Abstract
Simple Summary
Epigenetic changes in a cell’s genome can cause the inactivation of genes and affect how tumours respond to chemotherapy. These epigenetic changes do not alter the DNA sequence and small molecule inhibitors of maintenance of the epigenetic state can change the sensitivity of a tumour to chemotherapy. Epigenetic therapies are being clinically evaluated as single agents or in combination with chemotherapies, including those that induce DNA damage. DNA damage induced by chemotherapy may be repaired and the cell either survives or alternatively cell death pathways are engaged and the cell is eliminated. Epigenetic silencing of gene expression during tumour development may lead to sensitivity of the tumour to chemotherapy if DNA repair of the DNA lesion is inactivated, or to chemotherapy resistance if cell death responses are inactivated. This illustrates the clinical challenge of using epigenetic therapies in combination with chemotherapy. Will such epigenetic drugs confer chemosensitivity and increased efficacy of chemotherapy, or induce resistance? Is there an effect of epigenetic therapies on normal cell chemo-sensitivity leading to increased toxicity? This review will illustrate the clinical challenges in combination studies of epigenetic therapies with chemotherapy and discuss future perspectives on addressing these challenges.
Abstract
Epigenetic therapies describe drug molecules such as DNA methyltransferase, histone methyltransferase and histone acetylase/deacetylase inhibitors, which target epigenetic mechanisms such as DNA methylation and histone modifications. Many DNA damage response (DDR) genes are epigenetically regulated in cancer leading to transcriptional silencing and the loss of DNA repair capacity. Epigenetic marks at DDR genes, such as DNA methylation at gene promoters, have the potential to be used as stratification biomarkers, identifying which patients may benefit from particular chemotherapy treatments. For genes such as MGMT and BRCA1, promoter DNA methylation is associated with chemosensitivity to alkylating agents and platinum coordination complexes, respectively, and they have use as biomarkers directing patient treatment options. In contrast to epigenetic change leading to chemosensitivity, DNA methylation of DDR genes involved in engaging cell death responses, such as MLH1, are associated with chemoresistance. This contrasting functional effect of epigenetic modification on chemosensitivity raises challenges in using DNA-demethylating agents, and other epigenetic approaches, to sensitise tumours to DNA-damaging chemotherapies and molecularly targeted agents. Demethylation of MGMT/BRCA1 could lead to drug resistance whereas demethylation of MLH1 could sensitise cells to chemotherapy. Patient selection based on a solid understanding of the disease pathway will be one means to tackle these challenges. The role of epigenetic modification of DDR genes during tumour development, such as causing a mutator phenotype, has different selective pressures and outcomes compared to epigenetic adaptation during treatment. The prevention of epigenetic adaptation during the acquisition of drug resistance will be a potential strategy to improve the treatment of patients using epigenetic therapies.
Keywords: DNA repair, epigenetics, cancer, DNA methylation
1. Deoxyribonucleic Acid (DNA) Damage Response (DDR) Genes Are Epigenetically Regulated in Cancer, Affecting Chemosensitivity
Epigenetic modifications, such as gene promoter DNA hypermethylation, and subsequent changes in gene expression of DDR-associated genes, lead to a loss of DNA repair capacity and have been demonstrated in a variety of tumours and cell line models [1,2]. The loss of DNA repair activity in tumours may lead to chemosensitivity to DNA-damaging cytotoxic chemotherapy. One of the paradigms demonstrating the clinical relevance of epigenetic mechanisms involved in chemosensitivity is highlighted in glioblastoma. DNA methylation of MGMT, the DNA repair gene encoding O-6-methylguanine-DNA methyltransferase, leads to the loss of MGMT expression [1]. This leads to reduced DNA damage repair and subsequently increased sensitivity of cells to the alkylating agent, temozolomide [3]. Early clinical studies showed that glioma patients treated with temozolomide and radiotherapy and with a methylated MGMT gene promoter have a survival benefit compared to only radiotherapy. Patients with unmethylated MGMT promoters showed no statistically significant difference in survival [4]. Prospective randomised trials of glioblastoma patients for radiotherapy versus alkylating agent chemotherapy have demonstrated DNA methylation of MGMT is a clinically useful predictive biomarker to stratify patients, rather than just prognostic [3,5,6].
A second paradigm is the epigenetic alterations of homology recombination DNA repair (HR)-associated genes including breast cancer type 2 susceptibility proteins 1 and 2 (BRCA1/2) [7,8]. BRCA1 is frequently methylated in high-grade serous ovarian cancer (HGSOC) and can lead to HR-deficiency (HRD) which is associated with increased patient survival following platinum-based chemotherapy compared to patients with HR proficient tumours [8]. Sensitivity to platinum-based chemotherapy exploits HRD in HGSOC by introducing double-stranded breaks in DNA, leading to genomic instability and cell apoptosis [9]. However, non-homologous end joining (NHEJ) and base excision repair (BER) pathways, which require Poly (ADP-ribose) polymerase (PARP), can still be utilised to repair the damaged DNA. PARP inhibitors (PARPi) induce synthetic lethality in HR-deficient tumours by disrupting multiple DNA repair pathways simultaneously [10]. In breast and ovarian cancer, BRCA1/2 status is clinically useful to predict sensitivity to platinum-based chemotherapy and PARPi [11,12]. Furthermore, patients with BRCA1 methylated and HRD HGSOC have better prognosis than unmethylated HR proficient tumours [13].
In both the above paradigms, epigenetic regulation by DNA methylation during tumour development, prior to chemotherapy, leads to inactivation of DNA repair activity and drug sensitivity. However, epigenetic mechanisms have also been proposed as important drivers of acquired drug resistance adaptation during chemotherapy. This leads to increased epigenetic silencing in tumours at relapse compared to primary presentation [14]. For instance, loss of DNA mismatch repair (MMR) due to DNA methylation at the MutL Homolog 1 (MLH1) gene promoter has been associated with resistance to alkylating agents such as temozolomide and crosslinking agents such as cisplatin [15,16,17]. The presence of functional MMR has been proposed to lead to cell death due to futile repair cycles, generation of double-strand DNA breaks and engagement of apoptosis [18]. Thus, the absence of MMR leads to loss of engagement of cell death pathways by DDR pathways leading to drug resistance. In another example of epigenetic adaptation following chemotherapy, while sensitivity to PARP inhibitors of HGSOC is associated with DNA methylation at BRCA1, tumours recurring following chemotherapy restore BRCA1 expression associated with reduced DNA methylation. This supports a key role for DNA methylation changes during the acquisition of PARPi resistance [19]. FANCF, another DDR-associated gene closely linked to BRCA genes, is often methylated in several different cancer types including testicular [20], head and neck [21], lung [21], cervical [22] and ovarian [23,24]. Methylation of the FANCF promoter in ovarian cancer has been linked to platinum sensitivity, whereas demethylation of FANCF has been associated with platinum resistance and often occurs after platinum chemotherapy [24,25].
Whilst aberrant methylation of DDR genes has been shown in multiple cancers, other epigenetic mechanisms such as histone modifications at genomic regulatory regions, including enhancers and super enhancers, may also play an important role in response to chemotherapy. Studies show modest benefits of temozolmide treatment in patients with methylated MGMT in colorectal cancer and patient-derived xenograft (PDX) models of glioblastoma show high expression of MGMT linked to active enhancers, despite promoter methylation [26]. This suggests different epigenetic mechanisms are able to dynamically regulate gene expression. Furthermore, DDR-associated genes associated with drug response may themselves regulate the epigenetic landscape. BRCA1 mutations in breast cancer epithelial cells lead to the loss of H3K27ac at super enhancers and impair enhancer–promoter lopping [27], whereas BRCA2 depletion has been linked to chromatin remodelling [28]. MMR inactivation via MLH1 mutations has been shown to activate enhancers of genes associated with growth in colorectal cancer and may activate enhancers of genes associated with drug-resistance [29]. These observations highlight the potential interplay of different epigenetic mechanisms and DDR-associated genes in relation to chemosensitivity (Figure 1).
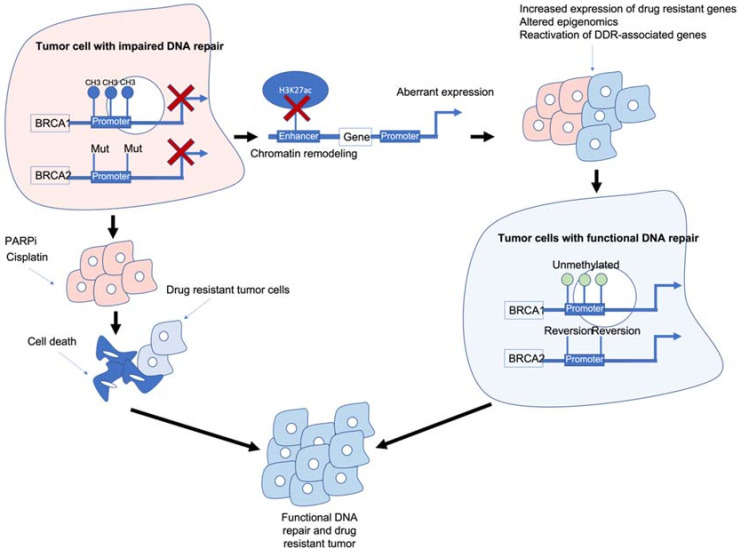
Figure 1.
Epigenetic interactions with breast cancer type 1/2 susceptibility (BRCA1/2) genes and drug resistance. BRCA1/2 deficiency caused by methylation and/or mutations results in impaired DNA repair and often sensitivity to poly (ADP-ribose) polymerase inhibitors (PARPi)/platinum-based chemotherapies. BRCA1/2 deficiency can also modulate chromatin accessibility and enhancers of drug-resistant and/or DNA damage response (DDR)-associated genes. Reactivation of BRCA1/2 either by demethylation or reversion mutations can result in tumour cells with functional DDR. Ultimately, these mechanisms result in tumour cells that are drug resistant, have functional DDR and no longer respond to previous PARPi/platinum chemotherapy.
BRCA1/2 deficiency can impair the HR pathway leading to increased sensitivity to DNA-damaging agents such as platinum chemotherapy and PARPi. However, BRCA1/2 deficiency can also lead to chromatin remodelling and reduced H3K27ac at regulatory regions such as enhancers/super-enhancers, leading to the aberrant expression of genes including those associated with drug resistance/DDR-associated genes. Reactivation of BRCA1/2 and/or other DDR-associated genes can result in tumour cells which are drug resistant and have functional DNA damage repair.
2. Can Epigenetic Therapies Reverse Epigenetically Driven Drug Resistance?
The clinically relevant examples above demonstrate how promoter DNA methylation may confer tumour chemosensitivity or chemoresistance. However, there is a wide spectrum of DDR genes whose epigenetic regulation can influence chemosensitivity [2]. Epigenetic therapies such as DNA-demethylating agents and inhibitors of maintenance of histone post-translational modification (for instance, histone deacetylase inhibitors) are now registered for clinical use, particularly in haematological malignancies. Furthermore, they remain the focus of many clinical trials in epithelial cancers [30]. However, careful patient selection will be key to demonstrating clinical efficacy of epigenetic therapies, especially when used in combination with other therapies.
This is exemplified in early clinical trials of DNA-demethylating agents in HGSOC. In cell line models, the loss of MMR due to MLH1 methylation results in failure to engage apoptotic responses and resistance to platinum coordination complexes and alkylating agents which can be reversed by DNA-demethylating agents such as 5-azacytidine and its derivatives [31]. DNA methylation has been used as a pharmacodynamic marker in surrogate tissues, such as blood, to demonstrate biological activity and guide the scheduling of combination studies with other therapies [32]. There have been two randomised phase II studies of DNA-demethylating agents and carboplatin in HGSOC with contrasting outcomes [33,34]. One study was closed early due to unacceptable toxicity and lack of efficacy of the combination compared to single-agent carboplatin [33]. The other trial showed an improvement in the 6-month progression-free survival of patients treated with the combination. However, this trial did not show statistically significant superiority for the primary endpoint of progression-free survival, potentially due to being statistically underpowered for the latter endpoint [34]. Both studies explored the combination of carboplatin with a DNA-demethylating agent during second-line chemotherapy. Glasspool et al., recruited partially platinum-sensitive patients recurring 6–12 months following the initial response to platinum-based chemotherapy while Oza et al., recruited women with recurrence within 6 months of the last platinum-containing regimen. It is possible that partially platinum sensitive patients may have a different proportion of women with tumours who are sensitive due to the methylation of HR genes, such as BRCA1, and for whom a demethylating agent may have an adverse effect. Neither study selected patient recruitment based on the methylation status of the patients’ tumours.
As previously mentioned, BRCA1 is frequently methylated in high-grade serous ovarian cancer and associated with increased patient survival following platinum-based and PARP inhibitor chemotherapy compared to patients with HR-proficient tumours. BRCA1/2 deficiency remains the strongest predictor of PARPi sensitivity [35] although abrogation of other key HR genes including FA Complementation Group A (FANCA) [36], DNA Repair Protein RAD51 homolog 1 (RAD51) [37], X-ray Repair Cross Complementing 2 (XRCC2) and X-ray Repair Cross Complementing 3 (XRCC3) [38] and DNA Polymerase Delta 4 (POLD4) [39] have been linked to platinum and/or PARPi responses. Furthermore, not all BRCA mutant tumours are HR deficient and many HR-proficient tumours can initially respond well to PARPi [40,41,42,43,44] which has been attributed to the involvement of PARP in other non-DDR associated mechanisms including chromatin remodelling [45]. Unfortunately, as with platinum-based chemotherapy, primary and acquired resistance to PARPi is common [35,40,46,47]. Reversion mutations can restore the function of HR-associated genes frequently mutated in HGSOC, including BRCA1/2 [48,49,50,51] and RAD51C/D [52]. Epigenetic mechanisms including histone modifications may also contribute to PARPi resistance although the exact mechanisms remain poorly understood [12,52,53,54,55,56,57].
Histone methylation has been linked to PARP inhibitor sensitivity in multiple cancers [58]. Enhancer of Zeste Homolog 2 (EZH2) and Euchromatic Histone Lysine Methyltransferase 2 (EHMT2) both maintain repressive H3K27 and H3K9 methylation histone marks, respectively, and are frequently overexpressed in cancer [59]. The inhibition of EZH2 alone has previously been linked to reducing the expression of multiple genes associated with DDR pathways in multiple cancers including prostate [60] and ovarian [61]. Furthermore, EZH2 inhibition has been shown to sensitise breast cancer cells to PARPi [58,61] and PARPi can regulate EZH2 expression [62] via PARPylation. EHMT2 has been linked with directly recruiting HR-associated factors, including BRCA1 to promote DNA damage repair [63]. The inhibition of EHMT2 promotes increased DNA damage and altered cell cycle regulation [64] and PARPi resistant cells treated with an EHMT1/2 inhibitor show significantly altered gene expression changes enriched in pro-survival pathways including, phosphatidylinositol 3 kinase(PI3K), protein kinase B (AKT) and mammalian target of rapamycin (mTOR) [64]. In BRCA1-depleted SUM149 breast cancer cells and PDX models, treatment with an EZH2i and PARPi reduced tumour growth more than single PARPi treatment; however, this effect was not seen in a BRCA2-depleted mouse model of breast cancer [65]. EZH2 inhibition alone may not be sufficient to modulate chromatin conformation [66] therefore dual EZH2/EHMT2 inhibitors in combination with PARPi would perhaps be of future interest. A clear theme regarding epigenetic therapies in combination with chemotherapy and their ability to modulate epigenetically driven drug resistance is that any future combination therapies must have clear stratification markers. The global epigenetic and mutational profile of DDR-associated genes must also be considered in order to maximise the positive outcomes for patients. A summary of key DDR genes associated with drug resistance and/or regulated by epigenetic mechanisms are shown in Table 1.
Table 1.
Summary of DDR-associated genes, how they can be epigenetically regulated and their involvement in drug response.
| Gene | Symbol | Summary | Reference Number |
|---|---|---|---|
| O6-Methylguanine-DNA Methyltransferase | MGMT | Methylated associated with increased sensitivity to temozolomide. Enhancer region associated with increased expression and resistance to temozolomide. | [1,3,5,6,26] |
| Breast Cancer type 1 susceptibility protein | BRCA1 | Methylated associated with sensitivity to PARPi/platinum and loss of H3K27ac at enhancer regions. | [7,8,11,12,27,35] |
| Breast Cancer type 2 susceptibility protein | BRCA2 | Deficiency causes chromatin conformation changes and increased sensitivity to PARPi/platinum. | [7,8,11,12,28] |
| MutL Homolog 1 | MLH1 | Unmethylated associated with temozolomide/platinum resistance and loss of MMR. | [15,16,17,31] |
| FA Complementation Group F | FANCF | Methylation associated with sensitivity to platinum, unmethylated associated with platinum resistance. | [24,25] |
| FA Complementation Group A | FANCA | Germline mutation associated with increased sensitivity to DNA damaging agents. | [36] |
| DNA Repair Protein RAD51 homolog 1 | RAD51 | High expression associated with platinum resistance. | [37] |
| X-ray Repair Cross Complementing 2 | XRCC2 | Low expression associated with sensitivity to PARPi. | [38] |
| X-ray Repair Cross Complementing 3 | XRCC3 | Low expression associated with sensitivity to PARPi. | [38] |
| DNA Polymerase Delta 4 | POLD4 | Low expression associated with sensitivity to PARPi/platinum. | [39] |
| RAD51 Paralog C | RAD51C | Reversion mutations associated with increased resistance to PARPi. | [52] |
| RAD51 Paralog D | RAD51D | Reversion mutations associated with increased resistance to PARPi. | [52] |
| Euchromatic Histone Lysine Methyltransferase 2 | EHMT2 | Maintains repressive H3K9 methylation marks. Recruits HR-associated factors, Inhibition of EHMT2 promotes DNA damage. | [58,59,63,64,66] |
| Enhancer of Zeste Homolog 2 | EZH2 | Maintains repressive H3K27 methylation marks. Controls expression of multiple DDR-associated genes. Inhibition of EZH2 sensitises cells to PARPi. | [58,59,60,61,66] |
3. Epigenetic Changes in Normal Tissue following Chemotherapy
The observations of increased DNA methylation at gene promoters in drug resistant tumours and cell line models following chemotherapy treatment could be due to the selection of cells epigenetically silenced that are present in the tumour before chemotherapy. Alternatively, DNA damage induced by the chemotherapy may be causing methylation changes. DNA damage such as platinum-induced and DNA double-strand breaks are recognized by DNA mismatch repair proteins [67]. These bind and recruit the DNA methylating enzyme encoded by the DNMT1 gene, resulting in aberrant DNA methylation [68,69,70,71]. At the time of relapse following platinum-based chemotherapy, changes in methylation at specific CpG sites in blood DNA are observed which mirror changes occurring in tumour DNA at relapse in ovarian cancer patients [72]. These changes can predict clinical outcome and identify patients with better overall survival. In contrast, blood samples taken at presentation prior to treatment show no association between methylation and survival. DNA methylation at specific CpGs in blood has been associated with environmental exposures including smoking and alcohol consumption [73,74]. Smoking-induced methylation changes at the ARRH gene have been associated with aberrant ARRH transcription in lung epithelial cells [60] and mediate the risk of developing lung cancer [75]. In 2020, it was estimated that 4.1% of all cancers can be attributed to alcohol consumption [76]. A 144 CpG DNA methylation signature has been used to identify heavy alcohol consumption in whole blood samples [77]. Whilst the functional consequences of methylation changes in blood remain unknown, they may be acting as a surrogate markers for changes in a more relevant tissue. Similarly, epigenetic changes occurring in normal tissues due to DNA-damaging agents such as chemotherapy can have long-term consequences for secondary tumours or altering immune responses [78].
4. Challenges and Future Prospects
Epigenetic drugs including inhibitors of DNA methyltransferases, histone methyltransferases and histone acetylases/deacetylases can all affect genome-wide transcription. Therefore, they may be a doubled-edged sword by globally activating or inhibiting DDR-associated genes or tumour suppressors/oncogenes or activating pathways which have negative impacts on other drug agents. Drugs targeting specific markers such as PARP inhibitors may also affect the global expression of genes. PARP1 can modulate chromatin confirmation and epigenetic regulation [79,80]; therefore, PARPi may cause off-target effects which are not currently clear. Reactions to PARPi have been documented with evidence suggesting that whilst patients with germline BRCA mutations may respond well to PARPi [10,81], their risk of developing other diseases including myelodysplastic syndrome and acute myeloid leukemia is increased [82]. HR or BRCA restoration may also negate any benefits of PARPi use [83,84], further highlighting the need to time treatment and develop biomarkers to predict not only the response but also the probability of HR restoration during treatment.
Epigenetic mechanisms underpin immune evasion in cancer. In pre-clinical models, the inhibition of DNMT1 and EZH2 improves the efficacy of immune checkpoint inhibitor therapy, slows tumour progression and increases TH1-cytokine production [85]. A key mechanism involves viral defence responses. DNMT1 inhibition triggers cytosolic sensing of double-stranded RNA (dsRNA), thereby inducing type I interferon responses. Many of the dsRNA species arise from the reactivation of heavily methylated endogenous retroviruses [86]. These data suggest that combining an immune checkpoint inhibitor with a DNA-demethylating agent may increase response rates. In 2017, the FDA approved the use of pembrolizumab for the treatment of malignancies with microsatellite instability (MSI-H) or mismatch repair deficiency (dMMR). It is hypothesised that a higher somatic mutational load leads to increased presentation of neoepitopes thus mediating immunotherapy responses in MSI-H tumours. In colorectal cancer, MSI-H tumours evolve gene expression changes (immunoediting) that may confer resistance to recognition by the immune system and hence resistance to pembrolizumab, despite high mutational load and frequent lymphocytic infiltration. Furthermore, these tumours also have genomic and DNA methylation events associated with activated WNT signalling and T-cell exclusion [87]. Tumours with dMMR during treatment may be resistant to checkpoint inhibitors due to epigenetic mechanisms driving immunoediting. Hence, the combination of pembrolizumab and a DNA-demethylating agent may be particularly beneficial to patients that have lost the expression of MMR genes due to DNA methylation.
The selection of patients most likely to benefit will be crucial for effective clinical evaluation of epigenetic therapies given alone or in combination with DNA-damaging agents. While many potential predictive biomarkers of drug sensitivity have been proposed in preclinical models, few have been tested in clinical trials of epigenetic therapies and none yet have been used as companion biomarkers during their clinical use. This is partly because most biomarkers identified from preclinical studies have been tested as prognostic rather than predictive biomarkers in clinical studies. This is partly due to the relative ease of doing prognostic studies that need considerably fewer patient samples. To exemplify this, a prognostic study in ovarian cancer to detect a hazard ratio of 2 at 80% power at p = 0.05 would require approximately 110 tumour samples. A predictive study to compare a biomarker between two treatment arms at similar statistical power would require over 1200 tumours. The challenges in identifying predictive biomarkers have previously been reviewed and potential roadmaps for their evaluation developed [88,89]. However, most clinical trial designs include the predictive biomarker late on in the clinical evaluation pathway. Perhaps the identification of molecular subgroups based on a biological understanding of the mechanism of action of an epigenetic therapy needs to be included earlier in the pathway, for instance, by selecting patients based on an epigenomic subtype of DDR genes, rather than histological subtype in phase II studies of combinations with DNA-damaging cytotoxics.
Trial designs which incorporate stratification biomarker evaluation will be particularly important in combining DNA-damaging therapies with epigenetic agents. This is clearly exemplified with DNA-demethylating agents where the demethylation of genes such as MGMT and BRCA1 could lead to resistance to cytotoxics and treatment failure, while demethylation of MMR genes and other pro-apoptotic genes could lead to increased efficacy in response to cytotoxics inducing DNA damage. The clinical timing of epigenetic intervention will also be important. Tumours at initial presentation will have genetic and epigenetic changes as a consequence of tumorigenesis where loss of DNA damage responses and genomic instability are important drivers. In contrast, tumours at the time of recurrence will have changes as a consequence of tumour adaptation during chemotherapy as well as tumorigenic evolution. Furthermore, DDR-associated genes can be dynamically regulated by more than one epigenetic mechanism. MGMT expression can still be high despite promoter DNA hypermethylation due to histone acetylation at active enhancer regions [26]. Therefore, one can envisage a scenario where a patient who has a methylated gene associated with drug resistance may appear as a candidate who could benefit from treatment with a DNMT inhibitor, yet this may fail due to histone modification having a greater effect on expression. Another particularly interesting speculation is whether epigenetic therapies could be used during remission as a way to prevent the emergence of epigenetic adaptation and resistance. Delaying recurrence or maintaining tumours in a chemotherapy sensitive state would have major effects on the overall survival of cancer patients. However, this assumes that epigenetic therapies do not have adverse effects on promoting tumour progression or normal cell toxicity.
5. Conclusions
Epigenetic marks play a key role in drug sensitivity/resistance. Epigenetic therapies, including DMNT inhibitors which target DNA methylation, HKMT inhibitors which target histone methylation and HDAC inhibitors which target histone deacetylases, could play an important role in enhancing chemotherapy by modifying epigenetic marks associated with resistance. Several DDR-associated genes can be regulated by epigenetic mechanisms such as DNA methylation, with recent evidence showing DDR-associated gene expression can be controlled by multiple epigenetic mechanisms including histone methylation and acetylation. Furthermore, specific DDR-associated genes inducing BRCA1/2 can directly modulate epigenetic mechanisms such as chromatin conformation [28] and enhancer activity [27,90] providing a strong rationale to combine epigenetic therapies with current chemotherapy in order to circumvent acquired resistance and enhance chemotherapy outcomes. The stratification of patients based on the mutation status of key DDR-associated genes, such as BRCA1/2 and MGMT, will require careful consideration with respect to their epigenetic state in order to predict a robust response to chemotherapy.
Author Contributions
I.M.G. and R.B. were involved in writing and editing the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Esteller M., Hamilton S.R., Burger P.C., Baylin S.B., Herman J.G. Inactivation of the DNA repair gene O(6)-methylguanine-DNA methyltransferase by promoter hypermethylation is a common event in primary human neoplasia. Cancer Res. 1999;59:793–797. [PubMed] [Google Scholar]
- 2.Knijnenburg T.A., Wang L., Zimmermann M.T., Chambwe N., Gao G.F., Cherniack A.D., Fan H., Shen H., Way G.P., Greene C.S., et al. Genomic and Molecular Landscape of DNA Damage Repair Deficiency across The Cancer Genome Atlas. Cell Rep. 2018;23:239–254.e6. doi: 10.1016/j.celrep.2018.03.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hegi M.E., Diserens A.-C., Gorlia T., Hamou M.-F., de Tribolet N., Weller M., Kros J.M., Hainfellner J.A., Mason W., Mariani L., et al. MGMT Gene Silencing and Benefit from Temozolomide in Glioblastoma. N. Engl. J. Med. 2005;352:997–1003. doi: 10.1056/NEJMoa043331. [DOI] [PubMed] [Google Scholar]
- 4.Wick W., Platten M., Meisner C., Felsberg J., Tabatabai G., Simon M., Nikkhah G., Papsdorf K., Steinbach J.P., Sabel M., et al. Temozolomide chemotherapy alone versus radiotherapy alone for malignant astrocytoma in the elderly: The NOA-08 randomised, phase 3 trial. Lancet Oncol. 2012;13:707–715. doi: 10.1016/S1470-2045(12)70164-X. [DOI] [PubMed] [Google Scholar]
- 5.Mansouri A., Hachem L.D., Mansouri S., Nassiri F., Laperriere N.J., Xia D., Lindeman N.I., Wen P.Y., Chakravarti A., Mehta M.P., et al. MGMT promoter methylation status testing to guide therapy for glioblastoma: Refining the approach based on emerging evidence and current challenges. Neuro Oncol. 2019;21:167–168. doi: 10.1093/neuonc/noy132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Weller M., Stupp R., Reifenberger G., Brandes A.A., Van Den Bent M.J., Wick W., Hegi M.E. MGMT promoter methylation in malignant gliomas: Ready for personalized medicine? Nat. Rev. Neurol. 2010;6:39–51. doi: 10.1038/nrneurol.2009.197. [DOI] [PubMed] [Google Scholar]
- 7.Bell D., Berchuck A., Birrer M., Chien J., Cramer D.W., Dao F., Dhir R., Disaia P., Gabra H., Glenn P., et al. Integrated genomic analyses of ovarian carcinoma. Nature. 2011;474:609–615. doi: 10.1038/nature10166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Stronach E.A., Paul J., Timms K.M., Hughes E., Brown K., Neff C., Perry M., Gutin A., El-Bahrawy M., Steel J.H., et al. Biomarker assessment of HR deficiency, tumor BRCA1/2 mutations, and CCNE1 copy number in ovarian cancer: Associations with clinical outcome following platinum monotherapy. Mol. Cancer Res. 2018;16:1103–1111. doi: 10.1158/1541-7786.MCR-18-0034. [DOI] [PubMed] [Google Scholar]
- 9.Dasari S., Bernard Tchounwou P. Cisplatin in cancer therapy: Molecular mechanisms of action. Eur. J. Pharmacol. 2014;740:364–378. doi: 10.1016/j.ejphar.2014.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lord C.J., Ashworth A. PARP inhibitors: Synthetic lethality in the clinic. Science. 2017;355:1152–1158. doi: 10.1126/science.aam7344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Faraoni I., Graziani G. Role of BRCA Mutations in Cancer Treatment with Poly(ADP-ribose) Polymerase (PARP) Inhibitors. Cancers. 2018;10:487. doi: 10.3390/cancers10120487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kondrashova O., Topp M., Nesic K., Lieschke E., Ho G.-Y., Harrell M.I., Zapparoli G.V., Hadley A., Holian R., Boehm E., et al. Methylation of all BRCA1 copies predicts response to the PARP inhibitor rucaparib in ovarian carcinoma. Nat. Commun. 2018;9:3970. doi: 10.1038/s41467-018-05564-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bonadio R., Fogace R., Miranda V., Diz M. Homologous recombination deficiency in ovarian cancer: A review of its epidemiology and management. Clinics. 2018;73:e450s. doi: 10.6061/clinics/2018/e450s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Brown R., Curry E., Magnani L., Wilhelm-Benartzi C.S., Borley J. Poised epigenetic states and acquired drug resistance in cancer. Nat. Rev. Cancer. 2014;14:747–753. doi: 10.1038/nrc3819. [DOI] [PubMed] [Google Scholar]
- 15.Wu F., Lu M., Qu L., Li D.Q., Hu C.H. DNA methylation of hMLH1 correlates with the clinical response to cisplatin after a surgical resection in Non-small cell lung cancer. Int. J. Clin. Exp. Pathol. 2015;8:5457–5463. [PMC free article] [PubMed] [Google Scholar]
- 16.Zeller C., Dai W., Steele N.L., Siddiq A., Walley A.J., Wilhelm-Benartzi C.S.M., Rizzo S., Van Der Zee A., Plumb J.A., Brown R. Candidate DNA methylation drivers of acquired cisplatin resistance in ovarian cancer identified by methylome and expression profiling. Oncogene. 2012;31:4567–4576. doi: 10.1038/onc.2011.611. [DOI] [PubMed] [Google Scholar]
- 17.Gallitto M., Cheng He R., Inocencio J.F., Wang H., Zhang Y., Deikus G., Wasserman I., Strahl M., Smith M., Sebra R., et al. Epigenetic preconditioning with decitabine sensitizes glioblastoma to temozolomide via induction of MLH1. J. Neurooncol. 2020;147:557–566. doi: 10.1007/s11060-020-03461-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.O’Brien V., Brown R. Signalling cell cycle arrest and cell death through the MMR System. Carcinogenesis. 2006;27:682–692. doi: 10.1093/carcin/bgi298. [DOI] [PubMed] [Google Scholar]
- 19.Swisher E.M., Kwan T.T., Oza A.M., Tinker A.V., Ray-Coquard I., Oaknin A., Coleman R.L., Aghajanian C., Konecny G.E., O’Malley D.M., et al. Molecular and clinical determinants of response and resistance to rucaparib for recurrent ovarian cancer treatment in ARIEL2 (Parts 1 and 2) Nat. Commun. 2021;12:2487. doi: 10.1038/s41467-021-22582-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Koul S., McKiernan J.M., Narayan G., Houldsworth J., Bacik J., Dobrzynski D.L., Assaad A.M., Mansukhani M., Reuter V.E., Bosl G.J., et al. Role of promoter hypermethylation in cisplatin treatment response of male germ cell tumors. Mol. Cancer. 2004;3:16. doi: 10.1186/1476-4598-3-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Marsit C.J., Liu M., Nelson H.H., Posner M., Suzuki M., Kelsey K.T. Inactivation of the Fanconi anemia/BRCA pathway in lung and oral cancers: Implications for treatment and survival. Oncogene. 2004;23:1000–1004. doi: 10.1038/sj.onc.1207256. [DOI] [PubMed] [Google Scholar]
- 22.Narayan G., Arias-Pulido H., Nandula S.V., Basso K., Sugirtharaj D.D., Vargas H., Mansukhani M., Villella J., Meyer L., Schneider A., et al. Promoter Hypermethylation of FANCF: Disruption of Fanconi Anemia-BRCA Pathway in Cervical Cancer. Cancer Res. 2004;64:2994–2997. doi: 10.1158/0008-5472.CAN-04-0245. [DOI] [PubMed] [Google Scholar]
- 23.Lim S.L., Smith P., Syed N., Coens C., Wong H., Van Der Burg M., Szlosarek P., Crook T., Green J.A. Promoter hypermethylation of FANCF and outcome in advanced ovarian cancer. Br. J. Cancer. 2008;98:1452–1456. doi: 10.1038/sj.bjc.6604325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Taniguchi T., Tischkowitz M., Ameziane N., Hodgson S.V., Mathew C.G., Joenje H., Mok S.C., D’Andrea A.D. Disruption of the Fanconi anemia-BRCA pathway in cisplatin-sensitive ovarian tumors. Nat. Med. 2003;9:568–574. doi: 10.1038/nm852. [DOI] [PubMed] [Google Scholar]
- 25.D’Andrea A.D. The Fanconi Anemia/BRCA Signaling Pathway: Disruption in Cisplatin-Sensitive Ovarian Cancers. Cell Cycle. 2003;2:289–291. doi: 10.4161/cc.2.4.413. [DOI] [PubMed] [Google Scholar]
- 26.Chen X., Zhang M., Gan H., Wang H., Lee J.-H., Fang D., Kitange G.J., He L., Hu Z., Parney I.F., et al. A novel enhancer regulates MGMT expression and promotes temozolomide resistance in glioblastoma. Nat. Commun. 2018;9:2949. doi: 10.1038/s41467-018-05373-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang X., Wang Y., Chiang H.-C., Hsieh Y.-P., Lu C., Park B.H., Jatoi I., Jin V.X., Hu Y., Li R. BRCA1 mutations attenuate super-enhancer function and chromatin looping in haploinsufficient human breast epithelial cells. Breast Cancer Res. 2019;21:51. doi: 10.1186/s13058-019-1132-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gruber J.J., Chen J., Geller B., Jäger N., Lipchik A.M., Wang G., Kurian A.W., Ford J.M., Snyder M.P. Chromatin Remodeling in Response to BRCA2-Crisis. Cell Rep. 2019;28:2182–2193.e6. doi: 10.1016/j.celrep.2019.07.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hung S., Saiakhova A., Faber Z.J., Bartels C.F., Neu D., Bayles I., Ojo E., Hong E.S., Pontius W.D., Morton A.R., et al. Mismatch repair-signature mutations activate gene enhancers across human colorectal cancer epigenomes. eLife. 2019;8:e40760. doi: 10.7554/eLife.40760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Miranda Furtado C.L., Dos Santos Luciano M.C., Da Silva Santos R., Furtado G.P., Moraes M.O., Pessoa C. Epidrugs: Targeting epigenetic marks in cancer treatment. Epigenetics. 2019;14:1164–1176. doi: 10.1080/15592294.2019.1640546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Plumb J.A., Strathdee G., Sludden J., Kaye S.B., Brown R. Reversal of drug resistance in human tumor xenografts by 2′-deoxy-5-azacytidine-induced demethylation of the hMLH1 gene promoter. Cancer Res. 2000;60:6039–6044. [PubMed] [Google Scholar]
- 32.Appleton K., Mackay H.J., Judson I., Plumb J.A., McCormick C., Strathdee G., Lee C., Barrett S., Reade S., Jadayel D., et al. Phase I and pharmacodynamic trial of the DNA methyltransferase inhibitor decitabine and carboplatin in solid tumors. J. Clin. Oncol. 2007;25:4603–4609. doi: 10.1200/JCO.2007.10.8688. [DOI] [PubMed] [Google Scholar]
- 33.Glasspool R.M., Brown R., Gore M.E., Rustin G.J.S., McNeish I.A., Wilson R.H., Pledge S., Paul J., MacKean M., Hall G.D., et al. A randomised, phase II trial of the DNA-hypomethylating agent 5-aza-2′-deoxycytidine (decitabine) in combination with carboplatin vs carboplatin alone in patients with recurrent, partially platinum-sensitive ovarian cancer. Br. J. Cancer. 2014;110:1923–1929. doi: 10.1038/bjc.2014.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Oza A.M., Matulonis U.A., Alvarez Secord A., Nemunaitis J., Roman L.D., Blagden S.P., Banerjee S., McGuire W.P., Ghamande S., Birrer M.J., et al. A Randomized Phase II Trial of Epigenetic Priming with Guadecitabine and Carboplatin in Platinum-resistant, Recurrent Ovarian Cancer. Clin. Cancer Res. 2020;26:1009–1016. doi: 10.1158/1078-0432.CCR-19-1638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bouwman P., Jonkers J. Molecular pathways: How can BRCA-mutated tumors become resistant to PARP inhibitors? Clin. Cancer Res. 2014;20:540–547. doi: 10.1158/1078-0432.CCR-13-0225. [DOI] [PubMed] [Google Scholar]
- 36.Wilkes D.C., Sailer V., Xue H., Cheng H., Collins C.C., Gleave M., Wang Y., Demichelis F., Beltran H., Rubin M.A., et al. A germline FANCA alteration that is associated with increased sensitivity to DNA damaging agents. Mol. Case Stud. 2017;3:a001487. doi: 10.1101/mcs.a001487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hoppe M.M., Jaynes P., Wardyn J.D., Upadhyayula S.S., Tan T.Z., Lie S., Lim D.G.Z., Pang B.N.K., Lim S.P.S., Yeong J., et al. Quantitative imaging of RAD51 expression as a marker of platinum resistance in ovarian cancer. EMBO Mol. Med. 2021;13:e13366. doi: 10.15252/emmm.202013366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Murai J., Huang S.Y.N., Das B.B., Renaud A., Zhang Y., Doroshow J.H., Ji J., Takeda S., Pommier Y. Trapping of PARP1 and PARP2 by clinical PARP inhibitors. Cancer Res. 2012;72:5588–5599. doi: 10.1158/0008-5472.CAN-12-2753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhang S., Chao H.H., Wang X., Zhang Z., Lee E.Y.C., Lee M.Y.W.T. Loss of the p12 subunit of DNA polymerase delta leads to a defect in HR and sensitization to PARP inhibitors. DNA Repair. 2019;73:64–70. doi: 10.1016/j.dnarep.2018.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ledermann J.A., El-Khouly F. PARP inhibitors in ovarian cancer: Clinical evidence for informed treatment decisions. Br. J. Cancer. 2015;113:S10–S16. doi: 10.1038/bjc.2015.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Domchek S.M., Aghajanian C., Shapira-Frommer R., Schmutzler R.K., Audeh M.W., Friedlander M., Balmaña J., Mitchell G., Fried G., Stemmer S.M., et al. Efficacy and safety of olaparib monotherapy in germline BRCA1/2 mutation carriers with advanced ovarian cancer and three or more lines of prior therapy. Gynecol. Oncol. 2016;140:199–203. doi: 10.1016/j.ygyno.2015.12.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Matulonis U.A., Penson R.T., Domchek S.M., Kaufman B., Shapira-Frommer R., Audeh M.W., Kaye S., Molife L.R., Gelmon K.A., Robertson J.D., et al. Olaparib monotherapy in patients with advanced relapsed ovarian cancer and a germline BRCA1/2 mutation: A multistudy analysis of response rates and safety. Ann. Oncol. 2016;27:1013–1019. doi: 10.1093/annonc/mdw133. [DOI] [PubMed] [Google Scholar]
- 43.Kim D.S., Camacho C.V., Nagari A., Malladi V.S., Challa S., Kraus W.L. Activation of PARP-1 by snoRNAs Controls Ribosome Biogenesis and Cell Growth via the RNA Helicase DDX21. Mol. Cell. 2019;75:1270–1285.e14. doi: 10.1016/j.molcel.2019.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Keung M.Y., Wu Y., Badar F., Vadgama J.V. Response of Breast Cancer Cells to PARP Inhibitors Is Independent of BRCA Status. J. Clin. Med. 2020;9:940. doi: 10.3390/jcm9040940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Luijsterburg M.S., de Krijger I., Wiegant W.W., Shah R.G., Smeenk G., de Groot A.J.L., Pines A., Vertegaal A.C.O., Jacobs J.J.L., Shah G.M., et al. PARP1 Links CHD2-Mediated Chromatin Expansion and H3.3 Deposition to DNA Repair by Non-homologous End-Joining. Mol. Cell. 2016;61:547–562. doi: 10.1016/j.molcel.2016.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Audeh M.W., Carmichael J., Penson R.T., Friedlander M., Powell B., Bell-McGuinn K.M., Scott C., Weitzel J.N., Oaknin A., Loman N., et al. Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and recurrent ovarian cancer: A proof-of-concept trial. Lancet. 2010;376:245–251. doi: 10.1016/S0140-6736(10)60893-8. [DOI] [PubMed] [Google Scholar]
- 47.Sonnenblick A., de Azambuja E., Azim H.A., Piccart M. An update on PARP inhibitors—Moving to the adjuvant setting. Nat. Rev. Clin. Oncol. 2015;12:27–41. doi: 10.1038/nrclinonc.2014.163. [DOI] [PubMed] [Google Scholar]
- 48.Sakai W., Swisher E.M., Jacquemont C., Chandramohan K.V., Couch F.J., Langdon S.P., Wurz K., Higgins J., Villegas E., Taniguchi T. Functional restoration of BRCA2 protein by secondary BRCA2 mutations in BRCA2-mutated ovarian carcinoma. Cancer Res. 2009;69:6381–6386. doi: 10.1158/0008-5472.CAN-09-1178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Dhillon K.K., Swisher E.M., Taniguchi T. Secondary mutations of BRCA1/2 and drug resistance. Cancer Sci. 2011;102:663–669. doi: 10.1111/j.1349-7006.2010.01840.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Norquist B., Wurz K.A., Pennil C.C., Garcia R., Gross J., Sakai W., Karlan B.Y., Taniguchi T., Swisher E.M. Secondary Somatic Mutations Restoring BRCA1/2 Predict Chemotherapy Resistance in Hereditary Ovarian Carcinomas. J. Clin. Oncol. 2011;29:3008–3015. doi: 10.1200/JCO.2010.34.2980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lin K.K., Harrell M.I., Oza A.M., Oaknin A., Ray-Coquard I., Tinker A.V., Helman E., Radke M.R., Say C., Vo L.T., et al. BRCA Reversion Mutations in Circulating Tumor DNA Predict Primary and Acquired Resistance to the PARP Inhibitor Rucaparib in High-Grade Ovarian Carcinoma. Cancer Discov. 2019;9:210–219. doi: 10.1158/2159-8290.CD-18-0715. [DOI] [PubMed] [Google Scholar]
- 52.Kondrashova O., Nguyen M., Shield-Artin K., Tinker A.V., Teng N.N.H., Harrell M.I., Kuiper M.J., Ho G.Y., Barker H., Jasin M., et al. Secondary somatic mutations restoring RAD51C and RAD51D associated with acquired resistance to the PARP inhibitor rucaparib in high-grade ovarian carcinoma. Cancer Discov. 2017;7:984–998. doi: 10.1158/2159-8290.CD-17-0419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Curry E., Zeller C., Masrour N., Patten D.K., Gallon J., Wilhelm-Benartzi C.S., Ghaem-Maghami S., Bowtell D.D., Brown R. Genes predisposed to DNA hypermethylation during acquired resistance to chemotherapy are identified in ovarian tumors by bivalent chromatin domains at initial diagnosis. Cancer Res. 2018;78:1383–1391. doi: 10.1158/0008-5472.CAN-17-1650. [DOI] [PubMed] [Google Scholar]
- 54.Wei S., Li C., Yin Z., Wen J., Meng H., Xue L., Wang J. Histone methylation in DNA repair and clinical practice: New findings during the past 5-years. J. Cancer. 2018;9:2072–2081. doi: 10.7150/jca.23427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sun J., Cai X., Yung M.M., Zhou W., Li J., Zhang Y., Li Z., Liu S.S., Cheung A.N.Y., Ngan H.Y.S., et al. miR-137 mediates the functional link between c-Myc and EZH2 that regulates cisplatin resistance in ovarian cancer. Oncogene. 2019;38:564–580. doi: 10.1038/s41388-018-0459-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Duan R., Du W., Guo W. EZH2: A novel target for cancer treatment. J. Hematol. Oncol. 2020;13:104. doi: 10.1186/s13045-020-00937-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Andronikou C., Rottenberg S. Studying PAR-Dependent Chromatin Remodeling to Tackle PARPi Resistance. Trends Mol. Med. 2021;27:630–642. doi: 10.1016/j.molmed.2021.04.010. [DOI] [PubMed] [Google Scholar]
- 58.Yamaguchi H., Du Y., Nakai K., Ding M., Chang S.S., Hsu J.L., Yao J., Wei Y., Nie L., Jiao S., et al. EZH2 contributes to the response to PARP inhibitors through its PARP-mediated poly-ADP ribosylation in breast cancer. Oncogene. 2018;37:208–217. doi: 10.1038/onc.2017.311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Rao Z.Y., Cai M.Y., Yang G.F., He L.R., Mai S.J., Hua W.F., Liao Y.J., Deng H.X., Chen Y.C., Guan X.Y., et al. EZH2 supports ovarian carcinoma cell invasion and/or metastasis via regulation of TGF-β 1 and is a predictor of outcome in ovarian carcinoma patients. Carcinogenesis. 2010;31:1576–1583. doi: 10.1093/carcin/bgq150. [DOI] [PubMed] [Google Scholar]
- 60.Yang Y.A., Yu J. EZH2, an epigenetic driver of prostate cancer. Protein Cell. 2013;4:331–341. doi: 10.1007/s13238-013-2093-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Karakashev S., Fukumoto T., Zhao B., Lin J., Wu S., Fatkhutdinov N., Park P.H., Semenova G., Jean S., Cadungog M.G., et al. EZH2 Inhibition Sensitizes CARM1-High, Homologous Recombination Proficient Ovarian Cancers to PARP Inhibition. Cancer Cell. 2020;37:157–167.e6. doi: 10.1016/j.ccell.2019.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Caruso L.B., Martin K.A., Lauretti E., Hulse M., Siciliano M., Lupey-Green L.N., Abraham A., Skorski T., Tempera I. Poly(ADP-ribose) Polymerase 1, PARP1, modifies EZH2 and inhibits EZH2 histone methyltransferase activity after DNA damage. Oncotarget. 2018;9:10585–10605. doi: 10.18632/oncotarget.24291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Yang Q., Zhu Q., Lu X., Du Y., Cao L., Shen C., Hou T., Li M., Li Z., Liu C., et al. G9a coordinates with the RPA complex to promote DNA damage repair and cell survival. Proc. Natl. Acad. Sci. USA. 2017;114:E6054–E6063. doi: 10.1073/pnas.1700694114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Watson Z.L., Yamamoto T.M., McMellen A., Kim H., Hughes C.J., Wheeler L.J., Post M.D., Behbakht K., Bitler B.G. Histone methyltransferases EHMT1 and EHMT2 (GLP/G9A) maintain PARP inhibitor resistance in high-grade serous ovarian carcinoma. Clin. Epigenet. 2019;11:165. doi: 10.1186/s13148-019-0758-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chen M. Efficacy of PARP inhibition combined with EZH2 inhibition depends on BRCA mutation status and microenvironment in breast cancer. FEBS J. 2021;288:2884–2887. doi: 10.1111/febs.15730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Curry E., Green I., Chapman-Rothe N., Shamsaei E., Kandil S., Cherblanc F.L., Payne L., Bell E., Ganesh T., Srimongkolpithak N., et al. Dual EZH2 and EHMT2 histone methyltransferase inhibition increases biological efficacy in breast cancer cells. Clin. Epigenet. 2015;7:84. doi: 10.1186/s13148-015-0118-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Li G.-M. Mechanisms and functions of DNA mismatch repair. Cell Res. 2008;18:85–98. doi: 10.1038/cr.2007.115. [DOI] [PubMed] [Google Scholar]
- 68.O’Hagan H.M., Wang W., Sen S., DeStefano Shields C., Lee S.S., Zhang Y.W., Clements E.G., Cai Y., Van Neste L., Easwaran H., et al. Oxidative Damage Targets Complexes Containing DNA Methyltransferases, SIRT1, and Polycomb Members to Promoter CpG Islands. Cancer Cell. 2011;20:606–619. doi: 10.1016/j.ccr.2011.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.O’Hagan H.M., Mohammad H.P., Baylin S.B. Double Strand Breaks Can Initiate Gene Silencing and SIRT1-Dependent Onset of DNA Methylation in an Exogenous Promoter CpG Island. PLoS Genet. 2008;4:e1000155. doi: 10.1371/journal.pgen.1000155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ding N., Bonham E.M., Hannon B.E., Amick T.R., Baylin S.B., O’Hagan H.M. Mismatch repair proteins recruit DNA methyltransferase 1 to sites of oxidative DNA damage. J. Mol. Cell Biol. 2016;8:244–254. doi: 10.1093/jmcb/mjv050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Cuozzo C., Porcellini A., Angrisano T., Morano A., Lee B., Di Pardo A., Messina S., Iuliano R., Fusco A., Santillo M.R., et al. DNA Damage, Homology-Directed Repair, and DNA Methylation. PLoS Genet. 2007;3:e110. doi: 10.1371/journal.pgen.0030110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Flanagan J.M., Wilson A., Koo C., Masrour N., Gallon J., Loomis E., Flower K., Wilhelm-Benartzi C., Hergovich A., Cunnea P., et al. Platinum-based chemotherapy induces methylation changes in blood DNA associated with overall survival in patients with ovarian cancer. Clin. Cancer Res. 2017;23:2213–2222. doi: 10.1158/1078-0432.CCR-16-1754. [DOI] [PubMed] [Google Scholar]
- 73.Shenker N.S., Polidoro S., van Veldhoven K., Sacerdote C., Ricceri F., Birrell M.A., Belvisi M.G., Brown R., Vineis P., Flanagan J.M. Epigenome-wide association study in the European Prospective Investigation Into Cancer And Nutrition (EPIC-Turin) identifies novel genetic loci associated with smoking. Hum. Mol. Genet. 2013;22:843–851. doi: 10.1093/hmg/dds488. [DOI] [PubMed] [Google Scholar]
- 74.Flanagan J.M., Brook M.N., Orr N., Tomczyk K., Coulson P., Fletcher O., Jones M.E., Schoemaker M.J., Ashworth A., Swerdlow A., et al. Temporal stability and determinants of white blood cell DNA methylation in the breakthrough generations study. Cancer Epidemiol. Biomark. Prev. 2015;24:221–229. doi: 10.1158/1055-9965.EPI-14-0767. [DOI] [PubMed] [Google Scholar]
- 75.Fasanelli F., Baglietto L., Ponzi E., Guida F., Campanella G., Johansson M., Grankvist K., Johansson M., Assumma M.B., Naccarati A., et al. Hypomethylation of smoking-related genes is associated with future lung cancer in four prospective cohorts. Nat. Commun. 2015;6:10192. doi: 10.1038/ncomms10192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Rumgay H., Shield K., Charvat H., Ferrari P., Sornpaisarn B., Obot I., Islami F., Lemmens V.E.P.P., Rehm J., Soerjomataram I. Global burden of cancer in 2020 attributable to alcohol consumption: A population-based study. Lancet Oncol. 2021;22:1071–1080. doi: 10.1016/S1470-2045(21)00279-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Liu C., Marioni R.E., Hedman A.K., Pfeiffer L., Tsai P.C., Reynolds L.M., Just A.C., Duan Q., Boer C.G., Tanaka T., et al. A DNA methylation biomarker of alcohol consumption. Mol. Psychiatry. 2018;23:422–433. doi: 10.1038/mp.2016.192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Jones P.A., Ohtani H., Chakravarthy A., De Carvalho D.D. Epigenetic therapy in immune-oncology. Nat. Rev. Cancer. 2019;19:151–161. doi: 10.1038/s41568-019-0109-9. [DOI] [PubMed] [Google Scholar]
- 79.Ummarino S., Hausman C., Di Ruscio A. The PARP Way to Epigenetic Changes. Genes. 2021;12:446. doi: 10.3390/genes12030446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Quénet D., El Ramy R., Schreiber V., Dantzer F. The role of poly(ADP-ribosyl)ation in epigenetic events. Int. J. Biochem. Cell Biol. 2009;41:60–65. doi: 10.1016/j.biocel.2008.07.023. [DOI] [PubMed] [Google Scholar]
- 81.Lord C.J., Tutt A.N.J., Ashworth A. Synthetic lethality and cancer therapy: Lessons learned from the development of PARP inhibitors. Annu. Rev. Med. 2015;66:455–470. doi: 10.1146/annurev-med-050913-022545. [DOI] [PubMed] [Google Scholar]
- 82.Morice P.M., Leary A., Dolladille C., Chrétien B., Poulain L., González-Martín A., Moore K., O’Reilly E.M., Ray-Coquard I., Alexandre J. Myelodysplastic syndrome and acute myeloid leukaemia in patients treated with PARP inhibitors: A safety meta-analysis of randomised controlled trials and a retrospective study of the WHO pharmacovigilance database. Lancet Haematol. 2021;8:e122–e134. doi: 10.1016/S2352-3026(20)30360-4. [DOI] [PubMed] [Google Scholar]
- 83.Edwards S.L., Brough R., Lord C.J., Natrajan R., Vatcheva R., Levine D.A., Boyd J., Reis-Filho J.S., Ashworth A. Resistance to therapy caused by intragenic deletion in BRCA2. Nature. 2008;451:1111–1115. doi: 10.1038/nature06548. [DOI] [PubMed] [Google Scholar]
- 84.Noordermeer S.M., van Attikum H. PARP Inhibitor Resistance: A Tug-of-War in BRCA-Mutated Cells. Trends Cell Biol. 2019;29:820–834. doi: 10.1016/j.tcb.2019.07.008. [DOI] [PubMed] [Google Scholar]
- 85.Peng D., Kryczek I., Nagarsheth N., Zhao L., Wei S., Wang W., Sun Y., Zhao E., Vatan L., Szeliga W., et al. Epigenetic silencing of TH1-type chemokines shapes tumour immunity and immunotherapy. Nature. 2015;527:249–253. doi: 10.1038/nature15520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Chiappinelli K.B., Strissel P.L., Desrichard A., Li H., Henke C., Akman B., Hein A., Rote N.S., Cope L.M., Snyder A., et al. Inhibiting DNA Methylation Causes an Interferon Response in Cancer via dsRNA Including Endogenous Retroviruses. Cell. 2015;162:974–986. doi: 10.1016/j.cell.2015.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Grasso C.S., Giannakis M., Wells D.K., Hamada T., Mu X.J., Quist M., Nowak J.A., Nishihara R., Qian Z.R., Inamura K., et al. Genetic mechanisms of immune evasion in colorectal cancer. Cancer Discov. 2018;8:730–749. doi: 10.1158/2159-8290.CD-17-1327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Dobbin K.K., Cesano A., Alvarez J., Hawtin R., Janetzki S., Kirsch I., Masucci G.V., Robbins P.B., Selvan S.R., Streicher H.Z., et al. Validation of biomarkers to predict response to immunotherapy in cancer: Volume II—clinical validation and regulatory considerations. J. Immunother. Cancer. 2016;4:77. doi: 10.1186/s40425-016-0179-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Prognostic/Predictive Biomarker Roadmap. [(accessed on 4 March 2022)]. Available online: https://www.cancerresearchuk.org/sites/default/files/prognostic_and_predictive.pdf.
- 90.Zhang X., Li R. BRCA1-Dependent Transcriptional Regulation: Implication in Tissue-Specific Tumor Suppression. Cancers. 2018;10:513. doi: 10.3390/cancers10120513. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.