Abstract
Lyme disease is the most common tick-borne disease in North America and Europe, however, current biomarkers inconsistently detect the disease. In this issue of the JCI, Gwynne et al. revealed how the Lyme disease agent Borrelia burgdorferi relies on host lipids for growth. The authors used a murine model to show that B. burgdorferi infection led to the production of antibodies against phospholipids, possibly as a consequence of incorporation into the spirochete membrane. Antibodies were induced against phosphatidic acid, phosphatidylcholine, and phosphatidylserine. Notably, no antibodies against cardiolipin were found, distinguishing Lyme disease from syphilis and some other diseases. Sera samples from patients with Lyme disease suggested that these antibodies may help diagnose B. burgdorferi infection and that antibody titers may effectively indicate the response to treatment. These findings suggest that B. burgdorferi–induced anti-lipid antibodies, in conjunction with a careful clinical assessment, may aid in the diagnosis of Lyme disease.
Insights into the challenges of Lyme disease diagnosis
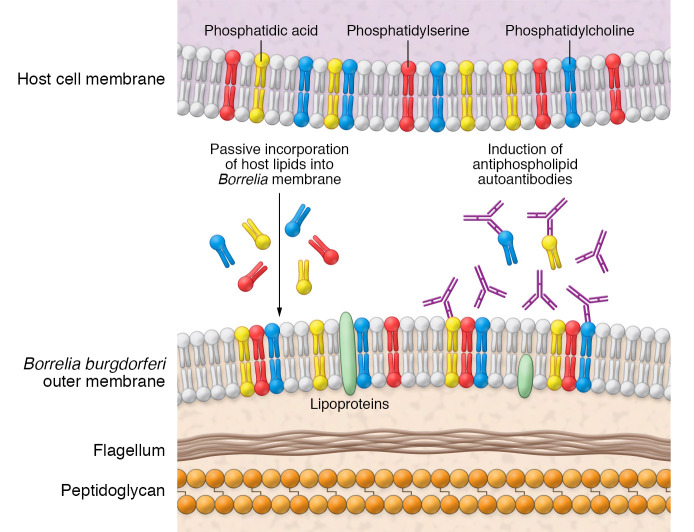
Lyme disease is the most common tick-borne infection in North America (1). Two-tiered serologic testing, consisting of an ELISA followed by immunoblotting against Borrelia antigens, is routinely used to aid clinicians in the diagnosis of Lyme disease. The sensitivity rates, however, are not always ideal, particularly during early infection, when Borrelia burgdorferi–specific antibodies have not yet developed (2, 3). In addition, the long-term presence of B. burgdorferi–specific antibodies after an initial infection can make serodiagnosis of a repeat infection difficult. In this issue of the JCI, Gwynne et al. demonstrate that mice developed antiphospholipid antibodies following B. burgdorferi infection (Figure 1 and ref. 4). Further, the authors found many of the same antibodies in the sera of patients with Lyme disease, mostly in early disease marked by erythema migrans. A further comparison with sera from patients with syphilis implied that some of these antiphospholipid antibodies were specific to B. burgdorferi infection. The findings suggest that antibodies against specific phospholipids could aid in the diagnosis of early or repeat infection by the Lyme disease spirochete.
Figure 1. Model for antiphospholipid antibody production following Borrelia infection.
Borrelia spirochetes incorporate host lipids into the Borrelia membrane. Subsequently, the host develops antiphospholipid antibodies against PC and PA. Elevated antiphospholipid antibodies may detect Lyme disease at early infection points in human serum.
Dependence of Borrelia on host lipids
The B. burgdorferi membrane includes lipoproteins and lipids, such as phosphatidylcholine (PC), phosphatidylglycerol (PG), and cholesterol (5–8). Because of an evolutionarily reduced genome, B. burgdorferi lack the metabolic pathways to synthesize many required lipids and rely on scavenging lipids from the host environment or culture medium (9). Cholesterol, for example, is taken directly from the host. Cholesterol and cholesterol-glycolipid rafts present on the B. burgdorferi surface can also interact with the lipid rafts of host cells and help Borrelia adhere to cells (10). Lipids are transferred between B. burgdorferi and host cells by direct contact and outer membrane vesicles released by the host (10). Using a delipidated medium, Gwynne et al. showed that B. burgdorferi was unable to grow in the absence of environmental lipids and that this growth was restored when fatty acids and cholesterol were added to the medium (4). The growth was also dependent on the concentration of these fatty acids and cholesterol. While the Borrelia membrane is known to contain PC and PG (11), Gwynne and colleagues revealed that other phospholipids — phosphatidylethanolamine (PE), phosphatidylserine (PS), and phosphatidic acid (PA) — were incorporated into borrelial membranes. Further, the study compared PC uptake in different bacteria and revealed that such uptake was specific to Borrelia and likely acquired by random diffusion. Borrelia lipase (bb0562) contributes to fatty acid scavenging and Borrelia survival under conditions in which free fatty acids are limited (12), but a comprehensive understanding of uptake will require further work. Gwynne and co-authors also demonstrated that the ratio of environmental lipids found in the growth media directly correlated with the proportion of lipids found in the Borrelia membrane (4). This lipid ratio agrees with previous work, demonstrating that Borrelia membrane lipid composition reflects the environmental lipid composition. It stands to reason that the membranes of Borrelia infecting a host would reflect this pattern, whereby the composition of lipids in the host environment would directly relate to the composition of lipids in the infecting Borrelia membrane. Borrelia may scavenge host lipids as a strategy to evade the host immune response directed against Borrelia antigens. The incorporation of host fatty acids into the membrane may allow the spirochetes to evade immune cells patrolling for pathogen lipids, making the spirochetes less immunostimulatory (13–15).
Antiphospholipid antibodies in different diseases
Gwynne and co-authors demonstrate that Borrelia infection may induce antibodies recognizing host phospholipids, revealing a potential biomarker of infection (4). Although antiphospholipid antibodies are most commonly associated with autoimmune disorders, such antibodies are sometimes generated following infection. Syphilis, caused by the spirochete Treponema pallidum, induces antibodies against the phospholipid cardiolipin (16). Interestingly, cardiolipin antibodies are also induced during autoimmune antiphospholipid syndrome (APS). Beyond syphilis, antiphospholipid antibodies have been reported in various infectious diseases including COVID-19, tuberculosis, malaria, leprosy, leptospirosis, and Lyme disease, as well as during Helicobacter pylori, hepatitis C, varicella, and other infections (16, 17). HIV infection is associated with antibodies against cardiolipin as well as PS, PC, and phosphatidylinositol (16, 18). Plasmodium infection similarly induces the development of PS-specific antibodies in mice and humans (19, 20). Such antiphospholipid antibodies are often due to erroneous activation of B cells that are induced because of inflammation and marked by expression of the T-bet transcription factor (21, 22). The broad development of these types of antibodies may limit their overall utility, however, anti-PA, anti-PS, and anti-PC antibodies might be used, in conjunction with a careful clinical assessment and current serologic tests, as biomarkers for Lyme disease.
Antiphospholipid-specific diagnosis of Lyme disease
Gwynne et al. showed that B. burgdorferi infection in a mouse model induced antibodies against PA and PC and against the phospholipids PS, PE, PG, and galactosylcholesterol (gC) (4). The mice did not develop antibodies against cardiolipin, the phospholipid associated with syphilis infection, implying that the detected antibodies were specific to Lyme disease. The authors then compared the diagnostic value of PA-, PS- and PC-specific antibody responses using standard assays in patients with early symptoms of Lyme disease. In a small number of patients, the diagnosis using antiphospholipid antibodies outperformed standard diagnostic tests and even correctly identified antibody responses in individuals who only presented with erythema migrans. In addition, the antiphospholipid antibodies peaked during B. burgdorferi infection in humans and declined following antibiotic treatment, which contrasts with the more prolonged antibody response toward Borrelia antigens. These antiphospholipid responses could depend on invariant natural killer T (iNKT) cells, which are capable of recognizing lipid antigens and assisting B cells with antibody production (23, 24). Future studies to validate antiphospholipid responses with B. burgdorferi infection should include a larger number of patients with Lyme disease and individuals with other infectious and autoimmune illnesses. The lipid autoantibodies described in the study by Gwynne and colleagues represent potentially promising biomarkers that could aid clinicians in the diagnosis of Lyme disease and meaningfully impact patient outcomes.
Acknowledgments
This work was support by grants from the NIH (AI126033 and AI138949, to EF).
Version 1. 03/15/2022
Electronic publication
Footnotes
Conflict of interest: EF has an equity interest in and serves as a consultant for L2 Diagnostics.
Copyright: © 2022, Arora et al. This is an open access article published under the terms of the Creative Commons Attribution 4.0 International License.
Reference information: J Clin Invest. 2022;132(6):e158254. https://doi.org/10.1172/JCI158254.
Contributor Information
Gunjan Arora, Email: gunjan.arora@yale.edu.
Erol Fikrig, Email: erol.fikrig@yale.edu.
References
- 1.Mead PS. Epidemiology of Lyme disease. Infect Dis Clin North Am. 2015;29(2):187–210. doi: 10.1016/j.idc.2015.02.010. [DOI] [PubMed] [Google Scholar]
- 2.Leeflang MM, et al. The diagnostic accuracy of serological tests for Lyme borreliosis in Europe: a systematic review and meta-analysis. BMC Infect Dis. 2016;16:140. doi: 10.1186/s12879-016-1468-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schutzer SE, et al. Direct diagnostic tests for Lyme disease. Clin Infect Dis. 2019;68(6):1052–1057. doi: 10.1093/cid/ciy614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gwynne PJ, et al. Antiphospholipid autoantibodies in Lyme disease arise after scavenging of host phospholipids by Borrelia burgdorferi. J Clin Invest. 2022;132(6):e152506. doi: 10.1172/JCI152506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wang XG, et al. Phospholipid synthesis in Borrelia burgdorferi: BB0249 and BB0721 encode functional phosphatidylcholine synthase and phosphatidylglycerolphosphate synthase proteins. Microbiology (Reading) 2004;150(pt 2):391–397. doi: 10.1099/mic.0.26752-0. [DOI] [PubMed] [Google Scholar]
- 6.Ben-Menachem G, et al. A newly discovered cholesteryl galactoside from Borrelia burgdorferi. Proc Natl Acad Sci U S A. 2003;100(13):7913–7918. doi: 10.1073/pnas.1232451100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hove PR, et al. Identification and functional analysis of a galactosyltransferase capable of cholesterol glycolipid formation in the Lyme disease spirochete Borrelia burgdorferi. PLoS One. 2021;16(6):e0252214. doi: 10.1371/journal.pone.0252214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Huang Z, London E. Cholesterol lipids and cholesterol-containing lipid rafts in bacteria. Chem Phys Lipids. 2016;199:11–16:S0009-3084(16)30036–6. doi: 10.1016/j.chemphyslip.2016.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ye M, et al. Genome reduction of Borrelia burgdorferi: two TCS signaling pathways for two distinct host habitats. Sci China Life Sci. 2016;59(1):19–21. doi: 10.1007/s11427-015-4996-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Crowley JT, et al. Lipid exchange between Borrelia burgdorferi and host cells. PLoS Pathog. 2013;9(1):e1003109. doi: 10.1371/journal.ppat.1003109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hossain H, et al. Structural analysis of glycolipids from Borrelia burgdorferi. Biochimie. 2001;83(7):683–692. doi: 10.1016/S0300-9084(01)01296-2. [DOI] [PubMed] [Google Scholar]
- 12.Kuhn HW, et al. BB0562 is a nutritional virulence determinant with lipase activity important for Borrelia burgdorferi infection and survival in fatty acid deficient environments. PLoS Pathog. 2021;17(8):e1009869. doi: 10.1371/journal.ppat.1009869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Toledo A, Benach JL. Hijacking and use of host lipids by intracellular pathogens. Microbiol Spectr. 2015;3(6) doi: 10.1128/microbiolspec.VMBF-0001-2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wolf BJ, et al. Identification of a potent microbial lipid antigen for diverse NKT cells. J Immunol. 2015;195(6):2540–2551. doi: 10.4049/jimmunol.1501019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Birge RB, et al. Phosphatidylserine is a global immunosuppressive signal in efferocytosis, infectious disease, and cancer. Cell Death Differ. 2016;23(6):962–978. doi: 10.1038/cdd.2016.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Asherson RA, Cervera R. Antiphospholipid antibodies and infections. Ann Rheum Dis. 2003;62(5):388–393. doi: 10.1136/ard.62.5.388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stelzer M, et al. The role of antiphospholipid antibodies in COVID-19. Curr Rheumatol Rep. 2021;23(9):72. doi: 10.1007/s11926-021-01041-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Silvestris F, et al. Antiphosphatidylserine antibodies in human immunodeficiency virus-1 patients with evidence of T-cell apoptosis and mediate antibody-dependent cellular cytotoxicity. Blood. 1996;87(12):5185–5195. doi: 10.1182/blood.V87.12.5185.bloodjournal87125185. [DOI] [PubMed] [Google Scholar]
- 19.Barber BE, et al. Antiphosphatidylserine immunoglobulin M and immunoglobulin G antibodies are higher in vivax than falciparum malaria, and associated with early anemia in both species. J Infect Dis. 2019;220(9):1435–1443. doi: 10.1093/infdis/jiz334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fernandez-Arias C, et al. Anti-self phosphatidylserine antibodies recognize uninfected erythrocytes promoting malarial anemia. Cell Host Microbe. 2016;19(2):194–203. doi: 10.1016/j.chom.2016.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Peng SL, et al. T-bet regulates IgG class switching and pathogenic autoantibody production. Proc Natl Acad Sci U S A. 2002;99(8):5545–5550. doi: 10.1073/pnas.082114899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rivera-Correa J, et al. Plasmodium DNA-mediated TLR9 activation of T-bet+ B cells contributes to autoimmune anaemia during malaria. Nat Commun. 2017;8(1):1282. doi: 10.1038/s41467-017-01476-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.King IL, et al. Invariant natural killer T cells direct B cell responses to cognate lipid antigen in an IL-21-dependent manner. Nat Immunol. 2011;13(1):44–50. doi: 10.1038/ni.2172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brennan PJ, et al. Invariant natural killer T cells: an innate activation scheme linked to diverse effector functions. Nat Rev Immunol. 2013;13(2):101–117. doi: 10.1038/nri3369. [DOI] [PubMed] [Google Scholar]