Abstract
BACKGROUND
Pulse wave velocity (PWV) is blood pressure (BP) dependent, leading to the development of the BP-corrected metrics cardio-ankle vascular index (CAVI) and CAVI0. We aimed to assess risk prediction by heart-to-ankle PWV (haPWV), CAVI, and CAVI0 in a US population.
METHODS
We included 154 subjects (94.8% male; 47.7% African American) with and without heart failure (HF). Left and right haPWV, CAVI, and CAVI0 were measured with the VaSera 1500N device. We prospectively followed participants for a mean of 2.56 years for the composite endpoint death or HF-related hospital admission (DHFA).
RESULTS
Left and right haPWV, CAVI, and CAVI0 values did not differ significantly. In unadjusted analyses, haPWV (left standardized hazard ratio [HR] = 1.51, P = 0.007; right HR = 1.66, P = 0.003), CAVI (left HR = 1.45, P = 0.012; right HR = 1.58, P = 0.006), and CAVI0 (left HR = 1.39, P = 0.022; right HR = 1.44, P = 0.014) significantly predicted DHFA. Predictive ability showed a decreasing trend from haPWV to CAVI to CAVI0; in line with the increasing amount of BP correction in these metrics. In Cox models, right-sided metrics showed a trend toward stronger predictive ability than left-sided metrics. After adjustment for baseline HF status, the Meta-Analysis Global Group in Chronic Heart Failure (MAGGIC) risk score, and systolic BP, right haPWV (HR = 1.58, P = 0.025) and CAVI (HR = 1.44, P = 0.044), but no other stiffness metrics, remained predictive.
CONCLUSIONS
Although conceptually attractive, BP-corrected arterial stiffness metrics do not offer better prediction of DHFA than conventional arterial stiffness metrics, nor do they predict DHFA independently of systolic BP. Our findings support PWV as the primary arterial stiffness metric for outcome prediction.
Keywords: arterial stiffness, blood pressure, hypertension, prospective observational study, risk prediction
Graphical Abstract
Graphical Abstract.

Arterial stiffness as measured by carotid–femoral pulse wave velocity (PWV) has been shown to predict cardiovascular events.1–3 However, PWV is known to depend on blood pressure at the time of measurement.4–6 This led to the development of arterial stiffness indices correcting for this blood pressure dependence, including cardio-ankle vascular index (CAVI).7 In 2017, CAVI was further refined into CAVI0,8,9 removing the residual acute blood pressure dependence that is potentially remaining in CAVI.
To our knowledge, the predictive ability of CAVI0 has not been assessed to date. Moreover, the predictive ability of CAVI was studied predominantly in Asian populations, with results suggesting a modest association between CAVI and incident cardiovascular risk.10 However, studies in European and US populations are lacking. This important gap of knowledge was identified in the Arterial Stiffness recommendations from the American Heart Association, which called for more stiffness studies in US-based populations.11
Recently, Tomiyama et al. observed that blood pressure correction may attenuate the predictive ability of PWV.12 In particular, when they performed correction using the same equation employed in CAVI, they observed reduced predictive ability for new onset of hypertension and new onset of retinopathy. However, instead of heart–ankle PWV (haPWV, as in CAVI), brachial–ankle PWV (baPWV) was used. Furthermore, the influence of CAVI0-based correction on prediction was not assessed.
In this study, we present the first US-based data regarding the prognostic value of CAVI and CAVI0, and aim to assess and compare risk prediction by haPWV, CAVI, and CAVI0.
METHODS
Study population
We enrolled 156 subjects with and without heart failure (HF) referred for a cardiac magnetic resonance imaging study at the Corporal Michael J. Crescenz VA Medical Center, Philadelphia, PA. The protocol was approved by the Philadelphia VA Medical Center Institutional Review Board, and written informed consent was obtained from all participants. Key exclusion criteria were as follows: (i) claustrophobia; (ii) presence of metallic objects or implanted medical devices in the body; (iii) atrial fibrillation, flutter, or significant arrhythmia at the time of enrollment, which may compromise the study measurements; and (iv) diagnosed or suspected infiltrative myocardial disease (cardiac or extracardiac amyloidosis or sarcoidosis).
Heart failure with reduced ejection fraction (HFrEF) was defined as a symptomatic HF in the presence of a left ventricular ejection fraction <50%. Heart failure with preserved ejection fraction (HFpEF) was defined as: (i) NYHA Class II–IV symptoms consistent with HF, in the absence of significant aortic stenosis; (ii) left ventricular ejection fraction >50%; (iii) a mitral E wave to annular e′ ratio >14,13 or at least 2 of the following: (a) a mitral E wave to annular e′ ratio >8; (b) treatment with a loop diuretic for control of HF symptoms; (c) left atrial volume index >34 ml/m2 of body surface area; (d) NT-pro B-type natriuretic peptide level >200 pg/ml; (e) left ventricular mass index >149 g/m2 in men and 122 g/m2 in women (measured by cardiac MRI). Subjects without HF had a left ventricular ejection fraction >50%, no significant valvular disease, and no symptoms and signs consistent with HF.14,15
We prospectively followed participants for the composite endpoint of death or HF-related hospital admission (DHFA). All events were formally adjudicated by physicians as per standardized criteria based on medical record review. The data underlying this article cannot be shared with investigators outside the Corporal Michael J. Crescenz VA Medical Center given that participants did not consent to wider sharing of their research data.
Measurements
Subjects underwent arterial stiffness assessments using the VaSera 1500N device (Fukuda Denshi, Tokyo, Japan). Before measurements were taken, subjects were rested comfortably for 10 minutes in the supine position. Measurements were taken using 2 brachial and 2 ankle cuffs (one per side), and a phonocardiograph, according to the manufacturer’s instructions. Cuffs were placed approximately 2 cm above the antecubital fossa on the arms and approximately 2 cm above the medial malleolus on the legs. Cuff size was adjusted according to arm size as per the manufacturer’s instructions. The VaSera device estimates haPWV by combining the heart-to-brachial transit time, which is obtained as the time difference between the closing of the aortic valve (from the phonocardiogram) and the dicrotic notch on the brachial cuff signal, and the brachial-to-ankle transit time, obtained as the foot-to-foot time difference between the brachial and ankle cuffs. For further details, see refs. 7,16. Data were transferred for analysis using the VaSera Data Management Software VSS-10U (Fukuda Denshi).
Calculations
Left and right haPWV (L-haPWV and R-haPWV) were calculated as described previously7,16:
| (1) |
| (2) |
tbR denotes the heart-to-right-brachial transit time; tbaL and tbaR denote the transit time from right-brachial to left and right ankle, respectively.16,17L is the estimated heart-to-ankle arterial pathway length.17 CAVI and CAVI0 are then calculated as7,8:
| (3) |
| (4) |
with ρ = 1,050 kg/m3 the blood mass density, and Pref = 100 mm Hg a reference pressure.8 Dimensional correctness was assured by converting all variables used in the CAVI/CAVI0 equations to standard (International System of Units, SI) units, i.e., Pa, m/s, and kg/m3, prior to calculations. Ps and Pd are the systolic and diastolic blood pressures, measured automatically by the VaSera device at the right brachial location. Consistent with the CAVI definition, right-brachial blood pressure is used in the computation of both left and right CAVI.16,17 As CAVI0 adopted this standard,18 left and right CAVI0 values were also computed using right-brachial blood pressures. a and b are CAVI-dependent scale factors.17,18 When L-haPWV is used for haPWV, L-CAVI and L-CAVI0 are obtained; when R-haPWV is used for haPWV, R-CAVI, and R-CAVI0 are obtained.
Statistical analysis
Continuous variables are presented as mean ± SD and compared using one-way analysis of variance (ANOVA, when normally distributed); or as median (25th, 75th percentile) and compared using a Kruskal-Wallis test (otherwise). Categorical variables are shown as total counts with percentages and were compared using Chi squared (χ 2) or Fisher’s exact tests. We used Cox proportional hazards regression to assess the relationship between various arterial indices and time to DHFA. For each parameter, we performed both unadjusted analyses and analyses that adjusted for key covariates. We adjusted for: (i) HF status and Meta-Analysis Global Group in Chronic Heart Failure (MAGGIC) risk score, which is an optimized prognostic multivariable score developed for both HFrEF and HFpEF19; and (ii) further adjusted for systolic blood pressure. Systolic blood pressure was obtained directly from the VaSera device (right brachial; Ps) and hence was measured simultaneously with the stiffness measurements. Besides systolic blood pressure, we did not include individual components of the MAGGIC score and other potential predictors of outcomes in order to avoid overfitting of statistical survival models, given the limited number of events. All hazard ratios are standardized (i.e., represent the relative risk per SD change in the predictor), to facilitate an intuitive comparison of the association between different indices. Statistical significance was defined as a 2-tailed P value < 0.05. Statistical analyses were performed using the MATLAB Statistics and Machine Learning Toolbox (MATLAB R2016b, MathWorks; Natick, MA) and SPSS for Mac v22 (SPSS, Chicago, IL).
RESULTS
Study population
Two cases were excluded from the analysis due to missing clinical data (n = 1) and lack of enough information for confident adjudication of HF status (n = 1), yielding n = 154 patients included in our further analyses. Our population was composed predominantly of males (95%), and nearly 50% of this population was of African American ethnicity (Table 1). Mean follow-up time was 2.56 years. During follow-up, 32 composite events (21 deaths/18 HF admissions) were recorded. Table 1 shows baseline study population characteristics stratified by HF status (no HF, HFrEF, and HFpEF). There were no significant differences in demographics and most clinical factors between the groups, although body mass index was significantly greater in the HFpEF group, whereas systolic blood pressure and the prevalence of diabetes were numerically greater (though statistically not significantly different) in the HFpEF group. The proportion of participants receiving beta blockers, angiotensin-converting enzyme inhibitors/angiotensin receptor blockers, and furosemide was greater in both HF groups, whereas spironolactone use was greater only in HFrEF, and hydralazine use was most common in HFpEF.
Table 1.
Baseline characteristics of study population
| No HF | HFrEF | HFpEF | P value | |
|---|---|---|---|---|
| n | 106 | 30 | 18 | — |
| Age [years] | 64.0 ± 10.9 | 67.3 ± 11.2 | 66.4 ± 10.7 | 0.2937 |
| Male sex | 100 (94.34%) | 30 (100.00%) | 16 (88.89%) | 0.2265 |
| Race | ||||
| White | 54 (50.94%) | 12 (40.00%) | 8 (44.44%) | 0.5412 |
| African American | 46 (43.40%) | 18 (60.00%) | 10 (55.56%) | 0.2185 |
| Other | 6 (5.66%) | 0 (0.00%) | 0 (0.00%) | 0.2433 |
| BMI [kg/m2] | 29.0 ± 6.1 | 28.7 ± 4.2 | 33.9 ± 7.8 | 0.0056 |
| Systolic blood pressure [mm Hg] | 138 (130,156) | 149 (131,169) | 150 (142,160) | 0.0819 |
| Diastolic blood pressure [mm Hg] | 87 (82,95) | 89 (84,103) | 92 (84,95) | 0.1432 |
| Hypertension | 78 (74.29%) | 23 (76.67%) | 17 (94.44%) | 0.1700 |
| Coronary artery disease | 27 (25.71%) | 9 (30.00%) | 8 (44.44%) | 0.2646 |
| COPD | 10 (9.62%) | 5 (16.67%) | 4 (22.22%) | 0.2438 |
| eGFR [ml/min/1.73 m2] | 86 (68,101) | 79 (61,98) | 85 (50,92) | 0.4656 |
| Diabetes mellitus | 42 (40.00%) | 11 (36.67%) | 11 (64.71%) | 0.1276 |
| Current smoking | 28 (27.72%) | 6 (20.69%) | 3 (16.67%) | 0.5083 |
| Medication use | ||||
| Beta blockers | 50 (47.17%) | 25 (86.21%) | 13 (72.22%) | 0.0003 |
| Aspirin | 54 (50.94%) | 20 (68.97%) | 11 (61.11%) | 0.1969 |
| ACE inhibitors or ARBs | 53 (50.00%) | 24 (82.76%) | 15 (83.33%) | 0.0006 |
| Furosemide | 2 (1.90%) | 18 (62.07%) | 12 (66.67%) | <0.0001 |
| Spironolactone | 6 (5.66%) | 7 (24.14%) | 1 (5.56%) | 0.0080 |
| Statins | 75 (70.75%) | 25 (86.21%) | 12 (66.67%) | 0.2003 |
| Long-acting nitrates | 8 (7.55%) | 2 (7.14%) | 3 (16.67%) | 0.4224 |
| Hydralazine | 2 (1.89%) | 1 (3.45%) | 3 (16.67%) | 0.0114 |
| Warfarin | 8 (7.55%) | 5 (17.24%) | 0 (0.00%) | 0.0979 |
| Calcium-channel blockers | 30 (28.30%) | 5 (17.24%) | 8 (44.44%) | 0.1304 |
| Thiazides | 23 (21.90%) | 3 (10.34%) | 3 (16.67%) | 0.3599 |
For continuous variables, values denote mean ± standard deviation (when normally distributed) or median (25th, 75th percentile) (otherwise). For discrete variables, values denote count (percentage). Systolic and diastolic blood pressures refer to the right-brachial blood pressures as obtained using the VaSera device. P values < 0.05 are printed in bold. Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; BMI, body mass index; COPD, chronic obstructive pulmonary disease; eGFR, estimated glomerular filtration rate; HF, heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction.
Arterial stiffness
Heart-to-ankle PWV
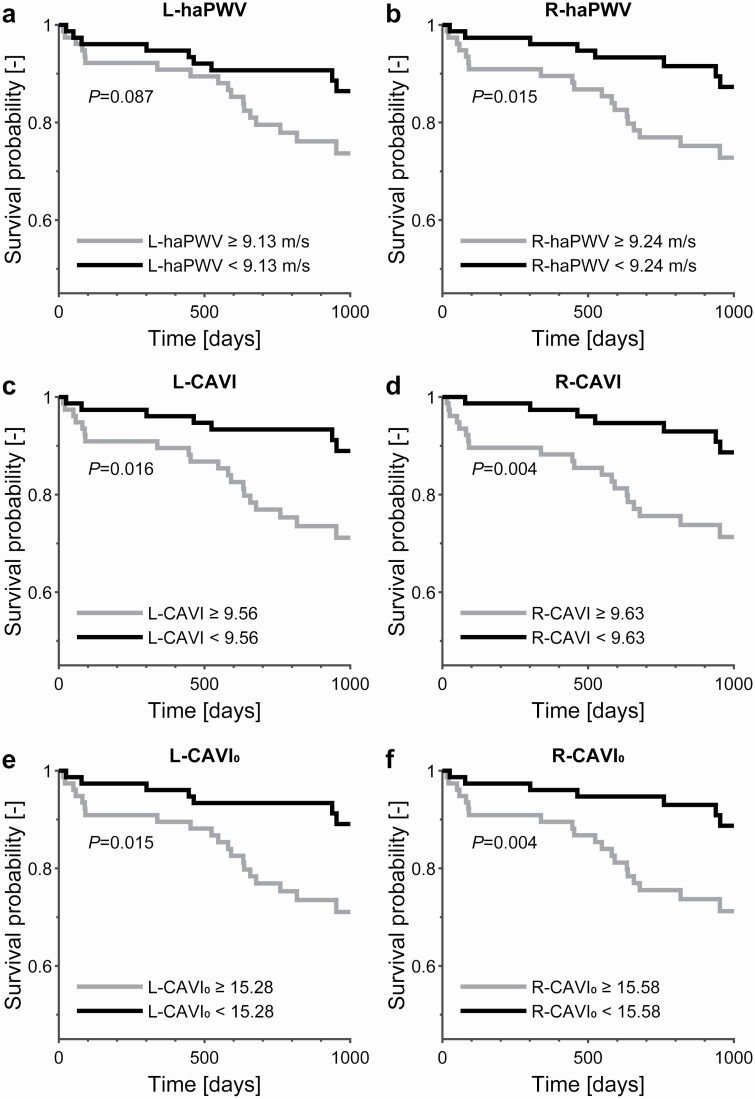
L-haPWV and R-haPWV did not differ significantly (9.45 vs. 9.38 m/s, P = 0.31). Survival analysis (Figure 1a,b), however, revealed no crude significance for L-haPWV (P = 0.087), in contrast to R-haPWV (P = 0.015). In Cox proportional hazards regression (Table 2), this trend in left–right differences persisted, with R-haPWV demonstrating consistently smaller P values for prediction of DHFA than L-haPWV. R-haPWV remained significant after adjustment for the MAGGIC risk score and HF status (P = 0.0280) and additional adjustment for systolic blood pressure (P = 0.0254). L-haPWV, however, was not a significant predictor after these adjustments (P = 0.1508 and P = 0.1395, respectively).
Figure 1.
Kaplan–Meier survival curves for left (L) and right (R) heart-to-ankle pulse wave velocity (haPWV; a and b, respectively), cardio-ankle vascular index (CAVI; c and d, respectively), and CAVI0 (e and f, respectively). Median values are used as cutoffs. P values are based on log-rank tests.
Table 2.
Cox proportional hazards regression results
| n | Standardized HR [95% CI] | P value | |
|---|---|---|---|
| Unadjusted | |||
| L-haPWV | 153 | 1.51 [1.12–2.04] | 0.0071 |
| R-haPWV | 154 | 1.66 [1.19–2.31] | 0.0026 |
| L-CAVI | 153 | 1.45 [1.08–1.93] | 0.0119 |
| R-CAVI | 154 | 1.58 [1.14–2.20] | 0.0058 |
| L-CAVI0 | 153 | 1.39 [1.05–1.84] | 0.0220 |
| R-CAVI0 | 154 | 1.44 [1.08–1.92] | 0.0141 |
| Adjusted for MAGGIC and HF status | |||
| L-haPWV | 153 | 1.25 [0.92–1.69] | 0.1508 |
| R-haPWV | 154 | 1.46 [1.04–2.05] | 0.0280 |
| L-CAVI | 153 | 1.21 [0.90–1.62] | 0.2123 |
| R-CAVI | 154 | 1.44 [1.02–2.04] | 0.0446 |
| L-CAVI0 | 153 | 1.16 [0.86–1.58] | 0.3302 |
| R-CAVI0 | 154 | 1.30 [0.95–1.78] | 0.1198 |
| Adjusted for MAGGIC, HF status, and SBP | |||
| L-haPWV | 153 | 1.27 [0.92–1.75] | 0.1395 |
| R-haPWV | 154 | 1.58 [1.06–2.36] | 0.0254 |
| L-CAVI | 153 | 1.21 [0.90–1.62] | 0.2155 |
| R-CAVI | 154 | 1.44 [1.01–2.06] | 0.0439 |
| L-CAVI0 | 153 | 1.16 [0.85–1.58] | 0.3363 |
| R-CAVI0 | 154 | 1.30 [0.93–1.80] | 0.1214 |
P values < 0.05 are printed in bold. Abbreviations: CI, confidence interval; HF, heart failure; HR, hazard ratio; MAGGIC, Meta-Analysis Global Group in Chronic Heart Failure; SBP, systolic blood pressure.
CAVI
L-CAVI and R-CAVI did not differ significantly (9.75 vs. 9.64, P = 0.27). In survival analysis (Figure 1c,d), both L-CAVI and R-CAVI predicted the risk of DHFA. In Cox regression (Table 2), right-sided values appeared to be stronger predictors of outcome than left-sided values. After adjustment for the MAGGIC risk score and HF status, only R-CAVI remained a significant predictor (P = 0.0446) of DHFA, which remained significant after further correction for systolic blood pressure (P = 0.0439).
CAVI0
Similar to haPWV and CAVI, no left–right CAVI0 difference was observed. CAVI0 crude survival curves were very similar to CAVI survival curves (compare Figure 1e vs. c and Figure 1f vs. d), with R-CAVI0 showing smaller P values for DHFA prediction than L-CAVI0. In Cox regression analyses (Table 2), left–right differences in P value persisted, and L-CAVI0 and R-CAVI0 were only significantly predictive in an unadjusted model.
Comparison of predictive powers of haPWV, CAVI, and CAVI0
Figure 2 shows the results of Cox regression from Table 2 as volcano plots. In all 3 panels, a trend emerged suggesting that, with increasing blood pressure correction (i.e., from haPWV to CAVI to CAVI0), predictive power decreases as visualized by the arrows (standardized hazard ratio decreases while P increases, noting that the y-axis shows negative log-transformed P values).
Figure 2.
Volcano plots for prediction of death or heart failure (HF)-related hospital admission using left (L) and right (R) heart-to-ankle pulse wave velocity (haPWV), cardio-ankle vascular index (CAVI), and CAVI0. Right-pointing triangles indicate right-sides measures; left-pointing triangles indicate left-sided measures. Small horizontal lines are drawn to clarify triangle direction. Long-tailed, diagonal arrows illustrate that, with increasing blood pressure correction, power to predict outcome decreases, suggesting that part of the predictive power of blood pressure-dependent stiffness metrics arises from the predictive power of blood pressure per se. This trend persists in unadjusted (a), MAGGIC and HF status-adjusted (b), and additionally SBP-adjusted (c) analyses. Abbreviations: HR, hazard ratio; MAGGIC, Meta-Analysis Global Group in Chronic Heart Failure; SBP, systolic blood pressure.
DISCUSSION
We assessed the ability of 3 arterial stiffness metrics based on heart-to-ankle transit time measurements to predict DHFA in a US-based population: haPWV, CAVI, and CAVI0. We observed that (i) going from haPWV to CAVI to CAVI0, predictive power shows a decreasing trend, and (ii) metrics obtained from the right body side appeared to show a stronger predictive ability than those from the left body side.
Arterial PWV, and hence haPWV, depends on blood pressure at the time of measurement.5,6 Already in 1923, Bramwell and Hill observed this dependence,4 which is caused by the nonlinear mechanical behavior of the artery wall, which can be summarized through an exponential relationship between arterial pressure and diameter.5,20,21 Several studies have since implemented a pressure correction of PWV, either statistically or through computation of pressure-corrected indices.22 One such index is CAVI, which, based on the Bramwell–Hill equation and a linearized form of the distensibility equation, corrects PWV for its blood pressure dependence.7 CAVI is essentially a form of stiffness index β, though now obtained by conversion of a transit time PWV (instead of β’s normal calculation based on a local distension and pressure measurement). In a study from 2017, CAVI was, on a theoretical/mathematical basis, suggested to show a residual acute blood pressure dependence.8 In that same study, CAVI0 was proposed, which differed from CAVI in 2 principal ways.23 First, and most important, whereas CAVI assumes a linearized, diastolic-to-systolic compliance term, CAVI0 uses a diastolic compliance term, based on the rationale that a (diastolic) foot-to-foot PWV metric such as haPWV acutely depends most strongly on diastolic blood pressure.4,24 Second, an additional term, (see equation (4)) was added, which overcomes an assumption in the derivation of stiffness index beta.25 CAVI0 values can easily be obtained through conversion of CAVI values,16,18 or through direct calculation by using equation (4).
In the initial CAVI0 study,8 regular CAVI was shown to be substantially less pressure dependent than PWV. From that study, it followed that acute blood pressure dependence decreases from PWV (most) to CAVI (less) to CAVI0 (least). Strikingly, in the present study, we show that predictive power of these indices also decreases, in the same order. This suggests that, the better the blood pressure correction, the weaker the predictive power of a stiffness metric. Indeed, this may make sense, as blood pressure per se is one of the most well-known predictors of mortality.26 PWV, hence, combines intrinsic arterial stiffness with blood pressure information, making it an excellent predictor.
Tomiyama et al. compared the predictive ability of baPWV with that of β-transformed baPWV.12 Note that this β transformation uses the exact same equation that is used to compute CAVI from haPWV, hence, their β-transformed baPWV may be regarded as a “BAVI,” a brachial–ankle vascular index, analogous to CAVI. They observed that in young normotensive men, regular baPWV was a better predictor of the development of hypertension and of retinopathy than the β-transformed baPWV, which may be due to the added predictive value of blood pressure present in baPWV. The present study confirms this trend, with CAVI0 additionally showing a trend toward an even further decreased predictive power than CAVI, which would be in line with its theoretically stronger blood pressure correction than CAVI. Kim et al. also compared 2 PWV metrics (haPWV and carotid–femoral PWV) with their β-transformed counterparts.27 For prediction of cardiovascular events, β-transformed parameters performed poorer than nontransformed PWVs. When predicting all-cause mortality, carotid–femoral PWV and β-transformed carotid–femoral PWV performed similarly overall, though CAVI showed improved performance over haPWV.
Studying the acute blood pressure correction performance of a stiffness measure is not trivial.28 Several studies have presented cross-sectional analyses where they studied the relationship between a stiffness measure and blood pressure (e.g., 29,30). However, as has been pointed out previously,22,29,31 such analysis does not prove or disprove the performance of a stiffness measure in correcting for acute blood pressure dependence. Despite this, CAVI0 has been criticized based on such analyses and the observation that cross-sectionally, CAVI0 correlates negatively with Pd in healthy subjects. While this was deemed “inexplicable,” 30 it can be readily explained using pulse pressure, which is higher in subjects with stiffer arteries,32 particularly at higher age.33 As Ps was normal (the subjects were healthy and classified normotensive), the Pd of subjects with stiffer arteries was probably relatively low, explaining the observed negative correlation.34 To accurately study the acute pressure dependence, short-term (acute) repeated-measures studies are needed in which stiffness measures are computed longitudinally, during a changing blood pressure pattern.
Shirai et al. performed such a study into the blood pressure dependence of CAVI, in which they elegantly manipulated blood pressure using 2 different drugs: a β 1-adrenoreceptor blocker (metoprolol), thought to only reduce cardiac contractility (and not affect arterial smooth muscle tone), and an α 1-adrenoreceptor blocker (doxazosin), thought to reduce blood pressure through vasodilation.35 Both drugs elicited a similar blood pressure reduction and both yielded a decrease in haPWV, yet only the α 1 blocker influenced CAVI, suggesting that CAVI is blood pressure independent. In a later report, Shirai et al. used the same study data to study the response of CAVI0 to these blood pressure stimuli; results were similar to those for CAVI (CAVI0 did not react to the β 1 blocker, but did to the α 1 blocker).36 They hence concluded that CAVI0 performed equally well as CAVI. However, due to the small sample size in this comparison study (n = 9), a lack of statistical power may have obscured potential CAVI–CAVI0 differences.
A more recent study in 60 individuals examined the responses of CAVI and CAVI0 to a blood pressure change induced by a cold pressor test and by isometric handgrip exercise. Preliminary results from this experimental study showed no blood pressure dependence of CAVI0 but a slight dependence of CAVI, confirming the previous theoretical/mathematical finding of additional blood pressure correction of CAVI0 over CAVI.8,37,38
We observed possible body-side differences in prediction using CAVI and CAVI0, which will require further study in larger samples. Notably, the left- and right-sided indices presented herein follow an identical arterial path from the heart to the femoral bifurcation (i.e., both left and right stiffness indices use the right brachial cuff in the haPWV calculation process).16,17 This implies that any body-side differences, if present, arise from differences in arterial stiffness of the left and right legs, and/or differences in measurement of the ankle blood pressure waveforms. A potential factor contributing to this difference could be unilateral lower-limb atherosclerosis, potentially affecting recording accuracy, warranting further studies. A previous cross-sectional study reported more pronounced body-side differences in haPWV related to cardiovascular disease.39 It is also known that body-side differences in blood pressure per se confer cardiovascular risk.40,41 However, we are unaware of studies comparing the predictive power of left- and right-sided arterial stiffness measurements. It should be noted that whereas we found that right-sided indices appear to be more robust predictors in our sample, this may have arisen by random noise. Given the small sample size, confidence intervals for indices on both sides were wide and partially overlapping; therefore, our findings should not be considered definitive in this regard, and body-side differences should be examined in future studies
Limitations
Our study included 154 subjects and recorded 32 composite events. Despite the clear and consistent trends observed in predictive power from haPWV to CAVI to CAVI0, this modest sample size limits our power to statistically discriminate between the different arterial stiffness indices. Also, although many potential additional confounders were recorded, we were unable to include all individually into our Cox regression. It is for this reason that we corrected using the MAGGIC score—a simple yet powerful score for risk stratification in HF.19 Our sample was derived from patients referred for a cardiac magnetic resonance imaging study at our institution and thus may not be generalizable to other clinical populations or to the general population. Furthermore, consistent with the patient population at VA medical centers, our sample was composed predominantly of men; future studies including larger proportions of women will hence be valuable. Also, since our study focused on HF, our adjudicated endpoint was DHFA; other studies will be valuable to assess for composite cardiorenal endpoints. We did not discern the relationship between cardiovascular and noncardiovascular death. Finally, all our stiffness metrics were based on the heart-to-ankle transit time, which was assessed using a combination of the phonocardiogram and 2 oscillometric cuffs, rather than carotid–femoral PWV, which is considered the noninvasive gold-standard for large artery stiffness assessments.7,9,16 The same blood pressure correction method can also be used for PWVs in other arterial beds and/or acquired using different techniques, such as a tonometric carotid–femoral PWV. Further studies are needed to assess whether our present finds can be extrapolated to such measurements.
We conclude that for arterial stiffness metrics, an increasing amount of blood pressure correction may weaken their predictive power, which may be due to the progressively smaller confounding prediction of blood pressure. In addition, our findings raise the possibility that right-sided arterial stiffness measurements may exhibit a more robust predictive ability for DHFA than left-sided measurements, which should be examined in future studies.
Perspectives
The dependence of PWV—the most commonly used clinical arterial stiffness metric—on blood pressure is often regarded as an artifact. Most studies therefore statistically correct PWV for blood pressure. Recent advances have led to more advanced arterial stiffness metrics such as CAVI and CAVI0 that are intrinsically corrected for blood pressure. Our study suggests a downside of such corrected metrics, in that their predictive power shows a decreasing trend with an increasing amount of blood pressure correction. Therefore, for maximally powered outcome prediction, PWV (even if statistically corrected for blood pressure) may be advantageous over CAVI, which in turn may be advantageous over CAVI0. PWV remains an excellent integrative predictor of outcome, presumably because it encapsulates arterial stiffness as well the intrinsic effect of blood pressure on the arterial wall.
FUNDING
This work was supported by an investigator-initiated grant from Fukuda Denshi (J.A.C). B.S. is supported by the European Union’s Horizon 2020 Research and Innovation program (grant 793805). J.A.C. is supported by NIH grants R01-HL 121510, U01-TR003734, 3U01TR003734 - 01W1, U01-HL160277, R33-HL-146390, R01-HL153646, K24-AG070459, R01-AG058969, R01-HL104106, P01-HL094307, R03-HL146874, R56-HL136730, R01 HL155599, R01 HL157264, R01HL155, and 1R01HL153646-01.
DISCLOSURE
J.A.C. has recently consulted for Bayer, Sanifit, Fukuda-Denshi, Bristol-Myers Squibb, JNJ, Edwards Life Sciences, Merck, NGM Biopharmaceuticals, and the Galway-Mayo Institute of Technology. He received University of Pennsylvania research grants from National Institutes of Health, Fukuda-Denshi, Bristol-Myers Squibb, and Microsoft, and Abbott. He is named as inventor in a University of Pennsylvania patent for the use of inorganic nitrates/nitrites for the treatment of Heart Failure and Preserved Ejection Fraction and for the use of biomarkers in heart failure with preserved ejection fraction. He has received payments for editorial roles from the American Heart Association, the American College of Cardiology, and Wiley. He has received research device loans from Atcor Medical, Fukuda-Denshi, Uscom, NDD Medical Technologies, Microsoft, and MicroVision Medical.
REFERENCES
- 1. Vlachopoulos C, Aznaouridis K, Stefanadis C. Prediction of cardiovascular events and all-cause mortality with arterial stiffness: a systematic review and meta-analysis. J Am Coll Cardiol 2010; 55:1318–1327. [DOI] [PubMed] [Google Scholar]
- 2. Chirinos JA, Segers P, Hughes T, Townsend R. Large-artery stiffness in health and disease: JACC state-of-the-art review. J Am Coll Cardiol 2019; 74:1237–1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Ben-Shlomo Y, Spears M, Boustred C, May M, Anderson SG, Benjamin EJ, Boutouyrie P, Cameron J, Chen CH, Cruickshank JK, Hwang SJ, Lakatta EG, Laurent S, Maldonado J, Mitchell GF, Najjar SS, Newman AB, Ohishi M, Pannier B, Pereira T, Vasan RS, Shokawa T, Sutton-Tyrell K, Verbeke F, Wang KL, Webb DJ, Willum Hansen T, Zoungas S, McEniery CM, Cockcroft JR, Wilkinson IB. Aortic pulse wave velocity improves cardiovascular event prediction: an individual participant meta-analysis of prospective observational data from 17,635 subjects. J Am Coll Cardiol 2014; 63:636–646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Bramwell JC, McDowall RJS, McSwiney BA. The variation of arterial elasticity with blood pressure in man (part I). Proc R Soc B 1923; 94:450–454. [Google Scholar]
- 5. Spronck B, Heusinkveld MH, Vanmolkot FH, Roodt JO, Hermeling E, Delhaas T, Kroon AA, Reesink KD. Pressure-dependence of arterial stiffness: potential clinical implications. J Hypertens 2015; 33:330–338. [DOI] [PubMed] [Google Scholar]
- 6. Segers P, Rietzschel ER, Chirinos JA. How to measure arterial stiffness in humans. Arterioscler Thromb Vasc Biol 2020; 40:1034–1043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Shirai K, Utino J, Otsuka K, Takata M. A novel blood pressure-independent arterial wall stiffness parameter; cardio-ankle vascular index (CAVI). J Atheroscler Thromb 2006; 13:101–107. [DOI] [PubMed] [Google Scholar]
- 8. Spronck B, Avolio AP, Tan I, Butlin M, Reesink KD, Delhaas T. Arterial stiffness index beta and cardio-ankle vascular index inherently depend on blood pressure but can be readily corrected. J Hypertens 2017; 35:98–104. [DOI] [PubMed] [Google Scholar]
- 9. Giudici A, Khir AW, Reesink KD, Delhaas T, Spronck B. Five years of cardio-ankle vascular index (CAVI) and CAVI0: how close are we to a pressure-independent index of arterial stiffness? J Hypertens 2021; 39:2128–2138. [DOI] [PubMed] [Google Scholar]
- 10. Matsushita K, Ding N, Kim ED, Budoff M, Chirinos JA, Fernhall B, Hamburg NM, Kario K, Miyoshi T, Tanaka H, Townsend R. Cardio-ankle vascular index and cardiovascular disease: systematic review and meta-analysis of prospective and cross-sectional studies. J Clin Hypertens 2019; 21:16–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Townsend RR, Wilkinson IB, Schiffrin EL, Avolio AP, Chirinos JA, Cockcroft JR, Heffernan KS, Lakatta EG, McEniery CM, Mitchell GF, Najjar SS, Nichols WW, Urbina EM, Weber T; American Heart Association Council on Hypertension . Recommendations for improving and standardizing vascular research on arterial stiffness: a scientific statement from the American Heart Association. Hypertension 2015; 66:698–722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Tomiyama H, Ohkuma T, Ninomiya T, Nakano H, Matsumoto C, Avolio A, Kohro T, Higashi Y, Maruhashi T, Takase B, Suzuki T, Ishizu T, Ueda S, Yamazaki T, Furumoto T, Kario K, Inoue T, Koba S, Takemoto Y, Hano T, Sata M, Ishibashi Y, Node K, Maemura K, Ohya Y, Furukawa T, Ito H, Chikamori T, Yamashina A. Brachial-ankle pulse wave velocity versus its stiffness index β-transformed value as risk marker for cardiovascular disease. J Am Heart Assoc 2019; 8:e013004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Nagueh SF, Smiseth OA, Appleton CP, Byrd BF III, Dokainish H, Edvardsen T, Flachskampf FA, Gillebert TC, Klein AL, Lancellotti P, Marino P, Oh JK, Popescu BA, Waggoner AD. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 2016; 29:277–314. [DOI] [PubMed] [Google Scholar]
- 14. Chirinos JA, Akers SR, Trieu L, Ischiropoulos H, Doulias PT, Tariq A, Vasim I, Koppula MR, Syed AA, Soto-Calderon H, Townsend RR, Cappola TP, Margulies KB, Zamani P. Heart failure, left ventricular remodeling, and circulating nitric oxide metabolites. J Am Heart Assoc 2016; 5:e004133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Chirinos JA, Sardana M, Ansari B, Satija V, Kuriakose D, Edelstein I, Oldland G, Miller R, Gaddam S, Lee J, Suri A, Akers SR. Left atrial phasic function by cardiac magnetic resonance feature tracking is a strong predictor of incident cardiovascular events. Circ Cardiovasc Imaging 2018; 11:e007512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Spronck B, Mestanik M, Tonhajzerova I, Jurko A, Jurko T, Avolio AP, Butlin M. Direct means of obtaining CAVI0—a corrected cardio-ankle vascular stiffness index (CAVI)—from conventional CAVI measurements or their underlying variables. Physiol Meas 2017; 38:N128–N137. [DOI] [PubMed] [Google Scholar]
- 17. Takahashi K, Yamamoto T, Tsuda S, Okabe F, Shimose T, Tsuji Y, Suzuki K, Otsuka K, Takata M, Shimizu K, Uchino J, Shirai K. Coefficients in the CAVI equation and the comparison between CAVI with and without the coefficients using clinical data. J Atheroscler Thromb 2019; 26:465–475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Spronck B, Mestanik M, Tonhajzerova I, Jurko A, Tan I, Butlin M, Avolio AP. Easy conversion of cardio-ankle vascular index into CAVI0: influence of scale coefficients. J Hypertens 2019; 37:1913–1914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Pocock SJ, Ariti CA, McMurray JJ, Maggioni A, Køber L, Squire IB, Swedberg K, Dobson J, Poppe KK, Whalley GA, Doughty RN; Meta-Analysis Global Group in Chronic Heart Failure . Predicting survival in heart failure: a risk score based on 39 372 patients from 30 studies. Eur Heart J 2013; 34:1404–1413. [DOI] [PubMed] [Google Scholar]
- 20. Hayashi K, Handa H, Nagasawa S, Okumura A, Moritake K. Stiffness and elastic behavior of human intracranial and extracranial arteries. J Biomech 1980; 13:175–184. [DOI] [PubMed] [Google Scholar]
- 21. Stefanadis C, Dernellis J, Vlachopoulos C, Tsioufis C, Tsiamis E, Toutouzas K, Pitsavos C, Toutouzas P. Aortic function in arterial hypertension determined by pressure-diameter relation: effects of diltiazem. Circulation 1997; 96:1853–1858. [DOI] [PubMed] [Google Scholar]
- 22. Spronck B, Delhaas T, Butlin M, Reesink KD, Avolio AP. Options for dealing with pressure dependence of pulse wave velocity as a measure of arterial stiffness: an update of cardio-ankle vascular index (CAVI) and CAVI0. Pulse 2018; 5:106–114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Spronck B. Stiff vessels approached in a flexible way: advancing quantification and interpretation of arterial stiffness. Artery Res 2018; 21:63–68. [Google Scholar]
- 24. Gao M, Cheng HM, Sung SH, Chen CH, Olivier NB, Mukkamala R. Estimation of pulse transit time as a function of blood pressure using a nonlinear arterial tube-load model. IEEE Trans Biomed Eng 2017; 64:1524–1534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Kawasaki T, Sasayama S, Yagi S, Asakawa T, Hirai T. Non-invasive assessment of the age related changes in stiffness of major branches of the human arteries. Cardiovasc Res 1987; 21:678–687. [DOI] [PubMed] [Google Scholar]
- 26. Ettehad D, Emdin CA, Kiran A, Anderson SG, Callender T, Emberson J, Chalmers J, Rodgers A, Rahimi K. Blood pressure lowering for prevention of cardiovascular disease and death: a systematic review and meta-analysis. Lancet 2016; 387:957–967. [DOI] [PubMed] [Google Scholar]
- 27. Kim ED, Ballew SH, Tanaka H, Heiss G, Coresh J, Matsushita K. Short-term prognostic impact of arterial stiffness in older adults without prevalent cardiovascular disease. Hypertension 2019; 74:1373–1382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Spronck B. Disentangling arterial stiffness and blood pressure. Heart Lung Circ 2021; 30:1599–1601. [DOI] [PubMed] [Google Scholar]
- 29. Kubozono T, Miyata M, Ueyama K, Nagaki A, Otsuji Y, Kusano K, Kubozono O, Tei C. Clinical significance and reproducibility of new arterial distensibility index. Circ J 2007; 71:89–94. [DOI] [PubMed] [Google Scholar]
- 30. Shirai K, Suzuki K, Tsuda S, Shimizu K, Takata M, Yamamoto T, Maruyama M, Takahashi K. Comparison of cardio-ankle vascular index (CAVI) and CAVI0 in large healthy and hypertensive populations. J Atheroscler Thromb 2019; 26:603–615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Benetos A. Assessment of arterial stiffness in an older population: the interest of the cardio-ankle vascular index (CAVI). Eur Heart J Suppl 2017; 19:B11–B16. [Google Scholar]
- 32. Niiranen TJ, Kalesan B, Mitchell GF, Vasan RS. Relative contributions of pulse pressure and arterial stiffness to cardiovascular disease. Hypertension 2019; 73:712–717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Mitchell GF, Wang N, Palmisano JN, Larson MG, Hamburg NM, Vita JA, Levy D, Benjamin EJ, Vasan RS. Hemodynamic correlates of blood pressure across the adult age spectrum: noninvasive evaluation in the Framingham Heart Study. Circulation 2010; 122:1379–1386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Spronck B, Jurko A, Mestanik M, Avolio AP, Tonhajzerova I. Reply to comments: using the cardio-ankle vascular index (CAVI) or the mathematical correction form (CAVI0) in clinical practice. Int J Mol Sci 2020; 21:2647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Shirai K, Song M, Suzuki J, Kurosu T, Oyama T, Nagayama D, Miyashita Y, Yamamura S, Takahashi M. Contradictory effects of β1- and α1- aderenergic receptor blockers on cardio-ankle vascular stiffness index (CAVI)—CAVI independent of blood pressure. J Atheroscler Thromb 2011; 18:49–55. [DOI] [PubMed] [Google Scholar]
- 36. Shirai K, Shimizu K, Takata M, Suzuki K. Independency of the cardio-ankle vascular index from blood pressure at the time of measurement. J Hypertens 2017; 35:1521–1523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Mestanik M, Spronck B, Jurko A Jr, Mestanikova A, Jurko T, Grendar M, Avolio AP, Tonhajzerova I. Enhanced cardio-ankle vascular index (CAVI0) is experimentally less blood pressure dependent than standard CAVI. J Hypertens 2019; 37:e209. [Google Scholar]
- 38. Mestanik M, Spronck B, Jurko A, Mestanikova A, Jurko T, Butlin M, Avolio A, Olexova LB, Visnovcova Z, Sekaninova N, Tonhajzerova I. P135 assessment of novel blood pressure corrected cardio-ankle vascular index in response to acute blood pressure changes. Artery Res 2020; 25:S173. [Google Scholar]
- 39. Yeragani VK, Kumar R, Bar KJ, Chokka P, Tancer M. Exaggerated differences in pulse wave velocity between left and right sides among patients with anxiety disorders and cardiovascular disease. Psychosom Med 2007; 69:717–722. [DOI] [PubMed] [Google Scholar]
- 40. Weinberg I, Gona P, O’Donnell CJ, Jaff MR, Murabito JM. The systolic blood pressure difference between arms and cardiovascular disease in the Framingham Heart Study. Am J Med 2014; 127:209–215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Agarwal R, Bunaye Z, Bekele DM. Prognostic significance of between-arm blood pressure differences. Hypertension 2008; 51:657–662. [DOI] [PubMed] [Google Scholar]