Abstract
Background
We aimed to estimate salt intake among an Iranian population using spot urine-based equations and a dietary-based method.
Methods
Adult men and women (n = 2069) were recruited from the Tehran Lipid and Glucose Study (2014–2017). Urinary sodium (Na), potassium (K), and creatinine (Cr) concentrations were measured in the morning spot urine samples. The 24-h urinary Na excretion and predicted salt intake was estimated using five equations, i.e., Kawasaki, Tanaka, Intersalt, Toft, and Whitton. A validated food frequency questionnaire (FFQ) was used to obtain dietary intake of salt. The agreement of each urinary- and FFQ-based salt estimation with the overall mean of the methods, considered as the gold standard, was assessed using the Bland–Altman method.
Results
Mean age of the participants was 45.6 ± 14.8 y, and 45.4% were men. Mean (SD) estimated salt intake, derived from the overall mean of the methods, was 9.0 ± 2.2 g/d (10.2 ± 2.1 and 7.9 ± 1.7 g/d in men and women, respectively). Mean bias of the estimations from the overall mean ranged from − 0.2.42 to 2.75 g/d, with the Tanaka equation having the least bias (mean bias = 0.13 ± 1.10, 95% CI − 2.37, 2.30 g/d). Tanaka estimated a mean salt intake of 8.9 g/d (range 2.1 to 18.7 g/d); accordingly, only 5.1% of participants adhered to the recommendation (< 5 g/d salt intake), whereas 26.8% and 2.4% exceeded the recommendation by 2- and threefold.
Conclusion
The Tanaka equation could provide a more accurate mean-population estimated salt intake from casual urinary Na concentration in our population. About 95% of the Iranian population exceeded the current recommendations of salt intake.
Keywords: Casual urinary sodium, Estimated population-salt intake, Tanaka equation
Introduction
Cardiovascular diseases (CVD) and hypertension (HTN) are growing global health problems; in 2019, 523 and 18.6 million prevalent cases of CVD and CVD deaths and 33% global age-standardized prevalence of HTN in adults were reported [1, 2]. The first‐line antihypertensive agents are thiazide diuretics, beta-blockers, calcium channel blockers, angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor antagonists, or alpha adrenergic blockers [3]. Complementary medicine and dietary approaches have also been proposed to manage CVD risk factors and HTN [4–7].
Excess salt intake is suggested to be responsible for a global 3 million deaths, and 70 million disability-adjusted life-years (DALYs) lost every year [8]. Current literature confirms the biological plausibility of the association between high intake of sodium (Na) and HTN and CVD events [9]. Although excessive Na intake remains undefined [10, 11], monitoring population salt intake is suggested as a global health priority [12, 13]. The World Health Organization (WHO) recommends less than 85 mmol/day of Na (< 2 g/d Na or ~ 5 g/d salt) for adults [14]. The Dietary Guidelines for Americans (DGA) recommends a reduction of Na intake to 100 mmol/day (< 2.3 g/day) and 65 mmol/day (1.5 g/day) in the general population and individuals with hypertension, diabetes, or chronic kidney disease [15]. The global estimation of mean intake of salt is around 2- to 3-fold higher than the current limit (10 g/d [16] to 14 g/d [8] of salt), and 95% of the World's population have a mean salt intake between 6 and 12 g/d [17].
Although the 24-h urine sampling is the gold standard for assessing Na excretion and estimating salt intake [18], the WHO considers spot urine sampling a less-burden and more feasible method of population salt intake monitoring [12]. Several equations have been developed to predict 24-h Na excretion from Na concentration of spot urine samples along with urinary creatinine and potassium (K) concentration, age, height, weight, and sex [19]; however, the validity and reliability of the equations are varied across populations due to diverse ethnicity and different patterns of Na intakes [19, 20]. Dietary methods tend to underestimate population salt intake [19, 21], and poor agreement is reported between estimated-Na from the food frequency questionnaire (FFQ) and 24‐h urine [22].
Choosing a valid method to monitor population salt intake over time as an alternative to more expensive and less feasible methods like 24-h urine collection or food-based estimation is now a research priority in the public health area. We aimed to estimate salt intake among an Iranian urban population using spot urine-based equations and the FFQ-based method.
Material and methods
Study population
This cross-sectional sectional study was conducted in the framework of the Tehran Lipid and Glucose Study (TLGS), an ongoing population-based cohort study initiated in 1999 on a representative sample of males and females aged ≥ 3 years to investigate and prevent non-communicable diseases [23]. For this study, adult men and women (age ≥ 19 years) were recruited from the sixth examination of the TLGS (2014–2017) and included in the analyses if they had completed measurements on spot urinary Na, K, and creatinine, as well as demographics, anthropometrics, biochemical measurements, and usual dietary intakes. Under- or over-reported daily energy intake (< 800 kcal/d or > 4200 kcal/d, respectively) were considered as exclusion criteria, and final analyses were conducted on 2069 subjects (940 men and 1129 women).
Demographic and anthropometric measurements
Detailed measurements of the variables, including demographics and anthropometric measurements, were reported elsewhere [24]. In brief, body weight was measured using a digital scale (Seca, Hamburg, Germany) while the participant was minimally clothed, without shoes, and was reported to the nearest 100 g. Height was measured using a tape meter in a standing position without shoes and was reported to the nearest 0.5 cm. Body mass index (BMI) was calculated as weight (kg) divided by the square of the height (m2). Waist circumference was measured to the nearest of 0.1 cm at the level of the umbilicus, over light clothing, using a soft tape meter, and without any pressure to the body.
The physical activity was assessed using the Modifiable Activity Questionnaire (MAQ); the frequency and time spent on light, moderate, hard, and very hard intensity activities according to the list of everyday activities of daily life over the past year were documented and physical activity levels expressed as metabolic equivalent hours per week (MET-hour/week) [25]. Reliability and validity of the Persian version of the MAQ have previously been investigated [26].
Systolic (SBP) and diastolic (DBP) blood pressures were measured using a standard mercury sphygmomanometer calibrated by the Institute of Standards and Industrial Research of Iran [27]. Blood pressure was measured twice on the participants' right arm, after a 15-min rest in a sitting position, with at least a 30-s interval between two measurements. The two measurements’ mean was considered the participant's BP.
Urine sampling and measurement of urinary metabolites
Casual urine samples were obtained between 7:00 and 9:00 AM following overnight fasting. Aliquots of the casual urinary samples were frozen and sent to the central laboratory of the TLGS. Urinary concentrations of Na and K were measured by flame photometry (Screen lyte, Hospitex Diagnostics, Florence, Italy). Intra- and inter-assay coefficients of variation (CVs) were ≤ 2.8% and 4.8% for Na and K, respectively. Spot urinary Cr concentrations were measured using the Jaffe method; both inter- and intra-assay CVs were ≤ 5%.
Estimation of 24-h Na from the spot urine sample
Although the 24-h urine sampling is the gold standard for estimation of Na intake [18], it is criticized for potential bias due to under- and over-collection of samples, imposing considerable burden for participants, and low response rates (~ 10–40%), which affects data collection in representative population-based studies [21, 28]. In contrast, spot urine sampling has received much recent attention because it can easily incorporate into population-based settings without potential inaccuracy of sample collections [21, 28].
Due to the lack of an accurate and reliable predictive equation developed explicitly for our population, we applied the most commonly used and validated equations to estimate 24-h urinary excretion (mg/d) from Na concentration (mmol/L) of spot urine sample, including Kawasaki [29], Tanaka [30], Intersalt (also includes spot urine K concentration) [31], Toft [32], and Whitton [20]. Table 1 provides details of the equations with a brief description. The Kawasaki, Tanaka, and Whitton equations have been developed among Asian populations, while Intersalt and Toft equations have been developed among Western populations [20, 29–32].
Table 1.
Commonly used equations for estimating 24-h urinary Na excretion from the spot urine sample
| Name of equation | Description | History |
|---|---|---|
| Kawasaki (1993) [29] |
Male = 23 × 16.3 × [0.1 × (15.12 × Weight + 7.39 × Height − 12.63 × Age − 79.9) × Spot Na ÷ Spot Cr]0.5 Female = 23 × 16.3 × [0.1 × (8.58 × Weight + 5.09 × Height—4.72 × Age − 74.5) × Spot Na ÷ Spot Cr]0.5 |
Initially developed among a Japanese population (159 healthy, free-living 20–79 y adults, using second morning voiding urine specimens and 24-h urine sampling (for 3–5 days) |
| Tanaka (2002) [30] | Both genders = 23 × 21.98 × (0.1 × [- 2.04 × Age + 14.89 × Weight + 16.14 × Height − 2244.45] × Spot Na ÷ Spot Cr)0.392 | Initially developed among a Japanese population (591 adults, 20–59 y) using casual urine specimens and 24-h urine sampling |
| Intersalt (2013) [31] |
Male = 23 × (25.46 + 0.46 × Spot Na—2.75 × Spot Cr − 0.13 × Spot K + 4.10 × BMI + 0.26 × Age) Female = 23 × (5.07 + 0.34 × Spot Na—2.16 × Spot Cr − 0.09 × Spot K + 2.39 × BMI + 2.35 × Age − 0.03 × Age2) |
Developed among the North American and European populations (5693 adults aged 20–59 y) |
| Toft (2014) [32] |
Male = 33.56 × [Spot Na ÷ Spot Cr × (− 7.54 × age + 14.15 × weight + 3.48 × height + 423.15]0.345 Female = 52.65 × [Spot Na ÷ Spot Cr × (− 6.13 age + 9.97 × weight + 1.24 × height + 342.73] 0.196 |
Developed among Danish individuals (473) using 24-h urine collection and a spot urine sampling |
| Whitton (2016) [20] | Both genders (for morning sample) = 23 × (88.66 + 0.55 × Spot Na—1.34 × Spot Cr—1.05 × Spot K − 0.87 × Age + 2.10 × BMI + 39.30 × Sex (male = 1, female = 0) + Ethnicity† (Malay = − 17.70, Indian = − 10.38, Chinese = 0) | Developed among Singaporean or permanent residents of Singapore (144 subjects, aged 18–79 y) using multiple spot urine sampling (morning, afternoon, evening) and a 24-h urine sampling |
Body weight in kg, BMI in kg/m2, spot Na in mmol/L, spot Cr in mmol/L, age in years, height in cm
†Considered similar to Chinese as 0
The values of Na intake (mg/d) were converted to salt intake (g/d) by multiplying the value of Na excretion (mg/d) by 0.00254 (2.54 ÷ 1000). The estimated 24-h urinary excretion from a single spot urine sample provided the Na status of the participants in a single day.
Dietary assessment
The usual dietary intakes of the participants over the previous year were assessed using a validated semi-quantitative 147-item FFQ. Details of dietary assessment in the TLGS were described elsewhere [33]. In brief, the frequency of food items consumed during the past year was asked daily, weekly, or monthly. Portion sizes of consumed foods reported in household measures were converted to the gram. Since the Iranian Food Composition Table is incomplete and has limited data on raw foods and beverages' nutrient content, the US Department of Agriculture Food Composition Table was used [34]. Usual dietary intake of Na and K were also obtained from nutritional data and are reported as mmol/d. The FFQ provided the mean intake of Na and K of the participants over the last year.
Statistical methods
Statistical analyses were conducted using SPSS for Windows version 20 (SPSS Inc., Chicago, IL, USA) and the GraphPad Prism version 8.00 for Windows (GraphPad Software, CA, USA). A two-tailed P value < 0.05 was considered statistically significant. Dietary intakes of Na and K were adjusted for total energy intake using the residuals’ method [35]. Mean, and standard deviation (SD) of values and the frequency (%) of characteristics of the participants were compared between men and women using an independent sample t-test or Chi-square test.
Due to the lack of accessibility to 24-h urine samples, as the gold standard, we compared each estimation with the mean of all estimations (spot urine-based equations and FFQ-based estimation). In the case of lacking a gold standard method for method comparison, mean of available assay methods can be considered as the reference [36, 37].
Bland–Altman difference plots were used to assess the agreement between each assay and the overall mean (i.e., provided as estimated mean bias and 95% CI). The regression equation (slope and intercept) for bias [difference of each estimation as dependent variable (y) vs. overall mean of 6 estimations as independent variables (x)] was determined using least squared perpendicular distance regression analysis (Deming’s method) [38], which is preferred over ordinary linear regression (OLR) for method comparison studies in which both variables are measured with error [39]. In OLR, it is assumed that random error, arising from inherent limitations of measurements, is constant over the range of the data whereas, in the Deming’s regression, random errors of both compared method are taken into account [40]. In Deming’s regression, both x and y variables are subject to error, and the squares of the perpendicular distances of the x and y points from the regression line are minimized [38].
Result
The mean age of the participants was 45.6 ± 14.8 y, and 45.4% were men. Table 2shows the characteristics of the study participants. Mean casual urinary Na concentration was 135 ± 56.5 mmol/L (143 ± 54.9 and 127 ± 56.8 in mmol/L, in men and women, respectively, P < 0.05); urinary Na-to-K ratio was higher in men compared to women (2.39 ± 1.45 vs. 2.19 ± 1.34, P < 0.05). The mean estimated Na intake was 144 ± 36.3 mmol/d (142 ± 37.3 and 145 ± 35.4, in men and women, respectively).
Table 2.
Characteristics of the study participants
| Total (n = 2069) |
Men (n = 940) |
Women (n = 1129) |
|
|---|---|---|---|
| Age, y | 45.6 ± 14.8 | 46.7 ± 15.7 | 44.6 ± 13.8* |
| BMI, kg/m2 | 27.9 ± 5.2 | 27.4 ± 4.4 | 28.2 ± 5.7* |
| Waist circumference, cm | 93.5 ± 12.5 | 95.9 ± 11.6 | 91.5 ± 12.9* |
| Systolic blood pressure, mmHg | 114 ± 17 | 119 ± 16 | 109 ± 17* |
| Diastolic blood pressure, mmHg | 75.6 ± 10.5 | 78.2 ± 10.4 | 73.4 ± 10.2* |
| Casual urinary Cr, mmol/L | 13.8 ± 6.1 | 14.2 ± 6.0 | 13.5 ± 6.2* |
| Estimated† 24-h urinary Cr, mmol/d | 13.0 ± 4.0 | 16.5 ± 3.3 | 10.0 ± 10.3* |
| Casual urinary Na, mmol/L | 135 ± 56.5 | 143 ± 54.9 | 127 ± 56.8* |
| Casual urinary K, mmol/L | 71.6 ± 36.2 | 72.8 ± 35.3 | 70.7 ± 36.9 |
| Casual urinary Na-to-K ratio | 2.28 ± 1.39 | 2.39 ± 1.45 | 2.19 ± 1.34* |
| FFQ-estimated‡ Na intake, mmol/d | 144 ± 36.3 | 142 ± 37.3 | 145 ± 35.4 |
| FFQ-estimated‡ K intake, mmol/d | 109 ± 28.5 | 104 ± 28.6 | 112 ± 28.2* |
| FFQ-estimated Na-to-K ratio | 1.48 ± 2.16 | 1.59 ± 3.14 | 1.38 ± 0.57 |
| Energy intake, kcal/d | 2300 ± 811 | 2501 ± 896 | 2134 ± 690* |
| Dietary protein, g/d | 90 ± 41.6 | 98.4 ± 44.9 | 83.0 ± 37.2* |
| Dietary fat, g/d | 75.7 ± 32.5 | 79.7 ± 36.7 | 72.4 ± 28.2* |
| Dietary carbohydrate, g/d | 342 ± 125 | 376 ± 135 | 313 ± 108* |
| Dietary fiber, g/d | 45.1 ± 22.3 | 49.3 ± 23.9 | 41.6 ± 20.2* |
| Physical activity, METs h/week | 69.4 ± 42.6 | 85.6 ± 46.5 | 56.1 ± 33.8* |
| Current smoking, % | 16.7 | 30.2 | 5.40* |
| Educational level | |||
| Illiterate, % | 1.50 | 1.00 | 2.00* |
| Diploma, % | 59.5 | 58.6 | 60.3 |
| Academic, % | 40.5 | 41.4 | 39.7 |
*P < 0.05 compared to men using independent sample t-test or Chi-square was used
†Based on Kawasaki equation
‡Residual-energy adjusted
BMI, body mass index; FFQ, food frequency questionnaire; Na, sodium; K, potassium; Cr, creatinine
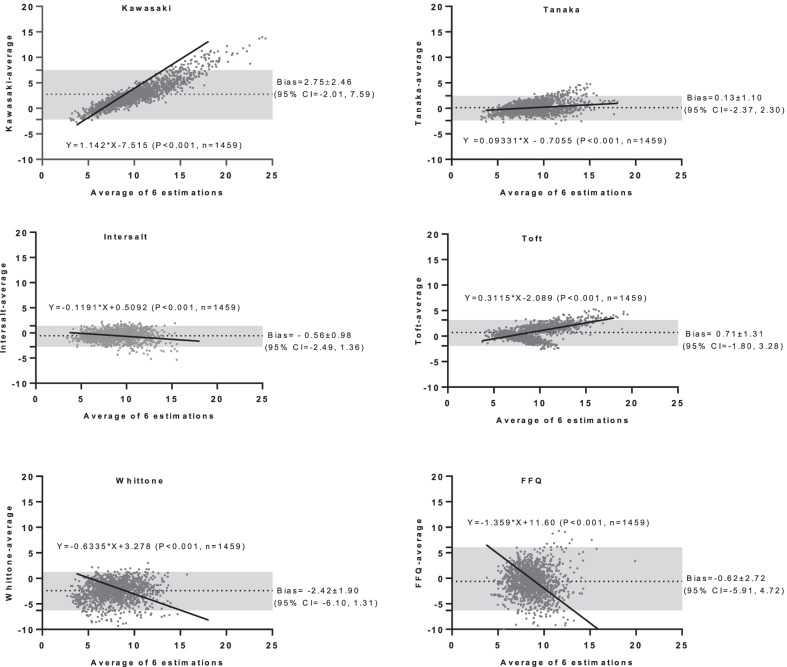
Mean (SD) estimated salt intake, derived from the overall mean of the methods, was 9.0 ± 2.2 g/d (10.2 ± 2.1 and 7.9 ± 1.7 in men and women, respectively). Compared with the gold-estimated salt intake, derived from the average of all methods, the mean bias ranged from − 2.42 to 2.75 g/d, with the Tanaka equation, had the least bias (mean bias = 0.13 ± 1.10, 95% CI − 2.37, 2.30 g/d) and the Kawasaki (mean bias = 2.75 ± 2.46, 95% CI − 2.01, 7.59 g/d) and Whitton (mean bias = − 2.42 ± 1.90, 95% CI − 6.10, 1.31 g/d) equations had the most bias (Fig. 1).
Fig. 1.
Mean bias, SD and 95% CI of each method compared to the gold-estimated salt intake (derived from the average of all methods), along with the slope and intercept for bias of each estimation vs. overall mean of 6 estimations
The slope and intercept for bias of each estimation vs. the overall mean of 6 estimates, obtained by the least squared perpendicular distance regression analysis, are reported in Table 3. The table also shows the least-biased point of each equation, i.e., the mean bias of the estimation to the overall mean was tended to be zero. The values showed that the most accurate equations for low- and high-salt intake levels are Intersalt and FFQ, respectively (least-biased point = 4.3 and 8.5 g/d salt intake). The least-biased point for the Tanaka equation was 7.6 g/d. The difference to overall mean at the low, medium, and high-salt intake for the estimations are provided in Table 3. The Tanka equation showed a mean bias < 1 g/d (range − 0.2 to 0.7 g/d) at all levels of salt intake. The Intersalt underestimated salt intake at low-, medium-, and high-level of salt intake, while Kawasaki, Toft, and Tanaka underestimated at low intake (< 5 g/d) and overestimated at high intake (> 15 g/d). Inversely, the Whitton equation and FFQ overestimated at low intake and underestimated at a high level of salt intake.
Table 3.
The slope and intercept for bias of each estimation vs. overall mean of 6 estimations
| Methods of estimation | Linear regression model | Zero difference to overall mean | Difference from overall mean at the low, medium and high salt intake (g/d) | |||
|---|---|---|---|---|---|---|
| Slope | Intercept | 5 | 10 | 15 | ||
| Kawasaki |
1.14 (1.10, 1.18) |
− 7.52 (− 7.91, − 7.12) |
6.6 | − 1.8 | 3.9 | 9.6 |
| Tanaka |
0.09 (0.06, 0.18) |
− 0.71 (− 1.02,− 0.39) |
7.6 | − 0.2 | 0.2 | 0.7 |
| Intersalt |
− 0.12 (− 0.15, 0.09) |
+ 0.51 (0.25, 0.76) |
4.3 | − 0.1 | − 0.7 | − 1.3 |
| Toft |
0.31 (0.27, 0.35) |
− 2.09 (− 2.45, − 1.73) |
6.7 | − 0.55 | 1.0 | 2.6 |
| Whitton |
− 0.63 (− 0.73, − 0.54) |
+ 3.28 (2.39, 4.16) |
5.2 | 0.1 | − 3.1 | − 6.2 |
| FFQ |
− 1.36 (− 1.44, − 1.28) |
+ 11.60 (10.88, 12.32) |
8.5 | 4.8 | − 2.0 | − 8.8 |
The Tanaka, the most fitted equation for salt estimation among our population, estimated a mean salt intake of 8.9 g/d (9.5 and 8.3 g/d, in men and women, respectively) with a range of 2.1 to 18.7 g/d. Only 5.1% (1.9% and 7.7% in men and women, respectively) adhered to WHO recommendation (< 5 g/d salt intake), whereas 26.8% (33.5% and 21.5% in men and women, respectively) and 2.4% (3.0% and 1.9% in men and women, respectively) exceeded the recommendation by more than 2- and threefold. About 6.6%, 12.0%, 15.4%, 16.9%, and 14.8% of the population exceeded the recommendation of salt intake by 1, 2, 3, 4, and 5 g/d, respectively.
Discussion
Our study indicated that the Tanaka equation, conducted based on urinary Na, K, and Cr concentrations from casual urine specimens may be a helpful method for estimating mean 24-h Na excretion and population salt intake. Compared with the gold-estimated salt intake, i.e., derived from the average of urine-based and FFQ-based procedures, the Tanaka equation provided the least biased estimation with a mean bias < 1 g/d at low-, medium-, or high-level of salt intake among our population. Population salt estimation using the Tanaka equation showed that most of the Iranian population (about 95%) exceeded the current recommended limits of salt intake.
The performance of usual equations used for 24-h Na excretion from spot urine samples has remained inconsistent among different populations [19]. The estimated 24-h urinary Na using the usual equations (i.e., Kawasaki, Tanaka, and Intersalt) were reported to be systematically biased with overestimation at lower levels and underestimation at higher levels of Na intake; other variables in the equations seem to contribute to the biased estimates since a similar systematic bias occurred where Na concentration was kept constant (41). Similar to our observation, the Kawasaki equation overestimates mean Na intake among several populations [41–43]. The Intersalt was more accurate than other equations estimating salt intake among different populations [41]. Dietary methods are also used to estimate population salt intake; however, they tend to underestimate Na intake due to the under-reporting of discretionary sources of salt [19, 21].
In our study, about 5.1% met salt intake recommendations, and about 26.8% and 2.4% exceeded by more than 2- and 3-fold. A recent national report of estimated Iranian salt intake indicated a mean population of 9.52 g/d (95% CI 9.48–9.56) and ~ 97.7% overconsumption of salt among the population [44].
It remains disputable whether there is a “healthy” or “normal range” of Na intake [45]; a range of 0.5–1 [46] up to 55 [47] g/d salt intake was reported (based on urinary Na excretion) among populations. The WHO technical report on “Reducing Salt Intake in Populations” in 2006 represents a wide range of 24-h urinary Na (from 0.8 to 299 mmol/d) among different populations; this report indicates that most people appear to have mean 24-h urinary Na over 100 mmol/d, and among the Asian countries this value exceeded 200 mmol/d [48]. Combining 197 datasets (69 799 subjects) over a 3-decade period indicates that human Na intake follows a classic normal distribution that depicts a narrow range with strict lower and upper limits of normality (mean = 159.2, range 90–248 mmol/d; mean after removal of outliers = 159.4, range 114–210 mmol/d) [17].
The WHO Global Non-communicable Diseases Action Plan 2013–2020 sets a 30% relative reduction in the mean population Na intake by 2025 [49]. Despite 4-decade comprehensive public policies, the distribution of population Na intake has not changed over time [17], and long-term low-Na-diet trials (85 mmol/d Na) could not achieve a significant reduction in Na intake [50]. Meta-analysis of both randomized clinical trials and observational studies resulted in a weak association between salt intake and BP, especially among non-obese individuals with normal BP [51, 52]; an effect size of 1 mm Hg in BP following a low-Na diet does not justify a general restriction for Na intake [52]. On the other hand, a salt intake lower than 5.8 g/d was reported to be associated with the activation of the renin–angiotensin–aldosterone system, increased plasma lipids, and increased mortality [51]; risk of myocardial infarction, cardiovascular diseases, and all-cause mortality was increased among hypertensive patients with increased plasma renin activity and low-urinary levels of Na [53, 54]. Furthermore, a meta-analysis of population Na intake showed a "U shape" relationship with the risk of mortality [55], and mean estimated Na intake was inversely associated with mortality at a level of < 4 g/d (10 g/d salt) [41]. Previous studies among our population also showed no significant association between dietary intake of Na and the risk of hypertension, CVD, and renal dysfunction [56, 57]. Current evidence relating Na intake to hypertension and CVD has significant limitations [9] and could not provide a strong statement on the adverse effect of Na intake on CVD outcomes and all-cause mortality [58].
Na intake appears to be set by human physiology, maintaining a minimal Na intake close to the lower limit of the normal range and approaching the upper limit obtained by global estimation of urinary Na excretion [45]. Such evidence may call for revisiting the dietary salt guidelines and maintaining optimal Na intake within the normal range identified by worldwide 24-h urinary Na surveys of populations [51].
Although it can be different among populations, about 75% of dietary Na is attributed to processed foods, 10–12% is naturally occurring in foods, and the rest of 10–15% is discretionary salt intake (salt used in home-cooking or at the table) [48, 59]. Bread products, cereal, and grains have been responsible for about 40% of total Na intake [48]; meat and dairy products are the major contributors to dietary Na intake in most populations [60]. More than half of their daily salt intake is from discretionary sources in some countries (i.e., Brazil, China, Costa Rica, Guatemala, India, Japan, Mozambique, Romania) [60].
As a strength, this was the first study among an Iranian urban population with a relatively large sample size that estimated salt intake using several urine-based methods and an FFQ-based approach. Our study also had some limitations; first, due to the lack of 24-h urinary samples as the gold standard method for measuring Na's urinary excretion and salt intake, we used the overall mean of the estimations to assess the performance of equations. The second and the most critical limitation of the present study was that the equations used for salt estimation were initially developed and validated for other populations; we, therefore, need to establish a best-fitted equation for our population. Finally, FFQ provided the mean Na intake of the participants over the last year, however, spot urine sample possibly provided Na status of a single day, and therefore, FFQ seems to be different from a urine sample in estimating Na intake.
Conclusion
In conclusion, our study also showed that about 95% of the Iranian population exceeded current salt intake recommendations. The study also indicated that the Tanaka equation might be the best model for population salt intake using spot specimens among our people. Since the performance of casual urine samples for estimating Na excretion was reported to be independent of the time of urine sample collection (i.e., overnight, morning, afternoon, and evening), this method seems to have priority as the best alternative of 24-h urine sample collection for estimating population salt intake.
Acknowledgements
We thank the Tehran Lipid and Glucose Study participants and the field investigators of the Tehran Lipid and Glucose Study for their cooperation and assistance in physical examinations, biochemical evaluation and database management. This study, was supported by the Research Institute for Endocrine Sciences of Shahid Beheshti University of Medical Sciences.
Abbreviations
- BMI
Body mass index
- Cr
Creatinine
- DALYs
Disability-adjusted life-years
- DBP
Diastolic blood pressure
- DGA
Dietary Guidelines for Americans
- FFQ
Food frequency questionnaire
- K
Potassium
- Na
Sodium
- OLR
Ordinary linear regression
- SBP
Systolic blood pressure
- SD
Standard deviation
- TLGS
Tehran Lipid and Glucose Study
- WHO
World Health Organization
Authors' contributions
ZB and AGh designed the study. ZB, RN and AGh analyzed the data. ZB, AGh, and PM wrote the manuscript. FA supervised the work. All authors read and approved the final manuscript.
Funding
Not applicable.
Availability of data and materials
Data will be presented upon forwarding the request to the corresponding author (ghasemi@endocrine.ac.ir) and confirmation of the director of RIES (azizi@endocrine.ac.ir).
Declarations
Ethical approval and consent to participate
We obtained written informed consent from all participants. Based on the ethical guidelines of the 1975 Declaration of Helsinki, the study protocol was approved by the Ethics Research Council of the Research Institute for Endocrine Sciences, Shahid Beheshti University of Medical Sciences.
Consent for publication
Not applicable.
Competing interests
The authors have no conflict of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Roth GA, Mensah GA, Johnson CO, Addolorato G, Ammirati E, Baddour LM, et al. Global burden of cardiovascular diseases and risk factors, 1990–2019: update from the GBD 2019 study. J Am Coll Cardiol. 2020;76(25):2982–3021. doi: 10.1016/j.jacc.2020.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Worldwide trends in hypertension prevalence and progress in treatment and control from 1990 to 2019: a pooled analysis of 1201 population-representative studies with 104 million participants. Lancet (London, England). 2021;398(10304):957–80. [DOI] [PMC free article] [PubMed]
- 3.Wright JM, Musini VM, Gill R. First-line drugs for hypertension. Cochrane Database Syst Rev. 2018;4(4):CD001841-CD. [DOI] [PMC free article] [PubMed]
- 4.Kolahdouz-Mohammadi R, Malekahmadi M, Clayton ZS, Sadat SZ, Pahlavani N, Sikaroudi MK, et al. Effect of egg consumption on blood pressure: a systematic review and meta-analysis of Randomized Clinical Trials. Curr Hypertens Rep. 2020;22(3):24. doi: 10.1007/s11906-020-1029-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rasad H, Entezari MH, Ghadiri E, Mahaki B, Pahlavani N. The effect of honey consumption compared with sucrose on lipid profile in young healthy subjects (randomized clinical trial) Clin Nutr ESPEN. 2018;26:8–12. doi: 10.1016/j.clnesp.2018.04.016. [DOI] [PubMed] [Google Scholar]
- 6.Mansouri M, Pahlavani N, Sharifi F, Varmaghani M, Shokri A, Yaghubi H, et al. Dairy consumption in relation to hypertension among a large population of university students: the MEPHASOUS study. Diabetes Metab Syndrome Obesity: Targets Therapy. 2020;13:1633. doi: 10.2147/DMSO.S248592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hadi V, Pahlavani N, Malekahmadi M, Nattagh-Eshtivani E, Navashenaq JG, Hadi S, et al. Nigella sativa in controlling Type 2 diabetes, cardiovascular, and rheumatoid arthritis diseases: molecular aspects. J Res Med Sci. 2021;26:20. doi: 10.4103/jrms.JRMS_236_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Health effects of dietary risks in 195 countries, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet (London, England). 2019;393(10184):1958–72. [DOI] [PMC free article] [PubMed]
- 9.Farquhar WB, Edwards DG, Jurkovitz CT, Weintraub WS. Dietary sodium and health: more than just blood pressure. J Am Coll Cardiol. 2015;65(10):1042–1050. doi: 10.1016/j.jacc.2014.12.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Alderman MH. Dietary sodium: where science and policy diverge. Am J Hypertens. 2016;29(4):424–427. doi: 10.1093/ajh/hpu256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.McGuire S. Institute of Medicine. 2013. Sodium Intake in Populations: Assessment of Evidence. Washington, DC: The National Academies Press, 2013. Advances in Nutrition. 2014;5(1):19–20. [DOI] [PMC free article] [PubMed]
- 12.World Health Organization. Reducing salt intake in populations: report of a WHO forum and technical meeting. Paris. France;2007:924159537X.
- 13.World Health Organization. Action plan for the prevention and control of noncommunicable diseases in the WHO European Region. 2016.
- 14.World Health Organization and Food and Agriculture Organization. Diet, nutrition, and the prevention of chronic diseases: report of a joint WHO/FAO expert consultation. World Health Organization, 2003 924120916X. [PubMed]
- 15.McGuire S. U.S. Department of Agriculture and U.S. Department of Health and Human Services, Dietary Guidelines for Americans, 2010. 7th Edition, Washington, DC: U.S. Government Printing Office, January 2011. Advances in nutrition (Bethesda, Md). 2011;2(3):293–4. [DOI] [PMC free article] [PubMed]
- 16.Organization WH. Guideline: Sodium intake for adults and children: World Health Organization; 2012. [PubMed]
- 17.McCarron DA, Kazaks AG, Geerling JC, Stern JS, Graudal NA. Normal range of human dietary sodium intake: a perspective based on 24-hour urinary sodium excretion worldwide. Am J Hypertens. 2013;26(10):1218–1223. doi: 10.1093/ajh/hpt139. [DOI] [PubMed] [Google Scholar]
- 18.WHO/PAHO Regional Expert Group for Cardiovascular Disease Prevention through Population-wide Dietary Salt Reduction. Protocol for population level sodium determination in 24-hour urine samples. WHO Press: World Health Organization., 2010.
- 19.Ginos BNR, Engberink R. Estimation of sodium and potassium intake: current limitations and future perspectives. Nutrients. 2020;12(11):3275. doi: 10.3390/nu12113275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Whitton C, Gay GMW, Lim RBT, Tan LWL, Lim W-Y, van Dam RM. Evaluation of equations for predicting 24-hour urinary sodium excretion from casual urine samples in Asian adults. J Nutr. 2016;146(8):1609–1615. doi: 10.3945/jn.116.232108. [DOI] [PubMed] [Google Scholar]
- 21.McLean RM. Measuring population sodium intake: a review of methods. Nutrients. 2014;6(11):4651–4662. doi: 10.3390/nu6114651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.McLean RM, Farmer VL, Nettleton A, Cameron CM, Cook NR, Campbell NRC, et al. Assessment of dietary sodium intake using a food frequency questionnaire and 24-h urinary sodium excretion: a systematic literature review. J Clin Hypertension. 2017;19(12):1214–1230. doi: 10.1111/jch.13148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Azizi F, Rahmani M, Emami H, Mirmiran P, Hajipour R, Madjid M, et al. Cardiovascular risk factors in an Iranian urban population: Tehran lipid and glucose study (phase 1) Soz Praventivmed. 2002;47(6):408–426. doi: 10.1007/s000380200008. [DOI] [PubMed] [Google Scholar]
- 24.Azizi F, Zadeh-Vakili A, Takyar M. Review of rationale, design, and initial findings: Tehran Lipid and Glucose Study. Int J Endocrinol Metab. 2018;16(4 (Suppl)):e84777. doi: 10.5812/ijem.84777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ainsworth BE, Haskell WL, Leon AS, Jacobs DR, Jr, Montoye HJ, Sallis JF, et al. Compendium of physical activities: classification of energy costs of human physical activities. Med Sci Sports Exerc. 1993;25(1):71–80. doi: 10.1249/00005768-199301000-00011. [DOI] [PubMed] [Google Scholar]
- 26.Momenan AA, Delshad M, Sarbazi N, Rezaei Ghaleh N, Ghanbarian A, Azizi F. Reliability and validity of the Modifiable Activity Questionnaire (MAQ) in an Iranian urban adult population. Arch Iran Med. 2012;15(5):279–282. [PubMed] [Google Scholar]
- 27.Askari S, Asghari G, Ghanbarian A, Khazan M, Alamdari S, Azizi F. Seasonal variations of blood pressure in adults: Tehran lipid and glucose study. Arch Iran Med. 2014;17(6):441–443. [PubMed] [Google Scholar]
- 28.Hawkes C, Webster J. National approaches to monitoring population salt intake: a trade-off between accuracy and practicality? PLoS ONE. 2012;7(10):e46727. doi: 10.1371/journal.pone.0046727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kawasaki T, Itoh K, Uezono K, Sasaki H. A simple method for estimating 24 h urinary sodium and potassium excretion from second morning voiding urine specimen in adults. Clin Exp Pharmacol Physiol. 1993;20(1):7–14. doi: 10.1111/j.1440-1681.1993.tb01496.x. [DOI] [PubMed] [Google Scholar]
- 30.Tanaka T, Okamura T, Miura K, Kadowaki T, Ueshima H, Nakagawa H, et al. A simple method to estimate populational 24-h urinary sodium and potassium excretion using a casual urine specimen. J Hum Hypertens. 2002;16(2):97–103. doi: 10.1038/sj.jhh.1001307. [DOI] [PubMed] [Google Scholar]
- 31.Brown IJ, Dyer AR, Chan Q, Cogswell ME, Ueshima H, Stamler J, et al. Estimating 24-hour urinary sodium excretion from casual urinary sodium concentrations in Western populations: the INTERSALT study. Am J Epidemiol. 2013;177(11):1180–1192. doi: 10.1093/aje/kwt066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Toft U, Cerqueira C, Andreasen AH, Thuesen BH, Laurberg P, Ovesen L, et al. Estimating salt intake in a Caucasian population: can spot urine substitute 24-hour urine samples? Eur J Prev Cardiol. 2014;21(10):1300–1307. doi: 10.1177/2047487313485517. [DOI] [PubMed] [Google Scholar]
- 33.Hosseini-Esfahani F, Hosseinpour-Niazi S, Asghari G, Bahadoran Z, Moslehi N, Golzarand M, et al. Nutrition and Cardio-Metabolic Risk Factors: Findings from 20 Years of the Tehran Lipid and Glucose Study. Int J Endocrinol Metab. 2018;16(4 (Suppl)):e84772. doi: 10.5812/ijem.84772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.US. Food Composition Table. Available at: http://www.ndbnalusdagov/ndb/search.
- 35.Willett WC, Howe GR, Kushi LH, et al. Adjustment for total energy intake in epidemiologic studies. Am J Clin Nutr. 1997;65(4):1220S–8S. doi: 10.1093/ajcn/65.4.1220S. [DOI] [PubMed] [Google Scholar]
- 36.Manley SE, Stratton IM, Clark PM, Luzio SD. Comparison of 11 human insulin assays: implications for clinical investigation and research. Clin Chem. 2007;53(5):922–932. doi: 10.1373/clinchem.2006.077784. [DOI] [PubMed] [Google Scholar]
- 37.Tohidi M, Arbab P, Ghasemi A. Assay-dependent variability of serum insulin concentrations: a comparison of eight assays. Scand J Clin Lab Invest. 2017;77(2):122–129. doi: 10.1080/00365513.2016.1278260. [DOI] [PubMed] [Google Scholar]
- 38.Ludbrook J. Linear regression analysis for comparing two measurers or methods of measurement: but which regression? Clin Exp Pharmacol Physiol. 2010;37(7):692–699. doi: 10.1111/j.1440-1681.2010.05376.x. [DOI] [PubMed] [Google Scholar]
- 39.Payne RB. Deming's regression analysis in method comparison studies. Ann Clin Biochem. 1985;22(Pt 4):430. doi: 10.1177/000456328502200419. [DOI] [PubMed] [Google Scholar]
- 40.Martin RF. General deming regression for estimating systematic bias and its confidence interval in method-comparison studies. Clin Chem. 2000;46(1):100–104. [PubMed] [Google Scholar]
- 41.He FJ, Ma Y, Campbell NRC, MacGregor GA, Cogswell ME, Cook NR. Formulas to estimate dietary sodium intake from spot urine alter sodium-mortality relationship. Hypertension (Dallas, Tex : 1979). 2019;74(3):572–80. [DOI] [PubMed]
- 42.Mill JG, Rodrigues SL, Baldo MP, Malta DC, Szwarcwald CL. Validation study of the Tanaka and Kawasaki equations to estimate the daily sodium excretion by a spot urine sample. Braz J Epidemiol. 2015;18(Suppl 2):224–237. doi: 10.1590/1980-5497201500060020. [DOI] [PubMed] [Google Scholar]
- 43.Allen NB, Zhao L, Loria CM, Van Horn L, Wang C-Y, Pfeiffer CM, et al. The validity of predictive equations to estimate 24-hour sodium excretion: the MESA and CARDIA Urinary Sodium Study. Am J Epidemiol. 2017;186(2):149–159. doi: 10.1093/aje/kwx056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Rezaei S, Mahmoudi Z, Sheidaei A, Aryan Z, Mahmoudi N, Gohari K, et al. Salt intake among Iranian population: the first national report on salt intake in Iran. J Hypertens. 2018;36(12):2380–2389. doi: 10.1097/HJH.0000000000001836. [DOI] [PubMed] [Google Scholar]
- 45.McCarron DA. What determines human sodium intake: policy or physiology? Adv Nutr. 2014;5(5):578–584. doi: 10.3945/an.114.006502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Oliver WJ, Cohen EL, Neel JV. Blood pressure, sodium intake, and sodium related hormones in the Yanomamo Indians, a "no-salt" culture. Circulation. 1975;52(1):146–151. doi: 10.1161/01.cir.52.1.146. [DOI] [PubMed] [Google Scholar]
- 47.Dahl LK. Possible role of salt intake in the development of essential hypertension. 1960. In J Epidemiol. 2005;34(5):967–72; discussion 72–4, 75–8. [DOI] [PubMed]
- 48.Elliott P, Brown I. Sodium intakes around the world. 2007.
- 49.World Health Organization. WHO Global Action Plan for the Prevention and Control of Noncommunicable Diseases 2013–20. Available online: http://apps.who.int/iris/bitstream/10665/94384/1/9789241506236_eng.pdf?ua=1. .
- 50.The Trials of Hypertension Prevention Collaborative Research Group. Effects of weight loss and sodium reduction intervention on blood pressure and hypertension incidence in overweight people with high-normal blood pressure. The Trials of Hypertension Prevention, phase II. Arch Internal Med. 1997;157(6):657–67. [PubMed]
- 51.Graudal N, Jürgens G. Conflicting evidence on health effects associated with salt reduction calls for a redesign of the salt dietary guidelines. Prog Cardiovasc Dis. 2018;61(1):20–26. doi: 10.1016/j.pcad.2018.04.008. [DOI] [PubMed] [Google Scholar]
- 52.Graudal NA, Galløe AM, Garred P. Effects of sodium restriction on blood pressure, renin, aldosterone, catecholamines, cholesterols, and triglyceride: a meta-analysis. JAMA. 1998;279(17):1383–1391. doi: 10.1001/jama.279.17.1383. [DOI] [PubMed] [Google Scholar]
- 53.Alderman MH, Madhavan S, Cohen H, Sealey JE, Laragh JH. Low urinary sodium is associated with greater risk of myocardial infarction among treated hypertensive men. Hypertension (Dallas, Tex : 1979). 1995;25(6):1144–52. [DOI] [PubMed]
- 54.Alderman MH, Ooi WL, Cohen H, Madhavan S, Sealey JE, Laragh JH. Plasma renin activity: a risk factor for myocardial infarction in hypertensive patients. Am J Hypertens. 1997;10(1):1–8. doi: 10.1016/s0895-7061(96)00301-9. [DOI] [PubMed] [Google Scholar]
- 55.Graudal N, Jürgens G, Baslund B, Alderman MH. Compared with usual sodium intake, low- and excessive-sodium diets are associated with increased mortality: a meta-analysis. Am J Hypertens. 2014;27(9):1129–1137. doi: 10.1093/ajh/hpu028. [DOI] [PubMed] [Google Scholar]
- 56.Mirmiran P, Bahadoran Z, Nazeri P, Azizi F. Dietary sodium to potassium ratio and the incidence of hypertension and cardiovascular disease: A population-based longitudinal study. Clin Exp Hypertension (New York, NY: 1993). 2018;40(8):772–9. [DOI] [PubMed]
- 57.Mirmiran P, Nazeri P, Bahadoran Z, Khalili-Moghadam S, Azizi F. Dietary sodium to potassium ratio and the incidence of chronic kidney disease in adults: a longitudinal follow-up study. Prevent Nutr Food Sci. 2018;23(2):87–93. doi: 10.3746/pnf.2018.23.2.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Committee on the Consequences of Sodium Reduction in P, Food, Nutrition B, Board on Population H, Public Health P, Institute of M. Sodium Intake in Populations: Assessment of Evidence. Strom BL, Yaktine AL, Oria M, editors. Washington (DC): National Academies Press (US) [PubMed]
- 59.Copyright 2013 by the National Academy of Sciences. All rights reserved.; 2013.
- 60.Brown IJ, Tzoulaki I, Candeias V, Elliott P. Salt intakes around the world: implications for public health. Int J Epidemiol. 2009;38(3):791–813. doi: 10.1093/ije/dyp139. [DOI] [PubMed] [Google Scholar]
- 61.Bhat S, Marklund M, Henry ME, Appel LJ, Croft KD, Neal B, et al. A systematic review of the sources of dietary salt around the World. Adv Nutr (Bethesda, Md) 2020;11(3):677–686. doi: 10.1093/advances/nmz134. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data will be presented upon forwarding the request to the corresponding author (ghasemi@endocrine.ac.ir) and confirmation of the director of RIES (azizi@endocrine.ac.ir).