Abstract
Vascular regeneration is a challenging topic in tissue repair. As one of the important components of the neurovascular unit (NVU), pericytes play an essential role in the maintenance of the vascular network of the spinal cord. To date, subtypes of pericytes have been identified by various markers, namely the PDGFR-β, Desmin, CD146, and NG2, each of which is involved with spinal cord injury (SCI) repair. In addition, pericytes may act as a stem cell source that is important for bone development and regeneration, whilst specific subtypes of pericyte could facilitate bone fracture and defect repair. One of the major challenges of pericyte biology is to determine the specific markers that would clearly distinguish the different subtypes of pericytes, and to develop efficient approaches to isolate and propagate pericytes. In this review, we discuss the biology and roles of pericytes, their markers for identification, and cell differentiation capacity with a focus on the potential application in the treatment of SCI and bone diseases in orthopedics.
Subject terms: Pathogenesis, Neurophysiology
Introduction
The pericyte (also called the Rouget cell or parietal cell) was first discovered by Rouget in 1873, and was named the pericyte by Zimmermann in 1923.1 The cytoplasm of the pericyte regularly forms an asteroid protuberance, and pericytes characteristically reside within the vascular basement membrane (which is usually continuous with the endothelial basement membrane), in proximity to, and often encircling, endothelial cells. Pericytes communicate with endothelial cells directly via gap junctions, tight junctions, focal adhesions, and soluble factors.2,3 For instance, the connection between the pericyte and the microvascular endothelium of the spinal cord is crucial in maintaining the tight junctions between endothelial cells and the structural integrity of the blood spinal cord barrier.4 Endothelial cells enclose the endothelial tubules, and pericytes abut the endothelial cells. Although the basement membrane mostly separates pericytes and endothelial cells, direct intercellular contacts between the two cell types occur at ‘peg-socket’ occlusions and adhesion plaques.1 The cell junction and basement membrane-like substance interweave the pericyte, endothelial cells, and the end-feet of the astrocyte (Fig. 1).5 Research suggests that pericytes are crucial for blood vessel growth and maturation via the activation of transforming growth factor-β (TGF-β) signaling, and the production of endothelial cell survival factor, angiopoietin 1 (Ang1), as well as by co-occupying endothelial tubules to provide physical stability and support for endothelial tubule function during the initial stage of angiogenesis.6 Therefore, the role of pericytes in the formation of the vasculature is of vital significance and is closely related to the form and function of endothelial cells.
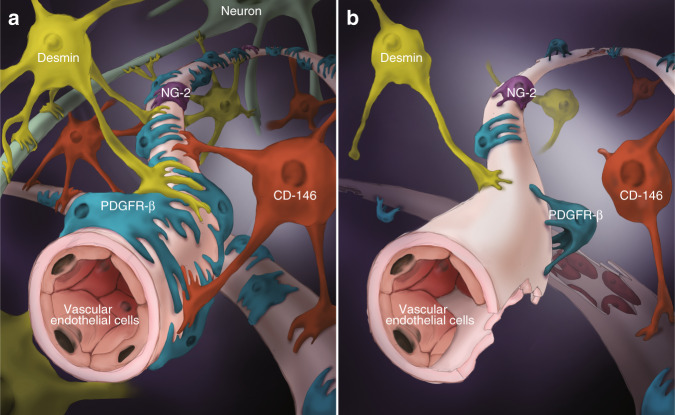
Fig. 1.
Pericytes in SCI. Schematic representation of the localization, morphology, and coverage of common subtypes of pericytes and vascular endothelial cells in normal spinal cord (a) and injured spinal cord (b)
Recent research points to the essential role of pericytes in angiogenesis and in the maintenance of vascular supply in SCI and bone fracture healing. Given the broad similarities in potential mechanisms of action of perivascular stem cells (PSC), including direct contribution to cellular regenerate, vasculogenic effects, fibrotic and anti-fibrotic activities, other paracrine effects in the spinal cord and in bone, we survey these two systems to discuss the role and potential application of PSC in the repair of SCI and bone fracture or defect. This review comprehensively defines the subtypes of pericytes, including their potential of stem cells, relationship with vascular homeostasis, and the unique function of the common subtypes of pericytes in the spinal cord and in bone, thereby providing a deeper theoretical basis for the processes of repair following spinal cord injury (SCI) and bone fracture or defect.
Pericytes have the potential of stem cells
PSC is a term that includes both pericytes and other perivascular progenitor cells with properties of mesenchymal stem cells (MSCs).7 The true nature of pericytes, described by characteristics such as their various sources, phenotypes, multipotency, and versatility, remains a controversial area of discussion. The similarities between pericytes and vascular smooth muscle cells are well documented, and whilst together they form the mural cells supporting blood vessels, selective markers, such as CD146, are used to identify phenotypic differences between the two cell types.8–10 For example, brain pericytes originate from the mesoderm and neural crest, and can differentiate into neural lineage cells and regulate the neurovascular network.11 Pericytes also demonstrate the capacity to differentiate into osteoblasts, chondrocytes, and adipocytes, providing evidence of their mesenchymal lineage differentiation potential.12,13 Pericytes and other cell types such as adventitial cells and vascular smooth muscle cells are thought to be responsible for ectopic calcification of blood vessels, heart valves, and skeletal muscle.12,14 Pericytes are also considered to be a potential source for cell-based therapeutic applications for the treatment of diseases, and of vital significance is the tissue-specific function and capacity for multilineage differentiation of pericytes.15 For instance, recent research is focused on identifying pericyte subsets with the capability to regenerate their tissue microenvironment, and/or maintain a heterologous differentiation potential.16 Research has shown that owing to their potential for myogenic differentiation, pericytes may be used to treat Duchenne muscular dystrophy.17 In line with this, skeletal pericytes have a greater osteogenic capacity than soft-tissue pericytes.16 Moreover, among soft-tissue pericytes, cells expressing CXCR4 were found to be osteoblastic, non-adipocytic precursor cells.16 Additional research identified a novel role for pericytes as stem cells capable of modifying the extracellular matrix microenvironment and promoting epidermal tissue regeneration. Pericytes were able to detach from the capillary basement membrane, differentiate into fibroblasts, and form collagen stroma during wound healing.8,18 Conversely, the collagen stroma can be fibrotic in chronically inflamed tissue and may form a fibrous stromal tumor, whereby pericytes may be therapeutically targeted.19,20 Taken together, the pericyte has stem cell potential, and alteration of the microenvironment will induce pericytes to differentiate towards a particular lineage, to attain various morphological features, and functional properties.13 On the other hand, studies of the multipotency of Tbx18-expressing pericytes appear to be inconsistent.21,22 Tbx18 was found to be expressed in vascular smooth muscle cells, pericytes, and glomerular mesangial cells, which appear to play a role in the vasculature development of the kidney.23 Whilst Tbx18-expressing cells exhibit differentiation properties of MSCs in vitro, they do not appear to function as stem cells in vivo in cell lineage-tracing experiments, such as transgenic mouse models, which indicates that Tbx18-expressing pericytes might not have significant regenerative and fibrogenic properties for therapeutic application.21,22 Nevertheless, exploring the source and differentiation cues of pericytes in depth, including the differentiation processes of subtypes of pericytes, are prime areas of research in vascular regenerative medicine as a potential source of the therapeutic cells across many disease entities.
The subtypes and hallmarks of pericytes
Pericytes share biological functions similar to stem cells and there are several subtypes of pericytes.1 Scientists have attempted to find pericyte-specific markers for the classification of the subtypes of pericytes, which has proven challenging. There are several reasons for the difficulty in the classification of pericytes: (1) the isolation of pericytes is difficult; (2) dissociation in the somatic environment may lead to changes in pericyte phenotypes; (3) the differentiation of pericytes makes it difficult to identify the subtypes, and (4) multiple markers can be present on one pericyte. For example, Desmin− pericytes lose CD13-expression during migration away from the blood vessels, so it is speculated that CD13 pericytes and Desmin+ pericytes may switch to each other when the microenvironment changes.24
Although it is difficult to classify the pericytes, one might attempt to distinguish the different pericyte markers, and their expression levels, by methods, such as immunofluorescence labeling. In this way, one can describe the pericyte subtypes according to their characteristic markers to evaluate their various properties of the function, location, source, and differentiation.
According to the existing research, pericytes express α-smooth muscle actin (SMA), smooth muscle myosin, tropomyosin, nestin, vimentin, desmin, neuron glial antigen 2 (NG2), connexin 43 (Cx43), CD146, endosialin, γ-glutamyl transpeptidase, alkaline phosphatase, protein kinase G (PKG), aminopeptidase A, regulator of G protein signaling 5 (RGS5), platelet-derived growth factor receptor-β (PDGFRβ), ATP-sensitive potassium Kir6.1, CD13, CD248, SUR2, Tbx18, and delta-like 1 homolog (DLK1); but do not express the endothelial cell marker, von Willebrand factor, nor the astrocyte marker, glial fibrillary acidic protein (GFAP).1,25,26
As pericytes and other cells could express similar biomarkers, how to distinguish between pericytes and other cells using immunofluorescence staining remains a challenging issue in pericyte research. Most of the existing studies use a variety of pericyte markers to co-stain the pericytes to determine their source, and to distinguish the pericyte subtypes. For example, pericytes could be identified and studied in relation to tissue-specific injury and angiogenesis using differential nestin/NG2 staining. Meanwhile, PDGFR-β, NG2, and CD146 have also been applied to immunofluorescence staining to distinguish pericytes from other cells.27,28 Therefore, it is feasible to distinguish between subtypes of pericytes by differential co-staining with multiple pericyte markers.
Pericyte regulation of vascular homeostasis
Pericytes are vital to vascular stability and to the regulation of vascular structure. Many reports show that the different subtypes of pericytes have different effects on the structure and function of blood vessels. It appears that PDGFR-β-type pericytes are derived from MSCs and differentiate into smooth muscle cells.26,29,30 PDGFRβ serves as a major pericyte marker and is involved in neurovascular functions, neuronal development, and aging.31–33 It regulates the development of cerebral microcirculation and the blood–brain barrier (BBB) and is associated with vascular disorders of the CNS.32,34,35 CD146 is an endothelial biomarker as well as a pericyte marker.13,35 CD146 type pericytes can be derived from CD146+ cells in bone marrow aspirates,36 or from neural precursor cells.33 Deletion of CD146 in pericyte caused an impairment in pericyte recruitment and the breakdown of BBB.33,37 Interestingly, CD146 coregulates with PDGFRβ in pericytes, which is required for pericyte recruitment, BBB development, and formation.33,37 It was revealed that the dimerization of CD146 is required for its association with PDGFRβ, which facilitate the auto-phosphorylation of PDGFRβ, the recruitment of PI3K-p110 to the p-PDGFRβ, and the downstream signaling of PDGFRβ, leading to pericyte recruitment and BBB integrity.33,37 CD146 also appears to be an orchestrator of the vascular niche that is involved in ischemic and atherosclerotic heart conditions.15,38,39 Desmin-type pericytes are derived from hematopoietic precursor cells and differentiate into fibrocytes.40,41 Like other pericyte markers, desmin is also involved in the atherosclerotic process via its regulation of stem cells and endothelial cells.42,43 NG2+ cells were identified in glia of the central nervous system (CNS),44 and NG2 is detected in arteriolar pericytes but not venular pericytes in the mesenteric microvascular system.45,46 In addition, NG2 is expressed in arterioles, capillaries, and venules pericytes in the retinal microvascular segments.47 NG2-type pericytes can also be derived from adipose-derived stem cells and differentiate into specialized adipose cells.48,49 Collectively, pericytes play a critical role in the formation and repair of the structural integrity of blood vessels. Pericyte deficiency or pathological alterations in pericytes could lead to dilated and hemorrhagic blood vessels, and is associated with disease processes.50
Deficiency of existing classification
To date, a single specific marker that could be used solely to label pericytes is undefined. The expression of pericyte makers varies between the different stages of pericyte development, and according to various pathological processes as well as in the cell culture environment. Therefore, the effective and accurate identification of pericytes is an important issue to be resolved. At present, pericytes are distinguished by a means of a combination of morphology, the relationship to endothelial cells, and labeling by greater than two pericyte markers. These criteria provide a basis by which to promote further research on the classification of pericyte subtypes.
In addition, different names of pericytes have been used and the controversies of pericyte identity remain to be fully addressed including specific versus non-specific markers. Herein, we have summarized some commonly described names and their putative biomarkers in neural and spinal cord as well as in bone systems (Table 1). Further, we have provided an update on various Cre lines generated and their usefulness for pericyte studies relevant to SCI and bone (Table 2).51–75
Table 1.
Classifications and biomarkers of perivascular stem cells
| Classifications | Biomarkers | PDGFRβ | αSMA | CD146 | desmin | NG2 | CD13 | CD44 | VIM | Nestin | CD56 | CD34 | CD31 | vWF | Sca-1 | References |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cell surface | + | + | + | + | + | + | + | + | ||||||||
| Uniprot no. for human | P09619 | P62736 | P43121 | P17661 | Q6UVK1 | P15144 | P16070 | P08670 | P48681 | P13591 | P28906 | P16284 | P04275 | |||
| Uniprot no. for mouse | P05622 | P62737 | Q8R2Y2 | P31001 | Q8VHY0 | P97449 | P15379 | P20152 | Q6P5H2 | P13595 | Q64314 | Q08481 | Q8CIZ8 | P05533 | ||
| Names descirbed (nomenclature) | ||||||||||||||||
| Perivascular cells at different locations | Pericytes in general | + | + | + | + | + | + | - | - | - | - | |||||
| Pericytes in brain tissue | + | + | + | + | + | + | 10 | |||||||||
| Capillary-associated pericytes. | + | - | + | + | + | 10,52 | ||||||||||
| Venules-associated pericytes | + | - | 52 | |||||||||||||
| Arterioles-associated pericytes | + | + | 13,46,51 | |||||||||||||
| Pericytes of microvessels | + | 52 | ||||||||||||||
| Perivascular cells at different stages | Perivascular cells | + | + | 52 | ||||||||||||
| Intermediate perivascular cells (PVC) | + | + | 52 | |||||||||||||
| Smooth muscle perivascular cells | + | - | 52 | |||||||||||||
| Bone marrow-derived pericyte precursors | + | + | 55 | |||||||||||||
| Attaching pericytes | + | +/− | +/− | +/− | 55 | |||||||||||
| Mature pericytes | +/− | +/− | +/− | +/− | 55 | |||||||||||
| Pericytic markers related cells or structure | Mural cells in all large vessels | + | + | 52 | ||||||||||||
| Arteriole vSMC | +/− | + | +/− | 55 | ||||||||||||
| vSMCs in large vessels | + | + | + | + | + | 10 | ||||||||||
| Mesenchymal stem cells | + | + | 52,54 | |||||||||||||
| Wharton’s jelly | + | - | 52 | |||||||||||||
| Associated cells with no pericytic markers | Endothelial cells | - | - | + | + | + | 52,94 | |||||||||
| Adventitial cells | - | + | + | 52,56 | ||||||||||||
| Adventitial perivascular cells | + | 52 | ||||||||||||||
| Other species (Chiken) pericytes | Pericytes | + | + | 53 | ||||||||||||
| Smooth muscle perivascular cells | + | + | + | 53 | ||||||||||||
Table 2.
Cre mouse lines and their usefulness for pericyte studies
| Cre mouse lines | Targets | Their usefulness for pericyte studies | References |
|---|---|---|---|
| In neural and SCI system | |||
| Wnt1-Cre, Sox10-Cre mice crossed to Rosa26(eYfp) | Tagging of Neural crest-derived MSC | Neural crest-derived perivascular cells | 61 |
| Foxg1(cre/+);Tgfbr2(flox/flox) (Tgfbr2-cKO) | Ko of Tgfbr2 | Brain vessel development | 64 |
| Glial fibrillary acidic protein (GFAP)-cre | Ko of beta1-integrin (beta1-itg) | Perivascular astrocyte, blood–brain barrier | 71 |
| Nestin-Cre | Ko of CLEC-2 | Maturation and integrity of the developing vasculature | 67 |
| Wnt1-cre | Ko of Ctnnb1 (beta-catenin) | Pituitary vasculature, neural crest-derived pericytes | 60 |
| NG2-CreERT:R26R-tdTomato | Ko of Ninj1 gene | Schwann cells and microvasculature | 69 |
| Zeb2 (Sip1/Zfhx1b) Ko with Tie2-Cre and Vav-iCre | Ko of Zeb2 (Sip1/Zfhx1b) | Pericyte coverage of the cephalic vasculature | 63 |
| R26R with SM22alpha-Cre | Ko of BMPR1A or MMP2 | Brain microvessels | 41 |
| In skeletal system | |||
| Tie2-Cre mice with R26Rosa-lox-Stop-lox-LacZ | Tagging of endothelial cells | Endothelial cells | 73 |
| Ocn-Cre, Dmp1-Cre and Cxcl12(gfp) | Tagging Cxcl12(gfp) expressing cells | Cxcl12-abundant reticular cells and arteriolar pericytes | 74 |
| Rosa26-YFP-Sox10-Cre | Tagging of YFP-positive PCs and vSMCs | PCs, vascular smooth muscle cells (vSMC) | 70 |
| Rosa26R-LacZ | Osterix-expressing osteoblast precursors | Coupled vascular and osteogenic transformation | 68 |
| Wnt1-Cre-Tom and GLAST-CreERT2-Tom | GLAST+ Wnt1-traced pericytes | Bone marrow pericytes | 75 |
| Msx1(lacZ), Msx2(lox) and Sm22alpha-Cre | Ko of Msx1(CreERT2) | Vascular smooth muscle cells (vSMC) in arteries | 66 |
| PDGFRalpha or PDGFRbeta Ko with SM22alpha-Cre | Ko of PDGFRalpha or PDGFRbeta | Vascular smooth muscle cells (vSMC) development | 62 |
| Bmp2 floxed/3.6Col1a1-Cre [Bmp2-cKO(od)] | Ko of Bmp2 gene in odontoblasts | Odontoblasts on vascular bed and associated pericytes | 72 |
| Alpha-SMA-GFP transgenic | Bone marrow stromal cells | Vascularization in bone microenvironment | 58 |
| Other pericyte-related systems | |||
| Myh11-Cre(ERT2)tdTomato | Ko of KLF4 | Smooth muscle cells or pericytes in adipose tissue | 57 |
| LysM-Cre | Ko of NG2 | Tumor vascularization | 59 |
| Foxd1-Cre; Rs26-tdTomato | Foxd1 progenitor-derived pericytes | Myofibroblast precursor | 65 |
The role of pericytes in SCI
SCI is a devastating and traumatic insult to the spinal nervous system, which is a population-health burden by the characteristics of high incidence, high morbidity, high cost, and presents a significant challenge for patients, care workers, and clinicians.76 The microcirculatory disturbance that occurs during the early stage of SCI causes localized edema, ischemia, and hypoxia, which leads to secondary injury, including anaerobic metabolism, tissue acidosis, free radical reaction, ion pump disorders, and a series of biochemical reactions, which further exacerbates the deterioration of spinal cord tissue.77,78 Meanwhile, vascular lesions, inflammation, and other factors will result in the formation of a necrotic cavity, spinal cord tissue softening, and possibly the formation of a glial scar, leading to the permanent loss of the spinal cord function.79,80 Although several methods, such as surgical anastomosis and physical rehabilitation, have relieved the pathological changes of SCI by varying degrees, none of these methods has a curative clinical effect and the prognosis of SCI is poor.81
Cell transplantation has been proposed as a potential treatment of SCI. However, due to the damage of local blood vessels, lack of oxygen and nutrients, and the apoptotic death of neural cells, the survival of the transplanted cells for the treatment of SCI is jeopardized without the support of the local blood vasculature.82 Thus, vascular regeneration is an important and challenging topic of SCI treatment. The neurovascular unit (NVU) of the spinal cord consists of the spinal cord vascular endothelial cells, vascular basement membranes, pericytes, glial cells, and adjacent neurons. Coordinated intercellular communication among neighboring cells is required to maintain the functional and structural stability of the NVU, which is mediated by autocrine and paracrine signaling mechanisms. Pericytes are favorably positioned in the NVU between endothelial cells, neurons, and astrocytes, to facilitate the coordination of intercellular communication, such as pericyte-endothelial signaling and pericyte-astrocyte signaling, which are involved with the regulation of vital functions of the CNS. Pericyte-signaling systems can regulate the integrity of the BBB, angiogenesis, phagocytosis, cerebral blood flow (CBF), capillary diameter, neuro-inflammation, and multipotent stem cell activity.35 The NVU is possibly the most fundamental structure maintaining the blood spinal cord barrier in the spinal microenvironment.83 The pericyte is a fundamental cell of the NVU, and performs vital functions, including neuroprotection, maintaining the pluripotency of stem cells, regulation of the BBB, and the promotion of angiogenesis and vessel maturation.84–86 The role of pericytes in the regulation of CBF, neurovascular coupling, and neurodegenerative conditions remain debatable. To date, the pericyte has been found to play an important role in maintaining the blood spinal cord barrier and vascular regeneration.86 Following SCI, new blood vessels are required to restore blood supply to damaged areas, relieve localized hypoxia, and reduce nerve apoptosis. To repair damaged blood vessels, the number and location of pericytes must also be restored. At present, owing to the absence of specific markers, there is considerable ambiguity of the distribution, number, and function of different subtypes of pericytes. Several subtypes of pericytes might have the potential for neural cell differentiation and are able to contribute to tissue repair and regeneration of SCI (Fig. 2). Further, the proper arrangement among subtypes of pericyte could provide a novel therapeutic basis to promote angiogenesis and axon regeneration following SCI.
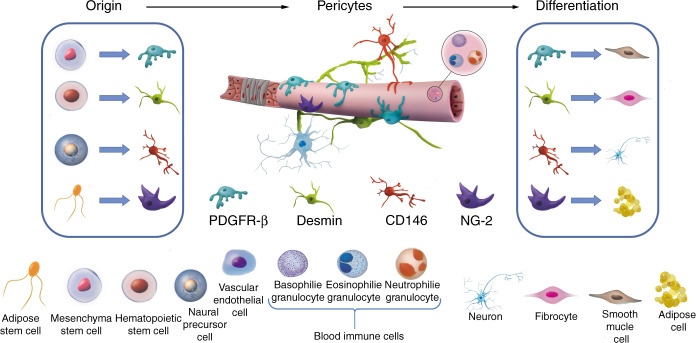
Fig. 2.
Schematic model showing pericytes by the expression of pericyte markers of PDGFRβ, CD146, desmin, and NG2 as examples, and their potential differentiation lineages
Pericytes are thought to regulate blood flow within the microvessels by constriction, and the flow of some particles in the blood vessel across the BBB, for the maintenance of the function of the nervous system and the integrity of the BBB.29 Without pericytes, large proteins can cross the spinal cord barrier by transcytosis, and affect the proliferation and differentiation of endothelial cells, leading to vascular malformations.83,87 Microvascular pericytes directly contact endothelial cells by gap junctions, thereby regulating endothelial cell proliferation and thus restricting vascular malformations.88 Accordingly, the pericyte is an important cell to help maintain the homeostasis of the vascular microenvironment following SCI. To date, relative to endothelial cells, our understanding of the roles and functions of pericytes is limited.
The functions of the subtypes of pericytes in SCI
The above common subtypes of pericytes participate in angiogenesis and functional protection from SCI. Previous research indicates that pericytes play an important role in several aspects of SCI (Fig. 3), including (1) nerve regeneration: CD146 type pericytes can differentiate into neurocyte and replace neurons in damaged areas; (2) microenvironment: the inhibition of pericyte contraction could effectively alleviate local tissue hypoxia after SCI;89 (3) post-SCI angiogenesis: NG2-type pericytes promote revascularization, vascular stability, and tissue healing;90 (4) fibrotic scar: PDGFR-β type pericytes are the source of the scar-forming cells and NG2-type pericytes promote scar formation.91 Although there are reports indicating that fibrotic scarring has a negative role in the repair of SCI, some pericyte-induced fibrotic scarring may have a positive effect on the recovery and tissue healing of SCI. Excessive inhibition or activation of certain types of pericytes can lead to an open tissue defect, whereas moderate pericyte proliferation contributes to wound closure and reduction of the fibrotic scar.92 Therefore, the pericyte is potentially a new target for the treatment of SCI. Determination of and promoting the therapeutic activation of the specific pericyte subtype ratio conducive to the recovery of SCI, represents a promising and challenging aim for future research. The promotion of angiogenesis following pericyte transplantation, and improvement of the microenvironment of the damaged zone during functional recovery are fundamental areas that must be solved for the application of pericyte-based treatments for SCI.
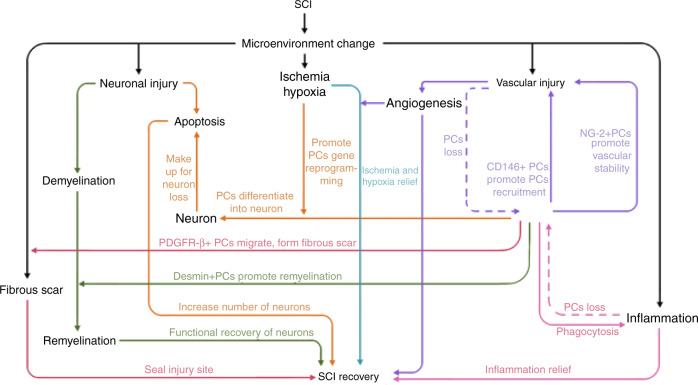
Fig. 3.
Relationships between different types of pericytes and the SCI microenvironment
Pericyte coverage of blood vessels in spinal cord
Pericytes are localized on the surface of the capillary, and play an important role in angiogenesis, vascular remodeling, and stability.12,93 Pericytes are relatively abundant in the spinal tissues as the coverage of pericytes in the spinal microvasculature is higher than that in the peripheral tissues. This phenomenon suggests that spinal cord pericytes may have a tissue-specific function. The decrease of pericyte coverage is accompanied by the decrease of vascular density in the peripheral tissues. The abnormal pathological changes of blood vessel characteristics, such as increased diameter, tortuosity, reduced perfusion, and depression may be the result of the changes that occur in the spinal cord microenvironment during the early stage of SCI.30 The coverage rate of pericytes is ~39.77% in ventral horn blood vessels and 13.34% in dorsal horn blood vessels.94 However, the proportion in the microvasculature of striated muscle is estimated at 1:10029. Our recent data also suggest that the locations and morphologies of common subtypes of pericyte vary in the normal spinal environment, as seen by immunofluorescence staining of spinal cord vessels and capillaries (unpublished observation). There appears a certain proportion and close physical connection among common subtypes of pericyte in the spinal cord. The size ratio of pericyte and vascular endothelial cells is ~1:2. In other words, the size of a pericyte is approximately equal to that of two vascular endothelial cells.29,30 This quantitative ratio might help us determine the total number of pericytes based on the number of endothelial cells in pericyte transplantation. By using the optimal number of transplanted cells with subtypes of pericytes, the goal of restoring the microenvironment of the SCI and recovering the function of the injured spinal cord can be achieved.
Application potential of pericytes in neurologic diseases and SCI repair
At present, there are two potential methods for treating diseases using the stem cell potential of pericytes: (1) exogenous pericyte cell transplantation, and (2) mobilization of endogenous pericyte cell migration. Current research mainly uses exogenous pericyte transplantation to repair tissue damage, and the endogenous pericytes mobilization method is still in the theoretical stage.95 Most pericyte transplantation mediates therapeutic effects by the secretory mechanism of pericytes to repair damaged nerve tissue. For instance, the therapeutic effects and mechanistic targets of saphenous vein-derived pericyte progenitor cells were investigated in an infarcted heart model, and found that the delivered cells could signal through a paracrine mechanism to reduce myocardial scarring, apoptosis, and fibrosis, while increasing vascular stability and attenuating permeability.96 Pericytes can promote the activation of stationary satellite cells by secreting insulin-like growth factor-1 or Ang1, and activated satellite cells may subsequently differentiate into myoblasts with tissue regenerative potential.95
Further, the repair and reconstruction of vascular tissues also depend on post-natal adult MSCs. There are many similarities between pericytes and stem cells. The pericyte is a source of precursor cell which has broad potential therapeutic applications and is an important member of the stem cell family, and has the capabilities of self-renewal, multi-directional differentiation, and immune suppression.29,30
Research has revealed that pericytes play a significant role by enhancing the structural integrity and functionality of the new blood vessels, particularly for the regulation of neurovascular function.19 MSC-derived pericytes appear to have potential in the treatment for Alzheimer’s disease by their ability to decrease amyloid-β-related pathology through protease-mediated degradation and to restore cerebral circulation.97 Consistently, pericytes appear to have an established protective role against neuronal damage in neurodegenerative diseases, via promoting a less aggressive neuronal environment and regulating neurovascular function in the CNS.98,99 These findings suggest that pericytes play a significant role in vascular regeneration and the regulation of neurovascular protection in the CNS. The widespread distribution of pericytes provides an adequate source of cells for the clinical application of autologous pericytes. At present, specific identification and isolation of pericytes have not been well established and the clinical application of pericytes is limited.
The microcirculatory disturbance in the early stage of SCI causes localized edema, ischemia, and hypoxia, leading to secondary injury including anaerobic metabolism, tissue acidosis, free radical reaction, and ion pump failure. This series of reactions give rise to ischemia and hypoxia in the spinal cord microenvironment, resulting in apoptosis and phagocytosis of spinal cord cells.100 The failure of endogenous angiogenesis directly limits the therapeutic effect of SCI treatments because new blood vessels are deprived of necessary and appropriate subsets of pericytes.93,101 Therefore, the pericyte has become a new target for SCI repair. Through gaining the knowledge and understanding of neuro-pathophysiology and the function of pericytes, the single subtype of pericyte transplantation appears to be the most common treatment of SCI and vascular regeneration.
The transplantation of pericytes may be ineffective owing to the low survival rate of transplanted pericytes in the transplant site where there is a lack of vascular support.102 The survival rate of transplanted pericytes has been shown to be independent of the cell density, and >80% of pericytes might die after transplantation for SCI.103 Therefore, methods to promote the survival and differentiation of the transplanted pericytes in the SCI microenvironment remains a challenge for the clinical application of pericyte-based therapies. At present, the survival rate of pericyte cell transplantation might be improved by combining pericytes with biological materials for delivery. For example, pericytes transplantation may be applied using a tissue scaffold for the treatment of spinal disease of the lumbar vertebra, and transplantation of a collagen matrix containing pericytes and endothelial cells may be effective for the promotion of angiogenesis during epidermal tissue regeneration.95
Controversy of pericytes in the formation of scarring of SCI
Contrary to the potential beneficial effect on SCI, pericytes were found to promote the formation of fibrotic scars and worsen spinal cord injuries.104 For instance, the number of new pericytes after SCI is twice that of astrocytes, whereas there are ten times as many astrocytes as pericytes in normal spinal tissue.26 These findings indicate that the PDGFR-β+ pericytes are a source of scar-forming cells in spinal cord lesions and inhibit axon regeneration in the adult spinal cord.26 Research also indicates that inhibiting the proliferation of the PDGFR-β+ pericytes reduces fibrotic scar formation by fibroblasts that are derived from pericytes, which promotes axon regeneration and functional recovery following SCI.92 Both studies used the Glast promoter-based transgene to select pericytes. However, the Glast gene is also expressed in other cell types, including astrocytes, Bergmann glia cells, Muller cells, and neural stem cells.105–108 Therefore, use of the Glast promoter-based transgene appears to be inconclusive and warrants further investigation. Immunofluorescence imaging shows that astrocytes have a greater basement membrane association as compared to the PDGFR-β+ pericytes.26,91 Furthermore, it appears that the mice in which the pericyte function is impaired, might sustain further CNS damage because of open SCI lesions. Moreover, pericytes are important for the formation of the BBB and vascular network, and removing pericytes from the damaged area might result in brain damage.91 The fibrotic scarring may help to isolate the inflammatory areas to maintain the integrity of the nerve tissue.91 Other evidence indicates that PDGFR-β+ pericytes play a positive role in sealing the lesion core following SCI, akin to the well-established protective role of reactive astrocytes.108 Recent research shows that the scar formation by pericytes occurs at the center of the injury, while scar formation by astrocytes is located at the periphery of the injury, indicating that when pericytes fail to proliferate, an open tissue defect may develop.109
The role of pericytes in bone development and fracture repair
Vascular regeneration is essential in bone development and repair, which is mediated by endothelial cells and pericytes.110–112 Endothelial cells are major components of the vasculature and form a crucial network between the bone system and blood circulation system. Pericytes, also known as mural cells, are perivascular cells surrounding blood vessel capillaries and endothelial cells, which facilitate the formation, maturation, and maintenance of the vascular network in bone.113–115 Pericytes also act as a stem cell source that is important for bone tissue regeneration, with several studies demonstrating their osteogenic potential.16,116–118 Recent research has focused on defining biomarkers that typify pericytes with the greatest potential for osteoblast formation.16 Importantly, CD146+ pericytes derived from skeletal tissue appear to have the most potential for osteoblast formation in vitro and skeletogenesis in vivo in comparison to pericytes derived from soft tissues, such as adipose or dermal tissue, suggesting the tissue-specific function of various pericyte populations.16 Nonetheless, pericytes derived from soft-tissue sources, such as adipose, have also shown a tremendous capacity for bone regeneration in clinical models.7,119–127
The role of perivascular progenitor cells in bone development and repair is emerging in vitro and in vivo. Early studies have shown that Gli-1-positive perivascular cells act as an innate osteogenic potential of perivascular progenitors involved in ischemia-induced vascular remodeling.14,128 During the process of embryonic endochondral ossification, Osx1 positive osteoprogenitor cells from limb mesenchyme with attachment to the blood vessels were able to invade the cartilaginous anlagen of long bones, indicating a role of MSCs-like cells, in bone development.129 In addition, endothelial and perivascular cells labeled with intravascular dyes were tracked to be present within new bone and cartilage in animal studies.130–132 Further, using lineage-tracing experiments, SMA tracked by an inducible reporter animal was found to act like osteochondroprogenitor cells.133 In this study, inducible SMA reporter mice were used to show that SMA-expressing cells were present in long bone fracture callus.133 Although SMA is not a definitive marker of pericytes (as it was also expressed in other cell types such as osteoblasts precursor cells, smooth muscle cells, and myofibroblasts), it is suggested that pericyte like cells play an important role in the early development of the skeletal system.134
More recently, pericytes have been identified as a cell source with osteogenic properties for bone fracture repair.118 In this study, pericytes were isolated from mouse embryos at 14.5–16.5 days post-coitus using CD146 as a cell surface marker. An enriched homogenous population of pericytes was also isolated by using dual markers of CD146 and NG2 with a lack of expression of CD31, CD45, and Ter119. These pericytes were also found to express high levels of PDGFRβ, which is not expressed in MSCs. Interestingly, all NG2, CD146, and PDGFRβ-positive pericytes showed osteogenic, adipogenic, and chondrogenic differentiation ability. Further, lineage-tracing experiments using the NG2-Cre or tamoxifen-inducible NG2-CreER mouse line showed that pericytes could differentiate into osteogenic cells in mice.118 In a bone fracture model, pericytes labeled with tdTomato-expressing were found to be recruited to the newly developed callus of the fractured femurs, which showed positive expression of Runx2 and type I collagen, in line with observed improved bone fracture repair.118
The therapeutic potential of pericytes in non-union fracture was also investigated using a rat tibial atrophic non-union model, in which pericytes were injected by a percutaneous route 3 weeks after the fibrous non-union procedures.125 It was revealed that injection of pericytes increased fracture callus size, accompanied by mineralization and osseous union. Taken together, it is likely that local injection of pericytes is useful for the repair of a delayed union or non-union bone fracture.125
The role of pericytes in bone formation and defect healing
The skeletal regenerative potential of exogenous PSCs derived from adipose tissue has been reported.121 The bone-forming capacity of purified PSCs was investigated using a murine calvaria defect model of bone healing.121 In this study, PSCs were isolated from a patient and implanted in a 3 mm non-healing calvaria defect centered in the parietal bone. Radiographic and histologic analysis showed that PSCs led to a significant increase in bony regeneration at the defect site with significant bone defect healing over time. In comparison, the unpurified stromal vascular fraction from the same patient had no statistically significant benefit in comparison to an acellular scaffold control.121 PSCs appear to have the potential to prevent atrophic non-union during fracture healing and could provide therapeutic benefit for a developing non-union.125
Consistently, ectopic bone formation has been demonstrated in implanted pericytes.126 For example, recent research showed that pericytes derived from human white adipose tissue with positive expression of alkaline phosphatase and bone matrix have osteogenic potential in an intramuscular mouse model.126 Further, PSCs have synergistic effects on ectopic bone formation when jointly used with bone morphogenetic protein 2 (BMP2).7 Similarly, in heterologous xenograft models, CD146+ pericytes derived from human adipose tissue were transplanted into animals and found to promote bone formation.135 In contrast, CD31+ endothelial cells were shown to suppress osteogenic differentiation.136,137
Collectively, pericytes or PSCs showed significantly greater potential for bone formation in comparison with unpurified stroma in both ectopic and orthotopic bone models. However, whether PSCs might induce bone defect healing through direct ossification of PSCs or indirect paracrine effects exerted by PSCs, or via the elimination of an inhibitory cell type within the heterogeneous stroma, remains to be elucidated. Further research is required to enrich tissue-specific PSCs that are optimized for skeletal tissue regeneration.
The role of pericytes in osteonecrosis
Pericytes could be an optimal source of stem cells in the treatment of osteonecrosis (avascular necrosis), the in situ death of a segment of bone, often affecting the femur or humerus.138,139 Although the loss or reduction of blood supply is necessary for osteonecrosis to occur, the pathogenesis of osteonecrosis remains incompletely understood.138,140 Osteonecrosis frequently affects younger patients aged 30–40 years, whereas its onset and progression are unpredictable.138,141 Osteonecrosis might be a disease initiated at a cellular level from bone marrow stromal cells and, therefore, stem cell therapy represents a promising treatment strategy.140,142 A recent systematic review and meta-analysis found that early autologous MSC implantation could attenuate disease progression, thereby alleviating the need for joint surgery.140 Additional studies suggest the potential benefit of stem cell therapy for the treatment of osteonecrosis.143–145 Despite the supporting evidence, further research is required to improve the effectiveness of stem cell therapy for osteonecrosis, specifically relating to optimal cell sources, number, and translation.144 Purified MSC-like pericytes (CD146+, CD34−, CD45−) represent a promising population in orthopedics and tissue engineering owing to their osteogenic, angiogenic, and paracrine activity for the secretion of vital growth and differentiation factors.110,126 Very little is known about the role of pericytes in osteonecrosis pathogenesis or treatment. Pericytes could be important cells for regulating the balance of physiological bone remodeling, with a loss of pericyte function implicated in osteonecrosis progression.146 Future research is necessary to further characterize tissue-specific pericytes that could both illuminate the pathogenesis of osteonecrosis and be an optimal stem cell source for therapeutic applications of osteonecrosis.
The role of pericytes in spinal fusion
Spinal fusion has become a major orthopedic procedure with a need for improved efficacy of various fusion techniques for various indications.147 Interestingly, PSCs were found to facilitate bone formation in a spinal fusion model and could potentially be applied for tissue regeneration using technologies, such as bone graft substitute or scaffold.124 Specifically, human PSCs were seeded in a demineralized bone matrix scaffold and tested in a rat posterolateral lumbar spinal fusion model, and found to increase endochondral ossification, bone deposition, and bone strength.124 Human PSCs were also found to have greater bone-forming potential in a posterolateral lumbar spinal fusion,124 as compared with the acellular control group in an athymic rat model. Using species-specific immunohistochemistry, PSCs were present within and around the newly formed bone tissue.124 Further, in a rat model of gonadectomy-induced osteoporosis, implanted PSCs showed protective effects on systemic osteoporosis.120 When PSCs were applied to a spinal fusion model in rats that had osteoporotic pathology induced ovariectomy, they showed a protective effect on bone-forming ability with a higher density of PSCs required.120
Despite recent progress showing the potential of pericytes in bone development, bone fracture, and bone defect repair, the isolation or purification of pericytes is still changing as many markers used are not pericyte-specific. For instance, CD146 used for pericytes is also expressed in MSCs,148 whereas NG2 used for pericytes is not expressed in MSCs,13,149 resulting in different subsets of PSCs. It remains a challenge to isolate and culture PSCs by avoiding the biologic property shift and regulatory measurements under cell culture conditions. Further, there were several subtypes of pericytes with different biomarkers. It will be important to use more specific markers to directly identify and isolate each specific subtype of perivascular progenitors, and to compare them individually and in combination for their therapeutic benefits in bone formation and tissue regeneration.
Future direction: proportional arrangements of pericytes for SCI repair and bone repair
It is speculated that the proportional arrangement of common subtypes of pericyte, such as PDGFR-β type, Desmin type, CD146 type, and NG2 type is of great significance for the stability of the spinal cord microenvironment. For instance, the PDGFR-β type pericytes may have the potential to close and seal lesions, thus preventing further damage to the CNS;91,150 the CD146 type pericyte may be able to secrete cell adhesion molecules and help pericytes to attach to endothelial cells;151 the NG2 type pericytes could possibly promote angiogenesis and vascular structural stability.90 Reports indicate that the different types of pericyte have different functions and that the intercellular communication among subtypes of pericyte may be of great significance, however, the specific mechanisms have not been clearly elucidated.29,90,91 Hypothetically, when the ratio of these pericytes is abnormal, CNS dysfunction might adversely occur. Similarly, it is postulated that different arrangements of pericytes will delay or facilitate bone repair. Further quantitative assessment on the proportional arrangements of subtypes of pericytes in a tissue-specific manner, will help to elucidate their approximate ratio for optimal vascular network and tissue regeneration. Collectively, we have highlighted common pericyte subtypes and their progenitors, and introduced a novel concept of the importance of maintaining proper pericyte subtype ratios in SCI and bone repair. This concept provides a theoretical basis for a potential new treatment method for SCI and bone fracture or defect, aiming to restore the number and proportion of the common pericyte subtypes through pericyte transplantation.
Conclusions
We summarize the current knowledge concerning the roles of common subtypes of pericytes in relation to the repair of SCI and bone defects. The heterogeneity in pericyte marker expression reflects the heterogeneity in function and differentiation potential of these cells. Advanced techniques are to be further employed to explore the function of pericytes in SCI, and to dissect the role of pericytes in scarring, angiogenesis, and axon regeneration. We have further outlined the lineages and differentiation paths of pericyte subtypes. We have raised an interesting and novel idea that optimal proportional ratios of different pericyte subtypes exist in spinal cord tissue, and thus may play a role in SCI recovery. Pericytes are also key components of the basic multicellular unit including endothelial cells, osteoclasts, osteoblasts, osteocytes, and bone lining cells of the skeletal system (Fig. 4). Understanding the role of pericytes in the bone microenvironment may help to develop novel therapeutic targets and diagnostic biomarkers for bone diseases, such as osteoporosis, osteonecrosis, osteoarthritis, and delayed fracture healing. Further unraveling the subtypes and functions of pericytes, developing approaches for pericyte isolation using specific cell surface markers, as well as the quantitative relation among subtypes are urgent issues to be solved in the potential clinical application of pericytes.
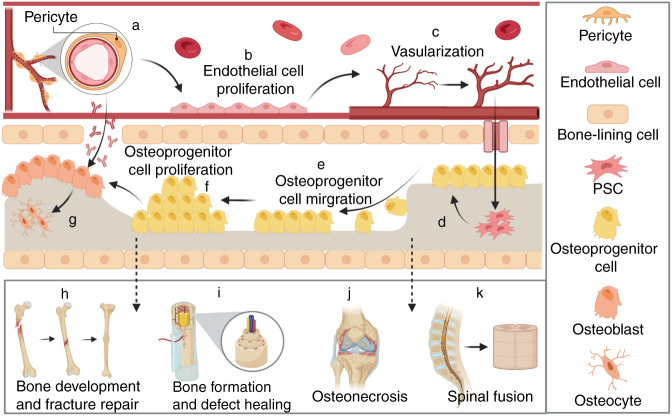
Fig. 4.
Schematic diagram proposing the role of pericytes in angiogenesis and bone repair. a Pericytes, located in the basement membrane of blood vessels, are the cells that surround endothelial cells in the capillary and veins of the body. b Pericytes communicate with endothelial cells through physical contact and paracrine signals, monitoring and stabilizing the maturation of endothelial cells. c Pericytes promote angiogenesis, which has an important role in maintaining intravascular homeostasis. d–k Pericytes are also an important source of stem cells for bone regeneration with osteogenic potential. d Mediating the differentiation of perivascular stem cells (PSCs) into osteoprogenitor cells. e Promoting the migration of osteoprogenitor cells. f Inducing the proliferation of osteoprogenitor cells. g Inducing the differentiation of osteoblasts via paracrine mode. h–k The proposed role of PSCs in bone development and fracture repair (h), bone formation and defect healing (i), osteonecrosis (j), and spinal fusion (k) as discussed in the manuscript
Acknowledgements
This study was partly supported by a research grant from the National Natural Science Funding of China (82172424,81972150,82172428), Zhejiang Medical and Health Science, and Technology Plan Project (2021KY212). This study was also supported in part by the Australian Health and Medical Research Council (NHMRC, No. 1107828). Dr. Sipin Zhu is a visiting scholar at UWA sponsored by Wenzhou Medical University.
Author contributions
S.Z. carried out most of the literature, data analysis, and participated in drafting the manuscript. M.C, Y.Y., Q.W., Z.H, W.N., X.W., and S.B. contributed to manuscript drafting and editing, and the formation of the figures. H.X., Jian Xiao, and Jiake Xu supervised the project. S.Z. and Jiake Xu conceptualized the study and revised the paper. All authors read and approved the final manuscript.
Competing interests
All authors declare no competing interests.
Contributor Information
Jian Xiao, Email: xfxj2000@126.com.
Jiake Xu, Email: jiake.xu@uwa.edu.au.
References
- 1.Armulik A, Genove G, Betsholtz C. Pericytes: developmental, physiological, and pathological perspectives, problems, and promises. Dev. Cell. 2011;21:193–215. doi: 10.1016/j.devcel.2011.07.001. [DOI] [PubMed] [Google Scholar]
- 2.Armulik A, et al. Pericytes regulate the blood-brain barrier. Nature. 2010;468:557–561. doi: 10.1038/nature09522. [DOI] [PubMed] [Google Scholar]
- 3.Dalkara T, Gursoy-Ozdemir Y, Yemisci M. Brain microvascular pericytes in health and disease. Acta Neuropathol. 2011;122:1–9. doi: 10.1007/s00401-011-0847-6. [DOI] [PubMed] [Google Scholar]
- 4.Winkler EA, et al. Blood-spinal cord barrier breakdown and pericyte reductions in amyotrophic lateral sclerosis. Acta Neuropathol. 2013;125:111–120. doi: 10.1007/s00401-012-1039-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhou W, et al. Targeting glioma stem cell-derived pericytes disrupts the blood-tumor barrier and improves chemotherapeutic efficacy. Cell Stem Cell. 2017;21:591–603 e594. doi: 10.1016/j.stem.2017.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Oudega M. Molecular and cellular mechanisms underlying the role of blood vessels in spinal cord injury and repair. Cell Tissue Res. 2012;349:269–288. doi: 10.1007/s00441-012-1440-6. [DOI] [PubMed] [Google Scholar]
- 7.James AW, et al. Perivascular stem cells: a prospectively purified mesenchymal stem cell population for bone tissue engineering. Stem Cells Transl. Med. 2012;1:510–519. doi: 10.5966/sctm.2012-0002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Paquet-Fifield S, et al. A role for pericytes as microenvironmental regulators of human skin tissue regeneration. J. Clin. Investig. 2009;119:2795–2806. doi: 10.1172/JCI38535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yao Y, Chen ZL, Norris EH, Strickland S. Astrocytic laminin regulates pericyte differentiation and maintains blood brain barrier integrity. Nat. Commun. 2014;5:3413. doi: 10.1038/ncomms4413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Smyth LCD, et al. Markers for human brain pericytes and smooth muscle cells. J. Chem. Neuroanat. 2018;92:48–60. doi: 10.1016/j.jchemneu.2018.06.001. [DOI] [PubMed] [Google Scholar]
- 11.Nakagomi T, Nakano-Doi A, Kawamura M, Matsuyama T. Do vascular pericytes contribute to neurovasculogenesis in the central nervous system as multipotent vascular stem cells? Stem Cells Dev. 2015;24:1730–1739. doi: 10.1089/scd.2015.0039. [DOI] [PubMed] [Google Scholar]
- 12.Collett GD, Canfield AE. Angiogenesis and pericytes in the initiation of ectopic calcification. Circ. Res. 2005;96:930–938. doi: 10.1161/01.RES.0000163634.51301.0d. [DOI] [PubMed] [Google Scholar]
- 13.Crisan M, et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell. 2008;3:301–313. doi: 10.1016/j.stem.2008.07.003. [DOI] [PubMed] [Google Scholar]
- 14.Kramann R, et al. Adventitial MSC-like cells are progenitors of vascular smooth muscle cells and drive vascular calcification in chronic kidney disease. Cell Stem Cell. 2016;19:628–642. doi: 10.1016/j.stem.2016.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chen WC, et al. Human myocardial pericytes: multipotent mesodermal precursors exhibiting cardiac specificity. Stem Cells. 2015;33:557–573. doi: 10.1002/stem.1868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xu J, et al. Comparison of skeletal and soft tissue pericytes identifies CXCR4+ bone forming mural cells in human tissues. Bone Res. 2020;8:22. doi: 10.1038/s41413-020-0097-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dellavalle A, et al. Pericytes of human skeletal muscle are myogenic precursors distinct from satellite cells. Nat. Cell Biol. 2007;9:255–267. doi: 10.1038/ncb1542. [DOI] [PubMed] [Google Scholar]
- 18.Bice BD, et al. Environmental enrichment induces pericyte and IgA-dependent wound repair and lifespan extension in a colon tumor model. Cell Rep. 2017;19:760–773. doi: 10.1016/j.celrep.2017.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cheng L, et al. Glioblastoma stem cells generate vascular pericytes to support vessel function and tumor growth. Cell. 2013;153:139–152. doi: 10.1016/j.cell.2013.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yamauchi M, Barker TH, Gibbons DL, Kurie JM. The fibrotic tumor stroma. J. Clin. Investig. 2018;128:16–25. doi: 10.1172/JCI93554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Birbrair A, et al. How plastic are pericytes? Stem Cells Dev. 2017;26:1013–1019. doi: 10.1089/scd.2017.0044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Guimaraes-Camboa N, et al. Pericytes of multiple organs do not behave as mesenchymal stem cells in vivo. Cell Stem Cell. 2017;20:345–359 e345. doi: 10.1016/j.stem.2016.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Xu J, Nie X, Cai X, Cai CL, Xu PX. Tbx18 is essential for normal development of vasculature network and glomerular mesangium in the mammalian kidney. Dev. Biol. 2014;391:17–31. doi: 10.1016/j.ydbio.2014.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lyle LT, et al. Alterations in pericyte subpopulations are associated with elevated blood-tumor barrier permeability in experimental brain metastasis of breast cancer. Clin. Cancer Res. 2016;22:5287–5299. doi: 10.1158/1078-0432.CCR-15-1836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Farrington-Rock C, et al. Chondrogenic and adipogenic potential of microvascular pericytes. Circulation. 2004;110:2226–2232. doi: 10.1161/01.CIR.0000144457.55518.E5. [DOI] [PubMed] [Google Scholar]
- 26.Göritz C, et al. A pericyte origin of spinal cord scar tissue. Science. 2011;333:238–242. doi: 10.1126/science.1203165. [DOI] [PubMed] [Google Scholar]
- 27.Birbrair A, et al. Type-1 pericytes accumulate after tissue injury and produce collagen in an organ-dependent manner. Stem Cell Res Ther. 2014;5:122. doi: 10.1186/scrt512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Birbrair A, et al. Type-2 pericytes participate in normal and tumoral angiogenesis. Am. J. Physiol. Cell Physiol. 2014;307:C25–C38. doi: 10.1152/ajpcell.00084.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Peppiatt CM, Howarth C, Mobbs P, Attwell D. Bidirectional control of CNS capillary diameter by pericytes. Nature. 2006;443:700–704. doi: 10.1038/nature05193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yemisci M, et al. Pericyte contraction induced by oxidative-nitrative stress impairs capillary reflow despite successful opening of an occluded cerebral artery. Nat. Med. 2009;15:1031–1037. doi: 10.1038/nm.2022. [DOI] [PubMed] [Google Scholar]
- 31.Daneman R, Zhou L, Kebede AA, Barres BA. Pericytes are required for blood-brain barrier integrity during embryogenesis. Nature. 2010;468:562–566. doi: 10.1038/nature09513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bell RD, et al. Pericytes control key neurovascular functions and neuronal phenotype in the adult brain and during brain aging. Neuron. 2010;68:409–427. doi: 10.1016/j.neuron.2010.09.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chen J, et al. CD146 coordinates brain endothelial cell-pericyte communication for blood-brain barrier development. Proc. Natl. Acad. Sci. USA. 2017;114:E7622–E7631. doi: 10.1073/pnas.1710848114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Winkler EA, Bell RD, Zlokovic BV. Pericyte-specific expression of PDGF beta receptor in mouse models with normal and deficient PDGF beta receptor signaling. Mol. Neurodegener. 2010;5:32. doi: 10.1186/1750-1326-5-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sweeney MD, Ayyadurai S, Zlokovic BV. Pericytes of the neurovascular unit: key functions and signaling pathways. Nat. Neurosci. 2016;19:771–783. doi: 10.1038/nn.4288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hassanpour M, et al. Autophagy modulation altered differentiation capacity of CD146+ cells toward endothelial cells, pericytes, and cardiomyocytes. Stem Cell Res. Ther. 2020;11:139. doi: 10.1186/s13287-020-01656-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen J, et al. CD146 is essential for PDGFRbeta-induced pericyte recruitment. Protein Cell. 2018;9:743–747. doi: 10.1007/s13238-017-0484-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen CW, et al. Human pericytes for ischemic heart repair. Stem Cells. 2013;31:305–316. doi: 10.1002/stem.1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wilkinson FL, et al. Contribution of VCAF-positive cells to neovascularization and calcification in atherosclerotic plaque development. J. Pathol. 2007;211:362–369. doi: 10.1002/path.2114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nehls V, Denzer K, Drenckhahn D. Pericyte involvement in capillary sprouting during angiogenesis in situ. Cell Tissue Res. 1992;270:469–474. doi: 10.1007/BF00645048. [DOI] [PubMed] [Google Scholar]
- 41.El-Bizri N, et al. SM22alpha-targeted deletion of bone morphogenetic protein receptor 1A in mice impairs cardiac and vascular development, and influences organogenesis. Development. 2008;135:2981–2991. doi: 10.1242/dev.017863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bruijn LE, van den Akker B, van Rhijn CM, Hamming JF, Lindeman JHN. Extreme diversity of the human vascular mesenchymal cell landscape. J. Am. Heart Assoc. 2020;9:e017094. doi: 10.1161/JAHA.120.017094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kelly-Goss MR, Sweat RS, Stapor PC, Peirce SM, Murfee WL. Targeting pericytes for angiogenic therapies. Microcirculation. 2014;21:345–357. doi: 10.1111/micc.12107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Binamé F. Transduction of extracellular cues into cell polarity: the role of the transmembrane proteoglycan NG2. Mol. Neurobiol. 2014;50:482–493. doi: 10.1007/s12035-013-8610-8. [DOI] [PubMed] [Google Scholar]
- 45.Alon R, Nourshargh S. Learning in motion: pericytes instruct migrating innate leukocytes. Nat. Immunol. 2013;14:14–15. doi: 10.1038/ni.2489. [DOI] [PubMed] [Google Scholar]
- 46.Murfee WL, Skalak TC, Peirce SM. Differential arterial/venous expression of NG2 proteoglycan in perivascular cells along microvessels: identifying a venule-specific phenotype. Microcirculation. 2005;12:151–160. doi: 10.1080/10739680590904955. [DOI] [PubMed] [Google Scholar]
- 47.Chan-Ling T, Hughes S. NG2 can be used to identify arteries versus veins enabling the characterization of the different functional roles of arterioles and venules during microvascular network growth and remodeling. Microcirculation. 2005;12:539–540. doi: 10.1080/10739680500253287. [DOI] [PubMed] [Google Scholar]
- 48.Volz, K. S. et al. Pericytes are progenitors for coronary artery smooth muscle. eLife4, e10036 (2015). [DOI] [PMC free article] [PubMed]
- 49.Ivanova E, Kovacs-Oller T, Sagdullaev BT. Vascular pericyte impairment and connexin43 gap junction deficit contribute to vasomotor decline in diabetic retinopathy. J. Neurosci. 2017;37:7580–7594. doi: 10.1523/JNEUROSCI.0187-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ahmed TA, El-Badri N. Pericytes: the role of multipotent stem cells in vascular maintenance and regenerative medicine. Adv. Exp. Med. Biol. 2018;1079:69–86. doi: 10.1007/5584_2017_138. [DOI] [PubMed] [Google Scholar]
- 51.Crisan M, et al. Perivascular multipotent progenitor cells in human organs. Ann. N. Y. Acad. Sci. 2009;1176:118–123. doi: 10.1111/j.1749-6632.2009.04967.x. [DOI] [PubMed] [Google Scholar]
- 52.Crisan M, Corselli M, Chen WC, Peault B. Perivascular cells for regenerative medicine. J. Cell Mol. Med. 2012;16:2851–2860. doi: 10.1111/j.1582-4934.2012.01617.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Fujimoto T, Singer SJ. Immunocytochemical studies of desmin and vimentin in pericapillary cells of chicken. J. Histochem. Cytochem. 1987;35:1105–1115. doi: 10.1177/35.10.3305702. [DOI] [PubMed] [Google Scholar]
- 54.Gronthos S, Simmons PJ, Graves SE, Robey PG. Integrin-mediated interactions between human bone marrow stromal precursor cells and the extracellular matrix. Bone. 2001;28:174–181. doi: 10.1016/s8756-3282(00)00424-5. [DOI] [PubMed] [Google Scholar]
- 55.Hall AP, Westwood FR, Wadsworth PF. Review of the effects of anti-angiogenic compounds on the epiphyseal growth plate. Toxicol. Pathol. 2006;34:131–147. doi: 10.1080/01926230600611836. [DOI] [PubMed] [Google Scholar]
- 56.Passman JN, et al. A sonic hedgehog signaling domain in the arterial adventitia supports resident Sca1+ smooth muscle progenitor cells. Proc. Natl. Acad. Sci. USA. 2008;105:9349–9354. doi: 10.1073/pnas.0711382105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bulut GB, et al. KLF4 (Kruppel-Like Factor 4)-dependent perivascular plasticity contributes to adipose tissue inflammation. Arterioscler. Thromb. Vasc. Biol. 2021;41:284–301. doi: 10.1161/ATVBAHA.120.314703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cai X, et al. Bone marrow derived pluripotent cells are pericytes which contribute to vascularization. Stem Cell Rev. Rep. 2009;5:437–445. doi: 10.1007/s12015-009-9097-6. [DOI] [PubMed] [Google Scholar]
- 59.Cejudo-Martin P, Kucharova K, Stallcup WB. Role of NG2 proteoglycan in macrophage recruitment to brain tumors and sites of CNS demyelination. Trends Cell Mol. Biol. 2016;11:55–65. [PMC free article] [PubMed] [Google Scholar]
- 60.Davis SW, et al. beta-catenin is required in the neural crest and mesencephalon for pituitary gland organogenesis. BMC Dev. Biol. 2016;16:16. doi: 10.1186/s12861-016-0118-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Foster K, et al. Contribution of neural crest-derived cells in the embryonic and adult thymus. J. Immunol. 2008;180:3183–3189. doi: 10.4049/jimmunol.180.5.3183. [DOI] [PubMed] [Google Scholar]
- 62.French WJ, Creemers EE, Tallquist MD. Platelet-derived growth factor receptors direct vascular development independent of vascular smooth muscle cell function. Mol. Cell Biol. 2008;28:5646–5657. doi: 10.1128/MCB.00441-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Goossens S, et al. The EMT regulator Zeb2/Sip1 is essential for murine embryonic hematopoietic stem/progenitor cell differentiation and mobilization. Blood. 2011;117:5620–5630. doi: 10.1182/blood-2010-08-300236. [DOI] [PubMed] [Google Scholar]
- 64.Hellbach N, et al. Neural deletion of Tgfbr2 impairs angiogenesis through an altered secretome. Hum. Mol. Genet. 2014;23:6177–6190. doi: 10.1093/hmg/ddu338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hung C, et al. Role of lung pericytes and resident fibroblasts in the pathogenesis of pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 2013;188:820–830. doi: 10.1164/rccm.201212-2297OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lopes M, et al. Msx genes define a population of mural cell precursors required for head blood vessel maturation. Development. 2011;138:3055–3066. doi: 10.1242/dev.063214. [DOI] [PubMed] [Google Scholar]
- 67.Lowe KL, et al. Podoplanin and CLEC-2 drive cerebrovascular patterning and integrity during development. Blood. 2015;125:3769–3777. doi: 10.1182/blood-2014-09-603803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Maes C, et al. Osteoblast precursors, but not mature osteoblasts, move into developing and fractured bones along with invading blood vessels. Dev. Cell. 2010;19:329–344. doi: 10.1016/j.devcel.2010.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Tomita Y, et al. Ninjurin 1 mediates peripheral nerve regeneration through Schwann cell maturation of NG2-positive cells. Biochem. Biophys. Res. Commun. 2019;519:462–468. doi: 10.1016/j.bbrc.2019.09.007. [DOI] [PubMed] [Google Scholar]
- 70.Trost A, et al. Characterization of dsRed2-positive cells in the doublecortin-dsRed2 transgenic adult rat retina. Histochem. Cell Biol. 2014;142:601–617. doi: 10.1007/s00418-014-1259-1. [DOI] [PubMed] [Google Scholar]
- 71.Venkatesan C, Birch D, Peng CY, Kessler JA. Astrocytic beta1-integrin affects cellular composition of murine blood brain barrier in the cerebral cortex. Int J. Dev. Neurosci. 2015;44:48–54. doi: 10.1016/j.ijdevneu.2015.05.005. [DOI] [PubMed] [Google Scholar]
- 72.Yang W, et al. Bmp2 is required for odontoblast differentiation and pulp vasculogenesis. J. Dent. Res. 2012;91:58–64. doi: 10.1177/0022034511424409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zeisberg EM, Potenta S, Xie L, Zeisberg M, Kalluri R. Discovery of endothelial to mesenchymal transition as a source for carcinoma-associated fibroblasts. Cancer Res. 2007;67:10123–10128. doi: 10.1158/0008-5472.CAN-07-3127. [DOI] [PubMed] [Google Scholar]
- 74.Zhang J, Link DC. Targeting of mesenchymal stromal cells by Cre-recombinase transgenes commonly used to target osteoblast lineage cells. J. Bone Min. Res. 2016;31:2001–2007. doi: 10.1002/jbmr.2877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sierra R, et al. Contribution of neural crest and GLAST+ Wnt1+ bone marrow pericytes with liver fibrogenesis and/or regeneration. Liver Int. 2020;40:977–987. doi: 10.1111/liv.14401. [DOI] [PubMed] [Google Scholar]
- 76.El Masri WS, Kumar N. Traumatic spinal cord injuries. Lancet (Lond., Engl.) 2011;377:972–974. doi: 10.1016/S0140-6736(11)60248-1. [DOI] [PubMed] [Google Scholar]
- 77.Fan B, et al. Microenvironment imbalance of spinal cord injury. Cell Transplant. 2018;27:853–866. doi: 10.1177/0963689718755778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Norden, D. M. et al. Bone marrow-derived monocytes drive the inflammatory microenvironment in local and remote regions after thoracic spinal cord injury. J. Neurotrauma36, 937–949 (2018). [DOI] [PMC free article] [PubMed]
- 79.Kumar N, Osman A, Chowdhury JR. Traumatic spinal cord injuries. J. Clin. Orthop. Trauma. 2017;8:116–124. doi: 10.1016/j.jcot.2017.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Taoka Y, Okajima K. Spinal cord injury in the rat. Prog. Neurobiol. 1998;56:341–358. doi: 10.1016/s0301-0082(98)00049-5. [DOI] [PubMed] [Google Scholar]
- 81.Silva NA, Sousa N, Reis RL, Salgado AJ. From basics to clinical: a comprehensive review on spinal cord injury. Prog. Neurobiol. 2014;114:25–57. doi: 10.1016/j.pneurobio.2013.11.002. [DOI] [PubMed] [Google Scholar]
- 82.Hurtado A, et al. Poly (D,L-lactic acid) macroporous guidance scaffolds seeded with Schwann cells genetically modified to secrete a bi-functional neurotrophin implanted in the completely transected adult rat thoracic spinal cord. Biomaterials. 2006;27:430–442. doi: 10.1016/j.biomaterials.2005.07.014. [DOI] [PubMed] [Google Scholar]
- 83.Lee JY, Kim HS, Choi HY, Oh TH, Yune TY. Fluoxetine inhibits matrix metalloprotease activation and prevents disruption of blood-spinal cord barrier after spinal cord injury. Brain. 2012;135:2375–2389. doi: 10.1093/brain/aws171. [DOI] [PubMed] [Google Scholar]
- 84.Kisler K, et al. Pericyte degeneration leads to neurovascular uncoupling and limits oxygen supply to brain. Nat. Neurosci. 2017;20:406–416. doi: 10.1038/nn.4489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Ryu, B. et al. Allogeneic adipose-derived mesenchymal stem cell sheet that produces neurological improvement with angiogenesis and neurogenesis in a rat stroke model. J. Neurosurg. 132, 442–455 (2019). [DOI] [PubMed]
- 86.Shaw I, Rider S, Mullins J, Hughes J, Peault B. Pericytes in the renal vasculature: roles in health and disease. Nat. Rev. Nephrol. 2018;14:521–534. doi: 10.1038/s41581-018-0032-4. [DOI] [PubMed] [Google Scholar]
- 87.Bergers G, Song S, Meyer-Morse N, Bergsland E, Hanahan D. Benefits of targeting both pericytes and endothelial cells in the tumor vasculature with kinase inhibitors. J. Clin. Investig. 2003;111:1287–1295. doi: 10.1172/JCI17929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Alarcon-Martinez, L. et al. Capillary pericytes express alpha-smooth muscle actin, which requires prevention of filamentous-actin depolymerization for detection. Elife7, e34861 (2018). [DOI] [PMC free article] [PubMed]
- 89.Cheng J, et al. Targeting pericytes for therapeutic approaches to neurological disorders. Acta Neuropathol. 2018;136:507–523. doi: 10.1007/s00401-018-1893-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Hesp ZC, et al. Proliferating NG2-cell-dependent angiogenesis and scar formation alter axon growth and functional recovery after spinal cord injury in mice. J. Neurosci. 2018;38:1366–1382. doi: 10.1523/JNEUROSCI.3953-16.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Starting the scar: a primary role for pericytes? Nat. Med.17, 1052–1053 (2011). [DOI] [PubMed]
- 92.Dias DO, et al. Reducing pericyte-derived scarring promotes recovery after spinal cord injury. Cell. 2018;173:153–165 e122. doi: 10.1016/j.cell.2018.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Caporali A, et al. Contribution of pericyte paracrine regulation of the endothelium to angiogenesis. Pharm. Ther. 2017;171:56–64. doi: 10.1016/j.pharmthera.2016.10.001. [DOI] [PubMed] [Google Scholar]
- 94.Yamadera M, et al. Microvascular disturbance with decreased pericyte coverage is prominent in the ventral horn of patients with amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. Frontotemporal Degener. 2015;16:393–401. doi: 10.3109/21678421.2015.1011663. [DOI] [PubMed] [Google Scholar]
- 95.Rozycka J, Brzoska E, Skirecki T. Aspects of pericytes and their potential therapeutic use. Postepy Hig. Med Dosw (Online) 2017;71:186–197. doi: 10.5604/01.3001.0010.3803. [DOI] [PubMed] [Google Scholar]
- 96.Katare R, et al. Transplantation of human pericyte progenitor cells improves the repair of infarcted heart through activation of an angiogenic program involving micro-RNA-132. Circ. Res. 2011;109:894–906. doi: 10.1161/CIRCRESAHA.111.251546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Tachibana M, Yamazaki Y, Liu CC, Bu G, Kanekiyo T. Pericyte implantation in the brain enhances cerebral blood flow and reduces amyloid-beta pathology in amyloid model mice. Exp. Neurol. 2018;300:13–21. doi: 10.1016/j.expneurol.2017.10.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Coatti GC, et al. Pericytes extend survival of ALS SOD1 mice and induce the expression of antioxidant enzymes in the murine model and in IPSCs derived neuronal cells from an ALS patient. Stem Cell Rev. 2017;13:686–698. doi: 10.1007/s12015-017-9752-2. [DOI] [PubMed] [Google Scholar]
- 99.Winkler EA, Bell RD, Zlokovic BV. Central nervous system pericytes in health and disease. Nat. Neurosci. 2011;14:1398–1405. doi: 10.1038/nn.2946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Oh JS, et al. Hypoxia-preconditioned adipose tissue-derived mesenchymal stem cell increase the survival and gene expression of engineered neural stem cells in a spinal cord injury model. Neurosci. Lett. 2010;472:215–219. doi: 10.1016/j.neulet.2010.02.008. [DOI] [PubMed] [Google Scholar]
- 101.Ding WG, Yan WH, Wei ZX, Liu JB. Difference in intraosseous blood vessel volume and number in osteoporotic model mice induced by spinal cord injury and sciatic nerve resection. J. Bone Min. Metab. 2012;30:400–407. doi: 10.1007/s00774-011-0328-y. [DOI] [PubMed] [Google Scholar]
- 102.Yokota K, et al. Periostin promotes scar formation through the interaction between pericytes and infiltrating monocytes/macrophages after spinal cord injury. Am. J. Pathol. 2017;187:639–653. doi: 10.1016/j.ajpath.2016.11.010. [DOI] [PubMed] [Google Scholar]
- 103.Hill J, Rom S, Ramirez SH, Persidsky Y. Emerging roles of pericytes in the regulation of the neurovascular unit in health and disease. J. Neuroimmune Pharm. 2014;9:591–605. doi: 10.1007/s11481-014-9557-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Tran AP, Warren PM, Silver J. The biology of regeneration failure and success after spinal cord injury. Physiol. Rev. 2018;98:881–917. doi: 10.1152/physrev.00017.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Ehm O, et al. RBPJkappa-dependent signaling is essential for long-term maintenance of neural stem cells in the adult hippocampus. J. Neurosci. 2010;30:13794–13807. doi: 10.1523/JNEUROSCI.1567-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Shibata T, et al. Glutamate transporter GLAST is expressed in the radial glia-astrocyte lineage of developing mouse spinal cord. J. Neurosci. 1997;17:9212–9219. doi: 10.1523/JNEUROSCI.17-23-09212.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Wynn TA. Common and unique mechanisms regulate fibrosis in various fibroproliferative diseases. J. Clin. Investig. 2007;117:524–529. doi: 10.1172/JCI31487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Narang A, Zheng B. To scar or not to scar. Trends Mol. Med. 2018;24:522–524. doi: 10.1016/j.molmed.2018.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Dias DO, Goritz C. Fibrotic scarring following lesions to the central nervous system. Matrix Biol. 2018;68-69:561–570. doi: 10.1016/j.matbio.2018.02.009. [DOI] [PubMed] [Google Scholar]
- 110.Chim SM, et al. Angiogenic factors in bone local environment. Cytokine Growth Factor Rev. 2013;24:297–310. doi: 10.1016/j.cytogfr.2013.03.008. [DOI] [PubMed] [Google Scholar]
- 111.Zhu S, et al. Endothelial cells produce angiocrine factors to regulate bone and cartilage via versatile mechanisms. Theranostics. 2020;10:5957–5965. doi: 10.7150/thno.45422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Lamagna C, Bergers G. The bone marrow constitutes a reservoir of pericyte progenitors. J. Leukoc. Biol. 2006;80:677–681. doi: 10.1189/jlb.0506309. [DOI] [PubMed] [Google Scholar]
- 113.Tang W, et al. White fat progenitor cells reside in the adipose vasculature. Science. 2008;322:583–586. doi: 10.1126/science.1156232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Krautler NJ, et al. Follicular dendritic cells emerge from ubiquitous perivascular precursors. Cell. 2012;150:194–206. doi: 10.1016/j.cell.2012.05.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Zhao H, et al. Secretion of shh by a neurovascular bundle niche supports mesenchymal stem cell homeostasis in the adult mouse incisor. Cell Stem Cell. 2014;14:160–173. doi: 10.1016/j.stem.2013.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Diaz-Flores, L., Gutierrez, R., Lopez-Alonso, A., Gonzalez, R. & Varela, H. Pericytes as a supplementary source of osteoblasts in periosteal osteogenesis. Clin. Orthop. Relat. Res. 280–286 (1992). [PubMed]
- 117.Doherty MJ, et al. Vascular pericytes express osteogenic potential in vitro and in vivo. J. Bone Min. Res. 1998;13:828–838. doi: 10.1359/jbmr.1998.13.5.828. [DOI] [PubMed] [Google Scholar]
- 118.Supakul, S. et al. Pericytes as a source of osteogenic cells in bone fracture healing. Int. J. Mol. Sci.20, 1079 (2019). [DOI] [PMC free article] [PubMed]
- 119.Meyers CA, et al. Early immunomodulatory effects of implanted human perivascular stromal cells during bone formation. Tissue Eng. Part A. 2018;24:448–457. doi: 10.1089/ten.tea.2017.0023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Lee S, et al. Brief report: human perivascular stem cells and nel-like protein-1 synergistically enhance spinal fusion in osteoporotic rats. Stem Cells. 2015;33:3158–3163. doi: 10.1002/stem.2103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.James AW, et al. An abundant perivascular source of stem cells for bone tissue engineering. Stem Cells Transl. Med. 2012;1:673–684. doi: 10.5966/sctm.2012-0053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.James, A. W. et al. Use of human perivascular stem cells for bone regeneration. J. Vis. Exp. e2952 (2012). [DOI] [PMC free article] [PubMed]
- 123.Askarinam A, et al. Human perivascular stem cells show enhanced osteogenesis and vasculogenesis with Nel-like molecule I protein. Tissue Eng. Part A. 2013;19:1386–1397. doi: 10.1089/ten.tea.2012.0367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Chung CG, et al. Human perivascular stem cell-based bone graft substitute induces rat spinal fusion. Stem Cells Transl. Med. 2014;3:1231–1241. doi: 10.5966/sctm.2014-0027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Tawonsawatruk T, et al. Adipose derived pericytes rescue fractures from a failure of healing–non-union. Sci. Rep. 2016;6:22779. doi: 10.1038/srep22779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.James AW, et al. Pericytes for the treatment of orthopedic conditions. Pharm. Ther. 2017;171:93–103. doi: 10.1016/j.pharmthera.2016.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.James AW, Peault B. Perivascular mesenchymal progenitors for bone regeneration. J. Orthop. Res. 2019;37:1221–1228. doi: 10.1002/jor.24284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Baker AH, Peault B. A Gli(1)ttering role for perivascular stem cells in blood vessel remodeling. Cell Stem Cell. 2016;19:563–565. doi: 10.1016/j.stem.2016.10.011. [DOI] [PubMed] [Google Scholar]
- 129.Jotereau FV, Le Douarin NM. The development relationship between osteocytes and osteoclasts: a study using the quail-chick nuclear marker in endochondral ossification. Dev. Biol. 1978;63:253–265. doi: 10.1016/0012-1606(78)90132-x. [DOI] [PubMed] [Google Scholar]
- 130.Salazar VS, Gamer LW, Rosen V. BMP signalling in skeletal development, disease and repair. Nat. Rev. Endocrinol. 2016;12:203–221. doi: 10.1038/nrendo.2016.12. [DOI] [PubMed] [Google Scholar]
- 131.Ahi EP. Signalling pathways in trophic skeletal development and morphogenesis: Insights from studies on teleost fish. Dev. Biol. 2016;420:11–31. doi: 10.1016/j.ydbio.2016.10.003. [DOI] [PubMed] [Google Scholar]
- 132.Diaz-Flores L, Jr, et al. Cell sources for cartilage repair; contribution of the mesenchymal perivascular niche. Front. Biosci. 2012;4:1275–1294. doi: 10.2741/s331. [DOI] [PubMed] [Google Scholar]
- 133.Grcevic D, et al. In vivo fate mapping identifies mesenchymal progenitor cells. Stem Cells. 2012;30:187–196. doi: 10.1002/stem.780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Diaz-Flores L, Gutierrez R, Gonzalez P, Varela H. Inducible perivascular cells contribute to the neochondrogenesis in grafted perichondrium. Anat. Rec. 1991;229:1–8. doi: 10.1002/ar.1092290102. [DOI] [PubMed] [Google Scholar]
- 135.Zhang X, et al. The Nell-1 growth factor stimulates bone formation by purified human perivascular cells. Tissue Eng. Part A. 2011;17:2497–2509. doi: 10.1089/ten.tea.2010.0705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Rajashekhar G, et al. IFATS collection: adipose stromal cell differentiation is reduced by endothelial cell contact and paracrine communication: role of canonical Wnt signaling. Stem Cells. 2008;26:2674–2681. doi: 10.1634/stemcells.2008-0277. [DOI] [PubMed] [Google Scholar]
- 137.Meury T, Verrier S, Alini M. Human endothelial cells inhibit BMSC differentiation into mature osteoblasts in vitro by interfering with osterix expression. J. Cell Biochem. 2006;98:992–1006. doi: 10.1002/jcb.20818. [DOI] [PubMed] [Google Scholar]
- 138.Mankin HJ. Nontraumatic necrosis of bone (osteonecrosis) N. Engl. J. Med. 1992;326:1473–1479. doi: 10.1056/NEJM199205283262206. [DOI] [PubMed] [Google Scholar]
- 139.Mafi R, Hindocha S, Mafi P, Griffin M, Khan WS. Sources of adult mesenchymal stem cells applicable for musculoskeletal applications - a systematic review of the literature. Open Orthop. J. 2011;5:242–248. doi: 10.2174/1874325001105010242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Papakostidis C, Tosounidis TH, Jones E, Giannoudis PV. The role of “cell therapy” in osteonecrosis of the femoral head. A systematic review of the literature and meta-analysis of 7 studies. Acta Orthop. 2016;87:72–78. doi: 10.3109/17453674.2015.1077418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Slobogean GP, Sprague SA, Scott T, Bhandari M. Complications following young femoral neck fractures. Injury. 2015;46:484–491. doi: 10.1016/j.injury.2014.10.010. [DOI] [PubMed] [Google Scholar]
- 142.Gangji V, Toungouz M, Hauzeur JP. Stem cell therapy for osteonecrosis of the femoral head. Expert Opin. Biol. Ther. 2005;5:437–442. doi: 10.1517/14712598.5.4.437. [DOI] [PubMed] [Google Scholar]
- 143.Zhao L, Kaye AD, Kaye AJ, Abd-Elsayed A. Stem cell therapy for osteonecrosis of the femoral head: current trends and comprehensive review. Curr. Pain. Headache Rep. 2018;22:41. doi: 10.1007/s11916-018-0700-x. [DOI] [PubMed] [Google Scholar]
- 144.Li R, et al. Stem cell therapy for treating osteonecrosis of the femoral head: From clinical applications to related basic research. Stem Cell Res. Ther. 2018;9:291. doi: 10.1186/s13287-018-1018-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Mao L, et al. Efficacy and safety of stem cell therapy for the early-stage osteonecrosis of femoral head: a systematic review and meta-analysis of randomized controlled trials. Stem Cell Res. Ther. 2020;11:445. doi: 10.1186/s13287-020-01956-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Cordova LA, et al. Severe compromise of preosteoblasts in a surgical mouse model of bisphosphonate-associated osteonecrosis of the jaw. J. Craniomaxillofac Surg. 2016;44:1387–1394. doi: 10.1016/j.jcms.2016.07.015. [DOI] [PubMed] [Google Scholar]
- 147.Deyo RA, Gray DT, Kreuter W, Mirza S, Martin BI. United States trends in lumbar fusion surgery for degenerative conditions. Spine. 2005;30:1441–1445. doi: 10.1097/01.brs.0000166503.37969.8a. [DOI] [PubMed] [Google Scholar]
- 148.Covas DT, et al. Multipotent mesenchymal stromal cells obtained from diverse human tissues share functional properties and gene-expression profile with CD146+ perivascular cells and fibroblasts. Exp. Hematol. 2008;36:642–654. doi: 10.1016/j.exphem.2007.12.015. [DOI] [PubMed] [Google Scholar]
- 149.Russell KC, et al. Cell-surface expression of neuron-glial antigen 2 (NG2) and melanoma cell adhesion molecule (CD146) in heterogeneous cultures of marrow-derived mesenchymal stem cells. Tissue Eng. Part A. 2013;19:2253–2266. doi: 10.1089/ten.tea.2012.0649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Fattal E, Kassinos SC. Editorial on the special issue “SimInhale”. Eur. J. Pharm. Sci. 2018;113:1. doi: 10.1016/j.ejps.2017.10.037. [DOI] [PubMed] [Google Scholar]
- 151.Iacobaeus E, et al. Dynamic changes in brain mesenchymal perivascular cells associate with multiple sclerosis disease duration, active inflammation, and demyelination. Stem Cells Transl. Med. 2017;6:1840–1851. doi: 10.1002/sctm.17-0028. [DOI] [PMC free article] [PubMed] [Google Scholar]