Abstract
Intraventricular application of vancomycin is an effective therapeutic regimen for the treatment of shunt-associated staphylococcal ventriculitis. We examined the in vitro activity of vancomycin at high concentrations against Staphylococcus aureus ATCC 25923 and Staphylococcus epidermidis ATCC 12228 in human cerebrospinal fluid samples. Time-kill curves revealed equal efficacies for concentrations of 10, 100, and 300 μg/ml, and incubation times of 24 to 48 h were needed to achieve a 3 log10 reduction of viable bacteria. A concentration of 5 μg/ml showed a slightly lower activity, but this difference was not significant. In an infant who was successfully treated for shunt-associated ventriculitis due to S. epidermidis by once-daily local administration of vancomycin (3 mg for 2 days and 5 mg for 4 days [0.5 to 0.8 mg/kg of body weight]) the in vivo kill kinetics were similar to those for the in vitro results. These results support time-dose regimens that provide trough vancomycin levels of 5 to 10 μg/ml.
Infections caused by Staphylococcus aureus and coagulase-negative staphylococci are a serious complication in patients with cerebrospinal shunting devices (4). Eradication of these pathogens frequently cannot be achieved by systemic application of antibiotics. If access to the intraventricular space (e.g., via a Rickham reservoir or external ventricular drainage) is possible, local administration of antibiotics can be performed. By intraventricular instillation of vancomycin, staphylococcal infections including those caused by multiresistant strains have been treated successfully without toxic side effects (4, 12).
This result has been related to the fact that this mode of treatment results in higher antibiotic concentrations in cerebrospinal fluid (CSF) than systemic application does (3–5). A recent study at our university hospital yielded mean CSF peak levels of vancomycin of 300 μg/ml after intraventricular instillation of 10 mg once daily for 5 to 13 days, while mean trough CSF levels were 7.6 μg/ml (11). However, due to lack of knowledge about the bactericidal efficacy of such high vancomycin concentrations, the most effective time-dose scheme for this kind of therapy has not been established to date.
It was the aim of this study to investigate the in vitro bactericidal activity of vancomycin in human CSF at a range of concentrations which can be found in patients after intraventricular instillation. In an infant suffering from ventriculitis, bacterial counts were performed during therapy and were compared with in vitro results.
MATERIALS AND METHODS
Reagents.
Vancomycin hydrochloride (Vancomycin Lilly; Eli Lilly, Gießen, Germany) was diluted in sterile double-distilled water to 15, 5, 0.5, 0.25, and 0.1 mg/ml. A total of 5 μl each of these stock solutions was further diluted in 250 μl of human CSF samples, yielding final concentrations of 300, 100, 10, 5, and 2 μg/ml, respectively (see below).
CSF samples.
Human CSF from patients undergoing lumbar puncture for various clinical indications was collected at the Department of Bacteriology. Specimens from patients receiving antibiotic therapy as well as samples showing bacterial contamination were excluded. Additionally, samples showing antimicrobial activity in a bioassay with Mueller-Hinton agar plates (Oxoid, Hampshire, England) enriched with 6 × 105 spores/ml of Bacillus subtilis ATCC 6633 (Difco, Detroit, Mich.) (9) were excluded from the study. By using vancomycin concentrations from 64 to 1 μg/ml in serial twofold dilutions in this bioassay, the relationship between the diameter of zone of inhibition and the logarithm of drug concentration was determined to be linear. A concentration of 0.5 μg/ml was the lower limit of detection in this assay, and plates were grown for 14 to 16 h at 36°C. Characteristics of the 40 CSF samples used for this study are depicted in Table 1. The bactericidal activity of vancomycin was tested in each individual sample.
TABLE 1.
Characteristics of human CSF samples (n = 40)
| Characteristic | Range | Median | 25–75 Quartiles |
|---|---|---|---|
| Age of patients (yr)a | 0.1–75 | 46 | 24–59 |
| Amt of glucose (mg/dl) | 46–91 | 82 | 60–86 |
| Amt of protein (mg/dl) | 22–331 | 68 | 50–102 |
| Amt of albumin (mg/dl) | 10–234 | 31 | 21–51 |
| No. of leukocytes/μl | 0–384 | 2 | 1–11 |
| No. of erythrocytes/μl | 0–57,000 | 2 | 0–22 |
16 female, 24 male.
Bacteria.
S. aureus ATCC 25923 and S. epidermidis ATCC 12228 were cultured on tryptic soy agar (Merck, Darmstadt, Germany). A clinical S. epidermidis isolate from the CSF of an infant with an infected intraventricular shunting device (see below) was also investigated. MICs of vancomycin were determined by the standard broth dilution method in Mueller-Hinton broth (Oxoid) (10) and by Etest (AB Biodisk, Solna, Sweden). MBCs were determined by subsequent evaluation of 99.9% kill (2).
Bactericidal activity of vancomycin.
Bacteria were grown overnight in tryptic soy broth at 37°C to approximately 109 CFU/ml, centrifuged for 10 min at 1,800 × g, and washed twice in 0.9% saline. Subsequent to 10-fold dilution in saline, 15 μl of bacterial suspension was added to 1.5 ml of CSF, yielding a final concentration of 3.3 × 105 to 1.2 × 106 CFU/ml. To 250-μl portions of these CSF suspensions, 5 μl of vancomycin stock solution (see above) was added to gain final concentrations of 2, 5, 10, 100, and 300 μg/ml, respectively. Control samples without vancomycin were treated the same way. Samples were incubated at 37°C. At 0, 8, 24, and 48 h aliquots of 50 μl were diluted in saline 10- and 100-fold. Quantitative cultures were performed by logarithmic plating of 50 μl of these dilutions in duplicate on Mueller-Hinton-agar plates by using a spiral plater (Whitley automatic spiral plater; Don Whitley Scientific Limited, West Yorkshire, England). CFU were counted after incubation at 37°C for 48 h.
Antibiotic carryover.
S. aureus ATCC 25923 and S. epidermidis ATCC 12228 were grown for 4 h in tryptic soy broth at 37°C and were washed twice in saline. Subsequent to 100-fold dilution in saline, 40 μl was added to 3.96 ml of 0.9% saline containing 0, 1, 10, 30, or 100 μg/ml vancomycin. Immediately afterwards, quantitative cultures were performed in duplicate by logarithmic plating of 50 μl of each solution.
Patient’s history.
A newborn male infant suffering from congenital occlusive hydrocephalus was treated by a ventriculo-peritoneal shunt (Holter valve system; Codman, Berkshire, United Kingdom). Five months later, at a weight of 6,300 g, he developed septic signs due to shunt-associated ventriculitis caused by infection with a methicillin-resistant S. epidermidis isolate susceptible to amikacin (MIC, 8 μg/ml), ciprofloxacin (MIC, 0.125 μg/ml), tetracycline (MIC, 0.5 μg/ml), rifampin (MIC, 0.002 μg/ml), and vancomycin (see Results), as determined by Etest, and to fosfomycin, as determined by the disc diffusion method DIN 58940 (1). The peritoneal catheter was removed from the abdominal cavity and drained externally. The patient was treated intravenously with 250 mg of ceftriaxone twice and 400 mg of fosfomycin three times daily for 5 days. Because of a diagnosed in vitro resistance of the pathogen against ceftriaxone (MIC > 256 μg/ml), this drug was replaced by amikacin (45 mg twice daily). Additionally, 5 mg of gentamicin (MIC, 64 μg/ml) was instilled intraventricularly for 7 days (7). Since S. epidermidis was still detectable in CSF, therapy was changed to 60 mg of intravenous vancomycin three times a day for the following 14 days. Nevertheless, CSF cultures remained positive. Intravenous vancomycin was continued, and additional intraventricular application of 3 mg of vancomycin diluted in 2 ml of sterile saline into the reservoir of the shunt was started once per day for 2 days and instillation of 5 mg per day was performed for 4 days more. Before each instillation, 1 ml of CSF was removed from the reservoir and centrifuged for 5 min at 16,000 × g. The supernatant was replaced by sterile saline to remove vancomycin, and quantitative cultures were performed as described above. The shunt was clamped for 1 h after each instillation.
Statistical methods.
The Student’s t test was used for comparison of CFU counts in antibiotic carryover experiments. Repeated measures analysis of variance and Dunnett’s Multiple Comparison test (Graphpad Software Inc.) were used to evaluate differences between the time-kill curves of bacteria for different concentrations of vancomycin. P values of < 0.05 were considered significant.
RESULTS
MICs and MBCs.
The MIC of vancomycin was 2 μg/ml against all strains used, by the broth dilution method as well as by Etest. MBCs of S. epidermidis ATCC 12228 and the clinical isolate of S. epidermidis were 2 and 4 μg/ml, respectively. For S. aureus ATCC 25923, the reduction of viable counts by 2 to 16 μg of vancomycin/ml in Mueller-Hinton broth ranged from 2.2 to 2.7 log10 after 24 h of incubation.
Antibiotic carryover.
Viable counts in saline solutions containing 1 and 10 μg of vancomycin/ml were 1.1 × 104 to 2.0 × 104/ml (S. aureus, n = 4) and 2.9 × 103 to 3.6 × 103/ml (S. epidermidis, n = 2), respectively. Counts of the controls without vancomycin were identical. The innermost spiral on the plate, where the plater releases more volume than in the peripheral region, showed a 25% reduction of bacterial growth when 30 μg of vancomycin/ml was used. This zone of inhibition was extended when the solution contained 100 μg of the antibiotic/ml. Therefore, in CSF samples containing 300 μg of vancomycin/ml additional 1:30 dilutions to 10 μg/ml were performed to avoid overestimation of drug activity.
Activity of vancomycin in human CSF in vitro.
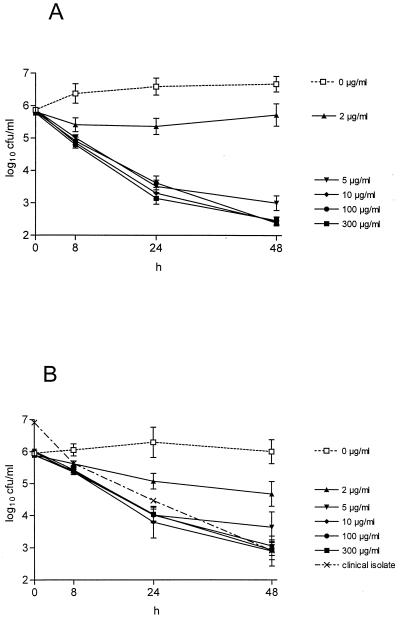
Kinetics of viable counts in human CSF samples are illustrated in Fig. 1 for S. aureus ATCC 25923 and S. epidermidis ATCC 12228. In the absence of vancomycin, both strains showed moderate multiplication over 24 h in CSF. The bactericidal action of vancomycin was slow. A 3 log10 reduction in CFU could be observed not earlier than 24 to 48 h for S. aureus and at 48 h for S. epidermidis. No differences in killing activity were detectable for vancomycin concentrations of 10, 100, and 300 μg/ml (P > 0.05), whereas a concentration of 2 μg/ml was significantly less bactericidal (P < 0.01). A concentration of 5 μg/ml showed a slightly lower activity than concentrations of 10 to 300 μg/ml after 48 h, but this difference was not significant.
FIG. 1.
In vitro activities of the indicated concentrations of vancomycin against S. aureus ATCC 25923 (A) and S. epidermidis ATCC 12228 (B) within human CSF samples at 37°C. Mean ± standard errors of the means of 10 (2, 5, 100, and 300 μg/ml) or 20 (0 and 10 μg/ml) samples each are depicted. Also shown in panel B is the mean activity for vancomycin concentrations of 10, 100, and 300 μg/ml in the infant’s CSF enriched with his clinical isolate of S. epidermidis.
Activity of vancomycin in human CSF in vivo.
In the infant suffering from shunt infection, decrease of S. epidermidis CFU in CSF was slow under intraventricular application of 3 to 5 mg of vancomycin (Table 2). Eradication of bacteria could be achieved after 4 days of local treatment. Vancomycin concentrations measured by bioassay from CSF samples immediately before each instillation on days 2 and 3 were 4 and 6 μg/ml, respectively. On day 7, the complete shunting device was replaced by a new one. Intravenous vancomycin was continued for 5 days more, and CSF remained sterile.
TABLE 2.
Quantitative cultures of S. epidermidis in CSF samples gained from the shunt reservoir during intraventricular therapya
| Day of therapy | CFU/ml |
|---|---|
| 1 (before the beginning of therapy) | 6 × 103 − 2 × 104 |
| 2 | 1 × 103 |
| 3 | Sterile |
| 4 | 4 × 102 |
| 5 | Sterile |
| 6 (end of therapy) | Sterile |
| 7 (exchange of the shunt system) | Sterile |
| 13 | Sterile |
Samples were taken immediately before each instillation of vancomycin.
When this patient’s CSF was enriched to vancomycin concentrations of 10, 100, and 300 μg/ml and spiked with S. epidermidis (patient’s own clinical isolate) up to 107 CFU/ml, time-kill curves matched those obtained with the ATCC strain and showed equipotent activity of vancomycin. The mean time-kill curve of the clinical isolate of S. epidermidis by 10, 100, and 300 μg of vancomycin/ml is compared to the time-kill curves for the ATCC strain in Fig. 1B.
DISCUSSION
The efficacy of local administration of vancomycin in the treatment of staphylococcal ventriculitis may be attributed to achieving drug concentrations as high as 300 to 800 μg/ml, while by systemic application peak values of 6 μg/ml are not exceeded (3–5). Our results, however, show that the microbicidal effect reaches a maximum at 5 to 10 μg/ml and cannot be improved by concentrations exceeding that. Similarly, Haworth et al. found no difference in the bactericidal activity of 30 and 400 μg of vancomycin/ml against a hemolytic, methicillin-resistant S. aureus isolate in human CSF in vitro (8). These findings are in keeping with the assumption of a saturation effect resembling the saturation curves in enzymology (6), which has been demonstrated in broth media for vancomycin in previous studies (6, 13).
Reduction of viable counts in human CSF by vancomycin concentrations of ≥10 μg/ml reached 3 log10 after 24 to 48 h. Testing the above-mentioned methicillin-resistant S. aureus isolate, Haworth et al. found that viable counts were never lowered by more than 1 log10 unit over a 24-h period by vancomycin concentrations of 30 to 400 μg/ml (8). It is difficult to explain this difference, since the S. aureus strains were similarly susceptible in Mueller-Hinton broth in both studies. It could be that the course of time-kill curves is influenced by the composition of individual CSF samples. However, contents of glucose, protein, and albumin as well as cell count do not seem to influence the activity of vancomycin since there were no significant differences in the reduction of viable counts between each of 40 unique samples tested in our study.
By using the logarithmic mode of automatic plating, carryover experiments showed no growth inhibition of our strains on Mueller-Hinton agar when the plated solution contained up to 10 μg of vancomycin/ml. This value exceeds the 5 μg/ml concentration found in a previous study, where manual plating was performed (6). Therefore, use of the spiral plater system in the logarithmic mode is advantageous not only for the determination of CFU counts but also for the avoidance of antibiotic carryover.
The activity of vancomycin in concentrations of ≥5 μg/ml in CSF proved to be significantly higher than that of 2 μg/ml (Fig. 1). This finding may be of importance for successful therapy of shunt infections. Eradication of S. epidermidis after 4 days of intraventricular treatment was achieved in the case of our patient despite previous ineffective intravenous application. As shown by Pfausler et al. (11), CSF trough levels of vancomycin in adults become not lower than 7.6 μg/ml within 24 h after local application of one single dose of 10 mg. CSF trough levels in our infant were similar (about 5 μg/ml). Since in vitro vancomycin concentrations of 5 to 10 μg/ml proved to be maximally effective against staphylococci, the success of intraventricular therapy seems to depend on trough levels of at least 5 μg/ml rather than on achieving high peak levels.
Recommendations for vancomycin single doses for this kind of therapy in adults vary from 5 to 20 mg (about 0.07 to 0.3 mg/kg of body weight) (3, 4, 11, 12). In children and infants, doses of up to 2 mg/kg have been used (12), and in our patient, doses of 0.5 to 0.8 mg/kg were successful. Because of these discrepancies and individual differences, measurement of CSF vancomycin trough levels is useful for surveillance of correct dosing. Therapy should be continued until CSF cultures become sterile and then reshunting should be performed. Probably, it will be possible to establish a generally valid dosage schedule enabling intraventricular therapy without the necessity of repeated drug concentration monitoring in the future. Further investigations of the in vivo bactericidal activity and pharmacokinetics of vancomycin are necessary to define the optimal dosing regimens and to reach this goal.
ACKNOWLEDGMENTS
This study was supported by the Austrian Science Fund (FWF), grant no. P12298-MED, and by the Legerlotz Foundation. The spiral plater was financed by the Jubiläumsfonds of the Austrian National Bank (project no. 6801/1).
REFERENCES
- 1.Allerberger F, Klare I. In-vitro activity of fosfomycin against vancomycin-resistant enterococci. J Antimicrob Chemother. 1999;43:211–217. doi: 10.1093/jac/43.2.211. [DOI] [PubMed] [Google Scholar]
- 2.Amsterdam D. Susceptibility testing of antimicrobials in liquid media. In: Lorian V, editor. Antibiotics in laboratory medicine. Baltimore, Md: Williams & Wilkins; 1996. pp. 52–111. [Google Scholar]
- 3.Arroyo J C, Quindlen E A. Accumulation of vancomycin after intraventricular infusions. South Med J. 1983;76:1554–1555. doi: 10.1097/00007611-198312000-00023. [DOI] [PubMed] [Google Scholar]
- 4.Bayston R, Hart C A, Barnicoat M. Intraventricular vancomycin in the treatment of ventriculitis associated with cerebrospinal fluid shunting and drainage. J Neurol Neurosurg Psychiatry. 1987;50:1419–1423. doi: 10.1136/jnnp.50.11.1419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cooper G C, Given D B. Vancomycin, a comprehensive review of 30 years of clinical experience. New York, N.Y: Park Row Publishers; 1986. [Google Scholar]
- 6.Flandrois J P, Fardel G, Carret G. Early stages of in vitro killing curve of LY146032 and vancomycin for Staphylococcus aureus. Antimicrob Agents Chemother. 1988;32:454–457. doi: 10.1128/aac.32.4.454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Guggenbichler J P, Menardi G, Hager J. Antimicrobial therapy of bacterial meningitis in premature and newborn infants and shunt infections. In: Guggenbichler J P, editor. New aspects for treatment with fosfomycin. Vienna, Austria: Springer; 1987. pp. 41–57. [Google Scholar]
- 8.Haworth C S, Sobieski M W, Scheld W M, Park T S. Staphylococcus aureus ventriculitis treated with single-dose intraventricular vancomycin or daptomycin ( LY146032): bacterial and antibiotic kinetics in hydrocephalic rabbits. Antimicrob Agents Chemother. 1990;34:245–251. doi: 10.1128/aac.34.2.245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kureishi A, Jewesson P J, Bartlett K H, Cole C D, Chow A W. Application of a modified bioassay for monitoring serum teicoplanin and vancomycin in febrile neutropenic patients. Antimicrob Agents Chemother. 1990;34:1642–1647. doi: 10.1128/aac.34.9.1642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.National Committee for Clinical Laboratory Standards. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically, 4th ed. Approved standard M7-A4. Wayne, Pa: National Committee for Clinical Laboratory Standards; 1997. [Google Scholar]
- 11.Pfausler B, Haring H P, Kampfl A, Wissel J, Schober M, Schmutzhard E. Cerebrospinal fluid (CSF) pharmacokinetics of intraventricular vancomycin in patients with staphylococcal ventriculitis associated with external CSF drainage. Clin Infect Dis. 1997;25:733–735. doi: 10.1086/513756. [DOI] [PubMed] [Google Scholar]
- 12.Swayne R, Rampling A, Newsom S W. Intraventricular vancomycin for treatment of shunt-associated ventriculitis. J Antimicrob Chemother. 1987;19:249–253. doi: 10.1093/jac/19.2.249. [DOI] [PubMed] [Google Scholar]
- 13.Verbist L. In vitro activity of LY146032, a new lipopeptide antibiotic, against gram-positive cocci. Antimicrob Agents Chemother. 1987;31:340–342. doi: 10.1128/aac.31.2.340. [DOI] [PMC free article] [PubMed] [Google Scholar]