Abstract
Background
Tuberculosis (TB) and diabetes mellitus (DM) have negative synergistic impact on each other. Global guidelines recommend collaborative efforts to address this synergy, but implementation has been slow. Part of the reason is lack of adequate evidence on the operational feasibility of existing tools and mechanisms of collaboration. The objective of this study was to assess the yield of DM screening among TB patients using risk scoring tools combined with blood tests as a feasible strategy for early detection to improve TB/DM treatment outcomes.
Methods
Between September 2020 and December 2021, we conducted a cross-sectional study among patients receiving TB treatment in public health facilities in Addis Ababa, Ethiopia. Trained health workers collected data on symptoms and risk scoring checklists before testing for random and fasting blood glucose levels. We used logistic regression analyses techniques to determine factors associated with increased DM detection. A receiver-operating characteristic curve was constructed to determine the performance of the risk scoring checklist.
Results
Of 2381 TB patients screened, 197 (8.3%) had DM of which 48.7% were newly diagnosed. Having a family history of DM predicted DM with Odds Ratio (OR) of 5.36 (95% Confidence Interval, [3.67, 7.83]) followed by age ≥ 45 years (OR = 4.64, [3.18, 6.76]). Having one or more “symptoms” of DM was a significant predictor (OR 2.88, 95% CI, 2.06–4.01). Combining risk scores with symptoms predicted DM diagnosis with sensitivity of 94.7%, but specificity was low at 29.4%. In patients with known treatment outcome status, death rate was high.
Conclusions
Almost a half of TB patients with DM did not know their status. A simple tool that combined risk factors with symptoms accurately predicted a subsequent diagnosis of DM. Such tools can help avoid high rates of death among TB patients suffering from DM through early detection.
Keywords: Diabetes mellitus, Risk scoring, Tuberculosis, Early detection, Mortality
1. Introduction
People living with diabetes mellitus (DM) are at increased risk to become infected with Mycobacterium tuberculosis (TB), progressing from latent tuberculosis infection to active TB disease, and suffering adverse TB treatment outcomes [1], [2] According to the 2021 report of the International Diabetes Federation (IDF), the global prevalence of diabetes among adults aged 20–79 was estimated to be 10.5% (537 million people) [3]. In the African region, about 1 in 22 adults (24 million) were estimated to be living with diabetes, 54% of these undiagnosed, and nearly half a million died in 2021 [3]. About 13.7% of patients with active TB are estimated to have DM [4].
Studies indicate that patients with TB/DM comorbidities have a two-fold higher risk of death during TB treatment and a higher risk of TB relapse after treatment mainly because either the diseases are detected late or there is poor glycemic control [5]. Earlier detection and treatment of DM has a beneficial effect in reducing the risk of complications to the individual patient, and to reduce the burden of disease on the health system [6]. Strategies for earlier detection often included recommendations to use risk scoring checklists followed by blood test, but such approaches were not evaluated in TB patients [7].
Ethiopia, with a population of 115 million is the second most populous country in Africa and is a high TB burden country with an estimated TB incidence of 132 per 100,000 population and mortality rate in drug-susceptible TB (DS-TB) patients of 19.2 per 100,000 population [8]. It is also estimated that there are over 2 million adults with undiagnosed diabetes in Ethiopia. But even when diagnosed and treated, a significant proportion of patients do not achieve optimal glycemic control. A study documented the prevalence of diabetes (8.3%) among Ethiopian TB patients to be twice higher than the diabetes prevalence (3.9%) reported in the general population at the time of the study [9]. Another locally published study indicated that 5.8% of DM patients were reported to have active TB [10].
The high proportion of people with DM and TB remaining undiagnosed in Ethiopia or being diagnosed at a late stage is mainly because of the lack of early detection of the diseases which leads to more severe disease presentations, higher mortality, and higher cost of treatment. If DM is diagnosed early in TB patients and optimal glycemic control is maintained during TB treatment, the treatment outcomes can be improved [11]. However, data on the rate of undiagnosed DM, treatment outcome and DM screening using risk scoring among TB patients is limited in Ethiopia.
The objective of this study was to assess the yield of DM screening among TB patients using risk scoring tools combined with blood tests as a feasible strategy for earlier detection to improve TB/DM treatment outcomes. We also assessed TB treatment outcomes among TB/DM co-morbid patients for whom outcome data was available.
2. Methods
2.1. Design and participants
This cross-sectional study was conducted in Addis Ababa, Ethiopia between September 2020 and December 2021. First, we conducted a baseline assessment to understand the availability of DM screening services in the public health facilities in Addis Ababa. Then we trained the health care workers, supplied glucometers and test strips, and conducted routine screening for DM amongst TB patients in public health facilities. All consenting adult patients with confirmed TB diagnosis were included irrespective of their phase of TB treatment. We asked each patient about current diagnosis of DM, risk factors for DM, and any symptom suggestive of DM or its complications. We used finger-prick blood tests to confirm DM diagnosis. A Random Blood Sugar (RBS) of ≥ 200 gm/dl or Fasting Blood Sugar (FBS) of ≥ 126 gm/dl on two separate occasions or previous diagnosis as confirmed by medical records, was considered confirmatory of DM diagnosis [12].
3. Data collection
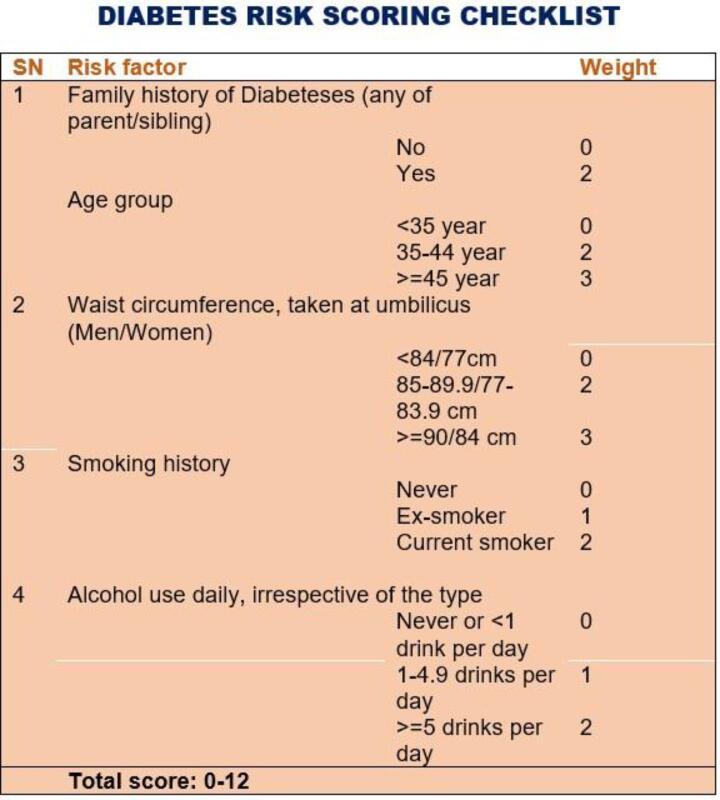
We recorded risk factors for and symptoms of DM in a standardized questionnaire before performing the blood tests. The risk factors included family history of DM, age, waist circumference, smoking history, and alcohol use. The scores for each risk factor ranged from 0 to 3. The values for each of the risk factors are shown in Fig. 1.. We also administered a checklist of nine symptoms known to be associated with acute or chronic complications of DM. These included three “poly symptoms” (excessive urination, thirst, and hunger), unexplained weight loss, blurred vision, fatigue, tingling/numbness in the limbs, frequent infection, and delayed wound healing. The symptoms had an equal weight of zero for no symptom and 1 otherwise.
Fig. 1.
Diabetes risk scoring checklist.
3.1. Data analysis
We used Statistical Package for Social Sciences (SPSS) version 25 for data entry and analysis [13]. We used descriptive analyses techniques to describe the baseline characteristics and TB/DM co-morbidity rates. We then computed median values for the cumulative risk scoring values to categorize the participants into “high risk” or “low risk” groups. We also categorized the participants as “symptomatic” or “asymptomatic” depending on the presence or absence of any one or more of the nine symptoms described above. We used binary logistic regression (LR) analyses with adjusted Odds Ratio (aOR) and 95% Confidence Interval (95%CI) to identify predictors of DM. In addition to the symptom and risk score categories, we included variables with a P-value of < 0.25 in a univariate analysis into the multivariate LR model.
For patients with known treatment outcomes, we calculated unfavourable treatment outcomes. Since treatment outcome was not known for all patients with diabetes co-morbidity, we performed sensitivity analysis using various scenarios.
We then tested the accuracy of the risk factors and symptoms against blood test results or known DM as reference standards. The results are presented as receiver operating characteristic curves (ROC) with areas under the curve (AUC).
4. Ethical considerations
The Ethics review committee of Addis Ababa City Council Health Bureau reviewed and approved the study protocol. Study participants provided informed verbal consent before being enrolled in the study. Patient data was handled confidentially, and anonymized data base was used for analysis. Patients who were diagnosed with diabetes received appropriate care according to the standard of care.
5. Results
5.1. Baseline characteristics
We screened 2,381 TB patients, their median age was 51 years and 52.6% were male. Of these, 2.7% had drug resistant (DR-TB). The HIV co-infection rate was 15%. Table 1 summarizes the baseline characteristics of the study participants.
Table 1.
Baseline characteristics of the respondents (n = 2,379).
| Characteristic | Value | |
|---|---|---|
| Age (in years) | Median, IQR | 31 (24,42) |
| Sex (n, %) | Male | 1247 (52.6) |
| Female | 1125 (47.4) | |
| Missing | 7 (0.3) | |
| Type of TB (n, %) | Extrapulmonary TB | 908 (38.2) |
| Bacteriologically confirmed pulmonary TB | 868 (36.5) | |
| Clinically diagnosed pulmonary TB | 528 (22.2) | |
| Drug resistant TB | 64 (2.7) | |
| Missing | 11 (0.5) | |
| HIV co-infected (n, %) | Yes | 357 (15) |
| No | 2005 (84.3) | |
| Unknown | 11 (0.5) | |
| Missing | 6 (0.3) | |
| Body weight (in Kg) | Median, IQR | 51 (45, 59) |
| Type of Health facility (n, %) | Hospital | 427 (17.9) |
| Health center | 1952 (82.1) |
5.2. Diabetes risk factors and “symptoms”
Assessment of presence of risk factors showed 22.2% had current or past history of tobacco smoking; 21.2% were aged 45 years and above; and family history of DM was reported in 8.4%. Table 2 summarizes the frequency of reported risk factors. The median cumulative risk score was 2 (interquartile range [IQR] = 0–3).
Table 2.
Frequency of risk factors for diabetes mellitus (n = 2,379).
| Risk factor | Number (%) | |
|---|---|---|
| Family history of DM | no | 2175 (91.4) |
| yes | 199 (8.4) | |
| missing | 5 (0.2) | |
| Age of patient | <35 yr | 1402 (58.9) |
| age 35–44 yr | 464(19.5) | |
| greater or equal to 45 | 504(21.2) | |
| missing | 9 (0.4) | |
| Waist circumference in centimeter (men/women) | <84/77 | 1728 (72.6) |
| 85 to 89.9 / 77 to 83.9 | 532 (22.4) | |
| greater or equal to 90 / 84 | 111 (4.7) | |
| missing | 8 (0.3) | |
| Smoking history | never | 1852 (77.8) |
| ex-smoker | 432 (18.2) | |
| current smoker | 94 (4) | |
| missing | 1 (0) | |
| History of alcohol use | ||
| (glasses per day) | never or < 1 | 2095 (88.1) |
| 1 to 5 | 234 (9.8) | |
| >=5 | 45 (1.9) | |
| Missing | 5 (0.2) |
Fatigue was the most frequently reported symptom at 27.3% followed by unexplained weight loss in 21.9%. Blurred vision was the third most commonly reported problem at 6.9%. Also, “poly symptoms” were reported by a significant minority of cases with excessive urination, thirst and hanger reported by 4%, 5.3% and 6.2% respectively. Neurologic complications such as numbness or tingling sensations were reported by 4.4% of the participants. History of frequent infections and delayed wound healing were the least frequently reported symptoms, 2.2% and 1.4% respectively. Overall, thirty-eight per cent of the participants reported current history of at least one symptom suggestive of possible diabetes mellitus or its complications.
5.3. Diabetes detection rate and associated factors
We detected 197 (8.3%) DM patients, of which 99 (50.3%) were diabetic patients under care and 98 (48.7%) were newly diagnosed.
Several factors were associated with higher diabetes co-morbidity rates as shown in Table 3. After adjusting for other potential risk factors, having a family history of diabetes was the strongest predictor of DM with Odds Ratio (OR) of 5.36 (95% Confidence Interval, [3.67, 7.83]) followed by age ≥ 45 years. Although men had higher rates of TB-DM co-infection than women (9.2% versus 7.5%), the difference was not statistically significant (P > 0.1). Having one or more “symptoms” of diabetes was a significant predictor of DM diagnosis (OR 2.88, 95% CI, 2.06–4.01).
Table 3.
Predictors of DM according to logistic regression analyses.
| Variable | Unadjusted OR (95%) | Adjusted OR (95% CI) |
|---|---|---|
| Sex (Female vs Male) | 1.26 (0.9, 1.69) | 1.23 (0.88, 1.72) |
| Family History of Diabetes (Yes vs No) | 7.3 (5.1, 10.3) | 5.36 (3.67, 7.83) |
| Waist circumference | ||
| <84/77 (Ref) | 1 | |
| 85–89.9/77–83.9 | 1.6 (1.2, 2.3) | 1.23 (0.85, 1.76) |
| ≥90/84 | 2.7 (1.6, 4.6) | 1.81 (0.99, 3.31) |
|
Smoking |
||
| Never (Ref) | 1 | |
| Ex-smoker | 0.99 (0.67, 1.45) | 1.17 (0.55, 2.51) |
| Current smoker | 1.63 (0.87, 3.06) | 0.91 (0.4, 2.06) |
|
Alcohol drinking |
||
| Never or < 1 drink per day (Ref) | 1 | |
| 1–4.9 drinks per day | 1.39 (0.89, 2.18) | – |
| ≥5 drinks per day | 2.12 (0.93, 4.83) | – |
| Age (in years) |
||
| <35 (Ref) | 1 | |
| 35–44 | 2.6 (1.7, 3.97) | 2.36 (1.53, 3.65) |
| ≥45 | 6.19 (4.34, 8.81) | 4.64 (3.18, 6.76) |
| Risk factors combined (≤2 versus > 2) | 4.87 (3.5, 6.8) | Not included |
| Any one or more diabetes “symptoms” (Yes vs No) | 3.5 (2.59, 4.80) | 2.88 (2.06, 4.01) |
5.4. Risk scoring and symptom prediction models
A risk factor score > 2 predicted DM diagnosis with moderate accuracy (AUC = 0.68, 95% CI = 0.6,0.73), and with 73% sensitivity and 63.1% specificity. When combined with symptom score, sensitivity increased to 94.7%, and the AUC was 0.71, but specificity dropped to 29.4%. Symptom alone had lower accuracy (Fig. 2).
Fig. 2.
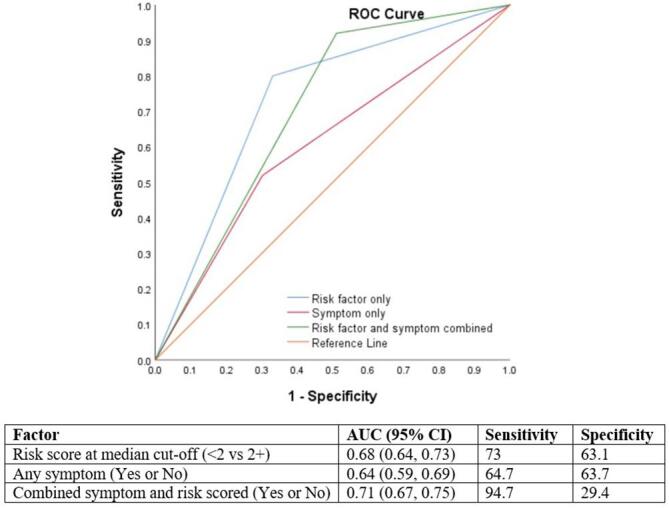
Receiver Operating Characteristic Curve showing the accuracy of different predictor models.
5.5. TB treatment outcome and cause of death
Treatment outcome data was available for 110 TB/DM co-infected patients. Of these, 11 (10%) died and one patient (0.1%) moved to MDR TB care. When this computation is limited to those with known outcome (n = 57), the death rate increases to 19.2%. Table 4 shows treatment outcome with sensitivity analysis on death under three scenarios.
Table 4.
TB treatment outcome among TB/DM patients.
| Outcome (n = 110) | Number (%) | |
|---|---|---|
| Under follow up | 53 (48) | |
| Cured | 24 (22) | |
| Completed | 21 (19) | |
| Died | 11 (10) | |
| Moved to MDR TB | 1 (0.1) | |
| Death rates under three scenarios | ||
| None of the “Under follow up” patients die | 11/110 (10%) | |
| 19.2% of “Under follow up” patients die (10 more patients die) | 21/110 (19.2%) | |
| Half of “Under follow up” patients die | 16/110 (14.5%) | |
| Possible cause of death (n = 11) | ||
| Not documented | 6 (54) | |
| Uncontrolled diabetes | 2 (18) | |
| COVID-19 | 1 (9) | |
| Renal failure | 1 (9) | |
| HIV | 1 (9) | |
6. Discussion
The observed detection rate of DM among patients with active TB was nearly twice the estimated rate in the general population. Combining socio-demographic and clinical risk factors with diabetes symptoms predicted DM diagnosis with a relatively high degree of accuracy. To our knowledge, ours is the first study to demonstrate improved accuracy of combining risk scoring with symptoms to predict DM in patients with active TB. Unfavorable treatment outcomes, particularly death, were high in TB/DM patients.
The findings clearly highlight the importance of routine screening to identify TB/DM co-morbid patients who are at high risk of unfavorable treatment outcomes. Despite many years of advocacy and availability of global guidelines [14] none of the TB clinics visited were doing routine screening for DM, requiring deliberate actions to initiate the practice. However, the clinicians in the TB clinics promptly learned how to do DM screening once training, tools and test kits were made available. Ensuring continuity of this practice will depend on the longer-term availability of resources including adequate funding and technical expertise.
As confirmed by the study results, undiagnosed DM was high among patients with active TB. In Nigeria, for example, 64% of DM patients in those with TB were newly diagnosed compared with 44% newly diagnosed DM in the non-TB population [15]. In a recent systematic review, the pooled prevalence of undiagnosed DM was 4.54% in the general population in Africa [16]. DM diagnosis can easily be missed due to the overlap in symptoms between TB and DM. Fatigue, for example, is a common symptom of both TB and DM which can be overlooked during clinical workup of patients for TB [17]. Raising the awareness of both patients and providers on the importance of these symptoms should be considered as part of the strategy to improve the management of TB/DM co-morbidities.
Combining symptoms with risk factors can serve as a useful tool to guide the early detection of undiagnosed DM. The use of risk scoring systems to predict undiagnosed DM has been studied in non-TB patient populations in the US, Europe, and Asian countries [18], [19], [20], [21]. However, these tools were used for predicting DM in the general population and were not specific for TB patients. Also, the parameters used as predictors varied from study to study. In the Finnish diabetes risk scoring system, for example, investigators used additional parameters such as body mass index, use of antihypertensive drugs, physical activity, daily consumption of fruits, and history of high blood glucose in their prediction model. Their goal was to predict a 10-year probability of developing DM rather than presence of current TB. To our knowledge, the only African study provided some hint about the potential utility of a risk scoring system was the one done by one of us (DJ) [22]. Our study provides further details and more specific accuracy data using larger sample size. We also demonstrated that combining risk scores with symptoms predicted DM with higher degree of overall accuracy, although specificity was minimal. With further validation and refining, the combined tool can be used to prioritize patients for confirmatory diagnostic tests in settings where routine screening of every patient is not feasible. The tool can also be developed into a self-screening tool with digital options.
The high rate of death among TB/DM co-morbid patients was not unexpected but a worrying finding. A review of 44 years’ worth of studies showed DM and other co-morbidities as important risk factors for death among TB patients which our result concurs with [23]. Given Ethiopia’s high treatment success rate of 96% in DS-TB patients, the 10% mortality rate in our study clearly shows the disproportionately high rate of death in this subgroup of patients. Using three different scenarios for the remaining half of patients whose treatment outcome was not known, we estimated that the death rate can be as high as 19.2%. This highlights the need for further disaggregation of data when TB treatment outcomes are reported. Such analyses can lead to targeted actions such as mortality audits and other quality improvement strategies.
The synergy between the Corona Virus Disease 2019 (COVID-19) and DM is an important factor worth mentioning here. The existing evidence clearly suggests that patients with DM and other chronic medical conditions are at increased risk of severe COVID-19 and subsequent death [24]. In our study, at least one of the 11 confirmed deaths was due to COVID-19. Since under-reporting of deaths is a common problem in COVID-19, as is with all disease reporting, [24], [25] the possibility of the additional six patients with undocumented cause of death being due to COVID-19 cannot be ruled out. Although the COVID-19 triggered disruptions in health services were believed to be the main underlying causes for the increase in TB associated deaths in high TB burden countries, [8] it is possible that direct impact of COVID-19 is the main cause of death for patients with co-morbidities. This needs further investigation using larger sample size.
The study has important limitations. Because of lack of laboratory capacity, we were not able to determine the glycemic control level of the patients using the latest methods such as HbA1C. We also did not include history of hypertension in our risk scoring checklist which could have enhanced the capacity of our model. Treatment outcome data were not available for all patients which precluded us from making a more thorough analysis of treatment outcomes and their predictors.
In conclusion, the rate of diabetes co-morbidity was more than double the prevalence estimate in the general population of Ethiopia. Almost a half of this was due to undiagnosed DM, further justifying the need for routine screening for DM in TB patients. Combining risk factors with symptoms accurately predicted a subsequent diagnosis of DM in adult TB patients deemed to be at “high risk” of developing DM. Every patient with TB should be screened for DM by combining risk scores with symptom checklists. The high rate of death in patients with known outcome status is a finding that requires further actions to improve the quality of care for TB/DM co-morbid patients.
We also suggest further studies on this topic. The impact of routine screening on treatment outcomes of both diseases should be further evaluated. The utility of the risk scoring system should be further validated in other settings. Assessing the feasibility of developing and using the risk scoring system as a digital self-screening tool is another area for further work.
7. Ethics Statement
The Ethics review committee of Addis Ababa City Council Health Bureau reviewed and approved the study protocol. Study participants provided informed verbal consent before being enrolled in the study. Patient data was handled confidentially, and anonymized data base was used for analysis. Patients who were diagnosed with diabetes received appropriate care according to the standard of care.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
We would like to thank the health workers in the health facilities, sub city coordinators and disease focal persons at Addis Ababa City Health Bureau, and the non-communicable diseases team at the Federal Ministry of Health for their inputs during the conception and implementation of this project. Dr Challa Negeri, the former technical director at KNCV Ethiopia office made important contributions to the designing and implementation of the project. We also wish to thank Dr Tenagne Kebede and Mr Melkamsew Aschalew for their contribution to data collection and site level support during the implementation of the project. This project was funded by Dr. C. de Langen Stichting voor Mondiale Tuberculosebestrijding (SMT).
References
- 1.Restrepo B.I., Schlesinger L.S. Impact of diabetes on the natural history of tuberculosis. Diabetes Res Clin Pract. 2014;106(2):191–199. doi: 10.1016/j.diabres.2014.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Salindri AD, Kipiani M, Kempker RR, Gandhi NR, Darchia L, Tukvadze N, et al., editors. Diabetes reduces the rate of sputum culture conversion in patients with newly diagnosed multidrug-resistant tuberculosis. Open Forum Infect Dis; 2016: 3(3):ofw126. [DOI] [PMC free article] [PubMed]
- 3.Sun H., Saeedi P., Karuranga S., Pinkepank M., Ogurtsova K., Duncan B.B., et al. IDF diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract. 2021;109119 doi: 10.1016/j.diabres.2021.109119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li M., Chen T., Hua Z., Yan H., Wang D., Li Z., et al. Global, regional, and national prevalence of diabetes mellitus in patients with pulmonary tuberculosis: a systematic review and meta-analysis. Diabetol Metab Syndr. 2021;13(1):127. doi: 10.1186/s13098-021-00743-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Huangfu P., Ugarte-Gil C., Golub J., Pearson F., Critchley J. The effects of diabetes on tuberculosis treatment outcomes: an updated systematic review and meta-analysis. Int J Tub Lung Dis. 2019;23(7):783–796. doi: 10.5588/ijtld.18.0433. [DOI] [PubMed] [Google Scholar]
- 6.Colagiuri S., Davies D. The value of early detection of type 2 diabetes. Curr Opin Endocrinol Diabetes Obes. 2009;16(2):95–99. doi: 10.1097/MED.0b013e328329302f. [DOI] [PubMed] [Google Scholar]
- 7.Bonora E., Dauriz M., Rinaldi E., et al. Assessment of simple strategies for identifying undiagnosed diabetes and prediabetes in the general population. J Endocrinol Invest. 2021;44(1):75–81. doi: 10.1007/s40618-020-01270-0. [DOI] [PubMed] [Google Scholar]
- 8.World Health Organization. WHO global lists of high burden countries for tuberculosis (TB), TB/HIV and multidrug/rifampicin-resistant TB (MDR/RR-TB), 2021–2025: background document. 2021.
- 9.Workneh M.H., Bjune G.A., Yimer S.A. Prevalence and Associated Factors of Diabetes Mellitus among Tuberculosis Patients in South-Eastern Amhara Region, Ethiopia: A Cross Sectional Study. PLoS ONE. 2016;11(1) doi: 10.1371/journal.pone.0147621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Feleke Y., Abdulkadir J., Aderaye G. Prevalence and clinical features of tuberculosis in Ethiopian diabetic patients. East Afr Med J. 1999;76(7):361–364. [PubMed] [Google Scholar]
- 11.Yoon Y.S., Jung J.-W., Jeon E.J., Seo H., Ryu Y.J., Yim J.-J., et al. The effect of diabetes control status on treatment response in pulmonary tuberculosis: a prospective study. Thorax. 2017;72(3):263–270. doi: 10.1136/thoraxjnl-2015-207686. [DOI] [PubMed] [Google Scholar]
- 12.International Diabetes Federation. IDF Clinical Practice Recommendations for managing Type 2 Diabetes in Primary Care. International Diabetes Federation. 2017.
- 13.IBM Corporation. SPSS Statistics for Windows. 25 ed. Armonk, NY2017.
- 14.World Health Organization. Collaborative framework for care and control of tuberculosis and diabetes. World Health Organization; 2011. Report No.: 9241502258. [PubMed]
- 15.Ogbera A.O., Kapur A., Chinenye S., Fasanmade O., Uloko A., Odeyemi K. Undiagnosed diabetes mellitus in tuberculosis: A Lagos report. Indian J Endocrinol Metab. 2014;18(4):475–479. doi: 10.4103/2230-8210.137488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Asmelash D., Asmelash Y. The Burden of Undiagnosed Diabetes Mellitus in Adult African Population: A Systematic Review and Meta-Analysis. J Diabetes Res. 2019;2019:4134937. doi: 10.1155/2019/4134937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kalra S., Sahay R. Diabetes Fatigue Syndrome. Diabetes Ther. 2018;9(4):1421–1429. doi: 10.1007/s13300-018-0453-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kahn H.S., Cheng Y.J., Thompson T.J., et al. Two risk-scoring systems for predicting incident diabetes mellitus in U.S. adults age 45 to 64 years. Ann Intern Med. 2009;150(11):741–751. doi: 10.7326/0003-4819-150-11-200906020-00002. [DOI] [PubMed] [Google Scholar]
- 19.Bang H., Edwards A.M., Bomback A.S., et al. Development and validation of a patient self-assessment score for diabetes risk. Ann Intern Med. 2009;151(11):775–783. doi: 10.7326/0003-4819-151-11-200912010-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Franciosi M., De Berardis G., Rossi M.C.E., Sacco M., Belfiglio M., Pellegrini F., et al. Use of the Diabetes Risk Score for Opportunistic Screening of Undiagnosed Diabetes and Impaired Glucose Tolerance. The IGLOO (Impaired Glucose Tolerance and Long-Term Outcomes Observational) study. Diabetes Care. 2005;28(5):1187–1194. doi: 10.2337/diacare.28.5.1187. [DOI] [PubMed] [Google Scholar]
- 21.Lindström J., Tuomilehto J. The diabetes risk score: a practical tool to predict type 2 diabetes risk. Diabetes Care. 2003;26(3):725–731. doi: 10.2337/diacare.26.3.725. [DOI] [PubMed] [Google Scholar]
- 22.Jerene D., Hiruy N., Jemal I., Gebrekiros W., Anteneh T., Habte D., et al. The yield and feasibility of integrated screening for TB, diabetes and HIV in four public hospitals in Ethiopia. Int Health. 2017;9(2):100–104. doi: 10.1093/inthealth/ihx002. [DOI] [PubMed] [Google Scholar]
- 23.Waitt C.J., Squire S.B. A systematic review of risk factors for death in adults during and after tuberculosis treatment. Int J Tub Lung Dis. 2011;15(7):871–885. doi: 10.5588/ijtld.10.0352. [DOI] [PubMed] [Google Scholar]
- 24.Dessie Z.G., Zewotir T. Mortality-related risk factors of COVID-19: a systematic review and meta-analysis of 42 studies and 423,117 patients. BMC Infect Dis. 2021;21(1):855. doi: 10.1186/s12879-021-06536-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Whittaker C., Walker P.G.T., Alhaffar M., et al. Under-reporting of deaths limits our understanding of true burden of covid-19. BMJ. 2021;375 doi: 10.1136/bmj.n2239. [DOI] [PubMed] [Google Scholar]