Abstract
Background
CD73 is an ectonucleotidase producing the immunosuppressor mediator adenosine. Elevated levels of circulating CD73 in patients with cancer have been associated with disease progression and poor response to immunotherapy. Immunosuppressive pathways associated with exosomes can affect T-cell function and the therapeutic efficacy of anti-programmed cell-death protein 1 (anti-PD-1) therapy. Here, we conducted a retrospective pilot study to evaluate levels of exosomal CD73 before and early during treatment with anti-PD-1 agents in patients with melanoma and its potential contribution to affect T-cell functions and to influence the clinical outcomes of anti-PD-1 monotherapy.
Methods
Exosomes were isolated by mini size exclusion chromatography from serum of patients with melanoma (n=41) receiving nivolumab or pembrolizumab monotherapy. Expression of CD73 and programmed death-ligand 1 (PD-L1) were evaluated on exosomes enriched for CD63 by on-bead flow cytometry. The CD73 AMPase activity was evaluated by mass spectrometry, also in the presence of selective inhibitors of CD73. Interferon (IFN)-γ production and granzyme B expression were measured in CD3/28 activated T cells incubated with exosomes in presence of the CD73 substrate AMP. Levels of CD73 and PD-L1 on exosomes were correlated with therapy response. Exosomes isolated from healthy subjects were used as control.
Results
Isolated exosomes carried CD73 on their surface, which is enzymatically active in producing adenosine. Incubation of exosomes with CD3/28 activated T cells in the presence of AMP resulted in a significant reduction of IFN-γ release, which was reversed by the CD73 inhibitor APCP or by the selective A2A adenosine receptor antagonist ZM241385. Expression levels of exosomal CD73 from serum of patients with melanoma were not significantly different from those in healthy subjects. Early on-treatment, expression levels of both CD73 and PD-L1 on exosomes isolated from patients receiving pembrolizumab or nivolumab monotherapy were significantly increased compared with baseline. Early during therapy exosomal PD-L1 increased in responders, while exosomal CD73 resulted significantly increased in non-responders.
Conclusions
CD73 expressed on exosomes from serum of patients with melanoma produces adenosine and contributes to suppress T-cell functions. Early on-treatment, elevated expression levels of exosomal CD73 might affect the response to anti-PD-1 agents in patients with melanoma who failed to respond to therapy.
Keywords: adenosine, melanoma, immunotherapy
Introduction
Melanoma is one of the most aggressive malignancies, with highly immunosuppressive features. Melanoma environment is characterized by elevated concentration of immunosuppressive soluble mediators, including exosomes, and cells with impaired functions, expressing high levels of immune checkpoints such as cytotoxic T-lymphocytes-associated protein 4 (CTLA-4) or programmed cell-death protein 1 (PD-1), which binding their own ligands on cancer cells, stromal cells or immune cells dampen the T-cell attack.1 Some current treatments are based on the use of immunotherapeutic agents that specifically block the immune checkpoints CTLA-4 or PD-1 to rejuvenate immune responses of the patients.2 Nivolumab or pembrolizumab are anti-PD-1 agents approved, alone or in combination strategies, for the treatment of metastatic melanoma. These agents importantly improved the survival of patients,2 however, about 55% of patients still do not respond to anti-PD-1 agents in monotherapy, while the percentage of patients not responsive to the combination with ipilimumab (anti-CTLA-4 monoclonal antibody) is about 42%.3 This evidence indicates that the effects of the immune checkpoints inhibitors are affected by additional suppressive factors, whose identification can be helpful to develop new therapeutic strategies and/or to monitor responses to immunotherapy.
Exosomes are extracellular vesicles that are between 30 and 150 nm, generated in the endosomal compartment of cancerous and non-cancerous cells, found in many biological fluids including blood.4 Exosomes functions in cell-to-cell communication are not yet fully understood, and their role can be complex according to the cancer type or context and their heterogeneity. Cancer exosomes can carry a variety of receptors, ligands, enzymes or other factors that can affect responses of tumor cells and stromal cells within tumor microenvironment, as well as responses of immune cells against tumor cells,4–6 largely resulting in pro-tumor and pro-metastatic effects. In melanoma, serum exosomes can be potentially useful as biomarkers of tumor progression and outcome having high expression of specific immunosuppressive markers.7–9 In particular, programmed death-ligand 1 (PD-L1) positive exosomes detected in plasma of patients with melanoma may predict the response to anti-PD-1 therapy,10 in addition to being a prognostic factor.11
CD73 is the main enzyme involved in the production of extracellular adenosine, a potent immunosuppressor, pro-angiogenic and pro-tumor factor that accumulates within tumor lesions.12 Specifically, this enzyme is able to hydrolyse AMP into adenosine and inorganic phosphate and it often works in concert with CD39, which produces AMP from ATP/ADP.13 The substrate for CD73 can also derive from an alternative pathway, involving the activity of CD38 and CD203a, which respectively convert NAD+ into ADP-ribose and ADP-ribose into AMP,14 while other enzymes including tissue-specific alkaline phosphatases such as prostate acid phosphatase, and tissue-non-specific alkaline phosphatases are able to convert extracellular AMP to adenosine. Considering its key role in the production of extracellular adenosine, CD73 has emerged as an attractive target for new anticancer therapeutic strategies, encouraging the development of clinical trials testing some monoclonal antibodies against CD73, including oleclumab, BMS-986179, CPI-006 and NZV930, alone or in combination with other treatments15 with some promising preliminary data from clinical trials of oleclumab showing efficacy.16 17 Furthermore, many studies have suggested the possibility to measure its expression and/or activity as a tool to predict the outcome of patients with cancer.18
Besides being expressed by many cell types, CD73 exists in a soluble form in peripheral blood.19 The soluble CD73, derived from the cleavage of the cell-membrane anchored CD73, shows a marked AMPase activity.19 Many studies have evaluated the role of the soluble CD73 in immunosuppression, being able to produces adenosine, and its correlation with response to anticancer therapeutic agents. We have demonstrated that the levels of activity and expression of the soluble CD73 in serum are significantly higher in patients with melanoma, having the potential as predictive biomarker for response to immunotherapy.20 21 The soluble CD73 can also include a form expressed on exosomes found in biological fluids, as plasma and serum22–24 and pleural fluid.25 Exosomes expressing CD73 may be generated by tumor cells or immune cells, including T regulatory cells or B cells or CD8 +T cells.23–30 Importantly, exosomal CD73 resulted enzymatically active.23–25 27 31 This evidence suggests an important role for exosomes in promoting an immunosuppressive environment that could likely lead to the generation of pro-tumorigenic niches and to the suppression of T-cell functions, impairing the immunotherapeutic efficacy.
In this study, we used serum from patients with metastatic melanoma receiving anti-PD-1 agents, nivolumab or pembrolizumab, to isolate exosomes and evaluate the functional relevance of exosomal CD73 in affecting T-cell functions ex vivo and as a potential biomarker of response to immunotherapy.
Methods
Serum samples
This retrospective study involved a total of 41 patients with melanoma. Patients were treated with nivolumab (n=21) or pembrolizumab (n=20) monotherapy in first, second or third line, and response to therapy was evaluated according to response evaluation criteria in solid tumors (RECIST) V.1.1 criteria. Tumor response was assessed every 12 weeks. Eighteen patients responded to therapy (partial response, PR=14; complete response, CR=4), while 23 patients did not benefit from treatment (progressive disease, PD=19; stable disease, SD=4).
Sera from patients were collected at Istituto Nazionale Tumori-IRCCS-Fondazione ‘G. Pascale’, Naples (Italy), prior to (baseline) treatment. For n=13 patients paired serum samples at baseline (before starting therapy) and on-treatment were available for analysis within each patient. Samples from healthy subjects were collected at random at the Transfusion and Stem Cell Transplantation Unit of the same institute for a total of 24 subjects (men, n=14 and women, n=10) aged >18 years. Written consent was obtained from each subject for blood donation.
All samples in 1 mL aliquots were stored at −80°C and thawed immediately prior exosomes isolation.
Exosomes isolation
Exosomes were isolated by using size exclusion chromatography (SEC) as described by Hong and collaborators.32 We modified the protocol as follows. Briefly, sera were thawed and filtered by using 70 µm pores filter. Then, the samples were centrifuged for 10 min at 2000 g, 4°C and then for 30 min at 10,000 g, 4°C. Then, samples were ultrafiltered by using Spin-X Centrifuge Tubes filter (Corning Incorporated) which are equipped with a 0.22 µm pore membrane. Exosomes can pass through the pores, while the bigger vesicles and/or other contaminants are retained on the top of the membrane. Volume of the samples was adjusted, if necessary, to 1 mL, adding phosphate-buffered solution (PBS). The samples were then loaded onto a column (Hercules Econo-pac, Bio-Rad) packed with 10 mL of Sepharose (CL-2B300, Sigma). We used 1 mL of PBS as eluent buffer for each fraction. As already described,24 32 33 exosomes are eluted mostly in the fraction # 4, that was used for all the experiments we describe here. Once collected, the fraction #4 was transferred to Amicon Ultra Centrifugal filter Units, MWCO 100 kDa (Millipore #UFC510024) to separate as much as possible exosomes from the presence of soluble factors.34
BCA-protein assay of isolated exosomal samples
The protein concentration of the purified exosomal samples was determined by Pierce BCA Protein Assay Kit (Thermo Scientific). Results were expressed as µg of proteins/mL of serum.
Exosomes proteins characterization by western blotting
Protein expression from purified exosomal samples was examined by western blot. Proteins amounting to 10 μg were prepared in Laemmli buffer and loaded into a 10% polyacrylamide gel. The primary antibodies used were the following: CD9 (1: 1000; C-4: sc-13118, SantaCruz Biotechnology), CD-81 (1:1000; 1.3.3.22: sc-7637, SantaCruz Biotechnology), Calregulin (1:1000; A-9: sc-166837, SantaCruz Biotechnology), detected with the secondary antibody goat anti-mouse IgG1k HRP-conjugated (1:20000; A90-105P, Bethyl Laboratories) using the chemiluminescence detection system. For CD73 we used an anti-CD73 antibody (1:1000; EPR6114; Abcam) and the goat anti-rabbit antibody (1:2000) as secondary antibody. Bands we visualized using Las4000 Imaging System (GE Healthcare Life Sciences).
Dynamic light scattering
Vesicle size distribution was measured by dynamic light scattering (DLS) using a Zetasizer V.7.01, Malvern Instrument (Malvern, UK). Each sample was dispersed in deionized water and the intensity of the scattered light was measured with a detector at 90° angle at room temperature. Mean diameter and size distribution were the mean of three measures.
Scanning electron microscope
Visualization of the isolated exosomal samples was performed by scanning electron microscope (SEM). Samples were diluted (1:2) in ultrapure water (Milli-Q UF Plus, Millipore, Molsheim, France) and spotted onto carbon-coated aluminum stabs. Solvent was evaporated under nitrogen flux overnight. SEM images of the samples were then acquired using a Tescan Solaris microscope equipped with secondary electron and backscattered electron detectors (TESCAN, Brno, Czech Republic). Analyses were conducted both at 1.5 and 5 keV without any coating of the particles, respectively.
Beads-assisted flow cytometry
To detect the expression of proteins on the exosomes surface by flow cytometry, it was required to conjugate exosomes with magnetic beads. Specifically, we used the ExoCap Streptavidin Kit (MEX-SA) from MBL. We adapted the protocol described by Theodoraki et al,24 as follows. Exosomes fraction #4 amounting to 50 µL were incubated for 2 hours with 0.5 µg of anti CD63 biotinylated antibody (BioLegend, Clone: H5C6, 353017), at room temperature on rotary mixer. Then, 10 µL of the streptavidin magnetic beads from the kit were added and samples were incubated for 2 hours at room temperature, on rotary mixer. During this incubation, the streptavidin on the beads bound the anti CD63 antibody, capturing the exosomes and creating in turn the complex. The exosomes complexes were captured using a magnet (EasySep Magnet, STEMCELL Technologies). Samples were then resuspended in 250 µL of PBS, and the suspension was divided into 5 aliquots of 50 µL, each used for the staining with one antibody. The antibodies used for these analyses were the following: anti-human CD73 FITC (Invitrogen, AD2, 11-0739-42) (0.250 µg/sample); anti-human PD-L1 APC (Invitrogen, B7-H1, 17-5983-42) (0.250 µg/sample); while the control isotypes were the following: mouse IgG1K APC (Invitrogen, 1 74 714–82) (0.250 µg/sample), mouse IgG1K FITC (Invitrogen, 17-4714-42) (0.250 µg/sample). Samples were incubated for 1 hour at room temperature, repaired from the light, on rotary mixer. Then, the supernatant was thrown away and exosomes were resuspended in 300 µL of PBS.
A total of 10,000 events for each sample was acquired using a BD FACScalibur (Becton Dickinson). Results were analyzed by using BD CellQuest Pro Software. The mean fluorescence intensity (MFI) of positive beads, gated on forward and sideward light scatter, was measured. Results are expressed as relative fluorescence intensity, calculated as ratio between the MFI of the target of interest and the MFI of the control isotype. Values greater than 1 were considered positive.
CD73 enzymatic activity measurement
To measure the activity of CD73 in purified exosomal samples, we evaluated the AMPase hydrolysis using multiple reaction monitoring mass spectrometry (MRM-MS), as described before.21 A total of 5 µg of exosomes proteins in 50 µL total volume of PBS were incubated at 37°C for 10 min, and then treated or not with CD73 inhibitors for 30 min at 37°C. Specifically, we used 5’ α,β-methylene-ADP (APCP; Sigma-Aldrich) (100 µM) or the anti-human CD73 mAb (Clone 7G2; Thermo Fisher Scientific) (5 µg/mL). Then, 15N-AMP substrate (10 µM; Sigma-Aldrich) was added and substrate and samples were incubated for 120 min at 37°C. Immediately after adding the substrate and after 120 min of reaction, 15 µL of each sample were collected and treated with ice-cold trichloroacetic acid (5% final concentration; Sigma-Aldrich) to quench the reaction. Samples were then centrifuged at 3000 g for 10 min, 4°C. The supernatants were dried in Concentrator Plus (Eppendorf) and dissolved in 25 µL of 10 mM Ammonium Acetate (AmAc, Sigma-Aldrich) containing 0.1% acetic acid (Sigma-Aldrich). The samples were used to perform ultra-performance liquid chromatography-electrospray ionization (UPLC-ESI)-MRM-MS analysis, in order to quantify the 15N-adenosine produced. Briefly, experiments were performed using a 6500 Q-TRAP from AB Sciex equipped with Shimadzu LC-20A and Autosampler systems, injecting 3 µL of each sample. UPLC separation was performed on a Luna Omega Polar 1.6 µm C18 100 Å column (50×2.10 mm, Phenomenex, Torrance, California, USA) at a flow rate of 400 µL/min and using 10 mM AmAc/0.1% Acetic acid in H2O (A) and 0.1% Acetic acid in methanol (B) as mobile phases. The gradient was set as follows: 0.5 min at 0% B, 0.5 min to 3 min at 5% B, 3 min to 5 min at 95% B, 5 min to 7 min at 95% B and then back to 0% B for a 5 min re-equilibration step.
15N-AMP was monitored through the 353.19/140.00 transition and 15N-Adenosine through the 273.00/141.00 one; the areas of their related peaks in each sample were measured using the Analyst Software from AB Sciex.
Functional assays
Peripheral blood mononuclear cells of healthy subjects were isolated by Ficoll density gradient (Histopaque-1077, Sigma–Aldrich). After isolation, cells were incubated in RPMI 1640 (Euroclone S.p.A.) supplemented with 10% (v/v) heat-inactivated fetal bovine serum (Euroclone S.p.A.), 1% (v/v) penicillin–streptomycin (Euroclone S.p.A.), 1% (v/v) MEM non-essential amino acids (Euroclone S.p.A.), and 1% (v/v) sodium pyruvate (Euroclone S.p.A.). One day after, cells (2×105/well) were incubated in supplemented RPMI without serum, and exosomes isolated from serum of patients with melanoma were added at concentration of 100 µg/mL. PBS was added to the cells instead of exosomes as control. AMP (50 µM) was added for 1 hour and then cells were activated using ImmunoCult Human CD3/CD28 T cell activator (25 µg/mL), according to the manufacturer’s instructions (STEMCELL Technologies). For some experiments, cells were pretreated with the CD73 inhibitor APCP (100 µM) or the A2A adenosine receptor antagonist ZM 241385 (1µM) (Sigma-Aldrich) before adding the substrate AMP. The levels of interferon (IFN)-γ were quantified in the cells supernatant 72 hours after treatments using DuoSet ELISA, following the manufacturer’s protocol (R&D Systems). Granzyme B expression was determined by western blot analysis using the primary antibody against granzyme B (1:1000; BD Pharmingen) in whole cell lysates collected 24 hours later treatments described above.
Statistical analyses
For statistical analysis, we used GraphPad Prism V.7.0. The Mann-Whitney test or analysis of variance test were used for two groups comparison or three or more groups comparison, respectively. For the cell culture experiments two-tailed unpaired t-test was used. To compare the expression of CD73 or PD-L1 between two time points, we used the Wilcoxon matched-pairs signed-rank test. Sample size (n) for each experimental group is indicated in each figure legend. Data are expressed as mean±SD. P values<0.05 were considered as significant.
Results
Characteristics of patients with melanoma
In this study, we used serum from patients with stage IV melanoma (85.4%) or stage III unresectable (14.6%) (VII edition of the American Joint Committee on Cancer, AJCC), including 26 men (63.4%) and 15 women (36.6%) with a median age of 64 years (range 23–85 years). Patients with melanoma were treated with nivolumab (n=21) or pembrolizumab (n=20) in monotherapy for a total of 41 patients; 17 patients received an anti-PD-1 agent as first-line of treatment; 17 patients received second-line PD-1 therapy and 7 patients in third-line. In details within the patients group receiving second-line treatment with anti-PD-1 agents 13/17 patients had previously received ipilimumab, while 4/17 patients received inhibitors of mitogen-activated protein kinases (MAPKs). Patients in third-line treatment with anti-PD-1 agents had previously received ipilimumab and then MAPK inhibitor. The clinic-pathologic characteristics of patients are reported in table 1.
Table 1.
Clinic-pathological characteristics of patients
| Nivolumab or pembrolizumab N (%) |
|
| Total | 41 (100) |
| Median age | |
| Range (23–85) | 64 |
| Gender | |
| Male | 26 (63.4) |
| Female | 15 (36.6) |
| Disease stage | |
| III | 6 (14.6) |
| IV | 35 (85.4) |
| Line of treatment | |
| I | 17 (41.5) |
| II | 17 (41.5) |
| >III | 7 (17) |
Purification and characterization of exosomes from serum and protein concentrations
Exosomes were isolated from serum samples of patients with melanoma. Exosomes from serum of healthy volunteers were also isolated as controls. Purification of exosomes from serum were performed by mini SEC as described in the Methods section and isolated exosomes in the fraction #4 were further characterized by evaluating their morphology, size distribution and expression of the characteristic exosomal markers tetraspanins CD9 and CD81.35 DLS showed a size distribution of exosomes with an average diameter of 105.5±21.99 nm (n=4). The representative image of size distribution of exosomes is shown in figure 1A. The SEM analysis confirmed the presence of particles within the size range measured by DLS and spherical morphology (figure 1B). Isolated exosomal samples express both CD9 and CD81, specific for exosomes, and are negative for the expression of calregulin, which is a cytoplasmic marker, as shown in the western blot representative images (figure 1C). We observed that the total protein content on purified exosomal samples from patients with melanoma were similar to those measured in healthy donors (data not shown).
Figure 1.
Exosomes characterization. (A) Size distribution profile of exosomes isolated from serum, measured by dynamic light scattering. (B) Exosomes morphology detected by scanning electron microscope. (C) Expression of proteins in the exosomes extract. The images are representative of n=4 independent experiments.
CD73 expression on exosomes and CD73-dependent AMPase activity
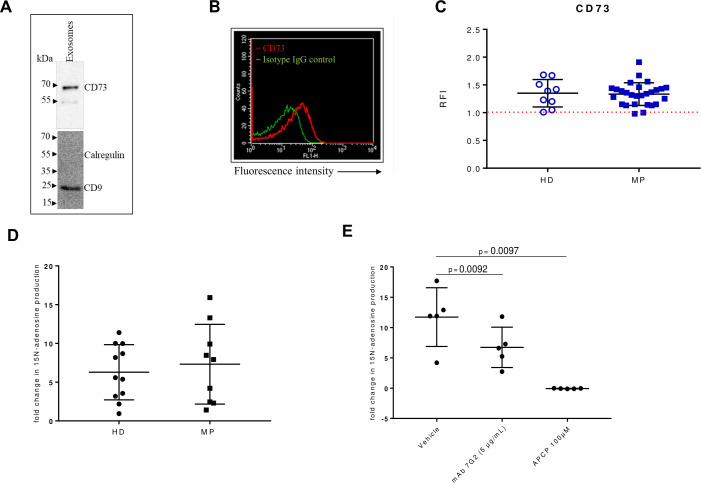
Previous studies have demonstrated that cancer exosomes express CD73.23–25 Analysis by western blotting indicated the presence of CD73 on isolated exosomes (figure 2A). To determine the expression of CD73 by flow cytometry, purified exosomes were captured for CD63 and bound to magnetic beads. Expression of CD73 on isolated exosomes was evaluated using a specific antibody anti CD73 in comparison with the isotype control (figure 2B). CD73 was detected on exosomes isolated from healthy subjects and patients with melanoma, with no significant difference between the two groups (figure 2C).
Figure 2.
CD73 expression and activity in serum-derived exosomes. (A) A representative western blot image for CD73 in purified exosomes (10 µg loaded protein). CD9 was used as specific exosomal marker and calregulin as negative marker. (B) A representative flow cytometry dot plot showing the expression of CD73 on isolated exosomes captured for CD63 and bound to magnetic beads, labeled with a specific antibody anti-CD73 (red line) or the isotype IgG control (green line). (C) Levels of CD73 expression as relative fluorescence intensity (RFI) values on exosomes isolated from healthy donors (HD) (n=9) and patients with melanoma (MP) at baseline (n=27). Comparison of the fold change in the 15N adenosine production, measured by mass spectrometry, after incubating for 2 hours 5 µg of exosomes isolated from serum of HD (n=6) or MP (n=9) with 15N AMP (10 µM) (D), with or without the anti-human CD73 mAb (5 µg/mL) or APCP (100 µM) (E), (n=5). Data are mean±SD (C, D, E). P values are obtained from a repeated measures (RM) one-way analysis of variance, followed by Dunnet’s multiple comparison test (E).
To evaluate the function of CD73 expressed on the surface of exosomes, we analyzed the AMPase activity by mass spectrometry in presence of 15N AMP. Exosomes from both healthy subjects and patients with melanoma at baseline produced 15N adenosine by hydrolysing 15N AMP (figure 2D). The results show that the AMPase activity in patients with melanoma at baseline was not statistically different from that in healthy donors. To test whether CD73 was the principal enzyme involved in the hydrolysis of 15N AMP, the AMPase activity was evaluated also in presence of the CD73 inhibitor APCP or the anti-CD73 functional antibody (clone 7G2), using exosomes isolated from patients with melanoma. We observed that the fold change in 15N adenosine production was significantly reduced in presence of the anti CD73 antibody or in presence of the false substrate of CD73, APCP (figure 2E), confirming that exosomes produce adenosine in a CD73-dependent manner. Moreover, analysis of the total serum AMPase activity in paired samples from patients with melanoma at baseline shows that the exosomal production of 15N-adenosine did not correlate with total serum production of 15N-adenosine (Pearson correlation r = −0,04131; p=0.9226, n=8) (online supplemental figure 1). These results suggest that in patients with melanoma at baseline there is no correlation between serum CD73 activity and exosomal CD73 activity.
jitc-2021-004043supp001.pdf (17.3KB, pdf)
Exosomal CD73 inhibits T-cell functions
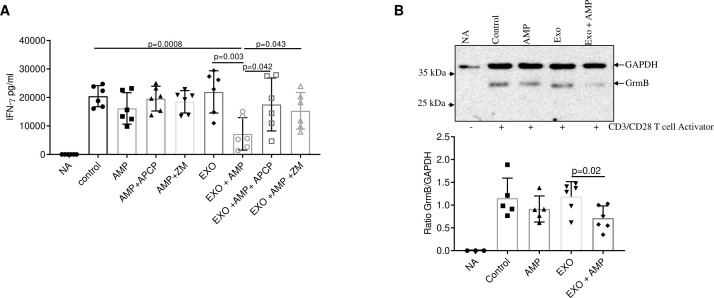
Producing adenosine, CD73 potently suppresses the function of T cells.36 To test the effect of exosomal CD73 on T-cell functions, we used human peripheral blood cells from healthy donors. T cells were activated with an anti CD3/CD28 T cell activator, in presence or not of exosomes derived from patients with melanoma and the CD73 substrate AMP. We measured the IFN-γ levels in the supernatants of cells 72 hours later treatment by ELISA. Control cells without CD3/28 activation (NA) did not produce at all IFN-γ (figure 3A). The results show that the levels of IFN-γ in cells pre-treated with exosomes were significantly reduced when adding the substrate AMP compared with control or activated cells treated with exosomes alone (figure 3A). In addition, this latter effect resulted abrogated in presence of the CD73 inhibitor APCP (figure 3A). Notably, treatment of cells with exosomes alone did not influence the production of IFN-γ in CD3/28 activated cells in comparison with activated control cells (figure 3A), ruling out any possible direct effects of exosomes on IFN-γ production in our experimental conditions. Furthermore, we also verified that, in our experimental conditions, treatment with melanoma exosomes, in presence or not of AMP, did not affect the cells viability (data not shown). Treatment of cells with AMP alone induces a trend to a reduction in IFN-γ production compared with CD3/28 activated control cells but not statistically significant, and remains unaltered in presence of APCP (figure 3A), suggesting that the inhibitory effects induced by AMP were independent of CD73 expressed on T cells, that could participate in hydrolysing the substrate AMP.
Figure 3.
Exosomes inhibit T-cell functions in presence of AMP. (A) Interferon (IFN)-γ levels were measured by ELISA in the supernatant of CD3/28 activated human peripheral cells from healthy donors with or without serum-derived exosomes (EXO) (100 µg/mL) in presence or absence of the CD73 substrate AMP (50 µM). The selective CD73 inhibitor APCP (100 µM) or the A2A adenosine receptor antagonist ZM241385 (ZM, 1 µM) were also used (n=6 independent biological experiments). Data are mean±SD. P values are from a two-tailed unpaired t-test. (B) Representative immunoblot (upper panel) and quantification (lower panel) of granzyme B (GrmB) by western blot analysis in CD3/28 activated human peripheral cells from healthy donors incubated with exosomes isolated from patients with melanoma (100 µg/mL) (n=6) with or without AMP (50 µM). GAPDH expression was evaluated as protein loading control. Non activated cells (NA) and CD3/28 activated cells were used as control. Data are mean±SD. P values are from a two-tailed unpaired t-test.
To further unravel the role of adenosine derived from AMP hydrolysis by exosomal CD73, we also performed experiments in the presence of the selective A2A adenosine receptor antagonist ZM 241385. A2A adenosine receptor is the main adenosine receptor expressed on T cells capable to suppress potently the functions of T cells.37 The results of these experiments revealed that treatment with ZM 241385 abrogated the inhibitory effects of AMP in presence of exosomes (figure 3A). These results suggest that adenosine generated from AMP by exosomes inhibits IFN-γ production via A2A adenosine receptor.
In addition, the expression of the granzyme B in CD3/28 activated cells is reduced in presence of exosomes when adding the substrate AMP (figure 3B).
Altogether these results indicate that exosomal CD73 suppresses T-cell functions in presence of AMP by producing adenosine, via A2A adenosine receptor.
Expression level of exosomal CD73/PD-L1 in association with response to therapy
To investigate the clinical relevance of exosomal CD73 as immune-regulatory molecule in serum of patients with melanoma undergoing anti-PD-1 monotherapy in association with treatment response, we analyzed the expression levels of CD73 prior to therapy (baseline) and during therapy (week 4), available for this study. Therefore, the expression levels of CD73 were analyzed on exosomes isolated from patients’ sera and correlated with treatment responses to immunotherapy. Patients were arbitrarily divided in two groups: responders (n=11), including patients with CR (n=3) or PR (n=8) and non-responders (n=14), including patients with PD (n=12) or SD (n=2). At baseline the levels of exosomal CD73 in clinical responders to anti-PD-1 agents were similar to those observed in non-responders (figure 4). Moreover, at baseline, the expression levels of exosomal CD73 were similar in patients receiving anti-PD-1 agents as first-line treatment to those in patients treated in second line (online supplemental figure 2).
Figure 4.
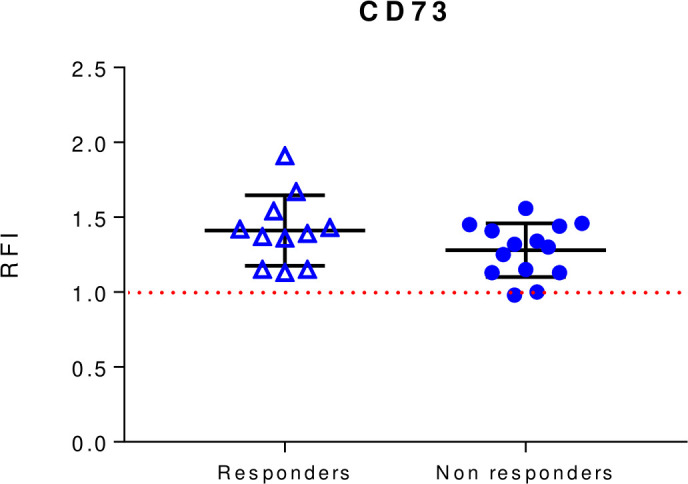
Baseline levels of exosomal CD73 expression in responders and non-responders to anti-PD-1 blockers nivolumab or pembrolizumab. CD73 expression was evaluated by beads-assisted flow cytometry on exosomes isolated from patients with melanoma prior to initiation of treatment. Responders n=11. Non-responders n=14. Data are mean±SD. PD-1, programmed cell-death protein 1; RFI, relative fluorescence intensity.
jitc-2021-004043supp002.pdf (41.6KB, pdf)
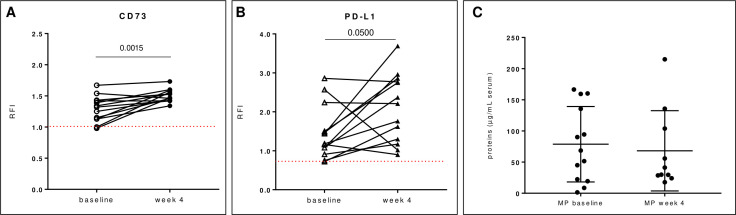
Notably, when we analyzed the expression of CD73 on exosomes isolated from serum of patients early during the therapy, we found that the levels of CD73 resulted significantly increased at week 4 compared with baseline (figure 5A). The previous treatment of patients did not influence the expression levels of exosomal CD73 at week 4 (online supplemental figure 2). Recent studies indicate that the expression levels of PD-L1 on circulating exosomes surface correlate with response to therapy10 in patients with melanoma undergoing PD-1 therapy (at week 3–6). Accordingly, the expression levels of PD-L1 on exosomes isolated from serum of patients with melanoma at week 4 resulted significantly increased compared with those at baseline (figure 5B). Analysis of the total protein content in exosomes isolated from patients with melanoma at baseline and at week 4 during therapy did not reveal any significant difference between the two groups (4 week mean: 68.15±20.35 µg/mL (n=10) vs baseline mean: 78.69±16.8 µg/mL (n=13)) (figure 5C).
Figure 5.
Levels of exosomal CD73 and PD-L1 on-treatment. Exosomes isolated from serum of patients with melanoma during treatment with anti-PD-1 agents at week 4 have significantly increased expression of CD73 (A) or PD-L1 (B) compared with those at baseline (n=15). (C) Total protein content on exosomes isolated from serum of patients at baseline and at week 4 during therapy was determined using the BCA Pierce kit assay. Data are mean±SD. P values are from Wilcoxon matched-pairs signed-rank test (A–B). MP, patients with melanoma; PD-L1, programmed death-ligand 1; RFI, relative fluorescence intensity.
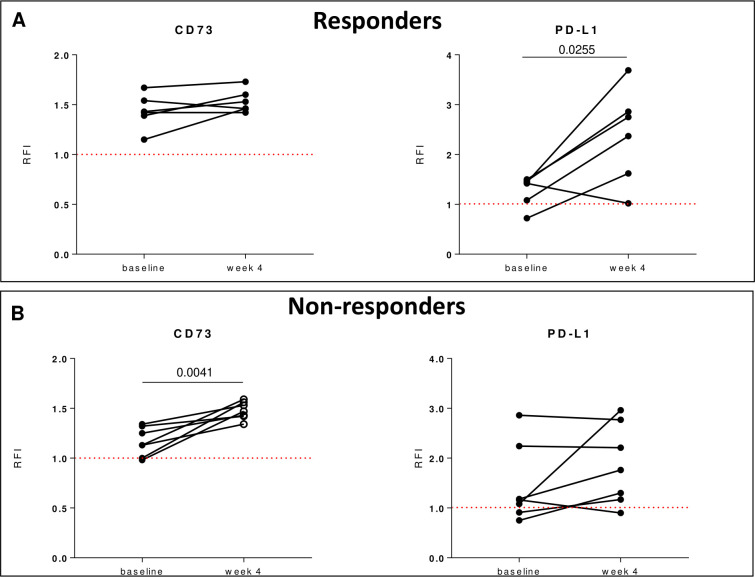
Analyzing in detail the exosomal expression levels of CD73 during treatment in responders and non-responders, we found that the levels of CD73 unchanged at week 4 of therapy in responders (figure 6A), while the expression of CD73 significantly increased at week 4 in non-responders (figure 6B). These results suggest that in non-responders there were increased levels of CD73 on exosomes early during treatment within 4 week of treatment. Moreover, in non-responders we observed that, in purified exosomes isolated from samples matched from the same patient with baseline and on treatment sera at week 4, the AMPase activity also increased on-treatment with a mean fold change in 15N adenosine production of 7.17±2.14 at baseline versus 13.65±1.62 at week 4 (n=5). These results suggest that in non-responders early-on-treatment also the exosomal CD73 activity enhanced compared with baseline.
Figure 6.
On-treatment changes in exosomal expression of CD73/PD-L1 in responders e non-responders. Levels of CD73 or PD-L1 on circulating exosomes within 4 weeks of therapy in responders (n=6) (A) and non-responders (n=7) (B). Data are mean±SD. P values are from Wilcoxon matched-pairs test. PD-L1, programmed death-ligand 1; RFI, relative fluorescence intensity.
In line with published data,10 the expression levels of exosomal PD-L1 resulted significantly increased at week 4 in the responders group (figure 6A) but not in non-responders (figure 6B). Altogether, these results suggest that changes in CD73 levels in circulating exosomes early during therapy would reflect a failure to respond to anti-PD-1 agents.
Discussion
We investigated the functional relevance of CD73 expressed on exosomes isolated from serum of patients with melanoma receiving nivolumab or pembrolizumab monotherapy. This study is the first to report an association between exosomal CD73 expression levels and patient responses to anti-PD-1 agents early during treatment of metastatic melanoma.
Despite the success of immunotherapy in the clinical practice, still many patients experienced primary or acquired resistance. Other subsets of patients who show clinical benefits to immunotherapy become refractory to anti-PD-1 or PD-L1 agents during therapy. The identification of factors influencing the effectiveness of treatment with immune-checkpoint inhibitors is critical to drive the clinical choices, and it is still an open field that requires more efforts.38 Growing evidence indicates that multiple mechanisms can be responsible of resistance, including altered expression of targeted molecules, resistance to IFN-γ, accumulation of immunosuppressive immune cells and suppressive mediators, or upregulation of additional immune checkpoints.39 40 Interestingly, circulating tumor-derived exosomes have emerged as a potential and interesting mechanism of tumor-induced immune suppression in melanoma.29
CD73 is the rate limiting enzyme in the production of the extracellular adenosine, a mediator able to strongly inhibit the CD8 +T cells functions. The A2A receptor is the main adenosine receptor subtype involved in the T-cell immunosuppressive effects of adenosine.41 The activation of this receptor on T cells is able to block the early signal transduction of the T-cell receptor (TCR) in a cAMP-dependent manner, attenuating in turn the T-cell responses, including proliferation and cytokines production.37 Many studies have demonstrated that CD73 is overexpressed in many human solid cancers, either expressed on tumor tissues or in a circulating form, soluble or exosomal, and often its overexpression is associated with poor prognosis.18 Preclinical studies demonstrated that blockade of CD73 can restore the antitumor immune response in combination with immunotherapeutic agents,42 providing the rationale for using CD73 inhibitors to enhance the effectiveness of anti-PD-1 therapy in the ongoing clinical trials.15 Interestingly, we have recently demonstrated the soluble form of CD73 as predictive peripheral blood biomarker of response to nivolumab or pembrolizumab in patients with metastatic melanoma.21 The serum contains multiple forms of CD73: the soluble CD73 which derives from the cleavage of the cell-membrane anchored CD73, and a form expressed on exosomes. Importantly, it is known that cancer exosomes carry CD73, which is enzymatically active in producing adenosine.23–25 31 CD73 +exosomes circulating in the body fluids have the capacity to regulate the production of adenosine, that in patients with cancer potently contribute to promote tumor progression and metastases. Investigating the different forms of soluble CD73 may help to understand their potential roles in inducing immune suppression in patients with cancer also during immunotherapy. Here, analyzing the exosomal CD73 isolated and purified by a combination of ultrafiltration and SEC, we observed that patients with melanoma have circulating exosomes expressing CD73 on their surface, which can hydrolyse AMP into adenosine, in line with previous studies. Exosomes from HDs serum also contain enzymatically active CD73 +exosomes.
Among the soluble proteins as peripheral blood biomarkers, the soluble form of PD-L1 has a strong prognostic value in different cancers as demonstrated by many studies.43 Recently, Chen and collaborators have demonstrated that the exosomal form of PD-L1 is markedly associated with response to pembrolizumab in patients with melanoma better than soluble one.10 In line with these studies, we found that the exosomal levels of PD-L1 increased in patients with melanoma who experienced clinical benefits from therapy. Here, we observed that the exosomal CD73 before starting therapy in responders resulted similar to those detected in non-responders. Interestingly, the expression levels of exosomal CD73 increased early during therapy with anti-PD-1 agents only in non-responder patients. It is reasonable to suppose that the enhanced levels of exosomal CD73 in patients with melanoma undergoing anti-PD-1 therapy could contribute to affect the response to therapy. Exosomal CD73 produces adenosine that potently suppresses the leukocytes functions through the activation of A2A adenosine receptor that blocks the early events in the TCR transduction cascade, resulting in a block of proliferation and production of cytokines from T cells, including IFN-γ. Here we confirmed that the IFN-γ production and granzyme B expression in activated T cells were significantly reduced after incubation with exosomes in presence of the CD73 substrate AMP and completely reversed by the CD73 inhibitor APCP. Furthermore, blockade of the adenosine receptor A2A with a selective receptor antagonist abrogated the inhibitory effects of AMP in activated cells incubated with exosomes, proving that CD73 expressed on the exosomes surface, by producing adenosine via its receptor A2A expressed on activated T cells, contributes to suppress the effector functions of these cells. Therefore, exosomes carrying CD73 are a source of adenosine that can affect T-cell functions and thus, indirectly, the therapeutic efficacy of anti-PD-1 agents. Since exosomal CD73 mediates T-cell suppression that can be reversed by a CD73 inhibitor, our results raise the possibility that blocking the exosomal CD73-derived adenosine may be an additional mechanism contributing to the antitumor activities of CD73 therapeutics. However, this intriguing possibility deserves further future investigations. Response to immune checkpoints inhibitors depends on effective T cell response, which in patients with melanoma is related to IFN-γ signature.44 While it is clear that the expression of PD-L1 is upregulated by IFN-γ early during PD-1 blockade therapy in exosomes as well as in their parent cancer cells following reinvigoration of T cells,10 45 46 in non-responders the levels of exosomal PD-L1 remain unchanged, probably as consequence of defects in IFN-γ signaling pathway or in T-cell response.47 48 Our unpublished results indicated that melanoma cells treated with IFN-γ show an upregulation of PD-L1 on their surface (data not shown in this manuscript), in line with previous data,10 while the expression of CD73 remains unchanged compared with control on IFN-γ treatment (data not shown in this manuscript). The potential mechanisms of induced CD73 expression on exosomes in patients who do not have clinical benefits from anti-PD-1 therapy are not clear from our study and require further investigations. It is known that within the tumor microenvironment the hypoxia-inducible factor 1α and inflammatory cytokine signaling can induce the upregulation of CD73, indicating that the expression of CD73 can be modulated during the development and progression of tumor in response to acute or chronic stress. Previous studies have reported that the CD73 expression, within tumor lesions, increased in subsets of patients with melanoma progressing under anti-PD-1 therapy,49 likely as an adaptive resistance mechanism, induced by damage and inflammation caused by immunotherapy.50 Although, these results have not yet been validated in a larger cohort of patients, requiring further investigations, it is clear that the expression of CD73 can change during immunotherapy and identification of the factors or drivers responsible of the regulation of CD73 could be crucial to understand the link between CD73 regulation and clinical outcomes. Our results on patients with melanoma-derived exosomes further suggest the dynamic nature of CD73, whose expression is modulated in patients resistant to therapy, reflecting a mechanism of immune evasion.
Conclusions
In summary, our results confirm the presence of the functional immunosuppressive molecule CD73 on exosomes from serum of patients with melanoma and suggest its potential clinical relevance during anti-PD-1 therapy, highlighting the emerging pivotal role of exosomes in regulating the immune response of patients with cancer. On the other hand, considering the crucial role of CD73 in generating the potent immunosuppressor adenosine, our results reinforce the therapeutic potential of targeting CD73. Although future studies will be necessary to validate these results in a larger patient cohort, and to fully evaluate the effects of exosomes expressing CD73 on systemic antitumor immunity, the assessment of exosomal immune checkpoint CD73 alongside other suppressive molecules might be informative to predict the therapy response and to evaluate potential combinatorial therapeutic strategies to overcome resistance to anti-PD-1 agents.
Acknowledgments
The authors would like to acknowledge Caterina Giraulo and Michele Bifolco for their technical support.
Footnotes
PAA and SM contributed equally.
Contributors: RT, PAA and SM conceived and designed the work; RT, EM, MCM, PDG and SM carried out the experiments; MC, GM, RA, ES and AS contributed to sample preparation; RT, EM, MCM, PDG, PAA and SM contributed to the analysis and interpretation of the results; RT and SM drafted the work; SM is guarantor; all authors provided critical revision of the manuscript for important intellectual content and approved the version to be published.
Funding: This work was supported by FARB 2020 University of Salerno (SM), P.O.R. Campania FESR 2007–2013 - O.O. 2.1 - OCKEY (SM), POR Campania, FESR 2014/2020 (Asse 1—Obiettivo specifico 1.2—Azione 1.2) Project: Campania OncoTerapie CUP: B61G18000470007 and Italian Ministry of Health (IT-MOH) through 'Ricerca Corrente' M2/M (PAA).
Competing interests: PAA has/had a consultant/advisory role for Bristol Myers Squibb, Roche-Genentech, Merck Sharp & Dohme, Novartis, Array, Merck Serono, Pierre-Fabre, Incyte, Medimmune, AstraZeneca, Syndax, Sun Pharma, Sanofi, Idera, Ultimovacs, Sandoz, Immunocore, 4SC, Alkermes, Italfamaco, Nektar, Boehringer Ingelheim, Eisai, Regeneron, Daiichi Sankyo, Pfizer, Oncosec, Nouscom, Takis and Lunaphore. He also received research funding from Bristol Myers Squibb, Roche-Genentech, Array and Sanofi, and travel support from MSD. All outside the submitted work. The other authors declare no potential competing interests.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available in a public, open access repository The raw data of the article is available on the Zenodo open access archive here: https://zenodo.org/record/7669879
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
This study was approved by the Ethics Committee of Istituto Nazionale Tumori—IRCSS—Fondazione 'G. Pascale', Naples, Italy (number 33/17 oss). Participants gave informed consent to participate in the study before taking part.
References
- 1. Marzagalli M, Ebelt ND, Manuel ER. Unraveling the crosstalk between melanoma and immune cells in the tumor microenvironment. Semin Cancer Biol 2019;59:236–50. 10.1016/j.semcancer.2019.08.002 [DOI] [PubMed] [Google Scholar]
- 2. Carlino MS, Larkin J, Long GV. Immune checkpoint inhibitors in melanoma. Lancet 2021;398:1002–14. 10.1016/S0140-6736(21)01206-X [DOI] [PubMed] [Google Scholar]
- 3. Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Five-Year survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med 2019;381:1535–46. 10.1056/NEJMoa1910836 [DOI] [PubMed] [Google Scholar]
- 4. Kalluri R. The biology and function of exosomes in cancer. J Clin Invest 2016;126:1208–15. 10.1172/JCI81135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Clayton A, Mason MD. Exosomes in tumour immunity. Curr Oncol 2009;16:46–9. 10.3747/co.v16i3.367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Ribeiro MF, Zhu H, Millard RW, et al. Exosomes function in pro- and anti-angiogenesis. Curr Angiogenes 2013;2:54–9. 10.2174/22115528113020020001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Logozzi M, De Milito A, Lugini L, et al. High levels of exosomes expressing CD63 and caveolin-1 in plasma of melanoma patients. PLoS One 2009;4:e5219. 10.1371/journal.pone.0005219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Peinado H, Alečković M, Lavotshkin S, et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through Met. Nat Med 2012;18:883–91. 10.1038/nm.2753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Pfeffer SR, Grossmann KF, Cassidy PB, et al. Detection of exosomal miRNAs in the plasma of melanoma patients. J Clin Med 2015;4:2012–27. 10.3390/jcm4121957 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Chen G, Huang AC, Zhang W, et al. Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature 2018;560:382–6. 10.1038/s41586-018-0392-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Daassi D, Mahoney KM, Freeman GJ. The importance of exosomal PDL1 in tumour immune evasion. Nat Rev Immunol 2020;20:209–15. 10.1038/s41577-019-0264-y [DOI] [PubMed] [Google Scholar]
- 12. Allard B, Allard D, Buisseret L, et al. The adenosine pathway in immuno-oncology. Nat Rev Clin Oncol 2020;17:611–29. 10.1038/s41571-020-0382-2 [DOI] [PubMed] [Google Scholar]
- 13. Yegutkin GG. Nucleotide- and nucleoside-converting ectoenzymes: important modulators of purinergic signalling cascade. Biochim Biophys Acta 2008;1783:673–94. 10.1016/j.bbamcr.2008.01.024 [DOI] [PubMed] [Google Scholar]
- 14. Horenstein AL, Chillemi A, Zaccarello G, et al. A CD38/CD203a/CD73 ectoenzymatic pathway independent of CD39 drives a novel adenosinergic loop in human T lymphocytes. Oncoimmunology 2013;2:e26246. 10.4161/onci.26246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Thompson EA, Powell JD. Inhibition of the adenosine pathway to potentiate cancer immunotherapy: potential for combinatorial approaches. Annu Rev Med 2021;72:6.1–6.18. 10.1146/annurev-med-060619-023155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Overman MJ, LoRusso P, Strickler JH, et al. Safety, efficacy and pharmacodynamics (PD) of MEDI9447 (oleclumab) alone or in combination with durvalumab in advanced colorectal cancer (CRC) or pancreatic cancer (panc). JCO 2018;36:4123–23. 10.1200/JCO.2018.36.15_suppl.4123 [DOI] [Google Scholar]
- 17. Martinez-Marti A, Majem M, Barlesi F, et al. LBA42 coast: an open-label, randomised, phase II platform study of durvalumab alone or in combination with novel agents in patients with locally advanced, unresectable, stage III NSCLC. Annals of Oncology 2021;32:S1320. 10.1016/j.annonc.2021.08.2121 [DOI] [Google Scholar]
- 18. Turiello R, Pinto A, Morello S. Cd73: a promising biomarker in cancer patients. Front Pharmacol 2020;11:11. 10.3389/fphar.2020.609931 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Schneider E, Rissiek A, Winzer R, et al. Generation and function of Non-cell-bound CD73 in inflammation. Front Immunol 2019;10:10. 10.3389/fimmu.2019.01729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Morello S, Capone M, Sorrentino C, et al. Soluble CD73 as biomarker in patients with metastatic melanoma patients treated with nivolumab. J Transl Med 2017;15:244–44. 10.1186/s12967-017-1348-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Turiello R, Capone M, Giannarelli D, et al. Serum CD73 is a prognostic factor in patients with metastatic melanoma and is associated with response to anti-PD-1 therapy. J Immunother Cancer 2020;8:e001689. 10.1136/jitc-2020-001689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Muller L, Hong C-S, Stolz DB, et al. Isolation of biologically-active exosomes from human plasma. J Immunol Methods 2014;411:55–65. 10.1016/j.jim.2014.06.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Schuler PJ, Saze Z, Hong C-S, et al. Human CD4+ CD39+ regulatory T cells produce adenosine upon co-expression of surface CD73 or contact with CD73+ exosomes or CD73+ cells. Clin Exp Immunol 2014;177:531–43. 10.1111/cei.12354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Theodoraki M-N, Hoffmann TK, Jackson EK, et al. Exosomes in HNSCC plasma as surrogate markers of tumour progression and immune competence. Clin Exp Immunol 2018;194:67–78. 10.1111/cei.13157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Clayton A, Al-Taei S, Webber J, et al. Cancer exosomes express CD39 and CD73, which suppress T cells through adenosine production. J Immunol 2011;187:676–83. 10.4049/jimmunol.1003884 [DOI] [PubMed] [Google Scholar]
- 26. Smyth LA, Ratnasothy K, Tsang JYS, et al. CD73 expression on extracellular vesicles derived from CD4+ CD25+ Foxp3+ T cells contributes to their regulatory function. Eur J Immunol 2013;43:2430–40. 10.1002/eji.201242909 [DOI] [PubMed] [Google Scholar]
- 27. Zhang F, Li R, Yang Y, et al. Specific decrease in B-cell-derived extracellular vesicles enhances post-chemotherapeutic CD8+ T cell responses. Immunity 2019;50:738–50. 10.1016/j.immuni.2019.01.010 [DOI] [PubMed] [Google Scholar]
- 28. Ludwig N, Yerneni SS, Azambuja JH, et al. Tumor-derived exosomes promote angiogenesis via adenosine A2B receptor signaling. Angiogenesis 2020;23:599–610. 10.1007/s10456-020-09728-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Sharma P, Diergaarde B, Ferrone S, et al. Melanoma cell-derived exosomes in plasma of melanoma patients suppress functions of immune effector cells. Sci Rep 2020;10:92. 10.1038/s41598-019-56542-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Schneider E, Winzer R, Rissiek A, et al. CD73-mediated adenosine production by CD8 T cell-derived extracellular vesicles constitutes an intrinsic mechanism of immune suppression. Nat Commun 2021;12:5911. 10.1038/s41467-021-26134-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Ludwig S, Floros T, Theodoraki M-N, et al. Suppression of lymphocyte functions by plasma exosomes correlates with disease activity in patients with head and neck cancer. Clin Cancer Res 2017;23:4843–54. 10.1158/1078-0432.CCR-16-2819 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Hong C-S, Funk S, Whiteside TL. Isolation of biologically active exosomes from plasma of patients with cancer. Methods Mol Biol 2017;1633:257–65. 10.1007/978-1-4939-7142-8_16 [DOI] [PubMed] [Google Scholar]
- 33. Sharma P, Ludwig S, Muller L, et al. Immunoaffinity-Based isolation of melanoma cell-derived exosomes from plasma of patients with melanoma. J Extracell Vesicles 2018;7:1435138. 10.1080/20013078.2018.1435138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Shu SL, Yang Y, Allen CL, et al. Purity and yield of melanoma exosomes are dependent on isolation method. J Extracell Vesicles 2020;9:1692401. 10.1080/20013078.2019.1692401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Théry C, Zitvogel L, Amigorena S. Exosomes: composition, biogenesis and function. Nat Rev Immunol 2002;2:569–79. 10.1038/nri855 [DOI] [PubMed] [Google Scholar]
- 36. Vijayan D, Young A, Teng MWL, et al. Targeting immunosuppressive adenosine in cancer. Nat Rev Cancer 2017;17:709–24. 10.1038/nrc.2017.86 [DOI] [PubMed] [Google Scholar]
- 37. Sorrentino C, Hossain F, Rodriguez PC, et al. Adenosine A2A receptor stimulation inhibits TCR-induced Notch1 activation in CD8+T-Cells. Front Immunol 2019;10:162–62. 10.3389/fimmu.2019.00162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Pilard C, Ancion M, Delvenne P, et al. Cancer immunotherapy: it's time to better predict patients' response. Br J Cancer 2021;125:927–38. 10.1038/s41416-021-01413-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Sharma P, Hu-Lieskovan S, Wargo JA, et al. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell 2017;168:707–23. 10.1016/j.cell.2017.01.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Nowicki TS, Hu-Lieskovan S, Ribas A. Mechanisms of resistance to PD-1 and PD-L1 blockade. Cancer J 2018;24:47–53. 10.1097/PPO.0000000000000303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Ohta A, Gorelik E, Prasad SJ, et al. A2A adenosine receptor protects tumors from antitumor T cells. Proc Natl Acad Sci U S A 2006;103:13132–7. 10.1073/pnas.0605251103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Vigano S, Alatzoglou D, Irving M, et al. Targeting adenosine in cancer immunotherapy to enhance T-cell function. Front Immunol 2019;10:925. 10.3389/fimmu.2019.00925 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Bailly C, Thuru X, Quesnel B. Soluble programmed death ligand-1 (sPD-L1): a pool of circulating proteins implicated in health and diseases. Cancers 2021;13:3034. 10.3390/cancers13123034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Grasso CS, Tsoi J, Onyshchenko M, et al. Conserved interferon-γ signaling drives clinical response to immune checkpoint blockade therapy in melanoma. Cancer Cell 2020;38:500–15. 10.1016/j.ccell.2020.08.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Garcia-Diaz A, Shin DS, Moreno BH, et al. Interferon receptor signaling pathways regulating PD-L1 and PD-L2 expression. Cell Rep 2017;19:1189–201. 10.1016/j.celrep.2017.04.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Poggio M, Hu T, Pai C-C, et al. Suppression of exosomal PD-L1 induces systemic anti-tumor immunity and memory. Cell 2019;177:414–27. 10.1016/j.cell.2019.02.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Zaretsky JM, Garcia-Diaz A, Shin DS, et al. Mutations associated with acquired resistance to PD-1 blockade in melanoma. N Engl J Med 2016;375:819–29. 10.1056/NEJMoa1604958 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Ni L, Lu J. Interferon gamma in cancer immunotherapy. Cancer Med 2018;7:4509–16. 10.1002/cam4.1700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Reinhardt J, Landsberg J, Schmid-Burgk JL, et al. Mapk signaling and inflammation link melanoma phenotype switching to induction of CD73 during immunotherapy. Cancer Res 2017;77:4697–709. 10.1158/0008-5472.CAN-17-0395 [DOI] [PubMed] [Google Scholar]
- 50. Hölzel M, Bovier A, Tüting T. Plasticity of tumour and immune cells: a source of heterogeneity and a cause for therapy resistance? Nat Rev Cancer 2013;13:365–76. 10.1038/nrc3498 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jitc-2021-004043supp001.pdf (17.3KB, pdf)
jitc-2021-004043supp002.pdf (41.6KB, pdf)
Data Availability Statement
Data are available in a public, open access repository The raw data of the article is available on the Zenodo open access archive here: https://zenodo.org/record/7669879