Keywords: aging, BH2, FMD, shear rate, vascular function
Abstract
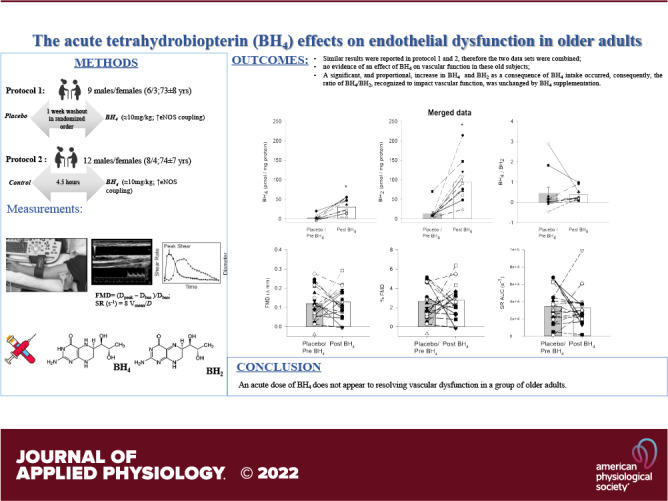
As a deficiency in tetrahydrobiopterin (BH4), a cofactor for endothelial nitric oxide synthase, has been implicated in the age-related decline in vascular function, this study aimed to determine the impact of acute BH4 supplementation on flow-mediated vasodilation (FMD) in old adults. Two approaches were used: 1) A multiday, double-blind, placebo-controlled, crossover design measuring, FMD [ΔFMD (mm), %FMD (%)] and shear rate area under the curve (SR AUC) in nine old subjects (73 ± 8 yr) with either placebo (placebo) or BH4 (≈10 mg/kg, post), and 2) a single experimental day measuring FMD in an additional 13 old subjects (74 ± 7 yr) prior to (pre) and 4.5 h after ingesting BH4 (≈10 mg/kg). With the first experimental approach, acute BH4 intake did not significantly alter FMD (ΔFMD: 0.17 ± 0.03 vs. 0.13 ± 0.02 mm; %FMD: 3.3 ± 0.61 vs. 2.9 ± 0.4%) or SR AUC (30,280 ± 4,428 vs. 37,877 ± 9,241 s−1) compared with placebo. Similarly, with the second approach, BH4 did not significantly alter FMD (ΔFMD: 0.09 ± 0.02 vs. 0.12 ± 0.03 mm; %FMD: 2.2 ± 0.6 vs. 2.9 ± 0.6%) or SR AUC (37,588 ± 6,753 vs. 28,996 ± 3,735 s−1) compared with pre. Moreover, when the two data sets were combined, resulting in a greater sample size, there was still no evidence of an effect of BH4 on vascular function in these old subjects. Importantly, both plasma BH4 and 7,8-dihydrobiopterin (BH2), the oxidized form of BH4, increased significantly with acute BH4 supplementation. Consequently, the ratio of BH4/BH2, recognized to impact vascular function, was unchanged. Thus, acute BH4 supplementation does not correct vascular dysfunction in the old.
NEW & NOTEWORTHY Despite two different experimental approaches, acute BH4 supplementation did not affect vascular function in older adults, as measured by flow-mediated vasodilation. Plasma levels of both BH4 and BH2, the BH4 oxidized form, significantly increased after acute BH4 supplementation, resulting in an unchanged ratio of BH4/BH2, a key determining factor for endothelial nitric oxide synthase coupling. Therefore, likely due to the elevated oxidative stress with advancing age, acute BH4 supplementation does not correct vascular dysfunction in the old.
INTRODUCTION
Globally, the number of old adults (>65 yr of age) is expected to double in the next several decades (1, 2), greatly increasing the prevalence of the elderly. With this demographic change, there is a growing need to improve the quality of life (3) and prevent adverse events in the older population. Indeed, aging is a major risk factor for developing both cardiovascular disease-related morbidity and mortality (4). Two principal and interconnected mechanisms are proposed as the biological contributors to the multifaceted process of aging, these are chronic, low-grade, inflammation, and increased cellular oxidative stress (5). Considerable evidence suggests that either of these two factors can lead to endothelial dysfunction (6, 7), a well-established early marker of cardiovascular disease (8, 9). Indeed, a recently proposed model, known as the “vascular health triad” has combined these phenomena to help better explain the vascular aging process (10). According to this model, inflammation-induced oxidative stress impairs endothelial function, through an attenuation of nitric oxide (NO) bioavailability (10).
Tetrahydrobiopterin (BH4), in addition to being an essential cofactor for the enzymes used in the degradation of the amino acid phenylalanine and the biosynthesis of many neurotransmitters, plays an important role in NO bioavailability and, therefore, NO-mediated vasodilation (11, 12). Indeed, BH4 appears to have multiple roles in NO-catalysis (11, 13–15), not only stabilizing the active conformation of the enzyme endothelial nitric oxide synthase (eNOS) (11, 14, 16) but also acting as an allosteric effector, enhancing substrate binding (11, 14, 16). Thereby, coupling substrate oxidation to NADPH consumption and lowering the ratio of superoxide to NO production (11, 13–15, 17). In addition, although predominantly from in vitro studies, BH4 has been reported to be a potent antioxidant. However, as a scavenger of oxygen-derived free radicals (18), high levels of oxidative stress provoke BH4 oxidation to 7,8 dihydrobiopterin (BH2) and the, subsequent, downregulation of eNOS activity (17). Furthermore, in vivo findings have suggested that a BH4 deficit results in eNOS uncoupling, implying a concurrent decrease in NO synthesis and an increase in the production of reactive oxygen species (i.e., superoxide anions) (18, 19). Thus, BH4 may be of major importance in the pathogenesis of endothelial dysfunction both as a consequence of NO bioavailability and the attenuation of oxidative vascular injury (11, 13–15).
In humans, there is considerable evidence that an acute BH4 supplementation can improve vascular function. Specifically, the beneficial effects of BH4 on endothelial function, assessed by flow-mediated dilation (FMD), have been documented in patients with type II diabetes, chronic obstructive pulmonary disease (COPD), hypercholesterolemia, and heart failure (20–24). With respect to aging, acetylcholine-induced increases in arm blood flow, across a spectrum of age (19–81 yr), were inversely related to age and were improved by BH4 infusion in an age-related manner (25). The BH4 augmented, acetylcholine-induced, blood flow, and the relationship with age were ablated by the coinfusion of NG-monomethyl-l-arginine, an NO synthase inhibitor, supporting the link between BH4, NOS activity, and aging (25). Furthermore, Eskurza et al. (26) documented a BH4-induced improvement in endothelial-dependent vasodilation in “relatively young” older subjects (62 ± 2 yr) who were sedentary, but, interestingly, not in their habitually exercise-trained, age-matched, counterparts. However, with an ever-growing life expectancy (1, 2), a better understanding of the impact of BH4 on vascular function in subjects more representative of the old population (≥65 yr) is warranted.
Therefore, this study sought to determine the impact of acute BH4 supplementation on vascular function, measured by FMD, in a group of relatively healthy, but not exercise-trained, old subjects (≥65 yr). We hypothesized that, even in the absence of other cardiovascular risk factors, vascular dysfunction in old adults would be attenuated as a result of acute BH4 supplementation.
MATERIALS AND METHODS
Participants
A total of 21 healthy participants took part in this investigation, which consisted of two protocols. The first protocol, modeled after a study of Eskurza et al., (26) included nine subjects, while the second protocol included 12 subjects. Subjects only participated in a single protocol. A preliminary visit included a self-reported medical history, blood drawn from the antecubital vein, and a prescreening FMD test. According to the literature, FMD values ≥5% are atypical in this target population (6), therefore higher than a 5% FMD value was included in the exclusion criteria. Other exclusion criteria were under the age of 65 yr, habitual exercise training, a history of cardiovascular or cerebrovascular disease, hypertension, hypercholesterolemia, diabetes mellitus, liver disease, renal disease, and being a current or former smoker. All females were postmenopausal and not currently taking hormone replacement therapy. Participants abstained from alcohol, caffeine, dietary supplements, and exercise for more than 12 h before laboratory visits (27). All procedures were approved by the University of Utah and Salt Lake City Veteran Affairs (VA) Medical Center Institutional Review Board and studies were performed at the VA Salt Lake City, Utah Vascular Research Laboratory (UVRL), under the principles outlined in the Declaration of Helsinki. Written informed consent was obtained before participation after an explanation of the nature, benefits, and risks of the study.
Study Design
Protocol 1.
For this protocol, we recruited nine subjects (73 ± 8 yr; 6/3, M/F) who went to a multiday, double-blind (investigators and subjects were blinded to the condition), randomized, crossover experimental design with two conditions, placebo (placebo) and BH4 (post). Randomization for the order of the condition (PLACEBO/BH4) was defined by an automatic generator, i.e., randomizer.org. On the two experimental days, the participants reported to the laboratory in the morning, having consumed a, provided, standardized low-fat breakfast and the oral BH4 [single dose of sapropterin dihydrochloride, 10 mg × kg−1, Kuvan (13), BioMarin Pharmaceutical Inc., Novato, CA] or placebo, independently, 4.5 h before their arrival at the UVRL. There was a washout period, of at least 5 days, before crossing over to the alternate condition. Experimental procedures consisted of supine blood pressure assessments and a blood draw followed by an FMD test on the contralateral (right) arm. All measurements were performed in quiet, comfortable conditions, and at the ambient temperature (∼22°C) of the laboratory. Of note, this protocol was modeled after a similar study of Eskurza et al. (26), where a similar dose of BH4 and a similar sample size were used.
Protocol 2.
Twelve participants (74 ± 7 yr; 8/4, M/F) were recruited for this protocol performed during a single-extended laboratory visit to better control the period of time following the BH4 intake and the, potentially, confounding environmental and physiological variations associated with assessments made on two different days in Protocol 1. During the single experimental visit, subjects arrived at the UVRL in the morning having consumed a, previously provided, low-fat standardized breakfast, and baseline measurements (pre) were taken. Subsequently, having consumed a second standardized breakfast and the oral BH4 (single dose of sapropterin dihydrochloride, 10 mg/kg, Kuvan (13), BioMarin Pharmaceutical Inc., Novato, CA), the subjects were retested 4.5 h post acute BH4 supplementation (post). During the 4.5 h break, between pre and post BH4 assessments, participants were kept under observation by a member of the research team. Every hour participants walked slowly around the laboratory to prevent the confounding effects of inactivity on vascular function (28). Experimental measurements consisted of supine blood pressure assessments and a blood draw followed by an FMD test performed on the contralateral (right) arm. All measurements were performed in quiet, comfortable conditions, and at the ambient temperature (∼22°C) of the laboratory.
Measurements and Data Analysis
BH4 and BH2.
Measurements of BH4 and BH2 were performed in a subgroup of subjects in both Protocol 1 and Protocol 2. Blood was collected in EDTA plasma tubes containing 1 mM dithioerythritol (DTE) in 0.9% saline to yield a final concentration of 0.1 mM DTE. The tubes were centrifuged at 3,000 rpm for 15 min at 4°C, and the plasma was collected and stored at −80°C. Plasma was thawed and extracted using differential oxidation with iodine, as previously described by Fukushima and Nixon (50), enabling the measurement of total biopterin and BH4. Under acidic conditions, BH4 and BH2 are oxidized to biopterin, whereas under alkaline conditions only BH2 is oxidized to biopterin and BH4 undergoes side-chain cleavage to form pterin. The actual BH4 levels can be determined by calculating the difference in biopterin content between the two oxidations. Prior to iodine oxidation, the plasma was deproteinized by adding 250 µL of 1 M trichloroacetic acid to 1 mL plasma. This solution was incubated in the dark at 4°C for 15 min and centrifuged at 20,000 g for 15 min at 4°C. High-performance liquid chromatography of biopterin was performed on an Acuity Arc system with a CORTECS C18, 2.7 µm column (4.6 × 150 mm), with a Vanguard precolumn, at a column temperature of 40°C. All equipment and supplies were from Waters Corporation (Milford, MA). Samples were run isocratically with 15 mM potassium phosphate buffer, pH 6.4 at a flow rate of 0.8 mL/min. A Waters 2475 Multi λ Fluorescence Detector was used to detect biopterin with excitation set to 350 nm and an emission setting of 440 nm.
Blood Pressure
Resting blood pressure measurements were performed on the upper arm, using a semiautomated blood pressure measuring device (Tango+, SunTech, Morrisville, NC), in triplicate, after 5 min in the upright seated position and after 20 min in the supine position.
Flow-Mediated Dilation
The brachial artery FMD test was performed following current guidelines (27, 29, 30). Specifically, subjects rested in a supine position for ≈20 min before FMD testing with a rapid inflation cuff (D.E. Hokanson Inc., Bellevue, WA) placed around the right forearm, immediately distal to the medial epicondyle. After 30 s of baseline data acquisition, after which the cuff was inflated to 250 mmHg for 5 min, then the cuff was rapidly deflated, and data were continuously acquired for 2 min. Continuous brachial artery diameter and blood velocity measurements were simultaneously obtained during all data acquisition periods with Doppler ultrasound (Duplex mode), with an imaging frequency of 15 MHz and Doppler frequency of 5 MHz (LOGIQ E9, GE Healthcare, Chicago, IL). The diameter measurements were made perpendicular to the long axis of the artery during diastole using a computer-based image analysis system (Brachial Artery Analyzer for Research, Medical Imaging Applications, LLC, Coralville, IA), which is largely independent of investigator bias. FMD was quantified as the maximal change in artery diameter after cuff release, expressed as absolute FMD, as well as a percentage increase above baseline (%FMD). Brachial artery shear rate (SR) was calculated post cuff release using the following equation:
SR (s−1) = 8 Vmean/D
where Vmean is average blood velocity and D is brachial artery diameter. The cumulative SR, corresponding to the reactive hyperemia post cuff release [total area under the curve (AUC) for SR from cuff release to time-to-peak, SR AUC], was also calculated as follows:
As FMD is primarily dependent on the endothelial-mediated response to the SR stimulus, both the FMD [ΔFMD (mm) and %FMD (%)] were normalized for SR AUC (FMD/SR AUC and %FMD/SR AUC) (31) to evaluate the sensitivity to this stimulus. Brachial artery blood flow (BF) was also calculated using the artery diameter (D) during diastole, and the mean blood velocity as:
BF (mL × min−1) = Vmean × π × (vessel D/2)2 × 60.
Cumulative BF AUC (mL) was integrated across the 2 min following cuff release.
Inflammation, Oxidative Stress, Free Radicals, and Antioxidant Capacity Assays
In Protocol 2, additional blood samples were obtained from the antecubital vein, pre and post acute BH4 supplementation, to assess inflammation, oxidative stress, free radicals (also assayed in Protocol 1), and antioxidant capacity. For these assays, serum, plasma, or whole blood samples were stored at −80°C until analysis. Systemic inflammation was assessed by measuring IL-6 and TNF-α in serum using a high-sensitivity enzyme-linked immunosorbent assay (32) (R&D Systems, Minneapolis, MN). Oxidative stress was assessed by quantifying plasma malondialdehyde (MDA) using colorimetry (Oxis Research/Percipio Bioscience, Foster City, CA) and protein carbonyl levels using a high-sensitivity enzyme-linked immunosorbent assay (Northwest Life Science Specialties, LLC Vancouver, WA). Endogenous antioxidant capacity in plasma, assessed by superoxide dismutase (SOD) and catalase activity, was assayed using colorimetry (33) (Cayman Chemical Company, Ann Arbor, MI). In addition, total antioxidant capacity was assessed by measuring the ferric reducing ability of plasma (FRAP), using the method described by Benzie and Strain (34). Electron paramagnetic resonance (EPR) spectroscopy was performed on whole blood samples to directly assess the concentration of free radicals, as previously described (2). Briefly, 1.0 mL of venous blood was collected into a vacutainer containing 0.5 mL of the spin trap α-phenyl-tert-butyl nitrone (PBN) (0.0140 mol/L). The PBN adduct was snap-frozen in liquid nitrogen and stored at −80°C. After thawing, the PBN adduct (1,000 μL) was pipetted into a glass tube and extracted with 500 µL toluene. After centrifugation, 300 µL of the toluene/PBN extract was pipetted into a precision-bore quartz EPR sample tube (Wilmad, Vineland, NJ). EPR spectroscopy was then performed at 21°C using an EMX X-band spectrometer (Bruker, Billerica, MA) and commercially available software (version 1.1 b.51, Bruker Xenon System), which was also used to calculate the area under the curve of the EPR spectroscopy signal by double integration.
Statistical Analyses
Statistical analyses were performed using commercially available software (SigmaStat version 3.13, Systat Software, RRID:SCR_010285). Normality of the data was tested with the Shapiro–Wilk test. To examine the effects of acute BH4 supplementation within each protocol, a paired Student’s t test was used. Across the two protocols, the effect of BH4 was assessed with unpaired Student’s t tests. Subject characteristics at baseline, across the two protocols, were assessed with unpaired Student’s t tests. Of note, as there were, predominantly, no differences in subject characteristics between Protocol 1 and 2 and there were no differences in the results of the two protocols (unpaired Student’s t tests), the data were combined for further analyses. In addition, to determine the effect of sex on the potential effect of the BH4 treatment, with the merged data set, a two-way ANOVA (treatment × sex) was performed on the FMD (% and Δ), the SR AUC, and the BF AUC data. A priori power analysis, based on a previous study (26), using G*Power version 3.1.9.7 (RRID:SCR_013726) (35), revealed that to ensure a statistical power ≥ 0.90, with significance set at 5%, a sample size of nine participants would be required. Subject characteristics are expressed as means ± SD, and all other data are expressed as means ± SE. Significance was established at P < 0.05 for all tests.
RESULTS
Subject Characteristics
The age range of the 21 subjects who took part in this study was 65–80 yr and 14 were male and seven were female. Protocol 1 included nine subjects (73 ± 8 yr; 6/3, M/F), whereas the second protocol included 12 subjects of similar age and gender mix (74 ± 7 yr; 8/4, M/F). Of note, although subjects were excluded during preliminary recruitment due to health conditions, no potential subjects were excluded following the preliminary FMD screening due to a >5% FMD. Subject characteristics are documented in Table 1. Subject characteristics for Protocol 1 and Protocol 2 did not differ in terms of age, height, weight, BMI, and BH4 dose administered. All subjects were normotensive and mean arterial blood pressure (MAP) did not differ between the two protocols (MAP: 94 ± 10 vs. 91 ± 5 mmHg, Protocol 1 and 2, respectively). Although the blood count, lipid panel, glucose panel, and estimated glomerular filtration rate (eGFR) were well within the normal range and not different between protocols, by chance, subjects in Protocol 2 exhibited a significantly lower total cholesterol than those that partook in Protocol 1 (187 ± 24 and 158 ± 30 mg/dL, Protocol 1 and 2, respectively; P = 0.03).
Table 1.
Subject characteristics
| Protocol 1 | Protocol 2 | P Values | Merged Data | |
|---|---|---|---|---|
| Subject, n (M/F) | 9 (6/3) | 12 (8/4) | – | 21 (14/7) |
| Age, yrs | 73 ± 8 | 74 ± 7 | 0.98 | 73 ± 7 |
| Height, cm | 173 ± 11 | 170 ± 9 | 0.42 | 171 ± 9 |
| Mass, kg | 72 ± 18 | 78 ± 21 | 0.73 | 76 ± 20 |
| BMI, kg/m2 | 24 ± 4 | 27 ± 5 | 0.33 | 26 ± 5 |
| MAP, mmHg | 94 ± 10 | 91 ± 5 | 0.1 | 92 ± 4 |
| BH4 dose, mg/kg | 10.1 ± 0.4 | 9.9 ± 0.4 | 0.38 | 10 ± 0.04 |
| Total cholesterol, mg/dL | 187 ± 24 | 158 ± 30 | 0.03 | 170 ± 31 |
| LDL, mg/dL | 100 ± 14 | 92 ± 23 | 0.33 | 95 ± 20 |
| HDL, mg/dL | 61 ± 16 | 50 ± 15 | 0.12 | 55 ± 16 |
| CHOL/HDL | 3 ± 1 | 3 ± 1 | 0.86 | 3 ± 1 |
| Triglycerides, mg/dL | 106 ± 47 | 94 ± 47 | 0.58 | 99 ± 46 |
| Plasma glucose, mg/dL | 85 ± 14 | 101 ± 35 | 0.23 | 94 ± 28 |
| Hemoglobin, g/dL | 14.6 ± 1.3 | 14.1 ± 1.4 | 0.41 | 14.3 ± 1.3 |
| HCT, % | 43.5 ± 1.3 | 41.9 ± 4.8 | 0.38 | 42.6 ± 4.2 |
| eGFR, mL/min/1.73m2 | 71 ± 13 | 72 ± 13 | 0.87 | 71 ± 12 |
BH4, tetrahydrobiopterin; BMI, body mass index; eGFR, estimated glomerular filtration rate; HCT, hematocrit; HDL, high-density lipoprotein; LDL, low-density lipoprotein; MAP, mean arterial pressure (upright seated). P values, statistical comparison between Protocol 1 and Protocol 2. Differences between protocols were tested with unpaired Student’s t tests. Data presented as means ± SD.
Tetrahydrobiopterin and 7,8 Dihydrobiopterin
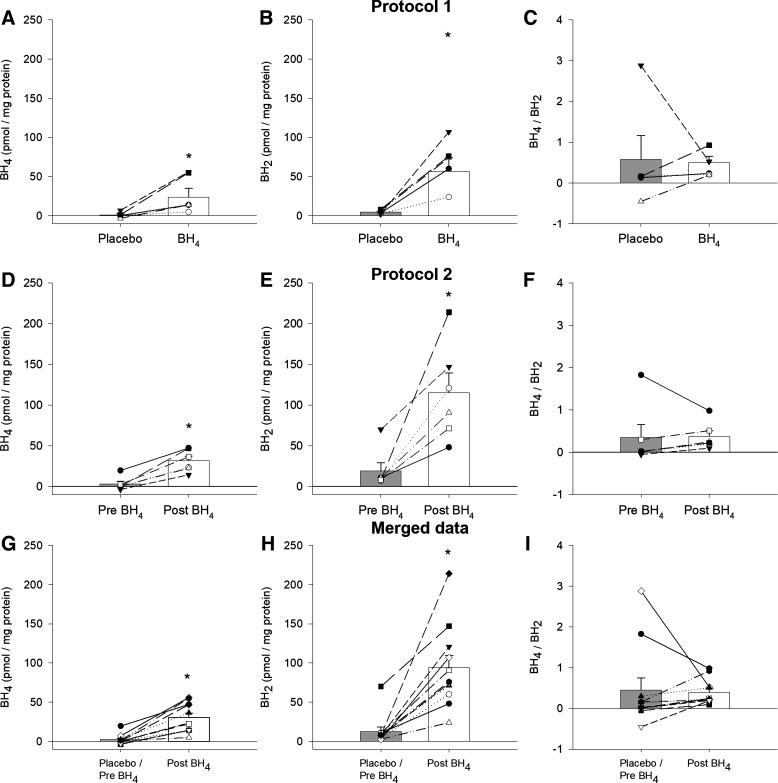
Plasma levels of BH4, BH2, and the ratio of BH4/BH2 for a subset of subjects who took part in Protocol 1 (n = 5/9), Protocol 2 (n = 6/12), and the merged data from these two protocols (n = 11/21), are illustrated in Fig. 1. In both Protocol 1 and 2, the acute BH4 supplementation significantly raised plasma BH4 levels (Protocol 1: 1.22 ± 1.63 vs. 28.80 ± 11.17 pmol, P = 0.05; Protocol 2: 3.09 ± 3.39 vs. 31.71 ± 5.64 pmol, P = 0.001, Fig. 1, A and D, respectively). Similarly, this single acute dose of BH4 significantly increased BH2 in both protocols (Protocol 1: 4.76 ± 1.18 vs. 56.83 ± 17.31 pmol, P = 0.009; Protocol 2: 18.94 ± 10.22 vs. 115.33 ± 24.40 pmol, P = 0.011, Fig. 1, B and E, respectively). Due to the, relatively, proportional increase in both BH4 and BH2, following acute BH4 supplementation, the ratio of BH4/BH2 was unaltered (Protocol 1: 0.57 ± 0.58 vs. 0.49 ± 0.15, P = 0.793; Protocol 2: 0.35 ± 0.29 vs. 0.37 ± 0.13, P = 0.905, Fig. 1, C and F, respectively). When the BH2 and BH4 data from Protocol 1 and 2 were merged, both significantly increased, again, leaving the ratio of the two unaltered (BH4: 2.24 ± 1.92 vs. 30.38 ± 5.56 pmol, P ≤ 0.001, BH2: 12.50 ± 5.80 vs. 93.90 ± 15.82 pmol, P ≤ 0.001, BH4/BH2: 0.45 ± 0.29 vs. 0.39 ± 0.09, P = 0.82, Fig. 1, G–I, respectively).
Figure 1.
Plasma concentrations of tetrahydrobiopterin, BH4 (A, D, and G), 7, 8-dihydrobiopterin, BH2 (B, E, and H), and the BH4/BH2 ratio (C, F, and I) assessed in Protocol 1, Protocol 2, and data from both protocols merged, merged data. Initial plasma concentrations prior to acute BH4 supplementation in Protocol 1, Protocol 2, and for the merged data are labeled as placebo/pre BH4 while 4.5 h after acute BH4 supplementation (10 mg/kg), plasma concentrations are labeled as post BH4. Figure 1 sample sizes are 5 (3 M/2F), 6 (4 M/2F), and 11 for Protocol 1, Protocol 2, and the merged data, respectively. The effect of BH4 supplementation, within each protocol, was tested with paired Student’s t test. Data are presented as means ± SE. *P< 0.05 different from placebo, pre, and placebo/pre.
Blood Pressure
Supine systolic and diastolic blood pressure data are documented in Table 2. Neither systolic nor diastolic blood pressure values were affected by the acute BH4 supplementation in Protocol 1 or Protocol 2, or when these blood pressure data were merged (Table 2).
Table 2.
Blood pressure and brachial artery flow-mediated dilation measures before and after acute tetrahydrobiopterin supplementation
|
Protocol 1
|
Protocol 2
|
Merged Data |
|||||
|---|---|---|---|---|---|---|---|
| Placebo | Post | Pre | Post | ΔMean P Value | Placebo/Pre | Post | |
| Systolic BP, mmHg | 123 ± 3 | 128 ± 2 | 122 ± 4 | 124 ± 2 | 0.78 | 122 ± 2 | 125 ± 3 |
| Diastolic BP, mmHg | 79 ± 2 | 80 ± 2 | 76 ± 2 | 73 ± 3 | 0.78 | 77 ± 3 | 76 ± 3 |
| Baseline diameter, mm | 4.64 ± 0.28 | 4.60 ± 0.29 | 4.68 ± 0.30 | 4.34 ± 0.35 | 0.42 | 4.66 ± 0.21 | 4.45 ± 0.23 |
| Peak diameter, mm | 4.81 ± 0.29 | 4.73 ± 0.30 | 4.77 ± 0.29 | 4.46 ± 0.36 | 0.60 | 4.78 ± 0.20 | 4.58 ± 0.24 |
| FMD, mm | 0.17 ± 0.03 | 0.13 ± 0.02 | 0.09 ± 0.02 | 0.12 ± 0.03 | 0.11 | 0.12 ± 0.02 | 0.13 ± 0.02 |
| %FMD, % | 3.25 ± 0.61 | 2.86 ± 0.42 | 2.17 ± 0.64 | 2.69 ± 0.59 | 0.28 | 2.64 ± 0.46 | 2.77 ± 0.38 |
| Time to peak, s | 78 ± 10 | 79 ± 12 | 63 ± 9 | 58 ± 11 | 0.68 | 69 ± 7 | 67 ± 8 |
| SR AUC, s−1 | 30280 ± 4428 | 37877 ± 9241 | 37588 ± 6753 | 28996 ± 3735 | 0.20 | 34456 ± 4284 | 32802 ± 4471 |
| FMD/SR AUC × 104, Δmm/s−1 | 0.10 ± 0.05 | 0.11 ± 0.05 | 0.05 ± 0.02 | 0.04 ± 0.01 | 0.43 | 0.07 ± 0.02 | 0.07 ± 0.02 |
| % FMD/SR AUC, %/s−1 | 0.21 ± 0.11 | 0.21 ± 0.12 | 0.11 ± 0.05 | 0.10 ± 0.02 | 1.00 | 0.15 ± 0.05 | 0.14 ± 0.05 |
| BF AUC, mL | 396 ± 74 | 413 ± 65 | 516 ± 115 | 377 ± 81 | 0.12 | 465 ± 72 | 392 ± 53 |
AUC, area under the curve; BF, blood flow; BH4, tetrahydrobiopterin; BP, blood pressure (supine); FMD, flow-mediated dilation; Placebo, pre, and placebo/pre, data collected without BH4 supplementation; post, data collected 4.5 h after acute BH4 supplementation (10 mg/kg); SR, shear rate; Δmean P value, the mean difference (post-pre) of each protocol was tested with unpaired Student’s t tests; data presented as means ± SE.
Brachial Artery Flow-Mediated Dilation
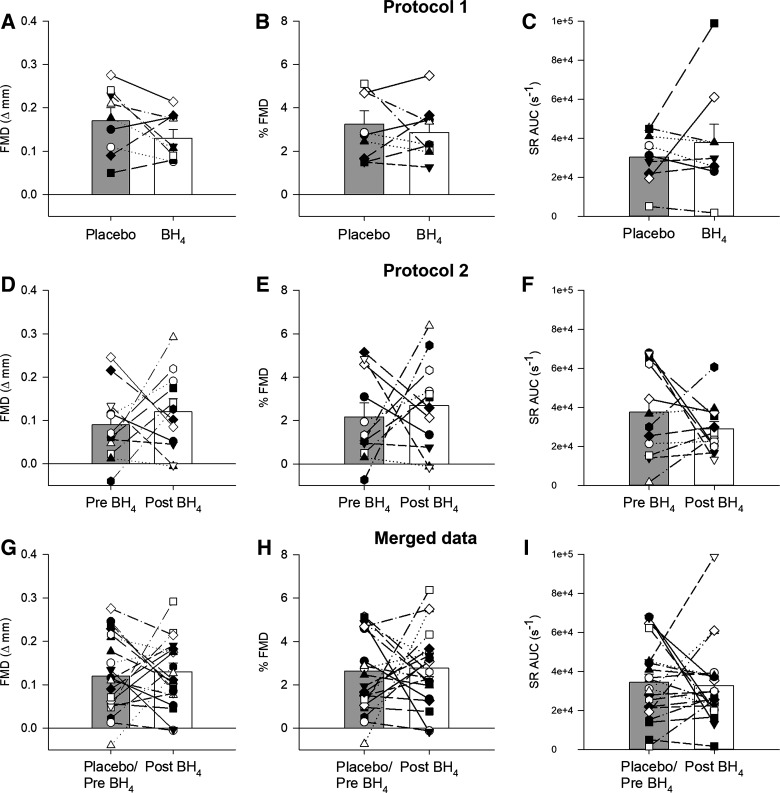
All brachial artery FMD data are documented in Table 2 and highlighted in Fig. 2. Neither baseline brachial artery nor peak diameter, during the FMD assessment, were affected by the acute BH4 supplementation in Protocol 1 and Protocol 2 or when these data were merged (Table 2 and Fig. 2, A, B, D, E, G, and H). Similarly, BH4 did not affect the absolute change in diameter (FMD), percentage change in diameter (%FMD), or time to peak diameter in either protocol or the merged data (Table 2). There was no effect of BH4 in terms of SR AUC (Fig. 2, C, F, and I) or the normalization of both absolute FMD and %FMD for SR AUC (Table 2). In addition, brachial artery BF AUC was not affected by the acute BH4 supplementation in either Protocol 1, Protocol 2, or the merged data (Table 2). As determined by two-way ANOVA, the effect of the BH4 treatment was not dependent on sex, therefore, there was no evidence of a statistically significant interaction between treatment and sex for FMD (P = 0.995), %FMD (P = 0.795), SR AUC (P = 0.494), and BF AUC (P = 0.610).
Figure 2.
Brachial artery flow-mediated dilation (FMD) expressed as an absolute value (A, D, and G), as a percentage change from baseline (B, E, and H), and shear rate area under the curve until peak FMD (SR AUC) (C, F, and I) assessed in Protocol 1, Protocol 2, and data from both protocols merged, merged data. Initial FMD responses prior to acute BH4 supplementation in Protocol 1, Protocol 2, and for the merged data are labeled as placebo/pre BH4 while 4.5 h after acute BH4 supplementation (10 mg/kg), FMD responses in Protocol 1, Protocol 2, and the merged data are labeled as post BH4. Figure 2 sample sizes are 9 (6 M/3F), 12 (8 M/4F), and 21 (14 M/6F) for Protocol 1, Protocol 2, and the merged data, respectively. The effect of BH4 supplementation, within each protocol, was tested with paired Student’s t test. Data are presented as means ± SE. BH4, tetrahydrobiopterin.
Inflammation, Oxidative Stress, Free Radicals, and Antioxidant Capacity Assays
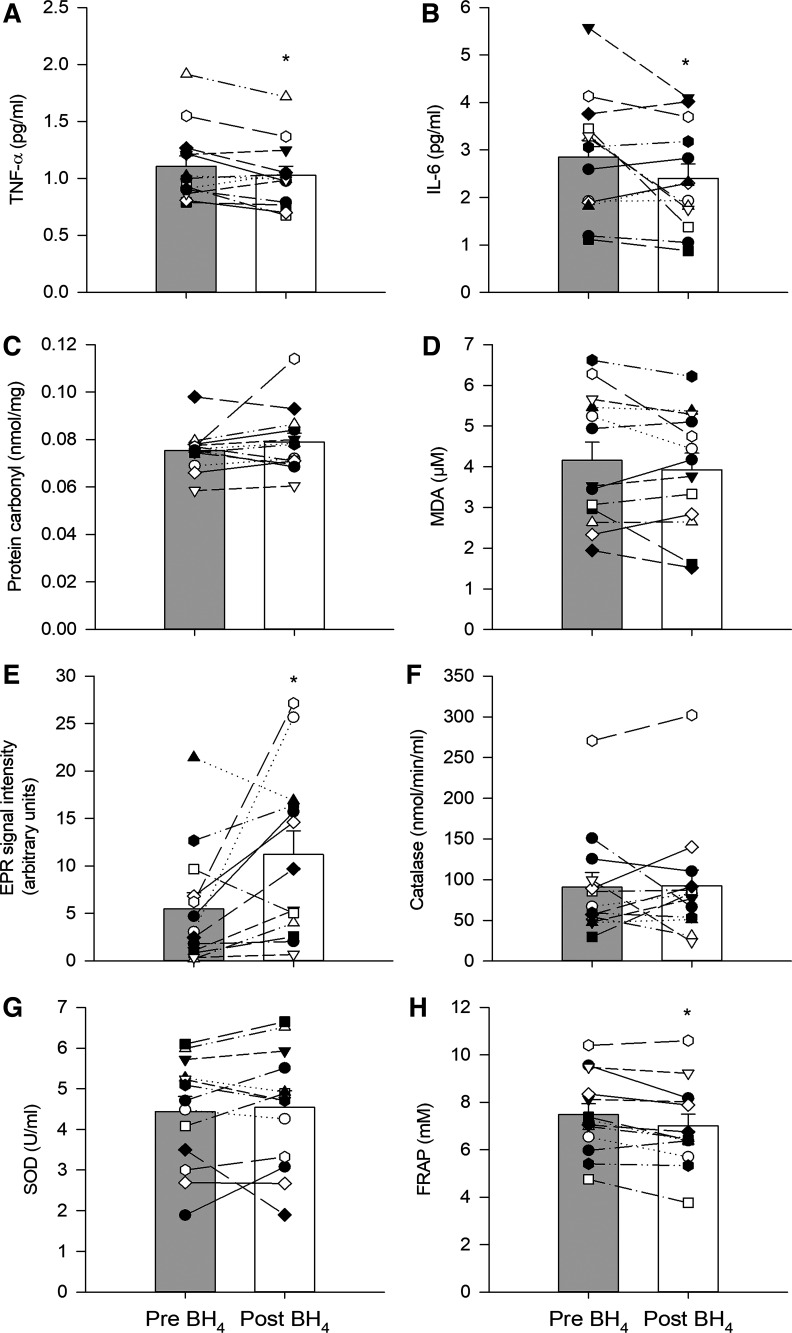
The blood biomarkers of inflammation, oxidative stress, free radicals, and antioxidant capacity from Protocol 2 are presented in Fig. 3. Following the acute BH4 supplementation there was a significant decrease in the inflammatory biomarkers, TNF-α (1.11 ± 0.09 vs. 1.03 ± 0.08 pg/mL; P = 0.03; Fig. 3A) and IL-6 (2.85 ± 0.35 vs. 2.40 ± 0.30 pg/mL; P = 0.04; Fig. 3B). There was no effect of the acute BH4 supplementation on the oxidative stress markers, protein carbonyl, and MDA (Fig. 3C and D). The acute BH4 supplementation significantly increased the EPR signal assessed in both Protocol 1 (placebo: 7 ± 5 vs. post BH4: 12 ± 8 AU, P = 0.08, data not illustrated) and Protocol 2 (6 ± 2 vs. 11 ± 2 AU, P = 0.03. Fig. 3E), which is proportional to free radical concentration. In terms of the antioxidant biomarkers, the acute BH4 supplementation did not affect the endogenous antioxidants, Catalase (Fig. 3F) and SOD (Fig. 3G), but significantly lowered the FRAP (7.48 ± 0.46 vs. 7.00 ± 0.49 mM; P = 0.003; Fig. 3H).
Figure 3.
Blood biomarkers, from Protocol 2, of inflammation [tumor necrosis factor-α (TNF-α), A; and interleukin 6 (IL-6), B; oxidative stress (protein carbonyl), C; and malondialdehyde (MDA), D; free radicals (electron paramagnetic resonance, EPR, signal intensity), E; endogenous antioxidant capacity (catalase activity), F; and superoxide dismutase (SOD), G; and total antioxidant capacity (ferric reducing ability of plasma, FRAP), H]. Initial blood biomarker data are labeled as pre BH4 intake (gray bars) while 4.5 h after acute BH4 supplementation (10 mg/kg), blood biomarkers are labeled as post BH4. Figure 3 sample size is 12 (8 M/4F). The effect of BH4 supplementation, within each protocol, was tested with paired Student’s t test. Data presented as means ± SE. *P< 0.05 different from pre.
DISCUSSION
As a deficiency in BH4, a cofactor for endothelial nitric oxide synthase (eNOS), has been implicated in the age-related attenuation in vascular function, this study aimed to determine the impact of acute BH4 supplementation on FMD in the old (>65 yr). Two approaches were employed: First, a multiday, double-blind, placebo-controlled, crossover design and second, a single experimental day approach with FMD assessed in control and BH4 conditions. Contrary to our hypothesis, regardless of the experimental approach, acute BH4 supplementation did not affect vascular function, as measured by FMD or %FMD, and did not alter either the stimulus for vasodilation, as assessed by SR AUC, or the microcirculation, as assessed by BF AUC. Moreover, when the two data sets were combined, by virtue of the lack of differences in the overall results of Protocol 1 and 2, affording a greater sample size, there was still no evidence of an effect of BH4 on vascular function in these old subjects. Importantly, the analysis of plasma levels of BH4 and BH2, the oxidized form of BH4, revealed a significant, and proportional, increase in both as a consequence of acute BH4 supplementation. Consequently, the ratio of BH4/BH2, recognized to impact vascular function, was unchanged by acute BH4 supplementation. Thus, acute BH4 supplementation does not correct vascular dysfunction in the old.
BH4, BH2, BH4/BH2 Ratio, eNOS, and Oxidative Stress
BH4 not only stabilizes the active conformation of eNOS but also acts as an allosteric effector enhancing substrate binding, thereby coupling substrate oxidation to NADPH consumption and lowering the ratio of superoxide (O2−) to NO production (11, 13–15, 17). Of note, although free radicals are clearly responsible for the oxidation of BH4 to BH2, it is likely not O2− that is responsible for this phenomenon as the rate constant of this reaction (3.9 × 105 M − 1·s−1) (36) is many orders of magnitude lower than that for NO with O2− (6.7 × 109 M −1·s−1), which is one of the fastest reactions in the body (37). A more likely mechanism for BH4 oxidation is the interaction with peroxynitrite (ONOO−), generated from the reaction between NO and O2−. Regardless, in oxidatively stressed endothelial cells, the depletion of BH4 can result in eNOS uncoupling and an increase in BH2. As the Km values of BH4 and BH2 are similar for eNOS, BH2 competes with BH4 for the eNOS binding site (38). Indeed, it is now recognized that the intracellular BH4/BH2 ratio, rather than the absolute concentration of BH4, is likely the key determinant of eNOS uncoupling (13, 36–38). As systemic oxidative stress plays a critical role in determining the degree of oxidation of BH4 to BH2, and, hence, the ratio of BH4:BH2, redox balance can alter the efficacy of BH4 supplementation (39). Certainly, in the presence of oxidative stress, even the acute supplementation with BH4 might tip the balance toward more BH2 such that any physiological benefit of a higher initial BH4 concentration is offset. Thus, unlike the current study (Fig. 1), it is somewhat surprising that the majority of previous studies examining the impact of BH4 on vascular function either only measure the concentration of BH4 or do not, actually, perform any BH4 or BH2 concentration measurements (21, 22, 24–26, 40). Such an assessment seems essential to fully understand the consequences of increasing BH4 concentration.
Acute BH4 Supplementation and Vascular Function with Healthy Aging
A major catalyst for the current study was the work by Eskurza et al. (26), which documented a BH4-induced improvement in endothelial-dependent vasodilation in relatively young, sedentary, older males (62 ± 2 yr), but, interestingly, not in their habitually exercise-trained, age-matched, counterparts, or sedentary young men. The goal of the current work was to extend these findings to an older population, in light of our ever-growing life expectancy. Interestingly, the cause of the initial attenuation in vascular function in the sedentary older subjects in the study by Eskurza et al. (26) was hypothesized to be related to oxidative stress, which had likely diminished BH4 bioavailability. Indeed, the plasma concentration of oxidized low-density lipoprotein (LDL), a somewhat insensitive systemic marker of oxidative stress, was higher in the older sedentary men compared with the young sedentary men, and the oxidized LDL of the young subjects was not different from that of the exercise-trained older subjects. However, there were no additional assessments of redox balance. Although rather speculative, due to the lack of comprehensive bioassays of redox balance and no measurement of BH4 or BH2, the authors attributed the restoration of vascular function in the sedentary older subjects to that of their exercise-trained counterparts to an overcoming of the oxidative stress-mediated attenuation in BH4 bioactivity by the acute BH4 supplementation (26). Furthermore, the lack of an effect of the BH4 supplementation in the habitually exercise-trained older subjects was attributed to an exercise-induced upregulation of enzymatic antioxidants (41), attenuating baseline oxidative stress (42), which, in turn, may have maintained BH4 activity (26).
The results of the current study contrast with the findings of Eskurza et al. (26), revealing a lack of an effect of acute BH4 supplementation on vascular function, as measured by FMD, in healthy, untrained, old subjects (74 ± 7 yr). Of note, the fundamental experimental design (multiday, double-blind, placebo-controlled, crossover design) of Protocol 1 in the current study was modeled after this prior work, with the same number of untrained, but older, subjects. Protocol 2, a single day experimental approach, with BH4 ingestion in the laboratory and similarly aged participants, prompted by the lack of an effect of acute BH4 supplementation in Protocol 1, and even the merging of the data from both protocols, yielded the same contrast with the findings of Eskurza et al. (26), no impact of acute BH4 supplementation. Therefore, the primary difference in the current study was the inclusion of a much older population, which may explain the differing results. Of note, a better understanding of this different effect across age groups is important for the design of future studies examining people across the lifespan. However, in addition to the age of the subjects, there are other important interstudy comparisons that are worthy of discussion. First, while the dosage of BH4 employed in the two studies was similar, Eskurza et al. (26) allowed 3 h while the current study allowed 4.5 h, however, both were guided by previous literature (43, 44). The impact of this difference in timing is unclear, but, based upon the previous studies (43, 44), which were used to determine the timing of the post BH4 assessments in each study, it is unlikely to explain the disparity between the findings. Third, the current study included both males and females, with a 2:1 ratio while the previous study was limited to males (26). As there was no effect of sex in the current study it is unlikely that this more optimal subject selection can explain the differing findings. Fourth, the initial %FMD assessments, before acute BH4 supplementation, were relatively low in both studies [≈2%–3% in the current study and ≈4% in the work of Eskurza et al. (26)], which contrast starkly with their age-matched, habitually trained subjects who exhibited a ≈6%FMD. These baseline FMD values imply that the current subjects had the clear potential for an improvement in vascular function, although such an improvement was not forthcoming in terms of either FMD, SR AUC, or BF AUC as a consequence of the acute BH4 supplementation. In combination, these comparisons between the current study and that of Eskurza et al. (26) suggest that these incongruent results are likely due to the age difference and, subsequently, the greater state of inflammation and oxidative stress experienced by the older subjects studied in the current investigation.
In addition to the assessment of vascular function and plasma BH4 levels, the current study incorporated a relatively comprehensive assessment of bioassays, including BH4, BH2, TNF-α, IL-6, protein carbonyl, MDA, EPR spectroscopy, catalase activity, SOD activity, and FRAP. These bioassays afford the opportunity to surmise the mechanisms responsible for the failure of the acute BH4 supplementation to improve vascular function in this old population. Specifically, with an initial focus on the data illustrated in Fig. 1, it is evident that likely due to relatively high levels of oxidative stress with advancing age, 4.5 h following the acute BH4 supplementation, there was significant oxidation of BH4 to BH2, which resulted in an unchanged BH4/BH2 ratio. With the recognition that the BH4/BH2 ratio, itself, rather than the absolute concentration of BH4, is probably the key determinant of eNOS uncoupling (13, 36–38), this is likely the fundamental explanation for the lack of an effect of the acute BH4 supplementation on vascular function in these old subjects (Fig. 2). However, the other bioassays of inflammation, oxidative stress, free radicals, and oxidative stress, performed at the same time point and illustrated in Fig. 3, can provide supporting evidence. Indeed, the documented oxidation of BH4 to BH2 likely results in greater free radical production, which is evident by the significant increase in EPR signal intensity [Protocol 1 data (not illustrated) and Fig. 3E] and the significant fall in FRAP (Fig. 3H). Furthermore, although seeming counterintuitive in the face of elevated free radical levels, the apparently attenuated level of inflammation, as documented by TNF-α and IL-6 (Fig. 3, A and B), is likely the result of the initial impact of the elevated BH4 levels and its antioxidant properties (45, 46). Of note, inflammation, as well as markers of oxidative stress (Fig. 3, C and D), have a much slower time course to that of free radical levels and antioxidant capacity and, therefore, both inflammation and evidence of oxidative stress would, ultimately, likely increase in response to the greater free radical load.
BH4 Supplementation and Vascular Function with Disease
The use of acute BH4 supplementation to restore impaired endothelial function in various forms of human pathology and adverse conditions, such as smoking, has been widely investigated and the preponderance of findings has supported a positive impact of BH4 (21, 23, 24, 40). However, as already noted, many of these prior investigations, that have targeted populations with conditions that result in a positive effect of BH4 on vascular function, have failed to appropriately assess the impact of acute BH4 supplementation on the BH4/BH2 ratio (21, 24–26, 40) and other important bioassays of inflammation, oxidative stress, free radicals, and antioxidant capacity, without which the interpretation of the findings is, somewhat, limited. Interestingly, in one of the, relatively, rare studies that did assess both BH4, BH2, and vascular function, in this case in a group of patients with coronary artery disease, it was documented that even after chronic BH4 supplementation (i.e., between 2 and 6 wk before coronary artery bypass surgery), the concentration of BH4 in the blood, as well as in the vasculature itself, increased significantly. However, this increase was also accompanied by systemic oxidation of BH4 to BH2, resulting in no change in the BH4/BH2 ratio (39), leaving the vascular function, as measured by arterial stiffness and brachial artery flow-mediated dilation, unaltered (39). Likewise, in the current study (Fig. 2), although the BH4 was delivered as just a single dose, the concomitant increase in both BH4 and BH2 resulted in no net effect on eNOS coupling and, ultimately, endothelial function. Thus, although currently devoid of other cardiovascular risk factors, the old subjects in the current study likely align more closely with patients with coronary artery disease (39), than their younger counterparts (26), especially in terms of oxidative stress and inflammatory state and, subsequently, the impact of acute BH4 supplementation.
Summary and Conclusion
In old subjects, there was no evidence of an effect of acute BH4 supplementation on vascular function. Interestingly, as a consequence of acute BH4 supplementation, plasma levels of both BH4 and BH2, the oxidized form of BH4, increased, significantly and proportionately. Consequently, the ratio of BH4/BH2, recognized to impact vascular function, was unchanged by single BH4 supplementation. Thus, acute BH4 supplementation does not appear to correct vascular dysfunction in the old.
Experimental Considerations
In this study, some experimental considerations must be taken into account. First, in contrast to Protocol 1, Protocol 2 did not include a placebo condition. Indeed, the experimental design for Protocol 2, as it was completed in a single day, includes just two conditions, pre and post BH4 intake. However, to merge the data, not only were the Δmean results between placebo/pre and post BH4 tested for differences between Protocols 1 and 2 but also the placebo (Protocol 1) and pre (Protocol 2) data were also tested for differences. This analysis revealed no significant differences between with and without a placebo (data not reported). The second experimental consideration is related to BH4 elimination and half-life. Kuvan prescription information describes a range of half-life from 3.9 to 17 h for patients with PKU, and from 3.0 to 5.3 h for young healthy subjects. However, since pharmacokinetic analysis in participants >49 yr of age has not been reported, the current experimental study design was based upon previous improved FMD responses, from our laboratory, reported in a cohort of patients with systemic sclerosis with a similar experimental approach (47). However, future studies, measuring BH4 concentrations, and vascular function at different time points following acute BH4 supplementation, to examine, potentially, differing pharmacokinetics in this population are warranted. Third, although plasma measurements of BH4 and BH2 have been included in the present manuscript, it is possible that these values do not fully reflect those in the vascular tissue, as previously demonstrated in patients with coronary artery disease (48), who, in terms of vascular responses, seem similar to the old population studied here. Moreover, how the biopterins are carried into the vascular tissue is not fully understood, especially in vivo in humans. A proposed mechanism is the rapid conversion of BH4 to BH2, which is then taken up by cells, and converted back to BH4 by the “salvage DHFR pathway” (49), further studies investigating the integrity of the dihydrofolate reductase (DHFR) salvage pathway, and the relationship between the intracellular and plasma ratio of BH4:BH2, likely necessary in various populations, would be required to fully elucidate this question. Importantly, it also needs to be recognized that the present findings are the result of acute BH4 supplementation and, therefore, different findings, such as a higher BH4/BH2 ratio, as a result of a longer supplementation (i.e., 1 wk) cannot be completely ruled out. Finally, although this study was not primarily powered to detect sex differences, a mixed-sex design does better represent the typical population than the common all-male approach. However, additional research is still needed to understand age-and sex-related differences in vascular senescence.
DATA AVAILABILITY
The data that support this study are available upon reasonable request from the corresponding author.
GRANTS
This work was supported by Veterans Administration Rehabilitation Research and Development Service Grants (E6910-R, E1697-R, E3207-R, E9275-L, E1572, I01RX001311, and I01CX002152) and a National Institutes of Health, Ruth L. Kirschstein National Research Service Award (T32HL139451) from the National Heart, Lung, and Blood Institute.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
AUTHOR CONTRIBUTIONS
A.V.B., R.M.B., and R.S.R. conceived and designed research; A.V.B., R.S.G., R.M.B., C.L.J., S.H.P., K.L.S., H.L.C., S.M.R., D.W.W., and R.S.R. performed experiments; A.V.B., R.S.G., R.M.B., C.L.J., S.H.P., K.L.S., H.L.C., S.M.R., V.R., J.Z., D.W.W., and R.S.R. analyzed data; A.V.B., R.S.G., R.M.B., C.L.J., S.H.P., K.L.S., H.L.C., S.M.R., V.R., J.Z., D.W.W., and R.S.R. interpreted results of experiments; A.V.B., R.S.G., R.M.B., C.L.J., S.H.P., K.L.S., H.L.C., S.M.R., D.W.W., and R.S.R. prepared figures; A.V.B., R.S.G., R.M.B., C.L.J., S.H.P., K.L.S., H.L.C., S.M.R., D.W.W., and R.S.R. drafted manuscript; A.V.B., R.S.G., R.M.B., C.L.J., S.H.P., K.L.S., H.L.C., S.M.R., V.R., J.Z., D.W.W., and R.S.R. edited and revised manuscript; A.V.B., R.S.G., R.M.B., C.L.J., S.H.P., K.L.S., H.L.C., S.M.R., V.R., J.Z., D.W.W., and R.S.R. approved final version of manuscript.
ACKNOWLEDGMENTS
The authors thank the subjects who graciously took part in this study.
REFERENCES
- 1.Steuerle E, Cosic D. Has Society Gotten Older or Younger? (Online). Urban Institute, 2019. https://www.urban.org/research/publication/has-society-gotten-older-or-younger. [2022 Feb 25] [Google Scholar]
- 2.Roberts L. 9 Billion? Science 333: 540–543, 2011. doi: 10.1126/science.333.6042.540. [DOI] [PubMed] [Google Scholar]
- 3.Franceschi C, Motta L, Motta M, Malaguarnera M, Capri M, Vasto S, Candore G, Caruso C; IMUSCE. The extreme longevity: the state of the art in Italy. Exp Gerontol 43: 45–52, 2008. doi: 10.1016/j.exger.2007.06.006. [DOI] [PubMed] [Google Scholar]
- 4.Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, et al. Heart disease and stroke statistics-2015 update: a report from the American Heart Association. Circulation 131: e29–e–322., 2015. [Erratum in Circulation 131: e535, 2015, and in Circulation 133: e417, 2016]. doi: 10.1161/CIR.0000000000000152. [DOI] [PubMed] [Google Scholar]
- 5.Chen JH, Hales CN, Ozanne SE. DNA damage, cellular senescence and organismal ageing: causal or correlative? Nucleic Acids Res 35: 7417–7428, 2007. doi: 10.1093/nar/gkm681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Celermajer DS, Sorensen KE, Spiegelhalter DJ, Georgakopoulos D, Robinson J, Deanfield JE. Aging is associated with endothelial dysfunction in healthy men years before the age-related decline in women. J Am Coll Cardiol 24: 471–476, 1994. doi: 10.1016/0735-1097(94)90305-0. [DOI] [PubMed] [Google Scholar]
- 7.Seals DR, Jablonski KL, Donato AJ. Aging and vascular endothelial function in humans. Clin Sci (Lond) 120: 357–375, 2011. doi: 10.1042/CS20100476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Broxterman RM, Witman MA, Trinity JD, Groot HJ, Rossman MJ, Park SY, Malenfant S, Gifford JR, Kwon OS, Park SH, Jarrett CL, Shields KL, Hydren JR, Bisconti AV, Owan T, Abraham A, Tandar A, Lui CY, Smith BR, Richardson RS. Strong relationship between vascular function in the coronary and brachial arteries: a clinical coming of age for the updated flow-mediated dilation test? Hypertension 74: 208–215, 2019. doi: 10.1161/HYPERTENSIONAHA.119.12881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Celermajer DS, Sorensen KE, Gooch VM, Spiegelhalter DJ, Miller OI, Sullivan ID, Lloyd JK, Deanfield JE. Non-invasive detection of endothelial dysfunction in children and adults at risk of atherosclerosis. Lancet 340: 1111–1115, 1992. doi: 10.1016/0140-6736(92)93147-F. [DOI] [PubMed] [Google Scholar]
- 10.Wadley AJ, Veldhuijzen van Zanten JJ, Aldred S. The interactions of oxidative stress and inflammation with vascular dysfunction in ageing: the vascular health triad. Age (Dordr) 35: 705–718, 2013. doi: 10.1007/s11357-012-9402-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kaufman S. New tetrahydrobiopterin-dependent systems. Annu Rev Nutr 13: 261–286, 1993. doi: 10.1146/annurev.nu.13.070193.001401. [DOI] [PubMed] [Google Scholar]
- 12.Vásquez-Vivar J. Tetrahydrobiopterin, superoxide, and vascular dysfunction. Free Radic Biol Med 47: 1108–1119, 2009. doi: 10.1016/j.freeradbiomed.2009.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bendall JK, Douglas G, McNeill E, Channon KM, Crabtree MJ. Tetrahydrobiopterin in cardiovascular health and disease. Antioxid Redox Signal 20: 3040–3077, 2014. doi: 10.1089/ars.2013.5566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Thöny B, Auerbach N, Blau N. Tetrahydrobiopterin biosynthesis, regeneration and functions. Biochem J 347: 1–16, 2000. [PMC free article] [PubMed] [Google Scholar]
- 15.Edgar KS, Galvin OM, Collins A, Katusic ZS, McDonald DM. BH4-mediated enhancement of endothelial nitric oxide synthase activity reduces hyperoxia-induced endothelial damage and preserves vascular integrity in the neonate. Invest Ophthalmol Vis Sci 58: 230–241, 2017. doi: 10.1167/iovs.16-20523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Werner-Felmayer G, Werner ER, Fuchs D, Hausen A, Reibnegger G, Schmidt K, Weiss G, Wachter H. Pteridine biosynthesis in human endothelial cells. Impact on nitric oxide-mediated formation of cyclic GMP. J Biol Chem 268: 1842–1846, 1993. doi: 10.1016/S0021-9258(18)53931-4. [DOI] [PubMed] [Google Scholar]
- 17.Kuzkaya N, Weissmann N, Harrison DG, Dikalov S. Interactions of peroxynitrite, tetrahydrobiopterin, ascorbic acid, and thiols: Implications for uncoupling endothelial nitric-oxide synthase. J Biol Chem 278: 22546–22554, 2003. doi: 10.1074/jbc.M302227200. [DOI] [PubMed] [Google Scholar]
- 18.Vásquez-Vivar J, Kalyanaraman B, Martásek P, Hogg N, Masters BS, Karoui H, Tordo P, Pritchard KA Jr.. Superoxide generation by endothelial nitric oxide synthase: the influence of cofactors. Proc Natl Acad Sci USA 95: 9220–9225, 1998. doi: 10.1073/pnas.95.16.9220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vásquez-Vivar J, Martásek P, Whitsett J, Joseph J, Kalyanaraman B. The ratio between tetrahydrobiopterin and oxidized tetrahydrobiopterin analogues controls superoxide release from endothelial nitric oxide synthase: an EPR spin trapping study. Biochem J 362: 733–739, 2002. doi: 10.1042/bj3620733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Heitzer T, Krohn K, Albers S, Meinertz T. Tetrahydrobiopterin improves endothelium-dependent vasodilation by increasing nitric oxide activity in patients with Type II diabetes mellitus. Diabetologia 43: 1435–1438, 2000. doi: 10.1007/s001250051551. [DOI] [PubMed] [Google Scholar]
- 21.Rodriguez-Miguelez P, Gregg J, Seigler N, Bass L, Thomas J, Pollock JS, Sullivan JC, Dillard TA, Harris RA. Acute tetrahydrobiopterin improves endothelial function in patients with COPD. Chest 154: 597–606, 2018. doi: 10.1016/j.chest.2018.04.028. [DOI] [PubMed] [Google Scholar]
- 22.Cosentino F, Hürlimann D, Delli Gatti C, Chenevard R, Blau N, Alp NJ, Channon KM, Eto M, Lerch P, Enseleit F, Ruschitzka F, Volpe M, Lüscher TF, Noll G. Chronic treatment with tetrahydrobiopterin reverses endothelial dysfunction and oxidative stress in hypercholesterolaemia. Heart 94: 487–492, 2008. doi: 10.1136/hrt.2007.122184. [DOI] [PubMed] [Google Scholar]
- 23.Setoguchi S, Hirooka Y, Eshima K, Shimokawa H, Takeshita A. Tetrahydrobiopterin improves impaired endothelium-dependent forearm vasodilation in patients with heart failure. J Cardiovasc Pharmacol 39: 363–368, 2002. doi: 10.1097/00005344-200203000-00007. [DOI] [PubMed] [Google Scholar]
- 24.Ueda S, Matsuoka H, Miyazaki H, Usui M, Okuda S, Imaizumi T. Tetrahydrobiopterin restores endothelial function in long-term smokers. J Am Coll Cardiol 35: 71–75, 2000. doi: 10.1016/s0735-1097(99)00523-9. [DOI] [PubMed] [Google Scholar]
- 25.Higashi Y, Sasaki S, Nakagawa K, Kimura M, Noma K, Hara K, Jitsuiki D, Goto C, Oshima T, Chayama K, Yoshizumi M. Tetrahydrobiopterin improves aging-related impairment of endothelium-dependent vasodilation through increase in nitric oxide production. Atherosclerosis 186: 390–395, 2006. doi: 10.1016/j.atherosclerosis.2005.07.025. [DOI] [PubMed] [Google Scholar]
- 26.Eskurza I, Myerburgh LA, Kahn ZD, Seals DR. Tetrahydrobiopterin augments endothelium-dependent dilatation in sedentary but not in habitually exercising older adults. J Physiol 568: 1057–1065, 2005. doi: 10.1113/jphysiol.2005.092734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Harris RA, Nishiyama SK, Wray DW, Richardson RS. Ultrasound assessment of flow-mediated dilation. Hypertension 55: 1075–1085, 2010. doi: 10.1161/HYPERTENSIONAHA.110.150821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Credeur DP, Miller SM, Jones R, Stoner L, Dolbow DR, Fryer SM, Stone K, McCoy SM. Impact of prolonged sitting on peripheral and central vascular health. Am J Cardiol 123: 260–266, 2019. doi: 10.1016/j.amjcard.2018.10.014. [DOI] [PubMed] [Google Scholar]
- 29.Thijssen DHJ, Bruno RM, van Mil ACCM, Holder SM, Faita F, Greyling A, Zock PL, Taddei S, Deanfield JE, Luscher T, Green DJ, Ghiadoni L. Expert consensus and evidence-based recommendations for the assessment of flow-mediated dilation in humans. Eur Heart J 40: 2534–2547, 2019. doi: 10.1093/eurheartj/ehz350. [DOI] [PubMed] [Google Scholar]
- 30.Thijssen DHJ, Black MA, Pyke KE, Padilla J, Atkinson G, Harris RA, Parker B, Widlansky ME, Tschakovsky ME, Green DJ. Assessment of flow-mediated dilation in humans: a methodological and physiological guideline. Am J Physiol Heart Circ Physiol 300: H2–H12, 2011. doi: 10.1152/ajpheart.00471.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pyke KE, Tschakovsky ME. The relationship between shear stress and flow-mediated dilatation: implications for the assessment of endothelial function. J Physiol 568: 357–369, 2005. doi: 10.1113/jphysiol.2005.089755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen X, Xun K, Chen L, Wang Y. TNF-α, a potent lipid metabolism regulator. Cell Biochem Funct 27: 407–416, 2009. doi: 10.1002/cbf.1596. [DOI] [PubMed] [Google Scholar]
- 33.Wheeler CR, Salzman JA, Elsayed NM, Omaye ST, Korte DW Jr.. Automated assays for superoxide dismutase, catalase, glutathione peroxidase, and glutathione reductase activity. Anal Biochem 184: 193–199, 1990. doi: 10.1016/0003-2697(90)90668-Y. [DOI] [PubMed] [Google Scholar]
- 34.Benzie IFF, Strain JJ. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal Biochem 239: 70–76, 1996. doi: 10.1006/abio.1996.0292. [DOI] [PubMed] [Google Scholar]
- 35.Faul F, Erdfelder E, Lang AG, Buchner A. G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39: 175–191, 2007. doi: 10.3758/bf03193146. [DOI] [PubMed] [Google Scholar]
- 36.Vásquez-Vivar J, Whitsett J, Martásek P, Hogg N, Kalyanaraman B. Reaction of tetrahydrobiopterin with superoxide: EPR-kinetic analysis and characterization of the pteridine radical. Free Radic Biol Med 31: 975–985, 2001. doi: 10.1016/S0891-5849(01)00680-3. [DOI] [PubMed] [Google Scholar]
- 37.Huie RE, Padmaja S. The reaction of no with superoxide. Free Radic Res Commun 18: 195–199, 1993. doi: 10.3109/10715769309145868. [DOI] [PubMed] [Google Scholar]
- 38.Crabtree MJ, Tatham AL, Al-Wakeel Y, Warrick N, Hale AB, Cai S, Channon KM, Alp NJ. Quantitative regulation of intracellular endothelial nitric-oxide synthase (eNOS) coupling by both tetrahydrobiopterin-eNOS stoichiometry and biopterin redox status. J Biol Chem 284: 1136–1144, 2009. doi: 10.1074/jbc.M805403200. [DOI] [PubMed] [Google Scholar]
- 39.Cunnington C, Van Assche T, Shirodaria C, Kylintireas I, Lindsay AC, Lee JM, Antoniades C, Margaritis M, Lee R, Cerrato R, Crabtree MJ, Francis JM, Sayeed R, Ratnatunga C, Pillai R, Choudhury RP, Neubauer S, Channon KM. Systemic and vascular oxidation limits the efficacy of oral tetrahydrobiopterin treatment in patients with coronary artery disease. Circulation 125: 1356–1366, 2012. doi: 10.1161/CIRCULATIONAHA.111.038919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Heitzer T, Brockhoff C, Mayer B, Warnholtz A, Mollnau H, Henne S, Meinertz T, Münzel T. Tetrahydrobiopterin improves endothelium-dependent vasodilation in chronic smokers: evidence for a dysfunctional nitric oxide synthase. Circ Res 86: E36–E41, 2000. doi: 10.1161/01.res.86.2.e36. [DOI] [PubMed] [Google Scholar]
- 41.Sen CK. Oxidants and antioxidants in exercise. J Appl Physiol (1985) 79: 675–686, 1995. doi: 10.1152/jappl.1995.79.3.675. [DOI] [PubMed] [Google Scholar]
- 42.Fukai T, Siegfried MR, Ushio-Fukai M, Cheng Y, Kojda G, Harrison DG. Regulation of the vascular extracellular superoxide dismutase by nitric oxide and exercise training. J Clin Invest 105: 1631–1639, 2000. doi: 10.1172/jci9551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fiege B, Ballhausen D, Kierat L, Leimbacher W, Goriounov D, Schircks B, Thöny B, Blau N. Plasma tetrahydrobiopterin and its pharmacokinetic following oral administration. Mol Genet Metab 81: 45–51, 2004. doi: 10.1016/j.ymgme.2003.09.014. [DOI] [PubMed] [Google Scholar]
- 44.Trinity JD, McDaniel J, Venturelli M, Fjeldstad AS, Ives SJ, Witman MA, Barrett-O'Keefe Z, Amann M, Wray DW. Impact of body position on central and peripheral hemodynamic contributions to movement-induced hyperemia: implications for rehabilitative medicine. Am J Physiol Circ Physiol 300: H1885–H1891, 2011. doi: 10.1152/ajpheart.00038.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Aboonabi A, Meyer RR, Singh I. The association between metabolic syndrome components and the development of atherosclerosis. J Hum Hypertens 33: 844–855, 2019. doi: 10.1038/s41371-019-0273-0. [DOI] [PubMed] [Google Scholar]
- 46.Rosenkranz-Weiss P, Sessa WC, Milstien S, Kaufman S, Watson CA, Pober JS. Regulation of nitric oxide synthesis by proinflammatory cytokines in human umbilical vein endothelial cells. Elevations in tetrahydrobiopterin levels enhance endothelial nitric oxide synthase specific activity. J Clin Invest 93: 2236–2243, 1994. doi: 10.1172/JCI117221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Machin DR, Clifton HL, Richardson RS, Wray DW, Donato AJ, Frech TM. Acute oral tetrahydrobiopterin administration ameliorates endothelial dysfunction in systemic sclerosis. Clin Exp Rheumatol 35: 167–172, 2017. [PMC free article] [PubMed] [Google Scholar]
- 48.Antoniades C, Shirodaria C, Crabtree M, Rinze R, Alp N, Cunnington C, Diesch J, Tousoulis D, Stefanadis C, Leeson P, Ratnatunga C, Pillai R, Channon KM. Altered plasma versus vascular biopterins in human atherosclerosis reveal relationships between endothelial nitric oxide synthase coupling, endothelial function, and inflammation. Circulation 116: 2851–2859, 2007. doi: 10.1161/CIRCULATIONAHA.107.704155. [DOI] [PubMed] [Google Scholar]
- 49.Crabtree MJ, Channon KM. Synthesis and recycling of tetrahydrobiopterin in endothelial function and vascular disease. Nitric Oxide 25: 81–88, 2011. doi: 10.1016/j.niox.2011.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fukushima T, Nixon JC. Analysis of reduced forms of biopterin in biological tissues and fluids. Anal Biochem 102: 176–188, 1980. doi: 10.1016/0003-2697(80)90336-X. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support this study are available upon reasonable request from the corresponding author.