Abstract
Purpose of review
Adverse pregnancy outcomes are associated with increased risk for future cardiovascular disease. The goal of this review is to share what is currently known about the increased risk and to identify areas for future research.
Recent findings
Severe studies have identified a strong association between adverse pregnancy outcomes and cardiovascular disease such as heart failure, valvular disease, ischemic heart disease, stroke, hypertension, and metabolic syndrome. The recognition of this increased risk is reflected in recent changes in prevention guidelines. The guidelines now recognize sex-specific risks such as preeclampsia and preterm delivery and recommend incorporating a pregnancy history to identify them earlier. However, no robust risk prediction tools incorporating these pregnancy risk factors have been developed and validated. While smaller clinical trials have been performed in reducing cardiovascular risk factors in the postpartum timeframe, there remains a paucity of large-scale randomized clinical trials that continue to show a risk reduction in these women.
Summary
While there is increasing recognition of the long-term cardiovascular risks associated with adverse pregnancy outcomes, there remains a need for interventional studies aimed at reducing this risk and for incorporation of pregnancy risk factors into traditional cardiovascular risk prediction tools.
Keywords: Adverse pregnancy outcomes, Preeclampsia, Gestational diabetes, Preterm delivery, Maternal cardiovascular disease, Aspirin
Introduction
Adverse pregnancy outcomes (APOs) are common and occur in 17–20% of all pregnancies in the USA [1, 2, 3•]. These include hypertensive disorders of pregnancy (HDP), such as preeclampsia and gestational hypertension, gestational diabetes mellitus (GDM), preterm delivery, and small for gestational age (SGA) infants. While APOs can result in short-term complications during pregnancy, APOs are increasingly associated with deleterious long-term complications, especially cardiovascular disease (CVD), such as coronary artery disease (CAD), stroke, or heart failure [4–6]. This has led to the hypothesis that pregnancy may act as a “stress test” for future CVD as it may unmask underlying risks of poor or suboptimal cardiovascular health in women.
Certainly, APOs and CVD share similar risk factors, including obesity, hypertension, metabolic syndrome, race, and low socioeconomic status (Fig. 1). It is unclear though, whether the increased CVD risk is related to these shared risk factors or whether other mechanisms and attributes contribute to the elevated long-term CVD risk associated with APOs. In this review, we explore the epidemiology for common APOs, what is currently known regarding their association with increased risk of CVD, and what remains to be learned.
Fig. 1.
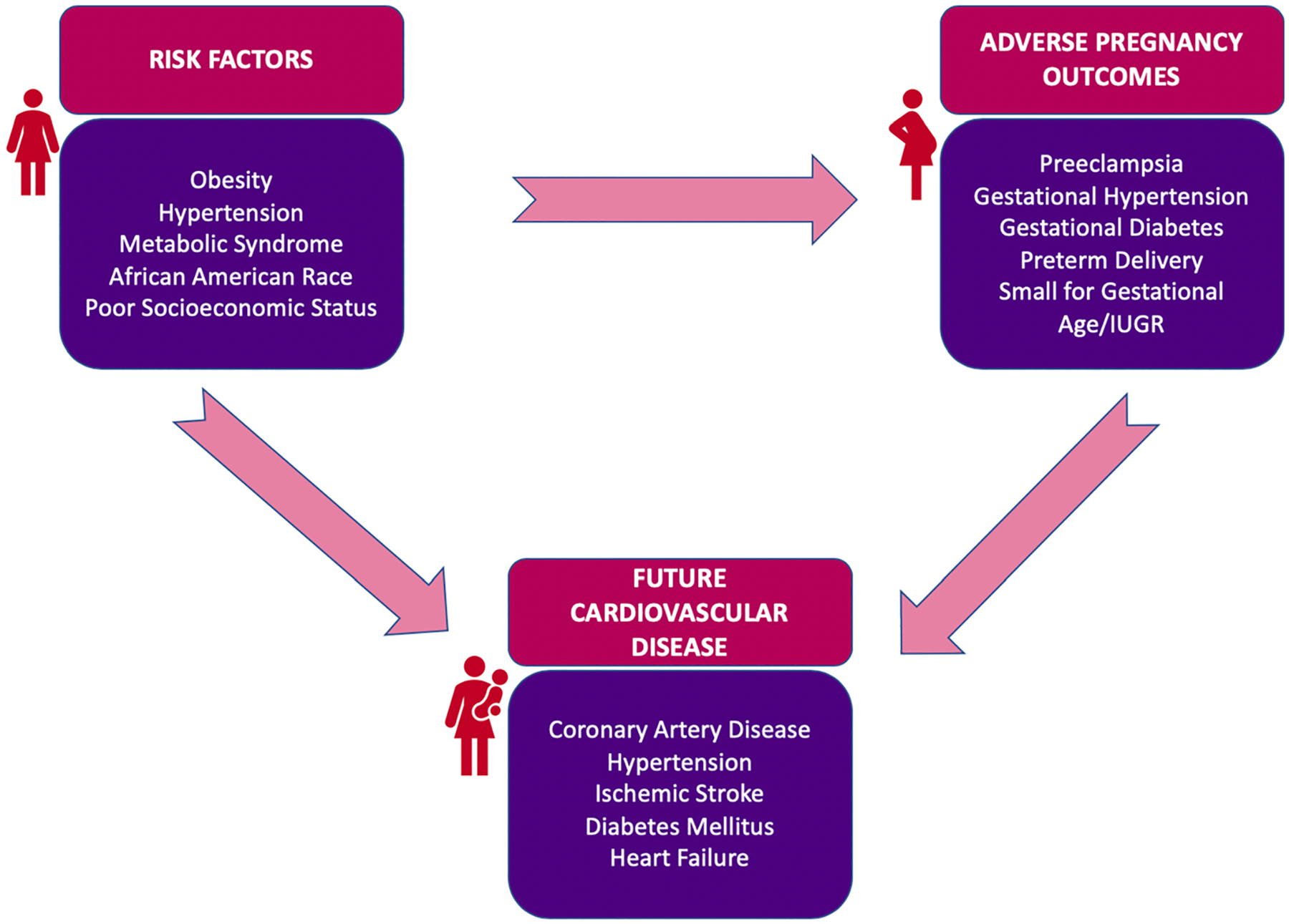
Shared risk factors between adverse pregnancy outcomes and cardiovascular disease.
Preeclampsia
Hypertensive disorders of pregnancy (HDP) are a common cause of morbidity during pregnancy and include chronic hypertension, gestational hypertension, preeclampsia, preeclampsia superimposed on chronic hypertension, HELLP syndrome, and eclampsia. Among the common variations of HDP, preeclampsia carries the greatest morbidity and mortality risk, affecting 5–10% of all pregnant women [7–9]. It is defined by new onset hypertension (systolic blood pressure > 140 mmHg or diastolic blood pressure > 90 mmHg) and end organ dysfunction, diagnosed after 20 weeks of gestation [10]. The pathogenesis underlying preeclampsia remains elusive, but abnormal placentation, an imbalance in angiogenic factors, and heightened inflammation are thought to be key mediators. In most women, the overt clinical manifestations of preeclampsia resolve in the immediate postpartum period. However, numerous studies show an increased long-term risk for CVD in women with a history of preeclampsia [11–14].
The observed association between CVD risk and preeclampsia may in part be due to shared risk factors between preeclampsia and CVD. Diabetes mellitus, hypertension, obesity, and smoking all increase risk for preeclampsia and are known risk factors for CVD [15–19]. Cohort studies have also demonstrated higher triglyceride levels, higher levels of atherogenic particles like ApoE, and higher ApoB/ApoA1 ratios, conferring increased risk, among women with preeclampsia [18, 20]. In the years after preeclampsia resolution, women are at higher risk for CVD. For instance, within the first year postpartum, women with HDP have a very high risk of developing chronic hypertension, with hazard ratios between 11.6 (95% CI 10.4–12.8) and 24.5 (95% CI 21.8–27.6) reported [21], and while this decreases over time, the risk remains twice that for women with normotensive pregnancies more than two decades later [21, 22].
Similarly, there is about a two-fold higher risk of ischemic heart disease (IHD) and stroke, with HR 1.8 (95% CI 1.3–2.6) reported for IHD and 2.98 (1.11–7.96) for non-fatal stroke after 10–15 years of follow-up [11, 23•, 24]. Subclinical markers of coronary disease, such as coronary artery calcium score (CACS), are also higher in women with prior preeclampsia [25–27]. Aside from IHD, women with a history of early-onset preeclampsia also have greater later life CVD risk factors, including diabetes, insulin, triglycerides, total cholesterol, and metabolic syndrome [26, 28–31].
In the acute setting, preeclampsia is associated with peripartum cardiomyopathy, possibly due to increased anti-angiogenic factors [32, 33]. Specifically, levels of angiogenic markers, soluble fms-like tyrosine kinase-1 (sFLT1), and soluble endoglin (sENG) correlate with myocardial dysfunction during preeclampsia [34]. Aside from the acute setting, several longitudinal studies also demonstrated an association between incident heart failure and history of HDP, with some study reporting HR 1.7 (95% CI 1.04–2.06) [23•].
A study in women with preeclampsia 1–2 years postpartum showed left ventricular hypertrophy and dysfunction, while another study in older women with prior HDP showed abnormal diastolic parameters and larger left atrial size [35, 36]. Interestingly, a recent, large observational study also reported an increased risk for valvular disease, including aortic stenosis (HR 2.9; 95% CI 1.5–5.4) and mitral regurgitation (HR 5.0; 95% 1.5–17.1) in women with HDP [23•].
Of note, women with recurrent preeclampsia are at much higher risk for future subclinical and clinical CVD when compared to women with only one pregnancy affected by preeclampsia. These patients have increased carotid-intima thickness, abnormal diastolic echocardiographic parameters, and elevated left ventricular mass [37–41]. Women with recurrent preeclampsia have an increased risk of hypertension (RR 2.3; 95% CI 1.9–2.9) and IHD (RR 2.4; 95% CI 2.2–2.7) [42, 43]. Higher incident heart failure among women with recurrent preeclampsia compared to controls (9.83 vs. 1.67 per 10,000 person-years) has also been reported [42].
Gestational diabetes mellitus
GDM is diabetes mellitus that manifests during the second or third trimester of pregnancy and is distinct from type 1 or type 2 diabetes mellitus that occurs prior to pregnancy [44]. In the USA, GDM has become increasingly prevalent, paralleling the increased prevalence in obesity [45•, 46], and now affects approximately 6–9% of pregnant women in the USA [47].
Women with GDM are at increased risk of developing CVD risk factors after pregnancy compared to women without GDM. Specifically, those with GDM are more than 7 times more likely to develop type 2 diabetes mellitus (RR 7.43 [95% CI 4.79, 11.51]) [48] and have nearly a twofold higher risk of developing hypertension and hyperlipidemia (hypertension OR 1.88 [95% CI 1.34, 2.64]; dyslipidemia OR 1.76 [95% CI 1.28, 2.44]) [49–51]. The risk of type 2 diabetes mellitus and hypertension is even higher in those with maternal obesity and GDM vs. no obesity and no GDM adjusted HR of 2.4 [95% CI 1.6, 3.5] compared to women with either GDM or gestational hypertension alone (adjusted HR 1.4; [95% CI 1.2, 1.7]) [49, 52].
Additionally, GDM is a risk factor for subsequent CVD, as it is associated with an increased risk of major cardiovascular events (RR 1.98 [95% CI 1.57, 2.50]) independent of the development of type 2 diabetes mellitus [53•]. Not surprisingly, the risk of CVD among women with GDM is amplified in the presence of other APOs. For example, women with both GDM and gestational hypertension have higher risks of CVD (adjusted HR 2.4 [95% CI 1.6, 3.5]) compared to women with either GDM or gestational hypertension alone (adjusted HR 1.4 [95% CI 1.2, 1.7]).
Epigenetics, subclinical inflammation, and endothelial/vascular dysfunction have all been proposed as mechanisms to explain the association between GDM and early-onset CVD [54, 55]. Inflammation has been associated with early atherosclerosis among women with GDM, as elevated levels of inflammatory markers, including C-reactive protein and interlukin-6, and lower levels of anti-inflammatory biomarkers, including adiponectin, have been demonstrated in women with GDM compared to normoglycemic women [56]. Endothelial dysfunction may also play a role, and women with GDM have been found to have increased carotid artery intimal thickness [57].
Preterm delivery
Preterm delivery, defined as birth between 20 and 37 weeks of gestation causes significant neonatal mortality worldwide and is a common reason for antenatal hospitalization [58]. In addition, preterm birth is associated with future maternal CVD risk, as shown in a recent large meta-analysis of more than 5.8 million women and 338,000 preterm births which highlights increased risk of future composite CVD (RR 1.43; 95% CI 1.18–1.72), cardiovascular mortality (RR 1.81; 95% CI 1.55–2.10), CAD (RR 1.49, 95% CI, 1.38, 1.60), and stroke (RR 1.65, 95% CI, 1.51, 1.79) [59•].
The most important potential confounder is the presence of hypertensive disorders of pregnancy. Two studies have compared spontaneous preterm birth with medically indicated preterm birth. A Norwegian study found that all gestational lengths shorter than 39 weeks were associated with future elevated CVD mortality [60]. Interestingly, spontaneous preterm delivery was associated with increased risk for CVD with HR up to 2.4 (95% CI 1.7–3.4), and medically indicated preterm delivery was associated with an increased CVD risk with HR up to 6.2 (95% CI 4.2–9.3) [60]. While the elevated future CVD mortality in the medically indicated group can be partially explained by the high prevalence of hypertensive disorders of pregnancy in this group, it is notable that the future CVD risk remains elevated in the spontaneous preterm labor group as well. Similarly, a study of Scottish women reported an association between preterm delivery and future risk of IHD. Women who have had medically indicated preterm delivery are at higher risk for future IHD events (HR 1.81; 95% CI 1.61–2.04) and IHD death (HR 2.49; 95% CI 1.89–3.30). However, women with a history of spontaneous preterm delivery were also at increased risk for IHD events (HR 1.46; 95% CI 1.33–1.61) and IHD death (2.14; 95% CI 1.70–2.70) [61]. These studies suggest that PTD remains a risk factor for future CVD even when accounting for the prevalence of hypertensive disorders of pregnancy.
Social disparities and APOs
Importantly, APOs disproportionately affect racial and ethnic minorities, particularly in the USA. These disparities are present not only in the prevalence of specific APOs but also in the cardiovascular sequelae of APOs and maternal mortality [62–64]. For example, African American and Native American women experience the highest rates of preterm birth, while Asian, Hispanic, and Native American women have the highest rates of GDM [65–67]. For HDP, there are conflicting data regarding racial disparities in disease prevalence [68]; however, African American women are more likely to suffer from severe maternal morbidity related to preeclampsia, including stroke, peripartum cardiomyopathy and cardiac arrest [69].
In addition, low socioeconomic status (SES) is also a significant risk factor for APOs, and this association is independent of race or ethnicity [64, 70, 71, 72•]. However, it is a weaker predictor of APOs than racial/ethnic group identification, and improving SES for racial/ethnic minorities does not eliminate disparities in APO rates between racial/ethnic minorities and white women [71, 72•]. Etiologies for racial disparities in APOs are multifactorial, with prior studies implicating differences in inflammatory markers, genetics and epigenetics, endothelial function, and psychosocial factors—including racial discrimination at individual and systems levels and other underlying risk factors for APOs [62, 66, 73]. Additional research is needed to further clarify etiologies for these disparities to pinpoint potential targets for solutions (Table 1).
Table 1.
Future directions and key research questions
| Areas of investigation | Future directions and key research questions |
|---|---|
| Preeclampsia |
|
| Gestational diabetes |
|
| Preterm delivery |
|
| Pathophysiology |
|
| Risk prediction and interventions |
|
APO adverse pregnancy outcome, CVD cardiovascular disease
Role of aspirin in the prevention of APOs
Mechanisms and biologic plausibility of aspirin
Because of the many adverse effects, there is great interest in reducing the incidence and severity of APOs. In addition to addressing social determinants of health, aspirin therapy has been increasingly utilized during pregnancy, particularly in preventing morbidity from preeclampsia [74]. Aspirin is a non-steroidal anti-inflammatory drug that inhibits cyclooxygenase isoenzymes 1 (COX-1) and 2 (COX-2) [75]. The latter converts arachidonic acid to prostaglandins, which have vasodilatory effects and inhibit platelet aggregation. In platelets, however, prostaglandin H2 is converted into thromboxane A2, which has the opposite effect of vasoconstriction and platelet activation [74, 75].
Aspirin preferentially inhibits COX enzymes in platelets rather than the endothelium, leading to inhibited thromboxane production without affecting prostacyclin production. One mechanism of preeclampsia is an imbalance in thromboxane A2 and prostacyclin production, resulting in uterine spiral arterial remodeling, vasoconstriction, and uteroplacental insufficiency [75, 76]. Ratios of thromboxane A2 to prostacyclin have been shown to be decreased after treatment with low-dose aspirin, which is proposed to be a mechanism by which aspirin may be beneficial in preeclampsia [76]. In addition to pathways of vasoconstriction, aspirin also decreases profibrotic factor activin A levels, which is associated with cardiac fibrosis and hypertrophy, as well as improve global longitudinal strain, suggesting possible mechanisms of prevention of cardiac dysfunction in women with preeclampsia [77].
Preeclampsia
Large randomized controlled trials of aspirin therapy report mixed results in women at elevated risk for preeclampsia or pregnancy-induced hypertension [83–86]. Two older trials failed to demonstrate differences in preeclampsia, perinatal death, preterm delivery, or intrauterine growth restriction (IUGR) with aspirin use [85, 86]. In contrast, the Aspirin for Evidence-Based Preeclampsia Prevention trial demonstrated that among 1776 women at high risk for preterm preeclampsia, aspirin reduced the incidence of preterm preeclampsia (OR 0.38 [95% CI 0.20, 0.74]) [84] The US Preventive Service Task Force (USPSTF) performed a systematic review investigating aspirin therapy in pregnancy in 2014 and reported a 24% RR reduction in preeclampsia (95% CI 0.62, 0.95), 14% reduction in preterm delivery (95% CI 0.76, 0.98), and a 20% reduction in IUGR (95% CI 0.65, 0.99) using pooled estimates [87•]. Aspirin was also noted to be relatively safe, with no change in placental abruption, perinatal mortality, or postpartum hemorrhage rates. Therefore, the USPSTF concluded that there is substantial net benefit of low-dose aspirin use and recommended starting low-dose aspirin therapy (81 mg/day) after 12 weeks of gestation for women at higher risk for preeclampsia [78], which is also endorsed by ACOG and the Society for Maternal and Fetal Medicine task forces [74] (Table 2).
Table 2.
Summary of indications for use of aspirin in pregnancy, shared pathophysiologic mechanisms, and emerging research
| Possible mechanisms | Society recommendations | Selected emerging research topics | |
|---|---|---|---|
| Preeclampsia | Inhibits thromboxane production and vasoconstriction → reduces uteroplacental ischemia | ACOG: Low-dose aspirin for women at high risk for PE from 12 to 28 weeks until delivery [74] USPSTF: Low-dose aspirin after 12 weeks for women at high risk for PE [78] |
Studies of biomarkers to understand the pathophysiologic benefits of platelet inhibition in PE and prevention of cardiac dysfunction [43, 77] |
| Preterm delivery | Not recommended in the absence of PE risk factors [74] | Newer trials and meta-analyses suggesting potential benefit [79, 80] | |
| IUGR | Not recommended in the absence of PE risk factors [75] | Clinical models to predict preterm delivery to identify women who might benefit from aspirin [81, 82] |
ACOG American College of Obstetricians and Gynecologists, PE preeclampsia, USPSTF United States Preventive Services Task Force, IUGR intrauterine growth restriction
Preterm delivery
In addition to preeclampsia, aspirin has been studied in the context of other APOs. Preterm delivery has been an outcome in many preeclampsia trials, with a few studies have reported the effect of aspirin on preterm delivery in women without risk factors for preeclampsia. In particular, the ASPIRIN trial, a recent large multi-national RCT, randomized 11,976 nulliparous women to low-dose aspirin between 6 weeks 0 days and 13 weeks 6 days of gestation and found an 11% relative reduction (95% CI 0.81, 0.98) in the risk of overall preterm delivery and a 25% relative reduction (95% CI 0.61, 0.93) in the risk of early preterm delivery < 34 weeks in women [79]. This study did not differentiate between spontaneous and iatrogenic preterm delivery. However, there were no differences in the rate of HDP or IUGR, the two main indications for iatrogenic preterm delivery, suggesting that the lower incidence of preterm delivery in the aspirin group may be due to lower rates of spontaneous preterm delivery. Additionally, there is evidence from other meta-analyses and preeclampsia trials that suggest a lower risk of spontaneous preterm delivery in women treated with aspirin [80, 88].
Intrauterine growth restriction
Finally, low-dose aspirin may also be effective at preventing IUGR, since both IUGR and preeclampsia share common pathophysiology underlying abnormal placentation [74]. However, clinical studies have reported conflicting results in women with small for gestational age fetuses [89, 90]. In contrast, patients with subsequent preeclampsia have consistently found a reduction in IUGR with aspirin therapy, especially when initiated prior to 17 weeks’ gestation [87, 91]. Ultimately, additional studies are needed to examine the mechanisms and effectiveness of aspirin in IUGR and to discover risk stratification tools, including biomarkers, that may identify women who are at higher risk of IUGR who may benefit from aspirin use (Table 1).
Interventions to reduce subsequent long-term CVD risk in women who have had APOs
Few clinical trials have been performed to investigate specific interventions to reduce long-term CVD risk in women with APOs. In one randomized controlled trial, postpartum women with a history of preeclampsia within the prior 5 years underwent an online intervention targeting modifiable risk factors, specifically diet and physical activity [92]. The intervention improved physical activity but did not decrease weight or blood pressure [92]. Since then, other clinical trials have started, including the RedCarRisk trial, which seeks to assess the efficacy of an intensive regular conditional workout on arterial stiffness in women after preeclampsia. (Table 1).
Because of the strong association between APOs with future CVD [93], the importance of early identification for women at risk of future CVD has been acknowledged by professional societies, including the AHA, ACOG, and the Society of Obstetricians and Gynecologists of Canada [93, 94•, 95]. Most recently, a joint presidential advisory from the AHA and ACOG urged that the postpartum time frame be considered a critical time for health promotion, cardiovascular health assessment, risk stratification, and focused counseling highlighting the need for screening and long-term follow-up [94•, 96]. More specifically, the ACOG Task Force on Hypertension in Pregnancy recommends periodic assessment for CVD risk factors (e.g., hypertension, hyperlipidemia, obesity, and diabetes) in women with a history of preeclampsia, especially for women with preterm or recurrent preeclampsia [10, 97•]. In addition, the recent 2019 American College of Cardiology (ACC)/American Heart Association (AHA) guideline on the Primary Prevention of Cardiovascular Disease has recommendations for women with HDP. The guidelines consider APOs to be CVD risk enhancers, which warrant further assessment of risk by obtaining a CACS or by considering intensified preventive interventions like statin therapy [98]. However, there are no specific recommendations on the frequency of screening. There are also no recommendations favoring chronic aspirin therapy.
Current knowledge gaps and future directions
Improvement in risk-prediction models
Current prevention guidelines for CVD have stressed the importance of pregnancy history in evaluating CVD risk, but few studies have supported the incorporation of pregnancy risk factors into risk prediction scores [99]. In particular, three studies have examined the incremental value of adding HDP, preterm delivery, IUGR, and low birth weight to well-established risk factors and have found no risk reclassification [35, 99–101]. In one study utilizing a Norwegian population-based registry, HDP and preterm delivery status did led to reclassification of CVD risk, but it was of minimal clinical significance [98]. In another study assessing a US cohort of women at low risk for CVD, incorporating HDP did not improve predictive discrimination despite being independently associated with increased 10-year CVD risk [99].
These studies used population-based registries and cohorts to create sample populations of parous women ≥ 40 years of age with no prior history of CVD, and thus a large proportion of women in these groups were beyond their reproductive years. Because the proportion of comorbidities, such as diabetes, obesity, dyslipidemia, and hypertension, increases with age, sampling of older women might have underestimated the risk attributed to APOs. In addition, APOs are not independent of each other and may occur simultaneously making it difficult to assess their individual risk. Hence, any future risk prediction modeling exercises should include risk factors that are independently highly predictive of CVD and should include populations that reflect the target population intended for screening (i.e., women of reproductive age) [97] (Table 1).
Cardiovascular dysfunction and atherosclerosis after APOs
For women who have had APOs, some studies reveal structural and functional cardiovascular changes as well as persistent anti-angiogenic, coagulopathic, and inflammatory states [35, 101].
Notably, one study in women with preeclampsia 5 years earlier are reported to have lower coronary flow reserve, higher carotid intima-media thickness, and higher high-sensitivity C-reactive protein values compared to normal controls [102]. Further, the Cardiovascular Risk Profile: Imaging and Gender-Specific Disorders (CREW-IMAGO) study has noted that 31% of women with previous preeclampsia aged 45–55 years had a CACS greater than zero, with 47% having coronary atherosclerotic plaques and 4.3% with significant coronary artery stenosis [26].
Of note, most of the studies assessing subclinical atherosclerosis have been performed in women of approximately 60 years, thus, by and large, 30–40 years after pregnancy. In addition, most of these findings are derived from single center studies. Thus, there should be future multicenter, phenomic and metabolomic studies that explore signals of early atherosclerosis, endothelial dysfunction, and early diastolic dysfunction in younger patients. These studies should also focus on the mechanistic link between APOs and cardiovascular dysfunction. In addition, the biological properties of aspirin, including immunomodulatory, anti-inflammatory, and antioxidant effects should be explored in future studies to further understand the mechanisms underlying potential therapeutic benefits in women who have had APOs (Table 1).
Behavioral interventions in women with APOs
Whether modifiable behavioral factors (e.g., exercise, weight loss) moderate CVD risk in women following an APO is an important question with clinical and public health implications. Women who have had a HDP and with higher BMIs have a higher risk of incident hypertension [103]. However, there are no large multicenter studies that have explored the effect of behavioral modifications on subclinical cardiovascular dysfunction. A potential study can assess the role of AHA’s Life’s Simple 7 recommendations on optimal control of CV risk factors, which target smoking, nutrition, exercise, weight loss, blood pressure, cholesterol, and diabetes, in the postpartum period.
Conclusions
Adverse pregnancy outcomes, including preeclampsia and HDP, GDM, and preterm delivery, are highly associated with future maternal CVD; however, it is unclear whether this is a causal relationship or whether APOs unmask shared risk factors with CVD. Unlocking the pathophysiologic link between an APO and the development of subclinical and overt CVD will help identify novel therapeutic targets and lifestyle recommendations for women with APOs. The role of aspirin in prevention of APOs and subsequent cardiovascular dysfunction will be important to explore as a possible therapeutic avenue. Timely recognition and aggressive risk factor modification and health promotion are vital, particularly among women with lower socioeconomic status. Future research should explore the efficacy of modifiable behavioral interventions in reducing CVD risk in women with APOs.
Footnotes
Human and animal rights and informed consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Conflict of interest
Anum S. Minhas, Wendy Ying, S. Michelle Ogunwole, Michael Miller, Sammy Zakaria, Arthur J. Vaught, Allison G. Hays, Andreea A. Creanga, Ari Cedars, Erin D. Michos, Roger S. Blumenthal, and Garima Sharma declare that they have no conflict of interest.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as:
• Of importance
- 1.Martin JA, Hamilton BE, Osterman MJK, Driscoll AK. Births: final data for 2018. Natl Vital Stat Rep. 2019;68:1–47. [PubMed] [Google Scholar]
- 2.Centers for Disease Control and Prevention. Preterm birth [Internet]. Available from: http://www.cdc.gov/reproductivehealth/maternalinfanthealth/pretermbirth.htm. Accessed 7/11/20.
- 3.•.Lane-Cordova AD, Khan SS, Grobman WA, Greenland P, Shah SJ. Long-term cardiovascular risks associated with adverse pregnancy outcomes. J Am Coll Cardiol. 2019;73:2106–16. [DOI] [PubMed] [Google Scholar]; This review highlights the epidemiology and possible mechanisms of adverse pregnancy outcomes and future CVD, incorporating recent data.
- 4.Kuklina E, Callaghan W. Chronic heart disease and severe obstetric morbidity among hospitalisations for pregnancy in the USA: 1995–2006: heart disease and severe morbidity in USA. BJOG Int J Obstet Gynaecol. 2011;118:345–52. [DOI] [PubMed] [Google Scholar]
- 5.Correa A, Bardenheier B, Elixhauser A, Geiss LS, Gregg E. Trends in prevalence of diabetes among delivery hospitalizations, United States, 1993–2009. Matern Child Health J. 2015;19:635–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sharma G, Lindley K, Grodzinsky A. Cardio-obstetrics. J Am Coll Cardiol. 2020;75:1355–9. [DOI] [PubMed] [Google Scholar]
- 7.Grill S, Rusterholz C, Zanetti-Dällenbach R, Tercanli S, Holzgreve W, Hahn S, et al. Potential markers of preeclampsia – a review. Reprod Biol Endocrinol. 2009;7:70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hogan MC, Foreman KJ, Naghavi M, Ahn SY, Wang M, Makela SM, et al. Maternal mortality for 181 countries, 1980–2008: a systematic analysis of progress towards Millennium Development Goal 5. Lancet. 2010;375:1609–23. [DOI] [PubMed] [Google Scholar]
- 9.Wanderer JP, Leffert LR, Mhyre JM, Kuklina EV, Callaghan WM, Bateman BT. Epidemiology of obstetric-related ICU admissions in Maryland: 1999–2008*. Crit Care Med. 2013;41:1844–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hypertension in Pregnancy: Executive Summary. Obstetrics & Gynecology. 2013;122(5):1122–31. [DOI] [PubMed] [Google Scholar]
- 11.Bellamy L, Casas J-P, Hingorani AD, Williams DJ. Preeclampsia and risk of cardiovascular disease and cancer in later life: systematic review and meta-analysis. BMJ. 2007;335:974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Silverberg O, Park AL, Cohen E, Fell DB, Ray JG. Pre-mature cardiac disease and death in women whose infant was preterm and small for gestational age: a retrospective cohort study. JAMA Cardiol. 2018;3:247–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wilson BJ, Watson MS, Prescott GJ, Sunderland S, Campbell DM, Hannaford P, et al. Hypertensive diseases of pregnancy and risk of hypertension and stroke in later life: results from cohort study. BMJ. 2003;326:845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ray JG, Schull MJ, Kingdom JC, Vermeulen MJ. Heart failure and dysrhythmias after maternal placental syndromes: HAD MPS study. Heart. 2012;98:1136–41. [DOI] [PubMed] [Google Scholar]
- 15.Thilaganathan B, Kalafat E. Cardiovascular system in preeclampsia and beyond. Hypertension. 2019;73:522–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kalafat E, Sukur YE, Abdi A, Thilaganathan B, Khalil A. Metformin for prevention of hypertensive disorders of pregnancy in women with gestational diabetes or obesity: systematic review and meta-analysis of randomized trials. Ultrasound Obstet Gynecol. 2018;52:706–14. [DOI] [PubMed] [Google Scholar]
- 17.Giannakou K, Evangelou E, Papatheodorou SI. Genetic and non-genetic risk factors for pre-eclampsia: umbrella review of systematic reviews and meta-analyses of observational studies: genetic and non-genetic risk factors for PE. Ultrasound Obstet Gynecol. 2018;51:720–30. [DOI] [PubMed] [Google Scholar]
- 18.Egeland GM, Klungsøyr K, Øyen N, Tell GS, Næss Ø, Skjærven R. Preconception cardiovascular risk factor differences between gestational hypertension and preeclampsia: cohort Norway study. Hypertension. 2016;67:1173–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Perry H, Khalil A, Thilaganathan B. Preeclampsia and the cardiovascular system: an update. Trends Cardiovasc Med. 2018;28:505–13. [DOI] [PubMed] [Google Scholar]
- 20.Serrano NC, Guio-Mahecha E, Quintero-Lesmes DC, Becerra-Bayona S, Paez MC, Beltran M, et al. Lipid profile, plasma apolipoproteins, and pre-eclampsia risk in the GenPE case-control study. Atherosclerosis. 2018;276:189–94. [DOI] [PubMed] [Google Scholar]
- 21.Behrens I, Basit S, Melbye M, Lykke JA, Wohlfahrt J, Bundgaard H, et al. Risk of post-pregnancy hypertension in women with a history of hypertensive disorders of pregnancy: nationwide cohort study. BMJ. 2017;358:j3078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Heida KY, Franx A, van Rijn BB, Eijkemans MJC, Boer JMA, Verschuren MWM, et al. Earlier age of onset of chronic hypertension and type 2 diabetes mellitus after a hypertensive disorder of pregnancy or gestational diabetes mellitus. Hypertension. 2015;66:1116–22. [DOI] [PubMed] [Google Scholar]
- 23.•.Honigberg MC, Zekavat SM, Aragam K, Klarin D, Bhatt DL, Scott NS, et al. Long-term cardiovascular risk in women with hypertension during pregnancy. J Am Coll Cardiol. 2019;74:2743–54. [DOI] [PMC free article] [PubMed] [Google Scholar]; This study showed that hypertensive disorders of pregnancy are associated with accelerated cardiovascular aging and may be useful to refine CVD risk assessments.
- 24.Leslie MS, Briggs LA. Preeclampsia and the risk of future vascular disease and mortality: a review. J Midwif Women’s Health. 2016;61:315–24. [DOI] [PubMed] [Google Scholar]
- 25.White WM, Mielke MM, Araoz PA, Lahr BD, Bailey KR, Jayachandran M, et al. A history of preeclampsia is associated with a risk for coronary artery calcification 3 decades later. Am J Obstet Gynecol. 2016;214:519.e1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zoet GA, Benschop L, Boersma E, Budde RPJ, Fauser BCJM, van der Graaf Y, et al. Prevalence of subclinical coronary artery disease assessed by coronary computed tomography angiography in 45- to 55-year-old women with a history of preeclampsia. Circulation. 2018;137:877–9. [DOI] [PubMed] [Google Scholar]
- 27.Beckman JP, Camp JJ, Lahr BD, Bailey KR, Kearns AE, Garovic VD, et al. Pregnancy history, coronary artery calcification and bone mineral density in menopausal women. Climacteric. 2018;21:53–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lykke JA, Langhoff-Roos J, Sibai BM, Funai EF, Triche EW, Paidas MJ. Hypertensive pregnancy disorders and subsequent cardiovascular morbidity and type 2 diabetes mellitus in the mother. Hypertension. 2009;53:944–51. [DOI] [PubMed] [Google Scholar]
- 29.Feig DS, Shah BR, Lipscombe LL, Wu CF, Ray JG, Lowe J, et al. Preeclampsia as a risk factor for diabetes: a population-based cohort study. Middleton P, editor. PLoS Med. 2013;10:e1001425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Veerbeek JHW, Hermes W, Breimer AY, van Rijn BB, Koenen SV, Mol BW, et al. Cardiovascular disease risk factors after early-onset preeclampsia, late-onset pre-eclampsia, and pregnancy-induced hypertension. Hypertension. 2015;65:600–6. [DOI] [PubMed] [Google Scholar]
- 31.Al-Nasiry S, Ghossein-Doha C, Polman S, Lemmens S, Scholten R, Heidema W, et al. Metabolic syndrome after pregnancies complicated by pre-eclampsia or small-for-gestational-age: a retrospective cohort. BJOG: Int J Obstet Gy. 2015;122:1818–23. [DOI] [PubMed] [Google Scholar]
- 32.Bello N, Rendon ISH, Arany Z. The relationship between pre-eclampsia and peripartum cardiomyopathy. J Am Coll Cardiol. 2013;62:1715–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Patten IS, Rana S, Shahul S, Rowe GC, Jang C, Liu L, et al. Cardiac angiogenic imbalance leads to peripartum cardiomyopathy. Nature. 2012;485:333–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen CW, Jaffe IZ, Karumanchi SA. Pre-eclampsia and cardiovascular disease. Cardiovasc Res. 2014;101:579–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Melchiorre K, Sutherland GR, Liberati M, Thilaganathan B. Preeclampsia is associated with persistent postpartum cardiovascular impairment. Hypertension. 2011;58:709–15. [DOI] [PubMed] [Google Scholar]
- 36.Scantlebury DC, Kane GC, Wiste HJ, Bailey KR, Turner ST, Arnett DK, et al. Left ventricular hypertrophy after hypertensive pregnancy disorders. Heart. 2015;101:1584–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tangren JS, Powe CE, Ankers E, Ecker J, Bramham K, Hladunewich MA, et al. Pregnancy outcomes after clinical recovery from AKI. JASN. 2017;28:1566–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Stergiotou I, Bijnens B, Cruz-Lemini M, Figueras F, Gratacos E, Crispi F. Maternal subclinical vascular changes in fetal growth restriction with and without pre-eclampsia: maternal vascular remodeling in FGR and PE. Ultrasound Obstet Gynecol. 2015;46:706–12. [DOI] [PubMed] [Google Scholar]
- 39.Sep S, Schreurs M, Bekkers S, Kruse A, Smits L, Peeters L. Early-pregnancy changes in cardiac diastolic function in women with recurrent pre-eclampsia and in previously pre-eclamptic women without recurrent disease: diastolic function early in recurrent pre-eclampsia. BJOG Int J Obstet Gynaecol. 2011;118:1112–9. [DOI] [PubMed] [Google Scholar]
- 40.Ghossein-Doha C, Spaanderman MEA, Al Doulah R, Van Kuijk SM, Peeters LLH. Maternal cardiac adaptation to subsequent pregnancy in formerly preeclamptic women according to recurrence of preeclampsia. Ultrasound Obstet Gynecol. 2016;47:96–103. [DOI] [PubMed] [Google Scholar]
- 41.Milic NM, Milin-Lazovic J, Weissgerber TL, Trajkovic G, White WM, Garovic VD. Preclinical atherosclerosis at the time of pre-eclamptic pregnancy and up to 10 years postpartum: systematic review and meta-analysis: preeclampsia and preclinical atherosclerosis. Ultrasound Obstet Gynecol. 2017;49:110–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Brouwers L, van der Meiden-van Roest A, Savelkoul C, Vogelvang T, Lely A, Franx A, et al. Recurrence of preeclampsia and the risk of future hypertension and cardiovascular disease: a systematic review and meta-analysis. BJOG: Int J Obstet Gy. 2018;125:1642–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Theilen LH, Meeks H, Fraser A, Esplin MS, Smith KR, Varner MW. Long-term mortality risk and life expectancy following recurrent hypertensive disease of pregnancy. Am J Obstet Gynecol. 2018;219:107.e1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.American Diabetes Association. Classification and diagnosis of diabetes: standards of medical care in diabetes—2019. Diabetes Care. 2019;42:S13–28. [DOI] [PubMed] [Google Scholar]
- 45.•.Committee on Practice Bulletins—Obstetrics. ACOG Practice Bulletin No. 190: Gestational Diabetes Mellitus. Obstet Gynecol. 2018;131:e49–64. [DOI] [PubMed] [Google Scholar]; ACOG’s practice bulletin on gestational diabetes mellitus provides updated guidance on management of GDM.
- 46.Garrison A. Screening, diagnosis, and management of gestational diabetes mellitus. Am Fam Physician. 2015;91:460–7. [PubMed] [Google Scholar]
- 47.Diabetes During Pregnancy | Maternal Infant Health | Reproductive Health | CDC [Internet]. [cited 2020 Apr 4]. Available from: https://www.cdc.gov/reproductivehealth/maternalinfanthealth/diabetes-during-pregnancy.htm.
- 48.Bellamy L, Casas J-P, Hingorani AD, Williams D. Type 2 diabetes mellitus after gestational diabetes: a systematic review and meta-analysis. Lancet. 2009;373:1773–9. [DOI] [PubMed] [Google Scholar]
- 49.Kaul P, Savu A, Nerenberg KA, Donovan LE, Chik CL, Ryan EA, et al. Impact of gestational diabetes mellitus and high maternal weight on the development of diabetes, hypertension and cardiovascular disease: a population-level analysis. Diabet Med. 2015;32:164–73. [DOI] [PubMed] [Google Scholar]
- 50.Carr DB, Utzschneider KM, Hull RL, Tong J, Wallace TM, Kodama K, et al. Gestational diabetes mellitus increases the risk of cardiovascular disease in women with a family history of type 2 diabetes. 2006 [DOI] [PubMed] [Google Scholar]
- 51.Retnakaran R, Qi Y, Connelly PW, Sermer M, Hanley AJ, Zinman B. The graded relationship between glucose tolerance status in pregnancy and postpartum levels of low-density-lipoprotein cholesterol and apolipoprotein B in young women: implications for future cardiovascular risk. J Clin Endocrinol Metab. 2010;95:4345–53. [DOI] [PubMed] [Google Scholar]
- 52.Pirkola J, Pouta A, Bloigu A, Miettola S, Hartikainen AL, Järvelin MR, et al. Prepregnancy overweight and gestational diabetes as determinants of subsequent diabetes and hypertension after 20-year follow-up. J Clin Endocrinol Metab. 2010;95:772–8. [DOI] [PubMed] [Google Scholar]
- 53.•.Kramer CK, Campbell S, Retnakaran R. Gestational diabetes and the risk of cardiovascular disease in women: a systematic review and meta-analysis. Diabetologia. 2019;62:905–14. [DOI] [PubMed] [Google Scholar]; This meta-analysis evaluated the impact of gestational diabetes on future CVD risk and demonstrated that the increased risk was independent of the subsequent development of type 2 diabetes.
- 54.Harreiter J, Dovjak G, Kautzky-Willer A. Gestational diabetes mellitus and cardiovascular risk after pregnancy. Women Health. 2014;10:91–108. [DOI] [PubMed] [Google Scholar]
- 55.Vrachnis N, Augoulea A, Iliodromiti Z, Lambrinoudaki I, Sifakis S, Creatsas G. Previous gestational diabetes mellitus and markers of cardiovascular risk. Int J Endocrinol. 2012;2012:1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Heitritter SM, Solomon CG, Mitchell GF, Skali-Ounis N, Seely EW. Subclinical inflammation and vascular dysfunction in women with previous gestational diabetes mellitus. J Clin Endocrinol Metab. 2005;90:3983–8. [DOI] [PubMed] [Google Scholar]
- 57.Bo S, Valpreda S, Menato G, Bardelli C, Botto C, Gambino R, et al. Should we consider gestational diabetes a vascular risk factor? Atherosclerosis. 2007;194:72–9. [DOI] [PubMed] [Google Scholar]
- 58.Practice Bulletin No. 171: Management of Preterm Labor. Obstetrics & Gynecology. 2016;128:e155–64. [DOI] [PubMed] [Google Scholar]
- 59.•.Wu P, Gulati M, Kwok CS, Wong CW, Narain A, O’Brien S, et al. Preterm delivery and future risk of maternal cardiovascular disease: a systematic review and meta-analysis. JAHA [Internet]. 2018. [cited 2020 Jul 9];7. Available from: 10.1161/JAHA.117.007809. [DOI] [PMC free article] [PubMed] [Google Scholar]; This study adds to prior literature on preterm delivery and maternal CVD by conducting a meta-analysis that incorporated more recent studies with larger sample sizes, showing that preterm delivery is associated wtih maternal CVD and mortality.
- 60.Rich-Edwards JW, Klungsoyr K, Wilcox AJ, Skjaerven R. Duration of pregnancy, even at term, predicts long-term risk of coronary heart disease and stroke mortality in women: a population-based study. Am J Obstet Gynecol. 2015;213:518.e1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hastie CE, Smith GC, MacKay DF, Pell JP. Maternal risk of ischaemic heart disease following elective and spontaneous pre-term delivery: retrospective cohort study of 750 350 singleton pregnancies. Int J Epidemiol. 2011;40:914–9. [DOI] [PubMed] [Google Scholar]
- 62.Bryant AS, Worjoloh A, Caughey AB, Washington AE. Racial/ethnic disparities in obstetric outcomes and care: prevalence and determinants. Am J Obstet Gynecol. 2010;202:335–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bentley-Lewis R, Powe C, Ankers E, Wenger J, Ecker J, Thadhani R. Effect of race/ethnicity on hypertension risk subsequent to gestational diabetes mellitus. Am J Cardiol. 2014;113:1364–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Silva LM, Coolman M, Steegers EA, Jaddoe VW, Moll HA, Hofman A, et al. Low socioeconomic status is a risk factor for preeclampsia: the Generation R Study. J Hypertens. 2008;26:1200–8. [DOI] [PubMed] [Google Scholar]
- 65.Caughey AB, Cheng YW, Stotland NE, Washington AE, Escobar GJ. Maternal and paternal race/ethnicity are both associated with gestational diabetes. YMOB. 2010;202:616.e1–5. [DOI] [PubMed] [Google Scholar]
- 66.Manuck TA. Racial and ethnic differences in preterm birth: a complex, multifactorial problem. Semin Perinatol. 2017;41:511–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Centers for Disease Control and Prevention (CDC). Racial/ethnic disparities in neonatal mortality–United States, 1989–2001. MMWR Morb Mortal Wkly Rep. 2004;53:655–8. [PubMed] [Google Scholar]
- 68.Tucker MJ, Berg CJ, Callaghan WM, Hsia J. The black-white disparity in pregnancy-related mortality from 5 conditions: differences in prevalence and case-fatality rates. American Journal of Public Health. Am Publ Health Assoc 2007;97:247–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Shahul S, Tung A, Minhaj M, Nizamuddin J, Wenger J, Mahmood E, et al. Racial disparities in comorbidities, complications, and maternal and fetal outcomes in women with preeclampsia/eclampsia. Hypertens Pregnancy. 2015;34:506–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Bo S, Menato G, Bardelli C, Lezo A, Signorile A, Repetti E, et al. Low socioeconomic status as a risk factor for gestational diabetes. Diabetes Metab. 2002;28:139–40. [PubMed] [Google Scholar]
- 71.Messer LC, Vinikoor LC, Laraia BA, Kaufman JS, Eyster J, Holzman C, et al. Socioeconomic domains and associations with preterm birth. Soc Sci Med. 2008;67:1247–57. [DOI] [PubMed] [Google Scholar]
- 72.•.Ross KM, Guardino C, Dunkel Schetter C, Hobel CJ. Interactions between race/ethnicity, poverty status, and pregnancy cardio-metabolic diseases in prediction of postpartum cardio-metabolic health. Ethn Health. 2018:1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]; This study assessed various socioeconomic and racial/ethnic factors in their association with pregnancy complications and cardiometabolic risk.
- 73.Nakimuli A, Chazara O, Byamugisha J, Elliott AM, Kaleebu P, Mirembe F, et al. Pregnancy, parturition and preeclampsia in women of African ancestry. Am J Obstet Gynecol. 2014;210:510–520.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.ACOG Committee Opinion No. 743: low-dose Aspirin use during pregnancy. Obstet Gynecol 2018;132:e44–52. [DOI] [PubMed] [Google Scholar]
- 75.Cadavid AP. Aspirin: the mechanism of action revisited in the context of pregnancy complications. Front Immunol. 2017;8:261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Schiff E, Peleg E, Goldenberg M, Rosenthal T, Ruppin E, Tamarkin M, et al. The use of Aspirin to prevent pregnancy-induced hypertension and lower the ratio of thromboxane A 2 to prostacyclin in relatively high risk pregnancies. N Engl J Med. 1989;321:351–6. [DOI] [PubMed] [Google Scholar]
- 77.Naseem H, Dreixler J, Mueller A, Tung A, Dhir R, Chibber R, et al. Antepartum aspirin administration reduces activin A and cardiac global longitudinal strain in preeclamptic women. JAHA. 2020;9:e015997. 10.1161/JAHA.119.015997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.LeFevre ML. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161:819. [DOI] [PubMed] [Google Scholar]
- 79.Hoffman MK, Goudar SS, Kodkany BS, Metgud M, Somannavar M, Okitawutshu J, et al. Low-dose aspirin for the prevention of preterm delivery in nulliparous women with a singleton pregnancy (ASPIRIN): a randomised, double-blind, placebo-controlled trial. Lancet. 2020;395:285–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.van Vliet EOG, Askie LA, Mol BWJ, Oudijk MA. Anti-platelet agents and the prevention of spontaneous preterm birth: a systematic review and meta-analysis. Obstet Gynecol. 2017;129:327–36. [DOI] [PubMed] [Google Scholar]
- 81.Tan MY, Poon LC, Rolnik DL, Syngelaki A, de Paco Matallana C, Akolekar R, et al. Prediction and prevention of small-for-gestational-age neonates: evidence from SPREE and ASPRE. Ultrasound Obstet Gynecol. 2018;52:52–9. [DOI] [PubMed] [Google Scholar]
- 82.Zhang J, Han L, Li W, Chen Q, Lei J, Long M, et al. Early prediction of preeclampsia and small-for-gestational-age via multi-marker model in Chinese pregnancies: a prospective screening study. BMC Preg Childbirth. 2019;19:304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.CLASP: a randomised trial of low-dose aspirin for the prevention and treatment of pre-eclampsia among 9364 pregnant women. CLASP (Collaborative Low-dose Aspirin Study in Pregnancy) Collaborative Group. Lancet. 1994;343:619–29. [PubMed] [Google Scholar]
- 84.Rolnik DL, Wright D, Poon LC, O’Gorman N, Syngelaki A, de Paco Matallana C, et al. Aspirin versus placebo in pregnancies at high risk for preterm preeclampsia. N Engl J Med. 2017;377:613–22. [DOI] [PubMed] [Google Scholar]
- 85.Caritis S, Sibai B, Hauth J, Lindheimer MD, Klebanoff M, Thom E, et al. Low-dose aspirin to prevent preeclampsia in women at high risk. N Engl J Med. 1998;338:701–5. [DOI] [PubMed] [Google Scholar]
- 86.Low-dose aspirin in prevention and treatment of intrauterine growth retardation and pregnancy-induced hypertension. Italian study of aspirin in pregnancy. Lancet. 1993;341:396–400. [PubMed] [Google Scholar]
- 87.•.Henderson JT, Whitlock EP, O’Connor E, Senger CA, Thompson JH, Rowland MG. Low-dose aspirin for prevention of morbidity and mortality from pre-eclampsia: a systematic evidence review for the U.S. Preventive Services Task Force. Ann Intern Med. 2014;160:695. [DOI] [PubMed] [Google Scholar]; In this USPSTF statement, the authors conduct a systematic review of low dose aspirin for the prevention of preeclampsia and analyze data that led to the current recommendations on the use of low dose aspirin in women at elevated risk for preeclampsia.
- 88.Andrikopoulou M, Purisch SE, Handal-Orefice R, Gyamfi-Bannerman C. Low-dose aspirin is associated with reduced spontaneous preterm birth in nulliparous women. Am J Obstet Gynecol. 2018;219:399.e1–6. [DOI] [PubMed] [Google Scholar]
- 89.McCowan LM, Harding J, Roberts A, Barker S, Ford C, Stewart A. Administration of low-dose aspirin to mothers with small for gestational age fetuses and abnormal umbilical Doppler studies to increase birthweight: a randomised double-blind controlled trial. Br J Obstet Gynaecol. 1999;106:647–51. [DOI] [PubMed] [Google Scholar]
- 90.Trudinger BJ, Cook CM, Thompson RS, Giles WB, Connelly A. Low-dose aspirin therapy improves fetal weight in umbilical placental insufficiency. Am J Obstet Gynecol. 1988;159:681–5. [DOI] [PubMed] [Google Scholar]
- 91.Roberge S, Sibai B, McCaw-Binns A, Bujold E. Low-dose aspirin in early gestation for prevention of preeclampsia and small-for-gestational-age neonates: meta-analysis of large randomized trials. Amer J Perinatol. 2016;33:781–5. [DOI] [PubMed] [Google Scholar]
- 92.Rich-Edwards JW, Stuart JJ, Skurnik G, Roche AT, Tsigas E, Fitzmaurice GM, et al. Randomized trial to reduce cardiovascular risk in women with recent preeclampsia. J Women’s Health. 2019;28:1493–504. [DOI] [PubMed] [Google Scholar]
- 93.Mann S, Hollier LM, McKay K, Brown H. What we can do about maternal mortality — and how to do it quickly. N Engl J Med. 2018;379:1689–91. [DOI] [PubMed] [Google Scholar]
- 94.•.Brown HL, Warner JJ, Gianos E, Gulati M, Hill AJ, Hollier LM, et al. Promoting risk identification and reduction of cardiovascular disease in women through collaboration with obstetricians and gynecologists: a presidential advisory from the American Heart Association and the American college of obstetricians and gynecology. Circulation. 2018;137:e843–52. [DOI] [PubMed] [Google Scholar]; This statement from the AHA and ACOG highlights the importance of coordinated healthcare delivery among OB/GYNs and cardiologists in multiple areas of preventive care.
- 95.ACOG Practice Bulletin No. 212: pregnancy and heart disease. Obstet Gynecol. 2019;133(5):e320–56. [DOI] [PubMed] [Google Scholar]
- 96.ACOG Committee on Obstetric Practice. Presidential Task Force on Redefining the Postpartum Visit Committee on Obstetric Practice. Obstet Gynecol. 2018;131(5):e140–50.29683911 [Google Scholar]
- 97.•.Grandi SM, Filion KB, Yoon S, Ayele HT, Doyle CM, Hutcheon JA, et al. Cardiovascular disease-related morbidity and mortality in women with a history of pregnancy complications: systematic review and meta-analysis. Circulation. 2019;139:1069–79. [DOI] [PubMed] [Google Scholar]; This systematic review incorporates a broader range of pregnancy complications outside of the commonly studied pre-eclampsia, gestational diabetes, or preterm delivery and demonstrates their increased association with future CVD.
- 98.Markovitz AR, Stuart JJ, Horn J, Williams PL, Rimm EB, Missmer SA, et al. Does pregnancy complication history improve cardiovascular disease risk prediction? Findings from the HUNT study in Norway. Eur Heart J. 2019;40:1113–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Stuart JJ, Tanz LJ, Cook NR, Spiegelman D, Missmer SA, Rimm EB, et al. Hypertensive disorders of pregnancy and 10-year cardiovascular risk prediction. J Am Coll Cardiol. 2018;72:1252–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Timpka S, Fraser A, Schyman T, Stuart JJ, Åsvold BO, Mogren I, et al. The value of pregnancy complication history for 10-year cardiovascular disease risk prediction in middle-aged women. Eur J Epidemiol. 2018;33:1003–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Grand’Maison S, Pilote L, Okano M, Landry T, Dayan N. Markers of vascular dysfunction after hypertensive disorders of pregnancy: a systematic review and meta-analysis. Hypertension. 2016;68:1447–58. [DOI] [PubMed] [Google Scholar]
- 102.Ciftci FC, Caliskan M, Ciftci O, Gullu H, Uckuyu A, Toprak E, et al. Impaired coronary microvascular function and increased intima–media thickness in preeclampsia. J Am Soc Hyperten. 2014;8:820–6. [DOI] [PubMed] [Google Scholar]
- 103.Timpka S, Stuart JJ, Tanz LJ, Rimm EB, Franks PW, Rich-Edwards JW. Lifestyle in progression from hypertensive disorders of pregnancy to chronic hypertension in Nurses’ Health Study II: observational cohort study. BMJ. 2017:j3024. [DOI] [PMC free article] [PubMed] [Google Scholar]


