Abstract
Clustered regularly interspaced short palindromic repeats/CRISPR-associated nuclease9 (CRISPR/Cas9) gene editing technology implements precise programming of the human genome through RNA guidance. At present, it has been widely used in the construction of animal tumor models, the study of drug resistance regulation mechanisms, epigenetic control and innovation in cancer treatment. Tumor immunotherapy restores the normal antitumor immune response by restarting and maintaining the tumor-immune cycle. CRISPR/Cas9 technology has occupied a central position in further optimizing anti-programmed cell death 1(PD-1) tumor immunotherapy. In this review, we summarize the recent progress in exploring the regulatory mechanism of tumor immune PD-1 and programmed death ligand 1(PD-L1) based on CRISPR/Cas9 technology and its clinical application in different cancer types. In addition, CRISPR genome-wide screening identifies new drug targets and biomarkers to identify potentially sensitive populations for anti-PD-1/PD-L1 therapy and maximize antitumor effects. Finally, the strong potential and challenges of CRISPR/Cas9 for future clinical applications are discussed.
Keywords: CRISPR/Cas9, PD-1, PD-L1, tumor immunity, immunotherapy
Introduction
Cancer is a genetic disease accompanied by the accumulation of a variety of mutations (1). Tumor immunotherapy has become one of the most promising therapeutic strategies in tumor treatment compared with the high recurrence rate of metastasis, drug resistance and poor prognosis of traditional therapies (2). Tumor cells actively evade immune detection by activating associated negative regulatory pathways (also known as checkpoints), thus inhibiting the immune response of the human body. Among the numerous checkpoints, cytotoxic T lymphocyte protein 4 (CTLA4) and programmed cell death protein 1 (PD-1) are by far received much attention. CTLA4 controls T cell activation by competing with the costimulatory molecule CD28 to bind to the shared ligands CD80 and CD86 (3). The binding of PD-1 on the surface of T cells to Programmed death ligand 1(PD-L1) on tumor cells is a major obstacle in the cancer immune cycle, inducing T cell apoptosis and inhibiting the activation and proliferation of T cells (4). Fortunately, genetic modification of tumor immunity can be achieved through the CRISPR/Cas9 system. The CRISPR/Cas9 system, derived from the acquired immune defense mechanism of bacteria, enables accurate editing of specific regions of the genome by simple and rapid Watson Crick base pairing between sgRNA and target genes (5, 6). Currently, the CRISPR/Cas9 system has derived many variants such as nickase Cas9 (nCas9), nuclease-deactivated Cas9 (dCas9) and so on, which have been widely used in a variety of cell types and organisms to achieve diversity and ease of use (7, 8). In this review, we summarize CRISPR/Cas9 technology centering on gene editing, gene screening and clinical application of tumor immunity PD-1/PD-L1, as well as its strong potential and existing problems in the field of future tumor therapy.
CRISPR/Cas9 Technology
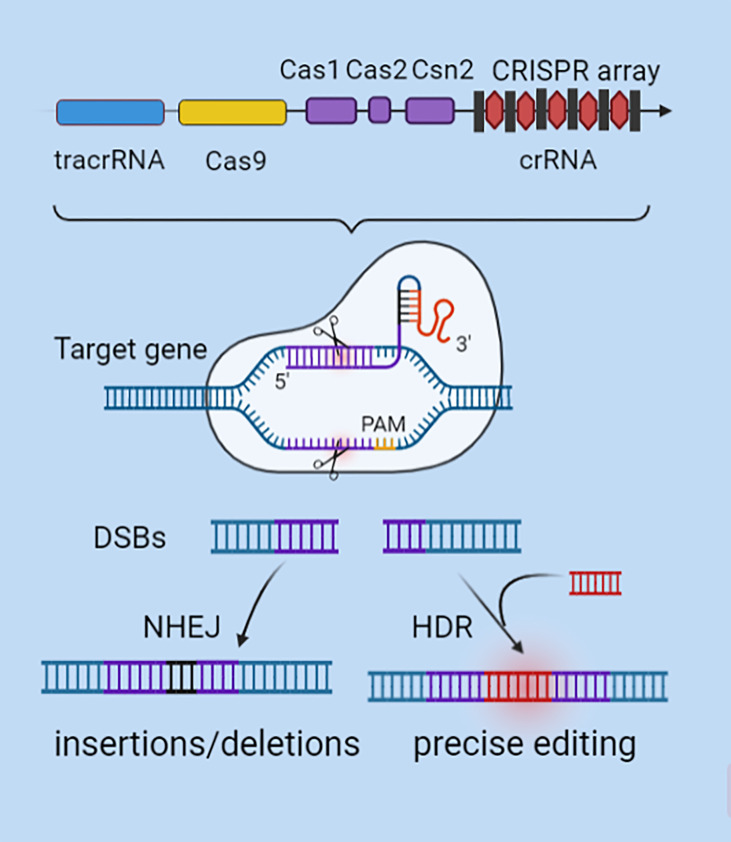
Tumors are characterized by highly heterogeneous molecular characteristics and carry a wide range of gene mutations (9). This may be the main cause of tumor drug resistance and recurrence (10). Gene editing technology is in urgent need of decoding the genetic code at a deeper level and implementing precision medicine. Genome editing can modify the human genome and change biogenesis, which has the potential of targeted therapy to cure diseases (11). The CRISPR/Cas9 system is a gene editing technology guided by the principle of RNA-DNA complementary pairing (12). CRISPR/Cas9 technology is used to repair mutations, knock in or knock out specific genes, and modify genes to explore the mechanisms of tumorigenesis, development, and metastasis for precision therapy. The CRISPR sequence is responsible for the production of crRNA and tracrRNA ( Figure 1 ). The two can be synthesized into a single-stranded guide RNA (sgRNA) (6, 13), which guides Cas9 nuclease to bind to the complementary target gene and cut near the protospacer adjacent motif (PAM) region to introduce DNA double strand breaks(DSBs) (5, 14). At DSBs, Non-homologous end-joining (NHEJ) repair pathway is activated to randomly insert or delete several bases, and efficiently produce INDEL mutations (15). Alternatively, Homologous directed repair (HDR) can occur under a DNA repair template with high homology (16). Compared with Zinc finger endonuclease (ZFN) (17) and Transcription activator-like effector nuclease (TALEN) (18) technologies, the CRISPR/Cas9 system abandons the traditional chimeric nuclease protein domain-DNA recognition design concept and avoids the multiple fusion process in multitarget gene editing (19–21). Therefore, it has the advantages of high specificity, strong operability and convenient systematic analysis, making it the preferred tool for gene editing in eukaryotes. CRISPR/Cas9 gene editing technology has been widely used in the construction of animal tumor models, the study of drug resistance regulation mechanisms, epigenetic control and tumor immunotherapy (22–24). In particular, CRISPR/Cas9 also shows great potential in exploring tumor immune mechanisms, biomarker screening, and clinical trials and therapies.
Figure 1.
CRISPR/Cas9 gene editing mechanism. CRISPR’s sequence consists of a leader, repeats, and spacers. SgRNA is formed by the combination of specific crRNA and tracrRNA that functions as a scaffold. SgRNA can recruit the Cas9 to anchor to the target gene by using RNA-DNA base pairing at the 5'-terminal specific binding sequence. After verifying the PAM motif, Cas9 plays a shearing role to generate DSBs. DSBs are activated by NHEJ or HDR mechanisms to repair mutations, knock in or knock out specific genes, and modify genes.
Tumor Immunity
The occurrence and development of tumours are closely related to the infiltration of immune cells, immune modification and immune escape in the tumour microenvironment (1, 25, 26). Tumour immunity promotes tumour progression by changing tumour biological characteristics (27), screening tumour cells adapted to the microenvironment for survival (28) or establishing a suitable tumour microenvironment (29), and so on. Among them, immune checkpoints such as PD-1 and CTLA4 play an important role. Under normal physiological conditions, the binding of PD-1 and PD-L1 can down-regulate the activity of T cells and prevent additional damage of cytotoxic effector molecule and autoimmunity, so it is called immune checkpoint (30) A Study confirmed that tumor cells often express negative costimulatory signals such as PD-L1, leading to the failure activation of T cell activation. At the same time, tumor-infiltrating lymphocytes (TILs) usually express elevated levels of PD-1 due to chronic stimulation of tumor antigens (31). PD-1/PD-L1 blockade enhances T cell response through multiple underlying mechanisms, altering the outlook for cancer treatment. For example, PD-1/PD-L1 blockade may depend on proliferation of "precursors of exhausted" T (TPEX) cells rather than reversal of T cell depletion procedures (32). It has been found that PD-L1 blockade in conjunction with Alarmin HMGN1 peptide seems to activate anti-tumor TPEX cells and promote their amplification, but does not deplete T cells (33). In addition, PD-1 posttranslational modifications such as glycosylation maintain its stability and membrane expression, and mediate immunosuppressive function. Antibodies targeting glycosylated PD-1 may recognize the heavy glycan moieties of PD-1 and have a higher affinity (34).
CRISPR/Cas9 in Tumor Immunity
Tumor immunity is regulated by many factors and ways. For example, iron poisoning can induce immunogenic apoptosis or necroptosis in cancer cells, which activates antitumor immunity (35). Autophagy regulation inhibits the response through selective degradation of MHC class I molecules, reduces the activity of the STING1 pathway, and promotes tumor immune escape (36). In recent years, CRISPR/Cas9 technology has received increasing attention in tumor immune regulation. CRISPR/Cas9 plays a role in the IFN signaling pathway, ERK/MAPK signaling pathway, Wnt/β-catenin signaling pathway, PI3K/AKT signaling pathway and other mechanisms in chronic inflammation and tumor immune resistance. For example, Wang et al. found that knockout of SHP2 using CRISPR/Cas9 gene editing inhibited SHP2 activity and enhanced tumor-intrinsic IFNγ signaling, resulting in increased chemoattractive cytokine release and cytotoxic T cell recruitment, as well as increased tumor cell surface Increased expression of MHC class I and PD-L1. In addition, SHP2 inhibition reduced the differentiation and suppressive function of immunosuppressive myeloid cells in the tumor microenvironment (37). The study by Yang et al. found that successful nuclear localization of CRISPR/Cas9 ensured efficient destruction of PD-L1 and PTPN2. Inhibition of PTPN2 can alleviate the inhibition of the JAK/STAT pathway and promote tumor susceptibility to CD8+ T cells dependent on IFN-γ, thereby further amplifying the adaptive immune response (38). Therefore, the combination of CRISPR/Cas9 technology with tumor immunotherapy has great potential for development. Immune checkpoint block is one of the most valuable individualized tumor treatment options. For example, the anti-PD-1/PD-L1 antibodies, nivolumab, pembrolizumab, and atezolizumab have shown significant advantages in melanoma, non-small-cell lung cancer (NSCLC), and urothelial carcinoma, and have been approved by the Food and Drug Administration (39–41). However, there remains inflammatory side effects (42, 43), and the overall survival rate is not significantly improved. In addition, CPISPR/Cas9 technology provides a multifunctional and convenient operation mode for the transformation of engineered T cells (44, 45). Genetically engineered T cells have the ability to kill tumor cells as well as significant therapeutic effects. Gene editing technology enables precise genetic modification of T cells, further improving chimeric antigen receptor-engineered T cell immunotherapy. This solves the problem of mass production of therapeutic immune cells and enables the genetic engineering of multiple T cells to meet the clinical needs for the treatment of complex types of cancer.
CRISPR/Cas9-Edited PD-1/PD-L1 in the Tumor Immune Evasion
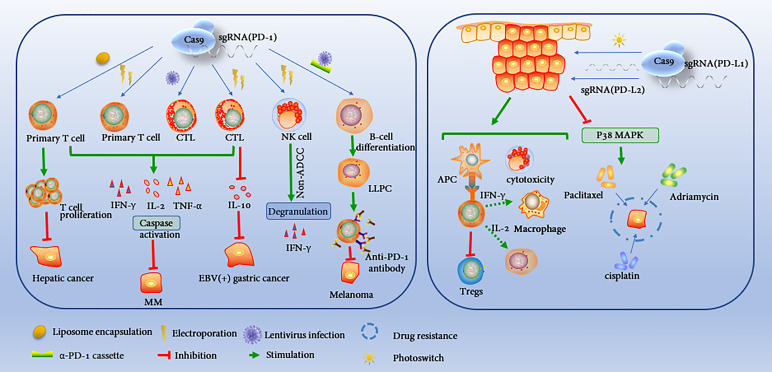
PD-1/PD-L1 has been identified as a negative immunomodulatory molecule that promotes immune evasion of tumor cells. Most patients treated with commercially targeted PD-1/PD-L1 antibodies have achieved a higher cure rate, prolonged survival and fewer recurrence and metastasis events (46–48). However, there are still some patients who are insensitive to targeted PD-1/PD-L1 antibodies or suffer side effects or drug-related inflammatory adverse events (49). The CRISPR/Cas9 technique maximizes PD-1/PD-L1 deletion at the genomic level, thereby saving the function of lymphocytes in the tumour microenvironment. However, whether it corrects the inactivation of targeted antibodies remains unclear ( Figure 2 ).
Figure 2.
CRISPR/Cas9 directly edits PD-1/PD-L1 to regulate tumor immunity. CRISPR/Cas9 technology can edit the PD-1 gene in primary T cells, CTLs, NK cells and B cells through different delivery methods to enhance antitumor immunity through different mechanisms. CRISPR/Cas9 knockdown of PD-L1 in tumor cells promotes tumor antigen presentation, immune cell proliferation and cytotoxicity in the tumor microenvironment, and improves tumor chemotherapy resistance.
CRISPR/Cas9 Engineered Immune Cells
In primary T cells, researchers electroperforated plasmids encoding sgRNA and Cas9 to knock out PD-1, which upregulated IFN-γ production and enhanced cytotoxicity without affecting the viability of primary T cells (50). Similarly, Lu specifically knocked out the PD-1 gene in primary T cells using CRISPR/Cas9 genome editing tools coated with liposomes. This greatly stimulated T cell activation by dendritic cells (DCs) and demonstrated enhanced anticancer potential both in vivo and in vitro (51). Some studies have confirmed that CRISPR/Cas9 is an effective system to knock out PD-1 in cytotoxic T lymphocytes (CTLs). On the one hand, PD-1 KO CTLs can reduce the number of Tregs or inhibit Treg activity and recruit more effector cells. On the other hand, it regulates the secretion of cytokines and activates caspase, thereby inhibiting tumor growth in vivo and in vitro and prolonging survival (52–54). Cas9/sgRNA specifically integrates the α-PD-1 box into the GAPDH site of B lymphocytes. Surprisingly, the edited B lymphocytes differentiated into typical long-lived plasma cells (LLPCs) both in vitro and in vivo. These cells continuously secrete new antibodies that inhibit human melanoma growth in xenograft tumor mice by antibody-mediated checkpoint blocking (55). Cas9 has also improved natural killer cell-based cancer immunotherapy. PD-1-deficient NK cells increased lethality through non-antibody-dependent cell-mediated cytotoxicity (ADCC) pathways and enhanced degranulation and cytokine production (56). In summary, CRISPR/Cas9 provides a powerful and effective protocol for editing PD-1 genes in a variety of immune cells to block immune checkpoints.
CRISPR/Cas9 Engineered Tumor Cells
PD-1/PD-L1 maintains high expression on the surface of various malignant tumors, which is associated with poor prognosis of the disease. PD-L1 leads to T cell dysfunction and tumor evasion from immune surveillance by binding to T cell suppressor receptors (57). After CRISPR/Cas9 reduced the expression level of PD-L1 on tumor cells, CD4 T cells, CD8 T cells, NK cells and CD11c M1 macrophages increased significantly in the tumor microenvironment, while regulatory T cells decreased. The mRNA levels of IFN-γ, TNF-α, interleukin (IL) -2, IL-12A, CXCL9 and CXCL10 were significantly increased, while the mRNA levels of IL-10, vascular endothelial growth factor, CXCL1 and CXCL2 were significantly decreased (58). Based on CRISPR/Cas9, scientists developed a photoactivated photoswitching system constructed from mg polyethylene imine derivative and plasmid PX330/SGPD-L1. The system effectively genetically destroys the PD-L1 gene in large cancer cells and even cancer stem cell-like cells (59). A vectorless multichannel gene editing system was designed in which multiple Cas9 RNPs inhibit both PD-L1 and programmed death ligand 2(PD-L2) ligands. It can significantly increase Th1 cytokine production of cytotoxic CD8 T cells, resulting in a synergistic cytotoxic effect (60). PD-L1 not only mediates tumor immune escape but also makes cancer cells resistant to chemotherapy. CRISPR/Cas9 modification of the tumor cell surface antigen PD-L1 can reduce chemotherapy resistance or produce synergistic effects in combination with chemoradiotherapy (61). For instance, the survival time of mice with the ovarian cancer in the PD-L1-KO group was significantly longer than that in the control group, and the therapeutic effect was enhanced when combined with cisplatin (58). Wu et al. found that CRISPR/cas9-mediated knockout of B7-H1 sensitized cancer cells to chemotherapy and targeting B7-H1 with a monoclonal antibody also sensitized cancer cells to chemotherapy. Furthermore, B7-H1 increases ERK activation in melanoma cells and maintains p38 MAPK activation in triple-negative breast cancer cells, and ERK activation plays a key role in cell survival and drug resistance (62). Targeting PD-L1 in osteosarcoma also increased drug sensitivity to adriamycin and paclitaxel. Doxorubicin and paclitaxel are commonly used to treat osteosarcoma. However, many patients with osteosarcoma are resistant to doxorubicin and paclitaxel chemotherapy. Liao et al. used the MTT assay to evaluate the role of PD-L1 in the resistance of osteosarcoma cells to doxorubicin and paclitaxel. They found that the results of the MTT assay indicated that PD-L1 may be involved in the drug resistance of osteosarcoma and may become a clinical potential target for therapy (63). In general, CRISPR/Cas9 technology not only improves the tumor immune microenvironment by knocking out PD-L1, but also breaks through tumor chemotherapy resistance.
Indirect Regulation of PD-1/PD-L1 by CRISPR/Cas9
The occurrence and development of tumors is a complex pathological process involving the interaction of multiple regulatory molecules and their downstream signaling pathways. CRISPR/Cas9 has enabled further understanding of the regulatory mechanism of immune checkpoint inhibitors (ICIs) and promising biomarkers to help patients stratify and coordinate targeted anti-PD-1/PD-L1 therapy.
Regulation of Antigen Presentation
Upregulation of antigen presenting-related genes of mouse MHC class I molecules, peptide transporters, peptide cleavages and transporter-MHC interactions was observed in lung squamous cell carcinoma lines with CRISPR deletion of WEE1 (64). This suggests the possibility of combining ICI with DNA damage induction therapy to treat laryngeal squamous cell carcinomas (LSCC). In addition, CRISPR-targeted EZH2 knockout reduced histone H3K27me3 modification on the B2 M promoter and restored antigen presentation. Anti-PD-1 antibodies also bypass the resistance of tumor cells to T cell-mediated killing (65). Therefore, the combination of EZH2-targeting with anti-PD-1 therapy may increase treatment susceptibility in head and neck squamous cell carcinoma (HNSCC). CRISPR/Cas9 promoted T cell infiltration in the tumor microenvironment by editing Atrx, and increased tumor cell antigen presentation after IFN-γ stimulation, thus enhancing the ICI response in NSCLC (66). Antigen presentation is a key step in initiating adaptive immunity, and CRISPR/Cas9 enhances antigen presentation to enhance antitumor immunity.
Regulation of Tumor-Associated Macrophages (TAM)
A study found that CRISPR/Cas9 knockdown of Cxcr2 reduced the level of PD-L1 by downregulating C-MYC, thereby accelerating the change in the phenotype of M1 macrophages and suppressing immune escape. Therefore, Cxcr2 knockout is a potentially effective method to block PD-L1 (67). It has also been found that immunocompetent mice with CRISPR/Cas9 knockdown of VEGFC are more likely to develop invasive tumors than immunodeficient mice. These tumors showed downregulation of activated lymphocytes and upregulation of M2 macrophage markers and PD-L1 (68). Thus, targeted therapy for VEGFC must be considered comprehensively. In addition, Li used CRISPR/Cas9 to develop histone chaperone Asf1a deficiency, which coordinated with anti-PD-1 immunotherapy by promoting macrophage M1 polarization and T cell activation (69). CRISPR/Cas9 downregulates the expression of PD-L1 and reverses the tumor-promoting state of TAMs by regulating upstream molecules, thus playing an antitumor role.
Regulating Signal Pathways
With the binding of PD-1 to PD-L1, tyrosine phosphatase is recruited to the cytoplasmic inhibitory motifs of PD-1 and dephosphorylates TCR, thereby inhibiting the downstream IFN, PI3K-AKT and Wnt/β-catenin signaling pathways to promote tumor progression (70, 71). CRISPR/Cas9 synergistically targets PD-1/PD-L1 therapy by regulating downstream signaling pathways. THE interferon (IFN) signaling pathway is a major component of natural immunity, and tumor cells may play a direct defensive role against IFN-mediated cytotoxicity by upregulating PD-L1. Knockout of SHP2 by CRISPR/Cas9 in cancer cells enhanced the expression of MHC class I and PD-L1 proteins in cancer cells through IFN-γ signaling and enhanced the response against PD-1 blockade in cogene mouse models (37). Programmable unlockable nanoencapsulation CRISPR technology (PUN) has hierarchical response characteristics. PUN mitigates inhibition of the JAK/STAT pathway by knocking out the protein tyrosine phosphatase PTPN2 in tumor cells, which relies on IFN-γ to promote antigen presentation and growth inhibition, further amplifying adaptive immunity to PD-L1 blockade (38). The PI3K-AKT signaling pathway is mainly involved in regulating the glycolysis process of immune cells (72). Studies found that CRISPR/Cas9-based GLO1 deletion changed glucose metabolism and redox homeostasis by modulating TXNIP, and PD-L1 expression was reduced at both the gene and protein levels (73). The Wnt//β-catenin pathway, which is extremely active in a variety of cancers, may also influence tumor immunity in addition to driving tumorgenesis and metastasis (74, 75). Researchers constructed an aptamer/peptide-functionalized genome-editing system, which significantly knocked out β-catenin and downregulated the Wnt//β-catenin pathway (76). After knockout, the expression of tumor immunosuppressor-related proteins (PD-L1 and CD47) and proliferation-related proteins (C-MYC and Cyclin D1) was significantly downregulated, which also reversed the immunosuppression induced by PD-L1 in the presence of IFN-γ. These findings provide a strong incentive for CRISPR/Cas9 to explore IFN, PI3K-Akt, and Wnt//β-catenin signaling pathways and strategies to enhance tumor immune invasion, as a means to increase tumor sensitivity to immune checkpoint blockade therapy.
CRISPR/Cas9 Screening in PD-1/PD-L1 Immunotherapy
The occurrence and progression of tumors are closely related to gene mutations (1). Genome-wide screening can comprehensively and fairly grasp the changes in genes in cells, reveal the genetic basis behind specific phenotypes and assist in the development of new therapies. Advances in whole-genome screening in mammalian systems have been hampered by time, size constraints and inefficient double-allelic mutations in mouse culture (77). In recent years, CRISPR/Cas9 technology has provided genome-scale gRNA libraries in the presence of gRNA-guided Cas9 endonucases to generate mutant cell libraries, which serve as a perfect platform for phenotypic screening (78). At present, Cas9 technology has been used in the laboratory, and various sgRNA libraries constructed have revealed the internal molecular regulatory mechanisms of tumor proliferation and metastasis (79), tumor drug resistance (80, 81), tumor immune escape (82) and other malignant behaviors. Below, we summarize the recent progress of CRISPR/Cas9 technology in screening PD-1/PD-L1 and potential regulatory molecules in immune-related cells and tumor cells.
CRISPR technology was used to conduct genome-wide screening of tumor-infiltrating CD8 T cells in vivo. They reidentified typical immunotherapy targets such as PD-1 and TIM-3, as well as other novel sites, and verified the region of CRISPR in vivo screening of CD8 T cells for tumour infiltration (83). Another study identified Fut8, a gene involved in the core focusing pathway, as a positive regulator of PD-1 expression on the cell surface through genome-wide functional loss screening of the CRISPR-Cas9 system. Inhibition of Fut8 can reduce PD-1 expression on the cell surface, enhance T cell activation, and eradicate tumors more effectively (84). Using genome-wide CRISPR and metabolic inhibitor screening, Wang determined that nicotinamide phosphoribotransferase (NAMPT) is required for T cell activation and demonstrated that NAD+ supplementation significantly enhances anti-PD-1 immunotherapy in a mouse solid tumor model (85). This demonstrates the feasibility of CRISPR/Cas9 screening technology in discovering PD-1-related regulatory factors and provides a more comprehensive strategy for in-depth exploration of the PD-1 regulatory mechanism.
In addition to screening in T cells, CRISPR/Cas9 also showed great strength in the study of PD-L1 expressed in tumor cells. At the transcriptional level, CRISPR/Cas9 screening confirmed that ALK activated STAT3 and eventually induced PD-L1 expression through the effects of the transcription factors IRF4 and BATF3 on PD-L1 gene enhancer regions (86). At the translation level, the overexpression of translation initiation factor eIF5B in lung adenocarcinoma allowed us to bypass the inhibitory upstream open reading frame of PD-L1 mRNA in the integrated stress response (ISR) activated by impaired haem production, leading to enhanced PD-L1 translation (87). CMTM6 prevents PD-L1 from lysosomal-mediated degradation, which colocalizes with PD-L1 in the plasma membrane and circulating endosomes (88). In addition, Aleksandra developed a pro-code/CRISPR screen that identified socs1 as a negative regulator of PD-L1 (89). In the same way, a study identified IRF2 in a CRISPR-based forwards genetic screen that controlled MHC-I Ag presentation and PD-L1 expression in HeLa cells (90). CRISPR/Cas9 screening found that the transcription regulator MLLT6 is required for effective PD-L1 protein expression and cell surface presentation in cancer cells. MLLT6 loss mitigates the inhibition of CD8 cytotoxic T cell-mediated lysis (91). In addition, CRISPR/Cas9 screening also found that editing Asf1a (69), PRMT1 and RIPK1 (92), MAN2A1 (93), IFNGR2 and JAK1 (94) increased the sensitivity of tumors to anti-PD-1 or PD-L1 treatment. Tumor cells disrupt T-cell-mediated immune monitoring by maintaining high levels of PD-L1, while CRISPR/Cas9 screening found that the PD-L1 transcription and level, translation levels and maintenance of its stability and expression of oncogenic factors, thus exploring the related regulatory mechanisms of PD-L1 to provide a potential biologic therapeutic target for disrupting PD-L1-mediated tumor tolerance therapy.
CRISPR/Cas9-Associated PD-1/PD-L1 Editing in Different Cancer Species
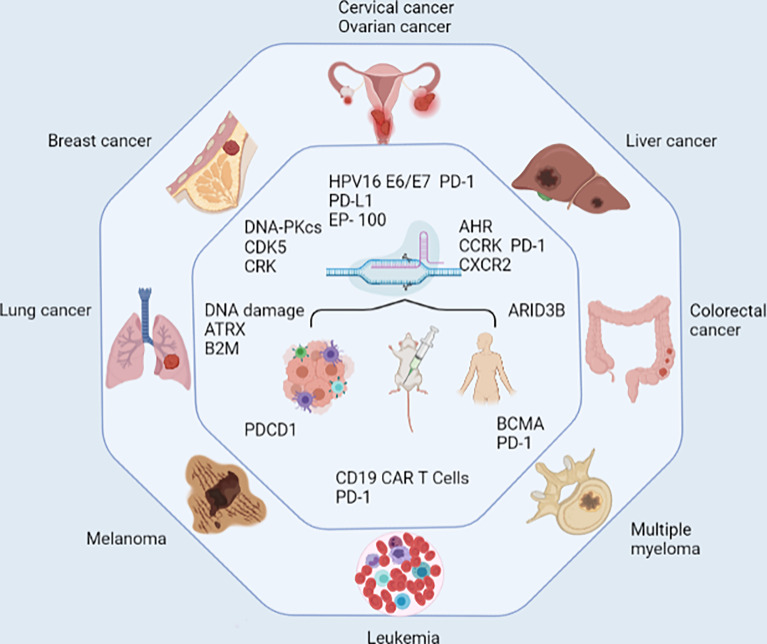
The dominant mechanism of tumor immunity is different in different tumors, which affects the occurrence and progression of tumors in various ways. In this review, we selected several cancers and summarized the role of CRISPR/Cas9 technology-associated PD-1/PD-L1 editing in tumor immunity ( Figure 3 ).
Figure 3.
Research progress of CRISPR/Cas9 in different cancer species. CRISPR/Cas9 has been tested at the cellular level, in animals, and even in humans in a variety of cancers, including leukaemia, multiple myeloma, colorectal cancer, liver cancer, cervical cancer, ovarian cancer, breast cancer, lung cancer, and melanoma. CRISPR/Cas9 enhances antitumor immunity by directly editing PD-1/PD-L1 or by knocking out other molecules in coordination with immune checkpoint blockade. Some novel molecules have been proposed as biomarkers for screening people susceptible to anti-PD-1 therapy.
Leukaemia
Leukaemia is a type of hematopoietic stem cell malignant clonal disease with obvious heterogeneity (95, 96). Currently, T cells modified with chimeric antigen receptors targeting CD19 have been developed, which can overcome many limitations of traditional therapies and achieve high remission rates in patients (97, 98). CRISPR/Cas9 technology provides easy access to further optimize the efficiency of CD19 Chimeric antigen receptor T cells (CAR T cells) to meet the needs of routine clinical use (99). The researchers constructed a disposable CRISPR system that easily harvested CD19 CAR T cells and interfered with PD-1 in CAR T cells. In a b-acute lymphoblastic leukaemia xenograft mouse model, these cells significantly increased their antileukaemia activity (100). This suggests that the combination of immune checkpoint disruption may improve the effectiveness of adoptive cell therapy for leukaemia.
Multiple Myeloma (MM)
MM is a plasma cell malignant proliferative disease with unknown etiology and no cure at present (101). Currently, PD-1/PD-L1 immune checkpoint blockade has shown remarkable efficacy in patients with intractable hematological malignancies (102). Zhao achieved PD-1-deficient CTLs based on CRISPR/Cas9 technology and found that the secretion of TNF-α and IFN-γ increased several times, promoted apoptosis of cocultured MM cells, inhibited tumor growth and prolonged survival within in vivo models of MM (54). CRISPR/Cas9 effectively knocked out PD-1 of in CTLs and enhanced their cytotoxicity. This laid the foundation for the potential use of CRISPR in the production of immune checkpoints targeting CAR T cells. However, CAR T cells targeting B cell maturation antigen (BCMA) on the plasma cell surface remain the mainstream treatment plan for MM (98, 103). The safety and efficacy of using CRISPR/Cas9 to edit PD-1 CAR T cells with MM remains to be investigated.
Breast Cancer
Triple-negative breast cancer is not sensitive to common endocrine therapy and targeted therapy, and resistance to radiotherapy and chemotherapy has become the main obstacle to prolonging the survival of cancer patients, so it is urgent to explore new effective therapeutic targets (104). By using CRISPR/Cas9, Wu found that B7-H1 is related to the catalytic subunit of DNA-dependent protein kinases (DNA-PKCs), which promotes or maintains the activation of ERK or P38 MAPK in cancer cells, thus restoring the sensitivity of cancer cells to chemotherapy (62). In addition, CRISPR/Cas9 knockdown of Cdk5 significantly reduced the expression of PD-L1 on tumor cells, thus effectively inhibiting lung metastasis of triple-negative breast cancer in mice (105). Crk adaptor protein also inhibited EMT and PD-L1 expression on tumor cells through CRISPR/Cas9 gene ablation and inhibited tumor growth and metastasis with anti-PD1 therapy (106). In conclusion, CRISPR/Cas9 compensates for the deficiency that ICIs are basically ineffective in inducing immunogenicity and treating breast cancer and effectively identifies upstream therapeutic targets of immunotherapy, which has considerable potential in exploring and verifying mechanisms of breast cancer metastasis and drug resistance.
Cervical and Ovarian Cancer
Cervical cancer is the most common gynecological malignant tumor and is highly correlated with Human Papillomavirus (HPV) infection (107). GDNA combined with targeting of PD-1 and HPV16 E6/E7 dramatically increased the number of dendritic cells, CD8+ and CD4+ T lymphocytes and hampered tumor growth (108). Therefore, a reasonable combination of CRISPR/Cas9-mediated PD1 blockade and HPV knockout can have a powerful synergistic effect by increasing the persistence of ICIs on the basis of the high sensitivity of targeted therapy for cervical cancer. In addition, cohort studies have been completed to evaluate the safety and efficacy of CRISPR/Cas9-HPV E6/E7 in the treatment of persistent HPV and HPV-associated cervical intraepithelial neoplasia I (NCT03057912). Ovarian cancer is the main cause of cancer death among gynecological malignant tumors, usually with advanced peritoneal or distal metastasis (109). CRISPR-mediated destruction of PD-L1 on the ovarian cancer cell surface regulates the production of cytokines and chemokines to inhibit the progression of ovarian cancer (58). On the other hand, EP-100 was found to induce PD-L1 expression and immune regulation in LHRH-R positive tumor cells. CRISPR/Cas9 revealed the internal biological mechanism of the synergistic action between anti-EP-100 and anti-PD-L1 by silencing the IL33 gene (110). In summary, CRISPR provides a basis for further clinical research on malignant diseases of the female reproductive system.
Hepatoma
Aflatoxin is a risk exposure factor for primary hepatocellular carcinoma, and CRISPR/Cas9 genomic screening identified aryl hydrocarbon receptor as essential for hepatocellular carcinoma (HCC) associated Aflatoxin toxicity, suggesting that an anti-PD-L1 immunosuppression regimen can be used as a treatment regimen for Aflatoxin-associated HCC (111). The CRISPR/Cas9 gene editing system disrupted the PD-1 gene in gypican-3 targeted second-generation CAR T cells (112). The role of CCRK and KDM1A in regulating PD-L1 levels on the surface of HCC cells was also explored (113, 114). Moreover, Huang found that Cas9 deletion of PD-1 combined with lentivirus transduction of human telomerase reverse transcriptase can prolong the lifespan of cytokine induced killer (CIK) cells and enhance their antitumor effects (115). In summary, as a more efficient genome editing technology, CRISPR/Cas9 has been widely used in the exploration of immunotherapy for liver cancer.
Colorectal Cancer
Colorectal cancer (CRC) is a common malignant tumor in the gastrointestinal tract. The occurrence and development of colorectal cancer involves multiple steps, stages and genes (116). Currently, ICIs have been used in the clinical treatment of colorectal cancer, but they are only effective for microsatellite instability MSI patients (117, 118). To explore the precise treatment of ICIs in microsatellite stabilized (MSS) CRCs, Liao discovered the atypical Notch pathway of PD-L1-driven ARID3B, revealing the immune avoidance mechanism of the CRC CMS4 subtype by CRISPR technology (119). These results support the use of ICIs for custom targeting of the CMS4 subtype to screen potential populations that may benefit from ICIs.
Lung Cancer
Anti-PD-1 immunotherapy has been approved as a first-line treatment for lung cancer patients. However, the response rate was poor in patients with lung squamous cell carcinoma and NSCLC. CRISPR/Cas9 has been explored in many influencing factors. Multiple gene editing of mouse pulmonary organs using the CRISPR/Cas9 system can effectively and rapidly generate LSCC to determine whether induced DNA damage can enhance the immunological characteristics of ICI (64). In addition, CRISPR also validated ATRX as a promising biomarker for ICIs in NSCLC, contributing to patient stratification and decision-making (66). In addition, the inability of antigen processing and presentation caused by damage to the main component of HLA class-I complex B2 M is also explained from the perspective of acquired resistance to anti-PD-1 or PD-L1 antibodies (120).
Melanoma
Melanoma is a highly malignant tumor derived from melanocytes that is prone to distant metastasis (121). Although ICIs are currently considered the mainstream immunotherapy for melanoma, the response rate of patients to monoclonal antibodies is still not optimistic (120). One study demonstrated the feasibility of CRISPR-edited PDCD1 in effect-memory CTLs with melanoma antigen specificity, and clearly demonstrated its superiority in delaying the growth of PD-L1-positive melanoma (122). Importantly, genes related to metabolism and cell signaling were actively expressed, but genes related to proliferation and DNA replication were downregulated in PD-1-deficient T cell clones. These results suggest that regulation of effector function is more critical than the ability of T cells to proliferate. To further elucidate the effect of metabolic status on the immune response, scientists performed proteomic analysis and CRISPR validation in patients with advanced melanoma who received anti-PD-1 therapy (123). It was finally proven that lipid metabolism, as a regulatory mechanism, increases the immunogenicity of melanoma by enhancing antigen presentation, thereby enhancing sensitivity to T cell-mediated killing in vivo and in vitro. Therefore, CRISPR can provide insights into the metabolic mechanisms of melanoma immunosuppression to identify therapeutic targets that improve the efficacy of anti-PD-1 immunotherapy.
Clinical Application of CRISPR Cas9-Related PD-1/PD-L1
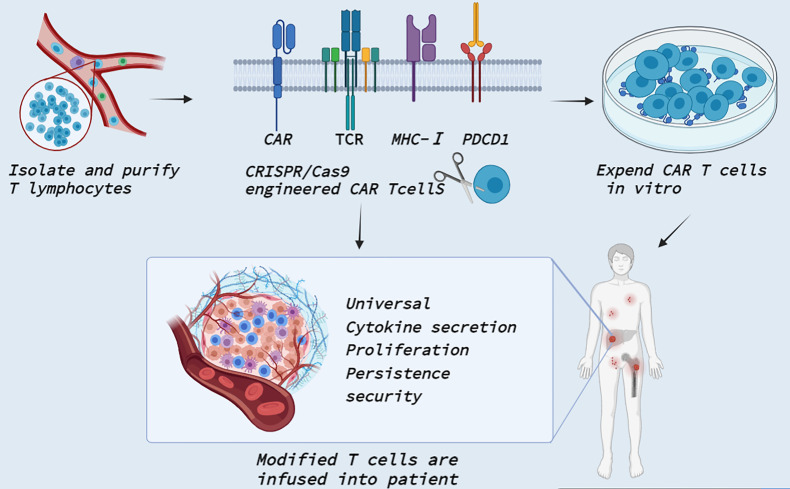
Immunotherapy has become a powerful treatment for many solid and hematological malignancies, including immune checkpoint blocking therapy, immune cell therapy and tumor vaccine therapy (124). Among them, CAR T cell therapy in immune cell therapy has shown great potential in the field of tumor therapy and made unprecedented breakthroughs in clinical practice (98, 125). CAR T therapy is administered by taking T cells from patients and genetically modifying them into chimeric antibody T cells with single-stranded fragment variants from the extracellular antigen recognition domain, the CD3ζ transmembrane domain, and the intracellular T cell activation domain (126). It can specifically recognize tumor cell surface-associated tumor-associated antigen, enhance its defense ability against tumor cells, and stimulate T cell proliferation, cytokine secretion, and lymphocyte recruitment, thereby playing an antitumor role. Fortunately, four generations of evolution have improved the time, difficulty and cost of obtaining CAR T cells, as well as the limitations on the number of patients’ T cells themselves (127). The use of CRISPR/Cas9 gene editing technology opens a new world for the upgrading of fourth-generation CAR T cells ( Figure 4 ). Recently, several clinical trials by Stadtmauer (128), Lu (129), Wang (130), and Simon (131) confirmed the clinical feasibility and safety of CRISPR/Cas9- modified T cells.
Figure 4.
Clinical application of CRISPR Cas9 in CAR T cells. T cells are isolated and purified from healthy persons or patients. CRISPR/Cas9 technology enables precise integration of CAR genes at specific T cell loci. CRISPR/Cas9 also knocked out TCR and MHC-I molecular genes to reduce allogenic reactions and avert GVHD to produce universal CAR T cells. In addition, CRISPR/Cas9 can edit PDCD1 to improve T cell proliferation activity and promote cytokine secretion. The modified T cells are massively amplified in vitro and then transfected back into the patient to reverse tumor immunosuppression.
First, CRISPR/Cas9 can be simultaneously transferred into multiple gDNAs to edit multiple genes at once, providing new ideas for general-purpose allogeneic CAR T cells. For example, Ren by combining CAR lentivirus delivery and electrical transfer of Cas9 mRNA and gRNA, simultaneously targeted endogenous TCR, B2 M and PD-1 and produced allogeneic CAR T cells lacking TCR, HLA class I molecules and PD1 gene destruction. It shows strong antitumor activity in vitro and in animal models, with reduced allograft T cell rejection, and does not cause graft-versus-host disease (45). At the same time, they developed a universal system for the generation of CAR T cells by rapid multigene editing, adding multiple gRNAs into CAR lentivirus vectors and redirecting T cells with antigen-specific CAR through a single CRISPR protocol (100). Second, CRISPR/Cas9-mediated disruption of immune checkpoint signaling, such as PD-1, enhances the therapeutic efficacy of CAR T cells in the immunosuppressive tumor microenvironment. Initially, three studies were completed in vitro in a preclinical model of human glioblastoma to demonstrate that engineered PD-1-deficient EGFRvIII CAR T cells (132, 133), or CD133-specific CAR T cells (134) showed similar levels of cytokine secretion and improved proliferation and cytotoxicity in vitro and enhanced tumor growth inhibition in a mouse model of glioma in situ. Similarly, the combination of positive stimulation from CAR and negative regulation of PD-1 by Cas9 blocking can induce T cells to be more persistent and invasive in vivo in triple-negative breast cancer TNBC (135). In addition, PD-1-deficient GPC3-CAR T cells significantly increased the phosphorylation of Akt and expression of the antiapoptotic protein Bcl-XL, thus preventing the depletion of PD-1-deficient GPC3-CAR T cells from confronting hepatocellular carcinoma cells expressing natural PD-L1 (112). Nevertheless, altering the proportion of PDCD1-deficient CARS affects T cell metastasis, meaning that a strong loss of PDCD1 may enhance CAR T cells in the short term but ultimately make the edited cells more likely to fail or function impaired. Therefore, further studies on the interaction of tumor load, T cell number and editing frequency in different tumors are needed to ensure that CRISPR/Cas9 plays a positive role (136). Subsequently, the application of Cas9-edited PD-1 in CAR T cells has been progressively implemented in clinical trials such as phase I clinical trials of multiple myeloma with mesothin-positive solid tumors (NCT03545815), multiple myeloma (NCT03399448), esophageal cancer (NCT03081715), metastatic NSCLC (NCT02793856), EBV (Epstein-Barr virus)-positive advanced stage malignancies (NCT03044743), advanced hepatocellular carcinoma (NCT04417764) and engineered TILs/CAR-TILs to treat advanced solid tumors (NCT04842812). Overall, CRISPR technology has brought new vitality to CAR T cells in the field of tumor immunotherapy and has gradually entered the stage of clinical trials.
Conclusion
In summary, CRISPR/Cas9 gene editing technology has great potential in the field of tumor PD-1-related immunotherapy. CRISPR/Cas9 gene editing technology can directly edit PD-1/PD-L1 or indirectly regulate antigen presentation and upstream and downstream signaling pathways to improve a variety of immune cells and the tumor microenvironment, and combat tumor immunosuppression. CRISPR genome-wide screening technology also provides new ideas for studying upstream and downstream regulatory mechanisms of PD-1/PD-L1, screening synergistic targeting molecules and identifying potentially sensitive patients. CRISPR/Cas9 has clinical value in the diagnosis and treatment of leukaemia, multiple myeloma, breast cancer, cervical cancer, ovarian cancer, liver cancer, colorectal cancer, lung cancer and melanoma. CRISPR/Cas9 produces allogeneic general-purpose CAR T cells combined with immune checkpoint knockout, expanding the clinical application of engineered cells.
However, the following problems need to be overcome when transforming from laboratory to clinical application: First, off-target effects: gene mutations caused by off-target cutting are prone to cellular malignancy but lead to cancer. Therefore, it is necessary to design sgRNA sequences to increase the specificity of target site cleavage (13) and develop Cas9 variants with PAM preference changes (137). Alternatively, the function of the P53 gene should be monitored to prevent toxicity caused by P53 mutations (138). Second, the activity and proliferation of edited cells should ideally be unaffected. Currently, gene editing and cell amplification can be performed in vitro and transfused back into patients for treatment combined with adoptive cell therapy. Third, safe and effective in vivo delivery methods were selected. At present, the most conventional delivery system is viruses, but they have the disadvantages of limited delivery scale and long-term accumulation easily leading to untargeted cutting. In addition, electroporation and nucleotide transfection techniques have been further explored in animal models. At present, lipid nanoparticle-mediated nonviral delivery without limitation of immunogenicity is more promising (139). Last, Cas9 is an inherent immunogenicity of foreign proteins. Current studies have shown that Cas9 can eliminate immune dominant epitopes through targeted mutations while retaining its function and specificity (140). In brief, although CRISPR/Cas9 gene editing technology shows great promise for PD-1/PD-L1-related therapies in tumor immunity, it is still in its infancy and requires a more comprehensive and in-depth investigation of its safety and reliability.
Author Contributions
ZS provided direction and guidance throughout the preparation of this manuscript. YX wrote and edited the manuscript. CC and YG reviewed and made significant revisions to the manuscript. SH collected and prepared the related papers. All authors read and approved the final manuscript.
Funding
This study was supported by The National Natural Science Foundation of China (81972663, 82173055, U2004112), The Excellent Youth Science Project of Henan Natural Science Foundation (212300410074), The Key Scientific Research Project of Henan Higher Education Institutions (20A310024), The Youth Talent Innovation Team Support Program of Zhengzhou University (32320290) and The Provincial and Ministry co-constructed key projects of Henan medical science and technology (SBGJ202102134).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
CRISPR/Cas9, Clustered regularly interspaced short palindromic repeats/CRISPR-associated nuclease9; PD-1, Programmed cell death 1; PD-L1, Programmed death ligand 1; CTLA4, Cytotoxic T lymphocyte protein 4; sgRNA, Single-stranded guide RNA; PAM, Protospacer adjacent motif; DSB, Double-strand break; NHEJ, Non-homologous end-joining; HDR, Homology-directed repair; TALEN, Transcription activator-like effector nuclease; TIL, tumor-infiltrating lymphocytes; NSCLC, non-small cell lung cancer; DC, dendritic cells; CTL, Cytotoxic T lymphocyte; ADCC, Antibody-dependent cell-mediated cytotoxicity; ICI, Immune checkpoint inhibitor; LSCC, Laryngeal squamous cell carcinomas; HNSCC, Head and neck squamous cell carcinoma; TAM, tumor-associated macrophages; NAMPT, nicotinamide phosphoribotransferase; ISR, integrated stress response; CAR T cell, Chimeric antigen receptor T cell; BCMA, B cell maturation antigen; DNA-PKC, DNA-dependent protein kinase; HPV, Human Papillomavirus; HCC, hepatocellular carcinoma; CIK, cytokine induced killer; CRC, Colorectal cancer; MSI, microsatellite instability; MSS, microsatellite stabilized.
References
- 1. Hanahan D, Weinberg RA. Hallmarks of Cancer: The Next Generation. Cell (2011) 144(5):646–74. doi: 10.1016/j.cell.2011.02.013 [DOI] [PubMed] [Google Scholar]
- 2. Mellman I, Coukos G, Dranoff G. Cancer Immunotherapy Comes of Age. Nature (2011) 480(7378):480–9. doi: 10.1038/nature10673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Walker LS, Sansom DM. The Emerging Role of CTLA4 as a Cell-Extrinsic Regulator of T Cell Responses. Nat Rev Immunol (2011) 11(12):852–63. doi: 10.1038/nri3108 [DOI] [PubMed] [Google Scholar]
- 4. Keir ME, Butte MJ, Freeman GJ, Sharpe AH. PD-1 and Its Ligands in Tolerance and Immunity. Annu Rev Immunol (2008) 26:677–704. doi: 10.1146/annurev.immunol.26.021607.090331 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Gasiunas G, Barrangou R, Horvath P, Siksnys V. Cas9-crRNA Ribonucleoprotein Complex Mediates Specific DNA Cleavage for Adaptive Immunity in Bacteria. Proc Natl Acad Sci USA (2012) 109(39):E2579–2586. doi: 10.1073/pnas.1208507109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E. A Programmable Dual-RNA-Guided DNA Endonuclease in Adaptive Bacterial Immunity. Science (2012) 337(6096):816–21. doi: 10.1126/science.1225829 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Gao Y, Xiong X, Wong S, Charles EJ, Lim WA, Qi LS. Complex Transcriptional Modulation With Orthogonal and Inducible Dcas9 Regulators. Nat Methods (2016) 13(12):1043–9. doi: 10.1038/nmeth.4042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Zong Y, Song Q, Li C, Jin S, Zhang D, Wang Y, et al. Efficient C-To-T Base Editing in Plants Using a Fusion of Ncas9 and Human APOBEC3A. Nat Biotechnol (2018). doi: 10.1038/nbt.4261 [DOI] [PubMed] [Google Scholar]
- 9. Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA, Jr., Kinzler KW. Cancer Genome Landscapes. Science (2013) 339(6127):1546–58. doi: 10.1126/science.1235122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Dagogo-Jack I, Shaw AT. Tumour Heterogeneity and Resistance to Cancer Therapies. Nat Rev Clin Oncol (2018) 15(2):81–94. doi: 10.1038/nrclinonc.2017.166 [DOI] [PubMed] [Google Scholar]
- 11. Doudna JA. The Promise and Challenge of Therapeutic Genome Editing. Nature (2020) 578(7794):229–36. doi: 10.1038/s41586-020-1978-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. van der Oost J, Westra ER, Jackson RN, Wiedenheft B. Unravelling the Structural and Mechanistic Basis of CRISPR-Cas Systems. Nat Rev Microbiol (2014) 12(7):479–92. doi: 10.1038/nrmicro3279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Slaymaker IM, Gao L, Zetsche B, Scott DA, Yan WX, Zhang F. Rationally Engineered Cas9 Nucleases With Improved Specificity. Science (2016) 351(6268):84–8. doi: 10.1126/science.aad5227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Jinek M, Jiang F, Taylor DW, Sternberg SH, Kaya E, Ma E, et al. Structures of Cas9 Endonucleases Reveal RNA-Mediated Conformational Activation. Science (2014) 343(6176):1247997. doi: 10.1126/science.1247997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Bibikova M, Carroll D, Segal DJ, Trautman JK, Smith J, Kim YG, et al. Stimulation of Homologous Recombination Through Targeted Cleavage by Chimeric Nucleases. Mol Cell Biol (2001) 21(1):289–97. doi: 10.1128/mcb.21.1.289-297.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. McConnell Smith A, Takeuchi R, Pellenz S, Davis L, Maizels N, Monnat RJ, Jr., et al. Generation of a Nicking Enzyme That Stimulates Site-Specific Gene Conversion From the I-AniI LAGLIDADG Homing Endonuclease. Proc Natl Acad Sci USA (2009) 106(13):5099–104. doi: 10.1073/pnas.0810588106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Urnov FD, Rebar EJ, Holmes MC, Zhang HS, Gregory PD. Genome Editing With Engineered Zinc Finger Nucleases. Nat Rev Genet (2010) 11(9):636–46. doi: 10.1038/nrg2842 [DOI] [PubMed] [Google Scholar]
- 18. Joung JK, Sander JD. TALENs: A Widely Applicable Technology for Targeted Genome Editing. Nat Rev Mol Cell Biol (2013) 14(1):49–55. doi: 10.1038/nrm3486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Deltcheva E, Chylinski K, Sharma CM, Gonzales K, Chao Y, Pirzada ZA, et al. CRISPR RNA Maturation by Trans-Encoded Small RNA and Host Factor RNase III. Nature (2011) 471(7340):602–7. doi: 10.1038/nature09886 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Hsu PD, Lander ES, Zhang F. Development and Applications of CRISPR-Cas9 for Genome Engineering. Cell (2014) 157(6):1262–78. doi: 10.1016/j.cell.2014.05.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Gaj T, Gersbach CA, Barbas CF, 3rd. ZFN, TALEN, and CRISPR/Cas-Based Methods for Genome Engineering. Trends Biotechnol (2013) 31(7):397–405. doi: 10.1016/j.tibtech.2013.04.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Hilton IB, D'Ippolito AM, Vockley CM, Thakore PI, Crawford GE, Reddy TE, et al. Epigenome Editing by a CRISPR-Cas9-Based Acetyltransferase Activates Genes From Promoters and Enhancers. Nat Biotechnol (2015) 33(5):510–7. doi: 10.1038/nbt.3199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Platt RJ, Chen S, Zhou Y, Yim MJ, Swiech L, Kempton HR, et al. CRISPR-Cas9 Knockin Mice for Genome Editing and Cancer Modeling. Cell (2014) 159(2):440–55. doi: 10.1016/j.cell.2014.09.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Yang Y, Qiu JG, Li Y, Di JM, Zhang WJ, Jiang QW, et al. Targeting ABCB1-Mediated Tumor Multidrug Resistance by CRISPR/Cas9-Based Genome Editing. Am J Transl Res (2016) 8(9):3986–94. [PMC free article] [PubMed] [Google Scholar]
- 25. Bader JE, Voss K, Rathmell JC. Targeting Metabolism to Improve the Tumor Microenvironment for Cancer Immunotherapy. Mol Cell (2020) 78(6):1019–33. doi: 10.1016/j.molcel.2020.05.034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Gajewski TF, Schreiber H, Fu YX. Innate and Adaptive Immune Cells in the Tumor Microenvironment. Nat Immunol (2013) 14(10):1014–22. doi: 10.1038/ni.2703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Balkwill F, Charles KA, Mantovani A. Smoldering and Polarized Inflammation in the Initiation and Promotion of Malignant Disease. Cancer Cell (2005) 7(3):211–7. doi: 10.1016/j.ccr.2005.02.013 [DOI] [PubMed] [Google Scholar]
- 28. Kerkar SP, Restifo NP. Cellular Constituents of Immune Escape Within the Tumor Microenvironment. Cancer Res (2012) 72(13):3125–30. doi: 10.1158/0008-5472.Can-11-4094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Zou W. Immunosuppressive Networks in the Tumour Environment and Their Therapeutic Relevance. Nat Rev Cancer (2005) 5(4):263–74. doi: 10.1038/nrc1586 [DOI] [PubMed] [Google Scholar]
- 30. Barber DL, Wherry EJ, Masopust D, Zhu B, Allison JP, Sharpe AH, et al. Restoring Function in Exhausted CD8 T Cells During Chronic Viral Infection. Nature (2006) 439(7077):682–7. doi: 10.1038/nature04444 [DOI] [PubMed] [Google Scholar]
- 31. Siddiqui I, Schaeuble K, Chennupati V, Fuertes Marraco SA, Calderon-Copete S, Pais Ferreira D, et al. Intratumoral Tcf1(+)PD-1(+)CD8(+) T Cells With Stem-Like Properties Promote Tumor Control in Response to Vaccination and Checkpoint Blockade Immunotherapy. Immunity (2019) 50(1):195–211.e110. doi: 10.1016/j.immuni.2018.12.021 [DOI] [PubMed] [Google Scholar]
- 32. Gonzalez NM, Zou D, Gu A, Chen W. Schrödinger's T Cells: Molecular Insights Into Stemness and Exhaustion. Front Immunol (2021) 12:725618:725618. doi: 10.3389/fimmu.2021.725618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Chen CY, Ueha S, Ishiwata Y, Shichino S, Yokochi S, Yang D, et al. Combining an Alarmin HMGN1 Peptide With PD-L1 Blockade Results in Robust Antitumor Effects With a Concomitant Increase of Stem-Like/Progenitor Exhausted CD8(+) T Cells. Cancer Immunol Res (2021) 9(10):1214–28. doi: 10.1158/2326-6066.Cir-21-0265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Sun L, Li CW, Chung EM, Yang R, Kim YS, Park AH, et al. Targeting Glycosylated PD-1 Induces Potent Antitumor Immunity. Cancer Res (2020) 80(11):2298–310. doi: 10.1158/0008-5472.Can-19-3133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Efimova I, Catanzaro E, van der Meeren L, Turubanova VD, Hammad H, Mishchenko TA, et al. Vaccination With Early Ferroptotic Cancer Cells Induces Efficient Antitumor Immunity. J Immunother Cancer (2020) 8(2). doi: 10.1136/jitc-2020-001369 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Yamamoto K, Venida A, Yano J, Biancur DE, Kakiuchi M, Gupta S, et al. Autophagy Promotes Immune Evasion of Pancreatic Cancer by Degrading MHC-I. Nature (2020) 581(7806):100–5. doi: 10.1038/s41586-020-2229-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Wang Y, Mohseni M, Grauel A, Diez JE, Guan W, Liang S, et al. SHP2 Blockade Enhances Anti-Tumor Immunity via Tumor Cell Intrinsic and Extrinsic Mechanisms. Sci Rep (2021. 1399) 11(1). doi: 10.1038/s41598-021-80999-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Yang J, Li Z, Shen M, Wang Y, Wang L, Li J, et al. Programmable Unlocking Nano-Matryoshka-CRISPR Precisely Reverses Immunosuppression to Unleash Cascade Amplified Adaptive Immune Response. Adv Sci (Weinh) (2021) 8(13):2100292. doi: 10.1002/advs.202100292 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Aguilar EJ, Ricciuti B, Gainor JF, Kehl KL, Kravets S, Dahlberg S, et al. Outcomes to First-Line Pembrolizumab in Patients With Non-Small-Cell Lung Cancer and Very High PD-L1 Expression. Ann Oncol (2019) 30(10):1653–9. doi: 10.1093/annonc/mdz288 [DOI] [PubMed] [Google Scholar]
- 40. Chism DD. Urothelial Carcinoma of the Bladder and the Rise of Immunotherapy. J Natl Compr Canc Netw (2017) 15(10):1277–84. doi: 10.6004/jnccn.2017.7036 [DOI] [PubMed] [Google Scholar]
- 41. Stege H, Haist M, Nikfarjam U, Schultheis M, Heinz J, Pemler S, et al. The Status of Adjuvant and Neoadjuvant Melanoma Therapy, New Developments and Upcoming Challenges. Target Oncol (2021) 16(5):537–52. doi: 10.1007/s11523-021-00840-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Naidoo J, Wang X, Woo KM, Iyriboz T, Halpenny D, Cunningham J, et al. Pneumonitis in Patients Treated With Anti-Programmed Death-1/Programmed Death Ligand 1 Therapy. J Clin Oncol (2017) 35(7):709–17. doi: 10.1200/jco.2016.68.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Postow MA, Sidlow R, Hellmann MD. Immune-Related Adverse Events Associated With Immune Checkpoint Blockade. N Engl J Med (2018) 378(2):158–68. doi: 10.1056/NEJMra1703481 [DOI] [PubMed] [Google Scholar]
- 44. Freen-van Heeren JJ. Using CRISPR to Enhance T Cell Effector Function for Therapeutic Applications. Cytokine X (2021) 3(1):100049. doi: 10.1016/j.cytox.2020.100049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Ren J, Liu X, Fang C, Jiang S, June CH, Zhao Y. Multiplex Genome Editing to Generate Universal CAR T Cells Resistant to PD1 Inhibition. Clin Cancer Res (2017) 23(9):2255–66. doi: 10.1158/1078-0432.Ccr-16-1300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Robert C, Ribas A, Wolchok JD, Hodi FS, Hamid O, Kefford R, et al. Anti-Programmed-Death-Receptor-1 Treatment With Pembrolizumab in Ipilimumab-Refractory Advanced Melanoma: A Randomised Dose-Comparison Cohort of a Phase 1 Trial. Lancet (2014) 384(9948):1109–17. doi: 10.1016/s0140-6736(14)60958-2 [DOI] [PubMed] [Google Scholar]
- 47. Sun C, Mezzadra R, Schumacher TN. Regulation and Function of the PD-L1 Checkpoint. Immunity (2018) 48(3):434–52. doi: 10.1016/j.immuni.2018.03.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Zielinski CC. A Phase I Study of MEDI4736, NNT-PD-L1 Antibody in Patients With Advanced Solid Tumors. Transl Lung Cancer Res (2014) 3(6):406–7. doi: 10.3978/j.issn.2218-6751.2014.08.07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Chiang CS, Lin YJ, Lee R, Lai YH, Cheng HW, Hsieh CH, et al. Combination of Fucoidan-Based Magnetic Nanoparticles and Immunomodulators Enhances Tumour-Localized Immunotherapy. Nat Nanotechnol (2018) 13(8):746–54. doi: 10.1038/s41565-018-0146-7 [DOI] [PubMed] [Google Scholar]
- 50. Su S, Hu B, Shao J, Shen B, Du J, Du Y, et al. CRISPR-Cas9 Mediated Efficient PD-1 Disruption on Human Primary T Cells From Cancer Patients. Sci Rep (2016) 6:20070. doi: 10.1038/srep20070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Lu S, Yang N, He J, Gong W, Lai Z, Xie L, et al. Generation of Cancer-Specific Cytotoxic PD-1(-) T Cells Using Liposome-Encapsulated CRISPR/Cas System With Dendritic/Tumor Fusion Cells. J BioMed Nanotechnol (2019) 15(3):593–601. doi: 10.1166/jbn.2019.2712 [DOI] [PubMed] [Google Scholar]
- 52. Zhang C, Peng Y, Hublitz P, Zhang H, Dong T. Genetic Abrogation of Immune Checkpoints in Antigen-Specific Cytotoxic T-Lymphocyte as a Potential Alternative to Blockade Immunotherapy. Sci Rep (2018. 5549) 8(1). doi: 10.1038/s41598-018-23803-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Su S, Zou Z, Chen F, Ding N, Du J, Shao J, et al. CRISPR-Cas9-Mediated Disruption of PD-1 on Human T Cells for Adoptive Cellular Therapies of EBV Positive Gastric Cancer. Oncoimmunology (2017) 6(1):e1249558. doi: 10.1080/2162402x.2016.1249558 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Zhao Z, Shi L, Zhang W, Han J, Zhang S, Fu Z, et al. CRISPR Knock Out of Programmed Cell Death Protein 1 Enhances Anti-Tumor Activity of Cytotoxic T Lymphocytes. Oncotarget (2018) 9(4):5208–15. doi: 10.18632/oncotarget.23730 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Luo B, Zhan Y, Luo M, Dong H, Liu J, Lin Y, et al. Engineering of α-PD-1 Antibody-Expressing Long-Lived Plasma Cells by CRISPR/Cas9-Mediated Targeted Gene Integration. Cell Death Dis (2020) 11(11):973. doi: 10.1038/s41419-020-03187-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Pomeroy EJ, Hunzeker JT, Kluesner MG, Lahr WS, Smeester BA, Crosby MR, et al. A Genetically Engineered Primary Human Natural Killer Cell Platform for Cancer Immunotherapy. Mol Ther (2020) 28(1):52–63. doi: 10.1016/j.ymthe.2019.10.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Ohaegbulam KC, Assal A, Lazar-Molnar E, Yao Y, Zang X. Human Cancer Immunotherapy With Antibodies to the PD-1 and PD-L1 Pathway. Trends Mol Med (2015) 21(1):24–33. doi: 10.1016/j.molmed.2014.10.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Yahata T, Mizoguchi M, Kimura A, Orimo T, Toujima S, Kuninaka Y, et al. Programmed Cell Death Ligand 1 Disruption by Clustered Regularly Interspaced Short Palindromic Repeats/Cas9-Genome Editing Promotes Antitumor Immunity and Suppresses Ovarian Cancer Progression. Cancer Sci (2019) 110(4):1279–92. doi: 10.1111/cas.13958 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Zhao L, Luo Y, Huang Q, Cao Z, Yang X. Photo-Enhanced CRISPR/Cas9 System Enables Robust PD-L1 Gene Disruption in Cancer Cells and Cancer Stem-Like Cells for Efficient Cancer Immunotherapy. Small (2020), e2004879. doi: 10.1002/smll.202004879 [DOI] [PubMed] [Google Scholar]
- 60. Ju A, Lee SW, Lee YE, Han KC, Kim JC, Shin SC, et al. A Carrier-Free Multiplexed Gene Editing System Applicable for Suspension Cells. Biomaterials (2019) 217:119298. doi: 10.1016/j.biomaterials.2019.119298 [DOI] [PubMed] [Google Scholar]
- 61. Ghebeh H, Lehe C, Barhoush E, Al-Romaih K, Tulbah A, Al-Alwan M, et al. Doxorubicin Downregulates Cell Surface B7-H1 Expression and Upregulates Its Nuclear Expression in Breast Cancer Cells: Role of B7-H1 as an Anti-Apoptotic Molecule. Breast Cancer Res (2010) 12(4):R48. doi: 10.1186/bcr2605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Wu X, Li Y, Liu X, Chen C, Harrington SM, Cao S, et al. Targeting B7-H1 (PD-L1) Sensitizes Cancer Cells to Chemotherapy. Heliyon (2018) 4(12):e01039. doi: 10.1016/j.heliyon.2018.e01039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Liao Y, Chen L, Feng Y, Shen J, Gao Y, Cote G, et al. Targeting Programmed Cell Death Ligand 1 by CRISPR/Cas9 in Osteosarcoma Cells. Oncotarget (2017) 8(18):30276–87. doi: 10.18632/oncotarget.16326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Hai J, Zhang H, Zhou J, Wu Z, Chen T, Papadopoulos E, et al. Generation of Genetically Engineered Mouse Lung Organoid Models for Squamous Cell Lung Cancers Allows for the Study of Combinatorial Immunotherapy. Clin Cancer Res (2020) 26(13):3431–42. doi: 10.1158/1078-0432.Ccr-19-1627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Zhou L, Mudianto T, Ma X, Riley R, Uppaluri R. Targeting EZH2 Enhances Antigen Presentation, Antitumor Immunity, and Circumvents Anti-PD-1 Resistance in Head and Neck Cancer. Clin Cancer Res (2020) 26(1):290–300. doi: 10.1158/1078-0432.Ccr-19-1351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Hou T, Jiang S, Wang Y, Xie Y, Zhang H, Feng Y, et al. Alpha Thalassemia/Intellectual Disability X-Linked Deficiency Sensitizes Non-Small Cell Lung Cancer to Immune Checkpoint Inhibitors. Front Oncol (2020) 10:608300. doi: 10.3389/fonc.2020.608300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Wan Y, Ge K, Zhou W, Lu J, Jia C, Zhu H. C-X-C Chemokine Receptor 2 (Cxcr2) Promotes Hepatocellular Carcinoma Immune Evasion via Regulating Programmed Death-Ligand 1 (PD-L1). Biol Chem (2021) 402(6):729–37. doi: 10.1515/hsz-2020-0328 [DOI] [PubMed] [Google Scholar]
- 68. Ndiaye PD, Dufies M, Giuliano S, Douguet L, Grépin R, Durivault J, et al. VEGFC Acts as a Double-Edged Sword in Renal Cell Carcinoma Aggressiveness. Theranostics (2019) 9(3):661–75. doi: 10.7150/thno.27794 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Li F, Huang Q, Luster TA, Hu H, Zhang H, Ng WL, et al. In Vivo Epigenetic CRISPR Screen Identifies Asf1a as an Immunotherapeutic Target in Kras-Mutant Lung Adenocarcinoma. Cancer Discovery (2020) 10(2):270–87. doi: 10.1158/2159-8290.Cd-19-0780 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Atefi M, Avramis E, Lassen A, Wong DJ, Robert L, Foulad D, et al. Effects of MAPK and PI3K Pathways on PD-L1 Expression in Melanoma. Clin Cancer Res (2014) 20(13):3446–57. doi: 10.1158/1078-0432.Ccr-13-2797 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Li J, Jie HB, Lei Y, Gildener-Leapman N, Trivedi S, Green T, et al. PD-1/SHP-2 Inhibits Tc1/Th1 Phenotypic Responses and the Activation of T Cells in the Tumor Microenvironment. Cancer Res (2015) 75(3):508–18. doi: 10.1158/0008-5472.Can-14-1215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Xu K, Yin N, Peng M, Stamatiades EG, Shyu A, Li P, et al. Glycolysis Fuels Phosphoinositide 3-Kinase Signaling to Bolster T Cell Immunity. Science (2021) 371(6527):405–10. doi: 10.1126/science.abb2683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Jandova J, Wondrak GT. Genomic GLO1 Deletion Modulates TXNIP Expression, Glucose Metabolism, and Redox Homeostasis While Accelerating Human A375 Malignant Melanoma Tumor Growth. Redox Biol (2021) 39:101838. doi: 10.1016/j.redox.2020.101838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Wang B, Tian T, Kalland KH, Ke X, Qu Y. Targeting Wnt/β-Catenin Signaling for Cancer Immunotherapy. Trends Pharmacol Sci (2018) 39(7):648–58. doi: 10.1016/j.tips.2018.03.008 [DOI] [PubMed] [Google Scholar]
- 75. Ganesh S, Shui X, Craig KP, Park J, Wang W, Brown BD, et al. RNAi-Mediated β-Catenin Inhibition Promotes T Cell Infiltration and Antitumor Activity in Combination With Immune Checkpoint Blockade. Mol Ther (2018) 26(11):2567–79. doi: 10.1016/j.ymthe.2018.09.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. He XY, Ren XH, Peng Y, Zhang JP, Ai SL, Liu BY, et al. Aptamer/Peptide-Functionalized Genome-Editing System for Effective Immune Restoration Through Reversal of PD-L1-Mediated Cancer Immunosuppression. Adv Mater (2020) 32(17):e2000208. doi: 10.1002/adma.202000208 [DOI] [PubMed] [Google Scholar]
- 77. Koike-Yusa H, Li Y, Tan EP, Velasco-Herrera Mdel C, Yusa K. Genome-Wide Recessive Genetic Screening in Mammalian Cells With a Lentiviral CRISPR-Guide RNA Library. Nat Biotechnol (2014) 32(3):267–73. doi: 10.1038/nbt.2800 [DOI] [PubMed] [Google Scholar]
- 78. Wang T, Wei JJ, Sabatini DM, Lander ES. Genetic Screens in Human Cells Using the CRISPR-Cas9 System. Science (2014) 343(6166):80–4. doi: 10.1126/science.1246981 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Chen S, Sanjana NE, Zheng K, Shalem O, Lee K, Shi X, et al. Genome-Wide CRISPR Screen in a Mouse Model of Tumor Growth and Metastasis. Cell (2015) 160(6):1246–60. doi: 10.1016/j.cell.2015.02.038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Huang K, Liu X, Li Y, Wang Q, Zhou J, Wang Y, et al. Genome-Wide CRISPR-Cas9 Screening Identifies NF-κb/E2F6 Responsible for EGFRvIII-Associated Temozolomide Resistance in Glioblastoma. Adv Sci (Weinh) (2019) 6(17):1900782. doi: 10.1002/advs.201900782 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Wei L, Lee D, Law CT, Zhang MS, Shen J, Chin DW, et al. Genome-Wide CRISPR/Cas9 Library Screening Identified PHGDH as a Critical Driver for Sorafenib Resistance in HCC. Nat Commun (2019) 10(1):4681. doi: 10.1038/s41467-019-12606-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Crowther MD, Dolton G, Legut M, Caillaud ME, Lloyd A, Attaf M, et al. Genome-Wide CRISPR-Cas9 Screening Reveals Ubiquitous T Cell Cancer Targeting via the Monomorphic MHC Class I-Related Protein MR1. Nat Immunol (2020) 21(2):178–85. doi: 10.1038/s41590-019-0578-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Dong MB, Wang G, Chow RD, Ye L, Zhu L, Dai X, et al. Systematic Immunotherapy Target Discovery Using Genome-Scale In Vivo CRISPR Screens in CD8 T Cells. Cell (2019) 178(5):1189–1204.e1123. doi: 10.1016/j.cell.2019.07.044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Yu JSL, Yusa K. Genome-Wide CRISPR-Cas9 Screening in Mammalian Cells. Methods (2019) 164-165:29–35. doi: 10.1016/j.ymeth.2019.04.015 [DOI] [PubMed] [Google Scholar]
- 85. Wang Y, Wang F, Wang L, Qiu S, Yao Y, Yan C, et al. NAD(+) Supplement Potentiates Tumor-Killing Function by Rescuing Defective TUB-Mediated NAMPT Transcription in Tumor-Infiltrated T Cells. Cell Rep (2021) 36(6):109516. doi: 10.1016/j.celrep.2021.109516 [DOI] [PubMed] [Google Scholar]
- 86. Zhang JP, Song Z, Wang HB, Lang L, Yang YZ, Xiao W, et al. A Novel Model of Controlling PD-L1 Expression in ALK(+) Anaplastic Large Cell Lymphoma Revealed by CRISPR Screening. Blood (2019) 134(2):171–85. doi: 10.1182/blood.2019001043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Suresh S, Chen B, Zhu J, Golden RJ, Lu C, Evers BM, et al. Eif5b Drives Integrated Stress Response-Dependent Translation of PD-L1 in Lung Cancer. Nat Cancer (2020) 1(5):533–45. doi: 10.1038/s43018-020-0056-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Burr ML, Sparbier CE, Chan YC, Williamson JC, Woods K, Beavis PA, et al. CMTM6 Maintains the Expression of PD-L1 and Regulates Anti-Tumour Immunity. Nature (2017) 549(7670):101–5. doi: 10.1038/nature23643 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Wroblewska A, Dhainaut M, Ben-Zvi B, Rose SA, Park ES, Amir ED, et al. Protein Barcodes Enable High-Dimensional Single-Cell CRISPR Screens. Cell (2018) 175(4):1141–1155.e1116. doi: 10.1016/j.cell.2018.09.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Kriegsman BA, Vangala P, Chen BJ, Meraner P, Brass AL, Garber M, et al. Frequent Loss of IRF2 in Cancers Leads to Immune Evasion Through Decreased MHC Class I Antigen Presentation and Increased PD-L1 Expression. J Immunol (2019) 203(7):1999–2010. doi: 10.4049/jimmunol.1900475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Sreevalsan S, Döring M, Paszkowski-Rogacz M, Brux M, Blanck C, Meyer M, et al. MLLT6 Maintains PD-L1 Expression and Mediates Tumor Immune Resistance. EMBO Rep (2020) 21(12):e50155. doi: 10.15252/embr.202050155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Hou J, Wang Y, Shi L, Chen Y, Xu C, Saeedi A, et al. Integrating Genome-Wide CRISPR Immune Screen With Multi-Omic Clinical Data Reveals Distinct Classes of Tumor Intrinsic Immune Regulators. J Immunother Cancer (2021) 9(2). doi: 10.1136/jitc-2020-001819 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Shi S, Gu S, Han T, Zhang W, Huang L, Li Z, et al. Inhibition of MAN2A1 Enhances the Immune Response to Anti-PD-L1 in Human Tumors. Clin Cancer Res (2020) 26(22):5990–6002. doi: 10.1158/1078-0432.Ccr-20-0778 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Williams JB, Li S, Higgs EF, Cabanov A, Wang X, Huang H, et al. Tumor Heterogeneity and Clonal Cooperation Influence the Immune Selection of IFN-γ-Signaling Mutant Cancer Cells. Nat Commun (2020) 11(1):602. doi: 10.1038/s41467-020-14290-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Devine SM, Larson RA. Acute Leukemia in Adults: Recent Developments in Diagnosis and Treatment. CA Cancer J Clin (1994) 44(6):326–52. doi: 10.3322/canjclin.44.6.326 [DOI] [PubMed] [Google Scholar]
- 96. John AM, Thomas NS, Mufti GJ, Padua RA. Targeted Therapies in Myeloid Leukemia. Semin Cancer Biol (2004) 14(1):41–62. doi: 10.1016/j.semcancer.2003.11.006 [DOI] [PubMed] [Google Scholar]
- 97. Maude SL, Frey N, Shaw PA, Aplenc R, Barrett DM, Bunin NJ, et al. Chimeric Antigen Receptor T Cells for Sustained Remissions in Leukemia. N Engl J Med (2014) 371(16):1507–17. doi: 10.1056/NEJMoa1407222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Skorka K, Ostapinska K, Malesa A, Giannopoulos K. The Application of CAR-T Cells in Haematological Malignancies. Arch Immunol Ther Exp (Warsz) (2020) 68(6):34. doi: 10.1007/s00005-020-00599-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Singh N, Shi J, June CH, Ruella M. Genome-Editing Technologies in Adoptive T Cell Immunotherapy for Cancer. Curr Hematol Malig Rep (2017) 12(6):522–9. doi: 10.1007/s11899-017-0417-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Ren J, Zhang X, Liu X, Fang C, Jiang S, June CH, et al. A Versatile System for Rapid Multiplex Genome-Edited CAR T Cell Generation. Oncotarget (2017) 8(10):17002–11. doi: 10.18632/oncotarget.15218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Pawlyn C, Davies FE. Toward Personalized Treatment in Multiple Myeloma Based on Molecular Characteristics. Blood (2019) 133(7):660–75. doi: 10.1182/blood-2018-09-825331 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Pardoll DM. The Blockade of Immune Checkpoints in Cancer Immunotherapy. Nat Rev Cancer (2012) 12(4):252–64. doi: 10.1038/nrc3239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Brudno JN, Maric I, Hartman SD, Rose JJ, Wang M, Lam N, et al. T Cells Genetically Modified to Express an Anti-B-Cell Maturation Antigen Chimeric Antigen Receptor Cause Remissions of Poor-Prognosis Relapsed Multiple Myeloma. J Clin Oncol (2018) 36(22):2267–80. doi: 10.1200/jco.2018.77.8084 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Lee KM, Giltnane JM, Balko JM, Schwarz LJ, Guerrero-Zotano AL, Hutchinson KE, et al. MYC and MCL1 Cooperatively Promote Chemotherapy-Resistant Breast Cancer Stem Cells via Regulation of Mitochondrial Oxidative Phosphorylation. Cell Metab (2017) 26(4):633–647.e637. doi: 10.1016/j.cmet.2017.09.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Deng H, Tan S, Gao X, Zou C, Xu C, Tu K, et al. Cdk5 Knocking Out Mediated by CRISPR-Cas9 Genome Editing for PD-L1 Attenuation and Enhanced Antitumor Immunity. Acta Pharm Sin B (2020) 10(2):358–73. doi: 10.1016/j.apsb.2019.07.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Kumar S, Davra V, Obr AE, Geng K, Wood TL, De Lorenzo MS, et al. Crk Adaptor Protein Promotes PD-L1 Expression, EMT and Immune Evasion in a Murine Model of Triple-Negative Breast Cancer. Oncoimmunology (2017) 7(1):e1376155. doi: 10.1080/2162402x.2017.1376155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Burd EM. Human Papillomavirus and Cervical Cancer. Clin Microbiol Rev (2003) 16(1):1–17. doi: 10.1128/cmr.16.1.1-17.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Zhen S, Lu J, Liu YH, Chen W, Li X. Synergistic Antitumor Effect on Cervical Cancer by Rational Combination of PD1 Blockade and CRISPR-Cas9-Mediated HPV Knockout. Cancer Gene Ther (2020) 27(3-4):168–78. doi: 10.1038/s41417-019-0131-9 [DOI] [PubMed] [Google Scholar]
- 109. Narod S. Can Advanced-Stage Ovarian Cancer Be Cured? Nat Rev Clin Oncol (2016) 13(4):255–61. doi: 10.1038/nrclinonc.2015.224 [DOI] [PubMed] [Google Scholar]
- 110. Kim MS, Ma S, Chelariu-Raicu A, Leuschner C, Alila HW, Lee S, et al. Enhanced Immunotherapy With LHRH-R Targeted Lytic Peptide in Ovarian Cancer. Mol Cancer Ther (2020) 19(11):2396–406. doi: 10.1158/1535-7163.Mct-20-0030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Zhu Q, Ma Y, Liang J, Wei Z, Li M, Zhang Y, et al. AHR Mediates the Aflatoxin B1 Toxicity Associated With Hepatocellular Carcinoma. Signal Transduct Target Ther (2021) 6(1):299. doi: 10.1038/s41392-021-00713-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Guo X, Jiang H, Shi B, Zhou M, Zhang H, Shi Z, et al. Disruption of PD-1 Enhanced the Anti-Tumor Activity of Chimeric Antigen Receptor T Cells Against Hepatocellular Carcinoma. Front Pharmacol (2018. 1118) 9:1118. doi: 10.3389/fphar.2018.01118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Zhou J, Liu M, Sun H, Feng Y, Xu L, Chan AWH, et al. Hepatoma-Intrinsic CCRK Inhibition Diminishes Myeloid-Derived Suppressor Cell Immunosuppression and Enhances Immune-Checkpoint Blockade Efficacy. Gut (2018) 67(5):931–44. doi: 10.1136/gutjnl-2017-314032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Wang Y, Cao K. KDM1A Promotes Immunosuppression in Hepatocellular Carcinoma by Regulating PD-L1 Through Demethylating MEF2D. J Immunol Res (2021) 2021:9965099. doi: 10.1155/2021/9965099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Huang K, Sun B, Luo N, Guo H, Hu J, Peng J. Programmed Death Receptor 1 (PD1) Knockout and Human Telomerase Reverse Transcriptase (hTERT) Transduction Can Enhance Persistence and Antitumor Efficacy of Cytokine-Induced Killer Cells Against Hepatocellular Carcinoma. Med Sci Monit (2018) 24:4573–82. doi: 10.12659/msm.910903 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB. Colorectal Cancer. Lancet (2019) 394(10207):1467–80. doi: 10.1016/s0140-6736(19)32319-0 [DOI] [PubMed] [Google Scholar]
- 117. Dienstmann R, Vermeulen L, Guinney J, Kopetz S, Tejpar S, Tabernero J. Consensus Molecular Subtypes and the Evolution of Precision Medicine in Colorectal Cancer. Nat Rev Cancer (2017) 17(2):79–92. doi: 10.1038/nrc.2016.126 [DOI] [PubMed] [Google Scholar]
- 118. Xiao Y, Freeman GJ. The Microsatellite Instable Subset of Colorectal Cancer Is a Particularly Good Candidate for Checkpoint Blockade Immunotherapy. Cancer Discovery (2015) 5(1):16–8. doi: 10.1158/2159-8290.Cd-14-1397 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Liao TT, Lin CC, Jiang JK, Yang SH, Teng HW, Yang MH. Harnessing Stemness and PD-L1 Expression by AT-Rich Interaction Domain-Containing Protein 3B in Colorectal Cancer. Theranostics (2020) 10(14):6095–112. doi: 10.7150/thno.44147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Gettinger S, Choi J, Hastings K, Truini A, Datar I, Sowell R, et al. Impaired HLA Class I Antigen Processing and Presentation as a Mechanism of Acquired Resistance to Immune Checkpoint Inhibitors in Lung Cancer. Cancer Discov (2017) 7(12):1420–35. doi: 10.1158/2159-8290.Cd-17-0593 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Zhang X, Sabio E, Krishna C, Ma X, Wang J, Jiang H, et al. Qa-1(B) Modulates Resistance to Anti-PD-1 Immune Checkpoint Blockade in Tumors With Defects in Antigen Processing. Mol Cancer Res (2021) 19(6):1076–84. doi: 10.1158/1541-7786.Mcr-20-0652 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Marotte L, Simon S, Vignard V, Dupre E, Gantier M, Cruard J, et al. Increased Antitumor Efficacy of PD-1-Deficient Melanoma-Specific Human Lymphocytes. J Immunother Cancer (2020) 8(1). doi: 10.1136/jitc-2019-000311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Harel M, Ortenberg R, Varanasi SK, Mangalhara KC, Mardamshina M, Markovits E, et al. Proteomics of Melanoma Response to Immunotherapy Reveals Mitochondrial Dependence. Cell (2019) 179(1):236–250.e218. doi: 10.1016/j.cell.2019.08.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Miller JF, Sadelain M. The Journey From Discoveries in Fundamental Immunology to Cancer Immunotherapy. Cancer Cell (2015) 27(4):439–49. doi: 10.1016/j.ccell.2015.03.007 [DOI] [PubMed] [Google Scholar]
- 125. Marofi F, Motavalli R, Safonov VA, Thangavelu L, Yumashev AV, Alexander M, et al. CAR T Cells in Solid Tumors: Challenges and Opportunities. Stem Cell Res Ther (2021) 12(1):81. doi: 10.1186/s13287-020-02128-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Jackson HJ, Rafiq S, Brentjens RJ. Driving CAR T-Cells Forward. Nat Rev Clin Oncol (2016) 13(6):370–83. doi: 10.1038/nrclinonc.2016.36 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Zhang C, Liu J, Zhong JF, Zhang X. Engineering CAR-T Cells. Biomark Res (2017) 5:22. doi: 10.1186/s40364-017-0102-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Stadtmauer EA, Fraietta JA, Davis MM, Cohen AD, Weber KL, Lancaster E, et al. CRISPR-Engineered T Cells in Patients With Refractory Cancer. Science (2020) 367(6481). doi: 10.1126/science.aba7365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Lu Y, Xue J, Deng T, Zhou X, Yu K, Deng L, et al. Safety and Feasibility of CRISPR-Edited T Cells in Patients With Refractory Non-Small-Cell Lung Cancer. Nat Med (2020) 26(5):732–40. doi: 10.1038/s41591-020-0840-5 [DOI] [PubMed] [Google Scholar]
- 130. Wang Z, Li N, Feng K, Chen M, Zhang Y, Liu Y, et al. Phase I Study of CAR-T Cells With PD-1 and TCR Disruption in Mesothelin-Positive Solid Tumors. Cell Mol Immunol (2021). doi: 10.1038/s41423-021-00749-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Lacey SF, Fraietta JA. First Trial of CRISPR-Edited T Cells in Lung Cancer. Trends Mol Med (2020) 26(8):713–5. doi: 10.1016/j.molmed.2020.06.001 [DOI] [PubMed] [Google Scholar]
- 132. Choi BD, Yu X, Castano AP, Darr H, Henderson DB, Bouffard AA, et al. CRISPR-Cas9 Disruption of PD-1 Enhances Activity of Universal EGFRvIII CAR T Cells in a Preclinical Model of Human Glioblastoma. J Immunother Cancer (2019) 7(1):304. doi: 10.1186/s40425-019-0806-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Nakazawa T, Natsume A, Nishimura F, Morimoto T, Matsuda R, Nakamura M, et al. Effect of CRISPR/Cas9-Mediated PD-1-Disrupted Primary Human Third-Generation CAR-T Cells Targeting EGFRvIII on In Vitro Human Glioblastoma Cell Growth. Cells (2020) 9(4). doi: 10.3390/cells9040998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. Hu B, Zou Y, Zhang L, Tang J, Niedermann G, Firat E, et al. Nucleofection With Plasmid DNA for CRISPR/Cas9-Mediated Inactivation of Programmed Cell Death Protein 1 in CD133-Specific CAR T Cells. Hum Gene Ther (2019) 30(4):446–58. doi: 10.1089/hum.2017.234 [DOI] [PubMed] [Google Scholar]
- 135. Hu W, Zi Z, Jin Y, Li G, Shao K, Cai Q, et al. CRISPR/Cas9-Mediated PD-1 Disruption Enhances Human Mesothelin-Targeted CAR T Cell Effector Functions. Cancer Immunol Immunother (2019) 68(3):365–77. doi: 10.1007/s00262-018-2281-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Rupp LJ, Schumann K, Roybal KT, Gate RE, Ye CJ, Lim WA, et al. CRISPR/Cas9-Mediated PD-1 Disruption Enhances Anti-Tumor Efficacy of Human Chimeric Antigen Receptor T Cells. Sci Rep (2017) 7(1):737. doi: 10.1038/s41598-017-00462-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Shen CC, Hsu MN, Chang CW, Lin MW, Hwu JR, Tu Y, et al. Synthetic Switch to Minimize CRISPR Off-Target Effects by Self-Restricting Cas9 Transcription and Translation. Nucleic Acids Res (2019) 47(3):e13. doi: 10.1093/nar/gky1165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Haapaniemi E, Botla S, Persson J, Schmierer B, Taipale J. CRISPR-Cas9 Genome Editing Induces a P53-Mediated DNA Damage Response. Nat Med (2018) 24(7):927–30. doi: 10.1038/s41591-018-0049-z [DOI] [PubMed] [Google Scholar]
- 139. Wilbie D, Walther J, Mastrobattista E. Delivery Aspects of CRISPR/Cas for In Vivo Genome Editing. Acc Chem Res (2019) 52(6):1555–64. doi: 10.1021/acs.accounts.9b00106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Ferdosi SR, Ewaisha R, Moghadam F, Krishna S, Park JG, Ebrahimkhani MR, et al. Multifunctional CRISPR-Cas9 With Engineered Immunosilenced Human T Cell Epitopes. Nat Commun (2019) 10(1):1842. doi: 10.1038/s41467-019-09693-x [DOI] [PMC free article] [PubMed] [Google Scholar]