Abstract
Background
Vitamin D deficiency has been associated with worse coronavirus disease 2019 (COVID-19) outcomes, but circulating 25-hydroxyvitamin D [25(OH)D] is largely bound to vitamin D–binding protein (DBP) or albumin, both of which tend to fall in illness, making the 25(OH)D status hard to interpret. Because of this, measurements of unbound (“free”) and albumin-bound (“bioavailable”) 25(OH)D have been proposed.
Objectives
We aimed to examine the relationship between vitamin D status and mortality from COVID-19.
Methods
In this observational study conducted in Liverpool, UK, hospitalized COVID-19 patients with surplus sera available for 25(OH)D analysis were studied. Clinical data, including age, ethnicity, and comorbidities, were extracted from case notes. Serum 25(OH)D, DBP, and albumin concentrations were measured. Free and bioavailable 25(OH)D were calculated. Relationships between total, free, and bioavailable 25(OH)D and 28-day mortality were analyzed by logistic regression.
Results
There were 472 patients with COVID-19 included, of whom 112 (23.7%) died within 28 days. Nonsurvivors were older (mean age, 73 years; range, 34–98 years) than survivors (mean age, 65 years; range, 19–95 years; P = 0.003) and were more likely to be male (67%; P = 0.02). The frequency of vitamin D deficiency [25(OH)D < 50 nmol/L] was similar between nonsurvivors (71/112; 63.4%) and survivors (204/360; 56.7%; P = 0.15) but, after adjustments for age, sex, and comorbidities, increased odds for mortality were present in those with severe deficiency [25(OH)D < 25 nmol/L: OR, 2.37; 95% CI, 1.17–4.78] or a high 25(OH)D (≥100 nmol/L; OR, 4.65; 95% CI, 1.51–14.34) compared with a 25(OH)D value of 50–74 nmol/L (reference). Serum DBP levels were not associated with mortality after adjustments for 25(OH)D, age, sex, and comorbidities. Neither free nor bioavailable 25(OH)D values were associated with mortality.
Conclusions
Vitamin D deficiency, as commonly defined by serum 25(OH)D levels (<50 nmol/L), is not associated with increased mortality from COVID-19, but extremely low (<25 nmol/L) and high (>100 nmol/L) levels may be associated with mortality risks. Neither free nor bioavailable 25(OH)D values are associated with mortality risk. The study protocol was approved by the London-Surrey Research Ethics Committee (20/HRA/2282).
Graphical Abstract
Keywords: vitamin D, COVID-19 mortality, vitamin D–binding protein, free vitamin D, bioavailable vitamin D
Introduction
Coronavirus disease 2019 (COVID-19) has accounted for nearly 200 million infections and over 4.25 million deaths worldwide as of 22 November 2021 (1). Factors associated with adverse outcomes include older age; male sex; comorbidities such as obesity, diabetes, and hypertension; and Black or Asian race (2). These factors are all independently associated with vitamin D deficiency (2) or, in males, with a reduced impact of vitamin D on the immune response (3). A role for vitamin D is further supported by correlations between latitude or UVB exposure and both COVID-19 infection growth rates (4) and age-adjusted mortality (5, 6), although such associations might be confounded by unmeasured factors.
Vitamin D, a cholesterol-derived steroid hormone, plays critical roles in musculoskeletal health and in modulating innate and adaptive immunity (6). In keeping with this, vitamin D status has been shown to influence the risk of acquiring respiratory viral infections (7). Vitamin D deficiency is also associated with the severity of respiratory disease: for example, the need for intensive care in infants hospitalized with bronchiolitis (8). Vitamin D deficiency upregulates proinflammatory cytokines such as TNF, type 1 IFN, and IL-6 (9, 10). Moreover, genetically modified mice lacking a vitamin D receptor show greatly increased susceptibility to lipopolysaccharide-induced experimental lung damage (11).
Various studies have examined associations between vitamin D status and the risk of acquiring COVID-19 or its severity (12, 13, 14, 15, 16, 17), but they are heterogenous and inconsistent. Studies have often been limited by sample size or by historical measurements of vitamin D, sometimes from many years before. Furthermore, studies of vitamin D concentrations in individuals who are already ill have to be interpreted with caution because of the possible fall in vitamin D–binding protein (DBP), and hence total 25-hydroxycholecalciferol [25(OH)D], as part of a negative acute-phase response (18, 19).
Vitamin D status is determined by the serum concentration of 25(OH)D, which is largely protein bound, with approximately 85% to DBP and 15% to albumin. Binding to albumin is less avid, and albumin-bound vitamin D is therefore commonly designated as “bioavailable.” Both serum DBP (20) and albumin (21) concentrations may fall in illness as a negative acute-phase response. Only 0.03% of circulating 25(OH)D is unbound or “free” (18). Measurement of free 25(OH)D is challenging due to its very low serum concentration, and has historically relied on cumbersome radioactive tracer–based methods (18). Thus, computational methods that rely on concentrations of total ligands and DBP and their in vitro measured affinity constants are often used to calculate free and bioavailable 25(OH)D (19). Both free 25(OH)D and active 1,25(OH)2D freely diffuse across the lipid bilayer of cell walls. Many cells and tissues, not just the kidneys, possess the 1α-hydroxylase-activating enzyme (CYP27B1). Perhaps consequently, adverse biological consequences of vitamin D deficiency tend to correlate with low serum concentrations of 25(OH)D but not with serum concentrations of activated 1,25(OH)2D (22).
Given these complexities, plus uncertainties around the 25(OH)D concentration used to define deficiency (23), it is perhaps not surprising that studies of vitamin D during COVID-19 have produced contradictory results. We have therefore investigated the associations between total, free, and bioavailable 25(OH)D and COVID-19 mortality.
Methods
We conducted a single-center study of COVID-19 patients who were hospitalized between 18 March and 2 November 2020 across the 2 acute hospital sites of the Liverpool University Hospitals NHS Foundation Trust. We included all patients who had surplus sera available for analyses of 25(OH)D concentrations. The population studied were unvaccinated. We collected baseline clinical information, including demographic data such as age, sex, BMI, and comorbidities (diabetes; hypertension; chronic respiratory illness, including asthma, chronic obstructive airways disease, and interstitial lung disease; chronic cardiovascular or cerebrovascular disease; neurological disease, including dementia or cerebrovascular accident; chronic kidney disease; chronic liver disease; and active malignancy). We used self-reported data on ethnicity, which is held in standard demographics of medical records. Data on clinical progress during hospitalization, including the need for supplemental oxygen, respiratory support in the form of continuous positive airways pressure or mechanical ventilation, and death within 28 days of a positive severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) test were recorded. We also collected information on medical treatment of COVID-19 (remdesivir and/or dexamethasone) or prescribed vitamin D supplementation prior to measurement of serum 25(OH)D levels.
Sample size
The sample size was determined by the number of suitable patients available for the study, and no formal power calculation was made. In total, data on 472 patients were available: of these, 360 were still alive 28 days after testing positive for SARS-CoV-2, and 112 had died. A retrospective power estimate based on the 112 patients who died and the 360 who survived suggested that we could have determined a difference of 6 nmol/L in the serum 25(OH)D levels between the survivor and nonsurvivor groups, using a standard significance level of 5% and a power of 80%. DBP was measured in 419 patients, as the remainder did not have sufficient residual serum to measure DBP. The primary outcome measure was mortality within 28 days of a positive SARS-CoV-2 test.
Measurement of serum 25(OH)D and DBP
Serum samples were aliquoted and stored at −80°C until they could be analyzed for DBP and total 25(OH)D. Concentrations of DBP were measured using a commercial polyclonal antibody ELISA kit (Immundiagnostik AG) according to the manufacturer’s protocol. The 25(OH)D2 and 25(OH)D3 concentrations were measured using established in-house LC-MS/MS methodology. The vitamin D2 and D3 values were summated to determine total 25(OH)D. Serum albumin concentrations were measured using a colorimetric bromocresol green–based method (Roche Diagnostics) using Roche Cobas c701 instrumentation. The levels of bioavailable 25(OH)D were calculated from total measured 25(OH)D, DBP, and serum albumin concentrations using the following equation, as described previously (19):
| (1) |
Here, Ka is the affinity constant. We used values of 7 × 108 M–1 for KaDBP and 6 × 105 M–1 for Kaalb.
Bioavailable 25(OH)D was calculated using the following affinity constants: DBP for 25(OH)D = 7 × 108 M–1, DBP for 1,25(OH)2D = 4 × 107 M–1, albumin for 25(OH)D = 6 × 105 M–1, and albumin for 1,25(OH)2D = 5.4 × 104 M–1 and the variables were concentrations of 25(OH)D, 1,25(OH)2D (assigned 0.1 nM in all cases), DBP, and albumin. A MATLAB program implements a system of coupled, nonlinear, algebraic equations for each vitamin D metabolite (19) for extracellular steady state free-ligand modeling, yielding calculated values for the bioavailable and free 25(OH)D metabolites of interest (19).
Statistical analyses
Initially, the baseline demographics and clinical characteristics were reported using standard summary statistics. As all the continuous variables, except patient age, were not normally distributed, the median values along with the IQRs were reported for these variables; counts and percentages were reported for categorical variables. Logistic regression models were then used to determine associations between the demographic and clinical variables and the primary outcome of 28-day mortality. Each possible explanatory variable was tested in a univariate analysis to determine its strength of association with the outcome of interest. Serum concentrations of total 25(OH)D were initially assessed as continuous variables and then by categories defined according to the various published definitions of vitamin D deficiency and sufficiency: that is, <25, 25–49, 50–74, and 75–99 nmol/L plus a category with high values ≥ 100 nmol/L and a group for high values ≥ 100 nmol/L, previously shown to be associated with increased fibroblast growth factor (FGF) 23, which suppresses vitamin D activation via the 1-alpha-hydroxylase (24, 25, 26), with significance determined in the univariate analysis using a chi-square goodness-of-fit. Free 25(OH)D, bioavailable 25(OH)D, and DBP were categorized into quintiles and similarly examined for associations with mortality. All variables identified as being possibly associated with the outcome (P < 0.1) were then taken forward into a multivariable model. The 2 clinical variables that were included in the final model (chronic kidney disease and neurological diseases) have been previously associated with worse clinical outcomes (27). Two different multivariable models were fitted, 1 with vitamin D as a continuous variable and the other with vitamin D as a categorized variable. A further analysis was performed by logistic regression, but with vitamin D as a continuous rather than categorized variable, again with adjustments for age and sex, and plotted with cubic spline smoothing using the package mgcv (28) in R (29). Correlations between 25(OH)D and biochemical parameters were assessed using Spearman rank correlation. Analyses were carried out using SPSS (Version 27.0, IBM Corp), or R version 4.1.1 (29). A 2-sided P value < 0.05 was considered statistically significant.
Ethical approval
The study protocol was approved by the London-Surrey Research Ethics Committee (20/HRA/2282).
Results
Cohort demographics
We included 472 patients with COVID-19, of whom 268 (56.8%) were men. This represented 47.5% of the 992 patients who had been admitted with COVID-19 during the study period of 18 March to 2 November 2020. We only included patients who had surplus sera available for 25(OH)D analysis (for patient flowchart, see Supplemental Figure 1). The 28-day mortality rate was 23.7%. The mean age at death was 73 years (SD, 19 years), and the 28-day mortality rates were 28.0% and 18.5% among men and women, respectively. The univariate analysis showed a significant association between an increased risk of mortality by 28 days and older age, male sex, and preexisting neurological disease ( Table 1). There were no significant associations found with BMI, ethnicity, and other comorbidities (Table 1). There was a trend towards increased mortality in patients with chronic kidney disease, although this was not significant (P = 0.06; Table 1). Increased mortality was recorded in patients with higher baseline concentrations of D-dimer, troponin, C-reactive protein (CRP), and creatinine and in those with lower baseline concentrations of albumin ( Table 2). Of the entire cohort, 76 patients (16.1%) received remdesivir and 128 (27.1%) received dexamethasone ( Table 3). Oxygen therapy was received by 69.1% during their hospital stay, and 76 (16.1%) were treated in the intensive care unit, among whom 56 (11.7% of total) received mechanical ventilation (Table 3).
TABLE 1.
Clinical characteristics of included subjects1
| Variable | Alive | Deceased | P value |
|---|---|---|---|
| N | 360 | 112 | |
| Age | |||
| Mean (SD) | 65 (26) | 73 (19) | 0.003 |
| Range | 19–95 | 34–98 | |
| Sex | |||
| Female | 167 (46.4%) | 37 (33.0%) | 0.02 |
| Male | 193 (53.6%) | 75 (67.0%) | |
| BMI, kg/m2 | |||
| Mean (SD) | 27 (9) | 26 (8) | >0.999 |
| Underweight | 25 (6.9%) | 12 (10.7%) | 0.627 |
| Normal | 97 (26.9%) | 30 (26.7%) | |
| Overweight | 74 (20.6%) | 22 (19.6%) | |
| Obese | 110 (30.6%) | 33 (29.5%) | |
| Not recorded | 54 (15%) | 15 (13.4%) | |
| Ethnicity | |||
| Black and minority | 41 (11.3%) | 12 (10.7%) | >0.999 |
| White | 302 (83.9%) | 94 (83.9%) | |
| Not recorded | 17 (4.7%) | 6 (5.3%) | |
| DM | |||
| No | 270 (75.0%) | 77 (69.8%) | 0.22 |
| Yes | 90 (25.0%) | 35 (30.2%) | |
| Hypertension | |||
| No | 203 (56.4%) | 55.(49.1%) | 0.19 |
| Yes | 156 (43.6%) | 57 (50.9%) | |
| Not recorded | 1 (0.2%) | — | |
| CKD | |||
| No | 301 (83.8%) | 77 (68.8%) | 0.06 |
| Yes | 58 (16.2%) | 25 (31.2%) | |
| Not recorded | 1 (0.2%) | — | |
| IHD | |||
| No | 294 (81.9%) | 88 (78.6%) | 0.49 |
| Yes | 65 (18.1%) | 24 (21.4%) | |
| Not recorded | 1 (0.2%) | — | |
| Smoking status | |||
| Non-/ex-smoker | 311 (86.4%) | 99 (88.4%) | 0.113 |
| Current smoker | 39 (10.8%) | 5 (4.5%) | |
| Not recorded | 10 (2.8%) | 8 (7.1%) | |
| Respiratory | |||
| No | 256 (71.1%) | 83 (74.1%) | 0.576 |
| Yes | 104 (28.9%) | 29 (25.9%) | |
| Neurological | |||
| No | 291 (81.1%) | 76 (67.9%) | 0.003 |
| Yes | 68 (18.9%) | 36 (32.1%) | |
| Not recorded | 1 (0.2%) | — | |
| Liver disease | |||
| No | 339 (94.4%) | 103 (92.0%) | >0.999 |
| Yes | 20 (5.6%) | 9 (8.0%) | |
| Not recorded | 1 (0.2%) | — | |
| Active malignancy | |||
| No | 330 (91.9%) | 102 (91.1%) | 0.84 |
| Yes | 29 (8.1%) | 10 (8.9%) | |
| Not recorded | 1 (0.2%) | — | |
A univariate analysis was performed to compare variables across alive and deceased categories using a chi-squared test for categorical variables and a Mann-Whitney U test for continuous variables. Categorical data are represented as counts and percentages. CKD, chronic kidney disease; DM, diabetes mellitus; IHD, ischemic heart disease.
TABLE 2.
Biochemical and radiological parameters among survivors and nonsurvivors1
| Variable | Alive, n = 360 (76.3%) | Deceased, n = 112 (23.7%) | P value |
|---|---|---|---|
| D-dimer, ng/ml, median (IQR) | 781 (836) | 1668 (2774) | <0.001 |
| Troponin, ng/L, median (IQR) | 21 (21.25) | 23 (37.5) | 0.003 |
| CRP, mg/L, median (IQR) | 49.5 (88.75) | 111 (125) | <0.001 |
| Albumin, g/L, median (IQR) | 38 (7) | 35 (8) | <0.001 |
| ALT, iu/L, median (IQR) | 24 (23) | 22 (21) | 0.47 |
| Creatinine, umol/L, median (IQR) | 73 (35) | 101.5 (76) | <0.001 |
| eGFR, ml/min/1.73m2 | |||
| ≤90 | 220 (62.0%) | 85 (77.3%) | |
| >90 | 135 (38.0%) | 25 (22.7%) | 0.003 |
| CXR changes | |||
| No | 63 (42.6%) | 7 (18.0%) | |
| Yes | 85 (57.4%) | 32 (82.0%) | 0.005 |
| CT changes | |||
| No | 4 (10.8%) | — | |
| Yes | 33 (89.2%) | 4 (100.0%) | >0.999 |
Values are based on values at hospital admission. COVID-19 changes on chest X-ray or CT scan were considered present on any imaging during hospital admission. A univariate analysis was performed to compare variables across alive and deceased categories using a chi-squared test for categorical variables and a Mann-Whitney U test for continuous variables. Categorical data are represented as counts and percentages and continuous variables as medians and IQRs. ALT, alanine aminotransferase; COVID-19, coronavirus disease 2019; CRP, C-reactive protein; CT, computed tomography; CXR, chest X-ray; eGFR, estimated glomerular filtration rate.
TABLE 3.
Treatments received by COVID-19 patients1
| Variable | Alive, n = 360 (76.3%) | Deceased, n = 112 (23.7%) | P value |
|---|---|---|---|
| Remdesivir | |||
| No | 305 (85.0%) | 91 (81.3%) | |
| Yes | 55 (15%) | 21 (18.7%) | 0.37 |
| Dexamethasone | |||
| No | 268 (74.7%) | 76 (67.9%) | |
| Yes | 92 (25.3%) | 36 (32.1%) | 0.18 |
| Vitamin D supplements | |||
| No | 256 (71.1%) | 80 (71.4%) | >0.999 |
| Yes | 100 (27.8%) | 31 (27.7%) | |
| Not recorded | 4 (1.1%) | 1 (0.8%) | |
| Oxygen | |||
| No | 144 (40.0%) | 2 (1.8%) | |
| Yes | 216 (60.0%) | 110 (98.2%) | <0.001 |
| ICU | |||
| No | 318 (88.3%) | 78 (68.8%) | |
| Yes | 42 (11.7%) | 34 (31.2%) | <0.001 |
| CPAP | |||
| No | 321 (89.2%) | 88 (78.6%) | |
| Yes | 39 (10.8%) | 24 (21.4%) | 0.48 |
| Mechanical ventilation | |||
| No | 334 (93.0%) | 82 (73.2%) | |
| Yes | 26 (7.0%) | 30 (26.8%) | <0.001 |
A univariate analysis was performed to compare variables across alive and deceased categories using a chi-squared test for categorical variables and a Mann-Whitney U test for continuous variables. Categorical data are represented as counts and percentages and continuous variables as medians and IQRs. COVID-19, coronavirus disease 2019; CPAP, continuous positive airways pressure; ICU, intensive care unit.
Vitamin D status
The overall prevalence of vitamin D deficiency [25(OH)D < 50 nmol/L] in the cohort was 58.1%, while 25.0% were severely deficient [25(OH)D < 25 nmol/L]. There was no significant difference between 25(OH)D concentrations amongst nonsurvivors (median, 39.5 nmol/L; IQR, 48.5 nmol/L) compared with survivors (median, 43.0 nmol/L; IQR, 41.0 nmol/L; P = 0.11; Table 4). Unadjusted, there was also no significant difference between frequencies of 25(OH)D vitamin D deficiency [25(OH)D < 50 nmol/L] amongst those who died (71/112; 63.4%) compared with survivors (204/360; 56.7%; P = 0.15). Total 25(OH)D, analyzed by 5 concentration categories according to the different definitions of sufficiency [<25, 25–49, 50–74, and 75–99 nmol/L, plus a group for high values ≥ 100 nmol/L, previously shown to be associated with increased FGF23 (24, 25, 26)], did suggest an association with 28-day mortality, with the lowest mortality for those with a 25(OH)D concentration of 50–74 nmol/L (P = 0.04; Table 4; Figure 1). This pattern remained after excluding patients who received prescribed vitamin D supplementation (Supplemental Figure 2). A similar analysis with 25(OH)D as a continuous variable was not significant. There was no significant association between free or bioavailable 25(OH)D and mortality (Table 4).
TABLE 4.
Levels of total 25(OH)D, DBP, and free and bioavailable 25(OH)D among COVID-19 patients1
| Variable | Alive | Deceased | P value |
|---|---|---|---|
| 25(OH)D, nmol/L, median (IQR) | 43.0 (41.0) | 39.5 (48.5) | 0.11 |
| 25(OH)D deficiency < 50 nmol/L, n (%) | 204 (56.7%) | 71 (63.4%) | 0.15 |
| 25(OH)D by categories | — | — | 0.04 |
| <25 nmol/L, n (%) | 85 (23.6%) | 33 (29.5%) | |
| 25–49 nmol/L, n (%) | 119 (33.1%) | 38 (33.9%) | |
| 50–74 nmol/L, n (%) | 99 (27.4%) | 16 (14.3%) | |
| 75–99 nmol/L, n (%) | 47 (13.0%) | 17 (15.2%) | |
| >100 nmol/L, n (%) | 10 (2.8%) | 8 (7.4%) | |
| Free 25(OH)D, pmol/L, median (IQR) | 3.31 (3.93) | 3.73 (4.92) | 0.28 |
| Free 25(OH)D by quintile categories | — | — | 0.32 |
| 0–1.55 pmol/L, n (%) | 66 (20.4%) | 19 (21.1%) | |
| 1.56–2.68 pmol/L, n (%) | 68 (21.0%) | 12 (13.3%) | |
| 2.69–4.10 pmol/L, n (%) | 67 (20.7%) | 18 (20.0%) | |
| 4.11–6.11 pmol/L, n (%) | 64 (19.8%) | 17 (18.9%) | |
| >6.11 pmol/L, n (%) | 59 (18.2%) | 24 (26.7%) | |
| Bioavailable 25(OH)D, nmol/L, median (IQR) | 1.07 (1.26) | 1.01 (1.34) | 0.81 |
| Bioavailable 25(OH)D by quintile categories | — | — | 0.39 |
| 0–0.459 nmol/L, n (%) | 62 (19.1%) | 20 (22.2%) | |
| 0.46–0.81 nmol/L, n (%) | 70 (21.6%) | 12 (13.3%) | |
| 0.82–1.3 nmol/L, n (%) | 64 (19.8%) | 23 (25.6%) | |
| 1.31–2.037 nmol/L, n (%) | 64 (19.8%) | 16 (18.9%) | |
| >2.038 pmol/L, n (%) | 64 (19.8%) | 19 (21.1%) | |
| DBP, mg/L, median (IQR) | 858 (707.8) | 707 (441.0) | 0.005 |
| DBP by quintile categories | — | — | 0.003 |
| 0–579.9 mg/L | 66 (20.3%) | 21 (22.3%) | |
| 580–733.6 mg/L | 69 (21.2%) | 16 (17.0%) | |
| 733.61–960.4 mg/L | 51 (15.7%) | 30 (31.2%) | |
| 960.41–1615 mg/L | 74 (22.8%) | 10 (11.9%) | |
| >1615 mg/L | 65 (20.0%) | 17 (18.1%) |
Note that sufficient sera for DBP, and hence free and bioavailable 25(OH)D, were only available for 325/360 (90.3%) of those who were alive and 94/112 (84%) of those who were deceased. A univariate analysis was performed using a chi-squared goodness-of-fit test. Categorical data are represented as counts and percentages and continuous variables as medians and IQRs. Levels of total 25(OH)D were categorized based on published definitions of adequacy and free 25(OH)D; bioavailable 25(OH)D and DBP were categorized as quintiles. COVID-19, coronavirus disease 2019; DBP, vitamin D–binding protein; 25(OH)D, 25-hydroxyvitamin D.
FIGURE 1.
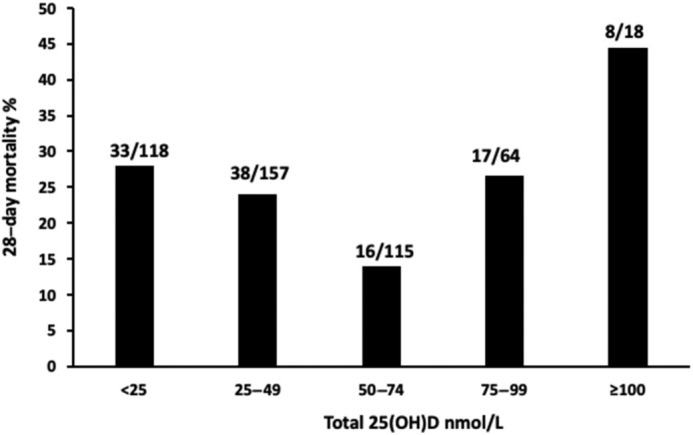
Bar graph of 28-day mortality (%) in patients, grouped according to serum 25(OH)D concentration on admission (unadjusted). It can be seen that the mortality rate was lowest among patients with a serum 25(OH)D concentration between 50–74 nmol/L. Serum 25(OH)D was measured in 472 individuals with COVID-19, among whom 360 were survivors and 112 were nonsurvivors. COVID-19, coronavirus disease 2019; 25(OH)D, 25-hydroxyvitamin D.
A multivariable analysis was therefore performed to investigate the associations between mortality and 25(OH)D concentration ranges, with adjustments for age, sex and comorbidities. This showed increased mortality in those with 25(OH)D concentrations < 25 nmol/L (OR, 2.37; 95% CI, 1.17–4.78; P = 0.016) and in those with 25(OH)D concentrations >100 nmol/L (OR, 4.65; 95% CI, 1.51–14.34; P = 0.007) compared with 25(OH)D concentrations of 50-74 nmol/L, with the latter selected as reference since it reflects the typical summer range for 25(OH)D in healthy adults (30). It is important to note, though, that the number of patients with 25(OH)D concentrations >100 nmol/l was small (n = 18). Levels of 25(OH)D between 25–49 and 75–99 nmol/L were not associated with significant differences in mortality compared with levels of 50–74 nmol/L ( Table 5). A logistic regression analysis of mortality, with adjustments for age and sex, but with vitamin D set as a continuous variable, did not show an association between mortality and 25(OH)D levels ( Figure 2). Free 25(OH)D and bioavailable 25(OH)D concentrations, grouped by quintiles, remained unassociated with mortality in a model adjusted for age, sex, and comorbidities (Supplemental Tables 1 and 2).
TABLE 5.
Multivariable analysis of age, sex, comorbidities, and total 25(OH)D with 28-day mortality1
| Variable | Regression Coefficient | OR | 95% CI | P value |
|---|---|---|---|---|
| Age | 0.041 | 1.04 | 1.03–1.06 | <0.001 |
| Male sex | 0.685 | 1.98 | 1.22–3.22 | 0.006 |
| CKD | −0.345 | 0.71 | 0.41–1.22 | 0.213 |
| Neurological disease | −0.280 | 1.35 | 0.45–1.27 | 0.288 |
| 25(OH)D, 50–74 nmol/L | — | 1.00 (ref) | — | 0.049 |
| 25(OH)D, <25 nmol/L | 0.861 | 2.37 | 1.17–4.78 | 0.016 |
| 25(OH)D, 25–49 nmol/L | 0.626 | 1.87 | 0.95–3.67 | 0.069 |
| 25(OH)D, 75–99 nmol/L | 0.786 | 2.19 | 0.97–4.98 | 0.060 |
| 25(OH)D, ≥100 nmol/L | 1.538 | 4.65 | 1.51–14.34 | 0.007 |
A logistic regression analysis was performed to determine associations between the variables and 28-day mortality. CKD, chronic kidney disease; 25(OH)D, 25-hydroxyvitamin D.
FIGURE 2.
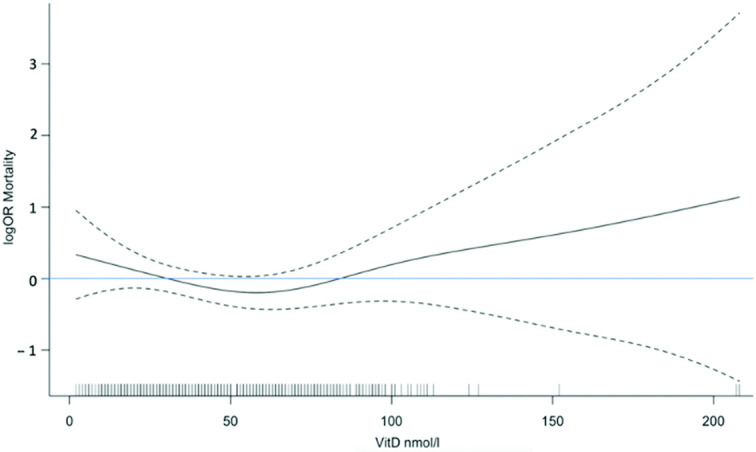
Log OR for 28-day mortality according to serum 25(OH)D concentration on admission, compared with mean vitamin D value (47.4 nmol/L) as the reference, adjusted for age and sex, analyzed by logistic regression with cubic spline smoothing, and showing 95% CIs. This analysis, performed with 25(OH)D as a continuous variable, did not show a significant association with mortality from COVID-19. Serum 25(OH)D was measured in 472 individuals with COVID-19, among whom 360 were survivors and 112 were nonsurvivors. COVID-19, coronavirus disease 2019; 25(OH)D, 25-hydroxyvitamin D.
DBP and mortality
On univariate analysis, median DBP concentrations were lower in patients who died (P = 0.005; Table 4; Figure 3). However, a multivariable analysis with adjustments for 25(OH)D, age, sex, and significant comorbidities did not show an association between DBP and mortality (Supplemental Table 3).
FIGURE 3.
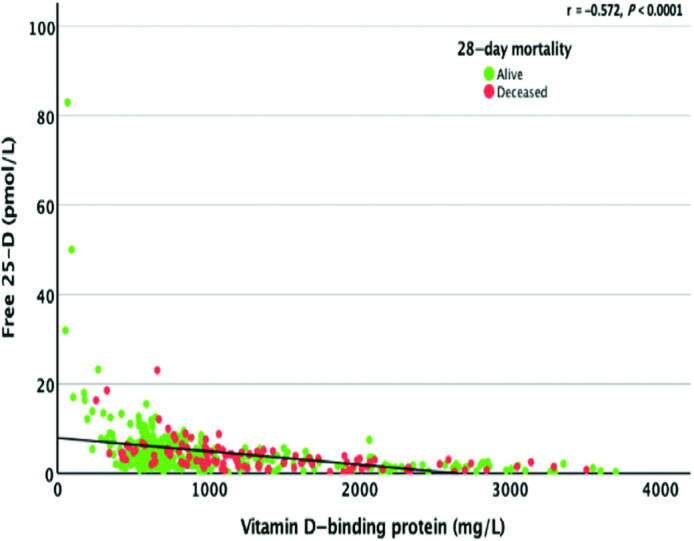
Association between serum-free 25(OH)D and DBP concentrations. The correlation between free 25(OH)D and DBP was assessed using the Spearman rank correlation. DBP and free 25(OH)D values were only available for 325/360 (90.3%) of those who were alive and 94/112 (84%) of those who were deceased. DBP, vitamin D–binding protein; 25(OH)D, 25-hydroxyvitamin D.
Associations of total, free, and bioavailable 25(OH)D with serum DBP, CRP, and albumin
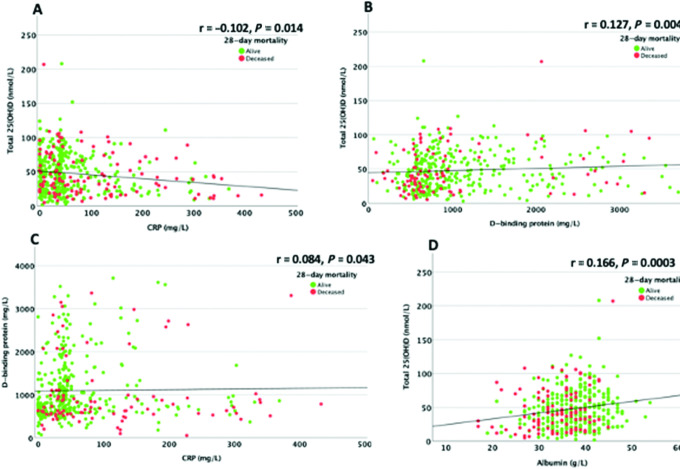
We investigated associations between total, free, and bioavailable 25(OH)D; DBP; and albumin, with baseline CRP as a marker of inflammation. The relationship between the DBP concentration and free 25(OH)D concentration was significant (Spearman’s rho = −0.572) but only sizeable at very low concentrations of DBP (<500 mg/L; Figure 3). There was no significant relationship between free 25(OH)D and serum albumin concentrations (Spearman’s rho = 0.048; Supplemental Figure 3).
Serum 25(OH)D concentrations correlated with baseline serum CRP values (Spearman’s rho = −0.102), in keeping with a negative acute-phase response but with a modest size effect; the mean 25(OH)D concentration fell from approximately 50 nmol/L at CRP levels < 5 mg/L to approximately 40 nmol/L at a CRP of 200 mg/L ( Figure 4A). Serum 25(OH)D concentrations correlated with DBP concentrations (Spearman’s rho = 0.127), but again with a modest size effect (Figure 4B). DBP concentrations showed a modest correlation with CRP (Spearman’s rho = −0.084; Figure 4C). Serum 25(OH)D concentrations correlated with serum albumin (Spearman’s rho = 0.166), with a stronger size effect than that observed for the DBP association, and the mean 25(OH)D concentration falling from approximately 50 nmol/L at an albumin concentration of 40 g/L to approximately 40 nmol/L at an albumin concentration of 30 g/L (Figure 4D). There was also an association between serum albumin concentration and baseline CRP value (Spearman’s rho = −0.212; Supplemental Figure 4). However, the difference in serum albumin concentrations between survivors (median, 38 g/L; IQR, −7 g/L) and nonsurvivors (median, 35 g/L; IQR, −8 g/L), although significant, was only modest (Table 2).
FIGURE 4.
Association between serum 25(OH)D and CRP serum albumin and associations between DBP and serum 25(OH)D and CRP. (A) Association between serum 25(OH)D concentrations and baseline CRP. There is only a modest correlation between CRP and total 25(OH)D. The correlation between total 25(OH)D and CRP was assessed using Spearman rank correlation. Both serum 25(OH)D and CRP were measured in 472 individuals with COVID-19, among whom 360 were survivors and 112 were nonsurvivors. (B) Associations between serum 25(OH)D concentrations and serum DBP. The correlation between free 25(OH)D and DBP was assessed using Spearman rank correlation. DBP and free 25(OH)D were only available for 325/360 (90.3%) of those who were alive and 94/112 (84%) of those who were deceased. (C) Association between serum DBP and CRP. The correlation between DBP and CRP was assessed using Spearman rank correlation. DBP measurements were only available for 325/360 (90.3%) of those who were alive and 94/112 (84%) of those who were deceased. (D) Association between serum 25(OH)D and serum albumin. The correlation between 25(OH)D and albumin was assessed using Spearman rank correlation. Both serum 25(OH)D and albumin were measured in 472 individuals with COVID-19, among whom 360 were survivors and 112 were nonsurvivors. COVID-19, coronavirus disease 2019; CRP, C-reactive protein; DRP, vitamin D–binding protein; 25(OH)D, 25-hydroxyvitamin D.
Vitamin D supplementation and mortality
Prescribed vitamin D supplements were being taken by 131 (27.8%) patients. Data were not available for use of supplements purchased over the counter. The mean age of death was significantly higher (80.2 years; SD, 11.9 years) amongst those taking prescribed supplements compared with those not taking prescribed supplements (71.1 years; SD, 12.0 years; P < 0.001), but the average age of all people taking prescribed supplements (71.5 years; SD, 16.0 years) was greater than that of those not taking prescribed supplements (62.0 years; SD, 18.1 years; P < 0.0001), and the analysis of 28-day mortality did not show a significant association with prescribed supplements after adjustments for age (OR, 0.67; 95% CI, 0.40–1.12; P = 0.12). There was an association between prescribed supplement usage and 25(OH)D concentrations (supplement usage of 0% in those with a 25(OH)D concentration ≤25 nmol/L; 23% in those with a concentration of 25–49 nmol/L; 32% in those with a concentration of 50–74 nmol/L; 59% in those with a concentration of 75–99 nmol/L; and 53% in those with a concentration ≥100 nmol/L).
Discussion
In this retrospective, single-center cohort study, we confirm that older age and male sex are associated with increased 28-day mortality from COVID-19, consistent with the results of other studies (30, 31, 32). We found no significant relationship between overall 25(OH)D deficiency (<50 nmol/L) and mortality from COVID-19. However, when 25(OH)D values on admission were categorized according to the varying definitions of deficiency (<25, <50, or <75 nmol/l) plus an additional category for high values > 100 nmol, a multivariable analysis, adjusted for age, sex, and comorbidities, showed increased mortality amongst those with very low (<25 nmol/l) or high (>100 nmol/l) serum 25(OH)D concentrations compared to those with 25(OH)D concentrations of 50–74 nmol/L, which was selected as the reference since it approximates the typical range for 25(OH)D seen from June to December in healthy, British adults (30). The increased mortality in patients with serum 25(OH)D concentrations < 25 nmol/L is consistent with that reported in some other studies (15, 31), but patients with high serum 25(OH)D concentrations ≥ 100 nmol/L also had increased mortality in our cohort. This needs to be interpreted with caution, though, since the number of patients was small (n = 18) in the highest category (>100 nmol/l). Moreover, a multivariable analysis with 25(OH)D as a continuous variable did not reach significance.
Many studies have reported on 25(OH)D status, in terms of deficient compared with sufficient concentrations, in association with COVID-19 outcomes in hospitalized patients, with contradictory findings (12, 13). There are also some data on COVID-19 outcomes in relation to 25(OH)D concentration ranges in hospitalized patients: a study from New York showed a significantly increased likelihood of needing oxygen support in patients with 25(OH)D concentrations < 50 nmol/L (16); a Brazilian study showed a nonsignificant trend towards increased mortality at both very low and very high 25(OH)D levels (17); an Italian study reported a progressive fall in mortality with high 25(OH)D levels but was based only on 9 deaths (14); and finally, another study from Italy reported a progressive increase in mortality with increasing 25(OH)D concentrations after adjustments for various factors, including baseline CRP and severe pneumonia (32).
U-shaped associations between serum 25(OH)D concentrations and various clinical outcomes, including all-cause mortality, have been reported previously but are highly controversial. The US Academy of Medicine noted in 2010 that several studies had reported such an association, and concluded that the “data are clearly suggestive of a U-shaped or reverse-J-shaped risk curve between serum 25OHD level and all-cause mortality; increases in risk are suggested at thresholds in the range of 75 to 120 nmol/L for the White population, with lower levels for the Black population” (33). A subsequent general-practice study from Denmark of 247,574 people, with an average age of 51 years who were followed over 3 years, confirmed a U-shaped association, with the lowest all-cause mortality rates again seen for those with 25(OH)D concentrations of 50–60 nmol/L (25). However, a study of 24,094 hospitalized patients from Boston showed a U-shaped association, with significantly increased mortality only seen at serum 25(OH)D concentrations ≥ 60 ng/ml, equivalent to 150 nmol/L (24). A U-shaped association between serum 25(OH)D and a 2.7-fold increased risk of bone fracture has also been shown in older men at 25(OH)D concentrations > 72 nmol/L (34), and a dose-ranging study of vitamin D supplementation for prevention of falls has reported worse outcomes with higher dosing regimens (35). Also, a meta-analysis of standardized serum 25(OH)D measurements in 26,916 individuals from 8 prospective European studies showed the lowest all-cause mortality rate for 25(OH)D concentrations of 75–100 nmol/l, but with no significant increase in mortality at levels >100 nmol/l (36). There is a biologically plausible explanation for the U-shaped curve: the phosphaturic hormone FGF23 is typically induced when the serum 25(OH)D concentration rises above 100 nmol/L (37), which leads to a marked suppression of the vitamin D–activating 1-alpha-hydroxylase.
In our study, there was no correlation between serum free vitamin D and mortality. There are several possible explanations for this: 1) protein-bound vitamin D can be taken up by immune cells expressing megalin (38) and activated by these cells through endogenous 1α-hydroxylase activity; and 2) serum concentrations of free 25(OH)D are more than 10,000-fold lower than those of total 25(OH)D, while bioavailable—that is, albumin-bound 25(OH)D—for which there was also no association with mortality in our patients, is present at even lower serum concentrations. A limitation of our study is that we did not directly measure free 25(OH)D levels but, as indicated previously, this is an extremely difficult assay. Moreover, previous studies have suggested that measurement of free and bioavailable 25(OH)D may not offer additional advantages over standard measurement of total 25(OH)D for diagnosing vitamin D deficiency (39).
Our study has several important strengths. Using a large sample size comprising 472 subjects has made this the largest study to date to investigate the association between serum vitamin D status and COVID-19 outcomes. In addition, we have also estimated DBP and free and bioavailable 25(OH)D concentrations. Importantly, vitamin D levels were measured contemporaneously, unlike in some other studies, where associations were derived from previously measured vitamin D levels (16, 40). There are, however, some limitations. Firstly, our study is retrospective and we only included patients with available surplus sera, which may have introduced bias. Moreover, the sample size, though considerable, is probably underpowered to detect small differences in overall serum 25(OH)D levels between the groups; thus, according to a retrospective analysis, to detect with statistical significance a difference of 3.5 nmol/l 25(OH)D, as found between survivors and nonsurvivors in this study, would have required over 500 patients per group. Our study was also probably underpowered to evaluate any interaction between COVID-19 severity, vitamin D levels, and other known risk factors, such as obesity, ethnicity, and the presence of comorbidities such as hypertension, diabetes, and chronic kidney disease. We noted a lower proportion of current smokers in patients who died from COVID-19, although this did not reach statistical significance. This is in keeping with a large UK study that showed a lower risk of COVID-19 mortality among current smokers (27), though other studies report an opposite association (41). Overall, these differences are likely explained by variations in sample size and patient populations.
In summary, we found no significant relationship between overall 25(OH)D deficiency (<50 nmol/L) and mortality from COVID-19, but there were associations noted when 25(OH)D status was assessed by 5 concentration categories, although these did not retain significance when vitamin D was analyzed as a continuous variable. There are several possible explanations: 1) there may be no relationship between vitamin D status and COVID-19 outcome; 2) vitamin D deficiency may impact the risk for hospitalization with COVID-19 but not its subsequent course; and 3) the relationship between vitamin D status and outcome may be complex, with both low and high 25(OH)D levels negatively impacting outcomes. Further studies, whether interventional or observational, should consider this last possibility as being at least plausible.
Acknowledgments
The authors’ responsibilities were as follows – SS, JMR, MP, JMT, AMM: were involved in the study design and initial drafting of the manuscript; SL: was involved in the data analysis and is the guarantor of the article; and all authors: revised the manuscript and read and approved the final manuscript. MP has received partnership funding for a MRC Clinical Pharmacology Training Scheme (cofunded by MRC and Roche, UCB, Eli Lilly, and Novartis); has received a PhD studentship jointly funded by EPSRC and Astra Zeneca; has received grant funding from Vistagen Therapeutics; has unrestricted educational grant support for the UK Pharmacogenetics and Stratified Medicine Network from Bristol-Myers Squibb and UCB; has developed an Human Leukocyte Antigen (HLA) genotyping panel with MC Diagnostics, but does not benefit financially from this; and is part of the IMI Consortium ARDAT (www.ardat.org), although none of these funding sources were used for the current paper. JMR holds a patent with the University of Liverpool and Provexis UK for use of a soluble fiber preparation as maintenance therapy for Crohn’s disease plus a patent for its use in antibiotic-associated diarrhea; and holds a patent with the University of Liverpool and others in relation to use of modified heparins in cancer therapy. SS has received speaker fees from MSD, Actavis, Abbvie, Dr Falk Pharmaceuticals, Janssen, Takeda, Boehringer-Ingelheim, and Shire; has received educational grants from MSD, Abbvie, and Actavis; and is an advisory board member for Abbvie, Dr Falk Pharmaceuticals, Takeda, Janssen, and Vifor Pharmaceuticals.
Data Availability
Data described in the manuscript will be made publicly and freely available without restriction at: https://drive.google.com/file/d/10lnd2MKQUhakKBERLpaIyxrz2Du502c2/view?usp=sharing. The code book and analytic code are available from the corresponding author on reasonable request.
Footnotes
This research was funded by an internal departmental grant from Liverpool University Hospitals NHS Foundation Trust.
The funders had no input into the study design or final analysis.
Supplemental Figures 1–4 and Supplemental Tables 1–3 are available from the “Supplementary data” link in the online posting of the article and from the same link in the online table of contents at https://academic.oup.com/ajcn/.
SUPPORTING INFORMATION
nqac027_supplemental_file
References
- 1.Johns Hopkins University; Baltimore, MD: 2020. Center for Systems Science and Engineering at Johns Hopkins University. COVID-19 dashboard [Internet]. Available from: https://coronavirus.jhu.edu/map.html. [Google Scholar]
- 2.Rhodes JM, Subramanian S, Laird E, Griffin G, Kenny RA. Perspective: Vitamin D deficiency and COVID-19 severity - plausibly linked by latitude, ethnicity, impacts on cytokines, ACE2 and thrombosis. J Intern Med. 2021;289(1):97–115. doi: 10.1111/joim.13149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Correale J, Ysrraelit MC, Gaitan MI. Gender differences in 1,25 dihydroxyvitamin D3 immunomodulatory effects in multiple sclerosis patients and healthy subjects. J Immunol. 2010;185(8):4948–4958. doi: 10.4049/jimmunol.1000588. [DOI] [PubMed] [Google Scholar]
- 4.Carleton T, Cornetet J, Huybers P, Meng KC, Proctor J. Global evidence for ultraviolet radiation decreasing COVID-19 growth rates. Proc Natl Acad Sci. 2021;118(1) doi: 10.1073/pnas.2012370118. e2012370118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rhodes J, Dunstan F, Laird E, Subramanian S, Kenny RA. COVID-19 mortality increases with northerly latitude after adjustment for age suggesting a link with ultraviolet and vitamin D. BMJ Nutr Prev Health. 2020;3(1):118–120. doi: 10.1136/bmjnph-2020-000110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Griffin G, Hewison M, Hopkin J, Kenny R, Quinton R, Rhodes J, Subramanian S, Thickett D. Vitamin D and COVID-19: Evidence and recommendations for supplementation. R Soc Open Sci. 2020;7(12):201912. doi: 10.1098/rsos.201912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jolliffe DA, Camargo CA, Sluyter JD, Aglipay M, Aloia JF, Ganmaa D, Bergman P, Borzutzky A, Damsgaard CT, Dubnov-Raz G et al. Vitamin D supplementation to prevent acute respiratory infections: Systematic review and meta-analysis of aggregate data from randomised controlled trials. medRxiv. [accessed 2022 February 9]. doi: 10.1101/2020.07.14.20152728. [DOI] [PubMed]
- 8.Vo P, Koppel C, Espinola JA, Mansbach JM, Celedon JC, Hasegawa K, Bair-Merritt M, Camargo CA., Jr Vitamin D status at the time of hospitalization for bronchiolitis and its association with disease severity. J Pediatr. 2018;203:416–422. doi: 10.1016/j.jpeds.2018.07.097. e1. [DOI] [PubMed] [Google Scholar]
- 9.Greiller CL, Martineau AR. Modulation of the immune response to respiratory viruses by vitamin D. Nutrients. 2015;7(6):4240–4270. doi: 10.3390/nu7064240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Greiller CL, Suri R, Jolliffe DA, Kebadze T, Hirsman AG, Griffiths CJ, Johnston SL, Martineau AR. Vitamin D attenuates rhinovirus-induced expression of intercellular adhesion molecule-1 (ICAM-1) and platelet-activating factor receptor (PAFR) in respiratory epithelial cells. J Steroid Biochem Mol Biol. 2019;187:152–159. doi: 10.1016/j.jsbmb.2018.11.013. [DOI] [PubMed] [Google Scholar]
- 11.Parekh D, Patel JM, Scott A, Lax S, Dancer RC, D’Souza V, Greenwood H, Fraser WD, Gao F, Sapey E, et al. Vitamin D deficiency in human and murine sepsis. Crit Care Med. 2017;45(2):282–289. doi: 10.1097/CCM.0000000000002095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kazemi A, Mohammadi V, Aghababaee SK, Golzarand M, Clark CCT, Babajafari S. Association of vitamin D status with SARS-CoV-2 infection or COVID-19 severity: A systematic review and meta-analysis. Adv Nutr. 2021;12(5):1636–1658. doi: 10.1093/advances/nmab012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bassatne A, Basbous M, Chakhtoura M, El Zein O, Rahme M, El-Hajj Fuleihan G. The link between COVID-19 and vitamin D (VIVID): A systematic review and meta-analysis. Metabolism. 2021;119:154753. doi: 10.1016/j.metabol.2021.154753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Barassi A, Pezzilli R, Mondoni M, Rinaldo RF, Dav IM, Cozzolino M, Melzi D, Eril GV, Centanni S. Vitamin D in severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) patients with non-invasive ventilation support. Panminerva Med[accessed 2022 February 9]. doi: 10.23736/S0031-0808.21.04277-4. [DOI] [PubMed]
- 15.Carpagnano GE, Di Lecce V, Quaranta VN, Zito A, Buonamico E, Capozza E, Palumbo A, Di Gioia G, Valerio VN, Resta O. Vitamin D deficiency as a predictor of poor prognosis in patients with acute respiratory failure due to COVID-19. J Endocrinol Invest. 2021;44(4):765–771. doi: 10.1007/s40618-020-01370-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gavioli EM, Miyashita H, Hassaneen O, Siau E. An evaluation of serum 25-hydroxy vitamin D levels in patients with COVID-19 in New York City. J Am Coll Nutr[accessed 2022 February 8]. doi: 10.1080/07315724.2020.1869626. [DOI] [PMC free article] [PubMed]
- 17.Reis BZ, Fernandes AL, Sales LP, Santos MD, Dos Santos CC, Pinto AJ, Goessler KF, Franco AS, Duran CSC, Silva CBR, et al. Influence of vitamin D status on hospital length of stay and prognosis in hospitalized patients with moderate to severe COVID-19: A multicenter prospective cohort study. Am J Clin Nutr. 2021;114(2):598–604. doi: 10.1093/ajcn/nqab151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bikle DD, Schwartz J. Vitamin D binding protein, total and free vitamin D levels in different physiological and pathophysiological conditions. Front Endocrinol. 2019;10:317. doi: 10.3389/fendo.2019.00317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chun RF, Peercy BE, Adams JS, Hewison M. Vitamin D binding protein and monocyte response to 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D: Analysis by mathematical modeling. PLoS One. 2012;7(1):e30773. doi: 10.1371/journal.pone.0030773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bouillon R, Schuit F, Antonio L, Rastinejad F. Vitamin D binding protein: A historic overview. Front Endocrinol. 2019;10:910. doi: 10.3389/fendo.2019.00910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kim S, McClave SA, Martindale RG, Miller KR, Hurt RT. Hypoalbuminemia and clinical outcomes: What is the mechanism behind the relationship? Am Surg. 2017;83(11):1220–1227. doi: 10.1177/000313481708301123. [DOI] [PubMed] [Google Scholar]
- 22.Chun RF, Shieh A, Gottlieb C, Yacoubian V, Wang J, Hewison M, Adams JS. Vitamin D binding protein and the biological activity of vitamin D. Front Endocrinol. 2019;10:718. doi: 10.3389/fendo.2019.00718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Roth DE, Abrams SA, Aloia J, Bergeron G, Bourassa MW, Brown KH, Calvo MS, Cashman KD, Combs G, De-Regil LM, et al. Global prevalence and disease burden of vitamin D deficiency: A roadmap for action in low- and middle-income countries. Ann NY Acad Sci. 2018;1430(1):44–79. doi: 10.1111/nyas.13968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Amrein K, Quraishi SA, Litonjua AA, Gibbons FK, Pieber TR, Camargo CA, Jr., Giovannucci E, Christopher KB. Evidence for a U-shaped relationship between prehospital vitamin D status and mortality: A cohort study. J Clin Endocrinol Metab. 2014;99(4):1461–1469. doi: 10.1210/jc.2013-3481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Durup D, Jorgensen HL, Christensen J, Schwarz P, Heegaard AM, Lind B. A reverse J-shaped association of all-cause mortality with serum 25-hydroxyvitamin D in general practice: The CopD study. J Clin Endocrinol Metab. 2012;97(8):2644–2652. doi: 10.1210/jc.2012-1176. [DOI] [PubMed] [Google Scholar]
- 26.Zittermann A, Iodice S, Pilz S, Grant WB, Bagnardi V, Gandini S. Vitamin D deficiency and mortality risk in the general population: A meta-analysis of prospective cohort studies. Am J Clin Nutr. 2012;95(1):91–100. doi: 10.3945/ajcn.111.014779. [DOI] [PubMed] [Google Scholar]
- 27.Williamson EJ, Walker AJ, Bhaskaran K, Bacon S, Bates C, Morton CE, Curtis HJ, Mehrkar A, Evans D, Inglesby P, et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature. 2020;584(7821):430–436. doi: 10.1038/s41586-020-2521-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wood S. 2nd edition. Chapman and Hall/CRC; Vienna, Austria: 2017. Generalized additive models: An introduction with R. [Google Scholar]
- 29.R Development Core Team . R Foundation for Statistical Computing; Vienna, Austria: 2021. R: A language and environment for statistical computing. [Google Scholar]
- 30.Hypponen E, Power C. Hypovitaminosis D in British adults at age 45 y: Nationwide cohort study of dietary and lifestyle predictors. Am J Clin Nutr. 2007;85(3):860–868. doi: 10.1093/ajcn/85.3.860. [DOI] [PubMed] [Google Scholar]
- 31.Baktash V, Hosack T, Patel N, Shah S, Kandiah P, Van Den Abbeele K, Mandal AKJ, Missouris CG. Vitamin D status and outcomes for hospitalised older patients with COVID-19. Postgrad Med J. 2021;97(1149):442–447. doi: 10.1136/postgradmedj-2020-138712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cereda E, Bogliolo L, Klersy C, Lobascio F, Masi S, Crotti S, De Stefano L, Bruno R, Corsico AG, Di Sabatino A, et al. Vitamin D 25OH deficiency in COVID-19 patients admitted to a tertiary referral hospital. Clin Nutr. 2021;40(4):2469–2472. doi: 10.1016/j.clnu.2020.10.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Institute of Medicine Committee to Review Dietary Reference Intakes for Vitamin D and Calcium . National Academies Press; Washington, DC: 2010. Dietary reference intakes for calcium and vitamin D. [Google Scholar]
- 34.Bleicher K, Cumming RG, Naganathan V, Blyth FM, Le Couteur DG, Handelsman DJ, Waite LM, Seibel MJ. U-shaped association between serum 25-hydroxyvitamin D and fracture risk in older men: Results from the prospective population-based CHAMP study. J Bone Miner Res. 2014;29(9):2024–2031. doi: 10.1002/jbmr.2230. [DOI] [PubMed] [Google Scholar]
- 35.Appel LJ, Michos ED, Mitchell CM, Blackford AL, Sternberg AL, Miller ER, 3rd, Juraschek SP, Schrack JA, Szanton SL, Charleston J, et al. The effects of four doses of vitamin D supplements on falls in older adults: A response-adaptive, randomized clinical trial. Ann Intern Med. 2021;174(2):145–156. doi: 10.7326/M20-3812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gaksch M, Jorde R, Grimnes G, Joakimsen R, Schirmer H, Wilsgaard T, Mathiesen EB, Njolstad I, Lochen ML, Marz W, et al. Vitamin D and mortality: Individual participant data meta-analysis of standardized 25-hydroxyvitamin D in 26916 individuals from a European consortium. PLoS One. 2017;12(2):e0170791. doi: 10.1371/journal.pone.0170791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zittermann A, Berthold HK, Pilz S. The effect of vitamin D on fibroblast growth factor 23: A systematic review and meta-analysis of randomized controlled trials. Eur J Clin Nutr. 2021;35(10):5339–5351. doi: 10.1038/s41430-020-00725-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Esteban C, Geuskens M, Ena JM, Mishal Z, Macho A, Torres JM, Uriel J. Receptor-mediated uptake and processing of vitamin D–binding protein in human B-lymphoid cells. J Biol Chem. 1992;267(14):10177–10183. [PubMed] [Google Scholar]
- 39.Peris P, Filella X, Monegal A, Guanabens N, Foj L, Bonet M, Boquet D, Casado E, Cerda D, Erra A, et al. Comparison of total, free and bioavailable 25-OH vitamin D determinations to evaluate its biological activity in healthy adults: The LabOscat study. Osteoporos Int. 2017;28(8):2457–2464. doi: 10.1007/s00198-017-4062-8. [DOI] [PubMed] [Google Scholar]
- 40.Hastie CE, Pell JP, Sattar N. Vitamin D and COVID-19 infection and mortality in UK Biobank. Eur J Nutr. 2021;60(1):545–548. doi: 10.1007/s00394-020-02372-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Clift AK, von Ende A, Tan PS, Sallis HM, Lindson N, Coupland CAC, Munafo MR, Aveyard P, Hippisley-Cox J, Hopewell JC. Smoking and COVID-19 outcomes: an observational and Mendelian randomisation study using the UK Biobank cohort. Thorax. 2022;77:65–73. doi: 10.1136/thoraxjnl-2021-217080. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
nqac027_supplemental_file
Data Availability Statement
Data described in the manuscript will be made publicly and freely available without restriction at: https://drive.google.com/file/d/10lnd2MKQUhakKBERLpaIyxrz2Du502c2/view?usp=sharing. The code book and analytic code are available from the corresponding author on reasonable request.