immobility time (± SD) in FST following a single i.p. dose of fraction “K” (0–25 mg/kg) or PXT (10 mg/kg) differed from both 1.0 mg/kg fraction “K” treatment and control (One-way ANOVA, p < 0.0001). Different letters indicate significant differences (Tukey’s HSD, p < 0.0001 all treatments except 25 mg/kg fraction “K” vs. PXT [p = 0.0021]).
Fraction “K” appears to affect 5-HT turnover in brain structures of Sub mice
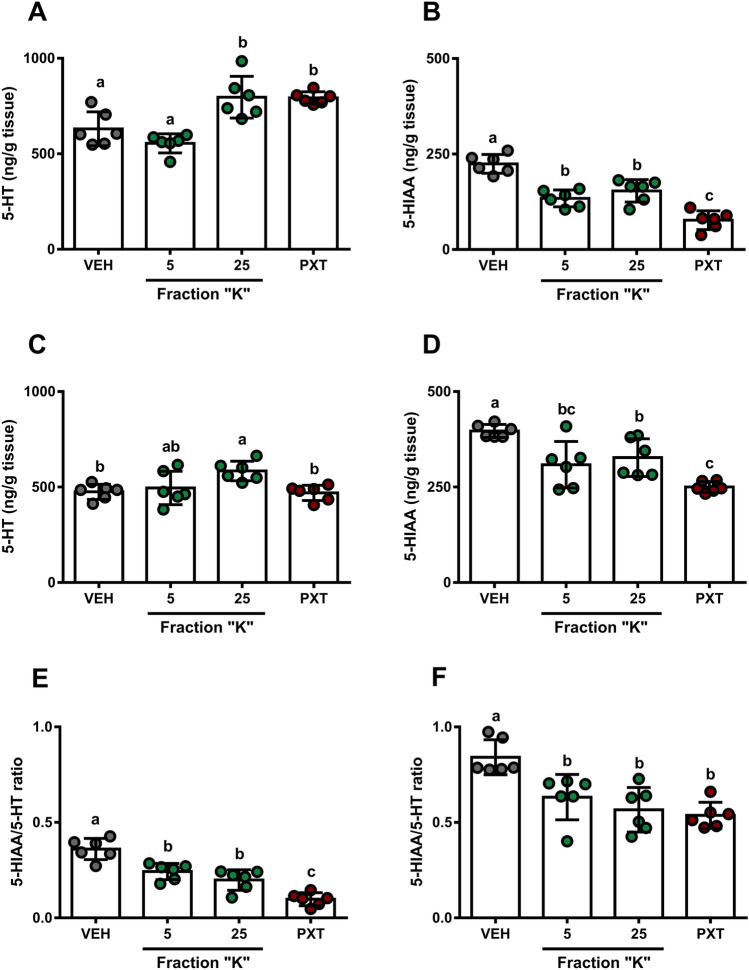
Neurochemical profiling following a single administration of pharmacologically-active doses of fraction “K” (5 and 25 mg/kg) using HPLC-ECD indicated a dose-dependent decrease in 5-HT content (One-way ANOVA with Tukey’s HSD: F[3,20] = 14.93, p < 0.0001, Fig. 6A; F[3,20] = 5.07, p = 0.0090, Fig. 6C). Doses of 25 mg/kg fraction “K” produced increases in 5-HT content comparable to PXT in prefrontal cortex (PFC), whereas the same dose produced increased 5-HT content in hippocampus (HPC), yet the dose of PXT used here did not. Concordantly, fraction “K” treatments reduced 5-HIAA content in both PFC (One-way ANOVA with Tukey’s HSD: F[3,20] = 34.62, p < 0.0001; Fig. 6B) and HPC (One-way ANOVA with Tukey’s HSD: F[3,20] = 13.22, p < 0.0001; Fig. 6D), with PXT treatment showing a more potent effect. Brain region-specific differences in PFC and HPC serotonin content were measured for all treatments, including vehicle control (5-HIAA/5-HT ratios. Two-way ANOVA: Brain region, F[1,40] = 339.2, p < 0.0001; treatment, F[3,40] = 28.38, p < 0.0001; interaction, F[3,40] = 1.239, p = 0.3081; Sidak test: p < 0.0001, all treatments; Fig. 6E,F).
Figure 6.
Regional brain tissue levels of 5-HT and 5-HIAA following fraction “K” and PXT treatments. (A,B,E) Prefrontal cortex. (C,D,F) Hippocampus. Mean (± SD; ng/g tissue) levels of prefrontal cortex (A) 5-HT, (B) 5-HIAA, and (E) 5-HIAA/5-HT ratios follow doses of fraction “K” (mg/kg dose indicated, all panels) or PXT (10 mg/kg, all panels). Mean (± SD; ng/g tissue) levels of hippocampus (C) 5-HT, (D) 5-HIAA, and (F) 5-HIAA/5-HT ratios follow doses of fraction “K” (mg/kg dose indicated, all panels) or PXT (10 mg/kg, all panels). One-way ANOVA followed by a Tukey’s HSD: p < 0.0001, all panels. Different letters indicate significant differences (p < 0.0332).
Phytochemical analysis of Fraction “K”
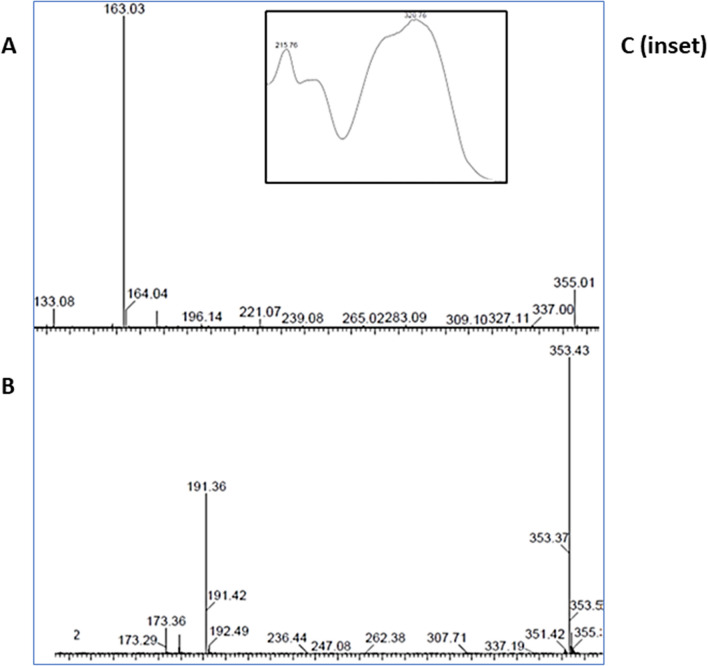
Two major components in fraction “K”, representing approximately 87% of the total content, were putatively identified based on positive and negative mass spectra as 49% chlorogenic acid (CGA) and 38% cynarine (Fig. 7A–C).
Figure 7.
Representative MS and UV spectra of Fraction “K” containing chlorogenic acid and cynarine. (A) ESI + mass spectra. (B) ESI—mass spectra. (C) UV spectra.
Discussion
Among the fractions from D. viscosa crude extract, we highlighted one fraction which we entitled “K”, that exhibited a strong cytoprotective effect by dose-dependently inhibiting H2O2-induced PC12 cell death determined by mitochondrial functionality. Since H2O2 is a potent apoptosis inducer, we may assume that fraction “K”-mediated cytoprotection may have been achieved by a direct anti-apoptotic effect or indirectly through antioxidative mechanisms similar to the effects shown by other plant extracts29,30. Although known antidepressants such as amitriptyline and fluoxetine show protective effects in a PC12 cell model31, we found that PXT in a non-cytotoxic concentration range for the drug alone produced increased H2O2-induced PC12 cell death. This is in agreement with other studies showing the cytotoxic activity of PXT against tumor cells of either murine or human origin32,33.
Despite the lack of a clear correlation between the cytoprotective effect of herbal/pharmacological agents in vitro and antidepressant activity in behavioral assays in vivo, several studies have reported such phenomena34,35. For example, antidepressant properties of Hypericum perforatum have been shown in vivo36,37 as well as neuroprotective effects observed in a number of in vitro studies38,39. We observed an antidepressant-like effect of fraction “K” in Sub mice possessing a strong depressive-like phenotype, which have been previously shown to be responsive to antidepressants, as well as plant-derived agents26,40 In our work, FST was applied as an acute environmental trigger for assessment of stress coping strategy in Sub mice.
The decreased immobility in the FST of Sub mice following fraction “K” administration was linked to a reduction in 5-HT turnover in PFC and HPC similar to PXT. PFC and HPC are considered to be hot-spot brain regions of the circuits involved in depression41 and are critically involved in the mechanism of SSRI action42. Both PFC and HPC are densely innervated by serotonergic fibers and the majority of 5-HT receptor subtypes are expressed in these regions43,44.
Thus, we suggest that fraction “K”-induced changes in the serotonergic system are associated with serotonin transporter (SERT) inhibition, similar to the established mechanism of PXT. PXT functions as a potent SERT inhibitor and reduces 5-HT reuptake, leading to a redistribution of 5-HT in favor of the synaptic cleft and a consequent decrease in presynaptic cytosolic concentrations. These events may trigger compensatory reduction of 5-HT degradation due to the disinhibition of the activity of tryptophan hydroxylase, reflected by decreased 5-HIAA content and increased 5-HT production45 as demonstrated here by decreased 5-HIAA/5-HT ratio.
We linked the observed biological activity of fraction “K” to CGA and cynarine, which were identified as major components of the fraction. CGA is a polyphenolic secondary metabolite produced by many plant species, including D. viscosa46. As a potent antioxidant, CGA shows high capability in modulating oxidative stress in cell- and animal-based models47,48. CGA is known to penetrate the blood–brain barrier49 exhibiting antidepressant and anxiolytic properties in rodent models50,51. CGA affects plasma β-endorphin52, serum 5-HT, and dopamine51 as well as colonic 5-HT levels53. It has also been shown in vitro that CGA stimulates axon and dendrite growth and promotes 5-HT release through augmenting synapsin I expression in rat raphe neurons49.
Cynarine has a broad repertoire of biological activities. It is a strong antioxidant compound54,55 that also produces effects on smooth muscle, which is thought to contribute to reported antihypertensive activity56,57. The antioxidant effects of cyanarine are most potently demonstrated in H2O2 cytotoxicity assays and is most likely the reason for cytoprotective effects of fraction “K” observed here. Cynarine also has the potential for neurological effects as the chemical has demonstrated anticholinesterase activity with a Ki in the low nM range55, however any potential psychotropic properties have not been studied.
In this work, we demonstrated, for the first time, the link between changes in the brain serotonergic system and behavioral phenotype following treatment with a D. viscosa plant root extract fraction (fraction “K”). Although fraction “K” is CGA and cynarine-enriched and despite the predominance of these two compounds in the isolated fraction, fraction “K” is still a crude preparation and we have not yet identified or have assessed the influences of other chemical agents present. Behavioral effects from fraction “K” treatment resemble those reported for CGA, however we cannot exclude the involvement of other, undetermined agents in the preparation. Further work is mandatory to understand the contribution of each compound in the observed biological activity, the contributions of as-yet unidentified compounds in the remaining 13% of the active fraction “K”, and their possible interactions. We believe the biggest challenge in future efforts will be the assessment of multiple chemical combinations from this bioactive fraction and identification of synergism effects. It is our intent to continue the study of D. viscosa root to isolate and characterize compounds with antidepressant action and other positive biological properties.
Conclusions
A CGA-enriched fraction of D. viscosa root extract suppressed the deteriorative effects of H2O2 on PC12 cells. The study provides the first evidence of a marked in vivo antidepressant-like activity of a CGA-enriched fraction of D. viscosa root extract in a mouse model of depressive-like behavior through modulation of the serotonergic system. Further studies are warranted to elucidate the contribution of other compounds within the active fraction to its pharmacological properties.
Materials and methods
Plant material collection and root extract preparation
Root samples of D. viscosa were collected from wild-growing plants in Ariel, Israel (lat 32.105766; long 35.211673) during June 2018. No national permits were required for plant collection (D. viscosa is a common, widespread Mediterranean plant and not on any protected species list). Plant authentication was performed by the National Natural History Collections Herbarium at Hebrew University, Jerusalem; a voucher specimen (HUJ-135000) was deposited (http://nnhc.huji.ac.il/).
To prepare the crude extracts, 100 g of D. viscosa roots were gently washed, cut into 1.0 cm sections, boiled in 1.0 L of distilled water (1.0 g/ 10 mL) for 1 h, cooled to room temperature, filtered, and frozen at -80 °C until use.
Extract fractionation
Fractionation of root crude extract was performed using preparative HPLC (CombiFlash EZ Prep) with Unisol (Agela Technologies) reverse phase C18 column (10 µm; 21.2 × 250 mm; 100 Å). A dual mobile phase gradient (mobile phase A [0.05% formic acid] and mobile phase B [acetonitrile]) was used to achieve appropriate separation. The gradient composition B:A was as follows: 5:95% (0 min); 15:85% (0–5 min); 20:80% (5–10 min); 25:75% (10–15 min); 30:70% (15–25 min); 35:65% (25–70 min). The flow rate was 21.0 mL·min−1 and the injection volume was 10 mL of crude extract.
Extract was separated into 14 fractions, each combining 5 min of flow, and assigned a letter (A-N). A UV detector visualized the peaks at wavelengths of 280 and 257 nm. Each fraction was lyophilized at − 80 °C for approximately 72 h and reconstituted to an aqueous solution. All reconstituted fractions were standardized to fit the amount of the major parallel peaks in the crude extract by diluting in n times (for example, reconstituted fraction “K” was diluted seven times to achieve its initial concentration in crude extract).
Data were collected and analyzed using ChromNAV 2.0 HPLC Software.
Chemicals and treatments
For in vivo and ex vivo studies, 5 mg/kg D. viscosa root crude extract or 1, 5, or 25 mg/kg of isolated fraction were used. The doses were chosen based on our pilot studies that identified pharmacological activity of the crude root extract or isolated fraction within this dose range. Saline was used as a solvent vehicle (VEH). Paroxetine (PXT, Sigma-Aldrich cat. PHR 1804S; 10 mg/kg i.p.), an antidepressant agent, was used for comparison with D. viscosa preparations since Sub mice with depressive-like behavior have shown antidepressant effects when treated with this drug26. All injections were performed i.p at a volume of 5 μL/g body weight, 60 min before the behavioral or neurochemical studies. For in vitro studies, cells were incubated in the presence of crude extract (250–5000 μg/mL), isolated fractions (250–1000 µg/mL), or PXT (10–100 µM) diluted in RPMI medium.
H2O2 cytotoxicity assay
Rat pheochromocytoma cells (PC12; ATCC, CRL-1721 were grown in RPMI medium supplemented with 10% heat-inactivated horse serum, 5% fetal bovine serum, and 0.1% penicillin/streptomycin at 37 °C in a humidified incubator with 5% CO2. For all experiments, cells were seeded into 96-well plates at a density of 5 × 104 cells/well/100 µL. Cell protection was assessed using the hydrogen peroxide (H2O2)-based oxidative stress cytotoxicity model. PC12 cells were treated with 400 µM H2O2 (IC50) for 12 h following 2 h of pretreatment with differing concentrations of D. viscosa extracts. Cell viability was determined by the XTT reduction assay (Biological Industries, Cat. No 20-300-1000). The 400 µM concentration was selected to produce 50% cell death as measured in the XTT assay.
Animals
Submissive (Sub) mice used in this study were selectively bred for 36 generations from an outbred Sabra strain (Envigo laboratories, Israel) using social interaction dominant-submissive relationship paradigm58. The behavioral profile of Sub mice possesses strong elements of depressive-like behavior and impaired stress coping strategies confirmed by different experimental approaches25–28.
Animals were housed in groups of five in a temperature (21 ± 2 °C), humidity (55 ± 5%), and light controlled room (lights on from 7 AM to 7 PM). Standard laboratory chow and water were available ad libitum. All experiments were conducted with male mice (age, 10 weeks), during the light phase of the day-night cycle between 9 AM and 3 PM. Behavioral and neurochemical studies were conducted on the separate cohorts of Sub mice. Housing, care, and experimental procedures involving animal use conformed to NIH/USDA and ARRIVE 2.0 guidelines. The experiments were approved and supervised by the Institutional Animal Care and Use Committee of Ariel University and the Israel Ministry of Health (Protocol IL-74-09-15).
Open field test (OF)
Spontaneous locomotor (horizontal) activity was assessed in the Open Field (OF) test using EthoVision (Noldus, Holland) as previously described27,59. Briefly, the OF apparatus consisted of a square black plastic chamber (40 cm × 40 cm). Each mouse was placed individually for 5 min in the center of the chamber and distance travelled was recorded. Between subjects, the apparatus was thoroughly washed with 70% ethanol and dried with tissue paper.
Forced swim test (FST)
FST was performed to access stress coping strategies following psychotropic interventions60. Mice (6 per group) were individually tested in a glass cylinder (30 cm in height, 10 cm in diameter) filled with water up to 25 cm height (25 ± 2 °C). Mice were tested for 6 min, the last 4 min were analyzed, where the total time spent immobile was recorded manually. Mice were considered “immobile” if they displayed no activity except for what is required to keep the head above water.
Neurochemical profiling
For sample preparation, mice were anesthetized in a CO2 chamber and decapitated immediately afterwards. Prefrontal cortex (PFC) and hippocampus (HPC) were dissected, frozen in liquid nitrogen and kept at − 80 °C until use. Serotonin (5-hydroxytryptamine, 5-HT) and its metabolite 5-hydroxyindoleacetic acid (5-HIAA) were assayed in brain tissue samples using HPLC with electrochemical detection (EICOM Co., Kyoto, Japan) as previously described45.
HPLC–MS/MS analysis
Chemical analysis of fractions was performed using a Waters 2695 Separation Module with a Photodiode Array Detector together with a Quattro Micro Mass Spectrometer. Chromatographic separation was achieved using a Luna (Phenomenex) reverse phase C18 (5 µm; 4.6 × 250 mm; 100 Å) column and the binary gradient described above (see Extract fractionation). MS acquisition was conducted in both ESI positive and negative ionization mode under the following conditions: capillary voltage—3.5 kV, cone voltage—45 V, extractor voltage − 3 V, RF lens − 0.2 V, source temperature − 120 °C, desolvation temperature − 350 °C, nitrogen flow rate of 700 L/h for desolvation and 50 L/h cone gas. Tentative identification of compounds was achieved based on comparisons with purchased standards (Cayman Chemical Company) as wells as spectra from an MS library (NIST 2017).
Statistical analysis
Data are expressed as means (± SD). All multiple comparisons were Bonferroni-corrected against α = 0.05 and all analyses were two-tailed. Multiple comparison analyses were performed by one-way ANOVA followed by a Tukey’s HSD or Dunnett’s test as appropriate or were compared by two-way ANOVA followed by a Sidak or Tukey’s HSD test based on the structure of the dataset. All analyses were performed in GraphPad Prism 7.0. Results of all analyses are presented in detail in Supplementary Table S1. Data from this study are publicly available in the Mendeley Data repository61.
Supplementary Information
Acknowledgements
We would like to thank Hagar Leschner of the National Natural History Collections at Hebrew University-Jerusalem for her assistance in registering plant samples in the Herbarium of Hebrew University.
Author contributions
K.M. performed the fractionation procedures, neurochemistry experiments, and behavioral experiments. N.C. performed field collections of plant material, root extraction procedures, and behavioral experiments. A. Pinkus performed cell bioassays. L.V. was responsible for animal care and assistance with behavioral experiments. M.K. performed data analysis and manuscript preparation. J.G. performed HPLC–MS/MS analyses. M.P., E.D., and A. Pinhasov served as technical advisors and supervisors of the project.
Funding
The research was supported by funding from the Israel Ministry of Science, Technology & Space (Grant # 3-12726).
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Elyashiv Drori, Email: droris@ariel.ac.il.
Albert Pinhasov, Email: albertpi@ariel.ac.il.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-022-04840-9.
References
- 1.World Health Organization, Depression and Other Common Mental Disorders: Global Health Estimates. 2017, World Health Organization; Licence: CC BY-NC-SA 3.0 IGO.
- 2.Friedrich MJ. Depression is the leading cause of disability around the world. JAMA. 2017;317(15):1517–1517. doi: 10.1001/jama.2017.3826. [DOI] [PubMed] [Google Scholar]
- 3.Hillhouse TM, Porter JH. A brief history of the development of antidepressant drugs: From monoamines to glutamate. Exp. Clin. Psychopharmacol. 2015;23(1):1–21. doi: 10.1037/a0038550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.El-Hage W, et al. Mechanisms of antidepressant resistance. Front Pharmacol. 2013;4:146. doi: 10.3389/fphar.2013.00146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ekor M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol. 2014;4:177. doi: 10.3389/fphar.2013.00177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li FS, Weng JK. Demystifying traditional herbal medicine with modern approach. Nat. Plants. 2017;3:17109. doi: 10.1038/nplants.2017.109. [DOI] [PubMed] [Google Scholar]
- 7.Garg V, et al. Facts about standardization of herbal medicine: A review. Zhong Xi Yi Jie He Xue Bao. 2012;10(10):1077–1083. doi: 10.3736/jcim20121002. [DOI] [PubMed] [Google Scholar]
- 8.Ng QX, Venkatanarayanan N, Ho CY. Clinical use of Hypericum perforatum (St John's wort) in depression: A meta-analysis. J. Affect. Disord. 2017;210:211–221. doi: 10.1016/j.jad.2016.12.048. [DOI] [PubMed] [Google Scholar]
- 9.Bent S, et al. Valerian for sleep: A systematic review and meta-analysis. Am. J. Med. 2006;119(12):1005–1012. doi: 10.1016/j.amjmed.2006.02.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hritcu L, Cioanca O. Prevalence of use of herbal medicines and complementary and alternative medicine in Europe. In: Grosso C, editor. Herbal Medicine in Depression: Traditional Medicine to Innovative Drug Delivery. Springer; 2016. pp. 135–181. [Google Scholar]
- 11.Yuan H, et al. The traditional medicine and modern medicine from natural products. Molecules. 2016;21(5):559. doi: 10.3390/molecules21050559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hirshler Y, Doron R. Neuroplasticity-related mechanisms underlying the antidepressant-like effects of traditional herbal medicines. Eur. Neuropsychopharmacol. 2017;27(10):945–958. doi: 10.1016/j.euroneuro.2017.07.008. [DOI] [PubMed] [Google Scholar]
- 13.Parolin P, Ion-Scotta M, Bresch C. Biology of Dittrichia viscosa, a Mediterranean ruderal plant: A review. Phyton Int. J. Exp. Bot. 2016;83(2):251–262. [Google Scholar]
- 14.Brullo S, Marco G. Taxonomical revision of the genus Dittrichia (Asteraceae) Portugaliae Acta Biologica. 2000;19:341–354. [Google Scholar]
- 15.Said O, et al. Ethnopharmacological survey of medicinal herbs in Israel, the Golan Heights and the West Bank region. J. Ethnopharmacol. 2002;83(3):251–265. doi: 10.1016/s0378-8741(02)00253-2. [DOI] [PubMed] [Google Scholar]
- 16.Ali-Shtayeh MS, Yaniv Z, Mahajna J. Ethnobotanical survey in the Palestinian area: A classification of the healing potential of medicinal plants. J. Ethnopharmacol. 2000;73(1–2):221–232. doi: 10.1016/s0378-8741(00)00316-0. [DOI] [PubMed] [Google Scholar]
- 17.Dafni A, Yaniv Z, Palevitch D. Ethnobotanical survey of medicinal plants in northern Israel. J. Ethnopharmacol. 1984;10(3):295–310. doi: 10.1016/0378-8741(84)90017-5. [DOI] [PubMed] [Google Scholar]
- 18.Krispil N. A Bag of Plants (The Useful Plants of Israel) [IN HEBREW] Yara Publishing House; 1987. [Google Scholar]
- 19.Seca AM, et al. The genus Inula and their metabolites: From ethnopharmacological to medicinal uses. J. Ethnopharmacol. 2014;154(2):286–310. doi: 10.1016/j.jep.2014.04.010. [DOI] [PubMed] [Google Scholar]
- 20.Craker LE, Simon JE. Herbs, Spices, and Medicinal Plants: Recent Advances in Botany, Horticultulture and Pharmacology. Oryx Press; 1986. [Google Scholar]
- 21.Targum SD, Fava M. Fatigue as a residual symptom of depression. Innov. Clin. Neurosci. 2011;8(10):40–43. [PMC free article] [PubMed] [Google Scholar]
- 22.Corfield EC, Martin NG, Nyholt DR. Co-occurrence and symptomatology of fatigue and depression. Compr. Psychiatry. 2016;71:1–10. doi: 10.1016/j.comppsych.2016.08.004. [DOI] [PubMed] [Google Scholar]
- 23.Sawynok J, Esser MJ, Reid AR. Antidepressants as analgesics: An overview of central and peripheral mechanisms of action. J. Psychiatry Neurosci. JPN. 2001;26(1):21–29. [PMC free article] [PubMed] [Google Scholar]
- 24.Ballenger JC. Anxiety and depression: Optimizing treatments. Prim. Care Companion J. Clin. Psychiatry. 2000;2(3):71–79. doi: 10.4088/pcc.v02n0301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nesher E, et al. Synapsin IIb as a functional marker of submissive behavior. Sci. Rep. 2015;5(1):10287. doi: 10.1038/srep10287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nesher E, et al. Differential responses to distinct psychotropic agents of selectively bred dominant and submissive animals. Behav. Brain Res. 2013;236(1):225–235. doi: 10.1016/j.bbr.2012.08.040. [DOI] [PubMed] [Google Scholar]
- 27.Gross M, Pinhasov A. Chronic mild stress in submissive mice: Marked polydipsia and social avoidance without hedonic deficit in the sucrose preference test. Behav. Brain Res. 2016;298(Pt B):25–34. doi: 10.1016/j.bbr.2015.10.049. [DOI] [PubMed] [Google Scholar]
- 28.Murlanova K, et al. Link between temperament traits, brain neurochemistry and response to SSRI: Insights from animal model of social behavior. J. Affect. Disord. 2020;282:1055–1066. doi: 10.1016/j.jad.2020.11.005. [DOI] [PubMed] [Google Scholar]
- 29.Qi G, et al. Neuroprotective action of tea polyphenols on oxidative stress-induced apoptosis through the activation of the TrkB/CREB/BDNF pathway and Keap1/Nrf2 signaling pathway in SH-SY5Y cells and mice brain. Food Funct. 2017;8(12):4421–4432. doi: 10.1039/c7fo00991g. [DOI] [PubMed] [Google Scholar]
- 30.Park HR, et al. Neuroprotective effects of Liriope platyphylla extract against hydrogen peroxide-induced cytotoxicity in human neuroblastoma SH-SY5Y cells. BMC Compl. Altern. Med. 2015;15:171. doi: 10.1186/s12906-015-0679-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kolla N, et al. Amitriptyline and fluoxetine protect PC12 cells from cell death induced by hydrogen peroxide. J. Psychiatry Neurosci. 2005;30(3):196–201. [PMC free article] [PubMed] [Google Scholar]
- 32.Cho YW, et al. Paroxetine induces apoptosis of human breast cancer MCF-7 cells through Ca(2+)-and p38 MAP kinase-dependent ROS generation. Cancers (Basel) 2019;11(1):64. doi: 10.3390/cancers11010064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chen WC, et al. Mechanism of paroxetine-induced cell death in renal tubular cells. Basic Clin. Pharmacol. Toxicol. 2008;103(5):407–413. doi: 10.1111/j.1742-7843.2008.00319.x. [DOI] [PubMed] [Google Scholar]
- 34.Liu P, et al. Potential antidepressant properties of Radix Polygalae (Yuan Zhi) Phytomedicine. 2010;17(10):794–799. doi: 10.1016/j.phymed.2010.01.004. [DOI] [PubMed] [Google Scholar]
- 35.Zhang Y, et al. Neuroprotective effects of sodium ferulate and its antidepressant-like effect measured by acute and chronic experimental methods in animal models of depression. J. Behav. Brain Sci. 2011;1(2):37–46. [Google Scholar]
- 36.Butterweck V, et al. Flavonoids from Hypericum perforatum show antidepressant activity in the forced swimming test. Planta Med. 2000;66(1):3–6. doi: 10.1055/s-2000-11119. [DOI] [PubMed] [Google Scholar]
- 37.Tian J, et al. Antidepressant-like activity of adhyperforin, a novel constituent of Hypericum perforatum L. Sci. Rep. 2014;4:5632. doi: 10.1038/srep05632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Oliveira AI, et al. Neuroprotective activity of hypericum perforatum and its major components. Front. Plant Sci. 2016;7:1004. doi: 10.3389/fpls.2016.01004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lu YH, et al. Neuroprotective effects of Hypericum perforatum on trauma induced by hydrogen peroxide in PC12 cells. Am. J. Chin. Med. 2004;32(3):397–405. doi: 10.1142/S0192415X04002053. [DOI] [PubMed] [Google Scholar]
- 40.Gross M, et al. Chronic food administration of Salvia sclarea oil reduces animals' anxious and dominant behavior. J. Med. Food. 2013;16(3):216–222. doi: 10.1089/jmf.2012.0137. [DOI] [PubMed] [Google Scholar]
- 41.Liu W, et al. The role of neural plasticity in depression: From hippocampus to prefrontal cortex. Neural Plast. 2017;2017:6871089–6871089. doi: 10.1155/2017/6871089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chang CH, Chen MC, Lu J. Effect of antidepressant drugs on the vmPFC-limbic circuitry. Neuropharmacology. 2015;92:116–124. doi: 10.1016/j.neuropharm.2015.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dale E, et al. Effects of serotonin in the hippocampus: How SSRIs and multimodal antidepressants might regulate pyramidal cell function. CNS Spectr. 2016;21(2):143–161. doi: 10.1017/S1092852915000425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Albert PR, Vahid-Ansari F, Luckhart C. Serotonin-prefrontal cortical circuitry in anxiety and depression phenotypes: Pivotal role of pre- and post-synaptic 5-HT1A receptor expression. Front. Behav. Neurosci. 2014;8:199–199. doi: 10.3389/fnbeh.2014.00199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Murlanova K, et al. Link between temperament traits, brain neurochemistry and response to SSRI: Insights from animal model of social behavior. J. Affect. Disord. 2021;282:1055–1066. doi: 10.1016/j.jad.2020.11.005. [DOI] [PubMed] [Google Scholar]
- 46.Grauso L, et al. Exploring Dittrichia viscosa (L.) Greuter phytochemical diversity to explain its antimicrobial, nematicidal and insecticidal activity. Phytochem. Rev. 2020;19(3):659–689. [Google Scholar]
- 47.Liang N, Kitts DD. Role of chlorogenic acids in controlling oxidative and inflammatory stress conditions. Nutrients. 2015;8(1):16. doi: 10.3390/nu8010016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Cha JW, et al. The polyphenol chlorogenic acid attenuates UVB-mediated oxidative stress in human HaCaT keratinocytes. Biomol. Therapeut. 2014;22(2):136–142. doi: 10.4062/biomolther.2014.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wu J, et al. Antidepressant potential of chlorogenic acid-enriched extract from eucommia ulmoides oliver bark with neuron protection and promotion of serotonin release through enhancing synapsin I expression. Molecules. 2016;21(3):260. doi: 10.3390/molecules21030260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ramalhete N, et al. Comparative study on the in vivo antidepressant activities of the Portuguese Hypericum foliosum, Hypericum androsaemum and Hypericum perforatum medicinal plants. Ind. Crops Prod. 2016;82:29–36. [Google Scholar]
- 51.Song J, et al. Modulation of gut microbiota by chlorogenic acid pretreatment on rats with adrenocorticotropic hormone induced depression-like behavior. Food Funct. 2019;10(5):2947–2957. doi: 10.1039/c8fo02599a. [DOI] [PubMed] [Google Scholar]
- 52.Park S-H, et al. Antidepressant-like effect of chlorogenic acid isolated from Artemisia capillaris Thunb. Anim. Cells Syst. 2010;14(4):253–259. [Google Scholar]
- 53.Wu Y, et al. Dietary chlorogenic acid regulates gut microbiota, serum-free amino acids and colonic serotonin levels in growing pigs. Int. J. Food Sci. Nutr. 2018;69(5):566–573. doi: 10.1080/09637486.2017.1394449. [DOI] [PubMed] [Google Scholar]
- 54.Erikel E, Yuzbasioglu D, Unal F. In vitro genotoxic and antigenotoxic effects of cynarin. J. Ethnopharmacol. 2019;237:171–181. doi: 10.1016/j.jep.2019.03.036. [DOI] [PubMed] [Google Scholar]
- 55.Topal M, et al. Antioxidant, antiradical, and anticholinergic properties of cynarin purified from the Illyrian thistle (Onopordum illyricum L.) J. Enzyme Inhib. Med. Chem. 2016;31(2):266–275. doi: 10.3109/14756366.2015.1018244. [DOI] [PubMed] [Google Scholar]
- 56.Hakkou Z, et al. Antihypertensive and vasodilator effects of methanolic extract of Inula viscosa: Biological evaluation and POM analysis of cynarin, chlorogenic acid as potential hypertensive. Biomed. Pharmacother. 2017;93:62–69. doi: 10.1016/j.biopha.2017.06.015. [DOI] [PubMed] [Google Scholar]
- 57.Xia N, et al. Artichoke, cynarin and cyanidin downregulate the expression of inducible nitric oxide synthase in human coronary smooth muscle cells. Molecules. 2014;19(3):3654–3668. doi: 10.3390/molecules19033654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Feder Y, et al. Selective breeding for dominant and submissive behavior in Sabra mice. J. Affect. Disord. 2010;126(1–2):214–222. doi: 10.1016/j.jad.2010.03.018. [DOI] [PubMed] [Google Scholar]
- 59.Gross M, et al. Social dominance predicts hippocampal glucocorticoid receptor recruitment and resilience to prenatal adversity. Sci. Rep. 2018;8(1):9595. doi: 10.1038/s41598-018-27988-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Can A, et al. The mouse forced swim test. J. Vis. Exp. 2012;59:e3638. doi: 10.3791/3638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kirby M. Dittrichia Viscosa Data. Mendeley Data; 2021. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.