Abstract
Purpose of Review
To synthesize evidence from randomized controlled trials on the effects of integrated behavioral interventions for comorbid obesity and depression in adults.
Recent Findings
Seven trials (n = 33 to 409) were included. The quality of evidence was mixed. In 2 trials, integrated interventions led to greater improvements in both obesity and depression over 12 months, compared with usual care. Of 4 trials comparing integrated interventions with a standalone obesity intervention, 2 showed incremental effects on depression only, and 2 did not detect a significant effect for either outcome. One 3-arm trial compared an integrated intervention with standalone obesity and depression interventions and only detected incremental effects on obesity when compared with a standalone depression intervention.
Summary
The effects of integrated interventions for comorbid obesity and depression are varied but promising. Implications for future research to guide intervention optimization and implement integrated interventions in clinical practice are provided.
Supplementary Information
The online version contains supplementary material available at 10.1007/s11892-022-01458-z.
Keywords: Obesity, Depression, Integrated behavioral intervention, Weight, BMI
Introduction
Obesity and depression are substantial public health concerns that are frequently comorbid. Adults with obesity are 55% more likely to develop depression, and adults with depression are 58% more likely to develop obesity, relative to their respective counterparts without the index condition [1]. Comorbid obesity and depression are associated with greater morbidity, poorer treatment adherence, and worse quality of life than either condition alone [2–4]. Changes in diet and physical activity levels [5, 6] and increased social isolation [7] during the coronavirus (COVID-19) pandemic elevated the concern that obesity and depression would affect more people than ever before. Indeed, weight gain during lockdown was reported by 11.1 to 72.4% of participants in a review of 36 observational studies among adults and adolescents older than 16 years [8], and the prevalence of depression symptoms grew from 8.5% in 2017–2018 to 27.8% in 2020 among adults [9]. As a result, the population affected by co-existing obesity and depression are expected to increase markedly, and effective interventions that treat these conditions together, namely, in an integrated manner, are critically needed to support recovery from the COVID-19 pandemic.
In a widely accepted form of treatment, behavioral interventions are traditionally designed to treat obesity [10] and depression [11] separately. However, these conditions share behavioral and psychosocial risk factors (e.g., physical inactivity, maladaptive eating behaviors, body dissatisfaction) [12–14] and biological mechanisms (e.g., epigenetics, inflammation, neural dysregulation) that are modifiable through interventions addressing these risk factors [15]. These overlapping risk factors and mechanisms suggest that integrated interventions designed to treat both conditions simultaneously hold great potential for synergistic and effective treatment of these comorbidities.
To inform clinical practice and future research, the purpose of this review is to synthesize evidence from randomized controlled trials (RCTs) on the effects of integrated behavioral interventions designed to treat comorbid obesity and depression in adults.
Methods
Review Design and Study Selection
An electronic literature search was conducted to identify relevant articles published through July 2021, with no beginning date limit imposed. PubMed, CINAHL, PsycInfo, and Cochrane databases were searched using database-specific variations on the following search terms: obesity AND depression AND (weight OR body mass index OR BMI OR waist OR fat). The search strategy is detailed in Supplementary Table 1. The study design was limited to RCTs that examined the effects of an integrated behavior change intervention designed for adults age 18 years or older with comorbid obesity and depression on both weight-related and depression outcomes relative to any type of control group. Integrated behavioral interventions were defined as those that included theory-based behavior change components specifically designed to address obesity and depression. Interventions designed to address obesity alone or depression alone were excluded. Any setting and length of follow-up were allowed. Studies that were unavailable in English were excluded. Duplicate records were removed using EndNote (version 8.2, Clarivate, London, UK). Two researchers (NL and EK) reviewed titles and abstracts against inclusion and exclusion criteria (Supplementary Table 2) to identify potentially eligible articles. The same two researchers then independently reviewed articles selected for full-text review to determine the final list of eligible studies. Disagreements were resolved through discussion and consultation with the senior researcher (JM).
Data Extraction and Risk of Bias Assessment
Data were extracted from each of the included articles by NL. Data elements included study design, study setting, study duration, eligibility criteria, baseline participant characteristics, study retention, characteristics of the treatment and control groups, treatment duration, and primary/secondary weight-related and depression outcomes. The Cochrane risk of bias tool was used to assess the quality of each study (high, low, or unclear risk of bias) on 7 domains: random sequence generation, allocation concealment, blinding of participants and research personnel, blinded outcome assessment, incomplete outcome data, selective outcome reporting, and other sources of bias [16]. Two researchers (NL and EK) independently assessed each study, and disagreements were resolved through discussion. The senior researcher was consulted when necessary. The consistency of outcome measures and research designs across studies was examined to determine whether a meta-analysis or qualitative synthesis was appropriate [17].
Results
Study Identification
Of the 1077 references identified, 1057 were excluded based on title, language, or study type (n = 924) and abstract reviews (n = 133). Candidate full-text articles (n = 20) were assessed for eligibility and 13 were excluded. Reasons for exclusion at all stages are depicted in Fig. 1. Reasons for exclusion on full-text review are detailed in Supplementary Table 3, which included primarily (1) participants do not have comorbid obesity and depression (n = 9), and (2) intervention has no behavior change component or is not designed for comorbid obesity and depression (n = 3). Table 1 details main features of the 7 studies, which are summarized below. Additional details of the studies can be found in Supplementary Table 4.
Fig. 1.
Results of search for relevant studies
Table 1.
Summary of integrated behavioral interventions for adults with comorbid obesity and depression
| Study | Treatment groups | Baseline measures | Primary outcome(s) | Secondary outcome(s) |
|---|---|---|---|---|
|
Linde et al., 2011 N = 203 |
G1: Combined intervention ○ Weight loss: behavior therapy for obesity ○ Depression: cognitive behavioral treatment, drawn from the Coping with Depression manual G2: Weight loss only intervention |
Weight, kg, mean (SD) G1: 103.4 (17.7) G2: 106.6 (22.3) SCL-20 score, mean (SD) G1: 1.9 (0.6) G2: 1.8 (0.6) |
At 12 months Weight change, kg, mean (SD) G1: − 2.3 (8.9) G2: − 3.1 (8.9) Between-group difference, mean (95% CI): not reported P = 0.55 SCL-20 score change, mean (SD) G1: − 0.7 (0.8) G2: − 0.5 (0.8) Between-group difference, mean (95% CI): not reported P = 0.25 |
At 6 months Weight change, kg, mean (SD) G1: − 1.8 (6.6) G2: − 2.8 (6.6) Between-group difference, mean (95% CI): not reported P = 0.26 SCL-20 score change, mean (SD) G1: − 0.5 (0.7) G2: − 0.5 (0.7) Between-group difference, mean (95% CI): not reported P = 0.70 |
|
Pagoto et al., 2013 N = 161 |
G1: Behavior therapy with lifestyle intervention (BA) ○ Weight loss: Diabetes Prevention Program ○ Depression: behavioral activation G2: Lifestyle intervention only (LI) |
Weight, kg, mean (SD) G1: 95.8 (11.9) G2: 92.5 (10.1) BDI-II score, mean (SD) G1: 21.1 (5.7) G2: 21.0 (5.9) HDRS score, mean (SD) G1: 17.9 (3.5) G2: 17.9 (3.5) |
At 6 months Weight change, %, mean (SE) G1: − 3.0 (0.7) G2: − 3.7 (0.6) Between-group difference, mean (95% CI): not reported P = 0.48 At 12 months Weight change, %, mean (SE) G1: − 2.6 (0.8) G2: − 3.1 (0.7) Between-group difference, mean (95% CI): not reported P = 0.72 |
At 6 months BDI-II score change, mean (SE) G1: − 12.5 (0.8) G2: − 9.2 (0.8) Between-group difference, mean (95% CI): not reported P = 0.005 HDRS score change, mean (SE) G1: − 8.9 (0.7) G2: − 6.9 (0.7) Between-group difference, mean (95% CI): not reported P = 0.07 At 12 months BDI-II score change, mean (SE) G1: − 12.6 (1.0) G2: − 9.9 (0.9) Between-group difference, mean (95% CI): not reported P = 0.045 HDRS score change, mean (SE) G1: − 8.9 (0.7) G2: − 7.6 (0.7) Between-group difference, mean (95% CI): not reported P = 0.22 |
|
Moncrieft et al., 2016 N = 111 |
G1: Community approach to lifestyle modification for diabetes (CALM-D) ○ Weight loss: Diabetes Prevention Program ○ Depression: cognitive behavioral and social learning approaches G2: Usual care |
Weight, kg, mean (SD) G1: 85.0 (12.2) G2: 85.6 (16.2) BDI-II score, mean (SD) G1: 19.3 (7.1) G2: 21.2 (7.1) |
At 12 months Weight, kg, mean (SD) G1: 82.0 (12.6) G2: 84.2 (15.5) Between-group difference, kg/month, mean (SE): − 0.322 (0.124) P = 0.01 BDI-II score, mean (SD) G1: 9.9 (8.9) G2: 16.0 (10.8) Between-group difference, unit score/month, mean (SE): −1.009 (0.226) P < 0.001 |
At 6 months Weight, kg, mean (SD) G1: 81.8 (12.6) G2: 85.2 (16.1) Between-group difference, kg/month, mean (SE): not reported P not reported BDI-II score, mean (SD) G1: 10.8 (7.8) G2: 16.1 (9.2) Between-group difference, unit score/month, mean (SE): not reported P not reported |
|
Faulconbridge et al., 2018 N = 76 |
G1: Combined treatment ○ Weight loss: Brownell’s lifestyle, exercise, attitudes, relationships, and nutrition program for weight management ○ Depression: Cognitive behavioral therapy G2: Cognitive behavioral therapy for depression (CBT-D) G3: Behavioral weight control (BWC) |
Weight, kg, mean (SD) G1: 101.4 (14.9) G2: 96.3 (12.3) G3: 99.5 (19.5) HDRS score, mean (SD) G1: 16.2 (3.1) G2: 15.8 (4.0) G3: 16.4 (3.7) |
At 20 weeks Weight change, %, mean (SD) G1: − 5.2 (1.2) G2: − 0.8 (1.3) G3: − 3.5 (1.3) Between-group difference, mean (95% CI): not reported G1 vs. G2 P < 0.02 G1 vs. G3 P = 0.21 G2 vs. G3 P = 0.13 |
At 20 weeks HDRS score change, mean (SD) G1: − 7.3 (1.3) G2: − 7.7 (1.3) G3: − 7.9 (1.3) Between-group difference, mean (95% CI): not reported G1 vs. G2 P > 0.05 G1 vs. G3 P > 0.05 G2 vs. G3 P > 0.05 At 46 weeks Weight change, %, mean (SD) G1: − 2.6 (1.4) G2: − 1.8 (1.4) G3: − 3.3 (1.5) Between-group difference, mean (95% CI): not reported G1 vs. G2 P > 0.05 G1 vs. G3 P > 0.05 G2 vs. G3 P > 0.05 |
|
Cooney et al., 2018 N = 33 |
G1: Cognitive behavioral therapy (CBT) and lifestyle modifications (LS) ○ Weight loss: behavioral counselling (self-monitoring, problem-solving, enlisting social support, and overcoming negative thoughts) ○ Depression: Cognitive behavioral therapy G2: Lifestyle modifications (LS) |
Weight, kg, median (IQR) G1: 100 (88–114) G2: 94 (77–111) CES-D score, median (IQR) G1: 23 (22–29) G2: 24 (19–27) |
At 8 weeks Weight change, kg, median (IQR) G1: − 1.4 (− 3.0 to − 0.6) G2: − 2.3 (− 3.2 to − 1.6) Between-group difference, mean (95% CI): not reported P = 0.60 At 16 weeks Weight change, kg, median (IQR) G1: − 3.2 (− 7.7 to − 2.1) G2: − 1.8 (− 3.8 to − 1.0) Between-group difference, mean (95% CI): not reported P = 0.08 |
At 8 weeks CES-D score change, median (IQR) G1: − 5 (− 11 to 8) G2: − 5 (− 13 to − 3) Between-group difference, mean (95% CI): not reported P = 0.95 At 16 weeks CES-D score change, median (IQR) G1: − 1 (− 9 to 8) G2: − 6 (− 12 to 3) Between-group difference, mean (95% CI): not reported P = 0.45 |
|
Ma et al., 2019 N = 409 |
G1: Integrated collaborative care intervention ○ Weight loss: Group Lifestyle Balance program adapted from Diabetes Prevention Program ○ Depression: The Program to Encourage Active, Rewarding Lives for Seniors (PEARLS) G2: Usual care |
BMI, kg/m2, mean (SD) G1: 36.7 (6.9) G2: 36.6 (5.8) SCL-20 score, mean (SD) G1: 1.5 (0.5) G2: 1.5 (0.6) |
At 12 months BMI, kg/m2, mean (SD) G1: 35.9 (7.1) G2: 36.6 (6.0) Between-group difference, mean (95% CI) − 0.7 (− 1.1, − 0.2) P = 0.01 SCL-20 score, mean (SD) G1: 1.1 (1.0) G2: 1.4 (1.3) Between-group difference, mean (95% CI) − 0.2 (− 0.4, − 0.0) P = 0.01 |
At 6 months BMI, kg/m2, mean (SD) G1: 36.0 (6.9) G2: 36.6 (6.2) Between-group difference, mean (95% CI) − 0.6 (− 0.9, − 0.3) P < 0.001 SCL-20 score, mean (SD) G1: 1.1 (0.7) G2: 1.4 (0.8) Between-group difference, mean (95% CI) − 0.3 (− 0.4, − 0.1) P < 0.001 |
|
Evans-Hudnall et al., 2020 N = 38 |
G1: HERO (Healthy Emotions and Improving Health BehavioR Outcomes) and MOVE! ○ Weight: Veterans Health Administration’s weight management program ○ Depression: cognitive behavioral therapy G2: Usual care and MOVE! |
Weight, lb, mean (SD) G1: 232.6 (43.2) G2: 263.0 (53.8) PHQ-9 score, mean (SD) G1: 11.3 (7.4) G2: 12.7 (7.1) |
NA |
At 8 weeks Weight, lb, mean (SD) G1: 234 (32.2) G2: 260 (47.3) Between-group difference, mean (95% CI): not reported P > 0.05 PHQ-9 score, mean (SD) G1: 13.1 (8.2) G2: 11.1 (6.7) Between-group difference, mean (95% CI): not reported P > 0.05 At 16 weeks Weight, lb, mean (SD) G1: 230.2 (35.4) G2: 264.8 (41.4) Between-group difference, mean (95% CI): not reported P > 0.05 PHQ-9 score, mean (SD) G1: 6.3 (7.1) G2: 13.4 (6.2) Between-group difference, mean (95% CI): not reported P = 0.02 |
Study Design
Participant Characteristics
A total of 1031 participants were included in the 7 studies (sample size range, 33 to 409). All studies included adults over 18 years with the mean age ranging from 45 to 59 years. One study reported the median age of 29 years in the integrated intervention group and 32 years in the lifestyle modification-only control group [18]. An upper age restriction of 65 to 70 years was imposed in 4 trials [19–22]. Participants were required to be female in 3 studies [18–20], while 3 studies that included both female and male reported a predominantly female sample [21–23], and 1 study that included the USA veterans reported a predominantly male sample [24]. More than half of participants were white in 5 of the 7 studies [18–20, 22, 23]. Study participants met baseline criteria for obesity (mean BMI range of 32.3 to 39.5) and depression assessed by several measures, including the 9-item Patient Health Questionnaire (PHQ-9), Structured Clinical Interview for DSM-IV (SCID-IV), Beck Depression Inventory-II (BDI-II), Hamilton Depression Rating Scale (HDRS), and Center for Epidemiologic Studies Depression Scale (CES-D).
Study Design and Setting
All included RCTs were conducted in the USA. Outcomes were assessed across a range of timepoints including 8 weeks [18, 24], 16 weeks [18, 24], 20 weeks [22], 6 months [19–21, 23], 46 weeks [22], and 12 months [19–21, 23]. Measures of obesity included BMI and weight. Measures of depression included the 20-item Depression Symptom Checklist (SCL-20) [25], HDRS [26], BDI-II [27], CES-D [28], and PHQ-9 [29]. SCL-20 scores range between 0 (best) and 4 (worst), with scores of > 1.5 to 2.0 and > 2.0 representing moderate depression and severe depression, respectively [23]. HDRS scores range from 0 (least severe) to 52 (most severe), with scores of 0 to 6 indicating minimal depressive symptoms, 7 to 17 indicating mild depression, 18 to 24 indicating moderate depression, and > 24 indicating severe depression [30]. BDI-II scores range from 0 to 63, with scores of 0 to 13 indicating minimal depression, 14 to 19 indicating mild depression, 20 to 28 indicating moderate depression, and ≥ 29 indicating severe depression [31]. CES-D scores range from 0 to 60, with scores of ≥ 16 indicating clinical depression [28]. PHQ-9 scores range from 0 to 27, with scores at or above 10, 15, and 20 corresponding to moderate, moderately severe, and severe depression, respectively [29]. The experimental integrated interventions were compared with usual care [21, 23] or standalone interventions (e.g., weight-loss or lifestyle-only intervention) [18–20, 22, 24]. All studies stated that analyses were conducted following intention-to-treat principles. However, randomization was overridden for 3 participants in one study [18].
Intervention Design
Integrated Intervention Intensity, Duration, Format, and Content
The 7 integrated interventions ranged in duration from 8 weeks to 1 year with varied frequency of contacts throughout the course of the program (e.g., weekly, biweekly, monthly). The number of sessions ranged from 15 to 38 during the entire intervention. Three 12-month interventions [19, 20, 23] had distinct intensive and maintenance phases, 6 months per phase. Two interventions [19, 22] utilized in-person group sessions, 2 interventions [18, 23] utilized in-person individual sessions, 2 interventions [20, 21] utilized a combination of in-person individual and group sessions, and 1 intervention [24] primarily utilized phone sessions. In addition to group and individual sessions, 2 interventions [20, 23] also utilized phone contacts during the maintenance phase.
All 7 integrated interventions included both an obesity and a depression treatment component. In 3 of them [20, 21, 23], the obesity treatment components were based on the Diabetes Prevention Program (DPP) [32]. The depression treatment components in these integrated inventions were various cognitive behavioral therapies (CBTs), including evidence-based strategies such as behavioral activation and problem-solving. Two interventions [20, 23] treated depression symptoms first with the obesity treatment component introduced in week 6 or week 9, 1 intervention [21] treated obesity first with the depression treatment component introduced in month 7, and the other 4 interventions [18, 19, 22, 24] treated obesity and depression concurrently. One integrated intervention [23] reported the use of as-needed antidepressant medications as a supplement to the first-line CBT for depression.
Theoretical Basis
Although all 7 behavioral lifestyle interventions used evidence-based cognitive-behavioral strategies (e.g., self-monitoring, goal setting, and problem-solving, and behavioral activation), only one study [23] reported social cognitive theory [33] as a theoretical basis for the Group Lifestyle Balance (GLB) program for weight loss which was adapted from the DPP and identified overlapping cognitive and behavioral principles with problem-solving treatment for depression [34].
Interventionists and Care Team
Four integrated interventions [19, 21–23] were delivered by interventionists who were cross-trained to deliver both the obesity and depression treatment components. Interventionists had varied prior experience (e.g., clinical psychologist, bachelor’s level health coach). In 3 interventions [18, 20, 24], the obesity and depression treatment components were delivered by separate trained interventionists (e.g., dietitian and exercise physiologist or trained counselor for obesity treatment and master’s or doctoral level counselor or clinical psychologist for depression treatment). Two interventions [19, 23] reported weekly care team meetings among interventionists and obesity or depression experts to conduct ongoing case reviews and discuss challenging cases or questions arising from treatment sessions.
Efficacy
Heterogeneity of the outcomes and research designs across the 7 eligible studies precluded a meta-analysis. Instead, we summarize the key findings of the individual studies.
Comparisons of Integrated Interventions to Usual Care
Two studies [21, 23] reported results on efficacy of the integrated interventions compared with usual care. Moncrieft et al. [21] reported that an integrated intervention combining a DPP-based behavioral weight loss intervention and cognitive behavioral and social learning therapy for depression resulted in decreased weight (between-group mean difference per month, mean ± SE = − 0.322 ± 0.124 kg, P = 0.01) and BDI-II scores (− 1.009 ± 0.226, P < 0.001) compared with usual care during the first 6 months. These improvements maintained at 12 months. Ma et al. [23] demonstrated that an integrated intervention combining the DPP-based Group Lifestyle Balance (GLB) for weight loss and problem-solving therapy with as-needed antidepressant medications for depression resulted in decreased BMI (between-group mean difference, mean [95% CI] = − 0.6 [− 0.9, − 0.3]; P < 0.001 for 6 months; − 0.7 [− 1.1, − 0.2], P = 0.01 for 12 months) and SCL-20 scores (− 0.3 [− 0.4, − 0.1], P < 0.001 for 6 months; − 0.2 [− 0.4, − 0.0], P = 0.01 for 12 months) compared with usual care at both 6 and 12 months.
Comparisons of Integrated Interventions to Standalone Interventions
Four studies [18–20, 24] reported results on efficacy of the integrated interventions compared with standalone weight loss interventions. In 2 studies [18, 19], weight loss and depression symptom changes did not differ significantly between treatment groups at varied time points from 8 weeks to 12 months. Two studies [20, 24] reported no difference in weight loss between treatment groups but significantly reduced depression symptoms in the integrated interventions compared with the standalone weight loss intervention. In Pagoto et al.’s study [20], participants in the integrated intervention showed significantly greater improvement in BDI-II scores compared with the standalone DPP at both 6 months (mean change ± SD, − 12.5 ± 0.85 vs. 9.2 ± 0.80, P = 0.005) and 12 months (− 12.6 ± 0.97 vs. − 9.9 ± 0.93, P = 0.045). In Evans-Hudnall et al.’s study [24], participants in the integrated intervention showed significantly lower PHQ-9 scores than participants in the MOVE! Program for weight loss at 16 weeks (mean score ± SD, 6.3 ± 7.1 vs. 13.4 ± 6.2, P = 0.02).
The only 3-arm RCT among the 7 included studies was a pilot study [22] that compared an integrated intervention with a standalone weight loss intervention and a standalone cognitive behavioral therapy for depression. In this study, participants in the integrated intervention lost significantly more weight than those in cognitive behavioral therapy for depression (mean % weight loss ± SD, − 5.2% ± 1.2% vs. − 0.8% ± 1.3%, P < 0.02) at 20 weeks; however, this effect on weight loss was not maintained at 46 weeks. In addition, improvements in depression symptoms did not differ between any of the treatment groups.
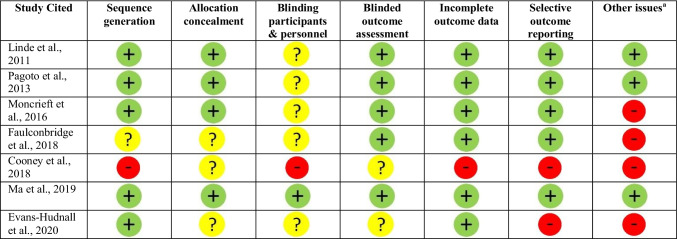
Risk of Bias Assessment
Scores on the Cochrane Risk of Bias Assessment are depicted in Table 2. One of the 7 studies had high risk of bias on generation of allocation sequence, blinding of study personnel, incomplete outcome data, selective outcome reporting, and other sources of bias such as low participant retention and short-term pilot study with small sample size [18]. One study had high risk of bias on selective outcome reporting and other sources of bias such as short-term pilot study with small sample size, no primary outcome specified, and inconsistent reporting of results and the number of participants [24]. This study also did not report enough information to enable determination of the risk of bias on allocation concealment, blinding of study personnel, and blinded outcomes assessment [24]. Another study also had high risk of bias on low participant retention, and unclear risk of bias on sequence generation, allocation concealment, and blinding of study personnel [22]. Three of the 7 studies had unclear risk of bias on blinding of study personnel [19–21]; among them, 1 study had high risk of bias on low participant retention [21]. Results from the studies with high risk of bias should be interpreted with caution.
Table 2.
Completed risk of bias tool
 , low risk of bias;
, low risk of bias;
 , high risk of bias;
, high risk of bias;
 , unknown risk of bias. aOther issues deemed high risk of bias include low participant retention (Moncrieff, Faulconbridge, Cooney), short-term pilot study with small sample size (Cooney, Evens-Hudnall), no primary outcome specified (Evens-Hudnall), and inconsistent reporting of results and the number of participants (Evens-Hudnall)
, unknown risk of bias. aOther issues deemed high risk of bias include low participant retention (Moncrieff, Faulconbridge, Cooney), short-term pilot study with small sample size (Cooney, Evens-Hudnall), no primary outcome specified (Evens-Hudnall), and inconsistent reporting of results and the number of participants (Evens-Hudnall)
Discussion
Multimorbidity (i.e., ≥ 2 chronic conditions [35]) involving mental and physical comorbidities is a major public health problem [36] that was severely exacerbated by the COVID-19 pandemic [37–42]. Obesity and depression, in particular, are highly comorbid, but traditionally treated separately despite their shared risk factors and biological mechanisms. Implementing integrated interventions that treat comorbid obesity and depression simultaneously could improve the overall health of a vulnerable population and enhance patients’ experiences of care. Integrated behavioral interventions for comorbid obesity and depression are emerging in the literature with varied results. This paper reviewed 7 RCTs of behavioral interventions among participants with mild [22] and moderate [18–21, 23, 24] depression. Of these studies, statistically significant effects were detected for both obesity and depression in 2 trials that compared the integrated interventions with usual care [21, 23]. The trial by Ma et al. [23] was the largest of the 7 and reported significant between-group mean differences for both BMI and SCL-20 scores over 12 months. The treatment effects were comparable to some of the prior trials that had separately tested either component of the integrated intervention in Ma et al. as standalone interventions, i.e., the Group Lifestyle Balance program for weight loss only [43] and the Program to Encourage Active, Rewarding Lives for Seniors (PEARLS) for depression only [44], compared with usual care. The other 5 trials [18–20, 22, 24] compared the integrated interventions with one or both of their standalone programs. Two of these trials detected significant effects of the integrated interventions on depression compared with the standalone behavioral interventions for weight loss only [20, 24], and one trial detected significant effects of the integrated intervention on weight loss compared with the standalone behavioral intervention for depression only [22]. However, none of the integrated interventions were more effective for weight compared with the standalone behavioral interventions for weight loss only or for depression compared with the standalone behavioral intervention for depression only, suggesting an additive rather than a synergistic effect of integrating the weight loss and depression interventions [18–20, 22, 24].
The varied results among these studies could be explained by the varied study and intervention designs. For example, both Ma [23] and Pagoto [20] integrated a DPP-based lifestyle intervention for weight loss and a behavioral therapy for depression that included behavioral activation, but the two studies employed different control conditions. Ma detected significant treatment effects on both weight loss and depression compared with usual care, while Pagoto only detected significant treatment effects on depression compared with the weight loss intervention only. In addition, intervention integration approaches varied across the 7 studies. Both Ma and Pagoto prioritized the depression treatment component first [20, 23], but others prioritized obesity treatment component first [21] or treated both conditions concurrently [18, 19, 22, 24]. There was also heterogeneity across intervention delivery models (i.e., individual sessions, group sessions, and a combination thereof) and modes (i.e., in-person, phone, and a combination of both during the intensive and maintenance phases of the interventions).
This systematic review has several limitations. First, our search was limited to indexed articles reported in English; we did not search for gray literature. Second, heterogeneity across study outcomes and design precluded the pooling of data for a meta-analysis. Third, although all studies were RCTs, several were designed to be pilot studies with small sample sizes and only reported within-group effects rather than between-group effects. Furthermore, results from the studies with unknown or high risk of bias related to personnel blinding, high rate of attrition, sequence generation, allocation concealment, and selective outcome reporting should be interpreted with caution.
Despite these limitations, the results of these studies have implications for clinical practice for people with comorbid obesity and depression.
When obesity and depression co-occur, an integrated collaborative care treatment approach may be effective. The results from the studies reviewed in this paper suggested an additive effect of integrating the weight loss and depression intervention. These integrated interventions enable providers to take a whole-person approach to treatment for these frequently comorbid conditions. This small but promising body of literature supports future implementation of integrated collaborative care interventions to move toward whole-person care [45] in clinical practice.
For patients with comorbid depression and obesity, it may be clinically important to first treat depression and then focus on weight. Both integrated interventions that treated depression symptoms first followed by obesity showed promising results on improving depression and weight outcomes [20, 23]. One study reported participants with better depression treatment response and remission lost greater weight [20]. This small body of evidence may support a need for a sequential treatment model for comorbid depression and obesity that can better engage patients with the comorbid conditions.
Transdisciplinary models of clinical practice may support future implementation of integrated interventions. Existing interventions were delivered by teams of interventionists who had specialized expertise in each of the conditions being treated. These studies employed varied team models, including delivery of intervention components by members with specialized expertise in each condition [18, 20, 24] and regular team meetings involving the cross-trained interventionist and providers with specialized expertise [19, 23]. Implementation of integrated interventions to treat multimorbidity in the future will require health systems that enable transdisciplinary team-based models of care.
The promising but heterogeneous results from these studies also point to areas for future research that advances integrated behavioral interventions for adults with comorbid obesity and depression. Four of the 7 studies had identifiable high risks of bias [18, 21, 22, 24], and 2 in particular [18, 24] raise considerable concern regarding the quality and interpretability of the study findings. Assurance of scientific rigor and transparent reporting is warranted in future research, focusing on several priority areas as follows.
Identifying moderators of treatment response may explain heterogeneity of effects and inform intervention tailoring based on baseline characteristics and clinical profiles. Adults with comorbid obesity and depression may vary in their baseline behavioral phenotype (e.g., one condition may be more severe than the other) [46]. However, few studies have examined these baseline phenotypic differences, and few integrated interventions have utilized baseline patient profiles to explore opportunities of targeting intervention to the phenotypic differences. Among the 7 studies included in this review, only 1 study [24] provided intervention tailored to the severity of patients’ baseline depression and anxiety symptoms. In this study, patients with moderate-to-severe depression received a depression management workbook, those with moderate-to-severe anxiety received an anxiety management workbook, and those with both conditions worked with the study clinician to determine which workbook to use based on the condition that was more problematic. More research is needed to advance our understanding on how baseline characteristics and clinical profiles can be used to optimize treatment for comorbid obesity and depression using a precision health framework [47].
Examining early patient engagement patterns and response to intervention will advance precise delivery of individualized care. Early patient engagement patterns (e.g., frequency of self-weighing) or intervention response (i.e., early trends in change on clinical outcomes) may be identified within the first 6 to 8 weeks of intervention. This information can be used to design augmentation or ancillary treatments at critical triage points that may bolster long-term outcomes [48]. By monitoring trajectories, future research can use innovative research methods, such as Sequential Multiple Assignment Randomized Trials (SMARTs) [49] or micro-randomization trials [50, 51], to design individualized adaptive integrated interventions using these early patterns or trajectories.
Evaluating hypothesized mechanisms underlying integrated interventions for comorbid obesity and depression will inform intervention optimization. Evaluating intervention effects on hypothesized mechanisms represents another important research area. For example, recent proof-of-mechanism trials have suggested that large-scale neural circuits, particularly the negative affect circuit, may predict or mediate treatment effects of an integrated intervention on obesity [52] and depression [53]. With more information on neural mechanisms, future trials may test neural target-driven enhancements of integrated interventions with noninvasive brain stimulation (e.g., transcranial direct current stimulation [54–56]) to augment the intervention for those who fail to show early engagement of the potential neural mediator. Mechanistic trials that use innovative approaches such as these are needed to optimize integrated interventions for treating comorbid obesity and depression.
Conclusions
Overall, this review demonstrates that integrated behavioral interventions for comorbid obesity and depression remain limited. Although the effects of existing integrated interventions for this population are varied, several are promising for improving obesity and/or depression among individuals with both conditions. This review suggests that there are considerable opportunities to build on the existing interventions using a precision medicine framework to better target and tailor interventions based on baseline phenotypes, early patient engagement and intervention response, and proven mechanisms. This important research will contribute to future optimization of integrated interventions that will lead to greater health and reduced healthcare costs among a growing population of adults with comorbid obesity and depression.
Supplementary Information
Below is the link to the electronic supplementary material.
Funding
This research was supported by the National Heart, Lung, and Blood Institute grant number T32 134634 (JM) and K23 159240 (EAK). The funder had no role in the design and conduct of the research; management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Declarations
Conflict of Interest
Dr. Jun Ma is a paid scientific consultant for Health Mentor, Inc. (San Jose, CA). Drs. Lv and Kringle declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Footnotes
This article is part of the Topical Collection on Obesity
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Nan Lv and Emily A. Kringle contributed equally to this paper.
Contributor Information
Nan Lv, Email: lvn2017@uic.edu.
Emily A. Kringle, Email: kringle@uic.edu
Jun Ma, Email: maj2015@uic.edu.
References
- 1.Luppino F, de Wit L, Bouvy P, Stijnen T, Cuijpers P, Penninx B, et al. Overweight, obesity, and depression: a systematic review and meta-analysis of longitudinal studies. Arch Gen Psychiatry. 2010;67(3):220–229. doi: 10.1001/archgenpsychiatry.2010.2. [DOI] [PubMed] [Google Scholar]
- 2.Opel N, Redlich R, Grotegerd D, Dohm K, Heindel W, Kugel H, et al. Obesity and major depression: body-mass index (BMI) is associated with a severe course of disease and specific neurostructural alterations. Psychoneuroendocrinology. 2015;51:219–226. doi: 10.1016/j.psyneuen.2014.10.001. [DOI] [PubMed] [Google Scholar]
- 3.Blasco BV, Garcia-Jimenez J, Bodoano I, Gutierrez-Rojas L. Obesity and depression: its revalence and influence as a prognostic factor: a systematic review. Psychiatry Investig. 2020;17(8):715–724. doi: 10.30773/pi.2020.0099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Colomer L, Anmella G, Vieta E, Grande I. Physical health in affective disorders: a narrative review of the literature. Braz J Psychiatry. 2020 doi: 10.1590/1516-4446-2020-1246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chew HSJ, Lopez V. Global impact of COVID-19 on weight and weight-related behaviors in the adult population: a scoping review. Int J Environ Res Public Health. 2021;18(4):1876. doi: 10.3390/ijerph18041876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Stockwell S, Trott M, Tully M, Shin J, Barnett Y, Butler L, et al. Changes in physical activity and sedentary behaviours from before to during the COVID-19 pandemic lockdown: a systematic review. BMJ Open Sport Exerc Med. 2021;7(1):e000960. doi: 10.1136/bmjsem-2020-000960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sepúlveda-Loyola W, Rodríguez-Sánchez I, Pérez-Rodríguez P, Ganz F, Torralba R, Oliveira D, et al. Impact of social isolation due to COVID-19 on health in older people: mental and physical effects and recommendations. J Nutr Health Aging. 2020;1–10. 10.1007/s12603-020-1469-2. [DOI] [PMC free article] [PubMed]
- 8.Bakaloudi DR, Barazzoni R, Bischoff SC, Breda J, Wickramasinghe K, Chourdakis M. Impact of the first COVID-19 lockdown on body weight: a combined systematic review and a meta-analysis. Clin Nutr. 2021;S0261–5614(21):00207–217. doi: 10.1016/j.clnu.2021.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ettman C, Abdalla S, Cohen G, Sampson L, Vivier P, Galea S. Prevalence of depression symptoms in US adults before and during the COVID-19 pandemic. JAMA Netw Open. 2020;3(9):e2019686. doi: 10.1001/jamanetworkopen.2020.19686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.LeBlanc E, Patnode C, Webber E, Redmond N, Rushkin M, O'Connor E. Behavioral and pharmacotherapy weight loss interventions to prevent obesity-related morbidity and mortality in adults: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;320(11):1172–1191. doi: 10.1001/jama.2018.7777. [DOI] [PubMed] [Google Scholar]
- 11.Farah WH, Alsawas M, Mainou M, Alahdab F, Farah MH, Ahmed AT, et al. Non-pharmacological treatment of depression: a systematic review and evidence map. BMJ Evid Based Med. 2016;21(6):214–221. doi: 10.1136/ebmed-2016-110522. [DOI] [PubMed] [Google Scholar]
- 12.Goldschmidt AB, Wall M, Choo T-HJ, Becker C, Neumark-Sztainer D. Shared risk factors for mood-, eating-, and weight-related health outcomes. Health Psychol. 2016;35(3):245. doi: 10.1037/hea0000283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hruby A, Manson JE, Qi L, Malik VS, Rimm EB, Sun Q, et al. Determinants and consequences of obesity. Am J Public Health. 2016;106(9):1656–1662. doi: 10.2105/AJPH.2016.303326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Choi KW, Chen C-Y, Stein MB, Klimentidis YC, Wang M-J, Koenen KC, et al. Assessment of bidirectional relationships between physical activity and depression among adults: a 2-sample mendelian randomization study. JAMA Psychiatry. 2019;76(4):399–408. doi: 10.1001/jamapsychiatry.2018.4175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Milaneschi Y, Simmons WK, van Rossum EF, Penninx BW. Depression and obesity: evidence of shared biological mechanisms. Mol Psychiatry. 2019;24(1):18–33. doi: 10.1038/s41380-018-0017-5. [DOI] [PubMed] [Google Scholar]
- 16.Cochrane Handbook for Systematic Reviews of Interventions. The Cochrane Collaboration; 2011. www.cochrane-handbook.org. Accessed 3 Mar 2022.
- 17.Muka T, Glisic M, Milic J, Verhoog S, Bohlius J, Bramer W, et al. A 24-step guide on how to design, conduct, and succesfully publish a systematic review an dmeta-analysis in medical research. Eur J Epidemiol. 2020;35:49–60. doi: 10.1007/s10654-019-00576-5. [DOI] [PubMed] [Google Scholar]
- 18.Cooney L, Milman L, Hantsoo L, Kornfield S, Sammel M, Allison K, et al. Cognitive-behavioral therapy improves weight loss and quality of life in women with polycystic ovary syndrome: a pilot randomized clinical trial. Fertil Steril. 2018;110(1):161–171. doi: 10.1016/j.fertnstert.2018.03.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Linde J, Simon G, Ludman E, Ichikawa L, Operskalski B, Arterburn D, et al. A randomized controlled trial of behavioral weight loss treatment versus combined weight loss/depression treatment among women with comorbid obesity and depression. Ann Behav Med. 2011;41:119–130. doi: 10.1007/s12160-010-9232-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pagoto S, Schneider K, Whited M, Oleski J, Merriam P, Appelhans B, et al. Randomized controlled trial of behavioral treatment for comorbid obesity and depression in women: the Be Active trial. Int J Obes. 2013;37:1427–1434. doi: 10.1038/ijo.2013.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Moncrieft A, Llabre M, McCalla J, Gutt M, Mendez A, Gellman M, et al. Effects of a multicomponent life-style intervention on weight, glycemic control, depressive symptoms, and renal function in low-income, minority patients with type 2 diabetes: Results of the community approach to lifestyle modification for diabetes randomized controlled trial. Psychosom Med. 2016;78:851–860. doi: 10.1097/PSY.0000000000000348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Faulconbridge L, Driscoll C, Hopkins C, Benforado B, Bishop-Gilyard C, Carvajal R, et al. Combined treatment for obesity and depression: a pilot study. Obesity. 2018;26:1144–1152. doi: 10.1002/oby.22209. [DOI] [PubMed] [Google Scholar]
- 23.Ma J, Rosas LG, Lv N, Xiao L, Snowden MB, Venditti EM, et al. Effect of integrated behavioral weight loss treatment and problem-solving therapy on body mass index and depressive symptoms among patients with obesity and depression: the RAINBOW randomized clinical trial. JAMA. 2019;321(9):869–879. doi: 10.1001/jama.2019.0557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Evans-Hudnall G, Odafe M, Johnson A, Armenti N, O'Neil J, Lawson E, et al. Using an adjunctive treatment to address psychological distress in a national weight management program: results of an integrated pilot study. Mil Med. 2020;185(9/10):e1662. doi: 10.1093/milmed/usaa145. [DOI] [PubMed] [Google Scholar]
- 25.Glass RM, Allan AT, Uhlenhuth E, Kimball CP, Borinstein DI. Psychiatric screening in a medical clinic: an evaluation of a self-report inventory. Arch Gen Psychiatry. 1978;35(10):1189–1195. doi: 10.1001/archpsyc.1978.01770340039003. [DOI] [PubMed] [Google Scholar]
- 26.Williams J. A structured interview guide for the Hamilton Depression Rating Scale. Arch Gen Psychiatry. 1988;45:742–747. doi: 10.1001/archpsyc.1988.01800320058007. [DOI] [PubMed] [Google Scholar]
- 27.Beck A, Steer R, Brown G. Manual for the beck depression inventory-II. San Antonio: The Psychological Corporation; 1996. [Google Scholar]
- 28.Lewinsohn P, Seeley J, Roberts R, Allen N. Center for Epidemiologic Studies Depression Scale (CES-D) as a screening instrument for depression among community-residing older adults. Psychol Aging. 1997;12(2):277–287. doi: 10.1037/0882-7974.12.2.277. [DOI] [PubMed] [Google Scholar]
- 29.Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606–613. doi: 10.1046/j.1525-1497.2001.016009606.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Frank E, Prien R, Jarrett R, Keller M, Kupfer D, Lavori P, et al. Conceptualization and rationale for consensus definitions of terms in major depressive disorder. Remission, recovery, relapse, and recurrence. Arch Gen Psychiatry. 1991;48:851–5. doi: 10.1001/archpsyc.1991.01810330075011. [DOI] [PubMed] [Google Scholar]
- 31.Beck A, Steer R, Ball R, Ranieri W. Comparison of Beck Depression Inventories - IA and -II in psychiatric outpatients. J Pers Assess. 1996;67(3):588–597. doi: 10.1207/s15327752jpa6703_13. [DOI] [PubMed] [Google Scholar]
- 32.The Diabetes Prevention Program (DPP) Research Group The Diabetes Prevention Program (DPP): description of lifestyle intervention. Diabetes Care. 2002;25(12):2165–71. doi: 10.2337/diacare.25.12.2165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bandura A. Social foundations of thought and action: a social cognitive theory. Englewood Cliffs: Prentice Hall; 1986. [Google Scholar]
- 34.Ma J, Yank V, Lv N, Goldhaber-Fiebert JD, Lewis MA, Kramer MK, et al. Research aimed at improving both mood and weight (RAINBOW) in primary care: a type 1 hybrid design randomized controlled trial. Contemp Clin Trials. 2015;43:260–278. doi: 10.1016/j.cct.2015.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Buttorff C, Ruder T, Bauman M. Multiple chronic conditions in the United States. 2017. https://www.rand.org/pubs/tools/TL221.html. Accessed 3 Mar 2022.
- 36.Walker ER, Druss BG. A public health perspective on mental and medical comorbidity. JAMA. 2016;316(10):1104–1105. doi: 10.1001/jama.2016.10486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Xiong J, Lipsitz O, Nasri F, Lui LMW, Gill H, Phan L, et al. Impact of COVID-19 pandemic on mental health in the general population: a systematic review. J Affect Disord. 2020;277:55–64. doi: 10.1016/j.jad.2020.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jia H, Guerin RJ, Barile JP, Okun AH, McKnight-Eily L, Blumberg SJ, et al. National and state trends in anxiety and depression severity scores among adults during the COVID-19 pandemic — United States, 2020–2021. MMWR Morb Mortal Wkly Rep. 2021;70:1427–32. doi: 10.15585/mmwr.mm7040e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ettman CK, Cohen GH, Abdalla SM, Sampson L, Trinquart L, Castrucci BC, et al. Persistent depressive symptoms during COVID-19: a national, population-representative, longitudinal study of U.S. adults. Lancet Reg Health Am. 2021:100091. 10.1016/j.lana.2021.100091. [DOI] [PMC free article] [PubMed]
- 40.Zachary Z, Brianna F, Brianna L, Garrett P, Jade W, Alyssa D, et al. Self-quarantine and weight gain related risk factors during the COVID-19 pandemic. Obes Res Clin Pract. 2020;14(3):210–216. doi: 10.1016/j.orcp.2020.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Centers for Disease Control and Prevention. Adult Obesity Prevalence Maps. 2021. https://www.cdc.gov/obesity/data/prevalence-maps.html. Accessed October 25 2021.
- 42.American Psychological Association. One year on: Unhealthy weight gains, increased drinking reported by Americans coping with pandemic stress. 2021. https://www.apa.org/news/press/releases/2021/03/one-year-pandemic-stress. Accessed October 25 2021.
- 43.Ma J, Yank V, Xiao L, Lavori P, Wilson S, Rosas L, et al. Translating the Diabetes Prevention Program lifestyle intervention for weight loss into primary care: A randomized trial. JAMA Intern Med. 2013;173(2):113–121. doi: 10.1001/2013.jamainternmed.987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ciechanowski P, Wagner E, Schmaling K, Schwartz S, Williams B, Diehr P, et al. Community-integrated home-based depression treatment in older adults: A randomized controlled trial. JAMA. 2004;291:1569–1577. doi: 10.1001/jama.291.13.1569. [DOI] [PubMed] [Google Scholar]
- 45.Thomas H, Mitchell G, Rich J, Best M. Definition of whole person care in general practice in the English language literature: A systematic review. BMJ Open. 2018;8:e023758. doi: 10.1136/bmjopen-2018-023758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ma J, Xiao L, Lv N, Rosas LG, Lewis MA, Goldhaber-Fiebert JD, et al. Profiles of sociodemographic, behavioral, clinical and psychosocial characteristics among primary care patients with comorbid obesity and depression. Prev Med Rep. 2017;8:42–50. doi: 10.1016/j.pmedr.2017.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ma J, Ronneberg CR, Lv N, Lewis MA, Kannampallil T. Precision Medicine. In: Boulton ML, editors. Evidence-based Public Health and Preventive Medicine. 16th ed. Chicago, IL: McGraw-Hill; 2020.
- 48.Lv N, Xiao L, Majd M, Lavori PW, Smyth JM, Rosas LG, et al. Variability in engagement and progress in efficacious integrated collaborative care for primary care patients with obesity and depression: within-treatment analysis in the RAINBOW trial. PLoS One. 2020;15(4):e0231743. doi: 10.1371/journal.pone.0231743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Almirall D, Nahum-Shani I, Sherwood NE, Murphy SA. Introduction to SMART designs for the development of adaptive interventions: with application to weight loss research. Transl Behav Med. 2014;4(3):260–274. doi: 10.1007/s13142-014-0265-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Golbus JR, Dempsey W, Jackson EA, Nallamothu BK, Klasnja P. Microrandomized trial design for evaluating just-in-time adaptive interventions through mobile health technologies for cardiovascular disease. Circ Cardiovasc Qual Outcomes. 2021;14(2):e006760. doi: 10.1161/CIRCOUTCOMES.120.006760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Klasnja P, Hekler EB, Shiffman S, Boruvka A, Almirall D, Tewari A, et al. Microrandomized trials: An experimental design for developing just-in-time adaptive interventions. Health Psychol. 2015;34(S):1220. doi: 10.1037/hea0000305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lv N, Lefferts WK, Xiao L, Goldstein-Piekarski AN, Wielgosz J, Lavori PW, et al. Problem-solving therapy–induced amygdala engagement mediates lifestyle behavior change in obesity with comorbid depression: a randomized proof-of-mechanism trial. Am J Clin Nutr. 2021;114(6):2060–73. 10.1093/ajcn/nqab280. [DOI] [PMC free article] [PubMed]
- 53.Goldstein-Piekarski AN, Wielgosz J, Xiao L, Stetz P, Correa CG, Chang SE, et al. Early changes in neural circuit function engaged by negative emotion and modified by behavioural intervention are associated with depression and problem-solving outcomes: A report from the ENGAGE randomized controlled trial. EBioMedicine. 2021;67:103387. doi: 10.1016/j.ebiom.2021.103387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Nord CL, Halahakoon DC, Limbachya T, Charpentier C, Lally N, Walsh V, et al. Neural predictors of treatment response to brain stimulation and psychological therapy in depression: a double-blind randomized controlled trial. Neuropsychopharmacology. 2019;44(9):1613–1622. doi: 10.1038/s41386-019-0401-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Goerigk SA, Padberg F, Bühner M, Sarubin N, Kaster TS, Daskalakis ZJ, et al. Distinct trajectories of response to prefrontal tDCS in major depression: results from a 3-arm randomized controlled trial. Neuropsychopharmacology. 2021;46(4):774–782. doi: 10.1038/s41386-020-00935-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Chalah MA, Ayache SS. Noninvasive brain stimulation and psychotherapy in anxiety and depressive disorders: a viewpoint. Brain Sci. 2019;9(4):82. doi: 10.3390/brainsci9040082. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.