Abstract
Introduction
We analyzed a cohort of patients with cancer and Sars-Cov-2 infection from the Veneto Oncology Network registry across two pandemic time periods.
Materials and methods
761 patients with cancer and SARS-CoV-2 infection were included.
Results
198 patients were diagnosed during the first pandemic time period (TP1; February 2020 September 2020), 494 during TP2 before the vaccination campaign (TP2/pre-vaccination; September 2020-21 February 2021) and 69 in TP2/post-vaccination (22 February 2021-15 May 2021). TP2 vs TP1 patients were younger (p = 0.004), showed more frequently a good performance status (p < 0.001) and <2 comorbidities (p = 0.002), were more likely to be on active anticancer therapy (p = 0.006). Significantly fewer patients in TP2 (3-4%) vs TP1 (22%) had an in-hospital potential source of infection (p < 0.001). TP2 patients were more frequently asymptomatic (p = 0.003). Significantly fewer patients from TP2 were hospitalized (p < 0.001) or admitted to intensive care unit (p = 0.006). All-cause mortality decreased from 30.3% in TP1, to 8.9% and 8.7% in the two TP2 periods (p < 0.001), reflected by a significant reduction in Sars-Cov-2-related mortality (15.2%, 7.5% and 5.8% in the three consecutive time periods, p = 0.004).
Conclusions
Differences in clinical characteristics and features of Sars-Cov-2 infection between TP1 and TP2 reflect the effects of protective measures and increased testing capacity. The lower mortality in TP2 is in line with a less frail population. However, the vast majority of death events in TP2 were related to COVID-19, reinforcing the priority to protect cancer patients.
Keywords: Sars-Cov-2, COVID-19, Cancer
1. Introduction
More than 280 million Severe acute respiratory syndrome coronavirus 2 (Sars-Cov-2) infection cases and more than 5,400,000 deaths have been reported worldwide as of December 2021 [1]. In Italy, the number of confirmed cases is more than 5,700,000, of which more than 600,000 are in the Veneto Region [2].
During the emergency of the first outbreak, the Veneto Oncology Network (Rete Oncologica Veneta [ROV]) licensed and subsequently updated a dedicated diagnostic-therapeutic pathway (PDTA), a workflow harmonizing protective measures for oncologic patients and staff members, while guaranteeing at the same time the continuity of oncologic care, across medical oncology units operating in the Veneto Region [3]. These efforts have likely contributed to mitigating the rate of Sars-Cov-2 infection and its consequences in cancer patients. However, several registry-based and population-based studies consistently documented a 30% rate of mortality for cancer patients infected with Sars-Cov-2 during the first surge [[4], [5], [6], [7], [8], [9]] In a multivariate analysis including more than 20,000 in-hospital COVID-19 patients, malignancy as a pre-existing condition was an independent factor associated with mortality [10]. These findings identified cancer patients as a vulnerable population, at a higher risk of the severe outcome of Sars-Cov-2 infection as compared to non-cancer patients. The Rete Oncologica Veneta COVID-19 (ROVID) is a prospective registry from the Veneto Region (Italy) including cancer patients with a documented Sars-Cov-2 infection. In a previous analysis involving 170 patients (February 2020–September 2020), all-cause and Sars-Cov-2-specific mortality rates were 33% and 17% [11].
In the past year, preparedness towards the pandemic has significantly improved, including optimization of COVID-19 clinical management, increased testing capacity and contact tracing ability, implementation of protective measures including vaccination. It is now clear that aiming for ‘zero COVID-19’ is an unrealistic target in the short-time period [12,13]. The pandemic continues to strike with new waves driven by the emerging Sars-Cov-2 variants [14]. Therefore, monitoring the performance of integrated care across pandemic phases [15], as well as the epidemiology and course of infection in cancer patients, is of paramount importance to assess the effectiveness of protective actions and plan future strategies.
In this update of the ROVID study, we evaluate the epidemiology, clinical characteristics, mortality rates for cancer patients with Sars-Cov-2 infection across the first and second pandemic time periods.
2. Methods
The ROVID (ROV COVID19) study is an observational study coordinated by the ROV and involves 26 medical oncology units. All consecutive patients with cancer diagnosis and documented SARS-CoV-2 infection (positive nasopharyngeal swab). The set of variables has been previously described (10).
This updated analysis includes cancer patients diagnosed with Sars-Cov-2 from February 2020 to May 2021.
We considered the following time periods, according to the Italian National Institute of Health's analysis of the pandemic in Italy [16]: first outbreak (February 2020–May 2020), transition phase (June 2020 – mid-September 2020), second time period (from mid-September 2020). For this analysis, we considered the first outbreak together with the transition phase (first time period [TP1]). Moreover, we divided the second time period (TP2) into pre- and post-vaccination campaigns for oncologic patients (cut-off: 22 February 2021, date of the official launch of the vaccination campaign for cancer patients in the Veneto Region). It has to be acknowledged that different pandemic time periods or waves are not synchronous in all countries. This is why, for our project, we applied a definition of pandemic time periods which is consistent with the epidemiology of the pandemic in Italy [16]. The database lock date for this analysis is 31 May 2021.
Testing policies for cancer patients in the Veneto Region initially recommended a nasopharyngeal swab only in case of symptoms or close contact criteria. Policies changed in late April 2020 recommending to test in all the following cases, irrespective of symptoms or close contact criteria: prior to surgical interventions, in case of hospitalization, prior to the administration of myelosuppressive treatments requiring granulocyte colony-stimulating factors, and in patients who had haematopoietic cell transplantation. Since April 2020, testing policies remained unchanged until the date of database lock for this analysis.
To compare variables across the different time-periods we used the Χ2 test or the Kruskal–Wallis test. Outcomes of interest were: all-cause mortality and Sars-Cov-2-related mortality (as determined by the investigators) occurring at any time during follow-up. Rates of mortality were compared by using the Χ2 test. Odds ratios and 95% confidence intervals (95%CIs) were calculated by logistic regression analysis. Statistical analyses were performed by using IBM® SPSS® Statistics (version 26). Level of significance was p < 0.05. The protocol was approved by the institutional review board of participating centres. All study procedures were in accordance with the precepts of Good Clinical Practice and the Declaration of Helsinki.
3. Results
3.1. Description of TP1 and TP2
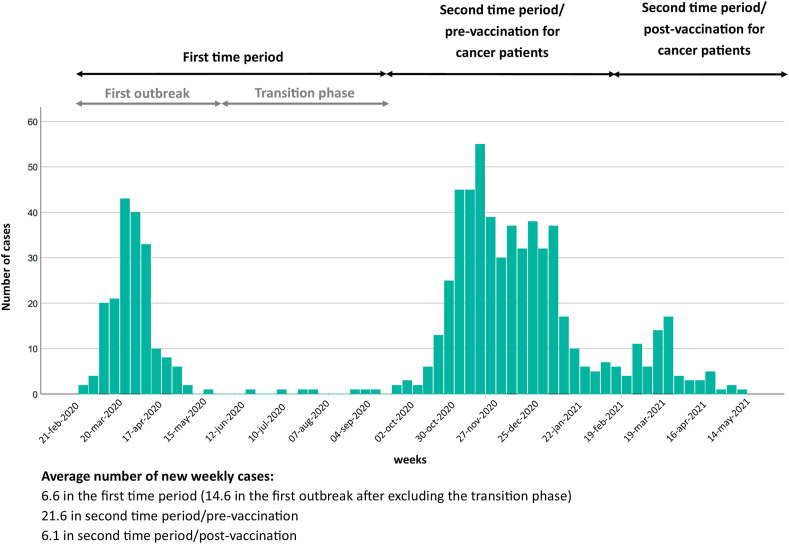
From February 2020 to May 2021, 761 patients were included: 198 from TP1 (191 in the first outbreak and 7 in the transition phase), 494 from TP2/pre-vaccination and 69from TP2/post-vaccination. Fig. 1 shows the number of cases per week. Although the date of the launch of the vaccination campaign for cancer patients in the Veneto Region is 22 February 2021, the first administrations of vaccine doses were planned in early March 2021. In our cohort, more than 76% (n = 53) of patients from TP2/post-vaccination got infected by the end of March 2021, thus likely either before receiving the first dose or in the interval between the two doses of vaccine. Full data on vaccination status are available for 26 patients from TP2/post-vaccination: 11 got infected before receiving any vaccine dose, 14 between the first and the second dose, and 1 after the 2-dose vaccination cycle.
Fig. 1.
Number of new weekly cases of cancer patients diagnosed with Sars-Cov-2 infection across centres participating to the ROVID study from February 2020 to May 2021. Relevant time periods considered in the present study are shown.
3.2. Patients’ characteristics
Patients from TP2 vs TP1 were younger, had more frequently an ECOG (Eastern Cooperative Oncology Group) Performance Status 0–1, and presented with fewer comorbidities (Table 1 ). The proportion of never smokers was lower in TP2 vs TP1 (data unavailable for 27.6% of patients Table 1).
Table 1.
Patient demographic, clinical and tumour characteristics according to pandemic time periods.
| TP1 N = 198 |
TP2/pre-vaccination n = 494 |
TP2/post-vaccination n = 69 |
Total N = 761 |
P value | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| n | % | N | % | n | % | n | % | |||
| Sex | Female | 107 | 54.0% | 260 | 52.6% | 45 | 65.2% | 412 | 54.1% | 0.743 |
| Male | 91 | 46.0% | 234 | 47.4% | 24 | 34.8% | 349 | 45.9% | ||
| Age (years), median (range) | 69 (25–95) | 66 (23–94) | 65 (31–91) | 67 (23–25) | 0.004 | |||||
| ECOG Performance Status | 0 | 63 | 36.6% | 255 | 58.9% | 26 | 46.4% | 344 | 52.0% | <0.001 |
| 1 | 61 | 35.5% | 127 | 29.3% | 21 | 37.5% | 209 | 31.6% | ||
| 2 | 27 | 15.7% | 34 | 7.9% | 6 | 10.7% | 67 | 10.1% | ||
| 3 | 15 | 8.7% | 14 | 3.2% | 3 | 5.4% | 32 | 4.8% | ||
| 4 | 6 | 3.5% | 3 | 0.7% | 0 | 0.0% | 9 | 1.4% | ||
| Cancer diagnosis | Breast | 49 | 24.9% | 127 | 25.9% | 21 | 30.4% | 197 | 26.0% | 0.240 |
| Gastrointestinal | 42 | 21.3% | 97 | 19.8% | 13 | 18.8% | 152 | 20.1% | ||
| Genitourinary | 24 | 12.2% | 76 | 15.5% | 8 | 11.6% | 108 | 14.3% | ||
| Gynecologic | 9 | 4.6% | 25 | 5.1% | 6 | 8.7% | 40 | 5.3% | ||
| Haematologic malignancy | 15 | 7.6% | 20 | 4.1% | 2 | 2.9% | 37 | 4.9% | ||
| Head and Neck | 6 | 3.0% | 7 | 1.4% | 1 | 1.4% | 14 | 1.8% | ||
| Lung | 24 | 12.2% | 61 | 12.4% | 5 | 7.2% | 90 | 11.9% | ||
| Melanoma | 10 | 5.1% | 48 | 9.8% | 6 | 8.7% | 64 | 8.5% | ||
| Other | 18 | 9.1% | 30 | 6.1% | 7 | 10.1% | 55 | 7.3% | ||
| Cancer Stage | I | 36 | 18.5% | 76 | 15.5% | 12 | 17.4% | 124 | 16.4% | 0.651 |
| II | 19 | 9.7% | 63 | 12.8% | 9 | 13.0% | 91 | 12.1% | ||
| III | 24 | 12.3% | 68 | 13.8% | 10 | 14.5% | 102 | 13.5% | ||
| IV | 116 | 59.5% | 284 | 57.8% | 38 | 55.1% | 438 | 58.0% | ||
| Documented lung metastases | No | 164 | 82.8% | 401 | 81.2% | 51 | 73.9% | 616 | 80.9% | 0.435 |
| Yes | 34 | 17.2% | 93 | 18.8% | 18 | 26.1% | 145 | 19.1% | ||
| Cancer setting | Curative | 82 | 41.4% | 207 | 42.2% | 31 | 44.9% | 320 | 42.2% | 0.872 |
| Palliative | 116 | 58.6% | 284 | 57.8% | 38 | 55.1% | 438 | 57.8% | ||
| Smoking habit | Never smoker | 105 | 77.2% | 273 | 74.4% | 27 | 56.3% | 405 | 73.5% | <0.001 |
| Current smoker | 31 | 22.8% | 47 | 12.8% | 9 | 18.8% | 87 | 15.8% | ||
| Former smoker | 0 | 0.0% | 47 | 12.8% | 12 | 25.0% | 59 | 10.7% | ||
| Selected comorbidities | Hypertension | 86 | 43.4% | 207 | 41.9% | 32 | 46.4% | 325 | 42.7% | 0.821 |
| Cardiac comorbidites | 57 | 28.8% | 75 | 15.2% | 10 | 14.5% | 142 | 18.7% | <0.001 | |
| Pulmonary comorbidites | 17 | 8.6% | 33 | 6.7% | 8 | 11.6% | 58 | 7.6% | 0.228 | |
| Autoimmune disorders | 12 | 6.1% | 12 | 2.4% | 0 | 0.0% | 24 | 3.2% | 0.010 | |
| Diabetes | 33 | 16.7% | 81 | 16.4% | 8 | 11.6% | 122 | 16.0% | 0.894 | |
| Chronic renal failure | 13 | 6.6% | 22 | 4.5% | 0 | 0.0% | 35 | 4.6% | 0.271 | |
| Presence of 1 or more comorbidities | No | 39 | 19.7% | 155 | 31.4% | 19 | 27.5% | 213 | 28.0% | 0.008 |
| Yes | 159 | 80.3% | 339 | 68.6% | 50 | 72.5% | 548 | 72.0% | ||
| Presence of 2 or more comorbidities | No | 97 | 49.0% | 308 | 62.3% | 39 | 56.5% | 444 | 58.3% | 0.002 |
| Yes | 101 | 51.0% | 186 | 37.7% | 30 | 43.5% | 317 | 41.7% | ||
| Obesity | No | 171 | 86.4% | 428 | 86.6% | 57 | 82.6% | 656 | 86.2% | 0.784 |
| Yes | 27 | 13.6% | 66 | 13.4% | 12 | 17.4% | 105 | 13.8% | ||
| Active anticancer therapy (any) | No | 92 | 46.5% | 167 | 33.9% | 23 | 33.3% | 282 | 37.1% | 0.006 |
| Yes | 106 | 53.5% | 326 | 66.1% | 46 | 66.7% | 478 | 62.9% | ||
| Active anticancer therapy (type) | Chemotherapy | 57 | 28.8% | 156 | 31.6% | 24 | 34.8% | 237 | 31.1% | 0.698 |
| Immunotherapy | 11 | 5.6% | 58 | 11.7% | 10 | 14.5% | 79 | 10.4% | 0.028 | |
| Targeted therapy | 24 | 12.1% | 89 | 18.0% | 6 | 8.7% | 119 | 15.6% | 0.067 | |
| Endocrine therapy | 24 | 12.1% | 65 | 13.2% | 10 | 14.5% | 99 | 13.0% | 0.938 | |
Abbreviations: TP, pandemic time period.
The proportion of patients on active therapy at the time of Sars-Cov-2 infection was higher in TP2, mainly driven by a higher rate of patients receiving immunotherapy (Table 1). Among 478 patients on anticancer treatment, 71.3% discontinued therapy because of Sars-Cov-2 infection, with similar proportions in the three time periods (68.6% in TP1, 72.7% in TP2/pre-vaccination and 74.5% in TP2/post-vaccination; p = 0.662). Anticancer therapy was resumed for 63.2% of patients (40.8% in TP1, 71.7% in TP2/pre-vaccination and 51.5% in TP2/post-vaccination, p < 0.001).
3.3. Sars-Cov-2 infection
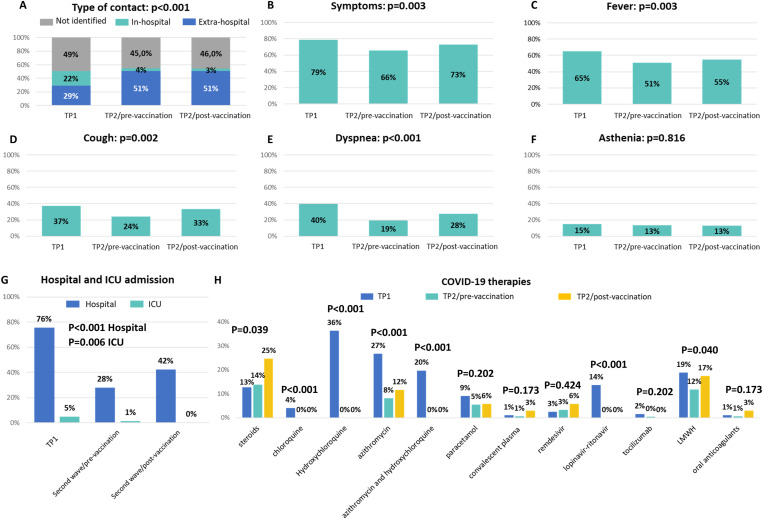
The potential source of Sars-Cov-2 infection was identified as in-hospital contact in 8.8% of the cases, with a significant decrease from TP1 (21.7%) to TP2 (4.5% pre-vaccination and 2.9% post-vaccination; p < 0.001, Fig. 2 A).
Fig. 2.
Characteristics of Sars-Cov-2 infection across pandemic time periods: type of contact (A), symptoms (B–F), hospitalization and ICU admission (G), and COVID-19 therapies. ICU: intensive care unit.
At the time of Sars-Cov-2 infection, 69.7% patients presented with COVID-19 symptoms, with a significantly higher proportion in TP1 (78.6%, as compared to 65.6% in TP2/pre-vaccination and 73.1% in TP2/post-vaccination, p < 0.001, Fig. 2B). Fever, cough and dyspnea were significantly more frequent among patients from TP1 (Fig. 2C–F).
More than 41% of patients were hospitalized and 2.2% were admitted to intensive care units (ICU), with a significant decline from TP1 to TP2 (Fig. 2G). Median duration of hospitalization was 13 days (95%CI 12–15), with a trend towards a reduced length from TP1 (median 14 days, 95%CI 12–17) to TP2 (median 12 days, 95%CI 11–15 pre-vaccination and median 11 days, 95%CI 6–17 post-vaccination; p = 0.085). The pattern of COVID-19 treatments significantly changed over time, with increasing use of steroids and a decline in the administration of antivirals, anti-malarials and azithromycin (Fig. 2H). The use of low molecular weight heparin was also significantly different in the three time periods, although potentially biased by administration for reasons other than COVID-19.
Overall, 613 (80.6%) patients had documented resolution of Sars-Cov-2 infection (67.2% in TP1, 86.4% in TP2/pre-vaccination, 76.8% in TP2/post-vaccination, P < 0.001). Median time to negative swab was 20 days (95%CI 19–21).
3.4. Mortality rates across time periods
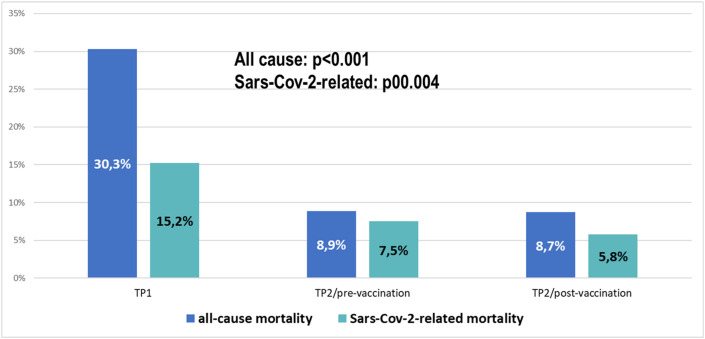
Overall, 110 patients had died (14.5%), being the cause of death directly correlated with Sars-Cov-2 infection in 71 cases (9.3% of the total study population, 64.5% of death events). Other causes of death were: cancer progression (n = 23) and other/unknown (n = 16Mortality rate significantly decreased over time: 30.3% in TP1, 8.9% in TP2/pre-vaccination and 8.7% in TP2/post-vaccination (P < 0.001; Fig. 3 ). Similarly, the rate of Sars-Cov-2-related mortality was significantly reduced: 15.2% in TP1, 7.5% in TP2/pre-vaccination and 5.8% in TP2/post-vaccination (P = 0.004; Fig. 3). Looking at the same data from a different perspective, we observed that in TP2, the proportion of patients dying from Sars-Cov-2 infection over the total deaths was significantly higher: 84.1% in TP2/pre-vaccination (n = 37 out of 44 death events), 66.7% in TP2/post-vaccination (n = 4 out of 6 death events) vs 50% in TP1 (n = 30 out of 60 death events, P = 0.002). In summary, the risk of dying from any cause and the risk of dying from Sars-Cov-2 infection were significantly lower in TP2 vs TP1. Nevertheless, among patients with a death event, the probability of dying for Sars-Cov-2 infection was higher in TP2. This observation accounts for the different clinical conditions, burden of comorbidity and thus, potential competing causes of death, of TP1 vs TP2 patients.
Fig. 3.
Mortality rates across the three pandemic time periods.
3.5. Factors associated with Sars-Cov-2 infection-related mortality
We evaluated factors potentially associated with Sars-Cov-2 infection-related mortality. For the purpose of this analysis, we considered TP2/pre-vaccination and TP2/post-vaccination in one single time period.
Sars-Cov-2 diagnosis in TP1, male gender, increased age, ECOG PS ≥ 2, >2 pre-existing comorbidities, stage IV cancer, palliative setting, haematologic or lung malignancy, COVID-19 symptoms at diagnosis, in-hospital contagion, hospitalization and ICU admission, all resulted associated in univariate analysis with the risk of Sars-Cov-2-related mortality (Table 2 ). At multivariate analysis, increased age, stage IV cancer, hospitalization and ICU admission were independent factors; when hospitalization and ICU admissions were excluded to include in the multivariable model only conditions that were present at the time of Sars-Cov-2 diagnosis, COVID-19 symptoms emerged as an independent factor (Table 2).
Table 2.
Univariate and multivariate logistic regression analysis evaluating the association of patient-associated, tumour-associated and infection-associated factors with Sars-Cov-2-related mortality.
|
Factors |
Univariate |
Multivariatea |
Multivariateb |
|||
|---|---|---|---|---|---|---|
| OR (95%CI) | P | OR (95%CI) | P | OR (95%CI) | P | |
| Time period (first vs second) | 2.27 (1.37–3.41) | <0.001 | 0.75 (0.39–1.46) | 0.398 | 1.61 (0.86–3.01) | 0.139 |
| Sex (male vs female) | 1.81 (1.10–2.97) | 0.019 | 1.02 (0.52–2.00) | 0.955 | 1.38 (0.75–2.54) | 0.309 |
| Age (continuous) | 1.06 (1.03–1.08) | <0.001 | 1.05 (1.02–1.09) | 0.005 | 1.06 (1.02–1.09) | 0.001 |
| ECOG PS (≥2 vs 0–1) | 2.41 (1.30–4.48) | 0.005 | 0.84 (0.39–1.80) | 0.653 | 1.30 (0.62–2.72) | 0.483 |
| Smoking habit (current/vs former or never) | 0.90 (0.40–2.06) | 0.799 | ||||
| Comorbidity (≥2 vs < 2) | 1.93 (1.18–3.15) | 0.009 | 1.01 (0.52–1.97) | 0.977 | 1.12 (0.61–2.07) | 0.711 |
| Obesity (yes vs no) | 1.32 (0.68–2.55) | 0.412 | ||||
| Cancer stage (IV vs I-III) | 2.78 (1.54–5.03) | 0.001 | 29.98 (1.49–604.46) | 0.027 | 21.04 (1.31–338.65) | 0.032 |
| Cancer setting (palliative vs curative) | 2.31 (1.32–4.02) | 0.003 | 0.12 (0.01–2.25) | 0.157 | 0.13 (0.10–1.96) | 0.140 |
| Cancer type (haematologic or lung vs others) | 3.42 (2.01–5.80) | <0.001 | 2.42 (1.19–4.92) | 0.015 | 2.41 (1.26–4.59) | 0.008 |
| Active anticancer therapy (yes vs no) | 1.26 (0.75–2.13) | 0.379 | ||||
| Chemotherapy ongoing (yes vs no) | 1.23 (0.74–2.05) | 0.432 | ||||
| Immunotherapy ongoing (yes vs no) | 0.78 (0.33–1.86) | 0.571 | ||||
| COVID-19 symptoms (presence vs absence) | 7.91 (2.85–22.0) | <0.001 | 1.55 (0.50–4.84) | 0.451 | 5.02 (1.75–14.38) | 0.003 |
| Source of infection: In-hospital vs Extra-hospitalc | 2.52 (1.09–5.81) | 0.030 | ||||
| Hospitalization (yes vs no) | 61.43 (14.9–352.7) | <0.001 | 68.27 (8.95–520.52) | <0.001 | ||
| ICU admission | 20.84 (7.45–58.3) | <0.001 | 9.61 (2.44–37.79) | 0.001 | ||
OR, odds ratio; CI, confidence interval; p, p value; ECOG, Eastern cooperative Oncology Group; PS, performance status; ICU, intensive care unit.
Multivariate analysis including those factors significantly associated with outcome in univariate analysis.
Multivariate analysis including those factors significantly associated with outcome in univariate analysis, after exclusion of hospital and ICU admission.
Not included in multivariate analysis due to high number of missing cases (contact not identified).
4. Discussion
We reported on Sars-Cov-2 infection in cancer patients across the first two pandemic time periods from a prospective database reflecting a homogeneous healthcare context.
Consistently with the pattern in the general population, we observed a larger diffusion of cases in TP2 vs TP1, coherently with the emergence of variants of concern driving the second surge in Europe, like the B.1.1.7 lineage [17,18]. Moreover, testing capacity was improved from TP1 to TP2, as described in the ‘methods’ section, potentially allowing the diagnosis of more asymptomatic cases.
The main result of our study is a three-fold decrease in all-cause mortality rates from TP1 to TP2, reflecting also a halving of Sars-Cov-2-related mortality. The increased testing capacity, the adoption of new testing policies for cancer patients and the implementation of measures to protect patients and the clinical staff explain the differences in characteristics of patients and Sars-Cov-2 infection observed in TP2 vs TP1 that may have driven a reduced mortality risk.
Patients from TP2 were younger, in better clinical conditions, and with fewer comorbidities. All these factors are known to be associated with all-cause mortality [4,6,19]. These features also determined a reduction in Sars-Cov-2-related mortality in our study, as they resulted significantly associated with this outcome. The proportion of patients receiving anticancer therapy at the time of Sars-Cov-2 infection was also higher in TP2, reflecting better clinical conditions allowing the administration of antineoplastic drugs as well as the efforts put in place to ensure the continuity of oncologic care.
Another key result is the dramatic decrease in in-hospital transmission as the source of infection, from 22% in TP1 to 3–4% in TP2, highlighting the efficacy of measures to protect the vulnerable population of hospitalized cancer patients, including restriction of access to visitors, testing for asymptomatic patients, as well as screening tests and vaccinations for healthcare providers. As compared to community transmission, patients with an identified in-hospital source of infection had a more than two-fold increase in the risk of Sars-Cov-2-related mortality, stressing the importance to prevent Sars-Cov-2 circulation within inpatient clinics. This is consistent with data from the OnCovid registry showing that pre-existing hospitalization is an independent poor prognostic factor [20].
Consistently with the increased testing capacity, TP2 patients were less frequently symptomatic. Accordingly, we also observed in TP2 a reduced rate of complicated/severe COVID19, by considering the rate of hospitalizations and ICU admissions as a proxy.
The pattern of anti-COVID-19 therapies changed over time, in line with the contemporary evidence [21]. The optimization of COVID-19 management might have contributed to improve clinical outcomes, although we were not able to formally assess its impact.
Several factors resulted associated with Sars-Cov-2-related mortality in our cohort by univariate analysis. Given the strict relation among them, only a few were independent predictors in multivariate analysis. We advocate for a comprehensive assessment of cancer patients diagnosed with Sars-Cov-2 infection in order to identify those at higher risk of severe illness deserving adequate clinical strategies to limit this risk. Importantly, we observed a lack of association of active anticancer therapy with mortality. This is a debated issue, since data from other registries are controversial [6,19,20,[22], [23], [24], [25]]. Based on our results, we support the opportunity of maintaining active anticancer treatments during the pandemic, provided the implementation of appropriate measures to prevent Sars-Cov-2 transmission and a risk-stratification approach.
Few other studies evaluated the evolution of Sars-Cov-2 infection in cancer patients beyond the first outbreak [19,20,22,26]. Comparison with these studies is limited by differences in healthcare contexts, time periods, definitions of pandemic time frames, patients’ selection and definition of outcome. Indeed, other studies did not present data on Sars-Cov-2-related mortality but reported either all-cause-related mortality or early all-cause-related mortality. The largest study evaluated around 200,000 patients included in a prospective UK registry until August 2021, of which about 20,000 had either a past history or current diagnosis of malignancy at the time of Sars-Cov-2 infection [26]. The authors did not observe a reduction in mortality rates for cancer patients despite a clear trend in lower mortality across time for non-cancer patients [26]. This study was focused on hospitalized COVID-19 patients, thus possibly skewed to a more critical population. Conversely, a significantly lower mortality rate across time in Sars-Cov-2-infected cancer patients was reported by other studies [19,20,22]. The OnCovid cohort demonstrated a significant reduction in 14-day mortality throughout 5 pre-specified time periods, from 29.8% in February–March 2020 to 14.5% in January–February 2021 [20]. Similar to our findings, this work described a higher prevalence of younger patients and with fewer comorbidities in the second outbreak, as well as a lower rate of hospitalization required for COVID-19 [20]. Differently to our study, there was a trend towards a higher rate of patients with pre-existing hospitalization at the time of Sars-Cov-2 infection diagnosis (27% in the second outbreak vs 22% in the first outbreak)19. The authors concluded that the implementation of widespread testing for Sars-Cov-2 is likely a major factor explaining the reduction in mortality. We agree that diffuse testing is key to more accurately unravel the effects of Sars-Cov-2 infection in cancer patients and to adequately maintain COVID-19-free pathways. We also believe that the reduction in mortality derives from the application of broader preventive measures, including those aimed at reducing in-hospital transmission that were homogeneously applied in the Veneto Region.
Our study has limitations. First, we were not able to capture the effects of a 2-dose full vaccination cycle. Further updates of the ROVID s will provide evidence in this regard. Second, data on sequencing of Sars-Cov-2 variants is not available.
A major strength of our study is the homogeneous healthcare context. This aspect is not trivial. Indeed, heterogeneity in the Italian territory has been documented, at least in terms of testing policies for cancer patients [27]. Moreover, differences in patients’ outcome have also been reported in patients from Europe vs United Kingdom [28]. The availability of data on Sars-Cov-2-related mortality as a pre-specified outcome collected in the database, thus not indirectly inferred.
In conclusion, we demonstrated that under the adoption of common guidelines and policies, the mortality of cancer patients with Sars-Cov-2 infection dramatically declined over time. Nevertheless, the vast majority of death events occurring in Sars-Cov-2 infected cancer patients in TP2 were related to COVID-19. Therefore, the protection of cancer patients from Sars-Cov-2 infection remains a priority, including vaccination. As of the time of this report, we are facing a new rapid spread of the contagion sustained by the Omicron variant, and thus, it is fundamental to acknowledge the evolutionary nature of the pandemic and be prepared for flexibility.
Conflict of interest statement
MVD reports personal fees from EliLilly, Exact Sciences, Novartis, Pfizer, Seagen, MSD, Gilead, outside the submitted work. G. Aprile reports personal fees from MSD, BMS, Eli Lilly, Amgen, Ipsen, Servier, outside of the work. MM reports personal fees from BMS, Pfizer, Merck Serono, Astellas, Janssen, MSD outside the submitted work. VG reports personal fees from EliLilly, Novartis, Roche, MSD, Gilead, GSK, outside the submitted work. EM reports personal fees from EliLilly, Novartis, Istituto gentili, outside the submitted work. VCS report personal fees from BMS, Novartis, Pierre-Fabre, Merck-Serono, outside the submitted work.
PFC reports personal fees from Daiichi Sankyo, Elililly, Roche/Genentech, Novartis, AstraZeneca, Tesaro, BMS, Celgene, Pfizer, and research funding (Institution) from Roche, BMS; Merck KGaA, all outside the submitted work. VZ reports personal fees from Bristol-Myers Squibb, MSD, Eisai, Italfarmaco, Roche, Astellas Pharma, Servier, AstraZeneca, Janssen, Ipsen, and research funding (Institution) from Bayer, Roche, Lilly, AstraZeneca, Bristol-Myers Squibb, Ipsen, Astellas Pharma, all outside the submitted work. SL reports personal fees from Amgen, Merck Serono, Lilly, Astra Zeneca, Incyte, Daiichi-Sankyo, Bristol-Myers Squibb, Servier, MSD, Roche, Servier, Merck Serono, Pierre-Fabre, GSK and research funding (Institution) from Amgen, Merck Serono, Bayer, Roche, Lilly. Astra Zeneca, Bristol-Myers Squibb, all outside the submitted work. All remaining authors have declared no conflicts of interest.
Author contribution statement
MVD: Conceptualization; Data curation; Formal analysis; Writing - original draft.
VG: Conceptualization; Funding acquisition; Supervision; Writing - original draft.
PFC: Conceptualization; Supervision; Writing - review & editing.
MC: Data curation; Methodology; Writing - review & editing.
All authors: Data curation; Writing - review & editing.
Financial support
The study was supported by a grant from Fondazione Cariparo, ID 55721 (2020.0146), to Prof. Valentina Guarneri.
References
- 1.https://covid19.who.int/
- 2.https://opendatadpc.maps.arcgis.com/apps/dashboards/b0c68bce2cce478eaac82fe38d4138b1
- 3.https://salute.regione.veneto.it/c/document_library/get_file?uuid=ce0e02aa-b449-49c6-8b37-018819e16532&groupId=534936
- 4.Garassino M.C., Whisenant J.G., Huang L.C., Trama A., Torri V., Agustoni F., Baena J., Banna G., Berardi R., Bettini A.C., Bria E., Brighenti M., Cadranel J., De Toma A., Chini C., Cortellini A., Felip E., Finocchiaro G., Garrido P., Genova C., Giusti R., Gregorc V., Grossi F., Grosso F., Intagliata S., La Verde N., Liu S.V., Mazieres J., Mercadante E., Michielin O., Minuti G., Moro-Sibilot D., Pasello G., Passaro A., Scotti V., Solli P., Stroppa E., Tiseo M., Viscardi G., Voltolini L., Wu Y.L., Zai S., Pancaldi V., Dingemans A.M., Van Meerbeeck J., Barlesi F., Wakelee H., Peters S., Horn L. TERAVOLT investigators. COVID-19 in patients with thoracic malignancies (TERAVOLT): first results of an international, registry-based, cohort study. Lancet Oncol. 2020 Jul;21(7):914–922. doi: 10.1016/S1470-2045(20)30314-4. Epub 2020 Jun 12. PMID: 32539942; PMCID: PMC7292610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.de Azambuja E., Brandão M., Wildiers H., Laenen A., Aspeslagh S., Fontaine C., Collignon J., Lybaert W., Verheezen J., Rutten A., Vuylsteke P., Goeminne J.C., Demey W., Van Beckhoven D., Deblonde J., Rottey S., Geukens T., Punie K., Bafort K., Belkhir L., Bossuyt N., Colombie V., Daubresse C., Dauby N., De Munter P., Delmarcelle D., Delvallee M., Demeester R., Delefortrie Q., Dugernier T., Holemans X., Louviaux I., Machurot P., Minette P., Mokrane S., Nachtergal C., Noirhomme S., Piérard D., Rossi C., Schirvel C., Sermijn E., Staelens F., Triest F., Van Beckhoven D., Van Goethem N., Van Praet J., Vanhoenacker A., Verstraete R., Willems E., Wyndham-Thomas C. Impact of solid cancer on in-hospital mortality overall and among different subgroups of patients with COVID-19: a nationwide, population-based analysis. ESMO Open. 2020 Sep;5(5) doi: 10.1136/esmoopen-2020-000947. PMID: 32978251; PMCID: PMC7520811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pinato D.J., Zambelli A., Aguilar-Company J., Bower M., Sng C., Salazar R., Bertuzzi A., Brunet J., Mesia R., Segui E., Biello F., Generali D., Grisanti S., Rizzo G., Libertini M., Maconi A., Harbeck N., Vincenzi B., Bertulli R., Ottaviani D., Carbo A., Bruna R., Benafif S., Marrari A., Wuerstlein R., Carmona-Garcia M.C., Chopra N., Tondini C., Mirallas O., Tovazzi V., Betti M., Provenzano S., Fotia V., Cruz C.A., Dalla Pria A., D'Avanzo F., Evans J.S., Saoudi-Gonzalez N., Felip E., Galazi M., Garcia-Fructuoso I., Lee A.J.X., Newsom-Davis T., Patriarca A., Garcia-Illescas D., Reyes R., Dileo P., Sharkey R., Wong Y.N.S., Ferrante D., Marco-Hernandez J., Sureda A., Maluquer C., Ruiz-Camps I., Gaidano G., Rimassa L., Chiudinelli L., Izuzquiza M., Cabirta A., Franchi M., Santoro A., Prat A., Tabernero J., Gennari A. Clinical portrait of the SARS-CoV-2 epidemic in European cancer patients. Cancer Discov. 2020 Jul 31;10(10):1465–1474. doi: 10.1158/2159-8290.CD-20-0773. Epub ahead of print. PMID: 32737082; PMCID: PMC7668225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Desai A., Gupta R., Advani S., Ouellette L., Kuderer N.M., Lyman G.H., Li A. Mortality in hospitalized patients with cancer and coronavirus disease 2019: a systematic review and meta-analysis of cohort studies. Cancer. 2021 May 1;127(9):1459–1468. doi: 10.1002/cncr.33386. Epub 2020 Dec 30. PMID: 33378122. [DOI] [PubMed] [Google Scholar]
- 8.de Joode K., Tol J., Hamberg P., Cloos M., Kastelijn E.A., Borgers J.S.W., Nuij V.J.A.A., Klaver Y., Herder G.J.M., Mutsaers P.G.N.J., Dumoulin D.W., Oomen-de Hoop E., van Diemen N.G.J., Libourel E.J., Geraedts E.J., Bootsma G.P., van der Leest C.H., Peerdeman A.L., Herbschleb K.H., Visser O.J., Bloemendal H.J., van Laarhoven H.W.M., de Vries E.G.E., Hendriks L.E.L., Beerepoot L.V., Westgeest H.M., van den Berkmortel F.W.P.J., Haanen J.B.A.G., Dingemans A.C., van der Veldt A.A.M., investigators D.O.C.C. Life-prolonging treatment restrictions and outcomes in patients with cancer and COVID-19: an update from the Dutch Oncology COVID-19 Consortium. Eur J Cancer. 2022 Jan;160:261–272. doi: 10.1016/j.ejca.2021.10.009. Epub 2021 Oct 25. PMID: 34799210; PMCID: PMC8542445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Saini K.S., Tagliamento M., Lambertini M., McNally R., Romano M., Leone M., Curigliano G., de Azambuja E. Mortality in patients with cancer and coronavirus disease 2019: a systematic review and pooled analysis of 52 studies. Eur J Cancer. 2020 Nov;139:43–50. doi: 10.1016/j.ejca.2020.08.011. Epub 2020 Sep 2. PMID: 32971510; PMCID: PMC7467090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Docherty A.B., Harrison E.M., Green C.A., Hardwick H.E., Pius R., Norman L., Holden K.A., Read J.M., Dondelinger F., Carson G., Merson L., Lee J., Plotkin D., Sigfrid L., Halpin S., Jackson C., Gamble C., Horby P.W., Nguyen-Van-Tam J.S., Ho A., Russell C.D., Dunning J., Openshaw P.J., Baillie J.K., Semple M.G. ISARIC4C investigators. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ. 2020 May 22;369:m1985. doi: 10.1136/bmj.m1985. PMID: 32444460; PMCID: PMC7243036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Guarneri V., Bassan F., Zagonel V., Milella M., Zaninelli M., Cattelan A.M., Vianello A., Gori S., Aprile G., Azzarello G., Chiari R., Favaretto A., Oliani C., Scola A., Pastorelli D., Mandarà M., Zustovich F., Bernardi D., Chiarion-Sileni V., Morandi P., Toso S., Di Liso E., Ziampiri S., Caccese M., Zampiva I., Puccetti O., Celestino M., Dieci M.V., Conte P., Veneto Oncology Network Epidemiology and clinical course of severe acute respiratory syndrome coronavirus 2 infection in cancer patients in the Veneto Oncology Network: the Rete Oncologica Veneta covID19 study. Eur J Cancer. 2021 Apr;147:120–127. doi: 10.1016/j.ejca.2021.01.021. Epub 2021 Feb 3. PMID: 33647547; PMCID: PMC7857033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Telenti A., Arvin A., Corey L., Corti D., Diamond M.S., García-Sastre A., Garry R.F., Holmes E.C., Pang P.S., Virgin H.W. After the pandemic: perspectives on the future trajectory of COVID-19. Nature. 2021 Aug;596(7873):495–504. doi: 10.1038/s41586-021-03792-w. Epub 2021 Jul 8. PMID: 34237771. [DOI] [PubMed] [Google Scholar]
- 13.Phillips N. The coronavirus is here to stay - here's what that means. Nature. 2021 Feb;590(7846):382–384. doi: 10.1038/d41586-021-00396-2. PMID: 33594289. [DOI] [PubMed] [Google Scholar]
- 14.Dyson L., Hill E.M., Moore S., Curran-Sebastian J., Tildesley M.J., Lythgoe K.A., House T., Pellis L., Keeling M.J. Possible future waves of SARS-CoV-2 infection generated by variants of concern with a range of characteristics. Nat Commun. 2021 Sep 30;12(1):5730. doi: 10.1038/s41467-021-25915-7. PMID: 34593807; PMCID: PMC8484271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pasello G., Menis J., Pilotto S., Frega S., Belluomini L., Pezzuto F., Caliò A., Sepulcri M., Cernusco N.L.V., Schiavon M., Infante M.V., Damin M., Micheletto C., Del Bianco P., Giovannetti R., Bonanno L., Fantoni U., Guarneri V., Calabrese F., Rea F., Milella M., Conte P. How the COVID-19 pandemic impacted on integrated care pathways for lung cancer: the parallel experience of a COVID-spared and a COVID-dedicated center. Front Oncol. 2021 Jun 28;11:669786. doi: 10.3389/fonc.2021.669786. PMID: 34262861; PMCID: PMC8273534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.https://www.iss.it/documents/20126/0/Report_ISS_Istat_2021_10_giugno+%281%29.pdf/3df35cd3-edd5-56e1-5867-d274f5032fb5?t=1623336923282
- 17.https://www.ecdc.europa.eu/sites/default/files/documents/COVID-19-risk-related-to-spread-of-new-SARS-CoV-2-variants-EU-EEA-first-update.pdf
- 18.https://www.epicentro.iss.it/coronavirus/pdf/sars-cov-2-monitoraggio-varianti-indagini-rapide-20-aprile-2021.pdf
- 19.Grivas P., Khaki A.R., Wise-Draper T.M., French B., Hennessy C., Hsu C.Y., Shyr Y., Li X., Choueiri T.K., Painter C.A., Peters S., Rini B.I., Thompson M.A., Mishra S., Rivera D.R., Acoba J.D., Abidi M.Z., Bakouny Z., Bashir B., Bekaii-Saab T., Berg S., Bernicker E.H., Bilen M.A., Bindal P., Bishnoi R., Bouganim N., Bowles D.W., Cabal A., Caimi P.F., Chism D.D., Crowell J., Curran C., Desai A., Dixon B., Doroshow D.B., Durbin E.B., Elkrief A., Farmakiotis D., Fazio A., Fecher L.A., Flora D.B., Friese C.R., Fu J., Gadgeel S.M., Galsky M.D., Gill D.M., Glover M.J., Goyal S., Grover P., Gulati S., Gupta S., Halabi S., Halfdanarson T.R., Halmos B., Hausrath D.J., Hawley J.E., Hsu E., Huynh-Le M., Hwang C., Jani C., Jayaraj A., Johnson D.B., Kasi A., Khan H., Koshkin V.S., Kuderer N.M., Kwon D.H., Lammers P.E., Li A., Loaiza-Bonilla A., Low C.A., Lustberg M.B., Lyman G.H., McKay R.R., McNair C., Menon H., Mesa R.A., Mico V., Mundt D., Nagaraj G., Nakasone E.S., Nakayama J., Nizam A., Nock N.L., Park C., Patel J.M., Patel K.G., Peddi P., Pennell N.A., Piper-Vallillo A.J., Puc M., Ravindranathan D., Reeves M.E., Reuben D.Y., Rosenstein L., Rosovsky R.P., Rubinstein S.M., Salazar M., Schmidt A.L., Schwartz G.K., Shah M.R., Shah S.A., Shah C., Shaya J.A., Singh S.R.K., Smits M., Stockerl-Goldstein K.E., Stover D.G., Streckfuss M., Subbiah S., Tachiki L., Tadesse E., Thakkar A., Tucker M.D., Verma A.K., Vinh D.C., Weiss M., Wu J.T., Wulff-Burchfield E., Xie Z., Yu P.P., Zhang T., Zhou A.Y., Zhu H., Zubiri L., Shah D.P., Warner J.L., Lopes G. Association of clinical factors and recent anticancer therapy with COVID-19 severity among patients with cancer: a report from the COVID-19 and Cancer Consortium. Ann Oncol. 2021 Jun;32(6):787–800. doi: 10.1016/j.annonc.2021.02.024. Epub 2021 Mar 19. PMID: 33746047; PMCID: PMC7972830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.OnCovid Study Group. Pinato D.J., Patel M., Scotti L., Colomba E., Dolly S., Loizidou A., Chester J., Mukherjee U., Zambelli A., Dalla Pria A., Aguilar-Company J., Bower M., Salazar R., Bertuzzi A., Brunet J., Lambertini M., Tagliamento M., Pous A., Sita-Lumsden A., Srikandarajah K., Colomba J., Pommeret F., Seguí E., Generali D., Grisanti S., Pedrazzoli P., Rizzo G., Libertini M., Moss C., Evans J.S., Russell B., Harbeck N., Vincenzi B., Biello F., Bertulli R., Ottaviani D., Liñan R., Rossi S., Carmona-García M.C., Tondini C., Fox L., Baggi A., Fotia V., Parisi A., Porzio G., Queirolo P., Cruz C.A., Saoudi-Gonzalez N., Felip E., Roqué Lloveras A., Newsom-Davis T., Sharkey R., Roldán E., Reyes R., Zoratto F., Earnshaw I., Ferrante D., Marco-Hernández J., Ruiz-Camps I., Gaidano G., Patriarca A., Bruna R., Sureda A., Martinez-Vila C., Sanchez de Torre A., Berardi R., Giusti R., Mazzoni F., Guida A., Rimassa L., Chiudinelli L., Franchi M., Krengli M., Santoro A., Prat A., Tabernero J., Van Hemelrijck M., Diamantis N., Gennari A., Cortellini A. Time-dependent COVID-19 mortality in patients with cancer: an updated analysis of the OnCovid registry. JAMA Oncol. 2021 Nov 24 doi: 10.1001/jamaoncol.2021.6199. Epub ahead of print. PMID: 34817562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Agarwal A., Rochwerg B., Siemieniuk R.A., Agoritsas T., Lamontagne F., Askie L., Lytvyn L., Leo Y.S., Macdonald H., Zeng L., Amin W., Burhan E., Bausch F.J., Calfee C.S., Cecconi M., Chanda D., Du B., Geduld H., Gee P., Harley N., Hashimi M., Hunt B., Kabra S.K., Kanda S., Kim Y.J., Kissoon N., Kwizera A., Mahaka I., Manai H., Mino G., Nsutebu E., Preller J., Pshenichnaya N., Qadir N., Sabzwari S., Sarin R., Shankar-Hari M., Sharland M., Shen Y., Ranganathan S.S., Souza J.P., Stegemann M., De Sutter A., Ugarte S., Venkatapuram S., Dat V.Q., Vuyiseka D., Wijewickrama A., Maguire B., Zeraatkar D., Bartoszko J.J., Ge L., Brignardello-Petersen R., Owen A., Guyatt G., Diaz J., Kawano-Dourado L., Jacobs M., Vandvik P.O. A living WHO guideline on drugs for covid-19. BMJ. 2020 Sep 4;370:m3379. doi: 10.1136/bmj.m3379. Update in: BMJ. 2020 Nov 19;371:m4475. Update in: BMJ. 2021 Mar 31;372:n860. Update in: BMJ. 2021 Jul 6;374:n1703. Update in: BMJ. 2021 Sep 23;374:n2219. PMID: 32887691. [DOI] [PubMed] [Google Scholar]
- 22.Wong Y.N.S., Sng C.C.T., Ottaviani D., Patel G., Chowdhury A., Earnshaw I., Sinclair A., Merry E., Wu A., Galazi M., Benafif S., Soosaipillai G., Chopra N., Roylance R., Shaw H., Lee A.J.X. Systemic anti-cancer therapy and metastatic cancer are independent mortality risk factors during two UK waves of the COVID-19 pandemic at university college london hospital. Cancers. 2021 Dec 2;13(23):6085. doi: 10.3390/cancers13236085. PMID: 34885194; PMCID: PMC8657102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Poortmans P.M., Guarneri V., Cardoso M.J. Cancer and COVID-19: what do we really know? Lancet. 2020 Jun 20;395(10241):1884–1885. doi: 10.1016/S0140-6736(20)31240-X. Epub 2020 May 29. PMID: 32479827; PMCID: PMC7259910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yekedüz E., Utkan G., Ürün Y. A systematic review and meta-analysis: the effect of active cancer treatment on severity of COVID-19. Eur J Cancer. 2020 Dec;141:92–104. doi: 10.1016/j.ejca.2020.09.028. Epub 2020 Oct 6. PMID: 33130550; PMCID: PMC7538140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lièvre A., Turpin A., Ray-Coquard I., Le Malicot K., Thariat J., Ahle G., Neuzillet C., Paoletti X., Bouché O., Aldabbagh K., Michel P., Debieuvre D., Canellas A., Wislez M., Laurent L., Mabro M., Colle R., Hardy-Bessard A.C., Mansi L., Colomba E., Bourhis J., Gorphe P., Pointreau Y., Idbaih A., Ursu R., Di Stefano A.L., Zalcman G., Aparicio T. GCO-002 CACOVID-19 collaborators/investigators. Risk factors for Coronavirus Disease 2019 (COVID-19) severity and mortality among solid cancer patients and impact of the disease on anticancer treatment: a French nationwide cohort study (GCO-002 CACOVID-19) Eur J Cancer. 2020 Dec;141:62–81. doi: 10.1016/j.ejca.2020.09.035. Epub 2020 Oct 8. PMID: 33129039; PMCID: PMC7543792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Drake T., Palmieri C., Turtle L., Harrison E., Docherty A., Greenhalf B., et al. Prospective data of >20,000 hospitalised patients with cancer and COVID-19 derived from the international severe acute respiratory and emerging infections consortium WHO coronavirus clinical characterisation consortium: CCP-cancer UK. Ann Oncol. 2021;32(suppl_5):S1283–S1346. doi: 10.1016/annonc/annonc741. [DOI] [Google Scholar]
- 27.Indini A., Pinotti G., Artioli F., Aschele C., Bernardi D., Butera A., Defraia E., Fasola G., Gamucci T., Giordano M., Iaria A., Leo S., Ribecco A.S., Rossetti R., Savastano C., Schena M., Silva R.R., Grossi F., Blasi L. Management of patients with cancer during the COVID-19 pandemic: the Italian perspective on the second wave. Eur J Cancer. 2021 May;148:112–116. doi: 10.1016/j.ejca.2021.01.040. Epub 2021 Feb 25. PMID: 33743478; PMCID: PMC7904514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pinato D.J., Scotti L., Gennari A., Colomba-Blameble E., Dolly S., Loizidou A., Chester J., Mukherjee U., Zambelli A., Aguilar-Company J., Bower M., Galazi M., Salazar R., Bertuzzi A., Brunet J., Mesia R., Sita-Lumsden A., Colomba J., Pommeret F., Seguí E., Biello F., Generali D., Grisanti S., Rizzo G., Libertini M., Moss C., Evans J.S., Russell B., Wuerstlein R., Vincenzi B., Bertulli R., Ottaviani D., Liñan R., Marrari A., Carmona-García M.C., Sng C.C.T., Tondini C., Mirallas O., Tovazzi V., Fotia V., Cruz C.A., Saoudi-Gonzalez N., Felip E., R Lloveras A., Lee A.J.X., Newsom-Davis T., Sharkey R., Chung C., García-Illescas D., Reyes R., Sophia Wong Y.N., Ferrante D., Marco-Hernández J., Ruiz-Camps I., Gaidano G., Patriarca A., Sureda A., Martinez-Vila C., Sanchez de Torre A., Rimassa L., Chiudinelli L., Franchi M., Krengli M., Santoro A., Prat A., Tabernero J., V Hemelrijck M., Diamantis N., Cortellini A. OnCovid study group. Determinants of enhanced vulnerability to coronavirus disease 2019 in UK patients with cancer: a European study. Eur J Cancer. 2021 Jun;150:190–202. doi: 10.1016/j.ejca.2021.03.035. Epub 2021 Apr 6. PMID: 33932726; PMCID: PMC8023206. [DOI] [PMC free article] [PubMed] [Google Scholar]