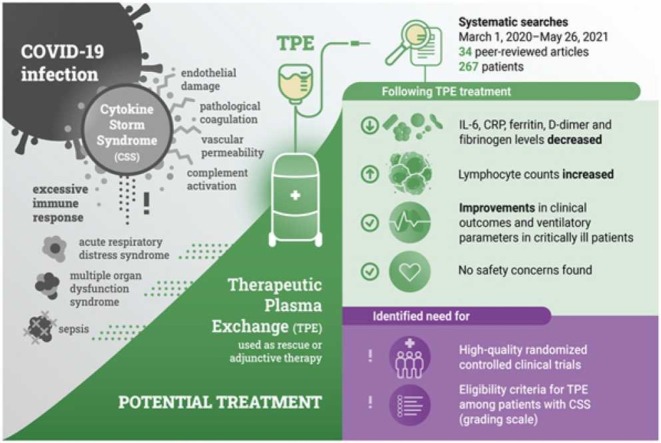
Abstract
The risk of mortality in patients with coronavirus disease 2019 (COVID-19) is largely related to an excessive immune response, resulting in a hyperinflammatory and hypercoagulable condition collectively referred to as cytokine storm syndrome (CSS). Management of critically ill patients with COVID-19 has included attempts to abate this process, prevent disease progression, and reduce mortality. In this context, therapeutic plasma exchange (TPE) offers an approach to eliminate inflammatory factors and cytokines, offset the pathologic coagulopathy, and reduce the CSS effects. The aim of this review is to analyze available data on the use of TPE for the treatment of CSS in patients with COVID-19. Systematic searches of PubMed, Scopus and COVID-19 Research were conducted to identify articles published between March 1, 2020 and May 26, 2021 reporting the use of TPE for the treatment of COVID-19-induced CSS. A total of 34 peer-reviewed articles (1 randomized controlled trial, 4 matched case-control series, 15 single-group case series, and 14 case reports), including 267 patients, were selected. Despite the low evidence level of the available data, TPE appeared to be a safe intervention for critically ill patients with COVID-19-induced CSS. Although inconsistencies exist between studies, they showed a general trend for decreased interleukin-6, C-reactive protein, ferritin, D-dimer, and fibrinogen levels and increased lymphocyte counts following TPE, supporting the immunomodulatory effect of this treatment. Moreover, TPE was associated with improvements in clinical outcomes in critically ill patients with COVID-19. While TPE may offer a valuable option to treat patients with COVID-19-induced CSS, high-quality randomized controlled clinical trials are needed to confirm its potential clinical benefits, feasibility, and safety. Moreover, clear criteria should be established to identify patients with CSS who might benefit from TPE.
Abbreviations: ADAMTS-13, a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13; ARDS, acute respiratory syndrome; ASFA, American Society for Apheresis; CCP, COVID-19 convalescent plasma; COVID-19, coronavirus disease 2019; CRP, C-reactive protein; CSS, cytokine storm syndrome; FFP, fresh frozen plasma; ICU, intensive care unit; IFN, interferon; IL, interleukin; LOS, length of stay; MAS, macrophage activation syndrome; MODS, multiple organ dysfunction syndrome; PaO2:FiO2, pressure of arterial oxygen to fractional inspired oxygen concentration; RCT, randomized controlled trial; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; sHLH, secondary hemophagocytic lymphohistiocytosis; SOFA, Sequential Organ Failure Assessment; TAMOF, thrombocytopenia-associated multiple organ failure; TNF, tumor necrosis factor; TPE, therapeutic plasma exchange
Keywords: Therapeutic plasma exchange, Cytokine storm syndrome, COVID-19, Efficacy, Safety, SARS-CoV-2, Penn grading
Graphical Abstract
1. Background
The coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is responsible for the pandemic declared by the World Health Organization on March 11, 2020 [1]. Up to June 22, 2021, COVID-19 has caused > 177 million infections and > 3.85 million deaths [2]. Patients with COVID-19 show a variety of symptoms, ranging from mild, flu-like symptoms (81% of cases) to severe (14%) and critical (5%) manifestations [3]. The risk of mortality in critically ill patients is attributed mainly to an excessive immune response rather than to the viral infection itself. Most patients with severe COVID-19 in the intensive care unit (ICU) have significant increases in cytokines and other inflammatory biomarkers, such as interleukins (ILs), interferons (IFNs), tumor necrosis factors (TNFs), colony-stimulating factors, growth factors, ferritin, C-reactive proteins (CRPs), and D-dimers [4], [5], [6], [7], [8]. This excessive and prolonged cytokine response can induce the recruitment of other immune cells (e.g., lymphocytes, monocytes/macrophages, dendritic cells), causing an exponential inflammatory growth [4], [9], [10]. This hyperinflammatory condition, often called cytokine storm syndrome (CSS) or cytokine release syndrome, causes complement activation, endothelial damage, pathologic activation of the coagulation system, and increased vascular permeability. Clinically, CSS may result in lung damage, acute respiratory distress syndrome (ARDS), multiple organ dysfunction syndrome (MODS), and sepsis [4], [11], [12], [13], [14], [15].
Currently, there is no approved specific treatment for COVID-19, but various therapeutic agents (e.g., tocilizumab, steroids) showed some level of effectiveness [16]. Besides supportive/standard care, management of patients with COVID-19 might also include timely control of the CSS to prevent disease aggravation and reduce mortality [4], [15]. In this context, potentially effective treatment approaches include administration of immunomodulators, cytokine antagonists, monoclonal antibodies, and anti-inflammatory drugs [4], [15], [17], [18]. Therapeutic plasma exchange (TPE) may also be a valuable option to control CSS by removing inflammatory markers and cytokines [4], [15], [19], [20]. The purpose of this review is to compile and analyze available data on the use of TPE for the treatment of CSS in patients with COVID-19.
2. Methods
A systematic literature search was conducted to identify studies using TPE in hospitalized patients with COVID-19. Systematic searches of PubMed, Scopus, and a Dialog database called COVID-19 Research were conducted for articles published between March 1, 2020 and May 26, 2021. The searches were performed with the following terms: ("plasma exchange" OR “plasmapheresis”) AND (“coronavirus” OR “COVID-19″ OR “SARS-CoV-2″ OR “2019-nCoV”). The search was done on April 24, 2020, and weekly updates were provided thereafter.
Identified articles were screened by one reviewer. Relevant papers were selected if at least one COVID-19 patient received TPE. Articles were excluded if COVID-19 was not the reason for TPE treatment initiation, the technique was unclear, or the article was not written in English. Systematic literature reviews and meta-analyses were excluded, but their reference lists were checked for relevant articles that might have been overlooked. Subsequently, data concerning the characteristics of patients and their disease, TPE procedures and adjunct treatments, outcomes (laboratory parameters and clinical outcomes), and TPE safety were extracted from selected articles.
Methodologic classification of articles was performed using the Oxford Centre for Evidence-Based Medicine levels by two assessors, with differences resolved by consensus [21]. The severity of COVID-19-induced CSS in the selected studies was evaluated using the Penn grading scale by two assessors [22].
3. Results
3.1. General information on search results
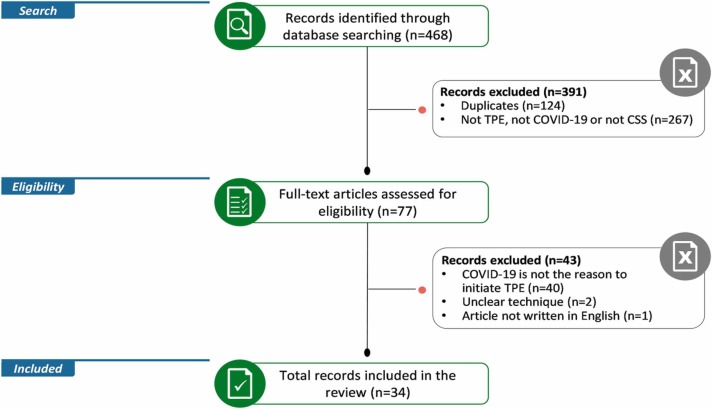
The systematic literature search identified 468 articles (344 after duplicates removal), of which 77 articles were selected for full-text screening ( Fig. 1). Of these, 43 articles did not meet selection criteria. In total, 34 peer-reviewed articles were included (1 randomized controlled clinical trial [RCT] [23], 4 matched case-control series [24], [25], [26], [27], 15 single-group case series [28], [29], [30], [31], [32], [33], [34], [35], [36], [37], [38], [39], [40], [41], [42], and 14 case reports including < 3 patients [43], [44], [45], [46], [47], [48], [49], [50], [51], [52], [53], [54], [55], [56]), disclosing outcomes for 267 TPE-treated patients ( Table 1).
Fig. 1.
PRISMA flowchart. COVID-19, coronavirus disease 2019; CSS, cytokine storm syndrome; n, number of records; TPE, therapeutic plasma exchange.
Table 1.
Characteristics of included studies and patients.
| First author, country, study group | Oxford level of evidence | Number of patients | Disease severity | ARDS, MODS and/or septic shock | Types of organ damage | Mechanical ventilation | SOFA score | CSS Penn grade* | Comorbidities |
|---|---|---|---|---|---|---|---|---|---|
| Randomized controlled clinical trial | |||||||||
| Faqihi, Saudi Arabia[23] TPE group | 3 | 43 | Life-threatening | ARDS (43), MODS (43), septic shock (43) | Pulmonary embolism (13) | MV (43) | Median (IQR): 10 (8–13) | Grade 4 (43) | Diabetes (10), hypertension (19), coronary artery disease (1) |
| Control group | 44 | Life-threatening | ARDS (43), MODS (43), septic shock (43) | Pulmonary embolism (6) | MV (44) | Median (IQR): 9 (6–12) | Grade 4 (44) | Diabetes (8), hypertension (16), coronary artery disease (1) | |
| Matched case-control series | |||||||||
| Arulkumaran, UK[24] TPE group | 4 | 7 | Critical | ARDS (7), MODS (NR), septic shock (NR) | Bilateral lung infiltrates (7) | MV (3), CPAP (4) | NR | Grade 3 (3), Grade 4 (4) | Asthma (3), obesity (2), nil (1), previous deep vein thrombosis (1), none (1) |
| Control group | 7 | Critical | NR | Bilateral lung infiltrates (7) | NR | NR | NR | NR | |
| Gucyetmez, Turkey[25] TPE group | 4 | 18 (12 after PSM) | Patients in ICU | NR | Pneumonia (18), AKI (6) | IMV (16), NIMV (1), HFOT (1) | Mean ± SD: 6 ± 1 | Grade 3 (1; 1 after PSM), Grade 4 (17; 11 after PSM) | NR |
| Control group | 35 (12 after PSM) | Patients in ICU | NR | Pneumonia (18), AKI (19) | IMV (30), NIMV (3) HFOT (2) | Median (IQR): 7 (3) | Grade 3 (2; 0 after PSM), Grade 4 (33; 12 after PSM) | NR | |
| Kamran, Pakistan[26] TPE group | 4 | 71 (45 after PSM) | Moderate (3), severe (20), critical (22) | NR | NR | IMV (3), CPAP (19) | NR | Grade 3 (19), Grade 4 (3), unknown (23) | Obstructive air way disease (2), ischemic heart disease (6), diabetes (11), hypertension (9), > 3 comorbidities (4), none (21) |
| Control group | 209 (45 after PSM) | Moderate (3), severe (20), critical (22) | NR | NR | IMV (3), CPAP (19) | NR | Grade 3 (19), Grade 4 (3), unknown (23) | Obstructive air way disease (2), ischemic heart disease (6), diabetes (11), hypertension (9), > 3 comorbidities (4), none (21) | |
| Khamis, Oman[27] TPE group | 4 | 11 | Critical | ARDS (10); septic shock (9); MODS (1) | Severe pneumonia (1) | IMV (10) | Median (IQR): 6 (3–9) | Grade 3 (1), Grade 4 (10) | Obesity (1), diabetes (8), hypertension (6), CKD (1) |
| Control group | 20 | Critical | ARDS (10), septic shock (10), MODS (3) | Severe pneumonia (10) | IMV (11) | Median (IQR): 3 (2–6) | Grade 3 (9), Grade 4 (11) | Obesity (2), diabetes (7), hypertension (6), CKD (3) | |
| Single-group case series | |||||||||
| Adeli, Iran[28] | 4 | 8 | Critical | ARDS (8), MODS (1), septic shock (8) | Pulmonary involvement (8) | MV (3), oxygen mask (5) | NR | Grade 3 (5), Grade 4 (3) | Hypertension (1), diabetes (2), none (5) |
| Alharthy, Saudi Arabia[29] | 5 | 3 | Life-threatening with associated thromboinflammation and CNS pathology | ARDS (3), MODS (NR), septic shock (NR) | Brain infarction (3), pulmonary embolism (3) | MV (3) | 8 (1), 9 (2) | Grade 4 (3) | Hypertension and diabetes (2), none (1) |
| Faqihi, Saudi Arabia[31] | 4 | 10 | Life-threatening | ARDS (10), MODS (10), septic shock (10) | Pulmonary embolism (2), AKI (1), pneumonia (10) | MV (10) | Median (IQR): 11 (8.9–11.5) | Grade 4 (10) | Diabetes (6), hypertension (5), cardiovascular disease (1), none (4) |
| Gluck, USA[32] | 4 | 10 | Critical | NR | NR | IMV (6) | NR | Grade 3 (4), Grade 4 (6) | Diabetes (3), hypertension (5), obesity (6) |
| Hashemian, Iran[33] | 4 | 15 | Critical | ARDS (15), MODS (NR), septic shock (NR) | NR | IMV (4), NIMV (11) | Mean ± SD: 9.6 ± 1.5 | Grade 3 (11); Grade 4 (4) | Hypertension (6), diabetes (5), cardiovascular disease (2), none (5) |
| Jaiswal, Dubai, United Arab Emirates[34] | 4 | 14 | Critical | ARDS (14), MODS (NR), septic shock (14) | AKI (5) | IMV (14) | NR | Grade 4 (14) | Hypertension (9), none (4) |
| Keith, USA[38] | 4 | 8 | Critical | ARDS (8), MODS (8); septic shock (8) | NR | MV (7), CPAP (1) | Mean (range): 6.8 (2–15) | Grade 4 (8) | Hypertension (4), hyperlipidemia and gastroesophageal reflux disease (1), cerebral palsy (1), diabetes (1), systemic lupus, erythematosus and benign prostatic hypertrophy (1), prostate cancer (1), obesity (2), dementia, pseudotumor cerebri, renal disease and stroke (1), obstructive sleep apnea, CKD, atrial fibrillation and diastolic heart failure (1) |
| Morath, Germany[35] | 4 | 5 | Critical | ARDS (5), MODS (5), circulatory shock or refractory fever (5) | AKI (5) | NR | NR | Grade 4 (5) | Diabetes (2), hypertension (3), coronary artery disease (1), schizophrenia and depression (1), atrial fibrillation (1), stroke (1), CKD, obesity (1), none (1) |
| Wang, China (Wuhan)[36] | 5 | 3 children | Critical | NR | AKI (3), pleural effusion (3), ascites (2), gastrointestinal involvement (2) | MV (3) | NR | Grade 4 (3) | Immunocompromised patient with acute lymphocytic leukemia (1) |
| Zhang, China[37] | 5 | 3 | Severe | ARDS (3), MODS (NR), septic shock (NR) | NR | HFOT (3) | NR | Grade 3 (3) | NR |
| De Prost, France[30] | 4 | 4 | Life-threatening with auto-antibodies against type I IFNs | ARDS (4), MODS (NR), septic shock (1) | Pneumonia (4) | IMV (3), NIMV (1), ECMO (2) | 7 (3), 8 (1) | Grade 3 (1), Grade 4 (3) | Obesity (2), pregnancy (1), hypertension and diabetes (1) |
| Fernandez, Spain[39] | 4 | 4 | Critical | MODS (3) | AKI (3), bilateral lung infiltrates (4), cardiac hypomotility (1), myocarditis (1), thrombotic events (1), acute limb ischemia (1), hepatic encephalopathy (1) | IMV (3) | 6, 7, 9, 11 | Grade 4 (4) | Obesity (2), diabetes (3), hypertension (4), liver transplantation (1), CKD (1), alcoholic liver cirrhosis (1) |
| Truong, USA[40] | 4 | 6 | Critical with hyperviscosity | ARDS (3), septic shock (4) | AKI (5), encephalopathy (3), cardiac arrhythmias (1), ischemia (1), shock liver (1), lower extremity deep venous thrombosis (1) | IMV | 5, 8, 14, 15 | Grade 4 (6) | Hypertension (3), diabetes (2), seizure disorder (1), COPD (2), coronary artery disease (1), cirrhosis (1) |
| Matsushita, Japan[41] | 4 | 5 | Severe | ARDS (1) | Bilateral consolidation in the lungs (1), ground-glass opacities in the lungs (3) | IMV (3) | NR | Grade 3 (2), Grade 4 (3) | End-stage renal disease on dialysis (2), malignancy (4), diabetes (1), hypertension (1), cerebral infarction and subdural hematoma (1) |
| Roshandel, Iran[42] | 4 | 5 | Critical | ARDS (5) | Severe pneumonia and/or ground-glass opacity (5) | MV (1), oxygen by mask (4) | NR | Grade 3 (4), Grade 4 (1) | Diabetes (2), hypertension (2), anemia (1), asthma (1), hypothyroidism (1), myocardial infarction (1), chronic lymphocytic leukemia (1), secondary hemophagocytic lymphohistiocytosis (1), hypercholesterolemia and coronary artery bypass grafting (1) |
| Case reports | |||||||||
| Akkoyunlu, Turkey[43] | 5 | 1 | Critical | NR | Bilateral multiple consolidations in the lungs | HFOT | NR | Grade 3 | Asthma, hypertension, diabetes |
| Altmayer, France[44] | 5 | 1 | Critical | ARDS | Bilateral interstitial infiltrates in the lungs | IMV | NR | Grade 4 | Hypertension, diabetes, overweight |
| Bagherzade, Iran[45] | 5 | 1 | Critical (respiratory arrest and loss of consciousness) | NR | Bilateral ground-glass opacities in the lungs | IMV | NR | Grade 4 | NR |
| Faqihi, Saudi Arabia[46] | 5 | 1 | Life-threatening | ARDS, sepsis | Peripheral neuropathy, peripheral bilateral ground-glass opacities in the lungs | IMV | NR | Grade 4 | None |
| Hua, China[47] | 5 | 1 | Critical | NR | Progressive lung infiltrates and diffuse gridding | IMV | NR | Grade 4 | COPD, hypertension, diabetes |
| Kamit, Turkey[48] | 5 | 1 child | Critical | ARDS, MODS, septic shock | Pneumonia, sinus tachycardia, metabolic acidosis, renal failure | IMV | NR | Grade 4 | Angelman syndrome and high-risk T cell acute lymphoblastic leukemia |
| Keith, USA[49] | 5 | 1 | Critical | ARDS, MODS, septic shock | Pneumonia, hypokinesis | CPAP | 7 | Grade 4 | Congestive heart failure, paroxysmal atrial fibrillation, obstructive sleep apnea, hypertension, obesity, diabetes |
| Lin, Taiwan[50] | 5 | 1 | Critical | NR | Pneumonia | IMV | NR | Grade 4 | NR |
| Ma, China[51] | 5 | 1 | Critical | NR | Bilateral ground-glass shadows in the lungs, dry gangrene in the finger, multiple cerebral infarctions, antiphospholipid syndrome | IMV | NR | Grade 4 | None |
| Ragab, Egypt[52] | 5 | 1 | Severe | ARDS | Diffuse bilateral patches of ground‐glass opacities in the lungs | HFOT | NR | Grade 3 | Diabetes, hypertension |
| Sadeghi, Iran[53] | 5 | 1 | Severe | NR | Bilateral multifocal peripheral ground-glass opacity, vasculopathy-related cutaneous manifestation and liver cholestasis | NR | NR | Grade 2 | None |
| Shi, China[54] | 5 | 1 | Critical | Septic shock | Rapidly progressive pulmonary lesions | HFOT | NR | Grade 4 | History of thyroid nodule |
| Tian, China[55] | 5 | 1 | Critical | NR | Ground-glass opacity with multiple patchy consolidations | HFOT | NR | Grade 3 | Diabetes |
| Yang, China[56] | 5 | 1 | Critical | ARDS | Pneumonia | IMV | NR | Grade 4 | None |
AKI, acute kidney injury; ARDS, acute respiratory distress syndrome; CKD, chronic kidney disease; CNS, central nervous system; COPD, chronic obstructive pulmonary disease; CPAP, continuous positive airway pressure; CSS, cytokine storm syndrome; ECMO, extracorporeal membrane oxygenation; HFOT, high-flow oxygen therapy; ICU, intensive care unit; IFN, interferon; IMV, invasive mechanical ventilation; IQR, interquartile range; MODS, multiple organ dysfunction syndrome; MV, mechanical ventilation; NIMV, non-invasive mechanical ventilation; NR, not reported; PSM, propensity score matching; SD, standard deviation; SOFA, Sequential Organ Function Assessment; TPE, therapeutic plasma exchange; UK, United Kingdom; USA, United States of America. * Evaluated by the authors of this review.
The RCT was categorized as Oxford level 3 (downgraded because of early termination and small sample size) [23]. The 4 matched case-control series and 12 single-group case series were categorized as Oxford level 4 [24], [25], [26], [27], [28], [30], [31], [32], [33], [34], [35], [38], [39], [40], [41], [42], and 3 single-group case series (downgraded because only 3 patients were included) and all case reports as Oxford level 5 [29], [36], [37], [43], [44], [45], [46], [47], [48], [49], [50], [51], [52], [53], [54], [55], [56].
3.2. Patients and disease characteristics
While most TPE-treated patients had critical or life-threatening COVID-19, a few patients with severe or moderate disease were also included in the selected studies (Table 1). Using the Penn grading scale [22], we estimated that all TPE-treated patients had grade 3 or 4 CSS (except one patient with grade 2 CSS). Available Sequential Organ Failure Assessment (SOFA) scores, which are based on the degree of organ dysfunction [57] and may help to predict outcomes in critically ill patients, ranged between 2 and 15 (median ranging from 3 to 11).
Many studies included patients with ARDS [23], [24], [27], [28], [29], [30], [31], [33], [34], [35], [37], [38], [40], [41], [42], [44], [46], [48], [49], [52], [56], MODS [23], [27], [28], [31], [35], [38], [39], [48], [49], and/or septic shock [23], [27], [28], [30], [31], [34], [35], [38], [40], [46], [48], [49], [54]. Other damaged organs included kidneys, liver, brain, heart, and gastrointestinal tract. Acute limb ischemia, neuropathies, and cutaneous manifestations were also reported [39], [46], [53]. Almost all patients received oxygen support before TPE administration (e.g., mechanical ventilation, continuous positive airway pressure, or high-flow oxygen therapy) (Table 1). Patients often had underlying comorbidities associated with poor clinical outcomes in COVID-19, such as diabetes, hypertension, obesity, cardiovascular disease, and/or chronic kidney disease (Table 1).
3.3. Treatment characteristics
The rationale for TPE treatment in patients with COVID-19 varied between studies, but the most frequent reasons were critical disease consisting of either severe ARDS with high and rapidly increasing ferritin or D-dimer levels [23], [24], [25], [27], [28], [29], [30], [31], [32], [33], [34], [35], [37], [38], [39], [40], [41], [42], [43], [44], [46], [47], [48], [50], [51], [52], [55], [56], and/or sepsis or MODS [23], [27], [28], [31], [34], [35], [36], [38], [40], [45], [46], [48], [49], [54] ( Table 2).
Table 2.
Treatment of COVID-19 patients who received TPE.
| First author, country, study group | Number of patients | Rationale for TPE | Replacement fluid and TPE system | Volume of replacement fluid | Number of TPE treatment | Frequency of TPE treatment | Adjunct immunoregulatory therapy | Other adjunct treatment linked to TPE | Other adjunct treatment not linked to TPE |
|---|---|---|---|---|---|---|---|---|---|
| Randomized controlled clinical trial | |||||||||
| Faqihi, Saudi Arabia[23] TPE group | 43 | Patients with life-threatening COVID-19 (ARDS and septic shock or MODS, and with ≥ 1 criteria defining CSS) | FFP or Octaplas | 1.5 plasma volume for the first session, then 1 plasma volume | Median (IQR): 3 (1–5) | Daily | None | Norepinephrine (1 patient) | Antivirals (ribavirin), antibacterial medications, dexamethasone, anticoagulation |
| Control group | 44 | Patients with life-threatening COVID-19 (ARDS and septic shock or MODS, and with ≥ 1 criteria defining CSS) | NA | NA | NA | NA | None | Norepinephrine (1 patient) | Antivirals (ribavirin), antibacterial medications, dexamethasone, anticoagulation |
| Matched case-control series | |||||||||
| Arulkumaran, UK[24] TPE group | 7 | Critically ill patients with severe respiratory failure and elevated thrombo-inflammatory markers | Octaplas LG; Spectra Optia Apheresis System | 3 L | 5–10 | Daily | None | None | Intermediate dose LMWH |
| Control group | 7 | NA | NA | NA | NA | NA | None | NA | Intermediate dose LMWH if confirmed thromboembolic event |
| Gucyetmez, Turkey[25] TPE group | 18 | Patients with pneumonia and D-dimer ≥ 2 mg/L | NR | NR | 3 | NR | Cytokine filters: 2 (16.7%); IL-6 blocker: 7 (58.3%); steroids: 7 (58.3%) | None | Therapeutic anticoagulation (UFH or LMWH), favipiravir, hydroxychloroquine, azithromycin |
| Control group | 35 | NA | NA | NA | NA | NA | Cytokine filters: 1 (8.3%); IL-6 blocker: 6 (50%); steroids: 7 (58.3%) | NA | NA |
| Kamran, Pakistan[26] TPE group | 45 | Patients with CSS | FFP and normal saline (2:1); COBE Spectra Apheresis System/ continuous flow centrifugation | 1.5 plasma volume | Median (IQR): 2.25 (1–5) | Daily | Steroids, methylprednisolone | None | Anticoagulation |
| Control group | 45 | NA | NA | NA | NA | NA | Steroids, methylprednisolone | NA | Anticoagulation |
| Khamis, Oman[27] TPE group | 11 | Patients with confirmed or imminent respiratory failure and ARDS, severe pneumonia, septic shock or MODS | FFP; Spectra Optia Apheresis System | Body weight (kg) × (1/13) × (100-hematocrit) | 5 | Daily | Tocilizumab: 55% of patients | NR | NR |
| Control group | 20 | NA | NA | NA | NA | NA | Tocilizumab: 30% of patients | NA | NR |
| Single-group case series | |||||||||
| Adeli, Iran[28] | 8 | Patients with septic shock and ARDS with poor response to antiviral treatment, corticosteroid therapy and interferon administration | 4 units of FFP, 5 vials of albumin and normal saline | 2 L | 3–5 | Daily | Interferon β, corticosteroid therapy (dexamethasone) | One or two 10–20 mL calcium gluconate (20%) | Hydroxychloroquine sulfate, antiviral drugs |
| Alharthy, Saudi Arabia[29] | 3 | Patients with life-threatening COVID-19 (ARDS, thromboembolic disease, and low GCS) and with a microangiopathic pattern | Octaplas; Spectra Optia Apheresis System | 1.5 plasma volume for the first dose and then 1 plasma volume | 5 | Daily | Hydrocortisone, interferon β-1b | Intravenous calcium replacement | Chlorpheniramine, ribavirin, antibiotics, therapeutic anticoagulation |
| Faqihi, Saudi Arabia[31] | 10 | Patients with life-threatening COVID-19 (ARDS and septic shock, and with > 3 risk factors for CSS) | Albumin 5% or FFP in patients with coagulopathy; Spectra Optia Apheresis System |
1.0–1.5 plasma volumes | 5–7 | Daily | Intravenous hydrocortisone | Norepinephrine, vasopressin | Hydroxychloroquine, antibiotics, prophylactic anticoagulation |
| Gluck, USA[32] | 10 | Patients with Penn class 3 or 4 CSS complicating COVID-19 | 5% albumin (9 patients) or FFP in patients with coagulopathy (1 patient); Spectra Optia Apheresis System | 1.0 plasma volume | 5 | Daily for 2 consecutive days then every other day | None | None | Hydroxychloroquine (2 patients) |
| Hashemian, Iran[33] | 15 | Patients with ARDS | 5% human albumin solution and 0.9% saline. 4 patients receive CCP; JMS fully automated SDS-20 hemodialysis machine | 40 mL/kg body weight | 3 | 3 times a week | CCP in 4 patients | None | Antiviral drugs, meropenem in patients with respiratory tract infection |
| Jaiswal, Dubai, United Arab Emirates[34] | 14 | Critically ill patients (ARDS, sepsis and septic shock) | FFP | 30–40 mL/kg bodyweight | 1 | NA | 500 mL of CCP 8 h after TPE; methylprednisolone | None | Enoxaparin |
| Keith, USA[38] | 8 | Critically ill patients (ARDS, sepsis and septic shock) | FFP; Spectra Optia Apheresis System | Approximately 1 plasma volume | 1–7 | Daily | CCP (4 patients); methylprednisolone (7 patients); tocilizumab (2 patients) | Vasopressor (8 patients) | Hydroxychloroquine (5 patients), azithromycin (7 patients), ivermectin (1 patient), anticoagulants (8 patients) |
| Morath, Germany[35] | 5 | MODS, ARDS, and AKI | FFP | Median: 3.39 L | 1 | NA | Tocilizumab (1 patient), interferon (1 patient), prednisolone (2 patients), immunoglobulins (1 patient), CCP (2 patients) | Vasopressor treatment (4 patients) | Antiviral treatment, antibiotics, antimycotics, hydroxychloroquine. |
| Wang, China (Wuhan)[36] | 3 children | Critically ill pediatric patients with AKI | NR | NR | 2–4 | Variable | Corticosteroid therapy, immunoglobulins | None | Anticoagulation (heparin), antibiotics, antiviral treatment. |
| Zhang, China[37] | 3 | Severe COVID-19 patients with ARDS | FFP; plasma separator multi-filtration system | About 3 L | 1 | NA | Interferon α-2b | None | Antiviral treatment, including arbidol. |
| De Prost, France[30] | 4 | Patients with life-threatening pneumonia (ARDS, high concentrations of neutralizing auto-antibodies against type I IFNs) | Albumin 5% and plasma in different proportions; continuous flow centrifugation (3 patients) or plasma filtration (1 patient) | Range: 32–57 mL/kg | 3–4 | Daily or every other day | Dexamethasone | None | None |
| Fernandez, Spain[39] | 4 | Critically ill adults with COVID-19 pneumonia that failed conventional interventions | Human albumin (5%) | 1.2 plasma volumes (range: 3.8–5 L) | 2–6 | Every other day | FFP and immunoglobulins (4 patients), dexamethasone (2 patients), methylprednisolone (2 patients), interferon β-1a (2 patients), tocilizumab (1 patient), anakinra and hydrocortisone (1 patient) | Norepinephrine (2 patients) | Hydroxychloroquine, antiviral drugs, antibiotics, heparin sodium |
| Truong, USA[40] | 6 | Critically ill patients with COVID-19-associated hyperviscosity | FFP | 1 plasma volume | 2–3 | Daily | NR | Vasopressors (2) | Anticoagulants (heparin, argatroban, bivalirudin, enoxaparin) |
| Matsushita, Japan[41] | 5 | ARDS and/or labored respiration and/or tracheal intubation. | FFP | 2.5–3 L | 3–7 | Daily or every other day | Glucocorticoid (5 patients), methylprednisolone pulse therapy (3 patients), hemodiafiltration (3 patients), hemoperfusion (2 patients) | NR | Anticoagulants (heparin), antiviral drugs, antibiotics |
| Roshandel, Iran[42] | 5 | Patients with ARDS | Albumin 5% + FFP for the 2 first sessions and CCP for the third session | 0.75 plasma volume | 3 | Daily | Steroids | NR | Antiviral, anti-fungal and antibacterial treatments, |
| Case reports | |||||||||
| Akkoyunlu, Turkey[43] | 1 | Critically ill patient whose clinical status worsened despite antiviral and tocilizumab treatments | FFP; multifiltrate model | 10 units | 1 | NA | Tocilizumab, prednisolone | None | Hydroxychloroquine, antibiotics, antiviral treatment, anticoagulation (enoxaparine) |
| Altmayer, France[44] | 1 | Patient with ARDS and CSS | Albumin 5%; Spectra Optia | 1.2 plasma volume | 4 | Every other day | NR | None | Antibiotics |
| Bagherzade, Iran[45] | 1 | COVID-19 patient with respiratory arrest and loss of consciousness | NR | NR | 5 | Daily | Corticosteroid, interferon β-1b, dexamethasone | Vasopressors (norepinephrine) | Hydroxychloroquine, antiviral treatment, antibiotics, prophylactic anticoagulation |
| Faqihi, Saudi Arabia[46] | 1 | Patient with life-threatening COVID-19 characterized by peripheral neuropathy, ARDS, sepsis, and hyperinflammation | Octaplas; Spectra Optia Apheresis System | 1.5 plasma volume for the first dose; then,1 plasma volume | 3 | Daily | Hydrocortisone, interferon β-1b | Intravenous vasopressors | Antiviral treatment, antibiotics, prophylactic anticoagulation |
| Hua, China[47] | 1 | Critical COVID-19 patient with prolonged IMV | FFP; Diapact CRRT system, and a filter membrane-based apparatus | 3 L | 3 | Daily | Methylprednisolone | Norepinephrine | Antiviral drugs |
| Kamit, Turkey[48] | 1 child | Child with ARDS with hyperferritinemic MODS, and CSS | FFP | 1.5 plasma volume for the 2 first doses; then 1 plasma volume | 4 | Daily | Tocilizumab, hydrocortisone, intravenous immunoglobulin | Epinephrine, norepinephrine | Antiviral drug, antibiotics, levetiracetam |
| Keith, USA[49] | 1 | Patient with pneumonia, septic shock and MOF | FFP | 4.5 L | 1 | NA | None | Norepinephrine and midodrine | Amiodarone with magnesium and potassium replacement, digoxin, home sotalol |
| Lin, Taiwan[50] | 1 | Critically ill COVID-19 patient with CSS | FFP | 0.065 × body weight × (1-hematocrit); 1 plasma (body weight × 40 mL); 1.5 plasma (body weight × 60 mL) | 3 | Daily | NR | NR | Continuous venovenous hemofiltration |
| Ma, China[51] | 1 | Critically ill COVID-19 patient with CSS | NR | NR | 3 | Daily | Gamma globulin | NR | Antibiotics, antiviral drugs, LMWH, aspirin |
| Ragab, Egypt[52] | 1 | Patient with severe COVID‐19, ARDS and CSS | FFP and CCP (400 mL) | (0.065 × body weight) × (1 − hematocrit as a fraction) | 1 | NA | CCP, corticosteroids, methylprednisolone, dexamethasone, tocilizumab | Hydroxychloroquine sulfate, antibiotics, antiviral drugs, anticoagulant (enoxaparin sodium) | |
| Sadeghi, Iran[53] | 1 | COVID-19 with vasculopathy-related cutaneous manifestation and liver cholestasis | CCP | NR | 3 | NR | One unit of washed packed cells injection, prednisolone | NR | Hydroxychloroquine, naltrexone, hydroxyzine, antibiotics |
| Shi, China[54] | 1 | Critically ill COVID-19 patient with CSS | FFP | 6 L | 4 | Daily | Human granulocyte-colony stimulating factor, thymalfasin, intravenous immunoglobulin, corticosteroids (methylprednisolone) | Vasopressors (dopamine, noradrenalin) | Antiviral drugs, antibiotics |
| Tian, China[55] | 1 | Critically ill COVID-19 patient | Multifiltrate bedside blood purifier and plasma separator | 2 L | 1 | NA | Thymalfasin, immune globulin, methylprednisolone | NR | Antiviral drugs, antibiotics, antimycotics, anticoagulant (enoxaparin) |
| Yang, China[56] | 1 | Critically ill COVID-19 patient with pneumonia that did not improve with tocilizumab and continuous renal replacement therapy | Albumin solution; double filtration plasmapheresis with a plasma separator and a plasma fractionator | 3 L | 3 | Daily | Multiple blood transfusion including 400 mL CCP, methylprednisolone, tocilizumab | Norepinephrine | NR |
AKI, acute kidney injury; ARDS, acute respiratory distress syndrome; CCP, COVID-19 convalescent plasma; COVID-19, coronavirus disease 2019; CRRT, continuous renal replacement therapy; CSS, cytokine storm syndrome; FFP, fresh frozen plasma; GCS, Glasgow Coma Scale; IFN, interferon; IQR, interquartile range; IL, interleukin; IMV, invasive mechanical ventilation; LMWH, low molecular weight heparin; MODS, multiple organ dysfunction syndrome; MOF, multiple-organ failure; NA, not applicable; NR, not reported; TPE, therapeutic plasma exchange; UFH, unfractionated heparin; UK, United Kingdom; USA, United States of America.
In general, three to five TPE sessions were performed [23], [25], [26], [27], [28], [29], [30], [32], [33], [36], [41], [42], [44], [45], [46], [47], [48], [50], [51], [53], [54], [56], with a daily or every other day frequency [23], [24], [26], [27], [28], [29], [30], [31], [32], [38], [39], [40], [41], [42], [44], [45], [46], [47], [48], [50], [51], [54], [56]. The most common replacement fluids were fresh frozen plasma (FFP) [23], [24], [27], [29], [30], [31], [32], [34], [35], [37], [38], [40], [41], [42], [43], [46], [47], [48], [49], [50], [52], [54] and 5% albumin solutions [30], [31], [32], [33], [39], [41], [42], [44], [56] (Table 2). In some studies, FFP was used only for patients with coagulopathies and 5% albumin was used for other patients [31], [32]. Different methods were used to determine the volume of replacement fluid, which was frequently expressed in plasma volume (range: 0.75–1.5) [23], [26], [29], [31], [32], [38], [39], [40], [42], [44], [46], [48] or in liters (range: 2–6) [24], [28], [35], [37], [41], [47], [49], [54], [55], [56].
Besides TPE, many patients received adjunct immunoregulatory therapies, such as cytokine filtration, IL-6 blockers, IL-1 receptor antagonists, corticosteroids, IFN-α, IFN-β, COVID-19 convalescent plasma (CCP), immunoglobulins, human granulocyte-colony stimulating factors, thymalfasin, washed packed cells, hemodiafiltrations, hemoperfusions, or blood transfusions (Table 2). Patients often also received other treatments, including systemic anticoagulants, antibiotics, antiviral drugs, antimycotics, anti-inflammatory drugs, vasopressors, and/or renal replacement therapy. Citrate infusions were used as standard anticoagulant during TPE sessions [58], and some patients received calcium infusions to prevent hypocalcemia and citrate toxicity.
3.4. Evolution of immune-inflammatory biomarkers
Since the main objectives of TPE are to decrease pro-inflammatory cytokines levels and correct coagulopathies, dynamic monitoring of these parameters is useful to evaluate the ability of TPE to abate the CSS.
The levels of IL-6, a pro-inflammatory cytokine playing an important role in CSS and used as cytokinemia marker [59], [60], decreased in most studies with available results [23], [24], [25], [27], [31], [32], [33], [35], [36], [37], [39], [42], [46], [47], [48], [51], [52], [55], [56] ( Table 3). This decrease was significant in six of eight case series evaluating statistical significance [23], [24], [25], [31], [32], [33], [35], [37]. In one case report, IL-6 levels remained stable after TPE [44].
Table 3.
Impact of treatment on laboratory parameters in COVID-19 patients who received TPE.
| First author, country, study group | Number of patients | Ferritin | D-dimer | CRP | IL-6 | Coagulation factor | Other laboratory parameters |
|---|---|---|---|---|---|---|---|
| Randomized controlled clinical trial | |||||||
| Faqihi, Saudi Arabia[23] TPE group | 43 | Median (IQR): from 987 ng/mL (319–1655) to 299 ng/mL (146–655)* | Median (IQR): from 4.9 mcg/mL (2.9–7.9) to 0.9 mcg/mL (0.5–1.4)* | Median (IQR): from 246 mg/L (157–356) to 45 mg/L (11–99)* | Median (IQR): from 458 pg/mL (225–1091) to 35 pg/mL (18–112)* | ADAMTS-13 activity: median (IQR): from 17% (6–38%) to 42% (29–56%)* | Lymphocytes: median (IQR): from 0.5 × 109/L (0.2–0.7) to 1.0 × 109/L (0.6–1.4)* |
| Control group | 44 | Median (IQR): from 320 ng/mL (75–675) to 287 ng/mL (106–468) | Median (IQR): from 2.5 mcg/mL (1.4–4.6) to 0.95 mcg/mL (0.6–3.2)* | Median (IQR): from 234 mg/L (109–359) to 78 mg/L (31–135)* | Median (IQR): from 122.5 pg/mL (48.7–262.8) to 27.0 pg/mL (17–144)* | ADAMTS-13 activity: median (IQR): from 37% (26–57%) to 32% (22–48%)* | Lymphocytes: median (IQR): from 0.6 × 109/L (0.2–1) to 0.7 × 109/L (0.3–1.1)* |
| Matched case-control series | |||||||
| Arulkumaran, UK[24] TPE group | 7 | Median (IQR): from 1003 ng/mL (514–3373) to 568 ng/mL (331–685)* | Median (IQR): from 4110 µg/L FEU (2690–6483) to 2385 µg/L FEU (968–3790)* | Median (IQR): from 300 mg/L (128–349) to 167 mg/L (38–271) | Median (IQR): from 27 pg/mL (8–52) to 18 pg/mL (10–117) | Median (IQR): fibrinogen: from 4.96 g/L (4.41–9.50) to 3.98 g/L (3.39–4.93)* ; ADAMTS-13 activity from 75% (66–83) to 79% (77–83) | Lymphocytes: median (IQR): from 0.91 × 109/L (0.53–1.10) to 1.40 × 109/L (0.90–1.95)* |
| Control group | 7 | NR | NR | NR | NR | NR | No significant recovery of lymphocytes among control group patients |
| Gucyetmez, Turkey[25] TPE group | 18 | Median (min–max): from 1268 ng/mL (399–6110) to 405 ng/mL (157–1650)* | Median (min-max): from 7.8 mg/L (2.1–35.2) to 1.3 mg/L (0.6–3.9)* | Median (min-max): from 11.8 (0.4–29.7) to 0.9 (0.3–7.2)* | Median (min-max): IL-6 from 161 (36.2–2958) to 24.5 (1.5–130)* | NR | Lymphocytes: median (IQR): from 0.91 × 109/L (0.5–1.3) to 1.02 × 109/L (0.77–1.27) |
| Control group | 35 | NR | NR | NR | NR | NR | NR |
| Kamran, Pakistan[26] TPE group | 45 | Median (range): 1500 ng/mL (336–7877) | Median (range): 350 ng/mL (150–1700) | Median (range):145 µg/mL (21–278) | Median (range): 78 (6–400) | Platelet count, median (range): 180 × 109/L (70–1100) | Lymphocyte: median (range): 700 × 109/L (200–2100) |
| Control group | 45 | Median (range): 1410 ng/mL (395–4500) | Median (range): 647 ng/mL (300–1100) | Median (range): 147 µg/mL (56–260) | Median (range): 104 (7–178) | Platelet count, median (range): 187 × 109/L (56–450) | Lymphocyte: median (range): 790 × 109/L (230–1400) |
| Khamis, Oman[27] TPE group | 11 | Range: from 221 to 2329 ng/mL to 143–1088 ng/mL (decrease in 9/11 patients) | Range: from 0.6 to 27 ng/mL to 0.89–4.9 ng/mL (decrease in 6/11 patients) | Range: from 49 to 344 mg/L to 12–416 mg/L (decrease in 9/11 patients) | Range: from 19 to 3415 pg/mL to 5–284 pg/mL (decrease in 6/8 patients) | NR | Lymphocyte: range: from 0.6 to 1.9 × 109/L to 0.6–3 × 109/L (increase in 9/11 patients) |
| Control group | 20 | NR | NR | NR | NR | NR | NR |
| Single-group case series | |||||||
| Adeli, Iran[28] | 8 | NR | NR | NR | NR | NR | NR |
| Alharthy, Saudi Arabia[29] | 3 | Range at baseline: 778–1289 µg/L, subtle decrease post-TPE | Range at baseline: 11.9–13.2 mg/L, subtle decrease post-TPE | Range at baseline: 142–201 mg/L, subtle decrease post-TPE | NR | ADAMTS-13 activity: range: from 8%− 15–22%− 28% | All inflammatory biomarkers and lymphocyte counts were equally normalized post-TPE |
| Faqihi, Saudi Arabia[31] | 10 | Median (IQR): from 1233 µg/L (799–1758) to 290 µg/L (201–322)* | Median (IQR): from 7.4 mg/L (4.9–11.7) to 0.9 mg/L (0.7–1.2)* | Median (IQR): from 71.3 mg/L (51.3–89.7) to 13.2 mg/L (7.2–26.4)* | Median (IQR): from 159.5 pg/mL (88.9–182.3) to 31.2 pg/mL (15.4–49.8)* | NR | Lymphocytes: median (IQR): from 0.6 × 109/L (0.45–0.8) to 1.15 × 109/L (0.8–1.4) |
| Gluck, USA[32] | 10 | NR | NR | Median: from 149.9 to 24.8 mg/L.* All patients demonstrated a reduction in CRP. | Median: from 32.04 to 5.92 pg/mL.* All patients demonstrated a reduction in IL-6. | NR | NR |
| Hashemian, Iran[33] | 15 | Mean ± SD: from 1027.3 ± 396.9 ng/mL to 654.0 ± 320.0 ng/mL* | NR | Mean ± SD: from 47.3 ± 17.7 mg/dL to 28.5 ± 20.5 mg/dL* | Mean ± SD: from 8.3 ± 1.8 pg/mL to 5.7 ± 1.3 pg/mL* | NR | T cell subset numbers were significantly decreased. The levels of all lymphocyte subsets increased to above the levels seen at baseline. |
| Jaiswal, Dubai, United Arab Emirates[34] | 14 | Mean ± SD: from 1416.25 ± 1150.62 ng/mL to 1051.42 ± 740.96 ng/mL | Mean ± SD: from 4.20 ± 5.46 mg/mL to 4.21 ± 5.93 mg/mL | Mean ± SD: from 86.74 ± 79.86 mg/dL to 30.56 ± 30.73 mg/dL | NR | NR | Lymphocyte: mean ± SD: from 0.70 ± 0.54 × 109/L to 1.04 ± 0.49 × 109/L |
| Keith, USA[38] | 8 | Ferritin levels decreased following 18/22 TPE treatments. Mean ± SD: from 1404.9 ± 696.3–984.4 ± 684.5 after the first TPE.* | D-dimer levels decreased following 15/23 TPE treatments. Mean ± SD: from 6187.3 ± 8758.9–3588.8 ± 3332.0 after the first TPE. | CRP levels decreased following 18/22 TPE treatments. Mean ± SD: from 266.1 ± 169.7–176.5 ± 162.6 after the first TPE.* | NR | NR | NR |
| Morath, Germany[35] | 5 | Significant reduction of ferritin (−49%)* | Significant reduction of D-dimer (−47%)* | Striking reduction of CRP (−47%)* | Striking reduction of IL-6 (−74%)* | NR | NR |
| Wang, China (Wuhan)[36] | 3 children | NR | NR | NR | Improved cytokine profile (IL-6 levels decreased) | NR | NR |
| Zhang, China[37] | 3 | NR | NR | Decreased from 84.8 to 196.3–5.2–24.4 mg/L* | Decreased from 12.14 to 142.9 pg/mL to 2.55–6.42 pg/mL | NR | The values of the neutrophil-to-lymphocyte ratio were significantly decreased. Lymphocytes (range): from 0.52 to 1.07 × 109/L to 1.03–2.91 × 109/L |
| De Prost, France[30] | 4 | NR | NR | NR | NR | NR | TPE decreased the concentrations of autoantibodies against type I IFN in all four patients whereas anti-SARS-CoV-2 antibody levels remained stable |
| Fernandez, Spain[39] | 4 | Range: from 1573 to 3137–394–627 | Range: from 3900 to 13200–2500–4600 | Range: from 0.4 to 10.81–0.4–2.60 | Decreased in 3 patients | Platelet count: range: from 120 to 462–30–360 | Lymphocytes: range: from 0.3 to 2.3 × 109/L to 0.5–3.1 × 109/L |
| Truong, USA[40] | 6 | NR | Median (range): from 5921 ng/mL (1134–60000) to 4893 ng/mL (620–7518) | Median (range): from 292 mg/L (136–329) to 84 mg/L (31–211) | NR | Median (range): viscosity: from 3.75 cP (2.6–4.2) to 1.6 cP (1.5–1.9); fibrinogen from 739 mg/dL (601–1188) to 359 mg/dL (235–461) | NR |
| Matsushita, Japan[41] | 5 | NR | NR | NR | NR | NR | NR |
| Roshandel, Iran[42] | 5 | NR | Range: from 0.4 to 14 mg/dL to 0.5–9 mg/dL at 1 day post-TPE | Range: from 3.2 to 80 mg/L to 5–61 mg/L at 1 day post-TPE | Range: from 42.5 to 109.4 pg/mL to 3.52–5.98 pg/mL at 7 days post-TPE | Fibrinogen; range: from 186 to 346 mg/dL to 186–303 mg/dL at 1 day post-TPE; Platelet count: range: from 121 to 529 × 1000/µL to 55–563 × 1000/µL | Lymphocyte: range: from 5%− 71–3%− 88% at 1 day post-TPE |
| Case reports | |||||||
| Akkoyunlu, Turkey[43] | 1 | From 106 to 37 and 13 ng/mL at 10 and 24 days post-TPE | From 1238 to 498 and 193 ng/mL at 10 and 24 days post-TPE | From 8.7 to 0.2 and 0.3 mg/L at 10 and 24 days post-TPE | NR | NR | Lymphocytes increased from 430 to 2770 and 2200 × 1000/µL at 10 and 24 days post-TPE |
| Altmayer, France[44] | 1 | NR | D-dimer levels decreased | CRP levels decreased | IL-6 levels remained stable, except for a patient with Pseudomonas aeruginosa pneumonia | Fibrinogen levels remained stable, except for a patient with Pseudomonas aeruginosa pneumonia | NR |
| Bagherzade, Iran[45] | 1 | NR | NR | No changes in CRP levels (25 mg/L) | NR | NR | Lymphocyte percentage: from 1.6% to 6.3%. |
| Faqihi, Saudi Arabia[46] | 1 | From 1123 to 382 ng/mL | From 3.6–0.8 µg/mL | From 247–18 mg/L | From 778–9.6 pg/mL | ADAMTS-13 activity: from 8% to 22% | Lymphocyte counts: from 0.51 to 1.1 × 109/L |
| Hua, China[47] | 1 | NR | NR | From 50.2 mg/L to 19.6 mg/L | From 3815–286.9 pg/mL | NR | NR |
| Kamit, Turkey[48] | 1 child | From > 100000–45268 ng/mL | From 19.44–8.7 mg/L | From 292.5–25.9 mg/L | From 25931–17140 pg/mL | Platelets: from 26000 to 28000; fibrinogen: from 4.5 to 1.27 g/L | NR |
| Keith, USA[49] | 1 | NR | NR | NR | NR | NR | NR |
| Lin, Taiwan[50] | 1 | NR | NR | NR | NR | NR | NR |
| Ma, China[51] | 1 | NR | NR | From 192.7 mg/L to 44.4 mg/L | From 236.3–92.05 pg/mL | NR | NR |
| Ragab, Egypt[52] | 1 | Ferritin levels decreased after TPE | D-dimer levels decreased after TPE | CRP started to decline the next day after TPE | IL-6 levels decreased after TPE | NR | Lymphocytes increased after TPE |
| Sadeghi, Iran[53] | 1 | NR | NR | NR | NR | NR | NR |
| Shi, China[54] | 1 | NR | NR | From 4.87–6.02 mg/L | NR | Platelet count: from 161 to 129 × 109/L | Lymphocytes: from 0.6 to 1.1 × 109/L |
| Tian, China[55] | 1 | NR | D-dimer remained elevated | NR | From 5.59 on day 2–1.69 on day 4 and 87.14 on day 6 | NR | Lymphocytes: from 228 on day 1–200 on day 3 and 585 on day 5 |
| Yang, China[56] | 1 | NR | D-dimer levels decreased | No changes in CRP levels | IL-6 levels decreased | Platelet counts increased | NR |
ADAMTS-13, a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13; cP, centipoise; CRP, C-reactive protein; IFN, interferon; IQR, interquartile range; IL, interleukin; NR, not reported; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; SD, standard deviation; TPE, therapeutic plasma exchange; UK, United Kingdom; USA, United States of America. *Statistically significant difference.
The levels of CRP, a marker of inflammation and cytokinemia [10], [60], decreased in most studies with available results [23], [24], [25], [27], [29], [31], [32], [33], [34], [35], [37], [38], [39], [40], [42], [43], [44], [46], [47], [48], [51], [52] (Table 3). This decrease was significant in 8/9 case series evaluating statistical significance [23], [24], [25], [31], [32], [33], [35], [37], [38]. However, in three case reports, no changes or slight increases in CRP levels were observed following TPE [45], [54], [56].
Ferritin is a marker of macrophage activation and vascular damage, whose gene transcription is elicited by IL-6. It represents a negative prognostic factor and is associated with increasing oxygen needs [60], [61], [62]. In 15 studies with available data, ferritin concentrations decreased after TPE [23], [24], [25], [27], [29], [31], [33], [34], [35], [38], [39], [43], [46], [48], [52] (Table 3). This decrease was significant in the seven studies evaluating statistical significance [23], [24], [25], [31], [33], [35], [38].
In most studies with available data, concentrations of D-dimer, a fibrin degradation product used as hypercoagulability marker [60], [63], decreased after TPE [23], [24], [25], [27], [29], [31], [35], [38], [39], [40], [42], [43], [44], [46], [48], [52], [56] (Table 3). This decrease was significant in five of six studies evaluating statistical significance [23], [24], [25], [31], [35], [38]. In one case series and one case report, D-dimer levels were stable [34], [55].
While three studies using FFP or artificial Octaplas LG (Octapharma, Manchester, UK; a pooled FFP product that has undergone pathogen inactivation) as replacement fluid showed that fibrinogen levels decreased following TPE [24], [40], [48], fibrinogen levels seemed stable in two studies using albumin or a mix of albumin and FFP as replacement fluid [42], [44] (Table 3). Four studies showed that the activity of disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS-13; von Willebrand factor-cleaving protease) [64] increased after TPE [23], [24], [29], [46]. Platelet count decreased in two studies [39], [54], but tended to increase in two other studies [48], [56]. In a fifth study, platelet count decreased in some patients but increased in others [42]. Viscosity decreased after TPE in a study in patients with COVID-19 hyperviscosity [40].
Several studies evaluated the effects of TPE on lymphopenia (i.e., low lymphocyte count, a marker of disease severity) [65] and showed an increase in the absolute lymphocyte count after TPE [23], [24], [25], [27], [29], [31], [33], [34], [37], [39], [42], [43], [45], [46], [52], [54], [55] (Table 3).
In the matched case-control study including the highest number of TPE-treated patients, lower D-dimer and IL-6 levels were observed in the TPE versus the control group, while no differences were observed in terms of ferritin, CRP, platelet, and lymphocyte count [26] (Table 3).
3.5. Clinical evolution
Mortality rates were highly variable between studies [23], [24], [25], [26], [27], [28], [29], [30], [31], [32], [33], [34], [35], [36], [37], [38], [39], [40], [41], [42] ( Table 4). In the RCT, the addition of TPE to standard treatment was associated with a lower 35-day mortality (20.9% versus 34.1%), but the difference did not reach statistical significance [23]. In the three matched case-control series with available results, mortality was reduced in patients following TPE versus matched controls [25], [26], [27]. In a single-group case series, a case-crossover design showed that TPE increased survival by 17% [33].
Table 4.
Safety of TPE and impact of treatment on clinical evolution in COVID-19 patients who received TPE.
| First author, country, study group | Number of patients | Mortality | Discharge rate | Length of hospitalization | Evolution of clinical symptoms | Ventilation status/oxygenation | TPE safety |
|---|---|---|---|---|---|---|---|
| Randomized controlled clinical trial | |||||||
| Faqihi, Saudi Arabia[23] TPE group | 43 | 35-day mortality: 20.9% | NR | ICU length of stay: median (IQR): 19 days (12–27) | SOFA score: median (IQR): from 10 (7–13) to 2 (1–3)* | PaO2:FiO2 ratio: median (IQR): 135 (72–198) to 300 (220–380)* Duration of MV: median (IQR): 15 days (8–22)* | No adverse events recorded |
| Control group | 44 | 35-day mortality: 34.1% | NR | ICU length of stay (days): median (IQR): 26 days (11.5–31.5) | SOFA score: median (IQR): from 9 (6–12) to 4.5 (3.5–5.5)* | PaO2:FiO2 ratio: median (IQR): 125 (75.5–174.5) to 255 (205–315)* ; duration of MV: median (IQR): 19 days (8–30)* | NA |
| Matched case-control series | |||||||
| Arulkumaran, UK[24] TPE group | 7 | 0% | 100% | NR | Positive impact on organ function; 3 patients deteriorated once TPE was stopped | Within 24 h, a further 2 patients required IMV; PaO2:FiO2 ratio increased in all patients from 87 (81–148) mm Hg to 136 (120–194) mm Hg | No major adverse events, including thrombotic or bleeding episodes, occurred during TPE |
| Control group | 7 | NR | NR | NR | NR | No significant improvement in PaO2:FiO2 ratio | NA |
| Gucyetmez, Turkey[25] TPE group | 18 | 8.3% | NR | LOS in ICU: 20 ± 10 days | NR | Duration of IMV: 316 h ± 271. PaO2:FiO2, mm Hg: from 108 (106) to 104 ± 32.4 in the first 48 h | NR |
| Control group | 35 | 58.3% | NR | LOS in ICU: 14 ± 5 days | NR | Duration of IMV: 278 h ± 139. PaO2:FiO2, mm Hg: from 125 (103) to 120 ± 32.5. in the first 48 h | NA |
| Kamran, Pakistan[26] TPE group | 45 | Overall survival: 91.1% (95% CI: 78.33–97.76) | NR | Median (range): 10 days (4–37) | Time for CSS resolution: median (range): 6 days (2–23)* | NR | Two patients developed TPE-related complications (femoral artery puncture, thrombophlebitis of femoral vein with DVT) |
| Control group | 45 | Overall survival: 61.5% (95% CI: 51.29–78.76) | NR | Median (range): 15 days (7–45) | Time for CSS resolution: median (range): 12 days (5–42) | NR | NA |
| Khamis, Oman[27] TPE group | 11 | 28-day mortality: 0%; all-cause mortality: 9.1% | ICU LOS: 14 days (8–20); total LOS: 19 days (9–21) | SOFA score: range: from 2 to 13–2–13. SOFA score decreased in 9/11 patients | Extubation rate: 73%. Of note, 9.1% of patients were not intubated. PaO2:FiO2 ratio: from 98 to 176–136–265 (increased in 10/10 patients) | Hypotension (1 patient) | |
| Control group | 20 | 28-day mortality 35%; all-cause mortality: 45% | ICU LOS: 6 days (1–14); total LOS: 11 days (8–15) | NR | Extubation rate: 20%. Of note, 45% of patients were not intubated. | NA | |
| Single-group case series | |||||||
| Adeli, Iran[28] | 8 | 14-day mortality: 1/9 patients died | Range: 8–22 days | At day 14, most patients showed no clinically important C0VID-19 symptoms | At day 14, respiratory status improved dramatically in 7/8 patients | No adverse events were observed | |
| Alharthy, Saudi Arabia[29] | 3 | 0% | 100% | Hospital LOS (range): 40–48 days; ICU LOS: 27–32 days | All patients gradually recovered and neurologically improved (GCS > 10). SOFA score: from 8 to 9 to < 4. | Extubation rate: 100%. SPO2:FiO2 ratio: from 120 to 140 to > 300. Duration of MV: 18–22 days |
No adverse events were reported |
| Faqihi, Saudi Arabia[31] | 10 | 28-day mortality: 10% | 20-day discharge rate: median (IQR): 90% (17.6–22.6) | ICU LOS: median (IQR): 15 days (13.2–19.6) | SOFA score: median (IQR): from 11 (8.9–11.5) to 2 (1.4–3.6). Radiologic findings: variable degrees of improvement. | Extubation rate: 9/10 patients. Duration of MV: median (IQR): 9 (7–12) days. PaO2:FiO2 ratio: median (IQR): from 110 (95.5–135.5) to 340 (310.5–370.6) | No TPE-related adverse events were observed |
| Gluck, USA[32] | 10 | 14-day mortality: 0% | NR | NR | Clinical benefit in 6/10 patients (4/4 Penn class 3 and 2/6 Penn class 4 patients) | Non-ventilated patients: 4/4 liberated from supplemental oxygen after a mean time of 5.25 days); ventilated patients: 2/6 extubated within 14 days. Average improvement of 78% in PaO2:FiO2 ratio and average improvement of 43% in OI | No TPE-related adverse events were observed |
| Hashemian, Iran[33] | 15 | Mortality rate: 40%. TPE had a significant effect on patient survival (P = 0.002) with an odds ratio of 1.171 | NR | ICU LOS: mean ± SD: 9.6 ± 2.3 days | NR | PaO2:FiO2 ratio: from 184.3 ± 56.1–224.0 ± 57.2. Two patients on NIMV required IMV. | NR |
| Jaiswal, Dubai, United Arab Emirates[34] | 14 | Day-7 mortality: 3 (21.4%); day-28 mortality: 4 (28.6%) | NR | Hospital LOS: mean ± SD: 35.64 ± 16.98 days; median (range): 18 (12–47) days; ICU LOS: mean ± SD: 26.43 ± 17.77 days; median (range): 12 (5–42) days | An improvement in symptoms (resolution of fever) in all patients | 10 patients were liberated from IMV (median duration: 8 [6–3]) days and 5.5 [3–36] days post-sequential therapy). PaO2:FiO2: from 138.89 ± 41.90–224.78 ± 136.35 | Transient hypotension (3 patients). |
| Keith, USA[38] | 8 | 25% | 75% | ICU LOS (range): 7–18 days. Hospital LOS (range): 14–35 days | SOFA score: mean ± SD: from 9.3 ± 4.5–6.4 ± 3.5 | All 7 MV patients were initially liberated from MV, but 2 patients required reintubation; duration of MV: 2–21 days | NR |
| Morath, Germany[35] | 5 | 40% | 60% | NR | Clinical improvement observed | 3/5 patients were extubated; OI increased in 4/5 patients | NR |
| Wang, China (Wuhan)[36] | 3 children | 33% | 33% (discharged from ICU) | Hospital LOS: 17 to > 60 days | Improvements in 2/3 patients | 2/3 patients were extubated | NR |
| Zhang, China[37] | 3 | 0% | 100% | Hospital LOS: 14–22 days | Improvements in all patients | Patients all changed from high-flow oxygen to ambient air breathing. PaO2:FiO2: range: from 93 to 178–259–319); mean: from 146 to 293 | NR |
| De Prost, France[30] | 4 | 28-day mortality: 0%; 2 patients died eventually | 50% patients discharged from ICU | LOS in ICU: 50 and 66 days | Improvement in 2 patients. Worsening in 2 patients | The non-intubated patient was intubated; 3 patients needed ECMO; duration of IMV: 15–49 days; respiratory status of 2 patients improved, while it did not improve for the 2 other patients. | No adverse events attributed to TPE |
| Fernandez, Spain[39] | 4 | 0% | 100% | Hospital LOS (range): 33–51 days; ICU LOS (range): 31–43 days | Clinical improvement observed in all patients | The 3 patients on IMV were extubated; respiratory function improved in all patients. Duration of IMV: 29–40 days. |
TPE was safe |
| Truong, USA[40] | 6 | 50% | 50% | Hospital LOS: 16, 29 and 34 days | Clinical status improved post-TPE in 4/6 patients, whose SOFA scores went from 5 to 15–4–10. | Tracheostomy (1 patient); weaned from ventilator (1 patient); extubated (2 patients); NR (2 patients). Duration of IMV: 1–39 days |
None |
| Matsushita, Japan[41] | 5 | 60% | Discharge rate from ICU: 20% | 40 and 62 days | Positive evolution in 40% of patients | 1/3 intubated patient was extubated Duration of IMV: 16–52 days |
NR |
| Roshandel, Iran[42] | 5 | 20% | 80% | NR | Body temperature: range: from 37.5° to 38.6°C to 36.5–38.8 °C at 1 day post-TPE | Oxygen saturation: range: from 68% to 89% to 79–96% at 1 day post-TPE Duration of IMV: 3 days Duration of oxygen by mask: range: 8–34 days |
NR |
| Case reports | |||||||
| Akkoyunlu, Turkey[43] | 1 | 0% | 100% | Hospital LOS: 24 days | Significant improvement in general health status. | Oxygen supplementation decreased gradually and stopped | NR |
| Altmayer, France[44] | 1 | 0% | NR | NR | Our patient rapidly improved | Weaned from MV and oxygen therapy stopped at 8 and 13 days after last TPE; PaO2:FiO2 ratio improved | NR |
| Bagherzade, Iran[45] | 1 | 0% | 100% | Hospital LOS: 7 days | Good general condition at discharge | Patient was extubated after 2 days in ICU | NR |
| Faqihi, Saudi Arabia[46] | 1 | 0% | 100% | Hospital LOS: 30 days; ICU LOS: 20 days | Gradual radiological improvement | Patient was extubated after 7 days in ICU; SpO2:FiO2 ratio exceeded 350 (from 100 to 330) post-TPE | No safety issue |
| Hua, China[47] | 1 | 0% | NR | NR | Chest CT: improvement of both lungs; circulatory efficiency: significantly improved | NR | NR |
| Kamit, Turkey[48] | 1 child | 100% | 0% | Patient died on day 7 | Organ dysfunction (pulmonary, hepatic, hematologic, cardiovascular): improved post-TPE, but patient died because of severe neurological dysfunction | Spontaneous breathing was preserved | NR |
| Keith, USA[49] | 1 | 0% | 100% | Hospital LOS: 13 days | SOFA score decreased from 7 to 3; rapid improvement | Respiratory status improved; patient slowly weaned to room air | NR |
| Lin, Taiwan[50] | 1 | 0% | 100% | NR | Clinical manifestations and radiographic images improved | NR | NR |
| Ma, China[51] | 1 | 0% | NR | NR | Patient remained clinically stable | Successfully weaned from ventilator after 10 days | NR |
| Ragab, Egypt[52] | 1 | 0% | 100% | Hospital LOS post-TPE: 15 days | General clinical condition had improved dramatically after 1 day | Gradual decrease in oxygen consumption. On day 13 after TPE, patient could breathe room air | NR |
| Sadeghi, Iran[53] | 1 | 0% | 100% | Hospital LOS: 17 days | All manifestations (cutaneous lesions and intrahepatic cholestasis) disappeared; pulmonary lesions significantly recovered | NR | NR |
| Shi, China[54] | 1 | 0% | 100% | Hospital LOS: 15 days | Symptoms were almost all alleviated; blood pressure was restored; patient recovered. | OI increased (oxygen saturation of 96% and patient breathing ambient air); PaO2:FiO2 increased to 302. Duration of HFOT: 4 days |
None |
| Tian, China[55] | 1 | 0% | 100% | Hospital LOS: 15 days | Overall condition was improved | Duration of HFOT: 5 days; Oxygen supplementation stopped after 19 days in the ICU |
NR |
| Yang, China[56] | 1 | 0% | Discharged from ICU | NR | Patient’s condition improved considerably; homeostatic parameters (blood pressure, heart rate, blood gas) and chest imaging recovered | ECMO was discontinued; SPO2 improved. Duration of ECMO: 11 days |
NR |
CI, confidence interval; COVID-19, coronavirus disease 2019; CSS, cytokine storm syndrome; CT, computed tomography; DVT, deep vein thrombosis; ECMO, extracorporeal membrane oxygenation; GCS, Glasgow Coma Scale; HFOT, high-flow oxygen therapy; ICU, intensive care unit; IQR, interquartile range; IMV, invasive mechanical ventilation; LOS, length of stay; MV, mechanical ventilation; NA, not applicable; NIMV, non-invasive mechanical ventilation; NR, not reported; OI, oxygenation index; PaO2:FiO2, pressure of arterial oxygen to fractional inspired oxygen concentration; SOFA, Sequential Organ Failure Assessment; SD, standard deviation; SP02, partial arterial pressure of oxygen; TPE, therapeutic plasma exchange; UK, United Kingdom; USA, United States of America. * Statistically significant difference.
Similarly to mortality rates, a high variability was observed in terms of lengths of stay (LOS) in the ICU or hospital (Table 4). In the RCT, ICU LOS was shorter for patients in the TPE versus the control group (19 versus 26 days) [23]. Results of matched case-control series were inconsistent, with two studies showing increased LOS in hospital or ICU [25], [27], and another study showing shorter hospitalizations [26] in TPE recipients versus matched controls.
The evolution of clinical symptoms was also variable. Improvements were observed in some or all patients in most studies [23], [24], [26], [27], [28], [29], [30], [31], [32], [34], [35], [36], [37], [38], [39], [40], [41], [42], [43], [44], [45], [46], [47], [49], [50], [52], [53], [54], [55], [56], but other patients did not show clinical improvements after TPE [26], [28], [30], [31], [32], [35], [36], [38], [41], [42], [48] (Table 4). In the RCT, a post hoc analysis revealed a significant reduction in SOFA score for TPE-treated patients (P < 0.05) compared with controls [23]. SOFA scores also tended to decrease after TPE in other studies [27], [29], [31], [38], [40], [49]. Of note, the discriminant accuracy of SOFA scores for mortality predictions seems poor in patients with COVID-19 [66]. In the matched case-control series with the highest number of patients, CSS symptom resolution time, based on the Pakistani National Guidelines for COVID-19 definition [67], was significantly reduced in the TPE versus the control group (6 versus 12 days) [26].
Concerning the evolution of the ventilation status, the pressure of arterial oxygen to fractional inspired oxygen concentration (PaO2:FiO2) ratios increased in both groups in the RCT [23]. Among matched case-control series, one study showed increases in PaO2:FiO2 ratios in TPE-treated patients but not in controls [24], while another study showed no changes in either group [25] (Table 4). Increases in PaO2:FiO2 ratios were also reported in TPE-treated patients in single-group case series and case reports [27], [29], [31], [32], [33], [34], [37], [44], [46], [54]. Improvements were observed in the ventilation status of some or all patients in the studies with available results [24], [27], [28], [29], [30], [31], [32], [33], [34], [35], [36], [37], [38], [39], [40], [41], [42], [43], [44], [45], [46], [47], [49], [50], [51], [52], [54], [55], [56]. The durations of mechanical ventilation, extracorporeal membrane oxygenation, and high-flow oxygen therapy were variable [23], [25], [30], [31], [32], [34], [38], [39], [40], [41], [42], [44], [45], [46], [51], [52], [54], [55], [56].
3.6. Safety of TPE
In almost all studies reporting safety results, no TPE-related adverse events were observed [23], [24], [28], [29], [30], [31], [32], [39], [40], [46], [54] (Table 4). In one study, two patients developed TPE-related complications linked to venous access (femoral artery puncture and thrombophlebitis of femoral vein with deep vein thrombosis) [26]. Hypotension was reported in one patient in one study [27] and three patients in another study [34].
4. Discussion
The pathophysiology of sepsis involves a complex interaction of inflammation, endothelial dysfunction, and pathologic activation of coagulation [68]. These dysregulations appear common to sepsis from multiple inciting pathogens, including SARS-CoV-2, and much of the morbidity is due to the abnormal host response rather than the infection itself [69], [70]. In contrast to many therapies that are targeting different components of this pathway [68], [69], TPE offers a potential non-specific therapeutic modality. Although evidence for TPE efficacy in sepsis is not robust, available data suggested potential clinical efficacy and safety [71], [72], [73], [74]. Based on these data, the American Society for Apheresis (ASFA) issued a Category III (optimum role of TPE is not established and decision making should be individualized), Grade 2B (weak recommendation based on moderate-quality evidence) recommendation for TPE to improve organ function by removing inflammatory and antifibrinolytic mediators and replenishing anticoagulant proteins, to reverse the pathobiological derangement, and to restore hemostasis in patients with sepsis with MODS, allowing for individual consideration on a case by case basis [75].
Considering the similar pathophysiology of severe COVID-19 and sepsis, TPE may be beneficial in patients with fulminant COVID-19 infection and it was utilized in selected cases since the onset of the pandemic [20], [76], [77]. Our literature review identified 267 patients included in 34 studies with published results. In these studies, TPE was almost exclusively utilized as rescue or adjunct therapy in patients with critical or life-threatening COVID-19 disease. While limited by the largely retrospective nature of available data, TPE was shown to be feasible, safe, and often clinically efficacious for these patients.
In the identified studies, the main reasons to initiate TPE were the presence of septic shock, MODS, and/or ARDS [23], [27], [28], [29], [30], [31], [33], [34], [35], [37], [38], [41], [42], [44], [46], [48], [49], [52]. The earliest reports of successful use of TPE to treat severe COVID-19 infections included case reports or small case series in patients with MODS, in line with the ASFA indications of sepsis with multiple organ failure. Keith et al. reported an early case of COVID-19-induced pneumonia complicated by ARDS, sepsis with vasopressor-dependent hypotension, acute renal failure, and viral cardiomyopathy, who responded to one TPE session using FFP [49]. Shi et al. reported another case of a patient with severe COVID-19, respiratory failure and vasopressor-dependent hypotension who had resolution of shock and organ failures after four TPE sessions [54]. In a large case series, 11 severely ill patients with COVID-19 (ARDS, severe pneumonia, septic shock, and/or MODS) responded favorably to TPE compared with patients who received standard care [27]. Patients receiving TPE experienced higher extubation rates (73% versus 20%; P = 0.018) and lower all-cause 28-day mortality rates (0 vs. 35%; P = 0.033). A single prospective RCT in 87 patients with life-threatening COVID-19 showed that the addition of TPE to standard care was associated with a statistically insignificant decrease in 35-day mortality (20.9% versus 34.1%; P = 0.09) and statistically significant decreases in number of days on mechanical ventilation and ICU LOS [23]. This study also supported the feasibility and safety of TPE in this setting. Other studies have shown variable clinical responses, as summarized in Table 4.
While ASFA criteria must be met (sepsis with MODS) to perform TPE in the United States [75], criteria are more variable and the decision is often at the discretion of the physician in other jurisdictions. In several studies, increases in serum markers of inflammation and coagulation, which are indicators of CSS, were used as triggers to initiate TPE [23], [24], [25], [26], [29], [31], [39], [44], [48], [51], [52], [56]. In a pilot study, ten COVID-19 patients meeting criteria for Penn class 3 or 4 CSS were identified as candidates for TPE and showed rapid improvements in oxygenation and significant reductions in biomarkers of cytokine load [32]. Kamran et al. retrospectively analyzed the clinical and biochemical effects of TPE in 90 patients with COVID-19-induced CSS (defined by specific biomarker levels) using propensity score matching [26]. TPE recipients demonstrated statistically significantly improved 28-day survival (91.1% versus 61.5%), shorter hospital LOS (10 versus 15 days), and shorter time to CSS resolution (6 versus 12 days). In the single prospective RCT, CSS-associated biomarkers decreased significantly with TPE [23]. Several other studies also reported an immunomodulatory effect of TPE through decreases in IL-6, CRP, ferritin, and D-dimer levels, and elevations in lymphocyte counts, even if these findings were not observed in all reports [23], [24], [25], [27], [29], [31], [32], [33], [34], [35], [36], [37], [38], [39], [40], [41], [42], [43], [44], [46], [47], [48], [51], [52], [55], [56]. While the time to CSS resolution was evaluated in one study [26], other studies focused on patient outcomes, symptom improvements, and evolution of immune-inflammatory markers, highlighting the need for standardized definitions of CSS resolution.
The strong systemic cytokine release in severely ill patients with COVID-19 generates numerous phenotypes that look similar to other diseases, often collectively referred to as cytokine storms [78], [79], [80]. These include macrophage activation syndrome (MAS), secondary hemophagocytic lymphohistiocytosis (sHLH), and thrombocytopenia-associated multiple organ failure (TAMOF) [78], [81]. Many of these diseases that COVID-19 can mimic were shown to improve with TPE and may be considered as separate entities [80], [82], [83], [84], [85], [86]. Although Gluck et al. utilized the Penn grading scale for CSS to identify patients eligible for TPE treatment [32], scales evaluating CSS severity [5], [22] were not used in the other studies to guide the therapeutic strategy for critically ill patients with COVID-19 due to need for quick treatment decision, lack of knowledge of these scales by clinicians, and their absence in international guidelines. The Penn grading scale is based on diagnostic and clinical aspects and distinguishes among mild, moderate, severe, and life-threatening CSS [22]. When we applied this scale to the other studies, we found that almost all TPE-treated patients met criteria for Penn class 3 or 4 CSS. Because the Penn grading scale or other grading scales are not specific for COVID-19-induced CSS, they may potentially be used to identify patients with CSS (caused by any condition) who could benefit from TPE [22].
While clinical and biochemical responses to TPE were often favorable, legitimate concerns were voiced. Many clinicians are worried that the removal of anti-SARS-CoV-2 neutralizing antibodies and other host defenses may be clinically detrimental [87]. The net effect on the host immune response cannot be interpreted through available data in this analysis, but very few adverse events were attributed to TPE and none were considered life-threatening. While these data do not directly address the concerns of those skeptical of the intervention, they reaffirm the safety of TPE in the context of sepsis.
Logistics of the TPE treatment(s) were highly variable. While a single TPE session was performed in some studies, the number of TPE sessions most often ranged from three to five [23], [25], [26], [27], [28], [29], [30], [32], [33], [36], [41], [42], [44], [45], [46], [47], [48], [50], [51], [53], [54], [56]. A daily frequency seemed optimal considering the short half-lives of cytokines, and TPE sessions were mainly performed daily or every other day [23], [24], [26], [27], [28], [29], [30], [31], [32], [38], [39], [40], [41], [42], [44], [45], [46], [47], [48], [50], [51], [54], [56]. In general, the volume of exchanged plasma was based on the total plasma volume of patients, and although different methods were used to determine the volume, it generally ranged between 0.75 and 1.5 plasma volume [23], [26], [29], [31], [32], [38], [40], [42], [44], [46], [48]. These observations are consistent with ASFA guidelines for the treatment of sepsis with MODS, where daily TPE sessions for 1–14 days, or until the resolution of symptoms, are recommended with an exchanged volume of 1–1.5 plasma volume [75]. The most frequently used replacement fluid were FFP or artificial Octaplas [23], [24], [27], [29], [31], [32], [34], [35], [37], [38], [40], [41], [43], [46], [47], [48], [49], [50], [54]. These replacement fluids offer potential superiority over albumin solutions based on the pathways previously described, manifesting as endothelial injury and microthromboses in multiple organs [29], [88]. When using FFP as replacement fluid, large, prothrombotic multimers are removed along with antibodies to ADAMTS-13, ADAMTS-13 is replenished, microthrombosis risk is theoretically reduced, and tissue perfusion is improved [29], [89]. CCP has also been used as partial replacement fluid to compensate the removal of anti-SARS-Cov-2 neutralizing antibodies [33], [42], [52], [53]. While the use of albumin may result in depletion of procoagulant factors and increased bleeding risk, some providers implement 5% albumin as replacement fluid (or a mixture of albumin and FFP) to avoid the replenishment of immune response effectors, such as complement, cytokines, and chemokines, and the decreases in coagulation factors [77]. A recent publication has reported an immunomodulatory effect of albumin through interaction with endosomal Toll-like receptors in leukocytes from patients with cirrhosis [90].
While promising, results from available studies are difficult to interpret due to multiple limitations. The biggest limitation is the retrospective nature of nearly all available data even if we attempted to ensure inclusion of only higher-quality reports. Our review may also be limited by the fact that the search, screening, and article selection were performed by one author. Interpretation is further limited because in the absence of a universally established standard of care for patients with COVID-19, TPE-treated patients often received other drugs, and treatment regimens were heterogeneous. A further limitation is the fact that positive results are more frequently reported in publications, leading to a risk of underreporting of data on unsuccessful interventions. Nevertheless, it is important to note that almost all studies consistently reported feasibility and safety while observing clinical and biochemical efficacy of TPE, despite geographical variations and discrepancies in terms of treatment regimen, study endpoints, and eligibility criteria. These observations lay the foundation and confirm the need for well-designed RCTs to evaluate the utility of TPE for the treatment of COVID-19-induced CSS.
5. Conclusion
Although the evidence level was low and treatment regimens were heterogeneous in the selected studies, available data suggest that TPE alone or in combination with other drugs should be considered as a safe and valuable option for the treatment of critically ill patients with COVID-19-induced CSS. While high-quality RCTs are needed to confirm the clinical benefits of this treatment, available data suggest that CSS should be considered as a standalone pathological manifestation caused by multiple underlying diseases. Therefore, clear criteria should be defined to classify patients with CSS and to facilitate the identification of those eligible for TPE treatment.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Funding
This work was supported by Terumo Blood and Cell Technologies Europe N.V. (Belgium), which was involved in all stages of the conduct and analysis of the review and covered the costs associated with the development and publishing of the present manuscript.
Authors’ contributions
All authors were involved in the literature review interpretation, critically revised the manuscript, and approved the final version.
Declaration of Competing Interest
M. Beraud and A. Bah are employees of Terumo Blood and Cell Technologies Europe N.V. (Belgium). M. Lozano on behalf of his institution, Clinic Research Foundation, has received research support from Terumo Blood and Cell Technologies and Sanofi-Genzyme, and speaker honoraria from Grífols. S. Al Hashami and Philip Keith have nothing to declare.
Acknowledgements
The authors acknowledge Melissa Kovac (Terumo Blood and Cell Technologies) for technical support regarding the search strategy and implementation. The authors thank Modis (c/o Terumo Blood and Cell Technologies) for editorial assistance and manuscript coordination. Claire Verbelen provided writing support and Sophie Timmery coordinated the manuscript development and provided editorial support.
References
- 1.Cucinotta D., Vanelli M. WHO Declares COVID-19 a Pandemic. Acta Biomed. 2020;91(1):157–160. doi: 10.23750/abm.v91i1.9397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.World Health Organization. Weekly Epidemiological Update. Available at: 〈https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports/〉 [Accessed on March 23, 2021].
- 3.Wu Z., McGoogan J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72 314 cases from the Chinese center for disease control and prevention. JAMA. 2020;323(13):1239–1242. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 4.Song P., Li W., Xie J., Hou Y., You C. Cytokine storm induced by SARS-CoV-2. Clin Chim Acta. 2020;509:280–287. doi: 10.1016/j.cca.2020.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nagant C., Ponthieux F., Smet J., Dauby N., Doyen V., Besse-Hammer T., et al. A score combining early detection of cytokines accurately predicts COVID-19 severity and intensive care unit transfer. Int J Infect Dis. 2020;101:342–345. doi: 10.1016/j.ijid.2020.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kermali M., Khalsa R.K., Pillai K., Ismail Z., Harky A. The role of biomarkers in diagnosis of COVID-19 - A systematic review. Life Sci. 2020;254 doi: 10.1016/j.lfs.2020.117788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Xu L., Mao Y., Chen G. Risk factors for 2019 novel coronavirus disease (COVID-19) patients progressing to critical illness: a systematic review and meta-analysis. Aging (Albany N Y) 2020;12(12):12410–12421. doi: 10.18632/aging.103383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Moore J.B., June C.H. Cytokine release syndrome in severe COVID-19. Science. 2020;368(6490):473–474. doi: 10.1126/science.abb8925. [DOI] [PubMed] [Google Scholar]
- 9.Shimabukuro-Vornhagen A., Gödel P., Subklewe M., Stemmler H.J., Schlößer H.A., Schlaak M., et al. Cytokine release syndrome. J Immunother Cancer. 2018;6(1):56. doi: 10.1186/s40425-018-0343-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lippi G., Plebani M. Cytokine “storm”, cytokine “breeze”, or both in COVID-19? Clin Chem Lab Med. 2021;59(4):637–639. doi: 10.1515/cclm-2020-1761. [DOI] [PubMed] [Google Scholar]
- 11.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Liu Y., Yang Y., Zhang C., Huang F., Wang F., Yuan J., et al. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci. 2020;63(3):364–374. doi: 10.1007/s11427-020-1643-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Huang Y., Tu M., Wang S., Chen S., Zhou W., Chen D., et al. Clinical characteristics of laboratory confirmed positive cases of SARS-CoV-2 infection in Wuhan, China: A retrospective single center analysis. Travel Med Infect Dis. 2020;36 doi: 10.1016/j.tmaid.2020.101606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yuki K., Fujiogi M., Koutsogiannaki S. COVID-19 pathophysiology: A review. Clin Immunol. 2020;215 doi: 10.1016/j.clim.2020.108427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ye Q., Wang B., Mao J. The pathogenesis and treatment of the `Cytokine Storm' in COVID-19. J Infect. 2020;80(6):607–613. doi: 10.1016/j.jinf.2020.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bokharee N., Khan Y.H., Khokhar A., Mallhi T.H., Alotaibi N.H., Rasheed M. Pharmacological interventions for COVID-19: a systematic review of observational studies and clinical trials. Expert Rev Anti Infect Ther. 2021 doi: 10.1080/14787210.2021.1902805. [DOI] [PubMed] [Google Scholar]
- 17.Tharmarajah E., Buazon A., Patel V., Hannah J.R., Adas M., Allen V.B., et al. IL-6 inhibition in the treatment of COVID-19: a meta-analysis and meta-regression. J Infect. 2021;82(5):178–185. doi: 10.1016/j.jinf.2021.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cron R.Q. COVID-19 cytokine storm: targeting the appropriate cytokine. Lancet Rheumatol. 2021;3(4):e236–e237. doi: 10.1016/S2665-9913(21)00011-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Reeves H.M., Winters J.L. The mechanisms of action of plasma exchange. Br J Haematol. 2014;164(3):342–351. doi: 10.1111/bjh.12629. [DOI] [PubMed] [Google Scholar]
- 20.Tabibi S., Tabibi T., Conic R.R.Z., Banisaeed N., Streiff M.B. Therapeutic plasma exchange: A potential management strategy for critically ill COVID-19 patients. J Intensive Care Med. 2020;35(9):827–835. doi: 10.1177/0885066620940259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Howick J., Chalmers I., Glasziou P., Greenhalgh T., Heneghan C., Liberati A., et al. The Oxford Levels of Evidence 2. Oxford Centre for Evidence-Based Medicine. Available at: 〈https://www.cebm.ox.ac.uk/resources/levels-of-evidence/ocebm-levels-of-evidence〉; 2011 [Accessed on March 23, 2021].
- 22.Porter D., Frey N., Wood P.A., Weng Y., Grupp S.A. Grading of cytokine release syndrome associated with the CAR T cell therapy tisagenlecleucel. J Hematol Oncol. 2018;11(1):35. doi: 10.1186/s13045-018-0571-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Faqihi F., Alharthy A., Abdulaziz S., Balhamar A., Alomari A., AlAseri Z., et al. Therapeutic plasma exchange in patients with life-threatening COVID-19: A randomized control clinical trial. Int J Antimicrob Agents. 2021;57(5) doi: 10.1016/j.ijantimicag.2021.106334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Arulkumaran N., Thomas M., Brealey D., Alwan F., Singh D., Lunn M., et al. Plasma exchange for COVID-19 thrombo-inflammatory disease. eJHaem. 2021;2(1):26–32. doi: 10.1002/jha2.140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gucyetmez B., Atalan H.K., Sertdemir I., Cakir U., Telci L. Therapeutic plasma exchange in patients with COVID-19 pneumonia in intensive care unit: A retrospective study. Crit Care. 2020;24(1):492. doi: 10.1186/s13054-020-03215-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kamran S.M., Mirza Z.E., Naseem A., Liaqat J., Fazal I., Alamgir W., et al. Therapeutic plasma exchange for coronavirus disease-2019 triggered cytokine release syndrome; a retrospective propensity matched control study. PLoS One. 2021;16(1) doi: 10.1371/journal.pone.0244853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Khamis F., Al-Zakwani I., Al Hashmi S., Al Dowaiki S., Al Bahrani M., Pandak N., et al. Therapeutic plasma exchange in adults with severe COVID-19 infection. Int J Infect Dis. 2020;99:214–218. doi: 10.1016/j.ijid.2020.06.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Adeli S.H., Asghari A., Tabarraii R., Shajari R., Afshari S., Kalhor N., et al. Therapeutic plasma exchange as a rescue therapy in patients with coronavirus disease 2019: a case series. Pol Arch Intern Med. 2020;130(5):455–458. doi: 10.20452/pamw.15340. [DOI] [PubMed] [Google Scholar]
- 29.Alharthy A., Faqihi F., Balhamar A., Memish Z.A., Karakitsos D. Life-threatening COVID-19 presenting as stroke with antiphospholipid antibodies and low ADAMTS-13 activity, and the role of therapeutic plasma exchange: A case series. SAGE Open Med Case Rep. 2020;8:1–7. doi: 10.1177/2050313X20964089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.de Prost N., Bastard P., Arrestier R., Fourati S., Mahévas M., Burrel S., et al. Plasma exchange to rescue patients with autoantibodies against type I interferons and life-threatening COVID-19 pneumonia. J Clin Immunol. 2021;41(3):536–544. doi: 10.1007/s10875-021-00994-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Faqihi F., Alharthy A., Alodat M., Kutsogiannis D.J., Brindley P.G., Karakitsos D. Therapeutic plasma exchange in adult critically ill patients with life-threatening SARS-CoV-2 disease: A pilot study. J Crit Care. 2020;60:328–333. doi: 10.1016/j.jcrc.2020.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gluck W.L., Callahan S.P., Brevetta R.A., Stenbit A.E., Smith W.M., Martin J.C., et al. Efficacy of therapeutic plasma exchange in the treatment of penn class 3 and 4 cytokine release syndrome complicating COVID-19. Respir Med. 2020;175 doi: 10.1016/j.rmed.2020.106188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hashemian S.M., Shafigh N., Afzal G., Jamaati H., Tabarsi P., Marjani M., et al. Plasmapheresis reduces cytokine and immune cell levels in COVID-19 patients with acute respiratory distress syndrome (ARDS) Pulmonology. 2020 doi: 10.1016/j.pulmoe.2020.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jaiswal V., Nasa P., Raouf M., Gupta M., Dewedar H., Mohammad H., et al. Therapeutic plasma exchange followed by convalescent plasma transfusion in critical COVID-19-An exploratory study. Int J Infect Dis. 2021;102:332–334. doi: 10.1016/j.ijid.2020.10.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Morath C., Weigand M.A., Zeier M., Speer C., Tiwari-Heckler S., Merle U. Plasma exchange in critically ill COVID-19 patients. Crit Care. 2020;24(1):481. doi: 10.1186/s13054-020-03171-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wang X., Chen X., Tang F., Luo W., Fang J., Qi C., et al. Be aware of acute kidney injury in critically ill children with COVID-19. Pediatr Nephrol. 2021;36(1):163–169. doi: 10.1007/s00467-020-04715-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zhang L., Zhai H., Ma S., Chen J., Gao Y. Efficacy of therapeutic plasma exchange in severe COVID-19 patients. Br J Haematol. 2020;190(4):e181–e183. doi: 10.1111/bjh.16890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Keith P.D., Scott L.K., Weaver K.E., Day M., Choe C., Perkins L., et al. Treatment of critically ill coronavirus disease 2019 patients with adjunct therapeutic plasma exchange: A single-center retrospective case series. Crit Care Explor. 2021;2(9) doi: 10.1097/CCE.0000000000000223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fernandez J., Gratacos-Ginès J., Olivas P., Costa M., Nieto S., Mateo D., et al. Plasma exchange: An effective rescue therapy in critically ill patients with coronavirus disease 2019 infection. Crit Care Med. 2020;48(12):e1350–e1355. doi: 10.1097/CCM.0000000000004613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Truong A.D., Auld S.C., Barker N.A., Friend S., Wynn A.T., Cobb J., et al. Therapeutic plasma exchange for COVID-19-associated hyperviscosity. Transfusion. 2021;61(4):1029–1034. doi: 10.1111/trf.16218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Matsushita Y., Kusaoi M., Hiki M., Murayama G., Abe Y., Nozawa K., et al. Combination therapy with plasma exchange and glucocorticoid may be effective for severe COVID-19 infection: A retrospective observational study. Ther Apher Dial. 2021:PMID: 33887110. [DOI] [PMC free article] [PubMed]
- 42.Roshandel E., Sankanian G., Salimi M., Jalili A., Salari S., Sadeghi A., et al. Plasma exchange followed by convalescent plasma transfusion in COVID-19 patients. Transfus Apher Sci. 2021 doi: 10.1016/j.transci.2021.103141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Akkoyunlu Y., Cetin G., Bolukcu S., Okay G., Ogun H., Durdu B., et al. The successful management of an elderly Covid-19 infected patient by plasmapheresis. Transfus Apher Sci. 2020;59(6) doi: 10.1016/j.transci.2020.102924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Altmayer V., Saheb S., Rohaut B., Marois C., Cao A., Gallo A., et al. Therapeutic plasma exchange in a critically ill Covid-19 patient. J Clin Apher. 2021;36(1):179–182. doi: 10.1002/jca.21830. [DOI] [PubMed] [Google Scholar]
- 45.Bagherzade M., Parham M., Zohali S., Molaei S., Vafaeimanesh J. Plasmapheresis with corticosteroids and antiviral: A life-saving treatment for severe cases of Covid 19. Caspian J Intern Med. 2020;11(Suppl 1):572–576. doi: 10.22088/cjim.11.0.572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Faqihi F., Alharthy A., Memish Z.A., Kutsogiannis D.J., Brindley P.G., Karakitsos D. Peripheral neuropathy in severe COVID-19 resolved with therapeutic plasma exchange. Clin Case Rep. 2020;8(12):3234–3239. doi: 10.1002/ccr3.3397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hua T., Li M., Li X. Therapeutic plasma exchange therapy support for critical COVID-19: A case report. Ther Apher Dial. 2020: 10.1111/744-9987.13586. [DOI] [PubMed]
- 48.Kamit F., Malbora B., Atay A., Bayirli D.T., Bektas M. A fatal case of COVID-19 in a child with ALL: A cytokine storm and hyperferritinemic MODS. J Child Sci. 2020;10(01):e240–e245. [Google Scholar]
- 49.Keith P., Day M., Choe C., Perkins L., Moyer L., Hays E., et al. The successful use of therapeutic plasma exchange for severe COVID-19 acute respiratory distress syndrome with multiple organ failure. SAGE Open Med Case Rep. 2020;8:2050313×20933473. [DOI] [PMC free article] [PubMed]
- 50.Lin J.H., Chen Y.C., Lu C.L., Hsu Y.N., Wang W.J. Application of plasma exchange in association with higher dose CVVH in Cytokine storm complicating COVID-19. J Formos Med Assoc. 2020;119(6):1116–1118. doi: 10.1016/j.jfma.2020.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ma J., Xia P., Zhou Y., Liu Z., Zhou X., Wang J., et al. Potential effect of blood purification therapy in reducing cytokine storm as a late complication of critically ill COVID-19. Clin Immunol. 2020;214 doi: 10.1016/j.clim.2020.108408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ragab D., Salah-Eldin H., Afify M., Soliman W., Badr M.H. A case of COVID-19, with cytokine storm, treated by consecutive use of therapeutic plasma exchange followed by convalescent plasma transfusion: A case report. J Med Virol. 2021;93(4):1854–1856. doi: 10.1002/jmv.26630. [DOI] [PubMed] [Google Scholar]
- 53.Sadeghi A., Dooghaie Moghadam A., Eslami P., Pirsalehi A., Salari S., Roshandel E. Vasculopathy-related cutaneous lesions and intrahepatic cholestasis as synchronous manifestations in a COVID-19 patient; a case report. Gastroenterol Hepatol Bed Bench. 2020;13(4):400–404. [PMC free article] [PubMed] [Google Scholar]
- 54.Shi H., Zhou C., He P., Huang S., Duan Y., Wang X., et al. Successful treatment with plasma exchange followed by intravenous immunoglobulin in a critically ill patient with COVID-19. Int J Antimicrob Agents. 2020;56(2) doi: 10.1016/j.ijantimicag.2020.105974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Tian H., Sui Y., Tian S., Zou X., Xu Z., He H., et al. Case report: clinical treatment of the first critical patient with coronavirus disease (COVID-19) in Liaocheng, Shandong Province. Front Med ((Lausanne)) 2020;7:249. doi: 10.3389/fmed.2020.00249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yang B., Yang J., Zhou L., Xue C., Li H., Hu W., et al. Inflammatory cytokine depletion in severe coronavirus disease 2019 infectious pneumonia: A case report. Medicine ((Baltimore)) 2020;99(49) doi: 10.1097/MD.0000000000023449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Vincent J.L., Moreno R., Takala J., Willatts S., De Mendonça A., Bruining H., et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the working group on sepsis-related problems of the European society of intensive care medicine. Intensive Care Med. 1996;22(7):707–710. doi: 10.1007/BF01709751. [DOI] [PubMed] [Google Scholar]
- 58.Lee G., Arepally G.M. Anticoagulation techniques in apheresis: From heparin to citrate and beyond. J Clin Apher. 2012;27(3):117–125. doi: 10.1002/jca.21222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhang C., Wu Z., Li J.-W., Zhao H., Wang G.-Q. Cytokine release syndrome in severe COVID-19: Interleukin-6 receptor antagonist tocilizumab may be the key to reduce mortality. Int J Antimicrob Agents. 2020;55(5) doi: 10.1016/j.ijantimicag.2020.105954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Webb B.J., Peltan I.D., Jensen P., Hoda D., Hunter B., Silver A., et al. Clinical criteria for COVID-19-associated hyperinflammatory syndrome: A cohort study. Lancet Rheumatol. 2020;2(12):e754–e763. doi: 10.1016/S2665-9913(20)30343-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cavezzi A., Troiani E., Corrao S. COVID-19: Hemoglobin, iron, and hypoxia beyond inflammation. A narrative review. Clin Pract. 2020;10(2):1271. doi: 10.4081/cp.2020.1271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ruscitti P., Giacomelli R. Ferritin and severe COVID-19, from clinical observations to pathogenic implications and therapeutic perspectives. Isr Med Assoc J. 2020;22(8):516–518. [PubMed] [Google Scholar]
- 63.Paliogiannis P., Mangoni A.A., Dettori P., Nasrallah G.K., Pintus G., Zinellu A. D-Dimer concentrations and COVID-19 severity: A systematic review and meta-analysis. Front Public Health. 2020;8:432. doi: 10.3389/fpubh.2020.00432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Doevelaar A.A.N., Bachmann M., Hölzer B., Seibert F.S., Rohn B.J., Bauer F., et al. von Willebrand factor multimer formation contributes to immunothrombosis in coronavirus disease 2019. Crit Care Med. 2021;49(5):e512–e520. doi: 10.1097/CCM.0000000000004918. [DOI] [PubMed] [Google Scholar]
- 65.Illg Z., Muller G., Mueller M., Nippert J., Allen B. Analysis of absolute lymphocyte count in patients with COVID-19. Am J Emerg Med. 2021;46:16–19. doi: 10.1016/j.ajem.2021.02.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Raschke R.A., Agarwal S., Rangan P., Heise C.W., Curry S.C. Discriminant accuracy of the SOFA score for determining the probable mortality of patients with COVID-19 pneumonia requiring mechanical ventilation. JAMA. 2021;325(14):1469–1470. doi: 10.1001/jama.2021.1545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Government of Pakistan. Ministry of National Health Services, Regulation & Coordination. Clinical management guidelines for COVID-19 infections. Available at: 〈http://www.nhsrc.gov.pk/SiteImage/Misc/files/Clinical-Management-nfection%20v2.pdf〉 [Accessed on June 15, 2021].
- 68.Remick D.G. Pathophysiology of sepsis. Am J Patho. 2007;170(5):1435–1444. doi: 10.2353/ajpath.2007.060872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Beltrán-García J., Osca-Verdegal R., Pallardó F.V., Ferreres J., Rodríguez M., Mulet S., et al. Sepsis and coronavirus disease 2019: common features and anti-inflammatory therapeutic approaches. Crit Care Med. 2020;48(12):1841–1844. doi: 10.1097/CCM.0000000000004625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Olwal C.O., Nganyewo N.N., Tapela K., Djomkam Zune A.L., Owoicho O., Bediako Y., et al. Parallels in sepsis and COVID-19 conditions: implications for managing severe COVID-19. Front Immunol. 2021;12 doi: 10.3389/fimmu.2021.602848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Busund R., Koukline V., Utrobin U., Nedashkovsky E. Plasmapheresis in severe sepsis and septic shock: A prospective, randomised, controlled trial. Intensive Care Med. 2002;28(10):1434–1439. doi: 10.1007/s00134-002-1410-7. [DOI] [PubMed] [Google Scholar]
- 72.Chang T., Tu Y.K., Lee C.T., Chao A., Huang C.H., Wang M.J., et al. Effects of polymyxin B hemoperfusion on mortality in patients with severe sepsis and septic shock: a systemic review, meta-analysis update, and disease severity subgroup meta-analysis. Crit Care Med. 2017;45(8):e858–e864. doi: 10.1097/CCM.0000000000002362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Long E.J., Taylor A., Delzoppo C., Shann F., Pearson G., Buckley D., et al. A randomised controlled trial of plasma filtration in severe paediatric sepsis. Crit Care Resusc. 2013;15(3):198–204. [PubMed] [Google Scholar]
- 74.Rimmer E., Houston B.L., Kumar A., Abou-Setta A.M., Friesen C., Marshall J.C., et al. The efficacy and safety of plasma exchange in patients with sepsis and septic shock: A systematic review and meta-analysis. Crit Care. 2014;18(6):699. doi: 10.1186/s13054-014-0699-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Padmanabhan A., Connelly-Smith L., Aqui N., Balogun R.A., Klingel R., Meyer E., et al. Guidelines on the use of therapeutic apheresis in clinical practice – Evidence-based approach from the writing committee of the American Society for Apheresis: The eighth special issue. J Clin Apher. 2019;34(3):171–354. doi: 10.1002/jca.21705. [DOI] [PubMed] [Google Scholar]
- 76.Keith P., Day M., Perkins L., Moyer L., Hewitt K., Wells A. A novel treatment approach to the novel coronavirus: an argument for the use of therapeutic plasma exchange for fulminant COVID-19. Critical Care. 2020;24(1):128. doi: 10.1186/s13054-020-2836-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Balagholi S., Dabbaghi R., Eshghi P., Mousavi S.A., Heshmati F., Mohammadi S. Potential of therapeutic plasmapheresis in treatment of COVID-19 patients: Immunopathogenesis and coagulopathy. Transfus Apher Sci. 2020;59(6) doi: 10.1016/j.transci.2020.102993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Osuchowski M.F., Winkler M.S., Skirecki T., Cajander S., Shankar-Hari M., Lachmann G., et al. The COVID-19 puzzle: Deciphering pathophysiology and phenotypes of a new disease entity. Lancet Respir Med. 2021;9(6):622–642. doi: 10.1016/S2213-2600(21)00218-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Hu B., Huang S., Yin L. The cytokine storm and COVID‐19. J Med Virol. 2021;93(1):250–256. doi: 10.1002/jmv.26232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Gao Y.M., Xu G., Wang B., Liu B.C. Cytokine storm syndrome in coronavirus disease 2019: A narrative review. J Intern Med. 2021;289(2):147–161. doi: 10.1111/joim.13144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Latimer G., Corriveau C., DeBiasi R.L., Jantausch B., Delaney M., Jacquot C., et al. Cardiac dysfunction and thrombocytopenia-associated multiple organ failure inflammation phenotype in a severe paediatric case of COVID-19. Lancet Child Adolesc Health. 2020;4(7):552–554. doi: 10.1016/S2352-4642(20)30163-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Bosnak M., Erdogan S., Aktekin E.H., Bay A. Therapeutic plasma exchange in primary hemophagocytic lymphohistiocytosis: Reports of two cases and a review of the literature. Transfus Apher Sci. 2016;55(3):353–356. doi: 10.1016/j.transci.2016.09.015. [DOI] [PubMed] [Google Scholar]
- 83.Pandey P.K., Kaul E., Agarwal N., Goel S. Effectiveness of therapeutic plasma exchange in a critically ill child with secondary hemophagocytic lymphohistiocytosis. Asian J Transfus Sci. 2019;13(2):145–147. doi: 10.4103/ajts.AJTS_45_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Nusshag C., Morath C., Zeier M., Weigand M.A., Merle U., Brenner T. Hemophagocytic lymphohistiocytosis in an adult kidney transplant recipient successfully treated by plasmapheresis: A case report and review of the literature. Med. ((Baltimore)) 2017;96(50) doi: 10.1097/MD.0000000000009283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Kinjo N., Hamada K., Hirayama C., Shimizu M. Role of plasma exchange, leukocytapheresis, and plasma diafiltration in management of refractory macrophage activation syndrome. J Clin Apher. 2018;33(1):117–120. doi: 10.1002/jca.21570. [DOI] [PubMed] [Google Scholar]
- 86.Fortenberry J.D., Nguyen T., Grunwell J.R., Aneja R.K., Wheeler D., Hall M., et al. Therapeutic plasma exchange in children with thrombocytopenia-associated multiple organ failure: the thrombocytopenia-associated multiple organ failure network prospective experience. Crit Care Med. 2019;47(3):e173–e181. doi: 10.1097/CCM.0000000000003559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Honore P.M., Barreto Gutierrez L., Kugener L., Redant S., Attou R., Gallerani A., et al. TPE seems to be a treatment that may improve outcomes by effectively removing fibrin degradation products and restoring coagulation status: fact or fiction? Crit Care. 2020;24(1):599. doi: 10.1186/s13054-020-03309-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Chen W., Pan J.Y. Anatomical and pathological observation and analysis of SARS and COVID-19: microthrombosis is the main cause of death. Biol Proced Online. 2021;23(1):4. doi: 10.1186/s12575-021-00142-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Favaloro E.J., Henry B.M., Lippi G. Increased VWF and decreased ADAMTS-13 in COVID-19: creating a milieu for (micro)thrombosis. Semin Thromb Hemost. 2021;47(4):400–418. doi: 10.1055/s-0041-1727282. [DOI] [PubMed] [Google Scholar]
- 90.Casulleras M., Flores-Costa R., Duran-Güell M., Alcaraz-Quiles J., Sanz S., Titos E., et al. Albumin internalizes and inhibits endosomal TLR signaling in leukocytes from patients with decompensated cirrhosis. Sci Transl Med. 2020;12(566):eaax5135. doi: 10.1126/scitranslmed.aax5135. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.