Abstract
Objective
This study aimed to determine population-based prevalence of chronic kidney disease (CKD) and its associated factors in Nepal.
Study design
The study was a nationwide population-based cross-sectional study.
Setting and participants
Cross-sectional survey conducted in a nationally representative sample of 12 109 Nepalese adult from 2016 to 2018 on selected chronic non-communicable diseases was examined. Multistage cluster sampling with a mix of probability proportionate to size and systematic random sampling was used for the selection of individuals aged 20 years and above.
Primary and secondary outcome measures
Primary outcome in this study was population-based prevalence of CKD in Nepal. A participant was considered to have CKD if the urine albumin-to-creatinine ratio was greater than or equal to 30 mg/g and/or estimated glomerular filtration rate is less than 60 mL/min/1.73 m2 at baseline and in follow-up using modification of diet in renal disease study equations. The secondary outcome measure was factors associated with CKD in Nepal. The covariate adjusted association of risk factors and CKD was calculated using multivariable binary logistic regression.
Results
The overall prevalence of CKD in Nepal was 6.0% (95% CI 5.5 to 6.6). Factors independently associated with CKD included older age (adjusted OR (AOR) 2.6, 95% CI 1.9 to 3.6), Dalit caste (AOR 1.6, 95% CI 1.1 to 2.3), hypertension (AOR 2.4, 95% CI 2.0 to 3.0), diabetes mellitus (AOR 3.2, 95% CI 2.5 to 4.1), raised total cholesterol (AOR 1.3, 95% CI 1.0 to 1.6) and increased waist-to-hip ratio (AOR 1.6, 95% CI 1.2 to 2.3).
Conclusion
This nationally representative study shows that the prevalence of CKD in the adult population of Nepal is substantial, and it is independently associated with several cardiometabolic traits. These findings warrant longitudinal studies to identify the causes of CKD in Nepal and effective strategies to prevent it.
Keywords: chronic renal failure, dialysis, glomerulonephritis, renal transplantation
Strengths and limitations of this study.
This is the first large scale nationwide population-based prevalence of chronic kidney disease (CKD) in Nepal.
Strict training processes and vigorous quality assurance programmes were used to ensure the quality of data collection
The use of standardised definitions of CKD facilitates international comparisons of CKD prevalence and risk factors.
High overall response rate of 91.7. % in first visit and 86.9% in follow-up visit.
Causal inference is unknown.
Background
Chronic kidney disease (CKD) is one of the most rapidly growing non-communicable diseases (NCDs) worldwide.1 According to the Global Burden of Disease (GBD) study, CKD was the 11th leading cause of death in 2019, up from 19th in 1990 accounting for 2.53% of total deaths.2 CKD is also associated with substantial morbidity, mortality and healthcare costs. Although it is largely preventable, over 9% of the population worldwide is estimated to be affected by the condition, particularly developing countries,3 4 and it is associated with diabetes type II, hypertension and obesity, which are also growing at an alarming rate. The rapid increase of CKD is likely to impose a great socioeconomic and public health burden in in resource-poor setting.5
The evidence on the prevalence of CKD in South East Asia is relatively limited, but it consistently shows a high prevalence of CKD6–8; however, data remain poorly characterised due to inconsistent assessment of kidney function and non-standard approaches which might distort the true estimates of CKD prevalence. There are few studies in Nepal already warned the higher prevalence of CKD9–11 and reported data largely depends on the population studied (rural/urban/or general/at high-risk group) methods and the lack of representativeness from the general population. Nepal has overcome many of the critical health challenges to survive the first 5 years of life, and that NCDs and burden of potential underlying risk factors such as obesity, hypertension and diabetes and unhealthy lifestyle habits including poor diets are highly prevalent.12
Understanding the burden and risk factors associated with CKD is important for making healthcare planning, designing screening strategies and prevention of these diseases in this resource constrains setting, where access to renal replacement therapy is costly. Therefore, this study aimed to determine the population-based prevalence of CKD and its associated factors in Nepal.
Methods
Study design and subjects
The Nepal Health Research Council designed and implemented the ‘National Population based Prevalence Survey of Selected NCDs in Nepal’. This was a population-based survey to investigate the prevalence of NCDs including CKD, chronic obstructive pulmonary disease carried out between 2016 and 2018 in adult’s ≥20 years old from seven provinces of the country. Full details of the design and protocols of the survey are available elsewhere.13
Participants, sample size and study setting
The sample size for the survey was calculated taking as reference the prevalence of raised blood glucose (p=4%) from NCD risk factors: STEPwise approach to Surveillance (STEPS) survey 2013,14 Z value of 1.96 at 95% CI and margin of error (d) of 20%, design effect of 2, adjusting the sample across three domains of the Terai, hills and mountains and adding a non-response rate of 20% yielded a sample size of 12 965. With a plan to enrol 33 participants in each cluster (400), the final sample size was 13 200. A ward (lowest administrative unit of the-then Village Development Committees and Municipalities) was considered as a cluster-primary sampling unit (PSU) of the study design. With the support from Central Bureau of Statistics, a total of 400 clusters were sampled. To select a representative sample of cluster, the rural and urban areas within each region were identified as the main sampling strata and the sample was selected in two stages in which ecological belts (Terai, Hill and Mountains) and five development regions (Central, Eastern, Mid-Western, Western and Far western), was taken into account.
The survey team members in the field used official or socially mapped household list to select 33 households (secondary sampling units) from each cluster using systematic random sampling. One participant out of the eligible candidates (≥20 years and above, resident in the study area at least 6 months and able to provide informed consent) was selected to take part in the survey using the “Kish method” . Of 13 200 participants who were approached for interview, 12 557 responded to the invitation in day 1. Among 12 557 who accepted the invitation, only 12 148 participants responded in day 2 of clinical setting. For the present study, data from 12 109 subjects were available for analyses with a response rate of 91.7%, 39 (0.32%) were excluded, because they refused providing blood and urine samples to evaluate renal function in day 2.
Data weighting was carried out to make the sampled population comparable to the national population. Data weighting was done using sampling weight. Sample weighting was carried out for probabilities of selection of PSU (ward/cluster), selection of households and selection of an individual in a household using 2011 population for Nepal with the support from Central Bureau of Statistics.
Data collection and measurements
Data collection was performed in 400 clusters (figure 1) within 72 districts of Nepal. Data collection teams at each site consisted of five member having academic background of nursing, general medicine, bachelor in medical laboratory technology or public health.
Figure 1.
Map of Nepal showing the study clusters (400 clusters)
Participants’ appointment for face-to-face personal interview at convenient and accessible site or at home on two occasions (day 1 and day 2): following written informed consent, from the participants in day 1, a structured questionnaire was administered to collect information about participants’ general health and sociodemographic characteristics. Participants self-reported their health and sociodemographic status, personal and family medical histories and history of medicine used. Information concerning their lifestyle factors (cigarette smoking, alcohol intake) was collected, and then physical and clinical measurements were performed. The clinical examination included measurements of height, weight, waist hip ratio and blood pressure (BP) following standard protocols. Height was measured in centimetres with a portable Bioplus stature metre and weight with a portable digital seca 874 weighing scale (Seca, Germany) and recorded in kilograms ensuring that the participant was wearing light clothes and was without footwear. Waist circumference was measured using Seca tape in cm at the level midway between the twelfth rib and the uppermost lateral border of the iliac crest during normal expiration. BP was measured at least three times with a minimum 3 min, and then averaged to be recorded by using an Omron digital automatic BP monitor model HEM-8712 (Omron Health Care Co., Japan) with appropriate sized cuffs.15 Participants were classified as hypertensive when the raised BP is defined as having systolic BP ≥140 mm Hg and/or diastolic BP ≥90 mm Hg during the study, or being previously diagnosed as having hypertension.
We used fasting, at least an 8-hour fast, and 2-hour post prandial (PP) blood sample to test various biochemical parameters. Following aseptic technique, blood samples were drawn by a trained enumerators using vacutainers to test laboratory parameters such as fasting blood glucose level, PP blood glucose level, serum creatinine, total cholesterol (TC), triglyceride (TG), high density lipoprotein (HDL) cholesterol and serum creatinine. The well-trained enumerators collected around 10 mL of blood sample at the fasting state and provided 82 g of glucose monohydrate (equivalent to 75 g of anhydrous glucose) in 250 mL water to drink. Second blood sample was collected at 2 hours of glucose intake. Fasting blood glucose, TC, TG, HDL cholesterol and serum creatinine were measured with the fasting sample and 2-hour PP sample was used for measuring PP blood glucose only. We carried out biochemical analysis with Biolyzer 100 Clinical Chemistry Analyzer (semiautomatic biochemistry analyzer, Analyticon, Germany) and used glucose oxidase-peroxidase method for blood glucose, cholesterol oxidase/phenol aminophenazone (CHOD-PAP) for TC, glycerol-3-phosphate oxidase/phenol aminophenazone for TG, HDL cholesterol by polyethylene glycol/cholesterol oxidase/phenol aminophenazone and serum creatinine measured by the Jaffe Reaction method. The laboratory supervisors calculated the low-density lipoprotein cholesterol by Freidewald’s Formula using TC, TG and HDL cholesterol.
Outcome measurements
A spot urine sample was collected from single-voided specimen to measure urine albumin and creatinine using the same semiautomatic biochemistry analyser (AnalyticonBiolyzer 100 Clinical Chemistry Analyzer, Germany). Laboratory supervisors measured urine albumin concentration by Turbidimetric tests and urine creatinine concentration by the Jaffe Reaction method and finally albumin-creatinine ration (ACR) was calculated and expressed in mg/g. Renal function was evaluated by using estimated GFR (eGFR), based on the widely used 4-variable Modification of Diet in Renal Disease Study (MDRD) equation. A participant was considered to have CKD if the urine albumin-to-creatinine ratio (uACR) was greater than or equal to 30 mg/g and/or e-GFR was less than 60 mL/min/1.73 m2 at baseline and in follow up.16 The secondary outcome measure was factors associated with CKD in Nepal
Follow-up of study participants to determine CKD
Participants having either ACR greater than or equal to 30 mg/g in the baseline and/or decreased eGFR (<60 mL/min/1.73 m2) were revisited again after 3 months in their place of residence to ensure the chronicity of the renal disease. Written consent was taken prior to blood and urine sample collection using similar procedures as mentioned above. Out of 1382 participants having a high ACR ratio and/or decreased eGFR, 1202 gave consent. However, data of ACR was missing for 8 participants making the availability of complete data for 1194 participants for analysis, with a response rate of 86.9%.
Data management and analysis
We used android mobile phones inbuilt with data collection software (REMO) to collect data digitally. Data cleaning including correction of inconsistencies was performed in SPSS V.20.0. We then exported the dataset to STATA V.13 for analysis. To adjust for unequal probability of selection, appropriate sampling weights with complex sampling analysis were used to provide valid estimates for the relevant Nepalese population which is based on 2011 national population and housing census data. Results are expressed as a percentage and OR with 95% CI. Bivariate analysis was conducted to analyse the unconditional association between each explanatory variable and CKD status. To be included in multivariable modelling, the level of significance α=0.25 was used during bivariate analysis (ie, p value 0.25 was not statistically significant). Independent variables included demographics (age, gender, ethnicity, education, province and place of residence), existing comorbidities (hypertension, diabetes, raised TC, increased waist hip ratio), any hypertension, any diabetes (self-reported or physician diagnosed (fasting blood glucose ≥126 mg/dL)), lifestyles (smoking and alcohol consumption), overweight or obesity (body mass index (BMI)≥25 kg/m2).
Ethnicity/caste data were self-reported and categorised into six major groups based on the caste coding by government of Nepal and STEPS survey Nepal, 2013 categorisation.17 (1) Upper caste groups (population with relatively higher socioeconomic and education status, mostly Brahmins, Chhetris, Thakuri and Sanyasi); (2) disadvantaged Janajatis (disadvantaged group of people and also indigenous, with relatively lower socioeconomic and education status); (3) Dalit (untouchables, most disadvantaged marginalised group of people, with relatively lower socioeconomic and education status); (4) disadvantaged non-Dalit Terai Caste Groups (disadvantaged group of people from the Terai, the lowlands, with relatively lower socioeconomic and education status but not the dalit groups); (5) religious minorities (Muslim, Christian, etc); (6) relatively advantaged Janajatis (indigenous group of people with relatively higher socioeconomic status, such as Gurung, Newar and Thakali).
Multicollinearity, the variance inflation factor was assessed for all the independent variables found to be statistically significant from the bivariate analysis. Multivariable logistic regression modelling was conducted with CKD as a dependent variable using the independent variables identified as being statistically significant from the bivariate analysis.
Results
A total of 12 109 subjects aged 20 years and above were included in the analyses. Among them 61.1% were female, two-fifth were adults in the age group 20–59 years (41.6%), one-third was from upper caste ethnic group (34.2%), and just above half of them were either illiterate or had no formal schooling (54.6%). Table 1 presents the unweighted numbers and proportions of different demographic characteristics of the study population as measured in the study.
Table 1.
Sociodemographic characteristics of the study participants (N=12 109)
| Characteristics | n | % |
| Age | ||
| 20–39 years | 4336 | 35.8 |
| 40–59 years | 5034 | 41.6 |
| 60 years and above | 2739 | 22.6 |
| Sex | ||
| Male | 4708 | 38.9 |
| Female | 7401 | 61.1 |
| Ethnicity | ||
| Upper caste group | 4144 | 34.2 |
| Disadvantaged janajati | 2546 | 21.0 |
| Relatively advantaged janajati | 2018 | 16.7 |
| Disadvantaged non-dalit terai caste | 1836 | 15.2 |
| Dalit | 1225 | 10.1 |
| Religious minorities | 340 | 2.8 |
| Education | ||
| Illiterate/No formal schooling | 6607 | 54.6 |
| Below secondary (<10 years) | 2742 | 22.6 |
| Secondary and above (≥10 years) | 2760 | 22.8 |
| Province | ||
| Province 1 | 2049 | 16.9 |
| Madhesh Province | 2003 | 16.5 |
| Bagmati Province | 3096 | 25.6 |
| Gandaki Province | 1315 | 10.9 |
| Lumbini Province | 2030 | 16.8 |
| Karnali Province | 588 | 4.9 |
| Sudurpaschim Province | 1028 | 8.5 |
| Place of residence | ||
| Rural | 6107 | 50.4 |
| Urban | 6002 | 49.6 |
Prevalence of CKD
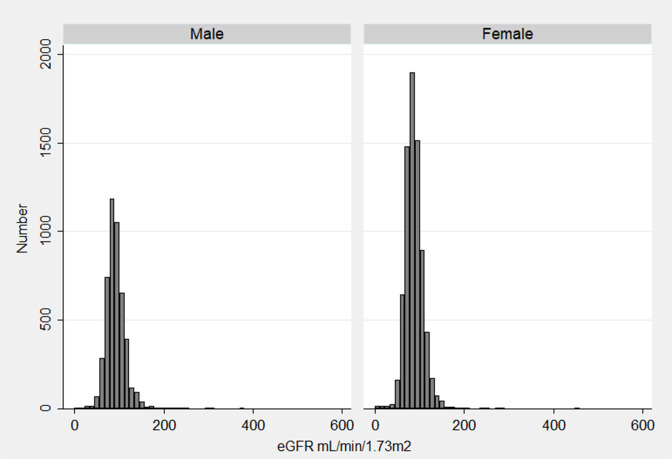
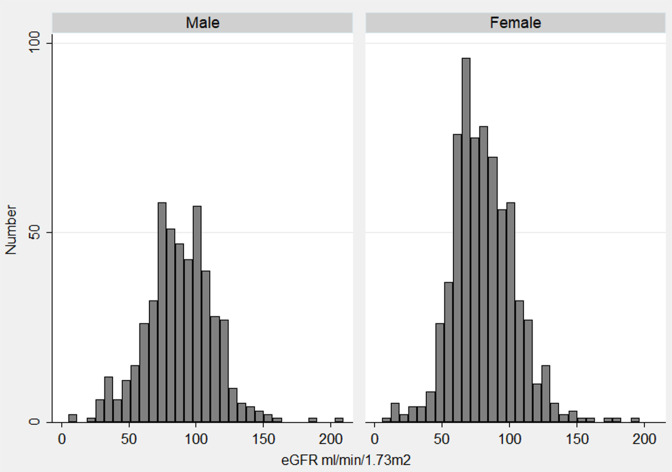
The overall prevalence of CKD based on the eGFR MDRD equation (uACR of ≥30 mg/g and/or glomerular filtration rate <60 both at baseline and follow-up) was 6.0% (95% CI 5.5 to 6.6). The prevalence was higher in males compared with female (6.5 vs 5.7) though not statistically significant. Prevalence increased with increasing age (11.5% among 60 years and above and 2.6% among 20–39 years age group) (table 2). The mean (±SD) eGFR in baseline was 92.6 (±22.1) mL/min/1.73 m2 for male and 88.2 (±20.5) mL/min/1.73 m2 for female (figure 2). The mean (±SD) eGFR was 88.0 (±25.7) mL/min/1.73 m2 for male and 81.1 (±23.4) mL/min/1.73 m2 for female in follow-up (figure 3).
Table 2.
Factors associated with chronic kidney disease (CKD) by univariate and multivariate analysis (N = 12 109)
| Characteristics | Count | CKD | ||
| N | CKD prevalence (n %) | COR (95% CI) | AOR (95% CI) | |
| Age | ||||
| 20–39 years | 4336 | 118 (2.6) | 1 | 1 |
| 40–59 years | 5034 | 294 (5.8) | 2.3 (1.8 to 3.0)‡ | 1.4 (1.0 to 1.8)* |
| 60 years and above | 2739 | 316 (11.5) | 4.9 (3.8 to 6.3)‡ | 2.6 (1.9 to 3.6)‡ |
| Sex | ||||
| Female | 7401 | 415 (5.7) | 1 | 1 |
| Male | 4708 | 313 (6.5) | 1.2 (1.0 to 1.4) | 1.2 (0.8 to 1.6) |
| Ethnicity | ||||
| Upper caste | 4144 | 212 (5.1) | 1 | 1 |
| Disadvantaged Janajati | 2546 | 138 (5.4) | 1.1 (0.8 to 1.4) | 1.1 (0.9 to 1.5) |
| Dalit | 1225 | 84 (7.1) | 1.4 (1.0 to 1.9)* | 1.6 (1.1 to 2.3)† |
| Disadvantaged non-Dalitterai caste | 1836 | 116 (6.4) | 1.3 (0.9 to 1.7) | 1.4 (0.9 to 2.1) |
| Religious minorities | 340 | 25 (8.0) | 1.6 (1.0 to 2.7) | 1.7 (1.0 to 2.9) |
| Relatively advantaged Janajati | 2018 | 153 (7.4) | 1.5 (1.1 to 2.0)* | 1.4 (1.0 to 1.9)* |
| Education | ||||
| Illiterate/no formal schooling | 6607 | 459 (7.0) | 1 | 1 |
| Below secondary (<10 years) | 2742 | 146 (5.4) | 0.8 (0.6 to 0.9)† | 1.1 (0.9 to 1.5) |
| Secondary and above (≥10 years) | 2760 | 123 (4.5) | 0.6 (0.5 to 0.8)‡ | 1.1 (0.7 to 1.4) |
| Province | ||||
| Karnali Province | 588 | 30 (4.7) | 1 | 1 |
| Province 1 | 2049 | 100 (5.2) | 1.1 (0.7 to 1.8) | 1.1 (0.6 to 2.0) |
| Madhesh Province | 2003 | 133 (6.6) | 1.4 (0.9 to 2.3) | 1.2 (0.6 to 2.2) |
| Bagmati Province | 3096 | 196 (6.5) | 1.4 (0.9 to 2.2) | 1.1 (0.6 to 1.9) |
| Gandaki Province | 1315 | 93 (6.8) | 1.5 (0.9 to 2.3) | 1.2 (0.7 to 2.0) |
| Lumbini Province | 2030 | 118 (5.7) | 1.2 (0.7 to 2.2) | 0.9 (0.5 to 1.6) |
| Sudurpashchim Province | 1028 | 58 (5.9) | 1.3 (0.7 to 2.2) | 1.4 (0.7 to 2.6) |
| Place of residence | ||||
| Rural | 6107 | 341 (5.8) | 1 | 1 |
| Urban | 6002 | 387 (6.5) | 1.2 (1.0 to 1.4) | 1.0 (0.8 to 1.3) |
| Smoking habit | ||||
| Non smoker | 8305 | 443 (5.3) | 1 | 1 |
| Smokers | 3804 | 285 (7.6) | 1.5 (1.2 to 1.7)‡ | 1.2 (1.0 to 1.5) |
| Alcohol consumption | ||||
| No | 9131 | 537 (6.0) | 1 | 1 |
| Yes | 2978 | 191 (6.2) | 1.0 (0.9 to 1.2) | |
| Blood pressure | ||||
| Normal | 7754 | 256 (3.3) | 1 | 1 |
| Raised | 4355 | 472 (10.8) | 3.6 (3.0 to 4.3)‡ | 2.4 (2.0 to 3.0)‡ |
| Body mass index (N=12 108)§ | ||||
| Normal | 6896 | 377 (5.5) | 1 | 1 |
| Underweight | 1494 | 88 (6.2) | 1.1 (0.9 to 1.5) | 1.1 (0.8 to 1.6) |
| Overweight and obese | 3718 | 263 (6.9) | 1.3 (1.1 to 1.5)† | 0.9 (0.7 to 1.1) |
| Total cholesterol (N=10 861)§ | ||||
| Normal | 7741 | 398 (5.2) | 1 | 1 |
| Raised | 3120 | 254 (8.2) | 1.6 (1.3 to 2.0)‡ | 1.3 (1.0 to 1.6)* |
| Waist hip ratio (N=11 979)§ | ||||
| Normal | 5095 | 282 (5.4) | 1 | 1 |
| Increased | 6884 | 440 (6.6) | 1.2 (1.0 to 1.5)* | 1.6 (1.2 to 2.3)* |
| Diabetes mellitus (N=11 271)§ | ||||
| No | 10 393 | 506 (4.8) | 1 | 1 |
| Yes | 878 | 172 (19.8) | 4.9 (3.8 to 6.2)‡ | 3.2 (2.5 to 4.1)‡ |
| Total | 12 109 | 6.0 (95% CI 5.5 to 6.6) | ||
Definition of CKD status: urinary albumin-to-creatinine ratio of ≥30 mg/g and/or glomerular filtration rate <60 ml/min/1.73 m2 both at baseline and follow-up).
AOR estimated from the stepwise multivariate logistic regression model with all the above variables added except alcohol consumption
*P value≤0.05.
†P value≤0.01.
‡P value≤0.0001.
§Missing value; refusal for blood sample collection led to variation in the sample size.
AOR, adjusted OR; COR, crude OR.
Figure 2.
Histogram of estimated glomerular filtration rate (eGFR) distribution among 12 097 participants in baseline by sex
Figure 3.
Histogram of estimated glomerular filtration rate (eGFR) distribution among 1194 participants in follow-up by sex
Factors associated with CKD
The multivariable analyses to investigate the association of independent factors and CKD are shown in table 2. In the bivariate analysis, age, smoking, BMI, ethnicity, education, having diabetes mellitus (DM), being hypertensive, increased waist hip ratio and having raised TC were found to be statistically significantly associated with CKD. Therefore, those nine factors including place of residence, province and sex of participants (p value≤0.25) were considered in the multiple logistic models. After adjustment, only six variables: age, ethnicity, diabetes, hypertension, raised TC and increased waist hip ratio, were found to be significant predictors of CKD. There was a strong positive association between age and CKD, that is, the odds of having CKD were about 1.4 (95% CI 1.0 to 1.8) and 2.6 (95% CI 1.9 to 3.6) times higher for people aged 40–59 years and ≥60 years respectively, compared with people aged 20–39 years. Participants with DM had about 3.2 (95% CI 2.5 to 4.1) times higher odds of occurrence of CKD than non-diabetic participants. Similarly, participants with raised BP had 2.4 (95% CI 2.0 to 3.0) higher odds of having CKD than non-hypertensive/normal participants.
Participants with raised TC had 1.3 (95% CI 1.0 to 1.6) times higher odds of having CKD relative to participants who had normal cholesterol level, whereas those with raised waist hip ratio had 1.6 (95% CI 1.2 to 2.3) times higher odds than normal participants to have CKD. Individuals who were classified as Dalit by ethnicity were 1.6 (95% CI 1.1 to 2.3) times or relatively advantaged janajati had 1.4 (95% CI 1.0 to 1.9) times higher odds of having CKD when compared with individuals with an upper caste.
Discussion
This is the first large-scale nationwide population-based representative study to report prevalence of and factors associated with CKD in Nepal among population aged 20 years and above. In the absence of population validated e-GFR equation for our population, MDRD equation as done in previous studies was chosen over the CKD-EPI equation to facilitate comparison of results. The findings show that approximately 1 in every 16 adults (6%) is affected by CKD in Nepal. This value is higher than the population-based study conducted in Morocco 5.1%18 and Vietnam 3.1%,19 almost similar to 6.8% in South Korea,20 east African countries including Uganda and Kenya21 but much lower than the prevalence of many Asian countries with 10.2% in China,2217.5% in Thailand,23 18.9% in Iran24 and 17.2% in India.25 Prevalence of CKD in Nepal is lower than the GBD study estimated prevalence of 8%–10%3 globally. However, these differences in the prevalence of CKD may be in part due to the differences in CKD diagnostic criteria, study design, laboratory methods and lack of validated measures for eGFR for Nepalese population.
Our multivariable analysis found that CKD was independently associated with older age, hypertension, diabetes, raised TC, dalit and relatively advantaged janajati ethnicity and increased waist hip ratio (p<0.05 for each) which are all major NCDs risk factors and consistent with previous findings.26–30Age is a well-established risk factor for development of CKD.31 As expected, age was found to be the most strongly associated risk factor in our study, we observed nearly three times higher odds of occurrence of CKD among people aged 60 years or older compared with people aged 20–39 years.
In line with other surveys in South East Asia, our findings show that the odds of CKD occurrence was significantly and independently higher by around twofold with the presence of hypertension; this supports previous reports32–35 indicating the importance of early detection and treatment of hypertension in Nepal. Similarly, people with DM had more than three times higher odds of occurrence of CKD. Studies around the world show diabetes to be independently associated with development of CKD.36 37 A survey across 10 Asian countries showed that the most common cause of end-stage renal disease in 9 out of 10 countries was diabetic nephropathy.38 In addition, elevated TC though marginal was associated with higher odds of CKD.
Nepal is an ethnically diverse country with more than 125 castes/ethnic groups. Interestingly ethnicity/caste was another factor associated with CKD in our study. Compared with upper caste participants, Dalit participants had a significantly higher prevalence of CKD. The mechanism underlying these differences might be multifactorial, including cultural differences such as smoking and drinking habit, lifestyle and genetic factors. By ethnicity, Dalit group of people are considered as one of the most marginalised in terms of socioeconomic, education, political and health indicators, resulting in decreased access to resources and higher vulnerability to poor health outcomes. This calls for future studies regarding CKD on ethnic differences.
Despite finding an association between CKD and several known established risk factors including waist hip ratio, no association was found between CKD and BMI. The correlation between CKD and waist hip ratio and not BMI is plausible since this ratio is a more sensitive marker for central obesity, metabolic syndrome and potentially less influenced by muscle mass than BMI, this is in agreement with earlier studies.28 39 40 The association between smoking and CKD was reported in bivariate analysis only in our study.
In general, the prevalence of CKD was higher among women compared with men, and this difference has been demonstrated in the past by several population based cross sectional studies.41 However, in the present study, though not statistically significant, we found slightly higher prevalence of CKD among male participants. These discrepancies in the prevalence may be due to the traditional risk factors such as hypertension and hyperglycaemia being prevalent among male participant in our study. We also could not find significant association between CKD and alcohol consumption, place of residence and provinces.
Traditional risk factors such as diabetes and hypertension are the major cause of CKD in most developed and developing countries,42 43 and several other non-traditional and environmental risk factors such as infectious diseases, analgesic abuse, exposure to heavy metal (lead, arsenic, mercury and uranium), pesticides, herbal medications and environmental pollution, impose an additional threat worsening CKD especially in developing countries44 including Nepal. However, we lack information about environmental and some of the non-traditional risk factor, which could be used to quantify the effects of these factors on CKD.
Our study poses several strengths. To the best of our knowledge, this is the first national survey with a large representative population-based sample of the adult population to report prevalence of CKD in Nepal as per Kidney Disease Outcomes Quality Initiative (KDOQI) clinical practice guideline; using (eGFR <60 mL/min/1.73 m2, presence of albuminuria ≥30 mg/g and chronicity confirmed by repeated testing after 3 months) to confirm presence of CKD. The study has used a validated standardised eGFR by the MDRD equation and all necessary protocols, standard operating procedures and quality assurance mechanism were followed to ensure the quality of data collection. Additional strengths of the study include a high overall response rate and analyses based on survey weights which ensure geographical representativeness of the study making the study findings generalisable to the general population. At the same time, potential limitations include dependency on estimation of GFR, rather than direct measurement using injection of an exogenous marker factors as well as cross-sectional design of the study which does not permit inferences regarding causal relationships.
Conclusions
This is the first population-based epidemiological survey of CKD using the protocols recommended by KDOQI in Nepal. The prevalence of CKD was found to be 6.0% with higher prevalence among male, participants with no formal education, urban residents, smokers and participants with overweight and obese. CKD was independently associated with older age, hypertension, DM, increased waist hip ratio, raised TC and individual belonging to the Dalit caste by ethnicity. Our findings highlight the need for early preventive measures to manage predisposing conditions such as diabetes and hypertension which could ultimately lead to CKD and to reduce the prevalence and mortality arising from the associated comorbidities in Nepal.
Supplementary Material
Acknowledgments
The authors would like to thank all the individuals and organisations that provided technical support for the design and implementation of the main survey. The authors are grateful Dr. Shiva raj Mishra, Research Fellow in Epidemiology and Intervention Melbourne School of Population and Global Health, for reviewing the manuscript.
Footnotes
Twitter: @PoudyalAnil, @Kriessh, @MeghnathDhimal
AP and KBK contributed equally.
Contributors: KBK, MD, KKA and AKJ were involved with the conception, design and implemented the study. AP, NS and NKM implemented the study on the field, completed data collection, overall supervision of the study. AP drafted the manuscript and conducted data analysis with input from NS, BB, LG, NKM, and DK. SKS, PG, VGL, UK and DAG contributed to the critical revision of the manuscript, provided feedback on the initial draft, and approved the final version. SKS, KKA, NS, PG and MD were also involved in responding to the comments raised by BMJ Open Journal reviewers; each authors reviewed the reversed manuscript critically and provided improvements, accepts accountability for their contributions. AP is the guarantors and undertake the full responsibility for the contents of the article submitted for publication.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Map disclaimer: The depiction of boundaries on this map does not imply the expression of any opinion whatsoever on the part of BMJ (or any member of its group) concerning the legal status of any country, territory, jurisdiction or area or of its authorities. This map is provided without any warranty of any kind, either express or implied.
Competing interests: None declared.
Patient and public involvement: Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement
Data are available upon reasonable request.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
The study protocol was approved by the Ethical Review Board (ERB) of Nepal Health Research Council, Government of Nepal (Reg.no.110/2016). Written informed consent was taken from all participants before proceeding for data collection. Participants gave informed consent to participate in the study before taking part.
References
- 1.Xie Y, Bowe B, Mokdad AH, et al. Analysis of the Global Burden of Disease study highlights the global, regional, and national trends of chronic kidney disease epidemiology from 1990 to 2016. Kidney Int 2018;94:567–81. 10.1016/j.kint.2018.04.011 [DOI] [PubMed] [Google Scholar]
- 2.Institute for health metrics and evaluation (IHME). GBD Compare Data 2019. [Google Scholar]
- 3.Bikbov B, Purcell CA, Levey AS, et al. Global, regional, and national burden of chronic kidney disease, 1990-2017: a systematic analysis for the global burden of disease study 2017. Lancet 2020;395:709–33. 10.1016/S0140-6736(20)30045-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stanifer JW, Muiru A, Jafar TH, et al. Chronic kidney disease in low- and middle-income countries. Nephrol Dial Transplant 2016;31:868–74. 10.1093/ndt/gfv466 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nugent RA, Fathima SF, Feigl AB, et al. The burden of chronic kidney disease on developing nations: a 21st century challenge in global health. Nephron Clin Pract 2011;118:c269–77. 10.1159/000321382 [DOI] [PubMed] [Google Scholar]
- 6.Agarwal SK, Dash SC, Irshad M, et al. Prevalence of chronic renal failure in adults in Delhi, India. Nephrol Dial Transplant 2005;20:1638–42. 10.1093/ndt/gfh855 [DOI] [PubMed] [Google Scholar]
- 7.Singh NP, Ingle GK, Saini VK, et al. Prevalence of low glomerular filtration rate, proteinuria and associated risk factors in North India using Cockcroft-Gault and modification of diet in renal disease equation: an observational, cross-sectional study. BMC Nephrol 2009;10:1–13. 10.1186/1471-2369-10-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jafar TH, Schmid CH, Levey AS. Serum creatinine as marker of kidney function in South Asians: a study of reduced GFR in adults in Pakistan. J Am Soc Nephrol 2005;16:1413–9. 10.1681/ASN.2004121100 [DOI] [PubMed] [Google Scholar]
- 9.Sharma SK, Zou H, Togtokh A, et al. Burden of CKD, proteinuria, and cardiovascular risk among Chinese, Mongolian, and Nepalese participants in the International Society of nephrology screening programs. Am J Kidney Dis 2010;56:915–27. 10.1053/j.ajkd.2010.06.022 [DOI] [PubMed] [Google Scholar]
- 10.Ene-Iordache B, Perico N, Bikbov B, et al. Chronic kidney disease and cardiovascular risk in six regions of the world (ISN-KDDC): a cross-sectional study. Lancet Glob Health 2016;4:e307–19. 10.1016/S2214-109X(16)00071-1 [DOI] [PubMed] [Google Scholar]
- 11.Sharma SK, Dhakal S, Thapa L, et al. Community-Based screening for chronic kidney disease, hypertension and diabetes in Dharan. JNMA J Nepal Med Assoc 2013;52:205–12. 10.31729/jnma.548 [DOI] [PubMed] [Google Scholar]
- 12.Dhimal M, Bista B, Bhattarai S. Non communicable disease risk factors: steps survey Nepal 2019.
- 13.Nepal Health Research Council . Population based prevalence of selected non-communicable disease in Nepal, 2019. Available: http://nhrc.gov.np/publication/population-based-prevalence-of-selected-non-communicable-diseases-in-nepal/
- 14.Aryal KK, Neupane S, Mehata S. Non communicable diseases risk factors: steps survey Nepal 2013 Kathmandu. Nepal Nepal Heal Res Counc Sch 2014. [Google Scholar]
- 15.Chobanian AV, Bakris GL, Black HR, et al. Seventh report of the joint National Committee on prevention, detection, evaluation, and treatment of high blood pressure. Hypertension 2003;42:1206–52. 10.1161/01.HYP.0000107251.49515.c2 [DOI] [PubMed] [Google Scholar]
- 16.Levey AS, Eckardt K-U, Tsukamoto Y, et al. Definition and classification of chronic kidney disease: a position statement from kidney disease: improving global outcomes (KDIGO). Kidney Int 2005;67:2089–100. 10.1111/j.1523-1755.2005.00365.x [DOI] [PubMed] [Google Scholar]
- 17.STEP I. Non communicable diseases risk factors: steps survey Nepal 2013.
- 18.Benghanem Gharbi M, Elseviers M, Zamd M, et al. Chronic kidney disease, hypertension, diabetes, and obesity in the adult population of Morocco: how to avoid "over"- and "under"-diagnosis of CKD. Kidney Int 2016;89:1363–71. 10.1016/j.kint.2016.02.019 [DOI] [PubMed] [Google Scholar]
- 19.Ito J, Dung DTK, Vuong MT, et al. Impact and perspective on chronic kidney disease in an Asian developing country: a large-scale survey in North Vietnam. Nephron Clin Pract 2008;109:c25–32. 10.1159/000134379 [DOI] [PubMed] [Google Scholar]
- 20.Jang SY, Kim I-H, Ju EY, et al. Chronic kidney disease and metabolic syndrome in a general Korean population: the third Korea National health and nutrition examination survey (KNHANES III) study. J Public Health 2010;32:538–46. 10.1093/pubmed/fdp127 [DOI] [PubMed] [Google Scholar]
- 21.Muiru AN, Charlebois ED, Balzer LB, et al. The epidemiology of chronic kidney disease (CKD) in rural East Africa: a population-based study. PLoS One 2020;15:e0229649. 10.1371/journal.pone.0229649 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang L, Wang F, Wang L, et al. Prevalence of chronic kidney disease in China: a cross-sectional survey. Lancet 2012;379:815–22. 10.1016/S0140-6736(12)60033-6 [DOI] [PubMed] [Google Scholar]
- 23.Ingsathit A, Thakkinstian A, Chaiprasert A, et al. Prevalence and risk factors of chronic kidney disease in the Thai adult population: Thai seek study. Nephrol Dial Transplant 2010;25:1567–75. 10.1093/ndt/gfp669 [DOI] [PubMed] [Google Scholar]
- 24.Hosseinpanah F, Kasraei F, Nassiri AA, et al. High prevalence of chronic kidney disease in Iran: a large population-based study. BMC Public Health 2009;9:1–8. 10.1186/1471-2458-9-44 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Singh AK, Farag YMK, Mittal BV, et al. Epidemiology and risk factors of chronic kidney disease in India – results from the seek (screening and early evaluation of kidney disease) study. BMC Nephrol 2013;14:1–10. 10.1186/1471-2369-14-114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hannan M, Ansari S, Meza N, et al. Risk factors for CKD progression: overview of findings from the CRIC study. Clin J Am Soc Nephrol 2021;16:648–59. 10.2215/CJN.07830520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tsuruya K, Yoshida H, Nagata M, et al. Association of the triglycerides to high-density lipoprotein cholesterol ratio with the risk of chronic kidney disease: analysis in a large Japanese population. Atherosclerosis 2014;233:260–7. 10.1016/j.atherosclerosis.2013.12.037 [DOI] [PubMed] [Google Scholar]
- 28.He Y, Li F, Wang F, et al. The association of chronic kidney disease and waist circumference and waist-to-height ratio in Chinese urban adults. Medicine 2016;95:e3769. 10.1097/MD.0000000000003769 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Alkerwi Ala’a, Sauvageot N, El Bahi I, et al. Prevalence and related risk factors of chronic kidney disease among adults in Luxembourg: evidence from the observation of cardiovascular risk factors (ORISCAV-LUX) study. BMC Nephrol 2017;18:1–10. 10.1186/s12882-017-0772-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ping H, Zhang X, Xing N. Prevalence of chronic kidney disease in China. Lancet 2012;380:216. 10.1016/S0140-6736(12)61213-6 [DOI] [PubMed] [Google Scholar]
- 31.O'Hare AM, Choi AI, Bertenthal D, et al. Age affects outcomes in chronic kidney disease. J Am Soc Nephrol 2007;18:2758–65. 10.1681/ASN.2007040422 [DOI] [PubMed] [Google Scholar]
- 32.Li Z-Y, Xu G-B, Xia T-A, et al. Prevalence of chronic kidney disease in a middle and old-aged population of Beijing. Clin Chim Acta 2006;366:209–15. 10.1016/j.cca.2005.10.011 [DOI] [PubMed] [Google Scholar]
- 33.Safarinejad MR. The epidemiology of adult chronic kidney disease in a population-based study in Iran: prevalence and associated risk factors. J Nephrol 2009;22:99. [PubMed] [Google Scholar]
- 34.Fiseha T, Kassim M, Yemane T. Prevalence of chronic kidney disease and associated risk factors among diabetic patients in southern Ethiopia. AJHR 2014;2:216–21. 10.11648/j.ajhr.20140204.28 [DOI] [Google Scholar]
- 35.Price CP, Finney H. Developments in the assessment of glomerular filtration rate. Clin Chim Acta 2000;297:55–66. 10.1016/S0009-8981(00)00233-3 [DOI] [PubMed] [Google Scholar]
- 36.Damtie S, Biadgo B, Baynes HW. Chronic kidney disease and associated risk factors assessment among diabetes mellitus patients at a tertiary Hospital, Northwest Ethiopia. Ethiop J Health Sci 2018;28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Duan J, Wang C, Liu D, et al. Prevalence and risk factors of chronic kidney disease and diabetic kidney disease in Chinese rural residents: a cross-sectional survey. Sci Rep 2019;9:1–11. 10.1038/s41598-019-46857-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lee G. End-Stage renal disease in the Asian-Pacific region. Semin Nephrol 2003;23:107–14. 10.1053/snep.2003.50009 [DOI] [PubMed] [Google Scholar]
- 39.Elsayed EF, Tighiouart H, Weiner DE, et al. Waist-to-hip ratio and body mass index as risk factors for cardiovascular events in CKD. Am J Kidney Dis 2008;52:49–57. 10.1053/j.ajkd.2008.04.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Silva MIB, Lemos CCdaS, Torres MRSG, et al. Waist-to-height ratio: an accurate anthropometric index of abdominal adiposity and a predictor of high HOMA-IR values in nondialyzed chronic kidney disease patients. Nutrition 2014;30:279–85. 10.1016/j.nut.2013.08.004 [DOI] [PubMed] [Google Scholar]
- 41.Bikbov B, Perico N, Remuzzi G. On behalf of the GBD genitourinary diseases expert group. disparities in chronic kidney disease prevalence among males and females in 195 countries: analysis of the global burden of disease 2016 study. Nephron 2018;139:313–8. [DOI] [PubMed] [Google Scholar]
- 42.Koye DN, Shaw JE, Reid CM, et al. Incidence of chronic kidney disease among people with diabetes: a systematic review of observational studies. Diabet Med 2017;34:887–901. 10.1111/dme.13324 [DOI] [PubMed] [Google Scholar]
- 43.Garofalo C, Borrelli S, Pacilio M, et al. Hypertension and prehypertension and prediction of development of decreased estimated GFR in the general population: a meta-analysis of cohort studies. Am J Kidney Dis 2016;67:89–97. 10.1053/j.ajkd.2015.08.027 [DOI] [PubMed] [Google Scholar]
- 44.Xu X, Nie S, Ding H, et al. Environmental pollution and kidney diseases. Nat Rev Nephrol 2018;14:313–24. 10.1038/nrneph.2018.11 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data are available upon reasonable request.