Abstract
The safety and antifungal activity of LY303366 (LY), a new broad-spectrum semisynthetic echinocandin, were studied against disseminated candidiasis in persistently neutropenic rabbits. In vitro time-kill assays demonstrated that LY has concentration-dependent fungicidal activity. The pharmacokinetics of LY in the plasma of nonneutropenic rabbits suggested a linear relationship between dose and area under the curve (AUC). The times spent above the MIC during the experimental dosing interval of 24 h were 4 h for LY at 0.1 mg/kg of body weight/day (LY0.1), 8 h for LY at 0.25 mg/kg/day (LY0.25), 12 h for LY at 0.5 mg/kg/day (LY0.5), and 20 h for LY at 1 mg/kg/day (LY1). Antifungal therapy was administered to infected rabbits for 10 days starting 24 h after the intravenous (i.v.) inoculation of 103 Candida albicans blastoconidia. Study groups consisted of untreated controls (UCs) and animals treated with amphotericin B (AmB; 1 mg/kg/day i.v.), fluconazole (FLU; 10 mg/kg/day i.v.), and LY0.1, LY0.25, LY0.5, or LY1 i.v. Rabbits treated with LY0.5, LY1, AmB, and FLU had similarly significant clearance of C. albicans from the liver, spleen, kidney, lung, vena cava, and brain in comparison to that for UCs. There was a dose-dependent clearance of C. albicans from tissues in response to LY. Among rabbits treated with LY0.1 there was a significant reduction of C. albicans only in the spleen. In animals treated with LY0.25 there was a significant reduction in all tissues but the brain. By comparison, LY0.5 and LY1 cleared all tissues, including the brain, of C. albicans. These in vivo findings were consistent with the results of in vitro time-kill assays. A dose-dependent effect of altered cell wall morphology was observed among UCs and animals treated with LY0.1, and LY0.25, with a progressive transition from hyphal structure to disrupted yeast forms. Serum creatinine levels were higher and serum potassium levels were lower in AmB-treated rabbits than in UCs and LY- and FLU-treated rabbits. LY0.5 and LY1 were well tolerated, displayed predictable pharmacokinetics in plasma, and had activities comparable to those of AmB and FLU in the treatment of disseminated candidiasis in persistently neutropenic rabbits.
Invasive candidiasis is an important cause of nosocomial infection in immunocompromised patients (2, 6, 23, 42). Despite recent advances in antifungal therapy, candidiasis is the most common hospital-acquired mycosis. Conventional amphotericin B (AmB), which binds to ergosterol and disrupts membrane integrity, remains the mainstay of therapy for serious Candida infections; however, its clinical utility may be thwarted by dose-limiting nephrotoxicity (12, 35). The advent of fluconazole provides new options for the treatment and prevention of invasive candidiasis (1, 15, 19, 32, 36, 41). However, the emergence of resistance to antifungal azoles poses a new challenge to our limited therapeutic armamentarium (11, 27, 33). New agents with potent antifungal efficacy and improved safety are clearly needed.
The echinocandins are a novel class of semisynthetic lipopeptide antifungal compounds which inhibit 1,3-β-d-glucan synthase. 1,3-β-d-Glucan is a major structural polymer of fungal cell walls. Inhibition of 1,3-β-d-glucan synthesis results in disruption of fungal cell wall biosynthesis, cell wall damage, and ultimately, cell death (7, 13, 22). Cilofungin or LY121019 (N-p-octyloxybenzoylechinocandin B nucleus) was the first echinocandin B developed for clinical trials. This early echinocandin had excellent in vitro activity against Candida spp. and was highly effective in animal models of disseminated candidiasis (16, 25, 28, 40); however, clinical development of cilofungin was discontinued because of toxicity due to the carrier vehicle (39).
During the past several years, a new generation of echinocandins has emerged. LY303366 (LY), a terphenyl-substituted echinocandin B, demonstrates potent and non-cross-resistant in vitro antifungal activity against Candida species and Pneumocystis carinii (3, 9, 10, 20, 21, 30). The antifungal activity of LY against other fungi such as Aspergillus fumigatus, Blastomyces dermatitidis, and Histoplasma capsulatum has also been observed (29, 43, 44). LY was well tolerated by healthy human volunteers (31).
Little is known, however, about the in vivo activity of LY in the treatment of disseminated candidiasis in immunocompromised hosts. We therefore investigated the efficacy and safety of LY in a persistently neutropenic rabbit model of disseminated candidiasis.
MATERIALS AND METHODS
MICs.
The MIC of LY for C. albicans NIH-8621 used in these experiments was determined by the broth microdilution method described in document M27-A of the National Committee for Clinical Laboratory Standards (26). A stock solution of LY (Eli Lilly & Co., Indianapolis, Ind.) was made by using 100% polyethylene glycol 400 (Sigma Chemical Co., St. Louis, Mo.). Serial twofold dilutions were further made with antibiotic medium 3 (AM3; National Institutes of Health Media, Bethesda, Md.). The final concentrations ranged from 1.0 to 0.001 μg/ml. The yeast inoculum size ranged from 0.5 × 103 to 2.5 × 103 CFU/ml. Dilutions of the inoculum were made with AM3. The 96-well plates were incubated at 37°C in air, and MICs were recorded at 24 and 48 h. Inoculum controls were also included, and growth in the presence of the drug was compared to the growth in those wells. The MIC was defined as the concentration that resulted in complete visual inhibition of fungal growth. The minimum lethal concentration (MLC) was determined by subculturing 100 μl from wells with concentrations of drug at and above the MIC. The lowest concentration at which no growth of C. albicans occurred was defined as the MLC. Candida krusei ATCC 6258 and Candida parapsilosis ATCC 22019 were included as quality control isolates. The LY MIC at 48 h for C. albicans was 0.015 μg/ml, and the MIC of AmB desoxycholate at 48 h was 0.125 μg/ml. The MLC of LY was 0.25 μg/ml, and that of AmB was 0.125 μg/ml.
Time-kill assay.
To characterize the fungicidal activities of LY and AmB, time-kill assays were performed with C. albicans NIH-8621. Three concentrations of LY and AmB (0.01, 0.1, and 0.25 μg/ml) were studied. These concentrations span the range of MICs and MLCs of the two compounds. The inoculum for the time-kill assay was prepared by growing the isolate for 48 h at 37°C on Sabouraud glucose agar (SGA), inoculating the colonies into a starter broth of 50 ml of Sabouraud glucose broth (SGB), and incubating the broth for 2 h in a gyratory water bath at 37°C. One milliliter of this suspension was transferred into 50 ml of fresh AM3 broth in each of four 250-ml Erlenmeyer flasks, and the flasks were incubated at 37°C for 16 h in gyratory water bath in order to generate logarithmic-phase growth. The suspension was centrifuged, the pellet was washed three times with normal saline, the concentration was adjusted with a hemacytometer, and the solution was inoculated into 250-ml Erlenmeyer flasks containing 50 ml of AM3 broth alone (untreated growth control) or AM3 plus antifungal compound. The final concentration of approximately 3.0 × 105 CFU/ml was confirmed by quantitative culture. The flasks were incubated simultaneously in a gyratory water bath at 37°C. Growth suspensions were sampled at times of 0, 2, 4, 6, and 24 h; and 100-μl aliquots were plated in dilutions of 10−2, 10−3, and 10−4 on one SGA plate/per aliquot. Colonies were counted after 48 h of incubation at 37°C, and the calculated number of CFU per milliliter was plotted for each time point. The lower limit of quantitation for the time-kill assay was 10 CFU/ml.
Animals.
Female New Zealand White rabbits (weight, 2.5 to 3.5 kg; Hazleton, Deutschland, Pa.) at the time of inoculation were used in all experiments. Rabbits were individually housed and were maintained according to National Institutes of Health guidelines for animal care and in fulfillment of the criteria of the American Association for Accreditation of Laboratory Animal Care (5). A total of 61 rabbits were used for all experiments. Vascular access was established in each rabbit by the surgical placement of a silastic tunneled central venous catheter (38). The silastic catheter permitted nontraumatic venous access for repeated blood sampling for study of biochemical and hematological parameters, study of pharmacokinetics in plasma, and administration of parenteral agents. Serum samples were drawn, when possible, from all rabbits at the initiation of immunosuppression, during the course of disseminated candidiasis, and before death. Rabbits were killed by intravenous (i.v.) administration of pentobarbital (500 mg/kg of body weight) at the end of each experiment, 24 h after administration of the last dose of the drug.
Antifungal compounds.
LY was provided by Eli Lilly & Company as a 10-mg/ml proprietary solution for parenteral administration. LY was administered by slow i.v. infusion at dosages of 0.1 mg/kg/day (LY0.1), 0.25 mg/kg/day (LY0.25), 0.5 mg/kg/day (LY0.5), and 1 mg/kg/day (LY1). Preliminary experiments also were performed with 5 mg/kg/day (LY5) and 10 mg/kg/day (LY10). The doses of 0.1, 0.25, 0.5, and 1 mg/kg were prepared by diluting the initial solution of 10 mg/ml with sterile 0.9% NaCl (Quality Biological, Inc., Gaithersburg, Md.) to a concentration of 1 mg/ml. LY5 and LY10 were administered directly in a concentration of 10 mg/ml. AmB (1 mg/kg/day; Squibb, Princeton, N.J.) was slowly administered i.v. (0.1 ml every 10 s). Fluconazole (FLU; Roerig-Pfizer, New York, N.Y.) was administered at 10 mg/kg/day i.v. Administration of all compounds was initiated 24 h after inoculation.
Single-dose pharmacokinetic studies.
Four groups of three immunocompetent New Zealand White rabbits each were studied. Animals received LY at 0.1, 0.25, 0.5, and 1.0 mg/kg as a single steady i.v. bolus. Serial plasma samples were drawn immediately before and at 0.16, 0.5, 1, 2, 4, 8, 12, 18, 24, 48, and 72 h after administration of the compound. Extensive sampling of neutropenic rabbits was not performed in order to minimize blood loss and anemia. Samples were stored at −80°C until assay. Levels of LY in plasma were determined by a sensitive, reproducible, and specific high-performance liquid chromatographic method developed and fully validated at the Drug Disposition Eli Lilly Research Laboratory (Indianapolis, Ind.).
In brief, LY and LY306168, the internal standard, were separated from plasma by using acetonitrile-ammonium acetate (50 mM) (pH 4.0)-based solvents, C8-bonded phase extraction cartridges (Varian Inc., Harbor City, Calif.), and a vacuum manifold (Supelco Inc., Bellefonte, Pa.). The eluant was dried in an evaporator (Zymark Corp., Hopkinston, Mass.) under a steady stream of nitrogen at 40°C and was reconstituted in 50:50 (vol/vol) methanol-ammonium acetate (50 mM) (pH 4.0) for injection. The average recovery of LY from rabbit plasma by the extraction procedure was >90% compared with the amount in the unextracted reagent standard. The mobile phase consisted of 50:50 (vol/vol) acetonitrile:ammonium acetate (50 mM) (pH 4.0) delivered at 0.5 ml/min. The injection volume was 75 μl. LY and LY306168 eluted at 6.3 and 4.1 min, respectively, from a 5-μm C8 analytical column (Zorbaz RX-C8; Rockland Technologics, Chadds Ford, Pa.), maintained at 50°C in conjunction with a precolumn filter containing a 2-μm insert. UV detection at 300 nm was used. Quantitation was performed with the peak height ratios of LY/LY306168 versus the LY concentrations of the external standard. Standard curves (20 to 5,120 ng/ml) were linear, with R2 values of ≥0.999. The lower limit of quantitation was 20 ng/ml. Accuracy was within ±0.4 to 3.2%, and intra- and interday variabilities (precisions) ranged from 1.2 to 4.7%.
Standard model-independent techniques were used to calculate the area under the plasma concentration-time curve (AUC) from 0 to 72 h (AUC0–72), apparent volume of distribution (V), total clearance (CL), elimination half-life (t1/2), and peak concentrations in plasma at 0 min (Cmax) (14). Trough levels at 24 h postdosing (Cmin) were obtained directly from the concentration-versus-time profiles.
Organism and inoculation.
C. albicans NIH-8621 from a granulocytopenic patient with autopsy-proven disseminated candidiasis was used for all experiments. Cultures of the isolate were stored at −40°C in skim milk suspension and at −70°C on potato dextrose agar slants. Cells from this suspension were streaked onto SGA plates. The plates were incubated at 37°C for 24 h. Several well-isolated colonies were sampled from a freshly grown culture and were suspended into 50 ml of Emmon’s modified of SGB (pH 7.0) in a 250-ml Erlenmeyer flask. This flask was incubated in a gyratory incubator for 2 h. The 2-h suspension was transferred to four Erlenmeyer flasks with fresh SGB, and the flasks were incubated in a gyratory incubator at 80 oscillations per min at 37°C for 18 h. The Candida suspension was centrifuged at 3,000 × g for 10 min and was washed three times with sterile normal saline, and the concentration was adjusted by use of hemacytometer counts and was confirmed by quantitative cultures of 10-fold serial dilutions. An inoculum of 103 blastoconidia suspended in a 5-ml volume of 0.9% NaCl was administered slowly to each rabbit via the indwelling silastic intravenous catheter. The inoculum size was confirmed by plating serial dilutions onto SGA plates. The pattern of infection of disseminated candidiasis permitted survival of nearly all rabbits through the full course of neutropenia.
Immunosuppression and maintenance of neutropenia.
Intravenous cytarabine (araC; Cytosar-U; The Upjohn Company, Kalamazoo, Mich.) was administered for induction and maintenance of granulocytopenia (<500 granulocytes/μl). Profound granulocytopenia (<100 granulocytes/μl) was achieved by an initial course of treatment with 440 mg of araC per m2 on days 1 to 5. A maintenance dose of 440 mg/m2 of araC per m2 was administered at intervals of 2 days during the experiment.
Ceftazidime (75 mg/kg given i.v. twice daily; Glaxo Pharmaceuticals, Division of Glaxo Inc., Research Triangle Park, N.C.) gentamicin (5 mg/kg given i.v. every other day; Elkins-Sinn, Inc., Cherry Hill, N.J.), and vancomycin (15 mg/kg i.v. given daily; Abbott Laboratories, North Chicago, Ill.) were administered from day 4 of chemotherapy for prevention of opportunistic bacterial infections during neutropenia. In order to prevent antibiotic-associated diarrhea due to Clostridium spiriforme, all rabbits received 50 mg of vancomycin per liter of drinking water. Total leukocyte counts and the percentages of granulocytes were monitored twice weekly with a Coulter Counter (Coulter Corporation, Miami, Fla.) and by peripheral blood smears and differential counts, respectively.
Treatment groups.
In a pilot study of LY1, LY5, and LY10 versus untreated control (UC) animals, the clearance of C. albicans was achieved in all tissues studied in all treatment groups. On the basis of these preliminary findings, subsequent experiments were conducted with the following experimental groups: animals treated with LY0.1, LY0.25, LY0.5, LY1, AmB, and FLU and UC animals. The distribution of weight was the same in all treatment groups. LY, AmB, and FLU therapies were continued throughout the course of the experiment for up to 10 days for surviving rabbits.
Assessment of in vivo antifungal efficacy.
Antifungal activity was determined by quantitative clearance of C. albicans from tissue. Representative sections of liver, spleen, kidney, lung, anterior vena cava, and brain were weighed and were then homogenized in sterile reinforced polyethylene bags (Tecmar Corp., Cincinnati, Ohio) (37). Each tissue homogenate was serially diluted 100-fold from 10 to 10−4 in sterile 0.9% saline. A 0.1-ml quantity of undiluted homogenate and of each dilution was separately plated onto Emmon’s modified SGA containing chloramphenicol and gentamicin. Culture plates were incubated at 37°C for 24 h, after which the numbers of CFU were counted and the number of CFU per gram of tissue was calculated for each organ. The microbiological response to antifungal treatment was evaluated by determination of the concentration (in CFU per gram) of C. albicans in tissue. The method was sensitive for detection of ≥10 CFU/g. The culture-negative plates were counted as having 0 CFU/g. Data were graphed as the mean ± standard error of the mean (SEM) of log10 CFU per gram.
Histopathology.
Representative sections of the liver, spleen, and kidney were prepared for histological studies. Tissue specimens were excised and fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned, and then stained with periodic acid-Schiff and Gomori methenamine silver stains.
Toxicity study.
The same infected neutropenic rabbits were used throughout all experiments, including the toxicity study. Chemical analyses that included determination of potassium, aspartyl aminotransaminase, alanine aminotransaminase, serum creatinine, alkaline phosphatase, and total bilirubin (Ani Lytics, Inc., Gaithersburg, Md.) levels were performed with the penultimate sample drawn from each rabbit.
Statistical analysis.
Values are expressed as means ± SEMs. All treatment groups were compared against the UC group by analysis of variance (ANOVA) with Bonferroni’s correction for multiple comparisons (six comparisons). The central hypothesis of this analysis is based upon the response of treated animals in comparison to that of UC animals. A two-tailed P value of <0.05, which has already been adjusted for multiple comparisons by Bonferroni’s method, was considered to be statistically significant. In order to investigate the antifungal efficacies of the different LY dosages versus that of AmB desoxycholate or FLU, we performed ANOVA with Bonferroni’s correction for multiple comparisons (four comparisons).
Differences between the means of pharmacokinetic parameters across the dosages were evaluated by the Kruskal-Wallis nonparametric ANOVA test. Simple linear regression and one-way analysis of variance were used to assess dose linearity. A two-tailed P value of <0.05 was considered to be statistically significant.
RESULTS
Time-kill assay.
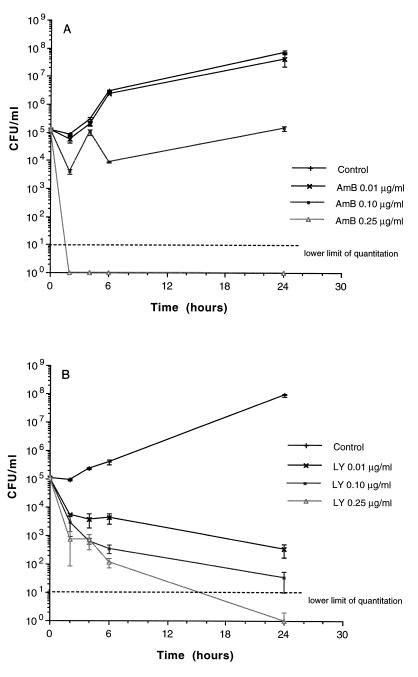
The fungicidal activity and rate of killing by LY and AmB were concentration dependent (Fig. 1). After 2 h, 90 to 99% killing was achieved with 0.25 μg of LY per ml and >99.9% killing was achieved with 0.25 μg of AmB per ml. At 4 to 6 h ≥99.9% killing was achieved, and sustained killing was achieved at 24 h with 0.25 μg of LY per ml. Killing of C. albicans at 24 h by LY was superior to that by AmB at comparable concentrations of 0.01 and 0.1 μg/ml.
FIG. 1.
Time-kill assay of AmB (A) and LY (B) against C. albicans in AM3. Growth in the presence of LY and AmB concentrations of 0.01, 0.1, and 0.25 μg/ml was studied in relation to the growth control. Data are plotted as the mean ± SEM for three separate experiments for each growth curve, including the control and AmB at 0.25 μg/ml. As the SEM was small for several time points, the error bars may not always be apparent in the time-kill curves.
Pharmacokinetics in plasma after administration of a single dose.
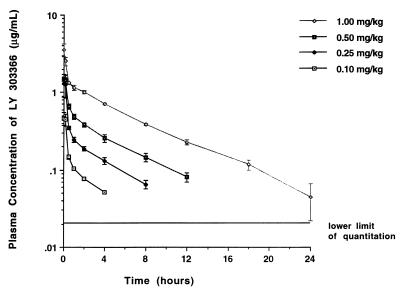
Plasma concentration-versus-time profiles for LY after administration of single doses of 0.1, 0.25, 0.5, and 1.0 mg/kg to immunocompetent rabbits are depicted in Fig. 2, and calculated pharmacokinetic parameters are listed in Table 1. The plasma of all animals was sampled for 72 h. The disposition of the drug appeared to be linear at the investigated dose range of 0.1 to 1.0 mg/kg, when the AUC0–4 was used for statistical analysis of departure from linearity. However, levels in plasma fell below assay limits between 8 and 24 h, which did not allow for direct comparison of AUC0–24, CL, or t1/2 but did permit comparison of Cmax and Cmin. Peak levels in plasma for all dosage groups were in excess of the MIC for the test strain used in the infection model. By extrapolating the concentration-versus-time profile for immunocompetent rabbits to those used for the infection model, the times spent above the MIC during the experimental dosing interval of 24 h were 4 h for the 0.1-mg/kg dose, 8 h for the 0.25-mg/kg dose, 12 h for the 0.5-mg/kg dose, and 20 h for the 1.0-mg/kg dose of LY.
FIG. 2.
Concentration-versus-time profiles in plasma after administration of LY to immunocompetent rabbits. Each point represents the mean ± SEM obtained for three rabbits assigned to each dosage group.
TABLE 1.
Noncompartmental pharmacokinetics in plasma after administration of a single dose of LY to healthy rabbitsa
| Drug dose (mg/kg) | Cmax (μg/ml)b | Cmin (at 24 h) (μg/ml) | AUC0–4 (μg · h/ml)c | AUC0–24 (μg · h/ml) | V (liters) | CL (liter/h) | t1/2 (h) |
|---|---|---|---|---|---|---|---|
| 0.10 | 0.464 ± 0.082 | <LOQd | 0.469 ± 0.030 | 0.565 ± 0.037 | 1.9 ± 0.13 | 0.432 ± 0.03 | 3.0 ± 0.11 |
| 0.25 | 1.298 ± 0.425 | <LOQ | 1.168 ± 0.109 | 1.559 ± 0.075 | 2.1 ± 0.15 | 0.375 ± 0.02 | 3.9 ± 0.23 |
| 0.50 | 1.488 ± 0.083 | <LOQ | 1.961 ± 0.153 | 3.474 ± 0.338 | 2.7 ± 0.16 | 0.387 ± 0.03 | 4.8 ± 0.33 |
| 1.00 | 3.563 ± 0.644 | 0.044 ± 0.022 | 4.577 ± 0.217 | 10.064 ± 0.786 | 2.3 ± 0.18 | 0.281 ± 0.02 | 5.8 ± 0.65 |
All values are given as means ± SEM for three rabbits at each dose.
P < 0.05 by Kruskal-Wallis nonparametric ANOVA.
P = 0.917 by ANOVA for departure from linearity after dose normalization.
<LOQ, less than the lower limit of quantitation.
Antifungal therapy.
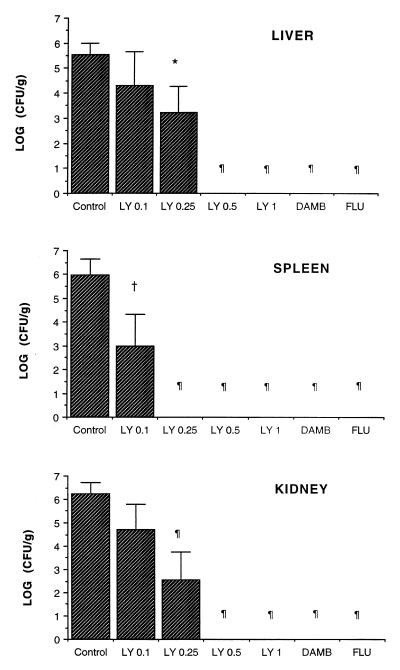
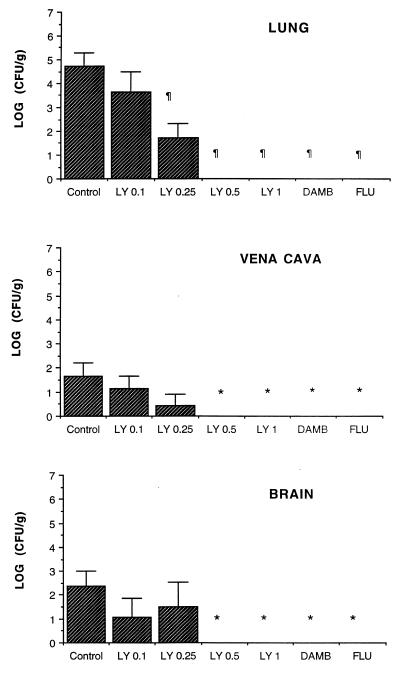
LY demonstrated a significant dose-dependent antifungal effect in treatment of disseminated candidiasis (Fig. 3 and 4). Rabbits treated with LY0.5, LY1, AmB, and FLU had similarly significant clearances of C. albicans from the liver (P ≤ 0.001), spleen (P ≤ 0.001), kidney (P ≤ 0.001), lung (P ≤ 0.001), vena cava (P < 0.05), and brain (P < 0.05) in comparison to those for UCs. In comparison with UC rabbits, LY0.25 demonstrated a significant (>10-fold to 106-fold) reduction of C. albicans (in CFU per gram) in all tissues but the brain, while in animals treated with LY0.1 there was a significant reduction of C. albicans (in CFU per gram) only in the spleen.
FIG. 3.
Response of disseminated candidiasis in persistently neutropenic rabbits to antifungal therapy measured as the mean log CFU per gram concentration of organism in the liver, spleen, and kidney for UCs (n = 13) and rabbits treated with LY0.1 (n = 6), LY0.25 (n = 6), LY0.5 (n = 6), LY1 (n = 6), AmB (DAMB, AmB desoxycholate) at 1 mg/kg/day (n = 6), and FLU at 10 mg/kg/day (n = 6). Values are given as means ± SEMs. ∗, P < 0.05; †, P < 0.01; ¶, P < 0.001 (P values were obtained by comparison to UCs by ANOVA with Bonferroni’s correction for multiple comparisons).
FIG. 4.
Response of disseminated candidiasis in persistently neutropenic rabbits to antifungal therapy measured as the mean log CFU per gram concentration of organism in the lung, vena cava, and brain for UCs (n = 13) and rabbits treated with LY0.1 (n = 6), LY0.25 (n = 6), LY0.5 (n = 6), LY1 (n = 6), AmB (DAMB, AmB desoxycholate) at 1 mg/kg/day (n = 6), and FLU at 10 mg/kg/day (n = 6). Values are given as means ± SEMs. ∗, P < 0.05; †, P < 0.01; ¶, P < 0.001 (P values were obtained by comparison to UCs by ANOVA with Bonferroni’s correction for multiple comparisons).
In comparison to AmB or FLU, LY was significantly less active at doses of 0.1 and 0.25 mg/kg in clearing C. albicans from the kidney and liver and at a dose of 0.1 mg/kg in clearing the organism from the lung and spleen (P < 0.05). On the other hand, there were no significant differences between the other doses of LY and AmB or FLU in clearing the organisms from the vena cava and brain.
Histopathology.
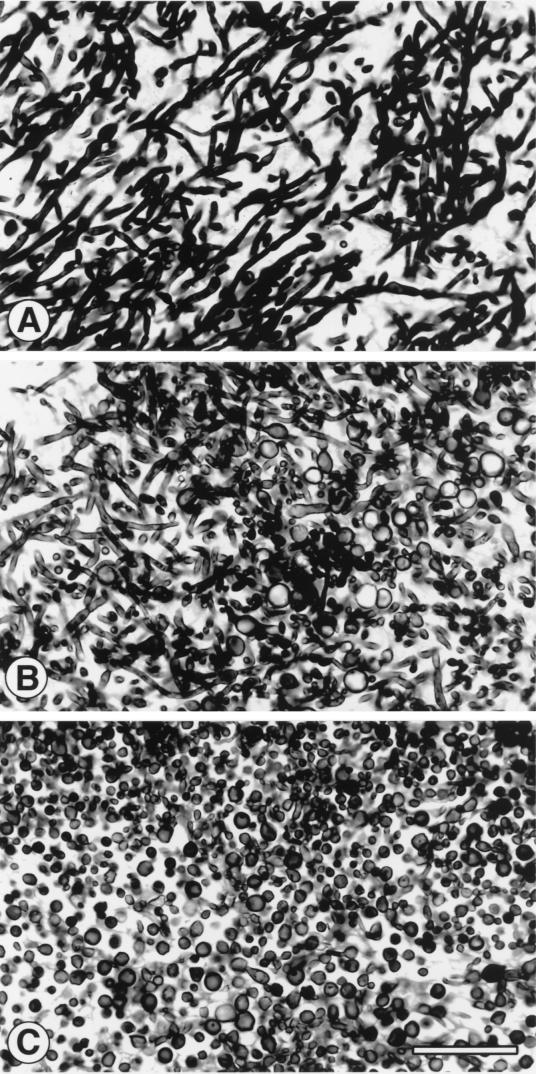
There was a concentration-dependent clearance of histologically detectable C. albicans in tissue. With LY0.1 and LY0.25 there was a dose-dependent reduction in the number of lesions, and with LY0.5 and LY1.0 there was complete eradication of lesions. Moreover, there was a dose-dependent effect on microscopic morphology of the Candida cell wall structure. As illustrated in Fig. 5, there was a transition from a predominance of hyphae and pseudohyphae in UCs to an increase in the proportion of large yeast-like structures in rabbits treated with LY0.1 to a predominance of these large yeast-like structures in rabbits treated with LY0.25. There was no histologic evidence of organisms in rabbits treated with ≥0.5 mg of LY per kg per day.
FIG. 5.
Dosage-dependent antifungal effect on microscopic morphology of the Candida cell structure from LY-treated rabbits. (A to C) Transition from a predominance of hyphae and pseudohyphae in UCs to a predominance of the large yeast-like structures with treatment with LY0.25. (A) UCs. (B) LY0.1. (C) LY0.25. Gomori methenamine silver stain was used. Magnification, ×550.
Safety.
AmB-treated rabbits had significant increases in the mean serum creatinine level (P < 0.001) and significant decreases in the serum potassium level (P < 0.01) in comparison to those for UCs, while LY-treated rabbits and FLU-treated rabbits had no changes in serum creatinine or serum potassium concentrations. There also were no differences in serum bilirubin, aspartyl aminotransaminase and alanine aminotransaminase levels in any of the treatment groups (Table 2).
TABLE 2.
Effects of LY, AmB, and FLU on serum creatinine and serum potassium concentrations in persistently neutropenic rabbits with disseminated candidiasis
| Treatment group (no. of animals) | Serum creatinine concn (mg/dl)a | Serum potassium concn (meq/liter)a |
|---|---|---|
| UC (n = 13) | 1.70 ± 0.26 | 3.88 ± 0.11 |
| LY (n = 30)b | 1.30 ± 0.02 | 3.98 ± 0.10 |
| AmB (n = 6) | 5.60 ± 1.42c | 3.02 ± 0.23d |
| FLU (n = 6) | 1.20 ± 0.09 | 4.15 ± 0.18 |
All values are expressed as means ± SEMs.
Mean serum creatinine and serum potassium concentrations for all dosage groups.
P < 0.001 (ANOVA with Bonferroni’s correction for multiple comparisons).
P < 0.01 (ANOVA with Bonferroni’s correction for multiple comparisons).
DISCUSSION
This study demonstrated that LY has activity comparable to that of AmB or FLU in the treatment of disseminated candidiasis in persistently neutropenic rabbits. These in vivo antifungal effects were dose dependent and corresponded to the in vitro concentration-dependent fungicidal effect shown in time-kill assays. A dosage-dependent effect on Candida cell wall morphology was also observed in infected tissues. Complete microbiological and histological clearance of infection from all tissues, including brain tissue, was found with LY at ≥0.5 mg/kg/day. Disposition of the drug given i.v. appeared to be linear at the dosage range of 0.1 to 1.0 mg/kg/day. Moreover, LY at this same dosage range had no adverse effects on renal or hepatic function, whereas AmB at 1.0 mg/kg/day caused significant azotemia and hypokalemia.
The concentration-dependent and dosage-dependent effects of this echinocandin are reflected in vitro and in vivo, respectively. The in vitro fungicidal effect of LY at concentrations of 0.01, 0.1, and 0.25 μg/ml was concentration dependent, resulting in reductions of approximately 105, 106, and 108 CFU/ml over 24 h. The control time-kill assay for AmB also demonstrated fungicidal activity most strikingly at 0.25 μg/ml. While the rate of killing was clearly greater at 0.25 μg/ml for AmB than for LY at 0.25 μg/ml, the echinocandin appeared to have more fungicidal activity than AmB at the lower concentrations of 0.01 and 0.1 μg/ml. These in vitro findings were predictive of the fungicidal effects of LY in persistently neutropenic rabbits with disseminated candidiasis.
The dose proportionality of LY differs from the plasma pharmacokinetic profile of its predecessor, cilofungin, which displayed nonlinear saturation plasma pharmacokinetics (40). While cilofungin had potent in vitro concentration-dependent fungicidal activity, its short t1/2 in plasma did not reflect this activity in vivo. Only with saturation of its clearance mechanism did the sustained levels in plasma result in sufficiently potent antifungal activity against disseminated candidiasis in vivo. By comparison, LY over the dosage range explored in these studies appears to display linear, nonsaturable kinetics in plasma which correlate both in vitro and in vivo with concentration-dependent and dosage-dependent anti-Candida effects, respectively. Whether this linearity is reflected at higher dosages remains to be investigated.
The pharmacokinetics of LY in plasma demonstrated Cmaxs ranging from a mean of 0.46 to 3.56 μg/ml at concentrations of between 0.1 and 1.0 mg/kg. These Cmaxs are similar to those observed in rabbits treated with AmB within this dosage range (24). Indeed, at 1.0 mg/kg, even V and CL of LY are virtually identical to those of conventional AmB. The similarities of the pharmacokinetic profiles of LY at 1.0 mg/kg and AmB at 1.0 mg/kg in plasma permit comparisons of the antifungal effect at comparable doses and peak concentrations in plasma. More recent studies in which minimum sampling methods were used were conducted in our laboratory and indicate that the pharmacokinetics of LY in plasma are similar between infected and noninfected rabbits.
The levels of plasma protein binding of LY are estimated to be approximately 75, 84, and 95% in rat, human, and dog plasma, respectively (8). The level of serum protein binding of LY in rabbits is unknown, and we have not determined it in our laboratories. We therefore analyzed the AUC/MIC ratio assuming a lower limit of 75 and an upper limit of 95% protein binding on the basis of these data. Table 3 presents the mean AUC/MIC ratio for total drug, for free drug assuming 75% plasma protein binding, and for drug assuming 95% plasma protein binding.
TABLE 3.
AUC and AUC/MIC ratio for total LY and free LY
| Dose (mg/kg) | Mean AUC0–24 (μg · h/ml) for total drug | AUC/MIC for total drug | AUC (μg · h/ml) for free drug corrected for 75% plasma protein binding | AUC/MIC for free drug assuming 75% plasma protein binding | AUC for free drug corrected for 95% plasma protein binding | AUC/MIC ratio for free drug assuming 95% plasma protein binding |
|---|---|---|---|---|---|---|
| 0.1 | 0.565 | 37.6 | 0.141 | 2 | 0.03 | 0.5 |
| 0.25 | 1.56 | 104 | 0.39 | 26 | 0.08 | 5.3 |
| 0.5 | 3.47 | 231 | 0.87 | 58 | 0.17 | 11.3 |
| 1.0 | 10.06 | 671 | 2.5 | 167 | 0.50 | 33.3 |
The concentration-dependent antifungal effect was observed in all tissues, which includes the liver, spleen, kidney, lung, vena cava, and brain. Microbiological and histological eradication was achieved at dosages of ≥0.5 mg/kg. With a dose of 0.25 μg/ml clearance in time-kill assays was achieved within 24 h in vitro. However, while peak concentrations in plasma of >0.25 μg/ml were achieved with doses of 0.1 and 0.25 mg/kg, the levels in plasma were sustained above the MLC (0.25 μg/ml) for less than 2 h. At the higher and more effective doses of 0.5 and 1.0 mg/kg, the times spent above the MLC were 5 and 12 h, respectively. Sustained killing of C. albicans in time-kill assays was also observed with lower concentrations of 0.1 and 0.01 μg/ml; the higher doses of 0.5 and 1.0 mg/kg maintained levels above these concentrations for approximately 12 and 20 h, respectively.
This dosage-dependent antifungal effect of LY was observed at all tissue sites, including the brain. Despite the relatively large molecular size of LY, this echinocandin was able to clear a central nervous system infection. Perhaps disruption of the blood-brain barrier by Candida organisms facilitated penetration of LY into the brain tissue. That an echinocandin has potential for treatment of Candida infections of the central nervous system may offer expanded treatment strategies against these often refractory infections (17, 18). There also was a lower threshold (0.25 mg/kg/day) for clearance of organisms from the spleen in comparison to clearance from other tissues, in which the effective dosage was 0.5 mg/kg/day. The reason for this greater response is unclear. There may be more accumulation of LY in the spleen. Alternatively, in synergy with LY, the residual splenic macrophages may be more effective than other populations of macrophages against C. albicans. Roilides et al. (34) recently reported that splenic macrophages demonstrated significantly greater antifungal activity against pseudohyphae of C. albicans than other macrophage populations in the liver and lungs. The dosage-dependent antifungal effect of LY and its linear plasma pharmacokinetics may permit a rational extrapolation of these in vivo findings to the design and interpretation of clinical trials for the treatment and prevention of invasive candidiasis.
Corresponding to the dosage-dependent microbiological response to LY, there was also a dosage-dependent clearance of histologically detectable C. albicans from tissue. In addition, the microscopic morphologies of Candida pseudohyphae and blastoconidia were profoundly altered with the lower dosages of 0.1 and 0.25 mg/kg/day. This altered morphology is most likely related to the inhibition of 1,3-β-d-glucan synthase in the echinocandin. At the lowest dosage (0.1 mg/kg/day), there was inhibition of hyphal and pseudohyphal formation, possibly due to the lack of 1,3-β-d-glucan synthesis and, thus, hyphal elongation. At the higher dosage, the cell wall structure appears to be further damaged, resulting in large distorted yeast forms. Such an effect of alteration of fungal cell wall morphology was reported by Cole et al. (4) for Candida cells in the gastrointestinal tracts of infected immunocompromised mice treated with cilofungin. However, this effect was attributed to the cyclophosphamide used in the murine model rather than to the cilofungin used to treat the disseminated experimental candidiasis. That sublethal concentrations of cilofungin may have contributed to this alteration in cell wall morphology cannot be excluded. We previously studied the effects of araC in vitro and in vivo and found no effects of the S-phase-specific inhibitor of DNA polymerase on the morphology of C. albicans. Our findings in vivo demonstrate that LY exerts a striking dosage-dependent effect on the cell walls of these organisms. Additionally, other laboratories found during the determination of MICs and MLCs that sublethal concentrations of echinocandins cause morphological alterations of Candida in vitro (22, 25, 44). Those observations correlate with the present in vivo observations.
There was no evidence of hepatic or renal toxicity due to LY administered at as much as 1.0 mg/kg/day. By comparison, AmB administered at 1 mg/kg/day, as anticipated, caused azotemia and hypokalemia. The serum creatinine level in infected, immunocompromised rabbits treated with AmB was greater than those previously reported in noninfected immunocompetent rabbits (24), probably as the result of concomitant gentamicin-induced nephrotoxicity in the former animals. The paucity of toxicity of these new echinocandins is in contrast to that of cilofungin, the carrier of which caused metabolic acidosis and renal insufficiency. Thus, LY, which demonstrated potent antifungal activity in the treatment of disseminated candidiasis in persistently neutropenic rabbits, represents a novel advance in antifungal therapy and merits further investigation in carefully designed clinical trials.
ACKNOWLEDGMENTS
We thank Joanne Peter for performing in vitro antifungal susceptibility studies and Robert L. Schaufele for technical assistance in those experiments. We also thank the staffs of the Veterinary Resources Branch and the National Cancer Institute Office of the Laboratory Animal Science for laboratory animal care.
REFERENCES
- 1.Anaissie E, Bodey G P, Kantarjian H, David C, Barnett K, Bow E, Defelice R, Downs N, File T, Karam G, et al. Fluconazole therapy for chronic disseminated candidiasis in patients with leukemia and prior amphotericin B therapy. Am J Med. 1991;91:142–150. doi: 10.1016/0002-9343(91)90006-j. [DOI] [PubMed] [Google Scholar]
- 2.Anaissie E J, Rex J H, Uzun O, Vartivarian S. Predictors of adverse outcome in cancer patients with candidemia. Am J Med. 1998;104:238–245. doi: 10.1016/s0002-9343(98)00030-8. [DOI] [PubMed] [Google Scholar]
- 3.Bartlett M S, Current W L, Goheen M P, Boylan C J, Lee C H, Shaw M M, Queener S F, Smith J W. Semisynthetic echinocandins affect cell wall deposition of Pneumocystis carinii in vitro and in vivo. Antimicrob Agents Chemother. 1996;40:1811–1816. doi: 10.1128/aac.40.8.1811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cole G T, Seshan K R, Phaneuf M, Lynn K T. Chlamydospore-like cells of Candida albicans in the gastrointestinal tract of infected, immunocompromised mice. Can J Microbiol. 1991;37:637–646. doi: 10.1139/m91-108. [DOI] [PubMed] [Google Scholar]
- 5.Committee on the Care and Use of Laboratory Animals of the Institute of Laboratory Animal Resources, Commission on Life Sciences, National Research Council. Guide for the care and use of laboratory animals. Washington, D.C: National Academy Press; 1996. [Google Scholar]
- 6.De Bock R. Epidemiology of invasive fungal infections in bone marrow transplantation. Bone Marrow Transplant. 1994;14(Suppl. 5):S1–S2. [PubMed] [Google Scholar]
- 7.Debono M, Gordee R S. Antibiotics that inhibit fungal cell wall development. Annu Rev Microbiol. 1994;48:471–497. doi: 10.1146/annurev.mi.48.100194.002351. [DOI] [PubMed] [Google Scholar]
- 8.Eli Lilly & Company. LY303366 Clinical investigator’s Brochure. Eli Lilly & Company, Indianapolis, Ind.; 1996. [Google Scholar]
- 9.Ernst M E, Klepser M E, Wolfe E J, Pfaller M A. Antifungal dynamics of LY303366, an investigational echinocandin B analog, against Candida spp. Diagn Microbiol Infect Dis. 1996;26:125–131. doi: 10.1016/s0732-8893(96)00202-7. [DOI] [PubMed] [Google Scholar]
- 10.Fromtling R A. LY303366. Drugs Future. 1994;19:338–342. [Google Scholar]
- 11.Frosco M B, Barret J F. Importance of antifungal drug-resistance: clinical significance and need for novel therapy. Exp Opin Invest Drugs. 1998;7:175–197. doi: 10.1517/13543784.7.2.175. [DOI] [PubMed] [Google Scholar]
- 12.Gallis H A, Drew R H, Pickard W W. Amphotericin B: 30 years of clinical experience. Rev Infect Dis. 1990;12:308–329. doi: 10.1093/clinids/12.2.308. [DOI] [PubMed] [Google Scholar]
- 13.Georgopapadakou N H, Tkacz J S. The fungal cell wall as a drug target. Trends Microbiol. 1995;3:98–104. doi: 10.1016/s0966-842x(00)88890-3. [DOI] [PubMed] [Google Scholar]
- 14.Gibaldi M, Perrier D. Pharmacokinetics. 2nd ed. New York, N.Y: Dekker; 1982. pp. 455–459. [Google Scholar]
- 15.Goodman J L, Winston D J, Greenfield R A, Chandrasekar P H, Fox B, Kaizer H, Shadduck R K, Shea T C, Stiff P, Friedman D J. A controlled trial of fluconazole to prevent fungal infections in patients undergoing bone marrow transplantation. N Engl J Med. 1992;326:845–851. doi: 10.1056/NEJM199203263261301. [DOI] [PubMed] [Google Scholar]
- 16.Gordee R S, Zeckner D J, Howard L C, Alborn W E, Jr, Debono M. Anti-Candida activity and toxicology of LY121019, a novel semisynthetic polypeptide antifungal antibiotic. Ann N Y Acad Sci. 1988;544:294–309. doi: 10.1111/j.1749-6632.1988.tb40415.x. [DOI] [PubMed] [Google Scholar]
- 17.Jafari H S, Saez-LLorens X, Severien C, Parras F, Friedland I, Rinderknecht S, Ehrett S, Olsen K D, Abramowsky C, McCracken G H., Jr Effects of antifungal therapy on inflammation, sterilization, and histology in experimental Candida albicans meningitis. Antimicrob Agents Chemother. 1994;38:83–89. doi: 10.1128/aac.38.1.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jafari H S, Saez-LLorens X, Grimprel E, Argyle J C, Olsen K D, McCracken G H., Jr Characteristics of experimental Candida albicans infection of the central nervous system in rabbits. J Infect Dis. 1991;164:389–395. doi: 10.1093/infdis/164.2.389. [DOI] [PubMed] [Google Scholar]
- 19.Kauffman C A, Bradley S F, Ross S C, Weber D R. Hepatosplenic candidiasis: successful treatment with fluconazole. Am J Med. 1991;91:137–141. doi: 10.1016/0002-9343(91)90005-i. [DOI] [PubMed] [Google Scholar]
- 20.Klepser M E, Ernst E J, Ernst M E, Messer S A, Pfaller M A. Evaluation of endpoints for antifungal susceptibility determinations with LY303366. Antimicrob Agents Chemother. 1998;42:1387–1391. doi: 10.1128/aac.42.6.1387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Krishnarao T V, Galgiani N J. Comparison of the in vitro activities of the echinocandin LY303366, the pneumocandin MK-0991, and fluconazole against Candida species and Cryptococcus neoformans. Antimicrob Agents Chemother. 1997;41:1957–1960. doi: 10.1128/aac.41.9.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kurtz M B, Douglas C M. Lipopeptide inhibitors of fungal glucan synthase. J Med Vet Mycol. 1997;35:79–86. doi: 10.1080/02681219780000961. [DOI] [PubMed] [Google Scholar]
- 23.Lecciones J A, Lee J W, Navarro E E, Witebsky F G, Marshall D, Steinberg S M, Pizzo P A, Walsh T J. Vascular catheter-associated fungemia in patients with cancer: analysis of 155 episodes. Rev Infect Dis. 1992;14:875–883. doi: 10.1093/clinids/14.4.875. [DOI] [PubMed] [Google Scholar]
- 24.Lee J W, Amantea M A, Francis P A, Navarro E E, Bacher J, Pizzo P A, Walsh T J. Pharmacokinetics and safety of a unilamellar liposomal formulation of amphotericin B (AmBisome) in rabbits. Antimicrob Agents Chemother. 1993;38:713–718. doi: 10.1128/aac.38.4.713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Morrison C J, Stevens D A. Comparative effects of cilofungin and amphotericin B on experimental murine candidiasis. Antimicrob Agents Chemother. 1990;34:746–750. doi: 10.1128/aac.34.5.746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.National Committee for Clinical Laboratory Standards. Reference method for broth dilution antifungal susceptibility testing of yeasts. Tentative standard. NCCLS document M27-T. Wayne, Pa: National Committee for Clinical Laboratory Standards; 1997. [Google Scholar]
- 27.Nguyen M H, Peacock J E, Jr, Morris A J, Tanner D C, Nguyen M L, Snydman D R, Wagener M M, Rinaldi M G, Yu V L. The changing face of candidemia: emergence of non-Candida albicans species and antifungal resistance. Am J Med. 1996;100:617–623. doi: 10.1016/s0002-9343(95)00010-0. [DOI] [PubMed] [Google Scholar]
- 28.Perfect J R, Hobbs M M, Wright K A, Durack D T. Treatment of experimental disseminated candidiasis with cilofungin. Antimicrob Agents Chemother. 1989;33:1811–1812. doi: 10.1128/aac.33.10.1811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pfaller M A, Marco F, Messer S A, Jones R N. In vitro activity of two echinocandin derivatives, LY303366 and MK-0991 (L-743,872), against clinical isolates of Aspergillus, Fusarium, Rhizopus, and other filamentous fungi. Diagn Microbiol Infect Dis. 1998;30:251–255. doi: 10.1016/s0732-8893(97)00246-0. [DOI] [PubMed] [Google Scholar]
- 30.Pfaller M A, Messer S A, Coffman S. In vitro susceptibilities of clinical yeast isolates to a new echinocandin derivative, LY303366, and other antifungal agents. Antimicrob Agents Chemother. 1997;41:763–766. doi: 10.1128/aac.41.4.763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rajman I, Desante K, Hatcher B, Hemingway J, Lachno R, Brooks S, Turik M. Program and abstracts of the 37th Interscience Conference on Antimicrobial Agents and Chemotherapy. Washington, D.C: American Society for Microbiology; 1997. LY303366 single intravenous dose pharmacokinetics and safety in healthy volunteers, abstr. F-74; p. 158. [Google Scholar]
- 32.Rex J H, Bennet J E, Sugar A M. A randomized trial comparing fluconazole with amphotericin B for the treatment of candidemia in patients without neutropenia. N Engl J Med. 1994;331:1325–1330. doi: 10.1056/NEJM199411173312001. [DOI] [PubMed] [Google Scholar]
- 33.Rex J H, Rinaldi M G, Pfaller M A. Resistance of Candida species to fluconazole. Antimicrob Agents Chemother. 1995;39:1–8. doi: 10.1128/aac.39.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Roilides E, Sein T, Gonzalez C, Lyman C, Walsh T J. Program and abstracts of the 37th Interscience Conference on Antimicrobial Agents and Chemotherapy. Washington, D.C: American Society for Microbiology; 1997. Antifungal activity of splenic, liver and pulmonary macrophages against Candida albicans and modulation by macrophage colony-stimulating factor (M-CSF), abstr. G-79; p. 280. [Google Scholar]
- 35.Sarosi G A. Amphotericin B: still the ‘gold standard’ for antifungal therapy. Postgrad Med. 1990;88:151–152. doi: 10.1080/00325481.1990.11716368. , 155–161, 165–166. [DOI] [PubMed] [Google Scholar]
- 36.Slavin M A, Osborne B, Adams R, Levenstein M J, Schoch H G, Feldman A R, Meyers J D, Bowden R A. Efficacy and safety of fluconazole prophylaxis for fungal infections after marrow transplantation—a prospective, randomized, double-blind study. J Infect Dis. 1995;171:1545–1552. doi: 10.1093/infdis/171.6.1545. [DOI] [PubMed] [Google Scholar]
- 37.Walsh T J, McEntee C, Dixon D M. Tissue homogenization with sterile reinforced polyethylene bags for quantitative culture of Candida albicans. J Clin Microbiol. 1987;25:931–932. doi: 10.1128/jcm.25.5.931-932.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Walsh T J, Bacher J, Pizzo P A. Chronic silastic central venous catheterization for induction, maintenance, and support of persistent granulocytopenia in rabbits. Lab Anim Med. 1988;38:467–470. [PubMed] [Google Scholar]
- 39.Walsh T J, Lee J W, Roilides E, Francis P, Backer J, Lyman C A, Pizzo P A. Experimental antifungal chemotherapy in granulocytopenic animal models of disseminated candidiasis: approaches to understanding investigational antifungal compounds for patients with neoplastic diseases. Clin Infect Dis. 1992;14(Suppl. 1):139–147. doi: 10.1093/clinids/14.supplement_1.s139. [DOI] [PubMed] [Google Scholar]
- 40.Walsh T J, Lee J W, Kelly P, Bacher J, Lecciones J, Thomas V, Lyman C, Coleman D, Gordee R, Pizzo P A. Antifungal effects of the nonlinear pharmacokinetics of cilofungin, a 1,3-β-glucan synthetase inhibitor, during continuous and intermittent intravenous infusions in treatment of experimental disseminated candidiasis. Antimicrob Agents Chemother. 1991;35:1321–1328. doi: 10.1128/aac.35.7.1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Walsh T J, Aoki S, Mechinaud F, Backer J, Lee J, Rubin M, Pizzo P A. Effects of preventive, early, and late antifungal chemotherapy with fluconazole in different granulocytopenic models of experimental disseminated candidiasis. J Infect Dis. 1990;161:755–760. doi: 10.1093/infdis/161.4.755. [DOI] [PubMed] [Google Scholar]
- 42.Wright W L, Wenzel R P. Nosocomial Candida. Epidemiology, transmission, and prevention. Infect Dis Clin N Am. 1997;11:411–425. doi: 10.1016/s0891-5520(05)70363-9. [DOI] [PubMed] [Google Scholar]
- 43.Zeckner D, Butler T, Boylan C, Boyll B, Lin Y, Rabb P, Schmidtke J, Current W. Program and abstracts of the 33rd Interscience Conference on Antimicrobial Agents and Chemotherapy. Washington, D.C: American Society for Microbiology; 1993. LY303366, activity against systemic aspergillosis and histoplasmosis in murine models, abstr. 364; p. 186. [Google Scholar]
- 44.Zhanel G G, Karlowsky J A, Harding G A, Balko T V, Zelenitsky S A, Friesen M, Kabani A, Turik M, Hoban D J. In vitro activity of a new semisynthetic echinocandin, LY-303366, against systemic isolates of Candida species, Cryptococcus neoformans, Blastomyces dermatitidis, and Aspergillus species. Antimicrob Agents Chemother. 1997;41:863–865. doi: 10.1128/aac.41.4.863. [DOI] [PMC free article] [PubMed] [Google Scholar]