Abstract
Human rhinoviruses (HRV) are recognized as the major etiologic agents of the common cold. Starting from the observation that local hyperthermic treatment is beneficial in patients with natural and experimental common colds, we have studied the effect of brief hyperthermic treatment (HT) on HRV replication in HeLa cells. We report that a 20-min HT at 45°C is effective in suppressing HRV multiplication by more than 90% when applied at specific stages of the virus replication cycle. Synthesis of virus proteins is not affected by HT, indicating that the target for treatment is a posttranslational event. The antiviral effect is a transient cell-mediated event and is associated with the synthesis of the 70-kDa heat shock protein hsp70. Unlike poliovirus, rhinovirus infection does not inhibit the expression of hsp70 induced by heat. The possibility that hsp70 could play a role in the control of rhinovirus replication is suggested by the fact that a different class of HSP inducers, the cyclopentenone prostaglandins PGA1 and Δ12-PGJ2, were also effective in inhibiting HRV replication in HeLa cells. Inhibition of hsp70 expression by actinomycin D prevented the antiviral activity of prostaglandins in HRV-infected cells. These results indicate that the beneficial effect of respiratory hyperthermia may be mediated by the induction of a cytoprotective heat shock response in rhinovirus-infected cells.
The human rhinoviruses (HRVs), members of the Picornaviridae family, are the major etiologic agents of the common cold (29). They include over 100 immunologically non-cross-reactive serotypes, classified into a minor and a major group according to membrane receptor recognition (5, 32). HRVs contain four nonglycosylated structural proteins, VP1, VP2, VP3, and VP4, forming a capsid with icosahedral symmetry. Within the capsid lies a single-stranded RNA genome which serves as a monocistronic mRNA for the synthesis of the structural and nonstructural proteins of the virus. Upon entry into the host cell, the viral RNA is translated into a large polyprotein which is subsequently cleaved by virus-encoded proteases (19). Unlike other types of picornaviruses, the human rhinoviruses are adversely affected by acidic pH and replicate optimally at 33°C or colder. This may partly account for their predilection for the cooler environment of the nasal mucosa, limiting rhinoviruses to upper respiratory infections.
Although there is an abundance of remedies for the common cold from nasal vasoconstrictors to vitamin C, no specific antiviral therapy has been found to be effective. Also, the large variety of immunologically non-cross-reactive rhinovirus serotypes and apparent antigenic drift in rhinoviral antigens cause major problems for the development of an effective vaccine (5). A different approach against rhinovirus infection was reported by Tyrrell et al., who demonstrated a beneficial role for local hyperthermia (20 to 30 min at 43°C) in improving the course of the disease in clinical trials in patients with natural and experimental common colds (31). In this case, brief hyperthermic treatment (HT) did not alter the frequency of antibody response in volunteers, suggesting either a direct effect of HT on rhinovirus replication or an inhibitory effect on inflammatory processes and the ensuing symptomatology. However, the mechanism of the anti-HRV activity of hyperthermia is not known.
Eukaryotic and prokaryotic cells respond to an increase in environmental temperature by expressing a specific set of cytoprotective proteins referred to as heat shock proteins (HSP) or stress proteins (13). HSP are utilized in the repair process following different types of injury to prevent damage resulting from the accumulation of nonnative proteins. In mammalian cells, HSP are induced in a variety of pathophysiological conditions, including fever, inflammation, oxidant injury, and virus infection (7). Induction requires the activation, translocation to the nucleus, and phosphorylation of a transregulatory protein, the heat shock transcription factor HSF (17). The 70-kDa heat shock proteins (hsp70) function as molecular chaperones and are encoded by a multigene family, including the constitutively expressed hsc70, the major inducible hsp70, the inducible hsp72, the glucose-regulated grp78/BiP, and the mithocondrial hsp75 (7, 13). A cytoprotective role of hsp70 in a variety of human diseases, including ischemia, inflammation, and infection, is widely recognized (7, 17).
In the case of viral infection, evidence for the presence of HSP in intact virions or association of HSP with virus proteins during infection, as well as for the modulation of HSP synthesis by viruses, has been reported (23). However, the role of HSP in viral infection is still controversial. The possibility that elevated levels of hsp70 may interfere with viral replication has been suggested by a variety of studies describing the antiviral activity of cyclopentenone prostaglandins and other inducers of the heat shock response in negative-strand RNA viruses (reviewed in references 23 and 24). In the case of picornaviruses, induction of the heat shock response has been studied mainly during poliovirus infection. Constitutive hsp70 was shown to be associated with newly synthesized capsid precursor P1 of poliovirus, and the hsp70-P1 complex was found to be part of an assembly intermediate (14). On the other hand, poliovirus infection was shown to inhibit constitutive or heat shock-induced hsp70 synthesis starting 2 to 3 h after infection (14, 18). Infection with poliovirus type 2 was recently shown to prevent HSP induction also by cyclopentenone prostaglandins (4).
Starting from the observation that HT is beneficial in patients with common colds, we have studied the induction of the heat shock response by hyperthermia and cyclopentenone prostaglandins during HRV infection in human cells. We provide evidence that, unlike poliovirus, rhinovirus infection does not inhibit the expression of hsp70 induced by heat or cyclopentenone prostaglandins and that both hyperthermia and prostaglandin treatment result in inhibition of HRV replication. The antiviral effect is a transient cell-mediated event, associated with hsp70 synthesis.
MATERIALS AND METHODS
Cell cultures.
HeLa (Ohio) cells were grown at 37°C in a 5% CO2 atmosphere in Eagle’s minimal essential medium (MEM) supplemented with 10% fetal calf serum (FCS), 2 mM glutamine, 100 IU of penicillin G per ml, and 100 μg of streptomycin per ml.
Virus infection and titration.
Confluent HeLa cell monolayers were infected with HRV serotype 1B or 14 (3) for 1 h at 33°C at a multiplicity of infection (MOI) of 1, 5, or 10 PFU/cell. The viral inoculum was removed, and cell monolayers were washed three times with phosphate-buffered saline (PBS) and incubated with MEM containing 2% FCS at 33°C. Prostaglandin A1 (PGA1) and 9-deoxy-Δ9, Δ12-13,14-dihydro-prostaglandin D2 (Δ12-PGJ2) (Cayman Chemical Co.) were stored in absolute ethanol and diluted to the appropriate concentration at the time of use. Control media contained the same concentration of ethanol diluent, which did not affect cell metabolism or virus replication. For the heating procedure, flasks were immersed in a temperature-controlled water bath (Grant Instruments) for 20 min at 45 ± 0.01°C, unless specified otherwise. HT (45°C, 20 min) was not cytotoxic to uninfected HeLa cells as shown by the trypan blue exclusion technique 24 h after heat shock (data not shown). Virus production was determined by plaque assay as described previously (3). Briefly, after three cycles of freezing and thawing, serial 10-fold dilutions of HRV were prepared and inoculated on confluent HeLa cell monolayers in 35-mm-diameter plates. After 1 h at 33°C, the inoculum was removed, and cells were washed three times with PBS before the addition of MEM containing 2% FCS and 1% SeaPlaque agarose (Miles). After 3 days of incubation at 33°C in a 5% CO2 atmosphere, plaques were stained with 0.33% neutral red solution. For virus purification, HeLa cells infected with 10 PFU of HRV serotype 1B were labeled with [35S]methionine (25 μCi/ml/5 × 105 cells, 20-h pulse) in the presence of actinomycin D (0.5 μg/ml) starting 5 h postinfection (p.i.). After three cycles of freezing-thawing and clarification at 6,000 × g for 20 min at 4°C, the supernatants diluted in PBS were centrifuged at 12,000 × g for 20 min and the virus was pelleted by centrifugation at 100,000 × g for 4 h at 4°C. Unless otherwise specified, HRV serotype 1B was utilized for the experimental protocols.
DNA, RNA, and protein synthesis.
Confluent monolayers of uninfected or virus-infected HeLa cells (10 PFU/cell) were labeled for 12 h, starting soon after virus infection, with [3H]thymidine, [3H]uridine, or [35S]methionine (Amersham International) at a concentration of 5 μCi/5 × 105 cells for DNA, RNA, or protein synthesis, respectively, and the radioactivity incorporated into trichloroacetic acid (TCA)-soluble (uptake) and -insoluble (incorporation) material was determined as described previously (16).
Protein synthesis and SDS-PAGE analysis.
Confluent cell monolayers were labeled with [35S]methionine (1-h pulse, 5 μCi/ml/5 × 105 cells) at hourly intervals from 0 to 11 h p.i. in methionine-free medium containing 2% dialyzed FCS. Cells were usually preincubated for 15 min in methionine-free medium. After labeling, cells were washed and lysed in lysis buffer (2% sodium dodecyl sulfate [SDS], 10% glycerol, 0.001% bromophenol blue, 0.1 M dithiothreitol, 0.0625 M Tris HCl [pH 6.8]) and the radioactivity incorporated into TCA-insoluble material was determined. Samples were analyzed by SDS-polyacrylamide gel electrophoresis (SDS-PAGE) in a vertical slab gel apparatus (3% stacking gel, 10 or 12% resolving gel) and processed for autoradiography, as described previously (2). Autoradiograms were quantified densitometrically with a laser beam densitometer (Ultroscan XL; LKB) (2), and bands were expressed as relative peak areas. Virus proteins were identified on the basis of Mr and in relation to the position of viral marker proteins from [35S]methionine-labeled purified HRV serotype 1B.
Immunoblot analysis.
For immunoblot analysis, an equal amount of protein from each sample was separated by SDS-PAGE and blotted onto nitrocellulose, as described previously (27). After transfer, the filters were incubated with anti-hsp70 monoclonal antibodies (diluted 1:500) from HeLa cells (Amersham) in Ten-Tween 20 buffer (0.05 M Tris-HCl [pH 7.4], 5 mM EDTA, 0.15 M NaCl, 0.05% Tween 20), and the bound antibody was detected by using horseradish peroxidase-linked sheep antimouse antibody (Amersham International). Molecular weights were calculated by using Bio-Rad low-Mr markers.
Statistical analysis.
Statistical analyses were performed by using Student’s test for unpaired data. Data are expressed as the means ± standard deviations (SDs) of at least duplicate samples. P values of <0.05 were considered significant.
RESULTS
Inhibition of HRV replication by brief HT in HeLa cells.
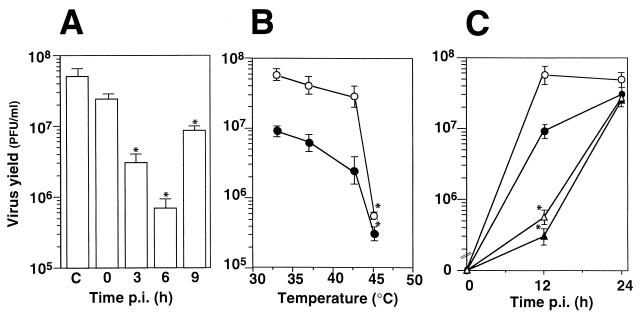
The effect of brief HT on rhinovirus production in human cells was evaluated under one-step growth conditions. HeLa cells infected with HRV serotype 1B (10 PFU/cell) were subjected to a 45°C HT (20 min) soon after the adsorption period (time 0) or at 3, 6, and 9 h p.i. Cells were incubated at 33°C, and virus yield was quantitated by plaque assay at 12 h p.i. Figure 1A shows that HT, when applied in specific stages of the virus cycle, was strongly effective in inhibiting HRV replication for at least 12 h. The most dramatic effect was observed at 6 h p.i., with a reduction in virus yield of more than 99% relative to that of the control. HT applied at later times of the virus growth cycle (9 h p.i.) resulted in a decreased inhibitory effect (approximately 80% reduction relative to the control), whereas treatment soon after infection (time 0) had no significant effect on virus yield, indicating that inhibition of rhinovirus replication is not due to an aspecific cytotoxic effect of heat.
FIG. 1.
Effect of HT during HRV replication in human cells. (A) HeLa cell monolayers were infected with HRV serotype 1B (10 PFU/cell) and subjected to HT (45°C, 20 min) soon after the adsorption period (time 0) or at 3, 6, or 9 h p.i. Virus titers were determined 12 h p.i. Bar C, untreated cells. Data represent the mean ± SD of duplicate samples of two independent experiments. (B) Temperature-dependent inhibition of HRV replication. HeLa cells were infected with HRV at 1 (●) or 10 ( ) PFU/cell and either kept at 33°C or subjected to 20 min of HT at different temperatures (37, 43, or 45°C) 6 h after infection. Virus titers were determined 12 h p.i. (C) HeLa cells were infected with HRV at 1 (● and ▴) or 10 ( and ▵) PFU/cell and either kept at 33°C ( and ●) or subjected to 20 min of HT at 45°C (▵ and ▴) 6 h after infection. Virus titers were determined 12 or 24 h p.i. Data represent the mean ± SD of at least duplicate samples. ∗, P < 0.05.
To investigate whether this effect was temperature dependent and was influenced by the MOI of the virus, confluent HeLa monolayers were infected with HRV at an MOI of 1 or 10 PFU/cell for 1 h at 33°C and, at 6 h p.i., were subjected to HT at 37, 43, or 45°C for 20 min. Inhibition of virus replication, determined 12 h p.i. by plaque assay, was found to be temperature dependent, and a 2-log reduction in HRV yield was obtained after treatment at 45°C (Fig. 1B). Heat treatment at 43°C appeared to be less effective in cells infected with 10 PFU of HRV (approximately 40% reduction in virus yield relative to untreated control) than in cells infected with 1 PFU (∼70% reduction), whereas heat treatment at 45°C was similarly effective at low and high MOIs (Fig. 1B). Inhibition of virus replication by HT is transient. In fact, when virus yields from parallel cultures were measured at 24 h p.i., virus titers of heat-stressed cells were comparable to that of untreated control cells (Fig. 1C), indicating that brief HT results in the delay, but not in the irreversible block, of HRV replication. The fact that inhibition of virus replication is transient further indicates that the reduction in virus yield is not caused by an aspecific irreversible cytotoxic effect of hyperthermia in HeLa cells.
To investigate whether the antirhinoviral activity of hyperthermia was a general effect or was specific for serotype 1B which belongs to the HRV minor group according to membrane receptor recognition, the effect of HT was tested on HRV serotype 14, a representative member of the major HRV group (32). HeLa cells infected with HRV serotype 14 (1 PFU/cell) were subjected to a 43°C HT (20 min) at 6 h p.i. Cells were incubated at 33°C, and virus yield was quantitated by plaque assay at 12 h p.i. HT was found to reduce the HRV serotype 14 yield by more than 90% at this time (control, 1.50 × 106 ± 0.40 × 106 PFU/ml; HT, 1.36 × 105 ± 0.50 × 105 PFU/ml).
Effect of brief HT on cellular and viral protein synthesis.
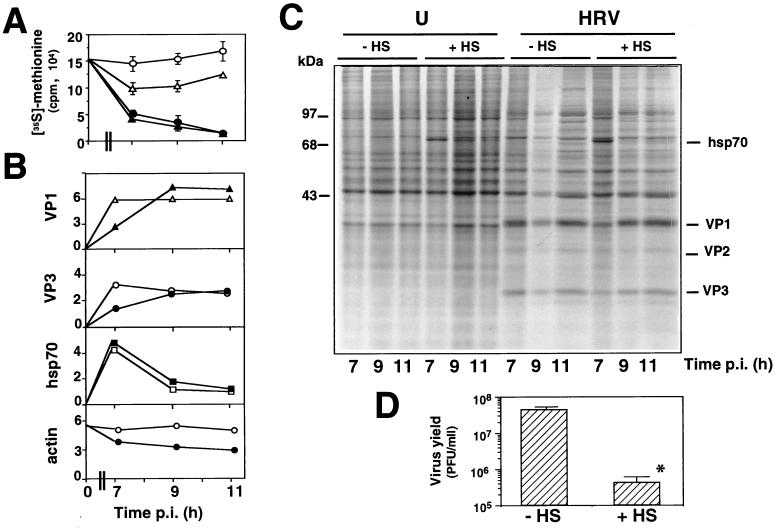
Inhibition of virus replication after HT has been previously associated with induction of HSP (6, 23). On the other hand, it has been shown that infection with different members of the Picornaviridae family, the polioviruses, prevents the expression of HSP stimulated by hyperthermia or chemical inducers of the heat shock response (4, 18). To investigate whether HRV infection could interfere with HSP expression and whether brief HT would affect HRV protein synthesis, HeLa cells infected with HRV (10 PFU/cell) were either kept at 33°C or subjected to HT (45°C, 20 min) at 6 h p.i. After a 1-h recovery period at 33°C, cells were labeled with [35S]methionine (1-h pulses at 33°C) at different times p.i. Uninfected cells were treated identically. Virus yield was quantitated by plaque assay at 12 h p.i. HT caused the expected reduction of virus yield at 12 h p.i. (Fig. 2D). As determined by [35S]methionine incorporation into TCA-insoluble material, heat stress was found to moderately (<30%) inhibit protein synthesis in uninfected HeLa cells for a period of approximately 3 h (Fig. 2A). Under the conditions described, rhinovirus infection caused progressive inhibition of HeLa cell protein synthesis, and no difference between untreated and HT-treated cells was detected (Fig. 2A). Samples containing an equal amount of radioactivity were processed for SDS-PAGE analysis and autoradiography. As expected, in uninfected HeLa cells, HT induced the synthesis of a 72-kDa protein, identified as hsp70 by Western blot analysis with anti-hsp70 monoclonal antibodies (data not shown). hsp70 synthesis started 1 h after heat shock and continued at a lower level for the following 3 to 4 h (Fig. 2B and C). HRV infection did not induce HSP synthesis in HeLa cells. HRV-infected cells were able to respond to HT by synthesizing hsp70 in an amount comparable to that of uninfected cells (Fig. 2B and C), indicating that rhinoviruses, unlike polioviruses, do not interfere with HSP expression even in relatively late stages of infection in human cells. Levels of actin synthesis were instead decreased in HRV-infected cells (Fig. 2B). Finally, even though it greatly reduced virus yield, HT caused only a modest inhibition of HRV protein synthesis at 7 h p.i., whereas no difference in viral protein synthesis was detected between untreated and HT-treated cells at later times of infection (Fig. 2B and C), suggesting that HT could affect a posttranslational event in the replication cycle, possibly by interfering with the virus assembly process or with virus release from infected cells.
FIG. 2.
Effect of HT on HRV protein synthesis. Cell monolayers infected with HRV (10 PFU/cell) were either kept at 33°C or subjected to HT (45°C, 20 min) 6 h after the adsorption period and labeled with [35S]methionine (1-h pulse) at 33°C at different times p.i. (A) Determination of radioactivity incorporated into TCA-insoluble material in uninfected ( and ▵) and infected (● and ▴) cells either untreated ( and ●) or treated with HT (▵ and ▴). (B) Quantitative determination of viral proteins VP1 (▵ and ▴) and VP3 ( and ●), as measured by densitometric analysis of autoradiographic patterns shown in panel C. Data are expressed as percentage of total proteins in untreated ( and ▵) or HT-treated (● and ▴) cells. Quantitative determination of hsp70 in uninfected (□) and HRV-infected (■) cells is also shown. Levels of actin in uninfected ( ) and HRV-infected (●) untreated cells are shown as a control. (C) SDS-PAGE analysis and autoradiography of samples containing an equal amount of radioactivity from uninfected (U) and HRV-infected (HRV) HeLa cells, maintained at 33°C (−HS) or subjected to HT (+HS). hsp70 and HRV proteins VP1, VP2, and VP3 are indicated. (D) In the same experiment, virus titers were determined 12 h p.i. by plaque assay. Data represent the mean ± SD of duplicate samples. ∗, P < 0.05.
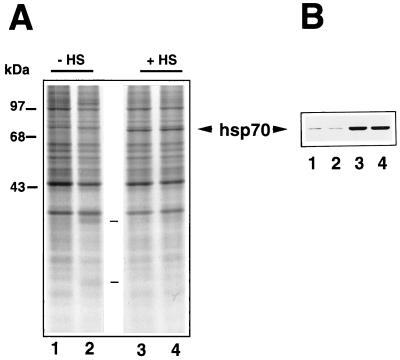
In a parallel experiment, HeLa cells were infected with HRV at a low MOI (1 PFU/cell) and, after the 1-h adsorption period, were either kept at 33°C or subjected to HT (45°C, 20 min) at 6 h p.i. After a 1-h recovery period at 33°C, the cells were labeled with [35S]methionine for the following 2 h at 33°C. Uninfected cells were treated identically. Samples containing an equal amount of radioactivity were processed for SDS-PAGE analysis and autoradiography. As shown in Fig. 3A, the levels of hsp70 synthesis were comparable in uninfected and HRV-infected cells. For hsp70 identification, equal amounts of protein from each sample were separated by SDS-PAGE and processed for immunoblot analysis using anti-hsp70 monoclonal antibodies (Fig. 3B). Although virus proteins were not detectable by SDS-PAGE under these conditions, the synthesis of two polypeptides (indicated in Fig. 3A), whose identification is presently under investigation, was evident in untreated, but not in HT-treated, infected cells.
FIG. 3.
Effect of HT on hsp70 synthesis in HRV-infected cells. Cell monolayers either uninfected (lanes 1 and 3) or infected with HRV at a low MOI (1 PFU/cell) (lanes 2 and 4) were kept at 33°C (lanes 1 and 2; −HS) or subjected to HT (45°C, 20 min) (lanes 3 and 4; +HS) 6 h after the adsorption period and labeled with [35S]methionine (2-h pulse) at 33°C 1 h after heat treatment. (A) Samples containing an equal amount of radioactivity were separated by SDS-PAGE analysis and processed for autoradiography. (B) Hsp70 was identified by immunoblot analysis using monoclonal anti-hsp70 antibodies.
Effect of PGA1 on rhinovirus infection.
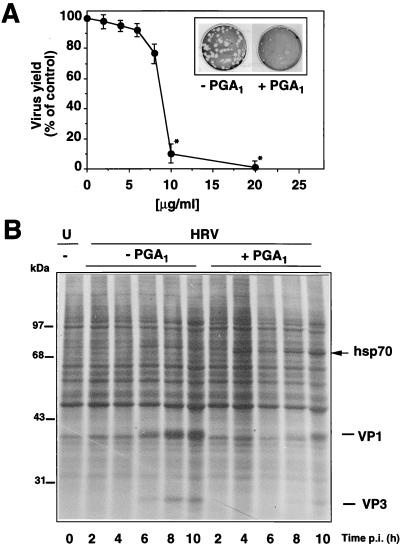
To investigate whether the antiviral effect of HT was a consequence of the induction of a heat shock response and HSP expression in the infected cell, we tested the effect of the cyclopentenone PGA1, which is a potent inducer of hsp70 synthesis (27), on HRV production after one cycle of virus growth. Confluent monolayers of HeLa cells were infected with HRV (10 PFU/cell) and, after the 1-h adsorption period, were treated with different concentrations of PGA1 or control diluent at 33°C. Virus yield was determined by plaque assay at 12 h p.i. PGA1 was found to reduce HRV production dose dependently, and an inhibition of more than 80% was observed at the concentration of 10 μg of PGA1/ml (Fig. 4A). As shown above for HT, the antiviral effect of PGA1 was transient, and, at 24 h p.i., the virus yield in cells treated with 10 μg of PGA1/ml (30 μM) was equal to 70% of that of control cells. However, the readdition of PGA1 to infected cells at 12 h p.i. resulted in a virus yield reduction of more than 80% up to 24 h p.i. (data not shown).
FIG. 4.
Effect of PGA1 on HRV replication in HeLa cells. (A) Dose-dependent inhibition of HRV replication by PGA1. Confluent monolayers of HeLa cells were infected with HRV (10 PFU/cell) for 1 h at 33°C and treated with different doses of PGA1 or control diluent soon after the 1-h adsorption period. Virus yield was determined 12 h p.i. by plaque assay. Data represent the mean ± SD of duplicate samples of a representative experiment. ∗, P < 0.05. Each experiment was repeated three times with the same results. The effect of PGA1 (10 μg/ml, 30 μM) (+PGA1) or ethanol diluent (−PGA1) added directly to the agar overlay on the reduction of HRV plaque size and number is shown in the inset. Approximately 100 plaques were measured in triplicate cultures for each sample. (B) Confluent monolayers of HeLa cells, either uninfected (U) or infected with HRV (5 PFU/cell) for 1 h at 33°C (HRV), were treated with PGA1 (10 μg/ml) (+PGA1) or control diluent (−PGA1) soon after the 1-h adsorption period and labeled with [35S]methionine (1-h pulse) at 33°C at different times p.i. Samples containing an equal amount of radioactivity were separated by SDS-PAGE analysis and processed for autoradiography. HRV proteins VP1 and VP3 are indicated. hsp70 is indicated by the arrow.
In a different type of experiment, HeLa cells were infected with HRV (100 PFU/dish) and, after the adsorption period (1 h, 33°C), were incubated with a semisolid medium containing agarose (1%) and PGA1 (10 μg/ml) or control diluent. The number and the size of viral plaques were determined after neutral red staining at 48 and 72 h p.i. Under these conditions, PGA1 treatment inhibited HRV replication and caused a dramatic reduction in the number of plaques at 48 h p.i. (control cells, 98 ± 12 plaques/dish; PGA1-treated cells, 8 ± 3 plaques/dish) (Fig. 4A, inset). At later times of infection (72 h p.i.), the number of plaques in PGA1-treated cells increased and was comparable to 60% of that in control cells; however, the plaque size was reduced by more than 50% in treated cells relative to that of the control.
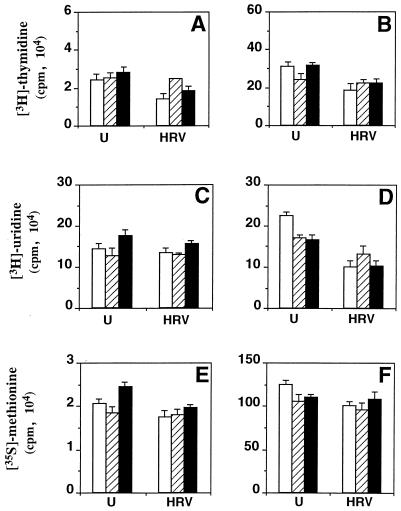
Under the conditions described above, PGA1 was shown not to be toxic to uninfected human cells, as determined by microscopic examination and vital dye uptake (Fig. 4A, inset). At the effective antiviral concentration, PGA1 also did not appear to affect nucleic acid or protein synthesis in HRV-infected cells at 12 h p.i. (Fig. 5). Confluent monolayers of HeLa cells uninfected or infected with HRV (10 PFU/cell) were treated with PGA1 (10 μg/ml) or with a different cyclopentenone prostaglandin, Δ12-PGJ2 (4 μg/ml), 1 h after the adsorption period and then labeled with [3H]thymidine, [3H]uridine, or [35S]methionine for the following 12 h, as described in Materials and Methods. The results shown in Fig. 5 indicate that, at the doses tested, neither prostaglandin significantly affected either the uptake of [3H]thymidine or [35S]methionine or DNA or protein synthesis in uninfected and HRV-infected cells. Both prostaglandins caused a modest reduction in RNA synthesis in mock-infected cells, which did not appear to be due to a reduction in the uptake of precursors, since intracellular [3H]uridine levels were not decreased in prostaglandin-treated cells (Fig. 5C and D).
FIG. 5.
Effect of PGA1 and Δ12-PGJ2 on DNA, RNA, and protein synthesis in uninfected and HRV-infected HeLa cells. Confluent cell monolayers, either uninfected (U) or infected with HRV (10 PFU/cell) for 1 h at 33°C (HRV), were treated with PGA1 (10 μg/ml, 30 μM) (▨), Δ12-PGJ2 (4 μg/ml, 12 μM) (■), or control diluent (□) soon after the 1-h adsorption period and then labeled with [3H]thymidine (A and B), [3H]uridine (C and D), or [35S]methionine (E and F) for the following 12 h. (A, C, and E) Uptake of precursors by uninfected or HRV-infected cells. (B, D, and F) Incorporation of precursors into DNA, RNA, and proteins, respectively. Data represent the mean ± SD of duplicate samples.
Effect of PGA1 on host cell and HRV protein synthesis.
To determine the effect of PGA1 treatment on the kinetics of HRV protein synthesis, HeLa cells were infected with HRV (5 PFU/cell) and treated with PGA1 (10 μg/ml) or control diluent after the 1-h adsorption period. Cells were then labeled with [35S]methionine (1-h pulse) at different times p.i. In uninfected cells, as already described for the same cell line kept at 37°C (4), treatment with PGA1 at 33°C did not greatly affect the overall electrophoretic protein profile, but it induced the synthesis of a 72-kDa cellular protein, which was identified as hsp70 by immunoblot analysis (data not shown). In HRV-infected cells, hsp70 synthesis started 4 h after PGA1 treatment and continued for up to 10 h (Fig. 4B), confirming that, unlike poliovirus, rhinovirus infection does not interfere with hsp70 expression. Under these conditions in control cells virus protein synthesis started 6 h after infection and several virus proteins were evident at 8 to 10 h p.i. PGA1 treatment was found to cause a delay in the synthesis of HRV proteins, which was not detected for up to 10 h p.i.
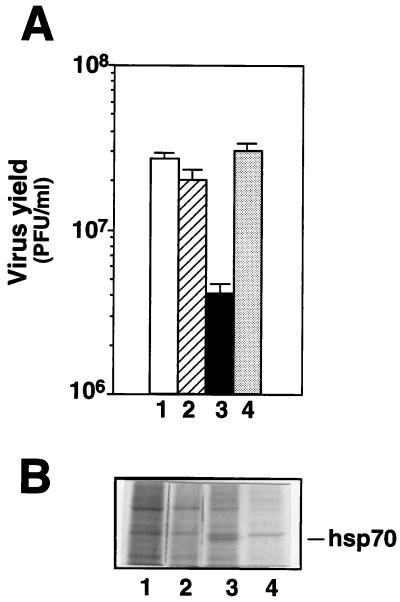
Since the antiviral activity of cyclopentenone prostaglandins in negative-strand RNA virus models has been shown to be dependent on the induction of hsp70 synthesis (2, 20), we investigated the effect of actinomycin D, which is known to inhibit PGA1-induced HSP expression (2, 16), on HRV production. HeLa cells infected with HRV (10 PFU/cell) were treated with PGA1 (10 μg/ml) or control diluent soon after the 1-h adsorption period in the presence or absence of actinomycin D (2 μg/ml). Virus yields were determined at 12 h p.i. As shown in Fig. 6A, treatment with actinomycin D by itself did not affect HRV replication. Actinomycin D, however, completely prevented the inhibitory effect of PGA1, indicating that the antiviral activity is dependent on efficient cellular transcription and translation. In a parallel experiment, HRV-infected cells treated with PGA1 or control diluent were labeled with [35S]methionine for 12 h in the presence or the absence of 2 μg of actinomycin D/ml. SDS-PAGE analysis of [35S]methionine-labeled proteins showed that treatment with actinomycin D prevented PGA1-induced hsp70 synthesis in HeLa cells (Fig. 6B), suggesting that hsp70 could be involved in the control of HRV replication.
FIG. 6.
Effect of PGA1 and actinomycin D on HRV replication. Confluent HeLa cell monolayers untreated (lanes 1 and 3) or pretreated with actinomycin D (2 μg/ml) (lanes 2 and 4) for 1 h at 37°C were infected with HRV (10 PFU/cell) for 1 h at 33°C and treated with PGA1 (10 μg/ml) (lanes 3 and 4) or control diluent (lanes 1 and 2). Actinomycin D was kept in the medium for the duration of the experiment. (A) Virus titers were determined 12 h p.i. by plaque assay. (B) In the same experiment, cells were labeled with [35S]methionine soon after PGA1 treatment for 12 h at 33°C. Samples containing an equal amount of radioactivity were separated by SDS-PAGE analysis and processed for autoradiography. A section of the fluorogram from native gels is shown. The position of hsp70 is indicated.
Effect of Δ12-PGJ2 on HRV replication.
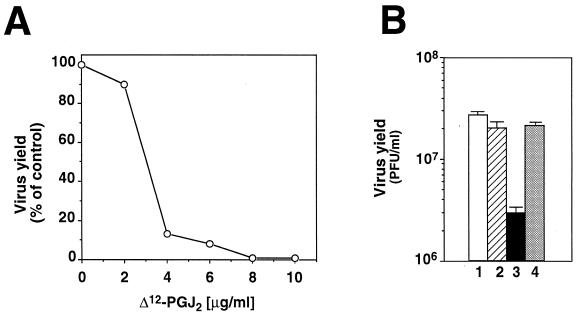
To determine whether cyclopentenone prostaglandins other than PGA1 could inhibit rhinovirus replication, we investigated the effect of Δ12-PGJ2 on HRV infection. HeLa cells infected with 10 PFU of HRV/cell were treated with different concentrations of Δ12-PGJ2 or ethanol diluent after the 1-h adsorption period. Virus yield was determined by plaque assay at 12 h p.i. Treatment with Δ12-PGJ2 was found to reduce HRV production dose dependently, and an inhibition of approximately 90% was observed at the concentration of 4 μg of Δ12-PGJ2/ml (12 μM) (Fig. 7A). At this concentration, Δ12-PGJ2 was not toxic to uninfected HeLa cells and, as described above, did not significantly affect nucleic acid and protein synthesis in either uninfected or HRV-infected cells (Fig. 5). To investigate whether the antiviral activity of Δ12-PGJ2 was also dependent on cellular protein expression, HRV-infected HeLa cells (10 PFU/cell) were treated with 4 μg of Δ12-PGJ2/ml in the presence or absence of actinomycin D (2 μg/ml) for 12 h after infection. Treatment with actinomycin D completely prevented Δ12-PGJ2-induced inhibition of rhinovirus replication, indicating that, as shown above for PGA1, the antiviral activity of Δ12-PGJ2 is cell mediated (Fig. 7B).
FIG. 7.
Effect of Δ12-PGJ2 on HRV replication. (A) Dose-dependent inhibition of HRV replication by Δ12-PGJ2. Confluent monolayers of HeLa cells were infected with HRV (10 PFU/cell) for 1 h at 33°C and treated with different doses of Δ12-PGJ2 or control diluent soon after the 1-h adsorption period. Virus yield was determined 12 h p.i. by plaque assay. (B) Confluent cell monolayers, either untreated (bars 1 and 3) or pretreated with actinomycin D (2 μg/ml) for 1 h at 37°C (bars 2 and 4), were infected with HRV (10 PFU/cell) for 1 h at 33°C and treated with Δ12-PGJ2 (4 μg/ml) (bars 3 and 4) or control diluent (bars 1 and 2). Actinomycin D was kept in the medium for the duration of the experiment. Virus titers were determined 12 h p.i. by plaque assay. Data represent the mean ± SD of duplicate samples of a representative experiment.
DISCUSSION
The results described in the present report indicate that brief HT, when applied at specific stages of the virus cycle, is effective in blocking rhinovirus replication during primary infection of human cells. The inhibitory effect of HT on HRV replication is temperature dependent, and, under one-step multiplication conditions, a 20-min treatment at 45°C was found to be extremely effective, independently of the MOI. The most dramatic effect was observed when HT was applied at 6 h p.i., with a reduction in virus yield of more than 99% relative to that of the control. HT applied at later times of the virus growth cycle (9 h p.i.) resulted in a decreased inhibitory activity, whereas no significant effect on virus replication was found when heat shock was applied soon after virus entry into the cells. These results indicate that the antiviral effect is not due to a general change in membrane fluidity or cell metabolism. In fact, a brief exposure to high temperature (45°C for 20 min) did not damage HeLa cells and only moderately (<30%) inhibited protein synthesis for a period of approximately 3 h in uninfected cells. HT also did not inhibit protein synthesis in HRV-infected HeLa cells (Fig. 2A).
In spite of the dramatic reduction of HRV yield after one cycle of virus growth, HT of HeLa cells did not significantly alter the synthesis of viral proteins at 9 and 11 h p.i., indicating that the target for HRV inhibition could be a posttranslational event. This hypothesis is also supported by the finding that no antiviral effect is observed when HT is applied soon after virus entry into the host cell. A posttranslational event was previously suggested as the target for the antiviral activity of brief HT in vesicular stomatitis virus-infected monkey epithelial cells (6). However, the mechanism by which brief HT can control HRV replication at specific stages of the virus cycle remains to be established. In different types of models of acute RNA virus infection, the antiviral activity of hyperthermia has been associated with the induction of a protective heat shock response and the synthesis of HSP in the infected cell (reviewed in references 23 and 24).
In the case of picornaviruses, increased levels of hsp70 have been detected in cultured neonatal myocardial cells from BALB/c mice after infection with two different picornaviruses, encephalomyocarditis virus and coxsackievirus B3 (11). Whether hsp70 is an unnecessary by-product of the viral infection or has a function in the viral life cycle is as yet unclear. hsp70 was also shown to be associated with newly synthesized capsid precursor P1 of poliovirus and coxsackievirus B1 in infected HeLa cells (14). The half-life of P1 was increased when bound to hsp70, and hsp70-P1 complexes were uncleavable by the viral protease. As anticipated in the introduction, infection with polioviruses, which are known to cause a dramatic shutoff of the host cell protein cap-dependent translation by proteolytically inactivating the cap-binding protein complex (22), inhibits constitutive as well as heat shock-induced hsp70 synthesis in human cells, even though to a minor extent in comparison to other cellular proteins (14, 18). We have recently shown that infection with poliovirus type 2 also prevents hsp70 synthesis after treatment with a different class of HSP inducers, the cyclopentenone prostaglandins (4). On the other hand, the translation of the glucose-regulated protein BiP was found to be increased in poliovirus-infected HeLa cells, at a time when cap-dependent translation of cellular mRNA is inhibited (28). We have now shown that, unlike polioviruses, rhinovirus infection does not induce the expression of glucose-regulated proteins and does not prevent heat shock-induced hsp70 synthesis in HeLa cells. Under the conditions described herein, comparable levels of this protein were detected in uninfected and HRV-infected cells. Based on the hsp70-P1 complex formation described in cells infected with other types of picornaviruses (14), it could be hypothesized that intracellular accumulation of high levels of hsp70 during specific stages of rhinovirus infection could impair virus maturation and/or release from the infected cells, possibly by hsp70 binding to viral polypeptides. Since HSP are known to be stable proteins (13), the relative time-specific effect of heat could be a consequence of the fact that hsp70 is utilized by the cell as a molecular chaperone as soon as it is synthesized. In this case, the availability of large amounts of newly synthesized protein could be essential for interaction with virus proteins or viral components at different times of the virus cycle.
It should be emphasized that inhibition of virus replication by HT is transient. If treatment is not repeated, HRV yields from HT-treated cells can reach the control level at later times of infection (24 to 48 h p.i.), confirming that the reduction in virus yield is not caused by an aspecific cytotoxic effect of hyperthermia in HeLa cells. The fact that hsp70 synthesis after heat shock is also transient, and it persists only for a period of 3 to 4 h after treatment, supports the possibility that high levels of hsp70 synthesis are necessary for the antiviral effect to persist. To investigate the possibility that hsp70 could play a role in the control of rhinovirus replication, we tested the effect of a different class of HSP inducers, the cyclopentenone prostaglandins, on HRV infection.
Cyclopentenone prostaglandins of the A and J type (PGA and PGJ) are known to possess a potent antiviral activity against a wide variety of DNA and RNA viruses in different types of mammalian cells, as well as in animal models (12, 21, 24–26). The antiviral activity of these molecules has been associated with their ability to function as a signal for the induction of hsp70 synthesis via cycloheximide-sensitive activation of the transcription factor HSF1 (1, 24). Micromolar concentrations of PGA1, which did not inhibit cell metabolism, significantly reduced HRV yield after one cycle of virus growth while inducing hsp70 synthesis starting 4 h after the beginning of treatment. It is interesting to note that the amount of hsp70 induced by PGA1 at 33°C, the temperature used for HRV infection, was comparable to that at 37°C, as determined by immunoblot analysis (data not shown). Unlike poliovirus (4), HRV infection did not prevent hsp70 induction by PGA1. As shown previously in other cell types (2), PGA1 was a more effective inducer of hsp70 than heat, since synthesis of hsp70 persisted for at least 10 h after the addition of PGA1 as compared to 3 to 4 h after HT under the conditions used. PAGE analysis of HRV proteins showed that PGA1 caused a delay of virus protein synthesis, which did not start before 10 h p.i. This indicates that, as suggested in other virus-host cell models (23), induction of the heat shock response may affect rhinovirus replication at more than one level. When HRV infection was allowed to proceed for up to 24 to 48 h p.i. without additional treatments, the virus yield progressively reached the level of mock-treated controls, indicating that the antiviral effect is reversible and not due to a cytotoxic effect of the drug. The possibility that hsp70 could be involved in the control of HRV replication is suggested by the fact that treatment with actinomycin D, which blocks hsp70 expression, prevented the inhibition of HRV production in PGA1-treated cells. Similar results were obtained with a different cyclopentenone prostaglandin, Δ12-PGJ2, a natural metabolite of PGD2, which presently occurs in human body fluids (10). Treatment with actinomycin D also prevented the antiviral effect of HT in HeLa cells (data not shown).
As anticipated in the introduction, Tyrrell and coworkers have previously described that naturally acquired as well as experimental colds benefit from local HTs (43°C for 20 to 30 min) in randomized double-blind clinical trials in humans (30, 31). A significant reduction in the mean symptom scores was observed in treated patients. A transient reduction in virus shedding was reported only on the day of treatment, whereas the mean titers in convalescence and the frequencies of antibody response were not significantly different in treated and control groups. Regulation of heat shock gene expression was then hypothesized to be a possible target for the antiviral activity of respiratory hyperthermia (31). On the other hand, Hendley et al. have shown that two nasal treatments with steam had no effect on viral shedding in volunteers with experimental rhinovirus infections (9). No beneficial effects from steam inhalation on common cold symptoms were detected in other studies (8, 15). Differences in the techniques of administering therapy and in the strains of rhinoviruses were both hypothesized to be responsible for the discrepancy in these studies (15). Our results, which show that HT at 45°C is effective in inhibiting HRV replication in vitro when applied at specific stages of the virus cycle, point out that differences in the temperature utilized and in the time of treatment should be considered. Our results also suggest the possibility that HSP and hsp70 in particular could participate in an intracellular defense strategy during HRV infection. However, several questions on the mechanism by which hsp70 can interfere with rhinovirus replication during heat stress remain to be answered. A better understanding of the role of HSP in virus replication could be useful in the comprehension of the beneficial effect of fever or artificial pyrexia during virus infection and could indicate new strategies in the treatment of the common cold.
ACKNOWLEDGMENTS
We thank Carlotta Volpi and Alessandra Fiocchetti for excellent technical assistance.
This work was supported by the Italian Ministry of Public Health, 1997 AIDS Research Project, and by the Italian National Research Council, P.F. “Biotechnology.”
REFERENCES
- 1.Amici C, Sistonen L, Santoro M G, Morimoto R I. Anti-proliferative prostaglandins activate heat shock transcription factor. Proc Natl Acad Sci USA. 1992;89:6227–6231. doi: 10.1073/pnas.89.14.6227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Amici C, Giorgi C, Rossi A, Santoro M G. Selective inhibition of virus protein synthesis by prostaglandin A1: a translational block associated with HSP70 synthesis. J Virol. 1994;68:6890–6899. doi: 10.1128/jvi.68.11.6890-6899.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Conti C, Orsi N, Stein M L. Effect of isoflavans and isoflavenes on rhinovirus 1B and its replication in HeLa cells. Antiviral Res. 1988;10:117–127. doi: 10.1016/0166-3542(88)90020-4. [DOI] [PubMed] [Google Scholar]
- 4.Conti C, Mastromarino P, Tomao P, De Marco A, Pica F, Santoro M G. Inhibition of poliovirus replication by prostaglandins A and J in human cells. Antimicrob Agents Chemother. 1996;40:367–372. doi: 10.1128/aac.40.2.367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Couch R B. Rhinoviruses. In: Fields B N, Knife D M, Melnick J L, Chanock R M, Roizman B, Shope R E, editors. Fields virology. New York, N.Y: Raven Press; 1985. pp. 795–816. [Google Scholar]
- 6.De Marco A, Santoro M G. Antiviral effect of short hyperthermic treatment at specific stages of vesicular stomatitis virus replication cycle. J Gen Virol. 1993;74:1685–1690. doi: 10.1099/0022-1317-74-8-1685. [DOI] [PubMed] [Google Scholar]
- 7.Feige U, Morimoto R I, Yahara I, Polla B S. Stress-inducible cellular responses. Basel, Switzerland: Birkhauser Verlag; 1996. [Google Scholar]
- 8.Forstall G J, Macknin M L, Yen-Lieberman B R, Medendrop S. Effect of inhaling heated vapor on symptoms of the common cold. JAMA. 1994;271:1109–1111. [PubMed] [Google Scholar]
- 9.Hendley J O, Abbott R D, Beasley P P, Gwaltney J M. Effect of inhalation of hot humidified air on experimental rhinovirus infection. JAMA. 1994;271:1112–1113. [PubMed] [Google Scholar]
- 10.Hirata Y, Hayashi H, Ito S, Kikawa Y, Ishibashi M, Sudo M, Miyazaki H, Fukushima M, Narumiya S, Hayaishi O. Occurrence of 9-deoxy-Δ9,Δ12-13,14-dihydroprostaglandin D2in human urine. J Biol Chem. 1988;263:16619–16625. [PubMed] [Google Scholar]
- 11.Huber S A. Heat shock protein induction in adriamycin and picornavirus-infected cardiocytes. Lab Investig. 1992;67:218–224. [PubMed] [Google Scholar]
- 12.Hughes-Fulford M, McGrath M S, Hanks D, Erickson S, Pulliam L. Effects of dimethyl prostaglandin A1on herpes simplex virus and immunodeficiency virus replication. Antimicrob Agents Chemother. 1992;36:2253–2258. doi: 10.1128/aac.36.10.2253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lindquist S, Craig E A. The heat-shock proteins. Annu Rev Genet. 1988;22:631–677. doi: 10.1146/annurev.ge.22.120188.003215. [DOI] [PubMed] [Google Scholar]
- 14.Macejac D J, Sarnow P. Association of heat shock protein 70 with enterovirus capsid precursor P1 in infected human cells. J Virol. 1992;66:1520–1527. doi: 10.1128/jvi.66.3.1520-1527.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Macknin M L, Mathew S, Medendrop S. Effect of inhaling heated vapor on symptoms of the common cold. JAMA. 1990;264:989–991. [PubMed] [Google Scholar]
- 16.Mastromarino P, Conti C, Petruzziello R, De Marco A, Pica F, Santoro M G. Inhibition of Sindbis virus replication by cyclopentenone prostaglandins: a cell-mediated event associated with heat-shock protein synthesis. Antiviral Res. 1993;20:209–222. doi: 10.1016/0166-3542(93)90021-a. [DOI] [PubMed] [Google Scholar]
- 17.Morimoto R I, Santoro M G. Stress-inducible responses and heat shock proteins: new pharmacologic targets for cytoprotection. Nat Biotechnol. 1998;16:833–838. doi: 10.1038/nbt0998-833. [DOI] [PubMed] [Google Scholar]
- 18.Muñoz A, Alonso M A, Carrasco L. Synthesis of heat shock proteins in HeLa cells: inhibition by virus infection. Virology. 1984;137:150–159. doi: 10.1016/0042-6822(84)90018-7. [DOI] [PubMed] [Google Scholar]
- 19.Palmenberg A C. Picornaviral processing: some new ideas. J Cell Biochem. 1987;33:191–198. doi: 10.1002/jcb.240330306. [DOI] [PubMed] [Google Scholar]
- 20.Pica F, De Marco A, De Cesare F, Santoro M G. Inhibition of vesicular stomatitis virus replication by Δ12-prostaglandin J2is regulated at two separate levels and is associated with induction of stress protein synthesis. Antiviral Res. 1993;20:193–208. doi: 10.1016/0166-3542(93)90020-j. [DOI] [PubMed] [Google Scholar]
- 21.Rozera C, Carattoli A, De Marco A, Amici C, Giorgi C, Santoro M G. Inhibition of HIV-1 replication by cyclopentenone prostaglandins in acutely infected human cells: evidence for a transcriptional block. J Clin Invest. 1996;97:1795–1803. doi: 10.1172/JCI118609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rueckert R R. Picornaviridae and their replication. In: Fields B N, Knipe D M, Chanock R M, Melnick J L, Roizman B, Shope R E, editors. Virology. 2nd ed. New York, N.Y: Raven Press; 1990. pp. 507–548. [Google Scholar]
- 23.Santoro M G. Virus infection. In: Feige U, Morimoto R I, Yahara I, Polla B S, editors. Stress-inducible cellular responses. Basel, Switzerland: Birkhauser Verlag; 1996. pp. 337–357. [Google Scholar]
- 24.Santoro M G. Antiviral activity of cyclopentenone prostanoids. Trends Microbiol. 1997;5:276–281. doi: 10.1016/S0966-842X(97)01066-4. [DOI] [PubMed] [Google Scholar]
- 25.Santoro M G, Benedetto A, Carruba G, Garaci E, Jaffe B M. Prostaglandin A compounds as antiviral agents. Science. 1980;209:1032–1034. doi: 10.1126/science.6157190. [DOI] [PubMed] [Google Scholar]
- 26.Santoro M G, Favalli C, Mastino A, Jaffe B M, Esteban M, Garaci E. Antiviral activity of a synthetic analog of prostaglandin A in mice infected with influenza A virus. Arch Virol. 1988;99:89–100. doi: 10.1007/BF01311026. [DOI] [PubMed] [Google Scholar]
- 27.Santoro M G, Garaci E, Amici C. Prostaglandins with antiproliferative activity induce the synthesis of a heat shock protein in human cells. Proc Natl Acad Sci USA. 1989;86:8407–8411. doi: 10.1073/pnas.86.21.8407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sarnow P. Translation of glucose-regulated protein 78/immunoglobulin heavy-chain binding protein mRNA is increased in poliovirus-infected cells at a time when cap-dependent translation of cellular mRNAs is inhibited. Proc Natl Acad Sci USA. 1989;86:5795–5799. doi: 10.1073/pnas.86.15.5795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sperber S J, Hayden F G. Chemotherapy of rhinovirus colds. Antimicrob Agents Chemother. 1988;32:409–419. doi: 10.1128/aac.32.4.409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tyrrell D A J. Hot news on the common cold. Annu Rev Microbiol. 1988;42:35–47. doi: 10.1146/annurev.mi.42.100188.000343. [DOI] [PubMed] [Google Scholar]
- 31.Tyrrell D A J, Barrow I, Arthur J. Local hyperthermia benefits natural and experimental common colds. Br Med J. 1989;298:1280–1283. doi: 10.1136/bmj.298.6683.1280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Uncapher C R, DeWitt C M, Colonno R J. The major and minor receptor families contain all but one human rhinovirus serotype. Virology. 1991;180:814–817. doi: 10.1016/0042-6822(91)90098-v. [DOI] [PubMed] [Google Scholar]