Abstract
Purpose
Tumor necrosis factor-like ligand 1A (TL1A), especially its secreted form, has been shown to contribute to eosinophilic inflammation and mucus production, cardinal features of asthma, through its receptor, death receptor 3 (DR3). However, the role of the TL1A-DR3 axis in asthma, especially in terms of airway remodeling, has not yet been fully understood.
Methods
The present study investigated the expression and secretion of TL1A in the lung and human bronchial epithelial cells. DR3 small interfering RNA (siRNA), TL1A siRNA, and truncated plasmids were used respectively to identify the function of the TL1A-DR3 axis in vitro. To further validate the roles of the TL1A-DR3 axis in asthma, we collected airway biopsies and sputa from asthmatic patients and constructed a mouse model following rTL1A administration, DR3 knockdown, and TL1A knockout, the asthma-related inflammatory response and the pathological changes in airways were analyzed using various experimental methods. Associated signaling pathways downstream of TL1A knockout in the mouse model were analyzed using RNA sequencing.
Results
TL1A, especially its non-secreted form (nsTL1A) was involved in the remodeling process in asthmatics’ airways. Knockdown of TL1A or its receptor DR3 decreased the expression of fibrosis-associated protein in BEAS-2B cells. Reversely, overexpression of nsTL1A in airway epithelial cells facilitated the transforming growth factor-β-induced remodeling progress. In the asthma mouse model, activating the TL1A-DR3 axis contributes to airway inflammation, remodeling, and tissue destruction. Reciprocally, DR3 knockdown or TL1A knockout partly reverses airway remodeling in the asthma model induced by ovalbumin.
Conclusions
Our results confirm differential TL1A expression (including its secreted and non-secreted form) in asthma, which modulates remodeling. The shared mechanism of action by which nsTL1A and secreted TL1A exert their effects on asthma development might be mediated via the nuclear factor-κB pathway. The TL1A-DR3 axis presents a promising therapeutic target in asthma.
Keywords: Asthma, airway remodeling; airway inflammation; tumor necrosis factor; TL1A; therapeutic target
INTRODUCTION
Asthma is a lifelong respiratory disease associated with reversible airway obstruction of varying degrees, airway inflammation, hyper-responsiveness (AHR), and remodeling.1 Along with clinical guidelines, the treatment of asthma has become more standardized, and patients’ compliance continues to improve.2 However, conventional therapies, including bronchodilators and inhaled glucocorticoids, sometimes have limited therapeutic efficacy, especially in severe cases, emphasizing the need for new therapeutic approaches and strategies.3
Currently, airway remodeling, as one of the main pathologies in bronchial asthma, has been confirmed to affect the outcome in asthma directly and has become a significant concern in therapy.4 Research results have increasingly suggested that structural changes linked to airway remodeling in patients with asthma lead to bronchial hyper-responsiveness and irreversible airflow obstruction.5 Even worse, features of airway remodeling increase with the duration of illness for patients with asthma.6 In the era of personalized treatment, one of the directions for new asthma treatment development has focused on finding reliable airway remodeling biomarkers to assist in asthma phenotype classification and selection of treatment options.7
Tumor necrosis factor (TNF) ligand-related molecule 1A (TL1A), also known as TNF superfamily member 15, is encoded by the TNFSF15 gene. It is a newly discovered subgroup of the TNF superfamily at the beginning of the 21st century.8,9,10 There are 2 forms of TL1A in humans, a non-secreted form (nsTL1A) and a secreted form (sTL1A), that has been found in plasma and bronchoalveolar lavage fluid (BALF).11 The primary receptor involved in the effects of TL1A is the death receptor 3 (DR3), but others, such as decoy receptor 3 (DcR3), also play a role.12 Upon binding to DR3, sTL1A could directly promote inflammatory responses by activating nuclear factor (NF)-κB signaling.13 It has also been proposed that sTL1A can specifically activate T cells and promote the secretion of inflammatory cytokines such as interferon-γ and granulocyte-macrophage colony-stimulating factor.8,14 Another member of the same family, LIGHT encoded by the TNFSF14 gene, was suggested to be essential for airway remodeling in asthma.15 Blocking LIGHT in mice can markedly improve pathologic airway fibrosis and relieve airway smooth muscle remodeling.15 At present, some studies have proven that the TL1A-DR3 axis is closely related to the progression of asthma and promotes eosinophilic inflammation and mucus production.16,17,18 However, the functions of TL1A in asthmatic airway remodeling are still not completely elucidated, particularly the nsTL1A. Identifying a role of the TL1A-DR3 axis in airway inflammatory responses and remodeling may open up a new area of research (Supplementary Fig. S1).
MATERIALS AND METHODS
Human tissue atlas
To identify the protein expression levels and organ localization of TL1A, we used the Human Tissue Atlas (http://www.proteinatlas.org/).
Lentivirus constructs
Lentiviral constructs were made in LV2N for shRNA knockdown using standard molecular biology procedures and verified by sequencing. Oligonucleotides encoding mouse DR3 shRNA targeting sequences were 5′-GCCCAAAGGGACACTACATGA-3′, 5′-GCTGCCAAACAGAACTCTTGG-3′, 5′-GGCTACAACACCAGTCCATGA-3′, and 5′-TTCTCCGAACGTGTCACGT-3′. The efficiency of the knockdown was established by measuring the protein levels of DR3 in the infected mouse lungs using immunoblots.
Plasmid construction
The truncated plasmids TL1A-M (Δ66-94) were constructed by Fenghui Shengwu Biotech Co., Ltd (Wuhan, China) using the primers (forward: 5′-ccagggagaggccccaagggcacacc-3′; reverse: 5′-ggtgtgcccttggggcctctccctgg-3′) for aa66–94 deletion with reference to the previously reported method.19 Correct sequence and orientation of the DNA inserts in the plasmid were confirmed by sequencing.
Patients
The diagnostic criteria for asthma were based on the Global Initiative for Asthma (GINA) guidelines (updated in 2021). Sputum induction was performed as previously described.20 All patients undergoing bronchoscopic biopsy did not have systemic inflammatory diseases or other respiratory diseases. The experiments were approved by local Ethics Committee (ethics review number: 2021-S923), and written informed consent was obtained from all patients. Control subjects were patients with benign pulmonary nodules with normal pulmonary function and no respiratory symptoms. The basic clinical information of all subjects is shown in Supplementary Tables S1 and S2.
Mice
Age and weight-matched (8-week-old, 20 ± 2 g) female C57BL mice (6 mice in each group) were purchased from PengYue Laboratory Animal Breeding Co., Ltd. (Jinan, China). TNFSF15-deficient mice (T011739) on C57BL/6 background were generated by the Model Animal Research Center of Nanjing University (Nanjing, China) using CRISPR/Cas9 technology. Mice were maintained in a standard laboratory animal facility for 1 week before the experiments. In the TL1A group, mice were challenged with 5 µg of recombinant TL1A (753006; BioLegend, San Diego, CA, USA) in 50 µL of phosphate buffered saline (PBS) intratracheally for 3 consecutive days. Control groups received sterile PBS at all time points. Bronchoalveolar lavage was performed as previously described.21
Animal model construction
Mice were sensitized to ovalbumin (OVA) by intraperitoneal (i.p.) injection of 50 µg of OVA and 4 mg of alum, Al(OH)3, in 0.5 mL of PBS, then boosted 2 weeks later with a second injection. OVA challenge was performed in the OVA, OVA-TL1A, OVA+shNC (OVA-induced + shDR3 lentivirus), and OVA+shDR3 group (OVA-induced + shDR3 lentivirus) by inhalation of 3% OVA aerosol on days 24, 25, and 26. For the lentivirus group, 48 hours prior to airway challenge, the mice were injected through the caudal vein with the lentivirus (1 × 108 TU/mouse) containing a control shRNA or the DR3 shRNA sequence. For OVA+TL1A experiments, rTL1A was instilled intratracheally after each nebulization. The airway hyper-reactivity experiment was used to evaluate whether the models were successful or failed and whether the assessment methods were performed as previously described by our research group.22 The schematic representation of the experimental methods used is presented in Supplementary Fig. S2.
Cell culture and treatment
BEAS-2B human bronchial epithelial cells were obtained from Shanghai Fuheng Biotechnology Co., Ltd. (Xian, China) and incubated in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal bovine serum (Gibco; ThermoFisher Scientific, Waltham, MS, USA). To construct the cellular model, BEAS-2B cells were stimulated with human interleukin (IL)-13 protein or human transforming growth factor (TGF)-β1 protein (Abbkine Scientific Co., Ltd., Wuhan, China) at various concentrations for the indicated time. BEAS-2B cell transfections were performed using the INTERFERin® reagent (Polyplus-Transfection SA, Paris, France) according to the manufacturer’s instructions.
Bioinformatics analysis
The data expression profile sets used were derived from the GEO database (https://www.ncbi.nlm.nih.gov/geo/), and one mRNA dataset of research on the human bronchial epithelial cell, GSE40374, was selected. Differential mRNAs were screened in an immortalized human bronchial epithelial cell line, M-BE cells, treated with and without human recombinant TGF-β1 (5 ng/mL) for 6 days. The Limma package (version: 3.40.2) of R software was used to study the differential expression of mRNAs. The adjusted P value was analyzed to correct for false-positive results in GEO datasets. Adjusted P < 0.01 and Log2 fold-change (FC) > 1.5 or Log2FC < −1.5 were defined as the thresholds for screening differential expression of mRNAs. The box plot was implemented by the R software package ggplot2. To confirm the expression mode of TL1A in asthmatic patients, we screened RNA profiling subjecting of patients and healthy controls from public databases (GSE18965).
RNA isolation and reverse transcription quantitative polymerase chain reaction (RT-qPCR) analysis
Total RNA was extracted from BEAS-2B cells using the RNA Fast 200 RNA Extraction kit (Fastagen Biotech, Shanghai, China) according to the manufacturer’s instructions. After quantification using spectrophotometry, 1 µg of RNA was used to synthesize cDNA by using an Evo M-MLV Mix Kit with gDNA Clean for qPCR (Accurate Biology Co., Ltd., Hunan, China). RT-qPCR was performed using the SYBR Green PCR Master Mix (Accurate Biotechnology Co., Ltd., Wuhan, China). The primers used are described in Supplementary Table S3. All samples were run in triplicate, and the mean values were used for quantification.
RNA-sequencing analysis
After modeling, the lung tissues of each TNFSF15 knockout and wild-type (WT) mouse were separated and stored at −80°C for RNA sequencing. RNA-sequencing analysis was carried out by Sinotech Genomics Co., Ltd. (Shanghai, China).
ELISA assays
IL-4, IL-5, IL-13, and TL1A levels in plasma or cell culture supernatant were measured using commercial ELISA kits. The ELISA kits used were as follows: Mouse IL-4 ELISA Kit (70-EK204/2-96; MultiSciences, Hangzhou, China), Mouse IL-5 ELISA Kit (70-EK105-96; MultiSciences), Mouse IL-13 ELISA Kit (70-EK204/2-96, 70-EK213/2-96; MultiSciences), Mouse IL-4 ELISA Kit, Mouse TNFSF15 ELISA Kit (ZC-55212; ZCI BIO, Shanghai, China), and Human TNFSF15 ELISA Kit (ZC-35762; ZCI BIO).
Immunoblotting
Total cellular proteins were extracted using RIPA and then subjected to western blot experiments for quantification. To carry out western blot experiments, total proteins were segregated using sodium-dodecyl sulfate polyacrylamide gel electrophoresis and transferred onto polyvinylidene difluoride membranes. After brief washing, Tris-buffered saline with 1% Tween 20 (TBST) containing 5% bovine serum albumin (BSA) was used for membrane blocking for 1 hour. Then, the membranes were incubated overnight at 4°C with the primary antibodies including anti-TL1A (1:1,000, ab85566; Abcam, Santa Cruz, CA, USA), anti-DR3/LARD antibody (1:1,000, ab189093; Abcam), Anti-COL1A1 antibody (1:1,000, BA0325; Boster, Wuhan, China), anti-fibronectin (1:1,000, JF0582; HUABIO, Hangzhou, China), anti-alpha smooth muscle actin (1:1,000, SY02-64; HUABIO), anti-E-cadherin (1:1,000, PB9561; Boster), anti-E-cadherin (1:1,000, ER0503; HUABIO), and anti-GAPDH (1:1,000, BA2913; Boster). The following day, the membranes were removed with tweezers and then washed in TBST 3 times and incubated with horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG (H+L) (1:3,000, GB23303; Servicebio, Wuhan, China) at room temperature for 1 hour. After washing with TBST again, the membranes were subjected to chemiluminescence by using enhanced chemiluminescence substrate.
Histological analysis
Immunohistochemical staining was performed as previously described. The human airway epithelial biopsy specimens and mouse lung tissues were fixed in 10% neutral formalin, paraffin-embedded, and cut into 5-µm sections. Sections were dewaxed, and rehydrated and used for hematoxylin and eosin (H&E), trichrome, and fluorescent immunostaining. Airway inflammation and collagen volume fraction were scored semi-quantitatively on the H&E staining or Masson-stained slides according to previously described methods.23,24
Cell immunofluorescence
BEAS-2B cells were plated on cell climbing slices in a 24-well plate. Following the designated treatments, cell climbing slices were washed with PBS, fixed with 4% paraformaldehyde, and then incubated with 0.5% triton X-100 for permeabilization. Then, the cells were blocked with 1% BSA in PBS for 1 hour at room temperature. The primary anti-TNFSF15 antibody (1:200, DF3053; Affinity, Jiangsu, China), anti-fibronectin antibody (1:200, JF0582; HUABIO), and anti-COL1A1 antibody (1:200, BA0325; Boster) were used to incubate the cell climbing slices overnight at 4°C. The primary antibody was then removed, and fresh PBS was added. After washing with PBS 3 times, climbing slices were incubated with the secondary antibody goat anti-rabbit IgG (1:500; Abbkine Scientific Co., Ltd.) and protected from light for 1 hour. The nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI) (1:500) for 5 minutes. After the final washing in PBS (3 × 10 minutes), the images were captured under an upright fluorescence microscope (Leica, Berlin, Germany).
Statistical analysis
All results are expressed as mean ± standard deviation; differences between 2 groups were analyzed using Student’s t test. A 2-tailed P value of < 0.05 was considered statistically significant.
RESULTS
TL1A is highly expressed in airway and lung tissues
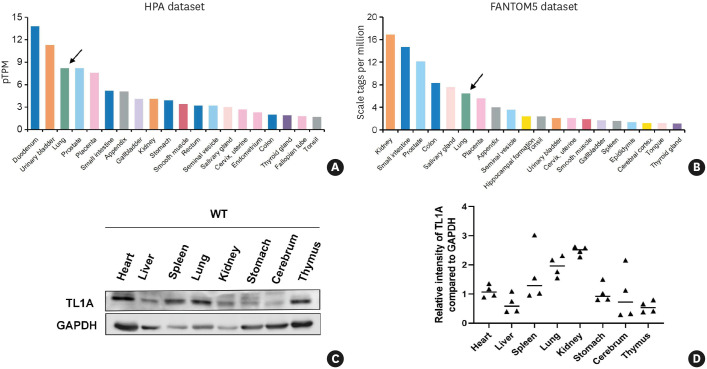
First, TL1A expression across the different tissues was examined using the Human Protein Atlas database. The results demonstrated that the protein expression of TL1A was limited in organs, but a high level of TL1A expression was observed in the airway and lung tissues (Fig. 1A and B). We also examined the expression of TL1A in various mouse tissues and organs by western blot analysis and confirmed high TL1A expression levels in the airway and lung tissues (Fig. 1C and D).
Fig. 1. TL1A is involved in various physiological and pathological processes in the respiratory system.
(A) TL1A protein expression levels of human tissues from the Human Protein Atlas. (B) TL1A expression levels of human tissues from FANTOM5 project database. (C and D) The TL1A expression level was measured by western blot analysis in tissues extracted from different mouse organs.
TL1A, tumor necrosis factor ligand-related molecule 1A; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; WT, wild-type.
TGF-β1 stimulation increases the expression of TL1A without affecting its secretion in bronchial epithelial cells
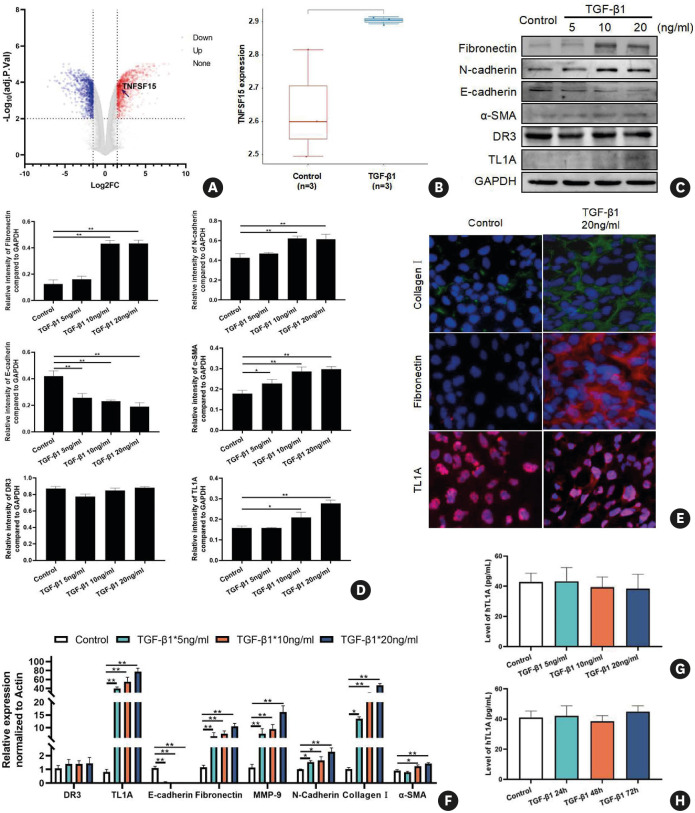
We next investigated whether the potential mechanism of increased airway remodeling is related to TL1A. In order to test this hypothesis, a microarray dataset, GSE40374, was retrieved from the GEO database, and bioinformatics analyses were conducted to determine the differences in gene expression in an immortalized human bronchial epithelial cell line with or without TGF-β1 stimulation. After screening (|Log2FC| ≥ 1.5 and adj-P value < 0.01), we found that 2,054 genes were differentially expressed, including 1,184 up-regulated and 867 down-regulated genes (Fig. 2A). The volcano plot and box plot showed that TL1A is one of the significantly up-regulated genes between control and TGF-β1-stimulated groups (Fig. 2A and B).
Fig. 2. TL1A levels are increased after TGF-β1 treatment in bronchial epithelial cells.
(A) Volcano plots were constructed using fold-change values and adjusted P. The red point in the plot represents overexpressed mRNAs and the blue point indicates down-regulated mRNAs with statistical significance. (B) The expression distribution of TNFSF15, where different colors represent different groups and the vertical axis represents the gene expression distribution. (C and D) Protein expression levels of E-cadherin, N-cadherin, fibronectin, DR3, TL1A, and α-SMA after treatment with different concentrations of TGF-β1 for 48 hours. (E) Immunofluorescence for collagen I, fibronectin, and TL1A after treatment with 20 ng/mL TGF-β1 for 48 hours. (F) The RNA levels of E-cadherin, collagen I, N-cadherin, fibronectin, DR3, TL1A, MMP-9. and α-SMA after treatment with different concentrations of TGF-β1 for 48 hours. (G-H) The levels of secreted TL1A remained unchanged in BEAS-2B cells treated with TGF-β1 at various concentrations and during various time courses. Data are expressed as means ± standard deviation.
TGF, transforming growth factor; TL1A, tumor necrosis factor ligand-related molecule 1A; DR3, death receptor 3; MMP-9, matrix metallopeptidase 9; α-SMA, α-smooth muscle actin; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
*P < 0.05, **P < 0.01 versus the control group.
Bioinformatics analysis highlighted the potential of TL1A in fibrosis, and biochemical experiments further validated these observations. Based on data in the literature and empirical experiments, we selected 48 hours as the optimal TGF-β1 stimulation duration to induce remodeling and epithelial-mesenchymal transition (EMT).22,25 After BEAS-2B cells were incubated with TGF-β1 at different concentrations (5, 10, and 20 ng/mL) for 48 h, the expression level of E-cadherin was reduced. In contrast, the levels of fibronectin, N-cadherin, and α-smooth muscle actin (α-SMA) significantly increased after treatment with 10 and 20 ng/mL TGF-β1 (Fig. 2C-F). Consistent with data from microarray analysis, TL1A expression significantly increased in protein and RNA at 48 hours after treatment with TGF-β1, while the level of DR3 remains unchanged.
Interestingly, the increased level of TL1A induced by TGF-β1 was not accompanied by a parallel increase in its secretion level in the supernatant (Fig. 2G). The same response was also observed in supernatants of BEAS-2B cells stimulated with 20 ng/mL TGF-β1 for prolonged time periods (Fig. 2H). The results presented above suggest that TL1A secretion by bronchial epithelial cells may be required for additional signaling mechanisms.
The activation of TL1A-DR3 axis is crucial for remodeling and EMT process in airway epithelial cells
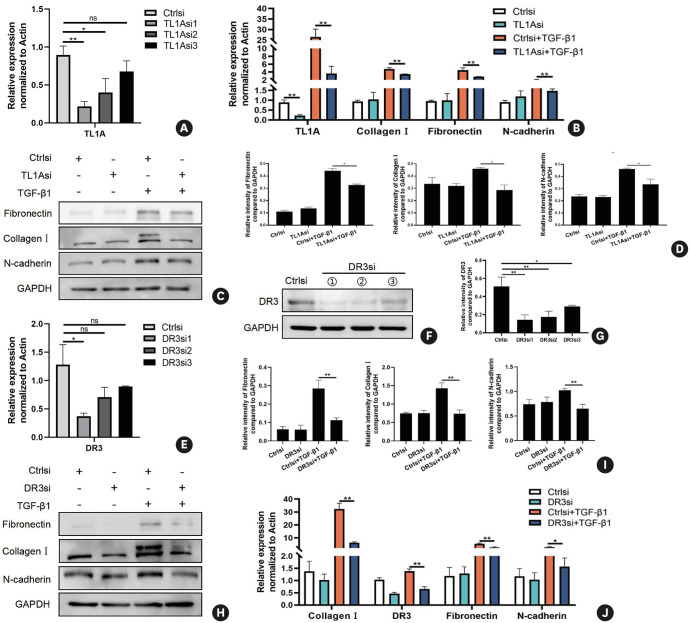
To determine whether TL1A-DR3 axis plays a role in the EMT and fibrosis process in normal bronchial epithelial cells, we first transfected BEAS-2B cells with small interfering RNA (siRNA) targeting TL1A. Given that TL1A protein expression was low in resting cells, RT-qPCR was performed to monitor the transfection efficiency (Fig. 3A). Among the 3 available TL1A siRNAs, siRNA1 exhibited the best transfection efficiency (Fig. 3A). As described above, TL1A was highly expressed in cells after TGF-β1 stimulation (Fig. 2). Therefore, we performed the knockdown experiments with TGF-β1 treatment and obtained the same result (Supplementary Fig. S2). Finally, the TL1Asi1 was selected and used in the following experiments. The following experiments found that the decreased TL1A expression had no impact on N-cadherin, fibronectin, or collagen I expression levels without TGF-β1 treatment (Fig. 3B-D). After TGF-β1 treatment, cells with TL1A down-regulation exhibited lower N-cadherin, fibronectin, and collagen I levels than those in NC-transfected cells (Fig. 3B-D; P < 0.05), in both protein and RNA. In order to further confirm the result, we observed above, siRNA sets of DR3, a primary TL1A receptor, were constructed. From both transcriptome and protein analyses, we observed evident down-regulation of DR3 by DR3 siRNA1 at the RNA and protein levels (Fig. 3E-G; P < 0.05). Combined with the above results, the TL1A-DR3 axis appeared to be associated with EMT and remodeling in airways (Fig. 3H-J; P < 0.05).
Fig. 3. Effects of TL1A/DR3 axis inactivation on EMT and remodeling indicators in bronchial epithelial cells.
(A) mRNA expression levels of TL1A after small interference RNA transfection. (B-D) Protein and mRNA expressions of fibronectin, collagen I, N-cadherin and TL1A after different treatments. (E-G) mRNA expression levels of DR3 after small interference RNA transfection. (H-J) Protein and mRNA expression of fibronectin, collagen I, N-cadherin, and DR3 after different treatments. Data are expressed as mean ± standard deviation.
TL1A, tumor necrosis factor ligand-related molecule 1A; DR3, death receptor 3; EMT, epithelial-mesenchymal transition; ns, not significant; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
*P < 0.05, **P < 0.01 versus the corresponding group.
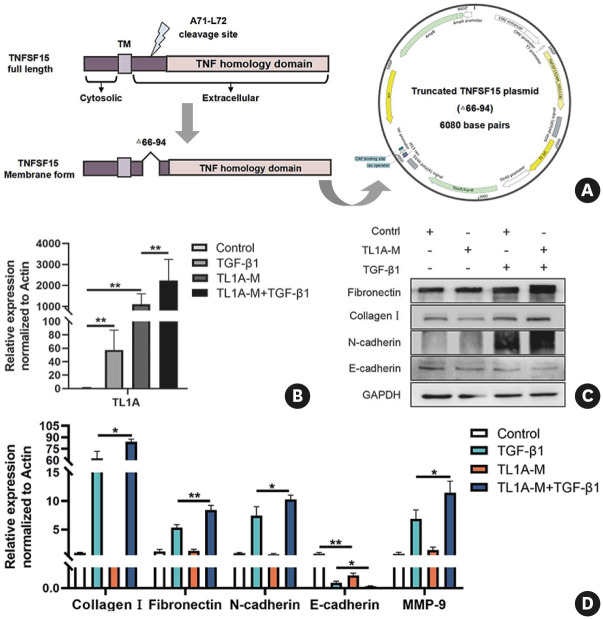
Existing studies have reported that the addition of exogenous recombinant TL1A protein leads directly or indirectly to the remodeling in airway epithelial cells. However, whether or not the nsTL1A had the same or opposite effect is not known. To address this, we constructed the truncated TL1A plasmid (TL1A-M, Δ66-94) for deletion of the cleavage sites without affecting its function of binding receptors as previously reported (Fig. 4A). As anticipated, in airway epithelial cells (BEAS-2B), the nsTL1A expression was up-regulated after transfecting TL1A-M and more pronounced in the TGF-β1 stimulated group (Fig. 4B). Intriguingly, we found that over-expressed nsTL1A alone could promote the remodeling-related alterations elicited by TGF-β1 in airway epithelial cells (Fig. 4C and D). These findings collectively emphasize that TL1A (its secreted and membrane forms) play an important role in airway epithelium during the early phase of the fibrosis process.
Fig. 4. Overexpression nsTL1A affects remodeling and EMT induced by TGF-β1.
(A) The construction of truncated TL1A plasmid for deletion aa66-94. (B) mRNA expression of TL1A after truncated TL1A plasmid transfection and TGF-β treatment. (B-D) Protein and mRNA expressions of fibronectin, collagen I, N-cadherin E-cadherin and MMP-9 after different treatments. Data are expressed as mean ± standard deviation.
nsTL1A, non-secreted tumor necrosis factor ligand-related molecule 1A; EMT, epithelial-mesenchymal transition; TGF, transforming growth factor; MMP-9, matrix metallopeptidase 9; TNF, tumor necrosis factor; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
*P < 0.05, **P < 0.01 versus the corresponding group.
TL1A is highly expressed in airway epithelium in asthma, with concomitant airway remodeling
Extensive studies have established that TL1A in innate lymphoid type 2 cells (ILC2) has long been associated with bronchial asthma and eosinophilic airway inflammation. However, the role of TL1A in the asthmatic airway epithelium has not been previously known. In light of the above consideration, we guess TL1A, especially its insoluble form, is involved in the pathogenesis of asthma in the airways. First, we constructed an asthma model induced by IL-13 and observed the change in TL1A levels accompanied by periostin, an indicator of eosinophilic inflammation, which lends credence to the validity of the association between asthma and TL1A in airway epithelium (Supplementary Fig. S4).
To further determine whether TL1A in airway epithelium correlated with asthma, we next analyzed the differential expressed genes and the RNA-seq value of TNFSF15 in GSE18965 dataset comprising 9 atopic asthmatics patients and 7 healthy subjects (Fig. 5A). As shown in the half-violin plot (Fig. 5B), TNFSF15 expression was more significantly up-regulated in the asthma group than the control group in airway epithelium. Next, airway biopsy tissues from patients with asthma and healthy controls were obtained by fiberoptic bronchoscopy. Masson staining and immunohistochemical analyses were performed to detect the TL1A level and visualize the deposition of collagen in the airways (Fig. 5). The results showed that levels of TL1A were significantly increased in the bronchial biopsy specimens from patients with asthma compared with healthy controls (Fig. 5C and D). In addition, we also observed increased collagen fibers in the thickened airway walls in the samples from asthmatics (Fig. 5C and D). To further investigate the correlation between TL1A and asthma, we next constructed murine asthma models using OVA (Supplementary Fig. S2). Fig. 5E-H show that the levels of TL1A were increased in lung tissue in OVA-challenged mice compared to control mice. Immunofluorescence analysis also revealed that elevated TL1A was localized near the airway epithelium in the OVA-induced mouse model (Fig. 5G and H). These results highlighted that nsTL1A might be a potential link to airway remodeling in asthma.
Fig. 5. Levels of TL1A in the asthma patients and murine model.
(A and B) TL1A expression profile graph in GSE18965 and differential expression analysis between asthmatics patients and healthy controls. (C) Expression of TL1A by immunohistochemical collagen levels by Masson staining in controls and subjects with asthma. Original magnification ×400. (D) Quantification of TL1A staining and collagen volume fraction between 2 groups. (E and F) Protein and mRNA expression of TL1A in lung tissue of mice 3 days after TL1A or PBS treatment. (G and H) Immunofluorescence detection of TL1A protein. (I) Expression of soluble TL1A in induced sputum detected by ELISA. (H) Expression of soluble TL1A in BALF detected by ELISA in each group (n = 10). Data are expressed as mean ± standard deviation.
TL1A, tumor necrosis factor ligand-related molecule 1A; PBS, phosphate buffered saline; BALF, bronchoalveolar lavage fluid.
*P < 0.05, **P < 0.01, ***P < 0.001 versus the corresponding group.
Elevated TL1A Level in sputum supernatant and BALF in asthmatic subjects
As stated above, nsTL1A was up-regulated in the airway epithelium of both animal models and human patients; however, whether the secreted form of TL1A is changed in the presence of the non-secreted form is not clear. To investigate this, we collected induced sputum samples from 13 asthmatics and 10 normal donors (Supplementary Table S2). The ELISA results indicated that the expression of TL1A was higher in the asthma group than in the control group (Fig. 5I). Similar results were obtained for BALF TL1A measurements from the OVA-induced animal model (Fig. 5J). However, in contrast, the level of secreted TL1A remains constant in the serum samples of the same batch of mice (Supplementary Fig. S5). We speculated that the inconsistencies between the BALF and serum could have been related to the local effect in the lungs.
Administration of TL1A directly promotes airway remodeling in mice
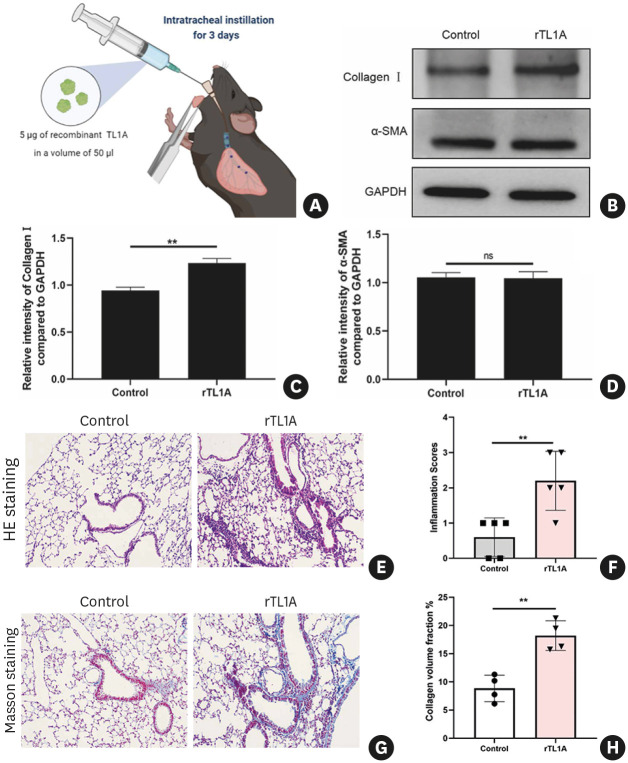
To assess if TL1A could directly contribute to airway remodeling in vivo, we first administered recombinant TL1A (rTL1A) (Fig. 6A). We selected a dose of 5 µg according to the previous study. After intratracheal delivery of 5 µg rTL1A for 3 days, inflammatory response and remodeling features became apparent (Fig. 6). Immunoblot analysis revealed a significant collagen increase in the lungs in the experimental group compared with normal histology in the control group (Fig. 6B-D). H&E and Masson & trichrome staining revealed significant infiltration of airway inflammatory cells, and collagen accumulation was observed around the bronchioles (Fig. 6E and G). Evaluation of airway inflammation with an inflammation score also confirmed that treating the airway with rTL1A promoted inflammatory cell infiltration (P < 0.01) (Fig. 6F). Surrounding the airways, the collagen volume fraction was also significantly increased in the TL1A group (P < 0.01) (Fig. 6H).
Fig. 6. Intratracheal administration of TL1A in WT mice.
(A) Schematic diagram of TL1A intratracheal instillation in mice. Part of the figure was generated with BioRender. (B-D) Protein expression of fibrosis indicator (collagen I and α-SMA) after treatment and subsequent quantitative analysis. (E) Representative images of murine lung tissue stained with H&E. (F) Inflammation score. (G) Representative images of Masson-stained lung tissues of mice. (H) Quantification of collagen volume fraction. Data are represented as mean ± standard deviation.
TL1A, tumor necrosis factor ligand-related molecule 1A; WT, wild-type; α-SMA, α-smooth muscle actin; ns, not significant.
**P < 0.01 versus the control group.
OVA-challenged mice exhibit increased airway inflammation, remodeling, and AHR after TL1A administration
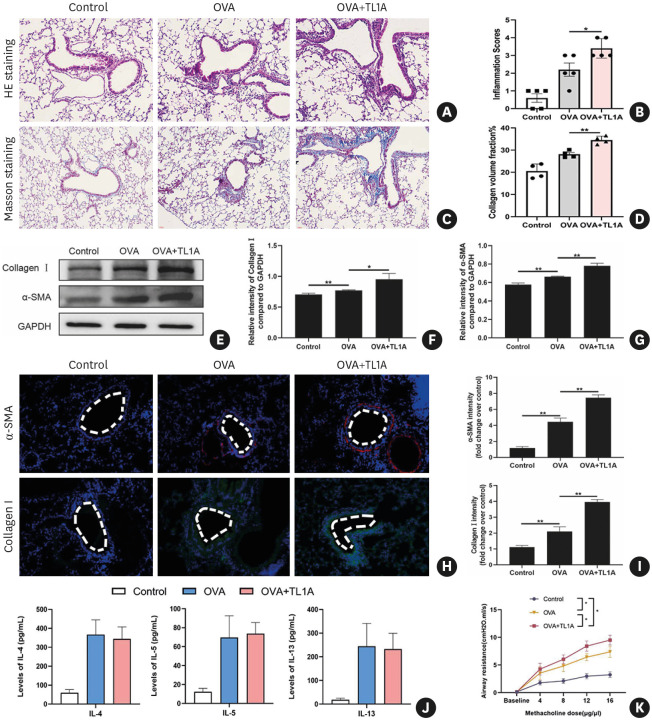
Next, to delineate the function of TL1A in asthma, we instilled TL1A intratracheally in the OVA-challenged mice asthma model. The OVA group exhibited evident accumulation of airway inflammatory cells as determined by H&E staining (Fig. 7A and B). Some peribronchial collagen deposition was also observed in asthmatic mice by Masson staining (Fig. 7C and D). Moreover, the administration of TL1A further exacerbated the histopathological changes induced by OVA (Fig. 7A-D). These histological data support the conclusion that TL1A plays a critical role in acute airway inflammation and asthmatic airway remodeling.
Fig. 7. Intratracheal administration of TL1A in the OVA-challenged mice.
(A-D) H&E and Masson staining were used to observe histopathological changes after TL1A treatment in the mouse model of asthma. Histopathological lesions were analyzed quantitatively. (E-G) Protein expression of fibrosis indicator (collagen I and α-SMA) after treatment and subsequent quantitative analysis. (H-I) Immunofluorescence detection of fibrosis “indicator” proteins and their quantitative analyses. (J) Expression of IL-4, IL-5, and IL-13 in the blood detected by ELISA. (K) AHR was measured by airway reactivity to increased doses of inhaled methacholine treatment. Administration of TL1A can increase methacholine-induced AHR.
TL1A, tumor necrosis factor ligand-related molecule 1A; OVA, ovalbumin; H&E, hematoxylin and eosin; α-SMA, α-smooth muscle actin; IL, interleukin; AHR, airway hyper-responsiveness; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
*P < 0.05, **P < 0.01.
In parallel with the analysis of histopathological changes, lung tissues of the 3 groups were homogenized, and proteins were extracted to evaluate the remodeling level by western blot analysis. Western blot analysis showed that collagen I and α-SMA were more significantly increased in lung tissue in the OVA+TL1A group (Fig. 7E-G). Immunofluorescent staining results were consistent with the immunoblotting ones (Fig. 7H and I). The level of Th2 cytokines (IL-4, IL-5, and IL-13) in plasma was evaluated by ELISA. In the OVA+TL1A group, administration of rTL1A did not impact the levels of TH2-related cytokines, similar to the OVA group (Fig. 7J). Additionally, we found that the administration of TL1A can augment OVA-induced AHR (Fig. 7J).
DR3 knockdown alleviates airway inflammation and remodeling
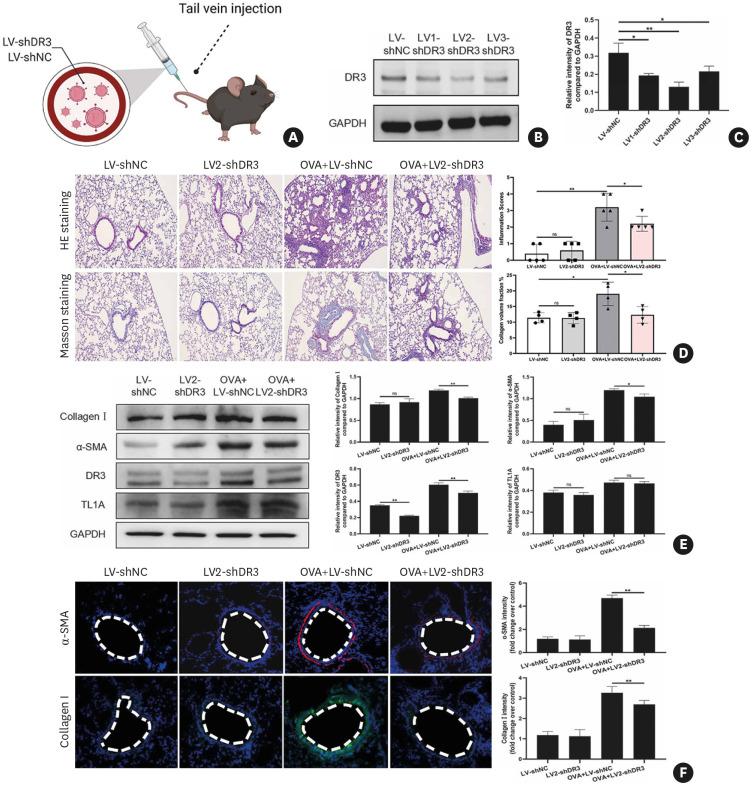
By analyzing human airway biopsy samples from patients with asthma and the animal model, we found the elevated TL1A in asthma, which may be involved in asthma development. We also found that the administration of TL1A could exaggerate airway inflammation and remodeling in vivo. To provide further evidence for the involvement of TL1A in asthma, we constructed 3 lentiviral shRNA vectors targeting DR3, the primary receptor of TL1A, to suppress the function of TL1A. Lentivirus was injected to C57BL/6 mice at 12 weeks of age via the caudal vein with either the LV-NC, LV1-shDR3, LV2-shDR3, or LV3-shDR3 (Fig. 8A). Mice were sacrificed 7 days after lentivirus administration, and lung tissues were collected for analysis. Immunoblot analysis indicated that manipulating of DR3 expression by lentivirus was successful and efficient (Fig. 8B and C). The LV2-shDR3 showed the best knockdown efficiency (Fig. 8B and C) without affecting the production of secreted TL1A (Supplementary Fig. S6), and it was used for the following experiments.
Fig. 8. DR3 in the OVA-challenged mice is knocked down with lentiviral-based shRNA.
(A) The diagram for lentivirus intratracheal injection. Part of the figure was generated with BioRender. (B and C) The effects of DR3 knockdown with lentivirus shRNAs were evaluated via immunoblots. (D) H&E and Masson staining were used following quantitative analysis to evaluate the histopathological changes after lentivirus treatment in the mouse model of asthma. (E) Protein expression of fibrosis indicator (collagen I and α-SMA) after treatment and subsequent quantitative analysis. (F) Immunofluorescence detection of fibrosis “indicator” proteins and their quantitative analysis.
DR3, death receptor 3; OVA, ovalbumin; H&E, hematoxylin and eosin; α-SMA, α-smooth muscle actin; ns, not significant.
*P < 0.05, **P < 0.01.
As shown in Fig. 8D, OVA-challenged +LV2-shDR3 mice had mild inflammatory cell infiltration around the small airways. Concomitantly, the degree of airway remodeling was more significantly attenuated compared with the OVA challenged+LV-shNC groups (Fig. 8D). Western blot and immunofluorescence analysis of fibrosis-related markers in the lung tissues also confirmed the same results (Fig. 8E and F). Overall, our research showed that the TL1A-DR3 axis might play an essential role in airway inflammation and remodeling in asthma.
TNFSF15 knockout may attenuate OVA-induced airway remodeling through the NF-κB pathway
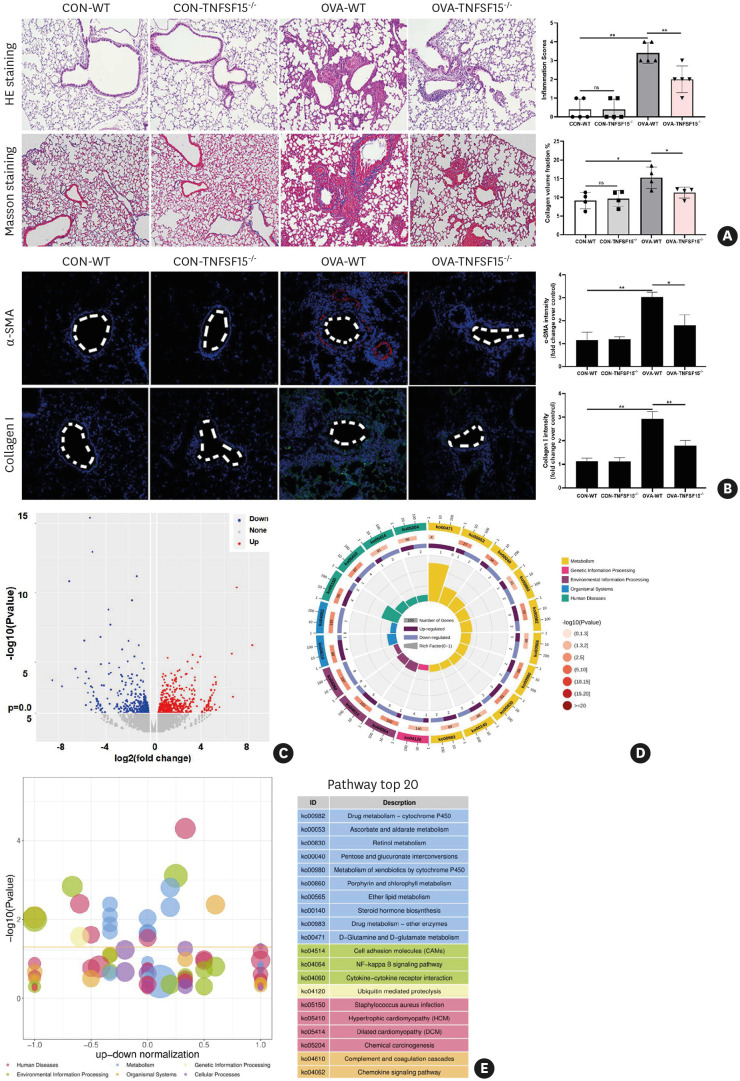
To further validate the authentic role of TL1A and characterize its signaling events related to downstream effector mechanisms in asthma progression, we generated TNFSF15-knockout mouse by CRISPR/Cas9 and tested our hypothesis using the knockout mouse model (Fig. 9). H&E and Masson staining as well as immunofluorescence analysis (α-SMA and collagen I) further proved the vital role of TL1A signaling. The results showed that along with diminished inflammation, markers of tissue remodeling were significantly attenuated, compared with OVA-induced WT controls (Fig. 9A and B). To further evaluate mechanisms by which TL1A loss of functions in asthma development, we next analyzed mRNA profiling in WT and knockout mouse models by RNA sequencing of total RNA extracted from the lung tissues. RNA-seq microarray analysis in the knockout mice identified over 370 differentially expression genes (DEGs), including 199 up-regulated and 177 down-regulated genes (Fig. 9C). The alterations in molecular mechanisms affected by these genes might explain the phenotypic difference between WT and knockout mice. To explore the characteristics of the 376 DEGs, KEGG pathway enrichment analyses were performed based on DEGs for this comparison (Fig. 9D-E), and NF-κB signaling pathways were significantly enriched in the KEGG pathway analysis, which is particularly marked for down-regulated genes. These results support that the effect observed with TNFSF15 knockout is dominated by inhibition of NF-κB signaling pathways.
Fig. 9. TL1A knockout effectively relieves OVA-induced airway inflammation and remodeling in mice.
(A) Following quantitative analysis, H&E and Masson staining were used to evaluate histopathological changes in the TNFSF15 WT and knockout mouse model. (B) Immunofluorescence detection of fibrosis “indicator” proteins (α-SMA and collagen I) and their quantitative analysis. (C) Volcano plot was used to present the results of RNA-seq from OVA-treated lung tissues between TNFSF15 WT and knockout mice (differential genes were determined using a P value of ≤ 0.05 and a |FC| > 1.2). (D and E) Bubble and circular maps of KEGG pathway enrichment.
TL1A, tumor necrosis factor ligand-related molecule 1A; OVA, ovalbumin; H&E, hematoxylin and eosin; WT, wild-type; α-SMA, α-smooth muscle actin; FC, fold-change.
*P < 0.05, **P < 0.01.
DISCUSSION
This study demonstrated that TL1A (both its secreted and non-secreted forms) was increased in asthmatic patients (induction sputum and airway epithelium) and the OVA-induced murine asthma model (lung tissues and BALF). The above results indicate that both nsTL1A and sTL1A may be involved in the asthma phenotype and development. In human bronchial epithelial cells induced by TGF-β1, TL1A expression was significantly increased, accompanied by fibrosis and EMT makers. Furthermore, down-regulation of TL1A or its receptor DR3 reversed the effects induced by TGF-β1, including the change in EMT and remodeling indicators. Some previous investigations have found that, in airway epithelial cells, the activation of the TL1A-DR3 axis using recombinant TL1A could alter airway remodeling. Our study demonstrated that elevated nsTL1A alone could directly promote the procession of remodeling induced by TGF-β1. These results confirmed that TL1A, especially its non-secreted form, plays a critical role in asthmatics airway remodeling processes. To further corroborate experimental results in vitro, we next carried out experiments in mice. In vivo experiments demonstrated that the intratracheal administration of TL1A could directly aggravate fibrosis in WT mice as an asthmatic mouse model, which could be reversed by the specific knockdown of DR3 or TL1A knockout. Inhibiting the airway remodeling and inflammatory response by TL1A knockout may be associated with down-regulation of the NF-κB signaling pathway. Collectively, in this study, the role of TL1A, especially its 2 distinct forms in asthmatic airway epithelium, was confirmed for the first time, further improving our understanding of the role of the TL1A-DR3 axis in asthma.
With the deterioration in air quality, the global incidence of asthma has been increasing.26 In that context, airway remodeling as an important common feature of asthma, especially severe refractory asthma, remains a challenge for treatment and clinical management.7 Currently, the airway epithelium is considered to play significant roles in the progression of asthma. In terms of airway remodeling, airway epithelium has also been assumed as an “initiator,” producing paracrine signals that induce proliferation, migration, and activation of inflammation. In our previous study, we also confirmed that elevated FSTL1 expression in the asthmatic airway epithelium is relevant to the functional significance of airway remodeling in asthma.22,27 So far, however, a complete scenario including detailed molecular mechanisms in airway epithelium pathological processes is not sufficiently understood, which hinders effective therapeutic strategies.
Given the role of TL1A in eosinophilic inflammation, much attention has been paid to the relationship between TL1A and asthma in recent years.18 TL1A is believed to act on Th2 cells and ILC2 to promote the production of Th2 cytokines.28,29 In addition to airway inflammation, excessive production of Th2 cytokines induced by TL1A in ILC2 also promotes mucus production, and interrupting TL1A/DR3 signaling decreases mucus production in an allergen-induced asthma model.30 Of great concern, one recent study revealed that TL1A and DR3 expression in patients with asthma significantly increased following allergen challenge.31 Given the important role of ILC2 in asthma, all these results emphasize the pivotal role of the TL1A/DR3 axis in asthma, especially in the progression of severe refractory asthma. However, the association of TL1A/DR3 signaling activity with airway remodeling in asthma has not been thoroughly investigated, especially with respect to the structural cells of the lungs, such as epithelial cells.
In conclusion, our study established a stronger link between the TL1A/DR3 axis and asthmatic airway remodeling. For the first time, we identify the sTL1A and nsTL1A together as being responsible for this process. Considering that TL1A could function via quite a few receptors, another receptor involved in this process and the complete mechanism by which TL1A affects asthmatic airway remodeling and function needs to be further investigated. This is the next step in our group’s research. To help answer this question, we have generated TNFSF15-deficient mouse lines by gene targeting and are trying to establish TNFSF15 transgenic (restricted membrane expression) mice. Future studies will include a detailed comparison between secreted versus non-secreted TL1A characteristics in asthma. Given the study’s exploratory nature, our research results will lay the foundation for further necessary research.
ACKNOWLEDGMENTS
This work was supported by grants from the National Natural Science Foundation of China (81770029), National Key Research and Development Project (2017YFC1310601) and Key Research Project of Shandong province (2017GSF218056). The funders had no role in the study design, data collection, data analysis, or preparation of the manuscript.
Footnotes
Disclosure: There are no financial or other issues that might lead to conflict of interest.
SUPPLEMENTARY MATERIALS
Characteristics of control subjects and patients with asthma
Characteristics of control subjects and asthmatic patients in sputum induction study
Primers for RT-qPCR
Overview of experimental design.
Detailed diagram for the animal model construction in this study.
Identification of TL1A knockdown efficiency upon TGF-β1 stimulation in BEAS-2B cells.
The level of TL1A RNA in the eosinophilic inflammation cellular model of asthma.
The concentration of the sTL1A in the model mouse blood.
Knockdown of DR3 with lentivirus in mice does not affect the expression of TL1A in the whole blood.
References
- 1.Hammad H, Lambrecht BN. The basic immunology of asthma. Cell. 2021;184:2521–2522. doi: 10.1016/j.cell.2021.04.019. [DOI] [PubMed] [Google Scholar]
- 2.Kim LH, Saleh C, Whalen-Browne A, O’Byrne PM, Chu DK. Triple vs dual inhaler therapy and asthma outcomes in moderate to severe asthma: a systematic review and meta-analysis. JAMA. 2021;325:2466–2479. doi: 10.1001/jama.2021.7872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cloutier MM, Dixon AE, Krishnan JA, Lemanske RF, Jr, Pace W, Schatz M. Managing asthma in adolescents and adults: 2020 asthma guideline update from the National Asthma Education and Prevention Program. JAMA. 2020;324:2301–2317. doi: 10.1001/jama.2020.21974. [DOI] [PubMed] [Google Scholar]
- 4.Liu G, Philp AM, Corte T, Travis MA, Schilter H, Hansbro NG, et al. Therapeutic targets in lung tissue remodelling and fibrosis. Pharmacol Ther. 2021;225:107839. doi: 10.1016/j.pharmthera.2021.107839. [DOI] [PubMed] [Google Scholar]
- 5.Elshaw SR, Henderson N, Knox AJ, Watson SA, Buttle DJ, Johnson SR. Matrix metalloproteinase expression and activity in human airway smooth muscle cells. Br J Pharmacol. 2004;142:1318–1324. doi: 10.1038/sj.bjp.0705883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Harmanci E, Kebapci M, Metintas M, Ozkan R. High-resolution computed tomography findings are correlated with disease severity in asthma. Respiration. 2002;69:420–426. doi: 10.1159/000064018. [DOI] [PubMed] [Google Scholar]
- 7.Zhang J, Dong L. Status and prospects: personalized treatment and biomarker for airway remodeling in asthma. J Thorac Dis. 2020;12:6090–6101. doi: 10.21037/jtd-20-1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hsu H, Viney JL. The tale of TL1A in inflammation. Mucosal Immunol. 2011;4:368–370. doi: 10.1038/mi.2011.20. [DOI] [PubMed] [Google Scholar]
- 9.Migone TS, Zhang J, Luo X, Zhuang L, Chen C, Hu B, et al. TL1A is a TNF-like ligand for DR3 and TR6/DcR3 and functions as a T cell costimulator. Immunity. 2002;16:479–492. doi: 10.1016/s1074-7613(02)00283-2. [DOI] [PubMed] [Google Scholar]
- 10.Haridas V, Shrivastava A, Su J, Yu GL, Ni J, Liu D, et al. VEGI, a new member of the TNF family activates nuclear factor-kappa B and c-Jun N-terminal kinase and modulates cell growth. Oncogene. 1999;18:6496–6504. doi: 10.1038/sj.onc.1203059. [DOI] [PubMed] [Google Scholar]
- 11.Clarke AW, Poulton L, Shim D, Mabon D, Butt D, Pollard M, et al. An anti-TL1A antibody for the treatment of asthma and inflammatory bowel disease. MAbs. 2018;10:664–677. doi: 10.1080/19420862.2018.1440164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yang CR, Hsieh SL, Teng CM, Ho FM, Su WL, Lin WW. Soluble decoy receptor 3 induces angiogenesis by neutralization of TL1A, a cytokine belonging to tumor necrosis factor superfamily and exhibiting angiostatic action. Cancer Res. 2004;64:1122–1129. doi: 10.1158/0008-5472.can-03-0609. [DOI] [PubMed] [Google Scholar]
- 13.Han F, Song J, Jia W, Yang M, Wang D, Zhang H, et al. TL1A primed dendritic cells activation exacerbated chronic murine colitis. Life Sci. 2020;262:118220. doi: 10.1016/j.lfs.2020.118220. [DOI] [PubMed] [Google Scholar]
- 14.Meylan F, Richard AC, Siegel RM. TL1A and DR3, a TNF family ligand-receptor pair that promotes lymphocyte costimulation, mucosal hyperplasia, and autoimmune inflammation. Immunol Rev. 2011;244:188–196. doi: 10.1111/j.1600-065X.2011.01068.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Doherty TA, Soroosh P, Khorram N, Fukuyama S, Rosenthal P, Cho JY, et al. The tumor necrosis factor family member LIGHT is a target for asthmatic airway remodeling. Nat Med. 2011;17:596–603. doi: 10.1038/nm.2356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ling C, Cai R, Gong Y, Ding X. DR3 and its ligands take roles in periodontium remodeling during orthodontic tooth movement. J Dent Sci. 2021;16:356–364. doi: 10.1016/j.jds.2020.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Herro R, Miki H, Sethi GS, Mills D, Mehta AK, Nguyen XX, et al. TL1A promotes lung tissue fibrosis and airway remodeling. J Immunol. 2020;205:2414–2422. doi: 10.4049/jimmunol.2000665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Skevaki C, Weckmann M. Activation of group 2 innate lymphoid cells via TL1A/DR3. A solution to corticosteroid resistance? Am J Respir Crit Care Med. 2020;202:1067–1069. doi: 10.1164/rccm.202007-2658ED. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Biener-Ramanujan E, Gonsky R, Ko B, Targan SR. Functional signaling of membrane-bound TL1A induces IFN-gamma expression. FEBS Lett. 2010;584:2376–2380. doi: 10.1016/j.febslet.2010.04.030. [DOI] [PubMed] [Google Scholar]
- 20.Fairs A, Agbetile J, Hargadon B, Bourne M, Monteiro WR, Brightling CE, et al. IgE sensitization to Aspergillus fumigatus is associated with reduced lung function in asthma. Am J Respir Crit Care Med. 2010;182:1362–1368. doi: 10.1164/rccm.201001-0087OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhang D, Qiao XR, Cui WJ, Zhang JT, Pan Y, Liu XF, et al. Syndecan-1 amplifies ovalbumin-induced airway remodeling by strengthening TGFβ1/Smad3 action. Front Immunol. 2021;12:744477. doi: 10.3389/fimmu.2021.744477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liu T, Liu Y, Miller M, Cao L, Zhao J, Wu J, et al. Autophagy plays a role in FSTL1-induced epithelial mesenchymal transition and airway remodeling in asthma. Am J Physiol Lung Cell Mol Physiol. 2017;313:L27–L40. doi: 10.1152/ajplung.00510.2016. [DOI] [PubMed] [Google Scholar]
- 23.Reuter S, Lemmermann NAW, Maxeiner J, Podlech J, Beckert H, Freitag K, et al. Coincident airway exposure to low-potency allergen and cytomegalovirus sensitizes for allergic airway disease by viral activation of migratory dendritic cells. PLoS Pathog. 2019;15:e1007595. doi: 10.1371/journal.ppat.1007595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hang P, Zhao J, Sun L, Li M, Han Y, Du Z, et al. Brain-derived neurotrophic factor attenuates doxorubicin-induced cardiac dysfunction through activating Akt signalling in rats. J Cell Mol Med. 2017;21:685–696. doi: 10.1111/jcmm.13012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Liu F, Shang YX. Sirtuin 6 attenuates epithelial-mesenchymal transition by suppressing the TGF-β1/Smad3 pathway and c-Jun in asthma models. Int Immunopharmacol. 2020;82:106333. doi: 10.1016/j.intimp.2020.106333. [DOI] [PubMed] [Google Scholar]
- 26.Bonato M, Gallo E, Bazzan E, Marson G, Zagolin L, Cosio MG, et al. Air pollution relates to airway pathology in children with wheezing. Ann Am Thorac Soc. 2021;18:2033–2040. doi: 10.1513/AnnalsATS.202010-1321OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Liu Y, Liu T, Wu J, Li T, Jiao X, Zhang H, et al. The correlation between FSTL1 expression and airway remodeling in asthmatics. Mediators Inflamm. 2017;2017:7918472. doi: 10.1155/2017/7918472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Meylan F, Hawley ET, Barron L, Barlow JL, Penumetcha P, Pelletier M, et al. The TNF-family cytokine TL1A promotes allergic immunopathology through group 2 innate lymphoid cells. Mucosal Immunol. 2014;7:958–968. doi: 10.1038/mi.2013.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Richard AC, Tan C, Hawley ET, Gomez-Rodriguez J, Goswami R, Yang XP, et al. The TNF-family ligand TL1A and its receptor DR3 promote T cell-mediated allergic immunopathology by enhancing differentiation and pathogenicity of IL-9-producing T cells. J Immunol. 2015;194:3567–3582. doi: 10.4049/jimmunol.1401220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Steele H, Sachen K, McKnight AJ, Soloff R, Herro R. Targeting TL1A/DR3 signaling offers a therapeutic advantage to neutralizing IL13/IL4Rα in muco-secretory fibrotic disorders. Front Immunol. 2021;12:692127. doi: 10.3389/fimmu.2021.692127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Machida K, Aw M, Salter BMA, Ju X, Mukherjee M, Gauvreau GM, et al. The role of the TL1A/DR3 axis in the activation of group 2 innate lymphoid cells in subjects with eosinophilic asthma. Am J Respir Crit Care Med. 2020;202:1105–1114. doi: 10.1164/rccm.201909-1722OC. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Characteristics of control subjects and patients with asthma
Characteristics of control subjects and asthmatic patients in sputum induction study
Primers for RT-qPCR
Overview of experimental design.
Detailed diagram for the animal model construction in this study.
Identification of TL1A knockdown efficiency upon TGF-β1 stimulation in BEAS-2B cells.
The level of TL1A RNA in the eosinophilic inflammation cellular model of asthma.
The concentration of the sTL1A in the model mouse blood.
Knockdown of DR3 with lentivirus in mice does not affect the expression of TL1A in the whole blood.