ABSTRACT.
Hemoglobin C is the second most common structural hemoglobinopathy in Africa, and carriers have a reduced risk of severe malaria. However, the effect of HbAC on the antibody response to malaria antigens in pregnancy has not been studied. Here, we measured PfEMP1-specific antibodies in plasma samples from 74 Beninese pregnant women with either HbAA or HbAC. IgG-mediated inhibition of VAR2CSA+ infected erythrocytes adhesion to chondroitin sulfate A (CSA) was also tested. PfEMP1-specific IgG levels to VAR2CSA were significantly lower in HbAC women, suggesting less exposure to VAR2CSA. In contrast, the percentage of VAR2CSA+-infected erythrocytes adhesion to CSA was not different between HbAA and HbAC women. Moreover, IgG levels to PfEMP1 variants associated with severe malaria were not significantly different between groups. The findings indicate similar exposure to Plasmodium falciparum parasites expressing PfEMP1 variants causing severe malaria, and justify more comprehensive studies of hemoglobinopathy-related qualitative and quantitative differences in PfEMP1-specific antibody responses.
Pregnant women are at high risk of malaria. In 2019, 12 million pregnant women in Africa were exposed to Plasmodium falciparum parasites during pregnancy.1 This can frequently lead to placental malaria (PM), a major cause of mother–offspring mortality and severe morbidity. In PM, infected erythrocytes (IEs) bind to oncofetal chondroitin sulfate A (CSA) in the placenta via VAR2CSA,2 a member of the P. falciparum erythrocyte membrane protein 1 (PfEMP1) family, expressed on the surface of IEs. Other PfEMP1 members enable adhesion of IEs to endothelial host receptors such as intercellular adhesion molecule 1 (ICAM-1) and endothelial protein C receptor (EPCR),3,4 which have been associated with severe malaria. Naturally acquired antibodies targeting PfEMP1 proteins are mainly acquired during childhood and can play a role in clinical protection in malaria.4
Malaria has exerted a strong evolutionary pressure on the human genome, selecting host polymorphisms that protect against the disease.5 These include structural variants of adult hemoglobin (HbA, wildtype) involving a single point mutation within the β-globin gene as in HbS, HbC, and HbE, or reduced production of globin as in thalassemias. HbC is the second most common structural Hb variant and is highly frequent in West Africa (15%). Carriers have a reduced risk of severe malaria by P. falciparum.5,6 This has been attributed to the reduced adhesiveness of HbC IEs to the endothelial cells and reduced PfEMP1expression on the IE surface.7,8 However, the effect of HbAC on the antibody response to VAR2CSA and other malaria-specific antigens in pregnancy has not been studied previously. Thus, we measured levels of PfEMP1-specific antibodies in Beninese pregnant women with either HbAA or HbAC, as well as IgG-mediated inhibition of VAR2CSA+ IEs-adhesion to CSA.
Plasma samples collected at delivery from pregnant women as part of the STOPPAM study in Comè District, Benin, between 2008 and 2011 were used.9 Declaration of voluntary, free-will participation in the study and written informed consent were obtained from all participants before enrollment. On the basis of the availability of samples, we carried out a case–control study with 74 plasma samples. Cases (HbAC) and controls (HbAA) were matched 1:1 according to age and gravidity because both are known to increase antibody levels to malaria antigens. The Hb genotyping was performed by polymerase chain reaction (PCR) as previously described.10 Peripheral blood samples were tested by real-time PCR to detect submicroscopic P. falciparum infection.11 PM was defined as the presence of malaria parasites in placental blood by microscopy or histological evidence of parasites in the placenta.
IgG reactivity at delivery against recombinant PfEMP1 antigens (100 ng/well) was measured by ELISA as described elsewhere.12 Antigens included the entire ectodomains of a VAR2CSA-type (IT4VAR04)12 and three non-VAR2CSA-type (HB3VAR06, IT4VAR09, IT4VAR60) PfEMP1 proteins, produced in baculovirus-transfected Sf9 insect cells as described previously.13 Two recombinant DBLβ domains containing an ICAM-1 binding motif (HB3VAR34, PFD1235w)3,14 and the R0 domain of glutamate-rich protein (GLURP),15 all produced in Escherichia coli, were also used. HB3VAR06 and IT4VAR60 bind uninfected erythrocytes, leading to formation of rosettes, which are a marker of parasites causing severe malaria. HB3VAR34 and PFD1235w are involved in adhesion of IEs to ICAM-1 and EPCR. IT4VAR09 causes rosetting and binds to human brain microvascular endothelial cells. Plasma samples from eight nonpregnant Danish women without P. falciparum exposure and a pool of 20 nonpregnant Ghanaian women previously exposed to P. falciparum during pregnancy were included as negative and positive controls, respectively. Specific antibody levels were calculated in arbitrary units (AU) using the equation 100 × [(ODSAMPLE – ODBLANK)/(ODPOS.CTRL – ODBLANK)], essentially as described elsewhere.12 Negative cutoff values were calculated as the mean AU values plus two standard deviations (SD) obtained with the negative control samples. Individuals were considered responders if the specific antibody level was higher than the cutoff. IgG reactivity to native VAR2CSA expressed on the surface of intact and unfixed IEs was analyzed by flow cytometry as described elsewhere.16 A CytoFLEX S (Beckman Coulter Life Sciences, Brea, CA) flow cytometer was used for data acquisition and FlowLogic software (Inivai Technologies, Mentone, Australia) for data analysis. To normalize data between plates, IgG binding to IEs is presented as normalized median fluorescence intensity (nMFI) using the equation (MFISAMPLE/MFINEG. CTRL) × 100.
The inhibition of IE adhesion to CSA (5 μg/mL decorin) by plasma samples collected at delivery was evaluated using a static adhesion assay described elsewhere.17 Briefly, 20% parasite suspension of late-stage VAR2CSA+ IEs was incubated with plasma (1:5) or soluble CSA (500 μg/mL). After removing nonadherent erythrocytes, the remaining bound cells were fixed with 1.5% glutaraldehyde in PBS, and then stained with 10% Giemsa. Adherent cells were quantified by light microscopy as the number of IEs bound/mm2. The positivity threshold was defined as the mean plus 3 SD obtained with plasma from 30 pregnant French women without P. falciparum exposure.
Data were analyzed and plotted using GraphPad Prism version 9.0 (GraphPad Software, San Diego, CA). Mann-Whitney U was used to compare two groups. Spearman’s rank correlation (rs) was used to assess the association between numeric variables. Fisher’s exact test was used to compare proportions. P values < 0.05 were considered statistically significant.
Matched plasma samples collected at delivery from 37 pregnant women in each group were analyzed. Overall, the women had a median age of 28 years (IQR 17–45 years) and three children on average (range 1–12 children). Two women in each group were primigravidae (5.4%), and 38% had at least one malaria episode during the current pregnancy (range 0–4). Women with PM (10.1%) had a higher number of infections during pregnancy (median 2 versus 0; P < 0.001, Mann-Whitney test). Whether those were new infections or recrudescence of previous episodes was not assessed. No significant differences were observed in demographic and clinical characteristics between the two groups (Table 1).
Table 1.
Characteristics of pregnant women according to the hemoglobin genotype
| Variable | HbAA (N = 37) | HbAC (N = 37) | P value* |
|---|---|---|---|
| n (%) | n (%) | ||
| Infection at enrollment | 3 (8.1) | 1 (2.7) | 0.61 |
| Infection during pregnancy | 15 (40.5) | 13 (35.1) | 0.81 |
| Placental malaria† | 4 (11.1) | 3 (9.1) | 0.99 |
| Parasite infection‡ | 16 (43.2) | 13 (35.1) | 0.63 |
| Number of parasite infections | |||
| 0 | 22 (59.5) | 24 (64.9) | 0.38 |
| 1 | 8 (21.6) | 10 (27.0) | |
| ≥ 2 | 7 (18.9) | 3 (8.1) |
P value using Fisher’s exact test.
Placental malaria data were available for 69 women (36 HbAA and 33 HbAS).
Women having at least one of these: infection at enrollment (most of the women were enrolled in the second trimester of pregnancy), during pregnancy, or placental malaria.
Antibody levels to CSA-adhering IEs and VAR2CSA-type PfEMP1 increase with parity and can inhibit adhesion of IEs to CSA.2,18,19 Here, we found that specific IgG levels to recombinant (18.0 versus 63.2, P = 0.022) and native (64.0 versus 70.3, P = 0.07) VAR2CSA correlated positively (rs = 0.47; P < 0.001) and were lower in HbAC than in HbAA pregnant women (Figure 1), suggesting a reduced exposure to VAR2CSA. One possible explanation is the decreased expression of VAR2CSA on the HbAC IE surface.8 In contrast, we have previously reported that levels of VAR2CSA-specific IgG in pregnant women with HbAS are comparable to those among HbAA women.16,20 Expression of VAR2CSA is also reduced in HbAS IEs, but the cytoadhesion to CSA seems to be less affected than in HbAC.8 As expected, levels of VAR2CSA-specific IgG increased with parity (rs = 0.27; P = 0.02) regardless of the Hb genotype. We also tested the ability of the plasma antibodies to inhibit IE adhesion to CSA (Figure 1). Most of the women (76%) had antibodies that inhibited the adhesion of IEs to CSA, and the percentage of inhibition was similar among HbAC and HbAA samples (49.1% versus 43.8%; P = 0.36, Mann Whitney test). However, levels of VAR2CSA-specific IgG correlated positively with the percentage of adhesion inhibition in HbAA women (rs = 0.43; P = 0.009) but not in HbAC women (rs = 0.27; P = 0.13). This indicates that VAR2CSA-specific IgG in both groups is a mixture of neutralizing (inhibited adhesion of IE) and non-neutralizing antibodies, and that IgG levels were sufficient to inhibit IE adhesion to CSA.
Figure 1.
Antibody response to VAR2CSA and inhibition of binding to chondroitin sulfate A (CSA). Recombinant and native VAR2CSA-specific IgG levels determined by an ELISA (n = 74) and flow cytometry (n = 67) are expressed in arbitrary units and normalized mean fluorescence intensity (nMFI), respectively. Inhibition of infected erythrocytes adhesion to CSA at delivery is expressed as a percentage (n = 70). IgG levels against R0 domain of glutamate-rich protein (GLURP) as determined by an ELISA (n = 74) are expressed in arbitrary units. Median, interquartile range (IQR), and whiskers (1.5 times the IQR) are shown. Dotted line indicates the threshold of positivity. P values using Mann-Whitney U test are also indicated. This figure appears in color at www.ajtmh.org.
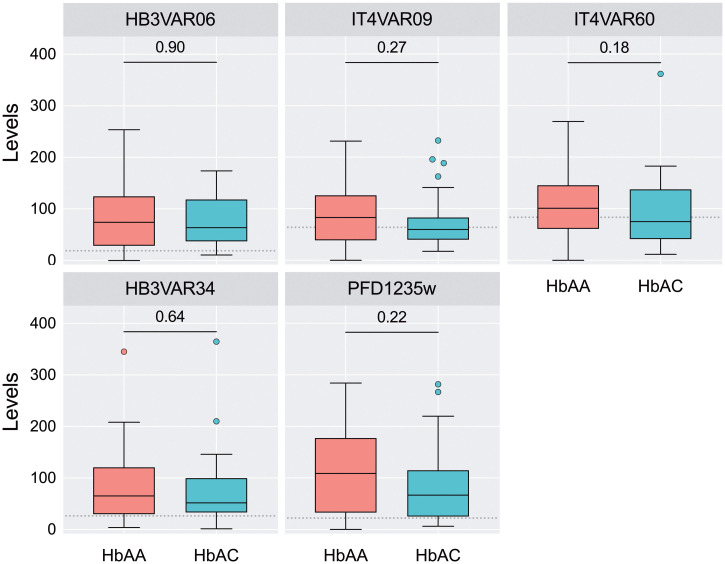
It has previously been reported that HbC does not protect against P. falciparum infection or uncomplicated malaria,5 and our finding of similar levels of GLURP-specific IgG in HbAA and HbAC women supports this observation (Figure 1). In contrast, HbC has been associated with decreased risk of severe malaria in children.5,6 We proceeded to compare levels of IgG specific for PfEMP1 variants previously associated with severe P. falciparum malaria. IgG levels against HB3VAR06, IT4VAR09, and IT4VAR60 were not significantly different between HbAA and HbAC women (Figure 2). Furthermore, no differences in levels of IgG specific for two ICAM-1-binding DBLβ domains were found (Figure 2). Finally, the breadth of the antibody response, defined as the number of antigens recognized was not significantly different between HbAA and HbAC women, with a median of five antigens in both groups (P = 0.40, Mann-Whitney test). The findings indicate similar exposure to P. falciparum parasites expressing these particular PfEMP1 variants and thus do not explain the previously reported enhanced clinical protection among HbC carriers. In contrast, the current data suggest a reduced exposure to VAR2CSA in HbAC women. Nevertheless, due to the small sample size, larger studies should be done to confirm the findings.
Figure 2.
Antibody response to recombinant non-VAR2CSA PfEMP1 proteins. IgG levels against full-length recombinant proteins HB3VAR06, IT4VAR09, and IT4VAR60, or single DBLβ domains of HB3VAR34 and PFD1235w were determined by ELISA (n = 74). In all panels, the values are expressed in arbitrary units. Median, interquartile range (IQR), whiskers (1.5 times the IQR), and outliers are shown. Dotted line indicates the threshold of positivity. P values using Mann-Whitney U test are also indicated. This figure appears in color at www.ajtmh.org.
In conclusion, our present and earlier data, combined with the strong evidence that severe P. falciparum malaria is related to the expression of particular PfEMP1 variants and controlled by PfEMP1-specific immunity, justify more comprehensive studies of hemoglobinopathy-related qualitative and quantitative differences in PfEMP1-specific antibody responses. Such investigations are currently in progress.
ACKNOWLEDGMENTS
We thank the Beninese women who participated in this study. We thank Anja TR Jensen and Michael Theisen (University of Copenhagen, Denmark) for providing the DBLβ domains and GLURP, respectively. We acknowledge Liz Stevenson (University of Copenhagen, Denmark) for help with the production of recombinant proteins. We thank Nadine Fievet, Achille Massougbodji, Philippe Deloron, and the staff of the IRCB of Benin for their facilitation and participation in fieldwork.
REFERENCES
- 1. WHO , 2020. World Malaria Report 2020: 20 Years of Global Progress and Challenges. Geneva, Switzerland: World Health Organization, 299. [Google Scholar]
- 2.Salanti A et al. 2004. Evidence for the involvement of VAR2CSA in pregnancy-associated malaria. J Exp Med 200: 1197–1203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lennartz F et al. 2017. Structure-guided identification of a family of dual receptor-binding PfEMP1 that is associated with cerebral malaria. Cell Host Microbe 21: 403–414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hviid L Jensen AT , 2015. PfEMP1—a parasite protein family of key importance in Plasmodium falciparum malaria immunity and pathogenesis. Adv Parasitol 88: 51–84. [DOI] [PubMed] [Google Scholar]
- 5.Taylor SM Parobek CM Fairhurst RM , 2012. Haemoglobinopathies and the clinical epidemiology of malaria: a systematic review and meta-analysis. Lancet Infect Dis 12: 457–468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Agarwal A et al. 2000. Hemoglobin C associated with protection from severe malaria in the Dogon of Mali, a West African population with a low prevalence of hemoglobin S. Blood 96: 2358–2363. [PubMed] [Google Scholar]
- 7.Fairhurst RM et al. 2005. Abnormal display of PfEMP-1 on erythrocytes carrying haemoglobin C may protect against malaria. Nature 435: 1117–1121. [DOI] [PubMed] [Google Scholar]
- 8.Kilian N Srismith S Dittmer M Ouermi D Bisseye C Simpore J Cyrklaff M Sanchez CP Lanzer M , 2015. Hemoglobin S and C affect protein export in Plasmodium falciparum-infected erythrocytes. Biol Open 4: 400–410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huynh BT Fievet N Gbaguidi G Dechavanne S Borgella S Guezo-Mevo B Massougbodji A Ndam NT Deloron P Cot M , 2011. Influence of the timing of malaria infection during pregnancy on birth weight and on maternal anemia in Benin. Am J Trop Med Hyg 85: 214–220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tetard M Milet J Dechavanne S Fievet N Dorin-Semblat D Elion J Fairhurst RM Deloron P Tuikue-Ndam N Gamain B , 2017. Heterozygous HbAC but not HbAS is associated with higher newborn birthweight among women with pregnancy-associated malaria. Sci Rep 7: 1414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cottrell G Moussiliou A Luty AJ Cot M Fievet N Massougbodji A Deloron P Tuikue Ndam N , 2015. Submicroscopic Plasmodium falciparum infections are associated with maternal anemia, premature births, and low birth weight. Clin Infect Dis 60: 1481–1488. [DOI] [PubMed] [Google Scholar]
- 12.Lopez-Perez M Larsen MD Bayarri-Olmos R Ampomah P Stevenson L Arevalo-Herrera M Herrera S Hviid L , 2018. IgG responses to the Plasmodium falciparum antigen VAR2CSA in Colombia are restricted to pregnancy and are not induced by exposure to Plasmodium vivax. Infect Immun 86: e00136–e18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stevenson L Laursen E Cowan GJ Bandoh B Barfod L Cavanagh DR Andersen GR Hviid L , 2015. alpha2-macroglobulin can crosslink multiple Plasmodium falciparum erythrocyte membrane protein 1 (PfEMP1) molecules and may facilitate adhesion of parasitized erythrocytes. PLoS Pathog 11: e1005022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Olsen RW Ecklu-Mensah G Bengtsson A Ofori MF Kusi KA Koram KA Hviid L Adams Y Jensen ATR , 2019. Acquisition of IgG to ICAM-1-binding DBLbeta domains in the Plasmodium falciparum erythrocyte membrane protein 1 antigen family varies between groups A, B, and C. Infect Immun 87: e00224–e19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Theisen M Vuust J Gottschau A Jepsen S Hogh B , 1995. Antigenicity and immunogenicity of recombinant glutamate-rich protein of Plasmodium falciparum expressed in Escherichia coli. Clin Diagn Lab Immunol 2: 30–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Lopez-Perez M, Viwami F, Seidu Z, Jensen ATR, Doritchamou J, Ndam N, Hviid L, 2021. PfEMP1-specific IgG reactivity among Beninese pregnant women with sickle cell trait. Open Forum Infect Dis 8: ofab527. [DOI] [PMC free article] [PubMed]
- 17.Fried M Duffy PE , 2002. Analysis of CSA-binding parasites and antiadhesion antibodies. Methods Mol Med 72: 555–560. [DOI] [PubMed] [Google Scholar]
- 18.Fried M Nosten F Brockman A Brabin BJ Duffy PE , 1998. Maternal antibodies block malaria. Nature 395: 851–852. [DOI] [PubMed] [Google Scholar]
- 19.Ricke CH Staalsoe T Koram K Akanmori BD Riley EM Theander TG Hviid L , 2000. Plasma antibodies from malaria-exposed pregnant women recognize variant surface antigens on Plasmodium falciparum–infected erythrocytes in a parity-dependent manner and block parasite adhesion to chondroitin sulfate A. J Immunol 165: 3309–3316. [DOI] [PubMed] [Google Scholar]
- 20.Chauvet M et al. 2019. Impact of hemoglobin S trait on cell surface antibody recognition of Plasmodium falciparum-infected erythrocytes in pregnancy-associated malaria. Open Forum Infect Dis 6: ofz156. [DOI] [PMC free article] [PubMed] [Google Scholar]