Abstract
Background
The effect of the anatomic location of HCC on the prognosis of patients after hepatectomy is currently unclear.
Methods
Patients who underwent hepatectomy were retrospectively enrolled and divided into the right tumour resection group (R group) and the left tumour resection group (L group) according to the tumour anatomic location. To avoid bias, 1:2 propensity score matching (PSM) analysis was used. Based on the survival data, disease-free survival (DFS) and overall survival (OS) were evaluated by the Kaplan–Meier method, and long-term survival analysis was performed. Cox proportional hazards regression was used to analyse the risk factors associated with postoperative prognosis.
Results
A total of 700 patients were enrolled in our study. After 1:2 PSM, 354 and 177 patients were enrolled in the R group and the L group, respectively, with comparable baseline characteristics. Survival analysis showed that patients in the L group had a significantly higher recurrence rate than patients in the R group (P = 0.036), but there was no significant difference in the survival rate (P = 0.99). Long-term survival analysis showed that the survival rate of the L group was lower than that of the R group (P < 0.01). Multivariate analysis showed that tumour location in the left liver was an independent risk factor for tumour recurrence (hazard ratio, 1.263; 95% CI, 1.005–1.587) and long-term survival (hazard ratio, 3.232; 95% CI, 1.284–8.134).
Conclusion
For HCC patients, the recurrence rate and long-term survival rate of left liver tumours were significantly higher than those of right liver tumours, indicating that the anatomical location of the tumour has a significant effect on the survival of HCC patients.
Trial registration Chinese Clinical Trial Registry, ChiCTR2100052407. Registered 25 October 2021, http://www.chictr.org.cn/showproj.aspx?proj=135500.
Keywords: Hepatocellular carcinoma, Anatomic location, Hepatectomy
Background
According to the latest cancer epidemiology survey, liver tumours are the fourth most deadly malignancy in the world, causing 841,080 related deaths in 2018 worldwide [1]. In addition, the death toll from liver tumours continues to rise, and this number is expected to increase to more than one million by 2030 [2]. As the most common malignant tumour, accounting for more than 90% of liver tumours, hepatocellular carcinoma (HCC) has many treatment options [3].
Currently, there are many ways to treat HCC, including hepatectomy, liver transplantation, local ablation and external radiation, transarterial therapies and systemic therapies [4, 5]. Among them, hepatectomy is widely practised in the treatment of HCC and is still an effective radical treatment for select patients [6–8]; however, the 5-year recurrence rate after HCC resection can be more than 70% [9], indicating that the high recurrence rate after surgery greatly limits the treatment of HCC [10]. Many risk factors have been widely reported and recognized for HCC prognosis, such as MVI, tumour size, and tumour number [11–13]. Among the numerous risk factors for recurrence, the relationship between the location of HCC and tumour recurrence has not been fully discussed. Whether there is a difference between the prognosis of left liver cancer and right liver cancer has been rarely discussed. The purpose of this study was to investigate whether the location of HCC affects the prognosis of liver cancer.
Methods
We retrospectively collected data from 856 patients who underwent hepatectomy at West China Hospital from 2008 to 2015. The diagnostic criteria for HCC referred to the American Association for the Study of Liver Diseases and EASL clinical practice guidelines: Management of HCC [4, 5]. Radical resection of HCC was defined as complete excision of the tumour with clear microscopic margins and no residual tumours demonstrated by computed tomography (CT) scan or angiography at 1 month after surgery [14]. The patients were divided into the left tumour resection group (L group) and the right tumour resection group (R group) according to the site of liver resection. To avoid bias, 1:2 propensity score matching (PSM) analysis between the two cohorts was performed [15]. The inclusion criteria were as follows: 1. patients without other treatment before hepatectomy; 2. hepatectomy limited to only the left or right liver; 3. patients with Child–Pugh stage A or B; and 4. patients with normal cardiopulmonary function and could withstand surgery. The exclusion criteria were as follows: 1. patients with a positive resection margin; 2. patients with recurrence within one month after the operation; 3. Patients who died within three months after surgery; 4. Patients with other malignancies; 5. Patients infected with HCV; and 6. Patients who underwent preoperative radiofrequency ablation or other intraoperative adjuvant treatments. Our study was approved by the West China Hospital of Sichuan University Biomedical Research Ethics Committee.
Intervention
For patients with a definite preoperative diagnosis, we routinely evaluated whether there were contraindications for surgery. If there were no contraindications, liver tumour resection was performed. Intraoperative ultrasound was routinely used to determine the lesion site, the number of tumours, and the relationship with important intrahepatic structures and to re-evaluate the scope of resection. If new lesions were found, resection or radiofrequency ablation was performed. Patients who underwent only radiofrequency ablation were excluded.
Follow-up
After the operation, abdominal ultrasound and alpha-fetoprotein (AFP) measurement were regularly performed every 3 months. If suspicious recurrent lesions were detected, contrast-enhanced computed tomography and enhanced magnetic resonance imaging were performed for further evaluation. Overall survival (OS) was defined as the time from the date of surgery to the patient's death or the last follow-up. Disease-free survival (DFS) was defined as the time from the date of surgery to the time of tumour recurrence. Long-term OS in our study were defined as 5-year OS. The end points of follow-up were OS and DFS.
Statistical analysis
For the clinical characteristics of patients, continuous variables were compared using the unpaired t test or the Mann–Whitney U test, and categorical variables were compared using the chi-squared (X2) test or Fisher’s exact test. A 1:2 PSM between the L group and R group was applied to overcome possible selection bias. All of the baseline clinical variables were used in PSM. Patient survival was analysed using the Kaplan–Meier method, and survival curves were compared using the log-rank test. Univariate analyses were carried out using a Cox proportional hazards stepwise model to identify independent factors related to OS and DFS. Stepwise multivariate analysis was performed if the variables were significant (P < 0.1). Analyses were performed by SPSS Statistics version 22.0 for Windows (IBM Corp) and R version 4.1.1 for Windows (32/64 bit).
Results
Patient characteristics
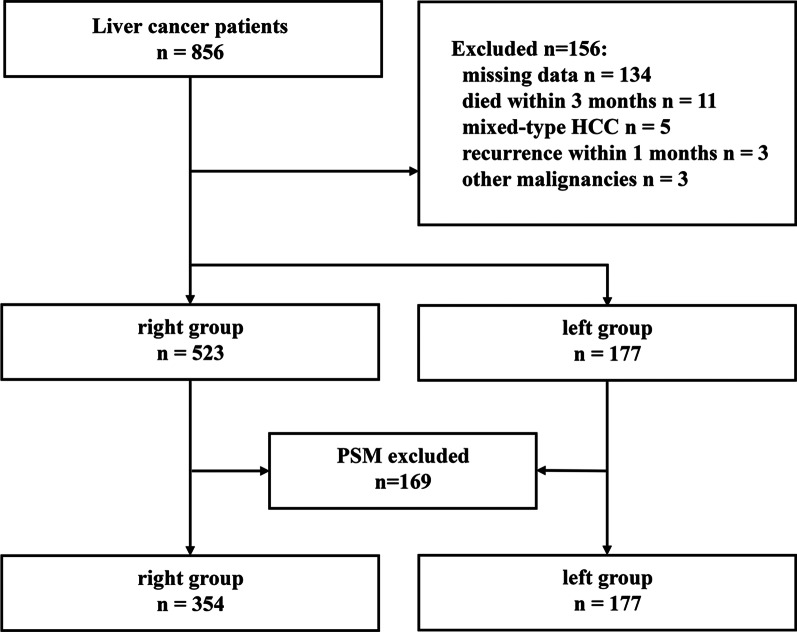
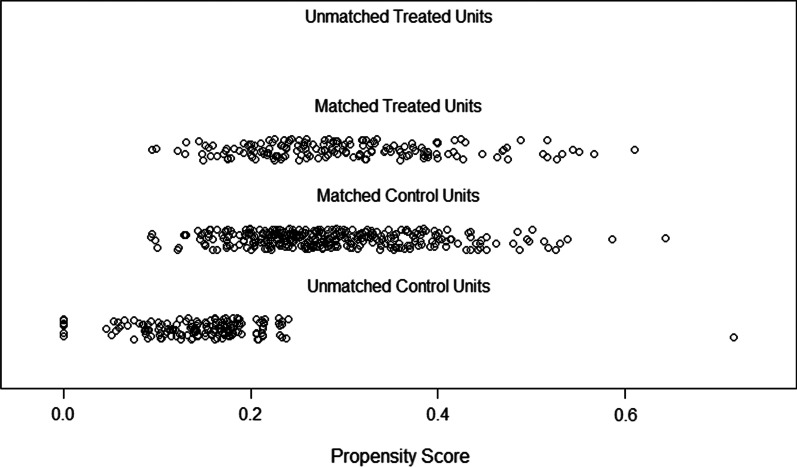
From January 2008 to April 2015, a total of 856 HCC patients who underwent hepatectomy at West China Hospital were retrospectively analysed. Among these patients, 700 patients meeting the inclusion and exclusion criteria were selected for comparison. For the excluded patients, the reasons for exclusion varied, including missing data (n = 134), other malignancies (n = 3), recurrence within 4 weeks (n = 3), death within 3 months (n = 11) and pathological confirmation of mixed-type HCC (n = 5). Finally, a total of 700 patients, consisting of 177 L group patients and 523 R group patients, were enrolled in the analysis. As shown in Table 1, the baseline characteristic data before PSM analysis showed significant differences, including differences in MVI (P = 0.003) and ALT level (P = 0.042). After 1:2 PSM, as shown in Table 2, there were 177 and 354 patients in L group and R group with comparable baseline characteristics (Fig. 1). The detailed scores of matched and unmatched unites in each group are shown in Fig. 2.
Table 1.
Baseline characteristics before propensity score matching
| Variable | R group | L group | P value |
|---|---|---|---|
| n = 523 | n = 177 | ||
| Sex (male) | 452 (86.4%) | 146 (82.5%) | 0.246 |
| Poor differentiation | 204 (39.0%) | 76 (42.9%) | 0.291 |
| Lymphatic metastasis | 2 (0.4%) | 2 (1.1%) | 0.573 |
| Cirrhosis | 327 (62.5%) | 121 (68.4%) | 0.191 |
| MVI | 156 (29.8%) | 75 (42.4%) | 0.003 |
| Satellite nodules | 77 (14.7%) | 24 (13.6%) | 0.797 |
| Tumor number (single) | 470 (89.9%) | 154 (87.0%) | 0.220 |
| GVI | 41 (7.8%) | 18 (10.2%) | 0.419 |
| Invading adjacent organs | 7 (1.3%) | 0 (0.0%) | 0.267 |
| Positive HBsAg | 463 (88.5%) | 151 (85.3%) | 0.320 |
| Positive HBeAg | 96 (18.4%) | 38 (21.5%) | 0.424 |
| AFP (< 400 ng/mL) | 317 (60.6%) | 98 (55.4%) | 0.255 |
| Age (year) (IQR) | 49.0 (42.0–58.0) | 49.0 (41.0–58.0) | 0.692 |
| Tumor diameter (cm) (IQR) | 5.0 (3.6–8.0) | 5.0 (3.5–8.0) | 0.772 |
| NEU count (109/L) (IQR) | 3.25 (2.46–4.23) | 2.99 (2.39–4.09) | 0.125 |
| LYM count (109/L) (IQR) | 1.45 (1.16–1.80) | 1.44 (1.05–1.83) | 0.339 |
| PLA count (109/L) (IQR) | 129.0 (89.0–184.0) | 128.0 (88.5–170.0) | 0.373 |
| TBIL level (mmol/L) (IQR) | 14.30 (10.70–18.30) | 13.40 (10.15–17.75) | 0.085 |
| ALT level (U/L) (IQR) | 42.00 (28.00–66.00) | 39.00 (26.00–57.00) | 0.042 |
| AST level (U/L) (IQR) | 39.00 (29.00–56.00) | 38.00 (29.00–52.00) | 0.389 |
| ALB g/dL (IQR) | 41.80 (39.10–44.90) | 41.60 (39.10–43.95) | 0.342 |
| BCLC | |||
| 0 | 235 (44.9%) | 86 (48.6%) | 0.124 |
| A | 209 (40.0%) | 55 (31.1%) | |
| B | 37 (7.1%) | 19 (10.7%) | |
| C | 42 (8.0%) | 17 (9.6%) | |
R group right tumor resection group, L group left tumor resection group, MVI microvascular invasion, GVI giant vascular invasion, AFP alpha fetoprotein, IQR interquartile range, NEU neutrophil granulocyte, LYM lymphocyte, PLA platelet, TBIL total bilirubin, ALT alanine aminotransferase, AST aspartate aminotransferase, ALB albumin, BCLC Barcelona Clinic Liver Cancer Staging
Table 2.
Baseline characteristics after propensity score matching
| Variable | R group | L group | P value |
|---|---|---|---|
| n = 354 | n = 177 | ||
| Sex (male) | 290 (81.9%) | 146 (82.5%) | 0.968 |
| Poor differentiation | 152 (42.9%) | 76 (42.9%) | 0.882 |
| Lymphatic metastasis | 2 (0.6%) | 2 (1.1%) | 0.859 |
| Cirrhosis | 248 (70.1%) | 121 (68.4%) | 0.764 |
| MVI | 133 (37.6%) | 75 (42.4%) | 0.330 |
| Satellite nodules | 42 (11.9%) | 24 (13.6%) | 0.676 |
| Tumor number (single) | 319 (90.1%) | 154 (87.0%) | 0.392 |
| GVI | 34 (9.6%) | 18 (10.2%) | 0.959 |
| Invading adjacent organs | 0 (0.0%) | 0 (0.0%) | 1.000 |
| Positive HBsAg | 305 (86.2%) | 151 (85.3%) | 0.895 |
| Positive HBeAg | 72 (20.3%) | 38 (21.5%) | 0.850 |
| AFP (< 400 ng/mL) | 205 (57.9%) | 98 (55.4%) | 0.642 |
| Age (year) (IQR) | 49.0 (41.0–59.0) | 49 (41.0–58.0) | 0.510 |
| Tumor diameter (cm) (IQR) | 5.0 (3.5–8.0) | 5.0 (3.5–8.0) | 0.743 |
| NEU count (109/L) (IQR) | 3.12 (2.42–4.13) | 2.99 (2.39–4.09) | 0.684 |
| LYM count (109/L) (IQR) | 1.41 (1.11–1.77) | 1.44 (1.05–1.83) | 0.983 |
| PLA count (109/L) (IQR) | 122.0 (84.0–173.0) | 128.0 (88.5–170.0) | 0.617 |
| TBIL level (mmol/L) (IQR) | 14.00 (10.60–17.83) | 13.40 (10.15–17.75) | 0.348 |
| ALT level (U/L) (IQR) | 40.00 (26.75–57.25) | 39.00 (26.00–57.00) | 0.618 |
| AST level (U/L) (IQR) | 38.00 (29.00–54.00) | 38.00 (29.00–52.00) | 0.695 |
| ALB g/dL | 41.70 (39.00–44.90) | 41.60 (39.10–41.60) | 0.562 |
| BCLC | |||
| 0 | 175 (49.4%) | 86 (48.6%) | 0.780 |
| A | 118 (33.3%) | 55 (31.1%) | |
| B | 29 (8.2%) | 19 (10.7%) | |
| C | 32 (9.0%) | 17 (9.6%) | |
R group right tumor resection group, L group left tumor resection group, MVI microvascular invasion, GVI giant vascular invasion, AFP alpha fetoprotein, IQR interquartile range, NEU neutrophil granulocyte, LYM lymphocyte, PLA platelet, TBIL total bilirubin, ALT alanine aminotransferase, AST aspartate aminotransferase, ALB albumin, BCLC Barcelona Clinic Liver Cancer Staging
Fig. 1.
Flow chart of the study participants
Fig. 2.
Distribution of propensity scores of L group (Treated Units) and R group (Control Units)
Association of tumour location and prognosis
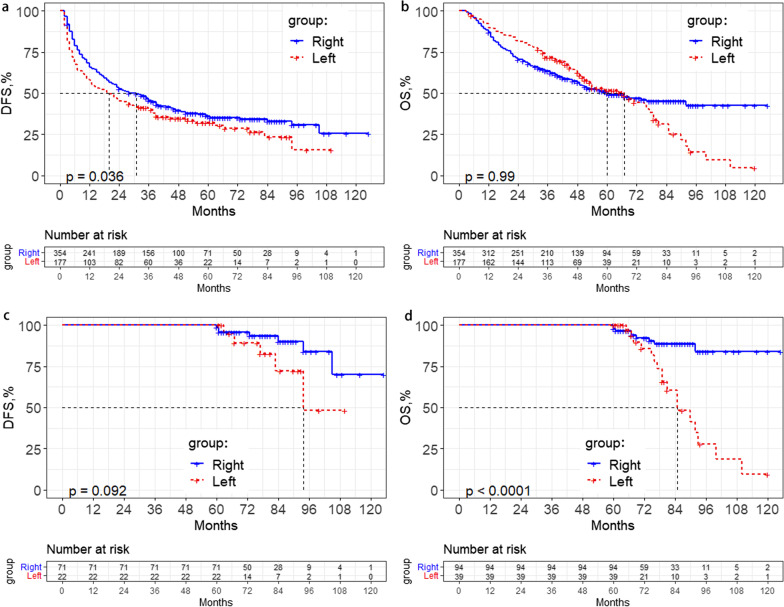
According to the tumour location of the patients before the operation, two groups were investigated: the L group and the R group. After the PSM analysis, for L group patients, the 1-, 3- and 5-year recurrence rates after surgery were 43.4%, 59.5% and 67.9%, while in the R group, the recurrence rates were 34.3%, 54.3% and 63.9%, respectively (P = 0.036) (Fig. 3a). However, this trend was not found for OS, and there was no significant difference between the two groups of patients. For L group patients, the 1-, 3- and 5-year mortality rates after surgery were 10.2%, 28.1% and 48.2%, while those of the R group were 13.0%, 36.4% and 50.2%, respectively (P = 0.990) (Fig. 3b). Multivariate analysis demonstrated that tumour location (hazard ratio, 1.263; 95% CI, 1.005–1.587), lymphatic metastasis (hazard ratio, 6.229; 95% CI, 2.228–17.412), MVI (hazard ratio, 1.711; 95% CI, 1.330–2.202), satellite nodules (hazard ratio, 1.449; 95% CI, 1.018–2.063), HBeAg (hazard ratio, 1.639; 95% CI, 1.255–2.139), and AFP levels (hazard ratio, 1.421; 95% CI, 1.131–1.786) were independent risk factors for DFS (Table 3). For OS, we failed to find that tumour location was an independent risk factor after multivariate analysis.
Fig. 3.
Kaplan–Meier analysis of disease-free survival (DFS) and overall survival (OS) for hepatocellular carcinoma patients with different anatomy locations: a DFS for the L group and R group after 1:2 PSM. b OS for the L group and R group after 1:2 PSM. c Long-term of DFS for the L group and R group after 1:2 PSM. d Long-term of OS for the L group and R group after 1:2 PSM
Table 3.
Uni- and multivariate analyses of disease-free survival (DFS) and overall survival (OS)
| Variable | Univariate | Multivariate | ||
|---|---|---|---|---|
| HR (95% CI) | P value | HR (95% CI) | P value | |
| DFS | ||||
| Location, left vs right | 1.271 (1.018–1.588) | 0.034 | 1.263 (1.005–1.587) | 0.046 |
| Differentiation, well vs poor | 0.712 (0.575–0.882) | 0.002 | ||
| Lymphatic metastasis, yes vs no | 6.729 (2.487–18.210) | < 0.001 | 6.229 (2.228–17.412) | < 0.001 |
| MVI, yes vs no | 2.390 (1.930–2.960) | < 0.001 | 1.711 (1.330–2.202) | < 0.001 |
| Satellite nodule, yes vs no | 2.504 (1.875–3.345) | < 0.001 | 1.449 (1.018–2.063) | 0.040 |
| GVI, yes vs no | 3.155 (2.299–4.329) | < 0.001 | ||
| BCLC, A vs 0 | 1.691 (1.325–2.159) | < 0.001 | ||
| BCLC, B vs 0 | 1.979 (1.371–2.856) | < 0.001 | ||
| BCLC, C vs 0 | 2.897 (1.325–5.806) | < 0.001 | ||
| HbeAg, positive vs negative | 1.481 (1.155–1.899) | 0.002 | 1.639 (1.255–2.139) | < 0.001 |
| AFP, > 400 vs < = 400 ng/mL | 1.762 (1.425–2.180) | < 0.001 | 1.421 (1.131–1.786) | 0.003 |
| Age, y (continuous) | 0.984 (0.975–0.993) | < 0.001 | ||
| Tumor diameter, cm (continuous) | 1.092 (1.066–1.119) | < 0.001 | ||
| NEU, 109/L (continuous) | 1.078 (1.000–1.162) | 0.049 | ||
| Platelet, 109/L (continuous) | 1.003 (1.001–1.005) | < 0.001 | ||
| AST, U/L (continuous) | 1.004 (1.002–1.006) | < 0.001 | ||
| Long-term OS | ||||
| Location, left vs right | 4.793 (2.132–10.780) | < 0.001 | 3.232 (1.284–8.134) | 0.013 |
| MVI, yes vs no | 4.117 (1.897–8.935) | < 0.001 | 3.161 (1.284–7.786) | 0.012 |
| Satellite nodule, yes vs no | 3.017 (1.029–8.845) | 0.044 | ||
| GVI, yes vs no | 4.215 (1.566–11.340) | 0.004 | ||
| BCLC, C vs 0 | 4.831 (1.696–13.765) | 0.003 | ||
| AFP, > 400 vs ≤ 400 ng/mL | 2.230 (1.016–4.891) | 0.046 | ||
HR hazard ratio, CI confidence interval, MVI microvascular invasion, GVI giant vascular invasion, BCLC Barcelona Clinic Liver Cancer Staging, AFP alpha fetoprotein, AST aspartate aminotransferase
Long-term survival analysis
We found potential differences after five years by observing the shape of the survival curve. Therefore, we wondered if different sites had an effect on long-term outcomes and analysed the survival outcome after five years. We did not find a significant difference in long-term DFS. For L group patients, the 5-, 8- and 10-year recurrence rates after surgery were 67.9%, 84.5% and 84.5%, while for R group patients, the recurrence rates were 63.9%, 69.3% and 74.4%, respectively (P = 0.092) (Fig. 3c). However, there were differences in long-term OS; for L group patients, the 5-, 8- and 10-year mortality rates after surgery were 48.2%, 85.7% and 95.2%, while for R group patients, the OS rates were 50.2%, 57.4% and 57.4% (P < 0.01) (Fig. 3d). Multivariate analysis demonstrated that tumour location (hazard ratio, 3.232; 95% CI, 1.284–8.134) and MVI (hazard ratio, 3.161; 95% CI, 1.284–7.786) were independent risk factors for long-term OS (Table 3).
Discussion
Although many risk factors have been widely reported to influence survival, few previous studies have reported the effect of the location of primary HCC lesions. Previously, surgeons’ understanding of liver anatomy was mainly for the innovation of the surgical method of liver resection. However, with the gradual deepening of understanding, the influence of anatomy on the prognosis of HCC should also be considered. In this study, we enrolled a large group of patients and analysed the relationship between the location of the primary HCC lesion and the survival of patients. We found that the postoperative recurrence rate and long-term mortality rate of the L group were significantly higher than those of the R group.
At present, late recurrence of HCC is generally considered 2 years after surgery [16, 17], while 5 years is usually regarded as the time point for long-term survival [18, 19]. The risk factors of early and late prognosis of HCC after hepatectomy are different and these issues have been discussed by many studies. Imamura et al. conducted research shows that factors related with elevated carcinogenesis, like higher grade of hepatitis activity, multiple tumours, and gross tumour classification, contributed to late phase prognosis [20]. Wu et al. found high viral loads and hepatic inflammatory activity were associated with late recurrence [21]. Zhang et al. revealed compared with women, men had greater late recurrence rate and rate of cancer-specific mortality [22]. From the perspective of liver anatomy, there are different structural systems of left and right hepatic venous outflow. First, most of the left hepatic vein and the middle hepatic vein converge before returning to the inferior vena cava, while the right hepatic vein exists independently [23, 24]. Fang, C. et al. conducted 3D reconstruction of hepatic veins in 200 patients and found a common trunk for the left hepatic and middle veins [25]. These main channels of venous outflow after hepatectomy may be affected to varying degrees among tumour locations, thus causing different degrees of liver congestion. Second, in addition to the different outflows of the main hepatic veins, there were significant differences in the distribution of short hepatic veins between the left and right liver. Mehran R. et al. divided the main short hepatic veins of the liver into four categories, most of which drained the right liver but not the left liver [26]. Furthermore, Sakaguchi et al. specifically analysed intrahepatic venovenous shunts of the right liver by hepatic venography [27], indicating that the right liver usually has a larger number and diameter of short hepatic veins.
The difference in the distribution of the main hepatic vein and the short hepatic vein can cause the difference in the left and right hepatic vein return. At present, whether there is a clear relationship between hepatic congestion and the prognosis of HCC after hepatectomy has not been reported in large numbers of patients, but liver congestion is widely regarded as a risk factor for the recurrence of liver tumours after liver transplantation and the survival of patients [28–30]. Although there is no direct evidence to prove it at present, it has been found in clinical observation that liver congestion after left liver surgery is more serious than that after right liver surgery.
This study has some limitations. First, objectively evaluating liver congestion at present is difficult, and the direct relationship between congestion and tumour prognosis needs to be confirmed by follow-up studies. Second, due to the retrospective nature of the data, many missing clinical data points could not be remedied, and important gaps remain in our knowledge of this process. The results need to be confirmed by prospective studies.
Conclusion
Tumour anatomic location has a significant impact on the recurrence and long-term survival of HCC patients after hepatectomy, while it does not seem to affect the short-term survival of patients.
Acknowledgements
Not applicable.
Abbreviations
- R group
Right tumour resection group
- L group
Left tumour resection group
- PSM
Propensity score matching
- DFS
Disease-free survival
- OS
Overall survival
- CI
Confidence interval
- HCC
Hepatocellular carcinoma
- MVI
Microvascular invasion
- HCV
Hepatitis C virus
- AFP
Alpha fetoprotein
- GVI
Giant vascular invasion
- IQR
Interquartile range
- NEU
Neutrophil granulocyte
- LYM
Lymphocyte
- PLA
Platelet
- TBIL
Total bilirubin
- ALT
Alanine aminotransferase
- AST
Aspartate aminotransferase
- ALB
Albumin
- BCLC
Barcelona clinic liver cancer staging
Authors’ contributions
MZ, and BL conceived the study and design, SQZ and PW were involved in patient care and collected data, LL and LLX drafted the manuscript, analysed and interpreted data for the work. All authors read and approved the final manuscript.
Funding
This work was supported by Grants from the National Natural Science Foundation of China (No. 71673193) and Key research and development project of science and technology department of Sichuan Province (2021YFS0106).
Availability of data and materials
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
We confirm that all methods were performed in accordance declaration of Helsinki with the relevant guidelines and regulations and our research has been approved by the West China Hospital of Sichuan University Biomedical Research Ethics Committee. In this retrospective study, verbal consent has been obtained from study participants and application form for exemption of informed consent has been approved by the West China Hospital of Sichuan University Biomedical Research Ethics Committee.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Ming Zhang, Email: 48284728@qq.com.
Bo Li, Email: cdhxlb@126.com.
References
- 1.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Villanueva A. Hepatocellular carcinoma. N Engl J Med. 2019;380(15):1450–1462. doi: 10.1056/NEJMra1713263. [DOI] [PubMed] [Google Scholar]
- 3.Llovet JM, Lencioni R, Di Bisceglie AM, Gaile PR, Dufour JF, Greten TF, Raymond E, Roskams T, De Baere T, Ducreux M, et al. EASL-EORTC Clinical Practice Guidelines: management of hepatocellular carcinoma. J Hepatol. 2012;56(4):908–943. doi: 10.1016/j.jhep.2011.12.001. [DOI] [PubMed] [Google Scholar]
- 4.Heimbach JK, Kulik LM, Finn RS, Sirlin CB, Abecassis MM, Roberts LR, Zhu AX, Murad MH, Marrero JA. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67(1):358–380. doi: 10.1002/hep.29086. [DOI] [PubMed] [Google Scholar]
- 5.European Association for the Study of the Liver. Electronic address eee, European Association for the Study of the L: EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol. 2018; 69(1):182–236. [DOI] [PubMed]
- 6.Llovet JM, Bru C, Bruix J. Prognosis of hepatocellular carcinoma: the BCLC staging classification. Semin Liver Dis. 1999;19(3):329–338. doi: 10.1055/s-2007-1007122. [DOI] [PubMed] [Google Scholar]
- 7.Forner A, Llovet JM, Bruix J. Hepatocellular carcinoma. Lancet. 2012;379(9822):1245–1255. doi: 10.1016/S0140-6736(11)61347-0. [DOI] [PubMed] [Google Scholar]
- 8.Marrero JA, Kulik LM, Sirlin CB, Zhu AX, Finn RS, Abecassis MM, Roberts LR, Heimbach JK. Diagnosis, staging, and management of hepatocellular carcinoma: 2018 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2018;68(2):723–750. doi: 10.1002/hep.29913. [DOI] [PubMed] [Google Scholar]
- 9.Ishizawa T, Hasegawa K, Aoki T, Takahashi M, Inoue Y, Sano K, Imamura H, Sugawara Y, Kokudo N, Makuuchi M. Neither multiple tumors nor portal hypertension are surgical contraindications for hepatocellular carcinoma. Gastroenterology. 2008;134(7):1908–1916. doi: 10.1053/j.gastro.2008.02.091. [DOI] [PubMed] [Google Scholar]
- 10.Poon RT, Fan ST, Ng IO, Lo CM, Liu CL, Wong J. Different risk factors and prognosis for early and late intrahepatic recurrence after resection of hepatocellular carcinoma. Cancer. 2000;89(3):500–507. doi: 10.1002/1097-0142(20000801)89:3<500::AID-CNCR4>3.0.CO;2-O. [DOI] [PubMed] [Google Scholar]
- 11.Poon RTP, Fan ST, Wong J. Risk factors, prevention, and management of postoperative recurrence after resection of hepatocellular carcinoma. Ann Surg. 2000;232(1):10–24. doi: 10.1097/00000658-200007000-00003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Imamura H, Matsuyama Y, Tanaka E, Ohkubo T, Hasegawa K, Miyagawa S, Sugawara Y, Minagawa M, Takayama T, Kawasaki S, et al. Risk factors contributing to early and date phase intrahepatic recurrence of hepatocellular carcinoma after hepatectomy. J Hepatol. 2003;38(2):200–207. doi: 10.1016/S0168-8278(02)00360-4. [DOI] [PubMed] [Google Scholar]
- 13.Dhanasekaran R, Limaye A, Cabrera R. Hepatocellular carcinoma: current trends in worldwide epidemiology, risk factors, diagnosis, and therapeutics. Hepat Med. 2012;4:19–37. doi: 10.2147/HMER.S16316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wang K, Liu JA, Yan ZL, Li J, Shi LH, Cong WM, Xia Y, Zou QF, Xi T, Shen F, et al. Overexpression of aspartyl-(Asparaginyl)-beta-hydroxylase in hepatocellular carcinoma is associated with worse surgical outcome. Hepatology. 2010;52(1):164–173. doi: 10.1002/hep.23650. [DOI] [PubMed] [Google Scholar]
- 15.Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46(3):399–424. doi: 10.1080/00273171.2011.568786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Portolani N, Coniglio A, Ghidoni S, Giovanelli M, Benetti A, Tiberio GA, Giulini SM. Early and late recurrence after liver resection for hepatocellular carcinoma: prognostic and therapeutic implications. Ann Surg. 2006;243(2):229–235. doi: 10.1097/01.sla.0000197706.21803.a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kim S, Shin J, Kim DY, Choi GH, Kim MJ, Choi JY. Radiomics on gadoxetic acid-enhanced magnetic resonance imaging for prediction of postoperative early and late recurrence of single hepatocellular carcinoma. Clin Cancer Res. 2019;25(13):3847–3855. doi: 10.1158/1078-0432.CCR-18-2861. [DOI] [PubMed] [Google Scholar]
- 18.Yoo HY, Patt CH, Geschwind JF, Thuluvath PJ. The outcome of liver transplantation in patients with hepatocellular carcinoma in the United States between 1988 and 2001: 5-year survival has improved significantly with time. J Clin Oncol. 2003;21(23):4329–4335. doi: 10.1200/JCO.2003.11.137. [DOI] [PubMed] [Google Scholar]
- 19.Wang XH, Long LH, Cui Y, Jia AY, Zhu XG, Wang HZ, Wang Z, Zhan CM, Wang ZH, Wang WH. MRI-based radiomics model for preoperative prediction of 5-year survival in patients with hepatocellular carcinoma. Br J Cancer. 2020;122(7):978–985. doi: 10.1038/s41416-019-0706-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Imamura H, Matsuyama Y, Tanaka E, Ohkubo T, Hasegawa K, Miyagawa S, Sugawara Y, Minagawa M, Takayama T, Kawasaki S, et al. Risk factors contributing to early and late phase intrahepatic recurrence of hepatocellular carcinoma after hepatectomy. J Hepatol. 2003;38(2):200–207. doi: 10.1016/S0168-8278(02)00360-4. [DOI] [PubMed] [Google Scholar]
- 21.Wu JC, Huang YH, Chau GY, Su CW, Lai CR, Lee PC, Huo TI, Sheen IJ, Lee SD, Lui WY. Risk factors for early and late recurrence in hepatitis B-related hepatocellular carcinoma. J Hepatol. 2009;51(5):890–897. doi: 10.1016/j.jhep.2009.07.009. [DOI] [PubMed] [Google Scholar]
- 22.Zhang H, Han J, Xing H, Li ZL, Schwartz ME, Zhou YH, Chen TH, Wang H, Gu WM, Lau WY, et al. Sex difference in recurrence and survival after liver resection for hepatocellular carcinoma: a multicenter study. Surgery. 2019;165(3):516–524. doi: 10.1016/j.surg.2018.08.031. [DOI] [PubMed] [Google Scholar]
- 23.van Leeuwen MS, Fernandez MA, van Es HW, Stokking R, Dillon EH, Feldberg MA. Variations in venous and segmental anatomy of the liver: two- and three-dimensional MR imaging in healthy volunteers. AJR Am J Roentgenol. 1994;162(6):1337–1345. doi: 10.2214/ajr.162.6.8191995. [DOI] [PubMed] [Google Scholar]
- 24.Nayak SB, Deepthinath R, Kumar N, Shetty P, Kumar V, Aithal A, Shetty SD. Evaluation of numerical and positional variations of the hepatic veins: a cadaveric study. J Cardiovasc Echogr. 2016;26(1):5–10. doi: 10.4103/2211-4122.178468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fang CH, You JH, Lau WY, Lai EC, Fan YF, Zhong SZ, Li KX, Chen ZX, Su ZH, Bao SS. Anatomical variations of hepatic veins: three-dimensional computed tomography scans of 200 subjects. World J Surg. 2012;36(1):120–124. doi: 10.1007/s00268-011-1297-y. [DOI] [PubMed] [Google Scholar]
- 26.Mehran R, Schneider R, Franchebois P. The minor hepatic veins: anatomy and classification. Clin Anat. 2000;13(6):416–421. doi: 10.1002/1098-2353(2000)13:6<416::AID-CA4>3.0.CO;2-H. [DOI] [PubMed] [Google Scholar]
- 27.Sakaguchi T, Suzuki S, Inaba K, Fukumoto K, Takehara Y, Nasu H, Kamiya M, Yamashita S, Ushio T, Nakamura S, et al. Analysis of intrahepatic venovenous shunt by hepatic venography. Surgery. 2010;147(6):805–810. doi: 10.1016/j.surg.2009.11.009. [DOI] [PubMed] [Google Scholar]
- 28.Suh SW, Lee JM, You T, Choi YR, Yi NJ, Lee KW, Suh KS. Hepatic venous congestion in living donor grafts in liver transplantation: is there an effect on hepatocellular carcinoma recurrence? Liver Transplant. 2014;20(7):784–790. doi: 10.1002/lt.23877. [DOI] [PubMed] [Google Scholar]
- 29.Guo HJ, Wang K, Chen KC, Liu ZK, Al-Ameri A, Shen Y, Xu X, Zheng SS. Middle hepatic vein reconstruction in adult right lobe living donor liver transplantation improves recipient survival. Hepatobiliary Pancreat Dis Int. 2019;18(2):125–131. doi: 10.1016/j.hbpd.2019.01.006. [DOI] [PubMed] [Google Scholar]
- 30.Rhu J, Kim JM, Jeong WK, Choi GS, Joh JW. Venous outflow congestion is related to poor recurrence-free survival of living donor liver transplantation recipients with hepatocellular carcinoma—a retrospective study. Transpl Int. 2021;34(2):272–280. doi: 10.1111/tri.13792. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.