Abstract
Background
Recent studies have shown that the excitatory amino acid transporters (EAATs) are associated with schizophrenia. The aim of this study was to investigate the relationship between the polymorphism of EAAT1 and EAAT2 genes and schizophrenia in Chinese Han population.
Methods
A total of 233 patients with schizophrenia and 342 healthy controls were enrolled. Two SNPs in EAAT1 gene (rs2269272, rs2731880) and four SNPs in EAAT2 gene (rs12360706, rs3088168, rs12294045, rs10836387) were genotyped by SNaPshot. Clinical features were collected using a self-made questionnaire. Psychotic symptoms of patients were measured by the Positive and Negative Syndrome Scale (PANSS), and patients’ cognitive function was assessed by Matrics Consensus Cognitive Battery (MCCB).
Results
Significant difference in allelic distributions between cases and controls was confirmed at locus rs12294045 (Ρ = 0.004) of EAAT2 gene. Different genotypes of rs12294045 were associated with family history (P = 0.046), in which patients with CT genotype had higher proportion of family history of psychosis. The polymorphism of rs12294045 was related to working operational memory (LNS: P = 0.016) and verbal learning function (HVLT-R: P = 0.042) in patients in which CT genotype had lower scores. However, these differences were no longer significant after Bonferroni correction.
Conclusions
Our study showed that the polymorphism of rs12294045 in EAAT2 gene may be associated with schizophrenia in Chinese Han population. CT genotype may be one of the risk factors for family history and cognitive deficits of patients.
Keywords: EAAT1, EAAT2, Schizophrenia, Symptom, Cognitive deficits
Introduction
Schizophrenia is a common psychiatric disorder with unclear etiology characterized clinically by positive symptoms, negative symptoms, and cognitive deficits [1]. Cognitive deficits are now generally recognized as core symptoms of schizophrenia, which have their own pathologic mechanisms and related to the abnormal development of the nervous system [2].
The glutamate hypothesis of schizophrenia suggests that the abnormal transmission of glutamate in the brain leads to cognitive and behavioral abnormalities related to schizophrenia [3, 4]. The changes of glutamate concentration in the central nervous system may damage the structural connection and integrity of neurons and cause programmed cell death and cell proliferation. Such changes may affect the ability to adapt to environmental changes and resist various physiological injuries. Approximately 40% of the synapses in the central nervous system are glutamatergic synapses [5, 6]. When glutamate is at normal level, it plays an important role in synaptic plasticity, learning and memory, neurodevelopment and so on [6]. The increased level of glutamate in the glutamatergic synaptic gap could cause excessive activation of the corresponding glutamate receptors, resulting in excessive increase of intracellular calcium concentration in neurons, leading to excitatory neuron damage or even nerve cell death [7, 8]. Relevant studies have shown that this excitotoxicity may be associated with the occurrence and development of a variety of neuropsychiatric disorders including schizophrenia, bipolar disorder, attention deficit hyperactivity disorder, epilepsy, stroke, amyotrophic lateral sclerosis, and idiopathic tremor [9–12]. On the other hand, the N-methyl-D-aspartate (NMDA) hypofunction hypothesis of schizophrenia indicated that the inhibition of synaptic NMDA receptors which cause decreased glutamate release was related to schizophrenia [13, 14]. And the glutamate level in the synaptic space is mainly regulated by excitatory amino acid transporters (EAATs) [15–17].
Previous studies have found that EAAT1 and EAAT2 genes are associated with schizophrenia. Region-specific increases in cortical EAAT1 and EAAT2 mRNA were involved in schizophrenia pathophysiology [18, 19]. The expression levels of EAAT2 protein in patients’ brain were lower than those in normal [20–22], and the impaired cognitive functions such as speech fluency and verbal learning function in patients were related to the decreased expression level of this gene [15, 23]. In addition, EAAT2 gene polymorphisms were found to be associated with schizophrenia in Japanese populatio [24], and the G allele of SNP rs4354668 was associated with poor clinical manifestations in abstract thinking and working memory [25, 26]. According to the glutamate hypothesis of schizophrenia [27–30], the researchers suggested that decreased levels of glutamate in schizophrenia may be associated with impaired cognitive and social functioning [31, 32].
Therefore, we conducted a case-control study to investigate the relationship between the polymorphisms of EAAT1 and EAAT2 genes and clinical characteristics, symptoms severity and cognitive deficits in Chinese Han patients with schizophrenia.
Methods
Participants
A total of 233 Patients, hospitalized in Shandong Mental Health Center of China from November 2015 to March 2018, who met the Diagnostic and Statistical Manual of Mental Disorders-Fourth Edition (DSM-IV) diagnosis of schizophrenia were enrolled in this study. Patients also met the following criteria: ages were range between 18 and 60 years old; biological parents were Chinese Han population; they were able to cooperate in completing the Matrics Consensus Cognitive Battery (MCCB) cognitive test; they have not been on antipsychotic medication for at least one month or receive modified electric convulsive therapy for six months; they had no history of drug or cigarette or alcohol abuse. Meanwhile, we included 342 healthy controls aged between 18 and 60 years old. They also met the criteria including their biological parents were Chinese Han population, they had no family history of mental illness, and they had no substance abuse and no pregnancy plan in the near future.
Assessments
A self-made general clinical data questionnaire was used to collect the participants’ data including age, gender, education level, occupation, age of onset, disease duration, family history, interpersonal relationship, marital status and other disease-related data. The Positive and Negative Symptom Scale (PANSS) was used to assess the symptoms severity of the patients. The patients’ cognitive function was assessed using Matrics Consensus Cognitive Battery (MCCB) which has been shown to be an effective tool for testing cognitive deficits of Chinese patients with schizophrenia [33].
SNPs selection
The SNPs information of EAAT1 and EAAT2 genes in Chinese Han population was downloaded from the International Genome Database and the Genome Reference Consortium Human Genome Build 38 (GRch38) edition and then were analyzed by Haploview software (version 4.2). Tag SNPs were screened according to MAF > 0.05 and Υ2 ≥ 0.8. Two loci of EAAT1 (rs2269272 and rs2731880) and four loci of EAAT2 (rs12360706, rs3088168, rs12294045 and rs10836387) were selected.
DNA extraction and SNPs genotyping
A total of 5 ml of peripheral venous blood collected from patients and healthy controls was placed in anticoagulant tubes containing 0.5 mol/L ethylenediamine teacetic acid. The tubes were then centrifuged for 10 min at the speed of 3000 rpm/min to remove white blood cells and serum. The deoxyribonucleic acid (DNA) of the blood sample was extracted using the modified potassium iodide method.
The primers were designed using the online primer design tool and synthesized by The Beijing Genomics Institute (Beijing, China) (Table 1). The PCR was carried out using 29-μl volume including 2 μl genomic DNA, 3 μl of 10 Ex Taq buffer (Takara), 1 μl of MIX Primer, 2 μl of dNTPs (2.5 mM each), 20.8 μl of H2O and 0.2 μl of Ex Taq [5 U/μl] (Takara). The reaction conditions of PCR were as follows: denaturation at 96 °C for 20 s after an initial step of 2 min at 96 °C; annealing at 54 °C for 10 s; extending at 72 °C for 30 s; 35 cycles in total. Then the amplified products were digested and purified. The SNaPshot extension reactions were performed with the SNaPshot Multiplex PCR Kit (Applied Biosystems) using 5 μl volume including 3 μl PCR product, 1 μl of MIX Primer (5 PM), 0.5 μl of SNaPshot MIX (ABI) and 0.5 μl of ddH2O. The reaction process was as follows: denaturation at 95 °C for 10 s after an initial step of 2 min at 95 °C; annealing at 50 °C for 5 s; extended at 60 °C for 30 s; 35 cycles in total. After the above reaction, 5ul of SNaPshot PCR production with 0.5 U of SAP was performed at 37 °C for 60 min and 75 °C for 15 min. Finally, genetic analysis of 8 μl volume including 1 μl SNaPshot product, 6.5 μl of Hi-Di Formamide and 0.5 μl of GS-120 LIZ (ABI) was carried out using the PRISM 3730 XI Genetic Analyzer (Applied Biosystems), and the results were analyzed with GeneMapper v4.1 software (Applied Biosystems).
Table 1.
List of primer pairs for multiple PCR
| Gene | Primer | Sequence (5′–3′) | Length (bp) |
|---|---|---|---|
| EAAT1 | rs2269272-F | TCCTTAGAATGAGGGAAAC | 258 |
| rs2269272-R | CAGCGTCTTTGACTGGATA | ||
| rs2269272-YF | ATAAGAGAAATGGTAGAAGATGAATCAGTATGAAGACACTGT | 42 | |
| rs2731880-F | TTTGTAAATGCTCCTCCTGC | 455 | |
| rs2731880-R | CAAACATTGAGCAACCACTG | ||
| rs2731880-YF | TGACGGCCTACTGCCAACAGAAGGTTATGATACTGT | 36 | |
| EAAT2 | rs12360706-F | GGGAAGTAACTCTTATGGA | 281 |
| rs12360706-R | AACTGACTGTTAGCCTTGT | ||
| rs12360706-YF | CCTCAGAGATGTGCTGGACCAACTTCCTTGGCTAGT | 36 | |
| rs3088168-F | TATAGATGCTCTGTGCTACGTGACT | 282 | |
| rs3088168-R | AAGGGTAAAGCCTACAATA | ||
| rs3088168-YF | TGAAAGGAGTTGAAGAAGCCACATTTTCAAGGAAAAATTAGCCTGTCCACCATA | 54 | |
| rs12294045-F | CATGCCCTCAAAGATCTAAGGTAAA | 300 | |
| rs12294045-R | CAGTTACAGCAGGCCAGAA | ||
| rs12294045-YF | ACTTGGGTTTCTCAAAGGGCAAGAATGAGAAAGAGAAGAATTAAAGTCTACTTAGTTGGTTTTCTC | 66 | |
| rs10836387-F | CTGCGTGAGTTGCTGATTC | 246 | |
| rs10836387-R | GTTGTCTTCTATTGCCTGA | ||
| rs10836387-YF | AATCTGTAGGGAGAAGCTGAGCTGCACTGGATGACTGTTATGCTCCCA | 48 |
Statistical analysis
Statistical analyses were carried out with the SPSS package (version 21.0). The goodness-of-fit Chi-square test was used to verify whether the allele and genotype frequencies of the members in case group and control group corresponded to the Hardy-Weinberg equilibrium (HWE). The chi-square test was used to analyze the differences between the various qualitative data of the subjects, including allele frequency and genotype frequency, gender, onset form, family history of psychosis, interpersonal relationship, premorbid characteristics, current marital status, occupational status, etc. One way analysis of variance (ANOVA) were used to compare the differences in age of onset, total disease duration, PANSS scores (except PANSS Positive scores) and cognitive function of patients with schizophrenia. PANSS Positive scores was carried out with Kruskal-Wallis test for non-parametric test. The Least Significant Difference (LSD) method was used to perform multiple analysis. Linkage disequilibrium analyses and haplotype analyses were performed by Haploview software (Version 4.2).
Results
A total of 575 subjects were enrolled in this study in which 233 were patients with schizophrenia and 342 were healthy controls. There was no significant difference in gender (χ2 = 0.041, Ρ = 0.084) and age (t = 1.858, Ρ = 0.064) between the two groups (Table 2). All of the six SNPs were in Hardy-Weinberg Equilibrium in both case and control groups.
Table 2.
The characteristics of participants
| Group | Case (n = 233) | Control (n = 342) | χ2/t | Ρ |
|---|---|---|---|---|
| Gender [n(%)] | ||||
| Male | 110 (47.21) | 160 (46.78) | 0.041 | 0.084 |
| Female | 123 (52.79) | 182 (53.22) | ||
| Age(Χ ± S) | 32.94 ± 10.77 | 31.30 ± 10.13 | 1.858 | 0.064 |
Genotypic and allelic distributions of EAAT1 and EAAT2 genes in patients and controls
Difference in the allelic distribution of rs2731880 in EAAT1 gene was observed (χ2 = 4.205, Ρ = 0.040), while there was no significance after Bonferroni correction (P < 0.008 correcting for 6 tests). The genotypic and allelic distributions of rs12294045 in EAAT2 gene were significantly different between cases and controls (χ2 = 8.054, Ρ = 0.018; χ2 = 8.144, Ρ = 0.004, respectively). When correcting for multiple testing, the difference of allele distribution remained significant and the effect of difference of genotypic distribution was only at a trend level of significance. No association was found between schizophrenia and other four SNPs tested (EAAT1:rs2269272; EAAT2: rs3088168, rs10836387, rs12360706).
The comparison of allelic and genotypic distributions of six SNPs in EAAT1 and EAAT2 genes between patients with schizophrenia and controls are presented in Tables 3 and 4 respectively.
Table 3.
Comparison of allelic distributions of six SNPs in EAAT1 and EAAT2 genes between patients and controls
| Gene | SNP | Allele | Case | Control | χ2 | P |
|---|---|---|---|---|---|---|
| EAAT1 | rs2269272 | C | 321 (0.689) | 474 (0.693) | 0.022 | 0.881 |
| T | 145 (0.311) | 210 (0.307) | ||||
| rs2731880 | C | 274 (0.588) | 443 (0.648) | 4.205 | 0.040 | |
| T | 192 (0.412) | 241 (0.352) | ||||
| EAAT2 | rs3088168 | C | 189 (0.406) | 313 (0.458) | 3.050 | 0.081 |
| T | 277 (0.594) | 371 (0.542) | ||||
| rs10836387 | G | 160 (0.343) | 252 (0.368) | 0.758 | 0.384 | |
| A | 306 (0.657) | 432 (0.632) | ||||
| rs12294045 | C | 330 (0.708) | 535 (0.782) | 8.144 | 0.004* | |
| T | 136 (0.292) | 149 (0.218) | ||||
| rs12360706 | G | 376 (0.807) | 559 (0.876) | 0.197 | 0.657 | |
| A | 90 (0.193) | 125 (0.124) | ||||
Notes: Bolded indicates statistically significant (p < 0.05)
*indicates the difference was still significant after Bonferroni correction
Table 4.
Comparison of genotypic distributions of six SNPs in EAAT1 and EAAT2 genes between patients and controls
| Gene | SNP | Genotype | Case | Control | χ2 | P |
|---|---|---|---|---|---|---|
| EAAT1 | rs2269272 | CC | 113 (0.485) | 164 (0.480) | ||
| TT | 25 (0.107) | 32 (0.094) | 0.394 | 0.821 | ||
| CT | 95 (0.408) | 146 (0.426) | ||||
| rs2731880 | CC | 82 (0.352) | 145 (0.424) | |||
| TT | 41 (0.176) | 44 (0.129) | 4.106 | 0.128 | ||
| CT | 110 (0.472) | 153 (0.447) | ||||
| EAAT2 | rs3088168 | CC | 37 (0.159) | 67 (0.196) | ||
| TT | 81 (0.348) | 96 (0.281) | 3.313 | 0.191 | ||
| CT | 115 (0.493) | 179 (0.523) | ||||
| rs10836387 | GG | 32 (0.137) | 41 (0.120) | |||
| AA | 105 (0.451) | 131 (0.383) | 4.043 | 0.132 | ||
| GA | 96 (0.412) | 170 (0.497) | ||||
| rs12294045 | CC | 117 (0.502) | 211 (0.617) | |||
| TT | 20 (0.086) | 18 (0.053) | 8.054 | 0.018 | ||
| CT | 96 (0.412) | 113 (0.330) | ||||
| rs12360706 | GG | 150 (0.644) | 227 (0.664) | |||
| AA | 7 (0.030) | 10 (0.029) | 0.249 | 0.883 | ||
| GA | 76 (0.326) | 105 (0.307) | ||||
Notes: Bolded indicates statistically significant (p < 0.05)
Linkage disequilibrium and haplotypes
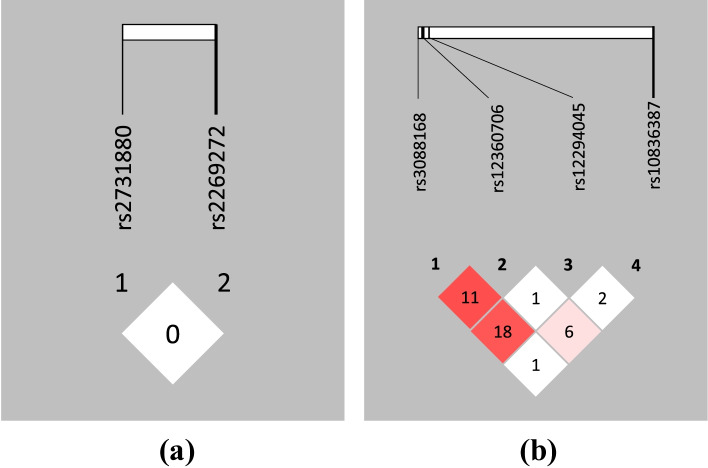
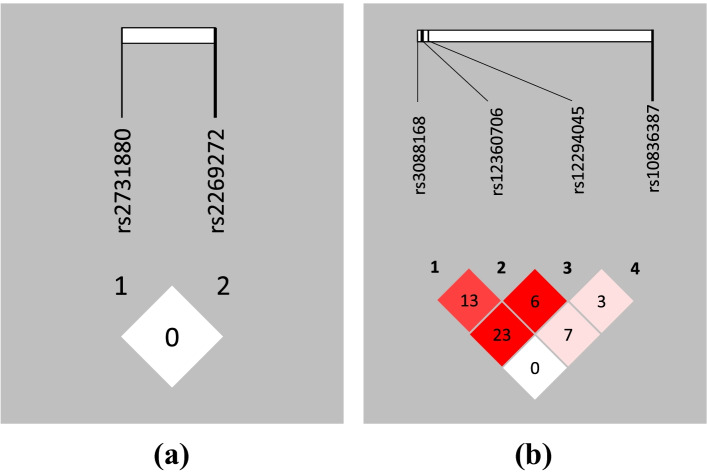
In both patients and controls pair-wise linkage disequilibrium (LD) between the investigated EAAT1 rs2269272 and rs2731880, and EAAT2 rs3088168, rs10836387, rs12294045 and rs12360706 SNPs was determined. LD blocks of EAAT1and EAAT2 genes SNPs and values of the correlation coefficient R2 for both groups are presented in Figs. 1 and 2.
Fig. 1.
Linkage disequilibrium analysis of the selected SNPs in patients. a is for EAAT1 gene and b is for EAAT2 gene. (Note: Numbers in the LD blocks are the values of R2 which are shown as figure of percentage. For example, 11 represents R2 = 0.11)
Fig. 2.
Linkage disequilibrium analysis of the selected SNPs in controls. Figure 1a is for EAAT1 gene and b is for EAAT2 gene. (Note: Numbers in the LD blocks are the values of R2 which are shown as figure of percentage. For example, 13 represents R2 = 0.13)
No haplotype was found by haplotype analysis using the current studied loci of either EAAT1 or EAAT2 gene.
Comparison of clinical features and symptoms severity with different genotypes of rs12294045
According to the above results, we found that the polymorphism of rs12294045 in EAAT2 gene may be associated with schizophrenia. As a result, we further examined the differences among three genotypes of rs12294045 in clinical data (gender, age of onset, onset form, total disease duration, family history of psychosis, interpersonal relationship, premorbid characteristics, current marital status, occupational status) and PANSS scores (PANSS positive scale score, PANSS negative scale score, PANSS psychopathology scale score, PANSS total score) of patients with schizophrenia. The results yielded nominally significant P value in family history (P = 0.046). Compared with patients without family history, patients with family history of psychosis had lower proportion of CC homozygous genotype and higher proportion of CT heterozygous genotype. However, P value did not withstand Bonferroni’s correction for multiple testing (P < 0.0038 correcting for 13 tests). There was no significant difference in other clinical data and PANSS scores among the three genotypes of rs12294045 (Table 5).
Table 5.
Comparison of clinical features and symptoms severity in patients with different genotypes of rs12294045
| Items | Classification | CC(117) | TT(20) | CT(96) | χ2/F | P |
|---|---|---|---|---|---|---|
| Gender |
Male(110) Female(123) |
53 (0.48) 64 (0.52) |
9 (0.08) 11 (0.09) |
48 (0.44) 48 (0.39) |
0.510 | 0.775 |
|
Age of onset (years, Χ ± S) |
24.06 ± 7.77 | 26.70 ± 7.14 | 23.99 ± 6.90 | 1.205 | 0.302 | |
| Onset form |
Acute(22) Subacute(28) Chronic(183) |
10 (0.46) 14 (0.50) 93 (0.51) |
4 (0.18) 0 (0) 16 (0.09) |
8 (0.36) 14 (0.50) 74 (0.40) |
5.560 | 0.235 |
|
Duration (months, Χ ± S) |
97.94 ± 96.04 | 109.00 ± 105.4 | 99.56 ± 96.45 | 0.111 | 0.895 | |
| Family history |
Positive(162) Negative(71) |
73 (0.45) 44 (0.62) |
14 (0.09) 6 (0.08) |
75 (0.46) 21 (0.30) |
6.162 | 0.046 |
| Interpersonal relationship |
Good(15) General(123) Poor(95) |
8 (0.53) 67 (0.54) 42 (0.44) |
2 (0.13) 11 (0.09) 7 (0.08) |
5 (0.34) 45 (0.37) 46 (0.48) |
3.794 | 0.435 |
| Premorbid character |
Extro(37) Neutral(14) Intro(182) |
20 (0.54) 7 (0.50) 90 (0.49) |
2 (0.05) 1 (0.07) 17 (0.09) |
15 (0.41) 6 (0.43) 75 (0.41) |
0.734 | 0.947 |
| Marital status |
Unmarried(116) Married(75) Living apart(10)Divorced(28) Loss of spouse(4) |
59 (0.51) 39 (0.52) 5 (0.50) 14 (0.50) 0 (0) |
8 (0.07) 6 (0.08) 3 (0.30) 2 (0.07) 1 (0.25) |
49 (0.42) 30 (0.40) 2 (0.20) 12 (0.43) 3 (0.75) |
11.423 | 0.179 |
| State of occupation |
Full-time(64) Part-time(38) Jobless*(93) Unemployment*(9) Retired(28) Other works(1) |
32 (0.50) 19 (0.50) 43 (0.46) 7 (0.78) 15 (0.54) 1 (1) |
5 (0.08) 3 (0.08) 9 (0.10) 0 (0) 3 (0.10) 0 (0) |
27 (0.42) 16 (0.42) 41 (0.44) 2 (0.22) 10 (0.36) 0 (0) |
5.027 | 0.889 |
| PANSS(Χ ± S) | Positive score | 27.74 ± 4.17 | 28.05 ± 4.05 | 28.52 ± 5.40 | 1.299 | 0.541 |
| PANSS(Χ ± S) | Negative score | 23.72 ± 5.45 | 24.10 ± 4.52 | 23.80 ± 5.11 | 0.046 | 0.955 |
| PANSS(Χ ± S) | psychopathology | 47.74 ± 7.02 | 49.80 ± 7.75 | 48.44 ± 7.12 | 0.798 | 0.452 |
| PANSS(Χ ± S) | Total score | 99.20 ± 11.46 | 101.95 ± 10.43 | 100.76 ± 11.35 | 0.801 | 0.450 |
Notes: Bolded indicates statistically significant (p < 0.05); Italics indicates Kruskal-Wallis test for non-parametric test, and the statistic is χ2. (Χ ± S) means (Mean ± Standard Deviation)
*“Jobless” represents patients whose job were lost because of disease. “Unemployment” represents patients who had never worked
Comparison of cognitive functions with different genotypes of rs12294045
One way ANOVA was performed to find out whether different genotypes of rs12294045 were related to cognitive functions of patients. Differences in Letter-Number Span Test (LNS) and Hopkins Verbal Learning Test-Revised (HVLT-R) were observed (LNS: Ρ = 0.016; HVLT-R: Ρ = 0.042). LSD analysis showed that patients with CT genotypes had significantly lower scores than those with CC and TT genotypes in terms of LNS representing working operational memory (Ρ1 = 0.042, Ρ2 = 0.011). In addition, patients with CT genotypes had significantly lower HVLT-R scores than those with TT genotypes (Ρ2 = 0.020) and lower scores than those with CC genotypes (Ρ1 = 0.098). These results suggested that the heterozygous status of rs12294045 (CT) might be considered as a risk factor for cognitive function of schizophrenia. However, after Bonferroni correction (P < 0.005 correcting for 10 tests), these differences were no longer significant. No significant difference was found in other subtests of MCCB (Table 6).
Table 6.
Comparison of cognitive test scores in patients with different genotypes of rs12294045
| Subtests | rs12294045 | F | Ρ | Ρ 1 | Ρ 2 | Ρ 3 | ||
|---|---|---|---|---|---|---|---|---|
| CT(n = 96) | CC(n = 116) | TT(n = 20) | ||||||
| TMT-A | 45.23 ± 7.86 | 46.28 ± 11.97 | 47.95 ± 7.54 | 0.704 | 0.495 | 0.442 | 0.274 | 0.500 |
| Symbol Coding | 42.00 ± 10.56 | 40.98 ± 10.10 | 44.60 ± 11.07 | 1.113 | 0.330 | 0.473 | 0.308 | 0.149 |
| Category Fluency | 47.75 ± 11.32 | 47.35 ± 11.25 | 48.55 ± 10.88 | 0.105 | 0.901 | 0.805 | 0.772 | 0.664 |
| CPT-IP | 41.84 ± 11.11 | 42.05 ± 10.75 | 45.25 ± 13.08 | 0.823 | 0.441 | 0.914 | 0.213 | 0.228 |
| LNS | 43.76 ± 10.97 | 46.96 ± 11.32 | 50.75 ± 9.89 | 4.184 | 0.016 | 0.042 | 0.011 | 0.149 |
| WMS-III | 44.34 ± 12.03 | 44.68 ± 12.93 | 47.10 ± 12.63 | 0.413 | 0.662 | 0.914 | 0.373 | 0.399 |
| HVLT-R | 42.22 ± 12.18 | 44.94 ± 10.88 | 48.80 ± 11.08 | 3.222 | 0.042 | 0.098 | 0.020 | 0.154 |
| BVMT-R | 43.89 ± 11.79 | 43.16 ± 11.16 | 44.30 ± 13.07 | 0.192 | 0.826 | 0.598 | 0.884 | 0.655 |
| NAB-MAZES | 43.02 ± 8.99 | 42.94 ± 11.05 | 42.65 ± 10.70 | 0.013 | 0.987 | 0.914 | 0.883 | 0.930 |
| MSCEIT | 47.03 ± 12.30 | 48.09 ± 11.91 | 51.75 ± 14.55 | 1.222 | 0.297 | 0.568 | 0.120 | 0.209 |
Note: Bolded indicates statistically significant (p < 0.05)
Ρ 1 represents the comparison between CT genotype patients and CC genotype patients; Ρ 2 represents the comparison between CT genotype patients and TT genotype patients; Ρ 3 represents the comparison between CC genotype patients and TT genotype patients.(TMT-A Trail Making Test A, CPT-IP Continuous Performance Test-Identical Pairs, LNS Letter-Number Span Test, WMS-III Wechsler Memory Scale-Third Edition, HVLT-R Hopkins Verbal Learning Test-Revised, BVMT-R Brief Visuospatial Memory Test-Revised, NAB Neuropsychological Assessment Battery, MSCEIT Mayer-Salovey-Caruso Emotional Intelligence Test).
Discussion
There are two objectives of this study. Firstly, the relationship between EAAT1 and EAAT2 genes and schizophrenia was explored. We found no significant association between EAAT1 gene polymorphism and schizophrenia. For EAAT2 gene, the polymorphism of rs12294045 may be associated with schizophrenia in Chinese Han population. Secondly, we investigated the relationship between the polymorphism of rs12294045 and clinical characteristics, symptoms severity and cognitive function in patients with schizophrenia. The results showed that rs12294045 locus may be associated with family history, working operational memory and verbal learning, in which the CT genotype was a risk factor. However, no significance remained after multiple testing.
In this study, only a trend was showed that the allele distribution of rs2731880 in EAAT1 gene may be associated with schizophrenia. Previous studies reported the relation between EAAT1 and schizophrenia. Spangaro M et al. found that rs2731880 in European population may be related to schizophrenia [25]. In addition, the levels of EAAT1 mRNA expression in the thalamus of subjects with schizophrenia was found significantly higher than healthy subjects [34]. Animal model studies [35] showed that compared with wild-type mice in the same nest, EAAT1 knockout mice have negative social behaviors, such as lack of pleasure, social withdrawal and self-neglect. Our inconsistent results may be because of the small sample size or the genetic differences among different races.
Our study found an evidence about the association between rs12294045 in EAAT2 gene and schizophrenia. There were few previous studies on rs12294045 and schizophrenia. Appenzeller S et al. [36] explored the polymorphism of rs12294045 in Parkinson’s disease and no association was found. For the relationship between EAAT2 gene and schizophrenia, recent studies have shown different results. A study on EAAT2 mRNA have found that region-specific increases in cortical EAAT2 mRNA were involved in schizophrenia pathophysiology [18]. A case-control study in Japanese population [24] reported the EAAT2 gene polymorphism was associated with schizophrenia, and concluded that at least one of the susceptibility locus for schizophrenia may reside in internal or nearby EAAT2. In contrast, no association was observed between the polymorphism − 181 A/C (rs4354668) in SLC1A2 (EAAT2) and onset of schizophrenia and its psychopathology in Polish population [37].
With regard to the relationship between EAAT2 gene and cognitive function in schizophrenia patients, our findings were fairly weak. Previous studies have shown that working memory represents the core content of changes in schizophrenia, it has been indicated that working memory can predict the severity of cognitive impairment and play a key role in the performance of other cognitive tasks in patients with schizophrenia [38, 39]. The results of prospective studies suggested that patients carrying the G allele of EAAT2 SNP rs4354668 had significantly reduced gray matter volume and impaired working memory function [26]. Spangaro M et al. [40] found that genotypes associated with low expression of EAAT2 were significantly associated with cognitive dysfunction such as executive function and working memory function. In agreement with the above findings, recent studies also suggested the genetic variation in EAAT2 may be involved in impaired working memory and executive function of patients with schizophrenia [41, 42]. Further study on the function of rs12294045 locus to explore how its polymorphism influences the expression of EAAT2 mRNA is needed, which could help us better understand the relationship between this locus and cognitive dysfunction in patients with schizophrenia.
Our findings are subject to a number of limitations. First, the sample size of this study was small which led to a relatively low genetic power. Second, our patients were identified through the inpatient department of hospital; we didn’t include schizophrenia patients in the outpatient clinics. Our findings may not generalize to patients who don’t seek treatment or those who don’t need hospitalization. Third, only one method (MCCB) was used to test the cognitive function of patients, so the patients’ cognitive dysfunction may not be fully explored. Fourth, we could not completely eliminate other possible factors, such as the previous medical treatment. Last but not least, EAAT1 and EAAT2 genes loci were still not identified by genome-wide association analysis in the most recent schizophrenia study [43]. Schizophrenia is a complex disease with unknown etiology and many factors could contribute to the pathogenesis of the disease. Therefore, genome-wide association study (GWAS) of schizophrenia based on larger sample size and interaction study between genes and other genetic and environmental factors are further needed.
Conclusions
In our study, the polymorphism of rs12294045 locus in EAAT2 gene may be associated with schizophrenia. CT genotype may be one of the risk factors for family history, the dysfunction of working operational memory and verbal learning in patients.
Acknowledgements
We thank all participants for their support in this study.
Abbreviations
- CNS
Central Nervous System
- SNPs
Single nucleotide polymorphisms
- EAAT
Excitatory Amino Acid Transporter
- NMDA
N-methyl-D-aspartate
- MAF
Minor Allele Frequency
- PCR
Polymerase Chain Reaction
- DSM-IV
Fourth Edition of the Diagnostic and Statistical Manual for Mental Disorders
- PANSS
Positive and Negative Syndrome Scale
- MCCB
Matrics Consensus Cognitive Battery
- HWE
Hard-Weinberg Equilibrium
- LD
linkage disequilibrium
- BVMT-R
Brief Visuospatial Memory Test-Revised
- CPT-IP
Continuous Performance Test-Identical Pairs
- HVLT-R
Hopkins Verbal Learning Test-Revised
- LNS
Letter-Number Span Test
- MSCEIT
Mayer-Salovey-Caruso Emotional Intelligence Test
- NAB
Neuropsychological Assessment Battery
- TMT-A
Trail Making Test A
- WMS-III
Wechsler Memory Scale-Third Edition
Authors’ contributions
Lanfen Liu and Lina Wang designed the research; Dongdong Qiao and Kaiyan Cui performed the experiments; Xiaojiao Bi, Chao Han, and Tantan Ma conducted sample collection and analyzed the data; Limin Yang, Mengmeng Sun and Lanfen Liu were responsible for the clinical diagnosis; Lina Wang and Tantan Ma were responsible for manuscript writing. All authors have read and approved the final manuscript.
Funding
This study was supported by Medicine and Health Science Technology Development Program of Shandong province (2015WS0252). The funding body played no role in the design of the study and collection, analysis, and interpretation of data or in writing the manuscript.
Availability of data and materials
Researchers interested in the study may contact corresponding author to obtain relevant data via email: liulf521@163.com.
Declarations
Ethics approval and consent to participate
This study was conducted in accordance with the Declaration of Helsinki. And the study was approved by the Ethics Committee of the Mental Health Center of Shandong Province, reference number: (2018) Ethics Review (R04). All participants signed the informed consent before participating in the study. We confirmed that all methods were performed in accordance with the relevant guidelines and regulations.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Lina Wang and Tantan Ma contributed equally to this work.
References
- 1.Coyle JT. Schizophrenia: Basic and Clinical. Adv Neurobiol. 2017;15:255–280. doi: 10.1007/978-3-319-57193-5_9. [DOI] [PubMed] [Google Scholar]
- 2.Smeland OB, Frei O, Kauppi K, et al. Identification of genetic loci jointly influencing schizophrenia risk and the cognitive traits of verbal-numerical reasoning, reaction time, and general cognitive function. JAMA Psychiatry. 2017;74(10):1065–1075. doi: 10.1001/jamapsychiatry.2017.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Adams B, Moghaddam B. Corticolimbic dopamine neurotransmission is temporally dissociated from the cognitive and locomotor effects of phencyclidine. J Neurosci. 1998;18(14):5545–5554. doi: 10.1523/JNEUROSCI.18-14-05545.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Olney JW, Farber NB. Glutamate receptor dysfunction and schizophrenia. Arch Gen Psychiatry. 1995;52:998–1007. doi: 10.1001/archpsyc.1995.03950240016004. [DOI] [PubMed] [Google Scholar]
- 5.Fairman WA, Amara SG. Functional diversity of excitatory amino acid transporters: ion channel and transport modes. Am J Physiol. 1999;277(4 Pt 2):F481–F486. doi: 10.1152/ajprenal.1999.277.4.F481. [DOI] [PubMed] [Google Scholar]
- 6.Suzuki M, Nelson AD, Eickstaedt JB, et al. Glutamate enhances proliferation and neurogenesis in human neural progenitor cell cultures derived from the fetal cortex. Eur J Neurosci. 2006;24(3):645–653. doi: 10.1111/j.1460-9568.2006.04957.x. [DOI] [PubMed] [Google Scholar]
- 7.Lucas DR, Newhouse JP. The toxic effect of sodium L-glutamate on the inner layers of the retina. Ama Arch Ophthalmol. 1957;58(2):193–201. doi: 10.1001/archopht.1957.00940010205006. [DOI] [PubMed] [Google Scholar]
- 8.Choi DW. Glutamate neurotoxicity and diseases of the nervous system. Neuron. 1988;1(8):623–634. doi: 10.1016/0896-6273(88)90162-6. [DOI] [PubMed] [Google Scholar]
- 9.Sheldon AL, Robinson MB. The role of glutamate transporters in neurodegenerative diseases and potential opportunities for intervention. Eurochem Int. 2007;51(6-7):333–355. doi: 10.1016/j.neuint.2007.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lee M, Cheng MM, Lin CY, et al. Decreased EAAT2 protein expression in the essential tremor cerebellar cortex. Acta Neuropathol Commun. 2014;2(1):1–11. doi: 10.1186/s40478-014-0157-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lin CL, Kong Q, Cuny GD, et al. Glutamate transporter EAAT2: a new target for the treatment of neurodegenerative diseases. Futur Med Chem. 2012;4(13):1689–1700. doi: 10.4155/fmc.12.122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rao JS, Kellom M, Reese EA, et al. Dysregulated glutamate and dopamine transporters in postmortem frontal cortex from bipolar and schizophrenic patients. J Affect Disord. 2017;220(1-2):156. doi: 10.1016/j.jad.2017.06.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Adell A. Brain NMDA receptors in schizophrenia and depression. Biomolecules. 2020;10(6):947. doi: 10.3390/biom10060947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nakazawa K, Sapkota K. The origin of NMDA receptor hypofunction in schizophrenia. Pharmacol Ther. 2019;205:107426. doi: 10.1016/j.pharmthera.2019.107426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Plitman E, Nakajima S, Camilo FS, et al. Glutamate-mediated excitotoxicity in schizophrenia: A review. Eur Neuropsychopharmacol. 2014;24(10):1591–1605. doi: 10.1016/j.euroneuro.2014.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Marcaggi P, Attwell D. Role of glial amino acid transporters in synaptic transmission and brain energetics. Glia. 2004;47(3):217–225. doi: 10.1002/glia.20027. [DOI] [PubMed] [Google Scholar]
- 17.Shan D, Lucas EK, Drummond JB, et al. Abnormal expression of glutamate transporters in temporal lobe areas in elderly patients with schizophrenia. Schizophr Res. 2013;144(1-3):1–8. doi: 10.1016/j.schres.2012.12.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Parkin GM, Gibbons A, Udawela M, et al. Excitatory amino acid transporter (EAAT)1 and EAAT2 mRNA levels are altered in the prefrontal cortex of subjects with schizophrenia. J Psychiatr Res. 2020;123. [DOI] [PubMed]
- 19.Walsh T, Mcclellan JM, Mccarthy SE, et al. Rare structural variants disrupt multiple genes in neurodevelopmental pathways in schizophrenia. Science. 2008;320(5875):539–543. doi: 10.1126/science.1155174. [DOI] [PubMed] [Google Scholar]
- 20.Ohnuma T, Augood SJ, Arai H, et al. Expression of the human excitatory amino acid transporter 2 and metabotropic glutamate receptors 3 and 5 in the prefrontal cortex from normal individuals and patients with schizophrenia. Brain Research. Mol Brain Res. 1998;56(1-2):207. doi: 10.1016/s0169-328x(98)00063-1. [DOI] [PubMed] [Google Scholar]
- 21.Ohnuma T, Tessler S, Arai H, et al. Gene expression of metabotropic glutamate receptor 5 and excitatory amino acid transporter 2 in the schizophrenic hippocampus. Mol Brain Res. 2000;85(1):24–31. doi: 10.1016/s0169-328x(00)00222-9. [DOI] [PubMed] [Google Scholar]
- 22.Egan MF, Straub RE, Goldberg TE, et al. Variation in GRM3 affects cognition, prefrontal glutamate, and risk for schizophrenia. Proc Natl Acad Sci U S A. 2004;101(34):12604–12609. doi: 10.1073/pnas.0405077101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Robinson MB. The family of sodium-dependent glutamate transporters: a focus on the GLT-1/EAAT2 subtype. Neurochem Int. 1998;33(6):479–491. doi: 10.1016/s0197-0186(98)00055-2. [DOI] [PubMed] [Google Scholar]
- 24.Deng X, Shibata H, Ninomiya H, et al. Association study of polymorphisms in the excitatory amino acid transporter 2 gene (SLC1A2) with schizophrenia. BMC Psychiatry. 2004;4(1):21. doi: 10.1186/1471-244X-4-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Spangaro M, Bosia M, Zanoletti A, et al. Exploring effects of EAAT polymorphisms on cognitive functions in schizophrenia. Pharmacogenomics. 2014;15(7):925–932. doi: 10.2217/pgs.14.42. [DOI] [PubMed] [Google Scholar]
- 26.Poletti S, Radaelli D, Bosia M, et al. Effect of glutamate transporter EAAT2 gene variants and gray matter deficits on working memory in schizophrenia. Eur Psychiatry. 2014;29(4):219–225. doi: 10.1016/j.eurpsy.2013.07.003. [DOI] [PubMed] [Google Scholar]
- 27.Javitt DC, Zukin SR. Recent advances in the phencyclidine model of schizophrenia. Am J Psychiatr. 1991;148(10):1301. doi: 10.1176/ajp.148.10.1301. [DOI] [PubMed] [Google Scholar]
- 28.Mohn AR, Gainetdinov RR, Caron MG, et al. Mice with reduced NMDA receptor expression display behaviors related to schizophrenia. Cell. 1999;98(4):427–436. doi: 10.1016/s0092-8674(00)81972-8. [DOI] [PubMed] [Google Scholar]
- 29.Olney JW, Newcomer JW, Farber NB. NMDA receptor hypofunction model of schizophrenia. J Psychiatr Res. 1999;33(6):523–533. doi: 10.1016/s0022-3956(99)00029-1. [DOI] [PubMed] [Google Scholar]
- 30.Steinpreis RE. The behavioral and neurochemical effects of phencyclidine in humans and animals: some implications for modeling psychosis. Behav Brain Res. 1996;74(1-2):45–55. doi: 10.1016/0166-4328(95)00162-x. [DOI] [PubMed] [Google Scholar]
- 31.Bustillo JR, Chen H, Gasparovic C, et al. Glutamate as a marker of cognitive function in schizophrenia: a proton spectroscopic imaging study at 4 tesla. Biol Psychiatry. 2011;69(1):19. doi: 10.1016/j.biopsych.2010.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Aoyama N, Théberge J, Drost DJ, et al. Grey matter and social functioning correlates of glutamatergic metabolite loss in schizophrenia. Br J Psychiatry J Ment Sci. 2018;198(6):448–456. doi: 10.1192/bjp.bp.110.079608. [DOI] [PubMed] [Google Scholar]
- 33.Zou Y, Cui J, Wang J, et al. Clinical reliability and validity of the Chinese version of measurement and treatment research to improve cognition in schizophrenia consensus cognitive battery. Chin J Psychiatry. 2009;42(1):29–33. [Google Scholar]
- 34.Smith RE, Haroutunian V, Davis KL, et al. Expression of excitatory amino acid transporter transcripts in the thalamus of subjects with schizophrenia. Am J Psychiatry. 2001;158(9):1393–1399. doi: 10.1176/appi.ajp.158.9.1393. [DOI] [PubMed] [Google Scholar]
- 35.Karlsson RM, Tanaka K, Saksida LM, et al. Assessment of glutamate transporter GLAST (EAAT1)-deficient mice for phenotypes relevant to the negative and executive/cognitive symptoms of schizophrenia. Neuropsychopharmacology. 2009;34(6):1578–1589. doi: 10.1038/npp.2008.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Appenzeller S, Schulte C, Thier S, et al. No association between polymorphisms in the glutamate transporter SLC1A2 and Parkinson's disease. Mov Disord. 2013;28(9):1305–1306. doi: 10.1002/mds.25330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Merk W, Kucia K, Mędrala T, Kowalczyk M, Owczarek A, Kowalski J. Association study of the excitatory amino acid transporter 2 (EAAT2) and glycine transporter 1 (GlyT1) gene polymorphism with schizophrenia in a Polish population. Neuropsychiatr Dis Treat. 2019;15:989–1000. doi: 10.2147/NDT.S194924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Perlstein WM, Carter CS, Noll DC, et al. Relation of prefrontal cortex dysfunction to working memory and symptoms in schizophrenia. Am J Psychiatry. 2001;158(7):1105–1113. doi: 10.1176/appi.ajp.158.7.1105. [DOI] [PubMed] [Google Scholar]
- 39.Silver H, Feldman P, Bilker W, et al. Working memory deficit as a core neuropsychological dysfunction in schizophrenia. Am J Psychiatry. 2003;160(10):1809–1816. doi: 10.1176/appi.ajp.160.10.1809. [DOI] [PubMed] [Google Scholar]
- 40.Spangaro M, Bosia M, Zanoletti A, et al. Cognitive dysfunction and glutamate reuptake: effect of EAAT2 polymorphism in schizophrenia. Neurosci Lett. 2012;522(2):151–155. doi: 10.1016/j.neulet.2012.06.030. [DOI] [PubMed] [Google Scholar]
- 41.Mazza E, Spa Ng Aro M, Poletti S, et al. Genetic variability of glutamate reuptake: Effect on white matter integrity and working memory in schizophrenia. Schizophr Res. 2019;208:457–459. doi: 10.1016/j.schres.2019.03.004. [DOI] [PubMed] [Google Scholar]
- 42.Zhang B, Guan F, et al. Common variants in SLC1A2 and schizophrenia: Association and cognitive function in patients with schizophrenia and healthy individuals. Schizophr Res. 2015;169(1-3):128–134. doi: 10.1016/j.schres.2015.10.012. [DOI] [PubMed] [Google Scholar]
- 43.The Schizophrenia Working Group of the Psychiatric Genomics, Consortium, Walters RS, O’Donovan JTR, Michael C. Mapping genomic loci prioritises genes and implicates synaptic biology in schizophrenia. medRxiv. 2020:09.12.20192922.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Researchers interested in the study may contact corresponding author to obtain relevant data via email: liulf521@163.com.