Abstract
Introduction
Studies have shown that exacerbation in chronic obstructive pulmonary disease (COPD) increases the risk of further exacerbations. Our aim was to investigate the impact of a single moderate exacerbation on the odds of subsequent exacerbations and death in GOLD B COPD patients.
Methods
This hospital-based nationwide, cohort study in Denmark included all patients ≥40 years of age with an in- and/or outpatient ICD-10 J44 diagnosis (COPD Register, 2008–2014). Index was date of first registered modified Medical Research Council (mMRC) score ≥2; baseline period was 12 months pre-index. At index, patients were grouped as: B0, no exacerbation; and B1, one moderate exacerbation during the previous year, and followed for three consecutive years in 2008–2017 for development of moderate- (short-term use of prednisolone or prednisone) and severe (emergency visit or hospitalization) exacerbations and death. Using B0 as reference, the odds ratio (OR) for exacerbation and death in GOLD B1 was estimated with multinominal logistic regression and a Cox model estimated the hazard ratio for exacerbation accounting for recurrent events.
Results
In total, 8,453 patients (mean age 70 years, 51% male) were included, of which GOLD B0 4,545 and GOLD B1 3,908 patients. During the 3-year follow-up, 34.1% and 24.9% of GOLD B0 and B1, respectively, had none or one moderate exacerbation whereas 61.9% and 71.2% of B0 and B1, respectively, had a severe trajectory with multiple moderate and/or a severe exacerbation or died. In B1 patients, the OR for 1 moderate, ≥2 moderate exacerbations, ≥1 severe exacerbation was 1.58 [CI 1.33–1.87], 2.60 [2.19–3.08], 2.08 [1.76–2.45], respectively, and 1.85 [1.57–2.17] for death compared with B0.
Conclusion
One moderate exacerbation in COPD patients with high symptom burden increases the odds of subsequent exacerbations and death during the three following years. The results emphasize the importance of preventing exacerbations in GOLD B patients.
Keywords: COPD, exacerbation, dyspnea, GOLD, mortality, epidemiology
Introduction
Chronic obstructive pulmonary disease (COPD) is one of the most important causes of morbidity and death in many countries.1 In most patients, COPD is a progressive disease where lung function and symptoms can be expected to worsen over time, a deterioration that may be accelerated by periods with exacerbations. It is well established, that exacerbations contribute to a more rapid disease progression, such as accelerated lung function decline, accelerated skeletal muscle loss, decreased health status, increased mortality, and imply a significant economic burden for the health care system as well - mainly driven by exacerbations.2–6 One single moderate exacerbation has been shown to increase the risk of hospitalization by 21%,7 and one out of five patients die within a year after their first hospitalization due to an exacerbation.8 To improve the prognosis of COPD and reduce the impact of the disease on the daily life of patients as well as the risk of long-term COPD-related morbidity and mortality, prevention of exacerbations is a key step in the COPD treatment goals.1
Due to the complexity and heterogeneity of COPD, and with the goal of improving clinical outcomes and prognosis for individual patients, the term “individualized” medicine has been proposed to define treatments targeted to the needs of individual patients based on eg individual biomarkers or phenotypic characteristics that distinguish a given patient from other patients with similar clinical presentations.9–11
The Global Initiative for Chronic Obstructive Lung Disease (GOLD) use clinical phenotyping to assess disease burden and future risk by grouping treatment naïve patients into GOLD A to D.1 GOLD recommends to use two components for individual COPD characterization and future risk assessment: exacerbation history (last 12 months) and symptom burden (CAT/ mMRC).1 However, it is recognized that COPD is a highly heterogenous and dynamic disease, even within the GOLD groups.12,13 This is particularly true for the GOLD B group, in which patients have a significant symptom burden (CAT ≥ 10 or a mMRC ≥ 2) and may have had either zero or one moderate exacerbation in the previous year. With this phenotyping, all patients characterized as GOLD B, irrespective of the level of symptoms above the cut-offs of CAT or mMRC, or whether they have had zero or one exacerbation, are considered to have the same disease impact and future risk. Hence, the treatment recommendations for this patient group are mainly focused on symptom treatment with either one or two long-acting bronchodilators.
Some GOLD B patients may deteriorate faster and in fact change GOLD category due to frequency of exacerbations.
The disease dynamics and future risk of the GOLD B risk group in real life is sparsely investigated outside clinical trials, which is mainly focused on frequent exacerbators. Therefore, the aim of this study was to investigate the impact of a single moderate exacerbation on the odds of subsequent exacerbations and death in GOLD B COPD patients by comparing patients with one moderate exacerbation during the previous year with those without.
Materials and Methods
Study Design and Data Sources
This was a population-based cohort study using various comprehensive national Danish health registries: (a) the National Hospital Registry covering all inpatient, non-psychiatric hospitalizations in Denmark since 1977 and all outpatient visits since 1995 with mandatory registration by the treating physician;14 (b) the National COPD Registry including all COPD patients followed in outpatient specialist clinics in Denmark since 2008;15 (c) the National Prescription Registry including all prescriptions redeemed at pharmacies since 1995 using anatomical therapeutic chemical (ATC) codes;16 (d) the Civil Registration System for information on vital status, migration and cohabitation,17 and (e) the Cause-of-death Register.18 Accurate individual linkage between the registries was possible by using the unique 10-digit personal identification number, assigned to all Danish residents. The study is purely registry-based, and all micro-data is anonymized, so no informed consent was needed. Data access was approved and delivered by Statistics Denmark (project number 704702). All analyses were performed at a secure research infrastructure server at Statistics Denmark, which has been set up to comply with the General Data Protection Regulation (GDPR) and the Danish Data Protection Agency guidelines for research on sensitive data.
Study Populations
The study base included all patients with COPD ≥40 years with a visit to a specialized hospital-based clinic for COPD in 2008–2014 (n=90,598). Patients with missing information on mMRC dyspnea score or incomplete registration of clinical outcome measures in the COPD registry were excluded (sFigure 1). To examine oral corticosteroid (OCS) use as a proxy for moderate COPD exacerbations, patients with a diagnosis of other conditions for which OCS is frequently prescribed were excluded (Crohn’s disease [K50], ulcerative colitis [K51], rheumatoid arthritis [M05], emphysema [J43], bronchiectasis [J47], cystic fibrosis [E84] or malignancy [C00–97]). Medical history data on OCS-related diagnoses were retrieved from the National Hospital Registry using ICD-10 codes from nine years prior to index date and current cancer diagnoses were identified in the 12 months prior to index. The ICD-10 was introduced in 1994 in Denmark and therefore covered the entire study period.
Baseline COPD Exacerbations and Dyspnea Score
To identify patients in GOLD group B, we used data available from the registry on the number and severity of COPD exacerbations in the 12 months prior to index date and data on index mMRC scores (sFigure 2) and combined those, in accordance with the most recent GOLD definition.1
Moderate exacerbations were captured using the Prescription Registry and defined as short-term use of OCS (corresponding to redeeming a prescription on a maximum of 20 defined daily doses (DDD) of 25mg prednisolone [ATC code H02AB06] or prednisone [ATC code H02AB07]). In Denmark, COPD exacerbations are treated with a daily Prednisolone dose between 25–37.5 mg (depending on weight, osteoporosis, age etc.) for 5–10 days. If patients collected more than one package of OCS on the same day, it was only counted as one exacerbation.
Severe exacerbations were identified in the Hospital Registry and defined as hospitalizations or emergency visits with ICD-10 code J40–44 as primary diagnosis or J13–18 (pneumonia) or J96 (respiratory insufficiency) as primary diagnosis in combination with a secondary diagnosis J40–44. Recurrent exacerbations within 28 days were combined and considered as one exacerbation. Patients with both a moderate and a severe exacerbation within 28 days were counted as having a severe exacerbation with the start date of the exacerbation that occurred first.
Information on dyspnea was obtained by the treating physician or nurse using the mMRC dyspnea scale19 and recorded in the National COPD Registry with a score ranging from 0 (not troubled by breathlessness except on strenuous exercise) to 4 (too breathless to leave the house, or breathless when dressing and undressing). The mMRC scores are used in clinical practice in Denmark and therefore these are available in the COPD Registry in contrast to CAT scores. By combining data on baseline exacerbations and mMRC scores, patients with mMRC ≥ 2 and zero or one moderate baseline exacerbation were identified and classified as GOLD B (n=8,453) (sFigure 1) and split into two groups; B0 (zero baseline exacerbations) and B1 (one moderate baseline exacerbation).
Comorbidity
Charlson Comorbidity Index (CCI)19 was used as a measure of comorbidities and based on diagnoses (ICD-10 codes) recorded up to 12 months prior to index as part of inpatient and outpatient hospital care.
Study Outcomes
Moderate and severe exacerbations were defined as described above and grouped as 0 exacerbations, 1 moderate exacerbation, ≥2 moderate exacerbations, ≥1 COPD-related hospital visit. All cause-mortality was investigated in each of the three years after index and over the total 3-year period. Causes of death were reported by main ICD-10 chapters over the total 3-year follow up period.
Statistical Analyses
Baseline characteristics were described as mean (standard deviation [SD]) for continuous variables and absolute and relative frequencies for categorical variables. Baseline characteristics were described during the baseline year or at the date of first mMRC measurement (index date) (sFigure 2). Baseline treatment was defined as respiratory medication collected during the four months prior to index.
Follow up for moderate exacerbations, severe exacerbations and death started on the day after the index date between January 1, 2008 and December 31, 2014. Follow up ended on the date of death, emigration, 36 months after index date, or end of study Dec 31, 2017, whichever occurred first. If a patient had several mMRC measurements during the study period, the first measurement was used as index. Patients who developed an OCS-related diagnosis were censored from analyses on the date of diagnosis.
Logistic regression models were applied to estimate odds ratios (OR) with 95% CIs of moderate exacerbation, severe exacerbation and death in B1 with B0 as reference for each year of follow up and adjusted for age, sex, cohabitation status, comorbidity, BMI and smoking. A cumulated measure of outcomes over the entire three year follow up was calculated with each patient being categorized according to the most severe event they experienced during follow up.
The hazard ratio of exacerbations accounting for recurrent exacerbations was estimated using a Cox proportional hazards model with 95% confidence intervals and GOLD B0 as the reference group. Recurrent events were included in the model as a covariate. Furthermore, the model was adjusted for age, sex, cohabitation status, comorbidity, BMI and smoking and death was handled as a competing event using the Fine & Gray method.20
All analyses were performed using SAS 9.4 TS Level 1m5, and all p-values <0.05 were considered statistically significant.
Results
Overall, 4,545 GOLD B0 patients (mean age 70.1 years, 51.1% male) and 3,908 GOLD B1 patients (mean age 69.9 years, 49.9% male) were included (Table 1). The mMRC score was similar in both groups with more than half of patients having a score of 2 and approximately 30% and 14% having an mMRC of 3 and 4, respectively (Table 1). 30.3% and 24.7% of patients in GOLD B0 and B1, respectively, did not collect any respiratory medication in the 4 months prior to index and 15.1% and 15.9% in GOLD B0 and B1 were only treated with one type of medication (Table 1). In GOLD B0, 4.6% collected LAMA/LABA and 33.2% collected triple treatment which was similar in GOLD B1 where 4.9% collected LAMA/LABA and 37.1% collected triple treatment.
Table 1.
Baseline Characteristics of Patients in GOLD B, Split into GOLD B0 and B1
| GOLD B0 | GOLD B1 | P-value | |||
|---|---|---|---|---|---|
| n | % | n | % | ||
| Patients | 4,545 | 3,908 | |||
| Sex | |||||
| Male | 2,323 | 51.1 | 1,951 | 49.9 | 0.276 |
| Female | 2,222 | 48.9 | 1,957 | 50.1 | |
| Cohabitation status | |||||
| Not cohabitating | 2,398 | 52.8 | 1,918 | 49.1 | <0.001 |
| Cohabitating | 2,147 | 47.2 | 1,990 | 50.9 | |
| Age (years) | |||||
| Mean age (SD) | 70.1 (10.2) | 69.9 (10.2) | 0.430 | ||
| 40–49 | 152 | 3.3 | 122 | 3.1 | 0.712 |
| 50–59 | 599 | 13.2 | 551 | 14.1 | |
| 60–69 | 1,264 | 27.8 | 1,100 | 28.1 | |
| 70–79 | 1,683 | 37.0 | 1,423 | 36.4 | |
| 80+ | 847 | 18.6 | 712 | 18.2 | |
| mMRC score | |||||
| 2 | 2,556 | 56.2 | 2,143 | 54.8 | 0.422 |
| 3 | 1,347 | 29.6 | 1,189 | 30.4 | |
| 4 | 642 | 14.1 | 576 | 14.7 | |
| FEV1 (% of predicted) | |||||
| 80+ | 185 | 4.1 | 127 | 3.2 | 0.114 |
| 50–79 | 1,722 | 37.9 | 1,436 | 36.7 | |
| 30–49 | 1,904 | 41.9 | 1,691 | 43.3 | |
| <30 | 734 | 16.1 | 654 | 16.7 | |
| BMI (kg/m2) | |||||
| <18 | 458 | 10.1 | 380 | 9.7 | 0.879 |
| 18–24 | 1,609 | 35.4 | 1,373 | 35.1 | |
| 25–29 | 1,328 | 29.2 | 1,129 | 28.9 | |
| 30–34 | 728 | 16.0 | 644 | 16.5 | |
| 35+ | 422 | 9.3 | 382 | 9.8 | |
| Smoking status | |||||
| Not current smoker | 2,903 | 63.9 | 2,519 | 64.5 | 0.576 |
| Current smoker | 1,642 | 36.1 | 1,389 | 35.5 | |
| Baseline treatment* | |||||
| No claims | 1,376 | 30.3 | 966 | 24.7 | <0.001 |
| LAMA | 561 | 12.3 | 440 | 11.3 | |
| LABA | 163 | 3.6 | 150 | 3.8 | |
| LABA/ICS | 727 | 16.0 | 709 | 18.1 | |
| LABA/LAMA | 211 | 4.6 | 193 | 4.9 | |
| LABA/LAMA/ICS | 1,507 | 33.2 | 1,450 | 37.1 | |
| Charlson Comorbidity Index (CCI) | |||||
| Mean (SD) | 0.36 (0.89) | 0.36 (0.88) | 0.7190 | ||
Note: *Medication claimed 4 months prior to index.
Abbreviations: SD, standard deviation; mMRC, modified Medical Research Council; FEV1, forced expired volume in the first second; BMI, body mass index; LAMA, long-acting muscarinic antagonists; LABA, long-acting beta-agonists; ICS, inhaled corticosteroids.
During each year of follow up, the proportion of B0 patients that had no exacerbations were between 32.5–43.9% and over the entire period 15% of B0 patients remained exacerbation-free (Table 2). For B1 patients the proportion of patients with no exacerbations in year 1, 2 and 3 were lower - between 24.9–32.9% and only 8% remained exacerbation-free during all three years. Over the three year follow up 34.1% of B0 patients and 24.9% of B1 patients had zero or one moderate exacerbation. The mean cumulative number of exacerbations during three years of follow up were 3.01 (SD 2.6) in GOLD B0 and 3.97 (2.9) in GOLD B1.
Table 2.
Frequency of Moderate and Severe Exacerbations and Death During Each Year of Follow Up and Cumulated Number Events Over All Three Years for GOLD B0 and GOLD B1 Patients
| Year 1 | Year 2 | Year 3 | Total Follow Up* | |||||
|---|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | n | % | |
| GOLD B0 | ||||||||
| No exacerbations | 1,996 | 43.9 | 1,785 | 39.3 | 1,477 | 32.5 | 681 | 15.0 |
| 1 moderate exacerbation | 936 | 20.6 | 742 | 16.3 | 719 | 15.8 | 869 | 19.1 |
| ≥2 moderate exacerbations | 502 | 11.0 | 470 | 10.3 | 398 | 8.8 | 619 | 13.6 |
| ≥1 Severe exacerbation | 516 | 11.4 | 502 | 11.0 | 468 | 10.3 | 893 | 19.6 |
| Dead | 501 | 11.0 | 912 | 20.1 | 1,306 | 28.7 | 1,306 | 28.7 |
| OCS censoring** | 94 | 2.1 | 134 | 2.9 | 177 | 3.9 | 177 | 3.9 |
| GOLD B1 | ||||||||
| No exacerbations | 1,284 | 32.9 | 1,110 | 28.4 | 975 | 24.9 | 321 | 8.2 |
| 1 moderate exacerbation | 894 | 22.9 | 742 | 19.0 | 619 | 15.8 | 653 | 16.7 |
| ≥2 moderate exacerbations | 698 | 17.9 | 588 | 15.0 | 587 | 15.0 | 774 | 19.8 |
| ≥1 Severe exacerbation | 537 | 13.7 | 563 | 14.4 | 455 | 11.6 | 888 | 22.7 |
| Dead | 403 | 10.3 | 783 | 20.0 | 1,121 | 28.7 | 1,121 | 28.7 |
| OCS censoring | 92 | 2.4 | 122 | 3.1 | 151 | 3.9 | 151 | 3.9 |
| p-value | <0.001 | <0.001 | <0.001 | <0.001 | ||||
Notes: *Cumulated number of events over the three years. Events were counted hierarchical, so an individual will count in the group with most severe events (moderate – severe – dead). **Censoring due to development of an OCS-related diagnosis during follow up (Crohn’s disease [K50], ulcerative colitis [K51], rheumatoid arthritis [M05], emphysema [J43], bronchiectasis [J47], cystic fibrosis [E84] or malignancy [C00–97]).
Abbreviation: OCS, oral corticosteroid.
In GOLD B0 and B1, 61.9% and 71.2% had a severe trajectory with two or more moderate exacerbations or one or more severe exacerbations or death (Table 2).
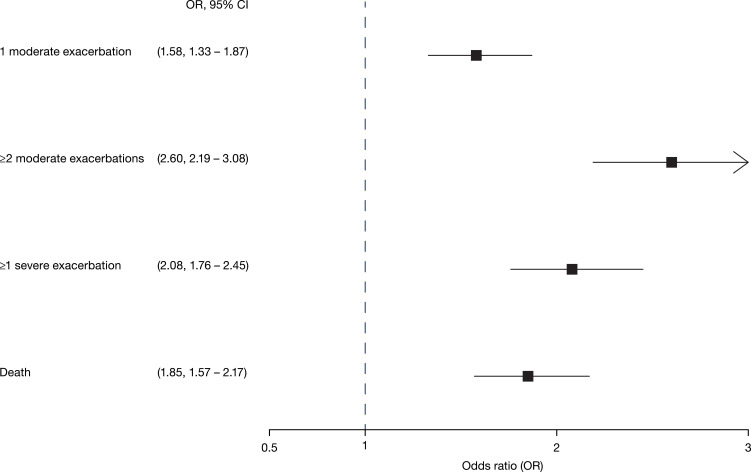
Having one moderate exacerbation in the previous year increased the odds of experiencing a new moderate exacerbation for each of the following years (Table 3) and over the three year-period the OR of a new moderate exacerbation in B1 patients was 1.58 (95% CI 1.33–1.87) compared to B0 patients adjusting for age, sex, cohabitation status, comorbidity, BMI and smoking (Figure 1). The same pattern was seen with more than doubled odds for ≥2 moderate exacerbations and for a severe exacerbation over the three year-period (Figure 1).
Table 3.
Odds Ratio (OR) of Exacerbations and Death in GOLD B1 Compared with GOLD B0 in Year 1, Year 2 and Year 3 Separately and in All Three Years Combined
| Year 1 | Year 2 | Year 3 | Total Follow Up§ | |
|---|---|---|---|---|
| OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | |
| 1 moderate | 1.48 (1.31–1.66) | 1.60 (1.41–1.82) | 1.29 (1.12–1.47) | 1.58 (1.33–1.87) |
| ≥2 moderate | 2.13 (1.86–2.44) | 2.00 (1.72–2.29) | 2.19 (1.88–2.60) | 2.60 (2.19–3.08) |
| ≥1 severe | 1.60 (1.39–1.84) | 1.80 (1.56–2.08) | 1.46 (1.25–1.70) | 2.08 (1.76–2.45) |
| Dead* | 1.26 (1.08–1.48) | 1.42 (1.24–1.62) | 1.32 (1.16–1.49) | 1.85 (1.57–2.17) |
Notes: §Cumulated number of events over the three years. Events were cumulated over the three-year period and counted hierarchical, so an individual with multiple events counted in the group with most severe events (moderate – severe – dead). All ORs are adjusted for age, sex, cohabitation status, comorbidity, BMI and smoking. All p-values comparing B0 and B1 were <0.05. *The number of dead patients was cumulated for all three years.
Figure 1.
Odds ratio (OR) with 95% CI of exacerbations and death after 3-years follow up in GOLD B1 with GOLD B0 as reference. Analyses adjusted for age, sex, cohabitation status, comorbidity, BMI and smoking.
Abbreviation: OR, odds ratio.
Taking recurrent exacerbations into account and adjusting for competing risk of dying, the hazard ratio of any exacerbation during follow up was still increased in B1 compared with B0 (HR 1.21, 1.18–1.25) (Table 4).
Table 4.
Risk of Exacerbation During the Three Year Follow Up, Accounting for Recurrent Events
| Hazard Ratio* | Lower 5% CI | Upper 95% CI | P-value | |
|---|---|---|---|---|
| GOLD B group | ||||
| B0 | Ref | |||
| B1 | 1.214 | 1.181 | 1.248 | <0.001 |
| Age | 1.004 | 1.003 | 1.006 | <0.001 |
| Sex | ||||
| Male | Ref | |||
| Female | 1.041 | 1.012 | 1.071 | 0.0051 |
| Cohabitation status | ||||
| Not cohabitating | Ref | |||
| Cohabitating | 1.079 | 1.050 | 1.110 | <0.001 |
| mMRC symptom score | ||||
| 2 | Ref | |||
| 3 | 1.055 | 1.023 | 1.087 | <0.001 |
| 4 | 1.145 | 1.100 | 1.191 | <0.001 |
| FEV1 (% predicted) | ||||
| 80+ | Ref | |||
| 50–79 | 1.074 | 0.984 | 1.173 | 0.1095 |
| 30–49 | 1.265 | 1.160 | 1.381 | <0.001 |
| <30 | 1.420 | 1.297 | 1.555 | <0.001 |
| BMI (kg/m2) | ||||
| <18 | Ref | |||
| 18–24 | 0.916 | 0.873 | 0.961 | <0.001 |
| 25–29 | 0.875 | 0.832 | 0.920 | <0.001 |
| 30–34 | 0.853 | 0.807 | 0.901 | <0.001 |
| 35+ | 0.893 | 0.839 | 0.951 | <0.001 |
| Smoking | ||||
| Smoking present | Ref | |||
| Never smoked | 0.853 | 0.807 | 0.901 | <0.001 |
| Smoking earlier | 0.893 | 0.839 | 0.951 | <0.001 |
| Comorbidity (CCI) | 1.010 | 0.994 | 1.027 | 0.2306 |
Note: *Adjusted for age, sex, cohabitation status, comorbidity, BMI and smoking.
Abbreviations: CI, confidence Interval; mMRC, modified Medical Research Council; FEV1, forced expired volume in the first second; BMI, body mass index.
Each year, approximately 10% of patients died and over the three years 28.7% of patients died in both groups. However, the OR of death was 1.85 (95% CI, 1.57–2.17) after three years in GOLD B1 compared with B0 when adjusting for age, sex, cohabitation status, comorbidity, BMI and smoking (Table 3). Causes of death were similar in the two groups with the main cause of death being COPD in both groups (B0, 43.8%; B1, 42.8%), followed by cardiovascular disease and neoplasms (sTable 1). Risk of death increased with higher mMRC score and with decreasing lung function and highest risk was seen in patients with FEV1 below 30%.
Discussion
In this nationwide cohort study, we showed that despite being low risk categorized as COPD GOLD B at the start of the study, more than a third of the patients had a severe disease trajectory with multiple moderate and/or severe exacerbations or death after three years. Among the GOLD B patients with one moderate baseline exacerbation, we demonstrated increased odds of subsequent moderate and severe exacerbations compared with patients with no exacerbations during the previous year. Furthermore, one moderate exacerbation during the baseline year was associated with an increased risk of death. Even when analyzing recurrent exacerbations with adjustment for competing risk of dying, the hazard of subsequent exacerbations remained significantly increased.
A major goal in the management of COPD is to prevent exacerbations since they are associated with increased morbidity and mortality.1 Patients characterized as GOLD B are regarded as a low-risk patient group. The results from the present study demonstrate that GOLD B is a heterogenous group, in which a large part of patients have a severe disease trajectory or even die, indicating the need to also look beyond the GOLD classification when deciding for treatment decisions at individual levels. In clinical practice in the Nordic countries the GOLD classification is utilized not only in the initial phase at diagnosis but throughout the disease management. COPD has multiple factors associated with increased risk of future exacerbations and mortality, such as smoking, chronic bronchitis, level of FEV1, dyspnea grade and COPD Assessment Test (CAT) score,21,22 which also should be included in the assessment of treatment for an individual patient.
There is mounting evidence showing the heterogeneity of clinical features and disease course variability in COPD, and that a single moderate exacerbation in many patients is associated with increased risk of future moderate or severe exacerbations and death.7,12,13,23 In the ECLIPSE cohort study, only 36% of GOLD B patients remained stable after a 1-year follow-up, 7% and 35% deteriorated to the higher risk categories, C and D respectively, and 22% improved to GOLD A.12 In the study by Lawrence et al, one quarter of the GOLD B patients progressed to GOLD group D during a one year follow-up,13 and in a recent long-term study, an almost linear relationship between exacerbation frequency and mortality was presented, with increased mortality risk already at an average of 0.2 moderate exacerbation per year, ie one moderate exacerbation every fifth year is associated with increased mortality.23
As most patients at some point experience multiple exacerbations, an analysis adjusted for competing risk of death taking all exacerbations into account showed that the risk of future exacerbations in GOLD B1 remained increased with 21% compared to GOLD B0.
A surprising finding in our hospital-based study is the fact that more than every fourth patient, despite being symptomatic, did not collect any inhalation medication in the four months prior to inclusion. Furthermore, approximately 15% only collected long-acting bronchodilator monotherapy (LABA or LAMA). These numbers are surprisingly high, considering the outpatient setting and the dyspnea-score (mMRC ≥ 2). The low collection could partly be due to poor adherence to the medication plan resulting in lower use, or medication stockpiling at home, but likely only valid for a limited number of patients. Alternatively, that patients were diagnosed with COPD for the first time at index and therefore did not collect medication before that date. However, as the study is hospital-based, this is likely only true for few patients. It has been shown previously that GOLD B patients to a large extent either do not use any treatment or only use short- or long acting bronchodilator monotherapy.24–26 These findings highlight that undertreatment among GOLD B patients seems to be a major issue as well as a consistent finding across studies. Therefore, more effort is needed to ensure correct medication and dosing adherence for symptomatic GOLD B patients.
Over the three year follow up, our study showed that the odds ratio of death was 1.85 among GOLD B1 patients compared with GOLD B0. Also, the overall mortality of 28.7% is similar to a previous Danish study, reporting a 3-year all-cause mortality of 23.8% in hospital GOLD B out-patients.27
The major strengths of this study are the high-quality nationwide registry data and personal identification numbers allowing accurate linkage of individual data in addition to clinical data such as mMRC scores from the National COPD Register enabling identification of GOLD B risk patients. However, when interpreting the results, one should be aware that patients included in this study were identified in a hospital setting. So, even though the study is population-based, it reflects a more symptomatic GOLD B population compared to patients followed only in primary care. In our study, approximately 15% in both groups had an mMRC score of 4. One should also note that there were differences in collection of baseline medication for B0 and B1 patients, which may reflect disease severity differences.
Conclusions
We have shown that one moderate exacerbation in GOLD B COPD patients increased the odds of subsequent exacerbations and death; and that the increased odds ratio was sustained during the three-year study period. This highlights the importance of identifying these patients and optimizing the treatment to prevent future negative outcomes. To improve the current management of COPD, a better risk assessment and a more individualized approach is required. The results from this study and others may have implications for treatment recommendations to guide therapy in COPD.
Funding Statement
This study was sponsored by AstraZeneca.
Data Sharing Statement
The dataset supporting the conclusions of this article are available from Public registries upon request and approval of access to national authorities.
Ethics Approval and Consent to Participate
This study was performed in accordance with the Declaration of Helsinki, the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use Good Clinical Practices, Good Pharmacoepidemiology Practice and the applicable legislation on non-interventional studies and/or observational Studies. All data accessed complied with relevant data protection and privacy regulations.
Consent for Publication
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
A.L., J.L., R.I., and O.H. reports no conflict of interests in relations to the current study. P.L. has participated in advisory boards for AstraZeneca, Boehringer Ingelheim, Chiesi and GSK, and has received research grants from AstraZeneca, Boehringer Ingelheim and GSK. S.d.F.L. and G.S. are full time employees of AstraZeneca. The authors report no other conflicts of interest in this work.
References
- 1.GOLD. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease; 2021.
- 2.Hansel TT, Barnes PJ. New drugs for exacerbations of chronic obstructive pulmonary disease. Lancet. 2009;374(9691):744–755. doi: 10.1016/S0140-6736(09)61342-8 [DOI] [PubMed] [Google Scholar]
- 3.Seemungal TA, Donaldson GC, Paul EA, Bestall JC, Jeffries DJ, Wedzicha JA. Effect of exacerbation on quality of life in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1998;157(5 Pt 1):1418–1422. doi: 10.1164/ajrccm.157.5.9709032 [DOI] [PubMed] [Google Scholar]
- 4.Soler-Cataluna JJ, Martinez-Garcia MA, Roman Sanchez P, Salcedo E, Navarro M, Ochando R. Severe acute exacerbations and mortality in patients with chronic obstructive pulmonary disease. Thorax. 2005;60(11):925–931. doi: 10.1136/thx.2005.040527 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Løkke A, Lange P, Lykkegaard J, et al. Economic burden of COPD by disease severity - A nationwide cohort study in Denmark. Int J Chron Obstruct Pulmon Dis. 2021;16:603–613. doi: 10.2147/COPD.S295388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mason SE, Moreta-Martinez R, Labaki WW, et al. Respiratory exacerbations are associated with muscle loss in current and former smokers. Thorax. 2021;76(6):554–560. doi: 10.1136/thoraxjnl-2020-215999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rothnie KJ, Mullerova H, Smeeth L, Quint JK. Natural history of chronic obstructive pulmonary disease exacerbations in a general practice-based population with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2018;198(4):464–471. doi: 10.1164/rccm.201710-2029OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ho TW, Tsai YJ, Ruan SY, Huang CT, Lai F, Yu CJ. In-hospital and one-year mortality and their predictors in patients hospitalized for first-ever chronic obstructive pulmonary disease exacerbations: a nationwide population-based study. PLoS One. 2014;9(12):e114866. doi: 10.1371/journal.pone.0114866 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Agustí A, Celli B. Natural history of COPD: gaps and opportunities. ERJ Open Res. 2017;3(4):00117–2017. doi: 10.1183/23120541.00117-2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ulrik CS, Vijverberg S, Hanania NA, Diamant Z. Precision medicine and treatable traits in chronic airway diseases - where do we stand? Curr Opin Pulm Med. 2020;26(1):33–39. doi: 10.1097/MCP.0000000000000639 [DOI] [PubMed] [Google Scholar]
- 11.Agusti A, Bel E, Thomas M, et al. Treatable traits: toward precision medicine of chronic airway diseases. Eur Respir J. 2016;47(2):410–419. doi: 10.1183/13993003.01359-2015 [DOI] [PubMed] [Google Scholar]
- 12.Agusti A, Edwards LD, Celli B, et al. Characteristics, stability and outcomes of the 2011 GOLD COPD groups in the ECLIPSE cohort. Eur Respir J. 2013;42(3):636–646. doi: 10.1183/09031936.00195212 [DOI] [PubMed] [Google Scholar]
- 13.Lawrence PJ, Kolsum U, Gupta V, et al. Characteristics and longitudinal progression of chronic obstructive pulmonary disease in GOLD B patients. BMC Pulm Med. 2017;17(1):42. doi: 10.1186/s12890-017-0384-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schmidt M, Schmidt SA, Sandegaard JL, Ehrenstein V, Pedersen L, Sorensen HT. The Danish National Patient Registry: a review of content, data quality, and research potential. Clin Epidemiol. 2015;7:449–490. doi: 10.2147/CLEP.S91125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lange P, Tottenborg SS, Sorknaes AD, et al. Danish register of chronic obstructive pulmonary disease. Clin Epidemiol. 2016;8:673–678. doi: 10.2147/CLEP.S99489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pottegard A, Schmidt SAJ, Wallach-Kildemoes H, Sorensen HT, Hallas J, Schmidt M. Data resource profile: the Danish National Prescription Registry. Int J Epidemiol. 2017;46(3):798–798f. doi: 10.1093/ije/dyw213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schmidt M, Pedersen L, Sorensen HT. The Danish civil registration system as a tool in epidemiology. Eur J Epidemiol. 2014;29(8):541–549. doi: 10.1007/s10654-014-9930-3 [DOI] [PubMed] [Google Scholar]
- 18.Helweg-Larsen K. The Danish register of causes of death. Scand J Public Health. 2011;39(7 Suppl):26–29. doi: 10.1177/1403494811399958 [DOI] [PubMed] [Google Scholar]
- 19.Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383. doi: 10.1016/0021-9681(87)90171-8 [DOI] [PubMed] [Google Scholar]
- 20.Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94(446):496–509. doi: 10.1080/01621459.1999.10474144 [DOI] [Google Scholar]
- 21.Punekar YS, Mullerova H, Small M, et al. Prevalence and burden of dyspnoea among patients with chronic obstructive pulmonary disease in five European countries. Pulm Ther. 2016;2(1):59–72. doi: 10.1007/s41030-016-0011-5 [DOI] [Google Scholar]
- 22.Casanova C, Marin JM, Martinez-Gonzalez C, et al. Differential effect of modified medical research council dyspnea, COPD assessment test, and clinical COPD questionnaire for symptoms evaluation within the new GOLD staging and mortality in COPD. Chest. 2015;148(1):159–168. doi: 10.1378/chest.14-2449 [DOI] [PubMed] [Google Scholar]
- 23.Larsson K, Janson C, Lisspers K, et al. The impact of exacerbation frequency on clinical and economic outcomes in Swedish COPD patients: the ARCTIC study. Int J Chron Obstruct Pulmon Dis. 2021;16:701–713. doi: 10.2147/COPD.S297943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sundh J, Åberg J, Hasselgren M, et al. Factors influencing pharmacological treatment in COPD: a comparison of 2005 and 2014. Eur Clin Respir J. 2017;4(1):1409060. doi: 10.1080/20018525.2017.1409060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Graf J, Jörres RA, Lucke T, Nowak D, Vogelmeier CF, Ficker JH. Medical treatment of COPD. Dtsch Arztebl Int. 2018;155(37):599–605. doi: 10.3238/arztebl.2018.0599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ding B, Small M, Holmgren U. A cross-sectional survey of current treatment and symptom burden of patients with COPD consulting for routine care according to GOLD 2014 classifications. Int J Chron Obstruct Pulmon Dis. 2017;12:1527–1537. doi: 10.2147/COPD.S133793 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gedebjerg A, Szépligeti SK, Wackerhausen LH, et al. Prediction of mortality in patients with chronic obstructive pulmonary disease with the new global initiative for chronic obstructive lung disease 2017 classification: a cohort study. Lancet Respir Med. 2018;6(3):204–212. doi: 10.1016/S2213-2600(18)30002-X [DOI] [PubMed] [Google Scholar]